Published online Apr 26, 2026. doi: 10.4252/wjsc.v18.i4.117652
Revised: February 10, 2026
Accepted: March 16, 2026
Published online: April 26, 2026
Processing time: 128 Days and 23.9 Hours
Osteoarthritis (OA) is marked by progressive cartilage degeneration and limited intrinsic repair. Mesenchymal stem cells (MSCs) show promise for cartilage regeneration, yet unstable chondrogenesis and a tendency towards hypertrophy constrain their efficacy. Dysregulated osteoarthritic microRNAs (miRNAs), inc
To determine whether transient ex vivo LNA-ASO silencing of OA-related miRNAs could improve the chondrogenesis of MSCs.
Rat bone-marrow MSCs were transfected with LNA-ASOs against miR-16-5p, miR-30b-5p, miR-146a-5p, and miR-193b-3p (single or combined) and induced into high-density micromass cultures. On day 11, quantitative real-time polymerase chain reaction assessed Sox9, Acan, Runx2, Mef2C. On day 21, histology (Safranin O, Alcian Blue/Fast Red, Masson’s trichrome, Picrosirius Red under polarised light) and DMMB (sulphated glycosaminoglycans normalised to dsDNA) quantified cartilage-like matrix. Transfection efficiency was measured by flow cytome
All LNA-ASOs achieved transfection efficiencies greater than 85%. The combination miR-30b-5p + miR-193b-3p produced the most favourable molecular profile at day 11, with higher Sox9 and Acan expression and lower Runx2 compared with the power inhibitor control, outperforming single-inhibitor conditions. miR-30b-5p alone and miR-30b-5p + miR-146a-5p also improved chondrogenic markers, whereas the miR-16-5p/miR-30b-5p combination showed a pattern prone to hypertrophy. At day 21, inhibition of miR-30b-5p/miR-193b-3p yielded a stronger Safranin O and Alcian Blue staining and more abundant, better-organized collagen on Masson’s trichrome and Picrosirius Red. DMMB analysis (sulphated glycosaminoglycans normalised to dsDNA) was highest with miR-30b-5p + miR-193b-3p, consistent with the histology results.
Brief ex vivo priming of MSCs with LNA-ASOs targeting anti-chondrogenic miRNAs promoted a hyaline-like programme and strengthened matrix deposition. Notably, the miR-30b-5p/miR-193b-3p pairing showed the most favourable profile, indicating promise for MSC-based strategies in cartilage regeneration.
Core Tip: Our study demonstrates that a brief ex vivo priming of mesenchymal stem cells (MSCs) with locked nucleic acid-antisense oligonucleotides targeting osteoarthritis-related microRNAs improved early chondrogenic programming. In rat MSC micromasses, the miR-30b-5p/miR-193b-3p combination produced higher Sox9 and Acan, lower Runx2, and stronger histological evidence of cartilage-like matrix than single inhibitors and controls. This transient, non-integrative strategy provides molecular fine-tuning of MSCs and has the potential to enhance the consistency and durability of MSC-based therapies for cartilage repair.
- Citation: Fontiveros-Palomino M, Álvarez-Iglesias I, González-González A, Alfonso-Fernández A, Pérez-Campo FM. Modulation of osteoarthritis-related microRNAs using locked nucleic acid-antisense oligonucleotides boosts chondrogenic lineage commitment of mesenchymal progenitors. World J Stem Cells 2026; 18(4): 117652
- URL: https://www.wjgnet.com/1948-0210/full/v18/i4/117652.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v18.i4.117652
Osteoarthritis (OA) is one of the most prevalent degenerative joint disorders and a leading cause of chronic pain, loss of mobility, and reduced quality of life. As the population ages and obesity rates increase, OA rates are expected to increase exponentially in the coming years. Current treatments focus mainly on symptom control (analgesics, nonsteroidal anti-inflammatory drugs, physical therapy, intra-articular injections) and, in severe cases, on joint replacement surgery[1,2]. However, none of these approaches can regenerate damaged cartilage or stop disease progression, underscoring the need for regenerative strategies that restore hyaline cartilage structure and function.
In the research for regenerative strategies, mesenchymal stem cells (MSCs) have emerged as a promising tool for cartilage repair. Beyond their potential to differentiate into chondrogenic, osteogenic, and adipogenic lineages, MSCs exert paracrine effects that can modulate inflammation and support tissue homeostasis via trophic factors and extracellular vesicles (EVs) that positively influence tissue repair and modulate inflammation[3]. Preclinical and early clinical studies report improvements in symptoms and joint milieu after intra-articular delivery or scaffold-based implantation of MSCs, although outcomes vary and durability remains a challenge, in part because MSCs frequently fail to maintain stable hyaline-like chondrogenesis and can drift towards hypertrophy, fibrocartilage, or premature ossi
MicroRNAs (miRNAs) are small, approximately 20-25 nucleotide noncoding RNAs that repress gene expression post-transcriptionally by binding to 3’-untranslated regions and recruiting RNA-induced silencing complexes. Aberrant miRNA profiles in OA influence apoptosis, autophagy, extracellular matrix (ECM) synthesis and degradation, inflammation, and hypertrophy[7]. A bioinformatic meta-analysis by Cong et al[8] identified a robust OA signature with up-regulation of miR-16-5p, miR-30b-5p, miR-146a-5p, and miR-193b-3p in human osteoarthritic cartilage, alongside down-regulation of protective miRNAs such as miR-140. These data, consolidated by subsequent reviews, support the view that specific OA-associated miRNAs act as anti-chondrogenic brakes, converging on nuclear factor kappa B, transforming growth factor-beta/SMAD, and Wnt pathways to promote catabolism and hypertrophy.
Given the influence of miRNAs on chondrogenic, catabolic and inflammatory pathways, targeting pathological miRNAs represents a compelling strategy to enhance MSCs performance. Among the various approaches for miRNA modulation, locked nucleic acid antisense oligonucleotides (LNA-ASOs) stand out due to their enhanced binding affinity, specificity, nuclease resistance, and favourable pharmacokinetics in vivo[9]. In fact, one of the pioneering demonstrations of miRNA-targeted therapy in OA showed that intra-articular delivery of LNA-miR-181a-5p ASO mitigated cartilage damage, reduced catabolic marker expression (e.g., matrix metalloproteinases, type II collagen degradation), and in
Among the different available modalities for miRNA modulation, LNA-ASOs are particularly attractive because of their high affinity, specificity, and resistance to nucleases. Proof-of-concept for anti-miR therapy in OA comes from studies showing that intra-articular LNA-based inhibition can reduce cartilage damage and catabolic markers and limit chondrocyte apoptosis in preclinical models and human explants[10]. However, direct intra-articular delivery has practical problems such as the rapid synovial clearance, limited penetration and/or retention in avascular cartilage, and limited control over which joint cell types are modulated, often needing repeated doses[11,12].
In this context, a brief, ex vivo priming of MSCs with LNA-ASOs offers a complementary route where miRNA in
Here, we implement this strategy by transiently priming MSCs with LNA-ASOs against four OA-associated, anti-chondrogenic miRNAs (miR-16-5p, miR-30b-5p, miR-146a-5p, and miR-193b-3p) delivered individually or in predefined combinations during the early chondrogenic induction. We then assess early lineage stability using Sox9 and Acan as chondrogenic read-outs and Runx2 and Mef2C as hypertrophic markers, and we evaluate ECM deposition by histology and sulphated glycosaminoglycan (sGAG) quantification. Our data show that the short-pulse combination inhibitionof miR-30b-5p and miR-193b-3p is able to enhance chondrogenic markers while attenuating hypertrophic markers and maintaining cell viability. Overall, the data point to brief ex vivo anti-miR priming as a practical complement to MSC therapy, with short-pulse, combinatorial miRNA inhibition directing chondrogenic fate in a translationally compatible way for cartilage regeneration.
Rat bone marrow-derived MSCs (rMSCs) were isolated from the femurs and tibiae of adult Wistar rats (8-10 weeks old) following a standard flushing protocol. Cells were expanded in low-glucose DMEM supplemented with 10% fetal bovine serum, 1% penicillin-streptomycin, and maintained at 37 °C with 5% CO2. MSC phenotype was confirmed by adherence to plastic, fibroblast-like morphology, and expression of surface markers (positive for CD90, CD29; negative for CD45).
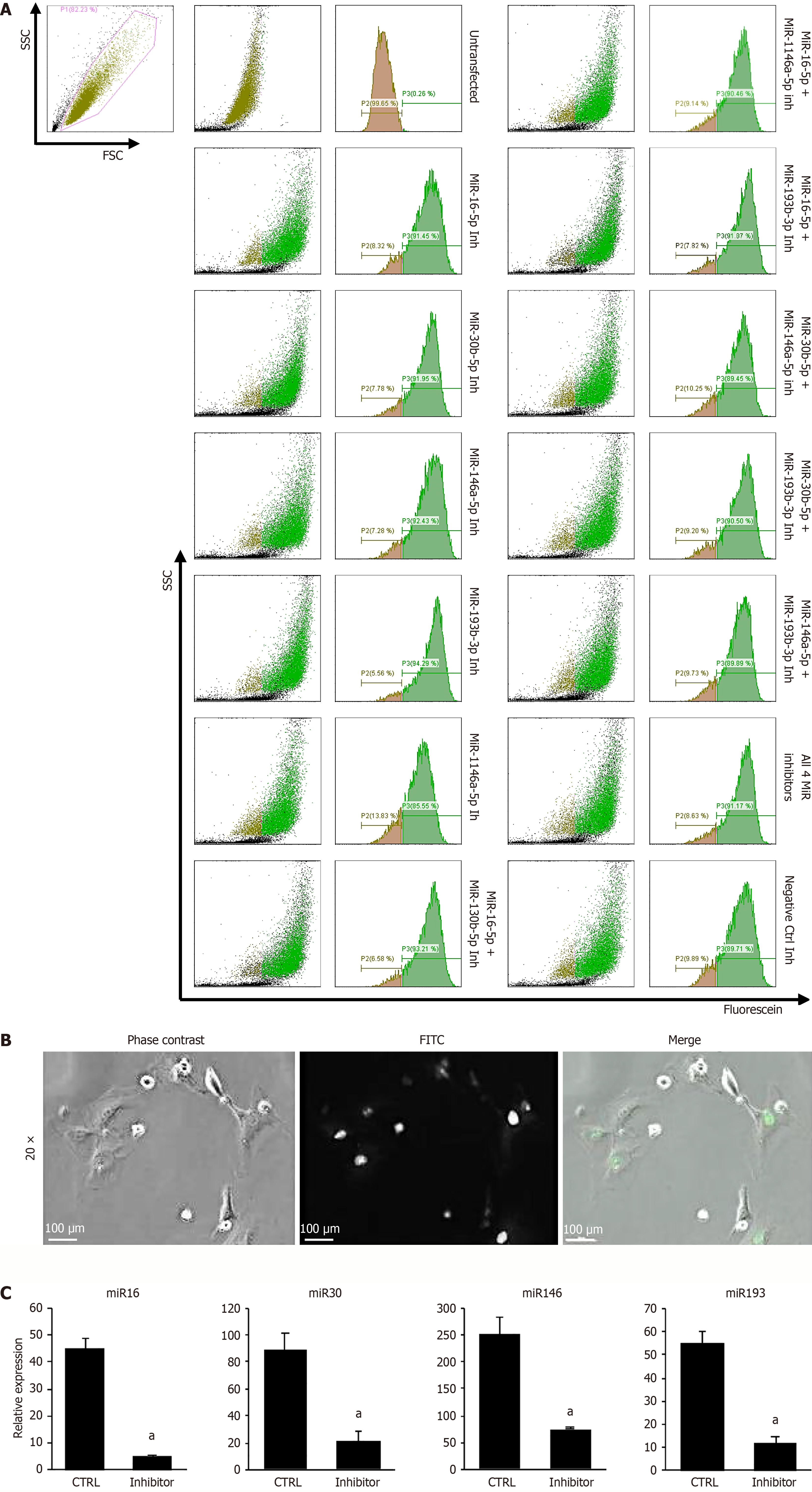
Rat MSCs were expanded, counted, and seeded at 22000 cells/cm2 in 6-well plates to obtain a near-confluent monolayer after 24 hours. To inhibit selected miRNAs we used miRCURY LNA miRNA inhibitors (QIAGEN, Hilden, Germany; formerly Exiqon), 20-22 nt in length and 6-FAM-labelled to enable estimation of transfection efficiency by flow cytometry. Delivery was performed by lipofection with DharmaFECT according to the manufacturer’s ratios. Two hours before transfection, culture media was replaced with reduced-serum Opti-MEM to favor cationic-lipid uptake. Inhibitors were delivered following the manufacturer’s recommended ratios. For single-inhibitor conditions we used miRCURY LNA Power Inhibitor (non-targeting negative control, Ref. 00199006), miR-16-5p (Ref. 04102067), miR-30b-5p (Ref. 04102485), miR-146a-5p (Ref. 04100679), and miR-193b-3p (Ref. 0202180). Combination conditions included all predefined pairwise mixes and an all-four mix. Non-transfected cells and cells transfected with the power inhibitor were used as controls. After 24 hours, an equal volume of complete medium was added to dilute the transfection mixture; after a further 48 hours the medium was fully replaced with standard culture medium. Transfection efficiency was assessed using the FACSCanto II flow cytometer. The results were analyzed using FACSDiva Software (BD Biosciences, San Diego, CA, United States). Rates ≥ 85% positive cells were considered acceptable.
Chondrogenesis was induced using a 3D micromass format. Control and transfected rMSCs were trypsinized, re
Micromass aggregates were transferred to 500 μL TRIzol (Thermo Fisher Scientific, MA, United States), snap-frozen at
For mRNA analysis, cDNA was synthesized with the PrimeScript Kit (Takara, Japan), and qPCR was performed on an Applied Biosystems 7300 using TaqMan™ Gene Expression Master Mix and probes. GAPDH served as the housekeeping gene and Sox9, Acan, Runx2 and MefC2 were quantified using specific TaqMan assays. For miRNA expression, 10 ng total RNA were reverse-transcribed with the TaqMan™ MicroRNA Reverse Transcription Kit (Thermo Fisher Scientific, MA, United States), followed by qPCR with TaqMan™ MicroRNA Assays for rno-miR-16-5p (assay ID rno481312_mir), rno-miR-30b-5p (rno481432_mir), rno-miR-146a-5p (rno481451_mir) and rno-miR-193b-3p (rno478306_mir). U6 snRNA was used as the reference. For gene expression analysis (HDAC3 and TGFB2), TaqMan™ assays Rn00584926_m1 and Rn00676060_m1 were used respectively. Results were expressed as Ct values and relative expression calculated by the
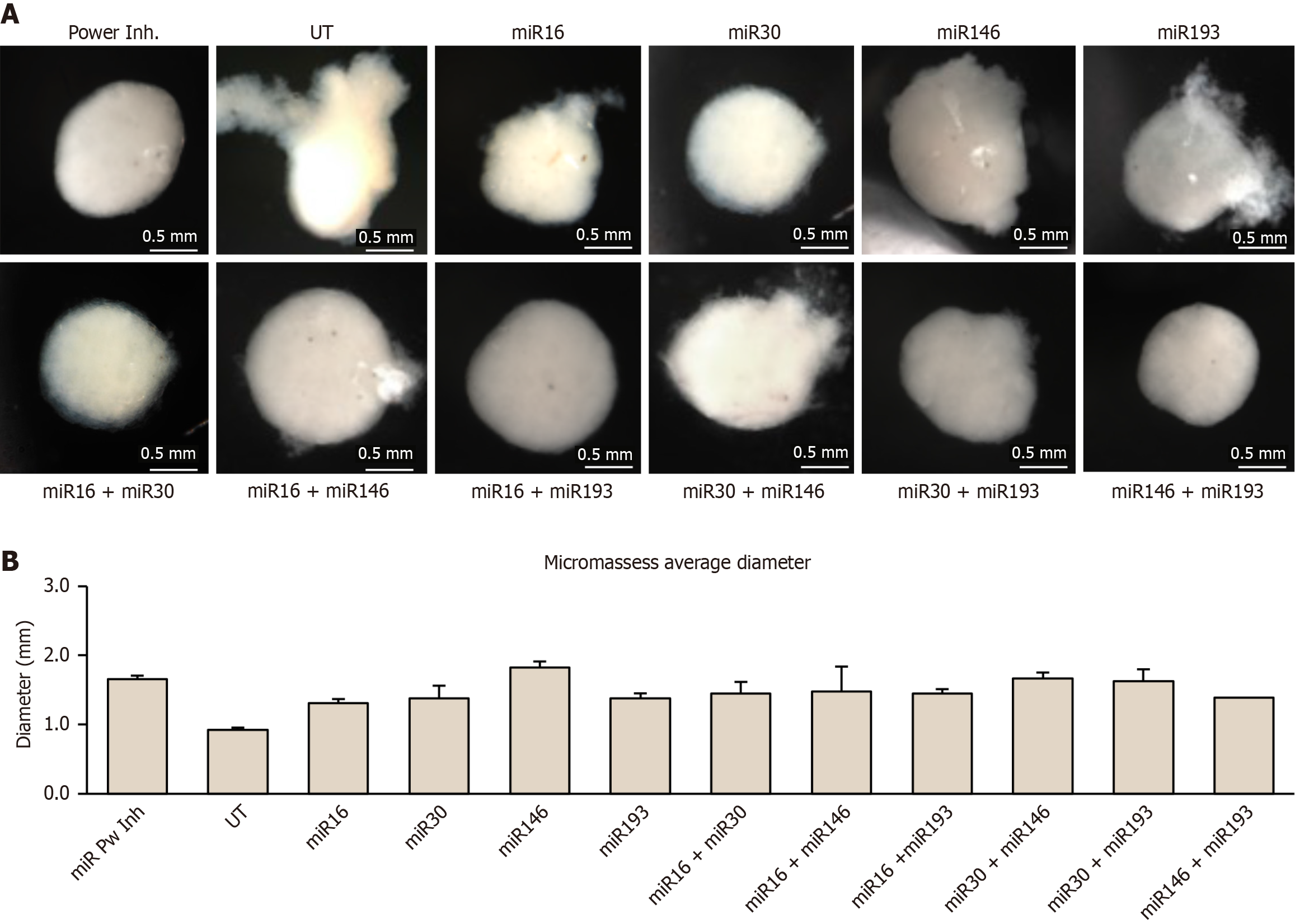
Micromasses were examined macroscopically using a stereomicroscope at 80 × magnification. Images were acquired with DXM1200F/ACT-1 v2.70 software (Nikon, Japan). A scale bar was added, and images were calibrated in ImageJ. Pellet size was quantified in ImageJ using the Line tool to measure the length of each micromass along its longest axis.
Micromasses were fixed in 10% formaldehyde for at least 24 hours, dehydrated through a graded ethanol series and xylene, and embedded in paraffin wax at 60 °C. Paraffin blocks were sectioned at 5 μm on a microtome. For histological staining, sections were deparaffinized in xylene, rehydrated through graded ethanols to distilled water, and stained with Safranin O, Alcian Blue/Fast Red, Masson’s trichrome, and Picrosirius Red following standard protocols. Finally, stained sections were dehydrated through graded ethanols, cleared in three changes of xylene, and coverslipped with DPX mounting medium for microscopy.
The 1,9-dimethylmethylene blue assay quantified sGAGs at 525 nm, using chondroitin sulfate as a standard[13]. DNA content was measured by PicoGreen assay (Invitrogen, MA, United States) to normalize sGAG levels.
Data were analyzed with GraphPad Prism 9.0 software. One-way ANOVA followed by Dunnett’s multiple comparison test was used. Results were expressed as mean ± SEM. P values < 0.05 were considered statistically significant.
Because gymnotic uptake of LNA-ASOs typically demands high doses, we combined the inhibitors with a lipidic transfection reagent to generate LNA-ASO-loaded nanoparticles and enhance delivery. Transfected inhibitors were 6-FAM-labelled, enabling direct read-out of intracellular uptake. Forty-eight hours after transfection, flow cytometry demonstrated uniform and efficient internalization across all inhibitor conditions, with approximately 89.45% to 94.29% of cells being positive (Figure 1A). Representative confocal micrographs revealed intense signal within the nuclear compartment, with a smaller fraction in the cytoplasm, consistent with vesicular entry followed by nuclear accumulation (Figure 1B).
To determine whether efficient uptake resulted in functional miRNA inhibition, endogenous miRNA levels were quantified by quantitative real-time polymerase chain reaction. A significant reduction of each target miRNA was observed following transfection with the corresponding LNA-ASO compared with cells treated with the non-specific power inhibitor control (Figure 1C; Supplementary Figure 1).
In addition, to further validate on-target activity, the expression of two well-established miR-193b-3p target genes, HDAC3 and TGFB2, was analyzed. Consistent with effective miRNA inhibition, both transcripts were significantly upregulated following miR-193b-3p silencing (Supplementary Figure 1). Collectively, these data demonstrate that lipofection enables efficient delivery of LNA-ASOs into rat MSCs (rMSCs) and achieves robust and specific suppression of the intended miRNA targets, providing a reliable foundation for downstream functional assays.
rMSCs (control and LNA-ASO-transfected) generated 3D micromasses in drop culture and were maintained in chondrogenic medium for 21 days. By 24-48 hours post-seeding, compact aggregates detached from the plastic and continued to grow. At day 21, stereomicroscopy revealed rounded, cohesive pellets across conditions. No clear differences in diameter were detected between groups (Figure 2). Qualitatively, micromasses derived from miR-16-5p or miR-30b-5p single inhibition, and from the miR-146a-5p/miR-193b-3p combination, appeared slightly smaller than the average. miR-146a-5p inhibition showed the largest pellets, whereas miR-16-5p inhibition showed the smallest. Increased translucency, compatible with a hyaline-like appearance, was most evident in micromasses from miR-16-5p, miR-30b-5p, miR-193b-3p, and the combinations miR-16-5p + miR-30b-5p and miR-30b-5p + miR-193b-3p.
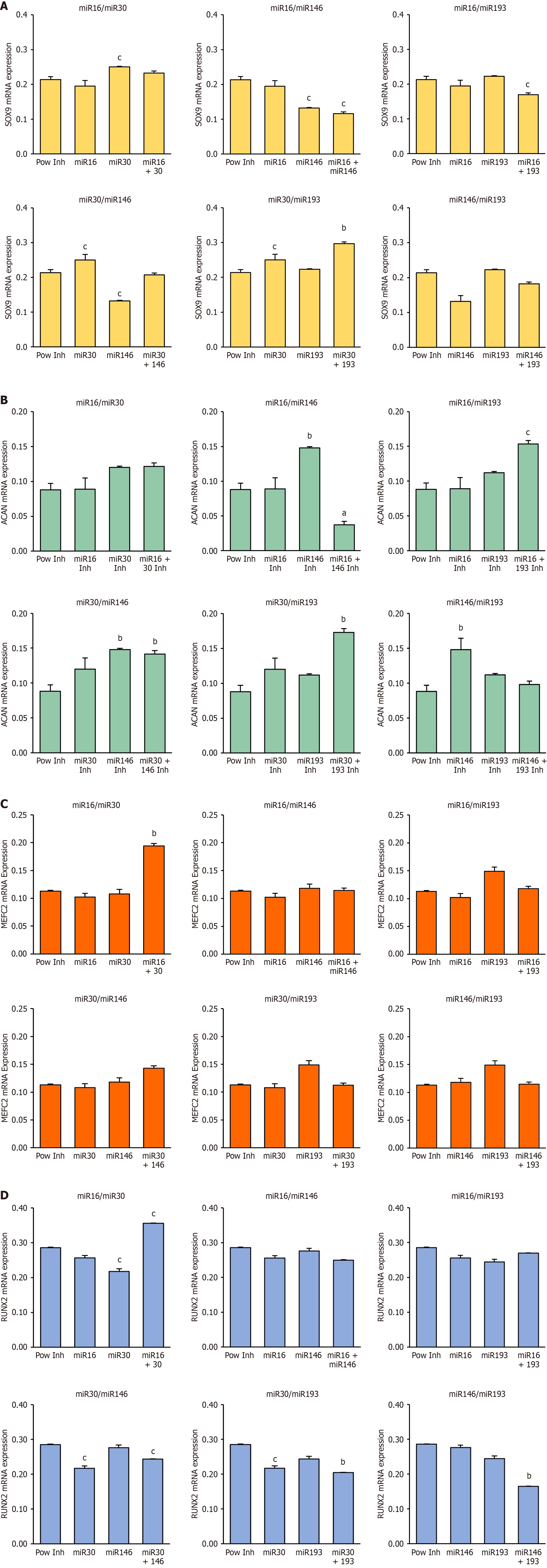
To assess how transient anti-miR treatment affects early lineage commitment, rMSCs were transfected with single LNA-ASO inhibitors or predefined combinations and then induced to form micromasses up to day 11, when mRNA was collected. This intermediate time-point was chosen to capture subtle shifts in expression rather than end-stage plateaus. Chondrogenic markers were Sox9 and Acan, whereas Runx2 and Mef2C were used as hypertrophy markers.
For Sox9, only miR-30b-5p and its combination with miR-193b-3p showed increases relative to the power inhibitor control. Of note, miR-30b-5p/miR-193b-3p exceeded the effect of miR-30b-5p alone, suggesting an additive interaction at this locus. In contrast, miR-146a-5p and its combination with miR16-5p inhibition was associated with lower expression (Figure 3A). For Acan, significant increases were detected in micromasses formed from MSCs transfected with the miR-146a-5p inhibitor. Moreover, all inhibitor combinations, except miR-146a-5p/miR-193b-3p, also produced increases in Acan expression, of varying magnitude (Figure 3B).
Hypertrophic markers showed the complementary trend. Mef2C remained largely unchanged across conditions, except for a rise under miR-16-5p + miR-30b-5p (Figure 3C). Statistical significance is indicated in each panel. However, Runx2 decreased with miR-30b-5p alone and with its combinations miR-30b-5p/ miR-146a-5p and, especially, miR-30b-5p/miR-193b-3p. The pair miR-146a-5p/miR-193b-3p also lowered Runx2. In contrast, miR-16-5p/ miR-30b-5p increased Runx2 (Figure 3D). In summary, miR-30b-5p/miR-193b-3p emerged as the most favourable combination at day 11, enhancing Sox9/Acan while reducing Runx2. Conversely, miR-16-5p/miR-30b-5p showed a hypertrophy-prone profile with a significant increase in Runx2 and Mef2C expression.
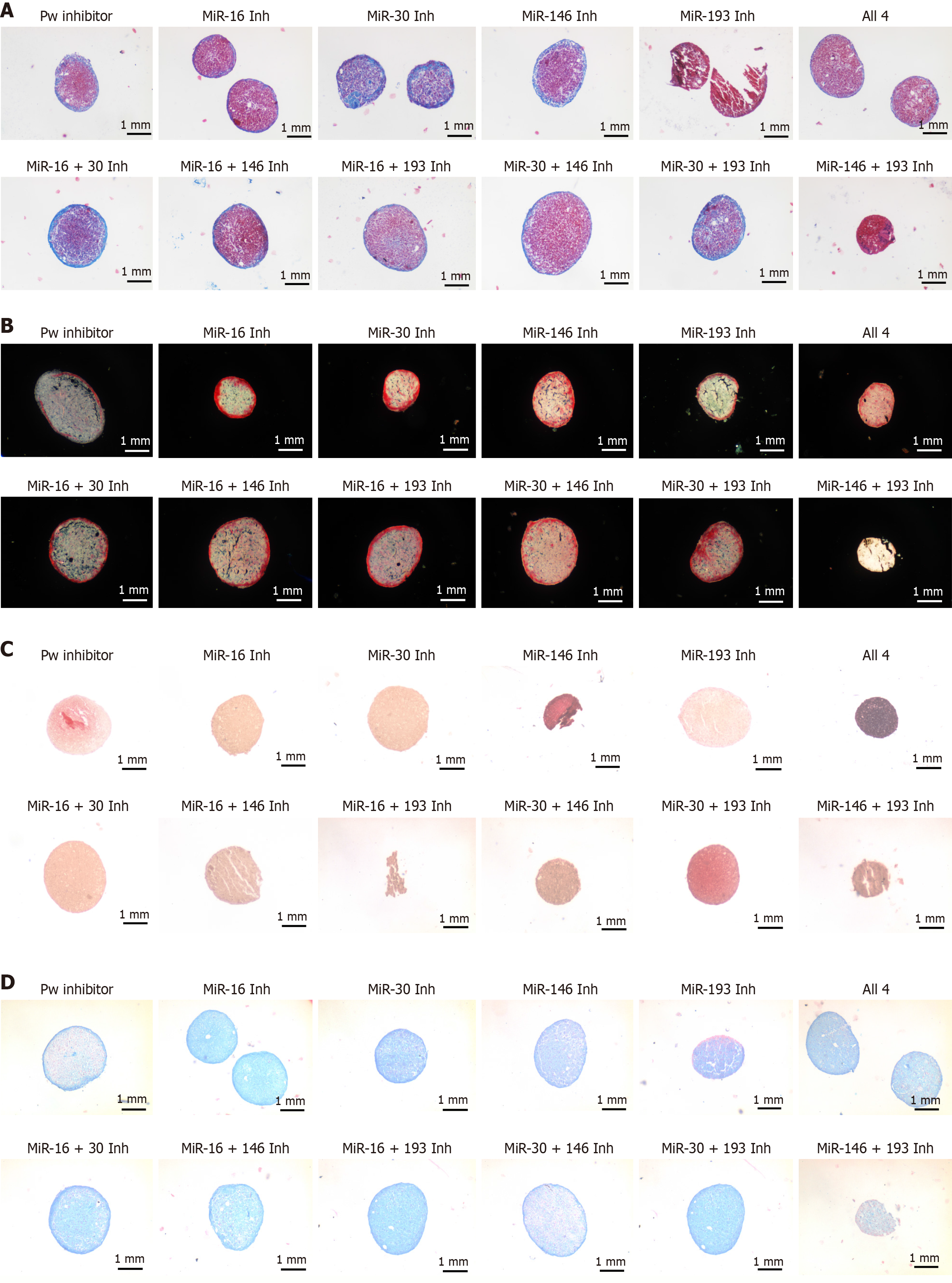
To visualize ECM deposition, sections were stained for collagen (Masson’s trichrome; Picrosirius Red under polarized light) and sGAGs/proteoglycans (Safranin O; Alcian Blue/Fast Red) (Figure 4). Masson’s trichrome highlighted collagen-positive rims in most groups, though miR-193b-3p alone and miR-146a-5p/miR-193b-3p were predominantly pink (reduced blue), suggesting lower collagen/mucosubstances. In conditions miR-30b-5p, miR-16-5p/miR-30b-5p and miR-30b-5p/miR-193b-3p the peripheral blue band appeared thicker and more continuous (Figure 4A). Picrosirius Red under polarized light revealed peripheral collagen fibrils aligned parallel to the micromass surface in most groups (consistent with chondrogenic organization), with the exception of miR-146a-5p/miR-193b-3p, where alignment was less evident. Notably, miR-30b-5p/miR-193b-3p displayed the most abundant, well-organized fibrils extending inside the micromass (Figure 4B).
Across conditions, sGAG/PG signal was detectable by Safranin O and Alcian Blue. Qualitatively, miR-30b-5p/miR-193b-3p showed the strongest Safranin O intensity relative to control, whereas micromasses formed with cells transfected with these individual inhibitors alone appeared fainter. With Alcian Blue/Fast Red, miR-16-5p and miR-30b-5p/miR-193b-3p exhibited the clearest light-blue staining, and miR-16-5p/miR-30b-5p showed a broad light-blue area centrally. Overall, histology indicates ECM formation in all conditions, with the miR-30b-5p/miR-193b-3p combination showing the most favourable profile (strong sGAG/PG staining and well-organized collagen).
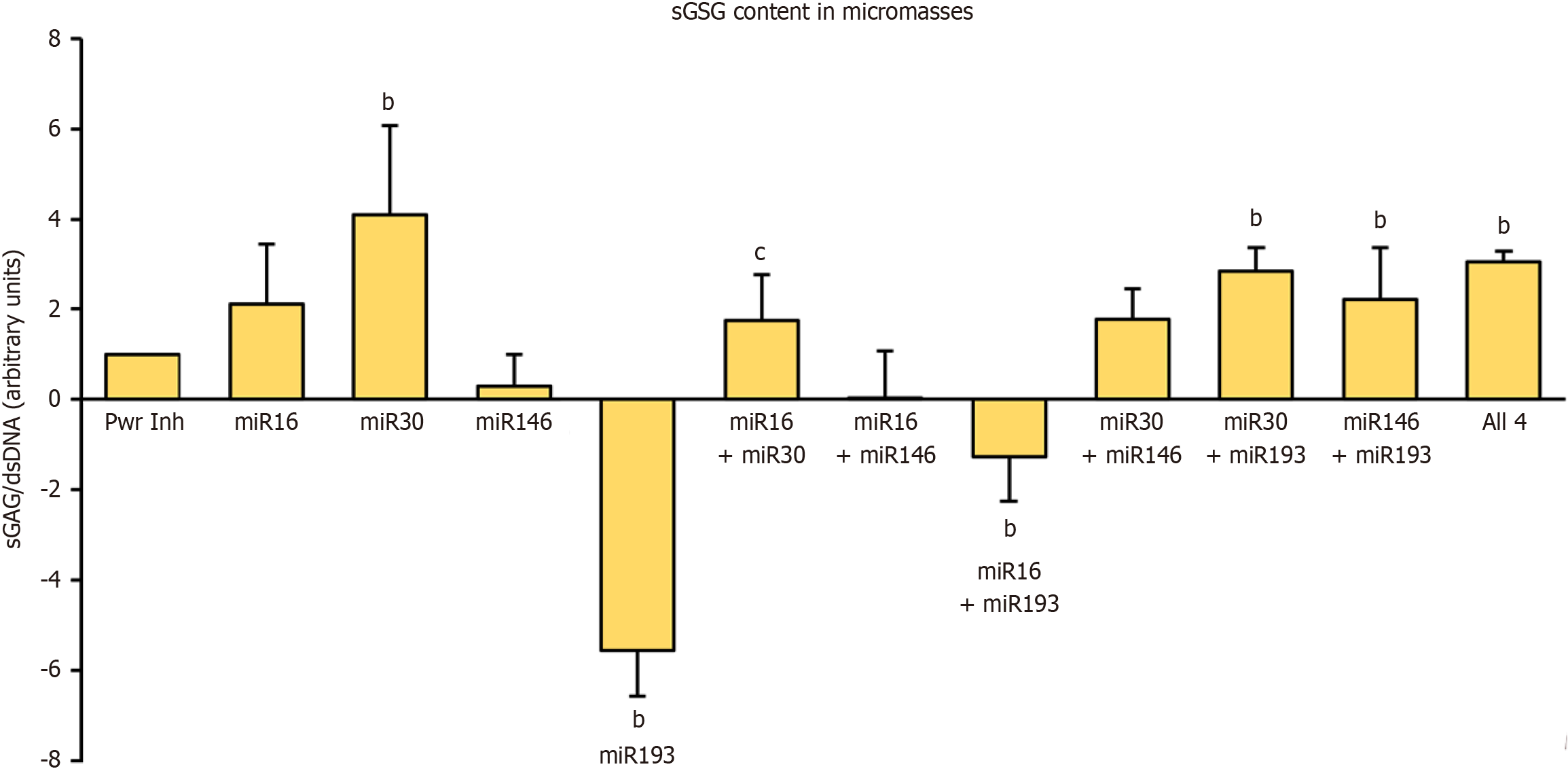
Papain-digested micromasses (day 21) were analyzed by DMMB to quantify sGAG, with values normalised to dsDNA (PicoGreen) to account for cell number. Notably, micromasses generated from MSCs transfected with miR-193b-3p alone, and with miR-30b-5p/miR-193b-3p, showed relatively high DNA content despite smaller macroscopic size, consistent with greater cellular condensation. Normalized sGAG content (sGAG/dsDNA) differed across groups (Figure 5). miR-30b-5p alone, as well as its combinations with miR-16-5p, miR-146a-5p or miR-193b-3p, yielded increased sGAG/dsDNA vs the power inhibitor control. miR-146a-5p alone and miR-146a-5p/miR-193b-3p also showed higher sGAG/dsDNA. The all-four inhibitor mix was likewise elevated. In contrast, miR-193b-3p alone and miR-16-5p/miR-193b-3p displayed reduced sGAG/dsDNA. These data align with the histological stains, in which micromasses from miR-30b-5p/miR-193b-3p exhibited stronger proteoglycan signals.
This study shows that short-pulse, ex vivo inhibition of anti-chondrogenic miRNAs in rMSCs can steer early lineage decisions and enhance matrix deposition, with the miR-30b-5p/miR-193b-3p inhibitor combination emerging as the most favorable. The finding that a short exposure to LNA-ASOs can already shift MSC fate is in line with the broader idea that miRNA networks define narrow windows during which chondrogenic commitment can be redirected. Importantly, this suggests that such windows can be leveraged without requiring any permanent genetic modification[14]. Notably, the aim of this strategy is not sustained miRNA silencing, but rather short, transient ex vivo priming during an early commitment window. In established chondrogenic differentiation models, early molecular cues are sufficient to irreversibly bias lineage commitment, even after the initiating signal has been withdrawn, supporting the translational rationale of a short-pulse priming approach[15,16]. Consistent with this conceptual framework, we show that LNA-ASO-mediated inhibition of miR-30b-5p and miR-193b-3p is progressively attenuated over time (Supplementary Figure 1), confirming the transient nature of the intervention. Notably, despite this gradual recovery of miRNA expression, chondrogenic differentiation proceeds robustly, indicating that early miRNA modulation is sufficient to prime MSCs towards a chondrogenic fate. From a translational point of view, transient priming fits well with advanced therapy considerations, since it minimizes oligonucleotide exposure while keeping manufacturing and release testing tractable[17,18].
Mechanistically, our findings align with reported roles for the miR-30 and miR-193 families in cartilage homeostasis and OA. Integrative human studies report dysregulation of miR-30 family members in OA cartilage with links to catabolic and apoptotic programmes, while restoration of their balance associates with pro-anabolic signalling. Similarly, miR-193 family members have been implicated in early chondrogenesis and matrix metabolism in developmental and OA contexts[8,19]. In this context, our finding that inhibiting miR-30b-5p alone increased Sox9 and lowered Runx2, while the combined inhibition of miR-30b-5p and miR-193b-3p produced a stronger rise in Acan and more robust collagen/GAG outputs, supports the idea that miRNAs can act cooperatively on shared pathways that regulate hypertrophy and matrix production[14,20]. Our histology (Safranin-O, Alcian Blue, Masson’s trichrome, Picrosirius Red) and DMMB results reinforce that combinatorial anti-miR fine-tuning can translate early transcriptional shifts into a macroscopic matrix phenotype underscored by the greater sGAG content and more organized collagen seen with miR-30b-5p/miR-193b-3p. These results align with the view that moderating key hypertrophic regulators such as Runx2 is essential for maintaining a stable, hyaline-like cartilage phenotype and preventing a shift toward fibrocartilage or early ossification[21,22].
We acknowledge that, while the functional and phenotypic outcomes observed in this study are robust, a deeper mechanistic dissection of miRNA-target interactions (e.g., luciferase reporter assays or Argonaute-based approaches) would further strengthen causal inference. These assays were not included here, and we therefore explicitly recognize this as a limitation of the present work. Importantly, the current study was intentionally designed as a first proof-of-concept to establish the feasibility and biological impact of transient miRNA-based MSC priming, rather than to provide an exhaustive molecular mapping of all downstream targets. We consider detailed mechanistic validation an important focus for future studies building upon this platform.
An important translational question is why to pursue ex vivo-primed MSCs rather than direct intra-articular injection of a given anti-miR. While anti-miR injections can be effective in preclinical OA, cartilage is an avascular tissue and is also highly charged. Due to these characteristics, oligonucleotides encounter rapid synovial clearance, limited penetration, and often require repeat dosing[11,12]. By contrast, briefly primed MSCs already lean towards cartilage formation and still release helpful signals[23] (paracrine, trophic and immunomodulatory). Together, this may help them keep a stable phenotype in the inflamed OA niche[17,24]. Moreover, if needed, anti-miR-primed MSCs can be integrated with cartilage-targeting biomaterials to enhance local persistence[25,26].
From a chemistry and safety perspective, LNA-ASOs provide high affinity and nuclease resistance with an increasingly well-defined non-clinical and clinical safety profile[23,27,28]. The short exposure used in this work aligns with the proposed best practice to minimize off-target and class-effect liabilities while preserving on-target potency. From a manufacturing standpoint, ex vivo priming can be integrated into standardized batch-release procedures such as covering identity, purity and potency, and incorporated into an advanced therapy medical product workflow alongside the established release assays used for MSCs[18,29]. Appropriate scrambled LNA controls delivered under identical lipofection conditions were included throughout to control for non-specific effects related to oligonucleotide delivery or transfection-induced cellular stress. Importantly, lipofection alone or scrambled LNA treatment did not induce chondrogenic differentiation or significantly alter chondrogenic or catabolic marker expression. The coherent and selective enhancement of chondrogenic markers and matrix deposition observed upon miR-30b-5p and miR-193b-3p inhibition therefore argues against a dominant contribution of non-specific or stress-related responses.
Our micromass model reproduces early chondrogenic commitment and the start of matrix formation. However, it does not mimic the biomechanical environment or the zonal architecture of a real joint. This limitation is well known for pellet/micromass systems and supports the need to validate our findings in more realistic 3D constructs and in vivo models of OA. Therefore, future work should: (1) evaluate how long the priming effect lasts after implantation; (2) optimize the dose and timing of anti-miR treatment to match the commitment phase of chondrogenesis; and (3) examine whether MSCs primed with anti-miRs cooperate with mechanobiological stimuli, known to restrain hypertrophy and promote hyaline-like matrix formation[30]. In particular, this model does not recapitulate the inflammatory and catabolic cues characteristic of osteoarthritic joints, such as interleukin-1β, tumor necrosis factor-α, or elevated matrix-degrading enzymes, which will need to be addressed in future in vivo and inflammatory challenge studies.
Our findings also relate to the growing interest in cell-free approaches. Brief modulation of MSC miRNA activity may shift the composition of their secretome and EVs in a pro-chondrogenic direction, potentially enabling cell-free products with lower immunogenicity and easier handling[24,31]. A comparative analysis of EVs from primed vs unprimed MSCs would help map the contribution of paracrine vs cell-intrinsic mechanisms to the matrix benefits we observe.
Finally, miRNAs function in networks. Although we deliberately restricted ourselves to a small, OA-relevant panel, the higher performance of the miR-30b-5p/miR-193b-3p combination suggests that rational combinations targeting non-redundant nodes can outperform single inhibitors. Using network-informed strategies that prioritize well-validated targets (e.g., HDAC3, TGFB2/TGFBR3) could boost efficacy without increasing the number of inhibitors required, helping keep the approach simple[20,32,33].
In summary, brief ex vivo anti-miR priming offers a practical way to fine-tune MSC fate before intra-articular delivery, enhancing chondrogenic commitment and matrix formation without introducing permanent genetic change. The convergence of our mechanistic data with independent evidence on miR-30b-5p/miR-193b-3p supports advancing this strategy into scaffold-supported 3D systems and relevant disease models, with a clear path toward clinically compatible manufacturing and safety frameworks[17,34]. By viewing MSCs as a programmable regenerative platform, a short pulse of combinatorial LNA-ASOs, most notably the miR-30b-5p/miR-193b-3p pair, can pre-commit cells towards stable chondrogenesis and improved matrix deposition while remaining compatible with current advanced therapy medical product workflows and avoiding the limitations of direct intra-articular oligonucleotide delivery. Supported by miRNA-network biology and by the clinical trajectory of oligonucleotide therapeutics, this approach provides a realistic way to enhance the consistency and durability of MSC-based therapies for OA and moves the field closer to achieving true structure-modifying outcomes[8,10].
This study shows that lipid-mediated delivery of LNA antisense oligonucleotides provides an efficient and specific strategy to silence selected miRNAs in rMSCs. Effective intracellular uptake was consistently achieved, and functional miRNA inhibition was confirmed both at the level of miRNA expression and through the derepression of established downstream target genes. Together, these results establish this approach as a robust and reproducible platform for modulating miRNA activity in rMSCs. By enabling controlled miRNA inhibition, this system offers a solid basis for future studies exploring the contribution of individual or combined miRNAs to MSC differentiation and regenerative responses in the musculoskeletal system.
During the preparation of this work, the author(s) used an AI language model (ChatGPT) to ensure grammatical accuracy in the English language text. After using this tool, the authors reviewed and edited the content as needed and take full responsibility for the content of the published article.
| 1. | Hunter DJ, Bierma-Zeinstra S. Osteoarthritis. Lancet. 2019;393:1745-1759. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3763] [Cited by in RCA: 3195] [Article Influence: 456.4] [Reference Citation Analysis (3)] |
| 2. | Martel-Pelletier J, Barr AJ, Cicuttini FM, Conaghan PG, Cooper C, Goldring MB, Goldring SR, Jones G, Teichtahl AJ, Pelletier JP. Osteoarthritis. Nat Rev Dis Primers. 2016;2:16072. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1629] [Cited by in RCA: 1379] [Article Influence: 137.9] [Reference Citation Analysis (0)] |
| 3. | González-González A, García-Sánchez D, Dotta M, Rodríguez-Rey JC, Pérez-Campo FM. Mesenchymal stem cells secretome: The cornerstone of cell-free regenerative medicine. World J Stem Cells. 2020;12:1529-1552. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 157] [Cited by in RCA: 139] [Article Influence: 23.2] [Reference Citation Analysis (0)] |
| 4. | Yu H, Huang Y, Yang L. Research progress in the use of mesenchymal stem cells and their derived exosomes in the treatment of osteoarthritis. Ageing Res Rev. 2022;80:101684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 105] [Article Influence: 26.3] [Reference Citation Analysis (0)] |
| 5. | Hwang JJ, Rim YA, Nam Y, Ju JH. Recent Developments in Clinical Applications of Mesenchymal Stem Cells in the Treatment of Rheumatoid Arthritis and Osteoarthritis. Front Immunol. 2021;12:631291. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 119] [Cited by in RCA: 105] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 6. | Barry F, Murphy M. Mesenchymal stem cells in joint disease and repair. Nat Rev Rheumatol. 2013;9:584-594. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 361] [Cited by in RCA: 312] [Article Influence: 24.0] [Reference Citation Analysis (0)] |
| 7. | Gu J, Rao W, Huo S, Fan T, Qiu M, Zhu H, Chen D, Sheng X. MicroRNAs and long non-coding RNAs in cartilage homeostasis and osteoarthritis. Front Cell Dev Biol. 2022;10:1092776. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 8. | Cong L, Zhu Y, Tu G. A bioinformatic analysis of microRNAs role in osteoarthritis. Osteoarthritis Cartilage. 2017;25:1362-1371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 51] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 9. | Nakamura A, Ali SA, Kapoor M. Antisense oligonucleotide-based therapies for the treatment of osteoarthritis: Opportunities and roadblocks. Bone. 2020;138:115461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 28] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 10. | Nakamura A, Rampersaud YR, Nakamura S, Sharma A, Zeng F, Rossomacha E, Ali SA, Krawetz R, Haroon N, Perruccio AV, Mahomed NN, Gandhi R, Rockel JS, Kapoor M. microRNA-181a-5p antisense oligonucleotides attenuate osteoarthritis in facet and knee joints. Ann Rheum Dis. 2019;78:111-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 87] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 11. | Bajpayee AG, Grodzinsky AJ. Cartilage-targeting drug delivery: can electrostatic interactions help? Nat Rev Rheumatol. 2017;13:183-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 277] [Cited by in RCA: 236] [Article Influence: 26.2] [Reference Citation Analysis (0)] |
| 12. | Roberts TC, Langer R, Wood MJA. Advances in oligonucleotide drug delivery. Nat Rev Drug Discov. 2020;19:673-694. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1781] [Cited by in RCA: 1489] [Article Influence: 248.2] [Reference Citation Analysis (0)] |
| 13. | Prosser A, Scotchford C, Roberts G, Grant D, Sottile V. Integrated Multi-Assay Culture Model for Stem Cell Chondrogenic Differentiation. Int J Mol Sci. 2019;20:951. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 24] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 14. | Jonas S, Izaurralde E. Towards a molecular understanding of microRNA-mediated gene silencing. Nat Rev Genet. 2015;16:421-433. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1694] [Cited by in RCA: 1476] [Article Influence: 134.2] [Reference Citation Analysis (0)] |
| 15. | Johnstone B, Hering TM, Caplan AI, Goldberg VM, Yoo JU. In vitro chondrogenesis of bone marrow-derived mesenchymal progenitor cells. Exp Cell Res. 1998;238:265-272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1954] [Cited by in RCA: 1706] [Article Influence: 60.9] [Reference Citation Analysis (0)] |
| 16. | Lefebvre V, Smits P. Transcriptional control of chondrocyte fate and differentiation. Birth Defects Res C Embryo Today. 2005;75:200-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 400] [Cited by in RCA: 361] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 17. | Galipeau J, Sensébé L. Mesenchymal Stromal Cells: Clinical Challenges and Therapeutic Opportunities. Cell Stem Cell. 2018;22:824-833. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1510] [Cited by in RCA: 1351] [Article Influence: 168.9] [Reference Citation Analysis (1)] |
| 18. | Mello DB, Mesquita FCP, Silva Dos Santos D, Asensi KD, Dias ML, Campos de Carvalho AC, Goldenberg RCDS, Kasai-Brunswick TH. Mesenchymal Stromal Cell-Based Products: Challenges and Clinical Therapeutic Options. Int J Mol Sci. 2024;25:6063. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 22] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 19. | Endisha H, Rockel J, Jurisica I, Kapoor M. The complex landscape of microRNAs in articular cartilage: biology, pathology, and therapeutic targets. JCI Insight. 2018;3:e121630. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 87] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 20. | Hausser J, Zavolan M. Identification and consequences of miRNA-target interactions--beyond repression of gene expression. Nat Rev Genet. 2014;15:599-612. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 543] [Cited by in RCA: 481] [Article Influence: 40.1] [Reference Citation Analysis (0)] |
| 21. | Balaskas P, Goljanek-Whysall K, Clegg PD, Fang Y, Cremers A, Smagul A, Welting TJM, Peffers MJ. MicroRNA Signatures in Cartilage Ageing and Osteoarthritis. Biomedicines. 2023;11:1189. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 22. | Stelcer E, Kulcenty K, Rucinski M, Jopek K, Richter M, Trzeciak T, Suchorska WM. The Role of MicroRNAs in Early Chondrogenesis of Human Induced Pluripotent Stem Cells (hiPSCs). Int J Mol Sci. 2019;20:4371. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 21] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 23. | Crooke ST, Baker BF, Crooke RM, Liang XH. Antisense technology: an overview and prospectus. Nat Rev Drug Discov. 2021;20:427-453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 654] [Cited by in RCA: 531] [Article Influence: 106.2] [Reference Citation Analysis (0)] |
| 24. | Cheng A, Choi D, Lora M, Shum-Tim D, Rak J, Colmegna I. Human multipotent mesenchymal stromal cells cytokine priming promotes RAB27B-regulated secretion of small extracellular vesicles with immunomodulatory cargo. Stem Cell Res Ther. 2020;11:539. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 65] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 25. | Li X, Dai B, Guo J, Zheng L, Guo Q, Peng J, Xu J, Qin L. Nanoparticle-Cartilage Interaction: Pathology-Based Intra-articular Drug Delivery for Osteoarthritis Therapy. Nanomicro Lett. 2021;13:149. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 111] [Cited by in RCA: 96] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 26. | Brown S, Kumar S, Sharma B. Intra-articular targeting of nanomaterials for the treatment of osteoarthritis. Acta Biomater. 2019;93:239-257. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 214] [Cited by in RCA: 167] [Article Influence: 23.9] [Reference Citation Analysis (0)] |
| 27. | Seth PP, Tanowitz M, Bennett CF. Selective tissue targeting of synthetic nucleic acid drugs. J Clin Invest. 2019;129:915-925. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 88] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 28. | Lundin KE, Højland T, Hansen BR, Persson R, Bramsen JB, Kjems J, Koch T, Wengel J, Smith CI. Biological activity and biotechnological aspects of locked nucleic acids. Adv Genet. 2013;82:47-107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 82] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 29. | Caplan H, Olson SD, Kumar A, George M, Prabhakara KS, Wenzel P, Bedi S, Toledano-Furman NE, Triolo F, Kamhieh-Milz J, Moll G, Cox CS Jr. Mesenchymal Stromal Cell Therapeutic Delivery: Translational Challenges to Clinical Application. Front Immunol. 2019;10:1645. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 260] [Cited by in RCA: 231] [Article Influence: 33.0] [Reference Citation Analysis (1)] |
| 30. | Zhang L, Su P, Xu C, Yang J, Yu W, Huang D. Chondrogenic differentiation of human mesenchymal stem cells: a comparison between micromass and pellet culture systems. Biotechnol Lett. 2010;32:1339-1346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 191] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 31. | Wong C, Stoilova I, Gazeau F, Herbeuval JP, Fourniols T. Mesenchymal stromal cell derived extracellular vesicles as a therapeutic tool: immune regulation, MSC priming, and applications to SLE. Front Immunol. 2024;15:1355845. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 32. | Meng F, Li Z, Zhang Z, Yang Z, Kang Y, Zhao X, Long D, Hu S, Gu M, He S, Wu P, Chang Z, He A, Liao W. MicroRNA-193b-3p regulates chondrogenesis and chondrocyte metabolism by targeting HDAC3. Theranostics. 2018;8:2862-2883. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 129] [Cited by in RCA: 128] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 33. | Hou C, Yang Z, Kang Y, Zhang Z, Fu M, He A, Zhang Z, Liao W. MiR-193b regulates early chondrogenesis by inhibiting the TGF-beta2 signaling pathway. FEBS Lett. 2015;589:1040-1047. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 56] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 34. | Yu H, Huang C, Kong X, Ma J, Ren P, Chen J, Zhang X, Luo H, Chen G. Nanoarchitectonics of Cartilage-Targeting Hydrogel Microspheres with Reactive Oxygen Species Responsiveness for the Repair of Osteoarthritis. ACS Appl Mater Interfaces. 2022;14:40711-40723. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 59] [Article Influence: 14.8] [Reference Citation Analysis (0)] |