INTRODUCTION
The human umbilical cord (UC) facilitates the transport of life-support essentials between the baby and the mother. The cord is composed of vessels surrounded by Wharton’s jelly (WJ), which reinforces and provides protection. WJ is increasingly popular as a potent source of mesenchymal stem cells (MSCs). An increasing amount of research is now being conducted to explore the optimal dosages, routes of administration, and possible medical conditions for which WJ-derived MSCs may be used[1].
Extracting these MSCs from WJ does not require any special surgical process; the cords that are usually thrown away after a healthy baby has been delivered are screened and can be processed to remove a good number of cells, which can be further cultured in a lab[2]. The benefits this structure provides to the fetus while still in the mother, as well as the potential benefits after delivery, make it an interesting research topic[3,4].
This study highlights the biological structure and function, biomechanical properties, and regeneration potential of WJ, with particular emphasis on WJ-acquired MSCs (WJ-MSCs) and their clinical translation. Recent studies indicate that WJ-MSCs are a better source compared to adult-tissue-derived stem cells owing to their capacity for immunomodulation, differentiation, and high proliferation. In this review, the authors aim to investigate the latest developments in regenerative medicine using WJ-MSCs and their potential applications in combination with other emerging therapies.
WJ-DERIVED MSCS
Isolation and characterization
WJ-MSCs may be isolated using several methods, including enzymatic digestion, explant culture, and mechanical dissociation. Recently published studies demonstrated that explant culture methods yield cells with greater proliferative capacity and relatively basic phenotype compared with enzymatic isolation. However, this approach requires longer initial culture periods[24]. Current consensus guidelines recommend isolation protocols that minimize enzymatic exposure while maximizing cell recovery and viability. Recent research compared collagenase digestion with tissue explant methods, showing that explant-derived cells reached confluence 5-7 days later but exhibited 23% higher expression of pluripotency markers (OCT4, NANOG) and maintained superior proliferation through 15 passages[24].
Phenotypically, WJ-MSCs satisfy standard MSC criteria established by the International Society for Cellular Therapy, expressing surface markers CD73, CD90, and CD105 while lacking expression of hematopoietic markers CD34, CD45, and HLA-DR[15]. WJ-MSCs also express embryonic stem cell markers, including SSEA-4, Oct-4, and Sox-2 at higher levels than bone marrow-derived MSCs, suggesting greater developmental plasticity[25,26]. Additionally, WJ-MSCs express cell adhesion molecules CD44 and CD166, which facilitate cell-matrix and cell-cell interactions and are important for therapeutic applications[27].
Proliferative capacity and senescence
In their molecular study, Heo et al[28] investigated characteristics of human MSCs from different sources. They used the enzymatic isolation techniques and observed that all tested sources had similar doubling times for the first two days and increased proliferation of placental MSCs for the following two days compared with other sources. However, this decreased doubling time was not statistically significant. Widowati et al[29] conducted a molecular investigation to assess how explant and enzymatic methods affect isolated WJ-MSCs. Based on the protocols followed by Widowati et al[29], they observed the shortest doubling times with both methods at the fifth passage.
Widowati et al[29] noted that MSCs began to change their shape to a more elliptical morphology in the 8th passage for both the enzymatic and explant methods of isolation. They observed a high expression of CD90 markers in isolated MSCs which is known for its role in differentiation and in controlling cancer cells through programmed cell death. Long-term expansion studies have shown that WJ-MSCs can undergo several population doublings before reaching senescence[24,30]. Previously published senescence marker analyses have revealed that WJ-MSCs accumulate senescence-associated markers (p16, p21, and beta-galactosidase activity) more slowly during extended culture compared to other MSC sources[31]. A previously conducted preclinical study demonstrated that WJ-MSCs maintain a stable profile when exposed to oxidative stress response pathways[32]. These findings support the use of WJ-MSCs for applications requiring extensive ex vivo expansion.
Differentiation potential
WJ-MSCs demonstrate the ability to undergo multilineage differentiation (bone, cartilage, or fat cells) and readily differentiate under appropriate conditions. Researchers noted the ability of WJ-MSCs to produce cartilage-like tissues with higher GAG content and collagen II expression than bone marrow MSCs[33]. This ability has generated an increased interest in WJ-MSC applications for cartilage repair and osteoarthritis treatment. Under chondrogenic culture conditions, WJ-MSCs commonly produce ECM components such as type II collagen and aggrecan, along with increased proteoglycan deposition detectable by alcian blue or safranin-O staining. These findings confirm activation of chondrogenic transcriptional programs, yet they do not necessarily demonstrate the formation of structurally and functionally equivalent articular cartilage[34].
Expression of cartilage-associated markers represents early or intermediate stages of chondrogenic differentiation, and the resulting cell populations often resemble chondrocyte-like cells rather than fully mature cartilage-forming cells capable of maintaining long-term cartilage architecture. Additionally, MSC differentiation capacity can vary depending on culture conditions, passage number, and signaling cues, and marker expression alone does not guarantee robust tissue formation[35]. Accordingly, most studies demonstrating WJ-MSC chondrogenesis should be interpreted as evidence of inducible lineage commitment in vitro, while long-term cartilage regeneration and durable integration into native cartilage tissue require further validation in vivo.
Studies have reported successful differentiation of WJ-MSCs toward neural lineages, producing cells that express neuronal markers, including nestin, beta-III tubulin, and neurofilament proteins[36,37]. Although WJ-MSCs can adopt neuron-like morphology and express neural markers such as nestin, GFAP, β-III-tubulin, or MAP2 under induction conditions, these observations should not be interpreted as evidence of fully functional neuronal differentiation. Expression of lineage-associated markers primarily reflects partial lineage commitment or transcriptional activation, rather than the acquisition of mature neuronal electrophysiological properties. Many differentiation protocols rely on short-term chemical induction and therefore evaluate early phenotypic conversion rather than long-term stability of the neural phenotype. For example, studies assessing neurogenic induction frequently demonstrate marker expression within hours to days but do not examine whether the phenotype persists after withdrawal of inducing stimuli or during extended culture[38,39].
Furthermore, increasing evidence suggests that the therapeutic benefits of WJ-MSCs in neural injury models may arise primarily from paracrine signaling and immunomodulatory mechanisms rather than true neuronal replacement. Experimental studies indicate that MSC-mediated neuroregeneration is often attributable to trophic factor secretion and neuroprotective effects rather than durable integration as mature neurons within host neural circuits[40]. Consequently, current evidence supports the interpretation that neurogenic differentiation of WJ-MSCs in vitro represents lineage-biased phenotypic induction, while stable neuronal maturation and long-term functional integration in vivo remain incompletely demonstrated.
Preclinical studies report hepatocyte-like differentiation of WJ-MSCs, demonstrating glycogen storage and urea production, with induced cells expressing albumin and alpha-fetoprotein, along with other markers[41,42]. While the functional maturity of these differentiated cells requires further validation, these findings suggest developmental plasticity exceeding typical MSC multipotency. Induction of WJ-MSCs toward hepatocyte-like cells is typically demonstrated by morphological changes and the expression of hepatic genes such as albumin, alpha-fetoprotein, HNF4A, CK18, and G6P after exposure to hepatogenic media. While these markers indicate progression toward an endodermal lineage, gene expression alone does not necessarily confirm the generation of fully mature hepatocytes capable of complex metabolic functions, including complete detoxification pathways or long-term albumin synthesis comparable to primary hepatocytes[43].
Most hepatic differentiation protocols evaluate cells after short induction periods (approximately 2-3 weeks) in vitro, which primarily reflects partial lineage specification rather than definitive hepatocyte maturation. Moreover, although hepatocyte-like cells derived from WJ-MSCs can display some functional properties in vitro, their capacity for long-term engraftment, stable hepatic functionality, and durable tissue replacement in vivo remains under active investigation[43]. Therefore, the available literature indicates that WJ-MSC hepatic differentiation models should be interpreted as in vitro lineage induction systems, useful for disease modeling and toxicity testing, but not yet equivalent to fully mature hepatocyte replacement therapies.
A 2023 study identified distinct subpopulations within WJ with varying lineage commitment, including subsets exhibiting vascular, fibroblastic, or pericyte-like transcriptional signatures[44]. These findings suggest that WJ contains heterogeneous cell populations at different stages of differentiation rather than a homogeneous stem cell population, which may influence therapeutic outcomes and manufacturing consistency.
Immunomodulatory properties
The immunomodulatory capabilities of WJ-MSCs are among their most therapeutically relevant characteristics. These cells suppress T-cell proliferation, inhibit B-cell differentiation, modulate natural killer cell activity, and promote the generation of regulatory T cells. Studies have demonstrated that WJ-MSCs inhibit T-cell activation through both cell-contact-dependent mechanisms and the paracrine secretion of immunosuppressive factors, including indoleamine 2,3-dioxygenase, prostaglandin E2, TGF-β, and HGF[45-47].
Karimi et al[48] found WJ-MSCs to be effective in reducing activity (causing cell death and shrinkage) in a variety of cancer cell lines, including ovarian, breast, and osteosarcoma cells. Gene expression analyses revealed that WJ-MSCs constitutively express higher levels of immunomodulatory factors, potentially explaining their high suppressive capacity; it was also noted that donor age may be an important factor in the immunomodulatory properties of WJ-MSCs[49].
The low immunogenicity of WJ-MSCs supports their use in allogeneic therapeutic applications. Previously published studies demonstrated that WJ-MSCs express minimal HLA class I molecules and lack HLA class II expression, thereby reducing recognition by recipient immune systems[46]. Additionally, WJ-MSCs express immune checkpoint molecules, including PD-L1 and PD-L2, that further suppress allogeneic immune responses[50]. Clinical trials using allogeneic WJ-MSCs have reported minimal adverse immune reactions, validating the clinical relevance of these immunological properties.
Paracrine mechanisms and secretome
Recently published preclinical studies indicate that the therapeutic effects of WJ-MSCs may primarily be attributed to paracrine mechanisms rather than direct cellular engraftment and differentiation. These mechanisms involve a multitude of secreted proteins, including growth factors, cytokines, chemokines, and ECM proteins[46]. Vascular endothelial growth factor (VEGF), basic fibroblast growth factor, HGF, insulin-like growth factor-1, and brain-derived neurotrophic factor are some of the key factors[46]. These factors promote angiogenesis, reduce apoptosis, modulate inflammation, and stimulate endogenous tissue repair.
Previously conducted comparative analyses report higher concentrations of pro-angiogenic and neurotrophic factors in WJ-MSC secretomes compared with those from adult MSC sources, potentially explaining superior therapeutic outcomes in some applications[51]. They observed elevated levels of cytokines and growth factors and inferred that WJ-MSCs secreted 2.1-fold higher VEGF, 1.8-fold higher basic fibroblast growth factor, and 3.2-fold higher HGF than bone marrow MSCs under identical culture conditions[51].
Previously published research has shown that WJ-MSC-derived exosomes contain microRNAs, mRNAs, and proteins that regulate recipient cell behavior[52,53]. These vesicles promote endothelial cell migration and tube formation, protect neurons from oxidative stress, and reduce inflammatory responses in target tissues[15]. The therapeutic potential of WJ-MSC-derived extracellular vesicles has generated significant interest as a cell-free alternative to cellular therapies.
Antimicrobial and antiviral properties
Recently published research has identified antiviral properties of WJ-MSCs, whereby they secrete interferon-beta upon inflammatory stimulation[54]. WJ-MSCs have demonstrated protective effects in viral infection models by reducing viral replication and attenuating inflammatory responses associated with viral pathogenesis. These properties have generated interest in WJ-MSC applications for viral infections, including coronavirus disease 2019 (COVID-19), with multiple clinical trials investigating this therapeutic approach[55,56].
REGENERATIVE MEDICINE APPLICATIONS
Wound healing and tissue repair
WJ-MSCs have been studied for their regenerative use in wound healing because of their ability to secrete bioactive molecules that affect tissue repair[15]. This secretome carries cytokines, extracellular vesicles, and growth factors that regulate inflammation, promote angiogenesis, and stimulate the proliferation and migration of fibroblasts and keratinocytes[15,57]. In their in vitro study, Yu et al[57] investigated the differences in functionality of exosomes extracted from WJ and adipose tissue by assessing the results from iTRAQ gel-free protein data. They observed that some proteins were more prevalent in exosomes derived from adipose tissue, while others were more prevalent in WJ-derived exosomes. They also conducted routine pathway and proliferation tests. WJ-MSC-derived exosomes were observed to perform better than exosomes of adipose origin in fibrinogen, coagulation, and especially in terms of proliferation and the migration of keratinocytes[57]. Recent preclinical investigations have demonstrated the potential of WJ-MSCs in a PF-127 hydrogel combined with sodium ascorbyl phosphate, which improved wound closure in a murine model of type 2 diabetes[58]. High dermal regeneration and collagen deposition were observed. Furthermore, this treatment promoted M2 tissue-healing, anti-inflammatory macrophages and enhanced neovascularization at the wound site[58]. Additionally, Kishta et al[59] conducted a randomized controlled trial (n = 110) in patients with diabetic foot ulcers to investigate the impact of the topical application of WJ-MSC-derived exosomes in combination with standard care. The researchers used SINBAD and WIFI systems (site, ischemia, neuropathy, bacterial infection, area, depth systems and wound, ischemia, and foot infection) along with measurement of size using photographs to ascertain the classification and outcome measures. The size of the ulcers and adverse events were noted periodically at 2 weeks, 4 weeks, 6 weeks and then again at 2 months, 4 months, 6 months. They noted improved wound closure, a shorter time to complete epithelialization and no serious adverse events[59]. It took approximately 6 weeks for the ulcer to heal in the treatment group, compared with an average of 20 weeks in the control group.
