Published online Mar 25, 2026. doi: 10.5501/wjv.v15.i1.118602
Revised: January 27, 2026
Accepted: March 9, 2026
Published online: March 25, 2026
Processing time: 65 Days and 20.3 Hours
Klebsiella species are important opportunistic pathogens responsible for various hospital- and community-acquired infections. The rise of multidrug-resistant strains, especially those producing extended-spectrum beta-lactamases (ESBL) and AmpC beta-lactamases, poses a major challenge to effective antimicrobial therapy and infection control.
To determine the antimicrobial susceptibility pattern of Klebsiella isolates and to detect the presence of ESBL and AmpC beta-lactamases.
A one-year cross-sectional study was conducted on 920 Gram-negative isolates, from which 130 non-repetitive Klebsiella isolates (14.13%) were selected. Antimicrobial susceptibility testing was performed, and ESBL and AmpC production were identified using standard phenotypic confirmatory methods.
Among 130 Klebsiella isolates, Klebsiella pneumoniae was the predominant species (n = 92, 70.76%). Most isolates were obtained from patients aged 45-60 years (n = 33, 25.38%) and from pus samples (n = 55, 42.3%), with the highest frequency from surgical departments (n = 36, 27.7%). Among the tested antibiotics, gentamicin showed the greatest susceptibility (n = 81, 62.3%). ESBL production was detected in 77 isolates (59.2%), while 62 isolates (47.69%) produced AmpC beta-lactamases. Co-production of ESBL and AmpC was found in 35 isolates (26.92%).
Klebsiella species are significant nosocomial and opportunistic pathogens characterized by high multidrug re
Core Tip: This study highlights the high prevalence of multidrug-resistant Klebsiella species in a tertiary care hospital, with 59.2% producing ESBL and 47.7% AmpC beta-lactamases. Co-production of both enzymes was observed in 26.9% of isolates, highlighting the urgent need for routine surveillance, phenotypic confirmation, and stringent infection control measures to guide targeted antimicrobial therapy and curb the spread of resistant strains. These findings provide critical insights for clinicians and microbiologists aiming to combat nosocomial Klebsiella infections effectively.
- Citation: Kaur H, Arora S, Sharma V, Kaur K, Kamboj S. Study of extended-spectrum beta-lactamases and AmpC beta-lactamases producing Klebsiella from clinical samples in tertiary care hospital of Punjab. World J Virol 2026; 15(1): 118602
- URL: https://www.wjgnet.com/2220-3249/full/v15/i1/118602.htm
- DOI: https://dx.doi.org/10.5501/wjv.v15.i1.118602
Klebsiella species, normally part of the human flora, are opportunistic pathogens that can cause a range of infections, including sepsis, urinary tract infections, pneumonia, and surgical wound infections[1]. These bacteria possess several virulence factors, such as capsules, pili, siderophores, and beta-lactamase enzymes, which enable them to cause disease and evade the host immune systems[2].
The overuse of beta-lactam antibiotics has led to the emergence of mutated beta-lactamases, such as extended-spectrum beta-lactamases (ESBLs), AmpC, and metallo-beta-lactamases, which present a significant therapeutic challenge in healthcare settings by hydrolyzing a wide range of beta-lactam antibiotics, including penicillins and cephalosporins, even carbapenems[3]. ESBLs, mediated by plasmids, can degrade third-generation cephalosporins, penicillins, and aztreonam, but do not affect cephamycin antibiotics like cefoxitin and cefotetan[4,5].
AmpC beta-lactamases primarily hydrolyze cephalosporins and cephamycins like cefoxitin and cefotetan and are resistant to inhibitors like clavulanic acid, sulbactam, and tazobactam. They also degrade oxyimino cephalosporins and monobactams, contributing to resistance to a broad range of beta-lactam antibiotics. Cloxacillin and oxacillin are effective inhibitors of AmpC, enhancing beta-lactam activity[6]. A key challenge with plasmid-mediated AmpC is its ability to mask the presence of ESBLs, limiting treatment options and underscoring the need for proper detection to prevent therapeutic failures[7].
The rise in ESBL and AmpC-producing Klebsiella strains poses a growing challenge to treatment options, highlighting the urgent need for effective detection and management strategies to prevent therapeutic failures[3]. This study aimed to determine the antimicrobial susceptibility patterns of Klebsiella isolates and to detect the presence of ESBL and AmpC beta-lactamases.
The current prospective study was conducted from January 2023 to December 2023 in the Microbiology Department of Guru Gobind Singh Medical College, Faridkot. According to the review of the last 6 months of data, a total of 920 clinical specimens were purposively collected from admitted patients, from which Klebsiella species were isolated.
Inclusion criteria: All clinical specimens (blood, urine, sputum, pus, endotracheal aspirate, catheter tip, pleural fluid, ascitic fluid, cerebrospinal fluid, and wound swab) were obtained from patients of all age groups and either sex admitted to the hospital.
Exclusion criteria: All clinical samples from the outpatient department were excluded from the study.
For this study, clinical specimens received in the Department of Microbiology were processed, and consecutive, non-repetitive Klebsiella species isolates obtained from all types of clinical samples were included. The study was initiated after obtaining approval from the Institutional Ethics Committee.
All the clinical specimens were collected under aseptic precautions using standard microbiological protocols and procedures. The identification of Klebsiella species was confirmed using Gram staining, colony morphology, and standard biochemical tests. The confirmed isolates were then subjected to antimicrobial susceptibility testing and assessed for ESBL and AmpC production.
