Published online Mar 6, 2026. doi: 10.12998/wjcc.v14.i7.116694
Revised: January 5, 2026
Accepted: February 11, 2026
Published online: March 6, 2026
Processing time: 107 Days and 19.1 Hours
Chronic subdural hematoma (cSDH) is typically surgically managed; however, middle meningeal artery embolization (MMAE) has recently gained attention as a minimally invasive alternative. Optimal embolization techniques and patient selection criteria remain under investigation. Here, we present a refined em
A 78-year-old female with bilateral symptomatic cSDH and significant mass effect was unsuitable for surgery due to advanced age, comorbidities, and religious considerations. Standalone MMAE using a refined layered technique achieved complete hematoma resolution and full brain re-expansion within 3 months, without complications. The layered embolization technique involved initial deep penetration of the distal neovascular branches using low-concentration particles, followed by proximal vessel occlusion with standard-concentration particles under continuous fluoroscopic guidance. This controlled approach optimizes distal embolization, minimizes premature reflux, and facilitates complete he
Standalone MMAE using layered embolization is safe and effective for managing cSDH with significant mass effect in high-risk surgical candidates.
Core Tip: This case demonstrates a refined layered embolization technique as a novel approach for treating chronic subdural hematoma with significant mass effect. By employing sequential low- and standard-concentration embolic suspensions, the technique achieves deep distal penetration and proximal sealing, facilitating complete hematoma clearance within three months. This approach expands the therapeutic role of middle meningeal artery embolization to high-risk or surgically unsuitable patients.
- Citation: Chen CY, Hsu YH. Complete resolution of massive chronic subdural hematoma with refined layered middle meningeal artery embolization: A case report. World J Clin Cases 2026; 14(7): 116694
- URL: https://www.wjgnet.com/2307-8960/full/v14/i7/116694.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i7.116694
Chronic subdural hematoma (cSDH) is a common neurological condition with an annual incidence of approximately 10 per 100000 individuals. Moreover, with population aging and the widespread use of antiplatelet and anticoagulant medications, the prevalence of cSDH is rising[1,2]. Surgical evacuation remains the standard treatment for symptomatic cases, particularly those with significant mass effect[1,2]. Middle meningeal artery embolization (MMAE) has recently emerged as a minimally invasive alternative to surgery[3-5]. However, technical refinements to enhance safety, efficacy, and hematoma clearance remain underexplored. Here, we report a case of massive cSDH with substantial mass effect successfully treated with standalone MMAE using a refined layered embolization technique, achieving complete he
A 78-year-old female presented with preserved consciousness, slurred speech, and right-sided weakness.
This 78-year-old woman had sustained a head injury after a fall two weeks earlier. She subsequently developed an acute onset of slurred speech and right-sided weakness during the night on the day of presentation.
The patient had a medical history of hypertension, dyslipidemia, and gout. She underwent surgery for a thyroid nodule 28 years ago and surgery for genital prolapse 12 years ago.
The patient is a 78-year-old female who is illiterate, widowed, and speaks Mandarin and Minnanese. She has no known drug or food allergies. She denies cigarette smoking, alcohol consumption, and betel nut use, and reports no travel history within the past three months. Her diet is regular, although she avoids beef. She received an influenza vaccination in 2023. Her family history is unremarkable, with no reported diabetes mellitus, hypertension, or cancer. She is primarily cared for by her three adult children (two sons and one daughter).
On admission, the patient was alert with a preserved mental status (Glasgow Coma Scale: E4V5M6). Neurological examination revealed right-sided weakness, with muscle strength in the right upper and lower limbs graded as 4/5 on the Medical Research Council scale, indicating movement against gravity with some resistance. Sensory examination, including pain perception, was normal.
Routine blood tests, including complete blood count, electrolytes, coagulation profile, blood biochemistry, and immune and infection markers, were all within normal limits. Routine urine analysis and urinary sediment examination revealed no abnormal findings. Routine fecal testing, including occult blood testing, was also unremarkable.
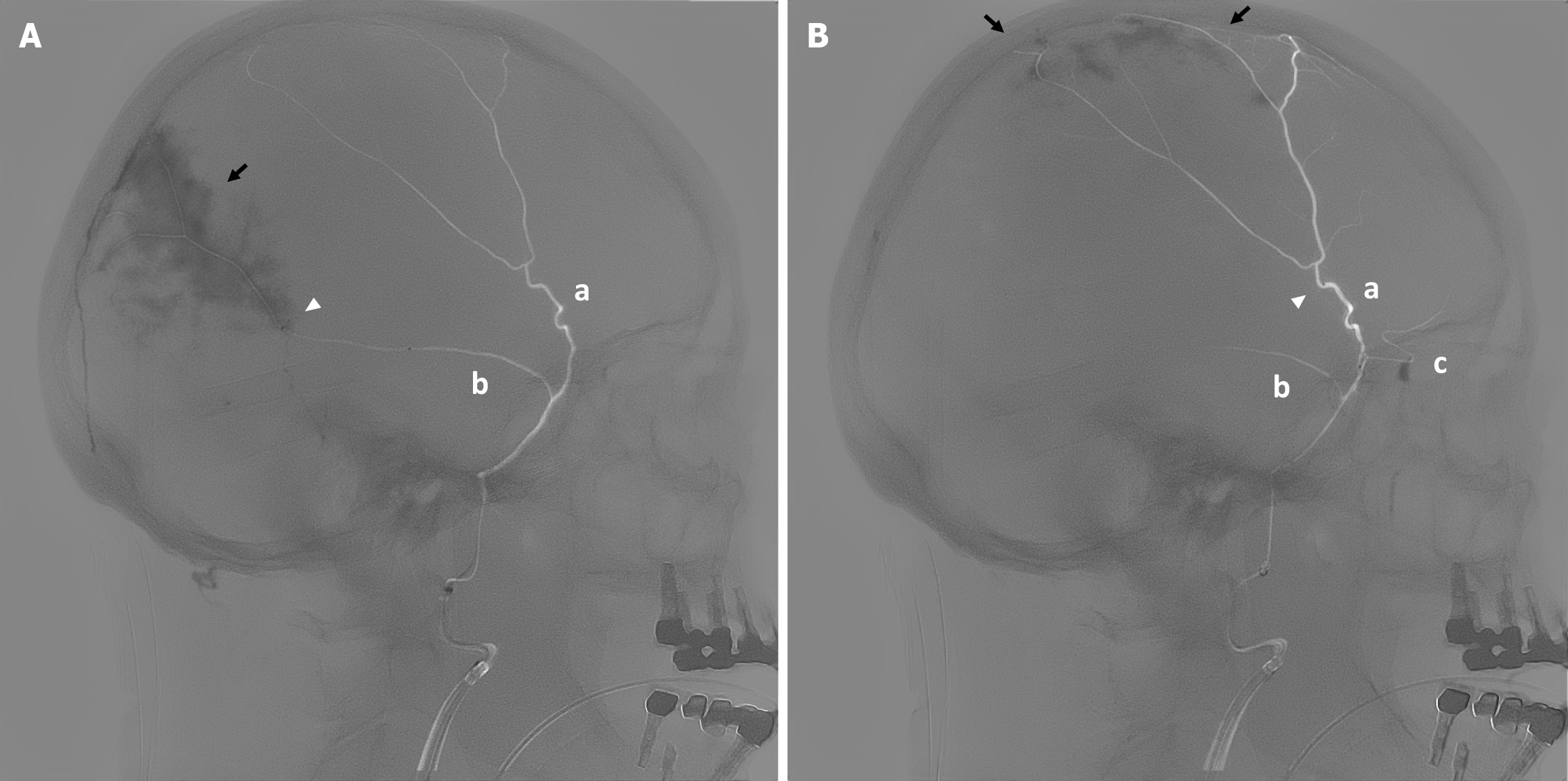
Brain computed tomography (CT) demonstrated bilateral cSDH with mass effect (3.3 cm on the right and 1.64 cm on the left). Selective pre-embolization angiography of the middle meningeal artery (MMA) revealed the anterior and posterior branches supplying the external membranes of the hematomas, demonstrating a contrast blush indicative of pathological neovascularization (Figure 1).
The final diagnosis was massive cSDH with substantial mass effect.
Given the patient’s advanced age, comorbidities, and personal religious considerations, MMAE was selected as the treatment instead of surgical evacuation. To minimize the risk of non-target embolization, hazardous anastomoses - particularly with the ophthalmic artery - were carefully identified, and the microcatheter was advanced beyond the origin of the meningo-ophthalmic artery to prevent reflux.
Embolization was performed using spherical polyvinyl alcohol (PVA) particles (Embozene™, 400 μm, 2 mL) initially suspended in 5 mL of non-ionic iodinated contrast medium (iohexol, 370 mg/mL), yielding a standard-concentration suspension with a 1:2.5 (v/v) particle-to-contrast ratio. To facilitate deeper distal penetration into the pathological microvascular network, a low-concentration suspension was prepared by diluting a 3.5-mL aliquot of the standard suspension (containing 1 mL of PVA particles) with an additional 3.5 mL of iohexol, resulting in a final 1:6 (v/v) ratio. The preparation protocols and clinical objectives for both embolic concentrations are summarized in Table 1.
| Suspension | PVA particles (mL) | Contrast medium (mL) | Ratio (v/v) | Clinical objective |
| Standard concentration | 2 | 5 | 1:2.5 | Proximal sealing and vessel stasis |
| Low concentration | 1 | 6 | 1:6 | Deep penetration of distal neovascular network |
A layered embolization technique was employed. The microcatheter was first advanced distally into the selected MMA branch, where the low-concentration suspension was slowly injected to achieve deep penetration of the neovascular network. This was followed by the administration of the standard-concentration suspension to fill and seal the proximal MMA branches, ensuring robust and complete vascular occlusion.
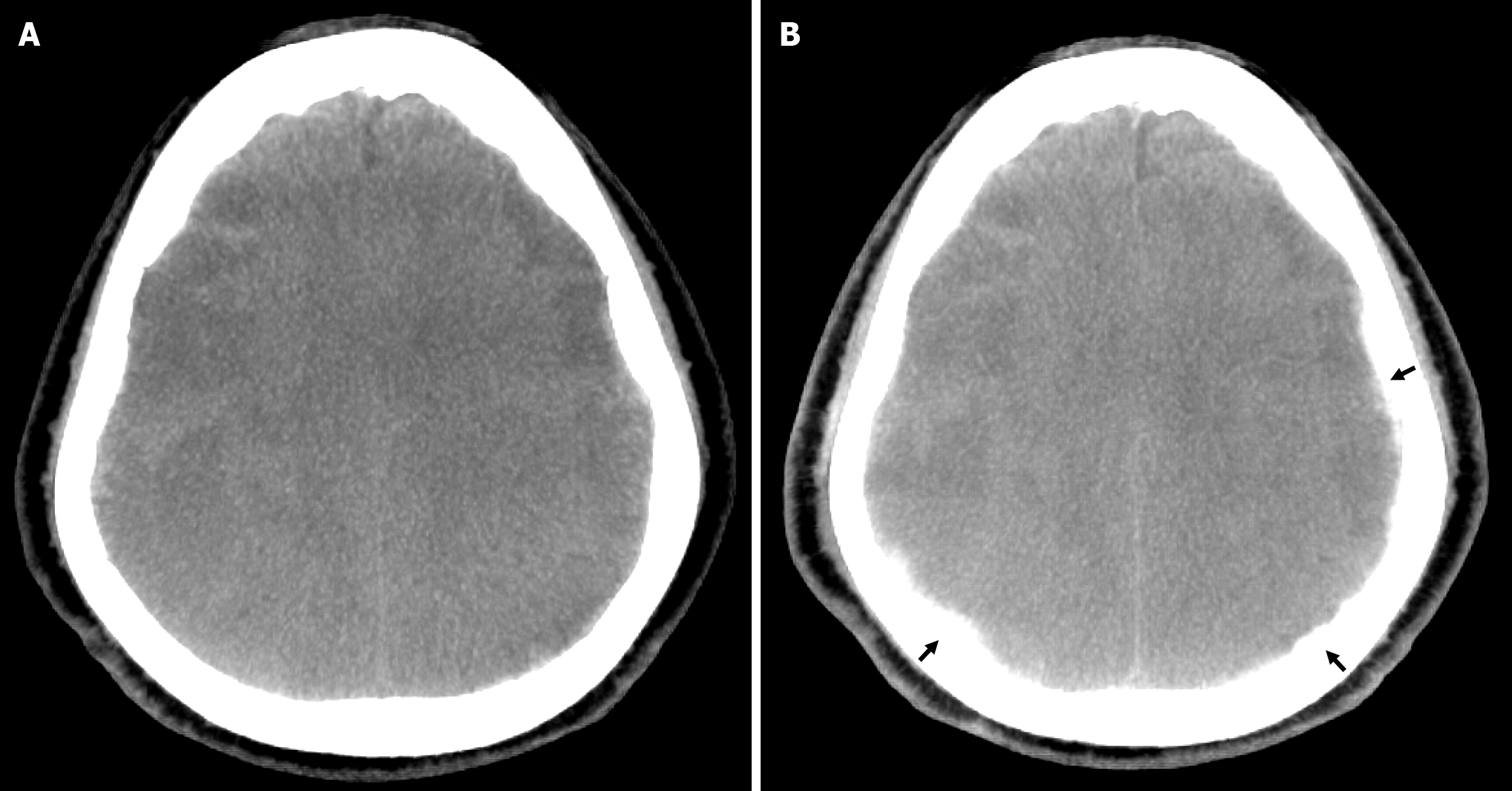
The injection proceeded under continuous fluoroscopic guidance until complete vessel stasis was confirmed angiographically. Intraprocedural cone-beam CT following MMAE demonstrated contrast penetration into the subdural space (Figure 2), consistent with deposition of PVA particles.
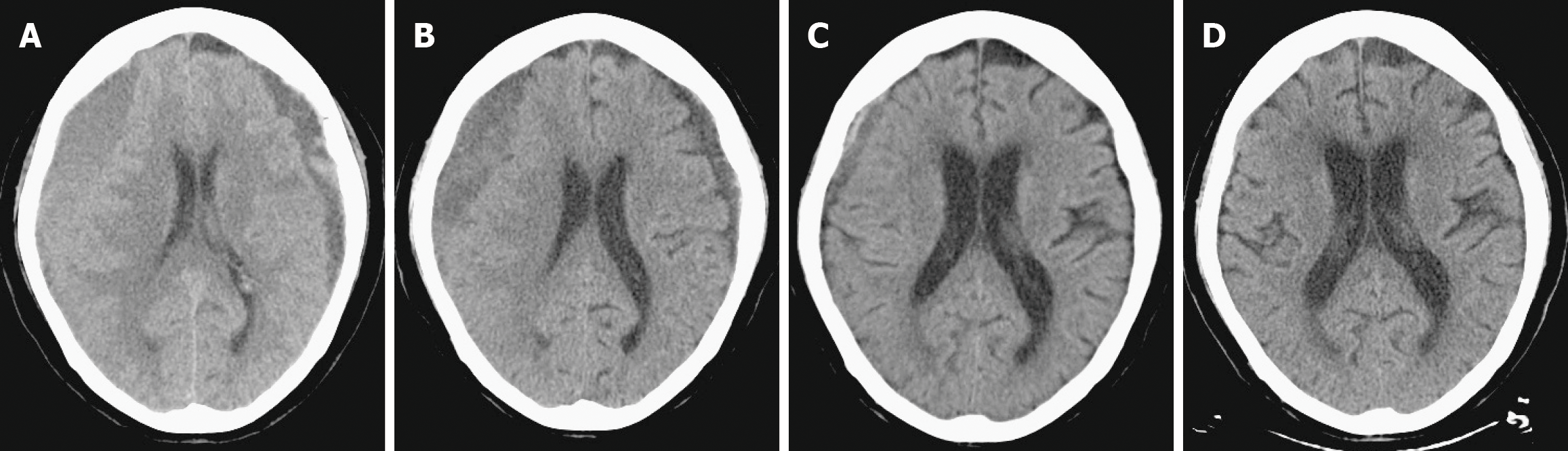
Follow-up CT scans at 1, 2, and 3 months post-procedure showed complete hematoma resolution and significant brain re-expansion (Figure 3). No procedure-related complications occurred.
MMAE is increasingly recognized as a safe and effective minimally invasive treatment for selected patients with cSDH[1,4,5]. Current guidelines recommend its use in patients with mild symptoms or small hematomas (< 10 mm thickness and < 5 mm midline shift), while surgical evacuation remains the first-line therapy for patients with large hematomas or neurological deficits[4,5]. Accordingly, the role of upfront MMAE in patients with significant mass effect is uncertain. Our case demonstrates the successful use of a refined layered embolization technique - designed to enhance distal penetration and proximal sealing - achieving complete hematoma resolution and potentially broadening the applicability of MMAE to high-risk or surgically unsuitable patients.
MMAE has been validated as an effective adjunct to standard therapy, significantly reducing recurrence and treatment failure, without increasing major complications[3-5]. The scanning transmission electron microscopy trial (a randomized controlled trial) further showed that, in the non-surgical standard-treatment subgroup, primary MMAE reduced the rate of treatment failure from 56% to 19%[5]. Additional studies support the favorable safety of standalone MMAE, reporting lower recurrence rates and fewer complications compared with surgery, particularly in older patients, those with multiple comorbidities, or individuals receiving long-term antiplatelet or anticoagulant therapy[1,2]. Nonetheless, patients with larger hematomas are typically directed to surgery, resulting in substantially larger hematomas in the surgical cohorts of current trials. Refinements in embolization technique remain underexplored, and studies evaluating technical modifications to enhance efficacy and safety are lacking. Our proposed layered embolization strategy improves the stability of particle deposition and, more importantly, expands the role of MMAE for patients currently considered unsuitable for standalone embolization.
Liquid embolic agents, such as Onyx and n-butyl cyanoacrylate, provide excellent distal penetration and real-time visualization. However, their clinical application is frequently constrained by high costs, technical complexity, and inherent risks, including dimethyl sulfoxide toxicity, catheter entrapment, and unintended proximal vessel sacrifice resulting from microcatheter reflux[6-8]. In contrast, particle-based agents offer a more cost-effective alternative with a favorable safety profile derived from their biocompatibility and technical simplicity[7,9]. Notably, comparative studies of MMAE for cSDH have demonstrated no significant differences in success, recurrence, or surgical rescue rates when comparing PVA particles to liquid agents[7,8,10].
Nevertheless, particle delivery often encounters the “log-jamming” effect, where inter-particle friction causes premature proximal occlusion, preventing distal saturation[8,11]. While microcatheter distalization has been shown to improve distal deposition and increase complete resolution rates[12,13], our layered embolization technique advances this concept by strategically modulating flow dynamics to enhance distal delivery.
Initially, a low-concentration suspension preserves laminar flow, ensuring that the 400-μm microspheres behave as discrete units. This minimizes inter-particle friction and facilitates delivery to the absolute geometric limits of the pathological vascular bed[11]. A subsequent transition to a standard-concentration suspension promotes proximal stacking, forming a dense embolic column. This sequential layering ensures comprehensive distal saturation and durable devascularization. By optimizing rheological delivery, this technique circumvents the limitations of conventional particle-based agents, achieving the deep penetration typically associated with liquids while maintaining the safety profile of particles.
The selection of 400-μm calibrated microspheres for MMAE optimizes safety by integrating anatomical morphometrics with neovascular pathophysiology[7,11,14]. This diameter provides a geometric barrier against non-target embolization through hazardous extracranial-intracranial anastomoses, specifically those supplying the ophthalmic circulation and the vasa nervorum of cranial nerves[7,15,16]. Because critical pathways - such as meningo-ophthalmic anastomoses (100-300 μm), the central retinal artery (approximately 160 μm), and microvascular nerve supplies - overlap with smaller particle sizes (150-250 μm), 400-μm microspheres are physically precluded from these vessels. This size threshold effectively eliminates the risk of iatrogenic blindness or ischemic neuropathy, such as facial nerve palsy, during potential hemodynamic reversals[7,15,16]. Additionally, by preventing distal penetration into micro-collateral networks (< 100 μm), 400 μm microspheres preserve physiological perfusion to the surrounding healthy tissue and scalp, mitigating the risk of ischemic necrosis[11,14,17,18].
The therapeutic target in cSDH is the neovascular outer membrane, characterized by fragile macrocapillaries (sinusoids) fed by hypertrophied arterial branches[1,14,19]. The 400-μm particle is precisely calibrated to the distal MMA trunks (400-800 μm) and the origins of neovascular feeders (100-400 μm)[15]. By occluding the arteriolar level, these spheres “close the tap” on the vascular nidus, arresting the exudative cycle[9,11,14]. Clinical evidence suggests that 300-500-μm particles maintain comparable efficacy and success rates while reducing the risk of non-target complications compared to smaller 150-250-μm PVA particles[7,9,11].
The paradigm shift toward MMAE for cSDH represents a transition toward targeted, low-stress solutions in personalized geriatric neurosurgery[2,14]. The decision to prioritize MMAE over conventional drainage often lies at the in
Beyond addressing frailty, MMAE reconciles the tension between clinical necessity and patient autonomy. As a minimally invasive alternative, it functions as an “active bridge” for patients who decline conventional surgery due to the perceived invasiveness of a craniotomy. Furthermore, MMAE maintains a superior safety profile regarding antithrombotic management, as it can often be performed without the complete cessation of antiplatelet or anticoagulant therapy[2]. This capacity to bypass the precarious balance between hemorrhagic and embolic risks is frequently the deciding factor in multidisciplinary consultations[2].
Previous studies have shown variable timelines for complete hematoma resolution after MMAE. Catapano et al[12] reported relatively rapid clearance, with 63% of patients achieving resolution at 3 months and 92% at 6 months, whereas Liu et al[22] observed complete resolution in approximately half of patients, typically requiring 5-6 months and up to 1 year in some cases. For burr-hole evacuation, Chang et al[23] reported a median resolution time of 161 days, with only 47.5% of patients achieving full resolution within 6 months. These discrepancies likely reflect differences in patient populations, hematoma characteristics, and follow-up protocols. Nevertheless, across both surgical and endovascular approaches, most patients achieve clearance within 6 months.
Comparative data support the superiority of MMAE over conventional management. Catapano et al[24] reported complete hematoma resolution in 63% of patients at 3-6 months after MMAE compared with 22% after surgery; additionally, a systematic review and meta-analysis by Sattari et al[14] demonstrated higher rates of complete resolution with MMAE than with conventional treatment (61.9% vs 26.4%; relative risk = 2.01, 95% confidence interval: 1.10-3.68, P = 0.02). This advantage reflects the ability of MMAE to interrupt the vascular supply to fragile neovessels within the hematoma membrane, whereas surgical or conservative approaches primarily evacuate the hematoma without ad
In our case, bilateral hematomas resolved progressively following MMAE, with the larger right-sided gradational type requiring a longer clearance period than the smaller left-sided homogeneous collection (Figure 3). Established predictors of clearance include baseline hematoma volume, with larger collections resolving more slowly[13,19,22], and internal architecture, as homogeneous and laminar types resolve faster than trabecular or separated types[17,22]. Advanced age also correlates with delayed resolution, and brain re-expansion is often incomplete in those with baseline cerebral atrophy, underscoring the role of patient-related factors[17,22]. Conversely, the type of embolic material used has not been shown to significantly influence resolution rates[13,17,22].
By ensuring robust distal penetration, our refined layered technique effectively targets the neovascular network sustaining hematoma persistence, achieving full clearance and brain re-expansion within three months, even in large symptomatic cases. However, the inherent limitations of a single case report preclude the direct generalizability of these findings to the broader cSDH population. Given the expanding role of MMAE in cSDH management, further technical refinement is warranted. Larger-scale prospective studies or randomized controlled trials are essential to validate this strategy, optimize patient selection, and evaluate long-term safety and efficacy.
This case demonstrates standalone MMAE using a refined layered embolization technique as a safe and effective al
| 1. | Ironside N, Nguyen C, Do Q, Ugiliweneza B, Chen CJ, Sieg EP, James RF, Ding D. Middle meningeal artery embolization for chronic subdural hematoma: a systematic review and meta-analysis. J Neurointerv Surg. 2021;13:951-957. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 176] [Article Influence: 35.2] [Reference Citation Analysis (6)] |
| 2. | Waqas M, Vakhari K, Weimer PV, Hashmi E, Davies JM, Siddiqui AH. Safety and Effectiveness of Embolization for Chronic Subdural Hematoma: Systematic Review and Case Series. World Neurosurg. 2019;126:228-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 112] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 3. | Davies JM, Knopman J, Mokin M, Hassan AE, Harbaugh RE, Khalessi A, Fiehler J, Gross BA, Grandhi R, Tarpley J, Sivakumar W, Bain M, Crowley RW, Link TW, Fraser JF, Levitt MR, Chen PR, Hanel RA, Bernard JD, Jumaa M, Youssef P, Cress MC, Chaudry MI, Shakir HJ, Lesley WS, Billingsley J, Jones J, Koch MJ, Paul AR, Mack WJ, Osbun JW, Dlouhy K, Grossberg JA, Kellner CP, Sahlein DH, Santarelli J, Schirmer CM, Singer J, Liu JJ, Majjhoo AQ, Wolfe T, Patel NV, Roark C, Siddiqui AH; EMBOLISE Investigators. Adjunctive Middle Meningeal Artery Embolization for Subdural Hematoma. N Engl J Med. 2024;391:1890-1900. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 182] [Article Influence: 91.0] [Reference Citation Analysis (0)] |
| 4. | Liu J, Ni W, Zuo Q, Yang H, Peng Y, Lin Z, Li Z, Wang J, Zhen Y, Luo J, Lin Y, Chen J, Hua X, Lu H, Zhong M, Liu M, Zhang J, Wang Y, Wan J, Li Y, Li T, Mao G, Zhao W, Gao L, Li C, Chen E, Cheng X, Zhang P, Wang Z, Chen L, Zhang Y, Tian B, Shen F, Lei Y, Wu Y, Li Y, Duan G, Xu L, Lv N, Yu J, Xu X, Du Z, Zhang H, Hu J, Li Z, Yuan Q, Zhou Y, Wu G, Zhang L, Gao C, Dai D, Wu X, Zhang Y, Jiang H, Zhao R, Su J, Xu Y, Ospel JM, Majoie CBLM, Goyal M, Li Q, Yang P, Gu Y, Mao Y; MAGIC-MT Investigators. Middle Meningeal Artery Embolization for Nonacute Subdural Hematoma. N Engl J Med. 2024;391:1901-1912. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 141] [Article Influence: 70.5] [Reference Citation Analysis (0)] |
| 5. | Fiorella D, Monteith SJ, Hanel R, Atchie B, Boo S, McTaggart RA, Zauner A, Tjoumakaris S, Barbier C, Benitez R, Spelle L, Pierot L, Hirsch JA, Froehler M, Arthur AS; STEM Investigators. Embolization of the Middle Meningeal Artery for Chronic Subdural Hematoma. N Engl J Med. 2025;392:855-864. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 156] [Article Influence: 156.0] [Reference Citation Analysis (0)] |
| 6. | Sioutas GS, Vivanco-Suarez J, Shekhtman O, Matache IM, Salem MM, Burkhardt JK, Srinivasan VM, Jankowitz BT. Liquid embolic agents for middle meningeal artery embolization in chronic subdural hematoma: Institutional experience with systematic review and meta-analysis. Interv Neuroradiol. 2023;15910199231183132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 22] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 7. | Ellens NR, Schartz D, Kohli G, Rahmani R, Akkipeddi SMK, Mattingly TK, Bhalla T, Bender MT. Safety and efficacy comparison of embolic agents for middle meningeal artery embolization for chronic subdural hematoma. J Cerebrovasc Endovasc Neurosurg. 2024;26:11-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 23] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 8. | Krothapalli N, Patel S, Fayad M, Elmashad A, Killory B, Bruno C, Sussman E, Alberts M, Ollenschleger M, Kureshi I, Mehta T. Outcomes of Particle versus Liquid Embolic Materials Used in Middle Meningeal Artery Embolization for the Treatment of Chronic Subdural Hematoma. World Neurosurg. 2023;173:e27-e36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 9. | Abdollahifard S, Farrokhi A, Yousefi O, Valibeygi A, Azami P, Mowla A. Particle embolic agents for embolization of middle meningeal artery in the treatment of chronic subdural hematoma: A systematic review and meta-analysis. Interv Neuroradiol. 2024;30:94-104. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 10. | Shehabeldin M, Amllay A, Jabre R, Chen CJ, Schunemann V, Herial NA, Gooch MR, Mackenzie L, Choe H, Tjoumakaris S, Rosenwasser RH, Jabbour P, Kozak O. Onyx Versus Particles for Middle Meningeal Artery Embolization in Chronic Subdural Hematoma. Neurosurgery. 2023;92:979-985. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 33] [Reference Citation Analysis (0)] |
| 11. | Sadasivan C, Dashti N, Marfoglio S, Fiorella D. In vitro comparison of middle meningeal artery embolization with Squid liquid embolic agent and Contour polyvinyl alcohol particles. J Neurointerv Surg. 2024;16:280-284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 12. | Catapano JS, Ducruet AF, Srinivasan VM, Rumalla K, Nguyen CL, Rutledge C, Cole TS, Baranoski JF, Lawton MT, Jadhav AP, Albuquerque FC. Radiographic clearance of chronic subdural hematomas after middle meningeal artery embolization. J Neurointerv Surg. 2022;14:1279-1283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 35] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 13. | Ma L, Hoz SS, Doheim MF, Fadhil A, Sultany A, Al-Bayati AR, Nogueira RG, Lang MJ, Gross BA. Impact of Embolisate Penetration, Type, and Technique on Results After Standalone Middle Meningeal Artery Embolization for Chronic Subdural Hematoma. Neurosurgery. 2024;95:1395-1406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 14. | Sattari SA, Yang W, Shahbandi A, Feghali J, Lee RP, Xu R, Jackson C, Gonzalez LF, Tamargo RJ, Huang J, Caplan JM. Middle Meningeal Artery Embolization Versus Conventional Management for Patients With Chronic Subdural Hematoma: A Systematic Review and Meta-Analysis. Neurosurgery. 2023;92:1142-1154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 68] [Article Influence: 22.7] [Reference Citation Analysis (0)] |
| 15. | Geibprasert S, Pongpech S, Armstrong D, Krings T. Dangerous extracranial-intracranial anastomoses and supply to the cranial nerves: vessels the neurointerventionalist needs to know. AJNR Am J Neuroradiol. 2009;30:1459-1468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 221] [Cited by in RCA: 263] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 16. | Hayreh SS. The blood supply of the optic nerve head and the evaluation of it - myth and reality. Prog Retin Eye Res. 2001;20:563-593. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 255] [Cited by in RCA: 264] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 17. | Uttam BK, Yuanyuan L, Bizhan A, Thorsten FR, Mazhar K, Marco C, Dheeraj G. Short-term follow-up pilot study of sole middle meningeal artery embolization for chronic subdural hematoma: influence of internal architecture on the radiological outcomes. Neuroradiology. 2023;65:1143-1153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 18. | Campos JK, Meyer BM, Zarrin DA, Khan MW, Collard de Beaufort JC, Amin G, Avery MB, Golshani K, Beaty NB, Bender MT, Colby GP, Lin LM, Coon AL. Immediate procedural safety of adjunctive proximal coil occlusion in middle meningeal artery embolization for chronic subdural hematomas: Experience in 137 cases. Interv Neuroradiol. 2024;15910199231224003. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 19. | Khorasanizadeh M, Chang YM, Enriquez-Marulanda A, Mizuhashi S, Salem MM, Gomez-Paz S, Siddiq F, Kan P, Moore J, Ogilvy CS, Thomas AJ. Morphological changes in chronic subdural hematomas following upfront middle meningeal artery embolization: sequence, timing, and association with outcomes. J Neurosurg. 2022;137:235-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 20. | Zaki PG, Bolger J, Rogowski B, Busch N, Elhamdani S, Jeong S, Li J, Leonardo J, Williamson R, Yu A, Shepard MJ. The Utility of the 5 Factor Modified Frailty Index in Outcome Prediction for Patients with Chronic Subdural Hematoma Treated with Surgical Drainage. World Neurosurg. 2023;179:e328-e341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 21. | Zolnourian A, Manivannan S, Edwards B, Chua A, Arora M, Akhigbe T, Durnford A, Hempenstall J, Nader-Sepahi A, Bulters D, Sadek AR. Factors affecting outcomes following burr hole drainage of chronic subdural hematoma: a single-center retrospective study. J Neurosurg. 2025;142:1606-1615. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 22. | Liu Z, Wang Y, Tang T, Zhang Y, Sun Y, Kuang X, Wei T, Zhou L, Peng A, Cao D, Hongsheng W, Qi W, Chenyi W, Shan Q. Time and Influencing Factors to Chronic Subdural Hematoma Resolution After Middle Meningeal Artery Embolization. World Neurosurg. 2023;179:e6-e14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 13] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 23. | Chang CL, Sim JL, Delgardo MW, Ruan DT, Connolly ES Jr. Predicting Chronic Subdural Hematoma Resolution and Time to Resolution Following Surgical Evacuation. Front Neurol. 2020;11:677. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 24. | Catapano JS, Ducruet AF, Nguyen CL, Cole TS, Baranoski JF, Majmundar N, Wilkinson DA, Fredrickson VL, Cavalcanti DD, Lawton MT, Albuquerque FC. A propensity-adjusted comparison of middle meningeal artery embolization versus conventional therapy for chronic subdural hematomas. J Neurosurg. 2021;135:1208-1213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/