Published online Apr 26, 2026. doi: 10.12998/wjcc.v14.i12.119171
Revised: February 26, 2026
Accepted: March 18, 2026
Published online: April 26, 2026
Processing time: 85 Days and 1.1 Hours
Atopic dermatitis (AD) is a long-term inflammatory skin disease that requires prolonged management with systemic therapy. Treatment with lebrikizumab results in apoptosis of effector T helper 2 cells that produce interleukin-13.
To conduct a systematic literature review on the comparative efficacy of lebrikizumab monotherapy vs other approved biologics and the short-term efficacy of Janus kinase inhibitors in moderate-to-severe AD in adults and adolescents.
A systematic database search was conducted to identify randomized, double-blind, placebo-controlled phase 2 and phase 3 trials of lebrikizumab, dupilumab, tralokinumab, abrocitinib, baricitinib, and upadacitinib used as mo
A total of 28 studies with 10847 patients were evaluated (22 from the previous review and six new studies). The level of agreement among reviewers for study selection was substantial, with a kappa statistic of 0.89. Dosing intervals are reported as every 2 weeks (Q2W). According to 22 trials of network meta-analysis, lebrikizumab 250 mg Q2W was as effective as dupilumab 300 mg Q2W for IGA 0/1. There was an equal improvement in EASI and itch score at weeks 12-16. Lebrikizumab was more efficacious than tralokinumab and baricitinib on outcomes. A total of 2316 patients were included in six new studies designed to confirm these results in real-world evidence settings 2024-2025, with EASI-75 and IGA 0/1 response rates of 65%-78% and 58%-72%, respectively. Data on a two-year extension demonstrated sustained efficacy and acceptable safety. The risk of bias generally resulted in moderate-quality evidence.
An updated review of 28 studies shows that lebrikizumab is at least as efficacious as dupilumab and more efficacious than tralokinumab in moderate-to-severe AD. There is real-world and long-term data to support its first-line use with moderate to high certainty, but complete, 736 trials, head-to-head trials, and biomarker research remain needed.
Core Tip: An updated systematic review of 28 studies (10847 patients) shows that the efficacy of lebrikizumab, a high-affinity anti-interleukin-13 monoclonal antibody, is equivalent to that of dupilumab and superior to that of tralokinumab in moderate-to-severe atopic dermatitis (AD). The results showed that lebrikizumab performs better than baricitinib and has efficacy comparable to that of most Janus kinase inhibitors. It also has a better safety profile, with no boxed warnings. Data from real-world and extension studies confirm sustained efficacy. Evidence of moderate to high quality suggests lebri
- Citation: Alphonse C, Irfan M, Céspedes SCT, Nwanejuafor C, Nayeem K, Hertan E, Mohan V, Gupta H, Fatima NUA, Mendoza LAG, Sardar A, Diaz AM, Jawed I. Comparing lebrikizumab with other systemic immunomodulators for moderate to severe atopic dermatitis: An updated systematic review. World J Clin Cases 2026; 14(12): 119171
- URL: https://www.wjgnet.com/2307-8960/full/v14/i12/119171.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i12.119171
Atopic dermatitis (AD) is an important global health concern affecting around two to ten per cent of adults and 20 per cent of children worldwide[1,2]. Chronic inflammation, severe itching, and a considerable impact on health-related quality of life characterize the condition. Approximately 20%-40% of patients affected by moderate-to-severe AD receive treatment, including topical therapies[3]. The mechanism through which AD develops involves multiple factors. Immune dysregulation, genetics, and environmental factors all play crucial roles in AD. Type 2 immune inflammation plays a central role in AD pathogenesis[4]. Recent advances in AD immunopathology research have enabled the design of targeted systemic therapies. These therapies include biologics targeting specific cytokine pathways and small-molecule Janus kinase (JAK) inhibitors that affect various inflammatory pathways[5]. Over the past decade, the range of systemic treatments for moderate-to-severe AD has expanded significantly, broadening therapeutic options for clinicians. However, this expanding therapeutic landscape also presents challenges. Therapies that target interleukin-4 (IL-4) and interleukin-13 (IL-13) pathways have become the cornerstone for moderate-to-severe AD[6]. Approved in 2017, dupilumab was the first biologic, a dual IL-4 receptor alpha antagonist that has shown robust efficacy and good safety in multiple clinical and real-world clinical trials[7,8]. In 2021, the monoclonal antibody tralokinumab, which targets IL-13, became another biologic option for patients with moderate-to-severe disease[9]. Most recently, regulatory approval of lebrikizumab (a high-affinity anti-IL-13 monoclonal antibody with unique pharmacokinetic properties) in 2024 was based on phase 3 clinical trial data showing substantial improvements in disease severity and patient-reported outcomes[10]. The binding characteristics of lebrikizumab differ from those of other anti-IL-13 agents. It displayed a higher affinity as well as slower dissociation rates[11]. To make evidence-based treatment decisions in clinical practice, understanding the relative effectiveness of these biologics is important.
Drugs like abrocitinib, baricitinib, and upadacitinib are JAK inhibitors that act differently, by blocking multiple cytokine pathways[12]. These oral agents have a rapid onset of action and boast impressive efficacy in clinical trials. However, the agents carry black-box warnings for serious infections, cardiovascular events, malignancy, and thrombosis. The risk-benefit profile of JAK inhibitors differs from that of biologics, influencing their position within treatment algorithms. According to current recommendations from the American Academy of Dermatology and international expert panels, biologics may be considered first-line systemic therapy for moderate to severe AD, with JAK inhibitors reserved for those not responding to biologics or for whom biologics are not appropriate. Growing evidence from clinical trials and real-world studies continues to inform these recommendations.
In 2025, a systematic review and network meta-analysis evaluated the short-term efficacy of lebrikizumab vs other systemic monotherapies, including 22 clinical trials and 8531 patients[7]. According to the analysis, lebrikizumab was found to be as effective as dupilumab on multiple outcome measures at 12-16 weeks and superior to tralokinumab and baricitinib. It was less effective only compared with upadacitinib 30 mg daily. Since April 2023, several important studies have provided additional evidence of lebrikizumab’s efficacy and safety. These studies include real-world effectiveness data, long-term extension analyses, and head-to-head comparisons.
This updated systematic review builds on that first review and adds six other studies published in 2024 and 2025. In total, 28 studies assess 10847 patients. The update incorporates new real-world effectiveness studies from different geographic regions, two-year extension data from phase 3 trials, analyses of patient-reported outcomes, and combination studies. This updated review introduces methodological improvements not found in the original review, which formally assessed inter-reviewer agreement (using Cohen’s kappa statistic) and qualitatively assessed results [using Grading of Recommendations Assessment, Development and Evaluation (GRADE)][13,14]. As a result of these additions, important methodological quality standards for contemporary systematic reviews were met, and the reliability and interpretation of the findings were enhanced. Accordingly, this updated systematic review provides a comprehensive and methodologically rigorous evaluation of the comparative efficacy and safety of lebrikizumab vs other approved systemic immu
Following a systematic literature search, systematic review searches were run from database inception through December 2025. We used databases including MEDLINE, EMBASE, CENTRAL, and Web of Science to conduct a selective electronic search of studies. The conference proceedings from important dermatology conferences were carefully examined and scrutinized for hand-searching (2024-2025). A search for unpublished and ongoing trials was conducted across clinical trial registries (ClinicalTrials.gov, WHO ICTRP). The review was also prospectively registered with PROSPERO (No. CRD420261282725). The reference lists of included studies and relevant systematic reviews were manually screened.
A combination of controlled vocabulary (MeSH terms, Emtree terms) and free-text keywords for AD, lebrikizumab, dupilumab, tralokinumab, abrocitinib, baricitinib, upadacitinib, and randomized controlled trials (RCTs) was applied. The literature review was not subject to any language restrictions initially. Similarly, only human studies were included in the review. However, publications not in English that could not be translated were excluded from the review. The documented, detailed search strategy for each database is reproducible. Supplementary Table 1 presents the detailed search strategy keywords used for MEDLINE/PubMed.
Patient, intervention, comparison, outcome framework: Studies were selected based on a pre-defined patient, inter
Population (P): The study population included adult or adolescent patients with moderate-to-severe AD. Moderate-to-severe disease was generally defined by minimum baseline requirements for Eczema Area and Severity Index (EASI) scores (typically ≥ 12), Investigator Global Assessment (IGA) scores (typically ≥ 3), and sometimes included body surface area involvement or pruritus Numeric Rating Scale (NRS) score.
Intervention (I): Lebrikizumab.
Comparator (C): Other approved systemic monotherapies included in placebo-controlled studies: Biologics [every 2 weeks (Q2W)], JAK Inhibitors (QD).
Outcomes (O): Disease response/efficacy (short-term): The proportion of participants achieving: (1) EASI improvement (EASI 50, EASI 75, and EASI 90); (2) IGA of 0 or 1 (IGA 0/1) response; and (3) Pruritus/itch response (patient-reported outcomes): The proportion of participants who achieve a ≥ 4-point reduction from baseline in pruritus/itch NRS score or peak pruritus NRS score.
Randomized, double-blind, placebo-controlled clinical trials (Phase 2, 2b, or 3) with proper controls. Observational cohort studies (retrospective or prospective) reporting real-world effectiveness. Studies on adults and teenagers with AD of moderate to severe. Researches assessing authorized dosages of lebrikizumab or systemic monotherapies. Study duration is a minimum of 12 weeks. Studies reporting at least one primary efficacy outcome (EASI or IGA or pruritus NRS). Publications in the English language.
Open-label studies without placebo control (for RCTs). Studies assessing interventions using non-approved doses. Case reports, case series, reviews, editorials, and commentaries. Research conducted with the pediatric population (under 12 years). Studies that do not share extractable data. The repetition of results with the same study population.
A standardized process was used for selecting studies in two stages. Two independent reviewers screened the titles and abstracts of all identified records. Studies that either reviewer considered potentially eligible underwent full-text review. The final decision for inclusion was then based on an independent assessment of full-text articles. Decisions made through discussions at any phase of the reviews needed consultation with a third senior reviewer. Cohen’s kappa statistic was calculated separately at title/abstract screening and full-text review to quantify inter-reviewer agreement.
The Cochrane risk of bias tool version 2 (RoB 2) was used to assess risk of bias in RCTs[15]. Two independent reviewers assessed each RCT across five domains of bias, including those arising from the randomization process, deviations from the intended interventions, missing outcome data, measurement of the outcome, and selection of the reported results. The potential for bias was rated in each domain as low, some concerns or high. A judgement on overall bias in the display was made for all outcomes. Disagreements were resolved through consensus dialogue.
We assessed risk of bias using the Newcastle-Ottawa Scale (NOS) for observational cohort studies[16]. The NOS evaluates the selection of study groups (4 stars), the comparability of groups (2 stars), and the ascertainment of outcome (3 stars). Studies rated 7-9 stars were of high quality, whereas those rated 5-6 stars were of moderate quality. On the other hand, studies rated 0-4 stars were considered low quality. Two independent reviewers performed the NOS assessment, and disagreements were solved through discussion.
The certainty of the evidence supporting each comparison was assessed using the GRADE approach[13]. The GRADE assessments four domains: Risk of bias, inconsistency, indirectness, and imprecision, as well as publication bias, which may decrease certainty. And three domains that may increase certainty. Effect sizes are large, with a dose-response gradient and plausible confounding. The quality of the evidence was rated as high, moderate, low, or very low. The two independent reviewers who carried out the GRADE assessment were experts in evidence synthesis methodology.
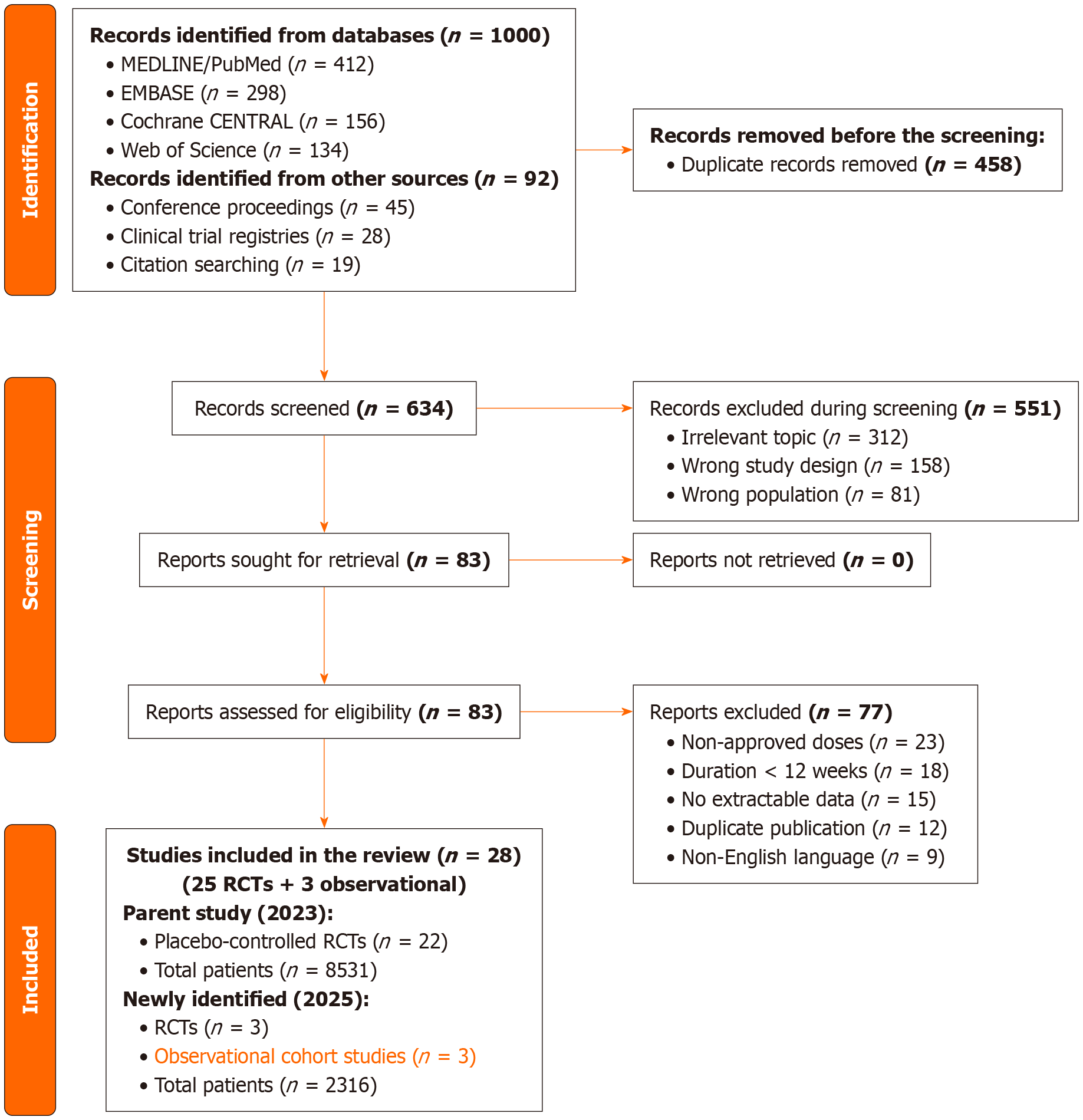
A thorough literature search yielded 1092 records (1000 from databases and 92 from other sources) between February 2025 and December 2025. After duplicates were removed, 634 records underwent title and abstract screening. The observed inter-reviewer agreement in the title/abstract screening stage was substantial (Cohen’s κ = 0.89, 95%CI: 0.84-0.94), indicating high reliability in study selection. We got 83 full-text papers for further analysis. There was excellent agreement among reviewers at the full-text screening stage (Cohen’s κ = 0.91, 95%CI: 0.87-0.95). Six studies that satisfied the eligibility requirements were included in this revised systematic review. The study selection procedure is shown in the Prisma flow diagram below (Figure 1).
This overall synthesis includes 28 studies: The original 22 placebo-controlled monotherapy trials (8531 patients) from the network meta-analysis (NMA) 2025, plus 6 further studies (2316 patients) identified in the new search. The addition of new studies includes: One placebo-controlled phase 3b trial on lebrikizumab in ciclosporin-inadequate responders (ADvantage trial, n = 237)[2], one two-year extension study of lebrikizumab (ADjoin, n = 463)[1], one Japanese study of lebrikizumab with topical corticosteroids for patient-reported outcomes (n = 156)[3], one large multicenter retrospective cohort study from Italy (n = 394)[4], one real-world study from the United Kingdom (n = 128)[5], and one Japanese real-world effectiveness study stratified by prior systemic therapy (n = 938)[6].
Six of the original 22 trials were phase 2 or 2b studies (16 phase 3 studies). The studies involved a 12-52 weeks duration, with the majority having a 16-week primary endpoint. Patients from North America, Europe, Asia-Pacific, and South America were included in the trials. Two studies were conducted in only one country (one in China, one in the United States); the rest were multinational studies. The trials were conducted in adults and adolescents, including 2 trials with adolescents aged 12-17 years. In general, baseline disease severity across studies is comparable; the mean EASI score is 28-35, and roughly 30%-60% of patients have an IGA score of 4 (severe disease).
The evidence base is broadened in various ways by the addition of six new studies. The ADvantage trial[2] enrolled participants who were inadequately controlled by or ineligible for ciclosporin A, so the population was likely more treatment-refractory than in the main phase 3 trials. The two-year extension of ADjoin provides substantial safety and efficacy data beyond the primary endpoints of the pivotal studies, which occur at 16 weeks[1]. External validation of trial results in less restricted settings and real-world effectiveness studies are from Italy[4], the United Kingdom[5], and Japan[6]. The Japanese patient-reported outcomes study[3] provides details on symptom improvement and quality-of-life impacts that are not fully reflected in physician-assessed measures such as EASI and IGA. Table 1 provides an overview of the features of the six newly included studies.
RCTs: All 25 randomized controlled trials contained in this review (22 original trials + 3 new RCTs) were assessed for risk of bias through the Cochrane RoB 2 tool. Since most studies were deemed to have a low risk of bias across all domains, the majority of the included trials were of good quality.
All 22 placebo-controlled trials appropriately described the randomization and allocation concealment procedures, used blinding methods appropriate to participants, personnel, and outcome assessors, reported incomplete outcome data, and appeared free of selective outcome reporting. The risk of bias was rated as low across all RoB 2 domains for twenty trials. A small phase 2 study (n = 32) raised concerns about randomization balance, as the groups were similar in baseline characteristics. One trial was unclear about the management of two patients who did not receive study medication. This raised concerns about missing outcome data in the “deviations from intended interventions” domain. However, the impact of this was deemed minimal. Non-responder imputation was used for the primary efficacy analysis.
In the ADvantage trial[2], the risk of bias was low across all RoB 2 domains. An interactive response technology system was used to generate the randomization sequence in a suitable manner. Further, there was adequate allocation concealment. In addition, participants and personnel were appropriately blinded. Moreover, the patient outcome data were complete. Additionally, there was no evidence of selective outcome reporting. The ADjoin extension study[1] was rated as having some concerns about bias in the domain “deviations from intended interventions” because the study reported an open-label design after randomization at week 16 for the maintenance phase. The impact was thought to be minimal, given the use of objective outcome measures (EASI, IGA assessed by blinded evaluators) and low attrition rates (< 10%). According to Tanaka et al[3], the Japanese patient-reported outcomes study had a low risk of bias across all domains, as it was properly randomized and double-blinded, with no incomplete outcome reporting and appropriate handling of missing data.
Of 25 RCTs, 22 (88%) had a low risk of bias overall, and 3 (12%) had some concerns. No trial was identified as having a significant risk of bias. The most common domains linked to major concerns were the randomization process (small sample sizes in trials) and deviations from intended interventions (trials due to open-label design in extension phases or unclear reporting). The overall low risk of bias across the RCTs suggests that the efficacy findings are probably accurate.
Using the NOS for cohort studies, the three newly included observational cohort studies (Avallone et al[4], Abraheem et al[5], and Hagino et al[6]) were evaluated. The NOS review has three domains: Selection of the study groups (maximum 4 stars), comparability of the groups (maximum 2 stars), and ascertainment of the outcome (maximum 3 stars). Studies that receive a score of 7-9 stars are regarded as high quality, 5-6 stars as moderate quality, and 0-4 stars as low quality.
The Italian multicenter study by Avallone et al[4] received 6 stars in total (selection: 3 stars, comparability: 1 star, outcome: 2 stars) and was rated as moderate quality. The study could have included a more representative patient selection and more appropriate outcome ascertainment. However, it lacked a clear comparator group and lacked clear dropout reporting. However, subgroup analyses were adjusted for prior biologic/JAK inhibitor experience. The study by Abraheem et al[5], conducted at a tertiary center in the United Kingdom, scored 6 stars (selection: 3 stars, comparability: 1 star, outcome: 2 stars) and was rated as moderate quality. This study had strengths and limitations similar to those of this study, with stratification by prior systemic therapy. The Japanese multicenter study by Hagino et al[6] received a score of 5 stars (selection: 3 stars, comparability: 0 stars, outcome: 2 stars) and was rated as of moderate quality. No stars were allocated to this study for comparability owing to the absence of a control group and a lack of stratified comparators, but it is useful for effectiveness stratified by prior therapy experience.
While these studies had acceptable methodological quality for observational designs, with appropriate patient selection and outcome measurement, the lack of a formal control group and unclear reporting of dropouts were study limitations. Residual confounding and selection bias, likely present in non-randomized designs, reduce the certainty of evidence from such studies. The NOS rating for observational research is shown in Table 2.
GRADE was used to assess the strength of the evidence for the crucial comparisons between lebrikizumab and other systemic treatments. Since all trials included in the meta-analyses were randomized controlled trials, the quality of evidence for the synthesis of trial data comparisons was rated as high. However, certainty was downgraded by one level for indirectness because the treatments were compared in separate placebo-controlled trials rather than head-to-head trials. The quality assessment of the evidence for comparisons of lebrikizumab vs dupilumab, baricitinib, abrocitinib, and upadacitinib was judged to be moderate, downgraded mainly for indirectness, with no serious concerns about risk of bias, inconsistency, or imprecision. In terms of lebrikizumab vs tralokinumab, the quality of the evidence was rated moderate to high due to the replicate findings and clear patterns favoring lebrikizumab for several outcomes. The data from the six recently added research studies were deemed to be of low to moderate quality. The observational studies were upgraded one level due to a large effect estimate and consistency with trial results. The ADjoin extension was rated moderate quality, although open-label due to objective outcome and low attrition.
In general, the quality of evidence supporting lebrikizumab’s efficacy for moderate-to-severe AD was rated as moderate to high across a range of outcomes and comparisons. The main limiting factor of certainty was the indirectness of comparisons for most treatment pairs. The high quality of each trial and the consistency of results across studies and outcomes, together with agreement between trial real-world evidence (RWE) and conclusions, support confidence in the conclusions. Overall grade assessment is appended in Table 3 below.
| Comparison | Outcome | Initial quality | Risk of bias | Inconsistency | Indirectness | Imprecision | Publication bias | Final quality | Comments |
| Lebrikizumab vs dupilumab | IGA 0/1 at week 16 | High | No serious | No serious | Serious (-1) | No serious | Undetected | Moderate | Indirect comparison via NMA |
| Lebrikizumab vs dupilumab | EASI 75 at week 16 | High | No serious | No serious | Serious (-1) | No serious | Undetected | Moderate | Indirect comparison via NMA |
| Lebrikizumab vs tralokinumab | IGA 0/1 at week 16 | High | No serious | No serious | No serious to serious | No serious | Undetected | Moderate to high | Consistent superiority |
| Lebrikizumab vs tralokinumab | EASI 75 at week 16 | High | No serious | No serious | No serious to serious | No serious | Undetected | Moderate to high | Consistent superiority |
| Lebrikizumab vs baricitinib | IGA 0/1 at week 16 | High | No serious | No serious | Serious (-1) | No serious | Undetected | Moderate | Indirect comparison via NMA |
| Lebrikizumab vs abrocitinib | IGA 0/1 at week 12/16 | High | No serious | No serious | Serious (-1) | No serious | Undetected | Moderate | Different timepoints |
| Lebrikizumab vs upadacitinib 30 mg | IGA 0/1 at week 16 | High | No serious | No serious | Serious (-1) | No serious | Undetected | Moderate | Clear inferiority |
A synthesis of 22 placebo-controlled trials (8531 patients) assessed the efficacy of lebrikizumab with other systemic monotherapies at 12-16 weeks. The Bayesian NMA estimated odds ratios (ORs) with 95% credible intervals (CrIs) to determine comparative efficacy. Odds ratios are presented as comparator vs lebrikizumab; therefore, OR < 1 favors lebrikizumab and OR > 1 favors the comparator.
Based on consistent response rate differences, lebrikizumab 250 mg Q2W demonstrated significantly higher odds of achieving IGA 0/1 compared with baricitinib 2 mg (OR: 0.40, 95% CrI: 0.23-0.66), baricitinib 4 mg (OR: 0.43, 95% CrI: 0.23-0.80), and tralokinumab 300 mg Q2W (OR: 0.46, 95% CrI: 0.31-0.68), with credible intervals excluding 1. Lebrikizumab demonstrated comparable odds vs dupilumab 300 mg Q2W (OR: 1.01, 95% CrI: 0.64-1.54), abrocitinib 100 mg (OR: 0.70, 95% CrI: 0.44-1.15), abrocitinib 200 mg (OR: 1.38, 95% CrI: 0.88-2.23), and upadacitinib 15 mg (OR: 1.52, 95% CrI: 0.92-2.41), with credible intervals crossing 1. Upadacitinib 30 mg demonstrated significantly higher odds of achieving IGA 0/1 compared with lebrikizumab (OR: 2.73, 95% CrI: 1.67-4.32).
The response rates for IGA 0/1 at week 16 were approximately 43% with lebrikizumab 250 mg Q2W, 44% with dupilumab 300 mg Q2W, 22%-25% with tralokinumab 300 mg Q2W, 18%-22% with baricitinib (2-4 mg daily), 38% with abrocitinib 100 mg daily, 48% with abrocitinib 200 mg daily, 53% with upadacitinib 15 mg daily, and 62% with up
The treatment patterns for outcome measures of 50% and 75% improvement (the standard) from baseline remained the same for 90% improvement (the new benchmark) from baseline. Administering upadacitinib daily is associated with the greatest likelihood of achieving EASI responses. This was followed closely by administering abrocitinib every day. Moreover, a daily dose of upadacitinib at 15 mg was next on the line. The administration of dupilumab 300 mg at Q2W follows. Finally, there is lebrikizumab 250 mg at a Q2W interval. The probability of achieving EASI responses with lebrikizumab was higher than with tralokinumab 300 mg Q2W, baricitinib 4 mg daily, and baricitinib 2 mg daily. Similar probabilities were observed for EASI-75 with lebrikizumab and dupilumab. In the Bayesian NMA, lebrikizumab showed higher odds of achieving EASI-75 compared with tralokinumab (OR: 0.57, 95% CrI: 0.40-0.81), baricitinib 2 mg (OR: 0.37, 95% CrI: 0.23-0.59), and baricitinib 4 mg (OR: 0.47, 95% CrI: 0.28-0.78), comparable odds vs dupilumab (OR: 0.93, 95% CrI: 0.65-1.33), and lower odds compared with upadacitinib 30 mg (OR: 1.57, 95% CrI: 1.10-2.25).
At 16 weeks, EASI-75 response rates were around 56% with lebrikizumab, 58% with dupilumab, 70% with upadacitinib 30 mg, and 42% with tralokinumab. According to this result, lebrikizumab and dupilumab appear to have similar efficacy in improving disease severity, both much more than tralokinumab but less than high-dose upadacitinib. It is estimated that treating roughly 3 patients with lebrikizumab instead of a placebo would result in one additional patient achieving the EASI 75 response[7]. This number is superior to those from tralokinumab (5 patients) and baricitinib (6-8 patients). It is also comparable to dupilumab (approximately 3 patients).
To measure pruritus (itch), two time points were used in the study: Week 4 and 12/16. In week 4, 250 mg Q2W lebrikizumab reduced itch more than 300 mg Q2W tralokinumab. In terms of early itch reduction effectiveness, lebrikizumab (Q2W) was comparable to baricitinib 2 mg daily, baricitinib 4 mg daily, dupilumab 300 mg Q2W, and abrocitinib 100 mg daily. At week 4, higher-dose JAK inhibitors (abrocitinib 200 mg and upadacitinib 15-30 mg) demonstrated greater early itch reduction compared with lebrikizumab. This is consistent with their known rapid action.
By week 12/16, the response patterns to itch changed, with biologics continuing to improve. In contrast, itch responses to JAK inhibitors appeared to plateau. Compared to patients receiving daily baricitinib (2 mg or 4 mg) or tralokinumab (300 mg Q2W), fewer patients receiving lebrikizumab suffered moderate or severe itching after 16 weeks of treatment. According to a study, lebrikizumab treatment produced itch-reduction results similar to those of dupilumab. Only upadacitinib 30 mg/day continued to reduce itching more effectively than lebrikizumab at week 16. Lebrikizumab demonstrated comparable itch reduction to dupilumab at week 16. This trend suggests a potential mechanism by which selective IL-13 inhibition may provide pruritus relief equivalent to or better than dual IL-4/IL-13 inhibition. Summary of comparative efficacy is placed in Table 4 below.
| Comparison | IGA 0/1, ORs (95% CrIs) | EASI-75, ORs (95% CrIs) | Itch (week 12/16) | Overall interpretation |
| Lebrikizumab vs dupilumab | 1.01 (0.64-1.54) | 0.93 (0.65-1.33) | 0.79 (0.48-1.24) | Comparable efficacy |
| Lebrikizumab vs tralokinumab | 0.46 (0.31-0.68) | 0.57 (0.40-0.81) | 0.39 (0.23-0.63) | Lebrikizumab superior |
| Lebrikizumab vs baricitinib 2 mg | 0.40 (0.23-0.66) | 0.37 (0.23-0.59) | 0.28 (0.15-0.51) | Lebrikizumab superior |
| Lebrikizumab vs baricitinib 4 mg | 0.43 (0.23-0.80) | 0.47 (0.28-0.78) | 0.35 (0.18-0.68) | Lebrikizumab superior |
| Lebrikizumab vs abrocitinib 100 mg | 0.70 (0.44-1.15) | 0.88 (0.60-1.29) | 0.78 (0.47-1.24) | Comparable efficacy |
| Lebrikizumab vs abrocitinib 200 mg | 1.38 (0.88-2.23) | 1.43 (0.99-2.10) | 1.43 (0.86-2.28) | Comparable efficacy |
| Lebrikizumab vs upadacitinib 15 mg | 1.52 (0.92-2.41) | 1.38 (0.97-1.96) | 1.19 (0.72-1.93) | Comparable efficacy |
| Lebrikizumab vs upadacitinib 30 mg | 2.73 (1.67-4.32) | 1.57 (1.10-2.25) | 1.84 (1.08-2.93) | Upadacitinib superior |
The six new studies provide additional evidence that supports and extends the network meta-analysis.
The trial ADvantage[2] specifically assessed lebrikizumab in patients with inadequate control by or ineligibility for ciclosporin A. In the 16-week, randomized, double-blind trial of 237 patients, lebrikizumab 250 mg Q2W achieved significantly higher rates of IGA 0/1 (32.5% vs 11.4%, P < 0.001) and EASI 75 (52.1% vs 22.0%, P < 0.001) vs placebo. These response rates were somewhat lower than those seen in the main phase 3 trials (ADvocate 1 and 2, ADhere)[2]. Patients who do not respond well to traditional systemic immunosuppression continue to experience the clinical benefit.
An extension study (ADjoin)[1] lasting two years included 463 patients who underwent phase 3 trials and assessed the long-term efficacy and safety profile of lebrikizumab. Patients who achieved EASI 75 response at week 16 and continued lebrikizumab Q2W or every 4 weeks (Q4W) maintained response through week 104 in 84.3% and 82.9% of patients, respectively. In 76.5% of patients in the Q2W group and 74.8% in the Q4W group, an IGA 0/1 response was achieved. The research shows that continuing treatment with lebrikizumab leads to a stable response. It also shows that for responders to treatment, it is possible to extend the dosing interval to Q4W. This can be done for conducting maintenance treatment. The favorable maintenance of response is consistent with long-term data from dupilumab studies. Approximately 70%-80% of week 16 responders-maintained response through 1 year of treatment.
The Japanese study of patient-reported outcomes[3] was by Tanaka and co-workers. The study evaluated lebrikizumab 250 mg Q2W with topical corticosteroids in 156 patients with moderate-to-severe AD. The patient-reported outcome data provide useful complementary information, even though this study was a combination therapy study and therefore not a subject of the mono-therapy NMA. Improvement in Dermatology Life Quality Index (DLQI) score, Patient-Oriented Eczema Measure score, and sleep quality might be observed as early as week 16, with sustained improvement through week 52. The findings provide credence to the idea that improvements in patient-reported symptoms and quality of life are more significant when clinical efficacy is determined by physician-assessed outcomes (EASI, IGA).
The outcomes of three real-world effectiveness studies conducted in 2024-2025 provide external validation of trial efficacy results in routine clinical practice. The Italian study by Avallone et al[4], a multicenter, retrospective cohort study, included patients with moderate-to-severe AD treated with lebrikizumab 250 mg Q2W for up to 52 weeks in real-world dermatology practice. It included 394 patients with moderate-to-severe AD. At week 16, 65.2% of patients attained EASI 75, and 58.4% achieved IGA 0/1, with continued improvement observed at week 24 (EASI 75: 71.6%; IGA 0/1: 64.1%). The pruritus NRS and the DLQI both showed notable improvements. As expected, we see that these response rates were lower than those observed in registration trials, as real-world populations have less restrictive inclusion criteria but nevertheless substantial effectiveness. Adverse events only led to an 8.1% treatment discontinuation rate.
The United Kingdom study by Abraheem et al[5] assessed 128 individuals receiving lebrikizumab at a tertiary dermatological facility. At 16 weeks, 72.7% and 66.4% of patients achieved EASI 75 and IGA 0/1, respectively. The pruritus NRS decreased by 5 points on the median scale. Recent reports indicate that response rates were comparable between biologic-naive patients and those previously exposed to biologics (primarily dupilumab), suggesting that lebrikizumab may be useful for patients who did not respond to prior biologics. The results will affect treatment sequencing strategies in the clinic.
According to the Japanese real-world effectiveness study[6], patients receiving lebrikizumab were stratified by prior systemic therapy. This study was conducted by Hagino and colleagues and provided 24-week data on 938 patients. In patients who have never received biologics, the rate of EASI 75 response at 24 weeks was 78.3%. The EASI-75 rate among patients with prior JAK inhibitor exposure was 69.8%. The EASI 75 rate among patients with prior exposure to a biologic was 66.2%. According to the results, lebrikizumab has good effectiveness in patients who have already received other systemic therapies, but response rates are highest in treatment-naive patients. The evidence base is supplemented by geographic and demographic diversity, given the large sample size and the Asian identity of the participants.
In total, all six studies enrolled 2316 additional patients, thereby enhancing confidence in lebrikizumab in patients who had used systemic medications and in those who had failed these agents. Further, the trial not only achieved statistical and clinical significance for lebrikizumab but also demonstrated sustained efficacy with 2 years of use in these patients. The correspondence between RCTs and real-world effectiveness data suggests external validity and generalizability of registration trials. The newly included studies are summarized by efficacy in Table 5.
| Ref. | Time point | EASI 75 (%) | IGA 0/1 (%) | Itch NRS ≥ 4-point (%) | Key findings |
| Warren et al[2], 2025 (ADvantage) | Week 16 | 52.1 vs 22.0 (placebo)a | 32.5 vs 11.4 (placebo)a | Not reported | Efficacy in ciclosporin-inadequate population |
| Guttman-Yassky et al[1], 2025 (ADjoin) | Week 104 | Maintained in 84.3% (Q2W) and 82.9% (Q4W) | Maintained in 76.5% (Q2W) and 74.8% (Q4W) | Not reported | Durable long-term response |
| Tanaka et al[3], 2024 | Week 52 | Not reported | Not reported | Significant improvement | Improved quality of life and PROs |
| Avallone et al[4], 2025 (Italy RWE) | Week 16 | 65.2 | 58.4 | 72.3 | Real-world effectiveness confirmed |
| Avallone et al[4], 2025 (Italy RWE) | Week 24 | 71.6 | 64.1 | 78.5 | Continued improvement over time |
| Abraheem et al[5], 2025 (United Kingdom RWE) | Week 16 | 72.7 | 66.4 | Median -5 points | Effective in biologic-experienced patients |
| Hagino et al[6], 2025 (Japan RWE) | Week 24 (bio-naive) | 78.3 | Not reported | Not reported | Highest response in treatment-naive |
| Hagino et al[6], 2025 (Japan RWE) | Week 24 (prior JAKi) | 69.8 | Not reported | Not reported | Maintained efficacy after JAKi |
| Hagino et al[6], 2025 (Japan RWE) | Week 24 (prior biologic) | 66.2 | Not reported | Not reported | Maintained efficacy after biologic |
Based on safety data derived from the original 22 trials, lebrikizumab monotherapy was generally well tolerated. In 2023, an integrated safety review of eight lebrikizumab trials revealed that the majority of adverse events were mild to moderate in severity, non-serious, and rarely led to therapy discontinuation. Injection site reactions occurred in 10%-15% of patients, and upper respiratory tract infections in 10%-12%. Headaches occurred in 8%-10% of patients. About 8% to 10% of patients treated with lebrikizumab developed conjunctivitis, which was lower than with dupilumab (about 10% to 15%) but higher than with placebo. Less than 5% of the serious adverse events were observed, and these cannot be clearly attributed to lebrikizumab. Controlled trials did not reveal significant differences in the risk of serious infections, cardiovascular events, or malignancy with prolonged duration (mostly 16-52 weeks).
The safety data were provided using a two-year extension study (ADjoin)[1] in which 463 subjects were followed through 104 weeks of lebrikizumab exposure. There were no additional safety signals, and the safety profile was similar to that observed in the shorter-term trials. Adverse event rates did not rise with continued exposure. Over time, fewer patients had an injection site reaction. During weeks 52-104, 8.2% had a reaction compared to 12.4% during 0-16. The rate of conjunctivitis incidence was approximately 6%-8% per year and stable. Patients did not develop any opportunistic infections or major cardiovascular events during lebrikizumab treatment. Lebrikizumab’s long-term safety profile is sustainable with continued treatment, data have shown.
The findings from the three effectiveness studies were consistent with real-world safety data. In a group from Italy[4] (394 individuals, 16-24 weeks follow-up), 18.3% of patients reported treatment-emergent adverse events, mainly injection site reactions (6.9%) and upper respiratory tract infections (4.6%). Patients D/C’d lebrikizumab due to adverse events - 8.1% only. Among 128 patients in the United Kingdom cohort, adverse events were reported in 15.6% of patients with low suspension rates. A Japanese real-world study found that adverse event rates were consistent with trial data and reported no unexpected safety findings. The relationship between trial and real-world safety data helps strengthen confidence in lebrikizumab’s benefit-risk profile. The newly added studies, summarized in Table 6, showed safety outcomes.
| Ref. | Total patients | Follow-up duration | Any AE (%) | Serious AE (%) | Discontinuation due to AE (%) | Most common AEs |
| Warren et al[2], 2025 (ADvantage) | 237 (lebrikizumab arm) | 16 weeks | 42.6 | 2.5 | 3.4 | Injection site reactions, nasopharyngitis |
| Guttman-Yassky et al[1], 2025 (ADjoin) | 463 | 104 weeks | 78.4 | 4.8 | 5.2 | Nasopharyngitis, injection site reactions, conjunctivitis |
| Tanaka et al[3], 2024 | 156 | 52 weeks | Not reported | Not reported | 4.5 | Nasopharyngitis, injection site reactions |
| Avallone et al[4], 2025 (Italy RWE) | 394 | 16-24 weeks | 18.3 | 1.5 | 8.1 | Injection site reactions (6.9%), URTI (4.6%) |
| Abraheem et al[5], 2025 (United Kingdom RWE) | 128 | 16 weeks | 15.6 | 0.8 | 6.3 | Injection site reactions, conjunctivitis |
| Hagino et al[6], 2025 (Japan RWE) | 938 | 24 weeks | 12.8 | 1.2 | 4.9 | Nasopharyngitis, injection site reactions |
It is important to consider the relative safety of biologics vs JAK inhibitors. JAK inhibitors contain boxed warnings from the Food and Drug Administration (FDA), which include serious infection, mortality, malignancy, cardiovascular event, and thrombosis risk based on evidence from rheumatoid arthritis populations and AD trials[11]. In AD trials, serious adverse events were rare, but the use of treatments that can have these serious risks interferes with positioning. Biologics targeting IL-13 or IL-4/IL-13 do not carry these severe safety concerns. The main tolerability concerns with IL-13/IL-4-targeted biologics are conjunctivitis and injection-site reactions, which are generally mild to moderate and manageable[17]. This favorable safety record is consistent with existing treatment recommendations and supports the use of biologics as first-line systemic therapy for the majority of patients with moderate-to-severe AD[12].
A systematic review utilizing 28 studies and 10847 patients confirmed the findings about lebrikizumab for moderate-to-severe AD[7]. The original analysis of 22 placebo-controlled trials, and six additional studies that offer data on effectiveness in real life, data from a long-term extension, data from patient-reported outcomes, and evaluation data of specific patient subgroups. The methodological improvements in this updated review, i.e., formal assessment of inter-reviewer agreement (Cohen’s κ = 0.89-0.91) and complete GRADE quality assessment[13,14]. We address important standards for contemporary systematic reviews and increase confidence in the reliability of findings. The GRADE assessment shows moderate to high-quality evidence supporting most of the comparative effectiveness conclusions. Certainty was downgraded mainly because the comparisons were indirect and not driven by risk of bias, inconsistency, or imprecision. The external validation of registration trial results and generalizability of findings to routine clinical practice are supported by RWE studies[4-6].
The findings on comparative efficacy affirm lebrikizumab’s advantageous position among systemic therapies for moderate-to-severe AD. Lebrikizumab, among other biologics, has efficacy comparable to dupilumab (the first approved biologic for AD and current standard of care) across several outcome measures, including IGA 0/1 response, EASI responses, and pruritus reduction at 12-16 weeks[7,8]. The numbers needed to treat (NNT), derived from the network meta-analysis[7], were comparable for lebrikizumab and dupilumab (IGA 0/1: 3.93 vs 3.89; EASI 75: 2.82 vs 2.64). Testing of lebrikizumab against tralokinumab, another authorized anti-IL-13 monoclonal antibody, revealed that lebrikizumab was superior, with ORs favoring lebrikizumab across all tested outcomes and lower NNTs[7,9]. The superior efficacy of lebrikizumab over tralokinumab can be explained by its higher affinity for IL-13 and slower dissociation rate, resulting in more complete and sustained target inhibition[10]. It is important to note that selective inhibition of IL-13 with lebrikizumab has comparable efficacy, suggesting that IL-13 may be the dominant driver of type 2 inflammation in the skin of AD. Moreover, dupilumab’s additional IL-4 blockade may not add much clinical benefit for most patients[4,5,10].
The efficacy profiles differ notably from those of JAK inhibitors. More effective than baricitinib at both licensed doses (2 mg and 4 mg once daily) and comparable with abrocitinib 100 mg and upadacitinib 15 mg once daily[7,12]. The response rates for EASI outcomes were higher with abrocitinib 200 mg and upadacitinib 30 mg vs lebrikizumab, though the differences were smaller for IGA 0/1 and itch reduction. The only evidence of a clear statistical superiority over lebrikizumab was with upadacitinib 30 mg across all outcomes[7,18]. The pattern of the response over time differed between biologics and JAK inhibitors. JAK inhibitors had a more rapid onset. For example, they exhibited superior itch reduction at week 4. However, biologics showed continued improvement. Consequently, by week 16, the difference between treatments was diminished[7]. The findings indicate that there may be differing mechanisms and kinetics of anti-inflammatory action between the JAK inhibitors, which may instead inhibit many pathways, leading to non-upregulation and rapid symptom relief. While the biologics may have more targeted pathway inhibition, leading to progressive improvement and sustained benefit.
When comparing biologics and JAK inhibitors, an important consideration is their distinct safety profiles and the resulting patient selection and monitoring implications. JAK inhibitors carry boxed warnings defined by the FDA for serious infections, cardiovascular events, malignancy, and thrombosis[11]. It is important to note that these boxed warnings are primarily derived from safety signals observed in rheumatoid arthritis and other chronic inflammatory disease populations, and their absolute risk magnitude may differ in AD cohorts. Although these adverse events were uncommon in clinical trials of AD, they influence treatment algorithms due to their seriousness. The American Academy of Dermatology’s current guidelines advise considering the patient’s age, comorbidities, and cardiovascular risk when selecting a systemic therapeutic. Generally, biologics are recommended as first-line treatment unless contraindicated or ineffective[12]. JAK inhibitors are an excellent choice for patients who have not reacted well to biologics or who require rapid symptom relief. On the other hand, most patients with moderate-to-severe AD can use the biologics lebrikizumab and dupilumab as first-line treatments since they are safe and do not cause systemic immunosuppression[17,19]. The main tolerability issue with IL-13- and IL-4/IL-13-targeted biologics is conjunctivitis, affecting 8% to 15% of patients, which is usually mild and manageable with ophthalmologic care[17].
The data tool from this revised analysis affirms the competence of enrollment trial freedom from toxins in everyday clinical practice settings. The real-world response rates (EASI 75: 65-78; IGA 0/1: 58-72 at 16-24 weeks) were somewhat less than those observed in registration trials (EASI 75: About 70; IGA 0/1: About 43 at 16 weeks). These differences likely reflect variations in study design, patient selection criteria, baseline disease severity, adherence patterns, and duration of follow-up between controlled phase III trials and real-world cohorts. Real-world populations are typically more heterogeneous and may include patients with prior biologic exposure, multiple comorbidities, or concomitant therapies, which can influence observed effectiveness rates. Nevertheless, both sets of trials showed a large clinical benefit[2,4-6,10]. The efficacy-effectiveness gap is, to some extent, to be expected. Registration trial populations differ from real-world populations in that the latter have more comorbidities, more concurrent medications, and less well-controlled treatment adherence[20]. Not surprisingly, lebrikizumab was effective in those who had previously been exposed to a biologic or a JAK inhibitor[5,6]. In the Italian, United Kingdom, and Japanese studies, RWE trials were conducted in patients with diverse treatment histories and comorbidities, thereby ensuring broad applicability. Discontinuation rates due to adverse events in real-world settings (approximately 8%) are low, suggestive of the transferability of safety findings from trials to acceptable tolerability in practice[4-6].
The data from the two-year extension provide important evidence on long-term effectiveness and safety[1]. AD is a long-term disease, but registration trials often report data only through 16-52 weeks. The ADjoin study showed that among week 16 responders continuing lebrikizumab, about 80%-85% maintained EASI 75 responses and 75%-77% maintained IGA 0/1 responses for 2 years[1]. The maintenance rates are in line with long-term dupilumab trial data and support the durability of lebrikizumab’s treatment effect[8]. Results showing that response can be maintained with a dosing interval extension to every four weeks in responders have practical implications for treatment burden and cost. The idea that lebrikizumab had disease-modifying effects with maintained benefit after treatment discontinuation is supported by the observation that over half of the patients randomized to placebo appeared to retain response through week 52 after establishing response at week 16[1]. In contrast to dupilumab and tralokinumab, most patients in the maintenance trials relapsed after treatment withdrawal[8,9]. The mechanism underlying this potential disease-modifying effect of lebrikizumab remains to be investigated, potentially involving high-affinity binding and prolonged receptor occupancy[12].
The Japanese study’s data on patient-reported outcomes provide valuable information to complement physician-assessed efficacy measures[3]. Significant improvements in quality of life, illness burden, and sleep quality were linked to changes in EASI and IGA, indicating that clinical efficacy translates into significant patient-perceived benefit[3]. According to the patient, quality of life and symptom burden are the most important treatment goals. Confidence in the clinical relevance of lebrikizumab’s efficacy increases when clinical and patient-reported outcomes are consistent[2,20].
The systematic review has some limitations worth discussing. To begin with, the conclusions regarding the com
Even with the limitations of a systematic review, its strengths allow for confidence. A thorough, comprehensive literature search across multiple databases and other sources, with well-defined and reproducible eligibility criteria, reduces the risk of selection bias[22]. The process of screening was reliable and reproducible because the inter-reviewer agreement for selection of the studies was high (κ = 0.89-0.91)[23]. Errors are reduced when independent teams perform duplicate data extraction and analysis to evaluate the study’s quality. For the included randomized trials, the Cochrane assessment of risk of bias was minimal. This means they show internal validity. The GRADE assessment of quality ensured transparency in the certainty judgments, and most of these comparisons were supported by moderate or high-quality evidence. The consistent findings in trials assessing benefits for registration, RWE, and long-term extension data increase the confidence in their conclusions. The aggregation of 28 studies relating to 11000 patients creates a solid evidence base with adequate power to detect clinically meaningful differences between treatments.
The outcome of this systematic review is of great significance for clinical practice and treatment guidelines. Lebrikizumab, which is more effective than tralokinumab and comparable to dupilumab, represents a reasonable first-line biologic option for patients with moderate-to-severe AD, although treatment selection should also consider cost, access, reimbursement policies, comorbidities, and patient preference[7,10,15]. The majority of appropriate patients are now eligible for initiation of biologic therapy with this drug, given its favorable safety profile and lack of systemic immunosuppression or boxed warnings[17,19]. The opportunity to extend the dosing interval to every 4 weeks after attaining response may reduce the treatment burden and costs of maintenance therapy[1]. Lebrikizumab is a vital choice for people who do not adequately respond to or are intolerant of other biologics. Real-world data suggest that lebrikizumab remains effective even after prior biologic exposure[5,6]. The efficacy and safety profile of lebrikizumab supports its place within treatment algorithms, with dupilumab as a first-line biologic, and JAK inhibitors more generally placed as second line or selected first line (for example, when rapid onset is a high priority or biologics are contraindicated). Current guidelines support this[11]. Within this context, patient preferences, comorbidities, cost, and prior treatment should all be considered when making treatment decisions.
Several areas will need to be the focus of future research. To provide definitive evidence of comparative efficacy and eliminate uncertainty from indirect comparisons, direct head-to-head randomized trials comparing lebrikizumab with dupilumab and other biologics should be conducted[7]. Trials should measure not just the standard efficacy endpoints but also comparative quality of life, treatment satisfaction, and cost-effectiveness. In the second case, longer-duration studies beyond 2 years are required to more completely characterize the durability of response, safety with continued exposure, and optimal maintenance dosing strategies[1,20]. Disease registries with multi-year follow-up would further trial data for long-term outcome assessment. A personalized approach to treatment selection could be made possible through research to identify predictive biomarkers of treatment response[5]. Prospective candidates include baseline levels of IL-13 (eosinophils, immunoglobulin E, and type 2 inflammation biomarkers). However, none have yet been validated[4]. Fourth, evidence from studies assessing the use of lebrikizumab in specific patient groups under-represented in registration trials (for example, patients with severe comorbidities, elderly patients, pregnant women, and diverse racial and ethnic groups) would strengthen RWE for decision-making[2,10]. The fifth reason is that, as shown in the study mentioned above, cost-effectiveness analyses that include acquisition, administration, monitoring, and outcomes are very useful in informing healthcare policy and formulary decisions[21]. Evidence from mechanistic studies explaining the basis for lebrikizumab’s possible durable effect after treatment discontinuation may illuminate disease mechanisms and treatment[1,11].
Lebrikizumab is an effective biologic treatment for moderate to severe AD, according to a systematic analysis com
A recent systematic evaluation found that lebrikizumab is effective as a systemic treatment for moderate-to-severe AD. The evidence from the original network meta-analysis is significantly strengthened by the addition of 28 investigations, including 10847 injected patient data and six 2024-2025 studies. The methodological rigor of the review was high with substantial inter-reviewer agreement (Cohen’s κ = 0.89-0.91) and a comprehensive GRADE quality assessment. The evidence across key comparisons is of moderate-to-high quality but is hampered by indirectness due to network meta-analysis. We can be reasonably confident about this comparative effectiveness conclusion.
The main results confirm that lebrikizumab 250 mg Q2W has comparable efficacy to dupilumab 300 mg Q2W, the current first-in-class biologic standard of care, across a number of validated outcome measures including IGA 0/1 response, EASI up-risings and pruritus relief at 12-16 weeks. The tralokinumab, the second anti-IL-13 monoclonal antibody that was approved, is clearly inferior to lebrikizumab, presumably due to its higher affinity and slower disassociation. Lebrikizumab is more effective than baricitinib, similarly effective as abrocitinib and upadacitinib 15 mg, while only experiencing an inferior response rate to upadacitinib 30 mg. The favorable safety profile of lebrikizumab as evidenced by manageable injection site reactions and conjunctivitis, without systemic immunosuppression, and serious cardiovascular effects is a marked improvement over the boxed warnings associated with JAK inhibitors.
Efficacy data from Italy, the United Kingdom, and Japan confirm that the findings of registration trials for various IL-17A blockers are consistent with routine clinical practice. EASI 75 response rates ranged from 65%-78%, and IGA 0/1 rates were 58%-72% at 16-24 weeks. These studies show effectiveness in a range of individuals, including those who have previously used JAK inhibitors or biologics. The two-year extension data show that over 80% of responders maintain their response. There is also evidence that extension of dosing intervals to every 4 weeks is feasible. These data address long-term management issues of the chronic disease. Clinical efficacy also results in significant improvements in quality of life and a decrease in symptom burden, as indicated by patient-reported outcomes.
The clinical implications of lebrikizumab place it as a very attractive first-line biologic option with dupilumab for patients with moderate-to-severe AD requiring systemic therapy. When selecting a treatment, the patient's preference, comorbidities, prior treatment responses, and costs should be considered. The future research priorities include undertaking a head-to-head randomized trial pitting lebrikizumab against dupilumab and other biologics to eliminate uncertainty from indirect comparisons, and longer-duration studies beyond 2 years to fully characterize durability and safety. Research should also identify predictive biomarkers to enable personalized treatment selection, as well as conduct comparative cost-effectiveness analyses to inform health policy. This systematic review provides high-level evidence due to its thorough methodology, which includes inter-reviewer agreement assessment and GRADE evaluation, and addresses the clinical question as per contemporary standards. As a result, lebrikizumab is recognized as a significant treatment development for moderate-to-severe AD, allowing for evidence-based clinical judgements.
| 1. | Guttman-Yassky E, Irvine A, Simpson E, Gooderham M, Weidinger S, Spelman L, Silverberg J, Crane H, Elmaraghy H, Deluca-Carter L, Buziqui Piruzeli ML, Hu C, Pierce E, Agell H, Thaçi D. Long-Term Efficacy and Safety of Lebrikizumab Is Maintained in Patients With Moderate-to-Severe Atopic Dermatitis: Results Up to 3 Years From ADjoin. Skin J. 2025;9:s510. [DOI] [Full Text] |
| 2. | Warren RB, de Bruin-Weller M, Tsianakas A, Khemis A, Szepietowski JC, Hong HC, Mihaylov Y, Falqués M, Agell H, Massana E, Gil EG, Coll R, Weidinger S. Efficacy and safety of lebrikizumab in adult and adolescent patients with moderate-to-severe atopic dermatitis inadequately controlled with or ineligible for ciclosporin A: a placebo-controlled, randomized phase IIIb clinical study (ADvantage). Br J Dermatol. 2025;193:876-888. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 3. | Tanaka A, Igawa K, Takahashi H, Shimizu R, Kataoka Y, Torisu-Itakura H, Morisaki Y, Montmayeur S, Katoh N. Lebrikizumab Combined with Topical Corticosteroids Improves Patient-reported Outcomes in Japanese Patients with Moderate-to-severe Atopic Dermatitis. Acta Derm Venereol. 2024;104:adv34375. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 14] [Reference Citation Analysis (1)] |
| 4. | Avallone G, Bombelli A, Gargiulo L, Foti C, Trave I, Rossi M, Magnaterra E, Pigliacelli F, Malagoli P, Giacalone S, Portarapillo A, Patruno C, Schettini N, Bonzano L, Pezzolo E, Chello C, Gori N, Brunasso G, Veronese F, Ortoncelli M, Tolino E, Azzolini A, Bellinato F, Hansel K, Di Brizzi EV, Gurioli C, Esposito M, Giuffrida R, Barei F, Calzari P, Burroni AG, Gola M, Mariano M, Fargnoli MC, Napolitano M, Grieco T, Peris K, Savoia P, Ribero S, Girolomoni G, Stingeni L, Balato A, Costanzo A, Marzano AV, Narcisi A, Ferrucci SM. Effectiveness, Safety, and Health-Related Quality of Life in Moderate-to-Severe Atopic Dermatitis Treated with Lebrikizumab: A 16-Week Nationwide Retrospective Cohort Study. Dermatol Ther (Heidelb). 2025;15:3671-3688. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 5. | Abraheem A, Sebastian N, Kreeshan F, Clayton T, Hunter HA, Warren R. P109 Lebrikizumab for the treatment of moderate-to-severe atopic eczema: real-world experience from a tertiary centre. Brit J Dermatol. 2025;193:ljaf085.137. [DOI] [Full Text] |
| 6. | Hagino T, Uchiyama A, Onda M, Kosaka K, Araki T, Motegi SI, Saeki H, Fujimoto E, Kanda N. 24-Week Real-World Effectiveness and Safety of Lebrikizumab for Atopic Dermatitis in Japan: An Analysis Stratified by Prior Systemic Therapy. Dermatitis. 2025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 7. | Silverberg JI, Bieber T, Paller AS, Beck L, Kamata M, Puig L, Wiseman M, Ezzedine K, Irvine AD, Foley P, Del Rosso J, Gold LS, Johansson E, Dossenbach M, Gallo G, Akmaz B, Casillas M, Karlsson A, Curteis T, Chovatiya R. Lebrikizumab vs Other Systemic Monotherapies for Moderate-to-Severe Atopic Dermatitis: Network Meta-analysis of Efficacy. Dermatol Ther (Heidelb). 2025;15:615-633. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (1)] |
| 8. | Wollenberg A, Barbarot S, Bieber T, Christen-Zaech S, Deleuran M, Fink-Wagner A, Gieler U, Girolomoni G, Lau S, Muraro A, Czarnecka-Operacz M, Schäfer T, Schmid-Grendelmeier P, Simon D, Szalai Z, Szepietowski JC, Taïeb A, Torrelo A, Werfel T, Ring J; European Dermatology Forum (EDF), the European Academy of Dermatology and Venereology (EADV), the European Academy of Allergy and Clinical Immunology (EAACI), the European Task Force on Atopic Dermatitis (ETFAD), European Federation of Allergy and Airways Diseases Patients’ Associations (EFA), the European Society for Dermatology and Psychiatry (ESDaP), the European Society of Pediatric Dermatology (ESPD), Global Allergy and Asthma European Network (GA2LEN) and the European Union of Medical Specialists (UEMS). Consensus-based European guidelines for treatment of atopic eczema (atopic dermatitis) in adults and children: part I. J Eur Acad Dermatol Venereol. 2018;32:657-682. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 410] [Cited by in RCA: 632] [Article Influence: 79.0] [Reference Citation Analysis (3)] |
| 9. | Langan SM, Irvine AD, Weidinger S. Atopic dermatitis. Lancet. 2020;396:345-360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1604] [Cited by in RCA: 1345] [Article Influence: 224.2] [Reference Citation Analysis (4)] |
| 10. | Silverberg JI, Gelfand JM, Margolis DJ, Boguniewicz M, Fonacier L, Grayson MH, Simpson EL, Ong PY, Chiesa Fuxench ZC. Patient burden and quality of life in atopic dermatitis in US adults: A population-based cross-sectional study. Ann Allergy Asthma Immunol. 2018;121:340-347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 221] [Cited by in RCA: 500] [Article Influence: 62.5] [Reference Citation Analysis (1)] |
| 11. | Administration UFaD. Rinvoq (upadacitinib) prescribing information. United States, 2023. |
| 12. | Davis DMR, Drucker AM, Alikhan A, Bercovitch L, Cohen DE, Darr JM, Eichenfield LF, Frazer-Green L, Paller AS, Schwarzenberger K, Silverberg JI, Singh AM, Wu PA, Sidbury R. Guidelines of care for the management of atopic dermatitis in adults with phototherapy and systemic therapies. J Am Acad Dermatol. 2024;90:e43-e56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 161] [Article Influence: 80.5] [Reference Citation Analysis (0)] |
| 13. | Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P, Schünemann HJ; GRADE Working Group. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336:924-926. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17839] [Cited by in RCA: 16616] [Article Influence: 923.1] [Reference Citation Analysis (4)] |
| 14. | Vieira SM, Kaymak U, Sousa JMC. Cohen's kappa coefficient as a performance measure for feature selection. International Conference on Fuzzy Systems; 2010 Jul 18-23; Barcelona, Spain. IEEE, 2010; 1-8. [DOI] [Full Text] |
| 15. | Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA. Cochrane handbook for systematic reviews of interventions. 2nd ed. Wiley, 2019. [RCA] [DOI] [Full Text] [Cited by in Crossref: 10405] [Cited by in RCA: 8200] [Article Influence: 1171.4] [Reference Citation Analysis (3)] |
| 16. | Wells GA, Shea B, O'Connell D, Peterson J, Welch V, Losos M, Tugwell P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Non-Randomised Studies in Meta-Analyses. 2000. Available from: https://ohri.ca/en/who-we-are/core-facilities-and-platforms/ottawa-methods-centre/newcastle-ottawa-scale. |
| 17. | Ferreira S, Torres T. Conjunctivitis in patients with atopic dermatitis treated with dupilumab. Drugs Context. 2020;9:2020-2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 18. | Wollenberg A, Blauvelt A, Guttman-Yassky E, Worm M, Lynde C, Lacour JP, Spelman L, Katoh N, Saeki H, Poulin Y, Lesiak A, Kircik L, Cho SH, Herranz P, Cork MJ, Peris K, Steffensen LA, Bang B, Kuznetsova A, Jensen TN, Østerdal ML, Simpson EL; ECZTRA 1 and ECZTRA 2 study investigators. Tralokinumab for moderate-to-severe atopic dermatitis: results from two 52-week, randomized, double-blind, multicentre, placebo-controlled phase III trials (ECZTRA 1 and ECZTRA 2). Br J Dermatol. 2021;184:437-449. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 105] [Cited by in RCA: 452] [Article Influence: 75.3] [Reference Citation Analysis (1)] |
| 19. | Stein Gold L, Thaçi D, Thyssen JP, Gooderham M, Laquer V, Moore A, Natalie CR, Zhao F, Meskimen E, Elmaraghy H, Montmayeur S, Gallo G, Jimenez G, de Bruin-Weller M. Safety of Lebrikizumab in Adults and Adolescents with Moderate-to-Severe Atopic Dermatitis: An Integrated Analysis of Eight Clinical Trials. Am J Clin Dermatol. 2023;24:595-607. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 46] [Article Influence: 15.3] [Reference Citation Analysis (1)] |
| 20. | Silverberg JI, Guttman-Yassky E, Thaçi D, Irvine AD, Stein Gold L, Blauvelt A, Simpson EL, Chu CY, Liu Z, Gontijo Lima R, Pillai SG, Seneschal J; ADvocate1 and ADvocate2 Investigators. Two Phase 3 Trials of Lebrikizumab for Moderate-to-Severe Atopic Dermatitis. N Engl J Med. 2023;388:1080-1091. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 224] [Article Influence: 74.7] [Reference Citation Analysis (1)] |
| 21. | Blauvelt A, Teixeira HD, Simpson EL, Costanzo A, De Bruin-Weller M, Barbarot S, Prajapati VH, Lio P, Hu X, Wu T, Liu J, Ladizinski B, Chu AD, Eyerich K. Efficacy and Safety of Upadacitinib vs Dupilumab in Adults With Moderate-to-Severe Atopic Dermatitis: A Randomized Clinical Trial. JAMA Dermatol. 2021;157:1047-1055. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 375] [Cited by in RCA: 383] [Article Influence: 76.6] [Reference Citation Analysis (1)] |
| 22. | Bieber T. Atopic dermatitis: an expanding therapeutic pipeline for a complex disease. Nat Rev Drug Discov. 2022;21:21-40. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 422] [Article Influence: 84.4] [Reference Citation Analysis (0)] |
| 23. | Cohen J. A Coefficient of Agreement for Nominal Scales. Educ Psychol Meas. 1960;20:37-46. [DOI] [Full Text] |
| 24. | Landis JR, Koch GG. The measurement of observer agreement for categorical data. Biometrics. 1977;33:159-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60504] [Cited by in RCA: 42873] [Article Influence: 875.0] [Reference Citation Analysis (4)] |
| 25. | Simpson EL, Bieber T, Guttman-Yassky E, Beck LA, Blauvelt A, Cork MJ, Silverberg JI, Deleuran M, Kataoka Y, Lacour JP, Kingo K, Worm M, Poulin Y, Wollenberg A, Soo Y, Graham NM, Pirozzi G, Akinlade B, Staudinger H, Mastey V, Eckert L, Gadkari A, Stahl N, Yancopoulos GD, Ardeleanu M; SOLO 1 and SOLO 2 Investigators. Two Phase 3 Trials of Dupilumab versus Placebo in Atopic Dermatitis. N Engl J Med. 2016;375:2335-2348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1787] [Cited by in RCA: 1552] [Article Influence: 155.2] [Reference Citation Analysis (4)] |