Antibiotic susceptibility testing was performed on Muller-Hinton agar using the Kirby-Bauer disk diffusion method. The antimicrobial agents tested were: Ampicillin (10 μg), amoxicillin (20 μg), amoxicillin-clavulanic acid (20/10 μg), gen
The detection of beta-lactamases was done by phenotypic methods.
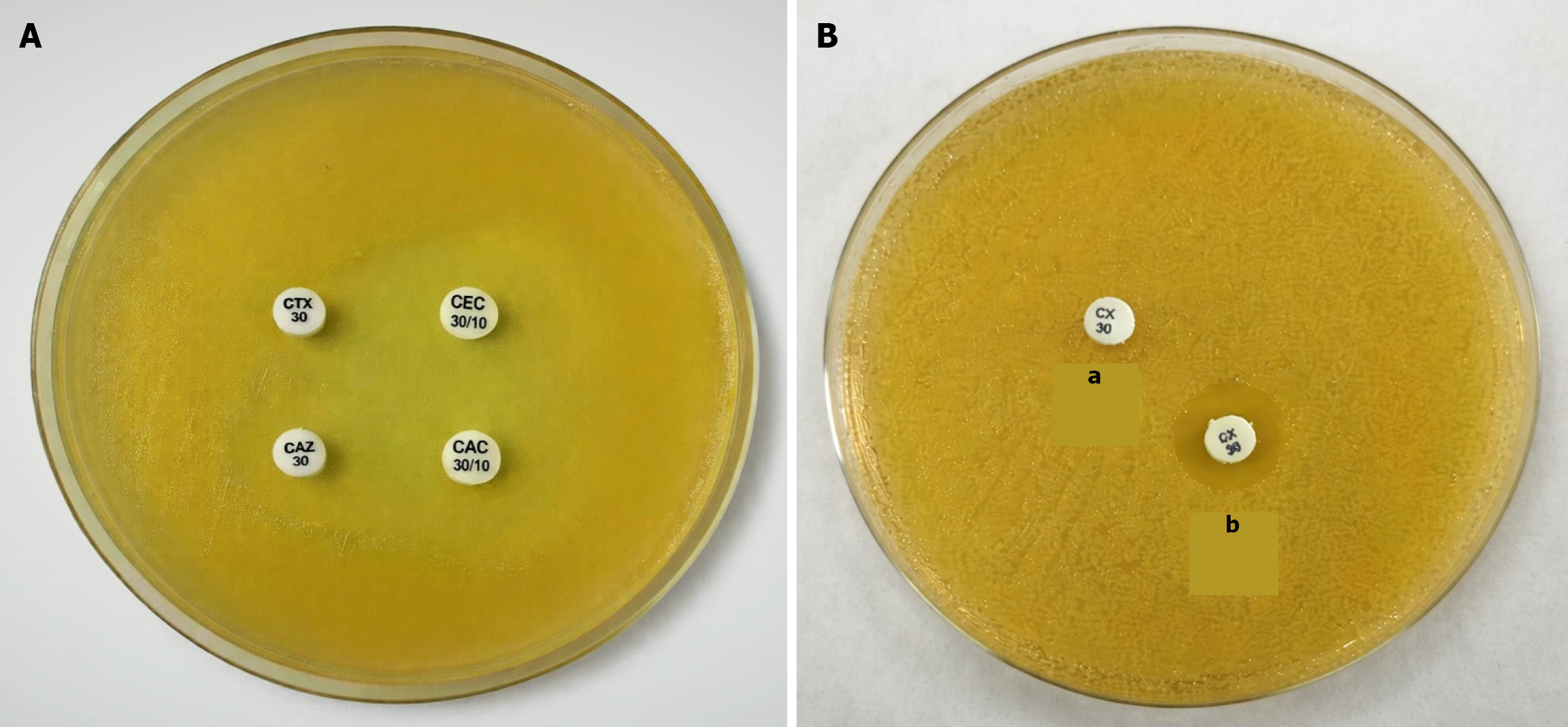
ESBL detection: Combined disc diffusion test[9]: ESBL production was screened by identifying isolates resistant to any third-generation cephalosporin. Screening-positive isolates were confirmed by the combined disc diffusion test. Mueller-Hinton agar plates were inoculated with the test strain, and discs of ceftazidime (30 μg) and cefotaxime (30 μg), alone and in combination with clavulanic acid (30/10 μg), were placed on the agar surface. Plates were incubated at T = 37 °C for t = 18 hours. An increase of ≥ 5 mm in the zone of inhibition around the combination discs compared to the corresponding cephalosporin discs alone was considered indicative of ESBL production (Figure 1A).
AmpC detection: AmpC beta-lactamase production was screened using a cefoxitin (30 μg) disc. Klebsiella isolates showing an inhibition zone diameter of < 18 mm were considered screen-positive and subjected to confirmatory testing. Phe
The descriptive statistics were used to summarize the distribution of Klebsiella isolates by age, gender, specimen type, department, and species. Frequencies and percentages were calculated for categorical variables. Comparative analysis between groups (such as ESBL producers vs non-ESBL producers and AmpC producers vs non-AmpC producers) was performed using the χ2 test to assess the significance of differences in antimicrobial susceptibility patterns. P < 0.05 was considered statistically significant.
The study adheres to the ethical principles outlined in the Declaration of Helsinki. Ethical approval was obtained from the Ethical Review Committee of G.G.S. Medical College and Hospital, Faridkot. Patient confidentiality was strictly maintained, and all data were anonymized prior to analysis.
Out of a total of 920 Gram-negative bacterial isolates, n = 130 (14.13%) were identified as Klebsiella species.
