Published online Mar 26, 2026. doi: 10.12998/wjcc.v14.i9.118634
Revised: February 12, 2026
Accepted: February 27, 2026
Published online: March 26, 2026
Processing time: 76 Days and 6.4 Hours
Hyponatremia is a common postoperative electrolyte disturbance in neuro
A 62-year-old woman underwent elective left supraorbital craniotomy and mi
This case highlights CBZ as a potentially reversible cause of significant postoperative hyponatremia after meningioma surgery. Early sodium monitoring after CBZ initiation and prompt substitution with alternative antiseizure therapy should be considered when euvolemic hypotonic hyponatremia develops in the postoperative period.
Core Tip: A 62-year-old woman developed progressive symptomatic hypotonic euvolemic hyponatremia after frontobasal meningioma resection shortly after starting carbamazepine (CBZ) for postoperative seizure prophylaxis. Despite isotonic saline, sodium fell to 119 mmol/L with somnolence; investigations supported a syndrome of inappropriate antidiuretic hormone secretion like pattern. Discontinuation of CBZ with substitution to levetiracetam, alongside hypertonic saline, led to clinical recovery and sustained normalization of serum sodium.
- Citation: Begagić E, Bečulić H, Mašović A, Alić F, Huremović M, Vranić S. Carbamazepine-induced hyponatremia following meningioma surgery: A case report. World J Clin Cases 2026; 14(9): 118634
- URL: https://www.wjgnet.com/2307-8960/full/v14/i9/118634.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i9.118634
Hyponatremia is among the most frequent electrolyte disturbances encountered in neurosurgical practice and occurs in a substantial proportion of patients, with reported rates varying widely according to the underlying diagnosis and procedure[1]. In the postoperative setting, even modest reductions in serum sodium can be clinically relevant because neurological symptoms such as lethargy, confusion, and seizures may overlap with, or mimic, intracranial complications, thereby complicating early postoperative assessment and decision-making[1]. The differential diagnosis of postoperative hyponatremia includes syndrome of inappropriate antidiuretic hormone secretion (SIADH), cerebral salt wasting, adrenal insufficiency, iatrogenic fluid administration, and medication-related mechanisms, with SIADH often cited as a pre
Carbamazepine (CBZ) remains widely used for epilepsy, neuralgia, and selected psychiatric indications, but it is a well-recognized cause of drug-induced hyponatremia[3]. The proposed mechanisms include antidiuretic effects mediated by enhanced vasopressin activity and/or increased renal sensitivity at the collecting duct, resulting in impaired free-water excretion[4]. Across heterogeneous populations, the reported incidence of CBZ-associated hyponatremia ranges broadly (approximately 1.8%-40%), reflecting differences in case definitions, comorbidities, concomitant medications, and dosing strategies[3]. In neurosurgical patients, this adverse effect is of particular concern when CBZ is introduced perioperatively for seizure prophylaxis, as the postoperative period itself may confer additional vulnerability to dysnatremias[1].
The present report describes a patient with clinically significant postoperative hyponatremia temporally associated with initiation of CBZ following meningioma surgery. The study also summarizes the current evidence on CBZ-associated hyponatremia. The aim is to highlight diagnostic considerations, practical management strategies, and preventive implications for postoperative antiseizure prophylaxis in patients undergoing intracranial tumor resection.
Increasing somnolence and cognitive slowing after left frontobasal meningioma surgery, beginning on postoperative day (POD) 2.
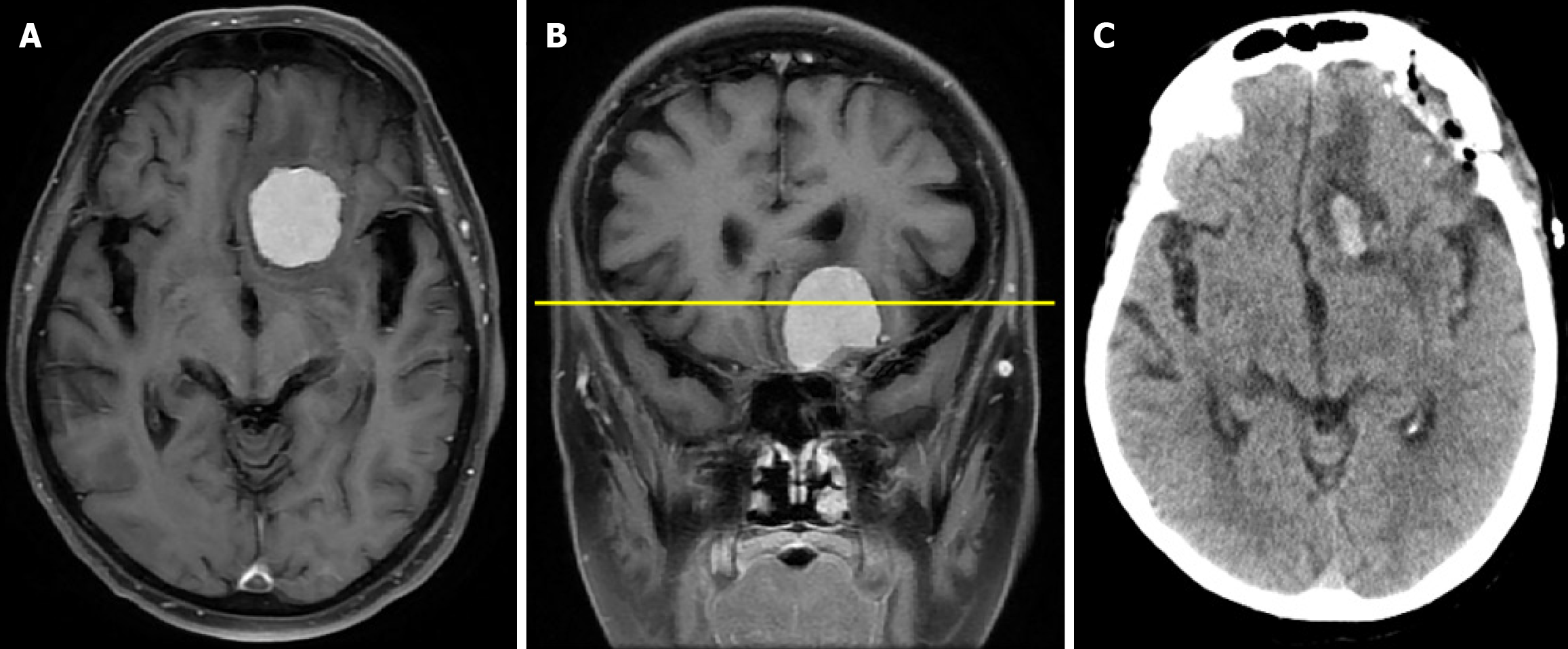
A 62-year-old woman was admitted for elective neurosurgical treatment of a left anterior skull-base (frontobasal) mass identified on pre-admission imaging. Approximately six weeks before admission, she experienced a brief transient loss of consciousness clinically consistent with a seizure; subsequent cranial computed tomography (CT) and magnetic resonance imaging (MRI) demonstrated a left frontobasal extra-axial lesion radiologically suggestive of meningioma (Figure 1A and B). She also reported several months of nausea without vomiting. She underwent a left supraorbital craniotomy with dural opening and microsurgical tumor resection; histopathology confirmed a World Health Organization grade 1 meningioma. A control cranial CT on POD 1 was satisfactory (Figure 1C). Postoperative antiseizure prophylaxis was initiated with CBZ 400 mg twice daily.
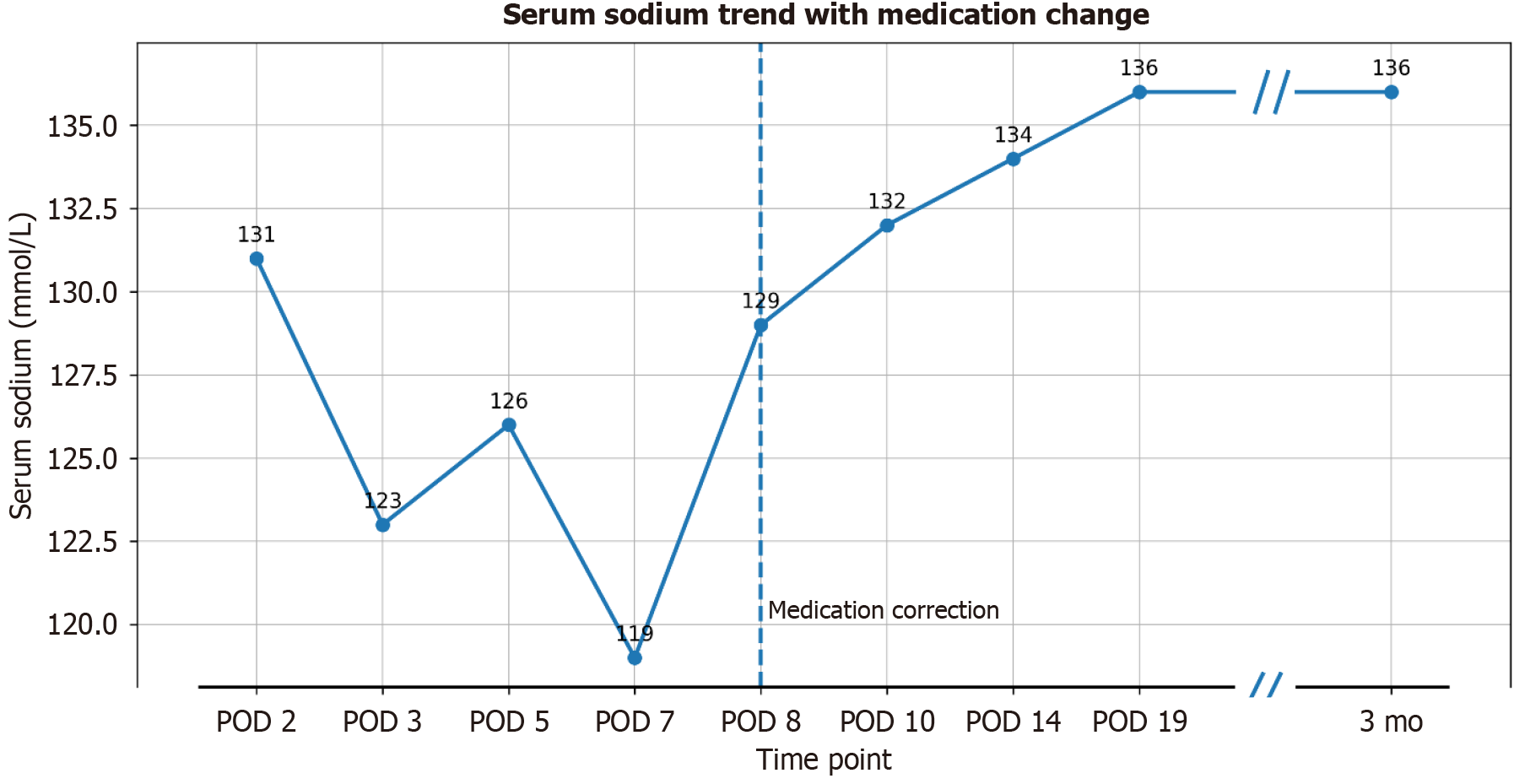
On POD 2, she developed increasing somnolence and cognitive slowing, and serum sodium was 131 mmol/L (Table 1). Intravenous isotonic saline was initiated. Serum sodium declined to 123 mmol/L on POD 3 despite supportive measures, and only partially improved to 126 mmol/L by POD 5 while symptoms persisted. On POD 7, sodium worsened to 119 mmol/L, prompting hypertonic saline therapy; sodium increased to 129 mmol/L the following day (Figure 2). A structured evaluation confirmed hypotonic hyponatremia with inappropriately concentrated urine and elevated urine sodium, normal thyroid and adrenal function, and clinical euvolemia, consistent with an SIADH-like physiology. Given the temporal relationship with CBZ initiation, CBZ-induced hyponatremia was considered the most likely etiology.
