Published online May 6, 2026. doi: 10.12998/wjcc.v14.i13.117403
Revised: March 5, 2026
Accepted: March 30, 2026
Published online: May 6, 2026
Processing time: 138 Days and 14.7 Hours
Neurocysticercosis (NCC) is the most common parasitic infection of the central nervous system and remains an important cause of neurological morbidity worldwide. Intraventricular NCC may lead to obstructive hydrocephalus and often requires surgical management. The fourth ventricle is the most frequently involved intraventricular location but poses specific diagnostic and therapeutic challenges.
A 60-year-old woman presented with persistent headache, dizziness, and recent onset nausea and vomiting. Brain magnetic resonance imaging revealed ob
Fourth ventricle NCC may require direct surgical removal when cerebrospinal fluid diversion alone is insufficient. Microsurgical excision remains an effective treatment option.
Core Tip: Intraventricular neurocysticercosis (NCC) may present with obstructive hydrocephalus requiring urgent management. Although endoscopic third ventriculostomy can relieve hydrocephalus, it may not address the underlying obstruction caused by the cyst. This case highlights the management dilemma between cerebrospinal fluid diversion and definitive cyst removal in fourth ventricle NCC. When symptoms persist or the lesion enlarges, microsurgical excision through a suboccipital telovelar approach remains a safe and effective treatment strategy.
- Citation: Creatura D, El Choueiri J, Anselmi L, Di Cosmo L, Bono BC, Rampini AD, Lanterna LAA. Fourth ventricle neurocysticercosis with hydrocephalus: A case report and literature review. World J Clin Cases 2026; 14(13): 117403
- URL: https://www.wjgnet.com/2307-8960/full/v14/i13/117403.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i13.117403
Neurocysticercosis (NCC) is the most common parasitic infection affecting the human central nervous system (CNS) and is caused by larvae of the pork tapeworm Taenia solium[1,2]. CNS NCC disease may be classified as intraparenchymal or extra-parenchymal. The extra-parenchymal type can be further categorized as intraventricular, subarachnoid, and spinal. Among the intraventricular CNS NCC, the fourth ventricle is the most frequent location, followed by the lateral and third ventricles. Consequently, this type of CNS NCC is usually associated with subacute onset of headache, visual im
In this paper, we describe a patient with an extra-parenchymal NCC in the fourth ventricle who underwent both an endoscopic third ventriculostomy (ETV) and microsurgical cyst removal. We discuss the treatment strategies and surgical indications for the rare condition of fourth ventricle NCCs.
A 60-year-old woman presented with persistent headache, dizziness, and recent onset nausea and vomiting.
She presented with a 5-month history of progressive headache and dizziness, followed by the recent onset of nausea and vomiting, prompting admission to the emergency department. No other neurological symptoms were reported.
No special notes.
No special notes.
Neurological examination was unremarkable at initial presentation.
Blood tests revealed the presence of anti-Taenia solium antibodies, supporting the suspected diagnosis; notably, lumbar puncture was not performed due to the risk of herniation for a fourth ventricular lesion.
