Published online Apr 26, 2026. doi: 10.12998/wjcc.v14.i12.119292
Revised: February 15, 2026
Accepted: March 20, 2026
Published online: April 26, 2026
Processing time: 82 Days and 5.6 Hours
Retroperitoneal fibrosis (RPF) is a rare fibro-inflammatory disorder, classically affecting middle-aged Caucasian men, and is characterized by the development of dense fibrotic tissue surrounding the abdominal aorta and adjacent retroperitoneal structures. Most cases are idiopathic or secondary to medications, infec
A 73-year-old woman presented with recurrent urinary tract infections and pro
This case highlights the need to consider occult malignancy in patients with extensive or refractory RPF. Timely imaging and histopathologic evaluation are essential to avoid diagnostic delay and ensure appropriate management, particularly when clinical or radiologic features are atypical.
Core Tip: Retroperitoneal fibrosis is a rare fibroinflammatory disorder that typically involves the infrarenal aorta and ureters, leading to insidious symptoms such as abdominal or back pain, constitutional features, and obstructive uropathy. Laboratory findings are often nonspecific, with elevated inflammatory markers and occasional autoimmune serologies. Cross-sectional imaging with computed tomography or magnetic resonance imaging is essential for diagnosis and for guiding biopsy to exclude malignancy. Glucocorticoids remain the cornerstone of therapy, with immunomodulatory agents reserved for refractory or relapsing disease. Long-term follow-up is critical due to high relapse rates despite treatment.
- Citation: Arora K, Tipmongkol S, Goyal MK, Matt-Amaral L. Retroperitoneal fibrosis mimicking benign disease - a paraneoplastic clue to small bowel adenocarcinoma: A case report. World J Clin Cases 2026; 14(12): 119292
- URL: https://www.wjgnet.com/2307-8960/full/v14/i12/119292.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i12.119292
Retroperitoneal fibrosis (RPF) is a rare condition characterized by the formation of fibroinflammatory tissue around the infrarenal aorta, iliac vessels, and ureters[1]. It occurs in approximately 1.3/100000, with a higher prevalence, nearly threefold in men aged 60 and above[1]. This data largely comes from Europe, and prevalence may vary by region. Although the exact cause remains unknown, it is proposed that the condition begins with an inflammatory process that activates fibroblasts, leading to the deposition of excessive fibromatous tissue[1]. This inflammatory insult can be related to toxins such as asbestos or smoke, drugs like ergots, or associated with malignancies, mostly gastric or breast cancer, and rarely hematologic neoplasms1. Recently, many cases previously identified as idiopathic have been associated with immunoglobulin G4 (IgG4)-related disease[1].
The accumulation of fibrotic tissue can compress retroperitoneal structures, causing vague symptoms like abdominal and back pain, fever, malaise, and weight loss[1]. Workup generally reveals renal insufficiency due to ureteral ob
Diagnosis of RPF can be challenging due to the ambiguous nature of the presenting symptoms, and it is crucial to determine whether the fibrosis is due to a malignant or benign etiology. Computed tomography (CT) and magnetic resonance imaging provide the extent of involvement of the disease and may help guide the best location for biopsy[1]. Histopathologic examination remains the most reliable, as it can help diagnose any underlying causes of fibrosis and help guide treatment, with the most common presentation being fibroblasts with lymphocytic infiltration[1].
In this case report, we highlight a case that presented as recurrent urinary tract infections (UTIs) in a female, with initial biopsy showing benign disease. When secondary workup was negative, repeat biopsy was pursued, which showed metastatic small bowel adenocarcinoma, in contrast to the commonly seen carcinomas, such as breast or gastric cancer. Small bowel adenocarcinoma is a rare malignancy in itself, and its association with RPF is reported infrequently, making this an uncommon but important diagnostic consideration. We will highlight the difficulties encountered in the diagnosis and management of this case report.
A 73-year-old female presented to the emergency department (ED) with fever, chills, nausea, vomiting, upper abdominal discomfort and productive cough for 2 days.
Symptoms started 2 days ago after a breast biopsy.
