Published online Mar 6, 2026. doi: 10.12998/wjcc.v14.i7.118719
Revised: January 27, 2026
Accepted: February 10, 2026
Published online: March 6, 2026
Processing time: 55 Days and 14.4 Hours
Direct peritoneal resuscitation (DPR) has been primarily studied in trauma and hemorrhagic shock, where it improves visceral perfusion and facilitates abdominal closure. Its application in sepsis and intra-abdominal infection (IAI) has emerged more recently, supported by limited experimental and clinical data. Persistent visceral hypoperfusion, intestinal barrier dysfunction, and inflammatory dysregulation in sepsis provide a biological rationale for peritoneal-based resuscitation strategies. However, the scope, characteristics, and consistency of the available evidence in septic settings remain unclear. We hypothesized that existing studies would be sparse, heterogeneous, and largely non-comparative.
To map and characterize existing evidence on DPR in sepsis and IAI.
A scoping review was conducted following Joanna Briggs Institute methodology and PRISMA-ScR guidelines. PubMed, EMBASE, Scopus, Web of Science, Cochrane Library, and LILACS were searched, supplemented by grey literature and citation tracking. Clinical, neonatal, and experimental studies evaluating DPR or related peritoneal-based resuscitation strategies in sepsis or IAI were eligible. Data were charted descriptively. No quantitative synthesis was performed.
From 1064 records, five studies met inclusion criteria: two adult clinical cohorts, two neonatal studies, and one experimental animal study. Adult and neonatal studies reported associations between DPR and improvements in selected physiological or metabolic parameters, including acid-base status, urine output, and illness severity scores. Adult cohorts also reported higher primary fascial closure rates following damage control surgery. Experimental data suggested protective effects on intestinal physiology and inflammatory markers. Study designs, populations, interventions, and outcomes were heterogeneous, and most evidence was observational or preclinical, with limited comparative data.
Current evidence suggests biological plausibility and feasibility of DPR in selected septic contexts, but data are limited, heterogeneous, and non-comparative, warranting prospective clinical investigation.
Core Tip: Direct peritoneal resuscitation has been proposed as an adjunctive strategy to address persistent visceral hypoperfusion, intestinal barrier injury, and dysregulated inflammation in sepsis. Although most human data derive from trauma populations, emerging experimental and limited clinical evidence suggest potential physiologic and abdominal benefits in selected septic patients, including improvements in metabolic parameters and facilitation of abdominal closure. However, current evidence remains limited and heterogeneous, and significant gaps must be addressed before wider clinical application in sepsis.
- Citation: Ribeiro Junior MAF, Dib Possiedi R, Stefani Pacheco L, de Cesaro Schpchacki N, Nafeesa Hashim S, Monteiro Tavares Pereira B. Direct peritoneal resuscitation in sepsis and intra-abdominal infection: A scoping review. World J Clin Cases 2026; 14(7): 118719
- URL: https://www.wjgnet.com/2307-8960/full/v14/i7/118719.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i7.118719
Sepsis, defined as a life-threatening organ dysfunction caused by a dysregulated host response to infection, remains a major global health challenge and is associated with high morbidity and mortality. Intra-abdominal infection (IAI) is a frequent source of sepsis and septic shock and often requires combined clinical and surgical management, as recom
A serious and potentially fatal complication in critically ill patients, particularly those with abdominal and systemic sepsis, is abdominal compartment syndrome[2]. It is defined as a sustained intra-abdominal pressure exceeding 20 mmHg associated with new organ dysfunction. Sepsis is a well-recognized precipitating factor for secondary abdominal compartment syndrome[2]. Management of this condition frequently requires maintaining the abdomen open, especially in unstable non-trauma patients, both to prevent progression and to allow adequate source control of infection. This approach, however, introduces additional challenges related to fluid balance disturbances, tissue edema, and complex wound management[3,4].
From a pathophysiological perspective, sepsis and shock are characterized by systemic and visceral hypoperfusion. Although conventional intravenous (IV) resuscitation is essential to restore intravascular volume and global hemo
The intestine plays a central role in the development and propagation of organ dysfunction in sepsis. The cytokine surge associated with sepsis increases intestinal permeability, thereby facilitating bacterial translocation and amplifying systemic inflammatory response, ultimately contributing to multiple organ failure[7]. Preservation of intestinal perfusion and barrier integrity has therefore emerged as a critical therapeutic target in septic patients.
Direct peritoneal resuscitation (DPR) has been proposed as an adjunctive resuscitation strategy that directly targets the peritoneal environment. DPR consists of the intraperitoneal infusion of a hypertonic solution, most commonly a peritoneal dialysis solution, administered in addition to standard IV resuscitation[5]. The peritoneum is a complex and metabolically active structure with barrier, immunomodulatory, and drainage functions, while the peritoneal cavity represents an immunologically active compartment housing most abdominal organs[8,9]. It should be emphasized that most of the mechanistic rationale and clinical experience supporting DPR derive from trauma and hemorrhagic shock populations. Although trauma and sepsis share features such as systemic hypoperfusion and microcirculatory dysfunction, their underlying pathophysiology, inflammatory dynamics, and temporal evolution differ substantially. Therefore, extrapolation of trauma-derived DPR findings to septic patients remains uncertain and should be regarded as biologically plausible but unproven.
Available experimental data suggest that DPR, primarily in trauma-related settings, acts as a hypertonic stimulus that promotes vasodilation, improves regional blood flow, and reduces ischemia and hypoxia in visceral organs following shock[5]. In experimental models of sepsis induced by severe IAI, most commonly using cecal ligation and puncture (CLP), DPR has demonstrated a protective effect on intestinal barrier integrity in controlled laboratory settings, with uncertain applicability to human sepsis. Reported benefits include reduction of intestinal edema, decreased serum D-lactate levels as a marker of permeability, restoration of epithelial and mucosal architecture, and modulation of the systemic inflammatory response, with reductions in lactate, tumor necrosis factor-alpha, and interleukin (IL)-6, alongside increased IL-10 levels[7].
The technique of DPR has been implemented using different perfusates. Experimental data suggest that glucose-pyruvate-based peritoneal dialysis solutions may be more effective than glucose-lactate-based solutions in mitigating inflammation and preserving intestinal barrier function in CLP-induced sepsis models[7]. This variability in perfusate composition may contribute to heterogeneity in reported outcomes across studies.
In human subjects, DPR has been studied predominantly in the context of trauma and hemorrhagic shock. In these settings, DPR has been associated with reduced visceral edema and earlier primary fascial closure following damage control surgery, benefits that are directly relevant to the management of the septic open abdomen[10].
Although most clinical evidence originates from trauma populations, the underlying mechanisms targeted by DPR, including microcirculatory dysfunction, intestinal hypoperfusion, and barrier failure, are also fundamental features of sepsis and severe IAI. Given the persistence of visceral ischemia, intestinal barrier injury, and dysregulated inflammation despite conventional resuscitation, DPR has been proposed as a potential adjunctive intervention in septic patients. However, the existing evidence remains limited, heterogeneous, and includes retracted publications[11]. Accordingly, this scoping review aims to map the spectrum of peritoneal-based resuscitation strategies applied in sepsis and septic shock across different populations and clinical contexts, integrating preclinical and clinical evidence and identifying key gaps to inform future research and clinical investigation.
This study was conducted as a scoping review following the methodological guidance of the Joanna Briggs Institute (JBI)[12] for scoping reviews. Reporting followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses Extension for Scoping Reviews (PRISMA-ScR) checklist[13]. A scoping review design was selected to map the extent, nature, and characteristics of the available evidence on DPR in sepsis and IAI, including clinical, neonatal, and experimental studies, and to identify knowledge gaps in this field.
The review protocol was prospectively registered in the Open Science Framework (https://doi.org/10.17605/OSF.IO/V4B75)[14]. Any deviations from the registered protocol were minor and related to refinement of database-specific search strategies, without changes to the eligibility criteria, review questions, or objectives.
In accordance with JBI recommendations for scoping reviews, the review questions were formulated using the population-concept-context framework[12]. The population comprised adults, neonates, and experimental models with sepsis, septic shock, or IAI. The concept of interest was DPR and related peritoneal resuscitative strategies. The context included sepsis, septic shock, IAI, damage control surgery, open abdomen management, and experimental sepsis models.
All original studies evaluating DPR or closely related peritoneal resuscitation strategies in the context of sepsis or IAI were eligible for inclusion. Eligible study designs included prospective and retrospective observational studies, case series, and experimental laboratory studies. For the purposes of this scoping review, peritoneal-based resuscitation strategies were considered eligible when their primary intent was to modulate intra-abdominal physiology, visceral perfusion, or the inflammatory response in sepsis or IAI, even if they predated contemporary hypertonic dialysate-based DPR protocols or differed in technical implementation. Early saline-based peritoneal drainage or lavage approaches were included as historically related peritoneal-based strategies but are conceptually and technically distinct from contemporary hypertonic dialysate-based DPR protocols; their inclusion was intended to map the broader landscape rather than imply therapeutic equivalence. Systematic reviews and narrative reviews were screened for mapping purposes and reference tracking but were not used as sources of primary outcome data. Editorials, conference abstracts without full text, and studies not addressing peritoneal resuscitation in a septic or IAI context were excluded. No restrictions were applied regarding publication year or language.
A comprehensive literature search was performed in PubMed/MEDLINE, EMBASE, Web of Science, Scopus, the Cochrane Library, LILACS, and Google Scholar. Searches were conducted using a combination of controlled vocabulary (e.g., MeSH and Emtree) and free-text terms appropriate to each database, and database-specific search strategies were developed to account for differences in indexing systems and search interfaces[13]. In line with JBI guidance for scoping reviews, the search process was iterative, with refinement of search terms and strategies occurring as familiarity with the literature increased[12]. The complete electronic search strategies for all databases are provided in the Supplementary material. The last search was conducted on December 20, 2025. In addition, reference lists of included studies and key reviews were manually screened to identify additional relevant publications.
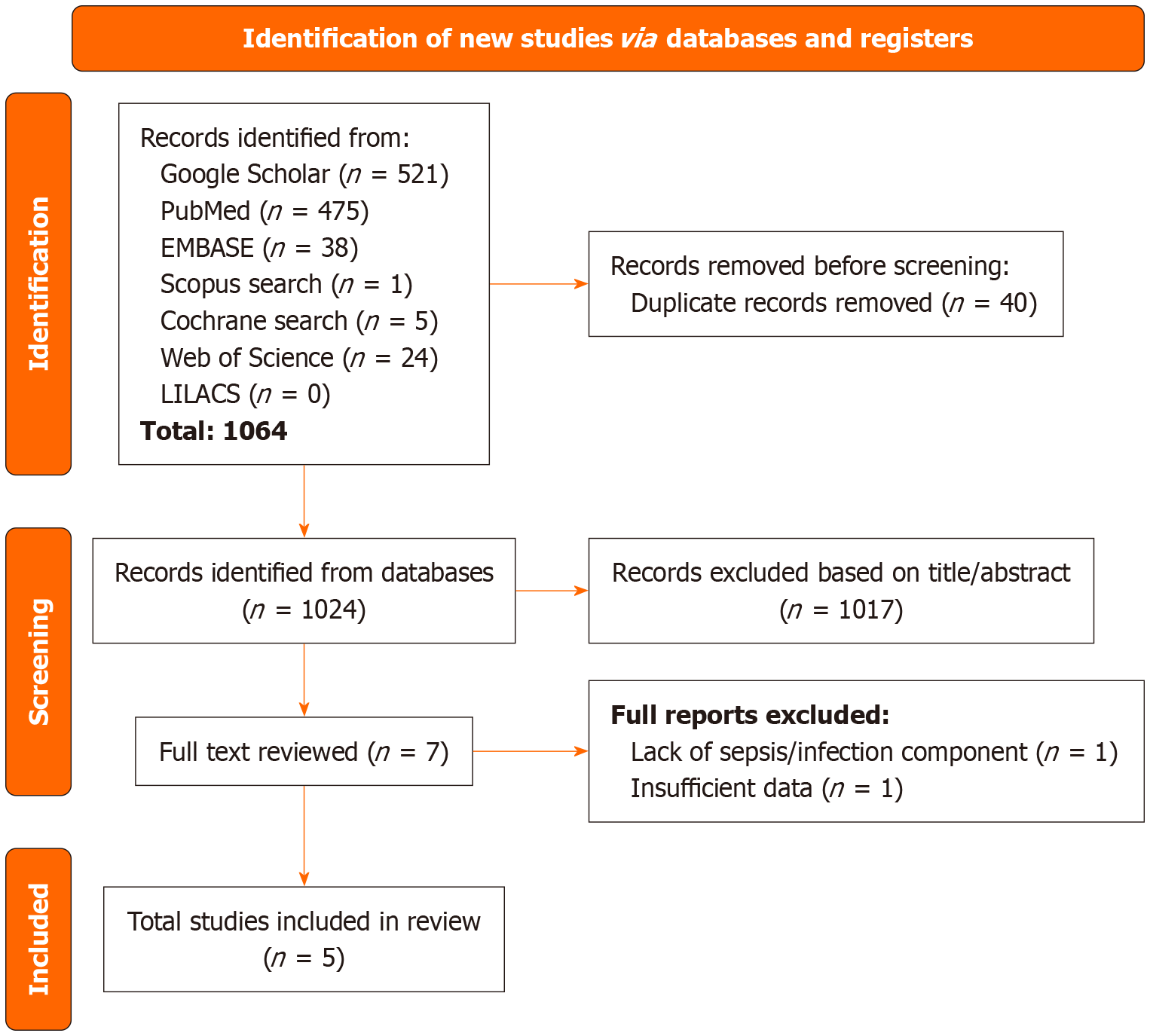
All retrieved records were imported into reference management software for deduplication using Zotero. Prior to formal screening, the reviewers conducted an initial calibration exercise to ensure consistent application of the eligibility criteria. Records were then uploaded to the Rayyan platform to facilitate study screening. Titles and abstracts were independently screened by two reviewers. When abstracts were unavailable, full-text articles were screened to determine eligibility. Full texts of potentially eligible studies were subsequently assessed for inclusion. Disagreements at any stage were resolved by discussion between the two reviewers, with adjudication by a third reviewer when consensus could not be reached, in accordance with JBI guidance[12]. The study selection process is summarized in a PRISMA-ScR flow diagram.
Data charting was performed using a standardized and pilot-tested form, consistent with JBI scoping review metho
Although critical appraisal is not mandatory in scoping reviews, an assessment of methodological quality was conducted for observational human studies to contextualize the available clinical evidence[12]. Methodological quality was assessed using the Newcastle–Ottawa Scale (NOS) for cohort and case-control studies[15]. Experimental laboratory studies, mechanistic studies, and case reports were not assessed using the NOS, as this tool is not applicable to non-observational human studies. The results of the quality assessment were not used to determine study eligibility or to weight findings, but rather to provide descriptive context regarding methodological strengths and limitations. The known limitations of the NOS, including concerns related to construct validity and the use of summary scores, were acknowledged[15].
Results were synthesized descriptively. No quantitative synthesis or meta-analysis was planned or performed, as scoping reviews aim to map the breadth of evidence rather than to generate pooled effect estimates, and because of substantial heterogeneity in study designs, populations, interventions, and outcomes[12,13]. Findings are presented narratively and in tabular form to map the existing evidence and highlight research gaps.
Of the 1064 records identified, 1024 were screened after removal of duplicates. Seven studies were assessed in full text, of which five met the inclusion criteria, as shown in the PRISMA-ScR flow diagram (Figure 1). All included studies were published as full reports. Three observational studies achieved higher NOS scores, ranging from 6 to 9[16-18], while two studies did not apply to this quality assessment[7,19]. Baseline characteristics are presented in Table 1, and key reported findings are summarized in Table 2.
| Ref. | Study design | Male sex at direct peritoneal resuscitation group | Age at direct peritoneal resuscitation group | Intervention | Total population | Context | Follow-up | Peritoneal solution | Direct peritoneal resuscitation duration |
| Smith et al[16], 2014 | Prospective cohort | 27 | 52 ± 12 years | Direct peritoneal resuscitation | Adults (n = 44 vs 44) | Sepsis patients undergoing damage control surgery | Early physiologic assessment at 24-48 hours; in-hospital follow-up | 2.5% DELFLEX | 800 mL bolus, then 400 mL/hour continuous infusion until repeat laparotomy |
| Ji et al[7], 2025 | Laboratory-controlled trial (in vivo) animal model | Not applicable | Not applicable | Direct peritoneal resuscitation | Rats (n = 48, 6 groups, n = 8 per group) | Sepsis induced by cecal ligation and puncture | 24 hours | Peritoneal lavage with normal saline, Lac-peritoneal dialysis solution (peritoneal lavage with 2.5% Glu-lac-peritoneal dialysis solution) and Pyr-peritoneal dialysis solution (peritoneal lavage with 2.5% Glu-Pyr-peritoneal dialysis solution) (80 mL/hour) | 3 hours (80 mL/hour) |
| Hopkins et al[19], 2011 | Two case-series | 1 (50%) | 31-week gestation and 27-week gestation | Direct peritoneal resuscitation | Infants (n = 2) | Necrotizing enterocolitis | Not mentioned | 2.5% dextrose-based peritoneal dialysis solution with added antiseptic (Clorpactin® WCS-90) ± antibiotic (cefazolin) | 25-50 mL or 25 mL/kg every 8-12 hours, intermittent, duration guided by clinical tolerance (up to 7 days) |
| Okumura et al[17], 2022 | Prospective single-arm cohort | 23 (62%) | Median age (interquartile range) = 62 (53-70) years | Direct peritoneal resuscitation | Adults (n = 37) | Ischemic bowel and sepsis undergoing damage control surgery | 30 days (in-hospital) | 2.5% glucose-based peritoneal dialysis solution (DELFLEX®) | Median 4 days (interquartile range 2-8) |
| Cheu et al[18], 1988 | Retrospective cohort | Not reported | 28.8 ± 3.5 weeks | Primary peritoneal drainage | Infants (n = 92) | Neonates with necrotizing enterocolitis | 2 months-3 years | Warm saline (intraoperative irrigation) | Intermittent drainage; no defined infusion duration |
| Ref. | Study design | Selection | Comparability | Outcome | Total |
| Smith et al[16], 2014 | Sequential prospective cohort | 4 | 2 | 3 | 9 |
| Okumura et al[17], 2022 | Prospective cohort | 4 | 1 | 3 | 8 |
| Cheu et al[18], 1988 | Retrospective cohort | 3 | 0 | 3 | 6 |
| Ji et al[7], 2025 | Not applicable (non-comparative/mechanistic) | Not applicable | Not applicable | Not applicable | Not applicable |
| Hopkins et al[19], 2011 | Case report (2 cases) | Not applicable | Not applicable | Not applicable | Not applicable |
The included studies comprised adult clinical cohorts[16,17], neonatal case series and cohorts[18,19], and one experimental animal model[7], with sample sizes ranging from small case series to cohorts of up to 92 participants. Sex distribution was inconsistently reported. In adult cohorts, mean or median age ranged from the early 50s to early 60s, with Smith et al[16] reporting a mean age of 52 ± 12 years and Okumura et al[17] a median age of 62 years (interquartile range: 53-70). In neonatal studies, reported gestational ages reflected extreme prematurity, including infants born at 27 and 31 weeks in a case series[19], and a mean gestational age of 28.8 ± 3.5 weeks in a retrospective cohort[18]. Age was not applicable to the experimental animal study[7].
Clinical contexts included sepsis and ischemic bowel in adults undergoing damage control surgery in trauma and non-trauma patients[16,17], necrotizing enterocolitis in neonates[18,19], and experimental sepsis in an animal model[7]. Follow-up duration ranged from short-term observation measured in hours to days, with longer-term follow-up reported in some studies, although incompletely described in others[16,17,19]. DPR interventions most commonly used 2.5% hypertonic dialysis solutions[16,17,19], while earlier neonatal protocols employed saline irrigation[18], and experimental models compared pyruvate- and lactate-based solutions[7].
Together, these studies map the populations, clinical contexts, interventions, reported outcomes, and evidence gaps related to DPR in sepsis and IAI, in direct alignment with the objectives of this scoping review.
The impact of DPR on abdominal wall closure has been mainly assessed in adults undergoing damage control surgery. Higher primary fascial closure rates were reported with DPR, with Smith et al[16] describing closure in 68% of patients in the DPR group compared with 43% in controls, together with a shorter time to definitive abdominal closure (5.9 days vs 7.7 days)[16]. Comparable results were observed by Okumura et al[17], who reported primary abdominal closure in 81% of patients treated with DPR for severe intra-abdominal conditions, including ischemic bowel and sepsis, in the absence of a contemporaneous control group[17].
Postoperative morbidity was variably reported in adult cohorts. Surgical-site infection was the most frequent complication among survivors, occurring in 38%, followed by fascial dehiscence in 28% and intra-abdominal abscess in 24%[17]. In a propensity-matched analysis, DPR was associated with a lower overall risk of abdominal complications, with rates of 27% compared with 47% in controls, corresponding to a relative risk of 0.57[16].
In neonatal populations, abdominal complications were described in the context of necrotizing enterocolitis management rather than abdominal closure. Additional complications included fistulae and adhesions, while short-gut syndrome and intra-abdominal abscesses were not observed among survivors treated with primary peritoneal drainage (PPD) alone[18].
Survival outcomes differed substantially according to population and clinical context. In the experimental animal study, post-intervention survival was higher among animals treated with peritoneal lavage using hypertonic dialysis solutions, with both lactate- and pyruvate-based solutions achieving survival rates of 75%, compared with 62.5% in the saline lavage group and 50% with conventional resuscitation[7]. In adult clinical studies involving patients with severe intra-abdominal pathology and sepsis managed with damage control laparotomy and DPR, reported 30-day mortality was 21.6%, corresponding to a survival rate of 78.4%[17].
In neonatal necrotizing enterocolitis, survival was reported in both very low-birth-weight infants treated with preoperative DPR in a small case series[19]. In a larger neonatal cohort, operative survival was 83% following primary laparotomy and 57% following PPD[18]. Outcomes varied by birth weight, with survival rates of 57% vs 52% in infants weighing < 1000 g and 86% vs 62% in those weighing > 1000 g after laparotomy and PPD, respectively[18]. Infants managed with PPD were significantly less mature and exhibited higher rates of intraventricular hemorrhage and patent ductus arteriosus, reflecting greater baseline illness severity[18]. When nine moribund infants treated with PPD as a last-resort intervention were excluded, survival in the drainage group increased to 93%[18].
Improvements in metabolic and hemodynamic parameters were reported primarily in adult and neonatal clinical studies. In adult patients, normalization of blood pH at 48 hours was observed following DPR, with a mean pH of 7.33 compared with 7.22 in control patients[16]. Reductions in illness severity were also documented in adult cohorts, with median Acute Physiology Score values decreasing from 31 to 19 and concurrent improvements in APACHE IV scores, reaching statistical significance among survivors[17].
In neonatal reports, peritoneal-based interventions were associated with increased urine output and reduced vasopressor requirements within 48 hours of initiation. Urine output increased from 0.4 to 10 mL/hour, accompanied by decreased dopamine support[19].
Hematological responses were reported only in pediatric populations. Despite rapid clinical improvement following peritoneal therapy, including improved skin color, spontaneous breathing, and urine output, persistent thrombocytopenia was observed, with platelet counts ranging from 23000/mm³ to 33000/mm³ during treatment. Platelet recovery occurred only after definitive surgical resection of necrotic bowel tissue[19].
Adverse events were variably reported across adult and neonatal studies. In adult cohorts, a lower overall proportion of abdominal complications was observed with DPR compared with controls, with rates of 27% vs 47% and a relative risk of 0.57[16]. Among survivors, surgical-site infection was the most frequently reported complication, followed by intra-abdominal abscess and fascial dehiscence, with some events occurring in association with 30-day mortality[17]. In neonatal cases, transient intraperitoneal fluid retention with abdominal distension and generalized edema were described during the course of peritoneal therapy, leading to adjustment and eventual discontinuation of peritoneal infusions[19].
This scoping review mapped the available experimental and clinical evidence on DPR and related peritoneal-based resuscitation strategies applied in sepsis and IAI. The identified literature is limited in volume and heterogeneous in terms of population, intervention characteristics, and outcomes, encompassing adult surgical cohorts, neonatal populations with necrotizing enterocolitis, and experimental animal models[7,16-19]. Accordingly, the findings of this review should be interpreted as a descriptive synthesis aimed at identifying patterns, mechanistic plausibility, and knowledge gaps rather than establishing causal effectiveness.
From a pathophysiological standpoint, the rationale for peritoneal-based resuscitation in sepsis is supported by growing evidence that conventional IV resuscitation may fail to adequately restore visceral microcirculatory flow and intestinal perfusion[20]. In the context of IAI, sepsis is a complex syndrome associated with impaired tissue perfusion, global hypoxia, and progression to multiple organ dysfunction, driven by an uncontrolled systemic inflammatory response[1]. The peritoneum is a biologically active compartment, and modulation of the peritoneal environment has been proposed as a strategy to influence regional perfusion, organ edema, and inflammatory mediator release[5,21].
In adult clinical populations, DPR has been evaluated primarily in the setting of open abdomen management following damage control surgery, where higher primary fascial closure rates and fewer abdominal complications have been reported[16]. Similarly, in adults with severe intra-abdominal sepsis or ischemic bowel, DPR was associated with a high rate of definitive abdominal closure[17]. Taken together, these observations are consistent with a potential role for DPR in reducing visceral edema and facilitating earlier closure in selected patients, while the non-comparative nature of the evidence precludes causal inference.
Beyond abdominal closure, adult studies also reported improvements in physiological and severity-related parameters following DPR, with higher arterial pH and lower severity scores observed within 48 hours compared with standard care, indicating early global physiological improvement after damage control surgery[16]. In severely ill adult patients undergoing damage control surgery, DPR was associated with significant reductions in Acute Physiology Score and APACHE IV scores among survivors during the early postoperative period[17]. These improvements were confined to physiological severity metrics and represent surrogate outcomes, not evidence of a mortality benefit.
Evidence in neonatal populations is substantially more limited and heterogeneous. In a small case series of very low-birth-weight infants with necrotizing enterocolitis, preoperative DPR was associated with early clinical stabilization, increased urine output, and reduced vasopressor requirements, suggesting improved visceral and renal perfusion[19]. However, clinically relevant adverse effects were also reported, including intraperitoneal fluid retention, abdominal distension, and generalized edema, which prompted adjustment or discontinuation of therapy. Moreover, hematological recovery lagged behind clinical improvement, with persistent thrombocytopenia resolving only after definitive surgical resection[19].
Historical data from neonatal cohorts further illustrate the complexity of interpreting peritoneal-based strategies in this population. In a retrospective study comparing primary laparotomy with PPD with saline lavage for necrotizing enterocolitis, survival outcomes varied according to baseline severity and birth weight, with infants managed with peritoneal drainage being significantly less mature and more severely ill at baseline[18]. Although survival after this approach appeared high after exclusion of moribund “last-resort” cases, this observation reflects substantial confounding by indication and underscores the difficulty of attributing outcome differences to the intervention itself[18]. Importantly, this historical strategy, based on decompression and saline lavage, differs technically and conceptually from contemporary hypertonic dialysate-based DPR, reinforcing the need to interpret neonatal data cautiously.
Experimental evidence provides underlying insight into the potential effects of DPR but has limited direct clinical translatability. In a rat model of sepsis induced by CLP, peritoneal lavage with glucose-based dialysis solutions resulted in higher survival compared with conventional resuscitation or saline lavage, while pyruvate- and lactate-based solutions demonstrated equivalent survival rates[7]. Notably, pyruvate-based solutions showed greater efficacy in preserving intestinal barrier integrity and reducing inflammatory mediators, including tumor necrosis factor-alpha and IL-6, as well as maintaining tight junction proteins such as occludin and claudin-1[7]. These findings suggest mechanistic advantages of specific perfusates, although their relevance to human sepsis remains uncertain.
From a technical standpoint, different intraperitoneal fluids have been used in DPR across experimental and clinical studies, with distinct physiological profiles. Contemporary DPR protocols most commonly employ hypertonic glucose-based peritoneal dialysis solutions, which promote visceral vasodilation, improve microcirculatory flow, and reduce organ edema, particularly in the setting of shock and open abdomen management[16,17,21]. Experimental models have further explored lactate- and pyruvate-buffered dialysates, with pyruvate-based solutions demonstrating greater preservation of intestinal barrier integrity and attenuation of inflammatory mediators compared with lactate-based formulations, although survival benefits appear comparable[7]. In contrast, early neonatal and historical approaches primarily relied on isotonic saline peritoneal drainage or lavage, aiming at decompression and contamination control rather than targeted microcirculatory resuscitation[18]. While saline-based strategies may reduce intra-abdominal pressure and provide temporary physiological stabilization, they lack the hypertonic and metabolic properties hypothesized to underlie the benefits of contemporary DPR and are therefore conceptually and technically distinct. The choice of perfusate thus represents a key variable influencing both physiological effects and potential risks, underscoring the need for standardized protocols in future clinical studies.
Complementary experimental and translational literature further supports the concept that modulation of the peritoneal environment may influence systemic inflammation and organ dysfunction. Experimental models have demonstrated that DPR can attenuate cytokine release, limit cellular injury markers, and preserve endothelial and epithelial integrity under conditions of shock and systemic inflammation[5,22]. Although these studies did not meet the predefined inclusion criteria for primary evidence in this scoping review, they are discussed here to provide biological context for interpreting the observed clinical and experimental findings.
From a practical perspective, implementation of DPR requires specific technical expertise and institutional infra
Several limitations inherent to the available evidence must be acknowledged. The literature is sparse and dominated by observational studies, case series, and experimental models, with no randomized controlled trials specifically evaluating DPR in sepsis. Interventions varied substantially with respect to solution composition, dosing, timing, duration, and clinical intent, and outcomes were inconsistently reported. Similarly, although acute physiological severity scores were reported in some adult studies, heterogeneity in patient populations, scoring systems, timing of assessment, and outcome definitions precluded meaningful pre- and post-intervention comparative analysis within the scope of this scoping review. Infectious outcomes related to DPR were inconsistently reported across the included studies, with limited detail regarding microbiological profiles or specific infectious complications, highlighting an important knowledge gap, including the potential risk of bacterial or fungal infections, that warrants further investigation. The intentional inclusion of historical peritoneal-based strategies alongside contemporary DPR protocols reflects the exploratory nature of this scoping review but limits comparability across studies. Confounding by indication, particularly in neonatal cohorts, and incomplete follow-up further constrain interpretation[12,13,18,19].
In summary, the existing evidence suggests that peritoneal-based resuscitation strategies, including DPR, are biologically plausible and may confer physiological and technical advantages in selected contexts of sepsis and IAI. However, the current evidence base is insufficient to support definitive conclusions regarding efficacy or optimal implementation. Future research should focus on clearly defining DPR protocols, standardizing outcomes, and evaluating safety and effectiveness in well-designed prospective clinical studies, particularly in non-trauma septic populations.
Available studies suggest biological plausibility and report associations with physiological improvement and facilitation of abdominal wall management in selected adult, neonatal, and experimental settings. However, the evidence base is sparse, methodologically diverse, and largely observational or preclinical, with substantial variability in interventions and outcomes. As a result, the role of DPR in sepsis remains undefined, and well-designed prospective clinical studies are needed to clarify its clinical applicability and safety, including appropriate patient selection, timing of intervention, safety endpoints, and standardized outcome measures.
| 1. | Sartelli M, Viale P, Catena F, Ansaloni L, Moore E, Malangoni M, Moore FA, Velmahos G, Coimbra R, Ivatury R, Peitzman A, Koike K, Leppaniemi A, Biffl W, Burlew CC, Balogh ZJ, Boffard K, Bendinelli C, Gupta S, Kluger Y, Agresta F, Di Saverio S, Wani I, Escalona A, Ordonez C, Fraga GP, Junior GA, Bala M, Cui Y, Marwah S, Sakakushev B, Kong V, Naidoo N, Ahmed A, Abbas A, Guercioni G, Vettoretto N, Díaz-Nieto R, Gerych I, Tranà C, Faro MP, Yuan KC, Kok KY, Mefire AC, Lee JG, Hong SK, Ghnnam W, Siribumrungwong B, Sato N, Murata K, Irahara T, Coccolini F, Segovia Lohse HA, Verni A, Shoko T. 2013 WSES guidelines for management of intra-abdominal infections. World J Emerg Surg. 2013;8:3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 222] [Cited by in RCA: 176] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 2. | Pereira B, Dorigatti A, Melek M, Dos Santos J, Ferreira M, Calderan T, Carmona C, Fraga G. Septic shock patients admitted to the intensive care unit with higher SOFA score tend to have higher incidence of abdominal compartment syndrome - a preliminary analysis. Anaesthesiol Intensive Ther. 2019;51:370-372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 3. | Coccolini F, Montori G, Ceresoli M, Catena F, Moore EE, Ivatury R, Biffl W, Peitzman A, Coimbra R, Rizoli S, Kluger Y, Abu-Zidan FM, Sartelli M, De Moya M, Velmahos G, Fraga GP, Pereira BM, Leppaniemi A, Boermeester MA, Kirkpatrick AW, Maier R, Bala M, Sakakushev B, Khokha V, Malbrain M, Agnoletti V, Martin-Loeches I, Sugrue M, Di Saverio S, Griffiths E, Soreide K, Mazuski JE, May AK, Montravers P, Melotti RM, Pisano M, Salvetti F, Marchesi G, Valetti TM, Scalea T, Chiara O, Kashuk JL, Ansaloni L. The role of open abdomen in non-trauma patient: WSES Consensus Paper. World J Emerg Surg. 2017;12:39. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 92] [Cited by in RCA: 74] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 4. | Pereira B, Duchesne J, Concon-Filho A, Leppãniemi A. Entero-atmospheric fistula migration: a new management alternative for complex septic open abdomen. Anaesthesiol Intensive Ther. 2020;52:56-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 5. | Weaver JL, Smith JW. Direct Peritoneal Resuscitation: A review. Int J Surg. 2016;33:237-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 6. | Ribeiro-Junior MAF, Costa CTK, de Souza Augusto S, Néder PR, Elia YGB, Rattan R; Peritoneal Resuscitation Study Group, Di Saverio S. The role of direct peritoneal resuscitation in the treatment of hemorrhagic shock after trauma and in emergency acute care surgery: a systematic review. Eur J Trauma Emerg Surg. 2022;48:791-797. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 7. | Ji J, Zhao Z, Ming L, Luo Z, Li M, Tian W, Yang F, Huang Q. Investigating the protective effect of direct peritoneal resuscitation on intestinal barrier function in rat models of sepsis. J Trauma Acute Care Surg. 2025;99:264-271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 8. | Isaza-Restrepo A, Martin-Saavedra JS, Velez-Leal JL, Vargas-Barato F, Riveros-Dueñas R. The Peritoneum: Beyond the Tissue - A Review. Front Physiol. 2018;9:738. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 68] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 9. | Liu M, Silva-Sanchez A, Randall TD, Meza-Perez S. Specialized immune responses in the peritoneal cavity and omentum. J Leukoc Biol. 2021;109:717-729. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 81] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 10. | Smith JW, Garrison RN, Matheson PJ, Franklin GA, Harbrecht BG, Richardson JD. Direct peritoneal resuscitation accelerates primary abdominal wall closure after damage control surgery. J Am Coll Surg. 2010;210:658-664, 664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 46] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 11. | Luo X, Jian D, Lv Z. The beneficial effect of direct peritoneal resuscitation on septic shock in rats. J Biomed Biotechnol. 2011;2011:743763. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 12. | Peters MD, Godfrey CM, Khalil H, McInerney P, Parker D, Soares CB. Guidance for conducting systematic scoping reviews. Int J Evid Based Healthc. 2015;13:141-146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5522] [Cited by in RCA: 4061] [Article Influence: 369.2] [Reference Citation Analysis (2)] |
| 13. | Tricco AC, Lillie E, Zarin W, O'Brien KK, Colquhoun H, Levac D, Moher D, Peters MDJ, Horsley T, Weeks L, Hempel S, Akl EA, Chang C, McGowan J, Stewart L, Hartling L, Aldcroft A, Wilson MG, Garritty C, Lewin S, Godfrey CM, Macdonald MT, Langlois EV, Soares-Weiser K, Moriarty J, Clifford T, Tunçalp Ö, Straus SE. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Ann Intern Med. 2018;169:467-473. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31615] [Cited by in RCA: 23917] [Article Influence: 2989.6] [Reference Citation Analysis (3)] |
| 14. | Dib Possiedi R, Junior MAFR, Pach LS, de Cesaro Schpchacki N, Hashim SN, Pereira BMT. Direct Peritoneal Resuscitation in Sepsis and Septic Shock: A Scoping Review Protocol. 2025. [DOI] [Full Text] |
| 15. | Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. 2010;25:603-605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14281] [Cited by in RCA: 13915] [Article Influence: 869.7] [Reference Citation Analysis (8)] |
| 16. | Smith JW, Neal Garrison R, Matheson PJ, Harbrecht BG, Benns MV, Franklin GA, Miller KR, Bozeman MC, David Richardson J. Adjunctive treatment of abdominal catastrophes and sepsis with direct peritoneal resuscitation: indications for use in acute care surgery. J Trauma Acute Care Surg. 2014;77:393-8; discussion 398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 40] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 17. | Okumura K, Latifi R, Smiley A, Lee JS, Shnaydman I, Zangbar B, Bronstein M, Con J, Prabhakaran K, Rhee P, Klein J, Shivaraj K, Klein MD, Miller DM. Direct Peritoneal Resuscitation (DPR) Improves Acute Physiology and Chronic Health Evaluation (APACHE) IV and Acute Physiology Score When Used in Damage Control Laparotomies: Prospective Cohort Study on 37 Patients. Surg Technol Int. 2022;41:85-93. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 18. | Cheu HW, Sukarochana K, Lloyd DA. Peritoneal drainage for necrotizing enterocolitis. J Pediatr Surg. 1988;23:557-561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 67] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 19. | Hopkins JW, Chandramouli B, Wall P. Preliminary resuscitation for perforated necrotizing enterocolitis: 2 cases treated with initial direct peritoneal resuscitation. J Pediatr Surg. 2011;46:237-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 20. | McGuirk M, Kajmolli A, Gachabayov M, Haider A, Bronstein M, Spatz D, Gwardshaladse C, Latifi R. Use of Direct Peritoneal Resuscitation for Intra-Abdominal Catastrophes: A Technical Note. Surg Technol Int. 2020;37:127-131. [PubMed] |
| 21. | Ribeiro Junior MAF, Pacheco LS, Duchesne JC, Parreira JG, Mohseni S. Damage control resuscitation: how it's done and where we can improve. A view of the Brazilian reality according to trauma professionals. Rev Col Bras Cir. 2025;51:e20243785. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 22. | Zhao Z, Ji J, Ming L, Luo Z, Li M, Chen Y, Sun R, Lu W, Tian W, Yang F, Huang Q. Direct Peritoneal Resuscitation and Melatonin in the Treatment of Abdominal Sepsis-Induced Lung Injury via Macrophage Metabolic Reprogramming. J Pineal Res. 2025;77:e70066. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/