Copyright: ©Author(s) 2026.
World J Clin Cases. Apr 26, 2026; 14(12): 119112
Published online Apr 26, 2026. doi: 10.12998/wjcc.v14.i12.119112
Published online Apr 26, 2026. doi: 10.12998/wjcc.v14.i12.119112
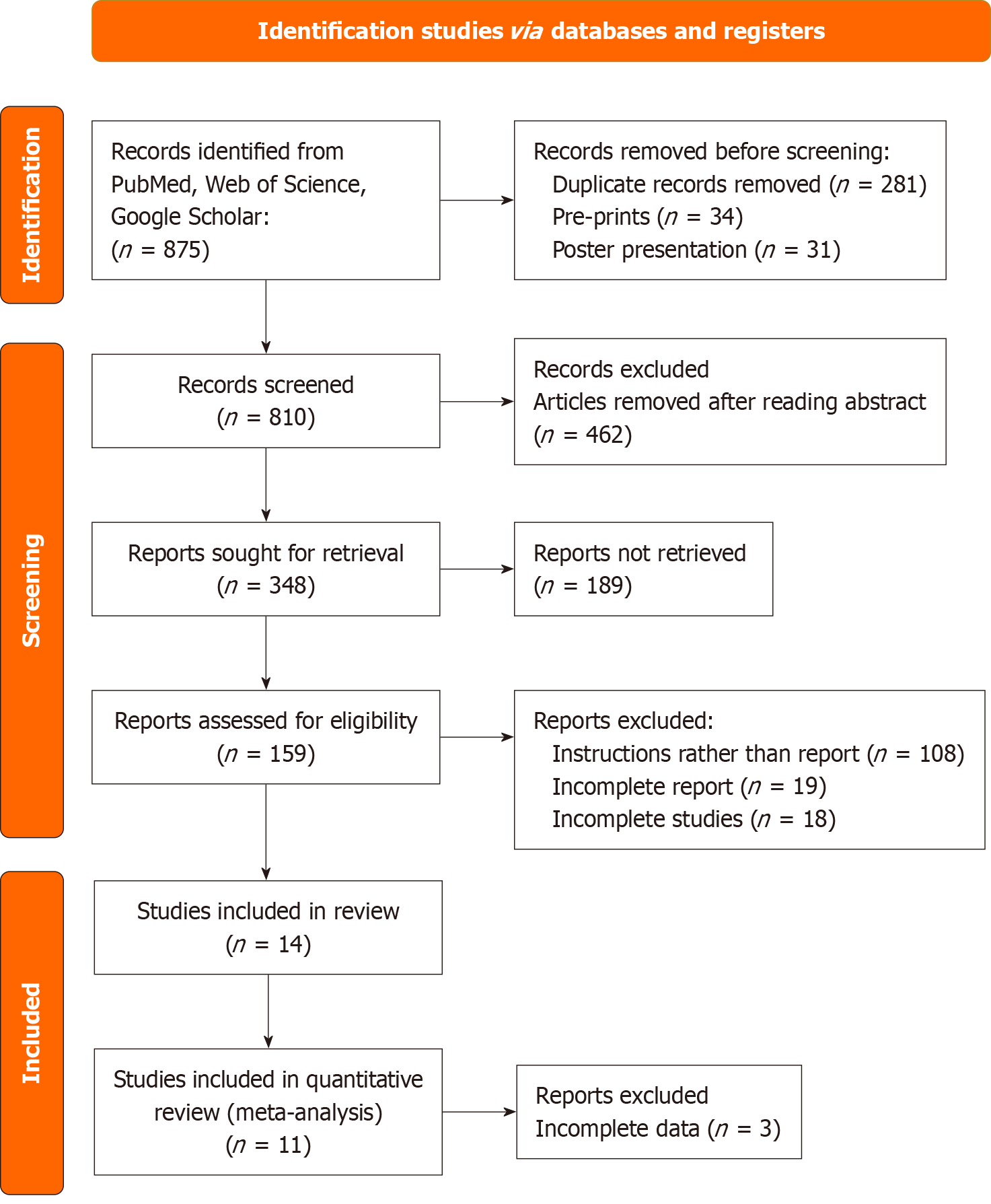
Figure 1 Preferred Reporting Items for Systematic Reviews and Meta-analyses flowchart for the review.