A 73-year-old female presented to the ED with fever, chills, nausea, vomiting, upper abdominal discomfort and productive cough for 2 days before admission. She underwent a right breast stereotactic biopsy a few days prior for a new nonpalpable density in the upper outer quadrant of the right breast and reported that the symptoms began after the procedure. There was no shortness of breath or chest pain, but there was a history of recurrent UTI. She had notable lower abdominal pain with cramps for 2-3 weeks before this episode and had lost 16 pounds over the past year.
She had a history of past medical history of diabetes (on insulin), hypertension (on lisinopril), hyperlipidemia (on simvastatin), hypothyroidism (on levothyroxine), renal stones and glaucoma. Family history was significant for breast cancer in her sisters and brain cancer in her brother.
In the ED, she was febrile, tachycardic, and hypertensive. On examination, she was not in any acute distress; her abdomen was non-distended, but abdominal tenderness was present in the left and right lower quadrants and the suprapubic area.
Initial labs were significant for white blood cell (WBC) 14.42 k/μL with absolute neutrophil count 12.46 k/μL, platelets 433 k/μL, hemoglobin 11.1 g/dL, albumin 2.8 g/dL, glucose 242 mg/dL, blood urea nitrogen/creatinine 20/1.26 mg/dL, estimated glomerular filtration rate 59 mL/minute/1.73 m2. Urinalysis was positive for glucose, hemoglobin, protein, nitrites, leukocyte esterase, WBC, red blood cell, and bacteria (Table 1).
| CMP | Admission | Day 2 | Day 3 | Day 4 | Day 5 | Day 6 |
| BUN | 20 | 17 | 14 | 15 | 14 | 16 |
| Creatinine | 1.26 | 1.2 | 1.42 | 1.46 | 1.38 | 1.24 |
| eGFR | 59 | 48 | 39 | 38 | 40 | 46 |
| CBC | ||||||
| Hemoglobin | 11.1 | 10 | 9.7 | 10 | 9.9 | 11.3 |
| Hematocrit | 33.4 | 31 | 30.3 | 30.1 | 31.2 | 36 |
| WBC | 14.42 | 13.84 | 8.29 | 8.15 | 5.93 | 6.48 |
| Platelets | 433 | 427 | 386 | 423 | 423 | 466 |
| Immunoglobulins | ||||||
| IgG | 407 | |||||
| IgG class 1 | 196.5 | |||||
| IgG class 2 | 131.8 | |||||
| IgG class 3 | 40.4 | |||||
| IgG class 4 | 9.4 | |||||
CT abdomen/pelvis showed extensive findings which included - ill-defined soft tissue infiltration throughout the upper abdominal periaortic retroperitoneum associated with obstruction of the right ureter at the uretero-pelvic junction, and associated hydroureteronephrosis. There was significant atrophy of the left kidney with possible renal artery stenosis. In addition, there was continuity with the pancreas and extension throughout the mesenteric base with encasement of the distal duodenum. Associated regional lymphadenopathy was seen. These findings were suggestive of the spectrum of RPF/IgG4-related disease. Additionally, duodenal regional gas and fluid collections, evidence of pancreatic atrophy, biliary ductal dilation, left renal artery stenosis, and some hypodensities in the liver were seen.
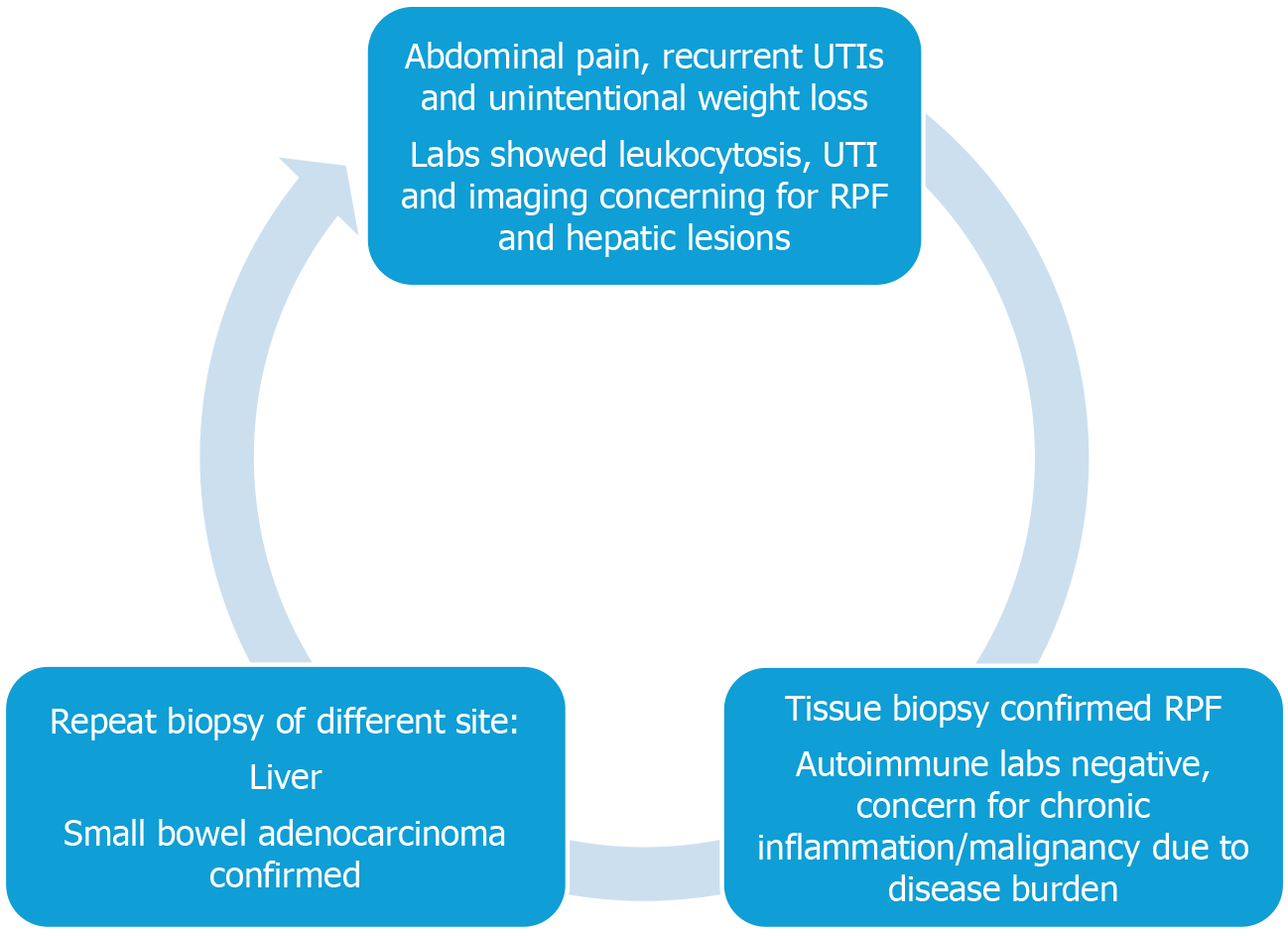
Percutaneous nephrostomy tubes were placed in the setting of RPF with hydronephrosis and, eventually, stent placement was done outpatient. Biopsy confirmed the diagnosis of RPF without any evidence of malignancy. Workup with IgG for ruling out an autoimmune cause of idiopathic RPF was unrevealing. The patient was stable and was subsequently discharged home.
The diagnosis was particularly challenging given nonspecific systemic symptoms, recurrent UTIs, weight loss, and although the imaging features were suggestive of benign fibroinflammatory disease, lymphadenopathy was concerning for lymphoma, metastatic solid organ malignancy, and infectious processes like tuberculosis and chronic bacterial or fungal infections in the setting of an immunocompromised state due to diabetes. Initial biopsy confirmed RPF without evidence of malignancy, and autoimmune workup was unrevealing, further supporting an idiopathic or IgG4-related process.
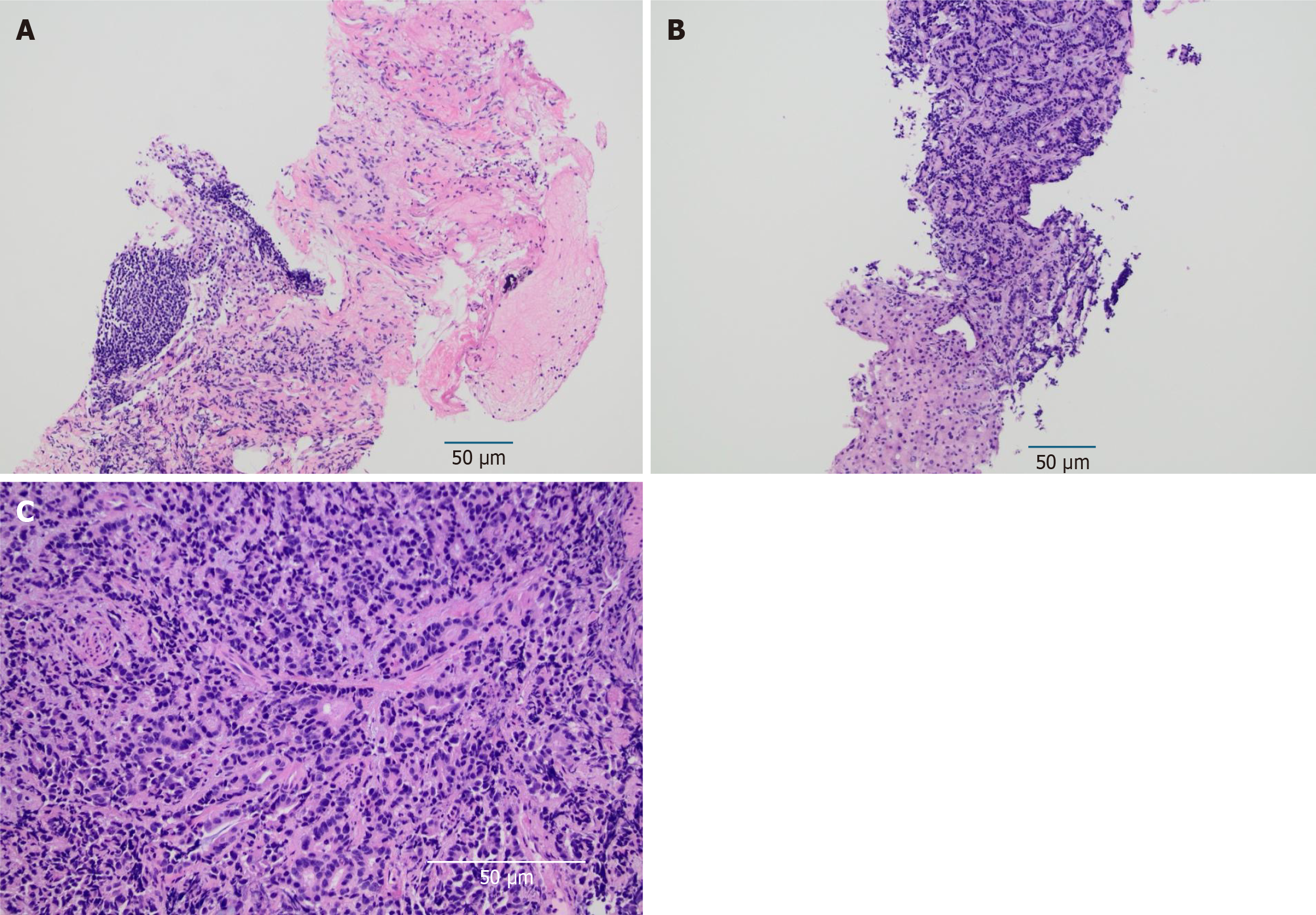
However, the extensive nature of the disease, regional lymphadenopathy, and hepatic lesions prompted further evaluation, and imaging-guided biopsy of the liver was performed. This revealed metastatic adenocarcinoma, with CK20, CDX2, and claudin 4 stain positivity, suggesting origin from a gastrointestinal primary. Cancer type ID resulted as 84% intestine, 83% small bowel (Figure 1).
The patient was diagnosed with RPF secondary to small bowel adenocarcinoma (Figure 2).
The patient received folinic acid, fluorouracil, and oxaliplatin chemotherapy outpatient.
There was a decrease in the size of the hepatic metastasis, a minimal increase in RPF, and stable biliary duct dilation, but there were significant side effects from the chemotherapy. Ultimately, the patient decided to undergo hospice and passed away (Table 2).
| Timeframe | Clinical events |
| Approximately 1 year prior to admission | Unintentional weight loss (16 pounds), abdominal discomfort and UTIs |
| 2-3 weeks prior to admission | Worsening lower abdominal pain and cramping |
| Few days prior to admission | Underwent right breast stereotactic biopsy for nonpalpable breast lesion |
| Hospitalization | Fever, chills, nausea, vomiting, abdominal discomfort, and productive cough × 2 days |
| Labs: Leukocytosis with neutrophilia; abnormal urinalysis consistent with UTI | |
| Imaging: Retroperitoneal soft tissue infiltration, hydroureteronephrosis, lymphadenopathy, and hepatic lesions | |
| Management: PNT placement and supportive care | |
| Biopsy: Fibroinflammatory tissue consistent with retroperitoneal fibrosis | |
| RPF workup: IgG4 and autoimmune serologies negative | |
| 4 months after discharge | Image-guided liver biopsy due to extensive disease and hepatic lesions |
| Final diagnosis | Metastatic small bowel adenocarcinoma causing secondary retroperitoneal fibrosis |
| Treatment | Initiation of FOLFOX chemotherapy |
| Follow-up | Partial radiographic response of hepatic metastases; progression of symptoms and chemotherapy-related toxicity |
| Outcome | Transition to hospice care and subsequent death |
The clinical presentation of RPF is often vague, depending on the extent and location of involvement, complicating the diagnosis of the disease. The higher acuity and concern for malignancy may mask the actual underlying diagnosis of RPF[2]. It may also present atypically with lower gastrointestinal bleeding, especially in the setting of IgG4-related disease[3]. The common symptoms of urinary involvement reported in cases include flank and abdominal pain, hematuria, and associated hydronephrosis or hydrocele, requiring interventions such as stent placement[1]. Spontaneous remission, although rare, has been reported[4]. Although uncommon, it is important to consider other symptoms, such as recurrent UTIs, as seen in our patient. In this case, urinary tract obstruction caused by RPF may have led to the patient's recurrent UTIs as an early presenting symptom.
This is further complicated by non-specific lab markers, such as elevation of ESR and CRP, which are commonly seen but do not provide any guidance in treatment[1]. Autoimmune labs, such as anti- nuclear antibodies, rheumatoid factor, anti-smooth muscle antibodies, and double-stranded DNA antibodies, are also sometimes positive[1]. Oftentimes, the titers of these antibodies are low and positivity can be nonspecific, but could also point toward another autoimmune process as the primary cause[1].
Steroids remain at the forefront of the treatment of RPF. While there are no consensus guidelines on steroid dosing, a randomized controlled trial by Vaglio et al[5] used 1 mg/kg for 1 month as induction therapy followed by a 0.5 mg/kg tapering dose. 36/40 patients on this induction regimen showed remission, and those receiving prednisone taper also showed low rates of relapse, compared to those receiving tamoxifen as maintenance therapy. A favorable response to steroids includes rapid symptomatic improvement in two weeks, improvement in renal function and markers of inflammation in 6 weeks, and eventual imaging documented regression in the size of the mass[6]. Such high steroid dosing also comes with side effects, with a need for exercising caution, especially in immunocompromised patients. A recent trial evaluating low-dose prednisone with methotrexate showed its non-inferiority compared to prednisone alone, paving the way for low steroid exposure and related side effects[7]. Tamoxifen monotherapy and the addition of mycophenolate mofetil or azathioprine to prednisone remain successful treatment options in steroid-refractory disease, while rituximab is favorable in the relapsed/refractory settings and IgG4-related disease[5,8,9]. Obstructive symptoms often require nephrostomy tubes or, rarely, surgical intervention[10].
Despite treatment, relapse rates remain high, with a risk of 21%, 41%, and 48% at 5 years, 10 years, and 15 years, respectively[11]. Males remain at a higher risk for relapse, along with ANA positivity and thoracic aorta involvement[11,12]. This necessitates follow-up imaging, like CT, at scheduled intervals, and when relapse is suspected[11].
Due to the rarity of RPF, maintaining a high suspicion and promptly initiating imaging and autoimmune testing remains of utmost importance for a timely diagnosis. In addition to these considerations, establishing standardized diagnostic and treatment protocols are crucial steps in improving prognosis of RPF.
RPF is a diagnostically challenging condition due to its nonspecific presentation and lack of standardized diagnostic criteria. While cross-sectional imaging is essential for initial evaluation, it may not reliably differentiate benign from malignancy-associated disease. Corticosteroids remain first-line therapy, but optimal management often requires multidisciplinary coordination. This case highlights the importance of maintaining suspicion for underlying malignancy in extensive or refractory RPF, even when the initial biopsy is benign. Persistent symptoms, atypical imaging features, or extra-retroperitoneal lesions should prompt reassessment and repeat tissue evaluation. Early multidisciplinary in
The authors would like to acknowledge Dr. Andrew Crabbe and Dr. Jason Lane of Department of Pathology, Cleveland Clinic Akron General, for help in the acquisition of pathology imaging and reports.
| 1. | Vaglio A, Salvarani C, Buzio C. Retroperitoneal fibrosis. Lancet. 2006;367:241-251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 584] [Cited by in RCA: 472] [Article Influence: 23.6] [Reference Citation Analysis (1)] |
| 2. | Altshuler E. Atypical Presentation of Idiopathic Retroperitoneal Fibrosis Effectively Treated With Colchicine After Lymphoma Misdiagnosis. Cureus. 2021;13:e14756. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 3. | Achrak E, Manville E, Ayyat M, Toribio RD. Atypical Presentation of Retroperitoneal Fibrosis Causing Colonic Obstruction: A Case Report. Cureus. 2024;16:e55621. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 4. | Kume H, Kitamura T. Spontaneous regression of bilateral hydronephrosis due to retroperitoneal fibrosis. Scand J Urol Nephrol. 2001;35:255-256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.2] [Reference Citation Analysis (1)] |
| 5. | Vaglio A, Palmisano A, Alberici F, Maggiore U, Ferretti S, Cobelli R, Ferrozzi F, Corradi D, Salvarani C, Buzio C. Prednisone versus tamoxifen in patients with idiopathic retroperitoneal fibrosis: an open-label randomised controlled trial. Lancet. 2011;378:338-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 155] [Cited by in RCA: 161] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 6. | van Bommel EF, Siemes C, Hak LE, van der Veer SJ, Hendriksz TR. Long-term renal and patient outcome in idiopathic retroperitoneal fibrosis treated with prednisone. Am J Kidney Dis. 2007;49:615-625. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 109] [Article Influence: 5.7] [Reference Citation Analysis (1)] |
| 7. | Peyronel F, Palmisano A, Maritati F, Alberici F, Urban ML, Gianfreda D, Rossi GM, Fenaroli P, Bettiol A, Moroni G, Vaglio A. Methotrexate and low-dose prednisone in idiopathic retroperitoneal fibrosis: a randomised clinical trial. J Autoimmun. 2025;157:103487. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 8. | van Bommel EF, Hendriksz TR, Huiskes AW, Zeegers AG. Brief communication: tamoxifen therapy for nonmalignant retroperitoneal fibrosis. Ann Intern Med. 2006;144:101-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 99] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 9. | Wallwork R, Wallace Z, Perugino C, Sharma A, Stone JH. Rituximab for idiopathic and IgG4-related retroperitoneal fibrosis. Medicine (Baltimore). 2018;97:e12631. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 36] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 10. | Raglianti V, Rossi GM, Vaglio A. Idiopathic retroperitoneal fibrosis: an update for nephrologists. Nephrol Dial Transplant. 2021;36:1773-1781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 43] [Article Influence: 7.2] [Reference Citation Analysis (1)] |
| 11. | Raffiotta F, da Silva Escoli R, Quaglini S, Rognoni C, Sacchi L, Binda V, Messa P, Moroni G. Idiopathic Retroperitoneal Fibrosis: Long-term Risk and Predictors of Relapse. Am J Kidney Dis. 2019;74:742-750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 24] [Article Influence: 3.4] [Reference Citation Analysis (2)] |
| 12. | Bond M, Bettiol A, Buttini EA, Trivioli G, Palazzini G, Fibbi I, Tesi M, Biancalana E, Dejaco C, Emmi G, Vaglio A. Predictors of remission and relapse in retroperitoneal fibrosis. J Intern Med. 2025;298:438-449. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |