Published online Apr 16, 2026. doi: 10.12998/wjcc.v14.i11.119413
Revised: February 28, 2026
Accepted: March 11, 2026
Published online: April 16, 2026
Processing time: 72 Days and 16 Hours
Diabetic retinopathy (DR) remains a major cause of preventable vision loss world
Core Tip: Gene therapy for diabetic retinopathy is developing as a method to diminish the want for regular intravitreal injections by offering prolonged intraocular modulation of disease pathways. Extensive experience with age-related macular degeneration provides compelling clinical evidence that sustained suppression of pathological vascular signaling is feasible, while preliminary studies on diabetic retinopathy are evaluating the potential for similar durability within the inflammatory diabetic retinal milieu. Significant obstacles to translation encompass delivery- and dose-associated intraocular inflammation, ambiguity concerning long-term efficacy and the practicality of repeated dosage, as well as the necessity for stringent patient selection and monitoring.
- Citation: Capobianco M, Nicolosi SG, Cappellani F, Khouyyi M, D’Esposito F, Gagliano C, Zeppieri M. Gene therapy for diabetic retinopathy. World J Clin Cases 2026; 14(11): 119413
- URL: https://www.wjgnet.com/2307-8960/full/v14/i11/119413.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i11.119413
Diabetic retinopathy (DR) is a common complication of diabetes mellitus and remains a leading cause of vision loss and blindness worldwide. Traditionally regarded as a microvascular disease, DR also involves retinal neurodegeneration and is driven by complex, interrelated mechanisms triggered by hyperglycemia, including oxidative stress, advanced glycation end products, inflammatory mediators, and vascular endothelial growth factor (VEGF) signaling[1-3]. Clinically, advanced disease includes proliferative DR, characterized by abnormal growth of new retinal blood vessels, and diabetic macular edema (DME), in which exudation and edema affect the central retina; DME can occur in eyes with nonproliferative or proliferative disease[2]. The likelihood of DR is strongly linked to longer diabetes duration, poor glycemic control, and hypertension, making optimization of blood glucose and blood pressure central to prevention and slowing progression[1-3].
Although classic syntheses report that approximately one-third of people with diabetes have DR, a recent systematic review and meta-analysis of 59 population-based studies estimated a global DR prevalence of 22.27% (95% confidence interval: 19.73%-25.03%) among individuals with diabetes and projected that the number of adults with DR will increase from 103.12 million in 2020 to 160.50 million by 2045, based on International Diabetes Federation population projections[2,3]. These trends underscore the ongoing public health burden of DR despite advances in awareness, screening (including retinal photography), and established therapies such as laser photocoagulation and intravitreal anti-VEGF treatment for DME associated with vision loss. However, anti-VEGF therapy primarily addresses later stages of disease, requires repeated intravitreal injections, and does not provide optimal benefit for all patients, reinforcing the need for additional therapeutic approaches - particularly those aimed at preventing progression from earlier stages and addressing patients for whom anti-VEGF is ineffective or inappropriate[1-3].
DR should not be viewed solely as a microvasculopathy. In diabetes, disruption of the retinal neurovascular unit (NVU) - the tightly coupled system of neurons, glia (Müller cells and astrocytes), immune cells (microglia/macrophages), and vascular cells (endothelial cells and pericytes) that maintains inner blood-retinal barrier (BRB) integrity and neurovascular coupling - emerges as an early pathogenic event[4]. Hyperglycemia-related pathways implicated in this early dysfunction include the accumulation of advanced glycation and lipoxidation end-products with downstream glial activation and cytokine production, alongside a pro-inflammatory milieu and oxidative stress-linked injury that can contribute to barrier impairment and neuronal damage[4].
Consistent with this framework, longitudinal human optical coherence tomography (OCT) data in individuals with diabetes and no or minimal clinically detectable DR demonstrate progressive thinning of inner retinal layers (nerve fiber layer and ganglion cell/inner plexiform complex) over time that is independent of glycated hemoglobin A1c and of DR presence or progression. Complementary evidence from human donor eyes showed significant nerve fiber layer thinning without a detectable reduction in retinal capillary density, and two diabetic mouse models exhibited early inner retinal thinning and later ganglion cell loss before clear microvascular loss markers (e.g., pericyte dropout/acellular capillaries) were observed[5]. Together, these findings support the concept that retinal diabetic neuropathy (RDN) can precede the established clinical and morphometric microvascular changes of DR and is not simply a late, ischemia-driven consequence of vascular damage[5]. Accordingly, contemporary perspectives increasingly frame DR as a tissue-specific neurovascular complication in which glial activation, neuronal dysfunction/apoptosis, and microvascular abnormalities are interdependent, and in which retinal neurodegeneration represents a clinically meaningful endpoint and potential therapeutic target - particularly when current treatments predominantly address later, vision-threatening stages[4,5].
Intravitreal anti-VEGF therapy is widely used as first-line therapy for DME, particularly when foveal involvement is accompanied by vision loss. In randomized clinical trials, anti-VEGF agents (ranibizumab, bevacizumab, and aflibercept), used alone or with laser, achieved larger visual acuity improvements than laser alone under intensive follow-up schedules that typically included frequent (often monthly) monitoring and near-monthly injections during the first year[6-9]. In routine care, however, treatment intensity is consistently lower: In the Intelligent Research in Sight Registry, most treatment-naïve patients with a diagnosis code for DME received no therapy within 28 days of diagnosis, and those treated promptly received a median of four injections over one year; similarly, electronic medical record (EMR)-based cohorts show that most eyes receive three or fewer injections in the first 12 months and only a small fraction approximate monthly dosing. Large real-world datasets likewise report that many eyes receive six or fewer injections at one year, with relatively few receiving 10-13 injections[6-9]. Correspondingly, mean 1-year visual gains in clinical practice are modest (roughly +4 letters to +6 letters) and meaningfully inferior to landmark trial outcomes; across real-world analyses, greater injection intensity is generally associated with better visual improvement, underscoring how reduced monitoring, missed visits, and lower dosing frequency can erode the benefits observed in trials[6-9].
DR reflects a highly complex retinal disorder, with diabetes affecting many retinal cell types and pathology extending beyond visible vascular lesions; microvascular dysfunction can progress to nonperfusion/hypoxia and is closely linked to BRB breakdown and macular edema. Despite the introduction of intravitreal VEGF inhibitors and steroids for DME, therapeutic options remain limited, and a substantial proportion of patients do not respond to current treatments (reported up to approximately 50% in the context of DME)[7,8]. In real-world cohorts specifically enriched for early “refractory” DME after a standard three-injection anti-VEGF loading phase, continuing anti-VEGF therapy was associated with minimal average functional or anatomic change at 12 months, whereas an early switch to a dexa
In this context, intraocular gene transfer has become a credible translational strategy. In inherited retinal disease, subretinal delivery of an adeno-associated virus (AAV) vector carrying RPE65 cDNA in patients with Leber congenital amaurosis type 2 (LCA2) showed an acceptable local and systemic adverse-event profile, along with modest improve
In treatment-experienced neovascular AMD, early clinical studies indicate that a single ocular gene-therapy administration can support sustained intraocular production of an anti-VEGF agent while maintaining functional and anatomic outcomes and markedly reducing the need for supplemental injections[14,15]. In the 2-year phase 1 safety and efficacy of ixoberogene soroparvovec in neovascular age-related macular degeneration in the United States (OPTIC) study, a single in-office intravitreal injection of ixoberogene soroparvovec (ixo-vec) - designed to enable long-term expression of aflibercept - was generally well tolerated, with no systemic treatment-related adverse events reported; ocular inflammation (e.g., anterior chamber/vitreous cells) was dose-dependent and responsive to topical corticosteroids[17]. Across dose cohorts, visual acuity and central subfield thickness remained stable over 2 years, while the annualized need for anti-VEGF rescue injections was substantially reduced, and measured aqueous aflibercept levels were stable through study completion in sampled participants[17].
Consistent proof-of-concept findings have also been reported with subretinal RGX-314 (AAV8), which encodes an anti-VEGF-A antigen-binding fragment intended for continuous VEGF-A suppression after a single administration[16,17]. In a phase 1/2a dose-escalation study, subretinal RGX-314 was generally well tolerated with no clinically recognized immune responses, and doses ≥ 6 × 1010 genome copies/eye were associated with sustained aqueous RGX-314 protein concentrations, alongside stable or improved best corrected visual acuity (BCVA) and central retinal thickness, and few or no supplemental anti-VEGF injections in many participants[18]. The study also documented dose-associated retinal pigmentary changes (more common at higher doses) and reported one serious event possibly related to RGX-314 with severe vision reduction at the highest dose level[18].
While most DR/DME gene-therapy programs currently in clinical development are designed to provide durable intraocular anti-VEGF activity, other investigational constructs are also being explored to address additional disease drivers - particularly retinal inflammation and vascular destabilization/BRB breakdown - with reported benefits on neurovascular integrity and retinal function in diabetic models[19-22]. Parallel advances in genome editing - most notably Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR)-Cas systems - are extending retinal gene therapy beyond gene supplementation toward direct, potentially permanent correction of pathogenic DNA, as exemplified by in vivo subretinal CRISPR-Cas9 editing for CEP290-associated retinal degeneration (EDIT-101)[23-25]. In the BRILLIANCE phase 1/2 study, EDIT-101 was not associated with treatment- or procedure-related serious adverse events or dose-limiting toxic effects, and a subset of participants showed meaningful improvements in cone-mediated function and/or other key visual outcomes, supporting further investigation of in vivo retinal gene editing[23]. Nevertheless, these approaches remain early-stage and require stringent safety evaluation - particularly for long-term risks such as off-target effects and immune responses to Cas effectors, guide RNAs, and delivery vectors - which can influence both efficacy and tolerability and are central considerations for clinical translation[24,25].
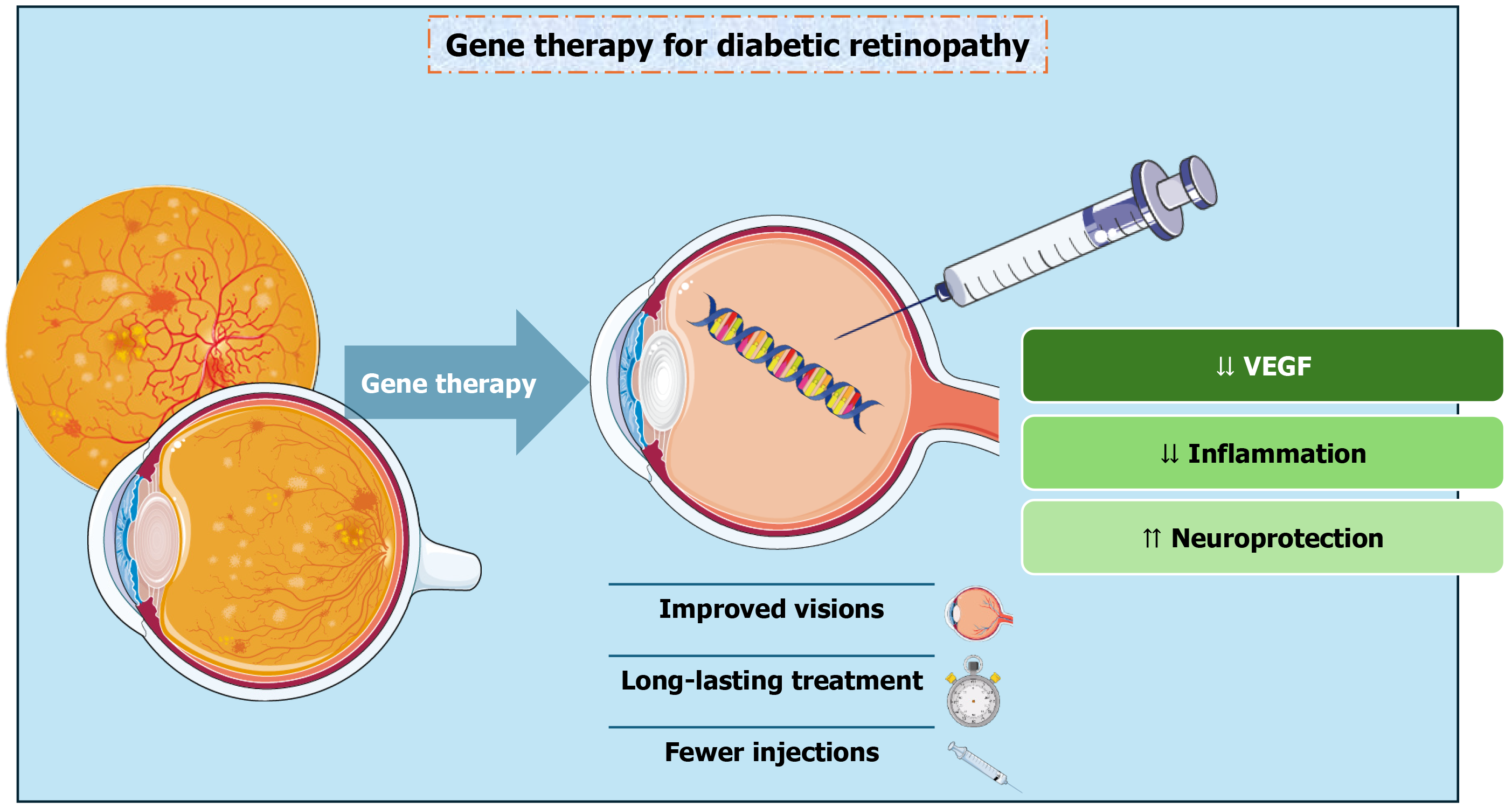
Figure 1 provides an at-a-glance overview of the rationale and translational framework for gene therapy in DR/DME, highlighting the ‘retinal biofactory’ concept, delivery routes, and key expected benefits and constraints (Figure 1). This narrative review aims to objectively assess the current status of gene therapy for DR and DME, incorporating molecular rationale, translational evidence, and new clinical data. This review synthesizes findings from preclinical studies, ongoing and completed clinical trials, and insights from related retinal conditions to position gene therapy not as a substitute for current treatments, but as an evolutionary advancement that could redefine long-term therapeutic objectives in diabetic retinal disease.
This manuscript is a narrative review that provides a comprehensive and critical evaluation of gene therapy and genome-editing strategies for DR and DME, with an emphasis on the mechanistic rationale, translational development, and emerging clinical evidence. A narrative approach was selected to enable contextual interpretation across heterogeneous sources - ranging from mechanistic and preclinical studies to early- and mid-phase clinical programs and real-world outcome analyses - whose variability in design, endpoints, and populations limits suitability for formal meta-analytic synthesis.
This study was conducted as a structured narrative review rather than a systematic review or meta-analysis; thus, a Preferred Reporting Items for Systematic Reviews and Meta-Analyses-style flow reporting has not been utilized. There was significant variability in the existing evidence, encompassing early-phase clinical trials, translational research, and platform-specific safety reports that cannot be effectively aggregated. The objective of the review has been to integrate mechanistic understanding with new clinical data to facilitate translational interpretation rather than statistical aggregation. Publications have been sourced from prominent biological databases and subsequently evaluated for their relevance to ocular gene transfer in diabetic retinal disease or to delivery systems with direct translational significance. Sources have been ranked based on their provision of mechanistic insights, safety data, or clinically interpretable results. This qualitative technique has been intentionally selected to prioritize interpretive significance over numerical comprehensiveness.
A literature search was conducted using PubMed/MEDLINE as the primary database, supplemented by targeted searches in Scopus and Web of Science. Search terms combined Medical Subject Headings and free-text keywords related to diabetic retinal disease and gene-based interventions, including “diabetic retinopathy”, “diabetic macular edema”, “ocular gene therapy”, “adeno-associated virus”, “AAV vectors”, “anti-VEGF gene delivery”, “retinal biofactory”, and “genome editing/CRISPR”. Searches were restricted to English-language publications available through December 2025. Reference lists of key reviews and pivotal primary studies were manually screened to identify additional relevant publications.
Eligible sources included peer-reviewed original research, clinical trial reports, translational platform studies, and high-quality narrative or systematic reviews addressing gene-based approaches in DR/DME or closely related retinal vascular diseases with direct translational relevance. Evidence from neovascular AMD was included when it informed shared vector platforms, delivery routes (intravitreal, subretinal, or suprachoroidal), durability of intraocular expression, or clinically observed safety liabilities pertinent to diabetic retinal disease. Conference abstracts without full peer-reviewed publication, non-indexed sources, and studies lacking a gene therapy or genome-editing component were excluded.
Included evidence was synthesized qualitatively, focusing on therapeutic targets, vector and delivery design, durability considerations, safety signals (particularly intraocular inflammation), and clinically meaningful outcomes such as best-corrected visual acuity, OCT-based anatomic measures (e.g., central retinal thickness), DR severity metrics [including DR severity score (DRSS) when applicable], and treatment-burden indicators (e.g., need for supplemental injections). Rather than pooling outcomes statistically, studies were evaluated thematically to identify convergent mechanisms, translational consistencies, and key uncertainties, with particular attention to how the inflammatory and metabolic milieu of the diabetic retina may shape efficacy, tolerability, and durability.
DR is increasingly understood as a complex, multifactorial disorder in which diabetes disrupts not only the retinal microvasculature but the tightly integrated retinal NVU. Chronic hyperglycemia activates interrelated biochemical and cellular pathways - including increased flux through the polyol pathway, mitochondria-driven oxidative stress with reactive oxygen species generation, advanced glycation end-product formation and advanced glycation end-linked signaling, and protein kinase C activation - alongside sustained inflammatory signaling, rather than operating through a single isolated mechanism. Within this milieu, early pericyte loss and endothelial dysfunction/vasoregression, impaired vascular autoregulation, and functional BRB impairment can emerge before the appearance of clinically obvious funduscopic lesions. In parallel, retinal glial and neuronal abnormalities - including reactive gliosis, microglial activation, and neurodegenerative changes - may be detectable when ophthalmoscopy still shows minimal or no vascular pathology. Taken together, these observations support the view that lesion-based clinical grading captures downstream manifestations of a broader, progressive neurovascular disease process, motivating therapeutic strategies aimed at durable modulation of upstream retinal signaling, inflammation, and cell-survival pathways rather than targeting only late-stage angiogenic endpoints[12,26].
Consistent with this framework, next-generation gene therapy approaches increasingly target NVU-relevant mechanisms beyond VEGF inhibition, recognizing that neuronal, glial, immune, and microvascular dysfunction are interdependent. Retinal neurodegeneration - encompassing reactive gliosis, early neuronal dysfunction, and neuroapoptosis - has been described as an early feature in diabetes and may precede, or evolve partly independently from, clinically visible microangiopathy; structural correlates (inner retinal/nerve fiber layer thinning on OCT) and functional correlates [e.g., multifocal electroretinogram (ERG) abnormalities] have been reported in patients with little or no fundus-detectable disease and may anticipate subsequent vascular change in a subset of individuals. Accordingly, neuroprotection is increasingly considered a clinically meaningful endpoint rather than merely a downstream consequence of microvascular rescue[3,4].
From a translational standpoint, this broader NVU perspective aligns with the growing emphasis on quantitative biomarkers for early phenotyping and monitoring. OCT angiography enables depth-resolved assessment of capillary plexus integrity and can reveal microvascular alterations even when fundus photography appears normal, while OCT- and function-based measures can capture neuroretinal injury not incorporated into traditional, lesion-driven DR grading scales[3,4]. Together, these observations support the rationale for gene-based strategies that aim to modulate inflammation, barrier stability, neuroglial signaling, and neurovascular coupling, positioning gene therapy not as a replacement for established anti-VEGF regimens, but as a potential platform to pursue durable, multi-pathway disease modification in diabetic retinal disease[3,4].
VEGF is a key effector within ischemic retinal disease, linking hypoxia-driven signaling to both vascular leakage and pathologic neovascularization. In ischemic retinopathies, stabilization of hypoxia-inducible factor 1 upregulates VEGF together with other hypoxia-regulated mediators, and VEGF is sufficient to induce marked vascular permeability and contributes critically to neovascular responses; accordingly, VEGF neutralization has become first-line therapy for macular edema and ocular neovascular complications in these settings. However, these conditions are inherently chronic, and clinical experience indicates that sustained control often requires repeated intravitreal dosing, leaving a persistent unmet need that has driven interest in both additional pathway targets and sustained-delivery approaches for VEGF antagonists[27].
Real-world outcomes further illustrate the practical consequences of this chronicity in DME. In Observation and Treatment in Adiabetic Macular Edema with Improved Nisual acuity (OBTAIN), most eyes with center-involving DME and very good baseline visual acuity maintained vision over 12 months, yet when intravitreal anti-VEGF was used, higher injection numbers (8-12) were associated with better visual outcomes than fewer injections, underscoring that treatment benefit is closely tied to treatment intensity and ongoing exposure rather than a one-time, durable effect[28]. Together, these observations support the rationale for strategies designed to provide longer-lasting intraocular pathway control compared with intermittent suppression alone[28].
From a molecular standpoint, ocular gene therapy is designed to achieve durable, intraocular expression of therapeutic proteins by transducing retinal cells with recombinant adeno-associated virus (AAV) vectors, thereby reducing the need for frequent re-treatment. AAV is widely used for ocular delivery because it can support long-term transgene expression while generally eliciting relatively mild immune responses. In the retina, vector tropism depends on serotype and delivery route; for example, AAV2 commonly transduces retinal pigment epithelium (RPE) and can also transduce Müller cells, ganglion cells, and photoreceptors. This sustained-expression strategy is particularly relevant for chronic, highly prevalent retinal vascular diseases such as DR and DME, where standard care can be effective but remains burdensome over the long term[25,27]. Importantly, AAV platforms under investigation in DR/DME are not limited to VEGF-pathway interference; preclinical and emerging clinical efforts also explore targets and constructs aimed at vascular hyperpermeability/BRB dysfunction, inflammation and oxidative stress pathways, and neurodegeneration-related mechanisms, often incorporating engineered capsids, tissue-specific promoters, and regulated expression systems to improve targeting and safety[29].
Beyond angiogenesis, diabetic retinal disease is increasingly understood as an early disruption of the retinal NVU, in which neurons, glia, immune cells, and microvasculature are functionally interdependent. Experimental studies, human donor data, and longitudinal imaging show that reactive gliosis and inner retinal neurodegeneration - including early vulnerability of retinal ganglion cells and other inner retinal neurons - can be detected before, or in parallel with, clinically evident microvascular retinopathy, with measurable structural thinning on OCT and functional abnormalities on electrophysiology and psychophysical testing. In people with diabetes and no or minimal retinopathy, progressive loss of inner retinal layers over time has been reported as largely independent of glycated hemoglobin A1c and retinopathy progression, and complementary donor-eye and animal-model observations support neuroretinal degeneration occurring without concomitant capillary loss or other early microvascular hallmarks. Mechanistically, glial activation may contribute to subclinical inflammation and barrier dysfunction, while an imbalance between neuroprotective factors and proinflammatory/innate immune mediators can promote neural injury. This framework broadens therapeutic thinking beyond late-stage anti-angiogenic interventions toward strategies aimed at preserving NVU integrity, combining risk-factor control with approaches that target inflammation and neuroprotection, while underscoring the need for improved early phenotyping and adequately powered long-term interventional studies[4,5,30].
Experience with other retinal disorders has yielded significant insights on vector performance and delivery practicality; however, diabetic retinal disease exhibits distinct biological differences in numerous crucial aspects. Chronic metabolic stress, persistent inflammatory activation, and pre-existing vascular instability characterize the diabetic retina, potentially affecting immune responses, tolerability, and the durability of gene-based therapies. Consequently, insights from other retinal conditions have mostly been used to assess feasibility rather than to predict therapy outcomes in diabetes cohorts.
Intravitreal anti-VEGF therapy has become the preferred first-line treatment for vision-impairing, center-involved DME, supported by randomized trials showing superior visual outcomes vs laser or sham/Laser-rescue strategies when delivered with frequent monitoring and repeated dosing (e.g., RISE/RIDE; VISTA/VIVID; DRCR.net Protocol T)[7,9]. In routine practice, however, sustaining the intensity used in trials is challenging. Large EMR-based real-world cohorts from the United States retina care consistently report fewer injections and less intensive follow-up than in landmark trials - commonly a median of around six injections in year 1 (with many eyes receiving ≤ 6, and only a minority approaching monthly dosing), and in some clinical-practice series, averages near three injections in the first year[7,8]. Correspondingly, mean 12-month visual gains in these real-world datasets are modest [approximately +4 Early Treatment Diabetic Retinopathy Study (ETDRS)-equivalent letters to +5 ETDRS-equivalent letters] and are explicitly inferior to outcomes reported in controlled trials. Given the short intraocular durability of current agents (≤ 9-day half-life), lower treatment intensity likely results in intermittent VEGF suppression and contributes to the persistent gap between real-world and trial outcomes, with additional complexity from incomplete longitudinal follow-up and ceiling effects (and potential vulnerability to vision loss) in eyes presenting with relatively good baseline acuity[7-9].
Requiring repeated intravitreal administration of VEGF-neutralizing agents creates important limitations that go beyond clinic scheduling. Although these drugs can deliver strong visual and anatomic benefits, maintaining those gains typically depends on continued, frequent dosing; when injection schedules become less intensive, outcomes can deteriorate. From a pharmacokinetic standpoint, intravitreal anti-VEGF activity is time dependent: Maximal binding activity occurs soon after injection and then progressively declines as drug levels fall, resulting in lower “trough” activity before the next dose. Modeling suggests that shortening the dosing interval primarily improves trough activity rather than peak activity, supporting the concept that more sustained VEGF suppression may better stabilize disease control than episodic exposure[28,30]. Together, the real-world burden of ongoing injections and the biological logic of avoiding low-activity intervals have driven interest in longer-acting strategies that can maintain VEGF inhibition with fewer retreatments. Gene therapy introduces a different therapeutic paradigm by enabling ocular tissues to produce a therapeutic protein over extended periods, effectively converting the eye into a local “biofactory”. Using AAV vectors, genes encoding VEGF-neutralizing proteins (or other biologically active factors) can be delivered so that transduced cells become a continuing source of the therapeutic agent. In early human experience with intravitreal AAV2-sFLT01, the VEGF-neutralizing transgene product was measurable in a subset of patients over long follow-up, illustrating the feasibility - and variability - of durable intraocular production after a single administration. For chronic retinal vascular diseases in which hypoxia-driven VEGF expression tends to persist and fuel permeability and neovascularization, approaches that aim for sustained VEGF suppression are conceptually aligned with the underlying biology and are being developed to reduce treatment burden while preserving efficacy[29,31-34].
Durable intraocular anti-VEGF expression has been demonstrated most clearly in gene-therapy programs developed for neovascular AMD. In the phase 1/2a, dose-escalation study of subretinal RGX-314, higher doses (≥ 6 × 1010 genome copies/eye) produced sustained intraocular anti-VEGF Fab expression measurable in aqueous humor, with generally stable or improved best-corrected visual acuity and central retinal thickness and, in many participants, few or no supplemental anti-VEGF injections over two years, without clinically evident immune responses beyond what would be expected after vitrectomy[18]. Similarly, in the two-year OPTIC phase 1 trial, a single in-office intravitreal administration of ixoberogene soroparvovec achieved durable intraocular aflibercept exposure, maintained visual and OCT stability, and reduced the annualized need for rescue anti-VEGF injections, with a substantial proportion of patients remaining injection-free at study completion[17]. Together, these results provide a practical proof-of-concept that one-time ocular gene transfer can deliver sustained, clinically meaningful VEGF suppression with a reduced treatment burden. On this basis, next-generation intravitreal platforms such as 4D-150 - engineered to express aflibercept while also inhibiting VEGF-C - have shown robust retinal transgene expression and anti-angiogenic activity in primate models and are being advanced into clinical programs that include DME, reflecting the shared VEGF-driven biology across neovascular retinopathies[35,36].
This translational pivot is now being tested directly in diabetic retinal disease, including the phase 2 Adjuvant Long-term Therapy for Improving Treatment Understanding in Diabetic Eye Disease (ALTITUDE) study evaluating a single suprachoroidal administration of ABBV-RGX-314 in DR with and without center-involved DME, the phase 2 INFINITY trial investigating intravitreal ixoberogene soroparvovec (ADVM-022) for DME, and the phase 2 SPECTRA program assessing intravitreal (4D-150) vs aflibercept in adults with DME[37-39].
Gene-transfer “biofactory” strategies are being developed primarily to achieve sustained intraocular VEGF suppression, an approach supported by the chronic, hypoxia-driven upregulation of VEGF in retinal/choroidal vascular diseases and by the gap between excellent outcomes in clinical trials with frequent dosing and the poorer results typically seen when injection frequency decreases in real-world practice. New sustained-delivery approaches - including long-acting devices, injectable depots, and viral-vector–mediated expression of VEGF-neutralizing proteins - aim to maintain more constant therapeutic exposure over time and thereby reduce the need for repeated retreatment. At the same time, important uncertainties remain, including the long-term consequences of prolonged VEGF suppression (e.g., whether it may influence the incidence of subretinal fibrosis or macular atrophy) and whether optimal outcomes in some patients - particularly in a subset with DME - may require additional pathway targeting beyond VEGF. For viral-vector biofactories specifically, immune responses represent a key barrier to durable efficacy and safety: AAV vectors can activate innate sensing pathways (including toll-like receptor 2 and toll-like receptor 9) with downstream cytokine release, and they can elicit capsid- and transgene-directed adaptive responses (T cells and neutralizing antibodies) that have been associated with intraocular inflammation and/or loss of effect in retinal gene-therapy studies, despite the eye’s immune-privileged environment[33,40].
By comparison, infectious endophthalmitis after conventional intravitreal anti-VEGF injections is uncommon (approximately 0.056% across > 350000 injections), with coagulase-negative Staphylococcus and Streptococcus species most frequently isolated; Streptococcus cases tend to have poorer visual outcomes[41].
Clinical experience in in vivo retinal gene therapy - including AAV-mediated gene augmentation trials for RPE65-associated Leber congenital amaurosis showing meaningful, durable visual gains with acceptable safety - provides an important translational precedent for adapting similar approaches to acquired retinal diseases such as DR and DME[42,43]. Recombinant AAV vectors are well-suited to ocular applications because they can be produced without viral genes and deliver an inverted terminal repeat-flanked expression cassette that predominantly persists as episomal DNA in the nucleus, supporting sustained transgene expression in low-turnover/post-mitotic retinal cells. Vector performance can be optimized through expression-cassette design within AAV packaging constraints (e.g., promoter selection) and through capsid choice or engineering (including discovery/engineering strategies that improve retinal tropism and approaches aimed at reducing susceptibility to neutralizing antibodies). At the same time, key challenges remain: Capsid- and transgene-directed immune responses (often influenced by pre-existing anti-AAV immunity) can reduce efficacy and complicate re-administration, and ocular delivery still requires continued refinement to achieve efficient, broader distribution with minimal procedure-related burden[42,43].
Early-phase clinical evidence in neovascular AMD suggests that AAV-based ocular gene transfer can enable sustained intraocular production of anti-VEGF biologics and substantially reduce the need for supplemental injections[41-43]. In a multicenter phase 1/2a dose-escalation study, subretinal AAV8-RGX-314 (ABBV-RGX-314) - encoding an anti-VEGF-A Fab similar to ranibizumab - produced measurable aqueous levels of the transgene product; at doses ≥ 6 × 1010 genome copies/eye, many participants showed control of exudation with stable (or improved) BCVA and OCT thickness metrics while requiring few or no rescue injections over 2-year follow-up, with safety findings that included dose-associated retinal pigmentary changes and a single serious event deemed possibly related at the highest dose[18]. Ixoberogene soroparvovec (ixo-vec; ADVM-022), delivered as a single intravitreal in-office administration to drive aflibercept expression, maintained vision and central subfield thickness over two years in OPTIC while markedly lowering annualized rescue injection frequency; intraocular inflammation was dose-dependent but generally responsive to topical corticosteroids[17]. In parallel, 4D-150 exemplifies a shift toward multi-target designs: A retinotropic intravitreal vector (R100) carrying an aflibercept transgene plus a VEGF-C - inhibitory microRNA produced robust retinal expression in nonhuman primates, reduced VEGF-A/VEGF-C signaling in human retinal cells, and prevented angiogenic lesions in a primate laser-choroidal neovascularization (CNV) model, supporting ongoing clinical development[35]. Contemporary syntheses of the field frame these programs (RGX-314, ixo-vec, and 4D-150) as leading examples of the “biofactory” approach aimed at lowering treatment burden while maintaining functional and anatomic stability[36].
The relevance of recent advances in retinal gene therapy to diabetic retinal disease rests on both shared and disease-specific biology. In DR and DME, VEGF-A signaling is a key driver of microvascular leakage and, in proliferative disease, pathological neovascularization; therefore, a sustained intraocular strategy that counteracts VEGF-mediated permeability and angiogenesis is mechanistically well aligned with established therapeutic targets in diabetic retinal disease[3]. At the same time, diabetes perturbs the retinal NVU and blood-retinal barrier through pericyte-endothelial dysregulation, endothelial dysfunction, and a cytokine-rich inflammatory milieu, with hyperglycemia-related oxidative stress further destabilizing vascular homeostasis[3]. This baseline environment could, in turn, influence vector performance and durability by increasing innate immune sensing and inflammatory mediator release, altering barrier permeability, and facilitating capsid- or transgene-directed adaptive responses - processes that have been implicated in ocular inflammation and loss of efficacy across AAV retinal gene-therapy studies[40]. Consequently, while AAV-based protein-expression approaches have been extensively developed in other retinal indications, translation to DR/DME should rely on indication-specific preclinical and clinical evidence rather than assuming comparable efficacy and immune tolerance across distinct retinal disease microenvironments[3,40].
Recent DR/DME-focused clinical programs underscore why indication-specific data matter. DR spans a wide clinical spectrum from mild nonproliferative diabetic retinopathy (NPDR) to advanced PDR with sight-threatening sequelae (e.g., neovascular glaucoma and tractional retinal detachment), and many patients remain asymptomatic until complications such as DME and/or PDR develop. Standard-of-care anti-VEGF therapy can be highly effective but typically demands frequent monitoring and repeated injections, and this cumulative burden is a practical driver of reduced adherence and worse real-world outcomes. In this context, current DR/DME gene-therapy strategies largely adopt a “retinal biofactory” paradigm - delivering transgenes that enable sustained intraocular expression of anti-angiogenic proteins that inhibit VEGF. Early readouts from trials such as SPECTRA (4D-150) and ALTITUDE (ABBV-RGX-314) suggest potential for DRSS improvement and meaningful reduction in treatment burden, whereas the INFINITY (ADVM-022/ixoberogene soroparvovec) experience highlights the counterbalancing safety risk, with severe inflammation and hypotony prompting discontinuation of development for DME. Together, these findings reinforce that DR/DME translation must balance durability goals against the liabilities of inflammation within the diabetic retinal milieu[19].
The route of administration is a key determinant of performance and tolerability in ocular gene delivery, because each approach changes (1) Where the vector distributes; (2) Which barriers it must cross; and (3) The immune milieu it en
Early clinical experience in DR/DME gene therapy aligns with that rationale. DR is a major cause of preventable vision loss and is often asymptomatic until vision-threatening complications (notably DME and/or PDR) arise; current standards (laser and intravitreal anti-VEGF) typically require ongoing monitoring and multiple injections, and treatment burden contributes to imperfect adherence and worse outcomes. Gene-therapy trials in DR/DME have therefore focused on retinal delivery of transgenes encoding anti-angiogenic proteins that inhibit VEGF, and preliminary updates report improvements in the DRSS and reductions in treatment burden in programs such as SPECTRA (4D-150) and ALTITUDE (ABBV-RGX-314). In DME, the phase 2 INFINITY study evaluates intravitreal ADVM-022 (AAV.7m8-aflibercept) as a single-administration strategy intended to support sustained intraocular aflibercept expression; in the posted record, the study enrolled 36 participants and, after unmasking for enhanced safety monitoring, reports (with that confounding caveat) improved efficacy across endpoints including reduced need for supplemental aflibercept and a clinically meaningful delay in worsening, while noting dose-limiting toxicities in some high-dose participants and mild-to-moderate intraocular inflammation as the most common related adverse event, generally responsive to corticosteroid drops and without reported fellow-eye or systemic events. Overall, these signals are promising but leave key questions open regarding patient selection, dosing, and durability/safety over long follow-up in chronic metabolic disease[19,38]. Key ongoing gene therapy programs targeting angiogenesis and related pathways in diabetic retinal disease are summarized in Table 1.
| Ref. | Program (vector) | Target indication/population | Delivery route | Vector/construct | Intended mechanism (as stated) | Trial phase/record in manuscript | Key points reported in the manuscript |
| Odio-Herrera et al[19], 2025 | ABBV-RGX-314 (RGX-314) | Diabetic retinopathy with and without center-involved DME; studies described as enrolling moderately severe or severe NPDR (and select mild PDR) with good baseline vision and no recent anti-VEGF exposure | Suprachoroidal (single administration) | Described as AAV8-RGX-314 (ABBV-RGX-314); encodes an anti-VEGF-A Fab (ranibizumab-like) in the broader discussion | Durable intraocular anti-VEGF activity via a “retinal biofactory” strategy | Phase 2 ALTITUDE; ClinicalTrials.gov NCT04567550[37] | Development framed as testing whether durable anti-VEGF expression can be achieved in the diabetic retinal milieu; preliminary updates described as suggesting DRSS improvement and treatment-burden reduction in programs including ALTITUDE |
| Odio-Herrera et al[19], 2025 | Ixoberogene soroparvovec (ADVM-022; ixo-vec) | DME | Intravitreal (single administration) | Described as AAV.7m8-aflibercept (AAV2.7m8 capsid driving aflibercept expression) | Sustained intraocular aflibercept exposure (“biofactory”) to reduce the need for repeated injections | Phase 2 INFINITY; ClinicalTrials.gov NCT04418427[38] | Manuscript highlights the counterbalancing safety risk in DME: Severe intraocular inflammation and hypotony (dose-dependent), prompting discontinuation of development for DME; ClinicalTrials.gov record described as 36 participants, unmasked April 2021 for enhanced safety monitoring after SUSAR in the high-dose arm; inflammation often mild–moderate and responsive to steroid drops, but dose-limiting toxicities impacted benefit–risk |
| Odio-Herrera et al[19], 2025 | 4D-150 | Adults with DME | Intravitreal | Engineered to express aflibercept and also inhibit VEGF-C (via a VEGF-C - inhibitory microRNA); described as a multi-target intravitreal platform | Sustained anti-angiogenic activity with broader pathway coverage (VEGF-A + VEGF-C modulation) | Phase 2 SPECTRA; ClinicalTrials.gov NCT05930561[39] | Manuscript states the translational pivot is being tested directly in diabetic retinal disease via SPECTRA (4D-150 vs aflibercept); preliminary updates described as suggesting DRSS improvement and treatment-burden reduction in programs including SPECTRA |
Although therapeutic progress in diabetic retinal disease has largely been driven by VEGF-A inhibition, the underlying pathology is increasingly framed as a broader neurovascular disorder in which BRB failure, vascular degeneration, and neural/glial dysfunction are shaped by inflammatory signaling within the retinal NVU. In diabetes, hyperglycemia-related redox imbalance and endothelial dysfunction occur alongside increased production of inflammatory cytokines and chemokines including tumor necrosis factor-α and interleukin (IL)-1β, with elevated interleukins such as IL-6/IL-8 and chemokines such as C-C motif chemokine ligand 2 reported in ocular fluids, derived from retinal-resident cells and adherent inflammatory cells. These mediators promote leukocyte-endothelial interactions (for example, via adhesion molecules such as intercellular adhesion molecule 1), disrupt endothelial junctional integrity, and contribute to inner BRB breakdown with increased vascular permeability, edema, and - in settings of ischemia - pathological neovascularization. Glial and immune cells participate actively: Diabetes-related Müller glial alterations can amplify inflammatory signaling and activate microglia, and microglial activation has been linked to neuroinflammation-driven BRB disruption. Clinically, anti-VEGF therapy is mainly effective in later stages, requires frequent intravitreal retreatment, and a substantial proportion of patients show incomplete responses, consistent with the contribution of VEGF-independent inflammatory mechanisms. Collectively, these observations support therapeutic strategies that complement VEGF-A suppression by also modulating inflammatory and barrier-regulating pathways to pursue more comprehensive and durable disease control[3,45,46].
Microvascular degeneration and BRB dysfunction are core features of DR and shape disease progression well before the proliferative stage. Contemporary syntheses emphasize that DR severity grading is largely based on visible vascular lesions, but the underlying trajectory includes progressive endothelial cell and pericyte loss, subsequent capillary closure/nonperfusion, and capillary basement-membrane thickening; together, these changes undermine barrier integrity and promote chronic vasopermeability and retinal swelling[45,46]. Persistent BRB breakdown can culminate in DME, a major cause of vision loss in diabetes, and - despite the availability of intravitreal anti-VEGF agents and steroids for DME - therapeutic responses remain incomplete in a substantial fraction of patients; for proliferative DR, laser photocoagulation is still widely used even though it is intrinsically destructive. Within these established mechanisms, angiopoietin-2 upregulation is highlighted as a diabetes-responsive pathway that can precede and contribute to pericyte dropout, reinforcing the rationale for therapeutic strategies that directly address vascular cell survival, vasodegeneration, and barrier stability rather than focusing only on late-stage angiogenic sequelae.
Beyond the VEGF axis, the endothelial Tie receptors and their angiopoietin ligands are described as a second vascular tissue-specific receptor tyrosine kinase system that governs vessel maturation and adult vascular homeostasis. In this framework, constitutive angiopoietin-1 (ANG1)-driven activation of TEK receptor tyrosine kinase (TIE2) supports a quiescent, stabilized endothelium that is characteristically associated with peri-endothelial cell recruitment (pericytes and smooth-muscle cells), and TIE2 signaling is dominated by phosphatidylinositol 3-kinase-protein kinase B survival pathways[12,47]. Conversely, endothelial ANG2 - produced by endothelial cells and stored in specialized granules for rapid availability - antagonizes baseline ANG1-TIE2 signaling and, via an autocrine-loop concept, increases endothelial cell responsiveness to diverse external cues, including angiogenic signals (such as VEGF), permeability mediators (e.g., histamine and bradykinin), and inflammatory factors (e.g., tumor necrosis factor-α). Importantly for the retina, the review also notes experimental evidence that ANG2 can destabilize established retinal vasculature, including dose-dependent pericyte loss after direct intraocular ANG2 exposure, supporting a mechanistic link between ANG2 predominance, pericyte dropout, and barrier fragility[12,47].
In parallel with this vasculopathy-centered model, RDN is increasingly supported as an early, progressive component of diabetic retinal disease. In a prospective longitudinal cohort of people with diabetes and no or minimal clinically detectable DR, Sohn et al[5] demonstrated significant, progressive thinning of inner retinal layers on OCT over four years - specifically the nerve fiber layer and the ganglion cell/inner plexiform complex - independent of glycated hemoglobin, age, and sex; donor-eye analyses similarly showed marked nerve fiber layer thinning in diabetes without a corresponding reduction in retinal capillary density. In two experimental mouse models, inner retinal thinning and later ganglion-cell loss were observed in a temporal window in which classical microvascular markers (including pericyte density differences and acellular capillaries) were not yet detectable, supporting the interpretation that early RDN is not simply a downstream consequence of retinal microvascular loss and may not be ischemic in origin[5].
Functionally, RDN has been associated with deficits measurable by electrophysiology and psychophysics (e.g., ERG abnormalities, dark-adaptation and contrast-sensitivity deficits, and perimetric/microperimetric changes), and the authors explicitly note that there are currently no established management options to mitigate RDN; they also raise the point that neuroprotective strategies - already explored in other neurodegenerative eye diseases - may warrant evaluation in diabetes, particularly in the setting of anti-VEGF exposure where neurodegenerative effects have been a concern in some rodent studies[5]. Consistent with this broader “survival signaling” perspective, the Augustin et al’s review[47] further discusses neuroprotective actions reported for ANG1 through phosphoinositide 3-kinase activation (e.g., anti-apoptotic effects and neurite outgrowth in experimental neuronal systems), while also stressing uncertainty about whether such neuronal effects are mediated by neuronal TIE2 in vivo vs alternative mechanisms (such as integrin-dependent interactions)[5,47].
Next-generation gene-therapy concepts for DR/DME are increasingly framed around the idea that sustained intraocular protein expression can be used not only to suppress VEGF-driven leakage and neovascularization, but also to address upstream processes within the diabetic retinal NVU. In this direction, multi-target vector designs are emerging as a logical extension of the “retinal biofactory” paradigm: For example, 4D-150 combines an aflibercept transgene with a VEGF-C - inhibitory microRNA, aiming to modulate multiple angiogenic/permeability axes with a single intravitreal injection[35,36]. Beyond angiogenesis alone, the broader DR biology - characterized by cytokine-rich inflammation, endothelial–pericyte dysregulation, and blood-retinal barrier fragility - supports strategies that couple anti-angiogenic activity with barrier-stabilizing and anti-inflammatory pathway modulation, including targets within Augustin et al’s signaling system[47] implicated in vascular maturation, survival signaling, and vessel stability[3,45-47].
This expanded targeting logic also aligns with evidence that neuroretinal dysfunction can be an early, progressive component of diabetic retinal disease, motivating interest in approaches that integrate vascular protection with neuronal preservation rather than treating only visible microvascular lesions[5,12]. However, most of these platforms remain preclinical or early translational, and the diabetic retinal microenvironment - already primed by inflammation and altered barrier permeability - may influence vector tolerability, immune activation, and durability of expression, underscoring the need for indication-specific evidence as these programs advance[3,40].
Genome editing extends ocular gene therapy beyond the “biofactory” model of sustained intraocular protein expression. However, the permanent nature of genome editing imposes a higher safety bar than reversible pharmacologic inhibition. In adult mouse eyes, endogenous VEGF produced locally by the RPE provides trophic support for retinal homeostasis: Conditional VEGFA deletion in adult RPE rapidly ablates the choriocapillaris and is accompanied by cone photoreceptor dysfunction and vision loss, indicating that near-complete removal of RPE-derived VEGF can compromise outer retinal function[48]. Consistently, experiments manipulating VEGF isoform bioavailability show that limiting RPE output to a poorly diffusible VEGF188-only state produces age-dependent degeneration of the RPE-Bruch’s membrane-choriocapillaris complex, with associated photoreceptor apoptosis and decline in visual function, reinforcing that adequate soluble RPE-derived VEGF signaling contributes to long-term maintenance of the choriocapillaris and outer retina[49].
Genome-editing techniques have significantly advanced ocular genetic medicine; yet, their direct applicability to diabetic retinal disease has been constrained. In contrast to monogenic hereditary illnesses, DR is a complex, multi
Beyond nuclease-based CRISPR editing, newer approaches such as base editing aim to correct pathogenic variants without inducing Cas9 double-strand breaks and without relying on homology-directed repair. In adult RD12 mice, subretinal delivery of a lentiviral vector expressing an adenine base editor and a guide targeting the Rpe65 nonsense mutation achieved measurable on-target correction with minimal indel formation, no detectable off-target editing above background at top candidate sites, restored RPE65 expression, and improved retinal and visual function readouts[50]. For CRISPR strategies aimed at suppressing ocular angiogenesis by directly disrupting VEGF, proof-of-concept studies in a laser-induced CNV model show that AAV-delivered CRISPR-Cas9 can reduce VEGF and suppress CNV, and that in vivo outcomes can differ between Cas9 ortholog systems even when delivery is comparable[51]. More broadly, reviews of CRISPR applications for ocular angiogenesis highlight potential advantages over conventional gene-therapy “biofactory” approaches - including the ability to target intracellular mediators or regulatory elements and to multiplex edits - while emphasizing key translational constraints such as uncertainty in therapeutic thresholds, challenges in cell-specific targeting, and persistent concerns regarding off-target effects, immune responses to CRISPR components, and the risk of permanently suppressing physiologic pathways like VEGF signaling[52].
The most clinically mature example of in vivo ocular genome editing comes from inherited retinal disease, where the first human trial was initiated in late 2019 using EDIT-101: An all-in-one AAV5 vector that packages Staphylococcus aureus Cas9 (SaCas9) together with a pair of CEP290 gRNAs and is delivered subretinally to generate productive edits (notably deletions or inversions) that remove the intronic mutation–containing region and thereby restore correct CEP290 splicing and expression[52]. The preclinical development program demonstrated editing activity in mature human photoreceptors (retinal explants), quantified on-target editing kinetics and dose–response after subretinal dosing in a humanized CEP290 knock-in mouse, and showed on-target editing in nonhuman primate photoreceptors, alongside a structured assessment of guide specificity and ocular tolerability - illustrating both the feasibility of retinal editing in vivo and the practical delivery trade-offs of localized subretinal administration[52].
By contrast, neovascular retinal disease (including conditions discussed under ocular angiogenesis such as proliferative DR and DME) is multifactorial, so CRISPR strategies are framed less as “mutation correction” and more as attempts to permanently suppress angiogenesis by targeting extracellular effectors (e.g., VEGF) and/or upstream intracellular mediators or regulatory elements (e.g., hypoxia-inducible factor 1-α or hypoxia-responsive control regions), potentially with cell-specific delivery and multiplexed gRNAs. At the same time, this translation is constrained by core concerns highlighted for ocular angiogenesis: Permanent suppression of physiologic pathways, the stochastic and hard-to-titrate nature of editing outcomes (mosaicism and uncertain therapeutic thresholds), and safety issues including off-target effects and immune considerations - particularly because VEGF-related signaling contributes to normal vascular and neural maintenance, and chronic VEGF suppression has been linked to atrophic complications in clinical contexts[53].
Preclinical work supports the feasibility of attenuating VEGF-A signaling by editing VEGFA directly in ocular cell types that contribute substantially to intraocular VEGF production[54]. In human ARPE-19 retinal pigment epithelial cells and MIO-M1 Müller cells, delivery of SpCas9 ribonucleoprotein (RNP) targeting exon 1 generated indels at the expected cut site and produced a robust reduction in secreted VEGF-A protein (approximately 43% in RPE and approximately 38% in Müller cells), without a detectable loss in cell counts in the tested conditions[54]. Because indiscriminate editing may expose non-target retinal compartments, cell-restricted strategies have also been explored. An RPE-selective CRISPR/Cas9 platform was created by placing Cas9 under the RPE-specific vitelliform macular dystrophy 2 promoter, yielding Cas9 expression and editing largely confined to the RPE in both the mouse retina and human retinal organoids.
Using this approach, RPE-specific VEGFA knockout achieved regression of laser-induced choroidal neovascularization while avoiding unwanted knockout in the neural retina, with efficacy comparable to ubiquitous VEGFA knockout in the same experimental setting[55]. Delivery modality remains a central determinant of both efficacy and tolerability. Beyond vector-based expression systems, multiple studies have pursued nonviral, “hit-and-run” editing via transient RNP delivery to limit nuclease exposure over time. In a subretinal, lipoplex-based paradigm using SpCas9 complexed with chemically modified sgRNAs, targeted indel formation in retinal tissue was achievable after a single administration, but higher RNP loads were associated with signs consistent with toxicity/autofluorescence in vivo - highlighting a dose-delivery trade-off that is critical for translation[56].
Immune recognition of editing components represents an additional translational constraint, particularly for approaches that entail persistent nuclease expression. Preclinical work in mice has shown that, even when AAV-CRISPR achieves initial editing, pre-existing immunity to Cas9 can precipitate a strong CD8+ T-cell response against Cas9-expressing cells, accompanied by apoptosis and tissue injury markers, followed by loss of vector genomes and near-complete elimination of edited cells over time, effectively erasing the intended editing effect[57]. While the eye benefits from anatomical barriers and relative immune privilege, ocular gene therapies can nonetheless elicit innate and adaptive immune responses that, under certain circumstances, can drive inflammation and reduce therapeutic efficacy[58].
Accordingly, mitigation strategies emphasized across the ocular gene-editing literature include minimizing duration of Cas exposure (e.g., transient delivery formats or self-limiting designs rather than sustained expression), restricting expression through cell-specific regulatory elements when vector expression is used, and reducing innate immune activation triggered by nucleic acids - for example, avoiding immunostimulatory guide-RNA features and favoring guide formats reported to be less immunogenic in mammalian cells[57,58].
In DR/DME, genome editing can be framed as a plausible next-generation strategy because it acts at the DNA level and, in principle, enables durable suppression of pro-angiogenic programs beyond the “biofactory” model of expressing VEGF antagonists. However, this is not simply a longer-lasting version of anti-VEGF gene therapy: The key translational concerns are the prospect of permanently suppressing physiologic pathways, the variable and mosaic nature of editing outcomes, and the risk of off-target changes - issues that are amplified when nucleases are delivered by viral vectors with sustained intraocular expression and have therefore tempered enthusiasm to date. Preclinical retina work with base editors helps explain both the promise and the bar for safety: In adult RD12 mice, subretinal lentiviral delivery of an adenine base editor plus sgRNA corrected a pathogenic Rpe65 nonsense mutation (reported up to approximately 29% in vivo) with low indel formation and no detectable off-target editing above background at the screened sites, restoring RPE65 expression and near-normal retinal/visual function; at the same time, the authors explicitly note that safer delivery approaches (e.g., alternative vector designs) and thorough, disease- and guide-specific efficacy/safety assess
Translating gene therapy from a promising concept to routine care for DR and DME depends on three tightly linked pillars: Ocular safety, durability of effect, and careful patient selection. In recent AAV-based programs, intraocular inflammation has emerged as a central safety signal, particularly after intravitreal delivery. Reported events range from anterior segment inflammation and vitritis to severe, dose-limiting reactions, and in DME, the INFINITY experience with intravitreal AAV2.7m8-aflibercept expression included severe inflammation and hypotony, ultimately leading to discontinuation of further development for DME[50-52]. Route-related differences in inflammatory phenotype have also been described in the context of gene therapy-associated uveitis (GTAU), with intravitreal administration reported at higher rates than subretinal or suprachoroidal delivery, and with vitritis/anterior segment inflammation being a common manifestation after intravitreal injection. These observations reinforce that dose, vector/capsid features, and route of delivery can materially shape ocular tolerability and inflammatory outcomes[51,52].
At the same time, the diabetic disease context is relevant to risk assessment. In DR/DME, chronic hyperglycemia is linked to oxidative stress, inflammation, and microvascular dysfunction, and clinical comparisons across indications suggest that a therapy/dose tolerated in neovascular AMD may not behave identically in DME. This supports a conservative approach to patient selection and prophylaxis, mirroring how current programs often define eligibility by disease stage and prior treatment behavior (e.g., DME eyes demonstrating responsiveness to VEGF inhibition; DR cohorts without center-involved DME) and frequently incorporate corticosteroid regimens and close monitoring for inflammatory signs. Durability is the major appeal of AAV-mediated “biofactory” strategies - continuous intraocular production of an anti-VEGF biologic could reduce the injection burden. In neovascular AMD gene-therapy programs, multi-year follow-up has been reported, with substantial reductions in rescue anti-VEGF injections, including cohorts in which many patients remained supplemental-injection-free at 2 years and subretinal anti-VEGF gene therapy showed reduced injection frequency over 4 years, supporting the plausibility of prolonged intraocular expression[19,29,43].
However, durability is not guaranteed. Recombinant AAV genomes generally persist as episomes, and transgene performance can be influenced by host immunity (capsid- and transgene-directed responses), pre-existing or induced neutralizing antibodies that can reduce efficacy, and by promoter/cassette behavior (including susceptibility to silencing in some contexts). During tissue division, episomes can also be diluted with cell replication; more broadly, immune-mediated elimination of transduced cells is a recognized mechanism that can shorten functional persistence. Against the background of diabetic retinal disease, where ongoing metabolic and vascular stressors are intrinsic to the patho
Long-term human follow-up of early retinal gene-augmentation trials shows that functional gains can be measurable yet incomplete and not always durable. In a phase 1-2 open-label study of subretinal recombinant AAV 2/2-RPE65 in 12 patients (6-23 years) with RPE65-associated Leber congenital amaurosis type, retinal sensitivity improved in a subset of treated eyes and generally peaked at approximately 6-12 months before declining during 3-year follow-up; no associated ERG improvement was detected, and visual acuity did not show a consistent benefit. Intraocular inflammation occurred in some participants (predominantly at the higher dose), and clinically significant visual acuity deterioration was observed in a minority[59].
Patient selection is pivotal for balancing potential benefit against procedural and biologic risk. In current gene-therapy development for diabetic eye disease, clinical programs have largely centered on two pragmatic populations: (1) Eyes with DME, where standard-of-care anti-VEGF therapy typically requires close monitoring and repeated intravitreal injections, and where reduced visit/injection frequency in real-world practice is linked to poorer long-term outcomes; and (2) Higher-risk NPDR without center-involved DME, as exemplified by suprachoroidal ABBV-RGX-314 studies enrolling moderately severe or severe NPDR (and select mild PDR) with good baseline vision and no recent anti-VEGF exposure. This focus aligns with the “biofactory” rationale - durable intraocular expression of an anti-VEGF strategy intended to reduce the cumulative injection burden - while recognizing that route of delivery is itself a key determinant of safety and distribution (e.g., office-based suprachoroidal delivery with broad posterior biodistribution, but outside a fully immune-privileged compartment). At the same time, the diabetic retina is increasingly understood as a neurovascular complication in which early dysfunction of the NVU is biologically relevant and better preventive/interventional strate
A further complexity is interindividual heterogeneity in disease biology and therapeutic response. Pharmacogenomic and candidate-gene literature suggest that DR susceptibility and severity are influenced by diverse genetic factors (e.g., human leukocyte antigen associations and multiple single-nucleotide polymorphisms across pathways) and that response to anti-VEGF therapy in DME shows marked variability, with evidence of molecular differences between responders and incomplete/nonresponders (including differences in transcriptional and cytokine-related signals and VEGF-pathway modulation). In parallel, a neurovascular framing of diabetic retinal disease emphasizes the need for improved pheno
Overall, successful translation of gene therapy for DR and DME will depend on demonstrating durable biologic activity with an acceptable safety profile in appropriately selected patients, alongside practical delivery approaches. While early clinical experience is encouraging in some programs, vigilance is warranted: Severe intraocular inflammation and hypotony have been reported in intravitreal gene-therapy development for DME, and even with alternative routes, immune responses and inflammation remain central concerns. In addition, chronic VEGF suppression raises theoretical safety questions because gene therapy is not readily “switchable off”, and future work has explicitly highlighted the need to better define prophylaxis/monitoring strategies and to identify biomarkers that can detect and quantify ocular immune responses. As longer follow-up accumulates and trials increasingly align enrollment and outcomes with the complex neurovascular biology of diabetic retinal disease, the field will be better positioned to judge whether gene therapy can deliver meaningful disease modification and sustained reduction in treatment burden, rather than simply re-packaging current anti-VEGF paradigms[4,19,44,60].
The interpretation of the available findings necessitated a clear examination of methodological limitations. A significant portion of studies on ocular gene therapy for diabetic retinal disease originated from early-phase or single-arm research, often characterized by small cohort sizes, brief follow-up periods, and varied outcome definitions. Consequently, evidence was evaluated with caution, and findings were assessed in terms of developmental trajectory rather than confirmed therapeutic efficacy.
Intravitreal anti-VEGF therapy has reshaped the management of DME and advanced DR, but the outcomes achieved in pivotal trials rely on frequent visits and repeated injections - an intensity that is difficult to reproduce in everyday care. Real-world EMR cohorts consistently show lower treatment density and more modest vision gains. In one integrated health-system cohort, follow-up was often irregular (only about three-fifths of eyes had at least quarterly visits, with monthly follow-up being rare), and most eyes received no more than three injections over 12 months, with an average gain of roughly five ETDRS-equivalent letters. In a much larger multi-site United States database, first-year treatment averaged about six to seven injections with a mean gain of approximately 4 letters; half of the eyes received six or fewer injections, and fewer than one-fifth received near-monthly dosing[7,8]. Importantly, visual outcomes generally improved as injection frequency increased (beyond the first couple of injections), while eyes starting with relatively good baseline acuity showed ceiling effects and were more likely to lose letters over one year. Together, these datasets explain why routine-practice outcomes often fall short of randomized trials and emphasize how visit burden and repeated injections constrain long-term disease control in DME[7,8].
Concurrently, mechanistic and clinical evidence have reframed diabetic retinal disease as a tissue-specific neuro
The strongest proof-of-concept for durable intraocular anti-VEGF expression in a common retinal disease is currently illustrated by subretinal AAV8 RGX-314 in neovascular AMD. In the open-label, multicenter phase 1/2a dose-escalation study, doses ≥ 6 × 1010 genome copies/eye produced sustained aqueous levels of the expressed anti-VEGF antigen-binding fragment through 2 years, with stable or improved BCVA and central retinal thickness and, in many participants, few or no supplemental anti-VEGF injections after a single administration[18]. Safety signals included dose-related retinal pigmentary changes (more frequent at higher doses) and one serious adverse event possibly related to the highest dose, while investigators reported no clinically recognized immune responses and no ocular inflammation beyond that expected after vitrectomy. Overall, the trial supports the feasibility of sustained VEGF-A suppression with a single subretinal gene-therapy procedure, while also showing that dose and long-term tolerability matter[18].
This AMD experience is relevant for DME largely because current anti-VEGF care remains vulnerable to under
However, diabetic retinal disease cannot be assumed to mirror AMD in safety or durability. The INFINITY phase 2 program testing intravitreal ADVM-022 (ixo-vec; AAV.7m8-aflibercept) in DME illustrates this point: While fewer ixo-vec–treated eyes required rescue aflibercept than control in early follow-up, the trial was complicated by dose-dependent inflammation, and the 6 × 1011 vg dose was associated with severe inflammation and hypotony, leading the sponsor to discontinue development for DME[19]. The ClinicalTrials.gov record further notes that the study was unmasked in April 2021 for enhanced safety monitoring after a suspected unexpected serious adverse reaction in the high-dose arm, and that although intraocular inflammation was often mild to moderate and generally responsive to corticosteroid drops, dose-limiting toxicities affected the high-dose benefit-risk profile[38]. Together, these data argue that “one-and-done” is not a default property of ocular gene therapy; it is a testable claim that must be re-established for each vector, dose, route, and disease context[19,38].
Finally, durability and re-treatment remain legitimate questions. In the RGX-314 study, protein-expression kinetics differed across dose cohorts (with uncertainty about why higher-dose cohorts showed a post-peak decline), and the authors explicitly highlight the value of longer follow-up for ongoing evaluation[18]. More broadly, vector immunology remains a practical constraint: While pre-existing anti-AAV8 immunity did not appear to reduce expression after a single subretinal RGX-314 administration, concerns persist that pre-existing or treatment-emergent neutralizing antibodies can reduce transduction efficiency and complicate re-administration strategies[18,19].
Patient selection will largely determine whether ocular gene therapy for DR is positioned primarily as a means to reduce the burden of repeated intravitreal anti-VEGF injections or as an intervention intended to improve retinopathy severity. Current clinical development has focused on eyes with DME that ordinarily require ongoing anti-VEGF treatment and on higher-risk nonproliferative DR populations (e.g., moderately severe to severe NPDR, or mild PDR without active center-involved DME), where clinically meaningful endpoints can be captured within conventional trial follow-up - such as the need for rescue injections and changes in the DRSS. In this setting, interim findings from programs such as SPECTRA (4D-150) and ALTITUDE (ABBV-RGX-314) are described as promising for both reducing treatment burden and improving DRSS, while experience from INFINITY (Ixo-vec/ADVM-022) underscores that severe intraocular inflammation and hypotony can occur and may be development-limiting in DME[19].
Taken together with the broader therapeutic landscape, this supports a cautious, stepwise conclusion. In DR/DME, targeting VEGF-driven microvascular pathology remains the only clearly successful therapeutic strategy to date, but it is typically deployed in later stages, requires regular intravitreal injections, and does not yield optimal responses in all patients - factors that collectively motivate the development of durable alternatives[3]. A pragmatic near-term translational role for gene therapy, consistent with current trials, is therefore the “biofactory” concept: Sustained intraocular production of anti-VEGF (or related anti-angiogenic) effectors to reduce monitoring and injection burden while potentially improving DR severity metrics[19]. However, the same durability that is attractive clinically also concentrates safety expectations, because inflammatory adverse events (including gene therapy-associated uveitis) have been reported, and chronic VEGF suppression cannot readily be “switched off”. Beyond transgene delivery, genome-editing strategies proposed for DR/DME (e.g., CRISPR/Cas approaches aimed at VEGF-pathway genes) introduce additional constraints highlighted in current evidence - variable editing efficiency, practical vector-packaging limitations for commonly used Cas9 systems, and concern for off-target mutations - making precise cell targeting and long-term safety surveillance central prerequisites for any future expansion toward earlier, asymptomatic stages[19].
This review has several limitations that reflect both the rapidly evolving nature of ocular gene therapy and the narrative methodology adopted. First, the clinical evidence base for gene therapy in DR and DME remains immature, dominated by early- to mid-phase studies, with much of the strongest human proof-of-concept derived from programs originally developed for neovascular AMD. As a result, long-term durability and safety in diabetic retinal disease remain incom
Second, substantial heterogeneity across studies restricts cross-trial interpretability and precludes meaningful quantitative synthesis. Programs differ in capsid/vector design, expression cassettes, administration routes (intravitreal, subretinal, suprachoroidal), dosing strategies, and prophylactic anti-inflammatory regimens - variables that materially influence ocular biodistribution, immunogenicity, and inflammatory risk. Moreover, enrolled populations vary in disease phenotype and stage (e.g., DME vs high-risk NPDR without center-involving DME), baseline vision, prior anti-VEGF exposure, and systemic metabolic status, each of which may modulate both efficacy and safety in the diabetic retinal microenvironment. These sources of variability require cautious interpretation of “positive signals” and support treating current evidence as hypothesis-generating rather than conclusive[19,40,44].
Third, the narrative design - selected to enable contextual integration across mechanistic, translational, and clinical domains - does not provide the structured rigor of systematic reviews or meta-analyses. Although the manuscript was built on a defined literature search strategy and qualitative appraisal of peer-reviewed studies, selection bias and publication bias remain possible, particularly in a field where unfavorable outcomes, terminated programs, or incomplete datasets may be less visible in the published record. Finally, because the evidence base is moving quickly, conclusions drawn here should be revisited as longer follow-up from ongoing DR/DME programs becomes available[19,38].
The existing literature appeared to be influenced by early-stage innovation programs, frequently supported by industry and occasionally presented initially as intermediate datasets. In these contexts, adverse or ambiguous results may be less likely to be published in their entirety in peer-reviewed journals, and the duration of follow-up may be inadequate to assess durability or safety. These considerations have heightened the likelihood of selective reporting and necessitated careful evaluation of seemingly effective signals. The outcomes of this research are thus characterized as tentative and dependent on validation by bigger, longitudinal studies.
Several convergent trends are likely to shape the next phase of gene therapy development for DR and DME. Continued optimization of AAV platforms - through capsid engineering and improved expression design - aims to enhance retinal transduction efficiency while mitigating immune activation, a priority given that intraocular inflammation has emerged as a defining safety constraint in multiple clinical programs, particularly after intravitreal delivery. In parallel, delivery innovation is expected to remain central: Suprachoroidal administration is being actively explored in DR/DME because it offers an outpatient, potentially scalable route with broad posterior-segment exposure, while also altering the immune and barrier context encountered by the vector compared with intravitreal injection[19,40,44].
Beyond “anti-VEGF biofactory” approaches, the field is moving toward broader pathway coverage that better reflects the neurovascular-unit framing of diabetic retinal disease. Multi-target constructs - already exemplified by platforms combining VEGF-A inhibition with modulation of additional angiogenic or permeability mediators - represent a logical extension of the durability paradigm and may help address VEGF-independent components of persistent disease activity in subsets of patients. Over time, more refined patient stratification is also likely to become increasingly important. Trial design and eventual clinical implementation will benefit from integrating quantitative ocular biomarkers and disease phenotyping (including severity staging and imaging-based assessment of neurovascular injury) together with systemic characteristics that influence inflammatory risk and treatment response, thereby improving the benefit–risk balance and clarifying “where” along the DR/DME continuum durable gene-based modulation is most appropriate[35,36,44].
Genome editing remains a more distant - but conceptually transformative - frontier. The permanent nature of editing imposes a substantially higher safety bar than reversible pharmacologic inhibition or protein-expression gene therapy, especially for pathways with physiologic roles in retinal and choroidal homeostasis. The translational constraints emphasized in the ocular gene-editing literature - off-target risk, mosaic/variable editing thresholds, and immune responses to Cas effectors or other editing components - are particularly relevant when considering approaches for multifactorial angiogenic disease rather than monogenic degeneration. Accordingly, if editing-based strategies are ever advanced toward DR/DME, they will likely require conservative target selection, stringent cell-restricted control, and delivery formats that minimize duration of nuclease exposure, coupled with prolonged safety monitoring[50,52,57,58].
Gene therapy has emerged as a credible translational strategy for diabetic retinal disease, driven by the clinical need for durability in conditions that currently demand repeated intravitreal anti-VEGF injections and intensive monitoring. The core therapeutic premise - turning ocular tissues into sustained producers of a therapeutic biologic - offers a potential path to reduce treatment burden while maintaining meaningful disease control, and multi-year experience in neovascular AMD provides proof-of-concept that durable intraocular anti-VEGF expression can be achieved after a single administration in selected settings. At the same time, DR and DME present disease-specific challenges that require indication-tailored validation rather than simple extrapolation from neovascular AMD. Despite the potential for prolonged intraocular therapeutic expression using gene-based methods, clinical advances in diabetic retinal disease have been constrained. Current evidence predominantly indicates exploratory or preliminary development, with long-term safety, durability, and generalizability still to be determined. Gene therapy should now be considered an evolving strategy, with its definitive position contingent on the validation of satisfactory safety profiles and consistent clinical benefits across broader diabetic populations.
The diabetic retina is characterized by chronic metabolic stress, blood-retinal barrier fragility, and a cytokine-rich inflammatory milieu, all of which may influence vector tolerability, immune activation, and the stability of transgene expression. Early DR/DME-focused development supports both sides of this balance: Ongoing programs have reported encouraging signals consistent with reduced treatment burden and improvements in clinically relevant measures in some settings, yet severe inflammation and hypotony in intravitreal DME development underscore that durability cannot be pursued at the expense of ocular safety and that route, dose, and vector design matter decisively.
In the near term, the most defensible trajectory is incremental: Positioning gene therapy as an evolutionary complement to established anti-VEGF paradigms, refined through improved vector engineering, safer and more scalable delivery routes, and biomarker-informed patient selection. Longer follow-up and appropriately powered late-phase trials will be essential to define durability, clarify long-horizon safety, and determine whether gene therapy can move beyond sustained VEGF suppression toward broader, mechanism-based disease modification aligned with the neurovascular biology of diabetic retinal disease.
| 1. | Wong TY, Cheung CM, Larsen M, Sharma S, Simó R. Diabetic retinopathy. Nat Rev Dis Primers. 2016;2:16012. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 933] [Cited by in RCA: 821] [Article Influence: 82.1] [Reference Citation Analysis (3)] |
| 2. | Teo ZL, Tham YC, Yu M, Chee ML, Rim TH, Cheung N, Bikbov MM, Wang YX, Tang Y, Lu Y, Wong IY, Ting DSW, Tan GSW, Jonas JB, Sabanayagam C, Wong TY, Cheng CY. Global Prevalence of Diabetic Retinopathy and Projection of Burden through 2045: Systematic Review and Meta-analysis. Ophthalmology. 2021;128:1580-1591. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1948] [Cited by in RCA: 1418] [Article Influence: 283.6] [Reference Citation Analysis (8)] |
| 3. | Antonetti DA, Silva PS, Stitt AW. Publisher Correction: Current understanding of the molecular and cellular pathology of diabetic retinopathy. Nat Rev Endocrinol. 2025;21:62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 4. | Simó R, Stitt AW, Gardner TW. Neurodegeneration in diabetic retinopathy: does it really matter? Diabetologia. 2018;61:1902-1912. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 499] [Cited by in RCA: 439] [Article Influence: 54.9] [Reference Citation Analysis (8)] |
| 5. | Sohn EH, van Dijk HW, Jiao C, Kok PH, Jeong W, Demirkaya N, Garmager A, Wit F, Kucukevcilioglu M, van Velthoven ME, DeVries JH, Mullins RF, Kuehn MH, Schlingemann RO, Sonka M, Verbraak FD, Abràmoff MD. Retinal neurodegeneration may precede microvascular changes characteristic of diabetic retinopathy in diabetes mellitus. Proc Natl Acad Sci U S A. 2016;113:E2655-E2664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 282] [Cited by in RCA: 508] [Article Influence: 50.8] [Reference Citation Analysis (2)] |
| 6. | Cantrell RA, Lum F, Chia Y, Morse LS, Rich WL 3rd, Salman CA, Willis JR. Treatment Patterns for Diabetic Macular Edema: An Intelligent Research in Sight (IRIS®) Registry Analysis. Ophthalmology. 2020;127:427-429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 39] [Article Influence: 5.6] [Reference Citation Analysis (1)] |
| 7. | Holekamp NM, Campbell J, Almony A, Ingraham H, Marks S, Chandwani H, Cole AL, Kiss S. Vision Outcomes Following Anti-Vascular Endothelial Growth Factor Treatment of Diabetic Macular Edema in Clinical Practice.. Am J Ophthalmol. 2018;191:83-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 83] [Article Influence: 10.4] [Reference Citation Analysis (1)] |
| 8. | Ciulla TA, Pollack JS, Williams DF. Visual acuity outcomes and anti-VEGF therapy intensity in diabetic macular oedema: a real-world analysis of 28 658 patient eyes. Br J Ophthalmol. 2021;105:216-221. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 123] [Article Influence: 20.5] [Reference Citation Analysis (1)] |
| 9. | Ciulla TA, Bracha P, Pollack J, Williams DF. Real-world Outcomes of Anti-Vascular Endothelial Growth Factor Therapy in Diabetic Macular Edema in the United States. Ophthalmol Retina. 2018;2:1179-1187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 127] [Article Influence: 15.9] [Reference Citation Analysis (1)] |
| 10. | Busch C, Fraser-Bell S, Iglicki M, Lupidi M, Couturier A, Chaikitmongkol V, Giancipoli E, Rodríguez-Valdés PJ, Gabrielle PH, Laíns I, Santos AR, Cebeci Z, Amphornphruet A, Degenhardt V, Unterlauft JD, Cagini C, Mané-Tauty V, D'Amico Ricci G, Hindi I, Agrawal K, Chhablani J, Loewenstein A, Zur D, Rehak M; International Retina Group. Real-world outcomes of non-responding diabetic macular edema treated with continued anti-VEGF therapy versus early switch to dexamethasone implant: 2-year results. Acta Diabetol. 2019;56:1341-1350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 61] [Article Influence: 8.7] [Reference Citation Analysis (1)] |
| 11. | Busch C, Zur D, Fraser-Bell S, Laíns I, Santos AR, Lupidi M, Cagini C, Gabrielle PH, Couturier A, Mané-Tauty V, Giancipoli E, Ricci GD, Cebeci Z, Rodríguez-Valdés PJ, Chaikitmongkol V, Amphornphruet A, Hindi I, Agrawal K, Chhablani J, Loewenstein A, Iglicki M, Rehak M; International Retina Group. Shall we stay, or shall we switch? Continued anti-VEGF therapy versus early switch to dexamethasone implant in refractory diabetic macular edema. Acta Diabetol. 2018;55:789-796. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 101] [Article Influence: 12.6] [Reference Citation Analysis (1)] |
| 12. | Stitt AW, Curtis TM, Chen M, Medina RJ, McKay GJ, Jenkins A, Gardiner TA, Lyons TJ, Hammes HP, Simó R, Lois N. The progress in understanding and treatment of diabetic retinopathy. Prog Retin Eye Res. 2016;51:156-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 990] [Cited by in RCA: 809] [Article Influence: 80.9] [Reference Citation Analysis (4)] |
| 13. | Maguire AM, Simonelli F, Pierce EA, Pugh EN Jr, Mingozzi F, Bennicelli J, Banfi S, Marshall KA, Testa F, Surace EM, Rossi S, Lyubarsky A, Arruda VR, Konkle B, Stone E, Sun J, Jacobs J, Dell'Osso L, Hertle R, Ma JX, Redmond TM, Zhu X, Hauck B, Zelenaia O, Shindler KS, Maguire MG, Wright JF, Volpe NJ, McDonnell JW, Auricchio A, High KA, Bennett J. Safety and efficacy of gene transfer for Leber's congenital amaurosis. N Engl J Med. 2008;358:2240-2248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1728] [Cited by in RCA: 1658] [Article Influence: 92.1] [Reference Citation Analysis (0)] |
| 14. | Russell S, Bennett J, Wellman JA, Chung DC, Yu ZF, Tillman A, Wittes J, Pappas J, Elci O, McCague S, Cross D, Marshall KA, Walshire J, Kehoe TL, Reichert H, Davis M, Raffini L, George LA, Hudson FP, Dingfield L, Zhu X, Haller JA, Sohn EH, Mahajan VB, Pfeifer W, Weckmann M, Johnson C, Gewaily D, Drack A, Stone E, Wachtel K, Simonelli F, Leroy BP, Wright JF, High KA, Maguire AM. Efficacy and safety of voretigene neparvovec (AAV2-hRPE65v2) in patients with RPE65-mediated inherited retinal dystrophy: a randomised, controlled, open-label, phase 3 trial. Lancet. 2017;390:849-860. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 931] [Cited by in RCA: 1479] [Article Influence: 164.3] [Reference Citation Analysis (1)] |
| 15. | Campochiaro PA, Nguyen QD, Shah SM, Klein ML, Holz E, Frank RN, Saperstein DA, Gupta A, Stout JT, Macko J, DiBartolomeo R, Wei LL. Adenoviral vector-delivered pigment epithelium-derived factor for neovascular age-related macular degeneration: results of a phase I clinical trial. Hum Gene Ther. 2006;17:167-176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 231] [Article Influence: 11.6] [Reference Citation Analysis (1)] |
| 16. | Campochiaro PA, Lauer AK, Sohn EH, Mir TA, Naylor S, Anderton MC, Kelleher M, Harrop R, Ellis S, Mitrophanous KA. Lentiviral Vector Gene Transfer of Endostatin/Angiostatin for Macular Degeneration (GEM) Study. Hum Gene Ther. 2017;28:99-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 150] [Article Influence: 15.0] [Reference Citation Analysis (1)] |
| 17. | Khanani AM, Boyer DS, Wykoff CC, Regillo CD, Busbee BG, Pieramici D, Danzig CJ, Joondeph BC, Major JC Jr, Turpcu A, Kiss S. Safety and efficacy of ixoberogene soroparvovec in neovascular age-related macular degeneration in the United States (OPTIC): a prospective, two-year, multicentre phase 1 study. EClinicalMedicine. 2024;67:102394. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 59] [Reference Citation Analysis (1)] |
| 18. | Campochiaro PA, Avery R, Brown DM, Heier JS, Ho AC, Huddleston SM, Jaffe GJ, Khanani AM, Pakola S, Pieramici DJ, Wykoff CC, Van Everen S. Gene therapy for neovascular age-related macular degeneration by subretinal delivery of RGX-314: a phase 1/2a dose-escalation study. Lancet. 2024;403:1563-1573. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 93] [Article Influence: 46.5] [Reference Citation Analysis (1)] |
| 19. | Odio-Herrera M, Orozco-Loaiza G, Wu L. Gene Therapy in Diabetic Retinopathy and Diabetic Macular Edema: An Update. J Clin Med. 2025;14:3205. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 20. | Cahoon JM, Rai RR, Carroll LS, Uehara H, Zhang X, O'Neil CL, Medina RJ, Das SK, Muddana SK, Olson PR, Nielson S, Walker K, Flood MM, Messenger WB, Archer BJ, Barabas P, Krizaj D, Gibson CC, Li DY, Koh GY, Gao G, Stitt AW, Ambati BK. Intravitreal AAV2.COMP-Ang1 Prevents Neurovascular Degeneration in a Murine Model of Diabetic Retinopathy. Diabetes. 2015;64:4247-4259. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 54] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 21. | Araújo RS, Bitoque DB, Silva GA. Dual-Acting Antiangiogenic Gene Therapy Reduces Inflammation and Regresses Neovascularization in Diabetic Mouse Retina. Mol Ther Nucleic Acids. 2020;22:329-339. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 22. | Rowe LW, Ciulla TA. Gene Therapy for Non-Hereditary Retinal Disease: Age-Related Macular Degeneration, Diabetic Retinopathy, and Beyond. Genes (Basel). 2024;15:720. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 34] [Reference Citation Analysis (1)] |
| 23. | Pierce EA, Aleman TS, Jayasundera KT, Ashimatey BS, Kim K, Rashid A, Jaskolka MC, Myers RL, Lam BL, Bailey ST, Comander JI, Lauer AK, Maguire AM, Pennesi ME. Gene Editing for CEP290-Associated Retinal Degeneration. N Engl J Med. 2024;390:1972-1984. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 134] [Article Influence: 67.0] [Reference Citation Analysis (1)] |
| 24. | Ewaisha R, Anderson KS. Immunogenicity of CRISPR therapeutics-Critical considerations for clinical translation. Front Bioeng Biotechnol. 2023;11:1138596. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 88] [Article Influence: 29.3] [Reference Citation Analysis (1)] |
| 25. | Huang J, Li J, Xu X, Li K. Adeno-Associated Virus Vectors in Retinal Gene Therapy: Challenges, Innovations, and Future Directions. Biomolecules. 2025;15:940. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 26. | Simó R, Hernández C. Novel approaches for treating diabetic retinopathy based on recent pathogenic evidence. Prog Retin Eye Res. 2015;48:160-180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 195] [Article Influence: 17.7] [Reference Citation Analysis (4)] |
| 27. | Busch C, Fraser-Bell S, Zur D, Rodríguez-Valdés PJ, Cebeci Z, Lupidi M, Fung AT, Gabrielle PH, Giancipoli E, Chaikitmongkol V, Okada M, Laíns I, Santos AR, Kunavisarut P, Sala-Puigdollers A, Chhablani J, Ozimek M, Hilely A, Unterlauft JD, Loewenstein A, Iglicki M, Rehak M; International Retina Group. Real-world outcomes of observation and treatment in diabetic macular edema with very good visual acuity: the OBTAIN study. Acta Diabetol. 2019;56:777-784. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 30] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 28. | Campochiaro PA. Molecular pathogenesis of retinal and choroidal vascular diseases. Prog Retin Eye Res. 2015;49:67-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 279] [Cited by in RCA: 441] [Article Influence: 40.1] [Reference Citation Analysis (1)] |
| 29. | Castro BFM, Steel JC, Layton CJ. AAV-Based Strategies for Treatment of Retinal and Choroidal Vascular Diseases: Advances in Age-Related Macular Degeneration and Diabetic Retinopathy Therapies. BioDrugs. 2024;38:73-93. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 24] [Reference Citation Analysis (1)] |
| 30. | Simó R, Hernández C. What else can we do to prevent diabetic retinopathy? Diabetologia. 2023;66:1614-1621. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 39] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 31. | Heier JS, Kherani S, Desai S, Dugel P, Kaushal S, Cheng SH, Delacono C, Purvis A, Richards S, Le-Halpere A, Connelly J, Wadsworth SC, Varona R, Buggage R, Scaria A, Campochiaro PA. Intravitreous injection of AAV2-sFLT01 in patients with advanced neovascular age-related macular degeneration: a phase 1, open-label trial. Lancet. 2017;390:50-61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 192] [Article Influence: 21.3] [Reference Citation Analysis (1)] |
| 32. | Xu D, Khan MA, Ho AC. Creating an Ocular Biofactory: Surgical Approaches in Gene Therapy for Acquired Retinal Diseases. Asia Pac J Ophthalmol (Phila). 2021;10:5-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 33. | Campochiaro PA, Akhlaq A. Sustained suppression of VEGF for treatment of retinal/choroidal vascular diseases. Prog Retin Eye Res. 2021;83:100921. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 84] [Article Influence: 14.0] [Reference Citation Analysis (1)] |
| 34. | Stewart MW, Rosenfeld PJ, Penha FM, Wang F, Yehoshua Z, Bueno-Lopez E, Lopez PF. Pharmacokinetic rationale for dosing every 2 weeks versus 4 weeks with intravitreal ranibizumab, bevacizumab, and aflibercept (vascular endothelial growth factor Trap-eye). Retina. 2012;32:434-457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 95] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 35. | Calton MA, Croze RH, Burns C, Beliakoff G, Vazin T, Szymanski P, Schmitt C, Klein A, Leong M, Quezada M, Holt J, Bolender G, Barglow K, Khoday D, Mason T, Delaria K, Hassanipour M, Kotterman M, Khanani AM, Schaffer D, Francis P, Kirn D. Design and Characterization of a Novel Intravitreal Dual-Transgene Genetic Medicine for Neovascular Retinopathies. Invest Ophthalmol Vis Sci. 2024;65:1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 16] [Reference Citation Analysis (1)] |
| 36. | Shirian JD, Shukla P, Singh RP. Exploring new horizons in neovascular age-related macular degeneration: novel mechanisms of action and future therapeutic avenues. Eye (Lond). 2025;39:40-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (1)] |
| 37. | AbbVie; REGENXBIO Inc.. RGX-314 Gene Therapy Administered in the Suprachoroidal Space for Participants With Diabetic Retinopathy (DR) With and Without Center Involved-Diabetic Macular Edema (CI-DME) (ALTITUDE®). [accessed 2026 January 24]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/study/NCT04567550 ClinicalTrials.gov Identifier: NCT04567550. |
| 38. | Adverum Biotechnologies, Inc.. ADVM-022 Intravitreal Gene Therapy for DME (INFINITY). [accessed 2026 January 24]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/study/NCT04418427 ClinicalTrials.gov Identifier: NCT04418427. |
| 39. | Clark J. 4D-150 in Patients With Diabetic Macular Edema. [accessed 2026 January 24]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/study/NCT05930561 ClinicalTrials.gov Identifier: NCT05930561. |
| 40. | Bucher K, Rodríguez-Bocanegra E, Dauletbekov D, Fischer MD. Immune responses to retinal gene therapy using adeno-associated viral vectors - Implications for treatment success and safety. Prog Retin Eye Res. 2021;83:100915. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 186] [Article Influence: 31.0] [Reference Citation Analysis (1)] |
| 41. | Fileta JB, Scott IU, Flynn HW Jr. Meta-analysis of infectious endophthalmitis after intravitreal injection of anti-vascular endothelial growth factor agents. Ophthalmic Surg Lasers Imaging Retina. 2014;45:143-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 178] [Article Influence: 14.8] [Reference Citation Analysis (1)] |
| 42. | Cepko CL, Vandenberghe LH. Retinal gene therapy coming of age. Hum Gene Ther. 2013;24:242-244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 39] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 43. | Naso MF, Tomkowicz B, Perry WL 3rd, Strohl WR. Adeno-Associated Virus (AAV) as a Vector for Gene Therapy. BioDrugs. 2017;31:317-334. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1138] [Cited by in RCA: 975] [Article Influence: 108.3] [Reference Citation Analysis (5)] |
| 44. | Lim Y, Campochiaro PA, Green JJ. Suprachoroidal Delivery of Viral and Nonviral Vectors for Treatment of Retinal and Choroidal Vascular Diseases. Am J Ophthalmol. 2025;277:518-533. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 45. | Semeraro F, Morescalchi F, Cancarini A, Russo A, Rezzola S, Costagliola C. Diabetic retinopathy, a vascular and inflammatory disease: Therapeutic implications. Diabetes Metab. 2019;45:517-527. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 177] [Cited by in RCA: 155] [Article Influence: 22.1] [Reference Citation Analysis (1)] |
| 46. | Tang J, Kern TS. Inflammation in diabetic retinopathy. Prog Retin Eye Res. 2011;30:343-358. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 924] [Cited by in RCA: 903] [Article Influence: 60.2] [Reference Citation Analysis (5)] |
| 47. | Augustin HG, Koh GY, Thurston G, Alitalo K. Control of vascular morphogenesis and homeostasis through the angiopoietin-Tie system. Nat Rev Mol Cell Biol. 2009;10:165-177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1229] [Cited by in RCA: 1084] [Article Influence: 63.8] [Reference Citation Analysis (1)] |
| 48. | Kurihara T, Westenskow PD, Bravo S, Aguilar E, Friedlander M. Targeted deletion of Vegfa in adult mice induces vision loss. J Clin Invest. 2012;122:4213-4217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 222] [Cited by in RCA: 279] [Article Influence: 19.9] [Reference Citation Analysis (1)] |
| 49. | Saint-Geniez M, Kurihara T, Sekiyama E, Maldonado AE, D'Amore PA. An essential role for RPE-derived soluble VEGF in the maintenance of the choriocapillaris. Proc Natl Acad Sci U S A. 2009;106:18751-18756. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 350] [Cited by in RCA: 427] [Article Influence: 25.1] [Reference Citation Analysis (0)] |
| 50. | Suh S, Choi EH, Leinonen H, Foik AT, Newby GA, Yeh WH, Dong Z, Kiser PD, Lyon DC, Liu DR, Palczewski K. Publisher Correction: Restoration of visual function in adult mice with an inherited retinal disease via adenine base editing. Nat Biomed Eng. 2020;4:1119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 51. | Chung SH, Mollhoff IN, Nguyen U, Nguyen A, Stucka N, Tieu E, Manna S, Meleppat RK, Zhang P, Nguyen EL, Fong J, Zawadzki R, Yiu G. Factors Impacting Efficacy of AAV-Mediated CRISPR-Based Genome Editing for Treatment of Choroidal Neovascularization. Mol Ther Methods Clin Dev. 2020;17:409-417. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 30] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 52. | Chung SH, Sin TN, Ngo T, Yiu G. CRISPR Technology for Ocular Angiogenesis. Front Genome Ed. 2020;2:594984. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 53. | Maeder ML, Stefanidakis M, Wilson CJ, Baral R, Barrera LA, Bounoutas GS, Bumcrot D, Chao H, Ciulla DM, DaSilva JA, Dass A, Dhanapal V, Fennell TJ, Friedland AE, Giannoukos G, Gloskowski SW, Glucksmann A, Gotta GM, Jayaram H, Haskett SJ, Hopkins B, Horng JE, Joshi S, Marco E, Mepani R, Reyon D, Ta T, Tabbaa DG, Samuelsson SJ, Shen S, Skor MN, Stetkiewicz P, Wang T, Yudkoff C, Myer VE, Albright CF, Jiang H. Development of a gene-editing approach to restore vision loss in Leber congenital amaurosis type 10. Nat Med. 2019;25:229-233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 323] [Cited by in RCA: 535] [Article Influence: 76.4] [Reference Citation Analysis (2)] |
| 54. | Ameri H, Murat C, Arbabi A, Jiang W, Janga SR, Qin PZ, Hamm-Alvarez SF. Reduced Expression of VEGF-A in Human Retinal Pigment Epithelial Cells and Human Muller Cells Following CRISPR-Cas9 Ribonucleoprotein-Mediated Gene Disruption. Transl Vis Sci Technol. 2020;9:23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 55. | Park J, Cui G, Lee H, Jeong H, Kwak JJ, Lee J, Byeon SH. CRISPR/Cas9 mediated specific ablation of vegfa in retinal pigment epithelium efficiently regresses choroidal neovascularization. Sci Rep. 2023;13:3715. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 56. | Holmgaard AB, Askou AL, Jensen EG, Alsing S, Bak RO, Mikkelsen JG, Corydon TJ. Targeted Knockout of the Vegfa Gene in the Retina by Subretinal Injection of RNP Complexes Containing Cas9 Protein and Modified sgRNAs. Mol Ther. 2021;29:191-207. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 57. | Li A, Tanner MR, Lee CM, Hurley AE, De Giorgi M, Jarrett KE, Davis TH, Doerfler AM, Bao G, Beeton C, Lagor WR. AAV-CRISPR Gene Editing Is Negated by Pre-existing Immunity to Cas9. Mol Ther. 2020;28:1432-1441. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 205] [Cited by in RCA: 173] [Article Influence: 28.8] [Reference Citation Analysis (1)] |
| 58. | Ren D, Fisson S, Dalkara D, Ail D. Immune Responses to Gene Editing by Viral and Non-Viral Delivery Vectors Used in Retinal Gene Therapy. Pharmaceutics. 2022;14:1973. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 37] [Article Influence: 9.3] [Reference Citation Analysis (1)] |
| 59. | Bainbridge JW, Mehat MS, Sundaram V, Robbie SJ, Barker SE, Ripamonti C, Georgiadis A, Mowat FM, Beattie SG, Gardner PJ, Feathers KL, Luong VA, Yzer S, Balaggan K, Viswanathan A, de Ravel TJ, Casteels I, Holder GE, Tyler N, Fitzke FW, Weleber RG, Nardini M, Moore AT, Thompson DA, Petersen-Jones SM, Michaelides M, van den Born LI, Stockman A, Smith AJ, Rubin G, Ali RR. Long-term effect of gene therapy on Leber's congenital amaurosis. N Engl J Med. 2015;372:1887-1897. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 506] [Cited by in RCA: 587] [Article Influence: 53.4] [Reference Citation Analysis (1)] |
| 60. | Agarwal A, Ingham SA, Harkins KA, Do DV, Nguyen QD. The role of pharmacogenetics and advances in gene therapy in the treatment of diabetic retinopathy. Pharmacogenomics. 2016;17:309-320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 1.0] [Reference Citation Analysis (1)] |