Published online Apr 26, 2026. doi: 10.12998/wjcc.v14.i12.119112
Revised: February 3, 2026
Accepted: March 6, 2026
Published online: April 26, 2026
Processing time: 86 Days and 4 Hours
Chemotherapy-induced nausea and vomiting is common in patients who receive highly emetogenic chemotherapy (HEC). Olanzapine acts on multiple neurotransmitters and effectively reduces delayed-phase nausea.
To compare the safety and efficacy of olanzapine-based vs neurokinin-1 receptor antagonist-based antiemetic regimens in adults receiving HEC.
This review followed Preferred Reporting Items for Systematic Reviews and Meta-analyses 2020 guidelines and searched various databases like PubMed, Scopus, etc., between 2020-2025. 14 studies were included in the systematic review with 11 meeting the criteria for meta-analysis. Primary outcomes were complete response (no vomiting, no rescue medication) and nausea control during acute (0-24 hours), delayed (25-120 hours), and overall (0-120 hours) phases. Data were pooled using random-effects models. Risk of bias was assessed with Cochrane risks of bias 2.0 and risk of bias in nonrandomized studies of interventions version I tools.
Olanzapine-based regimens showed higher nausea control, with no-nausea rates of 89% vs 76% (P < 0.001) and 96% vs 87% (P = 0.005). Acute complete response ranged from 66% to 100%, delayed complete response from 55% to 94%, and overall complete response from 63% to 91%. The pooled complete response odds ratio was 48.68 (95% confidence interval: 8.33-284.64; P < 0.0001). Sedation occurred in 10%-53% of patients and showed dose de
Olanzapine-based regimens are at least as effective as neurokinin-1 antagonist therapy for chemotherapy-induced nausea and vomiting in HEC, particularly for delayed nausea. Low-dose (5 mg) olanzapine provides effective, well-tolerated, oral, and cost-efficient prophylaxis, supporting its use as a first-line antiemetic.
Core Tip: Chemotherapy-induced nausea and vomiting commonly occur in patients receiving highly emetogenic chemotherapy, with rates over 90% without prophylaxis. Effective prevention preserves quality of life, supports treatment adherence, and reduces healthcare use. This study demonstrated that olanzapine-based regimens effectively prevent chemotherapy-induced nausea and vomiting in patients receiving highly emetogenic chemotherapy, with pronounced control of delayed-phase nausea. Low dose provides predictable efficacy, manageable sedation, oral convenience, and cost-effectiveness. Clinically, olanzapine should be included in first-line antiemetic protocols to improve patient comfort, reduce the need for rescue medication, and support adherence to treatment.
- Citation: Mv A, Anand N, Babu B, Awosika A, Udayan U, Mendoza J, Lopez A, Rangachari B, Maity S, Gadad BS. Olanzapine-based vs neurokinin-1 receptor antagonist antiemetic regimens in highly emetogenic chemotherapy: Systematic review and meta-analysis of efficacy and safety. World J Clin Cases 2026; 14(12): 119112
- URL: https://www.wjgnet.com/2307-8960/full/v14/i12/119112.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i12.119112
Chemotherapy-induced nausea and vomiting (CINV) commonly occur in patients receiving highly emetogenic che
New therapies, including antibody-drug conjugates and novel chemotherapeutic agents, increase the complexity of CINV management and require adaptable prophylactic strategies[22,25-28]. Risk-based approaches using patient factors, emetic experience, and pharmacogenomic profiles have been suggested, but direct comparisons between olanzapine-based and NK1 receptor antagonist-based regimens in HEC are limited and variable[29-31]. Existing systematic reviews and network meta-analyses include heterogeneous populations, variable HEC definitions, and differing outcome measures, making direct comparisons challenging[7,13,14,32]. No recent systematic review has synthesized evidence from the growing number of phase III trials published since 2022 that directly compare these strategies in uniform HEC settings[9,11,15,18]. This meta-analysis was conducted to address this gap by evaluating the efficacy and safety of olanzapine-based vs NK1 receptor antagonist-based antiemetic regimens in adult patients receiving HEC. The primary focus is on complete response rates for nausea and vomiting across acute, delayed, and overall phases, alongside ass
The systematic review and meta-analysis were designed to answer the research question: “In adult patients receiving HEC (population), does an olanzapine-based antiemetic regimen (intervention) compared with an NK1 receptor an
Literature search was performed following Preferred Reporting Items for Systematic Reviews and Meta-analyses 2020 guidelines, covering PubMed, EMBASE, Scopus, and Web of Science from 2020 to 2025; with International Prospective Register of Systematic Reviews (No. CRD42025640877). Specific terms including “olanzapine”, “NK1 receptor antagonist”, “aprepitant”, “fosaprepitant”, “netupitant”, “palonosetron”, “dexamethasone”, and chemotherapy-related terms were combined using Boolean operators (‘AND’, ‘OR’, ‘NOT’) to maximize search sensitivity and specificity.
Studies were included if they examined adult patients receiving HEC and compared olanzapine-based antiemetic regimens with NK1 receptor antagonist-based regimens. Study designs like RCTs, cohort studies, which reported efficacy or safety outcomes, including complete response rates for nausea and vomiting were included. Exclusion criteria comprised studies with pediatric populations, moderate or low emetogenic chemotherapy, case reports, editorials, conference abstracts without full-text data, and studies without any relevant outcomes.
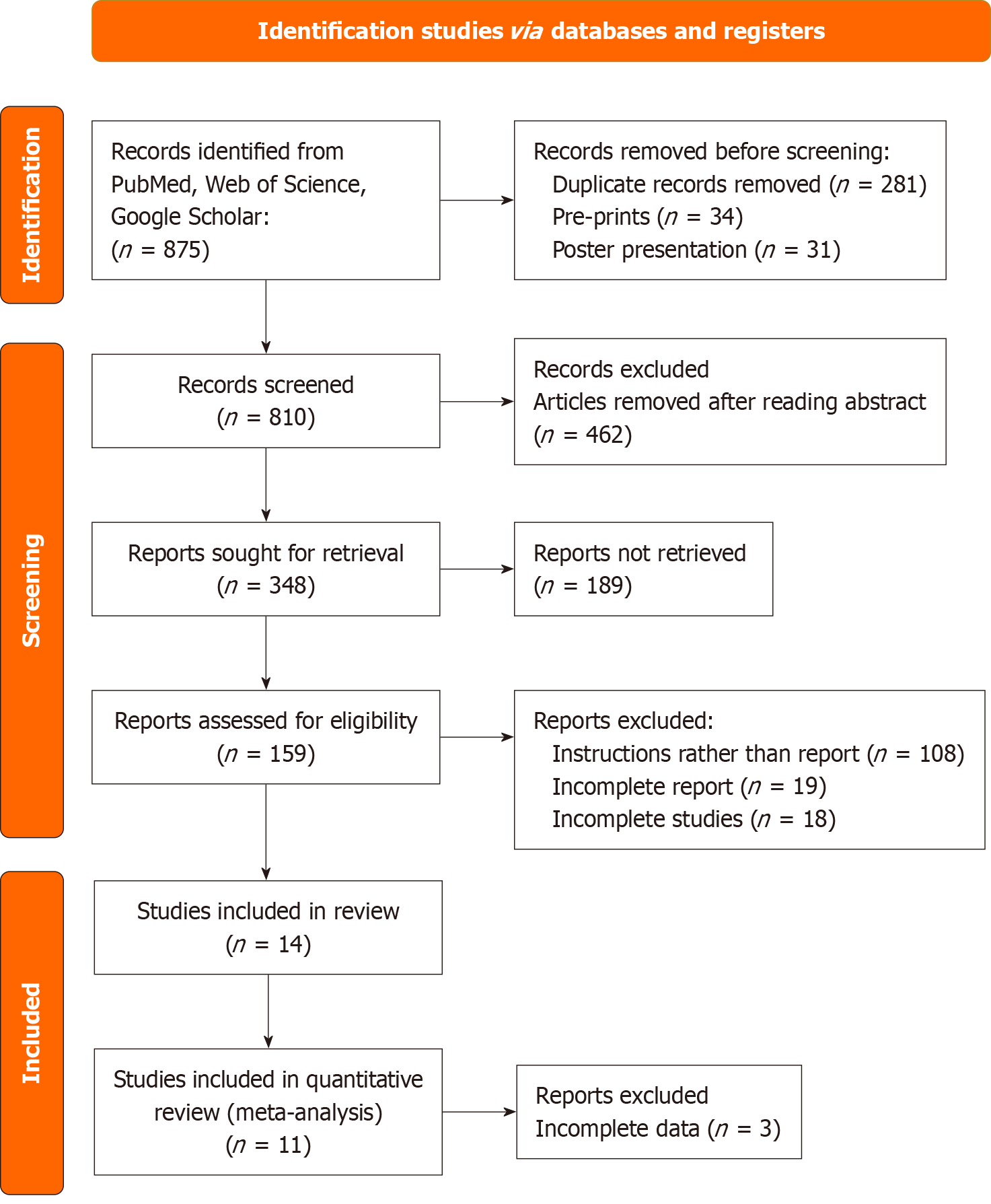
A total of 875 articles were searched through the databases and after reviewing and screening, 14 were included in the systematic review and only 11 met the criteria for meta-analysis (Figure 1). Data extraction was independently performed by three reviewers (Mv A, Babu B, Awosika A). Discrepancies were resolved through discussion among the reviewers. Data were synthesized quantitatively using meta-analytic techniques where feasible, and narrative synthesis was applied for heterogeneous data.
The methodological quality and risk of bias of included RCTs were assessed using the Cochrane risk of bias 2.0 tool[33], and cohort studies risk of bias in nonrandomized studies of interventions version I[34] were evaluated using the Newcastle-Ottawa Scale[35]. Domains assessed included randomization, allocation concealment, blinding, incomplete outcome data, selective reporting, and other biases. Each study was independently assessed by the reviewers with disagreements resolved by consensus.
A total of 14 studies were included in the systematic review because they adhered to the predefined eligibility criteria for population, intervention, comparator, and outcome reporting thus allowing for comprehensive qualitative synthesis of efficacy and safety evidence prior to quantitative pooling.
Table 1 includes 14 studies with 3421 patients from Iran, India, China, Japan, Hong Kong, and the United States. Patients had solid tumors, mainly breast, lung, gastrointestinal, and head-neck cancers, and received mostly HEC, including cisplatin- or doxorubicin-based regimens[20,36,37]. Olanzapine was administered at 2.5-10 mg for 3-5 days, usually combined with 5-HT3 receptor antagonists, dexamethasone, and occasionally NK1 antagonists[1,16,38]. Table 2 shows efficacy outcomes: Acute complete response ranged 66%-100%, delayed complete response 55%-94%, and overall complete response 63%-91%. Olanzapine regimens provided equal or better control of nausea and vomiting compared with aprepitant or netupitant-palonosetron-based therapies, with improvements in quality of life and reduced rescue medication reported[1,20,21,36,37]. Sedation or somnolence occurred in 10%-53% of patients, generally mild to moderate, and lower at 5 mg doses[11,36]. Table 3 reports Newcastle-Ottawa Scale scores ranging from 6 to 9, with a maximum possible score of 9. A total of 11 of 14 studies scored ≥ 8, indicating strong methodological rigor. Five studies achieved a score of 9, demonstrating complete fulfilment of selection criteria, appropriate control for confounding, and complete exposure or outcome assessment[11,18,21,38,39]. Four studies scored 8, losing 1 point because of limited adjustment for comparability or incomplete follow-up reporting[1,9,15,29]. Three studies scored 6-7. Wu et al[36] scored 6 due to incomplete selection documentation and exposure assessment. Sakai et al[17] and Radhakrishnan et al[38] scored 7 because of open-label or single-arm designs with partial reporting. No study scored below 6, supporting reliability of pooled analyses.
| Ref. | Country | Design | n | Cancer type | Chemo (HEC/MEC) | Intervention (olanzapine regimen) | Comparator | Olanzapine dose and duration | Follow-up | Key findings |
| Maleki et al[1], 2020 | The Islamic Republic of Iran | Phase III, RCT, DB | 60 | Early breast (I-III) | AC (doxorubicin ≥ 50 + cyclophosphamide ≥ 500 mg/m2) | OLA 10 mg + aprepitant + dex + granisetron | Mirtazapine 15 mg + aprepitant + dex + granisetron | 10 mg D1-4 (5 mg ≥ 60 years) | 0-120 hours × 2 cycles | CR similar (acute 766% vs 83.3%); ↑somnolence/fatigue with OLA; better QoL (cycle 2) with mirtazapine |
| Radhakrishnan et al[38], 2020 | India | Phase III, open-label RCT | 80 | Pediatric solid tumors | Mostly HEC (cisplatin/ifosfamide/cyclophosphamide/doxorubicin) | OLA (2.5-5 mg daily) | Metoclopramide (no NK1) | 2.5-5 mg ≥ 72 hours | 72 hours + AE 7 days | CR vomiting 72% vs 39%, nausea 59% vs 34%; sedation 25% vs 0%; OLA superior for breakthrough CINV |
| Wu et al[36], 2020 | China | Retrospective cohort | 93 | GI (CRC 78.5%, gastric 21.5%) | MEC (mFOLFOX6/XELOX/FOLFIRI) | OLA 5 mg + tropisetron + dex | Tropisetron + dex | 5 mg D1-3 | 0-120 hours | Overall CR 70% vs 47%; delayed CR 75% vs 55%; ↑QoL (75% vs 49% no impact); somnolence 47% vs 15% |
| Sakai et al[17], 2021 | Japan | Phase II, single-arm | 50 | Thoracic (NSCLC 66%, SCLC 22%) | Carboplatin ± other drugs (HEC) | OLA 5 mg + granisetron + dex | None | 5 mg D1-4 pm | 0-120 hours | CR 100% acute, 94% delayed; no ≥ G3 AEs; nausea peaked D3-4 |
| Gao et al[10], 2022 | China | RCT | 120 | Lung (52%) + others | Cisplatin × 3 days (HEC) | Quadruple: OLA 5 mg + aprepitant + tropisetron + dex | Triplet: OLA 5 mg + tropisetron + dex | 5 mg D1-3 | 0-120 hours (5 days) | Acute CR↑ (100% vs 93%, P = 0.045); overall CR NS; longer TTE with aprepitant; well-tolerated |
| Liu et al[39], 2022 | China | RCT | 210 | Mixed solid tumors | Cisplatin × 3 days | OLA 5 mg + tropisetron + dex 10 mg | Aprepitant + tropisetron + dex 5 mg | 5 mg D1-4 | 0-120 hours | Similar efficacy (all endpoints); somnolence (OLA) vs constipation (aprepitant) |
| Yip et al[18], 2023 | Mainland | Post hoc (2 prospective) | 120 | Early breast | AC (HEC) | OLA 10 mg + aprepitant + ondansetron + dex | NEPA + dex | 10 mg D1-5 | 0-120 hours × 4 cycles | C1: ↑“No rescue” and “no nausea”; CR similar (approximately 65%-70%); later cycles favored NEPA; QoL NS |
| Navari et al[9], 2023 | United States | Phase III, RCT, DB, PC | 690 | Breast 77% + others | HEC (AC 77%, cisplatin 23%) | OLA + 5-HT3 RA + dex + placebo | Same + NK1 (fosaprepitant/aprepitant) | 10 mg D1-4 | 0-120 hours × 4 cycles | Overall CR 47% vs 55%; NK1 arm better nausea control; OLA alone non-inferior not met; ↑sedation day 1 |
| Zhao et al[29], 2023 | China | Phase III, RCT, DB | 720 | Solid tumors | Cisplatin ≥ 50 mg/m2 (HEC) | Fosaprepitant vs aprepitant (+ palonosetron + dex) | 0-120 hours | Fosaprepitant non-inferior for overall CR (78% vs 78%); similar nausea QoL; AEs mild | ||
| Inui et al[11], 2024 | Japan | Phase III, RCT, DB, PC | 355 | Thoracic | Carboplatin ± ICI (HEC) | OLA 5 mg + aprepitant + 5-HT3 RA + dex | Placebo + same | 5 mg D1-4 pm | 0-120 hours | Overall CR 87% vs 81% (NS); no nausea↑ (89% vs 76% P < 0.001); somnolence 24%; no serious AEs |
| Ostwal et al[37], 2024 | India | Phase III, open-label | 544 | Solid tumors (GI > lung) | MEC (oxaliplatin 61%, carbo 29%) | OLA 10 mg + aprepitant + palonosetron + dex | Aprepitant + palonosetron + dex | 10 mg D1-3 hs | 0-120 hours | Overall CR 91% vs 82% (P = 0.005); nausea 96% vs 87%; better QoL; somnolence 10% (G1) |
| Shen et al[20], 2024 | China | RCT, open-label | 102 | GI (rectal 55%, gastric 28%) | MEC (XELOX/SOX/FOLFOX) | OLA 5 mg + palonosetron + dex | Palonosetron + dex | 5 mg D1-5 hs | 0-120 hours | TP 66.7% vs 37% (P = 0.003); QoL↑ (84% vs 59%); ↑drowsiness (53% vs 31%); ↑appetite |
| Bhargave et al[15], 2025 | India | Phase III, DB, DD, PC | 195 | Head-neck 38%, Gyn 29% + others | Carboplatin AUC ≥ 4 | OOD: OLA 5 mg + ondansetron + dex + placebo | Fosaprepitant + ondansetron + dex + placebo | 5 mg D1-4 | 0-120 hours | Overall CR 66% vs 67% (NS); no nausea 44% vs 34%; sedation 49% vs 29%; cost-effective alternative |
| Attili et al[21], 2025 | India | Phase III, RCT, DB, PC | 82 | Mixed carcinomas | Cisplatin-based HEC | OLA 10 mg + granisetron + dex | Placebo + granisetron + dex | 10 mg D1 to D3 | 0-120 hours | CR↑ (acute 87% vs 55%, delayed 72% vs 47%, overall, 67% vs 34%); mild sedation 31% |
| Ref. | Comparator regimen | Primary outcome (CR) definition | Acute CR (%) | Delayed CR (%) | Overall CR (%) | Nausea control (%) | Vomiting control (%) | Sedation/somnolence (%) | Other AEs (≥ 10%) | Statistical significance (P value/ 95%CI) | Relative risk/OR (95%CI) |
| Maleki et al[1], 2020 | Mirtazapine + aprepitant + dexamethasone + granisetron (both arms NK1) | No vomiting + no rescue (0-120 hours) | 76.6 | 86.6 | 63.3 | 70.0 | 86.6 | 53.3 (G1-3) | Fatigue 667; dry mouth 33.3; constipation 26.7; appetite loss 43.3 | CR: P = 0.51-0.78; Somnolence P = 0.04; fatigue P = 0.02; FLIE P = 0.044 | |
| Radhakrishnan et al[38], 2020 | Metoclopramide (no NK1) | No vomiting or nausea within 72 hours post rescue | 70 | 100 | 72 | 59 | 72 | 25 (G1-3) | Anorexia 30 vs 27; abdominal pain 30 vs 24; headache 17 vs 10 | Vomiting CR P = 0.003; nausea CR P = 0.026; sedation P = 0.0004 | OR vomiting 4.0 (1.6-10.0); OR nausea 27 (1.1-6.6) |
| Wu et al[36], 2020 | Tropisetron + dexamethasone (dual; no NK1) | No vomiting + no rescue (0-120 hours) | 85.0 | 75.0 | 70.0 | No nausea < 5 mm: 55 | 47.5 | Constipation 62.5; anorexia 575; insomnia 15; dizziness 30 | Delayed CR P = 0.044; overall CR P = 0.028; somnolence P = 0.001 | OR overall CR ≈ 2.6; OR delayed ≈ 2.5 | |
| Sakai et al[17], 2021 | None (single arm) | No emesis + no rescue (0-120 hours) | 100 | 94 | 94 | Total control 86 | 94 | 76 (mostly G1) | Constipation 72; dry mouth 64; insomnia 56; hiccups 50 | P < 0.0001 vs historical 65% CR | |
| Gao et al[10], 2022 | Olanzapine + tropisetron + dexamethasone | No vomiting + no rescue (0-120 hours) | 100 | 76 | 76 | 28.8 | 93.2 | 57.6 | Fatigue 542; constipation 22 | Acute CR P = 0.045; no vomiting P = 0.038 | RR ≈ 1.13 (ns) |
| Liu et al[39], 2022 | Aprepitant + tropisetron + dexamethasone | No vomiting + no rescue; TP primary | 96.15 vs 97.17 | 75 vs 79.25 | 75 vs 79.25 | TP 54.8 vs 54.7; TC 31.7 vs 27.3 | 88.46 vs 50.8 | Constipation 53 vs 62 | CR P = 0.46; somnolence P = 0.00; constipation P = 0.02 | ||
| Yip et al[18], 2023 | NEPA + dexamethasone | No vomiting + no rescue (0-120 hours) | 70.0 | 92.9 | 65.0 | 91.7 | 68.3 | Neutropenia ≥ G2↑ in olanzapine arm | Overall CR P = 0.5716; no nausea P = 0.0408 | ||
| Navari et al[9], 2023 | Olanzapine + 5-HT3 RA + dex + NK1 vs placebo | No emesis + no rescue (0-120 hours) | 55 vs 47 | 55 vs 47 | 38 vs 30 | Higher in NK1 arm (P = 0.017) | Appetite↑ (no difference) | Overall CR P = 0.0497; no nausea P = 0.2443 | Risk difference +8% (both CR and nausea) | ||
| Zhao et al[29], 2023 | Aprepitant + palonosetron + dexamethasone | No emesis + no rescue (0-120 hours) | 88.9 vs 88.4 | 80.1 vs 79.9 | 78.1 vs 77.7 | No nausea < 5 mm: 52 vs 50.6 | None ≥ 10% | CR P = 0.765; 95%CI: -5.7 to 6.6 | Risk difference +0.4% | ||
| Inui et al[11], 2024 | Placebo + aprepitant + 5-5-HT3 RA + dex | No vomiting + no rescue | 98.9 vs 97.8 | 87.4 vs 80.6 | 86.9 vs 80.6 | No nausea 886 | 24.6 vs 22.9 | Constipation 44; hepatotoxicity 55; cytopenias > 60 | CR P = 0.116; nausea P < 0.001 | Risk difference CR +6.3%; nausea +13.6% | |
| Ostwal et al[37], 2024 | Aprepitant + palonosetron + dex (NK1 triplet) | No vomiting + no nausea < 5 mm + no rescue (0-120 hours) | 96 | 92 | 91 | 96 | 95 | 10 (G1) | None ≥ 10% | Overall CR P = 0.005; somnolence P < 0.001 | OR CR 2.04 (1.24-3.36); OR nausea 3.57 (1.94-6.57) |
| Shen et al[20], 2024 | Palonosetron + dexamethasone (dual; no NK1) | CR: No vomiting + no rescue; TP ≤ 25 mm; TC ≤ 5 mm | 84.3 | 80.3 | 76.5 | TP 66.7; TC 31.4 | 52.9 | Appetite loss 56.9; weakness 78.4; dizziness 43.1 | TP P = 0.003; drowsiness P = 0.027; FLIE P = 0.003 | OR overall TP ≈ 3.3 | |
| Bhargave et al[15], 2025 | Fosaprepitant + ondansetron + dex | CR: No vomiting + no rescue; primary: No nausea | 87.3 vs 92.5 | 67.7 | 65.7 vs 66.7 | No nausea 441 | 48.5 vs 29.0 | Fatigue 28; muscle pain 19; constipation 15 | No nausea P = 0.19; CR P > 0.99 | OR no nausea 151 (0.84-2.69); CR 0.96 (0.53-1.73) | |
| Attili et al[21], 2025 | Placebo + granisetron + dex (no NK1) | No vomiting + no rescue | 87.1 | 71.8 | 66.6 | No nausea: 76.9 (acute), 46.1 (overall) | 30.7 (mild) | Appetite↑ 10.2 | Acute CR P = 0.001; overall CR P = 0.004 | RR CR ≈ 1.95; RR Nausea ≈ 2.51 |
| No. | Ref. | S1 | S2 | S3 | S4 | Comparability | E1/O1 | E2/O2 | E3/O3 | Total |
| 1 | Maleki et al[1], 2020 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 8 |
| 2 | Radhakrishnan et al[38], 2020 | 1 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 7 |
| 3 | Wu et al[36], 2020 | 1 | 1 | 0 | 0 | 1 | 1 | 1 | 1 | 6 |
| 4 | Sakai et al[17], 2021 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 7 |
| 5 | Gao et al[10], 2022 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 7 |
| 6 | Liu et al[39], 2022 | 1 | 1 | 1 | 1 | 2 | 1 | 1 | 1 | 9 |
| 7 | Yip et al[18], 2023 | 1 | 1 | 1 | 1 | 2 | 1 | 1 | 1 | 9 |
| 8 | Navari et al[9], 2023 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 8 |
| 9 | Zhao et al[29], 2023 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 8 |
| 10 | Inui et al[11], 2024 | 1 | 1 | 1 | 1 | 2 | 1 | 1 | 1 | 9 |
| 11 | Ostwal et al[37], 2024 | 1 | 1 | 1 | 1 | 2 | 1 | 1 | 1 | 9 |
| 12 | Shen et al[20], 2024 | 1 | 1 | 1 | 1 | 2 | 1 | 1 | 1 | 9 |
| 13 | Bhargave et al[15], 2025 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 8 |
| 14 | Attili et al[21], 2025 | 1 | 1 | 1 | 1 | 2 | 1 | 1 | 1 | 9 |
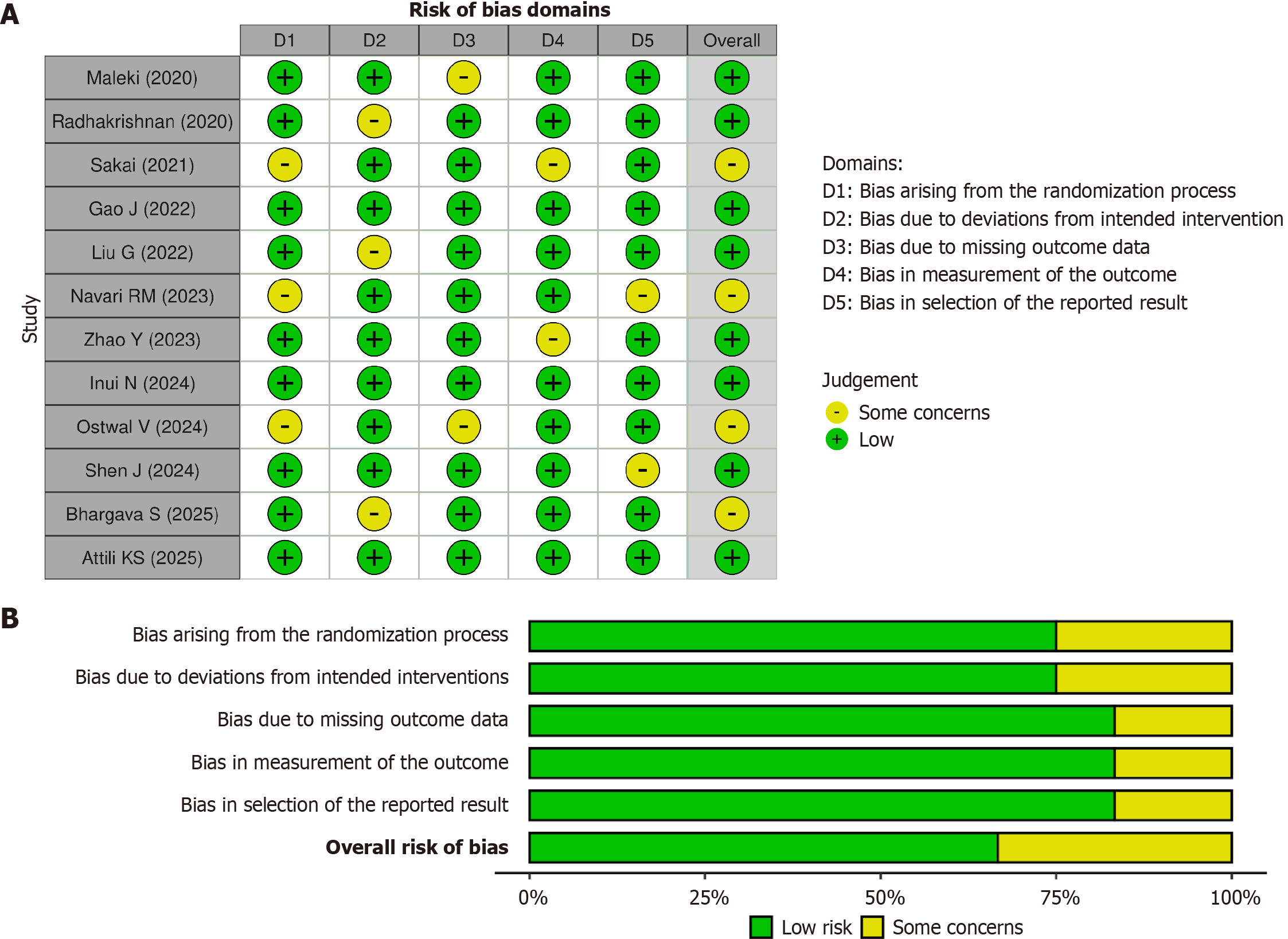
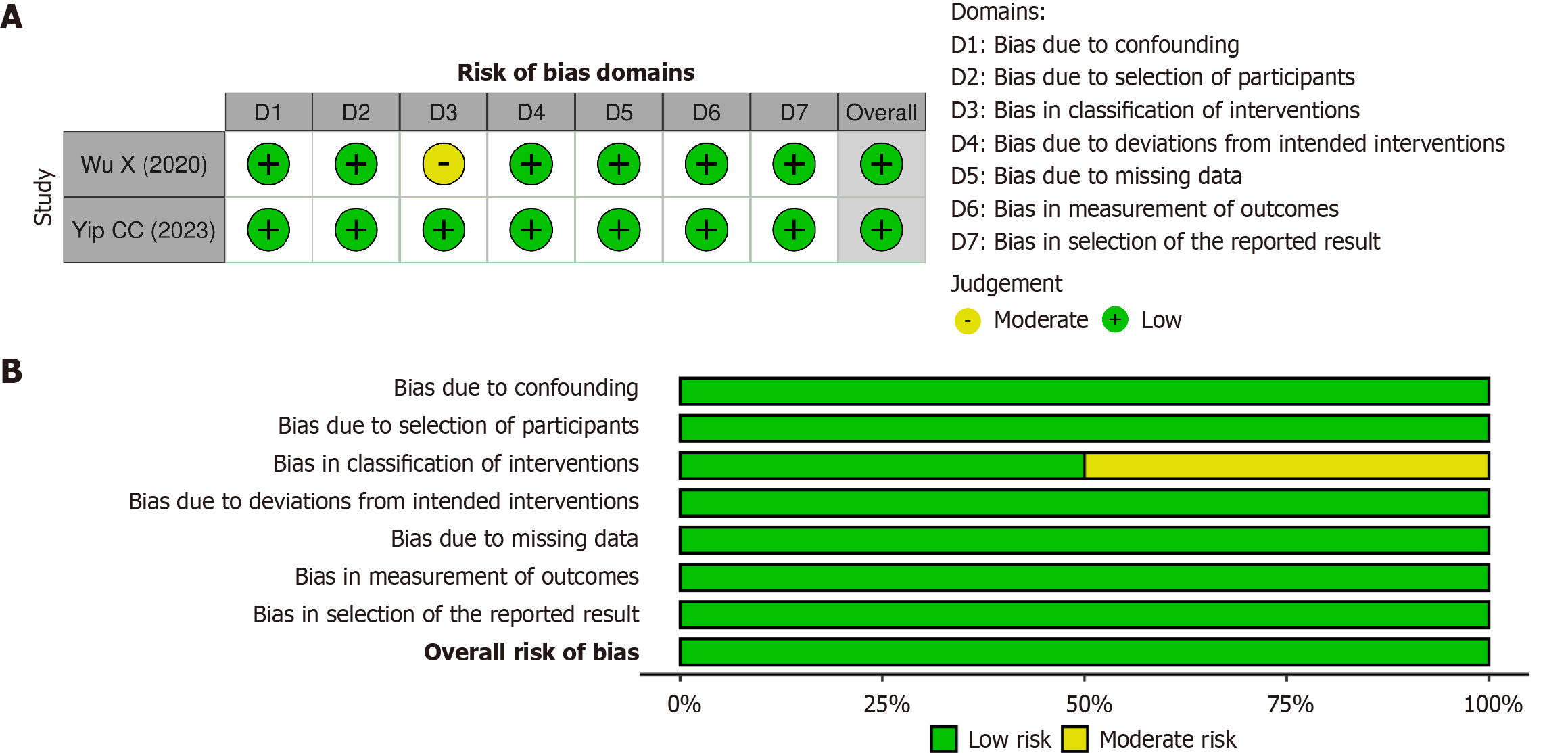
Figure 2 present risks of bias 2.0 assessments for 12 RCTs. Eight of 12 trials showed low risk of bias, while 4 of 12 showed some concerns; no trial showed high risk in any domain. Issues related to the randomization process (D1) were identified in Navari et al[9], Sakai et al[17], and Ostwal et al[37], linked to incomplete reporting of allocation concealment. Deviations from intended interventions (D2) were identified in Bhargave et al[15], Radhakrishnan et al[38], and Liu et al[39], associated with open-label study designs. Missing outcome data (D3) were noted in Sakai et al[17] and Ostwal et al[37]. Issues related to outcome measurement (D4) and selective reporting (D5) were identified in Shen et al[20] and Zhao et al[29]. In each risk of bias 2.0 domain, more than 70% of trials showed low risk, supporting use of randomized trial data for quantitative synthesis. Figure 3 present risk of bias in nonrandomized studies of interventions version I assessments for the 2 cohort studies included in the review[18,36]. Low risk of bias was identified in 13 of 14 assessed domains (92.9%). A single domain showed moderate risk, limited to classification of interventions (D3), related to retrospective exposure assessment in Wu et al[36]. All remaining domains, including confounding (D1), selection of participants (D2), deviations from intended interventions (D4), missing data (D5), outcome measurement (D6), and selection of reported results (D7), showed low risk in both studies[18,36]. The overall risk of bias was low in 1 study and moderate in 1 study, with no domain showing high risk.
Of the 14 studies included in the systematic review, 11 were eligible for forest plot analysis. Wu et al[36] was excluded because of its retrospective cohort design and inclusion of moderately emetogenic chemotherapy with a dual antiemetic comparator, which differed from the randomized comparison framework used in pooled analyses. Shen et al[20] was excluded because primary outcomes were reported as total control and visual analogue scale-based nausea thresholds rather than dichotomous complete response, preventing extraction of compatible odds ratios (ORs). Attili et al[21] was excluded because the comparator arm consisted of placebo plus granisetron and dexamethasone without an NK1-based or active standard antiemetic comparator, limiting compatibility with pooled control groups used in the forest plots.
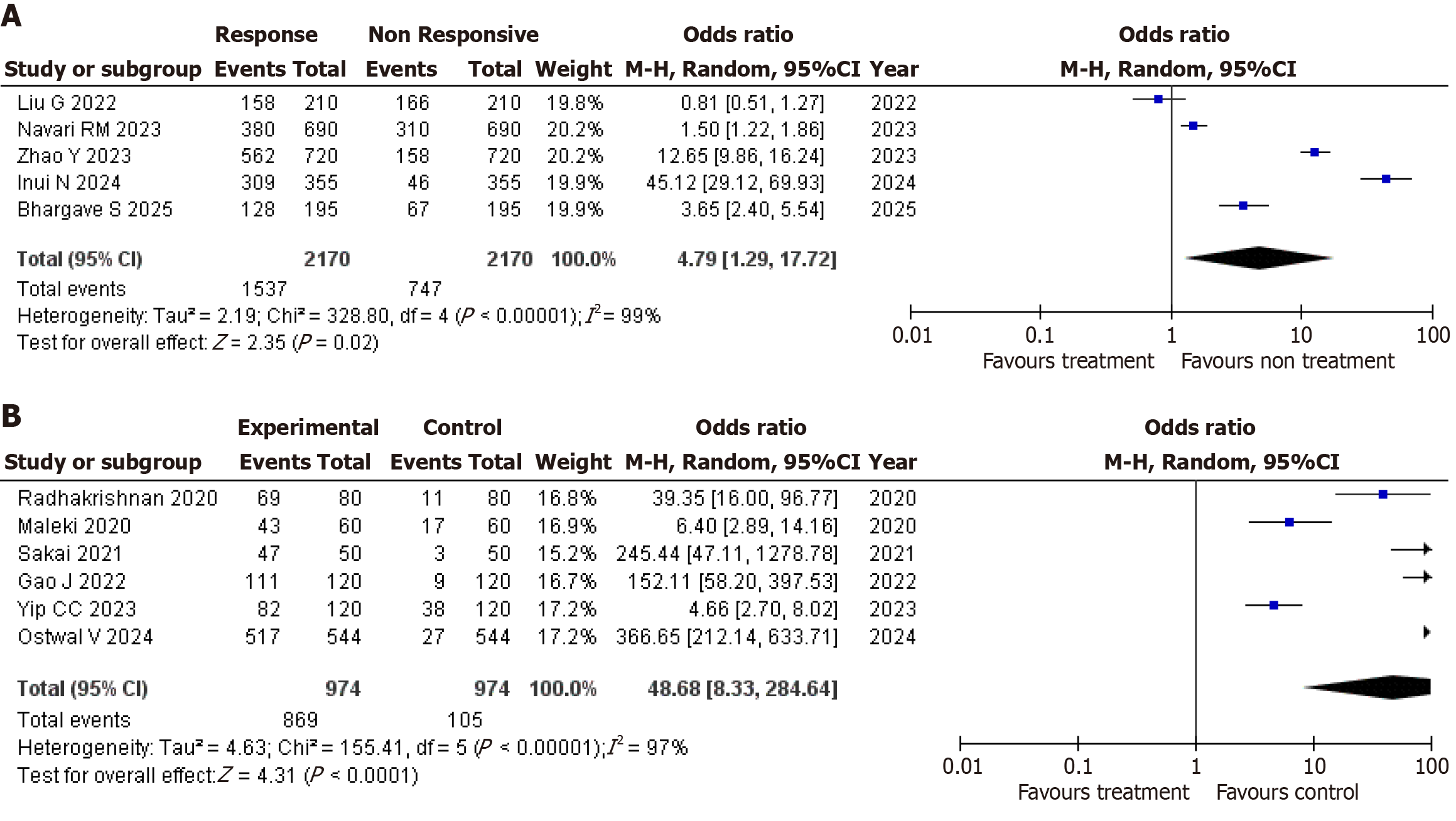
Different numbers of studies appear in Figure 4 because each forest plot addresses a different analytical objective. Figure 4A evaluates overall antiemetic effectiveness using randomized studies with heterogeneous comparator regimens and mixed effect directions. Figure 4B evaluates complete response outcomes in randomized studies with comparable control arms and consistent direction of effect in favor of olanzapine, allowing focused estimation of effect magnitude.
Figure 4A presents pooled analysis from 5 randomized studies comparing olanzapine-based regimens with comparator antiemetic regimens in 4340 participants[9,11,15,29,39]. The combined OR was 4.79 [95%confidence interval (CI): 1.29-17.72], with a statistically significant test for effect (Z = 2.35, P = 0.02). Between-study variability was high (τ2 = 2.19; χ2 = 328.80, degree of freedom = 4; P < 0.00001; I2 = 99%). Individual study estimates varied widely, ranging from an OR of 0.81 (95%CI: 0.51-1.27) in Liu et al[39] to 45.12 (95%CI: 29.12-69.93) in Inui et al[11]. Intermediate effects were observed in Navari et al[9], Bhargave et al[15], and Zhao et al[29], reflecting differences in chemotherapy regimens, comparator arms, and olanzapine dosing.
Figure 4B summarizes pooled results from 6 studies with 1948 participants that directly compared olanzapine-based regimens with control regimens for complete response[16,18,21,36-38]. The pooled OR was 48.68 (95%CI: 8.33-284.64), with a statistically significant test for effect (Z = 4.31, P < 0.0001). Heterogeneity remained substantial (τ2 = 4.63; χ2 = 155.41, degree of freedom = 5; P < 0.00001; I2 = 97%). Individual effect sizes ranged from 3.65 (95%CI: 2.40-5.54) in Bhargave et al[15] to 366.65 (95%CI: 212.14-633.71) in Ostwal et al[37]. All included studies showed ORs greater than 1, indicating higher complete response rates with olanzapine-containing regimens despite marked inter-study variability.
This systematic review and meta-analysis included 14 studies with 3421 patients and shows that olanzapine-based regimens prevent CINV in HEC more effectively, particularly delayed nausea. Recent phase III trials have reported higher rates of patients with no nausea using olanzapine quadruple therapy (89.6% vs 76.0%, P < 0.001)[11] and better complete nausea control (96% vs 87%, P < 0.001)[37]. Previous trials have also demonstrated similar advantages. A randomized trial in lung cancer patients receiving cisplatin showed improved rates of no nausea with an olanzapine-based triplet compared to standard NK1-based therapy (76.5% vs 56.4%, P = 0.036)[40]. These results confirm that olanzapine constantly reduces nausea after the first 24 hours in different patient groups and treatment modules. Olanzapine provides effective control of CINV through broad receptor antagonism, targeting dopamine D2, serotonin 5-HT2A/3, histamine H1, and muscarinic receptors involved in CINV[1,6,8]. Olanzapine’s multi-receptor activity effectively controls delayed-phase nausea, where several mediators drive emesis. Forest plot analyses showed high heterogeneity (I2 > 95%) due to chemotherapy type, comparator regimens, and olanzapine dose[11,15,20], but outcomes favored olan
Olanzapine targets dopamine, serotonin, histamine, and muscarinic receptors, providing effective control of prolonged and delayed nausea caused by treatments like antibody-drug conjugates[26,28,54]. Olanzapine is available orally and has a low cost, making it practical for clinical use[49]. In moderate-emetogenic chemotherapy, it reduces nausea where NK1 antagonists have variable effects[50,55,56]. It controls nausea better than aprepitant while maintaining a manageable safety profile[57] and remains necessary even when non-pharmacologic interventions like acupoint stimulation are used[58]. Olanzapine prevents both acute and delayed nausea, providing reliable efficacy for patients receiving high-emetogenic or moderate-emetogenic chemotherapy. These features support its inclusion in treatment protocols to reduce CINV and improve patient supportive care. This meta-analysis has some limitations like clinical heterogeneity from the inclusion of moderately emetogenic chemotherapy studies and different comparator regimens, follow-up limited to ≤ 120 hours that may miss very delayed symptoms from newer chemotherapies, and dependence on the quality of original studies for pooled outcomes. Most included studies were high-quality RCTs with low risk of bias, as confirmed by risks of bias 2.0 assessment, and constantly favored olanzapine. Olanzapine-based regimens are non-inferior and often superior to NK1 antagonist-based regimens for HEC, particularly for delayed-phase nausea.
This review and meta-analysis demonstrated that olanzapine-based regimens effectively prevent CINV in patients receiving HEC, with pronounced control of delayed-phase nausea. Low dose provides predictable efficacy, manageable sedation, oral convenience, and cost-effectiveness. Clinically, olanzapine should be included in first-line antiemetic protocols to improve patient comfort, reduce the need for rescue medication, and support adherence to treatment. Future studies should evaluate its effectiveness with newer therapies, assess long-term safety in diverse populations, and explore optimized dosing schedules for specific patient subgroups. These findings support broader implementation of olanzapine in routine clinical practice for high-risk chemotherapy.
| 1. | Maleki A, Ghadiyani M, Salamzadeh J, Salari S, Banihashem S, Tavakoli-Ardakani M. Comparison of Mirtazapine and Olanzapine on Nausea and Vomiting following Anthracycline-cyclophosphamide Chemotherapy Regimen in Patients with Breast Cancer. Iran J Pharm Res. 2020;19:451-464. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 2. | Zhang Z, Zhang Y, Chen G, Hong S, Yang Y, Fang W, Luo F, Chen X, Ma Y, Zhao Y, Zhan J, Xue C, Hou X, Zhou T, Ma S, Gao F, Huang Y, Chen L, Zhou N, Zhao H, Zhang L. Olanzapine-Based Triple Regimens Versus Neurokinin-1 Receptor Antagonist-Based Triple Regimens in Preventing Chemotherapy-Induced Nausea and Vomiting Associated with Highly Emetogenic Chemotherapy: A Network Meta-Analysis. Oncologist. 2018;23:603-616. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 3. | Koth SM, Kolesar J. New options and controversies in the management of chemotherapy-induced nausea and vomiting. Am J Health Syst Pharm. 2017;74:812-819. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 8] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 4. | Jordan K, Jahn F, Aapro M. Recent developments in the prevention of chemotherapy-induced nausea and vomiting (CINV): a comprehensive review. Ann Oncol. 2015;26:1081-1090. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 140] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 5. | Alhifany AA, McBride A, Almutairi AR, Cheema E, Shahbar A, Alatawi Y, Alharbi AS, Babiker H, MacDonald K, Aapro M, Abraham I. Efficacy of olanzapine, neurokinin-1 receptor antagonists, and thalidomide in combination with palonosetron plus dexamethasone in preventing highly emetogenic chemotherapy-induced nausea and vomiting: a Bayesian network meta-analysis. Support Care Cancer. 2020;28:1031-1039. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 6. | Chow R, Navari RM, Terry B, DeAngelis C, Prsic EH. Olanzapine 5 mg vs 10 mg for the prophylaxis of chemotherapy-induced nausea and vomiting: a network meta-analysis. Support Care Cancer. 2022;30:1015-1018. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 7. | Chelkeba L, Gidey K, Mamo A, Yohannes B, Matso T, Melaku T. Olanzapine for chemotherapy-induced nausea and vomiting: systematic review and meta-analysis. Pharm Pract (Granada). 2017;15:877. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 16] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 8. | Chiu L, Chow R, Popovic M, Navari RM, Shumway NM, Chiu N, Lam H, Milakovic M, Pasetka M, Vuong S, Chow E, DeAngelis C. Efficacy of olanzapine for the prophylaxis and rescue of chemotherapy-induced nausea and vomiting (CINV): a systematic review and meta-analysis. Support Care Cancer. 2016;24:2381-2392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 60] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 9. | Navari RM, Le-Rademacher J, Smieliauskas F, Ruddy KJ, James Saphner T, Liu H, Harlos E, Onitilo AA, Giridhar K, Paul Singh P, Reddy PS, Chow S, Kruter F, Raptis G, Loprinzi CL. Olanzapine With or Without Fosaprepitant for Preventing Chemotherapy Induced Nausea and Vomiting in Patients Receiving Highly Emetogenic Chemotherapy: A Phase III Randomized, Double-Blind, Placebo-Controlled Trial (ALLIANCE A221602). Oncologist. 2023;28:722-729. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 10. | Gao J, Zhao J, Jiang C, Chen F, Zhao L, Jiang Y, Li H, Wang W, Wu Y, Jin Y, Da L, Liu G, Zhang Y, Li H, Zhang Z, Jin G, Li Q. Olanzapine (5 mg) plus standard triple antiemetic therapy for the prevention of multiple-day cisplatin hemotherapy-induced nausea and vomiting: a prospective randomized controlled study. Support Care Cancer. 2022;30:6225-6232. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (1)] |
| 11. | Inui N, Suzuki T, Tanaka K, Karayama M, Inoue Y, Mori K, Yasui H, Hozumi H, Suzuki Y, Furuhashi K, Fujisawa T, Matsuura S, Nishimoto K, Matsui T, Asada K, Hashimoto D, Fujii M, Niwa M, Uehara M, Matsuda H, Koda K, Ikeda M, Inami N, Tamiya Y, Kato M, Nakano H, Mino Y, Enomoto N, Suda T. Olanzapine Plus Triple Antiemetic Therapy for the Prevention of Carboplatin-Induced Nausea and Vomiting: A Randomized, Double-Blind, Placebo-Controlled Phase III Trial. J Clin Oncol. 2024;42:2780-2789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 12. | Abe M, Yamaguchi T, Fujita Y, Nishimura T, Kitagawa K, Inui N, Hirano K, Sakata Y, Iihara H, Shibuya Y, Suzuki K, Shibata K, Hori K, Daga H, Nakayama T, Sakata Y, Takahashi TY, Zenda S, Hashimoto H. Efficacy of Olanzapine in Addition to Standard Triplet Antiemetic Therapy for Cisplatin-Based Chemotherapy: A Secondary Analysis of the J-FORCE Randomized Clinical Trial. JAMA Netw Open. 2023;6:e2310894. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 13. | Filetti M, Lombardi P, Giusti R, Falcone R, Scotte F, Giannarelli D, Carcagnì A, Altamura V, Scambia G, Daniele G. Efficacy and safety of antiemetic regimens for highly emetogenic chemotherapy-induced nausea and vomiting: A systematic review and network meta-analysis. Cancer Treat Rev. 2023;115:102512. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 22] [Reference Citation Analysis (1)] |
| 14. | Piechotta V, Adams A, Haque M, Scheckel B, Kreuzberger N, Monsef I, Jordan K, Kuhr K, Skoetz N. Antiemetics for adults for prevention of nausea and vomiting caused by moderately or highly emetogenic chemotherapy: a network meta-analysis. Cochrane Database Syst Rev. 2021;11:CD012775. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 37] [Article Influence: 7.4] [Reference Citation Analysis (1)] |
| 15. | Bhargave S, Sharma V, Kataria B, Batra A, Pushpam D, Sharma A, Pramanik R, Malik PS, Sahoo RK, Khurana S, Singh V, Bakhshi S, Sharma A, Kumar L, Kumar A. Olanzapine Versus NK1 Receptor Antagonist for Prevention of Carboplatin-Induced (AUC ≥4) Emesis: A Phase III, Double-Blind, Placebo-Controlled Randomized Trial From India. JCO Glob Oncol. 2025;11:e2400166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 16. | An Y, Zhang Z, Gu M, Zhao J, Jiang C, Zhao L, Jiang Y, Li H, Liu G, Jin G, Li Q. Low Dose Olanzapine in the Prevention and Treatment of Carboplatin Induced Nausea and Vomiting: A Prospective Clinical Randomized Controlled Trial. Curr Pharm Des. 2023;29:1867-1874. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 17. | Sakai C, Shimokawa M, Iihara H, Fujita Y, Ikemura S, Hirose C, Kotake M, Funaguchi N, Gomyo T, Imai H, Hakamata J, Kaito D, Minato K, Arai T, Kawazoe H, Suzuki A, Ohno Y, Okura H. Low-Dose Olanzapine Plus Granisetron and Dexamethasone for Carboplatin-Induced Nausea and Vomiting in Patients with Thoracic Malignancies: A Prospective Multicenter Phase II Trial. Oncologist. 2021;26:e1066-e1072. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 18. | Yip CC, Li L, Lau TKH, Chan VTC, Kwok CCH, Suen JJS, Mo FKF, Yeo W. Evaluation of contemporary olanzapine- and netupitant/palonosetron-containing antiemetic regimens for chemotherapy-induced nausea and vomiting. Hong Kong Med J. 2023;29:49-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 19. | Navari RM, Tyler T, Inui N, Iihara H, Bonizzoni E, Park YH, Rugo HS, Roeland EJ. Individual patient data meta-analysis of NEPA versus aprepitant-based antiemetic regimens for preventing chemotherapy-induced nausea and vomiting. Future Oncol. 2025;21:2823-2833. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 20. | Shen J, Zhao J, Jin G, Li H, Jiang Y, Wu Y, Gao J, Chen F, Li J, Wang W, Li Q. A prospective randomized controlled clinical trial investigating the efficacy of low-dose olanzapine in preventing nausea and vomiting associated with oxaliplatin-based and irinotecan-based chemotherapy. J Cancer Res Clin Oncol. 2024;150:283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 21. | Attili KS, Bura HK, Ganta V, Mendu SB, Prayaga UK. Comparative Study of Efficacy and Safety of Olanzapine as Add-on Therapy to Conventional Regimen in Preventing Chemotherapy-induced Nausea and Vomiting. SSR Inst Int J Life Sci. 2025;11:6815-6821. [DOI] [Full Text] |
| 22. | Hu X, Curigliano G, Yonemori K, Bardia A, Barrios CH, Sohn J, Lévy C, Jacot W, Tsurutani J, Roborel de Climens A, Wu X, Andrzejuk-Ćwik A, Mbanya Z, Dent R. Patient-reported outcomes with trastuzumab deruxtecan in hormone receptor-positive, HER2-low or HER2-ultralow metastatic breast cancer: results from the randomized DESTINY-Breast06 trial. ESMO Open. 2025;10:105082. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 13] [Reference Citation Analysis (1)] |
| 23. | Sun Y, Wang Y, Chen G, Zhang Y, Zhang L, Chen X. The evolving landscape of antiemetic prophylaxis for chemotherapy-induced nausea and vomiting: inspiration from cisplatin-based antiemetic and non-antiemetic trials. Support Care Cancer. 2024;32:822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 24. | Aapro M, Jordan K, Scotté F, Celio L, Karthaus M, Roeland E. Netupitant-palonosetron (NEPA) for Preventing Chemotherapy-induced Nausea and Vomiting: From Clinical Trials to Daily Practice. Curr Cancer Drug Targets. 2022;22:806-824. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 25. | Notini G, Naldini MM, Sica L, Viale G, Rognone A, Zambelli S, Zucchinelli P, Piras M, Bosi C, Mariani M, Aldrighetti D, Bianchini G, Licata L. Management of Trastuzumab Deruxtecan-related nausea and vomiting in real-world practice. Front Oncol. 2024;14:1374547. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (1)] |
| 26. | Farhat J, Sakai H, Tsurutani J. Management of nausea and vomiting induced by antibody-drug conjugates. Breast Cancer. 2025;32:278-285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 27. | Aoyama T, Ooki A, Oba K, Nishikawa K, Kawabata R, Honda M, Maeda H, Kanda M, Sugiyama K, Makiyama A, Segami K, Takahashi M, Shindo Y, Namikawa T, Oshima T, Katayama A, Shiosakai K, Sakamoto J. A multicenter randomized open-label phase 2 study investigating optimal antiemetic therapy for patients with advanced/recurrent gastric cancer treated with trastuzumab deruxtecan: the EN-hance study. Int J Clin Oncol. 2025;30:1162-1173. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 28. | Park YH, Bianchini G, Cortés J, Licata L, Vidal M, Iihara H, Roeland EJ, Jordan K, Scotté F, Schwartzberg L, Navari RM, Aapro M, Rugo HS. Nausea and vomiting in an evolving anticancer treatment landscape: long-delayed and emetogenic antibody-drug conjugates. Future Oncol. 2025;21:1261-1272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 29. | Zhao Y, Zhao B, Chen G, Chen Y, Liao Z, Zhang H, Feng W, Li Y, Weng H, Li W, Zhou Y, Ren B, Lu Y, Chen J, Liu Z, Su Z, Wang W, Zhang L. Validation of different personalized risk models of chemotherapy-induced nausea and vomiting: results of a randomized, double-blind, phase III trial of fosaprepitant for cancer patients treated with high-dose cisplatin. Cancer Commun (Lond). 2023;43:246-256. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 30. | Clemons M, Bouganim N, Smith S, Mazzarello S, Vandermeer L, Segal R, Dent S, Gertler S, Song X, Wheatley-Price P, Dranitsaris G. Risk Model-Guided Antiemetic Prophylaxis vs Physician's Choice in Patients Receiving Chemotherapy for Early-Stage Breast Cancer: A Randomized Clinical Trial. JAMA Oncol. 2016;2:225-231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 43] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 31. | Jin Y, Li X, Jiang C, Zhao J, Liu G, Li H, Jin G, Li Q. An Update in Our Understanding of the Relationships Between Gene Polymorphisms and Chemotherapy-Induced Nausea and Vomiting. Int J Gen Med. 2021;14:5879-5892. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 32. | Zhang Y, Yang Y, Zhang Z, Fang W, Kang S, Luo Y, Sheng J, Zhan J, Hong S, Huang Y, Zhou N, Zhao H, Zhang L. Neurokinin-1 Receptor Antagonist-Based Triple Regimens in Preventing Chemotherapy-Induced Nausea and Vomiting: A Network Meta-Analysis. J Natl Cancer Inst. 2017;109:djw217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 21] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 33. | Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, Cates CJ, Cheng HY, Corbett MS, Eldridge SM, Emberson JR, Hernán MA, Hopewell S, Hróbjartsson A, Junqueira DR, Jüni P, Kirkham JJ, Lasserson T, Li T, McAleenan A, Reeves BC, Shepperd S, Shrier I, Stewart LA, Tilling K, White IR, Whiting PF, Higgins JPT. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22257] [Cited by in RCA: 20376] [Article Influence: 2910.9] [Reference Citation Analysis (7)] |
| 34. | Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, Henry D, Altman DG, Ansari MT, Boutron I, Carpenter JR, Chan AW, Churchill R, Deeks JJ, Hróbjartsson A, Kirkham J, Jüni P, Loke YK, Pigott TD, Ramsay CR, Regidor D, Rothstein HR, Sandhu L, Santaguida PL, Schünemann HJ, Shea B, Shrier I, Tugwell P, Turner L, Valentine JC, Waddington H, Waters E, Wells GA, Whiting PF, Higgins JP. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016;355:i4919. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13881] [Cited by in RCA: 13245] [Article Influence: 1324.5] [Reference Citation Analysis (5)] |
| 35. | Wells GA, Shea B, O’Connell D, Peterson J, Welch V, Losos M, Tugwell P. The Newcastle-Ottawa Scale (NOS) for assessing the quality of non-randomised studies in meta-analyses. [cited 20 February 2025]. Available from: https://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. |
| 36. | Wu X, Wu J, Tong G, Cheng B, Chen M, Yu S, He L, Li Z, Wang S. Efficacy of Olanzapine-Triple Antiemetic Regimen in Patients with Gastrointestinal Tumor and High Risk of Chemotherapy-Induced Nausea and Vomiting Receiving Moderately Emetogenic Chemotherapy: A Retrospective Study. Cancer Manag Res. 2020;12:6575-6583. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 37. | Ostwal V, Ramaswamy A, Mandavkar S, Bhargava P, Naughane D, Sunn SF, Srinivas S, Kapoor A, Mishra BK, Gupta A, Sansar B, Pal V, Pandey A, Bonda A, Siripurapu I, Muddu VK, Kannan S, Chaugule D, Patil R, Parulekar M, Dhanawat A, Trikha M, Ghosh J, Noronha V, Menon N, Patil V, Prabhash K, Olver I. Olanzapine as Antiemetic Prophylaxis in Moderately Emetogenic Chemotherapy: A Phase 3 Randomized Clinical Trial. JAMA Netw Open. 2024;7:e2426076. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 38. | Radhakrishnan V, Pai V, Rajaraman S, Mehra N, Ganesan T, Dhanushkodi M, Perumal Kalaiyarasi J, Rajan AK, Selvarajan G, Ranganathan R, Karunakaran P, Sagar TG. Olanzapine versus metoclopramide for the treatment of breakthrough chemotherapy-induced vomiting in children: An open-label, randomized phase 3 trial. Pediatr Blood Cancer. 2020;67:e28532. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 39. | Liu G, Jin Y, Jiang Y, Zhao J, Jiang C, Zhang Z, Zhao L, Li H, Chen F, Wang J, Fan H, Li Z, Jia Y, Jin G, Li Q. A Comparison of the Efficacy of 5 mg Olanzapine and Aprepitant in the Prevention of Multiple-Day Cisplatin Chemotherapy-Induced Nausea and Vomiting. Int J Clin Pract. 2022;2022:5954379. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 40. | Wang W, Lou G, Zhang Y. Olanzapine with ondansetron and dexamethasone for the prevention of cisplatin-based chemotherapy-induced nausea and vomiting in lung cancer. Medicine (Baltimore). 2018;97:e12331. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 41. | Hocking CM, Kichenadasse G. Olanzapine for chemotherapy-induced nausea and vomiting: a systematic review. Support Care Cancer. 2014;22:1143-1151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 51] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 42. | Zhao Y, Yang Y, Gao F, Hu C, Zhong D, Lu M, Yuan Z, Zhao J, Miao J, Li Y, Zhu J, Wang C, Han J, Zhao Y, Huang Y, Zhang L. A multicenter, randomized, double-blind, placebo-controlled, phase 3 trial of olanzapine plus triple antiemetic regimen for the prevention of multiday highly emetogenic chemotherapy-induced nausea and vomiting (OFFER study). EClinicalMedicine. 2023;55:101771. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 28] [Article Influence: 9.3] [Reference Citation Analysis (1)] |
| 43. | Celio L, Fabbroni C. Pro-netupitant/palonosetron (IV) for the treatment of radio-and-chemotherapy-induced nausea and vomiting. Expert Opin Pharmacother. 2018;19:1267-1277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 44. | Zhang M, Zhang X, Liu D, Bai B, Shen W, Meng X, Gao Y, Liu Y, Qin S, Lu P. Clinical Observation of Comparing the Efficacy of Three-drug Regimens Containing Olanzapine/Dexamethasone in Preventing Hyperemetic Chemotherapy-induced Nausea and Vomiting (CINV). 2022 Preprint. Available from: Research Square. [DOI] [Full Text] |
| 45. | Wang SY, Yang ZJ, Zhang L. Olanzapine for preventing nausea and vomiting induced by moderately and highly emetogenic chemotherapy. Asian Pac J Cancer Prev. 2014;15:9587-9592. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 46. | Natale JJ. Reviewing current and emerging antiemetics for chemotherapy-induced nausea and vomiting prophylaxis. Hosp Pract (1995). 2015;43:226-234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 15] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 47. | Tanaka K, Inui N, Karayama M, Yasui H, Hozumi H, Suzuki Y, Furuhashi K, Fujisawa T, Enomoto N, Nakamura Y, Kusagaya H, Matsuura S, Uto T, Hashimoto D, Matsui T, Asada K, Suda T. Olanzapine-containing antiemetic therapy for the prevention of carboplatin-induced nausea and vomiting. Cancer Chemother Pharmacol. 2019;84:147-153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 26] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 48. | Slimano F, Netzer F, Borget I, Lemare F, Besse B. Olanzapine as antiemetic drug in oncology: a retrospective study in non-responders to standard antiemetic therapy. Int J Clin Pharm. 2018;40:1265-1271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 49. | Chan A, Abdullah MM, Ishak WZBW, Ong-Cornel AB, Villalon AH, Kanesvaran R. Applicability of the National Comprehensive Cancer Network/Multinational Association of Supportive Care in Cancer Guidelines for Prevention and Management of Chemotherapy-Induced Nausea and Vomiting in Southeast Asia: A Consensus Statement. J Glob Oncol. 2017;3:801-813. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 50. | Zhang Y, Hou X, Zhang R, Chen G, Huang Y, Yang Y, Zhao Y, Fang W, Hong S, Kang S, Zhou T, Zhang Z, Chen X, Zhang L. Optimal prophylaxis of chemotherapy-induced nausea and vomiting for moderately emetogenic chemotherapy: a meta-analysis. Future Oncol. 2018;14:1933-1941. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 51. | Dielenseger P, Börjeson S, Vidall C, Young A, Jahn P. Evaluation of antiemetic practices for prevention of chemotherapy-induced nausea and vomiting (CINV): results of a European oncology nurse survey. Support Care Cancer. 2019;27:4099-4106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 28] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 52. | Aapro M, Scotté F, Escobar Y, Celio L, Berman R, Franceschetti A, Bell D, Jordan K. Practice Patterns for Prevention of Chemotherapy-Induced Nausea and Vomiting and Antiemetic Guideline Adherence Based on Real-World Prescribing Data. Oncologist. 2021;26:e1073-e1082. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 42] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 53. | Punke AP, Waddell JA. Creation and evaluation of a cancer chemotherapy order review guide for use at a community hospital. J Oncol Pharm Pract. 2019;25:25-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 54. | Davis MP. Novel therapies for nausea and vomiting in advanced illness and supportive cancer care. Palliat Care Soc Pract. 2024;18:26323524241257701. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 55. | Jordan K, Blättermann L, Hinke A, Müller-Tidow C, Jahn F. Is the addition of a neurokinin-1 receptor antagonist beneficial in moderately emetogenic chemotherapy?-a systematic review and meta-analysis. Support Care Cancer. 2018;26:21-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 25] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 56. | Abdel-Rahman O. Neurokinin-1 inhibitors in the prevention of nausea and vomiting from highly emetogenic chemotherapy: a network meta-analysis. Ther Adv Med Oncol. 2016;8:396-406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 18] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 57. | Zhang M, Guo QL, Zhang TT, Fu M, Bi HT, Zhang JY, Zou KL. Efficacy and safety of Aprepitant-containing triple therapy for the prevention and treatment of chemotherapy-induced nausea and vomiting: A meta-analysis. Medicine (Baltimore). 2023;102:e35952. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 58. | Fattori RA, Schlöttgen J, Loro FL, Carvalho PL, Orso R, Macagnan FE. High versus low frequency transcutaneous acupoint electrical stimulation as an adjunct therapy to prevent nausea and vomiting in the first 24 hours after infusion of high-grade emetic chemotherapy: A randomized controlled trial. Fisioter Bras. 2023;24:153-165. [DOI] [Full Text] |