Published online Apr 16, 2026. doi: 10.12998/wjcc.v14.i11.119699
Revised: February 20, 2026
Accepted: March 12, 2026
Published online: April 16, 2026
Processing time: 65 Days and 7.9 Hours
A considerable proportion of patients diagnosed with hypertrophic cardiomyopathy (HCM) or HCM-like phenotypes lack an identifiable genetic mutation and are classified as non-familial cases. This observation suggests that hormonal, aut
A 45-year-old woman with a history of hypertension and hypothyroidism was evaluated and found to have hypokalemia, metabolic alkalosis, and asymmetric left ventricular hypertrophy. Transthoracic echocardiography revealed dynamic left ventricular outflow tract obstruction, while cardiac magnetic resonance ima
Endocrine and autoimmune disorders should be considered in patients presenting with HCM-like phenotypes.
Core Tip: Hypertrophic cardiomyopathy-like phenotypes without identifiable genetic mutations may arise from secondary systemic conditions rather than primary sarcomeric disease alone. This case highlights the rare coexistence of primary hyperaldosteronism, Sjögren’s syndrome, and obstructive uropathy contributing synergistically to myocardial hypertrophy and fibrosis. Recognition of endocrine and autoimmune contributors is essential, as targeted treatment may help limit disease progression and improve outcomes.
- Citation: Aydoğan E, Ülke S, Yumuşak P, Uygun İlikhan S, Karaahmetoğlu S. Hypertrophic cardiomyopathy in the context of primary hyperaldosteronism, Sjögren’s syndrome, and obstructive uropathy: A case report. World J Clin Cases 2026; 14(11): 119699
- URL: https://www.wjgnet.com/2307-8960/full/v14/i11/119699.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i11.119699
A substantial proportion of patients presenting with hypertrophic cardiomyopathy (HCM) do not have an identifiable sarcomeric mutation or a family history, suggesting that endocrine, autoimmune, and other systemic mechanisms may contribute to an HCM-like phenotype in selected individuals[1-3]. Chronic hormonal activation and immune-mediated inflammation can promote endothelial dysfunction, microvascular ischemia, and interstitial fibrosis, leading to secondary myocardial hypertrophy and, in some cases, dynamic left ventricular outflow tract (LVOT) obstruction[2,3]. Primary hyperaldosteronism (PHA) is a common and potentially treatable cause of secondary hypertension; its prevalence among hypertensive patients ranges from 4.6% to 13.0%, increasing to approximately 20% in those with resistant hypertension[4,5]. Because excess aldosterone is associated with adverse cardiovascular remodeling and fibrosis, early recognition is critical. Sjögren’s syndrome (SS) is a chronic autoimmune exocrinopathy that may involve multiple organs, and emerging data suggest an increased cardiovascular risk in SS, although overt cardiac involvement remains uncommon[6]. Here, we report a rare and clinically instructive case of an HCM-like phenotype in the setting of concomitant PHA, SS, and ob
A 45-year-old woman with known hypertension and hypothyroidism underwent routine assessment. The patient was admitted for routine evaluation and was asymptomatic at presentation.
Laboratory testing revealed hypokalemia and metabolic alkalosis, prompting further investigation.
The patient had a history of hypothyroidism treated with levothyroxine sodium. She had previously undergone me
There was no known family history of cardiomyopathy or sudden cardiac death.
Blood pressure was 169/97 mmHg, and heart rate was 60 beats per minute. No signs of heart failure were observed on physical examination.
Laboratory evaluation demonstrated hypokalemia (3.0 mEq/L), metabolic alkalosis (pH 7.49; bicarbonate 32.9 mmol/L), suppressed renin levels, and markedly elevated aldosterone concentrations with an increased aldosterone-to-renin ratio. Autoimmune serology was positive for antinuclear antibodies, anti-SS-A, anti-SS-B, and anti-Ro-52 antibodies. Given spontaneous hypokalemia, markedly elevated aldosterone levels, and suppressed renin activity, the biochemical findings were considered diagnostic of PHA; therefore, confirmatory suppression testing was not performed in accordance with current guideline recommendations.