The highest number of Klebsiella isolates was obtained from patients aged 45-60 years (n = 33, 25%), followed by those aged 15-30 years (n = 29, 22%) and 31-44 years (n = 27, 21%). Patients aged > 60 years accounted for n = 22 isolates (17%), while children aged < 14 years contributed n = 19 isolates (15%) of the total number of isolates.
A higher prevalence of Klebsiella species was observed in male patients, who contributed n = 75 cases (57.69%), compared to n = 55 cases (42.31%) in female patients.
The highest number of isolates was obtained from pus specimens (42.30%), followed by urine (20.8%), body fluids (16.15%), and sputum (9.23%), with the lowest proportion from catheter tips (0.76%) (Table 1).
| Specimen | ESBL producers (n = 77) | AmpC producers (n = 62) |
| Pus (n = 55) | 35 (63.63) | 27 (49) |
| Blood (n = 10) | 6 (60) | 2 (20) |
| Catheter tip (n = 1) | 1 (100) | 1 (100) |
| Body fluid (n = 21) | 12 (57.1) | 8 (38) |
| Endotracheal aspirate (n = 4) | 3 (75) | 4 (100) |
| Sputum (n = 12) | 6 (50) | 3 (25) |
| Urine (n = 27) | 14 (51.8) | 17 (62.96) |
| Total = 130 | 59.2% | 47.7% |
Among the isolates, Klebsiella pneumonia was the most frequently identified species (n = 92, 70.76%), followed by Klebsiella oxytoca (n = 23, 17.69%) and Klebsiella aerogenes (n = 15, 11.53%).
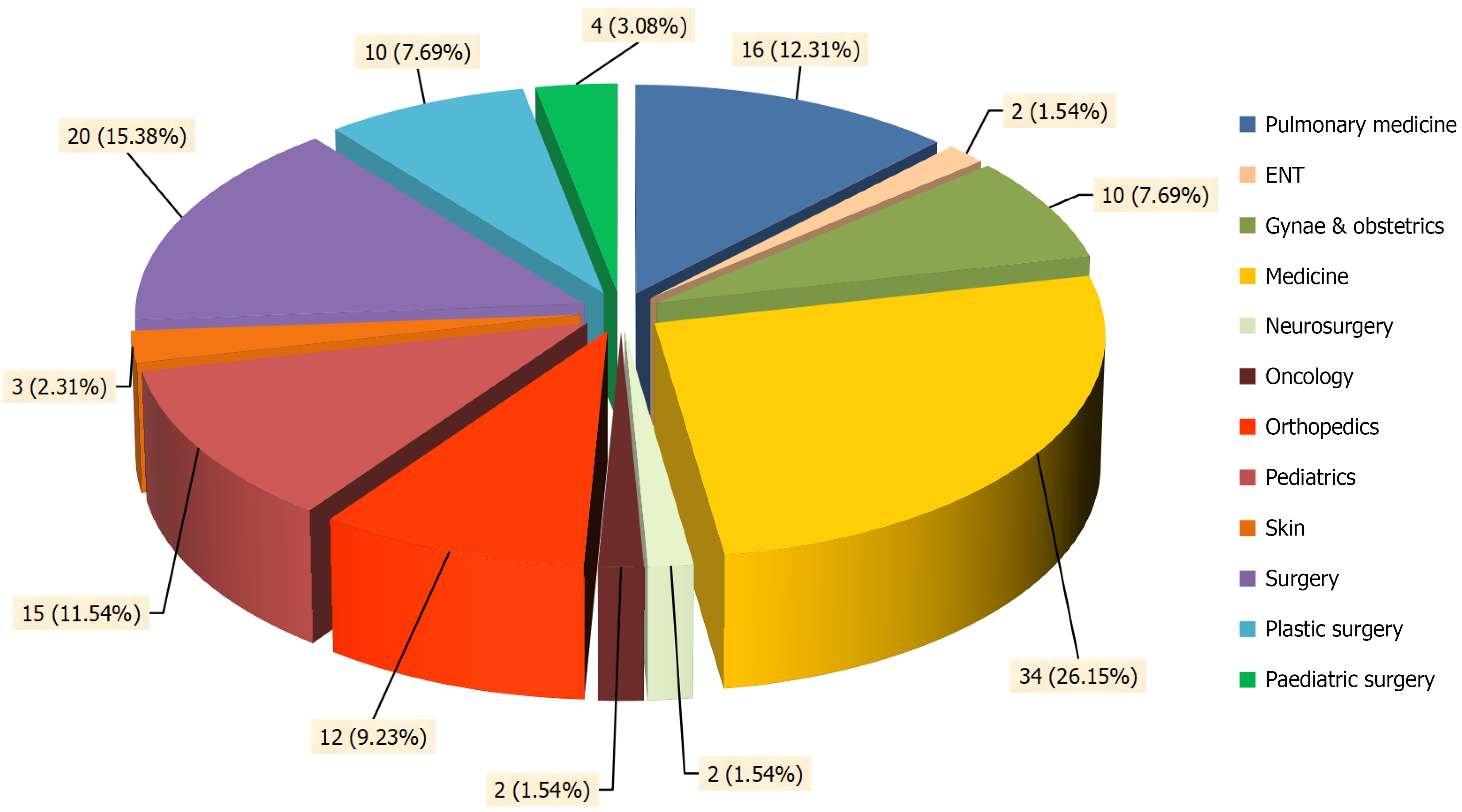
The majority of Klebsiella isolates were recovered from the surgical departments (including plastic surgery, pediatric surgery, and neurosurgery) (n = 36, 27.7%), followed by the medicine department (n = 34, 26.15%), pulmonary medicine (n = 16, 12.31%), and pediatrics (n = 15, 11.53%) (Figure 2).
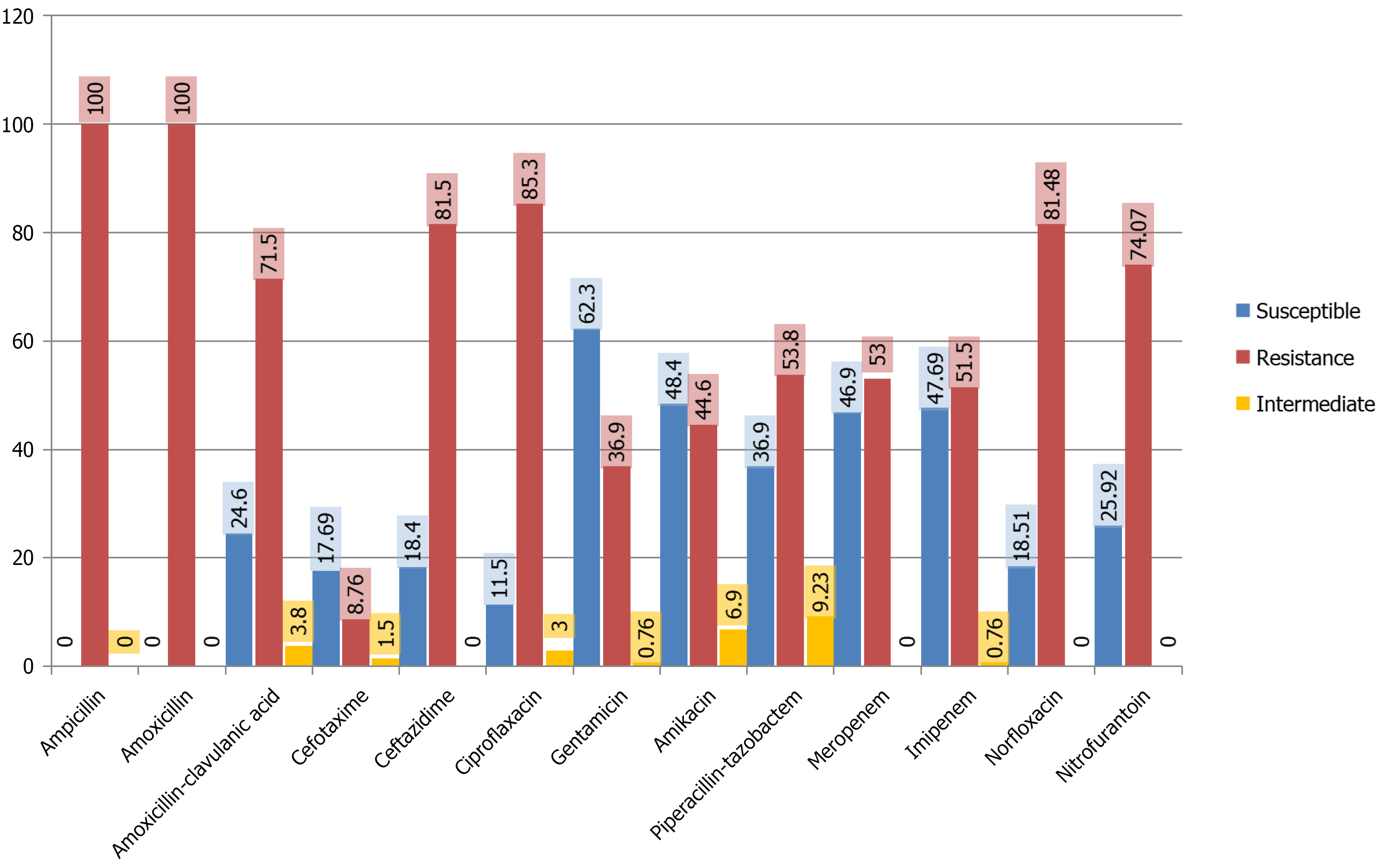
The aminoglycosides were the most effective antibiotics against Klebsiella, with gentamicin demonstrating higher sensitivity (n = 81, 62.3%) compared to amikacin (n = 63, 48.4%) (Figure 3).
Among 130 Klebsiella isolates, n = 77 (59.2%) were ESBL producers and n = 62 (47.7%) were AmpC producers.
ESBL producers were most common in pus samples (n = 35, 63.6%), while AmpC producers were highest in endotracheal aspirates (n = 4, 100%) and catheter tips (n = 1, 100%). Overall, beta-lactamase production was widespread across all clinical specimens (Table 1).
ESBL producers showed significantly higher resistance to cefotaxime (χ2 = 35.22, P < 0.001), ceftazidime (χ2 = 37.00, P < 0.001), ciprofloxacin (χ2 = 7.14, P = 0.03), and nitrofurantoin (χ2 = 10.78, P = 0.004). No significant differences were observed for ampicillin, amoxicillin, gentamicin, and amikacin (P > 0.05). These results highlight that ESBL production in Klebsiella is strongly associated with multidrug resistance, posing significant challenges for effective antibiotic therapy. P values were calculated using the χ2 test, comparing susceptibility patterns between ESBL-producing and non-ESBL-producing Klebsiella isolates. P < 0.05 was considered statistically significant (Table 2).
| Antibiotics | ESBL producers (n = 77) | Non ESBL producers (n = 53) | χ2 statistics | P value | ||||
| Susceptible | Resistance | Intermediate | Susceptible | Resistance | Intermediate | |||
| Ampicillin | 0 (0) | 77 (100) | 0 (0) | 0 (0) | 53 (100) | 0 (0) | 0 | 1 |
| Amoxicillin | 0 (0) | 77 (100) | 0 (0) | 0 (0) | 53 (100) | 0 (0) | 0 | 1 |
| Amoxicillin-clavulanic acid | 14 (18.2) | 59 (76.6) | 4 (5.2) | 18 (33.9) | 34 (64.1) | 1 (1.9) | 4.75 | 0.09 |
| Cefotaxime | 1 (1.3) | 75 (97.4) | 1 (1.3) | 22 (41.5) | 30 (56.6) | 1 (1.9) | 35.22 | < 0.0001a |
| Ceftazidime | 1 (1.3) | 76 (98.7) | 0 (0) | 23 (43.3) | 30 (56.6) | 0 (0) | 37 | < 0.0001a |
| Ciproflaxacin | 5 (6.5) | 71 (92.2) | 1 (1.3) | 10 (18.9) | 40 (75.4) | 3 (5.6) | 7.14 | 0.03a |
| Gentamicin | 46 (59.7) | 31 (40.3) | 0 (0) | 35 (66.0) | 17 (32.1) | 1 (1.9) | 2.22 | 0.33 |
| Amikacin | 38 (49.4) | 35 (45.5) | 4 (5.2) | 25 (47.2) | 23 (43.3) | 5 (9.4) | 0.88 | 0.64 |
| Piperacillin-tazobactam | 30 (39.0) | 41 (53.2) | 6 (7.8) | 18 (34.0) | 29 (54.7) | 6 (11.3) | 0.65 | 0.72 |
| Meropenem | 36 (46.8) | 41 (53.2) | 0 (0) | 25 (47.2) | 28 (52.8) | 0 (0) | 0 | 1 |
| Imipenem | 37 (48.1) | 39 (50.6) | 1 (1.3) | 25 (47.2) | 28 (52.8) | 0 (0) | 0.72 | 0.70 |
| Nitrofurantoin1 | 11 (78.6) | 3 (21.4) | 0 (0) | 2 (15.4) | 11 (84.6) | 0 (0) | 10.78 | 0.004a |
| Norfloxacin1 | 2 (14.3) | 12 (85.7) | 0 (0) | 5 (38.5) | 8 (61.5) | 0 (0) | 2.05 | 0.36 |
AmpC-producing Klebsiella isolates show significantly greater resistance to amoxicillin-clavulanic acid and imipenem compared to non-AmpC producers. The strongest effect was seen with amoxicillin-clavulanic acid, highlighting beta-lactamase-mediated resistance against beta-lactam/beta-lactamase inhibitor combinations.
The findings suggest that AmpC production contributes to multidrug resistance, particularly impacting beta-lactam antibiotic efficacy in Klebsiella infections. The χ2 test compared susceptibility patterns between AmpC and non-AmpC-producing Klebsiella isolates (P < 0.05 = significant) (Table 3).
| Antibiotics | AmpC producers (n = 62) | Non AmpC producers (n = 68) | χ2 statistics | P value | ||||
| Susceptible | Resistance | Intermediate | Susceptible | Resistance | Intermediate | |||
| Ampicillin | 0 (0) | 62 (100) | 0 (0) | 0 (0) | 68 (100) | 0 (0) | 0 | 1 |
| Amoxicillin | 0 (0) | 62 (100) | 0 (0) | 0 (0) | 68 (100) | 0 (0) | 0 | 1 |
| Amoxicillin-clavulanic acid | 7 (11.29) | 50 (80.64) | 5 (8.06) | 25 (36.76) | 43 (63.23) | 0 (0) | 15.41 | 0.0005a |
| Cefotaxime | 8 (12.9) | 52 (83.87) | 2 (3.22) | 15 (22.5) | 53 (77.94) | 0 (0) | 3.87 | 0.14 |
| Ceftazidime | 9 (14.51) | 53 (85.48) | 0 (0) | 15 (22.5) | 53 (77.94) | 0 (0) | 1.23 | 0.54 |
| Ciprofloxacin | 4 (6.451) | 57 (91.93) | 1 (1.61) | 11 (16.17) | 54 (79.41) | 3 (4.41) | 4.08 | 0.13 |
| Gentamicin | 37 (59.67) | 24 (38.70) | 1 (1.61) | 44 (64.70) | 24 (35.29) | 0 (0) | 1.33 | 0.51 |
| Amikacin | 27 (43.54) | 27 (43.54) | 8 (12.9) | 34 (50) | 31 (45.58) | 3 (4.41) | 3.08 | 0.21 |
| Piperacillin-tazobactam | 19 (30.64) | 39 (62.90) | 4 (6.451) | 29 (42.64) | 31 (45.58) | 8 (11.76) | 4.06 | 0.13 |
| Meropenem | 23 (37.09) | 39 (62.90) | 0 (0) | 38 (55.88) | 30 (44.11) | 0 (0) | 4.6 | 0.10 |
| Imipenem | 23 (37.09) | 38 (61.29) | 1 (1.61) | 39 (57.35) | 29 (42.64) | 0 (0) | 6.07 | 0.04a |
| Nitrofurantoin1 | 3 (17.64) | 14 (82.35) | 0 (0) | 2 (20) | 8 (80) | 0 (0) | 0.02 | 0.99 |
| Norfloxacin1 | 5 (29.41) | 12 (70.58) | 0 (0) | 2 (20) | 8 (80) | 0 (0) | 0.29 | 0.86 |
A total of n = 35 isolates (26.92%) exhibited the co-existence of both AmpC and ESBL enzymes, indicating a notable proportion of multidrug resistance among the Klebsiella species.
Klebsiella causes various infections like bacteremia, pneumonia, urinary tract infections, and meningitis, mainly acquired in healthcare settings, particularly affecting immunocompromised patients. These infections make up 5%-7% of all hospital-acquired infections, highlighting the significance of Klebsiella as a nosocomial pathogen. Drug-resistant strains, such as those producing ESBLs and AmpC beta-lactamases, are a growing concern globally, complicating treatment[6,12]. The current study focused on Klebsiella species and their prevalence, antibiotic resistance, and mechanisms of resistance. In this context, continuous regional surveillance is essential, as resistance patterns vary considerably across geographic locations and healthcare settings.
In a study of 920 g-negative bacteria isolated over a year, n = 130 (14.13%) consecutive Klebsiella isolates were con
Among the isolates, 25% (n = 33) were from individuals aged 45-60 years, 17% (n = 22) from those over 60 years, and 15% (n = 19) from children under 14 years. Sivaramakrishnan et al[16] also reported the highest Klebsiella infections in the 40-60 age group (36.5%), while Hymavathi et al[17] found higher rates in the elderly (37.4%). The study found that 58% (n = 75) of Klebsiella infection cases occurred in males, indicating a male predominance, consistent with previous studies by Sivaramakrishnan et al[16] and Hymavathi et al[17].
Pus specimens yielded the highest number of isolates, with n = 55 cases accounting for 42.3% of the total (Table 1), as reported by Biradar and Roopa[18]. The majority of Klebsiella isolates were from surgical departments (36, 27.7%) (Figure 2). Similar findings were reported in other studies[19,20]. The most common species identified was Klebsiella pneumonia (92, 70.76%), followed by Klebsiella oxytoca (23, 17.69%) and Klebsiella aerogenes (15, 11.53%). This is in concordance with the study done by Sivaramakrishnan et al[16].
Resistance patterns revealed that Klebsiella isolates showed complete resistance (130, 100%) to ampicillin, with high resistance to third-generation cephalosporins like cefotaxime(105, 80.76%) and ceftazidime (106, 81.5%) (Figure 3), similar to findings by Gupta et al[7]. However, the isolates were more susceptible to aminoglycosides, with gentamicin being the most effective (81, 62.3%). Better susceptibility to amikacin may be explained by the absence of routine use of amikacin as empirical therapy and its lack of considerable cross-resistance with beta-lactam groups of antibiotics[5].
ESBL production is acknowledged as one of the major resistance mechanisms in Klebsiella. In the present study, out of n = 105 isolates suspected of producing ESBLs, n = 77 (59.2%) were phenotypically confirmed to exhibit ESBL production. This corroborates the findings of Segar et al[21] (50.9%) and Ibadin et al[14] (54.4%). In contrast, some studies have documented lower frequencies of ESBL-positive isolates[22,23]. The high production rates were observed in catheter tip specimens (1, 100%) and endotracheal aspirates (3, 75%) (Table 1).
ESBL-producing Klebsiella isolates frequently exhibit broad antibiotic resistance across diverse antibiotic categories. This multidrug resistance is attributed to the plasmid-mediated nature of ESBL enzymes, which often carry resistance genes via plasmids, transposons, and integrons, facilitating transfer to other bacteria. These isolates frequently exhibit increased resistance to third-generation cephalosporins and other tested antimicrobial agents compared to non-ESBL producers. In our study, all 77 ESBL-positive isolates underwent primary and supplementary antibiotic susceptibility testing, revealing significant resistance to ampicillin (100%), amoxicillin (100%), ceftazidime (98.7%), cefotaxime (97.4%), ciprofloxacin (92.2%), and amoxicillin-clavulanic acid (76.62%). Conversely, ESBL producers exhibited higher sensitivity to gentamicin (59.74%), amikacin (49.35%), imipenem (48.05%), meropenem (46.75%), and piperacillin-tazobactam (38.96%) (Table 2). These findings are consistent with those reported by Soltani et al[24], who observed similar resistance to cefotaxime and ceftazidime and sensitivity to carbapenems (imipenem and meropenem) (96%) and piperacillin-tazobactam (84%). However, in contrast, El Bouamri et al[24] reported reduced susceptibility to gentamicin among ESBL producers.
AmpC beta-lactamases act primarily as cephalosporinases and are notably resistant to inhibition by clavulanic acid. They distinguish themselves from ESBLs by their ability to hydrolyze and render ineffective cephalosporins (such as ceftriaxone, cefotaxime, and ceftazidime), cephamycins (including cefoxitin and cefotetan), aminopenicillins, and monobactams. The resistance mechanisms of the enzyme include both chromosomal and plasmid-mediated pathways. Chromosomally mediated AmpC beta-lactamases are known to be inducible enzymes. Organisms that produce AmpC beta-lactamases can serve as a concealed reservoir for ESBLs. Moreover, the significant expression of AmpC enzymes can obscure the detection of ESBLs, potentially leading to inappropriate and ineffective antimicrobial treatment, which could have serious consequences. According to Clinical and Laboratory Standard Institute guidelines, there is currently no universally accepted technique established for the reliable detection of AmpC beta-lactamases[6]. Various studies have explored various phenotypic methods for detecting AmpC enzymes. In the present study, among 122 suspected AmpC producer n = 62 (47.69%) of Klebsiella isolates were confirmed as AmpC lactamase producers using boric acid and EDTA. The finding is in comparison with studies by Altun et al[25] Jena et al[12], who reported 47% and 62.13% as AmpC producers among Klebsiella isolates, respectively. In contrast, a study by Gajul et al[26] revealed a lower rate of AmpC production (12.5%). These variances may be attributed to several factors, such as geographical disparities, clinical diagnostic methods, antimicrobial prescribing practices, and infection control measures across institutions.
AmpC production was highest in catheter tip (n = 1) and endotracheal aspirate (n = 4) specimens (100%), followed by urine (n = 17, 62.96%), and lowest in blood samples (Table 1). In contrast, Ibadin et al[14] reported the highest AmpC rates in eye swabs (33.3%), urine (19.8%), and catheter tips (18.2%). Among AmpC producers, aminoglycosides were the most effective (27, 43.54% for amikacin, and 37, 59.67% for gentamicin), followed by carbapenems (23, 37.09%) for both meropenem and imipenem) and beta-lactam/beta-lactamase inhibitors (19, 30.64%). Susceptibility to ceftazidime and cefotaxime was low (9, 14.5%, and 8, 12.9%). Non-AmpC producers showed higher susceptibility overall (Table 3). A previous study reported higher sensitivity of AmpC producers to carbapenems (100%) and aminoglycosides (66.6%)[24].
Our study found a high co-expression rate for ESBLs and AmpC (n = 35, 26.92%), aligning with Kolhapure et al[15]. These findings underscore the growing problem of multidrug-resistant Klebsiella, which complicates treatment options. The co-existence of multiple beta-lactamases in a single isolate can mask detection and lead to therapeutic failure if not properly identified[9,24]. The study highlights the need for tailored antibiotic regimens and the importance of robust laboratory testing to inform treatment strategies.
While molecular techniques represent the gold standard for detecting various beta-lactamases, their routine implementation in diagnostic laboratories remains challenging. In contrast, despite their tendency to yield somewhat imprecise results, phenotypic methods are extensively utilized due to their simplicity and cost-effectiveness. In our study, although molecular techniques were not employed to detect beta-lactamases, the phenotypic methods successfully identified multiple beta-lactamases among the isolates.
The findings of this study have significant implications for both clinical practice and public health. The high prevalence of beta-lactamase-producing Klebsiella underscores the need for strict adherence to antibiotic stewardship and robust infection control measures. In resource-constrained laboratories, routine testing for beta-lactamase production is often not performed, allowing these resistant strains to persist and spread undetected within hospitals. Inappropriate or excessive use of antimicrobials further contributes to the increasing prevalence of beta-lactamase production. Therefore, judicious use of antibiotics is advised, and targeted therapeutic antibiotics should be prescribed for the shortest effective duration.
Several limitations must be considered. First, the single-center nature of the study limits the generalizability of the results. Second, this was an in vitro study, and no correlation with clinical outcomes was established. Third, only phenotypic methods were employed for beta-lactamase detection. The addition of genotypic profiling could have enriched the findings.
The current study highlights the high prevalence of ESBLs and AmpC beta-lactamases among Klebsiella isolates in our hospital. These isolates exhibit significant resistance to multiple classes of antibiotics, greatly limiting available therapeutic options. The coexistence of ESBL and AmpC production further underscores the need for regular screening and accurate confirmation of resistance patterns. Such efforts are essential to guide clinicians in selecting both empirical and definitive treatment regimens, thereby helping to prevent therapeutic failures.
In addition to antimicrobial resistance monitoring, the study emphasizes the urgent need to reassess existing infection control practices. Implementing a robust drug resistance surveillance system is crucial in combating the spread of resistant strains. The essential measures, like strict adherence to hand hygiene protocols, remain essential in preventing cross-infection and maintaining hospital safety standards.
We would like to express our heartfelt gratitude to the faculty and staff of the Department of Microbiology, Guru Gobind Singh Medical College and Hospital, Faridkot, for their constant support and encouragement during the course of this study.
| 1. | Dong N, Yang X, Chan EW, Zhang R, Chen S. Klebsiella species: Taxonomy, hypervirulence and multidrug resistance. EBioMedicine. 2022;79:103998. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 266] [Cited by in RCA: 207] [Article Influence: 51.8] [Reference Citation Analysis (1)] |
| 2. | Brisse S, Grimont F, Grimont PAD. The Genus Klebsiella. In: Dworkin M, Falkow S, Rosenberg E, Schleifer KH, Stackebrandt E. The Prokaryotes. New York: Springer, 2006. [DOI] [Full Text] |
| 3. | Khan MK, Thukral SS, Gaind R. Evaluation of a modified double-disc synergy test for detection of extended spectrum beta-lactamases in AMPC beta-lactamase-producing proteus mirabilis. Indian J Med Microbiol. 2008;26:58-61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 16] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 4. | Chaudhary U, Aggarwal R. Extended spectrum -lactamases (ESBL) - an emerging threat to clinical therapeutics. Indian J Med Microbiol. 2004;22:75-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 48] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 5. | Abayneh M, Tesfaw G, Abdissa A. Isolation of Extended-Spectrum β-lactamase- (ESBL-) Producing Escherichia coli and Klebsiella pneumoniae from Patients with Community-Onset Urinary Tract Infections in Jimma University Specialized Hospital, Southwest Ethiopia. Can J Infect Dis Med Microbiol. 2018;2018:4846159. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 49] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 6. | Singh P. The incidence of AmpC β-lactamases producing Klebsiella pneumoniae subspecies pneumoniae. Int J Res Med Sci. 2018;6:1169. [DOI] [Full Text] |
| 7. | Gupta V, Kumarasamy K, Gulati N, Garg R, Krishnan P, Chander J. AmpC β-lactamases in nosocomial isolates of Klebsiella pneumoniae from India. Indian J Med Res. 2012;136:237-241. [PubMed] |
| 8. | ANSI Webstore. Performance standards for antimicrobial susceptibility testing, 32nd Edition. [cited 3 August 2025]. Available from: https://webstore.ansi.org/standards/clsi/clsim100ed32. |
| 9. | Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing, 29th Edition. [cited 3 August 2025]. Available from: https://www.scribd.com/document/698721084/M100S-ed29. |
| 10. | Black JA, Moland ES, Thomson KS. AmpC disk test for detection of plasmid-mediated AmpC beta-lactamases in Enterobacteriaceae lacking chromosomal AmpC beta-lactamases. J Clin Microbiol. 2005;43:3110-3113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 149] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 11. | Ahmad KM, Alamen AAM, Saad SAM, Elzen AA. Emergence of AmpC β-Lactamase– and Metallo-β-Lactamase–producing Klebsiella pneumoniae in Sebha, Libya. Med J Islamic World Acad Sci. 2019;27:9-16. [DOI] [Full Text] |
| 12. | Jena J, Sahoo RK, Subudhi E, Debata NK. Prevalence of ESBL, MBL and AmpC β-lactamase-producing multidrug-resistant gram-negative bacteria in a tertiary care hospital. J Pure Appl Microbiol. 2014;8:4099-4105. |
| 13. | Oberoi L, Singh N, Sharma P, Aggarwal A. ESBL, MBL and Ampc β Lactamases Producing Superbugs - Havoc in the Intensive Care Units of Punjab India. J Clin Diagn Res. 2013;7:70-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 38] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 14. | Ibadin EE, Omoregie R, Igbarumah IO, Anogie NA, Ogefere HO. Prevalence of Extended Spectrum β-Lactamase, AmpC β-Lactamase and Metallo-β-Lactamase Among Gram Negative Bacilli Recovered From Clinical Specimens in Benin City, Nigeria. Int J Enteric Pathog. 2017;5:85-91. [DOI] [Full Text] |
| 15. | Kolhapure R, Kumar A, Rajkumar H. Coexpression of ESBL, Amp C and MBL in gram negative bacilli. Int J Res Med Sci. 2015;2698-2703. [DOI] [Full Text] |
| 16. | Sivaramakrishnan R, Leela KV, Sujith R, Naik S, Priya S. Antibiotic Resistance of Clinical Klebsiella Isolates from Tertiary Care Hospital. J Pure Appl Microbiol. 2022;16:2669-2678. [RCA] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 17. | Hymavathi R, Kumari SS, Swarnalatha G, Surekha A. Prevalence of ESBL-producing Klebsiella species isolated from respiratory samples at intensive care units. Ann Int Med Dent Res. 2018;4:MB6-MB10. |
| 18. | Biradar S, Roopa C. Isolation and Antibiogram of Klebsiella species from Various Clinical Specimens. Int J Curr Microbiol Appl Sci. 2015;4:991-995. |
| 19. | Vijayashree V, Saikeerthana D, Prabha P. Prevalence and antibiotic susceptibility pattern of Klebsiella species isolated from various clinical samples in a tertiary care hospital Coimbatore. Int J Community Med Public Health. 2021;8:4886-4890. [DOI] [Full Text] |
| 20. | Kaur S, Awari A. Prevalence and antibiogram of klebsiella species detected in clinical. Asian J Pharm Clin Res. 2024;17:121-124. [DOI] [Full Text] |
| 21. | Segar L, Kumar S, Joseph NM, Sivaraman U, Joseph NM. Prevalence of extended spectrum beta lactamases among enterobacteriaceae and their antibiogram pattern from various clinical samples. Asian J Pharm Clin Res. 2015;8:220-223. |
| 22. | Nepal K, Pant ND, Neupane B, Belbase A, Baidhya R, Shrestha RK, Lekhak B, Bhatta DR, Jha B. Extended spectrum beta-lactamase and metallo beta-lactamase production among Escherichia coli and Klebsiella pneumoniae isolated from different clinical samples in a tertiary care hospital in Kathmandu, Nepal. Ann Clin Microbiol Antimicrob. 2017;16:62. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 64] [Article Influence: 7.1] [Reference Citation Analysis (1)] |
| 23. | Soltani R, Ehsanpoor M, Khorvash F, Shokri D. Antimicrobial susceptibility pattern of extended-spectrum β-lactamase-producing bacteria causing nosocomial urinary tract infections in an Iranian referral teaching hospital. J Res Pharm Pract. 2014;3:6-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 24. | El Bouamri MC, Arsalane L, El Kamouni Y, Zouhair S. Antimicrobial susceptibility of urinary Klebsiella pneumoniae and the emergence of carbapenem-resistant strains: A retrospective study from a university hospital in Morocco, North Africa. Afr J Urol. 2015;21:36-40. [RCA] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 20] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 25. | Altun Ş, Tufan ZK, Yağcı S, Önde U, Bulut C, Kınıkl S, Demiroz AP. Extended spectrum beta-lactamases, AmpC and metallo beta-lactamases in emerging multidrug-resistant Gram-negative bacteria in intensive care units. Sci Rep. 2013;2:707. [DOI] [Full Text] |
| 26. | Gajul SV, Mohite ST, Mangalgi SS, Wavare SM, Kakade SV. Klebsiella Pneumoniae in Septicemic Neonates with Special Reference to Extended Spectrum β-lactamase, AmpC, Metallo β-lactamase Production and Multiple Drug Resistance in Tertiary Care Hospital. J Lab Physicians. 2015;7:32-37. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 15] [Article Influence: 1.4] [Reference Citation Analysis (1)] |