Published online May 6, 2026. doi: 10.12998/wjcc.v14.i13.117947
Revised: February 14, 2026
Accepted: April 1, 2026
Published online: May 6, 2026
Processing time: 125 Days and 11.8 Hours
Methylene blue (MB) is being investigated as a useful adjunct in the management of vasoplegic and septic shock due to its targeted inhibition of the nitric oxide-soluble guanylate cyclase pathway. Early studies demonstrated improved syste
Core Tip: Methylene blue (MB) is a mechanistically rational adjunct vasopressor that targets nitric oxide (NO)-mediated vasodilation in septic shock. When used early and within safe cumulative dose (≤ 4 mg/kg), it consistently reduces vasopressor requirements without toxicity. Unlike non-selective NO synthase inhibitors, MB selectively modulates the NO-guanylate cyclase-cyclic guanosine monophosphate pathway by inhibiting only the inducible NO synthase isoform of NO synthase, preserving physiological constitutive functions. Current evidence supports its use as an adjunct rather than rescue therapy, pending results from ongoing randomized trials. In this narrative review we present the facts and data on MB with no fluff!
- Citation: Bansal T. Methylene blue - an emerging vasopressor. World J Clin Cases 2026; 14(13): 117947
- URL: https://www.wjgnet.com/2307-8960/full/v14/i13/117947.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i13.117947
Methylene blue (MB) (methylthioninium chloride) (C16H18ClN3S) was discovered by Heinrich Caro in 19th century, a German scientist. Its use in septic shock has seen a tottering course till the Ibarra-Estrada trial in 2023 leading to an increasing number of publications, interest and use of MB as a vasopressor.
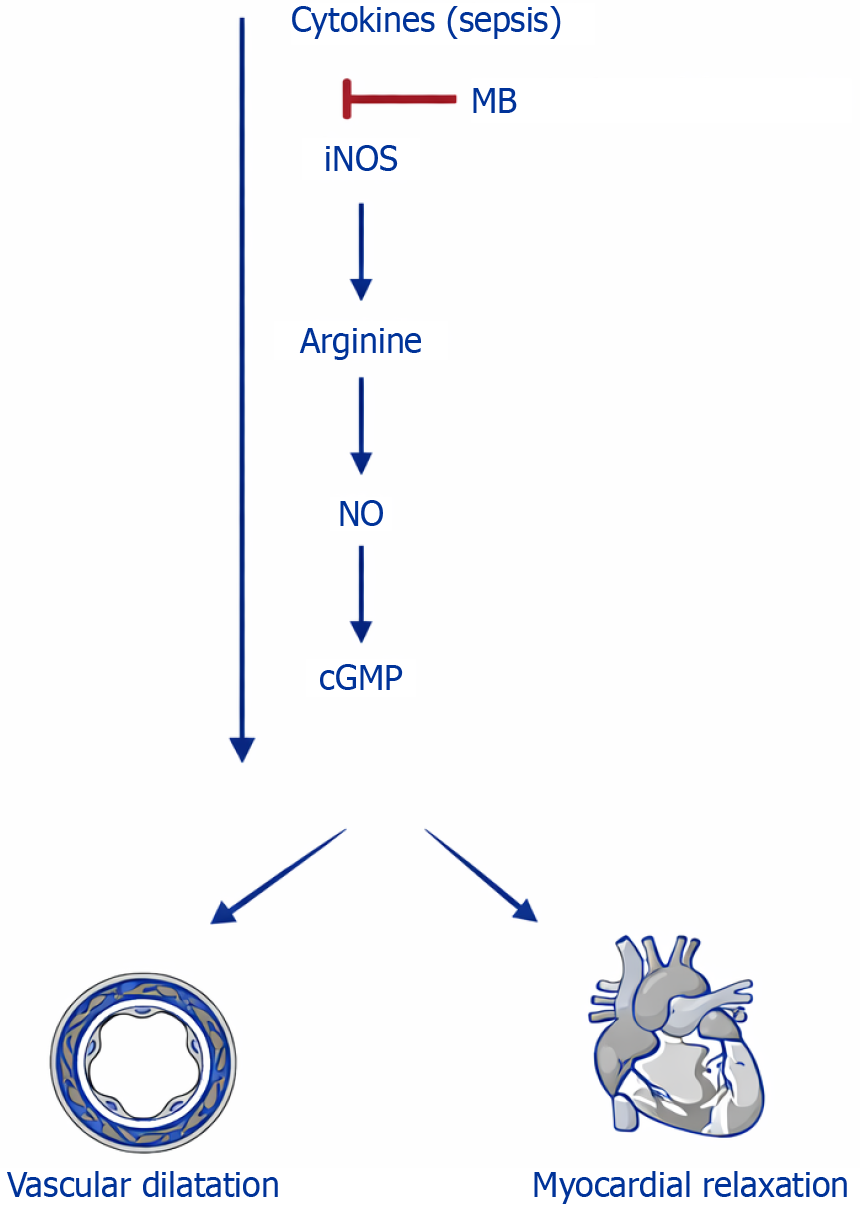
Septic shock is characterized by profound vasodilatation predominantly driven by excess nitric oxide (NO) production, with approximately 30% of patients also exhibiting concomitant myocardial depression. This pathological vasoplegia is primarily mediated by inducible NO synthase (iNOS), which is markedly upregulated during systemic inflammation.