Myocardial infarction
Myocardial infarction is a major cause of mortality worldwide, as it results in irreversible cardiomyocyte loss, fibrotic remodeling, and inflammation[60]. Despite advancements in reperfusion therapies, these approaches do not regenerate damaged myocardium; consequently, many patients develop chronic heart failure following an infarction. This limited regenerative capacity of adult cardiac tissue has led researchers to switch to stem cell-based therapies. WJ-MSCs are gaining popularity owing to their immunomodulatory and pro-angiogenic properties. Researchers note that WJ-MSCs produce high levels of growth factors that support angiogenesis and reduce inflammatory responses in ischemic tissues. These paracrine effects help preserve viable myocardium following infarction[61]. Collectively, it was observed that WJ-MSCs improve myocardial perfusion, attenuate adverse remodeling, and can be administered safely in cardiovascular settings.
A preclinical study investigated the role of WJ-MSCs in improving functional and structural outcomes after myocardial infarction[62]. The authors observed that combining a decellularized WJ matrix hydrogel with intramyocardial delivery of WJ-MSCs improved cardiac function and cardiomyogenesis, promoted microvascular renewal, and reduced scar formation. These findings suggest that WJ-MSCs can exert therapeutic effects through paracrine signaling, thereby creating a regenerative microenvironment in the injured myocardium[61,62].
WJ-MSC-derived extracellular vesicles contain bioactive molecules that modulate inflammation, promote angiogenesis, and enhance myocardial regeneration. A preclinical model demonstrated improved ejection fraction and other performance metrics, indicating therapeutic effects without direct cell engraftment[63]. Recent clinical studies further support the cardioprotective effects of WJ-MSCs. Intravenous infusion of WJ-MSCs in patients with cardiac failure was investigated[64]. In their phase 1/2 randomised controlled trial, the researchers investigated 30 participants, out of which 15 received a dose of 1 × 106 cells/kg. The participants were followed from baseline to 3 months, 6 months, and 12 months for safety and echocardiography. Improvements in overall quality of life and left ventricular function were observed[64].
Orthopedic applications (knee, soft tissue, and cartilage repair)
Degenerative orthopedic conditions, such as knee osteoarthritis, are influenced by inflammation, cartilage degradation, and limited intrinsic repair capacity. These conditions can cause disability and chronic pain, reducing the quality of life. Current treatments focus mainly on symptom management rather than tissue regeneration. As a result, regenerative approaches capable of creating cartilage structure have been gaining attention. Intra-articular administration of WJ-MSCs has resulted in improvements in functional and pain scores, especially in subchondral sclerosis, in patients with knee osteoarthritis[65].
Mechanistic studies have examined the role of WJ-MSCs in cartilage and soft-tissue repair. Encapsulation of WJ-MSCs in gelatin microspheres was observed to enhance cartilage regeneration in mouse models of osteoarthritis[66]. The joints treated with WJ-MSC showed increased GAG content and improved histological cartilage scores. A recent preclinical study demonstrated that WJ-MSC-derived exosomes improve cartilage repair by activating the TGF-β/Smad signaling pathway[67]. The researchers observed improved cartilage thickness and surface integrity in rabbits with microfractures. Thus, WJ-MSCs represent a promising regenerative strategy for orthopedic conditions.
COVID-19
COVID-19 is a respiratory disease caused by the severe acute respiratory syndrome coronavirus 2 virus, which rapidly became a global pandemic in 2019. This disease led to a large number of severe cases and deaths worldwide. Standard treatments include antiviral agents, supportive care, and corticosteroids. However, these approaches do not eliminate the risk of severe inflammation and long-term complications.
Clinical studies investigating WJ-MSC-derived extracellular vesicles in patients with COVID-19 and comorbidities, including diabetes mellitus and kidney disease, observed decreased nuclear factor kappa B signaling and cytokine production[68]. A recently conducted phase 1 clinical trial reported three intravenous administrations of WJ-MSCs in patients with severe COVID-19, which increased anti-inflammatory cytokines (IL-10 and stromal cell-derived factor 1)[69]. The study also demonstrated decreased levels of pro-inflammatory markers (IL-6 and TNF-α) and improvements in lung function[69].
Previously conducted studies determined that the secretome of WJ-MSCs significantly inhibits severe acute respiratory syndrome coronavirus 2 infection and replication, reducing viral activity in cell cultures by over 90% and demonstrating potential direct antiviral effects beyond immunomodulation[70]. Researchers note a significant overall survival advantage in patients with COVID-19 treated with WJ-MSC-based therapies compared with controls, underscoring their importance in the management of this disease[71].
Neurological diseases (Alzheimer’s disease and Parkinson’s disease)
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder that causes memory loss, cognitive decline, chronic neuroinflammation, amyloid-β plaque accumulation, and tau pathology. Neuroinflammation is mainly caused by activated microglia and elevated cytokines such as IL-1β and TNF-α, which play a major role in disease progression[72]. Preclinical studies indicate that WJ-MSC administration improves learning and memory outcomes in animal models of AD[72]. Furthermore, these cognitive benefits have been heavily associated with reduced amyloid-β burden and suppressed microglial activation. WJ-MSCs also increase anti-inflammatory cytokines, such as IL-10, which shifts the brain environment toward neuroprotection[73]. WJ-MSCs appear to have clinical benefits, slowing the progression of AD by targeting inflammation and neuronal dysfunction.
Parkinson’s disease (PD) is a neurodegenerative disorder that causes progressive loss of dopaminergic neurons in the substantia nigra, which causes motor and cognitive impairment. Current treatments, such as levodopa, provide only symptomatic relief and do not halt dopaminergic neuron degeneration[74]. As a result, regenerative approaches that modulate neuroinflammation and support neuronal survival have been seen as more promising.
Clinical studies in 6-OHDA-induced murine models of PD reported that systemic transplantation of WJ-MSCs improves motor function and restores hippocampal long-term potentiation, indicating increased neural connectivity[75]. The increased neuroprotection and restored neural function may be attributed to higher levels of neurotrophins, such as brain-derived neurotrophic factor and nerve growth factor, which are crucial for dopaminergic neuron survival.
MANUFACTURING AND CLINICAL TRANSLATION CONSIDERATIONS
Isolation and culture standardization
Past studies have demonstrated that cell isolation methods, culture media formulations, and passage protocols significantly influence cellular characteristics and therapeutic potency[24]. Scientific societies and regulatory bodies aim to establish standardized protocols to ensure product consistency and reproducibility. Key manufacturing considerations include enzymatic vs explant isolation methods, with studies showing trade-offs between yield, processing time, and cellular phenotype. Previously conducted preclinical studies have demonstrated that serum-free formulations can support WJ-MSC expansion while eliminating animal-derived components that raise regulatory and safety concerns[76]. They report improved growth kinetics and cellular characteristics compared with traditional cultures containing serum and animal components.
Previously conducted in vitro research demonstrated that early-passage WJ-MSCs (passages 3-5) exhibited superior immunomodulatory activity and differentiation potential compared to late-passage cells (passages 10-15), supporting the use of early passages for therapeutic applications[77]. However, large-scale manufacturing requirements may necessitate extended expansion, creating tension between cell quantity and quality. It is also important to note that, in some instances, later passages have been reported to perform well, particularly with regard to the specialization of cells to express bone-like characteristics[77]. Sypecka et al[38] reported that cutting the entire UC with a lancet, rather than using cylindrical sections, improves cell migration into the culture medium. They also noted that WJ-MSCs cultured under 5% oxygen produced a better cell density.
Quality control and potency testing
Establishing robust quality control and potency assays represents a major challenge for WJ-MSC product development. While standard identity markers (CD73, CD90, CD105 positivity; CD34, CD45 negativity) and basic differentiation assays satisfy minimal regulatory requirements, these tests poorly predict therapeutic efficacy. Previously published research has explored functional potency assays, including immunosuppression assays, angiogenesis assays, and disease-specific in vitro models, and has identified the strengths and limitations of each methodology and noted how different assays may be selected based on the purpose of the experiment[78].
Gene expression profiling and secretome analyses offer additional approaches for quality assessment. Previous studies identified gene expression signatures associated with therapeutic potency, including expression patterns of immunomodulation, angiogenic, and trophic factor genes[79]. Similarly, proteomic characterization of secreted factors has been proposed as a potency indicator, with specific growth factor and cytokine profiles correlating with therapeutic efficacy in preclinical models[79].
Contamination testing, including sterility, mycoplasma, and endotoxin assessments, is an essential quality control element. Additionally, karyotype stability assessment during manufacturing has long been used[80]. However, it is not as sensitive as fluorescence in situ hybridization or comparative genomic hybridization array; these processes have their own limitations, and researchers recommend using multiple methods in tandem, depending on the requirements of the samples and experiments, to achieve a better analysis[80].
Cryopreservation and banking
Effective cryopreservation, enabling cell banking and off-the-shelf availability, is a critical requirement for commercial WJ-MSC products. Previously conducted research indicates that lower temperatures reduce the viability and therapeutic potential of WJ-MSCs, especially over longer periods, regardless of the availability of cryoprotectants[38]. However, some studies have reported reduced immunomodulatory potency immediately post-thaw, with recovery of suppressive activity requiring a full day of culture[81]. These findings have important implications for clinical protocols and product design.
Researchers have noted that WJ-MSCs divide more efficiently when stored in suspension[38]. Cryopreservation medium optimization studies have explored alternatives to traditional cryoprotectants owing to concerns about the toxicity of dimethyl sulfoxide and the need for post-thaw washing procedures[82]. Research has demonstrated that formulations without dimethyl sulfoxide and animal content were able to maintain WJ-MSC viability and function significantly better than standard protocols, while potentially simplifying clinical administration[82]. Such alternatives may facilitate regulatory approval and ease clinical adoption.
Regulatory pathways and clinical development
WJ-MSC products face complex regulations that vary worldwide. In the United States, these products typically follow biologics regulations that require Investigational New Drug applications before proceeding with clinical trials that demonstrate safety and efficacy[83]. The European Medicines Agency classifies WJ-MSCs as Advanced Therapy Medicinal Products, which require similar comprehensive development programs[84]. Asian countries (e.g., South Korea, Japan, and China) have also established specific regenerative medicine regulatory frameworks with varying requirements[85].
An important factor that influences regulations is the autologous/allogeneic nature of the products. While autologous products may qualify for more streamlined pathways in some jurisdictions based on minimal manipulation criteria, allogeneic WJ-MSC products generally require full drug development programs[86]. However, the advantages of allogeneic products, including but not limited to scalability, immediate availability, and consistent manufacturing from characterized donors, make them a popular candidate for clinical use[86].
When designing a clinical trial, several factors should be considered, including sample size, outcome measures, dosing regimens, and delivery routes. Studies have explored various administration routes, including intravenous, intra-articular, intrathecal, and local injection, with optimal approaches varying by the health condition being investigated[2]. Generally, studies evaluate doses in cells per kilogram of body weight. However, the optimal dose for most health conditions remains unclear[2,38]. Another variable that requires systematic investigation is the efficacy of multiple vs single dose administrations.
Safety considerations
For clinical translation, safety is of utmost importance. The safety profiles of WJ-MSCs have generally been shown to be favorable through extensive preclinical safety evaluations and clinical safety monitoring across multiple trials. Previously conducted studies have demonstrated immunoprivileged characteristics of WJ-MSCs that mitigate concerns about allogeneic rejection[46]. Recent studies examining safety data from more than 50 clinical trials involving different MSC sources, including WJ-MSCs, demonstrated no major safety concerns, and serious adverse events directly related to cell administration were uncommon[87,88].
When preparing for a clinical intervention, some adverse events of particular concern include tumorigenicity, embolic events, immunological reactions, and transmission of infections or adventitious agents. It is important to note that long-term carcinogenicity studies in preclinical and previously published clinical investigations found no evidence of tumor formation or promotion[2,80]. However, all clinical studies must remain vigilant in monitoring these adverse events. Accordingly, manufacturing controls that prevent microbial contamination, along with final product testing, may provide additional safety assurances.
Clinical considerations
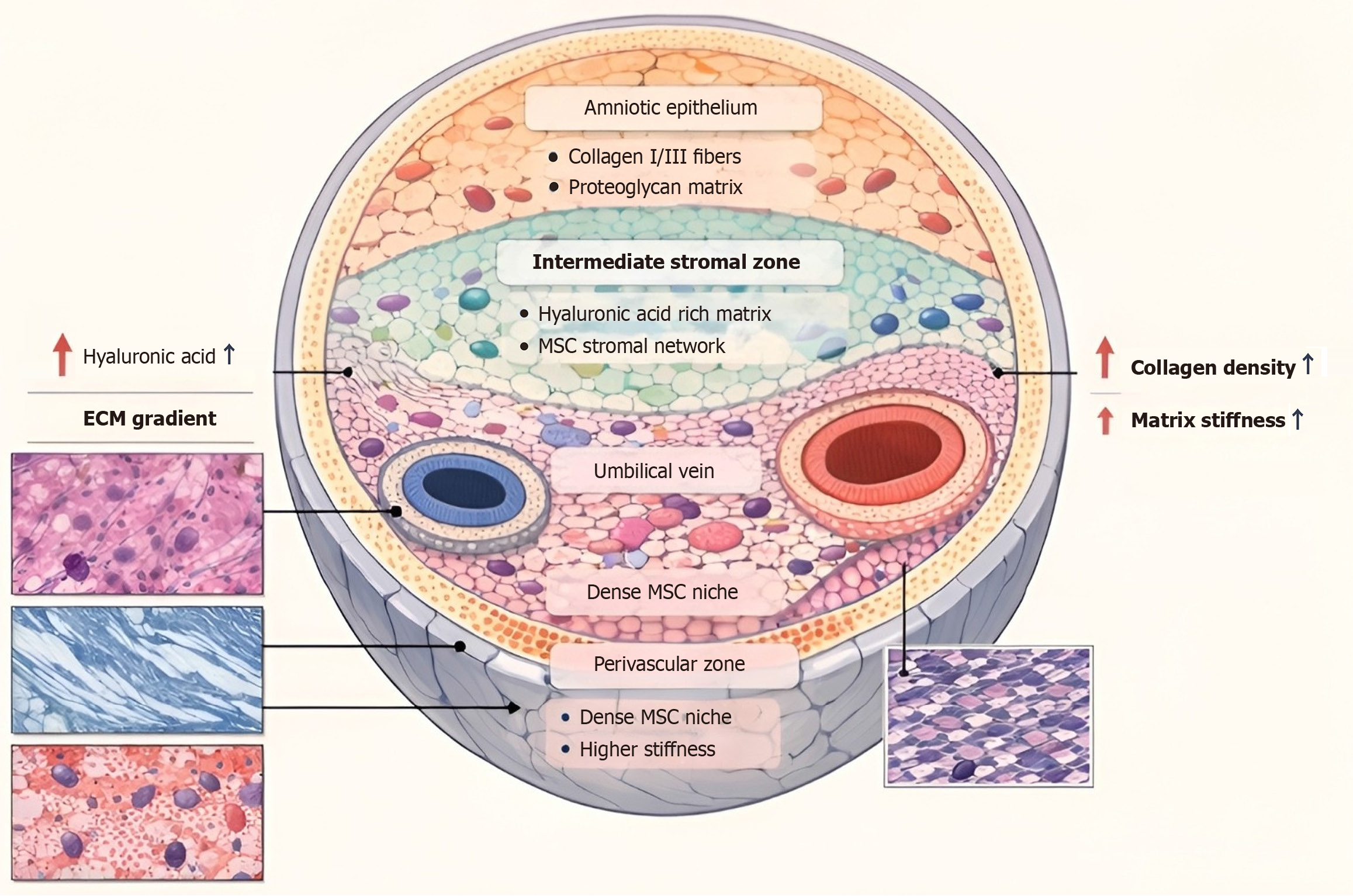
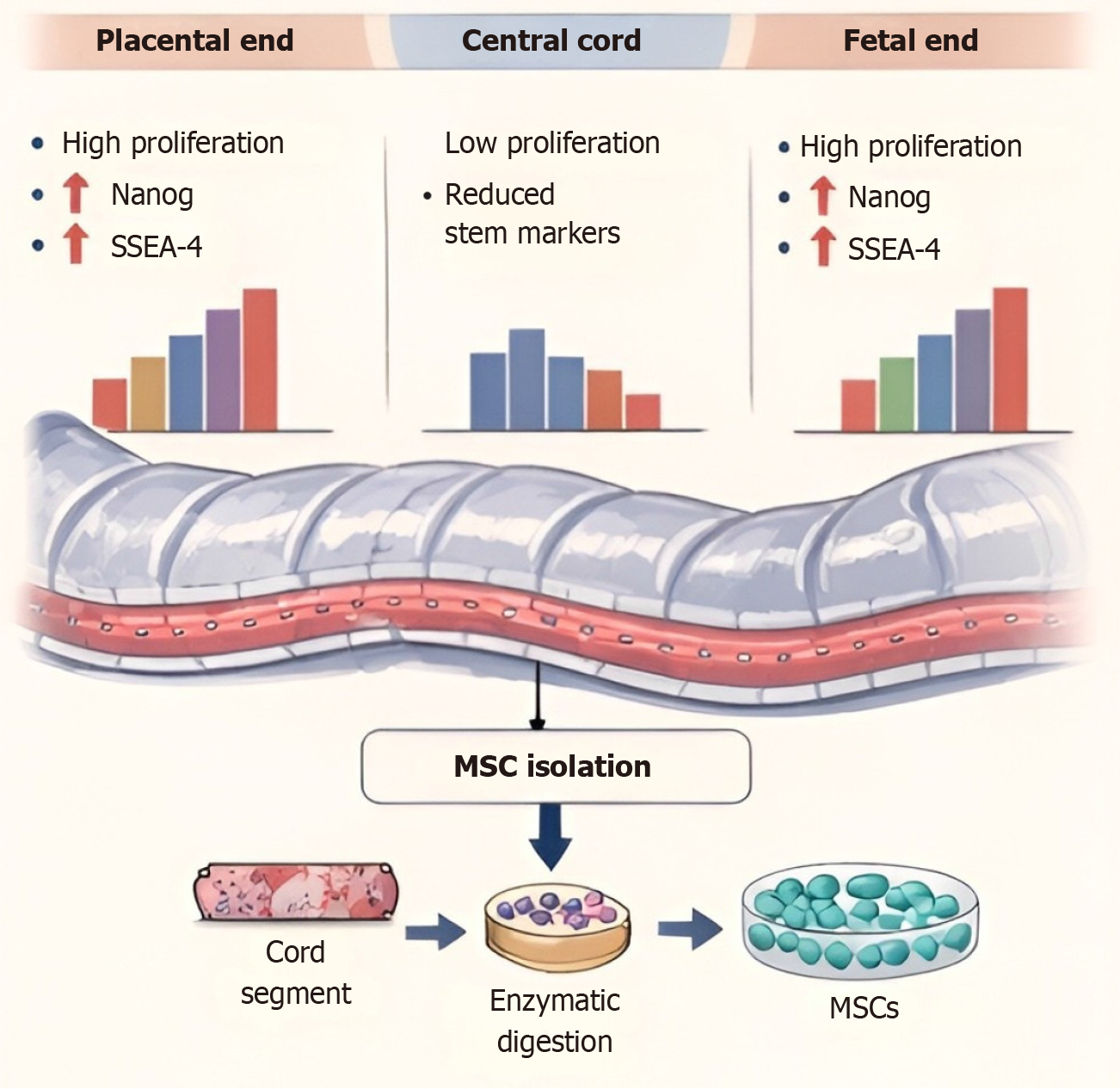
The UC matrix contains multiple anatomical regions. These spatial niches contribute to clonal and functional diversity within WJ-MSC cultures. A comprehensive single-cell and spatial transcriptomic study identified four major WJ-MSC subpopulations - proliferative MSCs, niche-supporting MSCs, metabolism-related MSCs, and biofunctional MSCs - each characterized by distinct gene expression signatures and predicted functional roles[44]. Notably, a S100A9+CD29+ CD142+ subpopulation showed enhanced wound-repair activity and was preferentially enriched in the fetal segment of the UC, illustrating how spatial origin within the cord can influence therapeutic phenotype. Such findings suggest that WJ-MSC cultures represent a composite of clonally distinct stromal progenitors, each contributing variably to proliferation, immunomodulation, angiogenesis, and tissue repair.
High-resolution omics technologies have demonstrated that WJ-MSC heterogeneity extends beyond anatomical location to include transcriptional and functional specialization among individual cells. Single-cell RNA-sequencing studies have shown that clusters of WJ-MSCs differ in pathways related to proliferation, immune regulation, metabolic activity, and ECM signaling[44]. These transcriptional differences likely underpin the variable differentiation potential and paracrine activity observed across cultures and donors. Importantly, lineage differentiation scores calculated from single-cell transcriptomic data indicate that individual WJ-MSC subpopulations exhibit differential bias toward specific differentiation pathways, suggesting that multipotency observed in bulk cultures may actually arise from a mixture of lineage-primed progenitors rather than uniform multipotency at the single-cell level[44].
The composition and potency of this secretome vary substantially depending on donor characteristics, culture conditions, and cell subpopulation composition. Proteomic and functional analyses demonstrate that WJ-MSC secretomes may differ in levels of factors such as VEGF, HGF, IL-6, and TGF-β, which can alter immunomodulatory and regenerative activity[15]. Consequently, differences in secretome profiles may significantly influence clinical outcomes, particularly in therapies relying on paracrine immunomodulation rather than engraftment.
Beyond intrinsic biological heterogeneity, manufacturing processes introduce additional layers of variability that complicate clinical translation. Even under good manufacturing practice (GMP) conditions, WJ-MSC production involves multiple steps - donor screening, tissue processing, isolation, expansion, cryopreservation, and thawing - each capable of influencing cell phenotype and potency. Studies of GMP-compliant WJ-MSC production demonstrate that while quality control criteria such as viability, immunophenotype, and differentiation capacity can be maintained, significant variability in yield, expansion kinetics, and functional characteristics can still occur between donor batches[89]. Donor-related factors - including maternal age, gestational conditions, and neonatal characteristics - have also been shown to affect WJ-MSC yield and biological properties, further contributing to inter-batch variability[90]. Variations in the literature regarding cellular heterogeneity across regions of WJ are more likely attributable to methodological differences rather than to fundamentally conflicting biological evidence. Analysis of study designs reveals several primary factors underlying these variations.
Definitions of tissue sampling vary considerably among studies. Some researchers distinctly separate perivascular areas adjacent to umbilical vessels from other regions of WJ, whereas others employ broader or ambiguous classifications. As perivascular regions are thought to contain a higher proportion of progenitor-like cells with unique characteristics, inconsistent definitions influence both the cell populations analyzed and the resulting interpretations.
Cell isolation techniques further contribute to the observed differences. Explant methods preferentially select for adherent and migratory cells, potentially excluding less adherent cell types. In contrast, enzymatic digestion can yield a broader spectrum of cells. However, outcomes can vary depending on the specific enzymes employed, digestion duration, and tissue handling procedures. These methodological choices determine the initial cell populations and subsequently influence experimental observations.
Culture conditions, including oxygen concentration, passage number, and media composition, also influence the characteristics and behavior of WJ-MSCs. For instance, hypoxic conditions help maintain stem cell properties and alter secretory profiles, whereas extended passaging can diminish regional distinctions and promote cellular homogeneity. Consequently, variations in culture protocols may either obscure or accentuate regional heterogeneity.
Extended in vitro expansion can alter MSC phenotype, gene expression, and differentiation capacity. Passage-dependent changes in transcriptional profiles and lineage propensity have been reported in WJ-MSCs, suggesting that later passages may represent functionally distinct cell populations. Clinical products may be administered as fresh or cryopreserved cells, and freeze-thaw processes can affect viability, metabolic state, and paracrine function. Clinical trials employ widely differing dosing strategies, ranging from single administrations to multiple infusions, with dose levels often reported per kilogram body weight [e.g., approximately (0.4-1.0) × 106 cells/kg in some trials][91].
There is no universally accepted potency assay for MSC products. Laboratories often rely on surrogate endpoints such as immunosuppressive capacity, cytokine secretion, or differentiation assays, which may not correlate with therapeutic efficacy. Trials frequently measure different endpoints - immunomodulation, tissue regeneration, inflammatory markers, or functional recovery - making cross-study comparisons difficult.
Collectively, these biological and methodological factors create a complex landscape in which WJ-MSC preparations from different laboratories or clinical trials may represent fundamentally different cellular products. Variability in clonal composition, transcriptional state, secretome profile, manufacturing process, dosing strategy, and outcome assessment significantly limits direct comparison between studies and complicates efforts to draw definitive conclusions regarding therapeutic efficacy.
Consequently, future clinical development of WJ-MSC therapies will likely require standardized manufacturing protocols, defined potency assays, and molecular characterization of functional subpopulations. Advances in single-cell omics and spatial transcriptomics may help identify therapeutically relevant MSC subsets, enabling more consistent and reproducible cell products for regenerative medicine applications.
The methodologies used to assess cellular differences are also significant. Studies relying primarily on standard MSC surface markers such as CD73, CD90, and CD105 typically report minimal regional variation. In contrast, investigations employing functional assays, gene expression profiling, or secretome analysis frequently identify substantial differences. These findings suggest that marker panels alone may be insufficient to capture the full extent of functional heterogeneity.
FUTURE DIRECTIONS AND EMERGING RESEARCH
Genetic engineering and cell engineering approaches
Recent research demonstrated that WJ-MSCs transduced with lentiviral vectors expressed higher levels of VEGF and exhibited improved angiogenic activity in ischemic disease models compared with unmodified cells[92]. Previous studies have explored enhanced expression of specific therapeutic genes, including growth and transcription factors, anti-inflammatory cytokines, and other protein structures[93]. Genetic modification of WJ-MSCs is an emerging strategy to enhance their therapeutic potency.
Proof-of-concept studies have demonstrated successful CRISPR-mediated gene knockout in WJ-MSCs, introducing the possibility of deleting specific regulatory factors or inserting therapeutic genes at specific loci to enhance their therapeutic potential[94]. However, regulatory pathways for genetically modified cellular therapies remain complex, and safety considerations, including off-target effects and immunogenicity, require thorough evaluation.
Keshtkar et al[95] conducted a study which demonstrated that hypoxic preconditioning significantly improved WJ-MSC therapeutic efficacy in pancreatic islet models, correlating with increased VEGF secretion and increased expression of anti-apoptotic factors. Ocansey et al[96] also reviewed cell engineering approaches, including permanent genetic manipulation and preconditioning strategies that enhance the therapeutic properties of WJ-MSCs. These studies pave the way to inspire future research on WJ-MSCs.
Biomaterial scaffolds and tissue engineering
Lech et al[97] evaluated various natural and synthetic scaffolds as carriers for WJ-MSC delivery, providing structural support, improving cell retention, and creating biomimetic microenvironments. In another study, Chen[98] observed that WJ-MSCs seeded on collagen-GAG scaffolds showed enhanced chondrogenic differentiation and produced cartilage-like tissues with superior mechanical properties compared with scaffold-free approaches. The findings from these studies indicate that combining WJ-MSCs with biomaterial scaffolds represents a promising approach for tissue engineering applications.
In a recent study, da Rosa et al[99] demonstrated successful bioprinting of WJ-MSCs in various bioink formulations, maintaining cell viability and function post-printing. A combination of these bioprinted scaffolds can also be used to precisely organize cells and materials, potentially enabling the fabrication of complex tissues. These may be used as skin substitutes, cartilage and organ constructs, and vascular grafts, though their clinical translation remains in early stages[99].
Ushakov et al[100] investigated decellularized ECM scaffolds and observed enhanced production of certain biochemical cues. Research has shown that WJ-MSCs cultured on decellularized cardiac matrix exhibited enhanced cardiac marker expression and improved therapeutic efficacy in heart injury models compared with cells cultured in standard conditions[101]. These findings suggest that biomimetic scaffolds can direct WJ-MSC behavior and enhance regenerative potential.
Personalized medicine and patient-specific approaches
While allogeneic WJ-MSCs offer manufacturing and practical advantages, personalized approaches using patient-specific cells represent another potential direction. Autologous UC banking at birth enables personal WJ-MSC repositories for potential future therapeutic use. However, the effects of long-term cryopreservation, cost-effectiveness, and actual clinical utility of the banked cords warrant further investigation and optimization.
Research suggests that patients’ inflammatory status influences WJ-MSC therapeutic responses, with highly inflammatory conditions potentially requiring higher cell doses or primed cells for optimal efficacy[102]. Developing patient stratification strategies and personalized dosing algorithms may optimize clinical outcomes. Hence, precision medicine approaches that match WJ-MSC products to specific patient populations based on biomarkers or disease characteristics represent another field of study worth looking into.
Previous studies have explored WJ-MSCs combined with physical therapy for musculoskeletal conditions, with immunosuppressive drugs for transplant applications, and with standard-of-care medications for various diseases[103]. These combination therapies integrating WJ-MSCs with conventional treatments represent another area of investigation and may provide synergistic benefits. However, they require careful evaluation of interactions and safety.
Consistent conclusions in current WJ-MSC research
There is a strong consensus that WJ-MSCs show potent immunomodulatory and anti-inflammatory effects largely through paracrine mechanisms, including release of secretomes and extracellular vesicles rather than long-term engraftment into host tissues. This paracrine activity appears central to their therapeutic potential across diverse preclinical and early clinical contexts. In addition, WJ-MSCs consistently exhibit low immunogenicity and ethical acceptability owing to their noninvasive UC origin. This supports their translational attractiveness relative to other MSC sources. The secretory effects, immune regulation, and favorable short-term safety are recurrent themes across recent reviews and clinical research summaries.
Divergences and evidence gaps
Despite these shared findings, important issues remain unresolved. Heterogeneity in WJ-MSC populations driven by differences in isolation methods, culture conditions, and subpopulation composition continues to impede direct comparisons between studies and the reproducibility of outcomes. Mechanistic understanding also varies significantly across models, especially regarding how secretome components vs cell-cell interactions mediate clinical effects. Evidence on long-term safety and durability is still insufficient, with few longitudinal clinical trials adequately powered to assess rare adverse events or sustained functional benefit. Additionally, some commercial UC products contain only a minimal number of viable MSCs, raising conceptual and regulatory questions about what constitutes true MSC-based therapy.
Currently, the evidence for the feasibility of isolation, multipotent differentiation potential, and immunomodulatory effects of MSCs from WJ tissue is consistent. However, certain aspects of the MSCs from WJ tissue, such as the heterogeneity of the tissue, the long-term integrative ability of the cells in the host tissue, and the relative contribution of the paracrine effect vs the long-term contribution of the cells themselves, are still unknown. In addition, certain aspects of the MSCs from WJ tissue, such as the ability of the cells to maintain their differentiation potential and their ability to replace the tissue in which they were transplanted, are still unsupported by sufficient evidence. The use of MSCs from WJ tissue in the clinic is further limited by the need for the development of standardized assays for potency and the ability to manufacture the cells in large enough quantities for use in the clinic.
Critical bottlenecks for future research
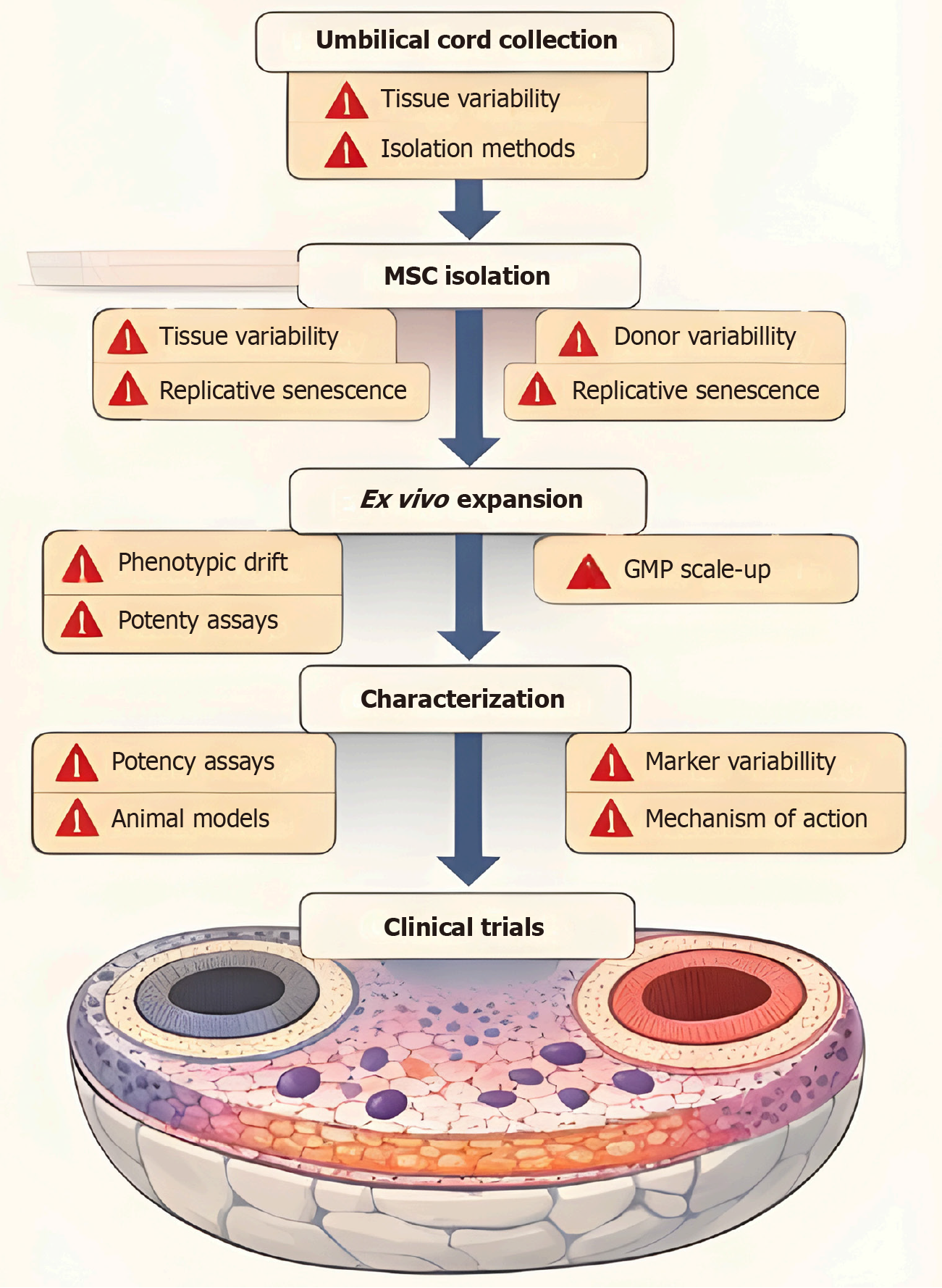
Two bottlenecks are especially pressing. First, standardization of product characterization and potency assays that link manufacturing variables to biological activity and clinical outcomes is essential to reduce heterogeneity and enable reliable cross-study interpretation. Second, rigorous longitudinal clinical studies with integrated mechanistic biomarkers are needed to clarify long-term safety, mechanisms of action under various disease conditions, and persistence of therapeutic effects (Figure 3). Addressing these bottlenecks will shift the field from accumulating isolated datasets toward an integrated, predictive, and clinically translatable science, thereby enabling more strategic trial and therapeutic application design.
Figure 3 Translational pathway and bottlenecks in the clinical translation of umbilical cord-derived mesenchymal stem cell therapy.
There are several steps in the clinical translation of umbilical cord-derived mesenchymal stem cell therapy, including tissue procurement, cell isolation and expansion, cell characterization, pre-clinical validation, and clinical validation. However, there are several bottlenecks in the clinical translation of umbilical cord-derived mesenchymal stem cell therapy, including donor variability, lack of standardization in cell isolation and potency, difficulties in large-scale manufacturing, and limited understanding of long-term safety and the mechanism of action. MSC: Mesenchymal stem cell.
CONCLUSION
UC-derived MSCs, particularly those from WJ, offer promise for regenerative and immunomodulatory treatments due to their ability to multiply, transform into various cell types, and cause low immune rejection. Early lab and clinical studies suggest they could help treat neurological, inflammatory, and degenerative diseases. However, the evidence is mixed and often limited by small sample sizes, varied isolation methods, and inconsistent characterization standards. Several major challenges limit their clinical use. First, biological differences between the cord and donors make it difficult to create standardized manufacturing methods. Second, it’s difficult to evaluate treatment outcomes because researchers are still unaware of how these cells function, integrate, or survive in the long term. Third, broader clinical use is still hampered by regulatory and GMP concerns, including increased production, quality assurance, and potency testing. We need additional studies on how these cells function, standardized procedures for separation and characterization, and carefully planned clinical trials to get around these restrictions. Even though UC-derived MSCs-based treatments have a lot of clinical potential, their future success hinges on resolving these issues and generating solid, trustworthy evidence to guarantee safe and efficient treatments.