| Postoperative day | Laboratory findings |
| POD 1 | Control cranial CT satisfactory (no laboratory values reported) |
| POD 2 | Serum Na 131 mmol/L; K 3.6 mmol/L; creatinine 60 μmol/L; urea 19 mmol/L; glucose 86 mmol/L |
| POD 3 | Serum Na 123 mmol/L |
| POD 5 | Serum Na 126 mmol/L |
| POD 7 | Serum Na 119 mmol/L |
| POD 8 | Serum Na 129 mmol/L; serum osmolality 254 mOsm/kg; glucose 54 mmol/L; total protein 69 g/L; triglycerides 1.3 mmol/L; urea 22 mmol/L; creatinine 58 μmol/L; uric acid 150 μmol/L; urine osmolality 560 mOsm/kg; urine Na 68 mmol/L; urine K 28 mmol/L; urine specific gravity 1.020; morning cortisol 520 nmol/L; TSH 1.6 mIU/L; free T4 14 pmol/L |
| POD 10 (discharge) | Serum Na 132 mmol/L |
| POD 14 | Serum Na 134 mmol/L |
| POD 19 | Serum Na 136 mmol/L |
| 3-month follow-up | Serum Na 136 mmol/L |
Arterial hypertension.
No relevant personal habits or family history contributing to hyponatremia were reported.
On admission, the patient was clinically stable. During the hyponatremia episode, examination demonstrated stable hemodynamics without orthostatic hypotension, no peripheral edema, and preserved mucous-membrane turgor; there were no focal neurologic deficits aside from somnolence.
On POD 2, serum sodium was 131 mmol/L (reference 136-146 mmol/L). Other values were: White blood cells 9.5 × 109/L, erythrocytes 4.16 × 1012/L, hemoglobin 120 g/L, platelets 256 × 109/L, mean corpuscular volume 82.0 fL, mean corpuscular hemoglobin 28.1 pg, mean corpuscular hemoglobin concentration 34 g/dL, mean platelet volume 9.60 fL, red cell distribution width 12.9%, glucose 8.6 mmol/L, urea 1.9 mmol/L, creatinine 60 μmol/L, calcium 2.30 mmol/L, potassium 3.6 mmol/L, chloride 102 mmol/L, and magnesium 0.87 mmol/L. Serum sodium subsequently declined to 123 mmol/L (POD 3), rose to 126 mmol/L (POD 5), and reached a nadir of 119 mmol/L (POD 7), with partial correction to 129 mmol/L after hypertonic saline.
During diagnostic evaluation, serum osmolality was 254 mOsm/kg; glucose 5.4 mmol/L; total protein 69 g/L; triglycerides 1.3 mmol/L; urea 2.2 mmol/L; creatinine 58 μmol/L; uric acid 150 μmol/L. Urine studies showed urine osmo
Carbamazepine-induced hypotonic euvolemic hyponatremia with SIADH-like pathophysiology following meningioma surgery.
Initial management included intravenous isotonic saline (0.9% NaCl 500 mL twice daily), increased dietary sodium intake, and escalation to 0.9% NaCl three times daily after internal-medicine consultation. With progression to severe hyponatremia (Na 119 mmol/L) and persistent symptoms, hypertonic saline (3% NaCl 250 mL twice daily) was administered, producing partial correction.
Given the SIADH-like profile and temporal association with CBZ, CBZ was discontinued and levetiracetam was initiated with titration to 500 mg twice daily.
After CBZ withdrawal and antiseizure substitution, somnolence and generalized weakness regressed. Serum sodium was 132 mmol/L at discharge (POD 10). Outpatient monitoring demonstrated sustained normalization: 134 mmol/L on POD 14 and 136 mmol/L on POD 19. At three-month follow-up, serum sodium remained 136 mmol/L, with no recurrence of altered consciousness or cognitive impairment.
Postoperative hyponatremia is a frequent and clinically important complication in neurosurgical patients, most commonly related to SIADH or cerebral/central salt wasting syndrome (CSWS); distinguishing these entities is essential because management differs and overly rapid correction can cause osmotic demyelination syndrome[1].
Although CBZ-associated hyponatremia is well recognized, early presentation during the immediate postoperative period after intracranial tumor surgery is less frequently reported. A prior report described severe CBZ-related hyponatremia after meningioma surgery; our case adds to this limited literature by illustrating a similar complication when CBZ is used for routine postoperative seizure prophylaxis after meningioma resection, underscoring the need for early electrolyte surveillance in this setting.
CBZ-induced hyponatremia has been repeatedly documented in the literature (summarized in Table 2). Its presentations range from incidental laboratory abnormalities to severe, symptomatic encephalopathy and seizures. Early evidence from a retrospective cohort showed that hyponatremia can occur during CBZ therapy even outside an acute illness context, with the lowest reported sodium reaching 113 mEq/L in patients receiving CBZ alone, and an apparent mitigating effect when lithium was co-administered (higher mean sodium and fewer episodes)[5]. Subsequent case reports and series consistently reinforced that clinically meaningful hyponatremia may develop under CBZ exposure and can produce neurologic manifestations that mimic primary seizure recurrence or postoperative neurologic deterioration rather than an electrolyte disorder[6-8].
| Ref. | Country | Study design | Indication for CBZ: Dose/regimen | Sodium values after CBZ exposure | CBZ discontinued? | Hyponatremia treatment | Patient outcomes |
| Kuz and Manssourian[3], 2005 | United States | CR (n = 1) | Psychiatric illness (on CBZ chronically): CBZ 400 mg BID (morning/noon) + 600 mg HS; took 1200 mg the night before admission + 200 mg the next morning | Serum Na 122 mEq/L, corrected to 136 mEq/L within 24 hours | Yes (withheld) | 0.9% NaCl IV: 1-L bolus, then another liter at 125 mL/hour | Seizures associated with acute hyponatremia; sodium normalized; discharged with counseling about avoiding large CBZ doses |
| Kaeley et al[4], 2019 | India | CR (n = 1) | Focal seizures with secondary generalization; CBZ 600 mg/day (200 mg TID) | Na 118 mEq/L at presentation; then 122 (day 3), 125 (day 4), 135 (after 7 days) | Yes | Fluid restriction + tolvaptan 30 mg PO daily; switched to levetiracetam 500 mg TID for seizures | Asymptomatic/ambulatory; sodium corrected to 135 mEq/L on follow-up |
| Vieweg et al[5], 1987 | United States | Retro (n = 33) | Psychiatric diagnoses (e.g., organic affective disorder, schizoaffective disorder, major depression with psychosis, bipolar disorder) and seizure disorders (generalized + partial complex seizures). CBZ dose/regimen not reported; ≥ 2 weeks of therapy before labs | Mean serum Na: 138.4 ± 4.3 mEq/L (CBZ alone) vs 141.8 ± 1.6 mEq/L (CBZ + Li). Lowest Na reported: 113 mEq/L (CBZ alone; associated with generalized seizures) vs 132 mEq/L (CBZ + Li) | No | Not an acute treatment study; lithium coadministration was associated with “protection against hyponatremia” (higher Na; fewer episodes) | The CBZ + Li group had a higher mean Na and fewer hyponatremia episodes; severe low Na (113) in the CBZ-alone group was linked to generalized seizures |
| Sordillo et al[6], 1978 | United States | CR (n = 1) | Trigeminal neuralgia; CBZ 600 mg/day; the day before hospitalization, CBZ 1200 mg was used; additional phenytoin 300 mg | 118 mEq/L | No | Addition of phenytoin (suppression of ADH) | Three days after hospitalization, and phenytoin dosage correction value of sodium 139 mEq/L |
| Scoccia and Scommegna[7], 1988 | United States | CR (n = 1) | Seizure disorder (grand mal); CBZ 1200 mg (300 mg QID); clonazepam 0.5 mg | Serum Na 123 mEq/L on ER admission (serum osmolality 246 mOsm/kg H2O) | NR | IV saline + furosemide overnight; discussion describes hypertonic IV saline until mildly hyponatremic + furosemide to prevent volume expansion | Nausea/vomiting then grand mal seizure; electrolytes/osmolality corrected within 36 hours |
| Krysiak and Okopień[8], 2007 | Poland | CR (n = 1) | Facial neuralgia; CBZ 1200 mg/day (600 BID) | Na 126 mmol/L after starting CBZ (initial mild hyponatremia); Na 112 mmol/L on admission; 3% saline stopped when Na reached 125 mmol/L; Na normal by hospital day 5 | Yes (discontinued on admission) | Fluid restriction + 3% hypertonic saline infusion | Presented with cerebral edema symptoms (incl. seizures); stabilized with correction; 12 months later, the patient remained stable without recurrent hyponatremia |
