Published online Apr 16, 2026. doi: 10.12998/wjcc.v14.i11.119674
Revised: March 4, 2026
Accepted: March 16, 2026
Published online: April 16, 2026
Processing time: 65 Days and 16.9 Hours
Corynebacterium striatum is a non-diphtherial corynebacterium commonly reg
A 46-year-old man with decompensated chronic liver disease (Child-Pugh C; Model for End-Stage Liver Disease-Na 36 on admission) and end-stage renal dis
Corynebacterium striatum should not be dismissed as a contaminant when isolated repeatedly from blood cultures in vulnerable patients. Early recognition, prompt echocardiographic evaluation, and aggressive multidisciplinary management are essential, as this seemingly be
Core Tip: Corynebacterium striatum, traditionally dismissed as a blood culture contaminant, can act as an aggressive endovascular pathogen in immunocompromised and dialysis-dependent patients. This case highlights how initial clinical quiescence and negative early investigations may delay recognition of infective endocarditis. However, serial blood cultures and repeat echocardiography were crucial in establishing the diagnosis. Persistent isolation of Corynebacterium striatum in the appropriate clinical context should prompt early consideration of infective endocarditis, timely antimicrobial opti
- Citation: Kataria S, Tiwari E, Ray S, Juneja D. Corynebacterium striatum and the deceptive diagnosis in a dialysis-dependent chronic liver disease patient: A case report. World J Clin Cases 2026; 14(11): 119674
- URL: https://www.wjgnet.com/2307-8960/full/v14/i11/119674.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i11.119674
Corynebacterium striatum is a non-diphtherial corynebacterium that forms part of the normal flora of the skin and upper respiratory tract and was long regarded as a benign contaminant when isolated from clinical specimens[1,2]. Over the past two decades, however, Corynebacterium striatum has emerged as an opportunistic pathogen capable of causing invasive infections, including pneumonia, meningitis, osteomyelitis, bloodstream infections, and both native and pro
A 46-year-old man presented with acute onset dyspnoea three days after discharge from a tertiary-care hospital, where he had been admitted for six weeks for severe bilateral lower-limb cellulitis requiring surgical debridement. That admission was complicated by acute-on-chronic renal failure, refractory fluid overload, malnutrition, and difficult vascular access, necessitating placement of a tunnelled right internal jugular haemodialysis catheter. During that hospitalization, he was treated with intravenous ceftriaxone and clindamycin. Blood and pus cultures at that time were sterile. No antibiotics were continued after discharge, and only local wound dressings were maintained.
Progressive shortness of breath and cough.
The patient had decompensated chronic liver disease due to alcohol-related cirrhosis (Child-Pugh class C), end-stage renal disease on maintenance haemodialysis, refractory ascites, recurrent hepatic encephalopathy, and a recent history of urethral stricture managed with cystoscopic dilatation.
There was no significant family history. The patient had a long-standing history of alcohol use disorder.
On admission, the patient was afebrile (36.8 °C) but tachycardic (104 beats/minute), tachypnoeic (28 breaths/minute), and hypotensive (100/60 mmHg). Oxygen saturation was 93% on room air. Examination revealed elevated jugular venous pressure, bilateral basal crepitations, tense ascites, and peripheral oedema. No peripheral stigmata of infective endocarditis were noted.
On admission, haemoglobin was 7.9 g/dL, platelet count 70 × 109/L, creatinine 9.0 mg/dL (on haemodialysis), sodium 125 mmol/L, and metabolic acidosis was present. Total bilirubin was 6 mg/dL (direct 4 mg/dL), albumin 1.4 g/dL, and international normalized ratio was 1.81. The Model for End-Stage Liver Disease-Na (MELD-Na) was 36. Leukocyte count was normal. Blood, urine, and ascitic cultures were sterile.
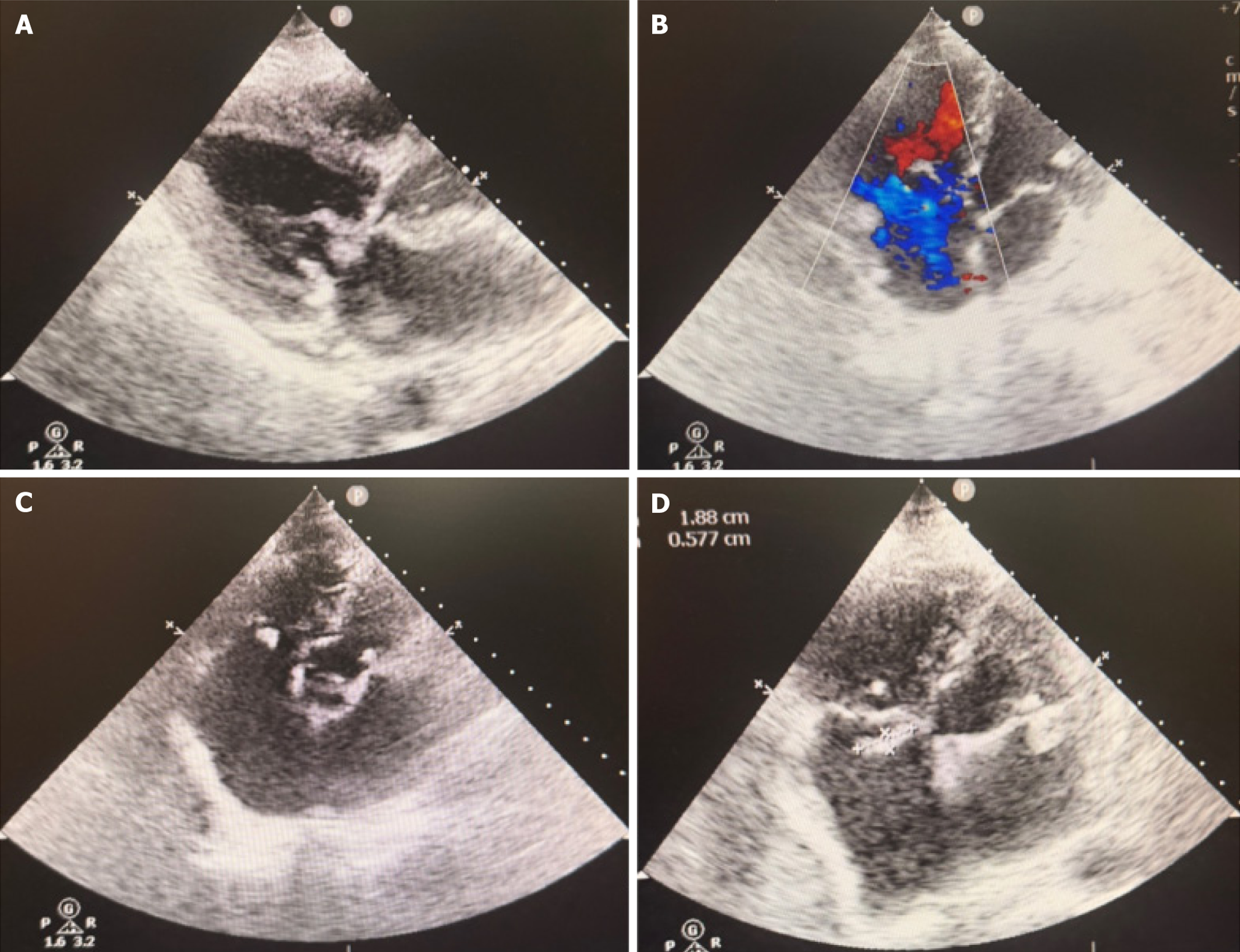
Baseline transthoracic echocardiography performed at admission, as well as echocardiographic imaging from the pre
During the first nine days of admission, the patient remained afebrile and haemodynamically stable with supportive care, and intermittent haemodialysis was continued for management of volume overload. No focal source of infection was identified during this period. Persistently elevated inflammatory markers were initially attributed to advanced cirrhosis and ongoing wound inflammation related to recent lower-limb cellulitis requiring continued dressing care, in the absence of clinical or microbiological evidence of sepsis. On hospital day 10, the patient developed a sudden high-grade fever and refractory septic shock with haemodynamic collapse. In view of the indwelling dialysis catheter and severe immunocompromise, paired blood cultures were obtained simultaneously from separate peripheral venepuncture sites and the tunnelled dialysis catheter lumen under strict aseptic precautions.
As part of point-of-care ultrasonography performed for evaluation of new-onset shock, urgent transthoracic echocardiography revealed mobile hyperechoic vegetations on the anterior mitral leaflet measuring 0.8 cm × 1.1 cm and on the septal leaflet of the tricuspid valve measuring 2.0 cm × 0.6 cm, associated with severe mitral regurgitation and moderate-to-severe tricuspid regurgitation (Figure 1). Given that transthoracic echocardiography at admission and during the prior hospitalisation had shown no valvular abnormalities, these findings confirmed the dynamic development of endocarditis during the current admission. A second paired set of blood cultures was obtained immediately following the echocardiographic findings.
Both peripheral and catheter-drawn cultures yielded Corynebacterium striatum, identified using the VITEK automated system. The catheter-drawn sample flagged positive approximately 30 minutes earlier than the peripheral sample. Although the organism was initially interpreted cautiously because of its commensal nature, concordant growth from multiple paired blood cultures obtained on the same diagnostic day, together with compatible echocardiographic findings and septic shock, established the diagnosis of multivalvular infective endocarditis. Transoesophageal echocardiography was deferred due to thrombocytopenia (platelet count 70 × 109/L) and coagulopathy (international normalized ratio: 2.68), given the procedural bleeding risk.
Multivalvular infective endocarditis caused by Corynebacterium striatum.
Empirical broad-spectrum antimicrobial therapy with meropenem and teicoplanin was initiated immediately after blood cultures were obtained, given the severity of septic shock and the echocardiographic findings. Once blood cultures identified Corynebacterium striatum, antimicrobial therapy was streamlined to intravenous vancomycin, based on susceptibility results. Vancomycin dosing was guided by therapeutic drug monitoring, with a trough concentration of 24.9 μg/mL, consistent with recommended targets for infective endocarditis. Despite appropriate therapy, paired blood cultures obtained on days 3 and 5 after vancomycin initiation continued to yield Corynebacterium striatum, indicating persistent bacteraemia. In view of persistent bacteraemia, adjunctive daptomycin was introduced empirically. The tunnelled dialysis catheter was removed due to concern for a possible catheter-associated source and replaced with a temporary catheter to continue haemodialysis. Catheter tip culture was not sent. Antimicrobial susceptibility testing was performed by disc diffusion; minimum inhibitory concentration values were not available for this isolate. Linezolid was not initiated due to pre-existing thrombocytopenia and advanced hepatic dysfunction, given the risk of further myelosuppression. Blood cultures subsequently cleared after 7 days of targeted therapy. Cardiothoracic surgical consultation was obtained. At that stage, the MELD-Na score was 40. Given advanced cirrhosis, ongoing septic shock, coagulopathy, and severe frailty, surgical intervention was not undertaken. The patient continued to deteriorate despite intensive medical management.
Despite appropriate antimicrobial therapy and maximal supportive care, the patient developed progressive multiorgan failure and died on hospital day 22.
Infective endocarditis caused by Corynebacterium striatum remains rare; however, this organism has increasingly transitioned from a presumed contaminant to an emerging pathogen of clinical importance in high-risk patient popu
Our patient had several well-recognised predisposing factors for invasive Corynebacterium striatum infection, including dialysis dependence with an indwelling vascular catheter, advanced chronic liver disease, recent prolonged hospitalisation, and multiple invasive procedures. Dialysis-dependent patients have a markedly increased risk of infective endocarditis compared with the general population, with reported incidence rates of approximately 200-300 cases per 100000 patient-years, nearly 20-40 times higher than in non-dialysis populations[11]. In parallel, cirrhosis is associated with cirrhosis-associated immune dysfunction, characterised by impaired opsonic activity, defective phagocyte function, and altered innate and adaptive immune responses, predisposing to bacteraemia and severe infections[12]. The con
This case highlights several important diagnostic challenges. The initial clinical course was atypical, with persistent inflammatory markers in the absence of fever or overt haemodynamic instability. Blunted inflammatory and febrile responses are well recognised in immunocompromised hosts, including patients with advanced cirrhosis, and may delay clinical suspicion of infective endocarditis[12]. Baseline and prior echocardiography were normal, indicating that the endocarditic process developed dynamically during the course of the current admission. The abrupt onset of septic shock appropriately prompted repeat blood cultures and urgent echocardiographic reassessment, which revealed new multi
Distinguishing true infection from contamination remains a central challenge when corynebacteria are isolated from blood cultures. Because Corynebacterium striatum colonises skin and mucosal surfaces, single positive cultures are frequently dismissed as contaminants. However, clinical studies demonstrate that a substantial proportion of Corynebacterium bloodstream isolates represent true bacteraemia, particularly when recovered from paired blood cultures in high-risk patients or when associated with intravascular devices[6,7]. In the present case, paired blood cultures obtained following echocardiographic detection of new valvular vegetations both yielded Corynebacterium striatum. Although the initial isolate was interpreted cautiously, given its commensal status, concordant growth in multiple paired culture sets obtained on the same diagnostic day, together with compatible echocardiographic findings and septic shock, established the diagnosis of infective endocarditis rather than contamination. Although the catheter-drawn sample flagged positive slightly earlier than the peripheral sample, the differential time to positivity was not sufficient to define catheter-related bloodstream infection and was not used as the basis for diagnosis. Subsequent serial blood cultures were appropriately used to assess microbiological response to therapy.
Accurate species-level identification of corynebacteria is essential but may be problematic with conventional laboratory techniques. Corynebacterium striatum shares the characteristic “diphtheroid” morphology with other corynebacterial species, and misidentification remains possible using routine biochemical methods. In this case, species-level identification was performed using the VITEK automated system. Antimicrobial susceptibility testing was conducted by disc diffusion according to laboratory protocol; minimum inhibitory concentration values were not available. The increasing availability of matrix-assisted laser desorption/ionisation time-of-flight mass spectrometry and molecular techniques such as 16S rRNA gene sequencing has markedly improved diagnostic accuracy. It is likely contributing to the growing recognition of Corynebacterium striatum as a true pathogen[1,5]. Several reported cases of Corynebacterium striatum endocarditis were definitively identified only after advanced microbiological analysis following initial misclassification[5].
Another notable feature of this case was multivalvular involvement. Multivalvular infective endocarditis is uncommon and is associated with more severe disease and poorer outcomes. In published series of Corynebacterium striatum endo
Management of Corynebacterium striatum endocarditis is challenging due to its propensity for multidrug resistance and biofilm formation. Contemporary isolates commonly exhibit resistance to β-lactams, macrolides, fluoroquinolones, clindamycin, and tetracyclines, leaving glycopeptides and oxazolidinones as the principal therapeutic options[1,2,13,14]. Vancomycin remains the cornerstone of therapy and was appropriately administered in this case with therapeutic drug monitoring. Persistent bacteraemia despite adequate vancomycin levels likely reflected established endocardial infection with large vegetations rather than inadequate antimicrobial exposure or ongoing catheter-related seeding, as the dialysis catheter had already been removed. Although alternative agents such as linezolid, rifampicin, or daptomycin have been successfully employed in selected refractory cases, optimal antimicrobial regimens remain poorly defined due to the rarity of this condition[13,14].
Adjunctive daptomycin was initiated due to ongoing bacteraemia. While daptomycin has been used successfully in selected cases of Corynebacterium striatum endocarditis, emerging reports describe reduced susceptibility and clinical resistance in some isolates, particularly after prior glycopeptide exposure[15]. Optimal regimens remain undefined given the rarity of the condition. Linezolid was not initiated because of baseline thrombocytopenia and advanced hepatic dysfunction, where additional myelosuppressive toxicity would have posed a significant risk.
Surgical intervention is often critical in complicated infective endocarditis, particularly in the presence of large vegetations, multivalvular involvement, heart failure, or persistent bacteraemia. Approximately 40%-50% of reported Corynebacterium striatum endocarditis cases have required valve surgery, with improved outcomes observed in patients receiving combined medical-surgical therapy[3,9]. Even with early source control, outcomes in such severely immunocompromised hosts remain poor. In this patient, cardiothoracic consultation was obtained. The MELD-Na score was 40 in the setting of septic shock and multiorgan dysfunction. Surgical intervention was not undertaken because of the high operative risk. Mortality in such cases reflects the combined impact of severe host factors and extensive valvular involvement. Mortality associated with Corynebacterium striatum endocarditis remains substantial, particularly among patients with delayed diagnosis, extensive valvular destruction, or inability to undergo surgical intervention[3,9]. The fatal outcome in our patient underscores the aggressive nature of this infection in immunocompromised hosts and high
From a broader perspective, this case has important implications for infection control and antimicrobial stewardship. Nosocomial transmission and hospital outbreaks of multidrug-resistant Corynebacterium striatum have been documented, particularly in dialysis units and intensive care settings[1,5]. Strict adherence to catheter care protocols, hand hygiene, and early removal of unnecessary intravascular devices remains essential to mitigate risk.
In summary, this case reinforces several key principles. High-risk patients with indwelling vascular devices and immune dysfunction are susceptible to invasive infection by Corynebacterium striatum. Repeated isolation of this organism from paired blood cultures in the appropriate clinical context should not be dismissed as contamination but should prompt thorough evaluation for infective endocarditis. Advanced microbiological identification techniques are invaluable for accurate diagnosis. Given the organism’s multidrug resistance, biofilm-forming capacity, and association with poor outcomes, early recognition, targeted antimicrobial therapy, and prompt multidisciplinary surgical assessment, when feasible, are critical to improving prognosis in this emerging and potentially lethal infection.
Corynebacterium striatum should no longer be considered a benign commensal in high-risk settings. This case un
| 1. | Campanile F, Carretto E, Barbarini D, Grigis A, Falcone M, Goglio A, Venditti M, Stefani S. Clonal multidrug-resistant Corynebacterium striatum strains, Italy. Emerg Infect Dis. 2009;15:75-78. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 58] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 2. | Datta P, Gupta V, Gupta M, Pal K, Chander J. Corynebacterium Striatum, an Emerging Nosocomial Pathogen: Case Reports. Infect Disord Drug Targets. 2021;21:301-303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 3. | Hong M, Xiang W, Kong L, Wang X, Fu L, Chen L, Liu F. Corynebacterium Striatum Infective Endocarditis: A Systematic Review. J Inflamm Res. 2025;18:17309-17318. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 4. | Otsuka Y, Ohkusu K, Kawamura Y, Baba S, Ezaki T, Kimura S. Emergence of multidrug-resistant Corynebacterium striatum as a nosocomial pathogen in long-term hospitalized patients with underlying diseases. Diagn Microbiol Infect Dis. 2006;54:109-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 77] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 5. | Orosz L, Sóki J, Kókai D, Burián K. Corynebacterium striatum-Got Worse by a Pandemic? Pathogens. 2022;11:685. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 6. | Yanai M, Ogasawasa M, Hayashi Y, Suzuki K, Takahashi H, Satomura A. Retrospective evaluation of the clinical characteristics associated with Corynebacterium species bacteremia. Braz J Infect Dis. 2018;22:24-29. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 7. | Martínez-Martínez L, Suárez AI, Rodríguez-Baño J, Bernard K, Muniáin MA. Clinical significance of Corynebacterium striatum isolated from human samples. Clin Microbiol Infect. 1997;3:634-639. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 43] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 8. | Fowler VG, Durack DT, Selton-Suty C, Athan E, Bayer AS, Chamis AL, Dahl A, DiBernardo L, Durante-Mangoni E, Duval X, Fortes CQ, Fosbøl E, Hannan MM, Hasse B, Hoen B, Karchmer AW, Mestres CA, Petti CA, Pizzi MN, Preston SD, Roque A, Vandenesch F, van der Meer JTM, van der Vaart TW, Miro JM. The 2023 Duke-International Society for Cardiovascular Infectious Diseases Criteria for Infective Endocarditis: Updating the Modified Duke Criteria. Clin Infect Dis. 2023;77:518-526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 287] [Cited by in RCA: 545] [Article Influence: 181.7] [Reference Citation Analysis (0)] |
| 9. | Souza Cd, Faria YV, Sant'Anna Lde O, Viana VG, Seabra SH, Souza MC, Vieira VV, Hirata Júnior R, Moreira Lde O, Mattos-Guaraldi AL. Biofilm production by multiresistant Corynebacterium striatum associated with nosocomial outbreak. Mem Inst Oswaldo Cruz. 2015;110:242-248. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 61] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 10. | Cahill TJ, Prendergast BD. Infective endocarditis. Lancet. 2016;387:882-893. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 835] [Cited by in RCA: 700] [Article Influence: 70.0] [Reference Citation Analysis (0)] |
| 11. | Bhatia N, Agrawal S, Garg A, Mohananey D, Sharma A, Agarwal M, Garg L, Agrawal N, Singh A, Nanda S, Shirani J. Trends and outcomes of infective endocarditis in patients on dialysis. Clin Cardiol. 2017;40:423-429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 38] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 12. | Albillos A, Lario M, Álvarez-Mon M. Cirrhosis-associated immune dysfunction: distinctive features and clinical relevance. J Hepatol. 2014;61:1385-1396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1026] [Cited by in RCA: 921] [Article Influence: 76.8] [Reference Citation Analysis (1)] |
| 13. | Fernández Guerrero ML, Molins A, Rey M, Romero J, Gadea I. Multidrug-resistant Corynebacterium striatum endocarditis successfully treated with daptomycin. Int J Antimicrob Agents. 2012;40:373-374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 14. | Shah M, Murillo JL. Successful treatment of Corynebacterium striatum endocarditis with daptomycin plus rifampin. Ann Pharmacother. 2005;39:1741-1744. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 33] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 15. | Mitchell KF, McElvania E, Wallace MA, Droske LE, Robertson AE, Westblade LF, Burnham CA. Evaluating the Rapid Emergence of Daptomycin Resistance in Corynebacterium: a Multicenter Study. J Clin Microbiol. 2021;59:e02052-e02020. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |