Published online Apr 26, 2026. doi: 10.4252/wjsc.v18.i4.117707
Revised: January 17, 2026
Accepted: March 9, 2026
Published online: April 26, 2026
Processing time: 127 Days and 19.5 Hours
Anti-aging for the preservation and recovery of physical and brain functions may be a major topic of regenerative medicine in the super-aging society. Stem cells and their secretory active molecules can play a central role of regenerative medi
To investigate whether extracellular vesicles (EVs) from amniotic membrane stem cells (AMSCs) enhance physical activity, including stamina, and cognitive fun
EVs were collected from conditioned media of AMSCs after hypoxic (2% O2, 5% CO2) cultivation for 3 days. The size and composition of EVs was analyzed via nanoparticle-tracking analysis and proteome/lipidome profiles, and functional molecules such as growth factors and neurotrophic factors were analyzed via enzyme linked immunosorbent assay. Male ICR mice were subcutaneously administered with D-galactose (300 mg/kg) for 6 weeks to induce facilitated aging, during which intravenously injected with EVs (6 × 108 exosome particles/body) at weeks 0, 2, 4, and 6. Physical activity and cognitive function were assessed through Rota-rod, forced swimming and passive avoidance performances, respectively. To clarify underlying mechanisms, acetylcholine (ACh), brain-derived neurotrophic factor (BDNF), sirtuin 1 (SIRT1), glial fibrillary acid protein (GFAP), glycogen, and thiobarbituric acid-reactive substances (TBARS) were analyzed in the brain and muscles.
Six-week injection of D-galactose decreased physical activity and impaired learning and memory function, along with the reduced ACh, BDNF, SIRT1, and glycogen in the brain and muscles, whereas brain GFAP and muscular TBARS increased. However, EV treatment recovered the D-galactose-induced neurobehavioral deficits not only by increasing BDNF and SIRT1 (regulating neuro-muscular adaptation and function) and enhancing brain ACh (governing memory acquisition), but also by restoring muscular glycogen (an energy source) and attenuating brain GFAP and muscular TBARS (inflammatory and oxidative injury markers).
EVs from AMSCs restored cognitive function of mice with facilitated brain-aging by increasing ACh, BDNF, and SIRT1. EVs also enhanced stamina not only by attenuating tissue injury, but also by strengthening the muscles through the production of glycogen and BDNF.
Core Tip: We obtained a large amount of extracellular vesicles containing high concentrations of growth factors and neurotrophic factors via hypoxic cultivation of amniotic membrane stem cells. The extracellular vesicles ameliorated physical and cognitive dysfunctions by recovering anti-aging factors and energy sources, in the brain and muscles, such as brain-derived neurotrophic factor, sirtuin 1, glycogen, acetylcholine, and antioxidation.
- Citation: Tsolmon KE, Bavuu Z, Kim SS, Jeong HS, Park D, Kim YB. Hypoxia-conditioned extracellular vesicles from human amniotic membrane stem cells attenuate D-galactose-induced aging in mice. World J Stem Cells 2026; 18(4): 117707
- URL: https://www.wjgnet.com/1948-0210/full/v18/i4/117707.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v18.i4.117707
Aging is a complex biological process that leads to a progressive decline in physiological functions, ultimately impacting physical activity, cognitive abilities, and overall quality of life. In addition to decreased physical activity, deterioration of learning and memory function is a characteristic feature in aging and Alzheimer’s disease (AD) patients[1], which has been linked to reduced neuroplasticity, increased oxidative stress, and a decline in regenerative capacity[2].
In AD patients, degeneration of presynaptic cholinergic system is one of the causes of cognitive dysfunction[3,4], in which loss of choline acetyltransferase activity and ensuing acetylcholine (ACh) depletion, has been reported[5-7]. As in AD, senescence-related brain aging leads to atrophy and degeneration of the cholinergic nervous system, resulting in profound neurobehavioral and cognitive dysfunction due to decreased ACh biosynthesis and reduced secretion of neurotrophins such as growth factors (GFs) and neurotrophic factors (NFs)[8,9]. Several hypotheses have been presented to explain the process of aging[10], in which oxidative stress was demonstrated to play a key role of senescence[11]. Increased oxidative stress causes mitochondrial dysfunction that produces high-level reactive oxygen species, acce
D-galactose (D-Gal) is a nutrient derived from lactose in milk. Lactose is hydrolyzed to monosaccharides glucose and galactose. However, excessive D-Gal reacts with free amines of amino acids in proteins and peptides to form advanced glycation end products that cause facilitation of oxidative damage linked to the pathogenesis of degenerative diseases[14]. Especially, D-Gal makes cellular constituents vulnerable to oxidative reaction, and reduces mitochondrial energy production[15]. In addition, D-Gal not only accelerates inflammatory brain injury through activation and degeneration of astrocytes, but also causes cognitive deficits by activating acetylcholinesterase (AChE)[15,16]. Since D-Gal administration has been widely used to mimic aging-related biochemical and behavioral alterations, including reduced motor performance, cognitive impairment, and oxidative brain damage, we adopted D-Gal as a model compound for facilitated brain aging.
In our former studies, it was found that transplantation of neural stem cells encoding choline acetyltransferase gene, an ACh-synthesizing enzyme, recovered cognitive function and physical activity of animal models of AD[17] and aging[18]. Notably, amniotic membrane stem cells (AMSCs) and adipose-derived stem cells (ADSCs) were differentiated into neurons in the brain, recovered ACh and neurotrophins, improved physical and cognitive functions, and finally extended lifespan[19]. Interestingly, it was demonstrated that among GFs and NFs, brain-derived neurotrophic factor (BDNF) played a central role in the stamina-enhancing activity of ADSCs[20].
However, using stem cells has some drawbacks. The cells have to be directly transplanted into the brain for enough efficiency, because they cannot penetrate blood-brain barrier following intravenous (IV) administration. In addition, repeated injections of live cells can induce risks of immunogenicity and tumor transformation. It has been confirmed that bioactive molecules are released in a form of extracellular vesicles (EVs) from functional cells such as inflammatory, immune, and stem cells. EVs are lipid-membrane nanoparticles secreted by stem cells to encapsulate and deliver hun
The present study aimed to investigate the anti-aging potential of AMSC-derived EVs (AMSC-EVs) in mice with facilitated brain aging induced by D-Gal. We assessed the effects of EVs on motor coordination, swimming stamina, cognitive function, and key biochemical markers such as ACh, BDNF, sirtuin 1 (SIRT1), and glycogen in the brain and muscles. By exploring the multifaceted impacts of AMSC-EVs, we sought to shed light on their therapeutic potential for treating aging-related motor and cognitive impairments.
Human placenta were obtained from a healthy pregnant female donor during Caesarean section with informed consent. Amniotic membrane tissue was digested with collagenase I and neutralized with a medium containing 10% fetal bovine serum (Biowest, Kansas City, MO, United States). After removing erythrocytes, the remaining cells were suspended in Keratinocyte serum-free medium (Invitrogen, Carlsbad, CA, United States) supplemented with 5% fetal bovine serum and antibiotics[22]. Culture of AMSCs were maintained under 5% CO2 at 37 °C in culture flask.
The separated AMSCs were suspended in the defined serum-free medium in Hyper flask (Nunc, Rochester, NY, United States), and cultivated under normoxic oxygen (20% O2, 5% CO2) or hypoxic oxygen (2% O2, 5% CO2) tensions at 37 °C for 3 days[22,23]. The media were filtered through a bottle-top vacuum filter system (0.22 μm, PES membrane, Corning, Glendale, CA, United States). The conditioned media were 30-fold concentrated using Vivaflow-200 to obtain final EVs (Sartorius, Hannover, Germany).
To analyze size distribution of EVs, nanoparticle-tracking analysis were performed using Nanosight NS300 equipped with v3.2.16 analytical software (Malvern Panalytical, Malvern, United Kingdom). Purified EVs were adjusted to 108 particles/mL according to the manufacturer’s recommendations in phosphate-buffered saline (PBS). The camera level was adjusted until the particle signal saturation did not exceed 20% and all particles were clearly visible, five images were captured in 60 seconds, and the size and particle concentration were analyzed[22,23].
Enzyme linked immunosorbent assay (ELISA) was conducted to analyze functional molecules, i.e., GFs and NFs related to cytoprotection and tissue regeneration in normoxic and hypoxic culture EVs[22,23]. ELISA kits for BDNF (ab212166; Abcam, United Kingdom), nerve GF (NGF, ab99986; Abcam, United Kingdom), platelet-derived growth factor (PDGF, ab100622; Abcam, United Kingdom), insulin-like growth factor (IGF, ab100695; Abcam, United Kingdom) or vascular endothelial growth factor (VEGF, ab222510; Abcam, United Kingdom) were used according to the manufacturer’s instructions. Briefly, EVs were incubated with the primary antibodies at room temperature, followed by secondary antibody, and color-developing substrate was applied for 10-30 minutes. After treatment with a stop solution, the absor
Proteome analysis, including CD markers of exosomes, was performed according to the standard protocol of KBio Health (Osong, Korea). For liquid chromatography-tandem mass spectrometry analysis of proteins, EVs were dissolved in 0.1% formic acid to make final 1 μg/μL. Protein concentration was determined using the BCA assay. Using a nano LC system, 3 μL (3 μg) EV sample was injected onto an LC analytical column (EASY-Spray 50 cm × 75 μm PepMap RSLC C18 2 μm) equipped with Q-Exactive™ Plus Hybrid Quadrupole-Orbitrap™ Mass Spectrometer (Thermo Fisher Scientific, Waltham, MA, United States), and separated with a linear gradient of solvent B (5%-95%) over 70 minutes at 300 nL/minute[23]. Data analysis was performed in Proteome Discoverer 2.4 (Thermo Fisher Scientific, Waltham, MA, United States), with searches run against the Homo sapiens database.
Lipidome analysis was performed according to the standard protocol of KBio Health. Using a UPLC system for analysis of lipids, 5 μL of sample was injected onto an LC analytical column (Agilent Poroshell EC-120 C18 2.1 mm × 100 mm) equipped with Q-Exactive™ Plus Hybrid Quadrupole-Orbitrap™ Mass Spectrometer, and separated with a linear gradient of solvent B (1%-99%) over 30 minutes at 0.25 mL/minute. Lipidomic database searching was performed using LipidSearch 4.2.21 (Thermo Fisher Scientific, Waltham, MA, United States). Results were refined by removing peaks predicted to be false positives.
Eight-week-old male ICR mice were procured from the Daehan-Biolink (Eumseong, Korea). The animals (n = 6/group) were maintained at a constant temperature (23 ± 2 °C), relative humidity of 55% ± 10%, and 12-hour light/dark cycle. The animals were fed with standard rodent chow and purified water ad libitum.
For the induction of facilitated aging, the animals were subcutaneously injected with D-Gal (300 mg/kg) for 6 weeks[14,16]. Treatment group mice received IV injections of EVs (6 × 108 EV particles/body) at 2-week intervals, that is, at weeks 0, 2, 4, and 6. In the animal study, we used only hypoxic culture EVs, but not normoxic culture EVs, since the effects of normoxia-cultivated EVs were minimal in the preliminary and previous studies.
Rota-rod performance: Motor balance and coordination were evaluated using a Rota-rod test system (Panlab technology, Barcelona, Spain) on days 1, 7, 21, and 35. Mice were placed on a rotating rod at a constant speed of 4 rpm, and the time taken for the mice to fall off the rod was recorded[19,20]. The endpoint was set to 600 seconds. The average latency was calculated from 3 measurements. Forced swimming performance: To analyze physical stamina, all mice were allowed to swim for 30 minutes for adaptation to a swimming pool with a constant water temperature of 25 ± 0.5 °C for 3 days before the measurement. All mice were subjected to a forced maximum swimming performance on days 2, 8, 22, and 36. The mice were loaded with a lead ring weighing 5% of their body weight attached to the tail, and then placed in the swimming pool. The test was performed by forcing animals to swim until exhaustion, which was determined by observing loss of coordinated movements and failure to return to the water surface within 7 seconds[19,20]. This 7-second cut off time was used as the criterion for the maximum swimming capacity of the animals.
For the evaluation of memory acquisition, the mice were subjected to a passive avoidance Shuttle box on day 43. Passive avoidance performance was assessed by 5 consecutive trials at 1-hour intervals to evaluate memory acquisition and retention. The Shuttle box apparatus consists of two compartments equipped with a lamp and a steel-grid floor for electric shock. On the trials, electric shock (0.5 mA for 2 seconds) was delivered when rats entered the dark compartment from the light room through a guillotine door[17-19]. The latency time of stay in the light room from light-on was recorded. The end-point was set at 300 seconds, denoting full acquisition of memory.
The mice were sacrificed 24 hours (on day 44) after the final forced swimming tests. The brain and gastrocnemius muscles were weighed and homogenized in PBS containing a cocktail of protease inhibitors (Sigma-Aldrich, St. Louis, MO, United States). After centrifugation, the supernatant was measured for ACh concentration with the Amplex Red ACh/AChE assay kit (Molecular Probes, Eugene, OR, United States) according to the manufacturer’s instructions[17-19]. In this assay, ACh is hydrolyzed by AChE to release choline, which is then oxidized by choline oxidase to betaine and H2O2. H2O2 interacts with Amplex Red (7-dihydroxyphenoxazine) in the presence of horseradish peroxidase to generate the highly fluorescent resorufin. The resulting fluorescence was measured in a fluorescence microplate reader using excitation in the range of 530-560 nm and emission at 590 nm.
Brain and gastrocnemius muscle extracts were analyzed using different mouse ELISA kits for the following proteins: BDNF (Antibodies, A1657; Cambridge, United Kingdom), SIRT1 (Abcam, ab206983, United Kingdom), and GFAP (Abcam, ab233621, United Kingdom). Brain tissues were homogenized in a lysis buffer containing protease and phosphatase inhibitors. Samples were centrifuged at 18000 × g for 20 minutes, and the supernatant was collected. All ELISA experiments followed the manufacturer’s protocols[24,25]. The optical densities of the samples were measured using a microplate reader at a wavelength of 450 nm, and the levels were calculated using the standard curves and normalized to the total protein content.
Muscle tissue (30 mg) was hydrolyzed in 0.3 mL of 30% potassium hydroxide solution in a boiling water bath for 30 minutes under intermittent shaking. After cooling to room temperature, 0.1 mL of 1 M Na2SO4 and 0.8 mL of ethanol were added to the sample, and boiled again for 5 minutes. The glycogen pellet was dissolved in 0.2 mL of water, and additional ethanol precipitations were performed twice. The final pellet was dried, dissolved in 0.2 mL of 0.3 mg/mL amyloglucosidase in 0.2 M sodium acetate buffer (pH 4.8), and then incubated for 3 hours at 40 °C. The reaction mixture was added to 0.2 mL of the glucose assay solution containing 0.3 M triethanolamine-potassium hydroxide (pH 7.5), 1 mmol/L ATP, 0.9 mmol/L β-NADP, and 5 μg of G6PDH/mL[26]. The absorbance at 340 nm was determined before and after addition of 1 μg of hexokinase. Glycogen content was expressed as μmol of glucosyl units/g.
Lipid peroxidation was measured by determining the formation of thiobarbituric acid-reactive substances (TBARS) in the gastrocnemius muscles. Briefly, the muscles were homogenized in cold PBS and centrifuged at 4 °C. SDS (500 μL of 8.1% solution) and acetic acid (1 mL of 20% solution; pH 3.5) were added to the muscle homogenate and centrifuged. Aliquots of the clear supernatant were mixed with an equal volume of TBA solution (0.8% w/v) and heated in a glass tube capped with aluminum foil at 95 °C for 30 minutes[20,26]. After cooling on ice, each sample was pipetted into 96-well plates, and the absorbance was read at 532 nm with a microplate reader.
The data were described as mean ± SEM. Statistical significance between the groups was analyzed by one-way analysis of variance with post-hoc tests (n = 6/group) using the SPSS statistical software 30.0.0. (SPSS Inc., Chicago, IL, United States). P-values of less than 0.05 were considered statistically significant.
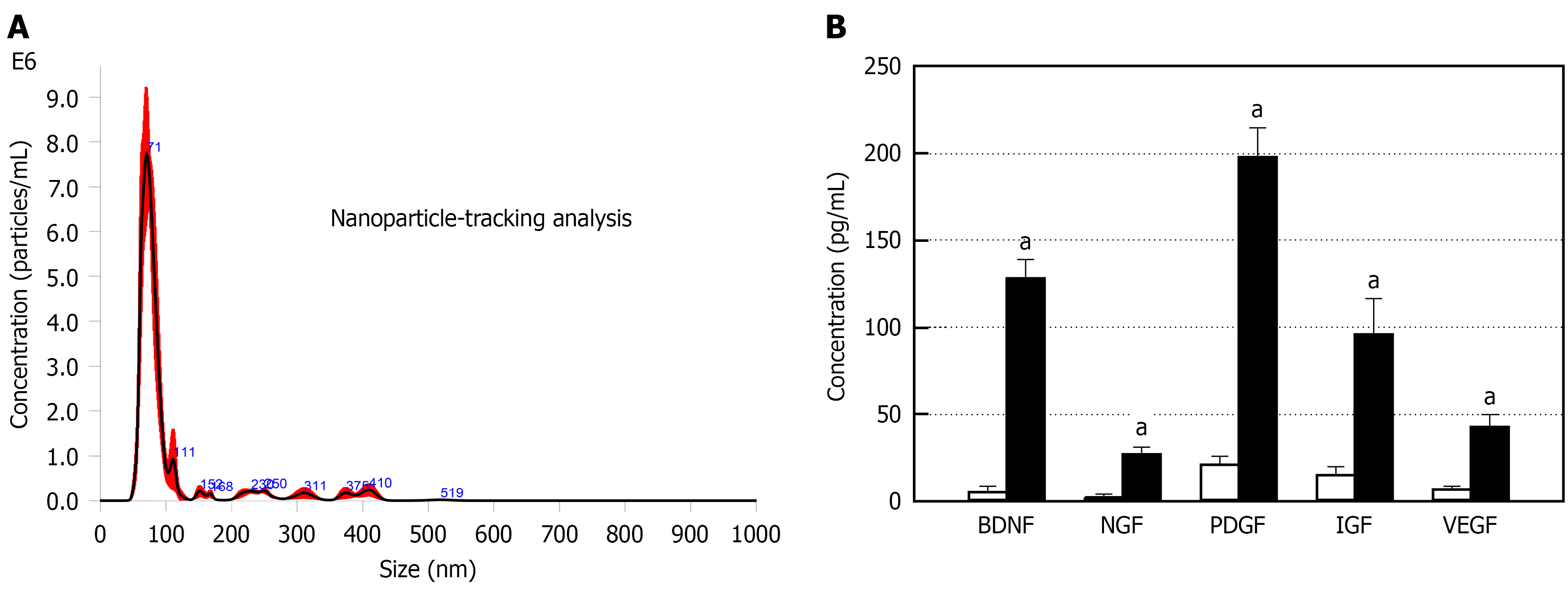
From nanoparticle-tracking analysis of the size distribution of AMSC-EVs, the major peak was observed at 71 nm (Figure 1A). It was found that the exosome particles in EVs were not aggregated or contaminated with larger EVs such as microvesicles and apoptotic bodies, since other considerable peaks were not observed. In ELISA, the concentrations of BDNF, NGF, PDGF, IGF, and VEGF which are neurotrophins related to neuroprotection and neuroregeneration, neuro-muscular adaptation, tissue regeneration, and angiogenesis were very low in normoxic (20% O2) culture medium. By comparison, it was confirmed that the GFs and NFs were very high in hypoxic (2% O2) culture EVs, reaching ten to a hundred times the normoxic culture EVs (Figure 1B).
From proteomic analysis of EVs, total 751 proteins were detected. The top 30 proteins are listed in Table 1, showing rich in cysteine (SPARC), collagen α-1 (I) chain, isoform 1 of fibronectin, filamin-A, collagen α-2 (I) chain, vimentin, collagen α-3 (VI) chain, collagen α-1 (VI) chain, fibrillin, and collagen α-1 (III) chain. Collagens and fibronectins are essential components for restoring tissue integrity from injury, and SPARC plays a critical role for proper assembly and maturation of collagen fibrils. Interestingly, the composition of AMSC-EVs was largely different from that of umbilical cord blood stem cell-derived EVs (UCBSC-EVs), in which isoform 1 of fibronectin, collagen alpha-3 (VI) chain, and collagen alpha-1 (XII) chain were the highest. Notably, CD9, CD63, and CD81 were detected in both AMSC-EVs and UCBSC-EVs, confirming the nature of exosomes.
| Accession | Protein name | Gene name | #PSM | Ratio | UCBSC (reference) | Remark |
| P09486 | SPARC | SPARC | 349 | 2.24% | 0.13% | |
| P02452 | Collagen alpha-1 (I) chain | COL1A1 | 312 | 2.00% | 0.51% | Collagen |
| P02751-1 | Isoform 1 of fibronectin | FN1 | 297 | 1.90% | 3.32% | Cytoskeleton |
| P21333 | Filamin-A | FLNA | 264 | 1.69% | 0.58% | Cytoskeleton |
| P08123 | Collagen alpha-2 (I) chain | COL1A2 | 214 | 1.37% | 0.23% | Collagen |
| P08670 | Vimentin | VIM | 207 | 1.33% | 0.74% | |
| P12111 | Collagen alpha-3 (VI) chain | COL6A3 | 204 | 1.31% | 2.31% | Collagen |
| P12109 | Collagen alpha-1 (VI) chain | COL6A1 | 187 | 1.20% | 0.56% | Collagen |
| P35555 | Fibrillin-1 | FBN1 | 180 | 1.15% | 0.72% | |
| P02461 | Collagen alpha-1 (III) chain | COL3A1 | 168 | 1.08% | 0.14% | Collagen |
| P60709 | Actin, cytoplasmic 1 | ACTB | 154 | 0.99% | 0.92% | |
| Q99715 | Collagen alpha-1 (XII) chain | COL12A1 | 150 | 0.96% | 1.04% | |
| Q15582 | Transforming growth factor-beta-induced protein ig-h3 | TGFBI | 150 | 0.96% | 0.66% | |
| A0A804HK76 | Filamin B | FLNB | 149 | 0.95% | 0.14% | |
| A0A804HII9 | Actinin alpha 1 | ACTN1 | 134 | 0.86% | 0.49% | |
| O00469-2 | Isoform 2 of procollagen-lysine, 2-oxoglutarate 5-dioxygenase 2 | PLOD2 | 131 | 0.84% | 0.12% | |
| P02787 | Serotransferrin | TF | 111 | 0.71% | ND | Iron transporter |
| O43707 | Alpha-actinin-4 | ACTN4 | 110 | 0.70% | 0.50% | |
| P04264 | Keratin, type II cytoskeletal 1 | KRT1 | 109 | 0.70% | 0.25% | |
| P68032 | Actin, alpha cardiac muscle 1 | ACTC1 | 107 | 0.69% | 0.56% | |
| P07585 | Decorin | DCN | 103 | 0.66% | 0.32% | |
| P01033 | Metalloproteinase inhibitor 1 | TIMP1 | 100 | 0.64% | 0.03% | Collagenase inhibitor |
| P08253 | 72 kDa type IV collagenase | MMP2 | 99 | 0.63% | 0.41% | |
| A0A7P0TA35 | Protein disulfide-isomerase | P4HB | 97 | 0.62% | 0.10% | |
| P05121 | Plasminogen activator inhibitor 1 | SERPINE1 | 96 | 0.62% | 0.36% | |
| P26038 | Moesin | MSN | 94 | 0.60% | 0.39% | |
| P09871 | Complement C1s subcomponent | C1S | 91 | 0.58% | 0.29% | |
| Q12841 | Follistatin-related protein 1 | FSTL1 | 91 | 0.58% | 0.06% | |
| P35908 | Keratin, type II cytoskeletal 2 epidermal | KRT2 | 90 | 0.58% | 0.21% | |
| A0A1B0GVI3 | Keratin 10 | KRT10 | 89 | 0.57% | 0.34% | |
| A0A7I2V3T0 | CD9 molecule | CD9 | 16 | 0.02% | 0.06% | Positive marker |
| F8VWK8 | Tetraspanin (fragment) | CD63 | 11 | 0.03% | 0.04% | Positive marker |
| H0YEE2 | CD81 molecule | CD81 | 20 | 0.03% | 0.07% | Positive marker |
From lipidomic analysis of EVs, 15 lipid species were identified (Table 2), wherein phosphatidylcholine (PC, 71.89%), ceramide (Cer, 14.31%), phosphatidylethanolamine (PE, 6.70%), and lysophosphatidylcholine (LPC, 2.37%) were the most abundant lipids, compared to the relatively-low levels of phosphatidylserine (PS), phosphatidylinositol (PI), and sphingomyelin (SM). Such high concentrations of PC, Cer, and PE may give the EV membranes fluidity and stability. In comparison, PC (51.70%), SM (21.44%), PI (10.36%), PS (3.92%), and PE (3.83%) were main lipid components in UCBSC-EVs, in which cell signaling- and apoptosis-related molecules were high.
| Lipid classes | Class | AMSC | UCBSC (reference) |
| Ceramide | Cer | 14.31 | 1.52 |
| Diacylglycerol | DG | 0.90 | 0.53 |
| Monohexosylceramide | HEX1 Cer | 0.18 | 0.38 |
| Dihexosylceramide | HEX2 Cer | ND | 0.05 |
| Lysophosphatidylcholine | LPC | 2.37 | 1.77 |
| Lysophosphatidylglycerol | LPG | 0.00 | ND |
| Lysophosphatidylinositol | LPI | 0.00 | 1.30 |
| Monoglyceride | MG | 1.70 | 0.42 |
| Phosphatidic acid | PA | 0.00 | ND |
| Phosphatidylcholine | PC | 71.89 | 51.70 |
| Phosphatidylethanolamine | PE | 6.70 | 3.83 |
| Phosphatidylglycerol | PG | 0.00 | 0.87 |
| Phosphatidylinositol | PI | 0.00 | 10.36 |
| Phosphatidylserine | PS | 0.00 | 3.92 |
| Sphingomyelin | SM | 0.32 | 21.44 |
| Triacylglycerol | TG | 1.63 | 0.40 |
| Total | 100.00 | 98.49 |
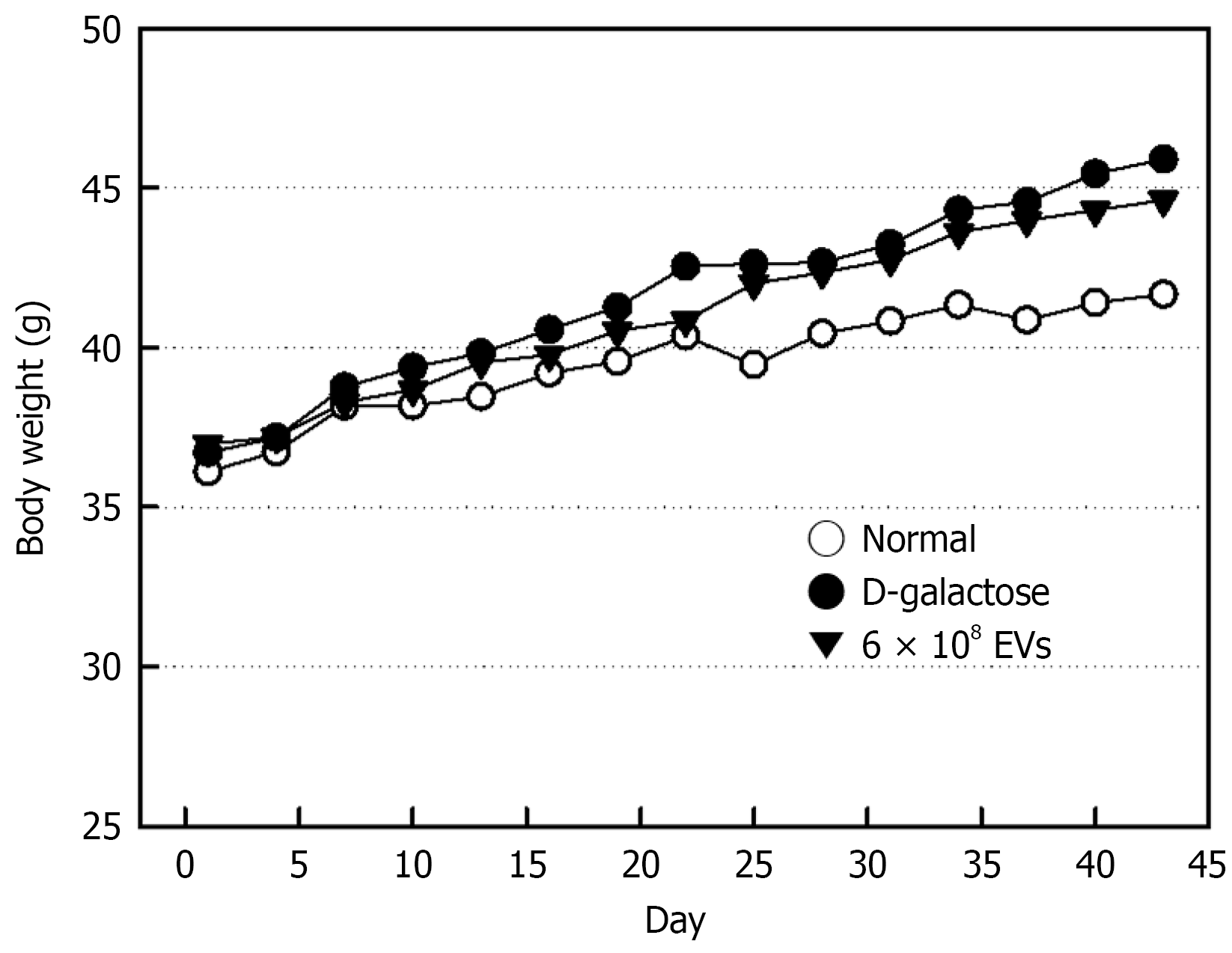
During 6-week treatment with D-Gal, the body weight gain was higher, to some extent, than that of normal animals (Figure 2). The effect of AMSC-EV treatment at 2-week intervals on D-Gal-induced increase was negligible, although the body weight gain was slightly attenuated.
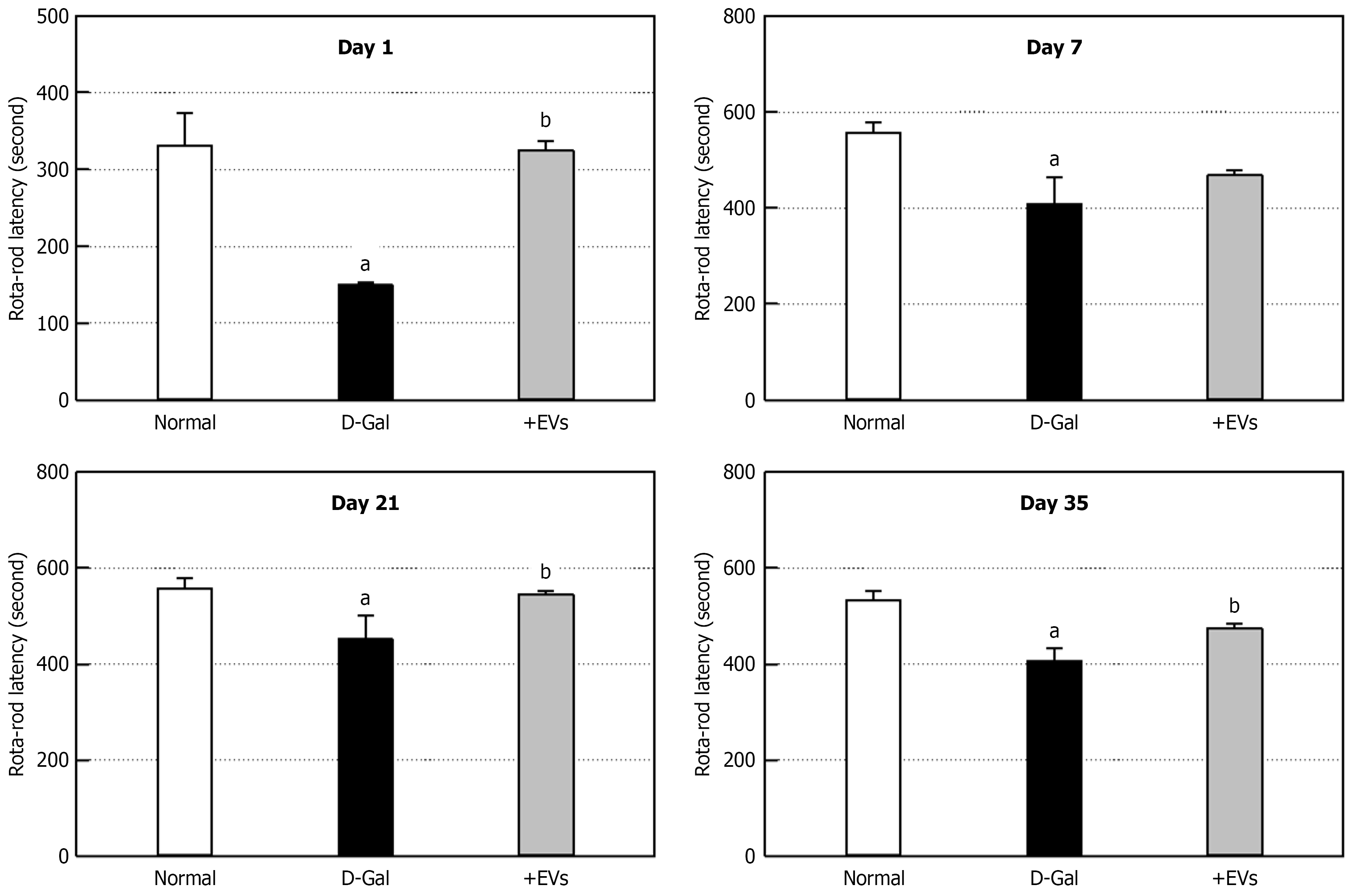
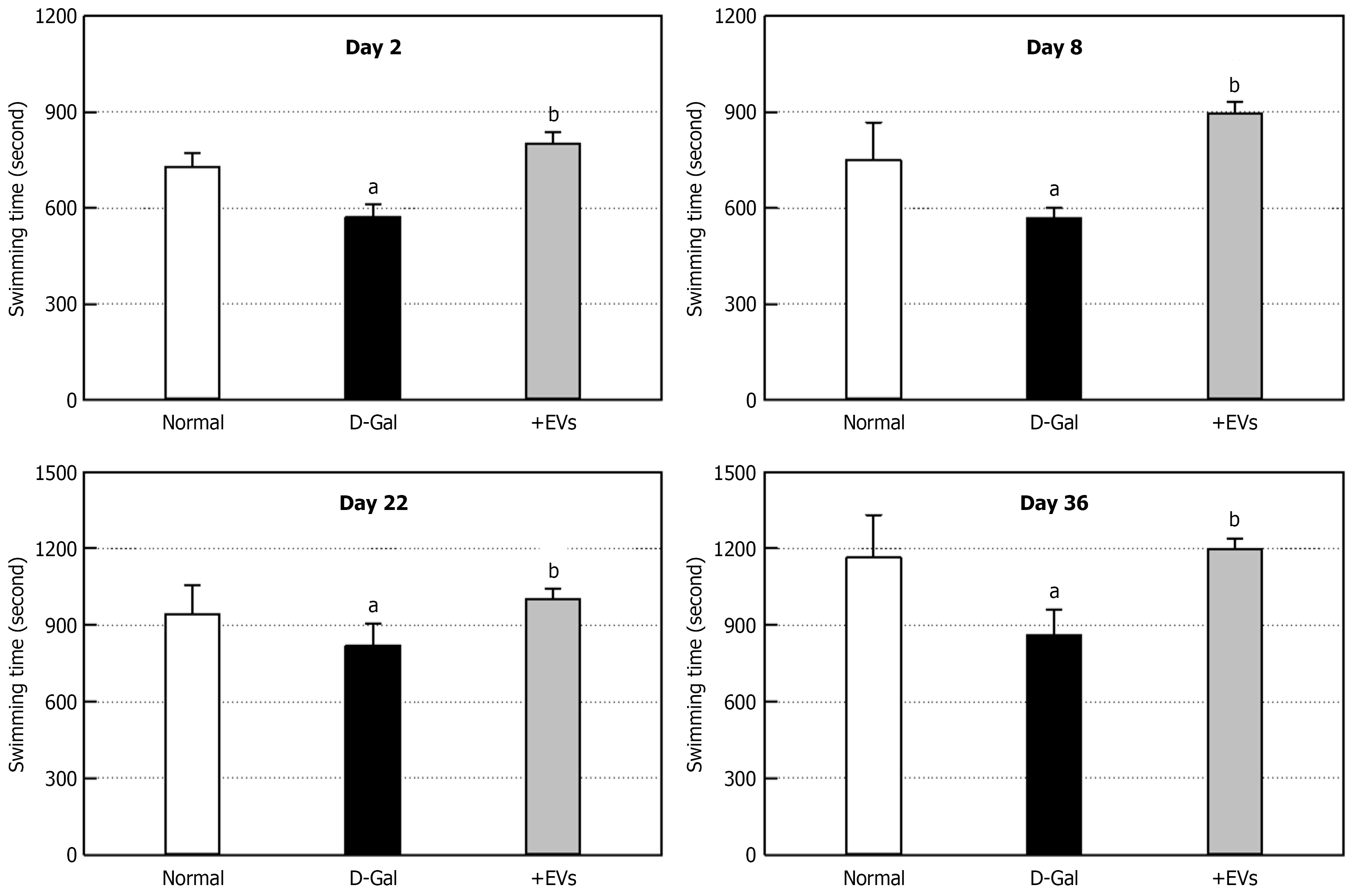
To investigate the effect of EVs on motor balance and coordination, a Rota-rod test was performed. On day 1 post-D-Gal administration (Figure 3), the latency time of D-Gal-administered rats markedly decreased to 45% of normal animals, indicating impaired motor coordination. Such an impaired movement lasted for 35 days (Figure 3). In contrast, the EV treatment remarkably recovered the latency time throughout the experimental period. Similarly, the maximum swimming time of D-Gal-challenged rats significantly decreased, compared to the normal group (Figure 4). However, treatment with EVs led to a great improvement of swimming performance, especially increasing the latency time longer than that of normal animals.
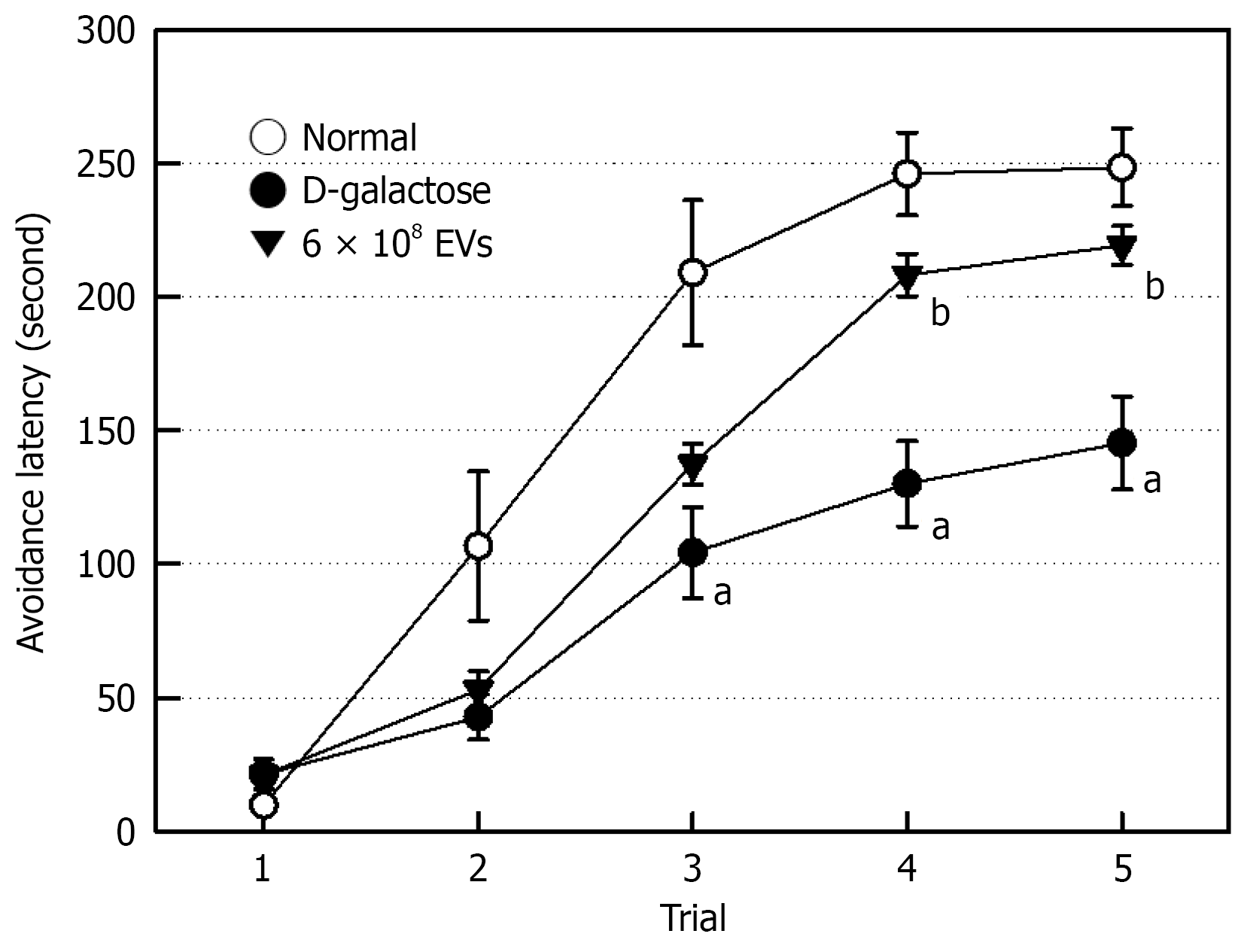
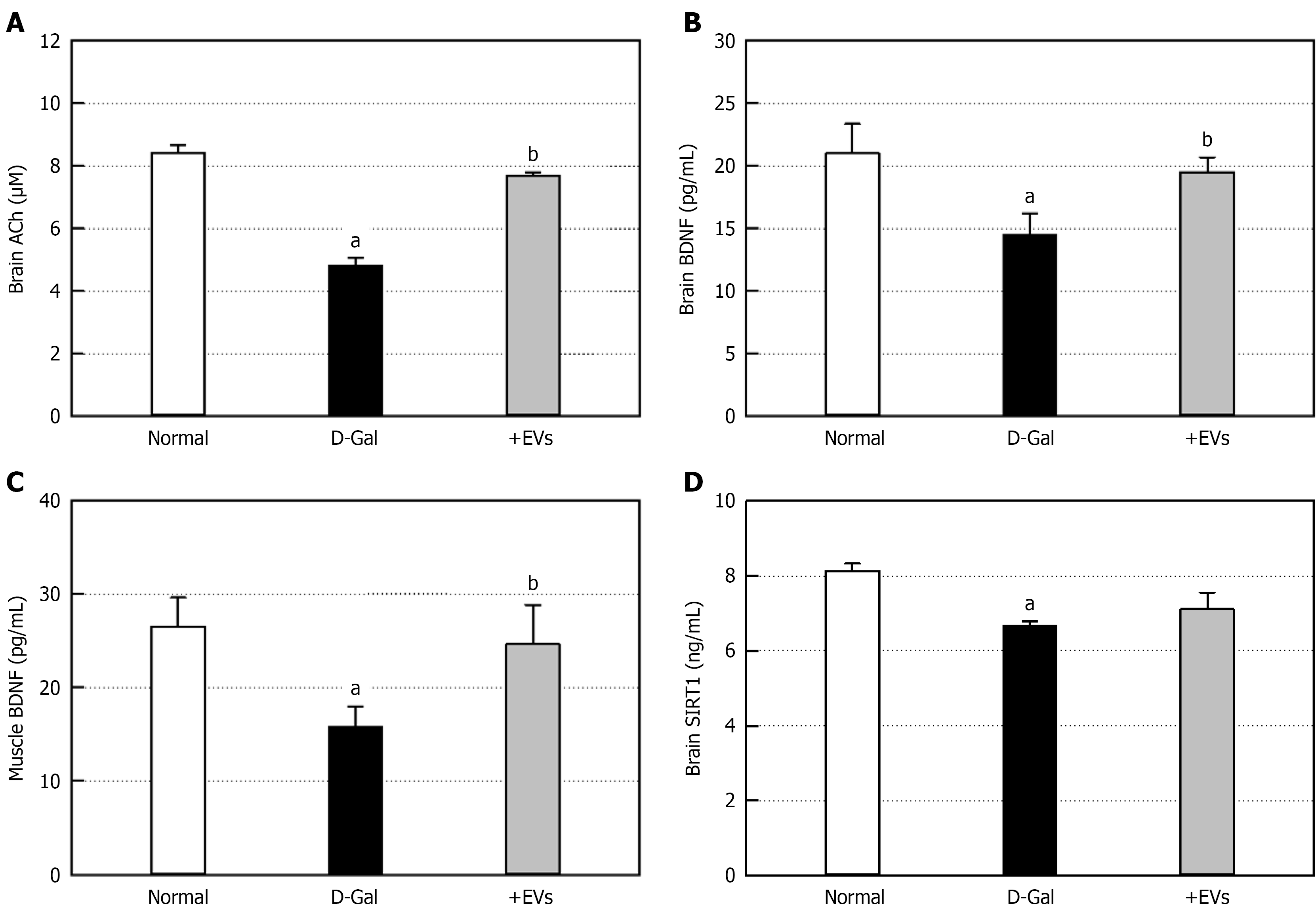
The cognition-enhancing effect of EVs was investigated via a passive avoidance performance. Six-week injection of D-Gal resulted in profound impairment in cognitive tasks, displaying significantly-delayed acquisition of information (Figure 5). Notably, EV treatment improved the learning and memory function. To confirm the relationship between memory deficits and brain ACh level, we analyzed the concentration of ACh, a neurotransmitter governing memory acquisition. Repeated administration of D-Gal decreased ACh concentration in the brain by 43% (Figure 6A), which was significantly restored by EV treatment, that is, near-fully recovering to a normal level.
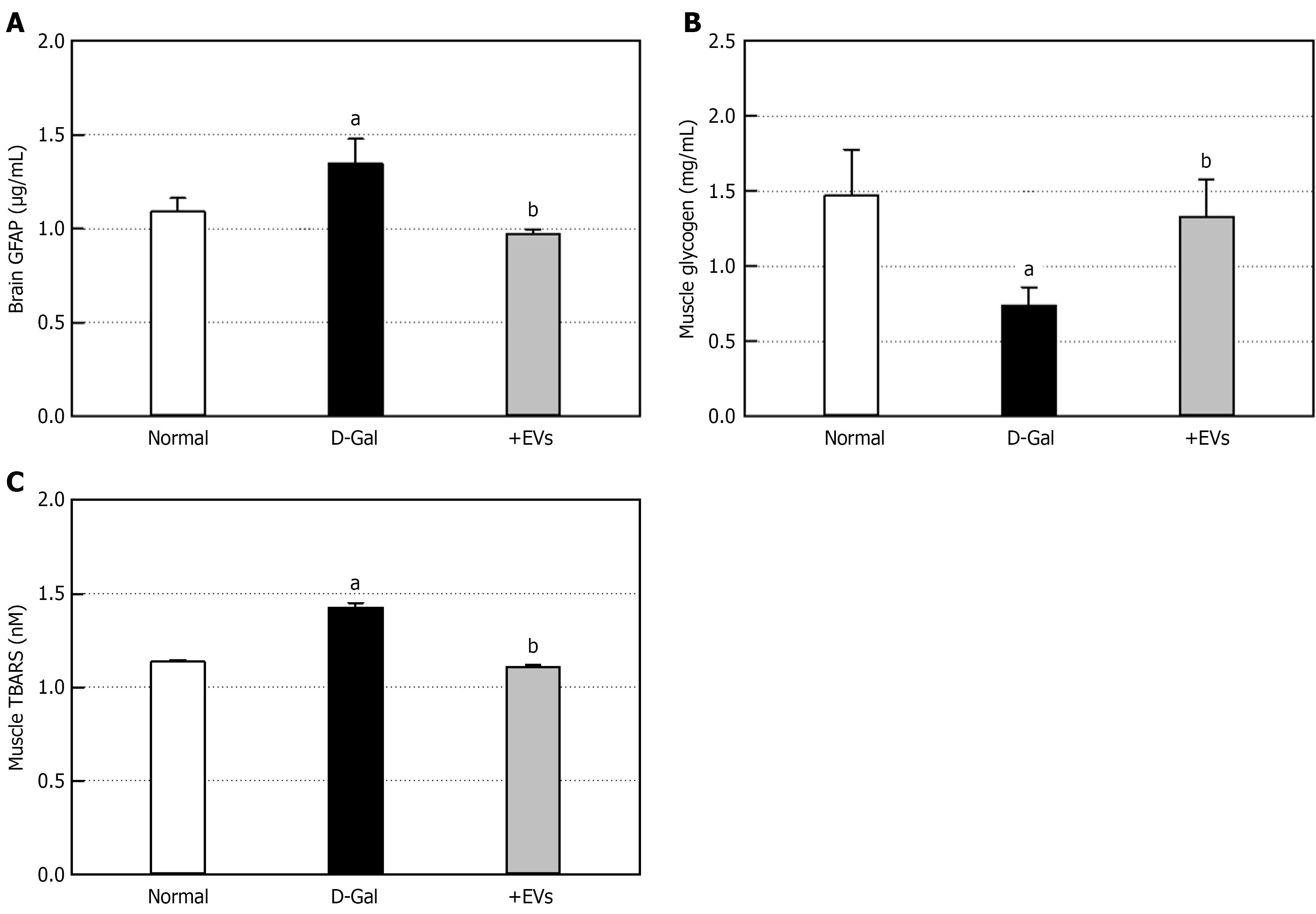
Following D-Gal challenge, the concentrations of brain and muscular BDNF decreased by 31% and 40.4%, respectively (Figure 6B and C). However, EV treatment significantly restored both the brain and muscular BDNF, a key molecule for neuroprotection and neuro-muscular adaptation, up to near-normal levels. The brain level of SIRT1, an anti-aging molecule, was also significantly reduced in the D-Gal-challenged animals. The D-Gal-induced decrease in SIRT1 was attenuated by treatment with EVs to some extent (Figure 6D). Next, we examined astrocytic response by measuring GFAP level, a specific marker of brain injury and aging. The ELISA showed significant increase in the brain GFAP via activation of astrocytes, following 6-week injection of D-Gal (Figure 7A). Notably, the GFAP increase due to brain injury was fully recovered by EV treatment. D-Gal treatment caused marked depletion of glycogen, an energy source, in gastrocnemius muscles to a half level (Figure 7B). However, EV treatment led to a considerable recovery. To assess muscular damage, lipid peroxidation was evaluated using TBARS analysis. The TBARS content in the gastrocnemius muscles of the D-Gal-challenged rats showed a significant increase by 25.4% compared to the normal animals (Figure 7C). In contrast, the EV treatment lowered the TBARS concentration to a normal level, in parallel with the recovery of muscular glycogen (Figure 7B).
It was demonstrated that AMSCs and ADSCs exerted anti-aging effects, improving physical activity, recovering cognitive function, and extending health span and lifespan[19]. Furthermore, ADSCs enhanced physical stamina, which was mediated by neuroprotective and neuro-muscular adaptive activities of GFs and NFs including BDNF[20]. Notably, BDNF-loaded exosomes displayed neuroprotective effects in vitro oxidative stress and in vivo stroke animal models[27].
In the present study, the exosomal nature of EVs was confirmed with specific markers CD9, CD63, and CD81 in proteomic analysis (Table 1)[22]. ELISA revealed that the EVs contain various neurotrophins such as BDNF, NGF, PDGF, IGF, and VEGF (Figure 1). BDNF plays a critical role in synaptic development and plasticity via strengthening excitatory (glutamatergic) synapses[28,29]. Indeed, BDNF released from ADSCs increased physical activity in aged animals[19,20] and over-expression of BDNF led to an early improvement in locomotor function in spinal cord injury model[30]. BDNF acts as a muscle- and endothelial cell-derived NF for motoneurons[24], in which it rescued motoneurons from programmed or injury-induced cell death during development[31]. Furthermore, application of BDNF to the gastrocnemius muscles significant increased electrical excitability of motoneurons[32]. Notably, BDNF can induce fast-twitch (type II) to slow-twitch (type I) transformation of muscle fibers, thus enhancing muscle strength[19,33]. Furthermore, it promotes revascularization[34], and serves a key role in maintaining the population of muscular progenitor cells in adult muscles via regulation of satellite cell differentiation and skeletal muscle regeneration[35,36].
Interestingly, BDNF and NGF were confirmed to play a critical role in the improvement of learning and memory functions[37], as observed in the present study. IGF plays myogenic and angiogenic roles, increasing muscle mass[19,38,39], which may be supported by angiogenesis, muscular adaptation, and tissue protection mediated by VEGF, BDNF, and PDGF[38,40-42], leading to the enhanced stamina in forced-swimming in the present study. In addition, VEGF and IGF enhance antioxidative capacity by up-regulating antioxidant enzymes[38,40,41,43]. Similarly, the oxidative injury of gastrocnemius muscles was significantly attenuated by EV treatment (Figure 7). Therefore, it is believed that the neurotrophins from EVs and preserved brain and muscles contributed to the enhanced stamina and cognitive function.
Although total 751 proteins and 15 lipid species were detected in AMSC-EVs (Tables 1 and 2), it is not reasonable to interpret the roles of each component, since there are too many molecules and relationship among them. In brief, AMSC-EVs were rich in SPARC as well as collagen and fibronectin components. SPARC is a critical cellular matrix protein that regulates the proper assembly and maturation of collagen fibrils. The abundant collagens and fibronectins are essential components for restoring tissue integrity from injury. Interestingly, the composition of AMSC-EVs was largely different from that of UCBSC-EVs, in which several fibronectin and collagen components were the highest. However, it is not possible for us to compare the different natures between the two EVs due to too many factors affecting composition and function.
More interesting point is the lipidomic profile of EVs (Table 2). AMSC-EVs showed a composition of high PC, Cer, and PE concentrations, but relatively-low levels of PS, PI, and SM. The high concentrations of PC, Cer, and PE may play a role for membrane fluidity and stability of the EV membranes, whereas PS, PI, and SM could be signals for apoptosis-related scavenging by inflammatory cells. Notably, PC, SM, PI, and PS were main lipid components in UCBSC-EVs, in which cell signaling- and apoptosis-related molecules are high. It is interesting to note that AMSCs do not express major histocompatibility complex class-II mediating immune response[44], and thereby can avoid immune-related elimination or adverse effects during repeated administration.
However, there are clear limitations in comparing the composition and function of AMSC-EVs with other stem cell EVs, because we obtained data from hypoxia-cultivated AMSC-EVs, but not from hypoxic culture UCBSC-EVs or ADSC-EVs. Actually, hypoxic culture leads to different particle size, composition, and functional molecules in EVs. In addition, content of microRNAs (miRNAs) in EVs and the role of each miRNA remain to be analyzed in a follow-up study, since hundreds of miRNA are included in EVs, and they function as cell and tissue regulators.
Interestingly, the particle size of AMSC-EVs was found to be 71 nm in diameter (Figure 1) which is much smaller than other stem cell EVs[45]. Indeed, it was found that the AMSC exosomes readily penetrated both the normal and damaged keratinocytes, fibroblasts, osteoblasts, and retinal ganglionic cells[22]. Importantly, we demonstrated the fate (distribution and clearance) of fluorescence dye-labeled EVs in vivo: i.e., the EVs readily entered the brain tissue of cerebral palsy animals following IV injection[23]. Therefore, it is believed that the tiny AMSC-EVs can more efficiently deliver functional molecules to the injured tissues including the brain and muscles.
Aging may come from the loss of regenerative capacity, determining the gradual tissue injury, degeneration, and longevity[2]. D-Gal caused significant deficits of physical performances, consistent with previous studies highlighting the impact of aging on motor function and physical stamina[14-16]. As biochemical parameters of tissue injury following facilitated aging, increased brain GFAP and muscular TBARS were confirmed (Figure 7). It was found out that such D-Gal-induced tissue damages came from depletion of brain and muscular BDNF as well as muscular glycogen, an energy source[20,26] (Figures 6 and 7). As expected, AMSC-EVs restored BDNF and glycogen in the brain and muscles, and thereby lowered brain GFAP and attenuated muscular lipid peroxidation. SIRT1 participates in a variety of cellular metabolic processes, exerting anti-aging effects. Indeed, stem cell EVs delayed brain aging through neuroprotective, anti-apoptotic, and antioxidative activities via SIRT1-signaling pathway in senescence-accelerated mice[25]. In the present study, AMSC-EVs restored the brain SIRT1 depleted by D-Gal, and might contributed to the neurobehavioral recovery (Figure 6).
Brain aging leads to cognitive dysfunction, which are closely related to depletion of ACh, a neurotransmitter governing memory acquisition. D-Gal markedly reduced the brain ACh level (Figure 6), and eventually impaired the passive avoidance performance (Figure 5). BDNF and SIRT1 play a central role in the improvement of learning and memory functions, in addition to neuroprotective activity[25,37,46]. Therefore, it is believed that AMSC-EVs recovered ACh by enhancing BDNF and SIRT1 (Figure 6), and thereby improved learning and memory functions.
The enhanced physical and cognitive activities by AMSC-EVs were inferred from the high GFs and NFs, since the neurotrophins are well known to exert tissue-protective and -regenerative activities[19,22]. In addition, AMSC-EV treatment led to a marked increase in muscle glycogen levels, indicating improved metabolic function and energy storage in muscles (Figure 7). Lipid peroxidation was also fully blocked by EVs containing antioxidative BDNF and IGF[38,40,41,43]. Therefore, diverse active ingredients of AMSC-EVs are considered to exert positive effects in reducing muscle fatigue and injury. Indeed, in our previous study, BDNF released from ADSCs enhanced physical stamina and exerted multiple effects including antioxidative activity, glycogen metabolism, muscle type differentiation[20]. Interestingly, it was found that BDNF induces transformation of fast-twitch muscles to slow-twitch fibers, which are resistant to long-term fatigue[20,33]. These findings suggest that AMSC-EVs not only protect the brain function, but may also enhance peripheral tissue activity, contributing to overall physical performances.
Taken together, the analytical results of neurobehavioral functional and biochemical parameters provide compelling evidences that EVs have multi-faceted potentials, effectively mitigating the deleterious effects of D-Gal-induced aging. The observed improvements in motor coordination, physical stamina, and cognitive function, in addition to molecular markers of brain health, suggest that AMSC-EVs may offer a promising strategy to delay aging-related cognitive decline and motor impairments. Although further follow-up studies are necessary to elucidate the underlying mechanisms, it is clear that the AMSC-EVs exert beneficial effects, and could be a candidate for clinical applications to delay aging progress or overcome age-related diseases.
| 1. | Terry RD, Davies P. Dementia of the Alzheimer type. Annu Rev Neurosci. 1980;3:77-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 301] [Cited by in RCA: 255] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 2. | Kirkwood TB. Understanding the odd science of aging. Cell. 2005;120:437-447. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1635] [Cited by in RCA: 1282] [Article Influence: 61.0] [Reference Citation Analysis (0)] |
| 3. | Whitehouse PJ, Price DL, Struble RG, Clark AW, Coyle JT, Delon MR. Alzheimer's disease and senile dementia: loss of neurons in the basal forebrain. Science. 1982;215:1237-1239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2672] [Cited by in RCA: 2453] [Article Influence: 55.8] [Reference Citation Analysis (0)] |
| 4. | Coyle JT, Price DL, DeLong MR. Alzheimer's disease: a disorder of cortical cholinergic innervation. Science. 1983;219:1184-1190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2384] [Cited by in RCA: 2144] [Article Influence: 49.9] [Reference Citation Analysis (0)] |
| 5. | Kása P, Rakonczay Z, Gulya K. The cholinergic system in Alzheimer's disease. Prog Neurobiol. 1997;52:511-535. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 311] [Cited by in RCA: 288] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 6. | Terry AV Jr, Buccafusco JJ. The cholinergic hypothesis of age and Alzheimer's disease-related cognitive deficits: recent challenges and their implications for novel drug development. J Pharmacol Exp Ther. 2003;306:821-827. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 873] [Cited by in RCA: 798] [Article Influence: 34.7] [Reference Citation Analysis (0)] |
| 7. | Musiał A, Bajda M, Malawska B. Recent developments in cholinesterases inhibitors for Alzheimer's disease treatment. Curr Med Chem. 2007;14:2654-2679. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 121] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 8. | Barnes CA. Memory deficits associated with senescence: a neurophysiological and behavioral study in the rat. J Comp Physiol Psychol. 1979;93:74-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1429] [Cited by in RCA: 1539] [Article Influence: 32.7] [Reference Citation Analysis (0)] |
| 9. | Rosenzweig ES, Barnes CA. Impact of aging on hippocampal function: plasticity, network dynamics, and cognition. Prog Neurobiol. 2003;69:143-179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 520] [Cited by in RCA: 562] [Article Influence: 24.4] [Reference Citation Analysis (0)] |
| 10. | Miquel J, Economos AC, Fleming J, Johnson JE Jr. Mitochondrial role in cell aging. Exp Gerontol. 1980;15:575-591. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 681] [Cited by in RCA: 606] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 11. | Floyd RA, Hensley K. Oxidative stress in brain aging. Implications for therapeutics of neurodegenerative diseases. Neurobiol Aging. 2002;23:795-807. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 556] [Cited by in RCA: 558] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 12. | Zeevalk GD, Bernard LP, Song C, Gluck M, Ehrhart J. Mitochondrial inhibition and oxidative stress: reciprocating players in neurodegeneration. Antioxid Redox Signal. 2005;7:1117-1139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 67] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 13. | Reddy PH. Mitochondrial dysfunction in aging and Alzheimer's disease: strategies to protect neurons. Antioxid Redox Signal. 2007;9:1647-1658. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 155] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 14. | Song X, Bao M, Li D, Li YM. Advanced glycation in D-galactose induced mouse aging model. Mech Ageing Dev. 1999;108:239-251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 296] [Cited by in RCA: 322] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 15. | Kumar A, Prakash A, Dogra S. Naringin alleviates cognitive impairment, mitochondrial dysfunction and oxidative stress induced by D-galactose in mice. Food Chem Toxicol. 2010;48:626-632. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 142] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 16. | Lei M, Hua X, Xiao M, Ding J, Han Q, Hu G. Impairments of astrocytes are involved in the d-galactose-induced brain aging. Biochem Biophys Res Commun. 2008;369:1082-1087. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 107] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 17. | Park D, Choi EK, Cho TH, Joo SS, Kim YB. Human Neural Stem Cells Encoding ChAT Gene Restore Cognitive Function via Acetylcholine Synthesis, Aβ Elimination, and Neuroregeneration in APPswe/PS1dE9 Mice. Int J Mol Sci. 2020;21:3958. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 30] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 18. | Park D, Yang YH, Bae DK, Lee SH, Yang G, Kyung J, Kim D, Choi EK, Lee SW, Kim GH, Hong JT, Choi KC, Lee HJ, Kim SU, Kim YB. Improvement of cognitive function and physical activity of aging mice by human neural stem cells over-expressing choline acetyltransferase. Neurobiol Aging. 2013;34:2639-2646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 85] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 19. | Kim D, Kyung J, Park D, Choi EK, Kim KS, Shin K, Lee H, Shin IS, Kang SK, Ra JC, Kim YB. Health Span-Extending Activity of Human Amniotic Membrane- and Adipose Tissue-Derived Stem Cells in F344 Rats. Stem Cells Transl Med. 2015;4:1144-1154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 20. | Yoon EJ, Seong HR, Kyung J, Kim D, Park S, Choi EK, Kim YB, Park D. Stamina-Enhancing Effects of Human Adipose-Derived Stem Cells. Cell Transplant. 2021;30:9636897211035409. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 21. | Zaborowski MP, Balaj L, Breakefield XO, Lai CP. Extracellular Vesicles: Composition, Biological Relevance, and Methods of Study. Bioscience. 2015;65:783-797. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1123] [Cited by in RCA: 976] [Article Influence: 88.7] [Reference Citation Analysis (1)] |
| 22. | Noh CH, Park S, Seong HR, Lee AY, Tsolmon KE, Geum D, Hong SC, Kim TM, Choi EK, Kim YB. An Exosome-Rich Conditioned Medium from Human Amniotic Membrane Stem Cells Facilitates Wound Healing via Increased Reepithelization, Collagen Synthesis, and Angiogenesis. Cells. 2023;12:2698. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 23. | Yoon EJ, Jeong J, Choi Y, Kim DH, Kim TM, Choi EK, Kim YB, Park D. Therapeutic effect of amniotic membrane mesenchymal stem cell-derived exosome-rich conditioned medium in cerebral palsy model. Stem Cells Transl Med. 2026;15:szaf071. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 24. | Cefis M, Chaney R, Quirié A, Santini C, Marie C, Garnier P, Prigent-Tessier A. Endothelial cells are an important source of BDNF in rat skeletal muscle. Sci Rep. 2022;12:311. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 25. | Zhang X, Liu T, Hou X, Zhou Z, Zhang F, Ma H, Wu X, Jiang J. Exosomes secreted by mesenchymal stem cells delay brain aging by upregulating SIRT1 expression. Sci Rep. 2023;13:13213. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 26. | Shin S, Yeon S, Park D, Oh J, Kang H, Kim S, Joo SS, Lim WT, Lee JY, Choi KC, Kim KY, Kim SU, Kim JC, Kim YB. Silk amino acids improve physical stamina and male reproductive function of mice. Biol Pharm Bull. 2010;33:273-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 27. | Zhu ZH, Jia F, Ahmed W, Zhang GL, Wang H, Lin CQ, Chen WH, Chen LK. Neural stem cell-derived exosome as a nano-sized carrier for BDNF delivery to a rat model of ischemic stroke. Neural Regen Res. 2023;18:404-409. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 109] [Reference Citation Analysis (0)] |
| 28. | Kang H, Schuman EM. Long-lasting neurotrophin-induced enhancement of synaptic transmission in the adult hippocampus. Science. 1995;267:1658-1662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 984] [Cited by in RCA: 1090] [Article Influence: 35.2] [Reference Citation Analysis (7)] |
| 29. | McAllister AK, Katz LC, Lo DC. Neurotrophins and synaptic plasticity. Annu Rev Neurosci. 1999;22:295-318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1071] [Cited by in RCA: 1005] [Article Influence: 37.2] [Reference Citation Analysis (0)] |
| 30. | Ziemlińska E, Kügler S, Schachner M, Wewiór I, Czarkowska-Bauch J, Skup M. Overexpression of BDNF increases excitability of the lumbar spinal network and leads to robust early locomotor recovery in completely spinalized rats. PLoS One. 2014;9:e88833. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 51] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 31. | Koliatsos VE, Clatterbuck RE, Winslow JW, Cayouette MH, Price DL. Evidence that brain-derived neurotrophic factor is a trophic factor for motor neurons in vivo. Neuron. 1993;10:359-367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 403] [Cited by in RCA: 427] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 32. | Gonzalez M, Collins WF 3rd. Modulation of motoneuron excitability by brain-derived neurotrophic factor. J Neurophysiol. 1997;77:502-506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 69] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 33. | Kubis HP, Hanke N, Scheibe RJ, Meissner JD, Gros G. Ca2+ transients activate calcineurin/NFATc1 and initiate fast-to-slow transformation in a primary skeletal muscle culture. Am J Physiol Cell Physiol. 2003;285:C56-C63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 62] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 34. | Kermani P, Rafii D, Jin DK, Whitlock P, Schaffer W, Chiang A, Vincent L, Friedrich M, Shido K, Hackett NR, Crystal RG, Rafii S, Hempstead BL. Neurotrophins promote revascularization by local recruitment of TrkB+ endothelial cells and systemic mobilization of hematopoietic progenitors. J Clin Invest. 2005;115:653-663. [PubMed] [DOI] [Full Text] |
| 35. | Clow C, Jasmin BJ. Brain-derived neurotrophic factor regulates satellite cell differentiation and skeltal muscle regeneration. Mol Biol Cell. 2010;21:2182-2190. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 103] [Cited by in RCA: 145] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 36. | Mousavi K, Jasmin BJ. BDNF is expressed in skeletal muscle satellite cells and inhibits myogenic differentiation. J Neurosci. 2006;26:5739-5749. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 161] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 37. | Blurton-Jones M, Kitazawa M, Martinez-Coria H, Castello NA, Müller FJ, Loring JF, Yamasaki TR, Poon WW, Green KN, LaFerla FM. Neural stem cells improve cognition via BDNF in a transgenic model of Alzheimer disease. Proc Natl Acad Sci U S A. 2009;106:13594-13599. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 718] [Cited by in RCA: 647] [Article Influence: 38.1] [Reference Citation Analysis (1)] |
| 38. | Sadat S, Gehmert S, Song YH, Yen Y, Bai X, Gaiser S, Klein H, Alt E. The cardioprotective effect of mesenchymal stem cells is mediated by IGF-I and VEGF. Biochem Biophys Res Commun. 2007;363:674-679. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 229] [Cited by in RCA: 226] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 39. | Sonntag WE, Lynch C, Thornton P, Khan A, Bennett S, Ingram R. The effects of growth hormone and IGF-1 deficiency on cerebrovascular and brain ageing. J Anat. 2000;197 Pt 4:575-585. [PubMed] [DOI] [Full Text] |
| 40. | Ferrara N. Molecular and biological properties of vascular endothelial growth factor. J Mol Med (Berl). 1999;77:527-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 828] [Cited by in RCA: 806] [Article Influence: 29.9] [Reference Citation Analysis (0)] |
| 41. | Rosenstein JM, Mani N, Silverman WF, Krum JM. Patterns of brain angiogenesis after vascular endothelial growth factor administration in vitro and in vivo. Proc Natl Acad Sci U S A. 1998;95:7086-7091. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 161] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 42. | Sakuma K, Yamaguchi A. The recent understanding of the neurotrophin's role in skeletal muscle adaptation. J Biomed Biotechnol. 2011;2011:201696. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 85] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 43. | Abid MR, Schoots IG, Spokes KC, Wu SQ, Mawhinney C, Aird WC. Vascular endothelial growth factor-mediated induction of manganese superoxide dismutase occurs through redox-dependent regulation of forkhead and IkappaB/NF-kappaB. J Biol Chem. 2004;279:44030-44038. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 86] [Article Influence: 3.9] [Reference Citation Analysis (1)] |
| 44. | Kang D, Kang MJ, Kong D, Lee JE, Lee AY, Geum DH, Kim BS, Kim YS, Hong SC. Effect of Human Amniotic Epithelial Stem Cell Transplantation on Preterm Premature Rupture of Fetal Membrane Using the Amniotic Pore Culture Technique in vitro. Gynecol Obstet Invest. 2022;87:333-343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 45. | Shin KO, Ha DH, Kim JO, Crumrine DA, Meyer JM, Wakefield JS, Lee Y, Kim B, Kim S, Kim HK, Lee J, Kwon HH, Park GH, Lee JH, Lim J, Park S, Elias PM, Park K, Yi YW, Cho BS. Exosomes from Human Adipose Tissue-Derived Mesenchymal Stem Cells Promote Epidermal Barrier Repair by Inducing de Novo Synthesis of Ceramides in Atopic Dermatitis. Cells. 2020;9:680. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 93] [Cited by in RCA: 144] [Article Influence: 24.0] [Reference Citation Analysis (0)] |
| 46. | Michán S, Li Y, Chou MM, Parrella E, Ge H, Long JM, Allard JS, Lewis K, Miller M, Xu W, Mervis RF, Chen J, Guerin KI, Smith LE, McBurney MW, Sinclair DA, Baudry M, de Cabo R, Longo VD. SIRT1 is essential for normal cognitive function and synaptic plasticity. J Neurosci. 2010;30:9695-9707. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 458] [Cited by in RCA: 443] [Article Influence: 27.7] [Reference Citation Analysis (1)] |