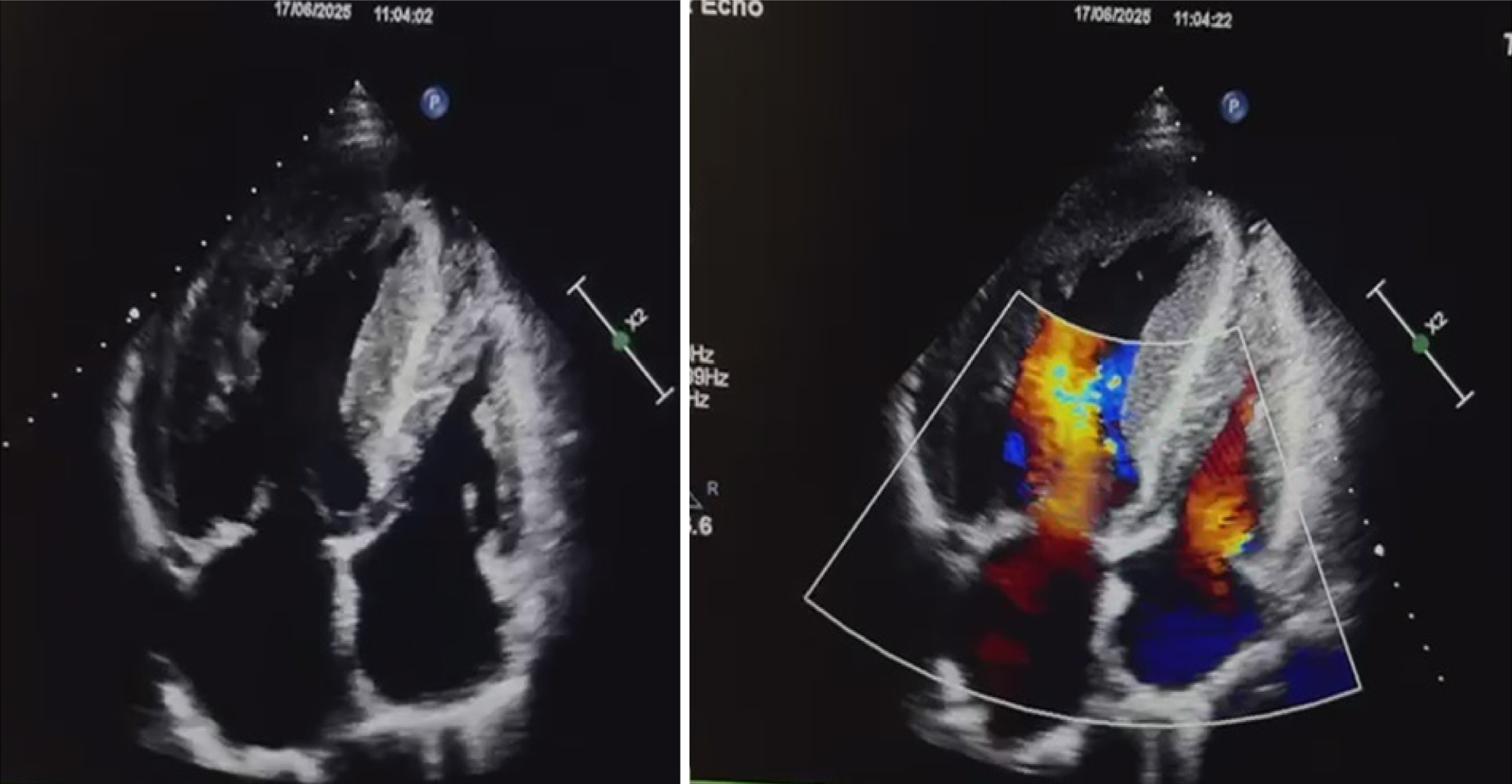
Electrocardiography demonstrated left ventricular hypertrophy with prominent U waves. Transthoracic echocardiography revealed asymmetric septal hypertrophy and dynamic LVOT obstruction, with a resting peak gradient of 90 mmHg, increasing to 130 mmHg during the Valsalva maneuver (Figure 1). Left ventricular systolic function was pre
HCM-like phenotype associated with PHA, SS, and obstructive uropathy.
The patient was started on spironolactone for the management of PHA. Hydroxychloroquine therapy was initiated following the diagnosis of SS. Cardiac management followed guideline-directed therapy for obstructive HCM.
The patient was followed for 9 months under multidisciplinary care involving cardiology, endocrinology, nephrology, and rheumatology services (Table 1). Blood pressure and serum potassium levels improved following mineralocorticoid receptor antagonist therapy, and the patient remained clinically stable without heart failure symptoms. However, repeat imaging has not yet demonstrated significant regression of septal hypertrophy.
| Time point | Clinical event |
| Initial evaluation | Routine assessment revealed hypokalemia and metabolic alkalosis |
| Week 1 | Hormonal evaluation demonstrated markedly elevated aldosterone levels with suppressed renin activity, confirming primary hyperaldosteronism |
| Week 2 | Transthoracic echocardiography revealed asymmetric septal hypertrophy and dynamic LVOT obstruction (resting gradient 90 mmHg; 130 mmHg with Valsalva) |
| Week 3 | Cardiac magnetic resonance imaging confirmed septal thickness of 26 mm, preserved ejection fraction (72%), and mid-wall late gadolinium enhancement |
| Week 4 | Autoimmune serology and minor salivary gland biopsy established Sjögren’s syndrome |
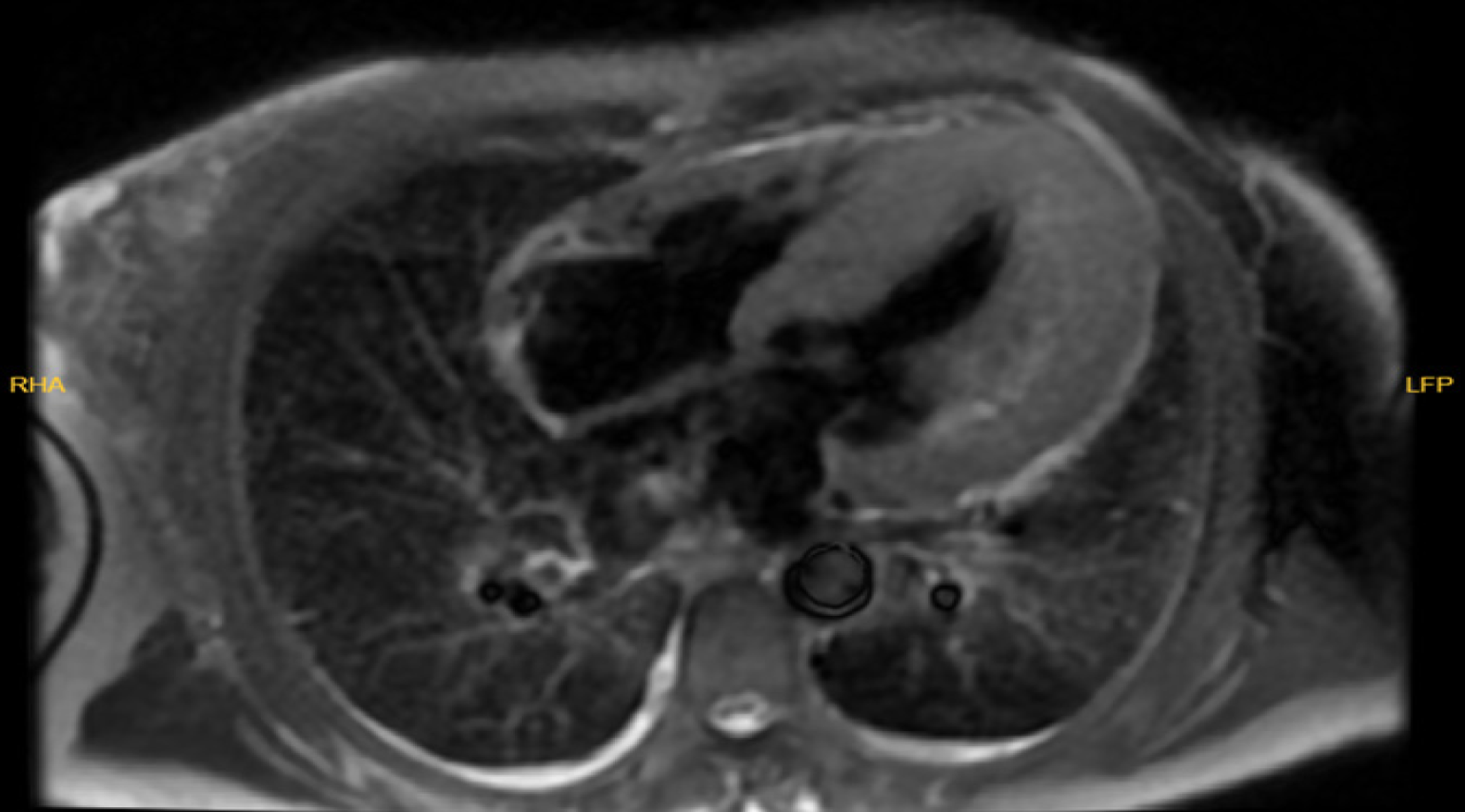
| Week 5 | Abdominal imaging identified right adrenal adenoma and left-sided obstructive uropathy |
| Week 6 | Spironolactone therapy was initiated; hydroxychloroquine treatment was started |
| Month 3 | Blood pressure and serum potassium levels improved under therapy |
| Month 9 | Clinical stability was maintained; repeat imaging showed no significant regression of septal hypertrophy |
HCM represents a heterogeneous clinical spectrum that includes inherited sarcomeric disease as well as non-familial phenotypes driven by systemic conditions. In patients without a clear family history or identifiable genetic variants, endocrine and immune-mediated mechanisms may promote hypertrophic remodeling and myocardial fibrosis, resulting in an “HCM-like phenotype” rather than primary sarcomeric HCM[1-3].
This case is notable for the rare coexistence of PHA, SS, and obstructive uropathy in a patient with asymmetric hyp
Excess aldosterone in PHA contributes not only to sodium retention and hypertension but also to myocardial fibrosis and structural remodeling via mineralocorticoid receptor–mediated pathways[7,8]. Patients with PHA have a higher burden of cardiometabolic events compared with those with essential hypertension, even when blood pressure levels are comparable, supporting a direct pathogenic role of aldosterone in adverse cardiovascular remodeling[7]. Mineralocorticoid receptor antagonists may mitigate remodeling and improve outcomes, reinforcing the importance of timely re
SS is primarily an autoimmune exocrinopathy; however, emerging data suggest increased cardiovascular risk, and reported cardiac manifestations include diastolic dysfunction, myocarditis, and arrhythmias[6,10,11]. Chronic systemic inflammation, endothelial dysfunction, and microvascular ischemia may plausibly contribute to hypertrophic remodeling and fibrosis, potentially amplifying the effects of aldosterone excess in susceptible patients.
The patient also had obstructive uropathy with marked unilateral renal dysfunction. Chronic renal impairment can contribute to cardiovascular stress through neurohormonal activation and increased afterload; nevertheless, the rela
Alternative explanations must be considered, particularly long-standing hypertension as an independent driver of left ventricular hypertrophy and even dynamic obstruction in some patients. However, the asymmetric pattern of hy
Clinically, this case underscores the importance of differentiating true sarcomeric HCM from secondary hypertrophic remodeling with obstruction. In patients with hypertrophic phenotypes accompanied by resistant hypertension, hy
This report has several limitations. First, genetic testing for sarcomeric mutations was not available; therefore, a genetic etiology cannot be completely excluded. Second, the relationship between obstructive uropathy and myocardial hyper
This case underscores the importance of considering endocrine and autoimmune disorders in patients presenting with unexplained left ventricular hypertrophy and obstructive physiology. Early recognition of potentially reversible con
| 1. | Basit H, Alahmadi MH, Rout P, Sharma S. Hypertrophic Cardiomyopathy. 2024 Jun 7. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2026 Jan. [PubMed] |
| 2. | Marian AJ. On genetic and phenotypic variability of hypertrophic cardiomyopathy: nature versus nurture. J Am Coll Cardiol. 2001;38:331-334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 29] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 3. | Weidemann F, Störk S, Herrmann S, Ertl G, Niemann M. [The various forms of left ventricular hypertrophy: diagnostic value of echocardiography]. Herz. 2011;36:713-723. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 4. | Eide IK, Torjesen PA, Drolsum A, Babovic A, Lilledahl NP. Low-renin status in therapy-resistant hypertension: a clue to efficient treatment. J Hypertens. 2004;22:2217-2226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 183] [Article Influence: 8.3] [Reference Citation Analysis (1)] |
| 5. | Calhoun DA, Nishizaka MK, Zaman MA, Harding SM. Aldosterone excretion among subjects with resistant hypertension and symptoms of sleep apnea. Chest. 2004;125:112-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 167] [Article Influence: 7.6] [Reference Citation Analysis (1)] |
| 6. | Farrukh L, Mumtaz A, Wajid S, Waqar HH, Peredo-Wende R. Cardiac Manifestations of Sjogren's Syndrome: A Review of Literature. Cureus. 2023;15:e41002. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 7. | Hundemer GL, Curhan GC, Yozamp N, Wang M, Vaidya A. Cardiometabolic outcomes and mortality in medically treated primary aldosteronism: a retrospective cohort study. Lancet Diabetes Endocrinol. 2018;6:51-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 402] [Cited by in RCA: 530] [Article Influence: 66.3] [Reference Citation Analysis (1)] |
| 8. | Funder JW, Carey RM, Mantero F, Murad MH, Reincke M, Shibata H, Stowasser M, Young WF Jr. The Management of Primary Aldosteronism: Case Detection, Diagnosis, and Treatment: An Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2016;101:1889-1916. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2449] [Cited by in RCA: 2101] [Article Influence: 210.1] [Reference Citation Analysis (1)] |
| 9. | Tang K, Liu S, Yang S, Yuan Q, Du Z. Diagnosis of hypertrophic cardiomyopathy accompanied with primary aldosteronism-Case report. Clin Case Rep. 2023;11:e8218. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 10. | Khalid H, Wang W, Ruzieh M. Dysphagia aortica presenting as failure of performing transoesophageal echocardiogram. Eur Heart J Case Rep. 2024;8:ytae007. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 11. | Chung MC, Lin CL, Wu MJ, Chen CH, Shieh JJ, Chung CJ, Li CY, Yu TM. Primary aldosteronism is associated with risk of urinary bladder stones in a nationwide cohort study. Sci Rep. 2021;11:7684. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 12. | Martin SS, Aday AW, Almarzooq ZI, Anderson CAM, Arora P, Avery CL, Baker-Smith CM, Barone Gibbs B, Beaton AZ, Boehme AK, Commodore-Mensah Y, Currie ME, Elkind MSV, Evenson KR, Generoso G, Heard DG, Hiremath S, Johansen MC, Kalani R, Kazi DS, Ko D, Liu J, Magnani JW, Michos ED, Mussolino ME, Navaneethan SD, Parikh NI, Perman SM, Poudel R, Rezk-Hanna M, Roth GA, Shah NS, St-Onge MP, Thacker EL, Tsao CW, Urbut SM, Van Spall HGC, Voeks JH, Wang NY, Wong ND, Wong SS, Yaffe K, Palaniappan LP; American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. 2024 Heart Disease and Stroke Statistics: A Report of US and Global Data From the American Heart Association. Circulation. 2024;149:e347-e913. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2192] [Cited by in RCA: 1807] [Article Influence: 903.5] [Reference Citation Analysis (0)] |