Published online Mar 26, 2026. doi: 10.12998/wjcc.v14.i9.118295
Revised: January 24, 2026
Accepted: February 26, 2026
Published online: March 26, 2026
Processing time: 85 Days and 17.7 Hours
The intestinal lumen harbors a dynamic sensory and responsive network com
Core Tip: Semaglutide,a leading glucagon-like peptide-1 receptor agonist, reshapes gut-brain communication by bidirectionally modulating the transforming growth factor-β/brain-derived neurotrophic factor signaling axis in the gut. It promotes neuroprotective brain-derived neurotrophic factor via phosphatidylinositol 3-kinase/protein kinase B/cyclic adenosine monophosphate-response element-binding protein while suppressing profibrotic transforming growth factor-β/Smad signaling. Combined with beneficial shifts in gut microbiota and anti-inflammatory effects, this action remodels enteric neural perception, extending its therapeutic potential beyond metabolism to neuropsychiatric and neurodegenerative disorders.
- Citation: Pan HY, Liang JX, Chen WW, Sheng YY, Zhang WJ, Zhu XW, Wang SY, Yang GH, Xu B, Xu TC. Beyond glycemic control: How semaglutide reshapes intestinal neuroception via transforming growth factor-β/brain-derived neurotrophic factor signaling hubs. World J Clin Cases 2026; 14(9): 118295
- URL: https://www.wjgnet.com/2307-8960/full/v14/i9/118295.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i9.118295
Traditionally, the primary action of glucagon-like peptide-1 receptor agonist (GLP-1RA) such as semaglutide has been understood as glucose-lowering[1]. However, emerging evidence reveals that these agents exert broader protective effects, particularly in mitigating multi-organ fibrosis. Central to these actions are two critical signaling hubs: The transforming growth factor-β (TGF-β) and brain-derived neurotrophic factor (BDNF) pathways. The TGF-β pathway is a key driver of fibrosis and inflammation, playing a pivotal role in chronic pathologies of organs such as the liver[2], kidney[3], and heart[4]. Meanwhile, BDNF is a crucial neurotrophic factor[5] influencing both central and enteric nervous system (ENS) function[6,7].
Studies indicate that GLP-1RAs like semaglutide effectively suppress the TGF-β/Smad pathway, reducing the extent of organ fibrosis[2,3], which may represent a fundamental molecular mechanism underlying their multi-organ protective effects[1]. Furthermore, these agents demonstrate neuroprotective properties, such as alleviating neuroinflammation in models of brain injury[8]. Although direct evidence linking semaglutide to BDNF modulation in the gut remains limited, BDNF is known to be involved in energy metabolism and interacts with factors such as interleukin-6 (IL-6). Notably, glucagon-like peptide-1 (GLP-1)-based therapies can modulate AMP-activated protein kinase activity[1,2], and IL-6 in turn influences GLP-1 expression. This leads us to hypothesize the existence of an intricate regulatory network in the gut involving GLP-1 signaling, cytokines (e.g., IL-6), BDNF, and energy-sensing pathways (e.g., AMP-activated protein kinase). This network likely orchestrates enteroendocrine function, neural sensing, and gut-brain communication.
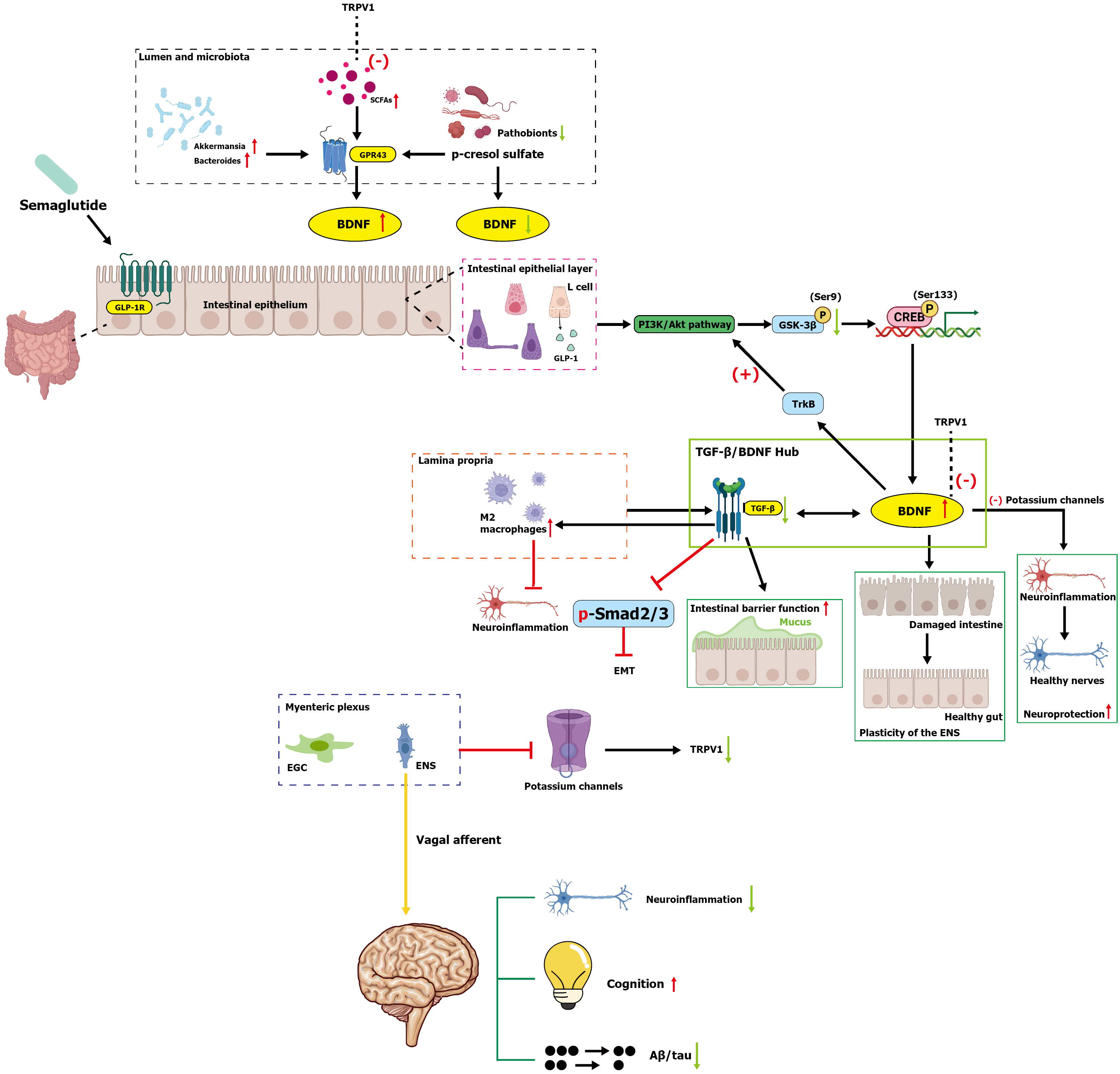
In summary, the pleiotropic effects of semaglutide are closely tied to its actions on the gut-brain axis (GBA) and the ENS, with the intestinal “TGF-β/BDNF signaling hub” playing a central role. Therefore, we believe that semaglutide may act on local glucagon-like peptide-1 receptor (GLP-1R) in the intestine to synergistically regulate TGF-β and BDNF signaling, thereby reshaping ENS function and gut-brain communication. This review will synthesize current evidence to explore the interplay between these two pathways, elucidating how semaglutide reshapes enteric neural perception, improves gut function, and coordinates systemic anti-fibrotic and neuroprotective responses. These insights may pave the way for novel therapeutic strategies in neurological disorders and future drug development (Figure 1).
TGF-β and BDNF are core signaling molecules of the GBA, collectively forming a hub that integrates neural, immune, and microbial signals. As a secreted polypeptide growth factor[9], TGF-β regulates enteric neuronal homeostasis, inhibits neuroinflammation[10,11], strengthens the intestinal mucosal barrier and gut microbiota balance, and mediates bidirectional gut-central nervous system communication via the ligand-receptor-Smad pathway[12]. It also serves as a key driver of organ fibrosis, with excessive activation exacerbating fibrosis in tissues such as the intestine, liver, and kidneys[13]. BDNF, a specific polypeptide neurotrophic factor, is widely expressed in various intestinal cells. It regulates neuronal survival, proliferation, and axon growth[14,15], exerts neuroprotective effects against inflammation/oxidative stress-induced damage[5], maintains intestinal barrier function, and mediates bidirectional GBA regulation[16,17]. An interaction exists between TGF-β and BDNF: BDNF may exert feedback regulation on TGF-β signaling by inhibiting Smad2/3 phosphorylation, and the two molecules collaboratively maintain intestinal homeostasis (Figure 1).
In the ENS, TGF-β and BDNF form a critical bidirectional regulatory axis that jointly maintains intestinal homeostasis. Dysregulation of this signaling network is a core mechanism underlying functional gastrointestinal disorders. TGF-β is a multifunctional core cytokine in the ENS, primarily produced by neurons, glial cells, and resident immune cells (e.g., macrophages). Its roles in the ENS can be categorized into two pathways: The classical Smad signaling pathway, where TGF-β activates Smad2/3 to form a complex with Smad4, regulating gene expression and modulating non-transcriptional processes[18]. In intestinal neuroregulation, TGF-β secreted by M2 macrophages specifically upregulates the oxytocin signaling system (oxytocin/oxytocin receptor) by activating the neuronal Smad2/3 pathway and inhibiting paternally expressed gene 3 expression, a mechanism confirmed during disease recovery[19]. The non-classical signaling pathway: Signal transducer and activator of transcription 3 serves as the integration point for neuro-immune dialogue. M1 mac
BDNF is a core signaling molecule in the ENS that regulates neuronal plasticity and function. It is primarily produced by neurons, glial cells, and neuro-supportive macrophages educated by TGF-β. Its key roles include: Binding to the tropomyosin receptor kinase B (TrkB) receptor on enteric neurons and enhancing neuronal excitability by inhibiting voltage-gated potassium channels, which forms the molecular basis for visceral hypersensitivity and pain amplification[22,23]; and stimulating secretomotor neurons to release vasoactive intestinal peptide and acetylcholine, thereby activating the cystic fibrosis transmembrane conductance regulator channel on intestinal epithelial cells, driving chloride ion and fluid secretion, and contributing to pathological diarrhea[5]. BDNF also serves as a critical mediator through which the gut microbiota influences ENS function: Beneficial metabolites such as short-chain fatty acids (SCFAs) can upregulate BDNF, whereas harmful metabolites like p-cresol sulfate downregulate its levels, thereby translating microbial signals into neural regulation. For example, Bifidobacterium bifidum CCFM1163 modulates the microbiota composition and SCFA levels to indirectly promote colonic BDNF expression[16,24]. In pathological conditions such as irritable bowel syndrome, abnormal enhancement of BDNF signaling directly leads to visceral hyperalgesia, secretory dysfunction, and forms a vicious cycle with symptoms like anxiety[25]. BDNF exhibits a “double-edged sword” characteristic in the ENS: It participates in physiological neural regulation under normal conditions, but drives visceral hy
Notably, in patients with depression, peripheral TGF-β levels are significantly positively correlated with BDNF levels, and TGF-β can upregulate BDNF expression[26], suggesting that the TGF-β/BDNF axis serves as a critical bridge con
Of particular significance, this hub serves as the core functional module for bidirectional communication in the “microbiota-GBA”. In autism model mice, localized intestinal inflammation was associated with downregulation of TGF-β and BDNF expression in multiple brain regions. Notably, the use of anti-α4β7 monoclonal antibodies targeting only the gut to eliminate intestinal inflammation could remotely and selectively reverse the expression defect of this hub in the brain, thereby rescuing social behavior[31]. This strongly demonstrates that gut-derived immune signals can specifically regulate the central TGF-β/BDNF protective axis. Similarly, various interventions such as chicoric acid and aerobic exercise can synergistically upregulate TGF-β and BDNF in multiple organs including the gut, spleen, and brain, further highlighting their central role in systemic regulation[32].
In summary, the existing evidence has delineated a clear picture: TGF-β/BDNF constitutes a highly synergistic “immunoneurotrophic” hub that can be regulated at the metabolic, behavioral, and peripheral organ levels. It integrates microglial phenotypes and neuroplasticity at the central level[27,29], and mediates the influence of intestinal and systemic states on brain function at the systemic level[31,32]. Therefore, we hypothesize that the TGF-β/BDNF regulatory network in the ENS can be regarded as a fully functional “brain-gut interface” unit. However, directly elucidating the cell-specific co-localization, local bidirectional dialogue mechanisms, and direct regulation of this hub on intestinal motility, secretion, and other functions within the intrinsic neurons and glial networks of the intestinal ENS remains a critical issue requiring breakthroughs in future research.
Semaglutide, as a long-acting GLP-1RA, activates the GLP-1R widely distributed on enteric neurons, enteroendocrine cells, and intestinal mucosal immune cells, initiating downstream signaling that precisely modulates the function and plasticity of the gut sensory neural circuitry. Its core mechanism involves the synergy of the following two pathways, which jointly target the pathological core of visceral hypersensitivity: Aberrant neural activity and the inflammatory microenvironment.
Upon binding to the GLP-1 receptor, semaglutide activates phosphatidylinositol 3-kinase (PI3K) and its downstream effector, protein kinase B (Akt). The phosphorylation of Akt subsequently leads to the phosphorylation of glycogen synthase kinase-3β (GSK-3β) at Ser9, thereby inhibiting its kinase activity. This pathway directly enhances enteric neuron function and stress resistance[33,34]. The inhibited GSK-3β can no longer suppress the transcriptional activity of cyclic adenosine monophosphate-response element-binding protein (CREB), allowing CREB to be phosphorylated at Ser133, translocate into the nucleus, and initiate the transcription of genes encoding neurotrophic factors, such as BDNF[35]. Within the ENS, the increased expression of BDNF is crucial for maintaining and repairing the survival, synaptic connectivity, and signal transduction of intrinsic sensory and interneurons. This pathway has been validated in studies of memory deficit disorders like sporadic Alzheimer’s disease[33], which elucidates how GLP-1RAs enhance synaptic plasticity and memory function by promoting CREB phosphorylation and BDNF expression. Similarly, in the gut, this pathway can enhance the stability and resilience of the enteric neuronal network, directly correcting aberrant electrical activity at the cellular level, thereby remodeling normal gut sensory thresholds and counteracting hypersensitivity states. Concurrently, BDNF is not only a neurotrophic factor but also possesses anti-inflammatory and neuroprotective properties. Elevated BDNF can further inhibit GSK-3β by reactivating the PI3K/Akt pathway through its TrkB receptor, forming a positive feedback loop that amplifies its own expression (Figure 1)[35].
Simultaneously, semaglutide exerts important anti-fibrotic and anti-inflammatory effects by bidirectionally modulating the TGF-β signaling pathway, although the direction of its effect is context-dependent. In the intestine, TGF-β is a central mediator regulating immune cell phenotype, inflammatory responses, and tissue repair, and its dynamic balance directly influences the microenvironment surrounding sensory nerve endings. In studies of cerebral ischemia/reperfusion injury, semaglutide promotes the polarization of microglia towards the anti-inflammatory M2 phenotype, upregulates the anti-inflammatory factor TGF-β, and suppresses pro-inflammatory factors like tumor necrosis factor-alpha, thereby mitigating neuroinflammation[8]. However, in fibrotic disease research, its effect is opposite, significantly reducing the expression of TGF-β1 and its receptor, and inhibiting the downstream epithelial-mesenchymal transition process, thus effectively alleviating fibrosis progression (Table 1)[36].
| Pathway name | Key regulatory nodes and direction | Regulatory direction | Primary biological functions | Ref. |
| Semaglutide/GLP-1R/PI3K/Akt/GSK-3β/CREB/BDNF pathway | Semaglutide activates the GLP-1R, leading to enhanced PI3K/Akt signaling. This results in the inhibition of GSK-3β, which in turn promotes CREB activation and increases the transcription of BDNF | Positive activation | Promotes neuronal differentiation, synaptogenesis, and plasticity. Improves memory and cognitive function. BDNF, via TrkB receptor, initiates a positive feedback loop that reinforces PI3K/Akt signaling, creating a neuroprotective cycle | [33,35] |
| Bidirectional regulation of TGF-β signaling by semaglutide | In neuroinflammatory contexts, semaglutide elevates anti-inflammatory TGF-β levels and promotes microglial polarization toward the M2 phenotype. In fibrotic disease contexts, it downregulates the expression of TGF-β and its receptors, thereby inhibiting the epithelial-mesenchymal transition process | Bidirectional regulation | Anti-inflammatory: Attenuates neuroinflammation and promotes tissue repair in models like cerebral ischemia/reperfusion injury. Anti-fibrotic: Inhibits excessive extracellular matrix deposition and mitigates fibrosis progression in models of the liver | [8,36] |
Evidence from both in vitro and in vivo experiments supports these mechanisms. In human neuroblastoma SH-SY5Y cells, treatment with GLP-1 significantly enhances the expression levels of α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors, N-methyl-D-aspartic acid receptors, and dopamine receptor D1. It also increases the expression of synaptic proteins such as synapsin 1, synaptophysin, and postsynaptic density protein 95, while reducing the expression of the proliferation marker vimentin. This effect can be blocked by the PI3K inhibitor LY294002, confirming the regulatory role of the PI3K/Akt signaling axis in this process[34]. This suggests that semaglutide may enhance the maturation and functional connectivity of enteric neuronal synapses via the same pathway, providing cytological basis for its ability to reshape gut neural perception and improve signal processing precision. In hepatic stellate cell models, components extracted from serum exosomes of semaglutide-treated diabetic patients significantly lower the expression of α-smooth muscle actin, phospho-Smad2, and connective tissue growth factor, directly demonstrating anti-fibrotic activity[37]. In a streptozotocin-induced mouse model of sporadic Alzheimer's disease, GLP-1RAs significantly downregulate hippocampal levels of phosphorylated extracellular signal-regulated kinase 1/2, elevate the expression of phosphorylated GSK-3β (p-GSK-3β, Ser9) and phosphorylated CREB protein (p-CREB, Ser133), and increase the content of BDNF. These changes subsequently reduce β-amyloid deposition and tau protein phosphorylation, exerting a neuroprotective effect[38]. This study demonstrated that GLP-1RAs can upregulate BDNF via the PI3K/Akt/GSK-3β/CREB pathway. This neuroprotective and plasticity-modulating mechanism is evolutionarily conserved, supporting the notion that se
| Evidence type | Experimental model | Intervention/treatment | Key findings and outcomes | Core targets/pathways involved | Ref. |
| In vitro | Human neuroblastoma SH-SY5Y cells | Treatment with GLP-1 | Upregulated synaptic protein and neuronal receptor expression; blocked by PI3K inhibitor LY294002 | PI3K/Akt signaling axis | [34] |
| Hepatic stellate cell line LX-2 | Exosomes derived from serum of T2DM patients treated with semaglutide | Reduced expression of fibrotic markers | TGF-β/Smad signaling pathway | [37] | |
| In vivo | Streptozotocin-induced mouse model of sporadic Alzheimer’s disease | GLP-1 receptor agonist treatment | Increased hippocampal p-GSK-3β (Ser9), p-CREB (Ser133), and BDNF levels; decreased Aβ deposition and tau phosphorylation | GSK-3β/CREB/BDNF neuroprotective pathway | [33] |
| High-fat diet-induced mouse model of diabetic cardiomyopathy | GLP-1RA | Inhibited cardiomyocyte lipid accumulation; downregulated TGF-β1, collagen I/III, and oxidative stress; attenuated myocardial fibrosis and improved cardiac function | TGF-β signaling; oxidative stress; fibrosis | [38] |
Current evidence chain’s core gaps lie in the mechanistic gaps of semaglutide in regulating the TGF-β/BDNF signaling hub and controversies in ENS plasticity: The BDNF-TrkB signaling pathway has been confirmed as a core target for neuroprotection in neurodegenerative diseases (such as Parkinson’s disease and Alzheimer’s disease), and activation of the GLP-1 receptor can improve cognitive function and inhibit neuronal damage through this pathway. This conclusion has been verified in studies on GLP-1-related drugs such as sitagliptin[40,41]. However, as a widely used clinical GLP-1 receptor agonist, the interaction mechanism between semaglutide and the TGF-β/BDNF axis still has significant research gaps: Existing literature has not yet clarified the direct regulatory effect of semaglutide on the expression of TGF-β and BDNF in the ENS, nor is there evidence confirming whether TGF-β plays an upstream mediating role in the pathway by which semaglutide regulates BDNF expression[41]. More critically, the interaction mode (synergistic activation or negative feedback regulation) between TGF-β and BDNF in the ENS microenvironment has not been elucidated, and direct reports on the quadruple synergistic effect of semaglutide, TGF-β, BDNF, and ENS are lacking. This poses a core obstacle to deciphering the GBA regulatory network of semaglutide. At the same time, the plasticity of the adult ENS itself is controversial: Some studies suggest that the adult ENS still has neurogenic potential, but direct experimental evidence is insufficient, and the regulatory direction of TGF-β/BDNF on ENS neurogenesis and gliogenesis has not been unified (some studies indicate that BDNF promotes neural proliferation, while TGF-β may exert an inhibitory effect)[42,43]. This controversy over plasticity further complicates mechanistic research, leaving the core scientific question of whether semaglutide affects ENS plasticity through regulating the TGF-β/BDNF axis and thereby exerts neuroprotective effects unresolved to this day.
Potential verification approaches primarily center on two core dimensions: The application of cutting-edge technologies in mechanism verification - providing feasible paths for addressing the aforementioned gaps - and the unresolved clinical translation bottlenecks of GLP-1RAs: The development of cutting-edge technologies such as single-cell and spatial omics, organoid culture, and meta-omics has provided feasible paths for filling the aforementioned mechanistic gaps. Single-cell RNA-seq technology has been successfully used to resolve the heterogeneity of ENS neuron subtypes and can accurately localize the molecular characteristics of specific functional neurons (such as TGF-β or BDNF-positive neurons)[44,45], while spatial transcriptomics technology can further clarify the spatial distribution of these neurons in intestinal tissues, providing a precise single-cell level tool for verifying the regulation of the local TGF-β/BDNF axis in the ENS by semaglutide. In addition, the co-culture system of intestinal organoids and ENS can simulate the interaction microenvironment between the mucosal barrier and nerves in vivo[46], which is expected to directly verify whether TGF-β-mediated BDNF expression is dependent on semaglutide activation; meta-omics technology can correlate the expression regulatory relationship between gut microbial metabolites (such as SCFAs) and TGF-β/BDNF by characterizing the metabolomic and transcriptomic profiles of the gut microbiota[46]. Real-time monitoring technologies such as voltage-sensitive dyes can also dynamically capture the activity changes of ENS neurons after semaglutide treatment, providing functional evidence for mechanism verification[47]. However, the clinical translation of GLP-1RAs in the treatment of neurological diseases still faces multiple bottlenecks: The low blood-brain barrier penetration rate limits the efficiency of drugs in regulating the central nervous system and local ENS signaling axes; the lack of efficacy-predictive biomarkers centered on TGF-β/BDNF makes precision medicine difficult to achieve; long-term safety data are insufficient, and large-sample clinical studies above phase 3 are scarce; at the same time, digestive dysfunction often associated with neurological disease patients may exacerbate the risk of gastrointestinal adverse reactions of GLP-1RAs[41,48]. These bottlenecks are intertwined with gaps in mechanistic research, and there is an urgent need to achieve breakthroughs through a closed-loop research of “technological innovation - mechanism elucidation - clinical translation”.
Another key focus lies in the interaction blind spots and research prospects of the microbiota-ENS-TGF-β/BDNF axis: The interaction between the gut microbiota and ENS is a core link in GBA regulation. Microbial metabolites (such as SCFAs) can affect ENS function by regulating the integrity of the mucosal barrier, but it remains unclear whether this process involves the mediation of the TGF-β/BDNF axis[44,46]. Current research gaps predominantly center on three fundamental dimensions. First, the causal nexus between key bacterial genera (e.g., Akkermansia) and the expression of TGF-β/BDNF in the ENS remains unestablished, thus precluding the definitive determination of whether microbial dysbiosis modulates BDNF expression via the modulation of TGF-β signaling. Notably, the directionality, specificity, and contextual dependence of this hypothesized interaction have yet to be rigorously validated through mechanistic studies. Second, the molecular mechanisms orchestrating the crosstalk between microbial metabolites and the TGF-β/BDNF axis remain inadequately elucidated, with a conspicuous paucity of sequential experimental validation for the proposed pathway ‘microbial metabolite (e.g., SCFAs) → TGF-β receptor binding → BDNF transcriptional activation’. In particular, the molecular mediators, post-translational modifications, and tissue-specific regulatory checkpoints within this cascade remain incompletely delineated. Third, whether semaglutide-mediated modulation of the gut microbiota indirectly modulates ENS function via the TGF-β/BDNF axis - thereby completing the “drug-microbiota-signaling axis-nerve” regulatory cascade - remains unresolved. Furthermore, the extent to which this cascade contributes to the therapeutic efficacy of semaglutide in pathological contexts (e.g., gastrointestinal dysfunction, metabolic disorders) necessitates systematic investigation using integrative in vitro, in vivo, and translational models. Future studies can use technologies such as single-cell spatial omics, organoid co-culture, and combined meta-omics analysis to focus on the aforementioned interaction blind spots. To directly test the hypothesis that “SCFAs modulate TGF-β signaling to influence BDNF expression in the ENS”, we propose a tiered experimental strategy. First, in vitro mechanistic dissection should utilize primary enteric neuron-macrophage co-cultures isolated from mouse jejunum[21], treated with physiological SCFA concentrations (acetate 10 mmol/L, propionate 2 mmol/L, butyrate 1 mmol/L) that reflect colonic luminal levels[49,50]. G protein-coupled receptor 43 (GPR43) (free fatty acid receptor 2) knockdown via specific small interfering RNA[51,52] will verify receptor specificity [with quantitative polymerase chain reaction PCR (qPCR) confirming a knockdown efficiency ≥ 70%], while 10 μM TGF-β receptor 1 inhibitor SB525334[53,54] determines if SCFA effects on BDNF are TGF-β-dependent, with phosphorylated Smad2/3 and BDNF measured at 12, 24, and 48 hours by qPCR and Western blot[33]. Complementary organoid-ENS assembloids cultured with integrated neural crest cells can then be exposed to SCFAs with/without semaglutide (100 nM[55]) spatial transcriptomics combined with immunofluorescence co-localization will map BDNF (neuron-specific β-III tubulin co-localization) and TGF-β1 (macrophage-specific CD68 co-localization) expression along the crypt-villus axis[56], with expected SCFA-induced reduction of TGF-β1 in macrophage-rich regions and BDNF upregulation in neuronal clusters, alongside detection of intestinal barrier molecules (occludin, zonula occludens-1)[51]. Second, in vivo causality testing via germ-free mouse colonization with defined bacterial consortia - including low-SCFA (Proteobacteria-dominant[57]), high-butyrate (Anaerostipes rhamnosivorans + Akkermansia muciniphila + Eubacterium rectale[58]) and high-propionate (Parabacteroides goldsteinii[49]) communities - should be combined with semaglutide treatment (30 nmol/kg/day subcutaneous injection[59]) for 4 weeks, followed by qPCR, 16S ribosomal RNA sequencing, gas chromatography-mass spectrometry-based SCFA quantification[60], and whole-mount phosphorylated Smad2/3 immunostaining of myenteric plexus. To isolate neuronal GPR43 contribution, conditional GPR43 knockout mice (GPR43ΔENS) generated using Cre-loxP recombination[61] should undergo colorectal distension-visceromotor response testing for visceral sensitivity and dorsal root ganglion BDNF-TrkB signaling analysis by Western blot[56]. Third, feedback loop validation can be achieved by intestinal luminal in situ injection of enteric-specific adeno-associated virus-TGF-β1 (serotype adeno-associated virus 9, promoter villin 1/synapsin 1[62]) in the germ-free model; if SCFA-mediated BDNF upregulation is suppressed, this confirms TGF-β negative feedback. Epigenetic crosstalk should be mapped via 13C-acetate stable isotope-resolved metabolomics[58], tracking histone H3 lysine 9 acetylation incorporation at BDNF promoter and TGF-β1 enhancer regions by chromatin immunoprecipitation sequencing[63]. Parallel clinical translation requires a pilot trial (n = 30 type 2 diabetes mellitus patients, excluding Inflammatory bowel disease/irritable bowel syndrome cases[56]) measuring fecal SCFAs (gas chromatography-mass spectrometry[60]), plasma TGF-β1 (enzyme-linked immunosorbent assay[64]), and serum BDNF (electrochemiluminescence[65]) before/after 12 weeks semaglutide treatment, with Pearson correlation analysis testing whether SCFA changes (especially butyrate) predict TGF-β/BDNF shifts as response biomarkers. Meanwhile, develop clinical biomarkers centered on the TGF-β/BDNF axis, optimize the dosage form and administration regimen of GLP-1RAs to improve ENS targeting, and ultimately provide “mechanism - technology - clinical” trinity theoretical and practical support for the application of semaglutide in the treatment of neurological diseases.
Semaglutide, a prominent agent among GLP-1 receptor agonists, owes its remarkable glucose-lowering and metabolic benefits largely to its ability to reshape the GBA. Current evidence suggests its core mechanism involves widespread activation of GLP-1 receptors, which enhances BDNF-centered neurotrophic and anti-inflammatory pathways while suppressing pro-inflammatory TGF-β signaling. These dual actions converge on intestinal neuro-sensory units, fine-tuning vagal communication with the brain, fortifying the gut barrier, and mitigating neural inflammation in both the gut and the brain[66,67].
Unraveling this mechanism carries significant clinical implications. It broadens therapeutic potential and inspires next-generation drug discovery. Given its neuroprotective properties, semaglutide could be repurposed for neurological disorders like epilepsy and multiple sclerosis, moving beyond metabolic diseases. And, it paves the way for personalized medicine. Future therapy could be tailored by using biomarkers like gut microbiota or TGF-β/BDNF levels to predict a patient’s individual response[68,69].
In conclusion, deepening our understanding of how semaglutide modulates gut neural perception via the TGF-β/BDNF axis not only illuminates the dynamic dialogue between gut and brain but also unlocks new possibilities in addressing the global burden of metabolic and neurological diseases.
| 1. | Ullah A, Shen B. Immunomodulatory effects of anti-diabetic therapies: Cytokine and chemokine modulation by metformin, sodium-glucose cotransporter 2 inhibitors, and glucagon-like peptide-1 receptor agonists (2013-2025). Eur J Med Chem. 2025;299:118065. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 2. | Hawary OA, Wadie W, El-Said YAM, Hassan OF. Repurposing of semaglutide by targeting SIRT1 and TGF-β/Smad signaling in hepatic fibrosis. Naunyn Schmiedebergs Arch Pharmacol. 2025. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 3. | Li YK, Ma DX, Wang ZM, Hu XF, Li SL, Tian HZ, Wang MJ, Shu YW, Yang J. The glucagon-like peptide-1 (GLP-1) analog liraglutide attenuates renal fibrosis. Pharmacol Res. 2018;131:102-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 69] [Article Influence: 8.6] [Reference Citation Analysis (3)] |
| 4. | Wang Z, Wang M, Hu X, Li Y, Ma D, Li S, Zhao G, Xie Y, Shu Y, Yang J. Liraglutide, a Glucagon-like Peptide-1 Receptor Agonist, Attenuates Development of Cardiac Allograft Vasculopathy in a Murine Heart Transplant Model. Transplantation. 2019;103:502-511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 5. | Singh A. Brain-derived neurotrophic factor - a key player in the gastrointestinal system. Prz Gastroenterol. 2023;18:380-392. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 6. | Ty D, Kania B, Sotelo A, Wisco J. Gut Dysbiosis: A Review of the Effects of a High‐Fat Diet on Iron Regulation and Brain‐Derived Neurotrophic Factor (BDNF) Expression in the Brain‐Gut Axis. FASEB J. 2022;36. [DOI] [Full Text] |
| 7. | Amagase Y, Kambayashi R, Sugiyama A, Takei Y. Peripheral Regulation of Central Brain-Derived Neurotrophic Factor Expression through the Vagus Nerve. Int J Mol Sci. 2023;24:3543. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 20] [Reference Citation Analysis (0)] |
| 8. | Mi R, Cheng H, Chen R, Bai B, Li A, Gao F, Xue G. Effects and mechanisms of long-acting glucagon-like peptide-1 receptor agonist semaglutide on microglia phenotypic transformation and neuroinflammation after cerebral ischemia/reperfusion in rats. Brain Circ. 2024;10:354-365. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 9. | Ahuja S, Zaheer S. Multifaceted TGF-β signaling, a master regulator: From bench-to-bedside, intricacies, and complexities. Cell Biol Int. 2024;48:87-127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 25] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 10. | Jiang M, Ding Z, Huang Y, Jiang T, Xia Y, Gu D, Gu X, Bai H, Yao D. TGF-β1 Improves Nerve Regeneration and Functional Recovery After Sciatic Nerve Injury by Alleviating Inflammation. Biomedicines. 2025;13:872. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 11. | Hatamzade Esfahani N, Day AS. The Role of TGF-β, Activin and Follistatin in Inflammatory Bowel Disease. Gastrointest Disord. 2023;5:167-186. [RCA] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 12. | Wang Z, Zhang Q, Jiang Y, Zhou J, Tian Y. ASI-RIM neuronal axis regulates systemic mitochondrial stress response via TGF-β signaling cascade. Nat Commun. 2024;15:8997. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 13. | Peng D, Fu M, Wang M, Wei Y, Wei X. Targeting TGF-β signal transduction for fibrosis and cancer therapy. Mol Cancer. 2022;21:104. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 924] [Cited by in RCA: 846] [Article Influence: 211.5] [Reference Citation Analysis (4)] |
| 14. | Antonijevic M, Dallemagne P, Rochais C. Inducing neuronal regeneration and differentiation via the BDNF/TrkB signaling pathway: a key target against neurodegenerative diseases? Neural Regen Res. 2024;19:495-496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 15. | Zhu ZH, Jia F, Ahmed W, Zhang GL, Wang H, Lin CQ, Chen WH, Chen LK. Neural stem cell-derived exosome as a nano-sized carrier for BDNF delivery to a rat model of ischemic stroke. Neural Regen Res. 2023;18:404-409. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 109] [Reference Citation Analysis (0)] |
| 16. | Molska M, Mruczyk K, Cisek-Woźniak A, Prokopowicz W, Szydełko P, Jakuszewska Z, Marzec K, Trocholepsza M. The Influence of Intestinal Microbiota on BDNF Levels. Nutrients. 2024;16:2891. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 35] [Reference Citation Analysis (3)] |
| 17. | O'Leary OF, Ogbonnaya ES, Felice D, Levone BR, C Conroy L, Fitzgerald P, Bravo JA, Forsythe P, Bienenstock J, Dinan TG, Cryan JF. The vagus nerve modulates BDNF expression and neurogenesis in the hippocampus. Eur Neuropsychopharmacol. 2018;28:307-316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 116] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 18. | Miyazawa K, Itoh Y, Fu H, Miyazono K. Receptor-activated transcription factors and beyond: multiple modes of Smad2/3-dependent transmission of TGF-β signaling. J Biol Chem. 2024;300:107256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 35] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 19. | Shi Y, Li S, Zhang H, Zhu J, Che T, Yan B, Li J, Liu C. The effect of macrophage polarization on the expression of the oxytocin signalling system in enteric neurons. J Neuroinflammation. 2021;18:261. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 20. | Shi CJ, Lian JJ, Zhang BW, Cha JX, Hua QH, Pi XP, Hou YJ, Xie X, Zhang R. TGFβR-1/ALK5 inhibitor RepSox induces enteric glia-to-neuron transition and influences gastrointestinal mobility in adult mice. Acta Pharmacol Sin. 2023;44:92-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 21. | Viola MF, Chavero-Pieres M, Modave E, Delfini M, Stakenborg N, Estévez MC, Fabre N, Appeltans I, Martens T, Vandereyken K, Theobald H, Van Herck J, Petry P, Verheijden S, De Schepper S, Sifrim A, Liu Z, Ginhoux F, Azhar M, Schlitzer A, Matteoli G, Kierdorf K, Prinz M, Vanden Berghe P, Voet T, Boeckxstaens G. Dedicated macrophages organize and maintain the enteric nervous system. Nature. 2023;618:818-826. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 82] [Article Influence: 27.3] [Reference Citation Analysis (0)] |
| 22. | Zhu S, Yu Q, Xue Y, Li J, Huang Y, Liu W, Wang G, Wang L, Zhai Q, Zhao J, Zhang H, Chen W. Bifidobacterium bifidum CCFM1163 alleviates cathartic colon by activating the BDNF-TrkB-PLC/IP(3) pathway to reconstruct the intestinal nerve and barrier. Food Funct. 2025;16:2057-2072. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 23. | Wang Y, Liang J, Xu B, Yang J, Wu Z, Cheng L. TrkB/BDNF signaling pathway and its small molecular agonists in CNS injury. Life Sci. 2024;336:122282. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 87] [Reference Citation Analysis (0)] |
| 24. | Denman CR, Park SM, Jo J. Gut-brain axis: gut dysbiosis and psychiatric disorders in Alzheimer's and Parkinson's disease. Front Neurosci. 2023;17:1268419. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 28] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 25. | Lobo B, Tramullas M, Finger BC, Lomasney KW, Beltran C, Clarke G, Santos J, Hyland NP, Dinan TG, Cryan JF. The Stressed Gut: Region-specific Immune and Neuroplasticity Changes in Response to Chronic Psychosocial Stress. J Neurogastroenterol Motil. 2023;29:72-84. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 26. | Lin J, Xiao L, Nie X, Wang Z, Luo Y, Zhang L, Liu Y. Investigating the role of TGF-β and BDNF in cancer-related depression: a primary cross-sectional study. Support Care Cancer. 2024;32:365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 27. | Yang Y, Yang J, Ma T, Yang X, Yuan Y, Guo Y. The role and mechanism of TGF-β1 in the antidepressant-like effects of tetrahydrocurcumin. Eur J Pharmacol. 2023;959:176075. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 28. | Philips AV, Robbins DJ, Coleman MS, Barkely MD. Immunoaffinity purification and fluorescence studies of human adenosine deaminase. Biochemistry. 1987;26:2893-2903. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 29. | Lu R, Zhang L, Wang H, Li M, Feng W, Zheng X. Echinacoside exerts antidepressant-like effects through enhancing BDNF-CREB pathway and inhibiting neuroinflammation via regulating microglia M1/M2 polarization and JAK1/STAT3 pathway. Front Pharmacol. 2022;13:993483. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 47] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 30. | Zhang Y, Liu K, Li Y, Ma Y, Wang Y, Fan Z, Li Y, Qi J. D-beta-hydroxybutyrate protects against microglial activation in lipopolysaccharide-treated mice and BV-2 cells. Metab Brain Dis. 2023;38:1115-1126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 31. | Butera A, De Simone R, Potenza RL, Sanchez M, Armida M, Campanile D, Di Carlo N, Trenta F, Boirivant M, Ricceri L. Effects of a gut-selective integrin-targeted therapy in male mice exposed to early immune activation, a model for the study of autism spectrum disorder. Brain Behav Immun. 2024;115:89-100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 32. | Wang N, Li R, Feng B, Cheng Y, Guo Y, Qian H. Chicoric Acid Prevents Neuroinflammation and Neurodegeneration in a Mouse Parkinson's Disease Model: Immune Response and Transcriptome Profile of the Spleen and Colon. Int J Mol Sci. 2022;23:2031. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 29] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 33. | Hassan RM, Elsayed NS, Assaf N, Budzyńska B, Skalicka-Wożniak K, Ibrahim SM. Limettin and PD98059 Mitigated Alzheimer's Disease Like Pathology Induced by Streptozotocin in Mouse Model: Role of p-ERK1/2/p-GSK-3β/p-CREB/BDNF Pathway. J Neuroimmune Pharmacol. 2025;20:55. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 34. | Yang JL, Lin YT, Chen WY, Yang YR, Sun SF, Chen SD. The Neurotrophic Function of Glucagon-Like Peptide-1 Promotes Human Neuroblastoma Differentiation via the PI3K-AKT Axis. Biology (Basel). 2020;9:348. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 35. | Singh AA, Katiyar S, Song M. Phytochemicals Targeting BDNF Signaling for Treating Neurological Disorders. Brain Sci. 2025;15:252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 26] [Reference Citation Analysis (0)] |
| 36. | Wu L, Zhan Y, Wang Y. Semaglutide May Ameliorate Fibrosis and Inhibit Epithelial-Mesenchymal Transition in Intrauterine Adhesion Models. Int J Mol Sci. 2024;25:6196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 37. | Scavo MP, Lisco G, Depalo N, Rizzi F, Volpe S, Arrè V, Carrieri L, Notarnicola M, De Nunzio V, Curri ML, De Pergola G, Piazzolla G, Giannelli G. Semaglutide Modulates Extracellular Matrix Production of LX-2 Cells via Exosomes and Improves Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD). Int J Mol Sci. 2024;25:1493. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (8)] |
| 38. | Kuo CY, Tsou SH, Kornelius E, Chan KC, Chang KW, Li JC, Huang CN, Lin CL. The protective effects of liraglutide in reducing lipid droplets accumulation and myocardial fibrosis in diabetic cardiomyopathy. Cell Mol Life Sci. 2025;82:39. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 39. | Cheng D, Yang S, Zhao X, Wang G. The Role of Glucagon-Like Peptide-1 Receptor Agonists (GLP-1 RA) in Diabetes-Related Neurodegenerative Diseases. Drug Des Devel Ther. 2022;16:665-684. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 66] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 40. | Dong Q, Teng SW, Wang Y, Qin F, Li Y, Ai LL, Yu H. Sitagliptin protects the cognition function of the Alzheimer's disease mice through activating glucagon-like peptide-1 and BDNF-TrkB signalings. Neurosci Lett. 2019;696:184-190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 46] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 41. | Khan MA, Haider N, Singh T, Bandopadhyay R, Ghoneim MM, Alshehri S, Taha M, Ahmad J, Mishra A. Promising biomarkers and therapeutic targets for the management of Parkinson's disease: recent advancements and contemporary research. Metab Brain Dis. 2023;38:873-919. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 42. | Zhou Y, Su Y, Li S, Kennedy BC, Zhang DY, Bond AM, Sun Y, Jacob F, Lu L, Hu P, Viaene AN, Helbig I, Kessler SK, Lucas T, Salinas RD, Gu X, Chen HI, Wu H, Kleinman JE, Hyde TM, Nauen DW, Weinberger DR, Ming GL, Song H. Molecular landscapes of human hippocampal immature neurons across lifespan. Nature. 2022;607:527-533. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 246] [Cited by in RCA: 224] [Article Influence: 56.0] [Reference Citation Analysis (0)] |
| 43. | Simard S, Rahimian R, Davoli MA, Théberge S, Matosin N, Turecki G, Nagy C, Mechawar N. Spatial transcriptomic analysis of adult hippocampal neurogenesis in the human brain. J Psychiatry Neurosci. 2024;49:E319-E333. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 44. | Jarret A, Jackson R, Duizer C, Healy ME, Zhao J, Rone JM, Bielecki P, Sefik E, Roulis M, Rice T, Sivanathan KN, Zhou T, Solis AG, Honcharova-Biletska H, Vélez K, Hartner S, Low JS, Qu R, de Zoete MR, Palm NW, Ring AM, Weber A, Moor AE, Kluger Y, Nowarski R, Flavell RA. Enteric Nervous System-Derived IL-18 Orchestrates Mucosal Barrier Immunity. Cell. 2020;180:50-63.e12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 142] [Article Influence: 23.7] [Reference Citation Analysis (2)] |
| 45. | Ma J, Mistareehi A, Madas J, Kwiat AM, Bendowski K, Nguyen D, Chen J, Li DP, B Furness J, L Powley T, Cheng ZJ. Topographical organization and morphology of substance P (SP)-immunoreactive axons in the whole stomach of mice. J Comp Neurol. 2023;531:188-216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 46. | Tilocca B, Pieroni L, Soggiu A, Britti D, Bonizzi L, Roncada P, Greco V. Gut-Brain Axis and Neurodegeneration: State-of-the-Art of Meta-Omics Sciences for Microbiota Characterization. Int J Mol Sci. 2020;21:4045. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 61] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 47. | Vignali S, Peter N, Ceyhan G, Demir IE, Zeller F, Senseman D, Michel K, Schemann M. Recordings from human myenteric neurons using voltage-sensitive dyes. J Neurosci Methods. 2010;192:240-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 16] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 48. | Yassine HN, Solomon V, Thakral A, Sheikh-Bahaei N, Chui HC, Braskie MN, Schneider LS, Talbot K. Brain energy failure in dementia syndromes: Opportunities and challenges for glucagon-like peptide-1 receptor agonists. Alzheimers Dement. 2022;18:478-497. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 49. | Fawad JA, Luzader DH, Hanson GF, Moutinho TJ Jr, McKinney CA, Mitchell PG, Brown-Steinke K, Kumar A, Park M, Lee S, Bolick DT, Medlock GL, Zhao JY, Rosselot AE, Chou CJ, Eshleman EM, Alenghat T, Hong CI, Papin JA, Moore SR. Histone Deacetylase Inhibition by Gut Microbe-Generated Short-Chain Fatty Acids Entrains Intestinal Epithelial Circadian Rhythms. Gastroenterology. 2022;163:1377-1390.e11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 115] [Cited by in RCA: 102] [Article Influence: 25.5] [Reference Citation Analysis (0)] |
| 50. | Tripp P, Davis EC, Gurung M, Rosa F, Bode L, Fox R, LeRoith T, Simecka C, Seppo AE, Järvinen KM, Yeruva L. Infant Microbiota Communities and Human Milk Oligosaccharide Supplementation Independently and Synergistically Shape Metabolite Production and Immune Responses in Healthy Mice. J Nutr. 2024;154:2871-2886. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 51. | Song M, Zhang S, Zhang Z, Guo L, Liang W, Li C, Wang Z. Bacillus coagulans restores pathogen-induced intestinal dysfunction via acetate-FFAR2-NF-κB-MLCK-MLC axis in Apostichopus japonicus. mSystems. 2024;9:e0060224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 52. | Zhang X, Liu W, Zhang S, Wang J, Yang X, Wang R, Yan T, Wu B, Du Y, Jia Y. Wei-Tong-Xin ameliorates functional dyspepsia via inactivating TLR4/MyD88 by regulating gut microbial structure and metabolites. Phytomedicine. 2022;102:154180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 53. | Huang P, Wei G, Kirkpatrick JD, Lin Y, Tan L, Matta H, Nasser I, Huang M, Chen L, Petitjean M, Skelton-Badlani D, Gao W, Vaid K, Zhao S, Lugovskoy A, Alenzi M, Chen X, Gores GJ, Popov YV. Transposon-based oncogene integration in Abcb4(Mdr2)(-/-) mice recapitulates high susceptibility to cholangiocarcinoma in primary sclerosing cholangitis. J Hepatol. 2025;82:84-96. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 54. | Decato BE, Ammar R, Reinke-Breen L, Thompson JR, Azzara AV. Transcriptome analysis reveals key genes modulated by ALK5 inhibition in a bleomycin model of systemic sclerosis. Rheumatology (Oxford). 2022;61:1717-1727. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 55. | Pinto S, Viegas J, Cristelo C, Pacheco C, Barros S, Buckley ST, Garousi J, Gräslund T, Santos HA, Sarmento B. Bioengineered Nanomedicines Targeting the Intestinal Fc Receptor Achieve the Improved Glucoregulatory Effect of Semaglutide in a Type 2 Diabetic Mice Model. ACS Nano. 2024;18:28406-28424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 56. | Gao J, Xiong T, Grabauskas G, Owyang C. Mucosal Serotonin Reuptake Transporter Expression in Irritable Bowel Syndrome Is Modulated by Gut Microbiota Via Mast Cell-Prostaglandin E2. Gastroenterology. 2022;162:1962-1974.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 95] [Article Influence: 23.8] [Reference Citation Analysis (0)] |
| 57. | Lemons JMS, Conrad M, Tanes C, Chen J, Friedman ES, Roggiani M, Curry D, Chau L, Hecht AL, Harling L, Vales J, Kachelries KE, Baldassano RN, Goulian M, Bittinger K, Master SR, Liu L, Wu GD. Enterobacteriaceae Growth Promotion by Intestinal Acylcarnitines, a Biomarker of Dysbiosis in Inflammatory Bowel Disease. Cell Mol Gastroenterol Hepatol. 2024;17:131-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 46] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 58. | Grefhorst A, Kleemann AS, Havik S, Troise AD, De Pascale S, Scaloni A, Nieuwdorp M, Bui TPN. Intestinal Myo-Inositol Metabolism and Metabolic Effects of Myo-Inositol Utilizing Anaerostipes rhamnosivorans in Mice. Int J Mol Sci. 2025;26:9340. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 59. | Hansen HH, Pors S, Andersen MW, Vyberg M, Nøhr-Meldgaard J, Nielsen MH, Oró D, Madsen MR, Lewinska M, Møllerhøj MB, Madsen AN, Feigh M. Semaglutide reduces tumor burden in the GAN diet-induced obese and biopsy-confirmed mouse model of NASH-HCC with advanced fibrosis. Sci Rep. 2023;13:23056. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 32] [Reference Citation Analysis (0)] |
| 60. | Pensinger DA, Fisher AT, Dobrila HA, Van Treuren W, Gardner JO, Higginbottom SK, Carter MM, Schumann B, Bertozzi CR, Anikst V, Martin C, Robilotti EV, Chow JM, Buck RH, Tompkins LS, Sonnenburg JL, Hryckowian AJ. Butyrate Differentiates Permissiveness to Clostridioides difficile Infection and Influences Growth of Diverse C. difficile Isolates. Infect Immun. 2023;91:e0057022. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 50] [Reference Citation Analysis (1)] |
| 61. | Ming Z, Liu F, Moran HR, Lalonde RL, Adams M, Restrepo NK, Joshi P, Ekker SC, Clark KJ, Friedberg I, Sumanas S, Yin C, Mosimann C, Essner JJ, McGrail M. Lineage labeling with zebrafish hand2 Cre and CreERT2 recombinase CRISPR knock-ins. Dev Dyn. 2026;255:86-105. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 62. | Sun L, Zhang HB, Jiang HC, Li W, Li MK, Yang XY, Cai YY, Xue KK, Gou YS, Liu XY, Liang Q, Zuo LG, Hu JG, Qian F. LMO7 drives profibrotic fibroblast polarization and pulmonary fibrosis in mice through TGF-β signalling. Acta Pharmacol Sin. 2025;46:1930-1945. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 63. | Hu M, Alashkar Alhamwe B, Santner-Nanan B, Miethe S, Harb H, Renz H, Potaczek DP, Nanan RK. Short-Chain Fatty Acids Augment Differentiation and Function of Human Induced Regulatory T Cells. Int J Mol Sci. 2022;23:5740. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 79] [Reference Citation Analysis (0)] |
| 64. | Xiao X, Zheng M, Zhao Y, Huang K, Shi Y, Ren Y, Chen L, Sun J, Ye R, Zheng Z, Ruan Q. Quercetin ameliorates diminished ovarian reserve via modulating intestinal BNIP3/dct-1-mediated mitophagy and subsequently activating the ovarian TGF-β signaling pathway. J Ethnopharmacol. 2026;355:120736. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 65. | Wulff BS, Kuhre RE, Selvaraj M, Rehfeld JF, Niss K, Fels JJ, Anna S, Raun K, Gerstenberg MK. Improved leptin sensitivity and increased soluble leptin receptor concentrations may underlie the additive effects of combining PYY and exendin-4 on body weight lowering in diet-induced obese mice. Heliyon. 2024;10:e32009. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 66. | Cheng CY, Lo SC, Huang CN, Yang YS, Wang YH, Kornelius E. Association Between GLP-1 Receptor Agonist Use and Epilepsy Risk in Type 2 Diabetes. Neurology. 2026;106:e214509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (2)] |
| 67. | Sadek MA, Kandil EA, El Sayed NS, Sayed HM, Rabie MA. Semaglutide, a novel glucagon-like peptide-1 agonist, amends experimental autoimmune encephalomyelitis-induced multiple sclerosis in mice: Involvement of the PI3K/Akt/GSK-3β pathway. Int Immunopharmacol. 2023;115:109647. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 44] [Reference Citation Analysis (0)] |
| 68. | Nunn E, Jaiswal N, Gavin M, Uehara K, Stefkovich M, Drareni K, Calhoun R, Lee M, Holman CD, Baur JA, Seale P, Titchenell PM. Antibody blockade of activin type II receptors preserves skeletal muscle mass and enhances fat loss during GLP-1 receptor agonism. Mol Metab. 2024;80:101880. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 84] [Article Influence: 42.0] [Reference Citation Analysis (0)] |
| 69. | Melson E, Ashraf U, Papamargaritis D, Davies MJ. What is the pipeline for future medications for obesity? Int J Obes (Lond). 2025;49:433-451. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 87] [Cited by in RCA: 180] [Article Influence: 180.0] [Reference Citation Analysis (1)] |