| Palacios Argueta et al[9], 2018 | Guatemala | CR (n = 1) | Pain complaint (left knee pain; reason CBZ prescribed “unknown”, likely chronic pain); CBZ initially 300 mg (daily) then self-increased to 600 mg approximately 7 days prior (also prednisone) | Na 119 mmol/L (initial); Na 128 mmol/L at discharge (day 3); Na 138 mmol/L at 1-week follow-up | Yes | Fluid restriction (1.5 L/day) + correction of hypokalemia with IV potassium | Fatigue/cramps resolved by follow-up; sodium/osmolality normalized; no neurologic deficits reported |
| Fadili et al[10], 2019 | Morocco | CR (n = 1) | Schizoaffective disorder; started risperidone + CBZ with progressive dose increases; at 1 week: Risperidone 8 mg + CBZ 1200 mg | Na 128 mmol/L at 1 week; Na 119 mmol/L at day 10; after treatment Na corrected to 134 mmol/L | Yes (stopped both drugs) | Fluid restriction; ICU transfer for severe hyponatremia; 3% NaCl hypertonic saline with lab monitoring | Symptoms included fatigue/somnolence; sodium corrected; then switched to clozapine with stable sodium (on clozapine 400 mg at 8 months follow-up) |
| Kamiyama et al[11], 1993 | Japan | CR (n = 1) | Low back pain; CBZ 400 mg (200 mg BID), after rechallenge 600 mg | Serum Na 120 mmol/L persistent; rechallenge to 117 mmol/L (day 2) | Yes | Mild water restriction (initially); diagnostic/treatment infusion of hypertonic saline (10% NaCl) described during evaluation | Sodium normalized after stopping CBZ; rechallenge reproduced acute symptomatic hyponatremia; authors diagnosed partial central diabetes insipidus (based on water restriction/desmopressin response) |
| Tanaka et al[12], 2008 | Japan | CR (n = 1) | Temporal lobe epilepsy; CBZ started 400 mg/day, then increased to 800 mg/day (treated approximately 6 years). Concurrent meds included warfarin + bucolome; hyponatremia developed after starting interferon-α2b + ribavirin | Blood Na 124 mEq/L (week after admission) | No (CBZ continued; adjunctive strategies used) | Water intake restriction (20 mL/kg) + dimethylchlortetracycline 900 mg/day; later lithium 400 mg/day (maintained approximately 1 year) | Hyponatremia improved; after interferon-α2b stopped, hyponatremia completely recovered by 6 months; later, Na 137 mEq/L reported |
| McCauley and Thiraviaraj[13], 2017 | United Kingdom | CR (n = 1) | Epilepsy (also myasthenia gravis; seizure-free for years); carbamazepine Tegretol-PR 400 mg BID (also on omeprazole + fluoxetine initially) | Sodium persistently 122-128 mmol/L despite treatment (per report summaries) | No (strong reluctance/“unable to forego” carbamazepine) | Stopped omeprazole + fluoxetine; fluid restriction; demeclocycline 300 mg BID (ineffective + worsened myasthenia); tolvaptan 15 mg twice weekly (marginal effect; thirst/nausea) | Clinically euvolemic and asymptomatic throughout; chronic refractory hyponatremia considered a “reset osmostat” baseline; elective thymectomy deferred, but authors argue surgery could proceed with closer peri-op surveillance |
| Ponte et al[14], 1990 | United States | CR (n = 1) | Postherpetic neuralgia; CBZ 600 mg/day (300 mg BID) | Na 133 mmol/L at discharge after starting CBZ; later Na 120 mmol/L on readmission; after approximately 4 days fluid restriction Na rose to 131 mmol/L (also 131 at discharge) | Yes | Fluid restriction; concomitant HCTZ/triamterene was discontinued | Symptoms (nausea/vomiting, ataxia/disequilibrium, syncope) improved after stopping CBZ; after fluid restriction, sodium/electrolytes normalized; discharged ambulatory with minimal residual ataxia; electrolytes normal at follow-up |
| Joseph et al[15], 2023 | United States | CR (n = 1) | Bipolar affective disorder; CBZ 600 mg (300 mg BID) (+ lorazepam 2 mg daily) | Serum Na 104 mmol/L on admission; 136 mmol/L by discharge | Yes | 3% saline for 24 hours (rate approximately 0.5 mEq/L/hour) + supportive therapy; later ICU care with ventilatory/supportive management | Improved and discharged day 5 after initial correction; returned approximately 2 weeks later with neurologic deficits and imaging consistent with osmotic demyelination; became conscious day 7 but rigidity/hyperreflexia persisted; discharged to ongoing rehab with follow-up |
| Inamura et al[16], 1999 | Japan | CS (n = 2) | Case 1 epilepsy; CBZ 600 mg/day → 1000 mg/day (with phenobarbital + phenytoin). Case 2 Facial pain after meningioma surgery; CBZ 400 mg/day (with valproate) | Case 1 Na 128 mmol/L. Case 2 Na 116 mmol/L | Yes (rapid improvement after withdrawal) | Case 1 CBZ withdrawal. Case 2 CBZ withdrawal + water restriction | Case 1 symptoms resolved promptly; Na corrected within approximately 3 days. Case 2 symptoms improved; Na corrected to approximately 138 mmol/L within approximately 3 days |
| Ballardie and Mucklow[17], 1984 | United Kingdom | CR (n = 1) | Epilepsy; CBZ 1100 mg/day; evaluated before/after demeclocycline | Hyponatremia episodes mentioned; sodium value 121 mmol/L | No (“partial reversal” strategy) | Demeclocycline added (1200 mg QID) | Water handling/osmolality improved, but not normalized |
| Brewerton and Jackson[18], 1994 | United States | CS (n = 6) | Psychiatric inpatients; CBZ rechallenge after normalization | Hyponatremia occurred on CBZ | Initially, yes (Na normalized after CBZ discontinuation), then CBZ rechallenged | Demeclocycline is used to prevent recurrent Na decline in 5/6 | Prevention was successful in 5/6 on rechallenge |
| Pham et al[19], 2021 | United States | CR (n = 1) | Bipolar illness/vascular dementia; recurrent CBZ-associated SIADH; CBZ dose N/A | Hyponatremia on two CBZ trials (numeric Na N/A) | No (strategy aimed to allow continued CBZ) | Low-dose lithium to correct/maintain Na | Prompt normalization was maintained for approximately 8 weeks |
| Morimoto et al[21], 2020 | Japan | CS (n = 3) | Localization-related epilepsy. Case 1 (A): Carbamazepine 300 mg/day; replaced with lacosamide 400 mg/day. Case 2 (B): Carbamazepine 400 mg/day; replaced with lacosamide 200 mg/day. Case 3 (C): Carbamazepine 800 mg/day; replaced with lacosamide 400 mg/day | Mean Na (mEq/L): On carbamazepine/after replacement - A: 132.0 ± 2.82/137.0 ± 2.83; B: 131.5 ± 0.70/138.5 ± 0.70; C: 133.0 ± 1.41/136.0 ± 1.41 | Yes | Gradual switch over approximately 3 months (taper carbamazepine while titrating lacosamide) | Serum sodium improved significantly in all 3; seizure frequency did not change substantially |
| Prakash et al[22], 2016 | India | CR (n = 1) | Epilepsy; “taking carbamazepine regularly” for 34 years (dose/regimen not reported), planned for post-burn debridement and skin grafting | Na 128 mEq/L, repeat 129 mEq/L; normalized pre-op to 142 mEq/L | No (continued; not stopped peri-op) | Increased oral salt intake + fluid restriction | Uneventful anesthesia and recovery after Na normalized; advised to consult a physician about drug-induced hyponatremia |
In our patient, hypotonic hyponatremia developed shortly after CBZ initiation for postoperative seizure prophylaxis, progressed despite isotonic saline, and resolved promptly after CBZ withdrawal and substitution with levetiracetam. Several reports describe hyponatremia emerging within days after CBZ initiation or dose escalation, supporting a relatively rapid onset in susceptible individuals. Sordillo et al[6] reported sodium of 118 mEq/L in the setting of trigeminal neuralgia with CBZ exposure and recent dose escalation, while Palacios Argueta et al[9] described sodium 119 mmol/L after self-increasing CBZ to 600 mg, with improvement after discontinuation and supportive measures. Fadili et al[10] documented progressive decline from 128 mmol/L to 119 mmol/L within roughly 10 days during titration to CBZ 1200 mg in a psychiatric patient, again with recovery after stopping the causative drugs. The reproducibility of the phenomenon was particularly strengthened by Kamiyama et al[11], who observed normalization after stopping CBZ and then recurrence to 117 mmol/L shortly after rechallenge, establishing a clear causal relationship. Although some cases reflect chronic exposure with later decompensation, such as long-term epilepsy treated for years before clinically significant hyponatremia was recognized, these reports still attribute the sodium derangement to CBZ-associated antidiuresis and show improvement with targeted strategies[12,13].
The biochemical changes described across the cases are consistent with an SIADH-like pathophysiology (Table 2), and our diagnostic profile fits squarely within that spectrum. Scoccia and Scommegna[7] reported low serum osmolality (246 mOsm/kg) with hyponatremia (Na 123 mEq/L) in a patient presenting with nausea/vomiting and seizure activity while on high-dose CBZ, consistent with impaired free-water excretion. Kamiyama et al[11] similarly described hypotonic hyponatremia that recurred rapidly on rechallenge, and several other reports treated the disorder explicitly as SIADH with water restriction and/or hypertonic saline when severe[8,10,14,15]. Importantly, the clinical context in our case - early postoperative neurosurgical recovery - creates a high-risk environment for misattribution, because somnolence and cognitive slowing can be interpreted as a surgical complication or cerebral edema unless electrolytes are checked promptly. In this sense, the temporal association between CBZ initiation and sodium decline becomes a critical diagnostic clue, as demonstrated across reports where symptoms improved only after the drug was stopped[8-10,14,16].
In neurosurgical patients, the key competing diagnoses for hypotonic hyponatremia with high urine sodium and inappropriately concentrated urine are SIADH and CSWS[1]. The distinction is clinically important because SIADH is characterized by relative water excess, while CSWS reflects renal sodium loss with effective circulating volume depletion. Hypertonic saline may be required for severe symptomatic hyponatremia in either condition; therefore, its use does not, by itself, confirm CSWS (Table 3)[1].
| Feature | SIADH | CSWS | Findings in this case |
| Volume status | Euvolemia or mild hypervolemia | Hypovolemia (effective volume depletion) | Clinically euvolemic; stable hemodynamics; no orthostasis |
| Urine output | Normal to low (variable) | Often increased (polyuria) | No polyuria documented; no negative fluid balance reported |
| Serum osmolality | Low | Low | Low (254 mOsm/kg) |
| Urine osmolality | Inappropriately high (> 100 mOsm/kg) | Often high | High (560 mOsm/kg) |
| Urine sodium | > 30-40 mmol/L | > 40 mmol/L | High (68 mmol/L) |
| Response to isotonic saline | Often minimal or may worsen | Improves sodium/volume | Sodium continued to decline despite 0.9% NaCl |
| Uric acid/FEurate | Often low uric acid; FEurate elevated (may normalize after correction) | Often low uric acid; FEurate elevated and may persist | Low uric acid (150 μmol/L) |
| Key therapeutic approach | Fluid restriction, treat trigger; hypertonic saline if severe symptoms | Volume and salt repletion, optional fludrocortisone, hypertonic saline if severe symptoms | Trigger removed (CBZ stopped) and hypertonic saline; sodium normalized |
In the present case, CSWS was considered less likely because the patient remained clinically euvolemic with stable hemodynamics and no orthostatic hypotension or clear evidence of dehydration, and serum sodium continued to decline despite isotonic saline administration; taken together with hypotonic hyponatremia, inappropriately concentrated urine, normal thyroid/adrenal function, and improvement after CBZ withdrawal, this supported a drug-induced SIADH-like mechanism[1].
Proposed mechanisms of CBZ-associated hyponatremia include enhanced vasopressin (AVP) action and impaired renal free-water excretion, resulting in an SIADH-like biochemical profile[2,3,6]. Postoperative factors (pain, nausea, stress, and perioperative fluids) may further increase AVP activity, potentially lowering the threshold for clinically significant hyponatremia in susceptible (often older) patients[1].
Management strategies summarized in Table 2 converge on CBZ discontinuation as the most consistently effective intervention, with supportive therapies tailored to severity and feasibility of stopping the drug. Several reports describe rapid correction of sodium and symptoms following withdrawal alone or with modest adjuncts, including Inamura et al[16], where both patients improved quickly after stopping CBZ, and in the post-meningioma case, sodium corrected to approximately normal within about three days. Ponte et al[14] documented improvement after CBZ cessation and fluid restriction, with symptomatic recovery and normalization of electrolytes on follow-up. Krysiak and Okopień[8] reported a severe presentation (nadir 112 mmol/L) requiring hypertonic saline and fluid restriction, with normalization by hospital day five after CBZ discontinuation. Similar approaches were used in ICU-level cases, including Fadili et al[10] and Joseph et al[15], where hypertonic saline was required for severe hyponatremia and neurologic symptoms, with Joseph et al[15] additionally illustrating the hazard of overly rapid correction through subsequent osmotic demyelination despite initial biochemical improvement. When CBZ could not be stopped or when rechallenge was pursued, alternative pharmacologic strategies were described, including demeclocycline to blunt antidiuresis in a partial-reversal or prevention approach and lithium used as a protective or corrective agent, aligning with Vieweg et al’s observational finding[5] and later case-based experiences aiming to preserve CBZ therapy[17-19]. Tolvaptan was used in selected cases with chronic or persistent hyponatremia, though results ranged from meaningful improvement to only marginal benefit[13].
CBZ was selected for postoperative seizure prophylaxis based on local practice and availability; however, the subsequent rapid sodium decline prompted reassessment. The drug was continued briefly while supportive measures were escalated and diagnostic work-up was completed, but once severe symptomatic hyponatremia with an SIADH-like profile was evident, CBZ was discontinued and replaced with levetiracetam[4,20].
Hypertonic saline and correction safety: When serum sodium reached 119 mmol/L with persistent neurocognitive symptoms, 3% NaCl was administered (250 mL twice daily) with close biochemical monitoring to avoid overly rapid correction and osmotic demyelination[15]. No clinical features suggestive of osmotic demyelination occurred during hospitalization or follow-up.
Our report is particularly aligned with the neurosurgical-relevant precedent provided by Inamura et al[16], who described CBZ-induced severe hyponatremia after meningioma surgery in a patient receiving CBZ for postoperative facial pain. That case and ours converge on several key points: Relatively low-to-moderate CBZ dosing can still pre
Plasma AVP (or copeptin) was not measured during the episode, which is a limitation. In routine practice, AVP/copeptin testing is often unavailable; therefore, diagnosis relies on clinical volume assessment, exclusion of endocrine causes, urine osmolality/sodium, and the response to removing the suspected trigger[1]. Future reports could incorporate AVP/copeptin where feasible to strengthen mechanistic attribution.
In conclusion, literature supports that CBZ can induce a reversible, SIADH-like hyponatremia across diverse indications and patient profiles, and that the safest and most reliable corrective step is withdrawal of CBZ when clinically feasible. The neurosurgical setting deserves special attention because postoperative neurologic symptoms can obscure early recognition and because hyponatremia has multiple competing etiologies after intracranial surgery; the rare but directly relevant precedent after meningioma surgery, combined with the consistent reversibility seen across reports, argues for low thresholds to monitor sodium soon after CBZ initiation and to promptly substitute CBZ if sodium declines or neurocognitive symptoms emerge.
| 1. | Hannon MJ, Thompson CJ. Neurosurgical Hyponatremia. J Clin Med. 2014;3:1084-1104. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 31] [Article Influence: 2.6] [Reference Citation Analysis (1)] |
| 2. | Dong X, Leppik IE, White J, Rarick J. Hyponatremia from oxcarbazepine and carbamazepine. Neurology. 2005;65:1976-1978. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 113] [Article Influence: 5.4] [Reference Citation Analysis (1)] |
| 3. | Kuz GM, Manssourian A. Carbamazepine-induced hyponatremia: assessment of risk factors. Ann Pharmacother. 2005;39:1943-1946. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 43] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 4. | Kaeley N, Kabi A, Bhatia R, Mohanty A. Carbamazepine-induced hyponatremia - A wakeup call. J Family Med Prim Care. 2019;8:1786-1788. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 5. | Vieweg V, Glick JL, Herring S, Kerler R, Godleski LS, Barber J, Yank G, Spradlin W. Absence of carbamazepine-induced hyponatremia among patients also given lithium. Am J Psychiatry. 1987;144:943-947. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 18] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 6. | Sordillo P, Sagransky DM, Mercado RM, Michelis MF. Carbamazepine-induced syndrome of inappropriate antidiuretic hormone secretion. Reversal by concomitant phenytoin therapy. Arch Intern Med. 1978;138:299-301. [PubMed] [DOI] [Full Text] |
| 7. | Scoccia B, Scommegna A. Carbamazepine-induced hyponatremia after transabdominal follicular ultrasound examination. Fertil Steril. 1988;50:984-985. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 3] [Article Influence: 0.1] [Reference Citation Analysis (1)] |
| 8. | Krysiak R, Okopień B. [Carbamazepine-induced hyponatremia]. Pol Arch Med Wewn. 2007;117:73-75. [PubMed] [DOI] [Full Text] |
| 9. | Palacios Argueta PJ, Sánchez Rosenberg GF, Pineda A. Walking hyponatremia syndrome of inappropriate antidiuretic hormone secretion secondary to carbamazepine use: a case report. J Med Case Rep. 2018;12:202. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 10. | Fadili A, Attouche N, Charra B, Alami KM, Agoub M. [Risperidone and carbamazepine-induced syndrome of inappropriate antidiuretic hormone secretion: case study]. Pan Afr Med J. 2019;32:78. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (1)] |
| 11. | Kamiyama T, Iseki K, Kawazoe N, Takishita S, Fukiyama K. Carbamazepine-induced hyponatremia in a patient with partial central diabetes insipidus. Nephron. 1993;64:142-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 4] [Article Influence: 0.1] [Reference Citation Analysis (1)] |
| 12. | Tanaka M, Kamoi K, Takahashi T. Interferon-alpha is a predisposing risk factor for carbamazepine-induced hyponatremia: A case of syndrome of inappropriate antidiuresis caused by interferon-alpha therapy. Int J Gen Med. 2008;1:21-25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (1)] |
| 13. | McCauley S, Thiraviaraj A. Pre-operative carbamazepine-induced hyponatraemia: Should we accept a reset hypo-osmolar baseline and proceed with surgery? Ulster Med J. 2017;86:123-124. [PubMed] |
| 14. | Ponte CD, Pollard S, Dattola RK. A suspected case of carbamazepine-induced hyponatremia. J Fam Pract. 1990;31:664, 666-667. [PubMed] |
| 15. | Joseph A, Sayeed T, Patel DK, Aiyadurai S, Shahbaz Z, Mettela SR, Garg T, Gadde R, Udoeyop D, Khan A. Central Pontine Myelinolysis With Carbamazepine-Induced Syndrome of Inappropriate Antidiuretic Hormone and Its Management: A Case Report and Literature Review. Cureus. 2023;15:e35816. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 16. | Inamura T, Kuba H, Morioka T, Muratani H, Muraishi M, Hisada K, Fukui M. [Carbamazepine induced hyponatremia]. No Shinkei Geka. 1999;27:85-87. [PubMed] |
| 17. | Ballardie FW, Mucklow JC. Partial reversal of carbamazepine-induced water intolerance by demeclocycline. Br J Clin Pharmacol. 1984;17:763-765. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.1] [Reference Citation Analysis (1)] |
| 18. | Brewerton TD, Jackson CW. Prophylaxis of carbamazepine-induced hyponatremia by demeclocycline in six patients. J Clin Psychiatry. 1994;55:249-251. [PubMed] |
| 19. | Pham VM, White JM, Gunther SR, Stewart JT. Lithium for Carbamazepine-Induced Syndrome of Inappropriate Antidiuretic Hormone. Clin Neuropharmacol. 2021;44:33-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (1)] |
| 20. | Pugonja R, Bečulić H, Begagić E, Skomorac R, Selimović E, Moranjkić M. Morphometric and Ki-67 proliferative index-related characteristics of meningiomas and their correlation with demographic, clinical, histopathological, and postoperative features. Med Glas (Zenica). 2024;21:132-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 21. | Morimoto M, Suzaki I, Shimakawa S, Hashimoto T, Nakatsu T, Hamada S, Kyotani S. Three cases in which drug-induced hyponatremia was improved by replacing carbamazepine with lacosamide. Clin Case Rep. 2020;8:1166-1170. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 22. | Prakash S, Bhatia PS, Raheja SG, Pawar M. Carbamazepine-induced hyponatremia. Brit J Anaesth. 2016;117:el_13266. [DOI] [Full Text] |