Published online Mar 26, 2026. doi: 10.12998/wjcc.v14.i9.118187
Revised: January 23, 2026
Accepted: March 2, 2026
Published online: March 26, 2026
Processing time: 88 Days and 12.9 Hours
Immune checkpoint inhibitors (ICIs) have transformed oncology by enhancing the immune response against tumor cells. Despite their ‘targeted’ action, these mono
To determine the incidence, clinical spectrum, and outcomes of ICIRCAE in a diverse Middle Eastern cohort of cancer patients receiving ICIs, providing critical insights into risk stratification, early recognition, and management strategies in this unique population.
A retrospective observational study was conducted at an advanced oncology center, including all adult cancer patients who received at least one dose of an ICI from January 2015 to January 2023. Demographic, clinical, and echocardiographic data of patients who developed ICIRCAE were extracted. STATA 18 was used for statistical analysis, and ICIRCAE were classified based on clinical presentation and diagnostic findings. Continuous variables were reported as mean ± SD, and categorical variables as n (%).
During the eight-year study period, 14 patients (2.1%) out of 672 ICI recipients (mean age 54.2 ± 13.3 years; 70.5% male) experienced ICIRCAEs. The most commonly implicated drug was pembrolizumab (78.6%). With a median onset of 122 days after therapy, the most common complications were heart failure (42.9%), acute coronary syndrome (28.5%), and pericardial effusion (21.4%). Elevated pulmonary artery systolic pressure and reduced global longitudinal strain were among the salient echocardiographic changes observed in affected patients. Overall mortality was 42.9%, and 71.4% of cases had persistent permanent cardiac dysfunction.
Compared to Western cohorts, delayed-onset heart failure, rather than myocarditis, was the most frequent presentation in this first extensive Middle Eastern study of ICIRCAEs. To maximize ICI safety and predictability of risk profiles across diverse populations, these results emphasize the need for multicenter prospective studies, early surveillance, and regional risk-stratification tools.
Core Tip: Immune checkpoint inhibitors (ICIs) have revolutionized treatments and outcomes in oncology, but can cause serious ICI-related cardiac adverse events. This first comprehensive Middle Eastern study revealed that delayed-onset heart failure, rather than myocarditis, predominated among affected patients, with pembrolizumab being the most common implicated ICI. The 2.1% incidence and high mortality necessiates the need for early cardiac surveillance and risk stratification.
- Citation: Chapra A, Ata F, Abdellatif A, Nofal M, Daniyal A, Mohammad Younus A, Shah JZ, Rasul K. Incidence and clinical course of immune checkpoint inhibitor-related cardiac adverse events: A descriptive study from the Middle East. World J Clin Cases 2026; 14(9): 118187
- URL: https://www.wjgnet.com/2307-8960/full/v14/i9/118187.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i9.118187
Immune checkpoint inhibitors (ICIs) have revolutionized the treatment of a broad spectrum of malignancies by unleashing cytotoxic T-cell responses against tumor cells. Agents targeting programmed cell death protein 1 (PD-1), programmed death ligand 1 (PD-L1), and cytotoxic T-lymphocyte-associated protein 4 have led to significantly improved management of cancers such as melanoma, non-small cell lung cancer, and renal cell carcinoma, amongst others[1]. Initially approved as a salvage cancer therapy, ICI therapy has rapidly advanced up the treatment ladder in various cancers, becoming a first-line treatment option[2]. However, the success of ICIs is accompanied by the risk of immune-related adverse events (irAEs), which most commonly affect the endocrine system but can involve nearly any organ system, including the cardiovascular system[3,4].
Although relatively rare, cardiac irAEs, also known as ICI-related cardiac adverse events (ICIRCAEs), are one of the most serious complications of ICI use, due to the significant morbidity and mortality[5]. ICIs have been reported to cause different types of cardiac adverse events, of which myocarditis is the most common and the most life-threatening. Others include heart failure, pericarditis/pericardial effusion, coronary artery disease, and various arrhythmias[4,6-8]. While myocarditis is recognized as the most common and severe ICIRCAE, the exact prevalence and pathophysiology of these events remain poorly understood due to limited data, with reported incidence ranging from 0.06% to 3.30%[4,9,10]. However, emerging data from real-world cohorts suggest cardiac adverse events may occur at higher rates, especially when routine prospective surveillance is performed[11].
Most large-scale studies on ICIRCAEs have been conducted in East Asian, European, or North American populations, leaving a significant gap in understanding how these adverse events unfold in Middle Eastern and South Asian populations, which may have different genetic and environmental influences. This study aimed to address this gap by leveraging data from the national healthcare system of the State of Qatar, which serves as the sole provider of ICI therapy in the country, thereby enabling comprehensive and centralized data extraction. Qatar’s healthcare population is among the most ethnically diverse, with over 170 nationalities represented, including significant representation from Western and Southern Asia, Northern Africa, and other regions, making this an excellent opportunity to examine ICIRCAEs in a diverse patient cohort[12].
The main objective of this study was to determine the prevalence, clinical characteristics, and outcomes of ICICAEs amongst recipients of ICI therapy in Qatar. We sought to determine whether our cohort differed in the incidence or presentation of ICIRCAE compared with that reported in existing international data.
This retrospective observational study included adult patients in Qatar who received ICIs for various types of malignancies from January 2015 to January 2023.
This study included adult patients (≥ 18 years of age) with various malignancies who received at least one dose of ICI. Patients were included if they were treated with at least one dose of any ICI (PD-1 inhibitor, PD-L1 inhibitor, or cytotoxic T-lymphocyte-associated protein 4 inhibitor) and subsequently developed a new diagnosis of a cardiac adverse event, such as myocarditis, pericarditis, arrhythmias, acute coronary syndrome, cardiomyopathy, or heart failure or had worsening of a preexisting cardiac illness out of the ones mentioned above. Patients who did not receive at least one dose of the scheduled ICI therapy were excluded from the study.
The de-identified data were exported from the electronic medical records (Oracle Cerner®) to an Excel sheet and then transferred to statistical software (stata version 17.0, StataCorp LLC, College Station, TX, United States). A sample size calculation was not performed, as all patients admitted during the study period were included. The data included demographic information, medical and cancer history, details of ICI treatment (type and doses), and subsequent cardiac complications. The diagnosis of ICIRCAE was established via a diagnosis assigned by cardiology or cardio-oncology in outpatient visits or discharge diagnosis of patients hospitalized after ICI use. The primary outcome was to assess the incidence of ICIRCAEs. The secondary outcomes included the pre- and post-ICI therapy echocardiographic findings, diagnostic features, therapeutic interventions, and clinical outcomes of patients who developed ICIRCAEs.
Descriptive statistics summarized the cohort’s characteristics, with continuous variables presented as means ± SD or medians with interquartile ranges (IQRs) depending on distributional normality. Categorical variables are presented as
The Institutional Review Board of the Hamad Medical Corporation approved the study (protocol ID MRC-01-24-488). Given the study’s retrospective nature, the Institutional Review Board waived informed consent. All patient data were deidentified to maintain confidentiality and compliance with ethical guidelines and regulations. The study adhered to the principles outlined in the Declaration of Helsinki.
The study cohort comprised 672 adult patients with various malignancies who received at least one dose of an ICI during the nine-year study period. The mean age at ICI initiation was 54.2 years (± 13.3), with 70.5% being male. The cohort was ethnically diverse, with Western (35.7%) and South Asian (29.3%) groups representing the largest proportions (Table 1). Pembrolizumab (61.5%) was the most commonly used ICI, followed by nivolumab (24.1%) and atezolizumab (12.5%), among others. The most common malignancies treated were lung cancer (24.1%) and gastrointestinal tumors (18.0%). Distribution of cancers among the study cohort is shown in Table 2.
| Characteristic | Overall (n = 672) |
| Age at visit, year | 54.2 ± 13.3 |
| Gender | |
| Males | 474 (70.5) |
| Females | 198 (29.5) |
| Ethnicity | |
| Western Asia | 240 (35.7) |
| Southern Asia | 197 (29.3) |
| Northern Africa | 115 (17.1) |
| South-Eastern Asia | 63 (9.4) |
| Eastern Africa | 16 (2.4) |
| Northern America | 12 (1.8) |
| Others | 29 (4.3) |
| First BMI result, kg/m2 | 26.2 ± 11.4 |
| Last BMI result, kg/m2 | 26.2 ± 11.6 |
| ICI therapy | |
| Atezolizumab | 84 (12.5) |
| Avelumab | 4 (0.6) |
| Durvalumab | 8 (1.2) |
| Ipilimumab | 1 (0.2) |
| Nivolumab | 162 (24.1) |
| Pembrolizumab | 413 (61.5) |
| Type of cancer | Number of patients (%) |
| Lung cancer | 162 (24.1) |
| GIT cancer | 121 (18.0) |
| Renal cancer | 64 (9.5) |
| Hepatobiliary cancer | 62 (9.2) |
| Oral cavity cancer | 47 (7.0) |
| Breast cancer | 40 (6.0) |
| Skin cancer | 36 (5.4) |
| Endometrial cancer | 17 (2.5) |
| Lymphoma | 14 (2.1) |
| Other | 109 (16.2) |
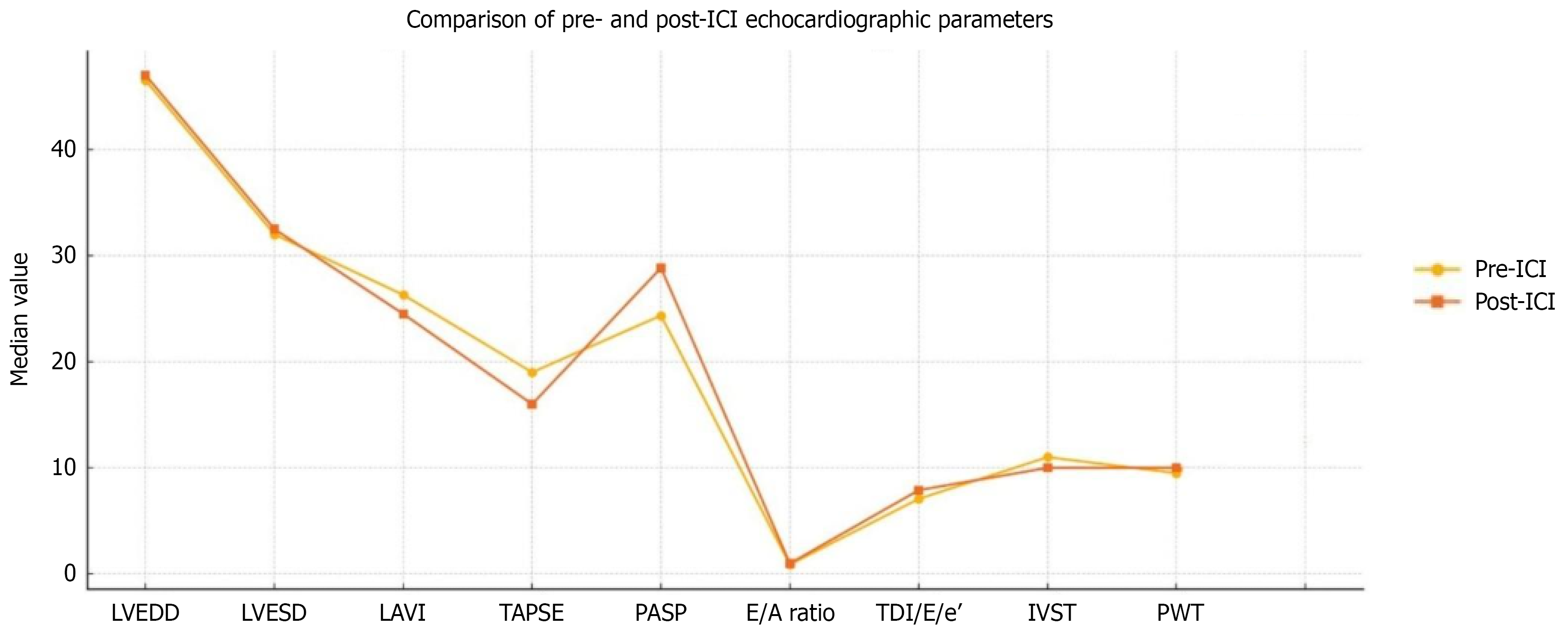
Cardiac complications occurred in 14 patients (Table 3), with a point prevalence of 2.1%. The median age of affected patients was 59.5 years (54-64), and 78.6% were male. Pembrolizumab was the most implicated ICI (78.6% of cases), followed by atezolizumab (14.3%) and nivolumab (7.1%). The median time from ICI initiation to onset of cardiotoxicity was 122 days (92-355). Heart failure (42.9%), acute coronary syndrome (28.5%), and pericardial effusion (21.4%) were the most frequently observed complications, followed by other less commonly observed cardiac complications (Table 4). Based on the National Cancer Institute Common Terminology Criteria for Adverse Events criteria, 21.4% were grade 2 or 4 each, and 28.6% were grade 3 and 5 each. Tables 5 and 6 summarize changes in key echocardiographic markers of cardiac structure and function before and after ICI therapy among the patients who ultimately developed cardiac complications. The trend is also shown in Figure 1.
| Characteristic | Value |
| Median age, years (IQR) | 59.5 (54-64) |
| Gender | |
| Male | 11 (78.6) |
| Female | 3 (21.4) |
| Nationality | |
| Filipino | 3 (21.4) |
| Jordanian | 3 (21.4) |
| Syrian | 2 (14.3) |
| Bahraini | 1 (7.1) |
| Bangladeshi | 1 (7.1) |
| Egyptian | 1 (7.1) |
| Indian | 1 (7.1) |
| Palestinian | 1 (7.1) |
| Qatari | 1 (7.1) |
| Comorbidities | |
| Hypertension | 9 (64.3) |
| Dyslipidemia | 7 (50.0) |
| Chronic kidney disease | 6 (42.9) |
| Type 2 diabetes | 5 (35.7) |
| Smoking | 4 (28.6) |
| Ischemic heart disease | 3 (21.4) |
| Hypothyroidism | 2 (14.3) |
| Type 1 diabetes | 1 (7.1) |
| Stroke | 1 (7.1) |
| Chronic heart failure | 1 (7.1) |
| Peripheral vascular disease | 1 (7.1) |
| Previous cardiomyopathy | 1 (7.1) |
| Previous arrhythmia | 1 (7.1) |
| Type of ICI administered | |
| Pembrolizumab | 11 (78.6) |
| Atezolizumab | 2 (14.3) |
| Nivolumab | 1 (7.1) |
| Characteristic | Value |
| Median time from ICI start to cardiotoxicity, days (IQR) | 122 (92-355) |
| Type of cardiac complication1 | |
| Myocarditis | 2 (14.3) |
| Atrial fibrillation | 1 (7.1) |
| Heart block (complete) | 1 (7.1) |
| Heart failure | 6 (42.9) |
| Cardiomyopathy (ischemic) | 1 (7.1) |
| Pericardial effusion | 3 (21.4) |
| Cardiac tamponade | 1 (7.1) |
| Cardiogenic shock | 1 (7.1) |
| ACS/CAD | 4 (28.5) |
| Sudden cardiac death | 1 (7.1) |
| NCI CTCAE grade | |
| Grade 1 | 0 |
| Grade 2 | 3 (21.4) |
| Grade 3 | 4 (28.6) |
| Grade 4 | 3 (21.4) |
| Grade 5 (death) | 4 (28.6) |
| Variable | Pre-ICI | Post-ICI |
| LVEDD (mm) | 46.5 (42-50) | 47 (37.5-52) |
| LVESD (mm) | 32 (28-35) | 32.5 (27-37.5) |
| Left atrial volume index (mL/m2) | 26.3 (21.9-30.9) | 24.5 (21.4-28.2) |
| TAPSE (mm) | 19 (15-22) | 16 (14-20) |
| PASP (mmHg) | 24.35 (21.84-32.04) | 28.84 (17.22-31.67) |
| Mitral E/A ratio | 0.89 (0.58-1.43) | 0.98 (0.47-1.07) |
| TDI E/e’ ratio | 7.07 (5.29-10.61) | 7.89 (7.8-8) |
| IVST (mm) | 11 (9-11.5) | 10 (8.5-11.5) |
| PWT (mm) | 9.5 (7.5-11) | 10 (8-11) |
| Global longitudinal strain (%) | -17 ± 1.7 | -12.1 ± 3.2 |
| Ejection fraction category (%) | Pre-ICI | Post-ICI |
| < 30 (severe dysfunction) | 1 (7.1) | 1 (7.1) |
| 30-39 (moderate dysfunction) | 0 (0.0) | 3 (21.4) |
| 40-49 (mild dysfunction) | 1 (7.1) | 3 (21.4) |
| 50-55 (borderline normal) | 5 (35.7) | 2 (14.3) |
| > 55 (normal) | 7 (50.0) | 5 (35.7) |
Of the 14 patients with post-ICI cardiovascular system complications, seven (50.0%) required hospitalization, with two admitted to the intensive care unit (Table 7). All 14 patients had ICI therapy permanently discontinued following the cardiac complication. Corticosteroid therapy was administered to 15.4% of patients, while 61.5% received adjunctive therapy, including beta-blockers, angiotensin-converting enzyme inhibitors, or angiotensin II receptor blockers, and 7.7% received spironolactone. Median troponin level was 41 ng/L (20-534), and median N-terminal pro-B-type natriuretic peptide was 494 pg/mL (373.5-3052) (Table 6). Cardiac magnetic resonance imaging features suggestive of myocarditis were seen in 1 patient (7.1%), while eight patients (57.1%) had reduced left ventricular ejection fraction on echocardiography. Despite treatment, only 1 patient (7.1%) experienced complete resolution of cardiac dysfunction, and 10 patients (71.4%) had persistent cardiac impairment at the last follow-up.
| Characteristic | Value |
| Median troponin-T level, ng/L | 41 (20-534) |
| Median NT-proBNP, pg/mL | 494 (373.5-3052) |
| Cardiac magnetic resonance imaging suggestive of myocarditis | 1 (7.1) |
| Reduced LVEF | 8 (57.1) |
| Medical management | |
| Beta-blockers | 8 (61.5) |
| ACEi/ARB/ARNI | 6 (46.2) |
| Aspirin | 5 (38.5) |
| Statin | 4 (30.8) |
| Clopidogrel | 3 (23.1) |
| SGLT2 inhibitors | 3 (23.1) |
| Corticosteroids | 2 (15.4) |
| Loop diuretics | 2 (15.4) |
| Spironolactone | 1 (7.7) |
| Anti-arrhythmias | 1 (7.7) |
| Ivabradine | 1 (7.7) |
| Hospitalized | 7 (50) |
| ICU admission | 2 (14.3) |
| ICI therapy discontinued | 14 (100) |
| Mortality | |
| In-hospital | 4 (28.6) |
| Died after discharge | 2 (14.3) |
| Permanent cardiac dysfunction | 10 (71.4) |
| Resolution of cardiac toxicity | 1 (7.1) |
This pioneering regional study revealed two notable trends. First, heart failure, rather than myocarditis, was the predominant manifestation of ICI-associated cardiotoxicity in our cohort. This contrasts with prior studies, which consistently identify myocarditis as the most frequent and life-threatening cardiac complication linked to ICI’s[9,13,14], including a recent systematic review by Shalata et al[15], which compiled 134 published cases and literature reviews. Second, the median time to onset of cardiotoxicity in our cohort was 122 days, significantly longer than the 17-65-day range reported in the literature, during which most immune-related cardiac events, particularly myocarditis, tend to occur[9,13-15]. These trends may reflect not only differences in malignancy types and treatment regimens but also differences in the population's genomic makeup, underscoring the importance of evaluating drug effects across popu
Data on ICI-associated toxicities at the regional level are limited. Before the current study, only one study conducted in Saudi Arabia specifically explored ICI-associated cardiotoxicity[19]. The authors reported that 1.2% of their 428-patient cohort suffered cardiac complications, most prominently induced by durvalumab, the anti-PD-L1. The median time to onset of cardiotoxicity was around 52 days. However, the study was unable to delineate the pattern of cardiac invol
The most commonly implicated ICI in our cohort was pembrolizumab, affecting 11 out of 14 patients, followed by atezolizumab and nivolumab. Such a pattern is consistent with data from large meta-analyses implicating those specific agents, as well as others from their class (PD-1 and PD-L1 antagonists), in the bulk of ICI-cardiotoxicity. This alignment with global data underscores the importance of vigilance when using these agents, especially in patients at higher risk.
The three echocardiographic parameters most affected by ICI therapy were global longitudinal strain (GLS), pulmonary artery systolic pressure, and tricuspid annular plane systolic excursion, all of which reflect ventricular dysfunction, consistent with our heart failure-dominant cohort. Consistent with prior studies, our affected patients had a greater mean reduction in GLS on echocardiography than in ejection fraction, underscoring the importance of GLS as an earlier and more reliable indicator of ventricular dysfunction and subclinical heart failure. This finding underscores the importance of GLS in alerting physicians to the patients who deserve closer monitoring and earlier initiation of appropriate therapy.
In terms of cardiac enzymes, our patients had a median high-sensitivity troponin-T level of 41 ng/L (20-534) (normal cutoff used was for males ≤ 15 ng/L and for females ≤ 10 ng/L), which is evidently lower than the levels mentioned in other myocarditis-predominant studies, as would be clinically expected. For example, Pereyra Pietri et al[20] reported a median troponin value of 351 ng/L (IQR: 1251) in definitively diagnosed ICI myocarditis. Likewise, Vasbinder et al[21] reported a median troponin value of 393 pg/mL (IQR: 110-1323 pg/mL) in ICI myocarditis. Many of the affected patients in this study also shared baseline risk factors previously associated with ICI-related cardiotoxicity, including hypertension, chronic kidney disease, diabetes mellitus, pre-existing cardiovascular disease, smoking, male sex, and age over 65, affirming their relevance in risk stratification tools[22-27].
Regarding outcomes, most of the patients in our cohort had permanent cardiac dysfunction (71.4%), and only one out of 14 patients experienced resolution of cardiac toxicity, which is overall consistent with the high ICI-cardiac morbidity and mortality reported in the literature. Due to the low incidence of myocarditis, only two patients were candidates for steroid therapy. Although the heart failure patients were appropriately initiated on medical management, including beta-blockers, angiotensin-converting enzyme inhibitors, angiotensin II receptor blockers, angiotensin receptor neprilysin inhibitors, and sodium-glucose cotransporter 2 inhibitors, this study lacked sufficient follow-up to assess their long-term response. While such therapies remain the cornerstone of heart failure management, their impact on reversing ICI-associated cardiac dysfunction remains uncertain. Future studies with more extended follow-up periods are needed to determine whether early detection and initiation of therapy can alter the trajectory of ICI-cardiomyopathy.
The main strength of this study lies in its ability to explore the cardiotoxic effects of ICIs in a highly diverse and previously understudied cohort, providing substantial data on myocardial changes, including echocardiographic patterns. Despite providing novel regional insights and encompassing a largely multi-ethnic population, our study has several limitations, pertinent to its retrospective nature. While the low incidence mirrors the real-world occurrence of ICI-cardiotoxicity, it limits the generalizability of our results to other populations that may differ. Several confounders may have influenced the ICIRCAE, but these could not be controlled due to the study’s design limitations. The male predominance of our cohort also limits applicability to female patients. Additionally, the single-center, retrospective design introduces selection bias and limits the external validity of our findings. Finally, cardiotoxicities linked to ICIs were identified from physician documentation in electronic health records, introducing potential subjectivity due to the absence of a standardized causality assessment using a predefined scoring system, which would make it more objective and less prone to bias.
Future research should prioritize subgroup analyses within larger, multi-center cohorts, stratified by the presence or absence of ICI-related cardiac complications, to assess critical clinical outcomes. While no globally accepted risk score currently exists, several retrospective models have been proposed. Studies by Heilbroner et al[26] and Chen et al[27], among others, have incorporated a range of baseline demographic variables (e.g., age, sex, smoking status, obesity), clinical comorbidities (such as chronic obstructive pulmonary disease, heart failure, and diabetes), laboratory markers (including cardiac enzymes, pro-B-type natriuretic peptide, and lymphocyte-to-monocyte ratio), and ICI treatment characteristics to develop risk prediction schemes. However, these models were designed to predict early-onset myocarditis and are primarily based on Western population data, which may limit their applicability across diverse clinical settings. As data evolves globally, more prospective real-world studies are needed to develop optimal guidelines for safe and effective use of ICI in the era of precision medicine.
This comprehensive regional study highlights the heterogeneity of cardiac involvement in ICI-associated cardiotoxicity in a previously unexplored patient population. This study reported a heart failure predominant pattern of ICI-associated cardiotoxicity with a later median onset of presentation, contrasting with the early-onset, myocarditis-predominant phenotype typically reported in Western cohorts. Consistent with prior literature, the most implicated agents in our cohort were PD-1 and PD-L1 inhibitors. Our findings also confirm the established relevance of baseline cardiovascular status, echocardiographic parameters of depressed ventricular function, and biochemical markers for risk stratification before ICI therapy. Primarily derived from Arab and Asian cohorts, our data provide valuable insights amid the paucity of regional studies addressing ICI cardiotoxicity. However, prospective cohort studies are imperative that focus on the special characteristics and risk profiles of underrepresented populations requiring ICI therapy. The development of more nuanced risk scores may enhance early detection, support timely management, and ultimately improve the outcomes of ICI-associated cardiotoxicity.
| 1. | Brahmer JR, Lacchetti C, Schneider BJ, Atkins MB, Brassil KJ, Caterino JM, Chau I, Ernstoff MS, Gardner JM, Ginex P, Hallmeyer S, Holter Chakrabarty J, Leighl NB, Mammen JS, McDermott DF, Naing A, Nastoupil LJ, Phillips T, Porter LD, Puzanov I, Reichner CA, Santomasso BD, Seigel C, Spira A, Suarez-Almazor ME, Wang Y, Weber JS, Wolchok JD, Thompson JA; National Comprehensive Cancer Network. Management of Immune-Related Adverse Events in Patients Treated With Immune Checkpoint Inhibitor Therapy: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2018;36:1714-1768. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3072] [Cited by in RCA: 2805] [Article Influence: 350.6] [Reference Citation Analysis (4)] |
| 2. | Rossi S, Masini S, Finocchiaro G, Lorenzi E, Toschi L, Santoro A. Retreatment with Immune Checkpoint Inhibitors in the New Scenario of Immunotherapy in Non-Small Cell Lung Cancer. Cancers (Basel). 2024;16:1683. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 3. | Ata F, Khan AA, Algorani E, Alsharafi AFM, Shdid RA, Nofal M, Ibrahim AR, Abdullah L, Annan KYE, Al-Bkoor TEH, Rasul KI, Elhadd T, Surchi H. The burden and clinical trajectory of immune checkpoint inhibitor-induced endocrinopathies: an 8-year experience. BMC Med. 2024;22:588. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 4. | Ball S, Ghosh RK, Wongsaengsak S, Bandyopadhyay D, Ghosh GC, Aronow WS, Fonarow GC, Lenihan DJ, Bhatt DL. Cardiovascular Toxicities of Immune Checkpoint Inhibitors: JACC Review Topic of the Week. J Am Coll Cardiol. 2019;74:1714-1727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 172] [Article Influence: 24.6] [Reference Citation Analysis (0)] |
| 5. | Zarifa A, Kim JW, Lopez-Mattei J, Palaskas N, Iliescu C, Kim PY. Cardiac Toxicities Associated with Immune Checkpoints Inhibitors: Mechanisms, Manifestations and Management. Korean Circ J. 2021;51:579-597. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 6. | Jensen G, Wang X, Kuempel J, Palaskas N, Chen Z, Yu W, Chen Y, Mohammad H, Luo W, Chang J. Immune checkpoint inhibitor-associated myocarditis: a historical and comprehensive review. Am J Physiol Heart Circ Physiol. 2025;328:H734-H751. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 7. | Lyon AR, Yousaf N, Battisti NML, Moslehi J, Larkin J. Immune checkpoint inhibitors and cardiovascular toxicity. Lancet Oncol. 2018;19:e447-e458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 498] [Cited by in RCA: 447] [Article Influence: 55.9] [Reference Citation Analysis (0)] |
| 8. | Palaskas N, Lopez-Mattei J, Durand JB, Iliescu C, Deswal A. Immune Checkpoint Inhibitor Myocarditis: Pathophysiological Characteristics, Diagnosis, and Treatment. J Am Heart Assoc. 2020;9:e013757. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 407] [Cited by in RCA: 373] [Article Influence: 62.2] [Reference Citation Analysis (0)] |
| 9. | Mahmood SS, Fradley MG, Cohen JV, Nohria A, Reynolds KL, Heinzerling LM, Sullivan RJ, Damrongwatanasuk R, Chen CL, Gupta D, Kirchberger MC, Awadalla M, Hassan MZO, Moslehi JJ, Shah SP, Ganatra S, Thavendiranathan P, Lawrence DP, Groarke JD, Neilan TG. Myocarditis in Patients Treated With Immune Checkpoint Inhibitors. J Am Coll Cardiol. 2018;71:1755-1764. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1309] [Cited by in RCA: 1208] [Article Influence: 151.0] [Reference Citation Analysis (0)] |
| 10. | Johnson DB, Balko JM, Compton ML, Chalkias S, Gorham J, Xu Y, Hicks M, Puzanov I, Alexander MR, Bloomer TL, Becker JR, Slosky DA, Phillips EJ, Pilkinton MA, Craig-Owens L, Kola N, Plautz G, Reshef DS, Deutsch JS, Deering RP, Olenchock BA, Lichtman AH, Roden DM, Seidman CE, Koralnik IJ, Seidman JG, Hoffman RD, Taube JM, Diaz LA Jr, Anders RA, Sosman JA, Moslehi JJ. Fulminant Myocarditis with Combination Immune Checkpoint Blockade. N Engl J Med. 2016;375:1749-1755. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1984] [Cited by in RCA: 1828] [Article Influence: 182.8] [Reference Citation Analysis (0)] |
| 11. | Isawa T, Toi Y, Sugawara S, Taguri M, Toyoda S. Incidence, Clinical Characteristics, and Predictors of Cardiovascular Immune-Related Adverse Events Associated with Immune Checkpoint Inhibitors. Oncologist. 2022;27:e410-e419. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 20] [Reference Citation Analysis (0)] |
| 12. | Zahid M, Khan AA, Ata F, Yousaf Z, Naushad VA, Purayil NK, Chandra P, Singh R, Kartha AB, Elzouki AYA, Al Mohanadi DHSH, Al-Mohammed AAAA. Medical Admission Prediction Score (MAPS); a simple tool to predict medical admissions in the emergency department. PLoS One. 2023;18:e0293140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 13. | Gan L, Liu D, Ma Y, Chen X, Dai A, Zhao S, Jin X, Gu G. Cardiotoxicity associated with immune checkpoint inhibitors: Current status and future challenges. Front Pharmacol. 2022;13:962596. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 24] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 14. | Zhou YW, Zhu YJ, Wang MN, Xie Y, Chen CY, Zhang T, Xia F, Ding ZY, Liu JY. Immune Checkpoint Inhibitor-Associated Cardiotoxicity: Current Understanding on Its Mechanism, Diagnosis and Management. Front Pharmacol. 2019;10:1350. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 82] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 15. | Shalata W, Abu-Salman A, Steckbeck R, Mathew Jacob B, Massalha I, Yakobson A. Cardiac Toxicity Associated with Immune Checkpoint Inhibitors: A Systematic Review. Cancers (Basel). 2021;13:5218. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 87] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 16. | Wang C, Zhao G, Zhang Z, Yang L, Liu S, Li G, Wang H, Huang J, Wang S, Li N. Immune checkpoint inhibitor-associated myocarditis: a systematic analysis of case reports. Front Immunol. 2023;14:1275254. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 17. | Jiménez-Alejandre R, Ruiz-Fernández I, Martín P. Pathophysiology of Immune Checkpoint Inhibitor-Induced Myocarditis. Cancers (Basel). 2022;14:4494. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 34] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 18. | Bomze D, Hasan Ali O, Bate A, Flatz L. Association Between Immune-Related Adverse Events During Anti-PD-1 Therapy and Tumor Mutational Burden. JAMA Oncol. 2019;5:1633-1635. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 120] [Cited by in RCA: 110] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 19. | Alwhaibi A, Alenazi MA, Alghadeer S, Mansy W, Alsaif RA, Abualreesh NE, Alanazi RJ, Alroumi A, Alanazi SA. A Real-World Comparison of the Safety Profile for Immune Checkpoint Inhibitors in Oncology Patients. J Clin Med. 2025;14:388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 20. | Pereyra Pietri M, Farina JM, Scalia IG, Mahmoud AK, Roarke M, Wasef B, Tagle-Cornell C, Kenyon CR, Abbas MT, Ali NB, Awad KA, Javadi N, Bismee NN, Larsen CM, Herrmann J, Arsanjani R, Ayoub C. Comparison of the diagnostic and prognostic value of criteria for immune checkpoint inhibitor related myocarditis. Cardiooncology. 2025;11:30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 21. | Vasbinder A, Chen Y, Procureur A, Gradone A, Azam TU, Perry D, Shadid H, Anderson E, Catalan T, Blakely P, Nelapudi N, Fardous M, Bretagne MC, Adie SK, Pogue KT, Leja M, Yentz S, Schneider B, Fecher LA, Lao CD, Salem JE, Hayek SS. Biomarker Trends, Incidence, and Outcomes of Immune Checkpoint Inhibitor-Induced Myocarditis. JACC CardioOncol. 2022;4:689-700. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 63] [Reference Citation Analysis (0)] |
| 22. | Marso SP, Baeres FMM, Bain SC, Goldman B, Husain M, Nauck MA, Poulter NR, Pratley RE, Thomsen AB, Buse JB; LEADER Trial Investigators. Effects of Liraglutide on Cardiovascular Outcomes in Patients With Diabetes With or Without Heart Failure. J Am Coll Cardiol. 2020;75:1128-1141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 92] [Article Influence: 15.3] [Reference Citation Analysis (3)] |
| 23. | Torrente M, Blanco M, Franco F, Garitaonaindia Y, Calvo V, Collazo-Lorduy A, Gutiérrez L, Sánchez JC, González-Del-Alba A, Hernández R, Méndez M, Cantos B, Núñez B, Sousa PAC, Provencio M. Assessing the risk of cardiovascular events in patients receiving immune checkpoint inhibitors. Front Cardiovasc Med. 2022;9:1062858. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 24. | de Jesus M, Chanda A, Grabauskas T, Kumar M, Kim AS. Cardiovascular disease and lung cancer. Front Oncol. 2024;14:1258991. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 21] [Reference Citation Analysis (0)] |
| 25. | Tan S, Sivakumar S, Segelov E, Nicholls SJ, Nelson AJ. Cardiovascular risk factor reporting in immune checkpoint inhibitor trials: A systematic review. Cancer Epidemiol. 2023;83:102334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 26. | Heilbroner SP, Few R, Mueller J, Chalwa J, Charest F, Suryadevara S, Kratt C, Gomez-Caminero A, Dreyfus B, Neilan TG. Predicting cardiac adverse events in patients receiving immune checkpoint inhibitors: a machine learning approach. J Immunother Cancer. 2021;9:e002545. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 27. | Chen X, Jiang A, Zhang R, Fu X, Liu N, Shi C, Wang J, Zheng X, Tian T, Liang X, Ruan Z, Yao Y. Immune Checkpoint Inhibitor-Associated Cardiotoxicity in Solid Tumors: Real-World Incidence, Risk Factors, and Prognostic Analysis. Front Cardiovasc Med. 2022;9:882167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 21] [Reference Citation Analysis (0)] |