Published online Mar 6, 2026. doi: 10.12998/wjcc.v14.i7.117882
Revised: January 26, 2026
Accepted: February 11, 2026
Published online: March 6, 2026
Processing time: 77 Days and 13.8 Hours
Neurogenic lower urinary tract dysfunction often requires long-term indwelling catheterisation, which is associated with catheter blockages and catheter-asso
To evaluate the feasibility and patient-reported outcomes of an acoustic wave device in patients with recurrent suprapubic catheter blockage.
This observational product evaluation included patients with neurogenic bladder and recurrent suprapubic catheter blockages. Participants used the UroShield™ device for 12 weeks. Catheter blockages, catheter satisfaction, pain, and sleep quality were assessed at baseline and during follow-up.
Ten patients were enrolled; six completed the 12-week evaluation. A reduction in reported catheter blockages and improvements in catheter satisfaction was observed. Four patients withdrew due to bladder spasms, skin irritation, or device practicality issues.
Use of an acoustic wave device was feasible in selected patients and was asso
Core Tip: Recurrent suprapubic catheter blockage is a major burden for patients with neurogenic bladder. This observational product evaluation explored the feasibility and patient-reported outcomes of an acoustic wave device designed to reduce biofilm formation on indwelling catheters. While limited by small sample size and lack of a control group, the findings suggest potential benefits in selected patients and support further controlled investigation.
- Citation: Tsikopoulos I, Knight SL, Katsimperis S, Riley C, Helal M, Nobrega R. Acoustic wave device for prevention of recurrent suprapubic catheter blockage in neurogenic bladder. World J Clin Cases 2026; 14(7): 117882
- URL: https://www.wjgnet.com/2307-8960/full/v14/i7/117882.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i7.117882
Many people with neurogenic lower urinary tract dysfunction secondary to spinal cord injury or multiple sclerosis rely on long-term indwelling catheters for bladder management[1]. Indwelling catheters (either supra-pubic or urethral) are indicated if patients are unable to perform intermittent catheterisation (National Institute for Health and Care Excellence, 2017). Catheter-associated urinary tract infections and catheter blockages are a major cause of morbidity and mortality in patients with indwelling catheters.
Urinary catheters can become colonized by bacteria soon after insertion, and prolonged catheterization increases the likelihood of biofilm development, which may contribute to urinary tract infections and catheter encrustation leading to blockage[2-4]. Urinary tract infections and catheter blockages can be a source of distress for patients and necessitate immediate attention by either the local district nursing or accident and emergency teams. Therefore, people with lower urinary tract dysfunction who rely on indwelling catheters to manage their bladder are susceptible to infections and blockages, which can decrease quality of life and increase healthcare costs due to frequent unplanned catheter changes and accident and emergency visits[5].
There is a variety of interventions that can be utilized to prevent and manage catheter blockages. The most common is the use of catheter irrigation/washout with either saline or a maintenance solution. However, there is insufficient evidence at present to determine whether these are beneficial in the long term[6]. The UroShield™ System (NanoVibronix, Ideal Medical Solutions, KT, United Kingdom) is a disposable medical device that has been developed to prevent bacterial biofilm formation by means of surface acoustic waves (SAW). The acoustic waves generated by the device interfere with the attachment of bacteria on the surface of the catheter, which is the initial step in biofilm formation. In addition, the device is reported to decrease the pain, discomfort, and spasm associated with indwelling urinary catheters. The device has recently been through Medical Technology Directive with National Institute for Health and Care Excellence.
Hereby, we report the results of a product evaluation of the Uroshield device in a group of patients with neurogenic lower urinary tract dysfunction who rely on an indwelling catheter to manage their neurogenic bladder and who were identified as having recurrent blockages and infections.
The Uroshield device has a Conformite Europeenne mark for use in the United Kingdom. We gained approval for a product evaluation through the hospital’s Clinical Governance Committee on Surgical Innovations, New Technologies and Treatments.
People with an indwelling catheter for management of neurogenic bladder who experienced recurrent blockages or infections were identified from the records of the complex catheter change clinic. All patients had tried alternative techniques for preventing blockages including increased frequency of change, use of Farco-fill® Aqua-Glycerol solution (FarcoPharm GmbH, Germany, no longer licensed in United Kingdom) in catheter balloon, regular washouts including using saline or Suby G (3.23% citric acid, Urotainer® Twin Suby G, B Braun Medical Limited, South Yorkshire, United Kingdom). After gaining informed consent, patients were entered into the Product Evaluation for a period of 12 weeks. They were given information about the device and full training in its use.
Baseline questionnaires regarding number of blockages and infections, quality of life, sleep quality, and catheter satisfaction were completed. Questionnaires were repeated at 4 weeks, 8 weeks and 12 weeks. Patient comments regarding the use of the device were also collected during and at the end of the trial period. Data were analyzed descriptively. Where appropriate, paired non-parametric comparisons were performed using the Wilcoxon signed-rank test.
A total of 10 patients were recruited into the product evaluation. 7 female and 3 male patients were included with an average age of 58 (range 30 years to 70 years). Full demographic details are shown in Table 1. All patients were recruited into the trial due to recurrent catheter blockages rather than recurrent infections. 4 patients did not complete the 12-week trial period. The reasons for withdrawal or non-compliance with the trial protocol encompassed bladder spasms (1 patient), charging and battery life not compatible with lifestyle (2 patients) and exacerbated skin issues (1 patient).
| Patient number | Sex | Age | Etiology | Catheter | Reason |
| 1 | Female | 55 | SCI | SPC | Blockages |
| 2 | Female | 69 | MS | SPC | Blockages |
| 3 | Male | 60 | SCI | SPC | Blockages |
| 4 | Female | 51 | Spina bifida | SPC | Blockages |
| 5 | Female | 64 | SCI | SPC | Blockages |
| 6 | Female | 60 | SCI | SPC | Blockages |
| 7 | Male | 29 | SCI | SPC | Blockages |
| 8 | Female | 62 | SCI | SPC | Blockages |
| 9 | Female | 67 | SCI | SPC | Blockages |
| 10 | Male | 58 | SCI | SPC | Blockages |
| Mean | 57.5 | ||||
| Sex | 70% female | ||||
| 30% male | |||||
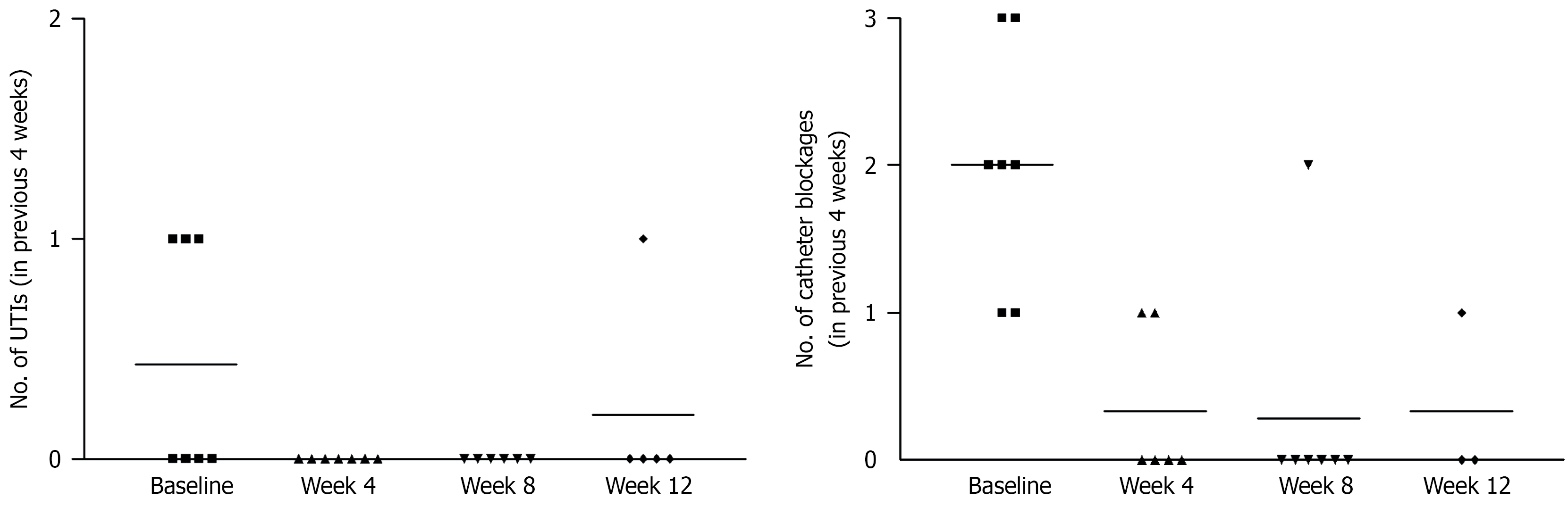
A reduction in catheter blockages was observed, decreasing from an average of two episodes per four-week period at baseline to fewer than one episode during follow-up (Figure 1). From baseline to four weeks, mean catheter satisfaction increased by 4.6 points on a ten-point scale (Tables 2 and 3). Modest improvements were also noted in catheter-related pain and sleep quality (Tables 4 and 5).
| Patient number | Baseline | Week 1 | Week 4 | Week 8 | Week 12 |
| 1 | 2 | 1 | 0 | 0 | |
| 2 | 1 | 0 | 0 | ||
| 3 | |||||
| 4 | 3 | 1 | 0 | 2 | 0 |
| 5 | 2 | 0 | 1 | 0 | 1 |
| 6 | 3 | 0 | 0 | ||
| 7 | |||||
| 8 | 2 | 2 | 0 | 0 | 0 |
| 9 | 1 | - | 0 | 0 | |
| 10 | 4 | 10 |
| Patient number | Baseline | Week 4 | Week 8 | Week 12 |
| 1 | 1 | 10 | 10 | 10 |
| 2 | 7 | 9 | 9 | 3 |
| 3 | 6 | 10 | 9 | |
| 4 | 4 | 6 | ||
| 5 | 4 | 9 | ||
| 6 | 4 | |||
| 7 | 5 | |||
| 8 | 2 | 9 | 10 | 10 |
| 9 | 5 | 8 | 10 | |
| 10 | 7 | 8 |
| Patient number | Baseline | Week 4 | Week 8 | Week 12 |
| 1 | 1 | 7 | 9 | 9 |
| 2 | 10 | 10 | 9 | 3 |
| 3 | 8 | 8 | 5 | |
| 4 | 8 | 1 | ||
| 5 | 1 | 5 | ||
| 6 | 3 | |||
| 7 | 6 | |||
| 8 | 4 | 5 | 5 | 9 |
| 9 | 2 | 8 | 9 | |
| 10 | 5 | 5 |
| Patient number | Baseline | Week 4 | Week 8 | Week 12 |
| 1 | 1 | 7 | 9 | 9 |
| 2 | 10 | 10 | 9 | 3 |
| 3 | 8 | 8 | 5 | |
| 4 | 8 | 1 | ||
| 5 | 1 | 5 | ||
| 6 | 3 | |||
| 7 | 6 | |||
| 8 | 4 | 5 | 5 | 9 |
| 9 | 2 | 8 | 9 | |
| 10 | 10 | 5 |
Four participants discontinued device use due to bladder spasms, skin irritation, or practical limitations related to device charging. Participants who completed the evaluation reported high levels of satisfaction and expressed a desire to continue using the device beyond the evaluation period.
The gold standard for bladder emptying for patients with a neurogenic bladder, is clean intermittent self-catheterization[7,8]. However, this is not feasible in all cases as some patients present with limited hand function, very small capacity bladder or are not fit/keen for any major reconstruction surgery. In such cases, an indwelling catheter (either supra-pubic or urethral) provides a safe and convenient method for bladder emptying[9-11]. Catheter-associated urinary tract infections and catheter blockages can pose significant challenges in certain patient populations, particularly in individuals with neurogenic bladders following spinal cord injury. This susceptibility may be exacerbated by alterations in metabolic processes associated with spinal cord injury, which can affect urinary function and increase the risk of complications[12,13]. Catheter blockages can result in urethral bypassing, and in some cases even life-threatening conditions like auto
SAW is not new to literature in terms of preventing biofilm formation and encrustation. A study conducted by Kopel et al[16], showed that SAW successfully eliminated over 85% of biofilm-residing bacteria when used in conjunction with an antibiotic against three clinically relevant species: Escherichia coli, Staphylococcus epidermidis, and Pseudomonas aeruginosa. Another study, conducted in 2013 examined the impact of SAW on human neutrophils’ ability to eliminate Staphylococcus epidermidis bacteria in both planktonic and biofilm forms. SAW at specific values, significantly boosted neutrophil activity against Staphylococcus epidermidis by enhancing chemotaxis in response to chemo-attractants. The authors suggested that CD18 may act as a mechanoreceptor for SAW, improving neutrophil chemotaxis and bacterial elimination[17].
Among indwelling medical devices, acoustic-wave-activated Foley catheters were studied in 2006. They featured surfaces that vibrate with both longitudinal and transverse dispersion vectors uniformly encompassing the catheter. The acoustic waves produced at the surface repel bacteria and disrupt the adhesion of planktonic microorganisms to solid surfaces, which is crucial in the early stages of microbial biofilm formation[18]. In similar fashion, the Uroshield device which is attached to an indwelling catheter delivers low frequency sound (acoustic) waves which prevent the build-up of biofilms on the catheter surface which may reduce infections and encrustation.
We performed a product evaluation of the device in a group of people who used an indwelling catheter to manage their neurogenic bladder and who experienced recurrent catheter blockages. Use of the device was associated with fewer reported episodes of catheter blockage and fewer unplanned catheter-related interventions during the evaluation period. In addition, there was an increase in catheter satisfaction, and quality of sleep, and a decrease in catheter associated pain. Although some patients did not complete the trial due to practical issues with the use of the device, many of the patients wished to continue with its use at the end of the trial period.
In our study, four participants discontinued device use due to bladder spasms, skin irritation, or practical limitations related to charging and device wearability. While these events cannot be conclusively attributed to the device, they highlight important usability considerations that should be addressed in future studies.
This study has some limitations that we acknowledge. First of all, only a small number of patients were included in the population and further recruitment is needed to enhance the power of the study. Apart from that, this was a single-center study as all patients were recruited from our specialized Spinal Cord Injury Centre. More extensive and multi-center studies are required for safe conclusions in the future. Last but not least, this was not a blind study as there was not a second leg (placebo) population for comparison.
This observational product evaluation suggests that an acoustic wave device may be feasible and acceptable for selected patients with neurogenic bladder and recurrent suprapubic catheter blockage. Given the methodological limitations, including small sample size and lack of a control group, conclusions regarding effectiveness cannot be drawn. Further controlled and multicenter studies are required.
The authors would like to thank the clinical and nursing staff of the London Spinal Cord Injury Centre for their support during patient identification and routine clinical care associated with this product evaluation.
| 1. | European Association of Urology. EAU Guidelines on Neuro-Urology. The Netherlands (Arnhem): EAU Guidelines Office, 2022. |
| 2. | Parkin J, Keeley FX. Indwelling catheter-associated urinary tract infections. Br J Community Nurs. 2003;8:166-167, 170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 3. | Getliffe K. Managing recurrent urinary catheter encrustation. Br J Community Nurs. 2002;7:574, 576, 578-580. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 16] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 4. | Tenke P, Köves B, Johansen TE. An update on prevention and treatment of catheter-associated urinary tract infections. Curr Opin Infect Dis. 2014;27:102-107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 54] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 5. | Zarb P, Coignard B, Griskeviciene J, Muller A, Vankerckhoven V, Weist K, Goossens M, Vaerenberg S, Hopkins S, Catry B, Monnet D, Goossens H, Suetens C; National Contact Points for the ECDC pilot point prevalence survey; Hospital Contact Points for the ECDC pilot point prevalence survey. The European Centre for Disease Prevention and Control (ECDC) pilot point prevalence survey of healthcare-associated infections and antimicrobial use. Euro Surveill. 2012;17:20316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 283] [Cited by in RCA: 334] [Article Influence: 23.9] [Reference Citation Analysis (0)] |
| 6. | Markowitz S, Rosenblum J, Goldstein M, Gadagkar HP, Litman L. The Effect of Surface Acoustic Waves on Bacterial Load and Preventing Catheter- associated Urinary Tract Infections (CAUTI) in Long Term Indwelling Catheters. Med Surg Urol. 2018;7:4. [DOI] [Full Text] |
| 7. | Guttmann L, Frankel H. The value of intermittent catheterisation in the early management of traumatic paraplegia and tetraplegia. Paraplegia. 1966;4:63-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 230] [Cited by in RCA: 208] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 8. | Lapides J, Diokno AC, Silber SJ, Lowe BS. Clean, intermittent self-catheterization in the treatment of urinary tract disease. J Urol. 1972;107:458-461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 886] [Cited by in RCA: 768] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 9. | Weld KJ, Dmochowski RR. Effect of bladder management on urological complications in spinal cord injured patients. J Urol. 2000;163:768-772. [PubMed] |
| 10. | Mitsui T, Minami K, Furuno T, Morita H, Koyanagi T. Is suprapubic cystostomy an optimal urinary management in high quadriplegics?. A comparative study of suprapubic cystostomy and clean intermittent catheterization. Eur Urol. 2000;38:434-438. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 71] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 11. | Larsen LD, Chamberlin DA, Khonsari F, Ahlering TE. Retrospective analysis of urologic complications in male patients with spinal cord injury managed with and without indwelling urinary catheters. Urology. 1997;50:418-422. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 76] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 12. | Lamid S, El Ghatit AZ, Melvin JL. Relationship of hypercalciuria to diet and bladder stone formation in spinal cord injury patients. Am J Phys Med. 1984;63:182-187. [PubMed] |
| 13. | Conley SB, Shackelford GD, Robson AM. Severe immobilization hypercalcemia, renal insufficiency, and calcification. Pediatrics. 1979;63:142-145. [PubMed] |
| 14. | Burr RG, Nuseibeh I. Diuresis and urinary saturation in patients with neurogenic bladder. Invest Urol. 1981;18:331-333. [PubMed] |
| 15. | Lavelle RS, Coskun B, Bacsu CD, Gliga LA, Christie AL, Lemack GE. Quality of life after suprapubic catheter placement in patients with neurogenic bladder conditions. Neurourol Urodyn. 2016;35:831-835. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 16. | Kopel M, Degtyar E, Banin E. Surface acoustic waves increase the susceptibility of Pseudomonas aeruginosa biofilms to antibiotic treatment. Biofouling. 2011;27:701-710. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 17. | Loike JD, Plitt A, Kothari K, Zumeris J, Budhu S, Kavalus K, Ray Y, Jacob H. Surface acoustic waves enhance neutrophil killing of bacteria. PLoS One. 2013;8:e68334. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 18. | Hazan Z, Zumeris J, Jacob H, Raskin H, Kratysh G, Vishnia M, Dror N, Barliya T, Mandel M, Lavie G. Effective prevention of microbial biofilm formation on medical devices by low-energy surface acoustic waves. Antimicrob Agents Chemother. 2006;50:4144-4152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 77] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/