Published online Mar 6, 2026. doi: 10.12998/wjcc.v14.i7.117431
Revised: January 13, 2026
Accepted: February 2, 2026
Published online: March 6, 2026
Processing time: 88 Days and 15.9 Hours
Extrahepatic portal venous obstruction is a major cause of non-cirrhotic portal hypertension in children and young adults. It most commonly results from portal vein thrombosis. While prothrombotic states are recognized contributors, com
A 27-year-old male presented with an episode of melena and had a background of portal cavernoma cholangiopathy and recurrent variceal bleeding since infancy. Previously, he underwent endoscopic sclerotherapy and proximal lienorenal shunt surgery and later developed shunt thrombosis and ischemic biliary strictures requiring Roux-en-Y hepaticojejunostomy. Upper gastrointestinal endoscopy (UGIE) revealed large esophageal and gastric varices that were managed by endoscopic variceal ligation and sclerotherapy. Following the UGIE, he suffered a seizure with deterioration of sensorium, followed by melena and spikes in temperature. Repeat UGIE revealed no active bleeding source and confirmed obliteration of the previously detected varices. Evaluation identified deficiencies in protein S, protein C, and antithrombin III, for which anticoagulation with enoxaparin was initiated. Despite recent sclerotherapy, anticoagulation precipitated melena. Sigmoidoscopy and capsule endoscopy revealed portal hypertensive colopathy with rectal varices. This was managed surgically by creating mesocaval shunts after which enoxaparin was restarted. No further recurrence of bleeding occurred; the patient was discharged in a stable condition.
This case highlighted the importance of thorough coagulation profiling in extrahepatic portal venous obstruction, particularly in young patients with recurrent thrombotic events. Clinicians must navigate the precarious balance between thrombosis prevention and hemorrhage risk.
Core Tip: Extrahepatic portal venous obstruction (EHPVO) is a leading cause of non-cirrhotic portal hypertension in young individuals. It is linked to procoagulable states; however, combined anticoagulant deficiencies are rare. This case described a 27-year-old adult with long-standing EHPVO complicated by recurrent thrombotic and hemorrhagic events in the setting of combined deficiencies of protein S, protein C, and antithrombin III. It highlighted the diagnostic uncertainty in distinguishing between acquired and inherited anticoagulant deficiencies in chronic portal vein thrombosis and underscored the clinical challenge of balancing anticoagulation against the risk of bleeding in EHPVO.
- Citation: Dhali A, Maity R, Sinha A, Singh P, Biswas J, Dhali GK. Combined anticoagulant deficiency causing extrahepatic portal venous obstruction: A case report. World J Clin Cases 2026; 14(7): 117431
- URL: https://www.wjgnet.com/2307-8960/full/v14/i7/117431.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i7.117431
Extrahepatic portal venous obstruction (EHPVO) is an essential cause of non-cirrhotic portal hypertension. According to the Asian Pacific Association for the Study of the Liver consensus (2006), EHPVO is “a vascular disorder of the liver, defined by obstruction of the extra-hepatic portal vein with or without involvement of the intra-hepatic portal veins or splenic or superior mesenteric veins”[1]. The most common presentations of EHPVO include upper gastrointestinal (GI) bleed, abdominal pain or lump, and lower GI bleed[2]. With a bimodal presentation age, it is the leading cause of GI bleeding in children and young adults in developing nations. Some of the etiologies comprise infections, prothrombotic states, and congenital anomalies (portal vein atresia, cleft lip and palate, etc), systemic prothrombotic states (prothrombin gene mutation, deficiencies of protein C, S, antithrombin III, nephrotic syndrome, etc), and local conditions (infections, surgery, trauma). According to the unifying hypothesis, a major thrombotic event occurring in the main portal vein at an early age leads to EHPVO, whereas repeated microthrombotic episodes later in life in the branches of the portal vein lead to idiopathic portal hypertension. A Doppler ultrasonography of the splenoportal axis demonstrating the presence of portal cavernomas is the investigation of choice in EHPVO[3].
Acute portal vein thrombosis (PVT) does not necessarily lead to cavernoma formation; hence, it is a distinct entity. EHPVO has been linked to procoagulant states caused by deficiencies in coagulation inhibitors, such as protein C, protein S, and antithrombin III[4,5]. While few cases of combined protein C and S deficiencies have been reported, triple anticoagulant deficiency is extremely rare and has not been extensively studied[6-9]. Here, we presented a case of a young adult with EHPVO suffering from recurrent thrombotic and hemorrhagic complications due to combined deficiencies of protein C, protein S, and antithrombin III.
A 27-year-old male presented with an episode of melena that had occurred overnight.
Over the preceding night, the patient noted passage of black, tarry stools associated with increasing fatigue, exertional dyspnea, and intermittent lightheadedness but no syncope or hematemesis. He denied abdominal pain, distension, jaundice, pruritus, or altered bowel habits and reported no recent fever, infections, trauma, surgery, prolonged immobili
The patient was born via normal vaginal delivery and had no history of abdominal trauma, surgical intervention or umbilical sepsis. He had an index presentation of recurrent, well-tolerated variceal bleeding when he was 6 months old. He was treated with endoscopic sclerotherapy near his residence. At ages 9 years and 13 years, he was hospitalized due to variceal bleeding. Investigations revealed a portal cavernoma with multiple collaterals near the splenic hilum. His coagulation profile was within normal limits. He was treated with multiple sessions of endoscopic sclerotherapy and secondary prophylaxis with a beta blocker.
Six years before presentation, he had recurrent episodes of cholangitis and was diagnosed with portal cavernoma cholangiopathy (PCC). He was treated with multiple sessions of endoscopic retrograde cholangiopancreatography with biliary endoprosthesis and underwent proximal lienorenal shunt with splenectomy 5 years prior. Subsequently, 7 months after surgery, he was diagnosed with shunt thrombosis. He was prescribed an oral anticoagulant; however, his coagulation state was not evaluated at that time. Oral anticoagulants were stopped 2 years prior as he was diagnosed with ischemic biliary stricture and underwent Roux-en-Y hepaticojejunostomy with cholecystectomy. Post-surgery, a triphasic contrast-enhanced computed tomography (CT) of the whole abdomen revealed mildly dilated intrahepatic biliary radicles due to pneumobilia and PVT extending into the superior mesenteric vein. Hence, he began taking oral anticoagulants again. They were stopped after an abdominal ultrasonography a few months after the surgery showed no thrombi in the portal vein.
The patient denied any family history of bleeding disorders or malignancies. He had no history of recent travel, smoking, or alcohol intake and was not on any regular medications (including nonsteroidal anti-inflammatory drugs, antiplatelets, or herbal drugs).
Upon physical examination, he had severe pallor but was alert, conscious, and oriented to time, place, and person and was hemodynamically stable (blood pressure 100/60 mmHg and pulse rate 98/minute). There were no signs of icterus or chronic liver disease. The systemic examination was unremarkable.
A complete blood count on the day of admission revealed a markedly low hemoglobin of 4.3 g/dL that was corrected by packed red blood cell transfusions (all routine blood tests performed during his hospital stay are listed in Table 1). Before the upper GI endoscopy (UGIE), the patient had an episode of seizure with postictal confusion. However, there was no deterioration in hemodynamic or neurological status.
| Parameter (units) | Day 1 | Day 3 | Day 5 | Day 7 | Day 11 | Day 14 | Day 30 |
| Hemoglobin (g/dL) | 4.3 | 6.8 | 9.4 | 9.4 | 8.2 | 7.7 | 9.0 |
| Total leucocyte count (cells/cubic mm) | 11370 | 21700 | 11500 | 14400 | 12600 | 15000 | 21600 |
| Differential leucocyte count (neutrophil/Lymphocyte) (%) | 90/7 | 80/10 | 93/3 | 83/2 | 87/8 | 87/0.6 | 93/4 |
| Platelet (cells/cubic mm) | 60000 | 60000 | 52000 | 60000 | 100000 | 220000 | 100000 |
| Bilirubin (total/direct) (mg/dL) | 0.5 | 2.8/0.6 | 1.9/0.7 | 1.2/0.4 | 1.4/0.4 | 2.5/0.5 | 2/0.5 |
| Total protein (g/dL) | - | 4.3 | 4.7 | 5.0 | 5.9 | 5.3 | 5.2 |
| Albumin (g/dL) | 3.1 | 2.6 | 2.7 | 2.6 | 3.8 | 3.7 | 2.7 |
| Globulin (g/dL) | - | 1.7 | 2.0 | 2.4 | 2.1 | 1.6 | 2.5 |
| ALT (IU/L) | 32 | 60 | 60 | 52 | 42 | 19 | 30 |
| AST (IU/L) | 58 | 122 | 73 | 64 | 39 | 31 | 53 |
| ALP (IU/L) | 65 | 87 | 92 | 89 | 69 | 65 | 64 |
| PT (second) | 13.4 | 14.5 | - | 12.0 | 25.0 | 20.0 | 16.0 |
| INR | 0.95 | 1.25 | - | 1.03 | 0.90 | 0.80 | 1.11 |
| Urea (mg/dL) | 28 | 63 | 36 | 28 | - | - | 13 |
| Creatinine (mg/dL) | 0.9 | 0.9 | 0.8 | 1.0 | - | - | 0.4 |
| Sodium (mEq/L) | 135 | 143 | 147 | 135 | 142 | - | 131 |
| Potassium (mEq/L) | 5.5 | 3.6 | 3.1 | 4.3 | 3.7 | - | 3.9 |
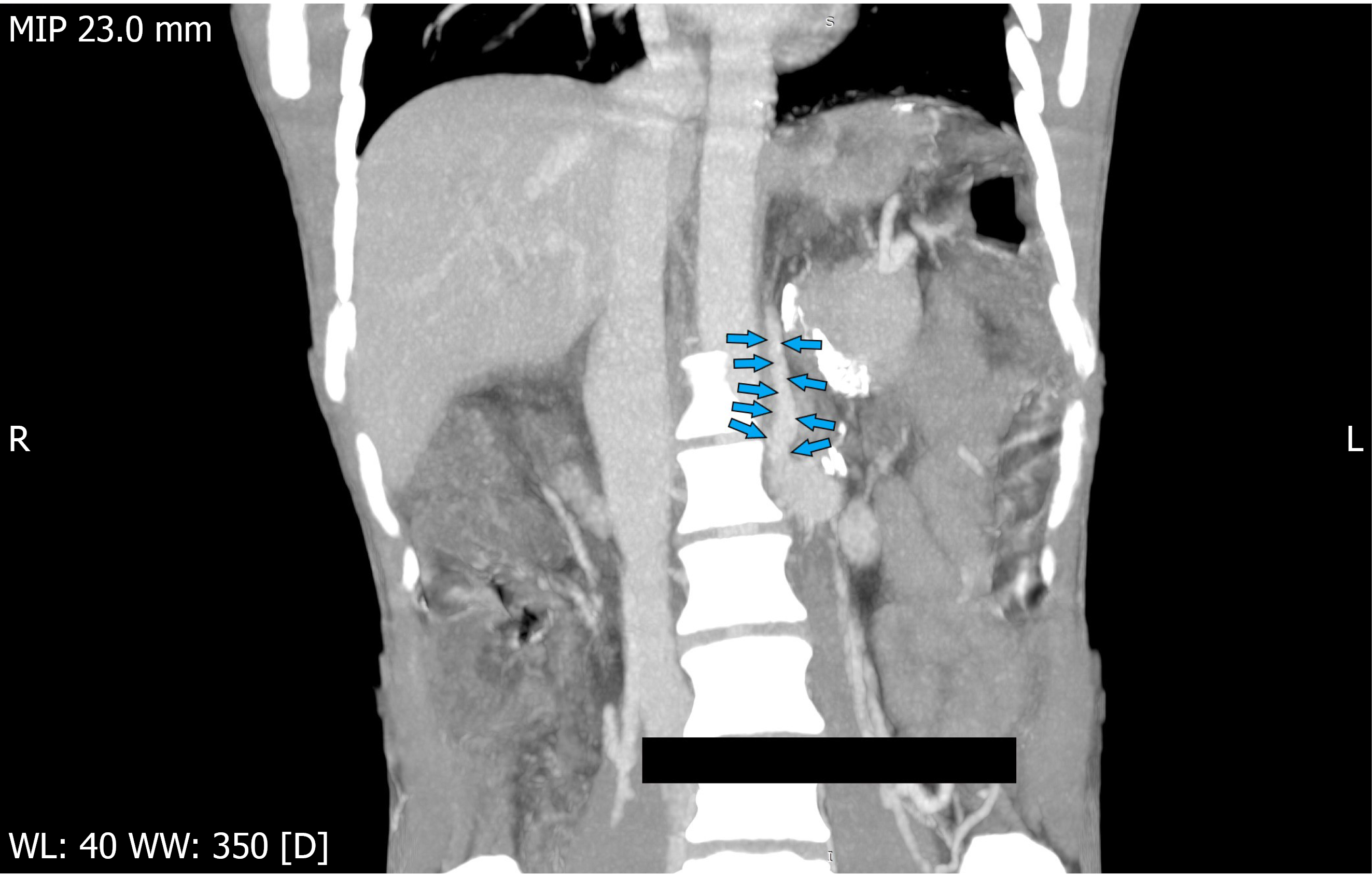
UGIE revealed one column of grade 3 esophageal varix and one GOV2F1 gastroesophageal varix (GOV2F1: Tortuous, extending from the gastric fundus towards the greater curvature) that were treated with endoscopic variceal ligation and endoscopic sclerotherapy (using cyanoacrylate glue injection), respectively. Following the UGIE, he had another episode of seizure with deterioration of sensorium without any focal neurological deficits. He was given intravenous antiepileptics. This was followed by melena along with a drop in hemoglobin. In view of his worsening vital parameters, he was shifted to the Intensive Therapy Unit, intubated, and put on mechanical ventilation. Repeat UGIE revealed no active bleeding sources and confirmed obliteration of gastric and gastroesophageal varices. Non-contrast CT of the head revealed a mega cisterna magna but no evidence of an infarct. CT angiography of abdominal vessels revealed a 6 mm spontaneously developed shunt arising from the left renal vein with obliteration of gastric and esophageal varices and bilaterally enlarged intrahepatic biliary radicles along with closure of the previously created shunt (Figure 1).
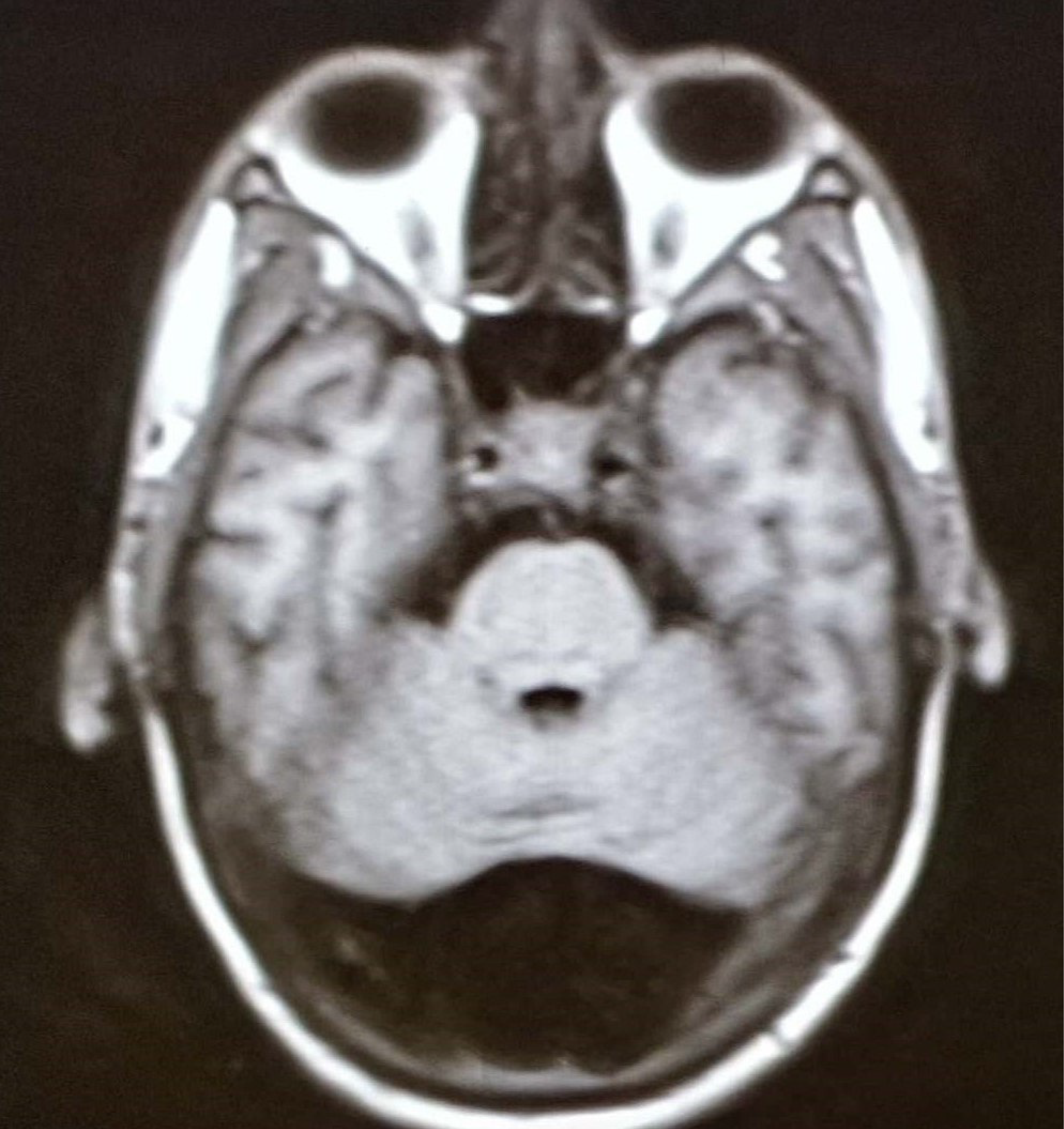
The patient had fever spikes without any neck rigidity or Kernig sign. Neurology input was sought, and the patient underwent electroencephalogram, magnetic resonance imaging (MRI) of the brain, and a lumbar puncture were performed. Lumbar puncture was within normal limits (total protein: 17 mg/dL; glucose: 111 mg/dL; cell count: 2 cells/cubic mm; no capsulated organisms seen on India ink; no fungal elements seen on wet mount; and no growth seen in culture). Electroencephalogram showed generalized slowing of waves without any evidence of spikes or triphasic waves. An MRI of the brain revealed mega cisterna magna, and magnetic resonance (MR) angiography of the brain and neck vessels was within normal limits (Figure 2).
Input from infectious disease was sought, and the antibiotic regimen was changed to ceftriaxone, acyclovir, and doxycycline. On the following day the patient responded to commands and was subsequently extubated. He subse
| Parameter (units) | Ascitic fluid | Pleural fluid |
| Total protein (g/dL) | 0.6 | 0.4 |
| Total albumin (g/dL) | 0.3 | 0.3 |
| Total cell count | 150 (neutrophil 60%, lymphocyte 30%, mesothelial cells 10%) | 80 (neutrophil 20%, lymphocyte 50%, mesothelial cells 30%) |
A comprehensive coagulation profile was performed, revealing severe protein S deficiency and mild deficiencies in protein C and antithrombin III with normal prothrombin time, activated partial thromboplastin time, and international normalized ratio levels (Table 3). Thus, the final diagnosis was EHPVO secondary to triple anticoagulant deficiency.
| Parameter | Observed value | Reference interval |
| PT | 14.5 s | 11.0-15.0 s |
| aPTT | 32 s | 25-35 s |
| INR | 1.25 | 0.80-1.20 |
| Thrombin time | 17 s | 14-19 s |
| Fibrinogen | 283 mg/dL | 200-400 mg/dL |
| D-Dimer | 1.30 µFEU/mL | < 0.5 µFEU/mL |
| Protein C, % | 42 | 70-140 |
| Protein S, % | 18 | 60-150 |
| Antithrombin III, % | 62 | 80-130 |
| Von Willebrand factor, % | 100 | 50-160 |
| Factor V Leiden mutation | Absent | - |
| Lupus anticoagulant | Negative | - |
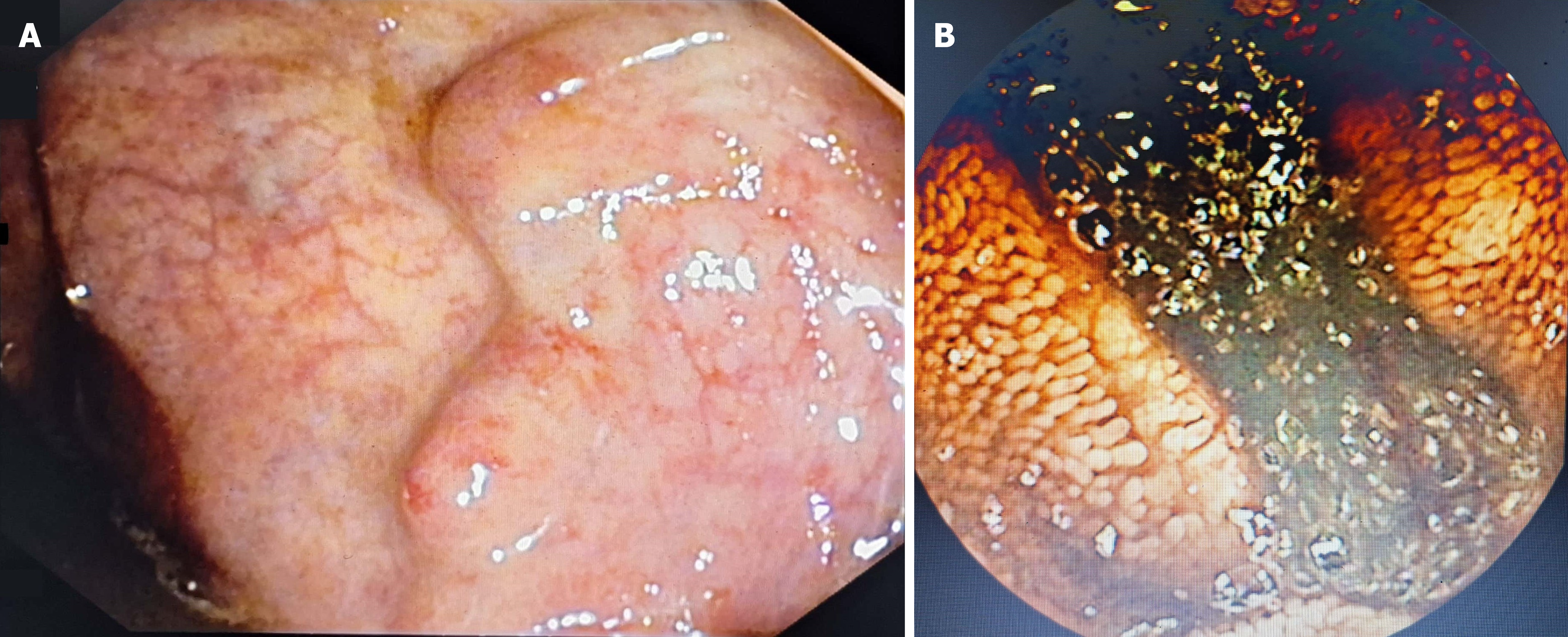
With obliterated varices and no fresh melena, low molecular weight heparin (LMWH) in the form of enoxaparin 1 mg/kg/day was initiated after consultation with the hematologist. The following day, the patient had three episodes of melena with a drop in hemoglobin. His enoxaparin dosage was lowered. However, the bleeding continued, and the anticoagulant was discontinued. Sigmoidoscopy revealed portal hypertensive colopathy with two columns of rectal varices (Figure 3A). With ongoing melena a wireless capsule endoscopy was performed, revealing multiple ectopic varices in the jejunum with active bleeding from some varices (Figure 3B). An urgent GI surgery consultation was sought for definitive management.
The patient underwent mesocaval shunt surgery using an ileocolic vein-inferior vena cava anastomosis and an inferior mesenteric vein-inferior vena cava interposition polytetrafluoroethylene graft. However, there was a drop in hemoglobin post-surgery. After 3 days, re-exploration was performed during which bleeding from the mesenteric site was controlled, and partial thrombosis of the polytetrafluoroethylene graft was corrected by removing the clot. Post-surgery, melena subsided, and no transfusion was required; therefore, enoxaparin was restarted. Due to the prolonged use of antimicrobials, he developed a Clostridium difficile infection (identified by the presence of glutamate dehydrogenase antigen and toxin A in his stool) that was managed with oral vancomycin and intravenous metronidazole.
There was no further recurrence of bleeding; the patient was discharged in a stable condition and was doing well on follow-up visits.
EHPVO is an essential cause of extrahepatic portal hypertension, which often presents in children and young adults with GI bleeding. It is a heterogeneous condition, most often resulting from PVT, and may be associated with prothrombotic states[10,11]. It is multifactorial and can be either due to a PVT or secondary to malignant invasion, compression, or encasement of the portal vein. Since PVT is a reasonably common occurrence in liver cirrhosis, it is crucial to determine whether liver cirrhosis is linked to PVT when evaluating an adult with PVT. In patients with cirrhosis PVT is associated with portal hypertension and the development of portosystemic shunts[10].
Following PVT, failure of complete recanalization may result in cavernomatous transformation of the portal vein with development of porto-portal collaterals. These collaterals are insufficient to decompress the portal system and may lead to complications such as PCC, which manifests due to compression of the biliary tree by collateral veins or ischemia of biliary venules[10,12]. This mechanism likely explains the patient’s prior ischemic biliary stricture requiring Roux-en-Y hepaticojejunostomy[13].
Most patients have well-preserved liver function; however, 4%-9% of people may have abnormal transaminase levels. Alkaline phosphatase and gamma-glutamyl transpeptidase levels rise when portal biliopathy develops, and hypoalbuminemia may occur during bleeding episodes, particularly in the setting of growth failure[3]. To diagnose EHPVO, Doppler ultrasound, CT, or MR angiography are recommended to detect portal vein obstruction, the presence of solid intraluminal material, or portal vein cavernoma[14]. The Baveno VII consensus suggests using contrast-enhanced CT or MR angiography to confirm a diagnosis made by Doppler ultrasonography[15].
In our case report the presence of a portal cavernoma, signifying an old thrombus and recurrent episodes of cholangitis due to PCC (a progressive condition with symptoms developing late in the course of portal hypertension), explains the protracted course of the disease process[6,12]. The development of collaterals could serve as a partial escape mechanism from the effects of an acute portal venous occlusion for some time. However, since they failed to relieve the portal hypertension, the patient ultimately experienced variceal bleeding. The patient also had seizures and a high spiking fever but with no clinical or laboratory evidence of meningoencephalitis. The episodes of seizures and altered sensorium raised concern for a possible cerebral venous thrombosis; however, in the absence of an MRI with venography, this diagnosis remains speculative. Normal MR angiography and cerebrospinal fluid analysis reduced the likelihood of arterial or infectious etiologies, but definitive exclusion of venous thrombosis was not possible. The recurrence of symptoms, implicating a recurrence of thrombosis, prompted an evaluation of various causes of hypercoagulability, including inherited thrombophilia, given the protracted course of the disease process and its recurrent manifestations dating back to childhood.
Deficiencies in natural anticoagulants are rare and can either be inherited or acquired. Protein C deficiency occurs in approximately 1 of every 200 to 500 people, whereas protein S deficiency can be expected in 1 of every 500 individuals. Antithrombin deficiency is the least common of the three deficiencies, occurring in approximately 1 in every 2000 to 5000 individuals[16]. There have been several case reports of PVT due to combined protein C and protein S deficiency[6-9]. However, the combined deficiency of protein C, protein S, and antithrombin III appears to be extremely rare.
Fisher et al[5] found that out of 20 patients with portal hypertension due to PVT, only 6 patients (21%) had combined deficiency of all three proteins. However, in the clinical setting of a portal cavernoma, plasma levels of coagulation inhibitors can be moderately decreased; hence, cautious interpretation of their levels is recommended[17,18]. This was further corroborated by the patient’s coagulation profile from his childhood. The profile was negative, likely ruling out hereditary causes. In a study of 20 children with portal vein obstruction, Dubuisson et al[4] found that while the levels of protein C, protein S, and/or antithrombin III were often lower in these individuals, they were mainly normal in their parents. This implies that anticoagulant deficiencies in EHPVO are probably not hereditary. Fisher et al[5] argue that deficiency of natural anticoagulant proteins is common in PVT and is perhaps a secondary phenomenon in most cases, occurring as part of a global disturbance of coagulation variables. The mechanism remains unclear but may result from a combination of reduced hepatic blood flow and portosystemic shunting. However, both Dubuisson’s and Fischer’s studies suffer from a lack of replicability due to their small sample sizes.
In the present case previously documented normal coagulation profiles during childhood, together with the long-standing portal hypertension and cavernomatous transformation, suggest that the observed deficiencies may represent an acquired process. Failure to conclusively establish whether the observed anticoagulant deficiencies were primary or secondary represents an important limitation. Genetic testing for inherited thrombophilia, including PROC, PROS1, and SERPINC1 mutations, was not performed in this patient. The absence of molecular confirmation further limits differentiation between inherited and acquired anticoagulant deficiencies and should be considered when interpreting this case. Against the background of the patient’s clinical findings, particularly the severely reduced protein S levels, this was something that could not be considered insignificant.
Levels of protein C, protein S, and antithrombin III are known to be influenced by acute illness, systemic inflammation, infection, active bleeding, and blood product transfusion[16,18]. Although liver synthetic function was largely preserved in this patient, the timing of testing during an acute hospitalization with gastrointestinal bleeding, sepsis, and transfusion may have contributed to reduced anticoagulant levels. Repeat testing after complete clinical stabilization was not performed and would have strengthened diagnostic certainty. This limitation should be acknowledged when interpreting the severity and clinical significance of the observed deficiencies.
The aim of treatment in PVT is to achieve recanalization of the portal vein. Systemic anticoagulation is the treatment of choice in recent non-cirrhotic PVT and should be initiated upon diagnosis at a therapeutic dosage. The preferred agent is LMWH, followed by vitamin K antagonists. Unfractionated heparin is generally avoided due to the risk of heparin-induced thrombocytopenia[15]. If anticoagulation fails, endovascular thrombolysis or surgical interventions become necessary and can be carried out via direct transhepatic or trans-splenic approaches or through transjugular access with or without transjugular intrahepatic portosystemic shunt placement[13]. In patients with chronic PVT leading to EHPVO, a multidisciplinary approach becomes necessary for managing variceal bleeding, portal biliopathy, and hypersplenism[19,20]. Since upper GI bleeding is the most common presentation and the second most common indication for surgery in EHPVO, anticoagulants must be prescribed cautiously[20]. However, it does not increase the risk of bleeding in patients with underlying thrombotic disorders[13]. Although the absence of ascites or splenomegaly at diagnosis does not rule out the presence of gastroesophageal varices, their presence is independently linked to such varices[21].
Portal decompression with a portosystemic shunt (such as the splenorenal shunt and mesocaval shunt) or TIPSS resolves the significant complications of EHPVO, i.e. variceal hemorrhage and hypersplenism[22]. Non-selective portosystemic shunts are not the ideal surgery in children due to the risk of hepatic encephalopathy and liver failure, thus lowering the postoperative quality of life[23]. TIPSS, despite having good patency rates and safety profiles in children, is challenging to perform in small patients[23,24]. The Rex shunt, a surgical procedure that restores hepatopetal portal blood flow to the liver via a bypass between the mesenteric vein and the left hepatic vein, is preferred in children because it can prevent liver dysfunction, enhance its metabolic function, and improve the patient’s quality of life[15,23]. In the presence of symptomatic portal biliopathy (indicated by jaundice, cholangitis, choledocholithiasis, or biliary stricture), biliary decompression becomes necessary[3]. However, shunt surgeries have been shown to correct biliary abnormalities in most patients by collapsing the enlarged varices in and around the biliary tree. Bilioenteric decompression becomes necessary in patients with non-shuntable veins, a previously performed blocked portosystemic shunt, or a failed shunt surgery[22].
In our case, therapeutic anticoagulation with enoxaparin was initiated at a dose of 1 mg/kg/day following hematology consultation to balance the high thrombotic risk against recent variceal intervention. A long-term anticoagulation strategy remains individualized, taking into consideration bleeding risk, shunt patency, and the recurrence of thrombosis. Although direct oral anticoagulants have emerging evidence in non-cirrhotic portal vein thrombosis, their role in patients with extensive portal hypertension, ectopic varices, and prior surgical shunts remains uncertain[13,25]. After the patient was initiated on LMWH, he had episodes of melena the very next day. The bleeding persisted even after decreasing the dose of LMWH, compelling its withdrawal. The patient had to undergo surgical interventions twice for this bleeding episode, and heparin was reinitiated once bleeding had stopped. Since the underlying pathophysiology is a prothrombotic state and the manifestation of the disease process is characterized by bleeding, management of such a scenario presents a constant challenge of treading a fine line between thrombotic and bleeding risks.
This case underscored the complexity of managing EHPVO with recurrent thrombotic manifestations and concurrent bleeding risk. Observed deficiencies of natural anticoagulants may represent an acquired phenomenon in the setting of chronic portal hypertension, emphasizing the need for cautious interpretation of coagulation profiles. Individualized, multidisciplinary management is essential to balance thrombosis prevention against hemorrhagic complications in such patients.
| 1. | Sarin SK, Sollano JD, Chawla YK, Amarapurkar D, Hamid S, Hashizume M, Jafri W, Kumar A, Kudo M, Lesmana LA, Sharma BC, Shiha G, Janaka de Silva H; Members of the APASL Working Party on Portal Hypertension. Consensus on extra-hepatic portal vein obstruction. Liver Int. 2006;26:512-519. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 225] [Cited by in RCA: 187] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 2. | Rathi P, Nair S. An Audit of Extrahepatic Portal Vein Obstruction: Experience from Tertiary Referral Center. J Assoc Physicians India. 2023;71:11-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 3. | Khanna R, Sarin SK. Idiopathic portal hypertension and extrahepatic portal venous obstruction. Hepatol Int. 2018;12:148-167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 52] [Article Influence: 6.5] [Reference Citation Analysis (4)] |
| 4. | Dubuisson C, Boyer-Neumann C, Wolf M, Meyer D, Bernard O. Protein C, protein S and antithrombin III in children with portal vein obstruction. J Hepatol. 1997;27:132-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 39] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 5. | Fisher NC, Wilde JT, Roper J, Elias E. Deficiency of natural anticoagulant proteins C, S, and antithrombin in portal vein thrombosis: a secondary phenomenon? Gut. 2000;46:534-539. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 76] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 6. | Usman A, Jibbran A, Ahmad U, Tariq F, Saleem MM, Mudassar M. Portal vein thrombosis due to inherited combined deficiency of protein C and S in a young adult: A case report. Radiol Case Rep. 2024;19:6235-6240. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 7. | Rodríguez-Leal GA, Morán S, Corona-Cedillo R, Brom-Valladares R. Portal vein thrombosis with protein C-S deficiency in a non-cirrhotic patient. World J Hepatol. 2014;6:532-537. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 8. | Hwang S, Kim DY, Kim M, Chon YE, Lee HJ, Park YN, Park JY, Ahn SH, Han KH, Chon CY. [Deficiencies in proteins C and S in a patient with idiopathic portal hypertension accompanied by portal vein thrombosis]. Korean J Hepatol. 2010;16:176-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 9. | Das SK, Ray A, Jana CK, Banerjee N, Khaskil S. Chronic portal vein thrombosis due to combined deficiency of protein C and protein S. J Indian Med Assoc. 2011;109:753-754. [PubMed] |
| 10. | Nery FG, Valla DC. Vascular diseases of the liver. In: Feldman M, Friedman LS, Brandt LJ, eds. Sleisenger and Fordtran’s Gastrointestinal and Liver Disease. Vol 2. 11th ed. Philadelphia (PA): Elsevier, 2020: 1324-1335. |
| 11. | Feldman AG, Suchy FJ. Portal Hypertension and Varices. In: Kliegman R, St Geme III JW, Blum NJ, Shah SS, Tasker RC, Wilson KM, eds. Nelson Textbook of Pediatrics. Vol 2. 21st ed. Philadelphia (PA): Elsevier, 2019: 2141-2143. |
| 12. | Dhiman RK, Saraswat VA, Valla DC, Chawla Y, Behera A, Varma V, Agarwal S, Duseja A, Puri P, Kalra N, Rameshbabu CS, Bhatia V, Sharma M, Kumar M, Gupta S, Taneja S, Kaman L, Zargar SA, Nundy S, Singh SP, Acharya SK, Dilawari JB. Portal cavernoma cholangiopathy: consensus statement of a working party of the Indian national association for study of the liver. J Clin Exp Hepatol. 2014;4:S2-S14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 74] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 13. | Intagliata NM, Caldwell SH, Tripodi A. Diagnosis, Development, and Treatment of Portal Vein Thrombosis in Patients With and Without Cirrhosis. Gastroenterology. 2019;156:1582-1599.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 311] [Cited by in RCA: 269] [Article Influence: 38.4] [Reference Citation Analysis (2)] |
| 14. | de Franchis R; Baveno VI Faculty. Expanding consensus in portal hypertension: Report of the Baveno VI Consensus Workshop: Stratifying risk and individualizing care for portal hypertension. J Hepatol. 2015;63:743-752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2652] [Cited by in RCA: 2372] [Article Influence: 215.6] [Reference Citation Analysis (2)] |
| 15. | de Franchis R, Bosch J, Garcia-Tsao G, Reiberger T, Ripoll C; Baveno VII Faculty. Baveno VII - Renewing consensus in portal hypertension. J Hepatol. 2022;76:959-974. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2244] [Cited by in RCA: 2063] [Article Influence: 515.8] [Reference Citation Analysis (15)] |
| 16. | Lipe B, Ornstein DL. Deficiencies of natural anticoagulants, protein C, protein S, and antithrombin. Circulation. 2011;124:e365-e368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 56] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 17. | DeLeve LD, Valla DC, Garcia-Tsao G; American Association for the Study Liver Diseases. Vascular disorders of the liver. Hepatology. 2009;49:1729-1764. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 799] [Cited by in RCA: 669] [Article Influence: 39.4] [Reference Citation Analysis (0)] |
| 18. | Condat B, Pessione F, Hillaire S, Denninger MH, Guillin MC, Poliquin M, Hadengue A, Erlinger S, Valla D. Current outcome of portal vein thrombosis in adults: risk and benefit of anticoagulant therapy. Gastroenterology. 2001;120:490-497. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 408] [Cited by in RCA: 336] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 19. | Arora A, Sarin SK. Multimodality imaging of primary extrahepatic portal vein obstruction (EHPVO): what every radiologist should know. Br J Radiol. 2015;88:20150008. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 23] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 20. | Das S, Manadal TS, Das S, Biswas J, Gupta A, Mukherjee S, Ray S. Surgical outcome of extrahepatic portal venous obstruction: Audit from a tertiary referral centre in Eastern India. Ann Hepatobiliary Pancreat Surg. 2023;27:350-365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 21. | Noronha Ferreira C, Seijo S, Plessier A, Silva-Junior G, Turon F, Rautou PE, Baiges A, Bureau C, Bosch J, Hernández-Gea V, Valla D, García-Pagan JC. Natural history and management of esophagogastric varices in chronic noncirrhotic, nontumoral portal vein thrombosis. Hepatology. 2016;63:1640-1650. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 81] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 22. | Bhavsar R, Yadav A, Nundy S. Portal cavernoma cholangiopathy: Update and recommendations on diagnosis and management. Ann Hepatobiliary Pancreat Surg. 2022;26:298-307. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 23. | Zhang J, Li L. Rex Shunt for Extra-Hepatic Portal Venous Obstruction in Children. Children (Basel). 2022;9:297. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 24. | Raissi D, Brahmbhatt S, Yu Q, Jiang L, Liu C. Transjugular intrahepatic portosystemic shunt for pediatric portal hypertension: A meta-analysis. J Clin Imaging Sci. 2023;13:18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 25. | Simonetto DA, Singal AK, Garcia-Tsao G, Caldwell SH, Ahn J, Kamath PS. ACG Clinical Guideline: Disorders of the Hepatic and Mesenteric Circulation. Am J Gastroenterol. 2020;115:18-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 196] [Cited by in RCA: 174] [Article Influence: 29.0] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/