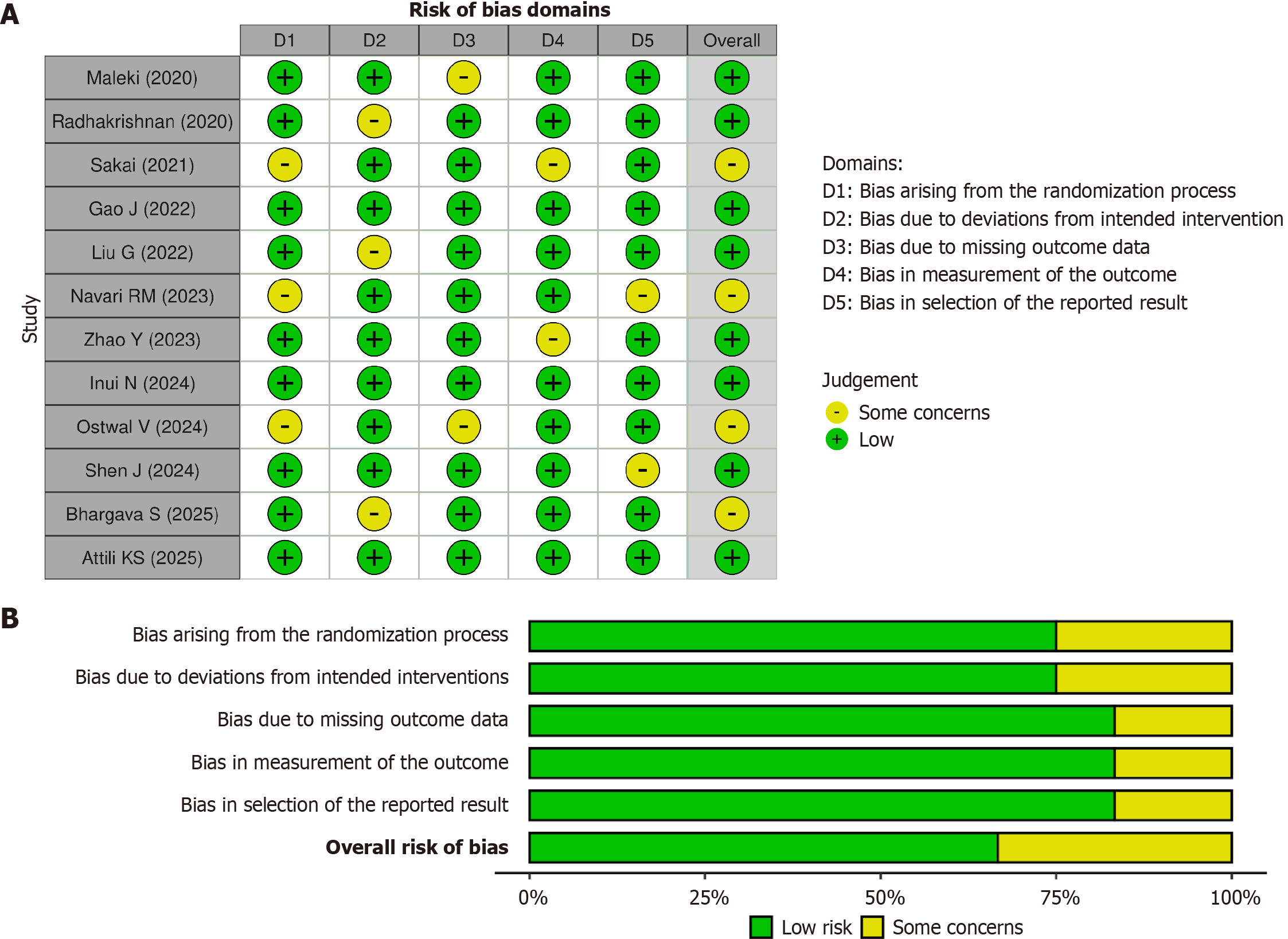
Figure 2 Risk of bias assessment for randomized control.
A: Individual studies using the risks of bias 2.0 tool; B: Overall studies using the risks of bias 2.0 tool.
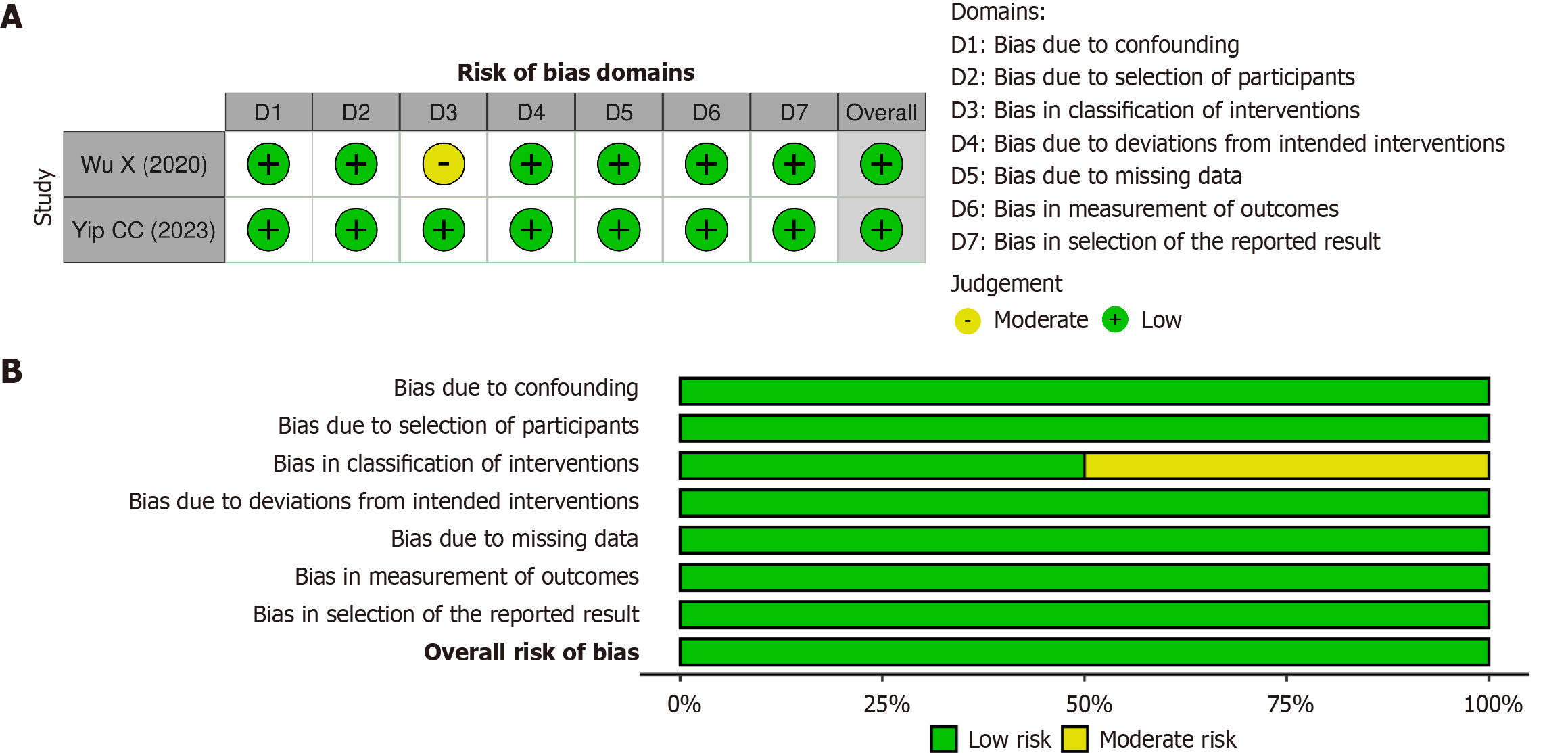
Figure 3 Risk of bias assessment for cohort.
A: Individual studies using the risk of bias in nonrandomized studies of interventions version I tool; B: Overall studies using the risk of bias in nonrandomized studies of interventions version I tool.
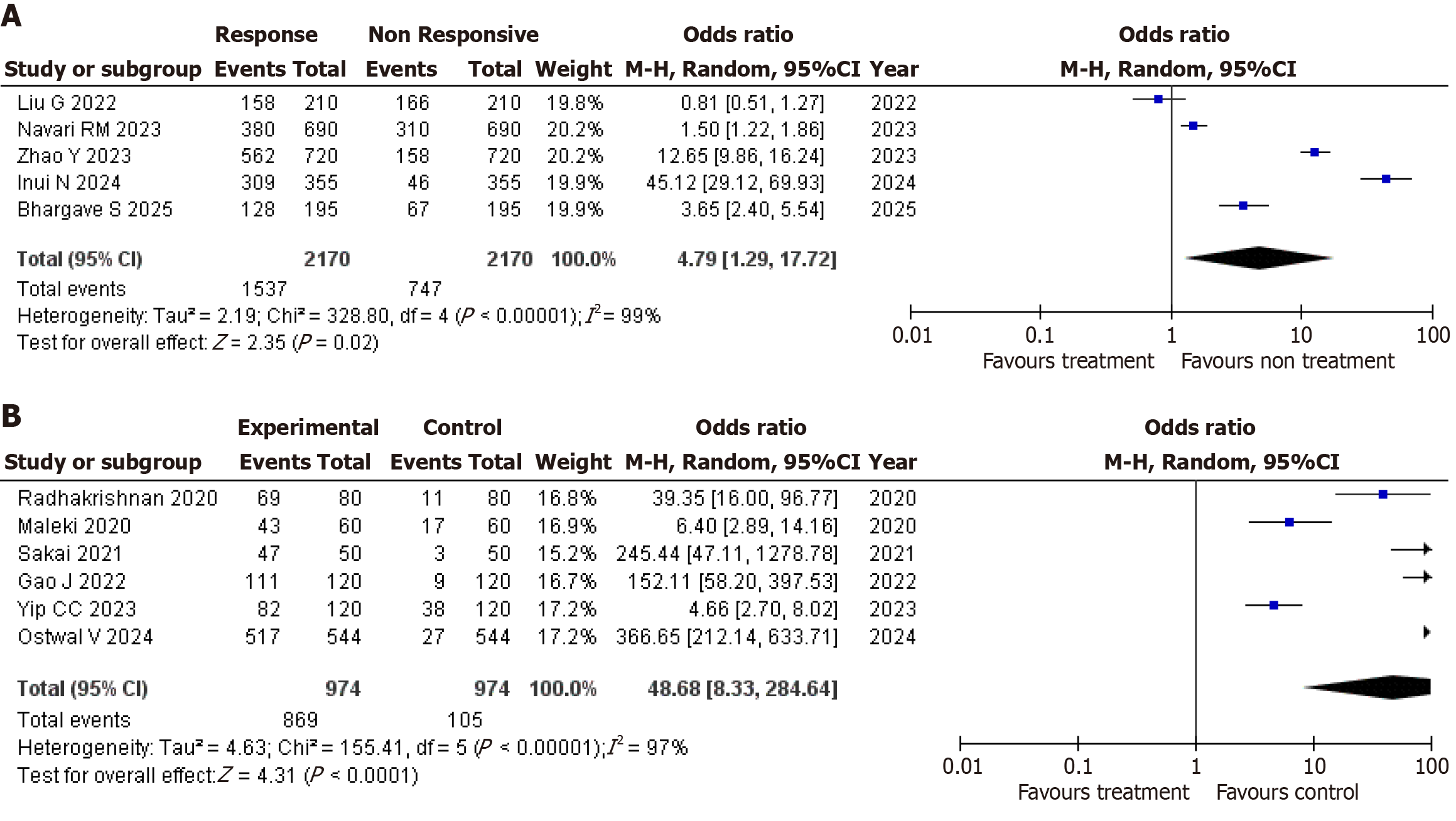
Figure 4 Forest plot.
A: Assessment comparing the effectiveness of antagonist antiemetic; B: Assessment comparing the mortality rate - antipsychotic drugs. CI: Confidence interval.
- Citation: Mv A, Anand N, Babu B, Awosika A, Udayan U, Mendoza J, Lopez A, Rangachari B, Maity S, Gadad BS. Olanzapine-based vs neurokinin-1 receptor antagonist antiemetic regimens in highly emetogenic chemotherapy: Systematic review and meta-analysis of efficacy and safety. World J Clin Cases 2026; 14(12): 119112
- URL: https://www.wjgnet.com/2307-8960/full/v14/i12/119112.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i12.119112