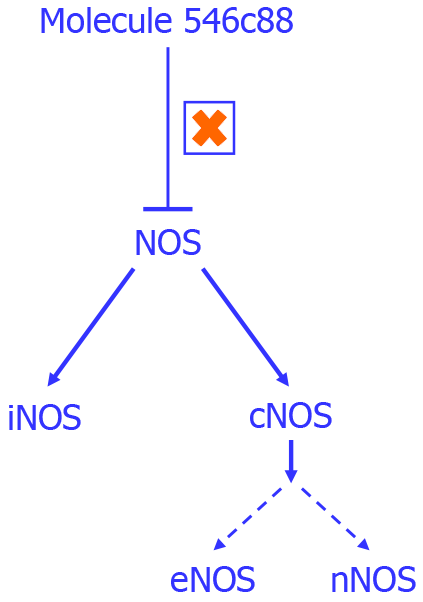
NO is synthesized by NO synthase (NOS), which exists in two major functional forms: Constitutive NOS (cNOS) and iNOS. cNOS comprises endothelial NOS and neuronal NOS and is responsible for physiological vascular homeostasis, endothelial integrity, basal blood pressure regulation, and neuronal signaling[1-3]. These pathways are essential for normal cardiovascular and cellular function and remain active under basal conditions.
In contrast, iNOS is minimally expressed in healthy states but is robustly induced during sepsis by inflammatory cytokines, endotoxins such as lipopolysaccharide, and other mediators. Once activated, iNOS generates large and sustained amounts of NO, leading to pathological vasodilation, impaired vascular responsiveness, and myocardial depression. Importantly, in septic shock, it is the excess NO derived from iNOS - rather than cNOS - that drives hemodynamic collapse, while cNOS pathways remain essential and largely unchanged for physiological homeostasis.
This distinction has critical clinical relevance. Prior attempts at non-selective NOS inhibition, most notably with N(G)-methyl-L-arginine hydrochloride (L-NMMA) (546C88), suppressed both iNOS and protective cNOS signaling and were associated with increased harm and mortality, leading to early termination of clinical trials[4]. These findings underscored the dangers of global NOS blockade and halted further development of non-selective strategies.
MB differs mechanistically from non-selective NOS inhibitors. Rather than globally suppressing NOS activity, MB preferentially attenuates iNOS-mediated NO signalling and its downstream effects, while preserving cNOS function. This selective modulation provides a mechanistic rationale distinct from prior failed approaches.
Downstream, NO activates guanylate cyclase, resulting in increased cyclic guanosine monophosphate (cGMP) production and subsequent relaxation of vascular smooth muscle cells and cardiomyocytes. MB inhibits guanylate cyclase and reduces cGMP generation; however, this effect is likely secondary and of lesser importance compared with its upstream attenuation of cytokine-driven iNOS activation and excessive NO production[5,6]. The differential effects of MB on the NO-guanylate cyclase-cGMP pathway, contrasted with non-selective NOS inhibition, are illustrated in Figures 1 and 2 which contrast selective inhibition of the iNOS-NO-cGMP pathway by MB with non-selective NOS inhibition by L-NMMA, which explains the divergent clinical outcomes observed and the need to understand the use of MB.
Beyond its effects on vascular tone, MB has been shown to exert direct effects on cardiac function and the pulmonary circulation. Excess NO is a well-recognized myocardial depressant and pulmonary vasodilator. In septic shock, inhibition of pathological NO excess by MB counteracts these effects, leading to improved left and right ventricular contractility and an increase in pulmonary vascular resistance.
These changes enhance ventriculo-arterial coupling and are associated with improvements in key hemodynamic indices, including cardiac index, left and right ventricular stroke work indices, mean pulmonary artery pressure, and pulmonary vascular resistance index. Such effects were consistently demonstrated in two well-conducted invasive hemodynamic studies by Kirov et al[7] and Donati et al[8], providing mechanistic support for the observed catecholamine-sparing effects of MB in septic shock.
The initial clinical use of MB as a vasopressor occurred in patients with vasoplegia following cardiopulmonary bypass during cardiac surgery. Although the underlying trigger differed, the pathophysiological mechanism was analogous to septic shock - excess NO production driven by cytokine release associated with surgical trauma and exposure to the bypass circuit rather than infection[9]. In this setting, a single bolus of MB was often sufficient to reverse vasoplegia, as postoperative vasoplegia typically resolves within 6 hours[10,11].
The first reported use of MB in septic shock was by Schneider in 1992, who successfully weaned vasopressors within 12 hours in two patients[12]. Subsequently, Brown et al[13] reported prolonged MB infusion for up to 44 hours with successful norepinephrine tapering in septic shock. These reports were followed by several case series and small observational studies suggesting hemodynamic benefit.
Two small randomized controlled trials by Kirov et al[7] and Memis et al[14] demonstrated reductions in vasopressor requirements with MB use. These remained the only randomized studies evaluating MB itself until the late 2000s. In contrast, a large multicenter randomized trial conducted by López et al[4] enrolled 797 patients with septic shock and followed earlier phase II studies[15,16]. This trial evaluated the non-selective NOS inhibitor L-NMMA (molecule 546C88), not MB. The study was terminated early because of increased 28-day mortality in the treatment arm (59% vs 49%, P < 0.001)[4]. Although no definitive mechanism was identified, the authors postulated that excessive vasoconstriction [target mean arterial pressure (MAP) 70-90 mmHg] and myocardial dysfunction may have contributed, as the excess mortality was predominantly cardiovascular.
Critically, L-NMMA inhibits both inducible and cNOS isoforms, suppressing protective cNOS signaling alongside pathological iNOS activity. In contrast, MB selectively attenuates iNOS-mediated NO signaling while sparing cNOS pathways essential for vascular and cellular homeostasis. Global inhibition of NO is known to have deleterious multisystem effects, and it is therefore plausible that the excess mortality observed in the López trial was related to non-selective NOS inhibition rather than NO modulation per se.
A large retrospective cohort study published in 2022 evaluated 223 patients who received MB for refractory shock, the majority of whom had septic shock[17]. The median bolus dose was 1.7 mg/kg, with a continuous infusion rate of 0.45 mg/kg/hour. Hemodynamic responders - defined by a MAP increase > 10% - demonstrated a reduction in nore
Notably, among responders receiving continuous renal replacement therapy (CRRT), intensive care units mortality was lower. The authors hypothesized that reduced clearance of MB during CRRT may lead to higher effective plasma concentrations. Supporting this observation, another study demonstrated a potential renoprotective effect of MB, with improved creatinine clearance and reduced urinary markers of tubular injury, which may partially explain improved outcomes in responders[18].
Overall, early case reports and small trials consistently demonstrated rapid catecholamine sparing with MB use; however, conclusions were limited by small sample sizes, heterogeneous dosing strategies, and variability in timing of administration.
The randomized trial by Ibarra-Estrada et al[19] in 2023 remains the most robust evidence supporting MB as an adjunct vasopressor in septic shock. This placebo-controlled randomized study enrolled 91 patients with early septic shock, with MB initiated within the first 24 hours. The trial was not powered to detect mortality differences.
All patients were receiving norepinephrine at enrollment. MB was administered as 100 mg infused over 6 hours daily for three consecutive days, resulting in a cumulative exposure of approximately 3.6 mg/kg. Vasopressin was added when norepinephrine requirements reached 0.25 μg/kg/minute.
MB significantly reduced vasopressor duration, shortening time to vasopressor discontinuation by nearly 25 hours. Vasopressor-free days at day 28 increased, and intensive care units and hospital length of stay were modestly reduced. There was no effect on ventilator duration or mortality, and importantly, no MB-related adverse events were observed. This trial re-established MB as a structured early adjunct rather than a rescue therapy and revived global interest in its use.
Following publication of the Ibarra-Estrada trial, few additional randomized studies have emerged. Post-2023 literature is dominated by pharmacologic reviews, meta-analyses, and small observational datasets. A consistent pattern is evident: MB is hemodynamically effective, appears safe, and is most beneficial when administered early.
Shaker et al[20] conducted a randomized trial in a cancer patient cohort with shock - predominantly septic - in which both low- and high-dose MB regimens were evaluated, demonstrating efficacy and safety. Multiple meta-analyses by Ballarin et al[21], Fernando et al[22], Huang et al[23], and Zhao et al[24], encompassing pooled patient numbers exceeding 1000, reached similar conclusions. MB reliably increases MAP, shortens vasopressor duration, and may confer a modest mortality benefit when used as a second-line adjunct rather than late rescue therapy. However, these analyses uniformly noted significant heterogeneity and the predominance of small studies, limiting the strength of conclusions.
Importantly, across studies evaluating MB itself, serious adverse effects remain rare (Tables 1 and 2). The López et al[4] trial remains a notable outlier, but it evaluated non-selective NOS inhibition rather than MB and should not be extrapolated to MB therapy.
| Ref. | Study design/population | Methylene blue regimen | Comparator | Major findings/clinical outcomes | Safety/adverse events | Key interpretation |
| Ibarra-Estrada et al[19], 2023 | Randomised controlled trial (n = 91); adult septic shock (sepsis-3) | 100 mg IV bolus infusion over 6 hour daily × 3 days (approximately 3.6 mg/kg total) | Placebo (saline) | Decreasing time to vasopressor discontinuation (69 hours vs 94 hours, P < 0.001); increasing vasopressor-free days; intensive care units and hospital LOS decrease; no mortality difference | No significant toxicity; no methemoglobinemia | Early adjunctive MB within 24 hours shortened vasopressor duration without added risk |
| Fernando et al[22], 2024 | Systematic review + meta-analysis (> 1000 patients) | Pooled 1-4 mg/kg bolus ± 0.25-1 mg/kg/hour infusion | Standard therapy | Decreasing vasopressor duration (approximately 20 hours); trend 28-day mortality decrease (RR: 0.82); mean arterial pressure increase | No increase in adverse events | MB benefit most pronounced when used early as adjunct |
| Ballarin et al[21], 2024 | Systematic review + meta-analysis (15 studies/1200 patients) | 1-4 mg/kg bolus ± continuous infusion | Placebo/standard care | Decreasing norepinephrine dose | No toxicity reported | Confirms hemodynamic benefit and renal protection |
| Arias-Ortiz and Vincent[25], 2024 | Narrative review/mechanistic update | Summative PK-PD review; discussed dose-response | - | Defined biphasic response; supports 2-4 mg/kg as effective and safe | Warns against > 7 mg/kg due to splanchnic vasoconstriction | Clarifies pharmacologic rationale and dose limits |
| Shaker et al[20], 2025 | Double-blind randomised controlled trial (n = 90) - cancer patients with septic shock | Low dose: 1 mg/kg bolus + 0.25 mg/kg/hour × 72 hour. High dose: 4 mg/kg bolus + 0.25 mg/kg/hour × 72 hours | Placebo (saline) | Both MB arms decrease vasopressor duration and increasing vasopressor-free days vs placebo (P < 0.05); trend mortality decrease (P = 0.083) | No methemoglobinemia or hemodynamic instability | Confirms efficacy up to 4 mg/kg; possible dose ceiling effect |
| Adverse effect | Comments/mechanism |
| Hemolysis (glucose-6-phosphate dehydrogenase deficiency) | Contraindicated; oxidative stress triggers hemolysis |
| Methemoglobinemia | < 2% incidence; dose-related |
| Serotonin syndrome | Occurs with MAOI/SSRI co-administration |
| Green-blue discoloration | Urine/secretions/skin; due to metabolites; affects pulse oximetry |
| Pulse oximetry artifacts | Spurious low SpO2; due to light absorption |
| Fall in P/F ratio | Possibly from pulmonary vasoconstriction → V/Q mismatch. Rare-high doses 31 |
| Splanchnic vasoconstriction | At doses > 7 mg/kg (possibly > 4 mg/kg) |
| Extravasation injury | Can cause necrosis; central line preferred |
| ECG changes, diaphoresis, dyspnea | Reported at higher doses |
| Pregnancy risk | Avoid use; insufficient safety data |
Recent pharmacologic analyses highlight why dosing and cumulative exposure are critical determinants of MB efficacy and safety. At lower doses, MB selectively modulates the NO-soluble guanylate cyclase-cGMP pathway, restoring vascular tone while preserving regional and microvascular perfusion. In contrast, higher cumulative doses may lead to excessive vasoconstriction, particularly in the splanchnic circulation, and increase the risk of dose-related toxicities such as methemoglobinemia[25].
Current practical dosing strategies fall into two broad categories. The most evidence-based regimen is derived from the Ibarra-Estrada trial, in which MB was administered as 100 mg infused over 6 hours daily for three consecutive days, resulting in a cumulative exposure of approximately 3.6 mg/kg. Alternative regimens reported in earlier studies include a bolus dose of 1-4 mg/kg, with or without a subsequent infusion for 12-24 hours[20]. Both approaches achieve comparable increases in MAP, provided cumulative exposure remains moderate.
At present, available data suggest that cumulative doses exceeding 7 mg/kg may be associated with adverse effects, while robust safety data beyond this threshold are lacking. Consequently, the optimal dosing strategy and the precise upper safe limit remain unsettled. The maximum duration of MB administration reported in the literature to date is approximately 120 hours[26].
MB follows a two-compartment pharmacokinetic model, with a distribution half-life of approximately 5-6 hours and a terminal elimination half-life of around 20 hours. Hemodynamic effects typically begin within the first hour of administration but are not immediate. MB undergoes hepatic reduction to leucomethylene blue, producing the characteristic blue-green discoloration of urine and secretions. Approximately 75% of the drug is excreted as metabolites and 25% unchanged[27].
Patients receiving CRRT may achieve higher effective MB concentrations, potentially explaining the enhanced hemodynamic responses observed in earlier observational cohorts. As noted by Naoum et al[17], “observed enhanced responses in CRRT patients may relate to altered volume of distribution or clearance; definitive pharmacokinetic data are lacking”. Dosing in hepatic dysfunction remains uncertain, given hepatic metabolism of the drug, as does the maximum safe duration beyond the approximately 120-hour exposure currently reported[28].
Across studies, MB consistently increases systemic vascular resistance, improves mean arterial pressure, preserves cardiac index, and reduces norepinephrine requirements[7,8,22,23,25]. Ballarin et al[21] reported an approximately 16% reduction in norepinephrine-equivalent dose within 6 hours of MB administration. Improvements in urine output and creatinine clearance have also been described in several cohorts.
However, improvements in macro-hemodynamic parameters do not necessarily translate into proven microcirculatory benefit. Whether MB meaningfully improves oxygen delivery or tissue oxygen utilization remains uncertain[25]. Modest reductions in serum lactate reported in some studies, including Zhao et al[24], are insufficient to establish a definitive microcirculatory effect because of small sample sizes and substantial heterogeneity.
MB remains a generally safe drug when used within recommended dosing ranges. Hemolysis in patients with glucose-6-phosphate dehydrogenase deficiency and serotonin syndrome with concomitant monoamine oxidase inhibitors or selective serotonin reuptake inhibitors represent the most clinically significant risks. Methemoglobinemia is uncommon and typically mild. Blue-green discoloration of urine, secretions, and skin is frequent and may interfere with pulse oximetry readings[28-31].
At cumulative doses above 7 mg/kg - and possibly above 4 mg/kg in susceptible individuals - splanchnic vasoconstriction and vasopressor-like toxicities may occur[32]. Tissue necrosis from extravasation underscores the importance of central venous administration.
MB has been successfully used in refractory anaphylactic shock, including reactions related to protamine, aprotinin, contrast agents, and antibiotics. In several reports, patients unresponsive to fluids, vasopressors, antihistamines, and corticosteroids demonstrated rapid hemodynamic improvement following MB bolus administration[33,34]. Histamine upregulates endothelial NOS expression, increasing NO production; inhibition of guanylate cyclase attenuates histamine-mediated vasodilation[35-38]. Del Duca et al[34] described two cardiac surgery patients with severe anaphylaxis in whom a single 100-mg MB bolus rapidly restored blood pressure and resolved urticaria.
MB has demonstrated benefit in vasoplegia associated with orthotopic liver transplantation, particularly during the reperfusion phase. Longhitano et al[39] conducted the only randomized controlled trial in this setting, administering 1.5 mg/kg MB before reperfusion in 36 patients, resulting in attenuation of reperfusion-related hypotension, increased systemic vascular resistance, lower lactate levels, and reduced epinephrine requirements. More recently Harding et al[40] reported further support for its utility in refractory post-reperfusion vasoplegia.
MB has been reported as adjunctive therapy in refractory shock due to calcium channel blocker overdose, particularly amlodipine toxicity[41], with occasional reports of benefit in metformin-associated shock.
MB has also been used in ifosfamide-induced encephalopathy, a toxic metabolic encephalopathy mediated by chloroacetaldehyde-induced mitochondrial dysfunction and oxidative stress. MB acts as an alternative electron acceptor, inhibits formation and effects of chloroacetaldehyde, and improves mitochondrial function. A review by Abahssain et al[42] identified 58 reported cases, with 65.5% receiving MB and 75.6% demonstrating clinical improvement, particularly when administered early. Robust prospective data, however, remain lacking.
MB inhibits pathological NO signaling primarily by attenuating cytokine-driven iNOS activation and, to a lesser extent, by blocking guanylate cyclase-mediated cGMP generation, thereby reducing vasodilation and myocardial depression. Over two decades of published data support MB as an adjunct vasopressor in vasodilatory shock - including septic, cardioplegic, and anaphylactic shock - without major adverse effects when used within recommended dosing ranges. Despite growing evidence, MB has not been incorporated into guidelines because of uncertainties regarding optimal dosing, duration, and its impact on oxygen delivery and utilization. Current evidence supports MB as an adjunct vasopressor in septic shock at doses of 100 mg infused over 6 hours daily for three days or a 2 mg/kg bolus followed by infusion (0.25-1 mg/kg/hour), with further clarification needed regarding standardized protocols. MB causes blue-green discoloration of secretions and skin and may interfere with pulse oximetry; higher doses increase the risk of methemoglobinemia, hemolysis (especially in glucose-6-phosphate dehydrogenase deficiency), and mesenteric ischemia. Ongoing and future studies will further define the role of MB as a targeted adjunct vasopressor.
Ongoing multicentre investigations, including pharmacokinetic-pharmacodynamic modelling studies (ClinicalTrials.gov NCT05941827), aim to individualize MB dosing using real-time hemodynamic monitoring. The Adjunctive MB in Septic SHock trial (NCT06900140) will assess feasibility prior to a large multicentre randomized trial, while additional studies are underway in Asia (NCT06481410, NCT0653224).
Accumulating evidence supports MB as a selective, mechanistically rational adjunct in septic shock. The most effective regimens employ early initiation (within 24 hours) and cumulative dosing up to approximately 4 mg/kg. Contemporary data demonstrate improvements in hemodynamic stability, vasopressor independence, myocardial performance, and renal perfusion without excess toxicity. Beyond septic shock, MB has utility in other forms of vasoplegia and selected toxicologic emergencies. Its low cost compared with newer vasopressors represents an additional advantage, particularly in low- and middle-income settings.
| 1. | Shanmugam G. Vasoplegic syndrome--the role of methylene blue. Eur J Cardiothorac Surg. 2005;28:705-710. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 107] [Article Influence: 5.1] [Reference Citation Analysis (1)] |
| 2. | Puntillo F, Giglio M, Pasqualucci A, Brienza N, Paladini A, Varrassi G. Vasopressor-Sparing Action of Methylene Blue in Severe Sepsis and Shock: A Narrative Review. Adv Ther. 2020;37:3692-3706. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 43] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 3. | Förstermann U, Sessa WC. Nitric oxide synthases: regulation and function. Eur Heart J. 2012;33:829-837, 837a. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3536] [Cited by in RCA: 3097] [Article Influence: 221.2] [Reference Citation Analysis (7)] |
| 4. | López A, Lorente JA, Steingrub J, Bakker J, McLuckie A, Willatts S, Brockway M, Anzueto A, Holzapfel L, Breen D, Silverman MS, Takala J, Donaldson J, Arneson C, Grove G, Grossman S, Grover R. Multiple-center, randomized, placebo-controlled, double-blind study of the nitric oxide synthase inhibitor 546C88: effect on survival in patients with septic shock. Crit Care Med. 2004;32:21-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 625] [Cited by in RCA: 600] [Article Influence: 27.3] [Reference Citation Analysis (0)] |
| 5. | Carvajal JA, Germain AM, Huidobro-Toro JP, Weiner CP. Molecular mechanism of cGMP-mediated smooth muscle relaxation. J Cell Physiol. 2000;184:409-420. [PubMed] [DOI] [Full Text] |
| 6. | Siegel GJ, Agranoff BW, Albers RW, Fisher SK, Uhler MD. Basic Neurochemistry: Molecular, Cellular and Medical Aspects. 6th ed. Philadelphia: Lippincott-Raven, 1999. |
| 7. | Kirov MY, Evgenov OV, Evgenov NV, Egorina EM, Sovershaev MA, Sveinbjørnsson B, Nedashkovsky EV, Bjertnaes LJ. Infusion of methylene blue in human septic shock: a pilot, randomized, controlled study. Crit Care Med. 2001;29:1860-1867. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 225] [Cited by in RCA: 177] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 8. | Donati A, Conti G, Loggi S, Münch C, Coltrinari R, Pelaia P, Pietropaoli P, Preiser JC. Does methylene blue administration to septic shock patients affect vascular permeability and blood volume? Crit Care Med. 2002;30:2271-2277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 65] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 9. | Levin RL, Degrange MA, Bruno GF, Del Mazo CD, Taborda DJ, Griotti JJ, Boullon FJ. Methylene blue reduces mortality and morbidity in vasoplegic patients after cardiac surgery. Ann Thorac Surg. 2004;77:496-499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 198] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 10. | Gomes WJ, Carvalho AC, Palma JH, Gonçalves I Jr, Buffolo E. Vasoplegic syndrome: a new dilemma. J Thorac Cardiovasc Surg. 1994;107:942-943. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 47] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 11. | Evora PR, Ribeiro PJ, de Andrade JC. Methylene blue administration in SIRS after cardiac operations. Ann Thorac Surg. 1997;63:1212-1213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 15] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 12. | Schneider F, Lutun P, Hasselmann M, Stoclet JC, Tempé JD. Methylene blue increases systemic vascular resistance in human septic shock. Preliminary observations. Intensive Care Med. 1992;18:309-311. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 75] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 13. | Brown G, Frankl D, Phang T. Continuous infusion of methylene blue for septic shock. Postgrad Med J. 1996;72:612-614. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 43] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 14. | Memis D, Karamanlioglu B, Yuksel M, Gemlik I, Pamukcu Z. The influence of methylene blue infusion on cytokine levels during severe sepsis. Anaesth Intensive Care. 2002;30:755-762. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 70] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 15. | Bakker J, Grover R, McLuckie A, Holzapfel L, Andersson J, Lodato R, Watson D, Grossman S, Donaldson J, Takala J; Glaxo Wellcome International Septic Shock Study Group. Administration of the nitric oxide synthase inhibitor NG-methyl-L-arginine hydrochloride (546C88) by intravenous infusion for up to 72 hours can promote the resolution of shock in patients with severe sepsis: results of a randomized, double-blind, placebo-controlled multicenter study (study no. 144-002). Crit Care Med. 2004;32:1-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 147] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 16. | Watson D, Grover R, Anzueto A, Lorente J, Smithies M, Bellomo R, Guntupalli K, Grossman S, Donaldson J, Le Gall JR; Glaxo Wellcome International Septic Shock Study Group. Cardiovascular effects of the nitric oxide synthase inhibitor NG-methyl-L-arginine hydrochloride (546C88) in patients with septic shock: results of a randomized, double-blind, placebo-controlled multicenter study (study no. 144-002). Crit Care Med. 2004;32:13-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 96] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 17. | Naoum EE, Dalia AA, Roberts RJ, Devine LT, Ortoleva J. Methylene blue for vasodilatory shock in the intensive care unit: a retrospective, observational study. BMC Anesthesiol. 2022;22:199. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 18. | Heemskerk S, van Haren FM, Foudraine NA, Peters WH, van der Hoeven JG, Russel FG, Masereeuw R, Pickkers P. Short-term beneficial effects of methylene blue on kidney damage in septic shock patients. Intensive Care Med. 2008;34:350-354. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 18] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 19. | Ibarra-Estrada M, Kattan E, Aguilera-González P, Sandoval-Plascencia L, Rico-Jauregui U, Gómez-Partida CA, Ortiz-Macías IX, López-Pulgarín JA, Chávez-Peña Q, Mijangos-Méndez JC, Aguirre-Avalos G, Hernández G. Early adjunctive methylene blue in patients with septic shock: a randomized controlled trial. Crit Care. 2023;27:110. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 62] [Article Influence: 20.7] [Reference Citation Analysis (1)] |
| 20. | Shaker EH, Soliman AM, Bedewy AAE, Elrawas MM. Comparative study between high and low dose methylene blue infusion in septic cancer patients: a randomized, blinded, controlled study. BMC Anesthesiol. 2025;25:15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 21. | Ballarin RS, Lazzarin T, Zornoff L, Azevedo PS, Pereira FWL, Tanni SE, Minicucci MF. Methylene blue in sepsis and septic shock: a systematic review and meta-analysis. Front Med (Lausanne). 2024;11:1366062. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 22. | Fernando SM, Tran A, Soliman K, Flynn B, Oommen T, Wenzhe L, Adhikari NKJ, Kanji S, Seely AJE, Fox-Robichaud AE, Wax RS, Cook DJ, Lamontagne F, Rochwerg B. Methylene Blue in Septic Shock: A Systematic Review and Meta-Analysis. Crit Care Explor. 2024;6:e1110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 23. | Huang X, Yan W, Chen Z, Qian Y. Effect of methylene blue on outcomes in patients with distributive shock: a meta-analysis of randomised controlled trials. BMJ Open. 2024;14:e080065. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 24. | Zhao CC, Zhai YJ, Hu ZJ, Huo Y, Li ZQ, Zhu GJ. Efficacy and safety of methylene blue in patients with vasodilatory shock: A systematic review and meta-analysis. Front Med (Lausanne). 2022;9:950596. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 25. | Arias-Ortiz J, Vincent JL. Administration of methylene blue in septic shock: pros and cons. Crit Care. 2024;28:46. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 26. | Dumbarton TC, Minor S, Yeung CK, Green R. Prolonged methylene blue infusion in refractory septic shock: a case report. Can J Anaesth. 2011;58:401-405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 27. | DiSanto AR, Wagner JG. Pharmacokinetics of highly ionized drugs. II. Methylene blue--absorption, metabolism, and excretion in man and dog after oral administration. J Pharm Sci. 1972;61:1086-1090. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 92] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 28. | Curry S. Methemoglobinemia. Ann Emerg Med. 1982;11:214-221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 225] [Cited by in RCA: 169] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 29. | Ng BK, Cameron AJ. The role of methylene blue in serotonin syndrome: a systematic review. Psychosomatics. 2010;51:194-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 19] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 30. | Sikka P, Bindra VK, Kapoor S, Jain V, Saxena KK. Blue cures blue but be cautious. J Pharm Bioallied Sci. 2011;3:543-545. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 33] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 31. | Gachot B, Bedos JP, Veber B, Wolff M, Regnier B. Short-term effects of methylene blue on hemodynamics and gas exchange in humans with septic shock. Intensive Care Med. 1995;21:1027-1031. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 69] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 32. | Juffermans NP, Vervloet MG, Daemen-Gubbels CR, Binnekade JM, de Jong M, Groeneveld AB. A dose-finding study of methylene blue to inhibit nitric oxide actions in the hemodynamics of human septic shock. Nitric Oxide. 2010;22:275-280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 72] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 33. | Daemen-Gubbels CR, Groeneveld PH, Groeneveld AB, van Kamp GJ, Bronsveld W, Thijs LG. Methylene blue increases myocardial function in septic shock. Crit Care Med. 1995;23:1363-1370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 80] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 34. | Del Duca D, Sheth SS, Clarke AE, Lachapelle KJ, Ergina PL. Use of methylene blue for catecholamine-refractory vasoplegia from protamine and aprotinin. Ann Thorac Surg. 2009;87:640-642. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 38] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 35. | Evora PR, Roselino CH, Schiaveto PM. Methylene blue in anaphylactic shock. Ann Emerg Med. 1997;30:240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 31] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 36. | Li H, Burkhardt C, Heinrich UR, Brausch I, Xia N, Förstermann U. Histamine upregulates gene expression of endothelial nitric oxide synthase in human vascular endothelial cells. Circulation. 2003;107:2348-2354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 72] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 37. | Kishi F, Nakaya Y, Takahashi A, Miyoshi H, Nomura M, Saito K. Intracellular and extracellular Ca2+ regulate histamine-induced release of nitric oxide in vascular endothelial cells as shown with sensitive and selective nitric oxide electrodes. Pharmacol Res. 1996;33:123-126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 11] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 38. | Champion HC, Kadowitz PJ. NO release and the opening of K+ATP channels mediate vasodilator responses to histamine in the cat. Am J Physiol. 1997;273:H928-H937. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 39. | Longhitano Y, Albuelkasem E, Voza A, La Via L, Zanza C, Planinsic RM. The Role of Methylene Blue and Cyanocobalamin for Vasoplegic Syndrome in Liver Transplantation. J Intensive Care Med. 2025;40:907-914. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 40. | Harding P, Nicholas T, Kassel C. The Use of Methylene Blue during Liver Transplantation for Vasoplegia. Case Rep Anesthesiol. 2021;2021:6610754. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 41. | Jang DH, Nelson LS, Hoffman RS. Methylene blue in the treatment of refractory shock from an amlodipine overdose. Ann Emerg Med. 2011;58:565-567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 48] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 42. | Abahssain H, Moukafih B, Essangri H, Mrabti H, Meddah B, Guessous F, Fadhil FZ, Souadka A, Errihani H. Methylene blue and ifosfamide-induced encephalopathy: Myth or reality? J Oncol Pharm Pract. 2021;27:143-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |