Published online May 16, 2026. doi: 10.12998/wjcc.v14.i14.120126
Revised: March 10, 2026
Accepted: April 2, 2026
Published online: May 16, 2026
Processing time: 67 Days and 14.5 Hours
Statin-associated necrotizing autoimmune myopathy (NAM) is an immune-mediated myopathy characterized by progressive weakness, markedly elevated creatine kinase (CK), and antibodies to 3-hydroxy-3-methylglutaryl-coenzyme A reductase (HMGCR). Unlike toxic statin myopathy, symptoms and CK elevation persist despite statin discontinuation and typically require immunosuppressive therapy. Although many patients improve with corticosteroids and intravenous immunoglobulin (IVIG), severe or refractory cases can occur with any statin ex
An 83-year-old woman with a history of bladder, breast, and lung cancers, cor
Persistent CK elevation after statin withdrawal warrants anti-HMGCR antibody testing and early immunosuppression. Delayed recognition may lead to disability or death.
Core Tip: Statin-associated necrotizing autoimmune myopathy (NAM) is a rare immune-mediated myopathy defined by progressive weakness, marked creatine kinase elevation, and persistence of disease activity despite statin withdrawal, most often associated with anti-3-hydroxy-3-methylglutaryl–coenzyme A reductase antibodies. Many patients respond to corticosteroids and intravenous immunoglobulin (IVIG), but refractory disease can occur, particularly when diagnosis is delayed or when severe systemic complications develop. We report an elderly woman receiving long-term rosuvastatin who developed persistent rhabdomyolysis with dialysis-dependent renal failure and died despite high-dose corticosteroids, IVIG, and rituximab. This case underscores the need for early antibody testing and prompt escalation of therapy when statin-associated NAM is suspected, to prevent severe outcomes.
- Citation: Thiriveedi M, Patel H, Reddy PJ. Refractory rosuvastatin-associated necrotizing autoimmune myopathy: A case report and review of literature. World J Clin Cases 2026; 14(14): 120126
- URL: https://www.wjgnet.com/2307-8960/full/v14/i14/120126.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i14.120126
Statin-associated necrotizing autoimmune myopathy (NAM), also referred to as anti-3-hydroxy-3-methylglu
This condition is strongly associated with autoantibodies directed against HMGCR, the pharmacologic target of statins, and anti-HMGCR antibodies are highly specific for the diagnosis of NAM in the appropriate clinical context[16-18]. Proposed mechanisms include statin-induced upregulation of HMGCR expression in regenerating muscle fibers and loss of immune tolerance, resulting in sustained antibody-mediated muscle injury even after the statin is stopped[7-10,16]. Predisposing factors are not clearly defined but may include older age, statin exposure, genetic susceptibility, and immune dysregulation in the setting of comorbidities[16-19].
Diagnostic evaluation typically includes CK measurement, myositis autoantibody testing, including anti-HMGCR, and consideration of electromyography (EMG), magnetic resonance imaging (MRI), and muscle biopsy when the diagnosis is uncertain or when alternative etiologies are suspected[10-12,20,21]. Treatment generally requires early immunosuppression, most commonly high-dose corticosteroids combined with steroid-sparing agents and/or intravenous immunoglobulin (IVIG); rituximab or other agents may be considered in refractory disease[22-26]. While many published cases improve with timely therapy, severe presentations complicated by rhabdomyolysis and acute kidney injury have also been reported[27-30].
Here, we describe a fulminant case of rosuvastatin-associated NAM in a frail elderly woman with complex comor
Progressive fatigue, generalized weakness, shortness of breath, and inability to ambulate following a fall.
An 83-year-old Caucasian woman presented with several days of worsening fatigue, shortness of breath, and profound generalized weakness, culminating in a fall in her bathroom. She reported remaining immobilized on the floor for approximately ten hours before obtaining assistance. She also reported black-brown stools for several days and increasing difficulty performing activities of daily living. The patient was admitted to the hospital for further management.
Her medical history included hyperlipidemia treated with rosuvastatin 40 mg daily for two years, chronic kidney disease stage 3b, coronary artery disease, bladder cancer status post cystectomy with ileal conduit (2013), right breast cancer status post mastectomy (1992), non-small cell lung cancer treated with radiation, prior deep vein thrombosis, right carotid artery stenosis status post endarterectomy (2023), and vitamin D deficiency.
She was a former smoker and denied alcohol or illicit drug use. No known drug allergies were reported. No history of cancer or autoimmune diseases in the family.
On physical exam, the vitals were as follows: Temperature 98.2 F; blood pressure 130/82 mmHg; heart rate 108 beats per minute; respiratory rate 16 breaths per minutes. She appeared pale and fatigued. Cardiorespiratory examination was significant for sinus tachycardia and mild basilar crackles. Neurologic examination revealed generalized weakness without focal deficits. Trace bilateral lower-extremity edema was present, greater on the left side. Ileal conduit was present and functioning.
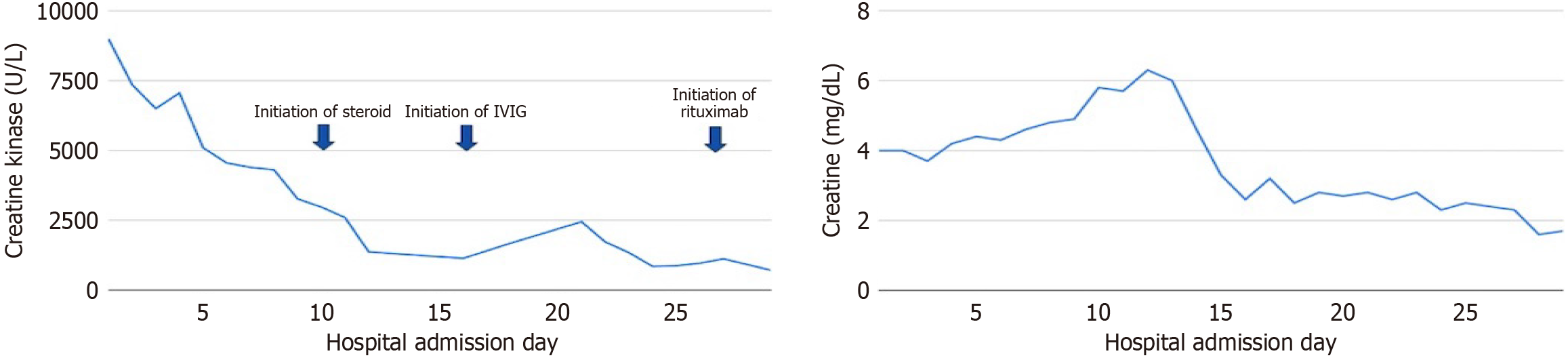
Laboratory data were significant for anemia (hemoglobin 8.1 g/dL), acute kidney injury (creatinine 4.0 mg/dL), and elevated liver enzymes (Table 1). Urinalysis demonstrated heme-positive urine with absent erythrocytes on microscopy, a pattern consistent with pigment nephropathy from myoglobin in the setting of rhabdomyolysis; formal urinary myoglobin testing was not obtained. The CK level was markedly elevated at 9012 U/L. Autoimmune testing and myositis panel were negative except for elevated anti-HMGCR antibodies at high titers (Table 1). Anti-signal recognition particle (anti-SRP) antibody testing was not performed. Figure 1 illustrates the temporal trends in CK and creatinine levels during hospitalization in relation to therapeutic interventions.
| Laboratory test | Result | Reference range |
| White blood cell count (cells/µL) | 12.92 × 10³ | 4.8-10.8 × 10³ |
| Hemoglobin (g/dL) | 8.1 | 12-16 |
| Hematocrit | 25.8% | 37%-47% |
| Mean corpuscular volume (fL) | 105.7 | 80-100 |
| Platelet count (cells/µL) | 348 × 10³ | 130-400 × 10³ |
| Sodium (mmol/L) | 138 | 136-145 |
| Potassium (mmol/L) | 3.7 | 3.5-5.1 |
| Chloride (mmol/L) | 107 | 98-107 |
| Bicarbonate (mEq/L) | 15 | 22-29 |
| Blood urea nitrogen (mg/dL) | 56 | 8-22 |
| Creatinine (mg/dL) | 4.0 | 0.7-1.2 |
| Glucose (mg/dL) | 91 | 70-104 |
| Calcium (mg/dL) | 8.3 | 8.8-10.2 |
| Total bilirubin (mg/dL) | 0.24 | 0.20-1.0 |
| Aspartate transaminase (U/L) | 365 | 10-30 |
| Alanine transaminase (U/L) | 280 | 10-36 |
| Alkaline phosphatase (U/L) | 74 | 32-104 |
| Creatine kinase (U/L) | 9012 | 24-173 |
| Erythrocyte sedimentation rate (mm/hour) | 23 | 0-20 |
| Urinalysis | Heme-positive urine with absent erythrocytes on microscopy; leukocytes present; 4+ bacteria | |
| Jo-1 antibody (U) | Negative | < 1.0 |
| SSA/Ro antibody (U) | Negative | < 1.0 |
| SSB/La antibody (U) | Negative | < 1.0 |
| Smith antibody (U) | Negative | < 1.0 |
| RNP antibody (U) | Negative | < 1.0 |
| Scl-70 antibody (U) | Negative | < 1.0 |
| Antinuclear antibody (U) | Negative | ≤ 1.0 |
| Cyclic citrullinated peptide antibody (U) | Negative | < 20.0 |
| HMG-CoA reductase antibody (CU) | Positive (348.8) | < 20.0 |
Chest radiography showed bilateral pulmonary edema. Lower-extremity venous Doppler ultrasound showed no evidence of deep vein thrombosis. Transthoracic echocardiography demonstrated preserved left ventricular ejection fraction. Computed tomography of the cervical and lumbar spine showed no evidence of metastatic disease. MRI of the brain and skeletal muscle was attempted; however, it could not be completed due to the patient’s claustrophobia.
Based on the clinical presentation of progressive weakness, markedly elevated CK levels despite statin discontinuation, and strongly positive anti-HMGCR antibodies with otherwise negative myositis serologies, the patient was diagnosed with IMNM, likely statin-associated NAM. The diagnosis was supported by a lack of response to supportive measures for presumed traumatic rhabdomyolysis and the progression of muscle weakness and renal failure despite appropriate measures. Muscle biopsy was not pursued, given the characteristic clinical phenotype and confirmatory serologic findings.
Upon admission, rosuvastatin was discontinued, and the patient was initially treated for presumed traumatic rhabdomyolysis with intravenous (IV) fluids and bicarbonate infusion, along with IV ceftriaxone for possible urinary tract infection, and proton pump inhibitor therapy. Due to persistent CK elevation and progressive weakness, immunosuppressive therapy was initiated after confirmation of NAM. She received high-dose IV methylprednisolone (1 g daily for 3 days starting on hospital day 10), followed by IVIG (2 g/kg in divided doses beginning on hospital day 16). Renal function continued to deteriorate, necessitating vascular catheter placement and initiation of hemodialysis. Given refractory disease, rituximab 500 mg IV was administered on hospital day 28 as salvage immunotherapy. A standard two-dose regimen was planned; however, further dosing was not pursued due to clinical deterioration and subsequent transition to comfort-focused care before additional treatment could be administered.
Despite escalation to triple immunotherapy with corticosteroids, IVIG, and rituximab, the patient demonstrated no meaningful clinical or functional recovery. Although the CK levels transiently decreased, she remained bed-bound with severe diffuse myalgias and dialysis dependence. Her hospital course was further complicated by intermittent atrial fibrillation during dialysis and intractable pain requiring escalating narcotic therapy. Given worsening pain, declining performance status, and lack of response to treatment, the palliative team was consulted. After discussion with her family, the patient elected a do-not-resuscitate status and comfort-focused care. Dialysis and disease-directed therapies were discontinued, and she died peacefully on hospital day 32.
Statin-associated NAM is a distinct immune-mediated myopathy that differs fundamentally from self-limited toxic statin myopathy. Whereas toxic statin myopathy is typically dose-related, may present with myalgias with normal or mildly elevated CK, and improves after statin discontinuation, NAM is characterized by progressive weakness, marked CK elevation (often thousands), and persistence of disease activity despite withdrawal of the statin[7-11]. This distinction is clinically crucial because NAM generally requires immunosuppressive therapy, and delayed recognition may result in prolonged disability, severe systemic complications, and, in rare cases, death.
Statin-associated NAM can occur at any time after initiation of therapy[17]. It is strongly linked to autoantibodies against HMGCR, the enzyme targeted by statins. Current models propose that statins upregulate HMGCR expression in skeletal muscle, particularly in regenerating fibers. In genetically predisposed individuals, this triggers loss of immune tolerance and sustained autoimmunity[7-10,16]. Once established, the autoimmune process may continue even after discontinuation of the statin because regenerating muscle continues to express HMGCR, creating a self-perpetuating cycle of injury and repair[7-10]. Genetic predisposition is supported by the association of anti-HMGCR myopathy with the HLA class II allele DRB1*11:01[31].
IMNM can occur with any statin, and the severity is variable. While pharmacokinetic differences between hydrophilic (rosuvastatin and pravastatin) and lipophilic (atorvastatin and simvastatin) statins have been discussed in relation to toxic statin myopathy, immune-mediated NAM is fundamentally driven by loss of immune tolerance and autoantibody-mediated injury rather than direct myotoxicity alone[7-10].
Clinically, patients often present with subacute progressive proximal weakness with or without myalgia; dysphagia, neck flexor weakness, and, in severe cases, respiratory muscle involvement have been described[10-14]. Laboratory findings typically show markedly elevated CK and transaminases (reflecting muscle injury rather than hepatic disease), and EMG may demonstrate an irritable myopathy pattern[10-14]. MRI may show muscle edema and can help localize biopsy targets, while muscle biopsy classically reveals necrotic and regenerating myofibers with relatively sparse inflammatory infiltrates compared with polymyositis or dermatomyositis[13,14,20,21]. However, in a patient with compatible clinical features and strongly positive anti-HMGCR antibodies, biopsy may not be required, particularly when it would not alter management[20,21].
The key diagnostic clue is a patient with ongoing weakness and persistent CK elevation despite discontinuation of statin therapy. In such cases, anti-HMGCR antibody testing should be obtained promptly. The present case illustrates several common diagnostic pitfalls. First, rhabdomyolysis following a fall and prolonged immobilization can plausibly explain an elevated CK. However, in uncomplicated traumatic rhabdomyolysis, CK generally declines with supportive care and removal of inciting factors. Second, a urine dipstick “blood” with absent erythrocytes on microscopy suggests pigment nephropathy due to myoglobin, supporting rhabdomyolysis as a major contributor to renal injury; nevertheless, it does not distinguish traumatic rhabdomyolysis from immune-mediated ongoing muscle necrosis. Thus, persistent CK elevation accompanied by progressive weakness after statin withdrawal should prompt evaluation for statin-associated NAM. Published studies emphasize that delayed recognition is associated with prolonged disability and may increase the likelihood of refractory disease[10-13,18].
There is no single universally accepted regimen for IMNM, but most expert approaches support early aggressive immunosuppression, often combining high-dose corticosteroids with IVIG and a steroid-sparing agent. IVIG is com
In the present case, the patient received methylprednisolone, IVIG, and a dose of rituximab, yet did not achieve meaningful clinical recovery. Several mechanisms may explain treatment refractoriness. First, the patient presented with severe systemic involvement, including dialysis-requiring renal failure and a substantial comorbidity burden, which may reflect advanced disease at the time immunotherapy was initiated. Second, rituximab often requires weeks to exert a clinical effect; administration late in the course, followed by rapid clinical decline, may preclude adequate assessment of response. Third, frailty, malnutrition, prolonged immobilization, and metabolic derangements can blunt functional recovery even if immune activity is partially controlled. Finally, persistent antigenic stimulation and ongoing muscle regeneration may sustain autoimmunity, requiring prolonged therapy and careful maintenance strategies, which were not feasible in this rapidly deteriorating clinical context.
Multiple case reports and series describe rosuvastatin-associated NAM confirmed by anti-HMGCR antibodies and/or biopsy with varying responses to therapy. Several reported patients improved with corticosteroids and IVIG-based regimens, although some required prolonged rehabilitation due to persistent muscle weakness[20,27]. A dermatomyositis-like rash associated with rosuvastatin exposure and high anti-HMGCR antibody levels has also been reported, highlighting potential diagnostic pitfalls and the importance of considering IMNM in atypical clinical presentations[32]. Another reported rosuvastatin-associated case with anti-HMGCR positivity and biopsy-confirmed muscle necrosis demonstrated relapse with hypoventilation requiring IVIG plus additional immunosuppressive therapy, illustrating the potential severity and relapsing nature of the disease[28].
Cases presenting with rhabdomyolysis complicated by acute kidney injury requiring hemodialysis have also been reported in association with rosuvastatin use, with patients demonstrating renal recovery following supportive care and discontinuation of the statin[29,30]. In contrast, the present case differs in three clinically important ways compared with previously reported rosuvastatin-associated cases: (1) Persistent CK elevation and progressive weakness despite statin withdrawal; (2) Dialysis dependence that did not resolve; and (3) Lack of meaningful clinical response to combined immunosuppressive therapy resulting in death. These findings highlight the heterogeneous clinical course of statin-associated NAM and suggest that advanced age, comorbidities, and delayed recognition may contribute to poorer outcomes.
A pharmacovigilance analysis of statin-associated NAM reports in the global drug safety database (VigiBase) identified atorvastatin as the most frequently reported statin, although substantial reporting with rosuvastatin was also observed. The authors noted that a true class effect cannot be excluded and that reporting frequency may be influenced by prescribing patterns and clinician awareness[33]. A more recent retrospective multi-site cohort study evaluating the incidence of IMNM among statin users further supports that this condition is rare but well recognized across different statin exposures. Importantly, many patients did not achieve normalization of CK levels or muscle strength at follow-up, reinforcing the potential for refractory disease despite treatment[34].
Although most patients improve with immunosuppressive therapy, fatal outcomes have been reported in statin-associated NAM. Mortality is often related to secondary complications such as severe dysphagia with aspiration or respiratory failure. An autopsy-confirmed report described rapid clinical deterioration due to bulbar involvement in atorvastatin-associated disease, and another report documented death related to persistent dysphagia and aspiration pneumonia in a patient receiving simvastatin[35,36]. Fatal outcomes specifically associated with rosuvastatin appear to be rarely reported. A comparison of previously reported cases of statin-associated NAM is summarized in Table 2, illu
| Ref. | Age/sex | Statin used | Diagnostic criteria | Therapy used | Outcome |
| Grable-Esposito et al[3] | 74/female | Atorvastatin | Elevated CK, muscle biopsy | Prednisone, methotrexate, mycophenolate, IVIG | Improved |
| Fink et al[20] | 65/female | Rosuvastatin | Elevated CK, MRI, anti-HMGCR antibodies | Prednisone, IVIG | Improved weakness and CK levels; required SNF |
| Gupta et al[24] | 61/female | Pravastatin | Elevated CK, anti-HMGCR antibodies, MRI, muscle biopsy | Prednisone, azathioprine, rituximab | Improved |
| Srinivasan et al[25] | 64/male | Atorvastatin, rosuvastatin | Elevated CK, EMG, muscle biopsy, anti-HMGCR antibodies | Prednisone, IVIG | Improved |
| Luong et al[27] | 55/male | Rosuvastatin | Elevated CK, anti-HMGCR antibodies, MRI, muscle biopsy | Prednisone, mycophenolate, IVIG | Improved |
| Yamamura et al[28] | 59/female | Rosuvastatin | Anti-HMGCR antibodies, elevated CK, MRI, muscle biopsy | Methylprednisolone pulse therapy, prednisolone | Improved |
| Chitralli et al[29] | 63/male | Atorvastatin, rosuvastatin | Elevated CK, EMG | Hemodialysis | Improved |
| Petreski et al[30] | 59/female | Rosuvastatin | EMG, muscle biopsy | Hemodialysis | Improved |
| Merlant et al[32] | 61/female | Rosuvastatin | Elevated CK, muscle biopsy, anti-HMGCR antibodies | Immunosuppressive therapy | Improved |
| Mirlesse et al[35] | 77/male | Atorvastatin | Elevated CK, MRI, anti-HMGCR antibodies | Methylprednisolone, IVIG | Died |
| Abusharar et al[36] | 73/male | Simvastatin | Elevated CK, muscle biopsy, anti-HMGCR antibodies | Prednisone, methylprednisolone, IVIG, methotrexate, azathioprine | Died |
This report has several limitations. Anti-SRP antibody testing was not performed; although anti-HMGCR and anti-SRP define distinct subtypes of IMNM, they are rarely co-expressed[10,16]. MRI of skeletal muscle and brain was attempted, but could not be completed due to claustrophobia, limiting additional supportive imaging data. Muscle biopsy was not obtained; while a biopsy can help confirm necrotizing myopathy and exclude alternative diagnoses, a strongly positive anti-HMGCR result in a compatible clinical syndrome supported the diagnosis and guided management. Finally, specific urinary myoglobin testing was not obtained, although urine heme positivity with absent erythrocytes supported pigment nephropathy in the setting of rhabdomyolysis.
From a clinical perspective, this case underscores several important lessons for practicing clinicians. First, persistent elevation of CK and progressive weakness after discontinuation of statin therapy should prompt early evaluation for IMNM, particularly with anti-HMGCR antibody testing. Second, although statin-associated NAM is rare, it should be considered in patients with unexplained rhabdomyolysis or prolonged recovery following presumed traumatic muscle injury. Third, early recognition and timely initiation of immunosuppressive therapy are critical, as delayed diagnosis may allow ongoing immune-mediated muscle injury and systemic complications to develop. Awareness of this entity and prompt diagnostic evaluation may improve outcomes and help prevent irreversible complications in affected patients.
Statin-associated NAM is a rare but potentially life-threatening complication of statin therapy that may persist despite drug discontinuation. This case highlights a fulminant, treatment-refractory presentation associated with rosuvastatin, resulting in persistent rhabdomyolysis, dialysis-dependent renal failure, and death despite aggressive immunosuppressive therapy. Persistent CK elevation after statin withdrawal should prompt early evaluation for anti-HMGCR antibodies, as delayed diagnosis may lead to irreversible organ damage and poor outcomes.
| 1. | Phillips PS, Haas RH, Bannykh S, Hathaway S, Gray NL, Kimura BJ, Vladutiu GD, England JD; Scripps Mercy Clinical Research Center. Statin-associated myopathy with normal creatine kinase levels. Ann Intern Med. 2002;137:581-585. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 403] [Cited by in RCA: 366] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 2. | Needham M, Fabian V, Knezevic W, Panegyres P, Zilko P, Mastaglia FL. Progressive myopathy with up-regulation of MHC-I associated with statin therapy. Neuromuscul Disord. 2007;17:194-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 168] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 3. | Grable-Esposito P, Katzberg HD, Greenberg SA, Srinivasan J, Katz J, Amato AA. Immune-mediated necrotizing myopathy associated with statins. Muscle Nerve. 2010;41:185-190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 230] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 4. | Dalakas MC. Inflammatory muscle diseases. N Engl J Med. 2015;372:1734-1747. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 394] [Cited by in RCA: 494] [Article Influence: 44.9] [Reference Citation Analysis (11)] |
| 5. | Vinci P, Panizon E, Tosoni LM, Cerrato C, Pellicori F, Mearelli F, Biasinutto C, Fiotti N, Di Girolamo FG, Biolo G. Statin-Associated Myopathy: Emphasis on Mechanisms and Targeted Therapy. Int J Mol Sci. 2021;22:11687. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 112] [Article Influence: 22.4] [Reference Citation Analysis (0)] |
| 6. | Mammen AL, Chung T, Christopher-Stine L, Rosen P, Rosen A, Doering KR, Casciola-Rosen LA. Autoantibodies against 3-hydroxy-3-methylglutaryl-coenzyme A reductase in patients with statin-associated autoimmune myopathy. Arthritis Rheum. 2011;63:713-721. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 564] [Cited by in RCA: 504] [Article Influence: 33.6] [Reference Citation Analysis (0)] |
| 7. | Mammen AL. Statin-Associated Autoimmune Myopathy. N Engl J Med. 2016;374:664-669. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 222] [Cited by in RCA: 261] [Article Influence: 26.1] [Reference Citation Analysis (0)] |
| 8. | Mohassel P, Mammen AL. Anti-HMGCR Myopathy. J Neuromuscul Dis. 2018;5:11-20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 94] [Cited by in RCA: 113] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 9. | Kurashige T. Anti-HMGCR myopathy: clinical and histopathological features, and prognosis. Curr Opin Rheumatol. 2021;33:554-562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 10. | Allenbach Y, Mammen AL, Benveniste O, Stenzel W; Immune-Mediated Necrotizing Myopathies Working Group. 224th ENMC International Workshop:: Clinico-sero-pathological classification of immune-mediated necrotizing myopathies Zandvoort, The Netherlands, 14-16 October 2016. Neuromuscul Disord. 2018;28:87-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 429] [Article Influence: 47.7] [Reference Citation Analysis (0)] |
| 11. | Stenzel W, Goebel HH, Aronica E. Review: immune-mediated necrotizing myopathies--a heterogeneous group of diseases with specific myopathological features. Neuropathol Appl Neurobiol. 2012;38:632-646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 68] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 12. | Selva-O'Callaghan A, Pinal-Fernandez I, Trallero-Araguás E, Milisenda JC, Grau-Junyent JM, Mammen AL. Classification and management of adult inflammatory myopathies. Lancet Neurol. 2018;17:816-828. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 363] [Cited by in RCA: 302] [Article Influence: 37.8] [Reference Citation Analysis (0)] |
| 13. | Yongchairat K, Tanboon J, Waisayarat J, Narongroeknawin P, Chevaisrakul P, Dejthevaporn C, Katchamart W. Clinical spectrums and outcomes of necrotizing autoimmune myopathy versus other idiopathic inflammatory myopathies: a multicenter case-control study. Clin Rheumatol. 2019;38:3459-3469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 14. | Mammen AL. Autoimmune Myopathies. Continuum (Minneap Minn). 2016;22:1852-1870. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 15. | Takami A, Terashima G, Tajima T, Yamashita K, Igarashi A. Trends in Reports of Statin-Associated Immune-Mediated Necrotizing Myopathy Using the Japanese Adverse Drug Event Report Database. Drugs Real World Outcomes. 2025;12:145-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 16. | Mohassel P, Mammen AL. Statin-associated autoimmune myopathy and anti-HMGCR autoantibodies. Muscle Nerve. 2013;48:477-483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 108] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 17. | Gawey B, Tannu M, Rim J, Sperling L, Henry TL. Statin-Induced Necrotizing Autoimmune Myopathy. JACC Case Rep. 2020;2:440-443. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 13] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 18. | Tiniakou E, Pinal-Fernandez I, Lloyd TE, Albayda J, Paik J, Werner JL, Parks CA, Casciola-Rosen L, Christopher-Stine L, Mammen AL. More severe disease and slower recovery in younger patients with anti-3-hydroxy-3-methylglutaryl-coenzyme A reductase-associated autoimmune myopathy. Rheumatology (Oxford). 2017;56:787-794. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 76] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 19. | Wang JX, Wilkinson M, Oldmeadow C, Limaye V, Major G. Outcome predictors of immune-mediated necrotizing myopathy-a retrospective, multicentre study. Rheumatology (Oxford). 2022;61:3824-3829. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 14] [Reference Citation Analysis (1)] |
| 20. | Fink NS, Abubshait L, Deshisky A. Anti-HMGCR (Hydroxy-3-Methylglutaryl-CoA Reductase) Myopathy: A Rare Cause of Proximal Muscle Weakness. Cureus. 2024;16:e61094. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 21. | Pinal-Fernandez I, Casal-Dominguez M, Mammen AL. Immune-Mediated Necrotizing Myopathy. Curr Rheumatol Rep. 2018;20:21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 204] [Article Influence: 25.5] [Reference Citation Analysis (0)] |
| 22. | Landon-Cardinal O, Allenbach Y, Soulages A, Rigolet A, Hervier B, Champtiaux N, Monzani Q, Solé G, Benveniste O. Rituximab in the Treatment of Refractory Anti-HMGCR Immune-mediated Necrotizing Myopathy. J Rheumatol. 2019;46:623-627. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 40] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 23. | Weeding E, Tiniakou E. Therapeutic management of immune-mediated necrotizing myositis. Curr Treatm Opt Rheumatol. 2021;7:150-160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 28] [Reference Citation Analysis (0)] |
| 24. | Gupta S, Rakhra A, Thallapally V, Nahas J. Rituximab use for refractory anti-HMGCR immune-mediated necrotizing myopathy: A case report. Intractable Rare Dis Res. 2021;10:122-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 25. | Srinivasan V, Prabu S, Sfeir JG, Muthusamy K. Statin-Induced Necrotizing Autoimmune Myopathy: Diagnosis and Treatment Approach. JCEM Case Rep. 2024;2:luae227. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 26. | Suh J, Amato AA. Effectiveness and Safety of IVIG for the Treatment of HMGCR Immune-Mediated Necrotizing Myopathy. Muscle Nerve. 2025;71:392-397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 27. | Luong MT, Danahy P, Ho GY. Spontaneous Development of Immune-Mediated Necrotizing Myopathy (IMNM) After Chronic Use of Statins: A Case Report. Cureus. 2023;15:e42266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 28. | Yamamura Y, Matsumoto Y, Tadokoro K, Ohta Y, Sato K, Yamashita T, Yamamura M, Sada KE, Abe K, Wada J. Recovery from hypoxemia and Hypercapnia following noninvasive pressure support ventilation in a patient with statin-associated necrotizing myopathy: a case report. BMC Pulm Med. 2020;20:156. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 29. | Chitralli D, Raheja R, Br K. Clinical Rhabdomyolysis With Acute Kidney Injury Secondary to High-Intensity Rosuvastatin Use: A Case Report. Cureus. 2020;12:e10932. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 30. | Petreski T, Piko N, Petrijan T, Dvoršak B, Hojs R, Bevc S. Statin-Associated Necrotizing Myopathy Leading to Acute Kidney Injury: A Case Report. Case Rep Nephrol Dial. 2021;11:129-135. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 31. | Mammen AL, Gaudet D, Brisson D, Christopher-Stine L, Lloyd TE, Leffell MS, Zachary AA. Increased frequency of DRB1*11:01 in anti-hydroxymethylglutaryl-coenzyme A reductase-associated autoimmune myopathy. Arthritis Care Res (Hoboken). 2012;64:1233-1237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 84] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 32. | Merlant M, Fite C, Kottler D, Maisonobe L, Dossier A, Deschamps L, Descamps V. [Dermatomyositis-like syndrome revealing statin-induced necrotizing autoimmune myopathy with anti-HMGCR antibodies]. Ann Dermatol Venereol. 2019;146:550-556. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 33. | Essers D, Schäublin M, Kullak-Ublick GA, Weiler S. Statin-associated immune-mediated necrotizing myopathy: a retrospective analysis of individual case safety reports from VigiBase. Eur J Clin Pharmacol. 2019;75:409-416. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 34. | Khoo T, Tan E, Limaye V, Gunawardena H, Sadler R, Lamb JA, Lyu X, Brusch A, Needham M, Austin K, Bahadori A, Buch MH, Tomaszewski M, Lilleker JB, Chinoy H. The incidence of anti-HMGCR immune-mediated necrotizing myopathy: an Australian and UK retrospective multi-site cohort study. Rheumatology (Oxford). 2025;64:4995-5003. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 35. | Mirlesse N, Egervari K, Bornand A, Lecluse J, Lobrinus JA, Scheffler M, Serratrice C, Prendki V, Cuvelier C. Statin-induced autoimmune necrotizing myopathy with pharyngeal muscles involvement. Age Ageing. 2020;49:883-884. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 36. | Abusharar SP, Moku P, Banks S, Khalid FM, Specht CS, Polimera HV. Immune mediated necrotizing myopathy: A rare complication of statin therapy. Clin Pract. 2020;10:1248. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |