Published online Mar 6, 2026. doi: 10.12998/wjcc.v14.i7.117943
Revised: January 30, 2026
Accepted: February 12, 2026
Published online: March 6, 2026
Processing time: 76 Days and 14.3 Hours
Age-related macular degeneration (AMD) is a major cause of central vision loss among older adults and is increasingly recognized as a condition with significant psychological consequences.
To synthesize current evidence on the prevalence, correlates, and management of depression and anxiety among individuals with AMD.
A comprehensive literature search of PubMed, Scopus, and Web of Science (2000-2025) identified 33 relevant systematic reviews, observational studies, and interventional trials. Extracted data included study design, popu
Prevalence of depression in AMD ranges from 15.7% to 44%, with pooled estimates around 22%-25%, while anxiety affects 9.6%-30.1% of patients. Risk factors include severity of visual loss, type of AMD (wet or dry), female gender, and limited social support. These comorbidities exacerbate disability, impair quality of life, and reduce adherence to ophthalmologic treatment. Psychosocial interventions - particularly cognitive-behavioral therapy, self-management programs, and behavioral activation - are effective in reducing depression and improving coping. Pharmacologic options such as selective serotonin reuptake inhibitors and serotonin-norepinephrine reuptake inhibitors may also be beneficial, though evidence remains limited.
An integrated, multidisciplinary approach combining psychological, pharmacological, and rehabilitative strategies is recommended. Future research should focus on anxiety-specific interventions and long-term outcomes to optimize mental health and visual rehabilitation in AMD patients.
Core Tip: Depression and anxiety are common but underrecognized in patients with age-related macular degeneration. Routine mental health screening, timely psychosocial interventions, and integration of psychiatric care within ophthalmology can improve overall patient outcomes and quality of life. This systematic review also synthesizes current evidence on the prevalence, correlates, and management of depression and anxiety among individuals with age-related macular degeneration.
- Citation: Aggarwal S, Morya AK, Nishant P, Sinha S, Kaur R, Sinha RK, Morya R. Psychiatric disorders like depression and anxiety in age-related macular degeneration. World J Clin Cases 2026; 14(7): 117943
- URL: https://www.wjgnet.com/2307-8960/full/v14/i7/117943.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i7.117943
Age-related macular degeneration (AMD) is a leading cause of irreversible central vision loss in older adults worldwide[1]. It not only impairs visual function but also profoundly affects psychological well-being. The global burden of AMD-related vision impairment nearly doubled between 1990 and 2021, with corresponding increases in disability-adjusted life years[2]. Advanced AMD, encompassing neovascular AMD and geographic atrophy, leads to significant disability, dependence, and emotional distress. Studies report that AMD increases the risk of depression by approximately 15%, with up to 20% of patients exhibiting clinically relevant depressive symptoms[3-6]. Despite advances in ophthalmologic treatment, the mental health consequences of AMD remain underappreciated in clinical settings[7-10]. Major depressive symptoms are often compounded by factors such as high treatment costs, frequent hospital visits, reduced social participation, and dependence on caregivers[6,11,12].
Furthermore, variability in disease prognosis and treatment outcomes heightens psychological vulnerability, while anxiety can exacerbate perceived disability and promote functional decline[5,10,13]. Although literature increasingly recognizes the bidirectional relationship between visual impairment and mental health, psychological morbidity in AMD remains underdiagnosed and undertreated[5,9,10,14]. This review aims to consolidate existing evidence on the prevalence and impact of depression and anxiety in patients with advanced AMD, explore contributing risk factors, and evaluate current therapeutic strategies for addressing mental health needs in this vulnerable population.
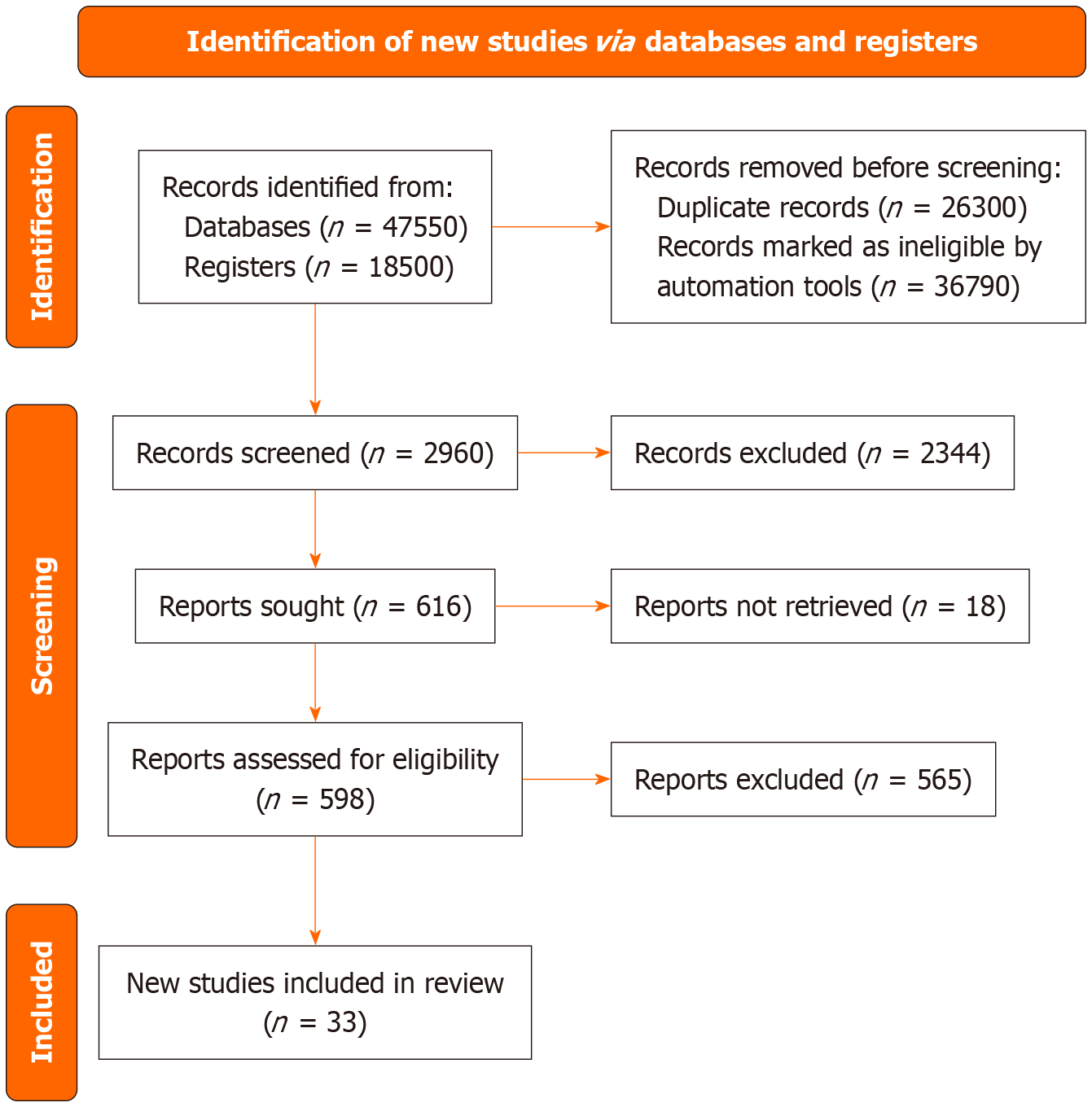
A comprehensive literature search was conducted using PubMed, Scopus, Web of Science, and Google Scholar between January 2000 and September 2025. Keywords included “age-related macular degeneration”, “depression”, “anxiety”, “mental health”, “psychological impact”, “retinal disorders”, “visual impairment”, and related thematic terms. Studies were included if they were published in English, involved adults aged ≥ 50 years, and evaluated depression or anxiety among patients diagnosed with AMD[4,9]. Thirty-three eligible publications were identified, comprising observational studies, randomized clinical trials, and systematic reviews (Figure 1). Case reports, commentaries, and studies lacking validated psychological assessment instruments were excluded[9].
Two reviewers independently screened titles and abstracts; disagreements were resolved in consultation with a third reviewer. Extracted data included study design, population characteristics, assessment tools, prevalence estimates, associated risk factors, and details of psychosocial or pharmacological interventions[4,10]. Quality appraisal was undertaken using standardized checklists appropriate to each design, such as the Newcastle-Ottawa Scale for observational studies[10]. Descriptive synthesis was performed because heterogeneity in study design and outcome measurement precluded meta-analysis. The synthesis followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Figure 1).
Depression and anxiety, diagnosed according to various validated psychological scoring and assessment systems, are significantly more prevalent in AMD populations than in age-matched controls (Table 1). Prevalence of depression in AMD ranges from 15.7% to 44%, with pooled estimates around 22%-25% (Table 2). Rates are generally higher in patients with wet AMD compared to those with dry AMD, reflecting the greater visual loss, functional dependency, and treatment burden associated[12,15,16]. Systematic reviews and meta-analyses confirm that depression rates in AMD significantly exceed those observed in the general older population[4,9,10,12]. In one review, depression prevalence reached 56% among those with wet AMD, 27% among those with dry AMD, and 20% among control groups[12]. The prevalence of anxiety in AMD is less consistent but remains noteworthy, ranging from 9.6% to 30.1%, with pooled estimates between 14% and 18% (Table 2). Although less frequently studied than depression, several studies suggest that anxiety often coexists with depressive symptoms, forming a combined psychological burden that exacerbates functional limitations and reduces social engagement[5,7,9,11,17,18].
| Diagnostic basis |
| Hospital Anxiety and Depression Scale |
| Centre for Epidemiological Studies - Depression scale |
| Structured Clinical Interview for DSM-IV |
| Geriatric Depression Scale |
| Goldberg Anxiety and Depression Scale |
| International Classification of Diseases |
| Patient Health Questionnaire |
| Condition | Prevalence range | Pooled estimate | Ref. |
| Depression | 15.7%-44% | 22%-25% | Tran et al[4], 2025; Cimarolli et al[5], 2015; Senra et al[6], 2019; Dawson et al[9], 2014; Olson et al[13], 2019; Fernández-Vigo et al[16], 2021; Lee et al[17], 2021; Casten and Rovner[19], 2008 |
| Anxiety | 9.6%-30.1% | 14%-18% | Tran et al[4], 2025; Cimarolli et al[5], 2015; Senra et al[6], 2019; Olson et al[13], 2019; Fernández-Vigo et al[16], 2021; Lee et al[17], 2021; Dawson et al[19], 2014 |
Thus, psychological morbidity is a substantial yet under-recognized component of AMD, particularly in advanced stages where vision loss is most disabling[6,7,9-12]. These comorbidities increase disability, impair quality of life, and complicate disease management, emphasizing the importance of incorporating routine psychological screening and multidisciplinary care into AMD management[6,7,10,12].
The severity of vision loss is one of the most consistent predictors of depression in AMD[5,8,9,12,16]. Patients with advanced or bilateral disease often experience greater dependence for daily tasks, loss of autonomy, and diminished self-esteem, all of which contribute to depressive symptoms. Although anxiety is less directly associated with the degree of visual impairment, individuals with more severe vision loss often report heightened emotional distress and uncertainty regarding disease progression[5,8,11,12]. Several studies have demonstrated that wet AMD is linked to higher rates of depression than dry AMD, likely due to its more aggressive course and the need for ongoing intravitreal therapy[6,9,12,16]. Sociodemographic variables play an important role as well. Female gender and advanced age have been independently associated with a higher likelihood of depressive or anxiety disorders in AMD[17,18]. These associations may reflect gender differences in emotional coping strategies and the cumulative psychological burden of age-related decline.
Perceived social support is another key determinant. Evidence suggests that low social support increases the risk of depression and anxiety independently of visual acuity[14]. The loss of social roles and limited engagement in meaningful activities may amplify loneliness and helplessness. Functional disability and treatment burden - including frequent hospital visits, uncertainty about treatment outcomes, and the financial costs of anti-vascular endothelial growth factor (VEGF) injections - are additional contributors to psychological distress[6,7,10,12,19]. These factors often interact to create a cycle of dependence, isolation, and emotional exhaustion that worsens the overall quality of life in AMD.
The coexistence of depression and anxiety in patients with AMD has far-reaching implications for quality of life, treatment outcomes, and overall disease management[6,7,9,10,12,15,16,18,19].
Quality of life: Depression and anxiety are strongly associated with reduced vision-related quality of life and heightened disability. Difficulty in performing routine activities such as reading, driving, and recognizing faces contributes to frustration, social withdrawal, and feelings of dependency. The psychological comorbidities increase the risk of falls, exacerbate functional impairment, and elevate the likelihood of institutionalization among older adults[9,10,15,16,18]. The compounding effect of emotional distress and sensory loss also magnifies the perception of visual handicap and limits participation in previously valued social and recreational activities[7,12,16,18]. Concomitant effects of alcohol consumption, dietary patterns, and other environmental factors, such as active or passive smoking and drug abuse, also affect the quality of life and exacerbate psychological duress as well as worsen retinal health; however, the effect of these factors coexisting with AMD has not been studied well.
Treatment adherence: Emotional disorders can negatively affect adherence to anti-VEGF regimens and satisfaction with ophthalmic care[6,9,12,16]. Depressed patients are less likely to comply with follow-up schedules, adhere to self-management programs, or pursue vision rehabilitation services[6,9,19]. Reduced motivation and hopelessness often contribute to premature treatment discontinuation and poorer visual outcomes.
Comorbidity: Depression and anxiety in AMD patients often coexist with other chronic conditions, including cardiovascular disease, diabetes, and cognitive impairment[7,9,10,17]. The presence of these psychiatric disorders further increases healthcare utilization, lengthens recovery times, and impairs coping capacity[9,10,17]. The heightened risk of falls may also be associated with concurrent systemic comorbidities like neurological and cardiovascular disease, as well as the use of medication, thereby developing a vicious circle of psychological duress and increased hospitalization, amplifying the pressure on healthcare systems to cater to patients with these unique needs. Ultimately, these conse
Psychosocial interventions play a central role in mitigating the emotional and functional burden associated with AMD[6,9,10,19-22]. Structured approaches such as cognitive-behavioral therapy (CBT), behavioral activation, and group self-management programs have demonstrated substantial efficacy in alleviating depressive symptoms and, to a lesser extent, anxiety[10,19,21,22].
CBT: CBT-based interventions aim to modify maladaptive beliefs and encourage adaptive coping behaviors. Studies show that CBT reduces depressive symptoms by enhancing patients’ sense of control and self-efficacy despite vision loss[10,19,22]. A stepped-care model incorporating self-help, problem-solving, and guided behavioral activation significantly lowered the incidence of depression and improved adjustment to vision impairment[21,22].
Behavioral activation and self-management: Behavioral activation, which encourages participation in meaningful daily activities, has been effective when combined with low vision rehabilitation[21,22]. Randomized controlled trials demonstrate that group self-management programs can both treat and prevent depression in AMD patients[21,22]. These programs typically include educational sessions, peer interaction, and coping-skills training to improve autonomy and emotional well-being[19,21].
Emotion-focused and self-help approaches: Non-randomized studies suggest that emotion-focused self-help interventions, including problem-solving therapy, can reduce depressive symptoms and enhance self-acceptance[10,19]. These approaches are valuable for individuals unable to access structured psychotherapy due to visual or logistical constraints.
Mindfulness-based stress reduction: Mindfulness interventions incorporating meditation and relaxation techniques improve resilience and emotional regulation. Mindfulness-based stress reduction, initially developed by Kabat-Zinn, has demonstrated benefits in anxiety and depression across various chronic illnesses[23,24]. In AMD, mindfulness may help patients reframe their emotional responses to visual decline and reduce anxiety related to disease uncertainty[24].
Interdisciplinary integration: The most successful psychosocial interventions combine psychological counseling with vision rehabilitation and social support frameworks, offering holistic management of both visual and emotional impairment[6,10,19-22]. However, despite robust evidence for depression reduction, anxiety outcomes remain underexplored, underscoring the need for targeted studies addressing anxiety in AMD[9,10,19,20].
Pharmacological management can complement psychosocial and rehabilitative interventions[6,7,9,10,19,25-28]. Although evidence remains limited, antidepressant therapy - particularly with selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) - has demonstrated potential benefits for mood stabilization, neuroprotection, and visual outcomes[25,26]. However, the use of SSRIs and SNRIs in the elderly is always a challenge, especially with other chronic medications, which can lead to electrolyte abnormalities and drug-drug interactions. Thus, it is pertinent to discuss in detail the current status of pharmacological therapy for anxiety and depression in AMD.
Antidepressants: Several studies report that the use of SSRIs (e.g., escitalopram) or SNRIs may improve depressive symptoms in patients with AMD, likely through mechanisms involving decreased inflammation, oxidative stress reduction, and upregulation of neurotrophic factors[25-27]. In a randomized clinical trial, escitalopram significantly alleviated depressive symptoms and improved quality of life, with minimal adverse effects[26]. Similarly, a pilot study of sertraline demonstrated safety and possible efficacy in reducing depression in AMD[27]. However, certain antidepressants may also affect ocular physiology. One observational study found that antidepressant use among patients with neovascular AMD was associated with an increased need for anti-VEGF retreatment, potentially due to medication-related upregulation of vascular endothelial growth factor or to the underlying depression itself[28]. Furthermore, isolated case reports have described cystoid macular edema associated with escitalopram use, emphasizing the importance of ophthalmic monitoring in patients receiving antidepressants[29]. Recent epidemiological analyses suggest that antidepressant exposure may be associated with a lower risk of AMD onset or progression, though causality remains uncertain[25]. These findings raise the possibility that certain antidepressants might exert neuroprotective effects within the retina, potentially by modulating oxidative and inflammatory pathways[25].
Anxiolytics: The role of anxiolytic medication in AMD remains less well defined. Benzodiazepines may provide short-term relief from severe anxiety or sleep disturbance, but their use in older adults carries risks of sedation, imbalance, and cognitive impairment[30]. Given the age profile of AMD patients, benzodiazepines should be prescribed cautiously and only for short durations[30]. Taken together, pharmacotherapy may be beneficial for selected patients with clinically significant depression or anxiety, but treatment decisions should consider the potential ocular and systemic risks. Close collaboration between psychiatrists and ophthalmologists is recommended to ensure safe and effective management[6,7,9,26-30].
Vision rehabilitation represents a critical component of AMD management, aimed at improving functional independence and emotional well-being in patients coping with central vision loss[6,9,10,19-22,31-33]. Rehabilitation strategies include the use of optical and electronic low-vision aids, occupational therapy, environmental modification, and training in adaptive behaviors to enhance daily functioning and quality of life[19-22,31,32].
Low-vision aids and assistive technology: Optical devices such as magnifiers, telescopic lenses, and electronic reading systems significantly enhance patients’ ability to perform routine activities, leading to reductions in frustration and depressive symptoms[21,31,32]. Recent advances in digital accessibility tools - including voice-assisted technology, contrast-enhancement software, and smartphone applications - have improved autonomy in such cases[32,33].
Occupational therapy: This includes activity modification, task adaptation, and environmental safety, enabling individuals to maintain independence despite central vision loss[21,31]. Incorporating psychosocial support into rehabilitation further strengthens outcomes by addressing emotional distress and enhancing motivation for therapy adherence[10,19,22,31].
Behavioral activation: Behavioral activation has shown measurable benefits for depression. Randomized controlled trials demonstrated that combined behavioral activation and low-vision rehabilitation can halve the incidence of depression compared with supportive therapy alone[21,22,31]. These effects are mediated through improved activity engagement, mastery experiences, and social re-engagement[19,21,22].
Holistic outcomes: Vision rehabilitation also improves psychological resilience and life satisfaction[6,9,10,19,31-33]. This evidence supports the inclusion of psychological assessment and therapy as integral elements within low-vision rehabilitation services, ideally delivered through a multidisciplinary team involving ophthalmologists, psychologists, and occupational therapists[6,9,10,19,21,22,31-33].
Social and community support systems are fundamental to maintaining psychological resilience and functional independence in patients with AMD[6,9,10,12,14,18,19,31,34]. Social support mitigates loneliness, enhances coping capacity, and provides emotional stability, thereby reducing both depression and anxiety[12,14,18,19].
Peer and support groups: Peer-support groups foster a sense of belonging and mutual understanding, significantly decreasing perceived isolation and helplessness[9,12,14,18]. Group interventions also improve coping strategies by facilitating the exchange of adaptive experiences and problem-solving methods among participants[19,31]. Regular peer interaction helps in coping with vision loss and reinforces adherence to rehabilitation plans[9,10,12,31].
Family and caregiver involvement: Family support is a critical protective factor against depression and anxiety[14,18,19,34]. Educating caregivers about the nature and psychological impact of AMD improves empathy, communication, and emotional support within the household[14,19,34]. In turn, this enhances treatment adherence and patient well-being. However, caregivers themselves frequently experience burden, fatigue, and psychological distress, especially when the patient’s dependency is high or disease progression is unpredictable[14,34].
Community and social services: Community-based vision rehabilitation programs and volunteer-driven initiatives offer opportunities for social reintegration, reducing isolation and dependence[9,19,31,34]. Partnerships between ophthalmic clinics, mental health professionals, and social welfare organizations create a continuum of care, addressing both visual impairment and its emotional sequelae[6,9,10,19,31,34]. However, the incorporation of structured peer and community interventions into standard ophthalmologic care remains limited. Addressing this gap could substantially improve both psychological outcomes and treatment adherence in AMD populations[9,10,14,18,19,31,34].
Lifestyle modifications play an important adjunctive role[6,9,10,19,32,35]. Physical activity, nutrition, and overall wellness behaviors influence both visual and psychological outcomes[32,35].
Physical activity: Regular physical exercise has been associated with lower levels of depression and anxiety in older adults and may also slow AMD progression through improved vascular and metabolic health[9,10,32,35]. Studies indicate that moderate-intensity aerobic exercise enhances cerebral and retinal perfusion, improves mood-regulating neurotransmission, and supports functional independence[32,35]. Tailored exercise programs adapted to vision-impaired individuals can improve mobility, confidence, and social interaction while reducing fear of falls[9,19,32].
Nutrition: Nutritional interventions with antioxidant-rich foods - such as fruits, leafy greens, and omega-3 fatty acids - contribute to both ocular and psychological health[6,9,19,32,35]. Dietary supplementation with lutein, zeaxanthin, and vitamins C and E has been linked with a reduced risk of AMD progression and may indirectly enhance mood by improving overall health perception[35]. Adequate vitamin D intake has also been associated with lower depressive symptom severity in older adults, although this relationship has not been specifically validated in AMD populations[32,35].
Sleep and stress management: Good sleep hygiene and stress reduction techniques, such as meditation, breathing exercises, and mindfulness-based practices, may further alleviate anxiety and emotional fatigue, improving coping and reducing perceived stress in visually impaired populations[9,24,32,35].
Integrated wellness approaches: A comprehensive wellness model combining physical activity, balanced nutrition, stress management, and social engagement provides the best outcomes for mental and visual health[6,9,10,19,24,32,35]. Lifestyle optimization promotes psychological well-being and complements the effects of medical and psychosocial interventions, leading to improved overall functioning and quality of life.
The available literature consistently indicates that depression and anxiety are highly prevalent yet underrecognized comorbidities among patients with AMD[4-7,9-12,14-16,18,19]. Prevalence estimates for depression range between 15% and 44%, and anxiety occurs in approximately 10%-30% of patients[4,9-12,16,18]. These rates exceed those in the general elderly population, suggesting that the psychological burden of AMD is comparable to that of other chronic disabling conditions such as stroke or Parkinson’s disease[6,9,12,15,17].
Emotional morbidity in AMD is multifactorial. Severity of vision loss, functional dependence, female gender, and limited social support are independent predictors of depression and anxiety[5,6,9,12,14,16-18]. Need for recurrent visits, intravitreal therapy, uncertainty about prognosis, and the financial burden of long-term care further exacerbate distress[6,7,10,12,19]. These stressors often lead to maladaptive coping behaviors and social withdrawal, reinforcing the cycle of depression and anxiety[9,14,18,19].
Psychological distress significantly affects treatment adherence, visual rehabilitation outcomes, and quality of life[6,9,10,12,16,18,19]. Depression is associated with poorer compliance to anti-VEGF therapy, reduced motivation for self-care, and lower engagement in vision rehabilitation[6,9,16,19]. Conversely, psychological interventions - particularly CBT, behavioral activation, and structured self-management programs - demonstrate substantial benefits in reducing depre
Although antidepressants such as SSRIs and SNRIs may offer benefits, their use requires caution due to age-related risks and possible systemic and ocular side effects in the elderly[26-30]. Evidence supporting pharmacotherapy is encouraging but limited, emphasizing the need for integrated psychiatric-systemic-ophthalmologic monitoring[6,9,10,19,26-30].
Vision rehabilitation, including low-vision aids, occupational therapy, and digital assistive technologies, has proven effective in improving autonomy and reducing psychological distress[19,21,22,31-33]. Incorporating psychosocial components such as group support and behavioral activation into these programs doubles their effectiveness in mitigating depression[21,22,31]. Likewise, community engagement, family education, and caregiver support play pivotal roles in maintaining mental health and social functioning[9,10,12,14,18,19,31,35-39].
Regular physical activity, a nutrient-rich diet, and mindfulness-based stress reduction appear to buffer against depression and anxiety while complementing medical and rehabilitative treatments[9,19,24,32,35,38,39].
Future research should focus on anxiety-specific interventions, longitudinal outcomes, and cost-effectiveness analyses of integrated care models[9,10,19,20,24,31]. Large-scale, multicenter studies are warranted to establish evidence-based protocols that combine ophthalmologic, psychiatric, and rehabilitative management. Incorporating validated mental health screening tools into routine ophthalmic assessments could also improve early detection and treatment of depression and anxiety[6,9,10,12,19].
Routine screening for depression and anxiety using validated tools should become standard practice in ophthalmologic clinics[6,9,10,12,19]. Early detection and prompt intervention can prevent deterioration in both mental health and visual functioning, ultimately improving long-term outcomes. By viewing AMD as both a visual and psychological disorder, clinicians can offer more holistic, patient-centered care that enhances recovery, preserves independence, and restores quality of life[9,10,12,19,31,35-39].
Depression and anxiety are common, clinically significant, and often underrecognized comorbidities among patients with AMD[4-7,9-12,14-16,18,19]. These disorders profoundly affect quality of life, treatment adherence, and visual rehabilitation outcomes[6,7,9,10,12,39]. An integrated, multidisciplinary approach - involving ophthalmologists, psychiatrists, psychologists, rehabilitation specialists, and social workers - is essential to address the complex interplay between vision loss and mental health[6,9,10,12,19,21,22,31-34]. CBT, behavioral activation, and structured self-management programs in reducing depression and improving coping[10,19-22,31]. Complementary strategies, including lifestyle optimization, community engagement, and family support, further enhance resilience and emotional well-being[9,14,18,19,24,32,35-39].
My teachers, family, and friends.
| 1. | Mitchell P, Liew G, Gopinath B, Wong TY. Age-related macular degeneration. Lancet. 2018;392:1147-1159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1387] [Cited by in RCA: 1160] [Article Influence: 145.0] [Reference Citation Analysis (0)] |
| 2. | Zhang S, Ren J, Chai R, Yuan S, Hao Y. Global burden of low vision and blindness due to age-related macular degeneration from 1990 to 2021 and projections for 2050. BMC Public Health. 2024;24:3510. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 54] [Article Influence: 27.0] [Reference Citation Analysis (0)] |
| 3. | Hwang S, Kang SW, Kim SJ, Han K, Kim BS, Jung W, Lim DH, Shin DW. Impact of Age-Related Macular Degeneration and Related Visual Disability on the Risk of Depression: A Nationwide Cohort Study. Ophthalmology. 2023;130:615-623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 32] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 4. | Tran E, Maher A, Basilious A, Malvankar-Mehta MS. Prevalence of anxiety and depression symptoms in age-related macular degeneration patients: A systematic review and meta-analysis. Arch Ment Health. 2025;26:14-28. [DOI] [Full Text] |
| 5. | Cimarolli VR, Casten RJ, Rovner BW, Heyl V, Sörensen S, Horowitz A. Anxiety and depression in patients with advanced macular degeneration: current perspectives. Clin Ophthalmol. 2016;10:55-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 45] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 6. | Senra H, Ali Z, Balaskas K, Aslam T. Psychological impact of anti-VEGF treatments for wet macular degeneration-a review. Graefes Arch Clin Exp Ophthalmol. 2016;254:1873-1880. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 68] [Cited by in RCA: 61] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 7. | Kim ES, Kim Y, Yu SY, Kim M. Depression and anxiety in age-related macular degeneration. Patient Prefer Adherence. 2016;10:1853-1854. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 8. | Augustin A, Sahel JA, Bandello F, Dardennes R, Maurel F, Negrini C, Hieke K, Berdeaux G. Anxiety and depression prevalence rates in age-related macular degeneration. Invest Ophthalmol Vis Sci. 2007;48:1498-1503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 96] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 9. | Dawson SR, Mallen CD, Gouldstone MB, Yarham R, Mansell G. The prevalence of anxiety and depression in people with age-related macular degeneration: a systematic review of observational study data. BMC Ophthalmol. 2014;14:78. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 91] [Cited by in RCA: 78] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 10. | Senra H, Macedo AF, Nunes N, Balaskas K, Aslam T, Costa E. Psychological and Psychosocial Interventions for Depression and Anxiety in Patients With Age-Related Macular Degeneration: A Systematic Review. Am J Geriatr Psychiatry. 2019;27:755-773. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 11. | Rezapour J, Schuster AK, Nickels S, Korb CA, Elbaz H, Peto T, Michal M, Münzel T, Wild PS, König J, Lackner K, Schulz A, Pfeiffer N, Beutel ME. Prevalence and new onset of depression and anxiety among participants with AMD in a European cohort. Sci Rep. 2020;10:4816. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 12. | Gouliopoulos N, Bouratzis N, Kympouropoulos S, Datseris I, Georgalas I, Theodossiadis P, Rouvas A. Mental Health Consequences of Age-Related Macular Degeneration: Exploring Depression Prevalence and Severity in Wet and Dry Forms. Clin Gerontol. 2026;49:168-176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 13. | Olson D, Zhang X, Ward MF, Le P, Ismail OM, Poole ZB, Donaldson JC, Van Buren E, Lin F, Ulrich JN, Davis RM. Rates of Comorbid Anxiety and Depression in Patients With Age-Related Macular Degeneration. J Vitreoretin Dis. 2019;3:211-214. [DOI] [Full Text] |
| 14. | Hernández-Moreno L, Senra H, Moreno N, Macedo AF. Is perceived social support more important than visual acuity for clinical depression and anxiety in patients with age-related macular degeneration and diabetic retinopathy? Clin Rehabil. 2021;35:1341-1347. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 15. | Berman K, Brodaty H. Psychosocial effects of age-related macular degeneration. Am J Ophthalmol. 2007;143:545. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 16. | Fernández-Vigo JI, Burgos-Blasco B, Calvo-González C, Escobar-Moreno MJ, Shi H, Jiménez-Santos M, Valverde-Megías A, Reche-Frutos J, López-Guajardo L, Donate-López J. Assessment of vision-related quality of life and depression and anxiety rates in patients with neovascular age-related macular degeneration. Arch Soc Esp Oftalmol (Engl Ed). 2021;96:470-475. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 17. | Lee CY, Chen HC, Huang JY, Lai CC, Lin HY, Yang SF, Wu WC. Increased probability of mood disorders after age-related macular degeneration: a population-based cohort study. Sci Rep. 2022;12:15222. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 18. | Papalamprou M, Georgakopoulos CD, Pharmakakis N. Quality of life: Investigating the impact of two major mental disorders, anxiety and depression, in patients with age-related macular degeneration. Br J Vis Impair. 2022;40:301-315. [DOI] [Full Text] |
| 19. | Casten R, Rovner B. Depression in Age-Related Macular Degeneration. J Vis Impair Blind. 2008;102:591-599. [DOI] [Full Text] |
| 20. | Cuijpers P, Miguel C, Ciharova M, Quero S, Plessen CY, Ebert D, Harrer M, van Straten A, Karyotaki E. Psychological treatment of depression with other comorbid mental disorders: systematic review and meta-analysis. Cogn Behav Ther. 2023;52:246-268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 21. | Brody BL, Roch-Levecq AC, Thomas RG, Kaplan RM, Brown SI. Self-management of age-related macular degeneration at the 6-month follow-up: a randomized controlled trial. Arch Ophthalmol. 2005;123:46-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 47] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 22. | Rovner BW, Casten RJ, Hegel MT, Massof RW, Leiby BE, Ho AC, Tasman WS. Low vision depression prevention trial in age-related macular degeneration: a randomized clinical trial. Ophthalmology. 2014;121:2204-2211. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 106] [Cited by in RCA: 89] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 23. | Bradshaw J, Siddiqui N, Greenfield D, Sharma A. Kindness, Listening, and Connection: Patient and Clinician Key Requirements for Emotional Support in Chronic and Complex Care. J Patient Exp. 2022;9:23743735221092627. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 30] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 24. | Hofmann SG, Gómez AF. Mindfulness-Based Interventions for Anxiety and Depression. Psychiatr Clin North Am. 2017;40:739-749. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 560] [Cited by in RCA: 368] [Article Influence: 40.9] [Reference Citation Analysis (4)] |
| 25. | Mahmoudzadeh R, Zaichik M, Farhani K, Salabati M, Randolph J. Antidepressant Use and Incidence and Progression of Age-Related Macular Degeneration in a National United States Database. Am J Ophthalmol. 2025;280:458-471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 26. | Brody BL, Field LC, Roch-Levecq AC, Moutier CY, Edland SD, Brown SI. Treatment of depression associated with age-related macular degeneration: a double-blind, randomized, controlled study. Ann Clin Psychiatry. 2011;23:277-284. [PubMed] |
| 27. | Rovner BW, Pasternak R, Casten R. A pilot study of sertraline for depression in age-related macular degeneration. Am J Geriatr Psychiatry. 2006;14:718-719. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 28. | Mantel I, Zola M, Mir O, Gaillard R, Behar-Cohen F. Antidepressant medication and ocular factors in association with the need for anti-VEGF retreatment in neovascular age-related macular degeneration. Br J Ophthalmol. 2019;103:811-815. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 29. | Civelekler M, Güven YZ, Akay F. Escitalopram-related bilateral cystoid macular edema. Indian J Ophthalmol Case Rep. 2024;4:143-145. [DOI] [Full Text] |
| 30. | Montes-Castrejon A, Moncayo-Samperio LG, Flores-Ramos M. Benzodiazepine Consumption, Functionality, Cognition, and Somnolence in Older Adults at a Tertiary Care Psychiatric Hospital in Mexico City. Cureus. 2024;16:e53252. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 31. | Erginturk Acar D, Batioglu F, Idil A, Sahli E, Goksuluk D. Rehabilitation Methods for Patients with Geographic Atrophy due to Age-Related Macular Degeneration and Effects of Rehabilitation on Quality of Life. J Ophthalmol. 2023;2023:3389750. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 32. | Gopalakrishnan S, Velu S, Raman R. Low-vision intervention in individuals with age-related macular degeneration. Indian J Ophthalmol. 2020;68:886-889. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 7] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 33. | Vukicevic M, Heraghty J, Cummins R, Gopinath B, Mitchell P. Caregiver perceptions about the impact of caring for patients with wet age-related macular degeneration. Eye (Lond). 2016;30:413-421. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 30] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 34. | McGuinness MB, Le J, Mitchell P, Gopinath B, Cerin E, Saksens NTM, Schick T, Hoyng CB, Guymer RH, Finger RP. Physical Activity and Age-related Macular Degeneration: A Systematic Literature Review and Meta-analysis. Am J Ophthalmol. 2017;180:29-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 93] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 35. | Lindgren EC, Källstrand J, Alftberg Å, Johansson P, Kristén L, Håman L, Ivarsson A, Carlsson IM. Empowerment-Based Physical Activity Intervention for People with Advanced Dry Age-Related Macular Degeneration: Mixed-Methods Protocol. Int J Environ Res Public Health. 2022;20:643. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 36. | Tolman J, Hill RD, Kleinschmidt JJ, Gregg CH. Psychosocial adaptation to visual impairment and its relationship to depressive affect in older adults with age-related macular degeneration. Gerontologist. 2005;45:747-753. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 52] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 37. | Heesterbeek TJ, van der Aa HPA, van Rens GHMB, Twisk JWR, van Nispen RMA. The incidence and predictors of depressive and anxiety symptoms in older adults with vision impairment: a longitudinal prospective cohort study. Ophthalmic Physiol Opt. 2017;37:385-398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 81] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 38. | Shah N, Tran E, Aly M, Phu V, Laughlin E, Malvankar-Mehta MS. Depression and Anxiety in Patients With Irreversible Vision Loss: Meta-Analysis and Systematic Review. Int J Psychiatry Med. 2025;912174251382653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 39. | Yüksel Elgin C, Elgin C. Beyond vision: economic toll of untreated mental health disorders in ophthalmic patients. BJPsych Open. 2025;11:e246. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/