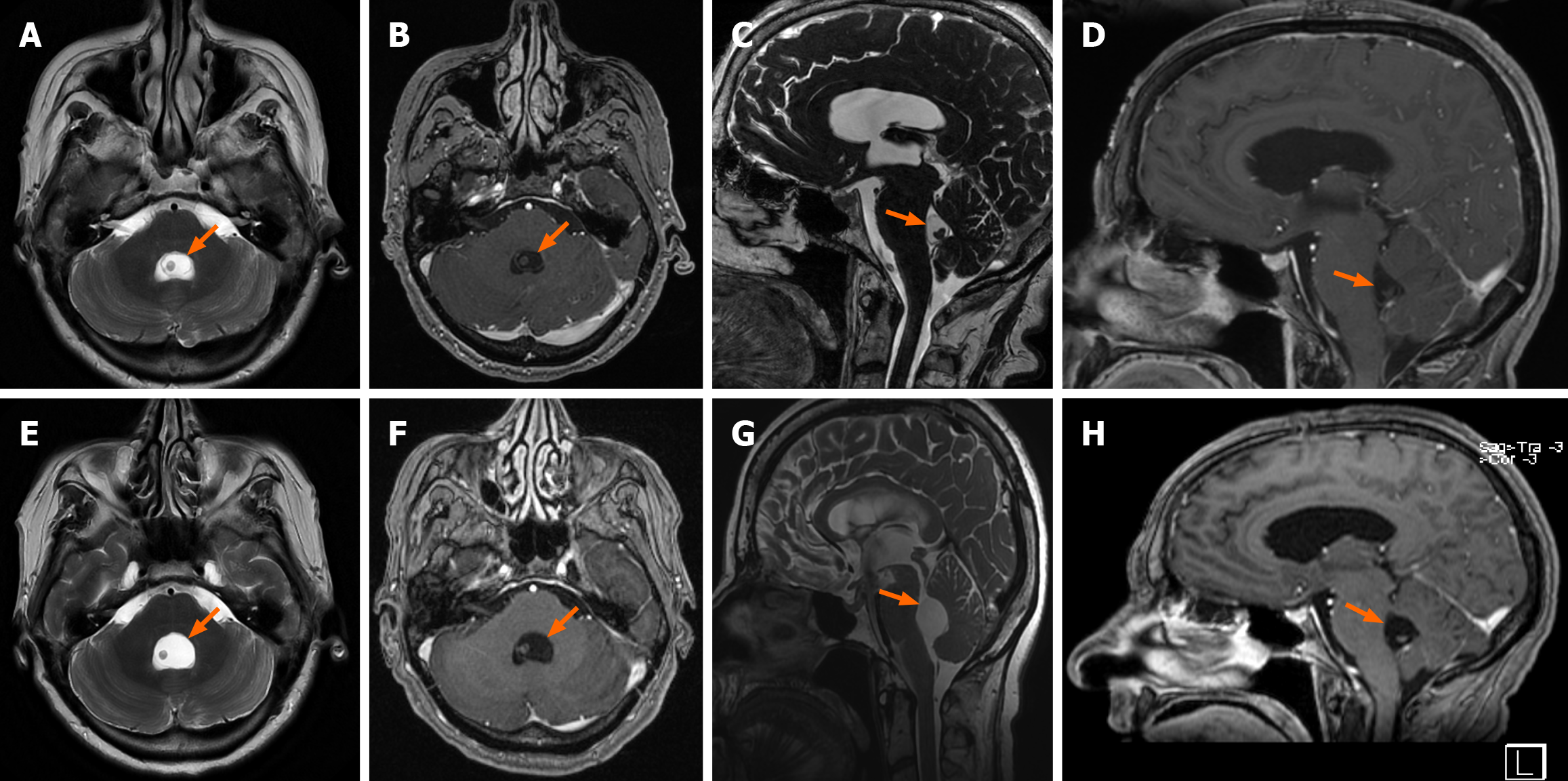
Magnetic resonance imaging (MRI) of the brain with gadolinium revealed a well-defined fourth ventricular cystic lesion, leading to tri-ventricular hydrocephalus (Figure 1). A small blurred-enhancing nodular component (5 mm) was observed within the cyst, adjacent to the right margin of the thickened cystic wall. A cerebral catheter angiogram was then performed to rule out a posterior fossa hemangioblastoma, initially considered in the differential diagnosis.
The MRI scans were carefully reviewed, and the radiological features—particularly the eccentrically located scolex within a thin-walled cyst—were highly suggestive of NCC in the vesicular stage.
An endoscopic third ventriculostomy (ETV) was first performed to treat hydrocephalus. The postoperative course was uneventful and, after multidisciplinary discussion, the patient was discharged without any anti-parasitic medication, as it was believed that such treatment could worsen cyst-related inflammation and swelling.
However, 5 weeks later, the patient was readmitted with recurrent headaches, vomiting, and gait ataxia. A repeat brain MRI demonstrated a significant increase in the size of the fourth ventricular cystic lesion (Figure 1).
A new multidisciplinary discussion with infectious disease specialists was conducted, and surgical removal of the lesion was considered the most appropriate treatment strategy to both excise the cyst and resolve the obstructive hydrocephalus.
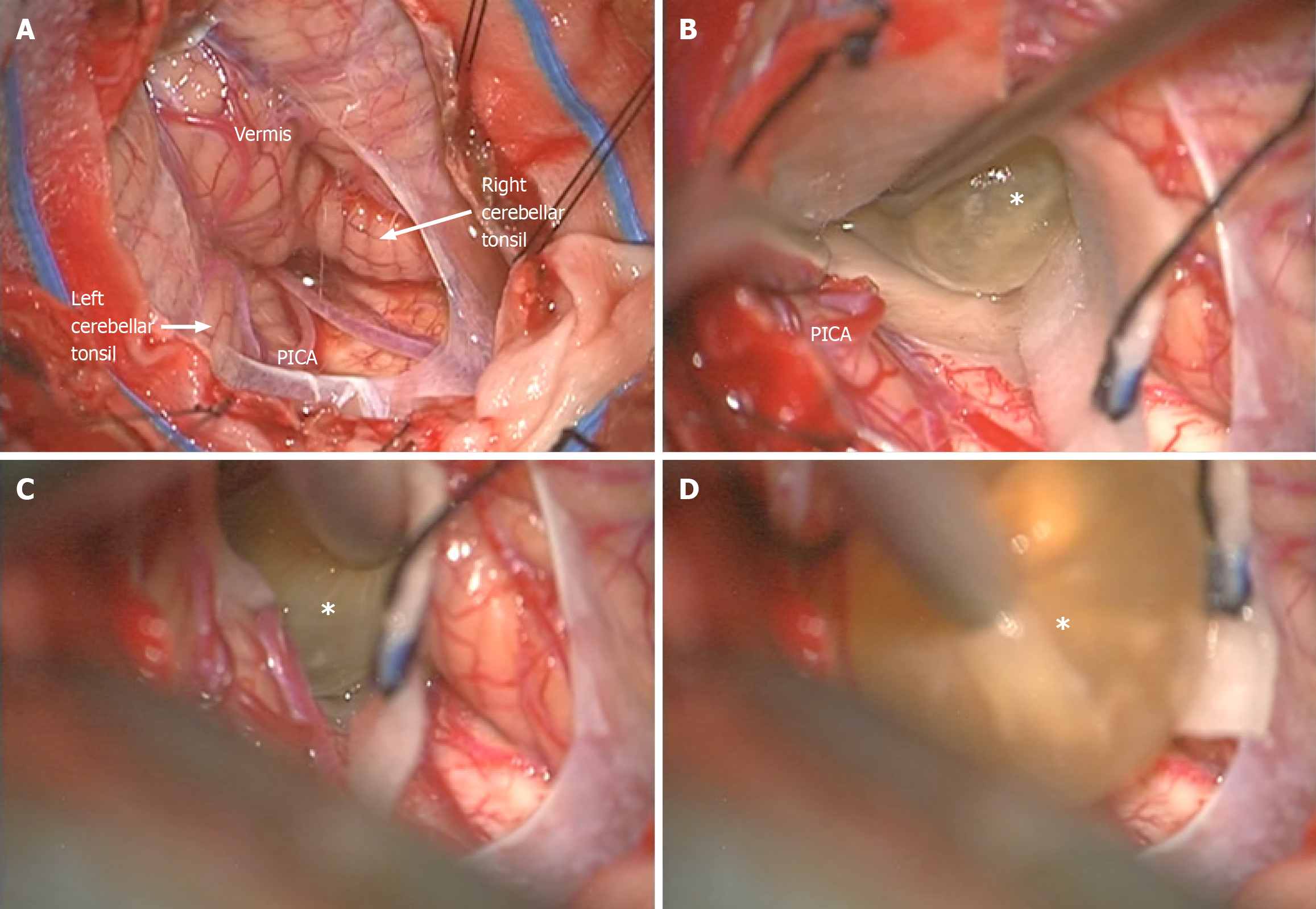
En bloc surgical resection of the lesion allowed definitive diagnosis of fourth ventricular neurocysticercosis (Taenia solium cyst) in the vesicular stage causing obstructive hydrocephalus.
The patient was placed in a prone position and a suboccipital telovelar approach was performed to reach the fourth ventricle. The cyst was identified and microsurgically dissected from the ependyma, allowing en bloc removal without any rupture (Figure 2). Cerebrospinal fluid (CSF) flow was successfully restored. Perioperative corticosteroid therapy was administered to reduce the inflammatory response.
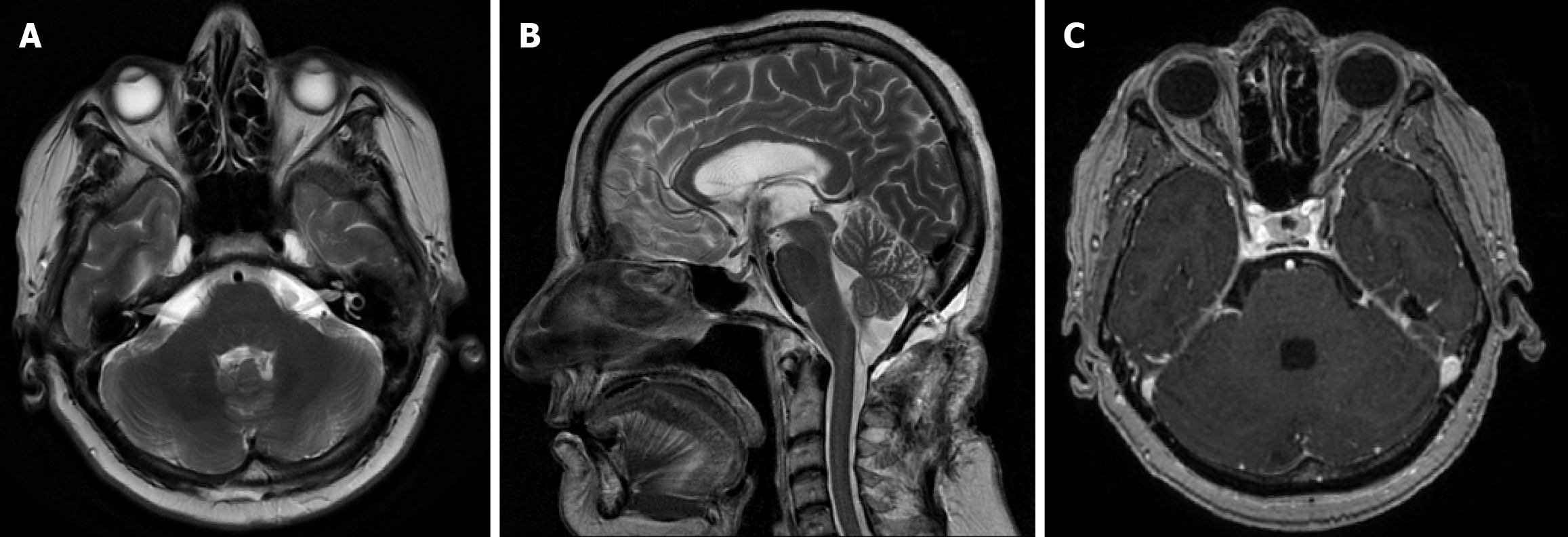
Postoperative MRI showed a gross total removal of the ventricular NCC and a marked reduction in ventricular size (Figure 3). Histopathological analysis confirmed the lesion as a Taenia solium cyst in the vesicular stage. After consultation with infectious disease specialists, antiparasitic therapy was withheld due to the lack of evidence supporting its benefit for solitary intraventricular cysts.
The postoperative course was uneventful, and the patient was discharged on postoperative day 8 without any neu
NCC is the most common parasitic infection affecting the CNS and remains a major cause of neurological morbidity worldwide[3-5]. It results from infection by pork tapeworm Taenia solium larvae and is typically acquired through ingestion of contaminated food or water[1].
NCC can be classified according to its anatomical location as parenchymal or extra-parenchymal disease. The extra-parenchymal form includes intraventricular, subarachnoid, and spinal involvement and is generally associated with more severe clinical manifestations, such as hydrocephalus, arachnoiditis, and ventriculitis[6,7]. Among intraventricular locations, the fourth ventricle represents the most frequent site (43%-70%), followed by the lateral and third ventricles[2]. In this setting, hydrocephalus is common and may result from direct obstruction of CSF pathways or inflammatory processes leading to aqueductal stenosis[8].
Intraventricular lesions may occasionally cause Bruns’ syndrome, characterized by intermittent obstruction of CSF flow due to a mobile intraventricular mass[9,10]. Although this condition can also be associated with tumors or other cystic lesions, it has been historically described in patients with fourth ventricle NCC[11].
Radiological findings play a crucial role in diagnosis. MRI typically demonstrates a cystic lesion with an eccentrically located scolex, which is highly suggestive of NCC in the vesicular stage. Nevertheless, differential diagnosis of cystic lesions within the fourth ventricle includes infectious conditions such as tuberculomas or hydatid cysts, as well as neoplastic and nonneoplastic lesions, including ependymomas, subependymomas, arachnoid cysts, or Blake’s pouch cysts[12-17]. Clinical history and epidemiological context can therefore help guide the diagnosis.
Management of intraventricular NCC remains challenging and often requires surgical intervention, particularly when hydrocephalus is present[18]. Treatment strategies aim to relieve CSF obstruction and, when feasible, remove the causative lesion. Historically, ventriculoperitoneal shunting has been commonly used for NCC-associated hydrocephalus; however, shunt malfunction and infection rates are relatively high, often requiring multiple revisions[2,19]. For this reason, ETV has been proposed as an alternative strategy for CSF diversion. Some series have reported success rates exceeding 90% with endoscopic CSF diversion procedures[20,21].
In our case, ETV was initially performed to treat obstructive hydrocephalus and to avoid permanent shunt placement. Although the patient initially improved, she returned 5 weeks later with recurrent symptoms and radiological evidence of cyst enlargement. This situation illustrates an important management dilemma in intraventricular NCC: While CSF diversion may provide temporary symptomatic relief, it does not eliminate the underlying obstructive lesion, and definitive cyst removal may ultimately be required.
Endoscopic removal has increasingly become the preferred approach for intraventricular cysts, particularly in the lateral and third ventricles[20,22]. However, removal of fourth ventricle cysts may be technically demanding depending on the anatomy of the aqueduct, the size of the cyst, and its potential adherence to the ependymal surface[23]. In these situations, a posterior fossa microsurgical approach remains a reliable and effective alternative.
Medical treatment of NCC remains a matter of debate. Anthelminthic therapy has demonstrated benefits in patients with active parenchymal disease by reducing viable cysts and preventing seizure recurrence[24-26]. Current guidelines from the Infectious Diseases Society of America and the American Society of Tropical Medicine and Hygiene recommend albendazole-based therapy depending on the number of parenchymal lesions[27]. However, in cases of intraventricular NCC, particularly when the cyst can be safely removed, surgical treatment remains the preferred option[22,27].
Moreover, some studies have discouraged the use of antiparasitic therapy before surgery because it may induce inflammatory changes or cyst rupture, potentially complicating surgical removal[27]. Corticosteroids are often ad
The suboccipital telovelar approach allows direct access to the fourth ventricle while avoiding splitting of the cerebellar vermis and provides adequate visualization for safe dissection of the cyst from surrounding structures. Several articles have discussed the risk factors and etiology of the disease[29,30], and many case series have reported favorable outcomes using this technique[31-35]. In our patient, microsurgical removal allowed complete en bloc resection of the cyst and restoration of CSF circulation.
Management of intraventricular NCC requires addressing both hydrocephalus and the obstructing lesion. Although ETV can effectively relieve hydrocephalus, it may not provide definitive treatment when the cyst continues to obstruct CSF pathways. This case highlights the clinical decision-making dilemma between cerebrospinal fluid diversion and definitive cyst removal in fourth ventricle NCC. When symptoms persist or the lesion enlarges, microsurgical excision through a suboccipital telovelar approach remains a safe and effective treatment option.
| 1. | Gripper LB, Welburn SC. Neurocysticercosis infection and disease-A review. Acta Trop. 2017;166:218-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 75] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 2. | Jensen TO, Post JJ. Intraventricular neurocysticercosis: Presentation, diagnosis and management. Asian Pac J Trop Med. 2016;9:815-818. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 18] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 3. | Psarros TG, Zouros A, Coimbra C. Neurocysticercosis: a neurosurgical perspective. South Med J. 2003;96:1019-1022. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 4. | Rizvi SA, Saleh AM, Frimpong H, Al Mohiy HM, Ahmed J, Edwards RD, Ahmed SS. Neurocysticercosis: A case report and brief review. Asian Pac J Trop Med. 2016;9:100-102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 5. | Del Brutto OH. Neurocysticercosis. Neurohospitalist. 2014;4:205-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 36] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 6. | Xiao A, Xiao J, Zhang X, You C. The Surgical Value of Neurocysticercosis: Analyzing 10 Patients in 5 Years. Turk Neurosurg. 2016;26:744-749. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 7. | DeGiorgio CM, Houston I, Oviedo S, Sorvillo F. Deaths associated with cysticercosis. Report of three cases and review of the literature. Neurosurg Focus. 2002;12:e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 32] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 8. | Fleury A, Escobar A, Fragoso G, Sciutto E, Larralde C. Clinical heterogeneity of human neurocysticercosis results from complex interactions among parasite, host and environmental factors. Trans R Soc Trop Med Hyg. 2010;104:243-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 69] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 9. | Shahani L, Garnes ND, Mejia R. Intraventricular Taenia solium cysts presenting with Bruns syndrome and indications for emergent neurosurgery. Am J Trop Med Hyg. 2015;92:1261-1264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 10. | Roque D, Hipólito-Reis J, Duarte-Batista P, Manuel Santos M. Fourth ventricle neurocysticercosis presenting with Bruns' syndrome: A case report. Surg Neurol Int. 2021;12:616. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 11. | White C. Intraventricular Neurocysticercosis and Bruns' Syndrome: A Review. J Rare Dis Res Treat. 2017;2:1-5. [DOI] [Full Text] |
| 12. | Westermaier T, Vince GH, Meinhardt M, Monoranu C, Roosen K, Matthies C. Arachnoid cysts of the fourth ventricle - short illustrated review. Acta Neurochir (Wien). 2010;152:119-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 18] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 13. | Mayorga-Corvacho J, Vergara-Garcia D, Riveros WM, Torres J. Tuberculoma in the Fourth Ventricle: An Unusual Location. Turk Neurosurg. 2021;31:658-660. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 14. | Yilmazlar S, Aksoy K. Approach via the floor of the fourth ventricle for hydatid cyst of the pons. Pediatr Neurosurg. 1999;31:326-329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 15. | Muly S, Liu S, Lee R, Nicolaou S, Rojas R, Khosa F. MRI of intracranial intraventricular lesions. Clin Imaging. 2018;52:226-239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 29] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 16. | Kimura-Hayama ET, Higuera JA, Corona-Cedillo R, Chávez-Macías L, Perochena A, Quiroz-Rojas LY, Rodríguez-Carbajal J, Criales JL. Neurocysticercosis: radiologic-pathologic correlation. Radiographics. 2010;30:1705-1719. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 113] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 17. | Mullins C, Muniz JA, Sandoval H, Padilla O, Ramos-Duran L. Neurocysticercosis of the fourth ventricle associated with hydrocephalus. Proc (Bayl Univ Med Cent). 2017;30:365-367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 18. | García HH, Evans CA, Nash TE, Takayanagui OM, White AC Jr, Botero D, Rajshekhar V, Tsang VC, Schantz PM, Allan JC, Flisser A, Correa D, Sarti E, Friedland JS, Martinez SM, Gonzalez AE, Gilman RH, Del Brutto OH. Current consensus guidelines for treatment of neurocysticercosis. Clin Microbiol Rev. 2002;15:747-756. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 302] [Cited by in RCA: 287] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 19. | Rajshekhar V. Surgical management of neurocysticercosis. Int J Surg. 2010;8:100-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 65] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 20. | Goel RK, Ahmad FU, Vellimana AK, Suri A, Chandra PS, Kumar R, Sharma BS, Mahapatra AK. Endoscopic management of intraventricular neurocysticercosis. J Clin Neurosci. 2008;15:1096-1101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 44] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 21. | Kaif M, Husain M, Ojha BK. Endoscopic Management of Intraventricular Neurocysticercosis. Turk Neurosurg. 2019;29:59-65. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 22. | Coyle CM. Neurocysticercosis: an update. Curr Infect Dis Rep. 2014;16:437. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 23. | Madrazo I, García-Rentería JA, Sandoval M, López Vega FJ. Intraventricular cysticercosis. Neurosurgery. 1983;12:148-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 73] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 24. | American Academy of Neurology. Evidence-based guideline: Treatment of parenchymal neurocysticercosis. [cited 1 August 2018]. Available from: https://www.aan.com/Guidelines/home/GetGuidelineContent/592. |
| 25. | Baird RA, Wiebe S, Zunt JR, Halperin JJ, Gronseth G, Roos KL. Evidence-based guideline: treatment of parenchymal neurocysticercosis [RETIRED]: report of the Guideline Development Subcommittee of the American Academy of Neurology. Neurology. 2013;80:1424-1429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 87] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 26. | Garcia HH, Pretell EJ, Gilman RH, Martinez SM, Moulton LH, Del Brutto OH, Herrera G, Evans CA, Gonzalez AE; Cysticercosis Working Group in Peru. A trial of antiparasitic treatment to reduce the rate of seizures due to cerebral cysticercosis. N Engl J Med. 2004;350:249-258. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 270] [Cited by in RCA: 238] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 27. | White AC Jr, Coyle CM, Rajshekhar V, Singh G, Hauser WA, Mohanty A, Garcia HH, Nash TE. Diagnosis and Treatment of Neurocysticercosis: 2017 Clinical Practice Guidelines by the Infectious Diseases Society of America (IDSA) and the American Society of Tropical Medicine and Hygiene (ASTMH). Am J Trop Med Hyg. 2018;98:945-966. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 82] [Cited by in RCA: 95] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 28. | Nash TE, Ware JM, Mahanty S. Intraventricular Neurocysticercosis: Experience and Long-Term Outcome from a Tertiary Referral Center in the United States. Am J Trop Med Hyg. 2018;98:1755-1762. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 41] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 29. | Ta R, Blond BN. The prevalence of and contributors to neurocysticercosis in endemic regions. J Neurol Sci. 2022;441:120393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 30. | Garcia HH, Gonzalez AE, Gilman RH. Taenia solium Cysticercosis and Its Impact in Neurological Disease. Clin Microbiol Rev. 2020;33:e00085-e00019. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 98] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 31. | Loyo M, Kleriga E, Estañol B. Fourth ventricular cysticercosis. Neurosurgery. 1980;7:456-458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 31] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 32. | Apuzzo ML, Dobkin WR, Zee CS, Chan JC, Giannotta SL, Weiss MH. Surgical considerations in treatment of intraventricular cysticercosis. An analysis of 45 cases. J Neurosurg. 1984;60:400-407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 100] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 33. | Hanak BW, Walcott BP, Codd PJ, Jones PS, Nahed BV, Butler WE, Asaad WF. Fourth ventricular neurocystercercosis presenting with acute hydrocephalus. J Clin Neurosci. 2011;18:867-869. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 11] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 34. | Franko LR, Pandian B, Gupta A, Savastano LE, Chen KS, Riddell J, Orringer DA. Posterior Fossa Craniotomy for Adherent Fourth Ventricle Neurocysticercosis. Oper Neurosurg. 2019;16:E154-E158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 35. | Simão D, Teixeira JC, Campos AR, Coiteiro D, Santos MM. Fourth ventricle neurocysticercosis: A case report. Surg Neurol Int. 2018;9:201. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |