Published online Apr 16, 2026. doi: 10.12998/wjcc.v14.i11.119896
Revised: February 19, 2026
Accepted: March 11, 2026
Published online: April 16, 2026
Processing time: 58 Days and 13.5 Hours
Scalp block is a well-established regional anaesthetic technique in neurosurgery, particularly for awake procedures such as deep brain stimulation (DBS). While generally safe, rare complications can occur.
This report details a case of a 39-year-old male with drug-refractory Parkinson’s disease who developed unilateral complete ptosis following a scalp block adm
This case underscores the importance of understanding the anatomical basis of scalp innervation and the potential for rare complications.
Core Tip: This case report describes a rare case of transient unilateral ptosis occurring during awake deep brain stimulation (DBS) following a scalp block. A 39-year-old man undergoing bilateral subthalamic nucleus DBS developed complete left-sided ptosis 30 minutes after local anaesthetic infiltration, with normal ocular movements and pupillary responses. Imaging excluded surgical complications, and the ptosis resolved spontaneously within 12 hours. The report highlights how inad
- Citation: Karan N, Patnaik R, Priyanka DS, Sharma AK. Transient drooping lid after scalp block during deep brain stimulation for Parkinson’s disease: A case report. World J Clin Cases 2026; 14(11): 119896
- URL: https://www.wjgnet.com/2307-8960/full/v14/i11/119896.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i11.119896
Awake neurosurgical procedures, such as deep brain stimulation (DBS), require meticulous anaesthetic management to achieve optimal analgesia while preserving patient cooperation and neurologic function. DBS is a highly precise procedure used to treat movement disorders, where the accuracy of electrode placement directly impacts clinical outcomes[1]. Performing DBS in the awake state enables real-time evaluation of motor, speech, and cognitive functions, ensuring precise electrode positioning and minimising the risk of postoperative complications. Young-onset Parkinson’s disease is relatively uncommon, accounting for only about 5%-10% of all cases. While younger patients often show different patterns of disease progression, current evidence does not establish a clear relationship between age at onset and responsiveness to medical therapy.
The scalp is richly innervated by cranial and cervical nerves, making it amenable to regional anaesthesia. A scalp block targets six principal nerves namely the supratrochlear, supraorbital, zygomaticotemporal, auriculotemporal, greater occipital, and lesser occipital nerves[2]. These nerves provide sensory innervation to the forehead, temples, lateral and posterior scalp, and areas around the ear. By blocking these nerves, effective analgesia is achieved for pin fixation, skin incision, and periosteal manipulation during neurosurgical procedures. Scalp blocks reduce the need for systemic sedatives or opioids, helping maintain patient cooperation during awake procedures such as DBS[2]. They also minimize sympathetic stimulation and improve hemodynamic stability[3]. Overall, the technique enhances patient comfort, facilitates surgical access, and allows reliable intraoperative neurologic testing.
While the scalp block is generally regarded as a safe and effective regional anaesthesia technique, potential complications have been reported. These include local anaesthetic systemic toxicity, hematoma formation, transient facial nerve palsy, and, rarely, unilateral ptosis, which is usually temporary and resolves spontaneously[4]. The objective of this case report is to describe a rare instance of transient unilateral ptosis that occurred during awake DBS following administration of a scalp block.
A man with early-onset, drug-refractory Parkinson’s disease presented with involuntary body movements for 12 years. Since he had exacerbation of symptoms in the past 5 months, he was posted for bilateral subthalamic nucleus DBS (STN-DBS).
A 39-year-old male with early-onset, drug-refractory Parkinson’s disease for 12 years presented with progressive involuntary movements and motor fluctuations despite optimal medical therapy. His clinical features consisted of resting tremor, rigidity, bradykinesia, and postural instability. He also reported orthostatic hypotension and sleep disturbances. Cognitive function was preserved, and there was no history of psychosis or dementia. He was taking tablet Carbidopa 25 mg four times daily (QID) and Levodopa 100 mg QID for the past 6 years. He was also taking tablet entacapone 200 mg QID, amantadine 100 mg three times daily (TID), pramipexole 0.5 mg twice daily (BID), trihexyphenidyl 2 mg once daily (OD), ropinirole 1 mg BID, and flupirtine 100 mg OD for the past 4 years. He was scheduled for bilateral STN-DBS.
There was no past illness.
There was no significant personal or family history.
On examination the patient was conscious, oriented, afebrile and alert. His vital signs were blood pressure of 122/88 mmHg, heart rate of 86 beats per minute, respiratory rate of 14 breaths per minute and Glasgow Coma Scale was E4V5M6. He had aggressive whole body involuntary movements.
Initial laboratory tests, including complete blood count, electrolytes, liver and renal function, and arterial blood gas analysis, were within normal limits.
Chest X-ray and head computed tomography performed on admission revealed no abnormalities.
The final diagnosis of drug-resistant young onset Parkinson’s disease was made. Bilateral STN-DBS was planned.
A day before surgery informed consent was obtained after detailed discussion of the procedure, potential risks, and expected outcomes. Preoperatively, stereotactic imaging was performed using the Brown-Roberts-Wells frame to accurately determine and confirm the three-dimensional coordinates of the STN, ensuring precise electrode placement during DBS.
On the day of surgery, patient was taken to the operation theatre. Standard anaesthesia monitors were attached and scalp block was performed using 25-gauge needle. Local infiltration was carried out meticulously. 1 mL of 0.25% bupivacaine was administered to each supraorbital and supratrochlear nerve, while 1.5 mL was injected around each of the remaining target nerves (zygomaticotemporal, auriculotemporal, lesser occipital, and greater occipital) bilaterally, yielding a total volume of approximately 16 mL. This dosage was calculated to remain within the safe upper limit of 2.5 mg/kg for bupivacaine. Sedation was initiated using intravenous dexmedetomidine, administered as a loading dose of 70 µg infused over 10 minutes, followed by a maintenance infusion at 0.5 μg/kg/hour to provide anxiolysis, cooperative sedation, and hemodynamic stability throughout the procedure.
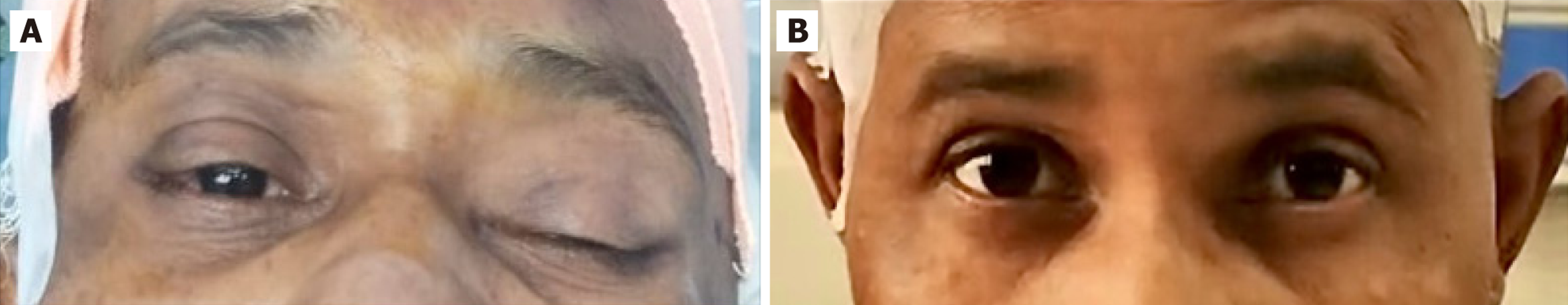
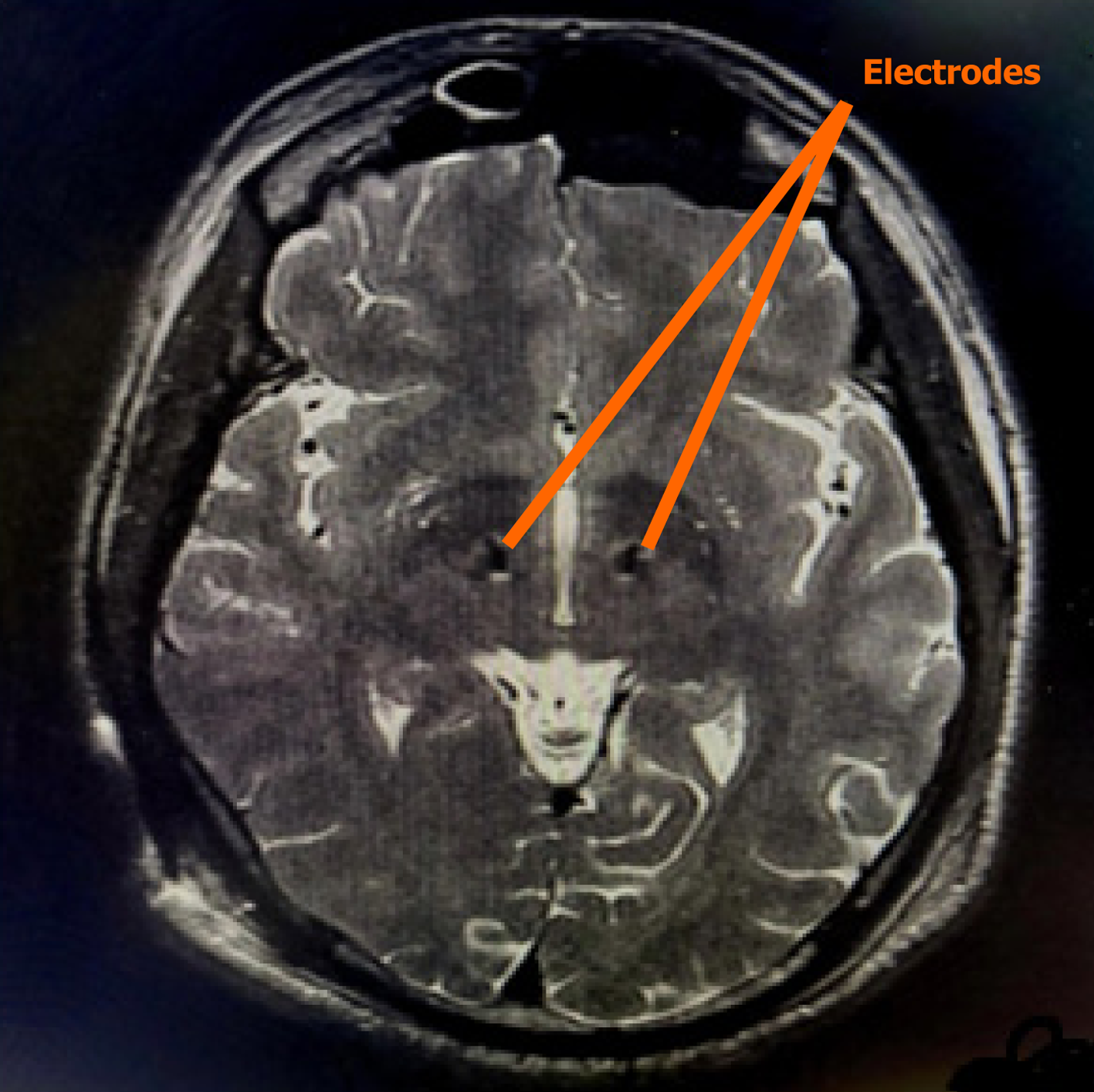
A Cosman-Roberts-Wells stereotactic frame was applied, and bilateral frontal burr holes were created. Guided by stereotactic coordinates and intraoperative neurophysiological mapping, electrodes were accurately implanted into the STN. Approximately 30 minutes into the procedure, the patient developed left-sided complete ptosis, while ocular movements and pupillary reflexes remained intact (Figure 1A). General anaesthesia, was then administered to implant the device at an infraclavicular pocket. The electrode extensions were tunnelled subcutaneously, secured in position, and connected to the pulse generator after confirming appropriate impedance. The device tested for functionality. Patient did not have any focal neurological deficits or any other intraoperative event.
Postoperative neurological assessment revealed no new deficits. The cranial nerve examination was entirely intact, specifically noting normal vision (acuity and visual fields) and intact extraocular muscle movements without diplopia. Pupillary size was equal and reactive bilaterally, and facial motor function was preserved. Higher mental functions, as well as the motor, sensory, and cerebellar systems, remained unremarkable. The patient was transferred to the neu
At the three-month follow-up, the patient showed no recurrence of ptosis during outpatient evaluation. His involuntary movements and motor fluctuations had significantly improved.
Transient unilateral ptosis following scalp block is a rare but documented complication. Hassan et al[5] reported complete mechanical ptosis following bilateral scalp block with 0.75% ropivacaine with adrenaline in an awake craniotomy. The ptosis developed about 30 minutes after block administration and resolved spontaneously by postoperative day 2 without steroid intervention.
The subgaleal space lies beneath the galea aponeurotica of the scalp and above the periosteum of the skull. It is a loose connective tissue plane that communicates with various fascial planes. The periorbital region contains delicate structures, including the levator palpebrae superioris muscle, Müller’s (superior tarsal) muscle, supraorbital and supratrochlear nerves, and sympathetic fibres controlling eyelid elevation and pupillary tone[6]. These structures are closely associated with soft tissue planes, so any infiltrated fluid can track along them leading to transient ptosis. Improper needle placement or subgaleal or periorbital infiltration can affect soft tissue planes and nearby motor or sensory nerves.
The temporal branch of the facial nerve innervates the frontalis and orbicularis oculi muscles, playing a key role in elevating the eyebrows and controlling eyelid closure. During a scalp block, inadvertent spread of local anaesthetic, particularly in the region of the auriculotemporal nerve, can temporarily anesthetize or impair this branch[7]. This may manifest as a transient drooping of the upper eyelid, clinically resembling ptosis. Such effects are typically short-lived, resolving spontaneously as the local anaesthetic wears off, and are considered a benign, self-limiting complication of the block.
During DBS surgery, burr holes are created in the skull to allow insertion of electrodes into target structures such as the STN. The STN and surrounding basal ganglia structures are located deep within the brain and are not directly involved in the neural pathways controlling eyelid elevation, which are primarily mediated by the levator palpebrae superioris (oculomotor nerve) and Müller’s muscle (sympathetic fibres). Therefore, unilateral ptosis resulting directly from the surgical procedure is highly unlikely[8]. Postoperative imaging, such as MRI, can confirm accurate electrode placement and exclude complications such as intracerebral haemorrhage, enema, or unintended injury, supporting that any observed eyelid drooping is unlikely to be related to the surgery itself and may instead be due to other causes, such as local anaesthetic effects from scalp block infiltration.
The absence of ophthalmoplegia or pupillary abnormalities supports a benign, anaesthesia-related mechanism rather than oculomotor nerve (cranial nerve III) involvement. In this case, the timing of symptom onset, approximately 30 minutes after the scalp block, and complete spontaneous resolution within 12 hours are consistent with transient local anaesthetic spread along soft tissue planes. Such a mechanism can temporarily affect periocular motor fibers or sympathetic pathways without causing structural nerve injury, highlighting the importance of recognizing reversible, block-related complications in the postoperative period[9].
The primary limitation of this case report is the absence of objective perioperative measurements such as real-time imaging of local anaesthetic spread, quantitative eyelid function assessment, or electrophysiologic monitoring. This limits deeper mechanistic insight into this phenomenon. Future studies comparing different local anaesthetic volumes, concentrations, and injection approaches may further guide best practices to minimize such events in neurosurgical anaesthesia.
In conclusion, careful attention to anatomical landmarks, precise needle placement, and appropriate dosing of local anaesthetic are essential to minimize the risk of inadvertent iatrogenic ptosis. Early recognition of this rare, self-limiting complication is equally important, as it allows clinicians to distinguish it from more serious neurologic events and prevents unnecessary diagnostic or therapeutic interventions in the neurosurgical setting.
| 1. | Kremer NI, van Laar T, Lange SF, Statius Muller S, la Bastide-van Gemert S, Oterdoom DM, Drost G, van Dijk JMC. STN-DBS electrode placement accuracy and motor improvement in Parkinson's disease: systematic review and individual patient meta-analysis. J Neurol Neurosurg Psychiatry. 2023;94:236-244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 2. | Guilfoyle MR, Helmy A, Duane D, Hutchinson PJA. Regional scalp block for postcraniotomy analgesia: a systematic review and meta-analysis. Anesth Analg. 2013;116:1093-1102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 119] [Article Influence: 9.2] [Reference Citation Analysis (1)] |
| 3. | Geze S, Yilmaz AA, Tuzuner F. The effect of scalp block and local infiltration on the haemodynamic and stress response to skull-pin placement for craniotomy. Eur J Anaesthesiol. 2009;26:298-303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 63] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 4. | Papangelou A, Radzik BR, Smith T, Gottschalk A. A review of scalp blockade for cranial surgery. J Clin Anesth. 2013;25:150-159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 30] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 5. | Hassan MH, Hassan WMNW, Kandasamy R, Chong SE. Unilateral Complete Ptosis after Scalp Block for Awake Craniotomy: A Rare Complication. J Neuroanaesth Crit Care. 2018;05:111-113. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 6. | Tremolada C, Candiani P, Signorini M, Vigano M, Donati L. The surgical anatomy of the subcutaneous fascial system of the scalp. Ann Plast Surg. 1994;32:8-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 28] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 7. | Singh MK, Haldar R, Kannaujia AK, Das KK. Iatrogenic Lagophthalmos or Lid Lag. An Uncommon Occurrence Following Scalp Block. Neurol India. 2021;69:526-527. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 8. | Nteli Chatzioglou G, Gayretli Ö, Özbilen KT, Önal V, Coşkun O, Kale A. Anatomical investigation of levator palpebrae superioris muscle and the distribution pattern of oculomotor nerve. Surg Radiol Anat. 2025;47:201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 9. | Situ S, Gupta P, Thirunavukkarasu M, Chaudhary G. Unilateral complete ptosis after scalp block: A rare complication of common procedure. Indian J Anaesth. 2020;64:1077-1078. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 0.7] [Reference Citation Analysis (1)] |