Published online May 6, 2026. doi: 10.12998/wjcc.v14.i13.117852
Revised: February 14, 2026
Accepted: March 25, 2026
Published online: May 6, 2026
Processing time: 127 Days and 1.7 Hours
Jugular paragangliomas (JPGLs) are rare, slow-growing neuroendocrine tumors originating from the adventitia of the vessels in the jugular foramen. They typi
We report the case of a 76-year-old woman with a long-standing history of right-sided hearing loss, intermittent chronic painless ear discharge, and lower motor facial nerve palsy, who presented with acute mastoid pain and otorrhea. Her symptoms were initially attributed to malignant otitis externa, given her diabetic status and history of a canal wall-up mastoidectomy. However, further evaluation with high-resolution computed tomography and contrast-enhanced magnetic re
This case underscores the importance of maintaining a broad differential diag
Core Tip: Jugular paragangliomas are rare skull base tumors that typically present with pulsatile tinnitus and lower cranial nerve palsies. This case highlights an atypical presentation marked by non-pulsatile tinnitus, sensorineural hearing loss, and isolated facial nerve involvement, leading to delayed diagnosis and intracranial extension. The report emphasizes the limitations of symptom-based diagnosis and underscores the critical role of early contrast-enhanced magnetic resonance imaging in patients with unilateral, progressive otologic symptoms to enable timely diagnosis and improved clinical outcomes.
- Citation: Patel H, Okuna Y, Ujima W. Atypical jugulotympanic paraganglioma presentation, diagnostic challenges, and role of imaging: A case report. World J Clin Cases 2026; 14(13): 117852
- URL: https://www.wjgnet.com/2307-8960/full/v14/i13/117852.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i13.117852
Paragangliomas are rare chromaffin cell tumors that arise from neural crest cells in the adventitia of blood vessels[1]. In the head and neck region, these tumors demonstrate a predilection for the jugular foramen [jugular paragangliomas (JPGLs)], middle ear cavity (tympanic paragangliomas), or combination (jugulotympanic paragangliomas), carotid body, and vagal nerve, with JPGLs being the second most common temporal bone tumors after vestibular schwannomas[2].
Although typically slow-growing and non-secretory, JPGLs can present with a wide range of symptoms based on their size and location[2]. Classic clinical features include pulsatile tinnitus, conductive hearing loss, and lower cranial nerve (CN) palsies (IX–XI), collectively referred to as jugular foramen syndrome[3]. However, in many patients, the pre
The diagnosis of JPGLs relies heavily on imaging, with contrast-enhanced magnetic resonance imaging (CEMRI) and computed tomography (CT) of the temporal bone playing complementary roles in identifying tumor extent, bony erosion, and vascular involvement[5]. Recent clinical guidelines recommend early CEMRI in patients presenting with sen
With surgical treatment, complete removal of the tumor is possible, often without recurrence. However, surgery carries a high risk of neurovascular complications, occurring in up to 60% of cases, most commonly CN injury and, less fre
A 76-year-old woman presented to the ear, nose, and throat clinic with a 4-day history of severe, throbbing right-sided mastoid pain, accompanied by purulent ear discharge and swelling over the right mastoid region.
The pain was acute in onset, progressive, and only partially relieved by over-the-counter analgesics. The discharge was moderate in amount, whitish-yellow, foul-smelling, and occasionally stained her pillow at night. Associated symptoms included right-sided non-pulsatile tinnitus, ear itchiness, dizziness, and subjective fever.
Notably, she denied any history of smoking, alcohol use, trauma, recent dental procedures, or systemic symptoms such as headache, visual disturbances, nausea, vomiting, or neurological deficits.
The patient has an 8-year history of well-controlled hypertension and newly diagnosed diabetes mellitus (hemoglobin A1c 7.6%). Eleven years before the current presentation (2024), the patient developed chronic right-sided otorrhea, intermittent otalgia, and progressive hearing loss. Preoperative imaging at that time demonstrated findings consistent with chronic suppurative otitis media with cholesteatoma, without radiologic evidence of a vascular skull base lesion. She subsequently underwent a right canal wall-up mastoidectomy. Histopathology confirmed cholesteatoma. There was no documentation of facial nerve palsy at that time.
In the years following surgery, the patient continued to experience intermittent right-sided otalgia and chronic discharge, with gradual worsening of hearing loss. Right-sided facial weakness developed several years postoperatively (exact onset unclear due to limited follow-up documentation) and progressively worsened to House-Brackmann grade V. The delayed onset and progressive nature of the facial nerve palsy suggest an evolving skull base process rather than an immediate postoperative complication.
During the intervening period, malignant otitis externa (MOE) was considered due to persistent otorrhea and progressive cranial nerve involvement. Microbiological confirmation, including cultures for Pseudomonas aeruginosa, was not documented. Furthermore, imaging did not demonstrate the characteristic skull base soft tissue infiltration or diffuse osteomyelitis typically seen in MOE.
The patient denied any family history of tumors.
On examination, she appeared toxic, with a fever of 38.6 °C and elevated blood pressure (186/109 mmHg). Otoscopy revealed the right post-auricular surgical scar, pre- and post-auricular induration with tenderness, and an otoscopy; a whitish, non-discharging mass completely obstructing the right external auditory canal was seen. Tuning fork tests demonstrated a left-lateralizing Weber test and a positive Rinne test (air conduction > bone conduction) on the right, consistent with right moderate to severe SNHL. The right facial nerve and cochlear nerve of VIII were affected, while CNs IX-XII were normal. The left ear and the remaining head and neck examination were unremarkable. Based on clinical findings, a diagnosis of MOE was made, and the patient was admitted.
No abnormality was found in routine blood analyses.
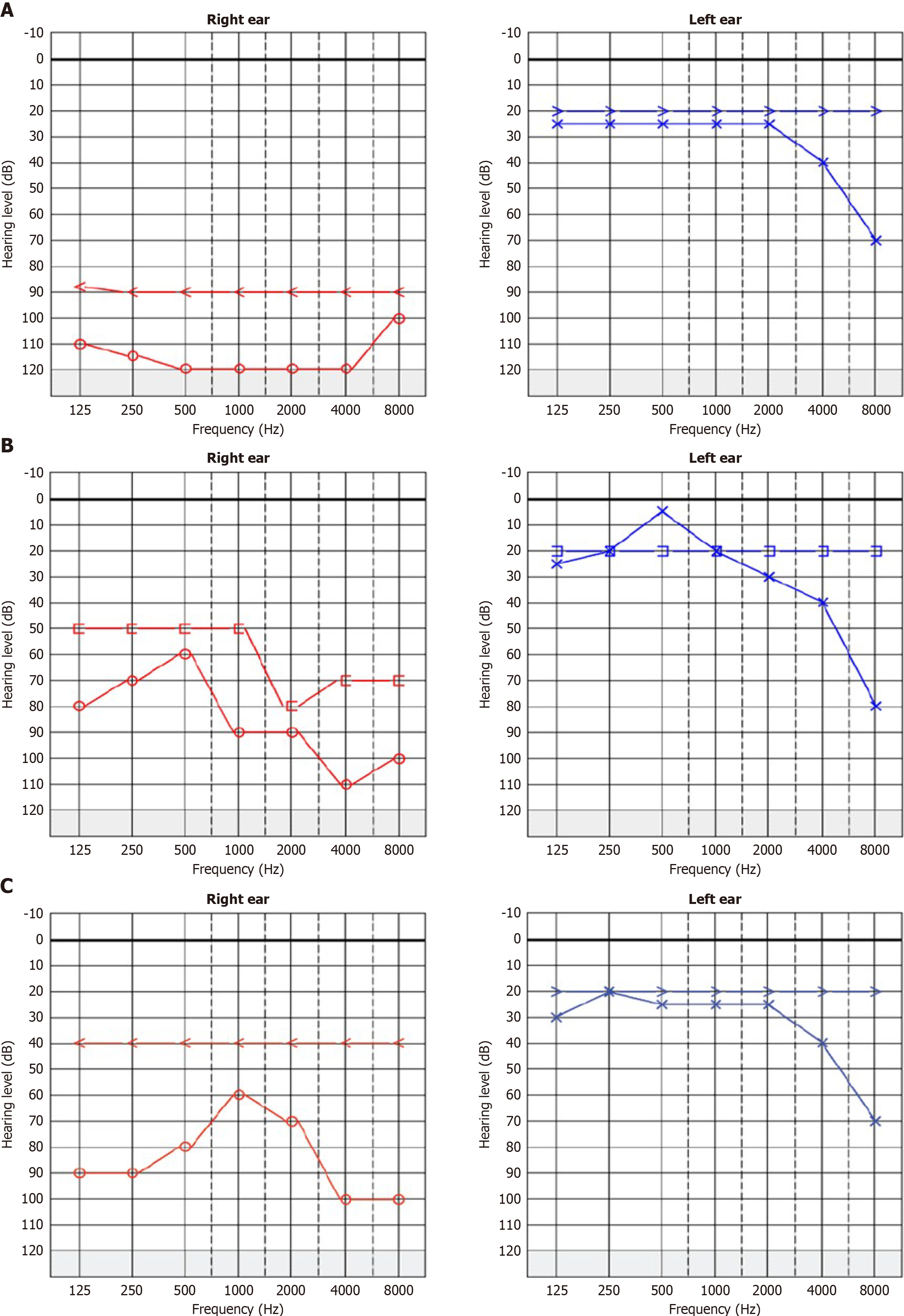
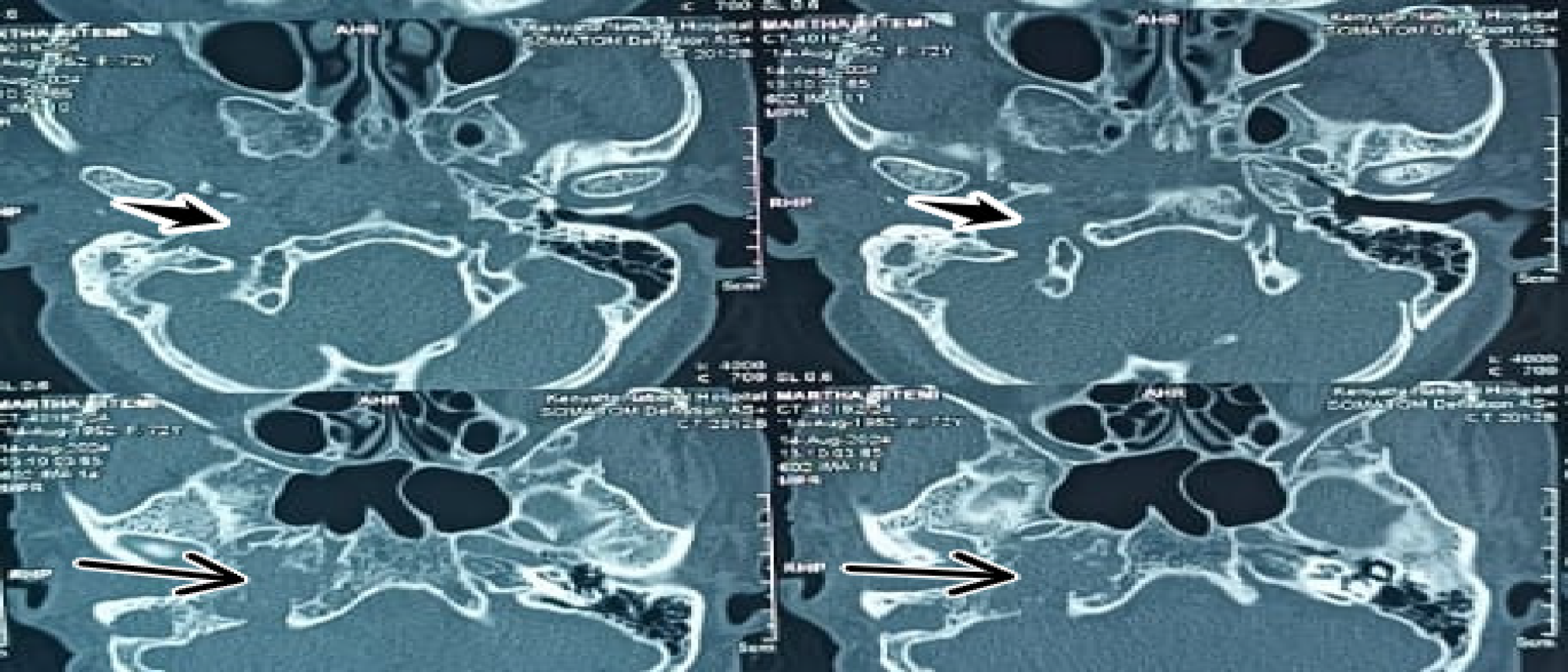
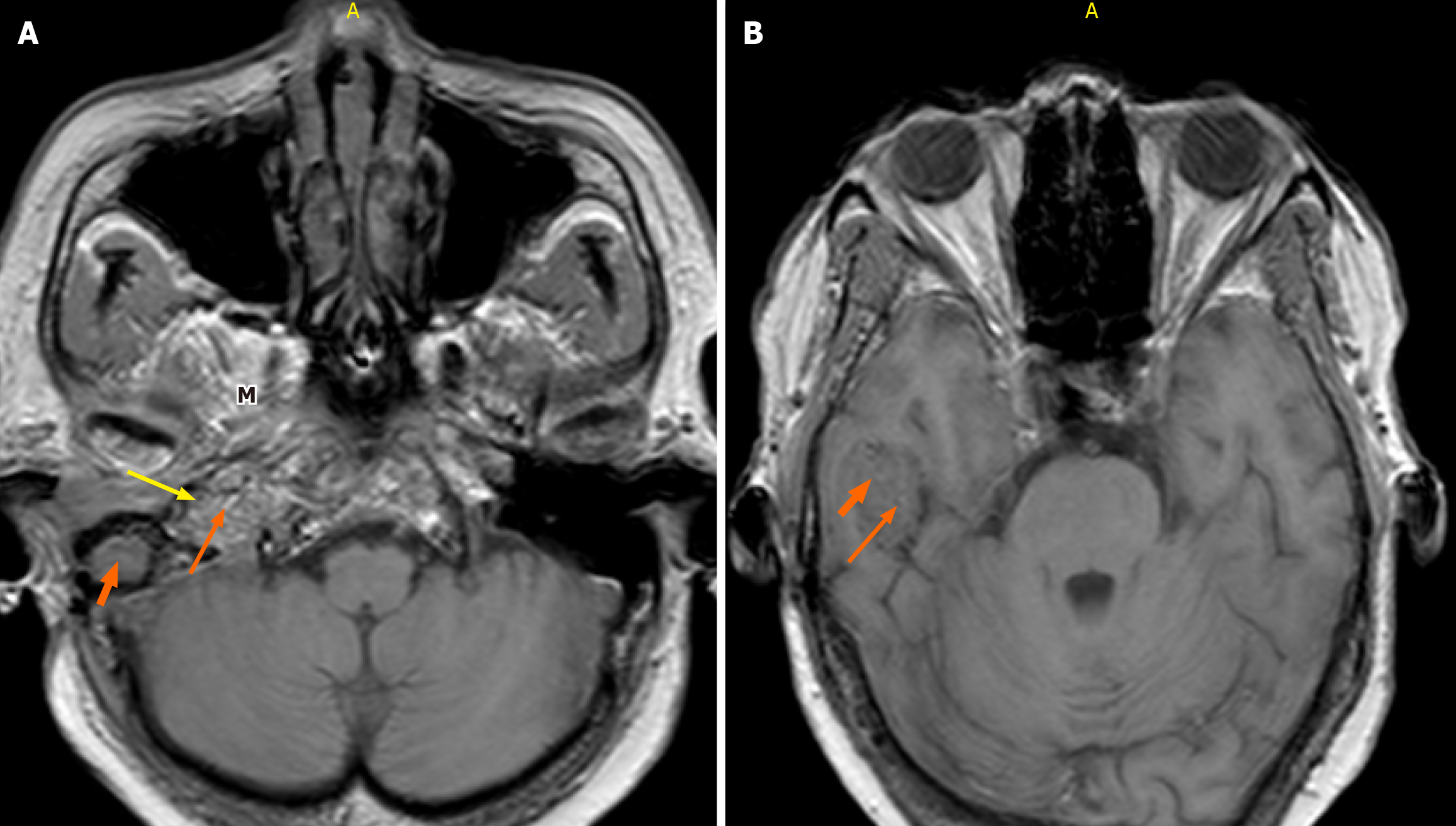
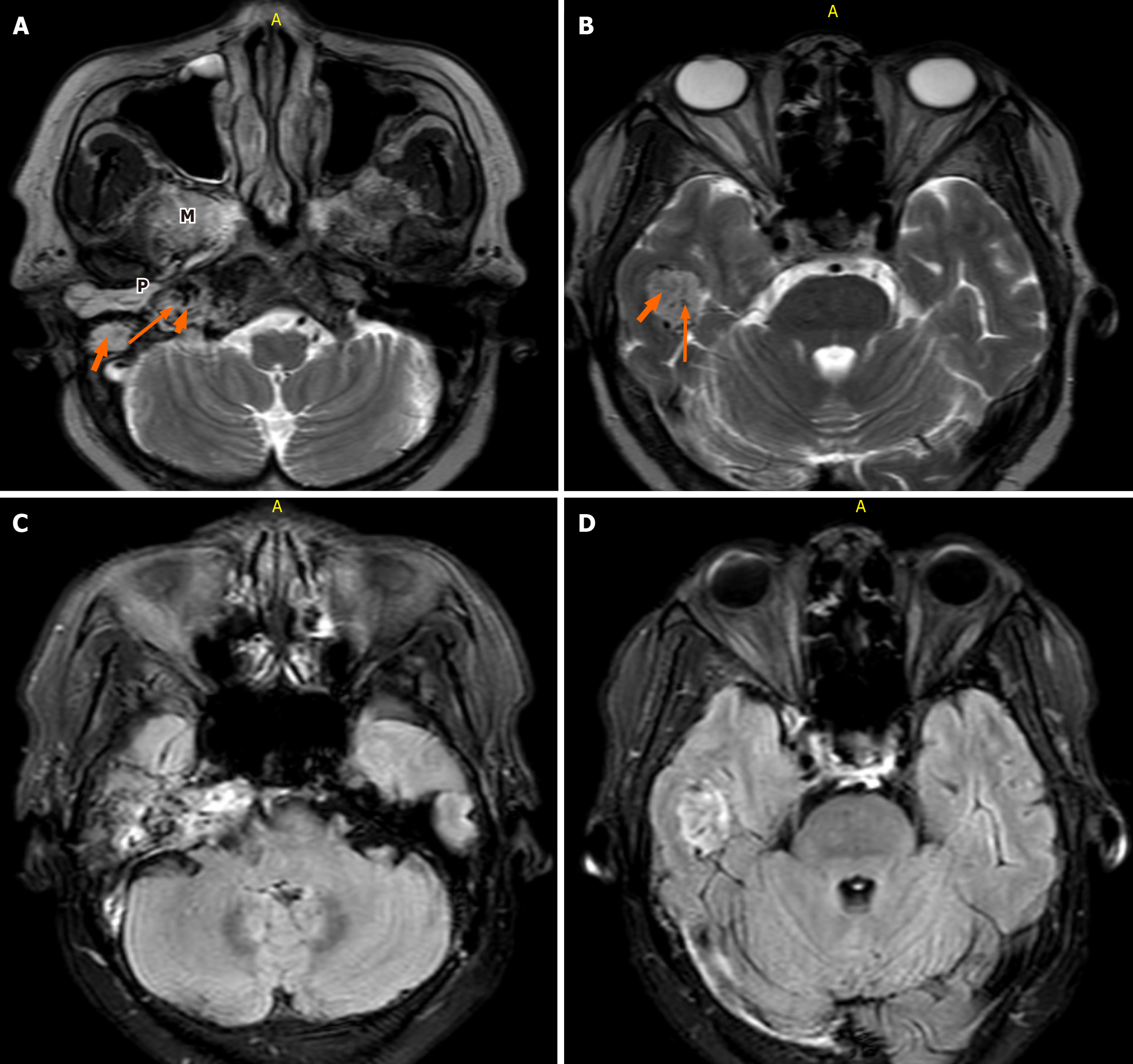
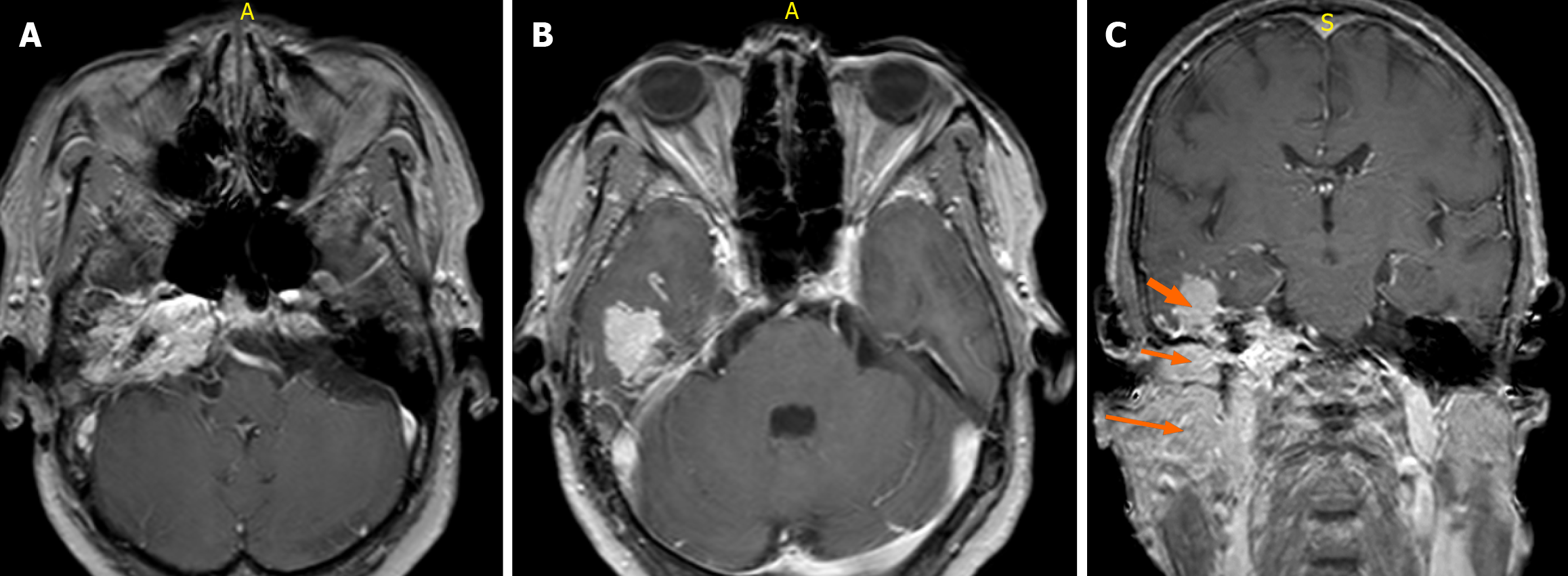
Pure-tone audiometry demonstrated moderately severe sensorineural hearing loss in the right ear, while the left ear exhibited isolated high-frequency hearing loss (Figure 1). A high-resolution CT (HRCT) scan of the temporal bone was obtained at admission, followed by a brain magnetic resonance imaging (MRI). Imaging revealed a highly vascular right jugulotympanic paraganglioma with superior and lateral extension into the middle ear and intracranial extension into the temporal lobe (Figures 2, 3, 4, and 5). A contrast-enhanced CT scan of the chest, abdomen, and pelvis was performed and demonstrated no evidence of distant metastasis. The patient was subsequently lost to follow-up; therefore, no definitive management plan was established.
The final diagnosis was jugulotympanic paraganglioma.
Initial stabilization included intravenous antibiotics, analgesia, blood pressure control, and glycemic management.
The patient was lost to follow-up and later returned with features suggestive of local intracranial extension. Further investigations, including serum metanephrines and chromogranin A levels, were not performed due to financial constraints, as the patient declined these tests. Additionally, due to the highly vascular nature of the tumor, a biopsy was not performed.
JPGLs present significant diagnostic challenges, particularly when classical jugular foramen syndromes are absent[2]. These tumors typically arise within the jugular foramen and may extend into the middle ear, temporal bone, or intracranially. Their clinical hallmark is pulsatile tinnitus, conductive hearing loss, and lower CN palsies (CN IX–XI), a constellation known as jugular foramen syndrome[8]. However, in this patient, such features were atypical, complicating timely diagnosis[9].
Our patient’s presentation with non-pulsatile tinnitus, SNHL, and long-standing facial nerve palsy deviated from the expected JPGL profile. Her case diagnosis was delayed and managed as chronic suppurative otitis media and cho
Interestingly, the tumor was non-pulsatile, likely due to multiple factors, including extensive erosion of the middle and inner ear structures and the associated conductive and sensorineural hearing loss. These changes may have masked the typical pulsatile nature of jugulotympanic paragangliomas, which is usually more apparent in lesions that preserve normal ossicular and vascular anatomy. The patient presented with isolated right facial nerve (CN VII) palsy without involvement of the lower cranial nerves (CN IX–XII). To our knowledge, this is the first reported case of a paraganglioma presenting with isolated CN VII palsy; previous reports have described primary involvement of the hypoglossal nerve (CN XII) or other lower cranial nerves[10].
Imaging revealed that the tumor primarily extended superiorly and laterally into the middle ear and temporal bone, involving the facial nerve canal, rather than inferiorly into the jugular foramen, where the lower cranial nerves exit. This selective involvement can be explained by the anatomic course of CN VII, which traverses the temporal bone via the fallopian canal, bringing it into direct contact with laterally and superiorly expanding tumors. Additionally, the slow-growing, hypervascular nature of jugulotympanic paragangliomas favors expansion along paths of least resistance, such as the middle ear and mastoid, rather than inferiorly toward the jugular bulb. These factors together account for the isolated facial nerve palsy and illustrate how tumor location, growth pattern, and vascularity determine cranial nerve involvement in skull base paragangliomas.
HRCT of the temporal bone revealed a soft-tissue mass in the right jugulotympanic region with avid contrast enhancement and permeative (“moth-eaten”) bone erosion of the jugular foramen and adjacent temporal bone, characteristic of a highly vascular skull base lesion. The following differential diagnoses were ruled out, as vestibular sch
This case underscores significant limitations in traditional diagnostic approaches that rely primarily on textbook symptom patterns. Adult patients presenting with unilateral, progressive ear complaints such as unexplained sen
HRCT is a valuable tool in the management of paraganglioma to evaluate bony erosion (moth-eaten pattern of the temporal bone) and surgical planning. In contrast, CEMRI provides superior contrast resolution, allowing visualization of tumor margins, vascularity, and involvement of surrounding neurovascular structures[5]. Magnetic resonance angio
In low-income countries, where advanced treatment modalities are often unavailable, surgery remains the cornerstone for both tumor management and the treatment of related complications[11]. It is the primary therapeutic option for patients presenting rapidly progressive intracranial hypertension and/or brainstem compression and continues to be the standard approach for young, otherwise healthy individuals who have already sustained complete functional loss of the affected CNs. Long-term follow-up has demonstrated that radical surgery can be highly effective in achieving a cure. Gross total resection is achieved in 40%-80% of cases, although local tumor control rates after surgical excision vary widely from 0% to 90%[12]. Despite refinements in microsurgical techniques and the use of intraoperative CN moni
Surgical morbidity, however, remains considerable, with complications ranging from cranial neuropathies to intracranial hematomas and cerebrospinal fluid leaks[3]. Deficits involving CNs IX–XII are reported in 31%-81% of patients, with some requiring vocal cord repair or gastrostomy. Facial nerve palsy is observed in nearly one-third to almost half of the cases, occasionally necessitating tarsorrhaphy, while hearing deterioration or loss is also frequently documented. Mortality rates remain significant, ranging from 4% to 6%. These risks are particularly pronounced in “complex” glomus jugulare tumors, which are characterized by features such as large size, multifocality, malignant histology, or catecholamine secretion[3].
This case highlights the diagnostic complexity of JPGLs, particularly when they present without classical features such as pulsatile tinnitus or lower CN palsies. Atypical symptoms like non-pulsatile tinnitus and isolated facial nerve involvement can mislead clinicians, especially patients presenting with CSOM. Inappropriate imaging in such cases can result in advanced tumor progression and limited therapeutic options. This underscores the importance of following evidence-based protocols, including early CEMRI in patients with unilateral SNHL and persistent ear symptoms. A high index of suspicion, timely imaging, and a multidisciplinary approach are crucial for early diagnosis, optimal man
We are grateful to the patient for his agreement of publication of this report and accompanying images.
| 1. | Brieger J, Bedavanija A, Gosepath J, Maurer J, Mann WJ. Vascular endothelial growth factor expression, vascularization and proliferation in paragangliomas. ORL J Otorhinolaryngol Relat Spec. 2005;67:119-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 2. | Sandow L, Thawani R, Kim MS, Heinrich MC. Paraganglioma of the Head and Neck: A Review. Endocr Pract. 2023;29:141-147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 66] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 3. | Antczak T, Borowy A, Suchcicka W, Bartoszewicz R. Analysis of clinical presentation and directions of extension of jugular foramen paragangliomas. Pol Otorhino Rev. 2023;12:14-20. [DOI] [Full Text] |
| 4. | Huang Z, Liu H, Huang W, Wang H, Liu J, Wu Z. Giant retroperitoneal paraganglioma: Challenges of misdiagnosis and high surgical risks, a case report. Int J Surg Case Rep. 2021;84:106081. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 5. | Lee KY, Oh YW, Noh HJ, Lee YJ, Yong HS, Kang EY, Kim KA, Lee NJ. Extraadrenal paragangliomas of the body: imaging features. AJR Am J Roentgenol. 2006;187:492-504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 196] [Cited by in RCA: 155] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 6. | Aliabadi H, Vredenburgh JJ, Everson RG, Desjardins A, Friedman HS, McLendon RE, Tucci DL, Sampson JH. Bevacizumab fails to treat temporal paraganglioma: discussion and case illustration. J Neurooncol. 2010;98:427-430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 7. | Chung SM, Kim HS, Jung J, Lee HK, Lee WS. Clinical presentation and management of jugular foramen paraganglioma. Clin Exp Otorhinolaryngol. 2009;2:28-32. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 9] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 8. | Eguchi S, Ono R, Sato T, Yada K, Umehara N, Narumi S, Ichihashi Y, Nozaki T, Kanomata N, Hasegawa T, Ozawa M, Hasegawa D. Hereditary paraganglioma presenting with atypical symptoms: Case report. Medicine (Baltimore). 2021;100:e27888. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 9. | Patel R, Allam YA, Shukairy MK, Kircher M. An Unusual and Discrepant Presentation of a Skull Base Paraganglioma. Cureus. 2024;16:e66394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 10. | Taïeb D, Hicks RJ, Hindié E, Guillet BA, Avram A, Ghedini P, Timmers HJ, Scott AT, Elojeimy S, Rubello D, Virgolini IJ, Fanti S, Balogova S, Pandit-Taskar N, Pacak K. European Association of Nuclear Medicine Practice Guideline/Society of Nuclear Medicine and Molecular Imaging Procedure Standard 2019 for radionuclide imaging of phaeochromocytoma and paraganglioma. Eur J Nucl Med Mol Imaging. 2019;46:2112-2137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 223] [Cited by in RCA: 282] [Article Influence: 40.3] [Reference Citation Analysis (0)] |
| 11. | Huy PT, Kania R, Duet M, Dessard-Diana B, Mazeron JJ, Benhamed R. Evolving concepts in the management of jugular paraganglioma: a comparison of radiotherapy and surgery in 88 cases. Skull Base. 2009;19:83-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 42] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 12. | Hafez RF, Morgan MS, Fahmy OM. The safety and efficacy of gamma knife surgery in management of glomus jugulare tumor. World J Surg Oncol. 2010;8:76. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 1.2] [Reference Citation Analysis (0)] |