Published online Mar 26, 2026. doi: 10.12998/wjcc.v14.i9.118210
Revised: February 1, 2026
Accepted: March 5, 2026
Published online: March 26, 2026
Processing time: 87 Days and 21 Hours
Chronic lymphocytic leukemia (CLL) is characterized by progressive humoral immune dysfunction, particularly hypogammaglobulinemia, leading to increased infection risk. While immunoglobulin replacement therapy (IgRT) is commonly used for infection prevention, its efficacy remains debated due to conflicting evidence.
To evaluate the association between IgRT and infection outcomes in patients with CLL by synthesizing evidence from randomized controlled trials (RCTs) and observational studies, using complementary dichotomous and rate-based meta-analytic approaches.
We conducted a systematic review and meta-analysis (International Prospective Register of Systematic Reviews-registered, Preferred Reporting Items for Sys
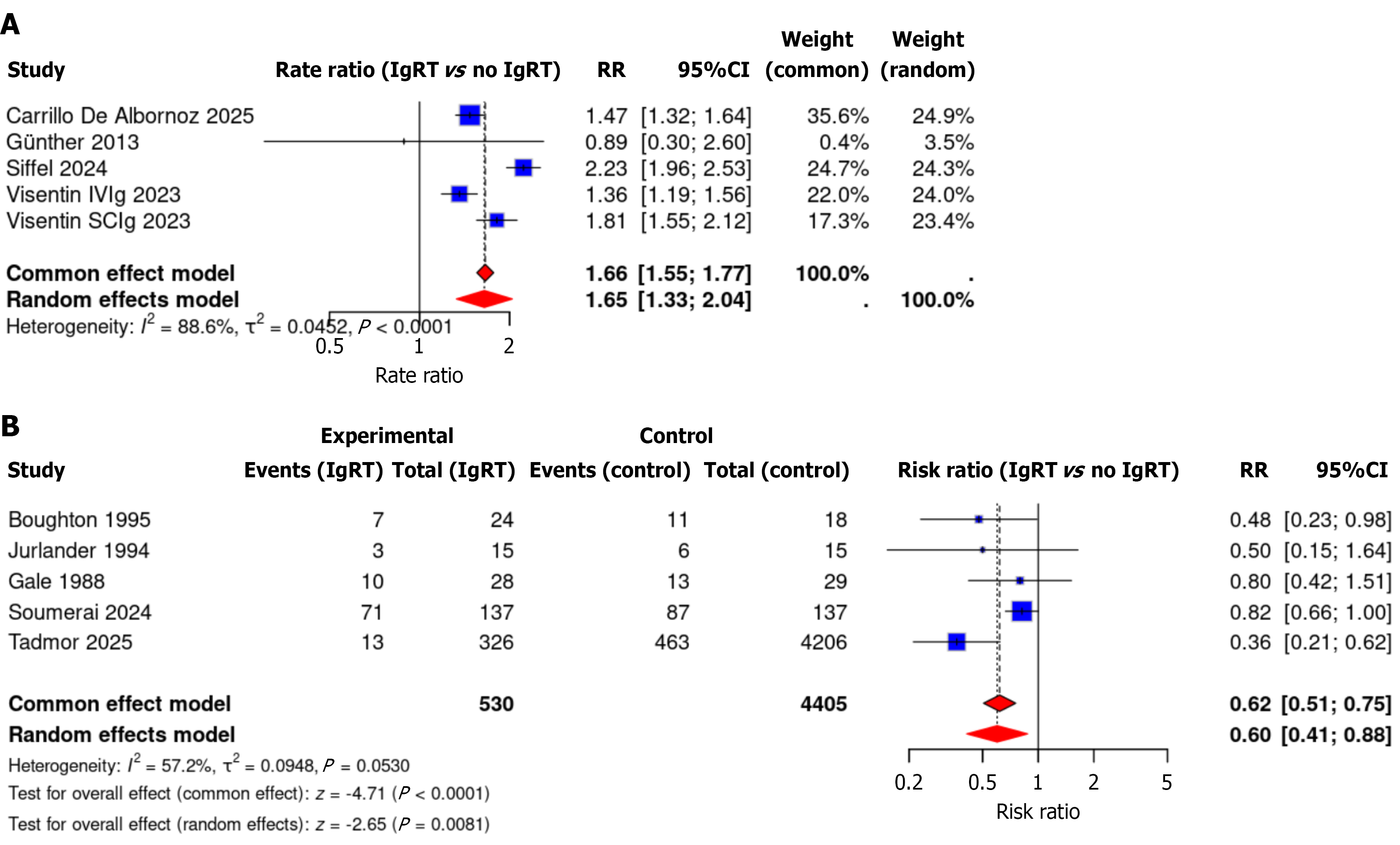
Nine studies were included in our meta-analysis. The dichotomous analysis (5 studies) showed a 40% reduction in infection risk with IgRT (RR = 0.60, 95% confidence interval: 0.41-0.88, P = 0.008; I2 = 57.2%). Conversely, rate-based analysis (5 studies) revealed a 65% increase in recurrent infections (rate ratio: 1.65, 95% confidence interval: 1.33-2.04, P < 0.001; I2 = 88.6%). Subgroup analyses demonstrated consistent effects across intravenous immunoglobulin (RR = 0.61) and subcutaneous immunoglobulin (RR = 0.52) for
IgRT reduces initial infections in CLL patients but may not prevent recurrences and could paradoxically increase infection rates in some settings. These findings support selective use in high-risk patients (such as patients with severe hypogammaglobulinemia) while highlighting the need for standardized outcome reporting and studies evaluating IgRT alongside modern therapies (e.g., Bruton’s tyrosine kinase inhibitors).
Core Tip: Immunoglobulin replacement therapy (IgRT) is widely used to prevent infections in patients with chronic lym
- Citation: Kalamara TV, Dodos K, Georgakopoulou VE. Immunoglobulin replacement therapy and infection risk in chronic lymphocytic leukemia: A systematic review and meta-analysis. World J Clin Cases 2026; 14(9): 118210
- URL: https://www.wjgnet.com/2307-8960/full/v14/i9/118210.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i9.118210
Chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma is a common adult B-cell malignancy of unknown etiology, characterized by clonal accumulation of small, mature-appearing but immunologically dysfunctional B lymphocytes in the blood, bone marrow, and lymphoid tissues[1]. CLL refers to disease with peripheral blood involvement (leukemic phase), whereas small lymphocytic lymphoma is characterized predominantly by nodal disease (lymphoma phase). The disorder typically follows an indolent course, and many patients are managed with a watch-and-wait strategy in early stages; however, progressive disease often requires targeted therapies such as Bruton’s tyrosine kinase inhibitors or B-cell lymphoma-2 inhibitors[1].
A central feature of CLL is progressive immune dysfunction, most notably humoral immunodeficiency manifesting as hypogammaglobulinemia, with reduced levels of immunoglobulin G (IgG), particularly IgG3 and IgG4 subclasses. This is accompanied by impaired function of nonclonal CD5-negative B cells, T lymphocytes, natural killer cells, neutrophils, and complement pathways. As a result, patients have a diminished ability to mount effective antibody responses, leading to recurrent and severe infections[2]. Infection-related complications account for a substantial proportion of morbidity and mortality in CLL, contributing to approximately 30%-50% of deaths[3].
Standard infection-prevention strategies include vaccination against seasonal influenza, Streptococcus pneumoniae, and Haemophilus influenzae[4], as well as antibiotic prophylaxis in patients with symptomatic antibody deficiency[4,5]. Despite these measures, residual infection risk remains high in patients with profound hypogammaglobulinemia and recurrent infections[4]. In this context, immunoglobulin replacement therapy (IgRT), administered intravenously as intravenous immunoglobulin (IVIg) or subcutaneously as subcutaneous immunoglobulin (SCIg), has emerged as a passive immunotherapeutic approach. IVIg, derived from pooled donor plasma, provides broad-spectrum antibodies, including natural antibodies, and is widely used in primary and secondary immunodeficiencies, autoimmune disorders, and CLL-asso
Multiple randomized and crossover studies have demonstrated a reduction in infection rates with IVIg in CLL patients with hypogammaglobulinemia. In a landmark randomized controlled trial (RCT) of 84 patients, IVIg administered at 400 mg/kg every three weeks for one year reduced bacterial infections by 45% compared with placebo, with a 61% reduction among patients completing the full treatment course[6]. Earlier crossover studies using lower doses (e.g., 300 mg/kg every four weeks) similarly showed significant decreases in infection episodes during IVIg prophylaxis[5,7].
These findings have been supported by meta-analyses. A pooled analysis of nine randomized trials in CLL and mul
More recent real-world studies suggest that the effectiveness of IgRT may depend on structured immunological monitoring. A large observational study from Mass General Brigham reported fewer infections and reduced antimicrobial use among CLL and non-Hodgkin lymphoma patients undergoing regular IgG monitoring and receiving IgRT[10]. However, emerging data warrant caution. A 2025 Australian cohort study found no reduction in hospitalizations for severe infections among CLL patients receiving regular IgRT, and in some cases observed higher infection rates during treatment periods[11].
CLL-associated hypogammaglobulinemia confers a substantial risk of infection. IVIg provides broad antibody supplementation and has demonstrated efficacy in reducing infection incidence in selected patient populations, yet its clinical benefit remains debated. Importantly, most previous studies have evaluated infections as a composite outcome without distinguishing between first infectious episodes and recurrent infections, despite the distinct clinical implications of these entities for disease burden, healthcare utilization, and patient prognosis. The heterogeneity of existing studies, small sample sizes, and evolving therapeutic landscapes further limit definitive conclusions. A systematic review and meta-analysis are therefore warranted to synthesize available evidence, specifically addressing this methodological gap, quantify the impact of IVIg on both first and recurrent infection outcomes, and clarify its optimal role in the management of patients with CLL.
This systematic review and meta-analysis were conducted in accordance with the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines[12]. The review protocol was registered on the International Prospective Register of Systematic Reviews (No. CRD420251273640).
A search was conducted for studies enrolling CLL patients of any age. The included studies could be RCTs, prospective non-randomized trials, crossover or retrospective studies. The intervention group included CLL patients receiving IgRT compared to CLL patients not receiving IgRT, assessing the incidence of infections of any type. We excluded case reports, case series, former meta-analyses, editorials, opinion papers, and narrative reviews.
A comprehensive literature search was conducted to identify studies evaluating immunoglobulin-based strategies in the context of infection among patients with CLL. The PubMed/MEDLINE search was performed from database inception to August 12, 2025, using a combination of controlled vocabulary [Medical Subject Headings (MeSH)] and free-text terms for three key concepts: CLL, infection, and immunoglobulin therapy. Specifically, the CLL concept was captured using “chronic lymphocytic leukemia” (MeSH) and the corresponding free-text term (“chronic lymphocytic leukemia”); infection was captured using “infection” (MeSH) and the free-text term (“infection”); and immunoglobulin therapy was captured using “immunoglobulin” (MeSH) and free-text terms (“immunoglobulin”, “immunoglobulin replacement treatment”, and “gammaglobulin”). These concepts were combined using Boolean operators (OR within concepts; AND across concepts). The final PubMed search string was: {[“Chronic lymphocytic leukemia” (MeSH terms) OR “Chronic lymphocytic leukemia”] AND [“Infection” (MeSH terms) OR “Infection”]} AND {[“immunoglobulin” (MeSH terms) OR “immunoglobulin”] OR “immunoglobulin replacement treatment” OR “gammaglobulin”}, yielding 1079 records. In parallel, additional searches were undertaken in Cochrane Central Register of Controlled Trials, Scopus (articles only), and Google Scholar (screening the first 800 results) using equivalent keyword strategies adapted to each database’s syntax: (“Chronic lymphocytic leukemia” OR “Chronic lymphocytic leukaemia” OR “CLL”) AND “infection” AND (“immunoglobulin” OR “intravenous immunoglobulin” OR “gammaglobulin” OR “immunoglobulin replacement treatment”).
Study selection and data extraction were performed by two reviewers (Kalamara TV and Dodos K) independently. All articles from the electronic searches were assessed and citations that met the initial predefined selection criteria were obtained. Following deduplication, the remaining reports were reviewed at a title and abstract level by the two reviewers and all potentially eligible studies were full text assessed. Relevant information was extracted and recorded on a data collection form developed in Microsoft Excel©. Extracted information included the following: First author, year of study conduction, country of origin, design, time period of the study, study sample size, population (number of CLL patients and inclusion criteria in the IgRT group), type of intervention (IgRT type, timing, dosage), comparator, key clinical outcome (incidence of infections). Any disagreements between the two reviewers at any stage were resolved by discussion, consensus or arbitration by a third senior reviewer (Georgakopoulou VE).
Two authors (Kalamara TV and Dodos K) independently evaluated the risk of bias for each study using the tools described in the Cochrane Handbook for Systematic Reviews of Interventions, more specifically the Cochrane risk of bias 2 (RoB 2) tool for RCTs and the risk of bias in nonrandomized studies of interventions version I (ROBINS-I) tool for observational studies[13]. For RCTs the assessment covered five domains: Randomization process, deviations from intended interventions, missing outcome data, measurement of the outcome, and selection of the reported result. Each domain was rated as “low risk”, “high risk”, or “unclear risk” of bias. Similarly, for observational studies ROBINS-I was applied across seven domains: Confounding, selection of participants, classification of interventions, deviations from intended interventions, missing data, measurement of outcomes, and selection of the reported result. For selective reporting, study protocols were reviewed when available, while in their absence, the risk was judged as unclear. The results of the independent assessments were compared, and divergent views among reviewers were settled through discussion with a third senior reviewer (Georgakopoulou VE).
The primary outcome of infection incidence was analyzed using two complementary approaches to comprehensively assess the impact of (IgRT). For studies reporting recurrent infection data, we performed a rate-based analysis calculating rate ratios with 95% confidence intervals (CIs) for infections per patient-year. For studies reporting binary outcomes, we conducted a dichotomous analysis computing risk ratios (RRs) with 95%CI for the proportion of patients experiencing one or more infections. Several included observational studies employed retrospective or before-after designs to evaluate infection outcomes before and after initiation of IgRT. It is to be acknowledged that such designs are susceptible to time-related biases, including regression to the mean, immortal time bias, and changes in infection risk over time unrelated to the intervention. Formal adjustment for these biases was not feasible at the meta-analysis level due to heterogeneity in study design, follow-up duration, and reporting of pre- and post-intervention periods.
To address these limitations, time-related bias and regression to the mean were explicitly considered within the risk-of-bias assessment, particularly under the domains of confounding, participant selection, and outcome measurement. Studies with before-after designs or insufficient temporal adjustment were therefore more likely to be rated as having moderate or serious risk of bias. These considerations informed both the interpretation of the rate-based analyses and the emphasis placed on randomized and contemporaneous comparator evidence when drawing conclusions.
For sparse data in dichotomous analyses, we applied a 0.5 continuity correction to zero-event cells. Rate ratios were pooled from study-specific infection rates (events/patient-year), using the same correction for zero-event arms. The restricted maximum-likelihood estimator was used for all random-effects models, chosen for its superior performance in estimating between-study variance with limited studies. All meta-analyses were performed using both fixed-effect and random-effects models, employing Mantel-Haenszel weighting for dichotomous data and inverse-variance weighting for rate-based data. The random-effects results were prioritized in our interpretation due to anticipated clinical and methodological variability across studies. The I2 statistic was used for the evaluation of statistical heterogeneity. Forest plots were created for a graphical representation of the effect estimate[13]. All analyses were performed at the 0.05 level of sig
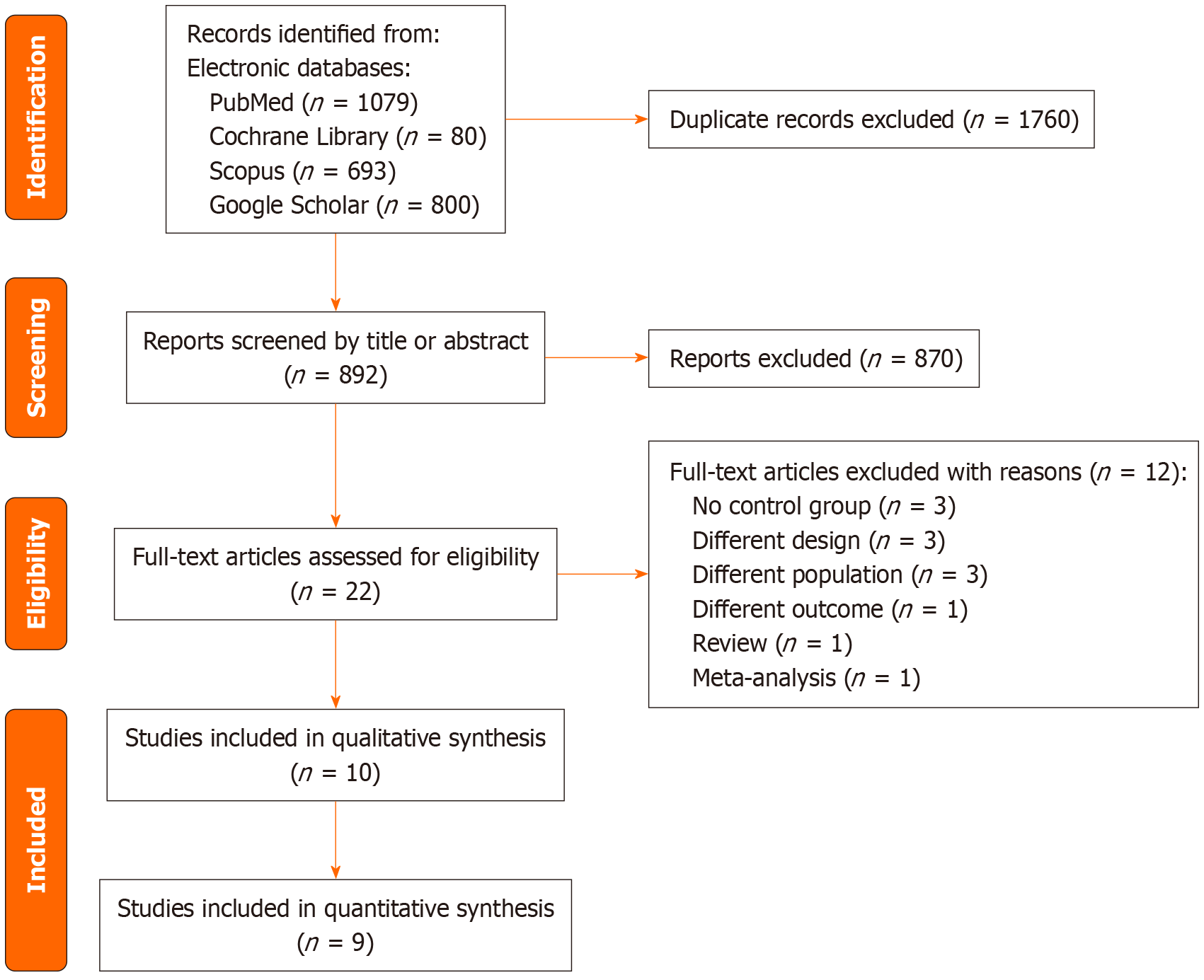
As shown in the corresponding PRISMA flow diagram (Figure 1), our search strategy retrieved 2652 results in total. After deduplication, we initially screened 892 records at the title and abstract level. Finally, we assessed 22 records in full text. Ten of them were eligible for inclusion in the qualitative synthesis[7,10,11,15-21]. Of these, 9 studies were included in our quantitative synthesis[10,11,15-21]. Twelve studies[8,22-32] were excluded for various reasons. Three studies were excluded due to the lack of a control group[22-24]. The results presented by one study[25] were also excluded, because it assessed a different outcome (levels of pneumococcal antibodies). Three studies, designed for IgRT dosage testing, were not included in our synthesis[26-28]. Moreover, three studies were excluded, as they examined a different population[29-31]. Finally, the study by Raanani et al[8] was excluded as meta-analysis and the study by Morell and Barandun[32], was excluded as review.
A detailed description of participants’ baseline characteristics is provided in Table 1. A total of 9 studies met the inclusion criteria of our meta-analysis, comprising RCTs, prospective cohorts, retrospective cohorts, and before-after IgRT observational designs. Two early RCTs[15,17], compared IVIg with placebo over 12 months, reporting infection events as binary outcomes (patients with ≥ 1 infection). In the study, 35.7% (10/28) of IVIg patients developed infections compared with 44.8% (13/29) in the placebo group[17]. Similarly, in Boughton et al[15], 29.2% (7/24) of IVIg patients experienced infections vs 61.1% (11/18) of those receiving albumin placebo. Several observational studies evaluated infection burden prior to and during IgRT, with patients receiving IgRT serving as their own controls. Jurlander et al[16] reported a re
| Ref. | Study design | Setting | Intervention vs comparator | Number of participants IgRT/non-IgRT | IgRT administration and indications | Outcome | Incidence of infections |
| Boughton et al[15] | Prospective, double-blind RCT | Multicenter, United Kingdom, 12-month duration | IVIg vs albumin placebo | IVIg: 24/18 (placebo) | 18 g IVIg every 3 weeks in patients with serum IgG levels < 5.5 g/L and a history of two or more recent infections | Number of patients experiencing infections and severe infections whilst treated with IVIg or albumin placebo | IVIg group: 7 failures1/24 albumin placebo: 11 failures/18 |
| Carrillo de Albornoz et al[11] | Retrospective cohort | Australia, January 2008 to December 2022, 12-month duration | IVIg vs no IVIg | IVIg: 524/524 | IVIg | Proportion of patients exhibiting serious infections | IVIg group: 0.672 events/patient/year. No IVIg group (cessation period): 0.456 events/patient/year |
| Jurlander et al[16] | Uncontrolled before-after observational study | Single center, Denmark | IVIg vs no IVIg | IVIg: 15/15 | IVIg 10 g every 3 weeks in patients with serum IgG level below lower reference limit, a history of recurrent infections and a performance status enabling outpatient setting | Number of infection-related events (antibiotic prescriptions, hospital admissions due to infections, febrile episodes, severe infections) in the 12-month period that preceded IgRT compared to the number of infection-related events during the period of IgRT | IVIg group: 3/15. No IVIg group: 6/15 |
| Cooperative Group for the Study of Immunoglobulin in Chronic Lymphocytic Leukemia[17] | Prospective double-blind RCT | International, multicenter, Germany, Luxembourg, United States, United Kingdom, Italy, 12 months duration | IVIg vs 0.9% sodium chloride solution placebo | IVIg: 28/29 (placebo) (completed 12 months of study) | IVIg 0.4 g/kg body weight every 3 weeks in patients with IgG < 50% LLN for the hospital laboratory or a history of one or more serious infections requiring systemic antibacterial therapy | Number of patients experiencing infections whilst treated with IVIg or sodium chloride placebo | IVIg group: 10/28. Sodium chloride placebo: 13/29 |
| Günther and Dreger[18] | Prospective cohort | Single center, Germany, April 1997 to November 2010 | IVIg vs no IVIg | IVIg: 5/5 | IVIg 0.35 g/kg body weight every 3-4 weeks in patients with secondary immune deficiency with recurrent serious bacterial infections | Incidence of bacterial infections before and after starting IVIg | IVIg group: 8 events/patient/year. No IVIg group: 9 events/patient/year |
| Molica et al[7] | Cross sectional | Multicenter, Italy, 24-month duration | IVIg vs no IVIg | 300 mg/kg IVIg every 4 weeks for at least 6 months in patients with IgG levels < 600 mg/dL and/or a history of at least one serious infectious episode in the 6-month period preceding entry into the study | Number and type of infections occurring. During the 24-month treatment period | ||
| Siffel et al[19] | Retrospective cohort | Single center, United States, October 2015 to March 2020, 12-month duration | IgRT vs no IgRT | IgRT: 118/118 | serum IgG levels < 5.0 g/L, hypogammaglobulinemia diagnosis codes, and ≥ 1 major infection | Number of infections, severe infections, inti-infective use, hospitalizations, length of hospital stay for IgRT-treated and no-IgRT matched cohorts of patients with SID at 12-month follow-up | IgRT group: 7.93 events/patient/year. No IgRT group: 3.56 events/patient/year |
| Soumerai et al[10] | Retrospective cohort | Multicenter, United States, January 2010 and February 15, 2023, 12-month duration | IgRT vs no IgRT | IgRT: 137/137 | Immune globulin infusion (human) 10% | Rate of infections, severe infections and associated antimicrobial use were compared 3 months, 6 months, and 12 months before vs after the index date | IgRT group: 71/137. No IgRT group: 87/137 |
| Tadmor et al[20] | Retrospective cohort | Single center, Israel | IVIg vs no IVIg | IVIg: 326/4206 | IVIg monthly Infection prevention in patients with IgG < 500 mg/L and recurrent infections (2 infections in 6 months or 3 in a year) | Pneumonia episodes over one year, hospitalizations for pneumonia | IVIg group: 13/326. No IVIg group: 463/4206 |
| Visentin et al[21] | Retrospective cross sectional | Multicenter, Italy | IVIg vs no IVIg SCIg vs no SCIg | IVIg: 49/49; SCIg: 88/88 | SCIG or IVIG every 3 weeks or 4 weeks to patients with hypogammaglobulinemia and recurrent infections according to AIFA indications | Rate of bacterial or mycotic infections of any grade before and after treatment with SCIg or IVIg. Incidence of grade ≥ 3 infections | IVIg group: 3.14 events/patient/year. No IVIg group: 2.31 events/patient/year. SCIg group: 2.59 events/patient/year. No SCIg group: 1.43 events/patient/year. The incidence of grade ≥ 3 infections remained stable with IVIg (0.80 events/patient the year before and during IVIg), while it decreased from 1.43 to 0.64 with SCIg |
The measured outcome was the incidence of infections. This meta-analysis evaluated the impact of IgRT on infection outcomes in patients CLL through two complementary approaches: A rate-based analysis of infections per patient/year and a dichotomous analysis of patients experiencing at least one infection. The results of the studies comparing IgRT in CLL patients to no IgRT or placebo, are depicted as forest plots (Figure 2).
The dichotomous and rate-based analyses in this study address related but distinct estimates and should therefore be interpreted as complementary rather than contradictory. Methodological approaches in the different excluded studies that contribute to different characteristics and heterogeneity are presented in Table 2. The dichotomous analysis estimates the relative effect of IgRT on the probability that a patient experiences at least one infection during follow-up (i.e., time to first infectious event). This estimate captures whether IgRT reduces the proportion of patients who cross the threshold from no infection to any infection. In this analysis, IgRT was associated with a lower likelihood of experiencing a first infection (RR = 0.54), consistent with a protective effect against initial infectious events.
| Ref. | Index date | IgRT definition | Person-time counted | Pre-IgRT infections excluded? | Outcome ascertainment |
| Boughton et al[15] | Randomization/trial entry | Assigned treatment arm (IVIg 18 g every 3 weeks vs albumin placebo); dose escalation or crossover after ≥ 3 infections | Total follow-up time from randomization over a fixed 12-month period (intention-to-treat framework) | Yes (only infections occurring after trial entry were counted) | Prospectively collected, standardized clinical definition using predefined scoring system; infections recorded at 3-weekly visits and via patient diaries; serious infections predefined |
| Carrillo de Albornoz et al[11] | Varies by analysis: CLL diagnosis (overall cohort) or first IgRT episode (IgRT users) | Time-varying IgRT exposure defined from hospital procedure codes (on-IgRT = IgRT use in prior 30 days; off-IgRT = no use in prior 30 days); regular vs intermittent IgRT defined by frequency and gaps | Person-time accrued longitudinally from index date until death or censoring (December 31, 2022); segmented into on-IgRT and off-IgRT periods for recurrent-event analyses | No (serious infections prior to IgRT were included and modeled as predictors of IgRT initiation and as time-varying covariates) | Serious infections identified retrospectively via ICD-10-AM and AR-DRG codes for multi-day infection-related hospitalizations |
| Jurlander et al[16] | Initiation of low-dose IVIg therapy | Fixed low-dose IVIg (10 g every 3 weeks) administered during treatment period only; no concurrent control group | Aggregated person-time compared between two periods: 12 months before IVIg (168 patient-months) vs during IVIg treatment (169 patient-months); rates implicitly calculated from total events over person-time | No (pre-treatment infections explicitly included as the comparator period) | Prospectively recorded clinical events: Antibiotic prescriptions, hospital admissions due to infection, febrile episodes; severe infections defined clinically; microbiology reported for selected events |
| Cooperative Group for the Study of Immunoglobulin in Chronic Lymphocytic Leukemia[17] | Randomization and initiation of IVIg or placebo infusion (trial enrollment start) | IVIg administered at 400 mg/kg every 3 weeks. Compared against placebo (albumin infusion) | Participants were followed prospectively for 12 months. Person-time accrued from randomization until end of follow-up, withdrawal, death, or study completion | Only infections occurring after trial entry were counted. Infections before enrollment were not included in outcome measurement | Prospectively monitored. Clinically documented. Confirmed through medical record review. Categorized as bacterial infections requiring antimicrobial therapy. Assessed during scheduled follow-up visits and interim clinical reports |
| Günther and Dreger[18] | Initiation of IVIg therapy | IVIg exposure defined as active treatment period only; standard dose approximately 0.35 g/kg every 3-4 weeks; no concurrent control group | Person-time compared between two periods: Infections in the 3 months prior to IVIg initiation (extrapolated to annualized rates) vs infections accrued during IVIg treatment over long-term follow-up (total 528 patient-months) | No (pre-IVIg infections explicitly included as comparator period) | Clinically documented serious bacterial infections recorded during routine care; infection type, treatment, and duration prospectively documented; some baseline data retrospectively abstracted from medical records |
| Molica et al[7] | Entry into study/randomization | IVIg 300 mg/kg every 4 weeks during assigned treatment periods; patients crossed over between IVIg prophylaxis and observation (no IVIg), acting as their own controls | Person-time accrued from study entry until death, loss to follow-up, or study end; segmented into observation (no IVIg) and IVIg treatment periods (36 vs 376 patient-months overall; 321 vs 292 patient-months for 6-month completers; 206 vs 215 patient-months for 12-month completers) | No (infections during observation periods served as the comparator by design) | Prospectively assessed before each infusion using predefined clinical criteria; infections graded as trivial, minor, or major; serious infections defined by need for antibiotics, hospitalization, or IV therapy |
| Siffel et al[19] | IgRT analysis: Re-indexed to first IgRT claim (IgRT cohort) or pseudo-index date (no-IgRT cohort) | Receipt of IgRT identified from claims; IgRT-treated patients required ≥ 2 IgRT administrations post-index; comparator was SID patients without IgRT | Fixed 12-month post-index follow-up (minimum 3 months for survival); outcomes summarized per patient over follow-up, not as continuous person-time rates | No (baseline infections and infection burden during the pre-index period were included and differed substantially between groups) | Infections identified via ICD-10-CM diagnosis codes in claims/EHR; severity inferred from antibiotic escalation, intravenous therapy, hospitalization, unusual pathogens or complications |
| Soumerai et al[10] | Date of first IgRT administration | IgRT initiation identified in EHR; patients required ≥ 3 months of follow-up before and after index | Fixed windows (3 months, 6 months, 12 months before vs after IgRT initiation); no continuous time-at-risk modeling | No (pre-IgRT infections are intentionally included and serve as the comparator) | Infections identified via ICD-9/10 codes; severe infections defined by hospitalization or IV antimicrobials; antimicrobial use captured via prescriptions within 30 days |
| Tadmor et al[20] | Date of CLL diagnosis | Monthly IVIg administered for hypogammaglobulinemia (< 500 mg/L) with recurrent infections; modeled as a time-dependent exposure | Person-time accrued from CLL diagnosis until event or censoring; patients contributed unexposed time before IVIg initiation and exposed time after initiation | Yes, for exposed time (events prior to IVIg initiation were not attributed to IgRT-exposed person-time) | Pneumonia identified via ICD-9 codes combined with antibiotic prescriptions (outpatient) or imaging and hospitalization records (inpatient); prospectively recorded within electronic health records |
| Visentin et al[21] | Start of IgRT (IVIg or SCIg initiation) | Continuous IgRT exposure (IVIg every 3-4 weeks or SCIg weekly/biweekly); patients switching from IVIg to SCIg reclassified at switch | Person-time accrued from IgRT initiation only until infection, discontinuation, death, or last follow-up | Yes (baseline infection rates were analyzed separately but not included in post-IgRT person-time) | Infections prospectively abstracted from clinical records; bacterial and mycotic infections of any grade; rates expressed as events per patient-year and cumulative incidence (time-to-first and second infection) |
In contrast, the rate-based analysis estimates the relative difference in the frequency of infections per unit of person-time among patients who remain at risk, thereby capturing the burden of recurrent infections. This estimate reflects infection dynamics after the first event and is particularly influenced by patients with persistent susceptibility. The observed higher infection rates in the IgRT group (rate ratio 1.81) therefore do not imply an increased risk of initial infection, but rather indicate that IgRT does not reduce - and may be associated with higher observed - recurrent infection frequency among patients who continue to experience infections.
The rate-based analysis of five studies demonstrated that IgRT was associated with significantly higher infection rates compared to no IgRT, with a pooled random-effects rate ratio of 1.65 (95%CI: 1.33-2.04, P < 0.001). However, this analysis showed high heterogeneity (I2 = 88.6%), suggesting substantial variation across studies. Subgroup analyses revealed consistent findings across different IgRT formulations, with IVIg showing a rate ratio of 1.42 (95%CI: 1.11-1.82) and SCIg showing rate ratio = 1.81 (95%CI: 1.55-2.12). Study design subgroups demonstrated similar effects, with before-after IgRT studies showing a rate ratio of 1.55 (95%CI: 1.02-2.36) and retrospective studies showing rate ratio = 1.72 (95%CI: 1.23-2.40).
In contrast, the dichotomous analysis of five studies found that IgRT significantly reduced the risk of patients experiencing at least one infection, with a pooled random-effects RR of 0.60 (95%CI: 0.41-0.88, P = 0.008). This analysis showed a moderate heterogeneity (I2 = 57.2%). Subgroup analyses revealed that observational studies showed a stronger protective effect (RR = 0.54, 95%CI: 0.33-0.88) compared to RCTs (RR = 0.63, 95%CI: 0.37-1.07), though the latter did not reach statistical significance. Both IVIg (RR = 0.61, 0.38-0.99) and non-IVIg formulations (RR = 0.52, 95%CI: 0.28-0.96) de
When interpreted jointly, these findings suggest that IgRT may reduce the likelihood of a first infection in patients with CLL, while offering limited benefit in preventing recurrent infections among those who remain susceptible. The high heterogeneity observed in the rate-based analysis further indicates that estimates of recurrent infection burden are strongly influenced by study design, patient selection, and time-related biases inherent to observational data. Accordingly, the two analyses describe different clinical constructs and should not be interpreted as measuring the same underlying infection risk.
Sensitivity analyses demonstrated robust findings. For the rate-based analysis, excluding small studies or before-after designs did not substantially alter the results, with rate ratios remaining between 1.66-1.72. Similarly, the dichotomous analysis results remained consistent when excluding small studies (RR = 0.55, 95%CI: 0.36-0.83) or analyzing only RCTs (RR = 0.63, 95%CI: 0.37-1.07).
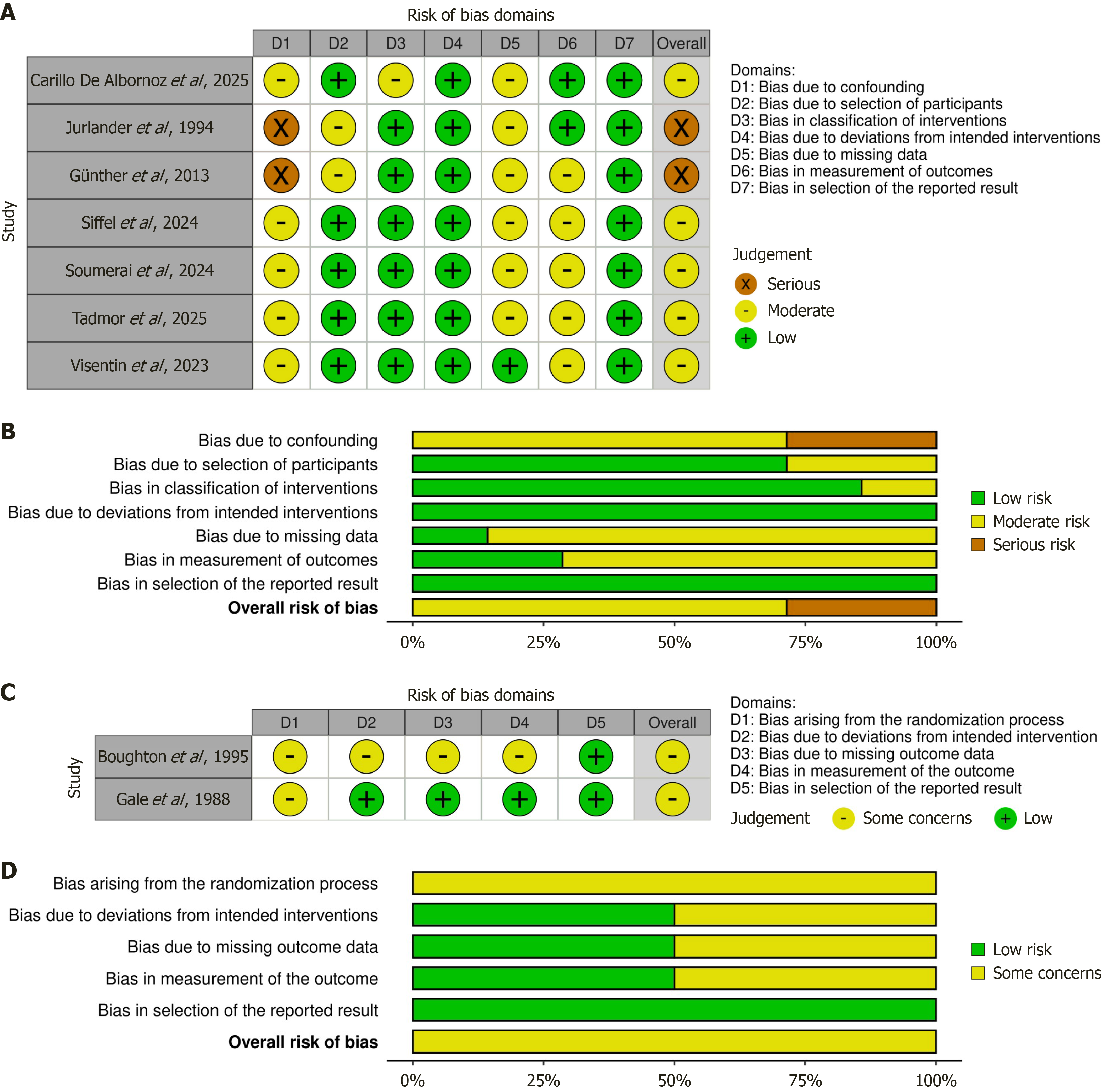
The risk of bias is represented with a “traffic light” plot for each domain and provided in Figure 3. An online tool was used for the graphical illustration of the quality assessment[33]. The quality assessment of included studies was performed using the RoB 2 tool for RCTs and the ROBINS-I tool for observational studies.
Among the observational studies assessed with ROBINS-I (Figure 3A), most demonstrated a moderate risk of bias overall, with common concerns relating to confounding (D1) and missing data (D5). Specifically, the studies by Jurlander et al[16] and Günther and Dreger[18] were judged to be at serious risk of bias, primarily due to confounding and par
For the RCTs, the RoB 2 assessment (Figure 3B) indicated that the study by Boughton et al[15] and the study by Cooperative Group for the Study of Immunoglobulin in Chronic Lymphocytic Leukemia[17] were rated as having some concerns, particularly due to issues in the randomization process and deviations from intended interventions. These trials contributed principally to the dichotomous outcome analyses and supported the observed reduction in first infection risk.
Taken together, the body of evidence included in this review is characterized by a predominance of observational cohort studies with moderate risk of bias, alongside two RCTs with some concerns arising. The overall quality assessment for each domain is presented in Figure 3C and D. Importantly, conclusions regarding the reduction of first infections are supported by both randomized and lower-risk observational evidence, whereas findings related to recurrent infection rates are driven largely by observational studies with moderate to serious risk of bias. These considerations underscore the need for cautious interpretation of the rate-based results, particularly in light of residual confounding and other methodological limitations inherent to non-randomized designs.
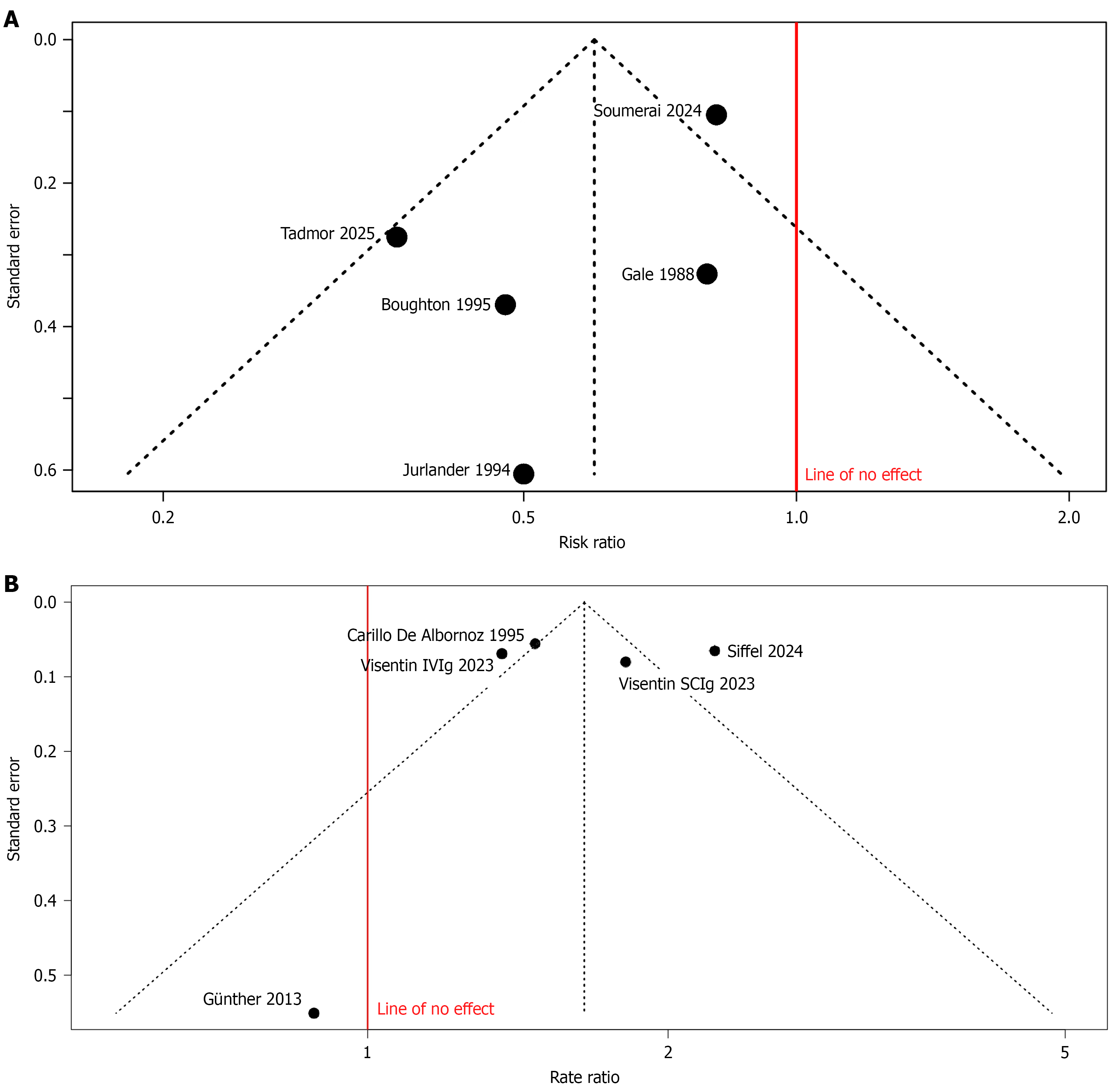
Publication bias was assessed using funnel plots for both the dichotomous and the rate-based analyses. Visual inspection of the funnel plots did not suggest the presence of publication bias. For the dichotomous analysis, Egger’s regression test did not indicate funnel plot asymmetry (intercept = -1.65, 95%CI: -3.68 to 0.39; t = -1.584; P = 0.211). Similarly, for the rate-based analysis, Egger’s test did not demonstrate evidence of asymmetry (intercept = -0.70, 95%CI: -8.03 to 6.64; t = -0.186; P = 0.864). Overall, these findings suggest a low likelihood of publication bias in the included studies (Figure 4).
This systematic review and meta-analysis aimed to integrate and assess the published evidence regarding IgRT for infection prevention in patients with CLL. By employing a systematic approach in accordance with PRISMA criteria and utilizing meta-analysis to synthesize data from multiple independent studies, our investigation reveals a complex and seemingly contradictory relationship between IgRT and infection risk, a paradox resolvable only through careful consideration of outcome hierarchy and study methodology. Our dual analytical approach - incorporating both dichotomous (proportion of patients with ≥ 1 infection) and rate-based (infections per patient-year) measures - captured distinct dimensions of risk, yielding divergent pooled results that reflect fundamental differences in how IgRT influences initial vs recurrent infections, as well as critical methodological variations across the evidence base.
The dichotomous analysis, which included data from both RCTs and observational studies, demonstrates that IgRT is associated with a 40% reduction in the proportion of patients experiencing at least one infection (RR = 0.60, 95%CI: 0.41-0.88). This finding aligns with the seminal 2009 meta-analysis by Raanani et al[8], which concluded intravenous IgRT significantly reduced major infections (RR = 0.45). The highest-confidence evidence for this protective effect originates from RCTs, which consistently show IgRT reduces the probability of a first or clinically significant bacterial infection. These trials, by design, are less susceptible to confounding by indication, immortal time bias, and differential sur
In stark contrast, the rate-based analysis - largely informed by observational studies and before-after designs evaluating recurrent infections - did not demonstrate a reduction in infection frequency. Instead, it found IgRT was associated with a 65% increase in infection rates per patient-year (RR = 1.65, 95%CI: 1.33-2.04). This observation must be interpreted with extreme caution, as it reflects a profound associative finding rather than evidence of causation. IgRT is typically initiated in real-world practice precisely in those patients with more advanced immunodeficiency, pre-existing recurrent infections, or greater disease severity. This creates overwhelming confounding by indication, where recipients are intrinsically at a higher baseline risk. Surveillance bias further contributes, as patients on IgRT have more frequent healthcare encounters, increasing the detection of infections. Therefore, the observed higher infection rates during IgRT exposure should not be interpreted as evidence that IgRT worsens infection risk. Rather, they manifest the residual confounding, indication bias, and methodological heterogeneity inherent to the observational studies that populate this analysis. These data suggest that once chronic infectious susceptibility is established, IgRT should not be expected to eliminate recurrent infections, as this burden in CLL likely reflects multifactorial immune failure - including T-cell dysfunction, neutropenia, and treatment-related immunosuppression - that passive antibody replacement cannot fully correct.
The interpretation of these divergent results is heavily influenced by substantial heterogeneity across studies, particularly in the rate-based analysis (I2 = 88.6%). This high heterogeneity is not merely a statistical limitation but a reflection of genuine and systematic clinical and methodological diversity. Key sources include: (1) Variable infection definitions and severity thresholds, ranging from microbiologically documented severe infections to all antibiotic-treated events; (2) Critical differences in exposure timing and person-time attribution. RCTs accrue person-time from randomization, excluding pre-enrollment events, while observational studies often initiate IgRT after patients have demonstrated recurrent infections, inherently capturing periods of heightened baseline risk; (3) Varied study designs, including po
These methodological differences explain the apparent discrepancy between our analyses. They underscore why dichotomous outcomes (any first infection) from RCTs provide the most reliable evidence of efficacy, while rate-based outcomes (recurrent events) from observational data are most informative for understanding the limitations of IgRT in advanced disease states and the powerful role of confounding. Our findings, therefore, support a targeted, risk-stratified approach: IgRT appears best suited to reduce the risk of a first or early serious bacterial infection and delay progression to a recurrent infection-prone status in high-risk patients. Conversely, it should not be expected to normalize recurrent infection frequency, replace vaccination or antimicrobial prophylaxis, or reverse advanced immune dysregulation.
A key limitation of this synthesis, and of the field at large, is that the majority of randomized evidence for IgRT predates contemporary targeted CLL therapies, including Bruton’s tyrosine kinase inhibitors, B-cell lymphoma-2 inhibitors, and phosphatidyl-inositol-3-kinase inhibitors. These agents fundamentally alter infection risk by modifying T-cell function, innate immunity, and susceptibility to opportunistic pathogens. Consequently, historical estimates of IgRT benefit may not fully translate to modern practice. Determining the role of IgRT in patients receiving these novel agents represents a critical unmet research need. Future studies must incorporate time-dependent exposure modeling, standardized infection definitions (explicitly distinguishing first, severe, and recurrent events), and stratification by immune phenotype and targeted therapy use to clarify which contemporary patients derive the greatest net benefit.
In patients with CLL and hypogammaglobulinemia, the most reliable evidence indicates that IgRT reduces the likelihood of experiencing an initial or severe bacterial infection. However, it does not appear to meaningfully reduce the burden of recurrent infections, and observational data associate its use with higher infection rates - a finding largely attributable to confounding by indication and methodological bias rather than a causal harmful effect. These results support a targeted and individualized role for IgRT, prioritizing its use for prevention of first serious infections in high-risk patients (e.g., those with profound hypogammaglobulinemia and prior infection history) rather than routine universal administration. IgRT should not be expected to eliminate recurrent infections once chronic susceptibility is established. Importantly, the applicability of historical trial data to patients treated with modern targeted therapies remains uncertain, underscoring an urgent need for contemporary randomized studies that employ standardized outcome frameworks to define the precise role of IgRT in the current treatment era.
| 1. | Mukkamalla SKR, Taneja A, Malipeddi D, Master SR. Chronic Lymphocytic Leukemia. 2023 Mar 7. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan. [PubMed] |
| 2. | Ahmed J, Choi Y, Ko T, Lim J, Hajjar J. Use of Immunoglobulin Replacement Therapy in Clinical Practice: A Review. J Immunother Precis Oncol. 2025;8:34-46. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 3. | Perez EE. Immunoglobulin use in immune deficiency and autoimmune disease states. Am J Manag Care. 2019;25:S92-S97. [PubMed] |
| 4. | Boqué C, Sánchez-Ramón S, Córdoba R, Moreno C, Cabezudo E. Current Approach in the Management of Secondary Immunodeficiency in Patients with Hematological Malignancies: Spanish Expert Consensus Recommendations. J Clin Med. 2023;12:6356. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 5. | Grywalska E, Zaborek M, Łyczba J, Hrynkiewicz R, Bębnowska D, Becht R, Sosnowska-Pasiarska B, Smok-Kalwat J, Pasiarski M, Góźdź S, Roliński J, Niedźwiedzka-Rystwej P. Chronic Lymphocytic Leukemia-Induced Humoral Immunosuppression: A Systematic Review. Cells. 2020;9:2398. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 6. | Bunch C. Immunoglobulin replacement in chronic lymphocytic leukaemia. Nouv Rev Fr Hematol (1978). 1988;30:419-422. [PubMed] |
| 7. | Molica S, Musto P, Chiurazzi F, Specchia G, Brugiatelli M, Cicoira L, Levato D, Nobile F, Carotenuto M, Liso V, Rotoli B. Prophylaxis against infections with low-dose intravenous immunoglobulins (IVIG) in chronic lymphocytic leukemia. Results of a crossover study. Haematologica. 1996;81:121-126. [PubMed] |
| 8. | Raanani P, Gafter-Gvili A, Paul M, Ben-Bassat I, Leibovici L, Shpilberg O. Immunoglobulin prophylaxis in chronic lymphocytic leukemia and multiple myeloma: systematic review and meta-analysis. Leuk Lymphoma. 2009;50:764-772. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 108] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 9. | Baratè C, Scortechini I, Ciofini S, Picardi P, Angeletti I, Loscocco F, Sanna A, Isidori A, Sportoletti P. Management of infections for patient treated with ibrutinib in clinical practice. Front Oncol. 2024;14:1428464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 10. | Soumerai JD, Yousif Z, Gift T, Desai R, Huynh L, Ye M, Banatwala A, Clear L, Pinaire M, Belsky G, Hsieh YG, Herrick C, Darnell EP, Duh MS, Sanchirico M, Murphy SN. IgG testing, immunoglobulin replacement therapy, and infection outcomes in patients with CLL or NHL: real-world evidence. Blood Adv. 2024;8:4239-4249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 11. | Carrillo de Albornoz S, Zhang X, Arnolda R, MacPhail A, Irving A, Higgins AM, Wood EM, McQuilten ZK, Petrie D. Immunoglobulin use, survival, and infection outcomes in patients with chronic lymphocytic leukemia. Blood Adv. 2025;9:5367-5377. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 12. | Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9803] [Reference Citation Analysis (0)] |
| 13. | Cochrane Handbook for Systematic Reviews of Interventions. 2nd Edition. Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, editors. Chichester (UK): John Wiley & Sons, 2019. |
| 14. | R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing. 31 Oct 2025. Available from: https://cran.r-project.org/doc/manuals/r-release/fullrefman.pdf. |
| 15. | Boughton BJ, Jackson N, Lim S, Smith N. Randomized trial of intravenous immunoglobulin prophylaxis for patients with chronic lymphocytic leukaemia and secondary hypogammaglobulinaemia. Clin Lab Haematol. 1995;17:75-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 72] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 16. | Jurlander J, Geisler CH, Hansen MM. Treatment of hypogammaglobulinaemia in chronic lymphocytic leukaemia by low-dose intravenous gammaglobulin. Eur J Haematol. 1994;53:114-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 44] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 17. | Cooperative Group for the Study of Immunoglobulin in Chronic Lymphocytic Leukemia; Gale RP, Chapel HM, Bunch C, Rai KR, Foon K, Courter SG, Tait D. Intravenous immunoglobulin for the prevention of infection in chronic lymphocytic leukemia. A randomized, controlled clinical trial. N Engl J Med. 1988;319:902-907. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 285] [Cited by in RCA: 252] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 18. | Günther G, Dreger B. Post-marketing observational study on 5% intravenous immunoglobulin therapy in patients with secondary immunodeficiency and recurrent serious bacterial infections. Microbiol Immunol. 2013;57:527-535. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 9] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 19. | Siffel C, Richter J, Anderson-Smits C, Kamieniak M, Ren K, Shah D, Davids MS. Treatment patterns and burden of infection in patients with chronic lymphocytic leukemia and secondary immunodeficiency: a retrospective database study. Ann Hematol. 2024;103:4567-4580. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 20. | Tadmor T, Hardak E, Melamed G, Alapi H, Rokach L. Chronic lymphocytic leukemia and associated chronic lung diseases. Haematologica. 2025;110:2673-2682. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 21. | Visentin A, Molinari MC, Pravato S, Cellini A, Angotzi F, Cavaretta CA, Ruocco V, Imbergamo S, Piazza F, Proietti G, Mauro FR, Trentin L. A Retrospective Study on the Efficacy of Subcutaneous Immunoglobulin as Compared to Intravenous Formulation in Patients with Chronic Lymphocytic Leukemia and Secondary Antibody Deficiency. Curr Oncol. 2022;30:274-283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 22. | Keegan A, Dennington PM, Dhondy N, Mulligan SP. Immunoglobulin replacement therapy in chronic lymphocytic leukaemia patients with hypogammaglobulinaemia and infection. Eur J Haematol. 2022;108:460-468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 23. | Dimou M, Iliakis T, Maltezas D, Bitsani A, Kalyva S, Koudouna A, Kotsanti S, Petsa P, Papaioannou P, Kyrtsonis MC, Panayiotidis P. Efficacy-safety of Facilitated Subcutaneous Immunoglobulin in Immunodeficiency Due to Hematological Malignancies. A Single-Center Retrospective Analysis. Anticancer Res. 2018;38:4187-4191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 24. | Paassen PV, Pittrow D, Scheidegger C, Klotsche J, Ellerbroek PM. Use of recombinant human hyaluronidase-facilitated subcutaneous immunoglobulin in elderly patients. Immunotherapy. 2020;12:131-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 25. | Sklenar I, Schiffman G, Jønsson V, Verhoef G, Birgens H, Boogaerts M, Ferrant A, Christensen BE, Hasle H, Drivsholm A. Effect of various doses of intravenous polyclonal IgG on in vivo levels of 12 pneumococcal antibodies in patients with chronic lymphocytic leukaemia and multiple myeloma. Oncology. 1993;50:466-477. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 35] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 26. | Gamm H, Huber C, Chapel H, Lee M, Ries F, Dicato MA. Intravenous immune globulin in chronic lymphocytic leukaemia. Clin Exp Immunol. 1994;97 Suppl 1:17-20. [PubMed] |
| 27. | Chapel H, Dicato M, Gamm H, Brennan V, Ries F, Bunch C, Lee M. Immunoglobulin replacement in patients with chronic lymphocytic leukaemia: a comparison of two dose regimes. Br J Haematol. 1994;88:209-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 57] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 28. | 40th International Symposium on Intensive Care & Emergency Medicine 2020 - Part 2 : e-ISICEM 2020 15-18 September 2020. Crit Care. 2020;24:533. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 29. | Griffiths H, Brennan V, Lea J, Bunch C, Lee M, Chapel H. Crossover study of immunoglobulin replacement therapy in patients with low-grade B-cell tumors. Blood. 1989;73:366-368. [PubMed] |
| 30. | Angelotti F, Capecchi R, Giannini D, Mazzarella O, Rocchi V, Migliorini P. Long-term efficacy, safety, and tolerability of recombinant human hyaluronidase-facilitated subcutaneous infusion of immunoglobulin (Ig) (fSCIG; HyQvia((®))) in immunodeficiency diseases: real-life data from a monocentric experience. Clin Exp Med. 2020;20:387-392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 31. | Dimou M, Speletas M, Milito C, Pyzik A, Huscher D, Kamieniak M, Pittrow D, Borte M. Insights into Facilitated Subcutaneous Immunoglobulin Use in Patients with Secondary Immunodeficiency Diseases: A FIGARO Subgroup Analysis. Cancers (Basel). 2023;15:4524. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 32. | Morell A, Barandun S. Prophylactic and therapeutic use of immunoglobulin for intravenous administration in patients with secondary immunodeficiencies associated with malignancies. Pediatr Infect Dis J. 1988;7:S87-S91. [PubMed] |
| 33. | McGuinness LA, Higgins JPT. Risk-of-bias VISualization (robvis): An R package and Shiny web app for visualizing risk-of-bias assessments. Res Synth Methods. 2021;12:55-61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4021] [Cited by in RCA: 3618] [Article Influence: 723.6] [Reference Citation Analysis (4)] |