Published online Mar 25, 2026. doi: 10.5501/wjv.v15.i1.116055
Revised: January 1, 2026
Accepted: February 11, 2026
Published online: March 25, 2026
Processing time: 132 Days and 3.2 Hours
In the past 40 years, much effort has been made to develop a vaccine that can prevent human immunodeficiency virus (HIV) infection. But till today, this continues to be an unresolved challenge. The main obstacle in developing a vac
Core Tip: The article primarily focuses on the hurdles in developing a successful vaccine for human immunodeficiency virus-1, lessons learnt from some of the landmark trials, and the targets to be induced by a successful vaccine, and briefly touches on some current advances.
- Citation: Varatharajan S, Krishnasai S, Girish C. Human immunodeficiency virus vaccines: Advances, challenges and future perspectives. World J Virol 2026; 15(1): 116055
- URL: https://www.wjgnet.com/2220-3249/full/v15/i1/116055.htm
- DOI: https://dx.doi.org/10.5501/wjv.v15.i1.116055
Human immunodeficiency virus (HIV)-1 has remained a global health concern over the last four decades. Globally, an estimated 40.8 million people were living with HIV-1 in 2024, according to the United Nations Programme on HIV/acquired immune deficiency syndrome (UNAIDS) 2025 epidemiological estimates. Among these, around 77% were on antiretroviral therapy (ART)[1]. There has been substantial progress in treating HIV-1, and with modern antiretroviral regimens, the life expectancy of people living with HIV (PLHIV) on effective treatment is expected to approach that of the general population[2]. However, ART is a double-edged sword. It maintains viral suppression, increases CD4+ T-cell counts, halts disease progression and improves the quality of life of PLHIV on one hand. On the other hand, it also causes various adverse effects on the gastrointestinal, metabolic, dermatological and cardiovascular systems. These toxicities may compromise treatment adherence and diminish quality of life, particularly in ageing patients on lifelong therapy[3]. Additionally, current regimens do not eradicate infection. Therefore, there is a need for interventions that prevent the acquisition of infection. Accordingly, the development of a safe and effective HIV-1 vaccine remains a major focus of current research. Historically, vaccines have helped eradicate infections and reduce transmission of several infectious diseases. The quest for an HIV-1 vaccine has been going on for the past four decades, with only modest success to date.
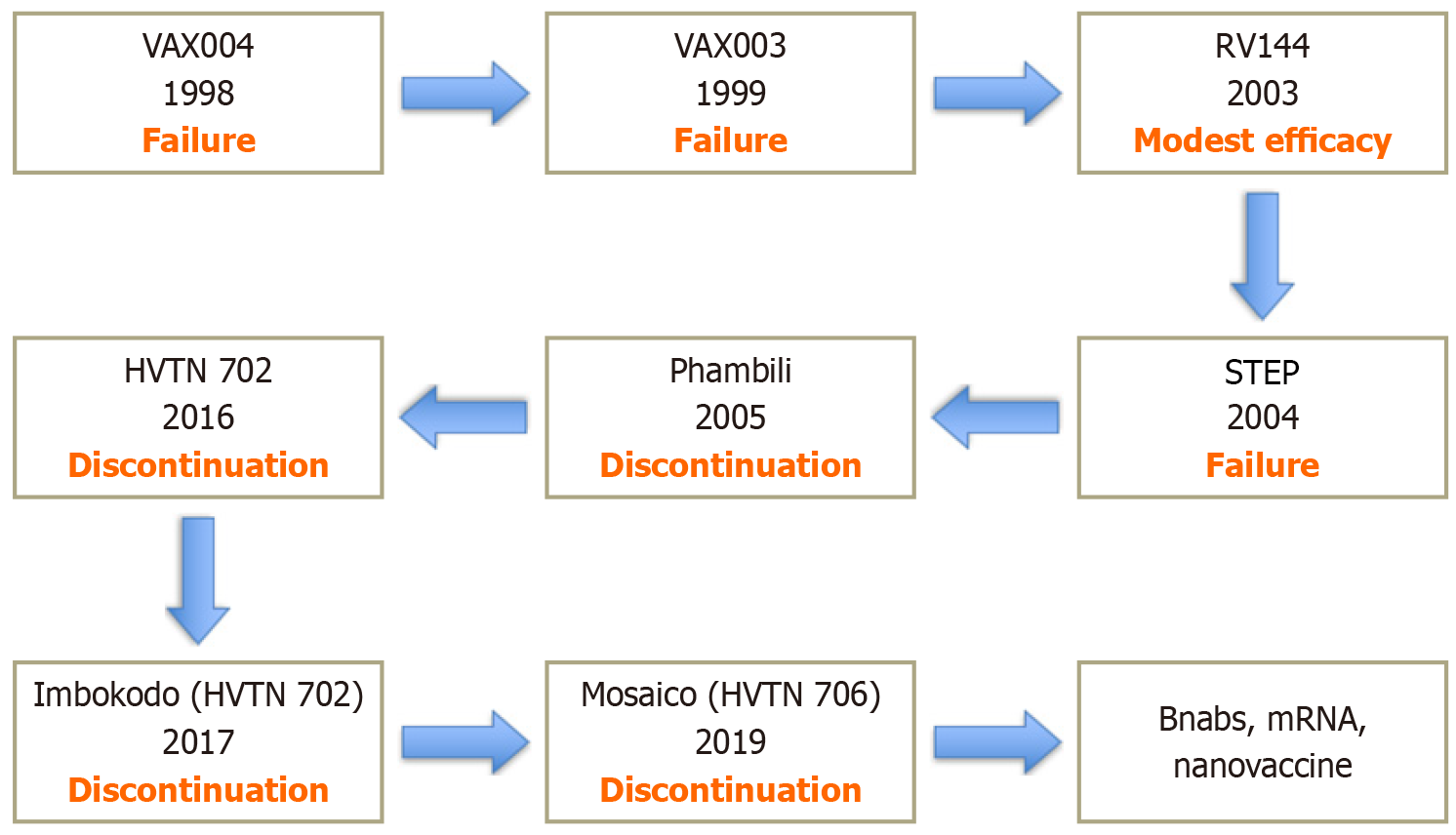
The pursuit of an effective HIV-1 vaccine has been ongoing for the last four decades. The inaugural human HIV-1 phase 1 vaccine trial in 1987 tested the recombinant vaccinia gp160 protein in HIV-negative volunteers and was found capable of inducing HIV-specific antibodies and cytotoxic T-lymphocytes[4]. VAX004 was the first large-scale trial initiated in 1998 to test the AIDSVAX B/B vaccine (two recombinant gp120 envelope proteins from subtype B isolates), but it did not demonstrate a protective effect[5]. HIV-specific CD8+ T-cell responses were higher among HIV-negative volunteers who subsequently acquired infection[6]. Subsequently, the VAX003, a phase 3 trial initiated in 1999, utilized the AIDSVAX B/E vaccine (gp120 Env proteins from clade B and clade E), which demonstrated no significant difference compared to the placebo[7]. In 2003, the Thai and the United States government jointly initiated RV144, a phase 3 trial to evaluate a novel HIV vaccine strategy commonly referred to as prime-boost, where the ALVAC-HIV (a canarypox vector vaccine coding for HIV-1 Env, Gag, and Pol) was the prime and AIDSVAX B/E (a gp120 subunit vaccine) was the boost. To date, this was one of the largest vaccine trials that enrolled 16000 participants showing a modest protection efficacy of 31.2% compared to the placebo group[8]. Overcoming the previous setbacks, the STEP (2004) and Phambili (2007) trials were designed as a phase IIb randomised double-blind trial, targeted at eliciting strong cellular immunity by using a modified adenovirus type 5 vector that delivered HIV-1 genes for the Gag, Pol, and Nef proteins from subtype B. However, both these trials were halted due to a lack of efficacy and an increased risk of infection[9,10]. HVTN 702 (Uhambo, 2016) was a large phase 2b/3 trial that used an RV144-like prime-boost regimen, where ALVAC-HIV (canarypox vector) served as the prime, and a bivalent gp120 protein subunit vaccine with MF59 adjuvant as the boost. This trial was halted in 2020 for non-efficacy reasons after an interim analysis[11]. The trials Imbokodo (2017) and Mosaico (2019) from Janssen tested an Ad26-based mosaic prime-boost regimen combined with soluble clade C gp140 (Imbokodo) or bivalent clade C/mosaic gp140 (Mosaico) Env protein with aluminium phosphate adjuvant. Both were halted due to futility after the interim analysis[12,13] (Figure 1).
The Env (gp120) protein dominates HIV vaccine targets for neutralising antibodies. Developing HIV-1 Env vaccines is exceptionally difficult due to various reasons. The HIV-1 virus exhibits extraordinary genetic variability, allowing it to undergo mutation (3.4 × 10-5 mutations per base in one replication cycle) and recombination, creating a rapidly shifting swarm of variants that makes neutralisation difficult[14]. The Env glycoprotein undergoes mutations due to the error-prone nature of reverse transcriptase[15]. The high conformational flexibility, low density of Env protein (approximately 7-14 spikes per virion), protection of the conserved regions of Env by a layer of glycan shield constituting about 50% of gp120 mass, the ability of Env to maintain its structural integrity while undergoing recurrent mutations, extraordinary genetic diversity of HIV-1 Env protein worldwide- all make Env protein a “difficult to neutralise” target[16-19]. This results in millions of Env variants in a single individual within 24 hours of replication[20]. HIV-1 is classified into main (M), outlier (O), non-M/non-O (N), and pending (P) groups with group M subdivided into nine subtypes/clades denoted by the letters A, B, C, D, F, G, H, J, and K. This results in amino acid variations within subtypes as high as 30% and up to 42% between subtypes[21]. In regions like Africa, around 10%-20% of the PLHIV are infected with two or more strains[22] making virus neutralization difficult. There is no animal model that recapitulates all salient features of HIV-1 infection in humans, limiting translational value[23]. Pharmaceutical companies generally show low investment in HIV-1 vaccine research and development due to high risks, scientific challenges, and uncertain financial returns compared to other pharmaceutical products[24]. Broad neutralising antibodies can neutralise a vast array of HIV-1 strains. These antibodies develop after years of infection due to somatic hypermutation in precursor cells. Producing these antibodies by traditional vaccination methods is difficult. Combinations of 2-3 broadly neutralising antibodies (bnAbs) can neutralise > 95% of strains, with ongoing trials testing them for prevention and cure alongside latency reversers[25-27]. Germline-targeting immunogens, such as eOD-GT8, act as a special bait to capture naive B cells that can eventually produce potent HIV-fighting antibodies called bnAbs. Phase 1 (IAVI G001) trial demonstrated that this approach is practical, with up to 97% of participants experiencing activation of these starter cells following vaccination[28]. Another issue is that immunogens that activate multiple bnAb precursors that target distinct epitopes on the Env protein may suffer competition[29]. The ability of the virus to remain dormant in CD4+ T-lymphocytes for years escaping from being recognized by immune system is another barrier. Virus-infected patients produce antibodies or activated CD4 T-cells which do not eradicate/neutralise the virus, questioning the benchmarks of protection against the virus. CD4 T-cells are required for eliciting a primary antibody response or a cytotoxic T-cell response. But the ability of the virus to downregulate CD4 T-cells makes them less reliable markers[30,31]. Several HIV-1 vaccine trials have been completed, with only one trial, the RV-144 demonstrating modest efficacy[8]. Immune correlates of protection in HIV vaccine trials vary across studies and are not universally specific or consistent, reflecting differences in vaccine designs, populations, and endpoints[32] (Figure 1 and Table 1).
| Ref. | Trial (years) | Vaccine or strategy | Population | Lessons learnt |
| [7] | AIDSVAX B/E (1999-2003) | AIDSVAX B/E (gp120 protein, clade B/E) | 2545 people who inject drugs in Bangkok, Thailand | The gp120 protein alone is insufficient, better immunogen design and/or vectors are needed |
| [5] | VAX004 (1998-2002) | AIDSVAX B/B (gp120 protein, clade B) | About 5400 MSM (men who have sex with men) and high-risk women in N. America/Europe | Protein-only approaches didn’t protect; highlighted the limits of the then available Env designs |
| [9] | STEP (2004-2007) | Modified adenovirus type 5 vector to deliver genes for gag/pol/nef (T-cell vaccine) | About 3000 MSM and high-risk women in the Americas/Australia | Vector pre-immunity and target biology matter. Avoid rAd5 strategies in at-risk subgroups |
| [10] | Phambili/HVTN 503 (2007) | Same rAd5 construct | Adults in South Africa (mostly heterosexual, clade C region) | Stopped early after STEP futility; no efficacy |
| [8] | RV144 (2003-2009) | Prime-boost: ALVAC-HIV (canarypox vector coding for HIV-1 Env, Gag, Pol) + AIDSVAX B/E (gp120 subunit) | 16402 general-population adults in Thailand | Correlates: V1/V2-specific IgG linked to lower risk; high Env-IgA linked to higher risk |
| [11] | HVTN 702 (2016-2020) | RV144-like pox-protein regimen adapted for clade C | Men and women in South Africa | Vaccine components such as adjuvants, dosing schedules, and immune correlates of protection may need to be tailored to different viral subtypes and regional epidemic settings |
| [12] | HVTN 705 (Imbokodo) (2017-2021) | Ad26 mosaic prime + gp140 boost | 2600+ young women in sub-Saharan Africa | No sufficient protection; trial ended. Elicit broadly neutralising activity and/or stronger functional antibody profiles |
| [13] | HVTN 706 (Mosaico) (2019-2023) | Ad26 mosaic prime + gp140 boost (modified) | About 3900 MSM and TG in America/Europe | No efficacy and trial discontinued. Results consistent with Imbokodo. The same vaccine platform cannot protect different populations |
With the lack of efficacy observed in the HVTN 702, HVTN 705, and HVTN 706 studies the focus has now moved towards finding new vaccine strategies that induce broad neutralising antibodies[33]. Several strategies aim to induce bnAbs against HIV-1 by activating rare naive B-cell precursors and guiding their maturation through somatic hy
Env immunogens are developed from longitudinal analyses of bnAbs from infected individuals. In the lineage vaccine strategy, these Env immunogens (priming immunogen) show affinity for a single unmutated common ancestor of the bnAbs’ lineage. Following this, vaccination with Env proteins that are capable of inducing somatic hypermutation is administered. These strategies have been tested in knock-in mice and non-human primates, where Env immunogens that bind to precursor B-cells of either the CD4-binding site or V3-glycan bnAbs lineage were found to be capable of inducing a bnAbs response[45]. HVTN 300 (NCT04864072) is an ongoing phase 1 trial evaluating stabilised CH505 TF chTrimer immunogens (native-like Env trimers) with 3M-052-AF (novel synthetic toll-like receptor 7/8 agonist adjuvant) and alum adjuvant to prime CH103-like bnAb B-cell lineages targeting the CD4-binding site[46].
The germline vaccine strategy involves priming naive B cells using engineered Env immunogens (e.g., eOD-GT8) that bind to diverse, unmutated, common ancestors within a specific bnAbs class (e.g., VRC01-class), followed by boosts with native-like trimers or lineage-specific immunogens[43,45]. Ray et al[47] demonstrated that targeting the membrane-proximal external region (MPER) epitope of the gp41 subunit can drive germinal-centre selection and maturation of B-cell precursors toward bnAbs lineages. Wang et al[48] demonstrated that mRNA-LNP delivery of the soluble, self-assembling nanoparticle immunogen eOD-GT8 successfully activated and evolved VRC01-class precursor B cells toward bnAbs lineages in humanised mice. IAVI G003 (NCT05414786) is a phase 1, open-label trial testing the safety of the germline-targeting eOD-GT8 60mer mRNA vaccine (mRNA-1644, 100 μg dose) administered at weeks 0 and 8 in HIV-uninfected adults. The study evaluates a two-dose vaccine regimen developed to stimulate VRC01-class immune cells in African participants. Participants are screened for eligibility up to 56 days before giving first dose and after receiving two doses they are followed up for 6 months. The main goal of the study is safety evaluation[49]. IAVI G002 (NCT05001373) is a phase 1, randomised, first-in-human, open-label trial evaluating a sequential combo of eOD-GT8 60mer (mRNA-1644, 100 μg) for priming, followed by core-g28v2 60mer (mRNA-1644v2-Core, 100 μg) for boosting to stimulate the VRC01-class and V3-glycan bnAb precursors in 56 HIV-uninfected adults. The study includes four arms: EOD-GT8 alone, eOD-GT8 followed by core-g28v2, core-g28v2 followed by eOD-GT8 and core-g28v2 alone based on the hypothesis that sequential germline-targeting via mRNA guides bnAb B-cell maturation[50].
In this strategy, immunogens that target the neutralising epitope on HIV-1 Env protein are presented, masking the non- neutralising epitopes. Structurally characterised bnab epitopes on HIV-1 include CD4-binding site at the V2 trimer apex, V3-glycan supersite and the MPER[51]. A phase 1 trial, IAVI G001, tested eOD-GT8 nanoparticles targeting the CD4bs epitope, with results showing 97% of recipients activated VRC01-class bnAbs precursors[28]. HVTN 139 (NCT05182125) is an ongoing phase 1 trial evaluating the safety and immunogenicity of chimpanzee-derived adenovirus vectors (AdC6-HIVgp140, AdC7-HIVgp140) expressing clade C gp140, boosted by CH505TF gp120/GLA-SE protein in healthy, HIV-uninfected adults. It aims to determine the safety and tolerability of these vector and protein combinations[52].
The next advancement is the development of SOSIP trimers[53]. SOSIP trimers are engineered HIV-1 proteins that resemble the native Env protein, essential for eliciting bnAbs[54]. Using computer software, the I55P9 point mutation was incorporated into the pre-fusion form of gp41, followed by linking their gp120 and gp49 with a covalent disulfide bond[53]. This resulted in the development of a stable SOSIP timer resembling the Env protein[54]. SOSIP timers obtained from different HIV-1 strains can be combined to increase the breadth and potency of the immune response[55]. Animal studies have shown that this SOSIP trimer can induce broad neutralising antibodies[56]. BG505 SOSIP.664 was one of the first SOSIP trimers used[57]. SOSIP trimers have been developed from multiple strains across different clades[55]. Studies have shown that antibody responses often target exposed glycan-hole regions on these trimers, such as those identified in BG505 SOSIP.664[56]. Rabbits immunised with SOSIP trimer (JR-FL_SOSIP) utilising the DNA prime and protein boost technique produced neutralising antibodies to sensitive strains[58]. ACTHIVE-001 was a phase 1 trial that tested ConM SOSIP.v7 Env trimers with MPLA as adjuvant in 24 HIV-uninfected adults. The vaccine has proved to be safe and immunogenic, inducing strong autologous tier-1/2 neutralizing antibodies targeting V1V2, CD4bs, and base epitopes in all participants after multiple doses. Notably, females showed superior antibody responses compared to males, highlighting sex-based differences in immunogenicity[59]. Currently, SOSIP trimers have been used in combination with various other methods of vaccine delivery, including mRNA and nanoparticle technology, to maximise efficacy[60]. As with advantages, there are some issues with the use of SOSIP trimers. Animal studies have shown that low pH or a DNA component, if present in adjuvants, can result in destabilisation of SOSIP trimers[61]. SOSIP trimers use the non-native tPA (tissue plasminogen activator) signal peptide (SP) coding sequence primarily for superior endoplasmic translocation efficiency. As native SP has a role in Env folding, presentation of Env and overall glycosylation, these factors might be altered in SOSIP trimer[62]. Exposure of the gp41 trimer base in the SOSIP trimer, which is usually masked by the viral membrane, is another limitation resulting in off-target bnAbs directed to these sites[63]. Another debated feature is that the SOS bond itself may introduce non-native, structural changes[61]. Thus, the SOSIP trimer has proved to be a key design in presenting the Env spike in a form that closely resembles the native protein.
This technique uses the virus’s genetic material to produce a protein of interest, which stimulates the virus’s immune system[64]. In one such approach, mRNA is coupled with nanoparticles, which can protect it from degradation by RNases and help with delivery. With the success of the coronavirus disease 2019 vaccines, the development of mRNA vaccines for HIV-1 has generated hope. There are two types of mRNA platforms in the development of HIV-1 vaccines. Conventional non-replicating mRNA that encodes Env, Gag, or immunogenic trimers, such as SOSIP gp140, is preferred for its enhanced stability and resemblance to native folding[65]. The other one is the saRNA, which encodes an RNA-dependent RNA polymerase derived from alphaviruses. This polymerase can recognise and amplify specific elements in the saRNA within the host cells[66].
Two AGS-004 mRNA studies (NCT00672191 and NCT00833781) evaluated dendritic cell-based immunotherapy in which autologous dendritic cells are transfected with RNA encoding HIV antigens (including Gag, Nef, Rev, Vpr) from the patient’s own virus and then returned to the participant, with both studies failing to stimulate an antiviral response. Viral rebound occurred in approximately 4 weeks in both[67,68]. The HIVACATTriMIX mRNA study (NCT02888756) tested iHIVARNA01, which is an mRNA-based therapeutic vaccine combining a conserved T-cell immunogen with TriMix (mRNAs for CD40 L, constitutively active TLR4, and CD70) against TriMix alone and placebo in chronic HIV-1-infected patients on stable ART. The Phase IIa trial was terminated after an interim analysis because it did not demon
The essential advantages of mRNA vaccines include the rapid design flexibility, low cost, safety over DNA and viral vector vaccines, and potent immune activation[64,65]. Lipid nanoparticles deliver mRNA encoding the HIV-1 Env or Gag protein, which act as antigens, triggering an immune response by the body[64,65]. The main challenges include the short-lived nature of antibody responses[65]. The glycan shield of the Env protein helps in evasion of the immune response, making vaccine development difficult[16]. The difficulty in delivering mRNA due to its unstable nature limits the widespread application[64]. Carefully determined immunisation regimens are needed to elicit broad neutralising antibodies[37].
Nanoparticles utilise proteins or lipids in the form of tiny particles to deliver antigens to the immune system. These particles can enhance the immune response by presenting genetic material in the form of natural infection. Nanotechnology enables precise antigen delivery, enhances antigen presentation, and promotes potent immune system activation by mimicking the structural and functional properties of pathogens[78,79]. Nanovaccine platforms offer improved stability, controlled antigen release, and enhanced presentation to antigen-presenting cells[80]. Nanoparticle vaccines can be designed to present conserved regions of the HIV-1 Env protein, which are less prone to mutation[79]. This strategy aims to elicit bnAbs that can neutralise a wide range of HIV-1 strains[79]. Broadly, nanovaccines are divided into non-viral and viral platforms[78,80]. The non-viral systems include inorganic, polymeric, and lipidic nanoparticles[78]. Among inorganic systems, gold nanorods and silica-coated calcium phosphate nanoparticles have demonstrated the ability to stimulate Env-specific antibodies and T-cell responses in experimental HIV-1 immunisation models[81-83]. Polymeric nanoparticles such as poly lactic-co-glycolic acid (PLGA) and poly-methyl methacrylate can be tailored for mucosal targeting, and PLGA-Eudragit microparticles have been shown to elicit strong mucosal IgA and IgG responses following intra-colorectal immunisation[84,85]. Lipid nanoparticles encapsulating Env mRNA have enabled efficient intracellular delivery and rapid antigen expression, activating both humoral and cellular immune arms[78,79]. Self-assembling protein nanoparticles, such as ferritin, E2p, and I53-50 scaffolds, allow for the controlled multivalent display of HIV-1 Env trimers, like BG505 SOSIP, thereby improving immunogenicity[78,79]. Ferritin nanoparticles displaying eight Env trimers per particle induced higher binding-antibody titres and improved germinal-centre B-cell responses compared with soluble Env trimers[86]. These advances highlight nanotechnology’s expanding role in next-generation HIV-1 vaccine design. In a preclinical study, a self-assembling protein nanoparticle (1c-SApNP) conjugated to BG505 uncleaved prefusion-optimised (UFO) was used to induce bnAbs in mice and rabbits. These 1cSApNPs presenting the nativelike BG505 UFO trimers induced improved antibody responses in mice and rabbits compared with soluble trimers[87]. An eOD-GT8 60-mer nanoparticle was designed, which was capable of priming VRC01-class HIV-1-specific B cells for bNAbs production in mice[88]. Recently the same group revealed that eOD-GT5 60-mer induced a CD4 T-cell response in mice, thereby emphasizing the highly immunogenic nature of nanoparticle vaccine[89]. Another important advantage is that these nanoparticles can be combined with adjuvants and such adjuvants can increase the production of antibodies. In a preclinical study, ConM SOSIP trimers were evaluated as free proteins and presented on ferritin nanoparticles with different adjuvants (squalene emulsion, ISCOMATRIX, GLALSQ, and MPLA liposomes) in rabbits. Compared with soluble trimers, the trimers presented on the ferritin nanoparticles enhanced neutralising antibody responses, with the strength of this effect depending on the adjuvant, with ISCOMATRIX and squalene emulsion generally providing stronger responses than GLA-LSQ[90]. In NHPs the use of an imidazoquinoline adjuvant 3M-052 when paired with PLGA nanoparticle-based vaccines induced sustained tier 1 nAb production[91].
The CRISPR/Cas9 technique, in the context of HIV-1, does not refer to vaccines but rather to curative gene-therapy approaches designed to eliminate or disable the virus in people already suppressed on ART[92]. Rather than preventing infection, these methods aim to cut out the integrated HIV DNA (the provirus) or disrupt essential host or viral factors[92]. As of 2025, there are no CRISPR-based HIV vaccines in clinical trials. Preclinical studies have shown strong potential. Using multiple guide RNAs targeting conserved HIV regions, CRISPR/Cas9 can excise the entire proviral genome in cell cultures and suppress viral replication in humanised mouse models[93]. Clinical results have been far more limited. The first human trial, EBT-101 (NCT05144386), used an AAV9-delivered CRISPR therapy in ART-suppressed individuals. The treatment was generally safe and well tolerated, but in participants who stopped ART, HIV rebounded within weeks, indicating that only a small fraction of the viral reservoir was edited[94]. No phase 2 trial has yet been announced.
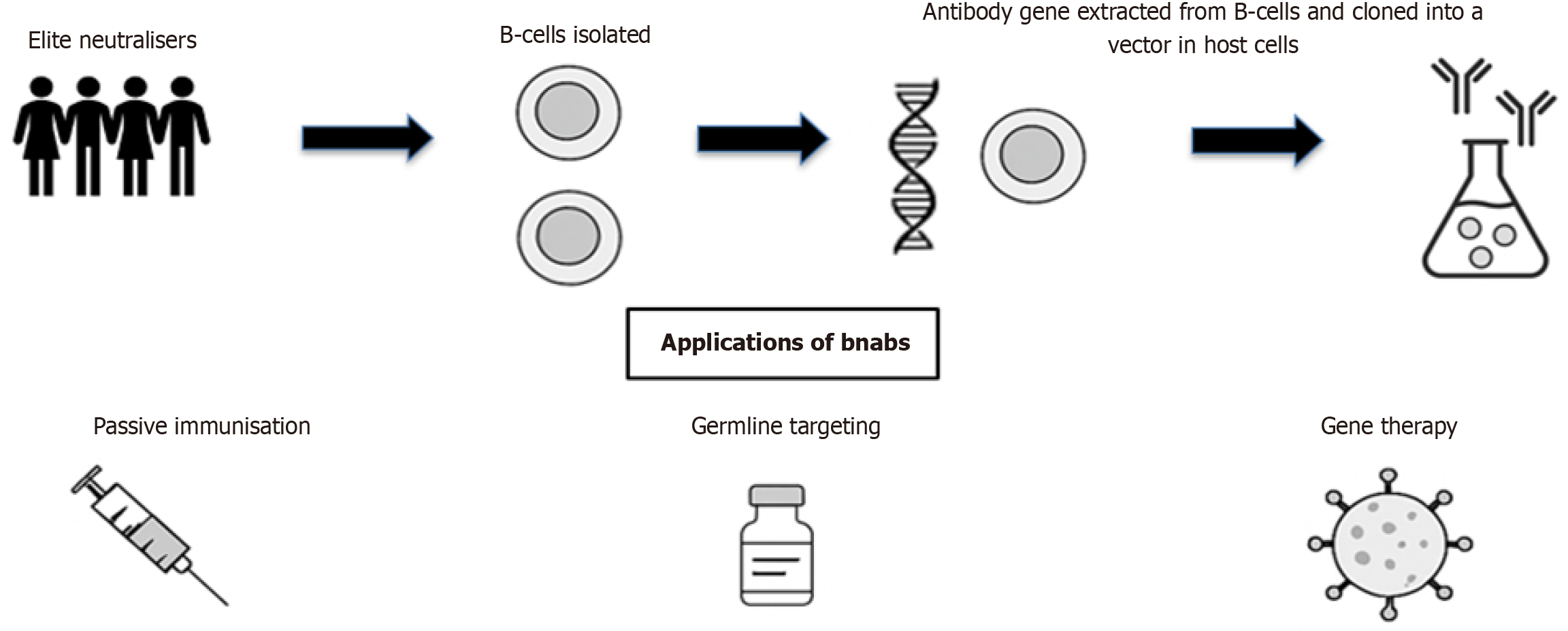
In the past years the therapeutic potential of bnAbs has been demonstrated in humanized mice, non-human primates, and humans. Initially, the first generation bnAbs such as 2G12, 2F5, and 4E10 reduced viral load in HIV infected patients only to be deferred by the development of escape mutants and emergence of resistance[95-97]. After ART interruption in PLHIV, administration of bnAbs delayed viral rebound, with observed delays ranging from approximately 3 to 24 weeks across different studies, depending on the antibody used and participant characteristics[98,99]. However, the undesirable features, including autoreactivity exhibited by 2F5 and 4E10 bnAbs, whilst 2G12 demonstrates low potency with resistance, limiting their usage[100,101]. Even though we have moved to second-generation bnAbs after the development of single-cell antibody cloning techniques in 2009[102], the first generation remains the foundation for future therapies. The second generation bnAbs with improved breadth and potency target highly conserved regions as discussed before including the CD4 binding site (CD4bs), the V1V2 glycan region, the V3-glycan region. Others include the gp120-gp41 interface, the gp120 silent face, the gp41 MPER, and the gp41 fusion domain[103-106]. Early phase 1 trials in PLHIV with bnAbs targeted either the CD4-binding site (3BNC117 and VRC01) or the V3-glycan region (10-1074 and PGT121). In bnAbs-sensitive virus-infected PLHIV, a single infusion of 3BNC117, VRC01, PGT121, or 10-1074 produced a considerable reduction in viral copies from baseline, and viremia remained suppressed up to 28 days, only to be followed by viral rebound due to escape mutants[107-111]. In patients with ART interruption with reduced viral levels, the efficacy of bnAbs is enhanced. After ART discontinuation, VRC01 or 3BNC117 monotherapy demonstrated effective viremia control and delayed viral rebound for 4 and 10 weeks. Rebound viruses arose from a single clone either due to waning immunity or due to the emergence of resistance. About 30% of 3BNC117 participants remained virally suppressed. 3BNC117 exerts significant selective pressure on emerging viruses during ATI in humans[112-115]. Talking about combination bnAbs therapy, several studies showed that a combination of bnAbs achieved durable viral suppression in animal models. After ART interruption, the combination of 3BNC117 and 10-1074 delayed viral rebound to a median of about 21 weeks in participants whose virus was sensitive to both antibodies in HIV-positive individuals. In untreated viremic patients without pre-existing bnAbs resistance, the same combination suppressed viral replication for up to approximately 3 months[116,117]. Provided adequate concentration is maintained, a combination of 3BNC117 and 10-1074 offers some degree of viral suppression in patients with bNAb-sensitive viruses. To conclude, optimal bNAb combinations and concentrations are necessary to achieve durable viral suppression in HIV-infected individuals.
Artificial intelligence and computational tools accelerate HIV vaccine development by predicting epitopes, designing immunogens, analysing trial data, and optimising trials for speed and equity.
Epitope prediction: Ragon institute/Massachusetts institute of technology (MUNIS, 2025) applies deep learning to over 650000 HLA-bound peptides to predict CD8+ T cell epitopes accurately thereby spotting new HIV and Epstein-Barr virus targets to support T cell vaccines. It outperforms predictors such as NetMHCpan-4.1 in identifying likely presented and immunogenic epitopes and has been validated on influenza, HIV, and Epstein-Barr virus datasets, identifying both known and novel epitopes to support Tcell vaccine design efforts[118]. NetMHCpan-4.1, along with transformer models, predicts peptide-MHC matches for over 13000 alleles, including glycan effects on stable Env trimer regions[119].
Immunogen design: Protein language models process HIV sequences and mutations to create bnAbs mimics, thereby predicting the binding strength and further improving designs obtained from elite controllers, using genomic and population data for local variants[120]. Artificial intelligence algorithms are trained to rapidly select Env mutants capable of boosting bnAb lineages to heterologous breadth and potency. The path to a successful vaccine lies in learning to engineer the immune system to stimulate and mature rare neutralising antibodies that infrequently occur in PLHIV. Formulation of a multivalent immunogen is the next step to be used in a trial. Currently, work is going on to design Env immunogens with many bnAb triggering sites on the same immunogen[118-121].
Phase 1 HIV-1 trials mainly assess safety and immune priming and as a result face fewer ethical concerns. Moving for
After the discontinuation of Imbokodo and Mosaico studies due to lack of efficacy[12,13], there has been a shift in the landscape of HIV vaccine trials. Studies like IAVI G001 (NCT03547245) are the landmark proof-of-concept studies for germline targeting immunization. It demonstrated that bnAb precursor activation in humans is feasible[127]. The IAVI G003 (NCT05414786) study tests the safety/immunogenicity of two doses (100 μg) of eOD-GT8 60mer mRNA (mRNA-1644) to induce VRC01-class B-cells[49]. The results might instil confidence in mRNA-only germline targeting techniques. The HVTN 302 (NCT05217641) study evaluated the safety and immunogenicity of mRNA-encoded envelope trimers. Membrane-anchored trimers elicited tier 2 neutralizing antibodies in 80% of vaccinees compared to the mere 4% vaccinees who received soluble trimer vaccine[72]. Going further, the HVTN 300 (NCT04864072) evaluates whether bnAbs of a predictable B-cell lineage can be safely induced using carefully chosen immunogens[128]. Further studies like ACTHIVE-001 demonstrated that females exhibited higher Env-specific antibody responses than males. It highlighted the importance of considering sex-based difference in HIV vaccine immunogenicity[59]. The HVTN 137A (NCT04177355) demonstrated the first clinical evaluation of a good manufacturing practice produced BG505 SOSIP.664 soluble native-like trimer along with a novel toll-like-receptor 7/8 signalling adjuvant. It induced autologous tier-2 nAbs in 5/17 (about 30%) participants. The regimen used here appeared safe[129]. The HVTN-139 (NCT05182125) study is unique in that it pairs a chimpanzee adenoviral vector as a prime with a CH505 transmitted/founder gp120 protein[130]. In another phase 1, first-in-human trial, the HIV-1 prefusion-stabilized Env candidate trimer 4571 proved to be safe, well-tolerated, and immunogenic in healthy adults, inducing specific antibody responses. When administered with alum, it promises to be a stable approach for eliciting targeted immune responses[131]. Another novel study IAVI C114 (NCT06617091) uses a gorilla adenovirus vector to induce T-cell responses against highly networked, mutationally constrained HIV epitopes to limit viral escape[132] (Table 2).
| Ref. | Clinical trial | Vaccine concept | Platform | Vaccine description | Status |
| [127] | NCT03547245 (IAVIG001) | Germline targeting (CD4bs) | Protein nanoparticle + adjuvant (Phase 1) | eOD-GT8 60mer nanoparticle + AS01B (liposome-based adjuvant containing MPLA + QS-21) | Completed |
| [50] | NCT05001373 (IAVI G002) | Sequential germline targeting | mRNA-LNP (Phase 1) | eOD-GT8 60mer mRNA + core-g28 version 2 60mer mRNA nanoparticle | Active, not recruiting |
| [49] | NCT05414786 (IAVI G003) | Germline targeting (CD4bs) | mRNA-LNP (Phase 1) | eOD-GT8 60mer mRNA alone | Recruiting |
| [71] | HVTN 302 NCT05217641 | Stabilized BG505 MD39.3 Env trimers | mRNA-LNP (Phase 1) | BG505 MD39.3, BG505 MD39.3 gp151, and BG505 MD39.3 gp151 CD4KO mRNA (3 groups) | Active, not recruiting |
| [46] | NCT04864072 (HVTN 300) | B-cell lineage (CD4bs) | Protein + adjuvant (Phase 1) | CH505 transmitted/founder chimeric trimer + 3M-052 aqueous formulation with aluminum hydroxide | Active, not recruiting |
| [59] | NCT03961438 (ACTHIVE-001) | Native-like trimer | Protein + adjuvant (Phase 1) | Consensus M SOSIP version 7 gp140 + MPLA liposomes(as adjuvant) | Completed |
| [129] | HVTN 137A (NCT04177355) | Native-like trimer | Protein + adjuvant (Phase 1) | BG505 SOSIP.664 + 3M-052 aqueous formulation with aluminium hydroxide | Completed |
| [52] | NCT05182125 (HVTN 139) | B-cell lineage priming | Adenoviral vector + protein (Phase 1) | Adenovirus chimpanzee serotype 6/7 expressing HIV gp140 envelope glycoprotein+ CH505 transmitted/founder gp120 glycoprotein adjuvanted with glucopyranosyl lipid A stable emulsion | Recruiting |
| [131] | NCT03783130 (Trimer 4571) | Prefusion-stabilized trimer | Protein + adjuvant (Phase 1) | BG505 DS-SOSIP.664 (Trimer 4571) + alum | Completed |
| [132] | IAVI C114 (NCT06617091) | T -cell-inducing preventive/therapeutic HIV vaccine | Non-replicating Gorilla Adenovirus (Phase 1) | Gorilla adenovirus vectored HIV networked epitopes 1 encode highly networked, mutationally constrained HIV epitopes from stable structural regions (less prone to mutate) | Not yet recruiting |
HIV-1 vaccine development has faced decades of setbacks due to the virus’s extreme genetic variability and extensive glycan shielding. Developments in germline-targeting immunogens such as the eOD-GT8, which activates up to 97% of VRC01-class naïve B-cell precursors together with stabilised SOSIP Env trimers and mRNA delivery platforms (for example, HVTN 302), support a stepwise vaccination strategy. AI-based tools like MUNIS and NetMHCpan can improve the prediction of CD8 T-cell targets. Computer-guided refinement of bnAb lineages from elite controllers may help optimise early-phase trial design. In this PrEP era, the progression to phase 3 efficacy trials is also accelerated, possibly by testing vaccines in PLHIV. Overall, artificial intelligence-guided vaccines which target multiple neutralisable regions of HIV such as the CD4 binding site, V3-glycan region, and MPER, including bispecific designs maintained with booster doses, hold promise for the future. This should be followed by sustained long-term engagement from industry supported by robust public-private partnerships.
| 1. | United Nations Programme on HIV/AIDS. Global HIV and AIDS statistics-Fact sheet. [cited 21 January 2026]. Available from: https://www.unaids.org/en/resources/fact-sheet. |
| 2. | Wandeler G, Johnson LF, Egger M. Trends in life expectancy of HIV-positive adults on antiretroviral therapy across the globe: comparisons with general population. Curr Opin HIV AIDS. 2016;11:492-500. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 361] [Cited by in RCA: 321] [Article Influence: 32.1] [Reference Citation Analysis (0)] |
| 3. | Oguntibeju OO. Quality of life of people living with HIV and AIDS and antiretroviral therapy. HIV AIDS (Auckl). 2012;4:117-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 109] [Article Influence: 7.8] [Reference Citation Analysis (1)] |
| 4. | Redfield RR, Birx DL, Ketter N, Tramont E, Polonis V, Davis C, Brundage JF, Smith G, Johnson S, Fowler A. A phase I evaluation of the safety and immunogenicity of vaccination with recombinant gp160 in patients with early human immunodeficiency virus infection. Military Medical Consortium for Applied Retroviral Research. N Engl J Med. 1991;324:1677-1684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 174] [Cited by in RCA: 169] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 5. | Flynn NM, Forthal DN, Harro CD, Judson FN, Mayer KH, Para MF; rgp120 HIV Vaccine Study Group. Placebo-controlled phase 3 trial of a recombinant glycoprotein 120 vaccine to prevent HIV-1 infection. J Infect Dis. 2005;191:654-665. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 740] [Cited by in RCA: 709] [Article Influence: 33.8] [Reference Citation Analysis (1)] |
| 6. | Jones NG, DeCamp A, Gilbert P, Peterson ML, Gurwith M, Cao H. AIDSVAX immunization induces HIV-specific CD8+ T-cell responses in high-risk, HIV-negative volunteers who subsequently acquire HIV infection. Vaccine. 2009;27:1136-1140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 21] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 7. | Pitisuttithum P, Gilbert P, Gurwith M, Heyward W, Martin M, van Griensven F, Hu D, Tappero JW, Choopanya K; Bangkok Vaccine Evaluation Group. Randomized, double-blind, placebo-controlled efficacy trial of a bivalent recombinant glycoprotein 120 HIV-1 vaccine among injection drug users in Bangkok, Thailand. J Infect Dis. 2006;194:1661-1671. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 663] [Cited by in RCA: 632] [Article Influence: 31.6] [Reference Citation Analysis (1)] |
| 8. | Karasavvas N, Billings E, Rao M, Williams C, Zolla-Pazner S, Bailer RT, Koup RA, Madnote S, Arworn D, Shen X, Tomaras GD, Currier JR, Jiang M, Magaret C, Andrews C, Gottardo R, Gilbert P, Cardozo TJ, Rerks-Ngarm S, Nitayaphan S, Pitisuttithum P, Kaewkungwal J, Paris R, Greene K, Gao H, Gurunathan S, Tartaglia J, Sinangil F, Korber BT, Montefiori DC, Mascola JR, Robb ML, Haynes BF, Ngauy V, Michael NL, Kim JH, de Souza MS; MOPH TAVEG Collaboration. The Thai Phase III HIV Type 1 Vaccine trial (RV144) regimen induces antibodies that target conserved regions within the V2 loop of gp120. AIDS Res Hum Retroviruses. 2012;28:1444-1457. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 192] [Cited by in RCA: 176] [Article Influence: 12.6] [Reference Citation Analysis (1)] |
| 9. | Buchbinder SP, Mehrotra DV, Duerr A, Fitzgerald DW, Mogg R, Li D, Gilbert PB, Lama JR, Marmor M, Del Rio C, McElrath MJ, Casimiro DR, Gottesdiener KM, Chodakewitz JA, Corey L, Robertson MN; Step Study Protocol Team. Efficacy assessment of a cell-mediated immunity HIV-1 vaccine (the Step Study): a double-blind, randomised, placebo-controlled, test-of-concept trial. Lancet. 2008;372:1881-1893. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1414] [Cited by in RCA: 1352] [Article Influence: 75.1] [Reference Citation Analysis (1)] |
| 10. | Gray GE, Allen M, Moodie Z, Churchyard G, Bekker LG, Nchabeleng M, Mlisana K, Metch B, de Bruyn G, Latka MH, Roux S, Mathebula M, Naicker N, Ducar C, Carter DK, Puren A, Eaton N, McElrath MJ, Robertson M, Corey L, Kublin JG; HVTN 503/Phambili study team. Safety and efficacy of the HVTN 503/Phambili study of a clade-B-based HIV-1 vaccine in South Africa: a double-blind, randomised, placebo-controlled test-of-concept phase 2b study. Lancet Infect Dis. 2011;11:507-515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 324] [Cited by in RCA: 301] [Article Influence: 20.1] [Reference Citation Analysis (0)] |
| 11. | Moodie Z, Dintwe O, Sawant S, Grove D, Huang Y, Janes H, Heptinstall J, Omar FL, Cohen K, De Rosa SC, Zhang L, Yates NL, Sarzotti-Kelsoe M, Seaton KE, Laher F, Bekker LG, Malahleha M, Innes C, Kassim S, Naicker N, Govender V, Sebe M, Singh N, Kotze P, Lazarus E, Nchabeleng M, Ward AM, Brumskine W, Dubula T, Randhawa AK, Grunenberg N, Hural J, Kee JJ, Benkeser D, Jin Y, Carpp LN, Allen M, D'Souza P, Tartaglia J, DiazGranados CA, Koutsoukos M, Gilbert PB, Kublin JG, Corey L, Andersen-Nissen E, Gray GE, Tomaras GD, McElrath MJ. Analysis of the HIV Vaccine Trials Network 702 Phase 2b-3 HIV-1 Vaccine Trial in South Africa Assessing RV144 Antibody and T-Cell Correlates of HIV-1 Acquisition Risk. J Infect Dis. 2022;226:246-257. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 41] [Article Influence: 10.3] [Reference Citation Analysis (1)] |
| 12. | Gray GE, Mngadi K, Lavreys L, Nijs S, Gilbert PB, Hural J, Hyrien O, Juraska M, Luedtke A, Mann P, McElrath MJ, Odhiambo JA, Stieh DJ, van Duijn J, Takalani AN, Willems W, Tapley A, Tomaras GD, Van Hoof J, Schuitemaker H, Swann E, Barouch DH, Kublin JG, Corey L, Pau MG, Buchbinder S, Tomaka F; Imbokodo/HVTN 705/HPX2008 Study Group. Mosaic HIV-1 vaccine regimen in southern African women (Imbokodo/HVTN 705/HPX2008): a randomised, double-blind, placebo-controlled, phase 2b trial. Lancet Infect Dis. 2024;24:1201-1212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 32] [Article Influence: 16.0] [Reference Citation Analysis (1)] |
| 13. | Buchbinder SP, Spinosa Guzman S, Sanchez J, Willems W, Stieh DJ, van Duijn J, van Rosmalen MGM, Hendriks J, Nijs S, Lavreys L, Paez CA, Grinzstejn B, Hutter J, Mann P, Sierra Madero JG, Cahn P, Castagna A, Truyers C, Roels S, Gilbert PB, Carone M, Luedtke A, Corey L, Pau MG, Tomaka F; HVTN 706/HPX3002/Mosaico Study Team. Efficacy and safety of a mosaic HIV-1 vaccine regimen in men who have sex with men and transgender individuals (HVTN 706/HPX3002/Mosaico): a global, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet HIV. 2025;12:e823-e835. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 14. | Mansky LM, Temin HM. Lower in vivo mutation rate of human immunodeficiency virus type 1 than that predicted from the fidelity of purified reverse transcriptase. J Virol. 1995;69:5087-5094. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 866] [Cited by in RCA: 784] [Article Influence: 25.3] [Reference Citation Analysis (1)] |
| 15. | Van Duyne R, Kuo LS, Pham P, Fujii K, Freed EO. Mutations in the HIV-1 envelope glycoprotein can broadly rescue blocks at multiple steps in the virus replication cycle. Proc Natl Acad Sci U S A. 2019;116:9040-9049. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 44] [Article Influence: 6.3] [Reference Citation Analysis (1)] |
| 16. | Crispin M, Ward AB, Wilson IA. Structure and Immune Recognition of the HIV Glycan Shield. Annu Rev Biophys. 2018;47:499-523. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 147] [Cited by in RCA: 124] [Article Influence: 15.5] [Reference Citation Analysis (1)] |
| 17. | Zhu P, Liu J, Bess J Jr, Chertova E, Lifson JD, Grisé H, Ofek GA, Taylor KA, Roux KH. Distribution and three-dimensional structure of AIDS virus envelope spikes. Nature. 2006;441:847-852. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 675] [Cited by in RCA: 611] [Article Influence: 30.6] [Reference Citation Analysis (1)] |
| 18. | Alexiev I, Dimitrova R. The Origins and Genetic Diversity of HIV-1: Evolutionary Insights and Global Health Perspectives. Int J Mol Sci. 2025;26:10909. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 19. | Wang Q, Finzi A, Sodroski J. The Conformational States of the HIV-1 Envelope Glycoproteins. Trends Microbiol. 2020;28:655-667. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 94] [Cited by in RCA: 80] [Article Influence: 13.3] [Reference Citation Analysis (1)] |
| 20. | Perelson AS, Neumann AU, Markowitz M, Leonard JM, Ho DD. HIV-1 dynamics in vivo: virion clearance rate, infected cell life-span, and viral generation time. Science. 1996;271:1582-1586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2706] [Cited by in RCA: 2056] [Article Influence: 68.5] [Reference Citation Analysis (1)] |
| 21. | Liu Y, Jia L, Su B, Li H, Li Z, Han J, Zhang Y, Zhang T, Li T, Wu H, Li J, Li L. The Genetic Diversity of HIV-1 Quasispecies Within Primary Infected Individuals. AIDS Res Hum Retroviruses. 2020;36:440-449. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 22. | Hemelaar J. The origin and diversity of the HIV-1 pandemic. Trends Mol Med. 2012;18:182-192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 346] [Cited by in RCA: 300] [Article Influence: 21.4] [Reference Citation Analysis (1)] |
| 23. | Hatziioannou T, Evans DT. Animal models for HIV/AIDS research. Nat Rev Microbiol. 2012;10:852-867. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 298] [Cited by in RCA: 263] [Article Influence: 18.8] [Reference Citation Analysis (1)] |
| 24. | Tatoud R, Brander C, Hwang C, Kennelly J, Lu S, O'Neil K, Safrit JT, Benhayoun I, Firmat J, Barriere N. Biotech's role in advancing HIV vaccine development. Emerg Microbes Infect. 2024;13:2384460. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 25. | Klein F, Diskin R, Scheid JF, Gaebler C, Mouquet H, Georgiev IS, Pancera M, Zhou T, Incesu RB, Fu BZ, Gnanapragasam PN, Oliveira TY, Seaman MS, Kwong PD, Bjorkman PJ, Nussenzweig MC. Somatic mutations of the immunoglobulin framework are generally required for broad and potent HIV-1 neutralization. Cell. 2013;153:126-138. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 538] [Cited by in RCA: 460] [Article Influence: 35.4] [Reference Citation Analysis (1)] |
| 26. | Hsu DC, Mellors JW, Vasan S. Can Broadly Neutralizing HIV-1 Antibodies Help Achieve an ART-Free Remission? Front Immunol. 2021;12:710044. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 27. | Wagh K, Bhattacharya T, Williamson C, Robles A, Bayne M, Garrity J, Rist M, Rademeyer C, Yoon H, Lapedes A, Gao H, Greene K, Louder MK, Kong R, Karim SA, Burton DR, Barouch DH, Nussenzweig MC, Mascola JR, Morris L, Montefiori DC, Korber B, Seaman MS. Optimal Combinations of Broadly Neutralizing Antibodies for Prevention and Treatment of HIV-1 Clade C Infection. PLoS Pathog. 2016;12:e1005520. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 190] [Cited by in RCA: 174] [Article Influence: 17.4] [Reference Citation Analysis (1)] |
| 28. | Leggat DJ, Cohen KW, Willis JR, Fulp WJ, deCamp AC, Kalyuzhniy O, Cottrell CA, Menis S, Finak G, Ballweber-Fleming L, Srikanth A, Plyler JR, Schiffner T, Liguori A, Rahaman F, Lombardo A, Philiponis V, Whaley RE, Seese A, Brand J, Ruppel AM, Hoyland W, Yates NL, Williams LD, Greene K, Gao H, Mahoney CR, Corcoran MM, Cagigi A, Taylor A, Brown DM, Ambrozak DR, Sincomb T, Hu X, Tingle R, Georgeson E, Eskandarzadeh S, Alavi N, Lu D, Mullen TM, Kubitz M, Groschel B, Maenza J, Kolokythas O, Khati N, Bethony J, Crotty S, Roederer M, Karlsson Hedestam GB, Tomaras GD, Montefiori D, Diemert D, Koup RA, Laufer DS, McElrath MJ, McDermott AB, Schief WR. Vaccination induces HIV broadly neutralizing antibody precursors in humans. Science. 2022;378:eadd6502. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 255] [Cited by in RCA: 214] [Article Influence: 53.5] [Reference Citation Analysis (1)] |
| 29. | Xie Z, Wang X, Yan Y, Steichen JM, Ma KM, Cottrell CA, Melzi E, Bottermann M, Villavicencio PM, Rantalainen K, Schiffner T, Warner JE, Weldon SR, Prum T, Ellis-Pugh JR, Torres JL, Jackson AM, Flynn CT, Ozorowski G, Himansu S, Carfi A, Ward AB, Nair U, Schief WR, Batista FD. Simultaneous priming of HIV broadly neutralizing antibody precursors to multiple epitopes by germline-targeting mRNA-LNP immunogens in mouse models. Sci Immunol. 2025;10:eadu7961. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 30. | Marsden MD, Zack JA. Double trouble: HIV latency and CTL escape. Cell Host Microbe. 2015;17:141-142. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 31. | Burgers WA, Manrique A, Masopust D, McKinnon LR, Reynolds MR, Rolland M, Blish C, Chege GK, Curran R, Fischer W, Herrera C, Sather DN. Measurements of immune responses for establishing correlates of vaccine protection against HIV. AIDS Res Hum Retroviruses. 2012;28:641-648. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 32. | Tomaras GD, Plotkin SA. Complex immune correlates of protection in HIV-1 vaccine efficacy trials. Immunol Rev. 2017;275:245-261. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 104] [Cited by in RCA: 94] [Article Influence: 10.4] [Reference Citation Analysis (1)] |
| 33. | Nkolola JP, Barouch DH. Prophylactic HIV-1 vaccine trials: past, present, and future. Lancet HIV. 2024;11:e117-e124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 38] [Article Influence: 19.0] [Reference Citation Analysis (1)] |
| 34. | Atabey T, Sanders RW, Aldon Y. Germline-targeting Strategies to Induce bNAbs against HIV-1. Curr HIV Res. 2025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 35. | Doores KJ. The HIV glycan shield as a target for broadly neutralizing antibodies. FEBS J. 2015;282:4679-4691. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 121] [Cited by in RCA: 105] [Article Influence: 9.5] [Reference Citation Analysis (1)] |
| 36. | Mascola JR, Haynes BF. HIV-1 neutralizing antibodies: understanding nature's pathways. Immunol Rev. 2013;254:225-244. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 447] [Cited by in RCA: 399] [Article Influence: 30.7] [Reference Citation Analysis (1)] |
| 37. | Haynes BF, Wiehe K, Borrow P, Saunders KO, Korber B, Wagh K, McMichael AJ, Kelsoe G, Hahn BH, Alt F, Shaw GM. Strategies for HIV-1 vaccines that induce broadly neutralizing antibodies. Nat Rev Immunol. 2023;23:142-158. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 318] [Cited by in RCA: 277] [Article Influence: 92.3] [Reference Citation Analysis (1)] |
| 38. | Zhou P, Wang H, Fang M, Li Y, Wang H, Shi S, Li Z, Wu J, Han X, Shi X, Shang H, Zhou T, Zhang L. Broadly resistant HIV-1 against CD4-binding site neutralizing antibodies. PLoS Pathog. 2019;15:e1007819. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 24] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 39. | Steinhardt JJ, Guenaga J, Turner HL, McKee K, Louder MK, O'Dell S, Chiang CI, Lei L, Galkin A, Andrianov AK, A Doria-Rose N, Bailer RT, Ward AB, Mascola JR, Li Y. Rational design of a trispecific antibody targeting the HIV-1 Env with elevated anti-viral activity. Nat Commun. 2018;9:877. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 66] [Article Influence: 8.3] [Reference Citation Analysis (1)] |
| 40. | Xu L, Pegu A, Rao E, Doria-Rose N, Beninga J, McKee K, Lord DM, Wei RR, Deng G, Louder M, Schmidt SD, Mankoff Z, Wu L, Asokan M, Beil C, Lange C, Leuschner WD, Kruip J, Sendak R, Kwon YD, Zhou T, Chen X, Bailer RT, Wang K, Choe M, Tartaglia LJ, Barouch DH, O'Dell S, Todd JP, Burton DR, Roederer M, Connors M, Koup RA, Kwong PD, Yang ZY, Mascola JR, Nabel GJ. Trispecific broadly neutralizing HIV antibodies mediate potent SHIV protection in macaques. Science. 2017;358:85-90. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 245] [Cited by in RCA: 237] [Article Influence: 26.3] [Reference Citation Analysis (1)] |
| 41. | Fidler S. A Randomised Placebo Controlled Trial of ART Plus Dual Long-acting HIV-specific Broadly Neutralising Antibodies (bNAbs). (RIO) [cited 21 January 2026]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/study/NCT04319367 ClinicalTrials.gov Identifier: NCT04319367. |
| 42. | Gilead Sciences. Gilead Presents New HIV Research Data at EACS 2025 – Driving Scientific Innovation in Treatment and Prevention. [cited 21 January 2026]. Available from: https://www.gilead.com/news/news-details/2025/gilead-presents-new-hiv-research-data-at-eacs-2025--driving-scientific-innovation-in-treatment-and-prevention. |
| 43. | Haynes BF, Kelsoe G, Harrison SC, Kepler TB. B-cell-lineage immunogen design in vaccine development with HIV-1 as a case study. Nat Biotechnol. 2012;30:423-433. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 465] [Cited by in RCA: 409] [Article Influence: 29.2] [Reference Citation Analysis (1)] |
| 44. | Davenport TM, Gorman J, Joyce MG, Zhou T, Soto C, Guttman M, Moquin S, Yang Y, Zhang B, Doria-Rose NA, Hu SL, Mascola JR, Kwong PD, Lee KK. Somatic Hypermutation-Induced Changes in the Structure and Dynamics of HIV-1 Broadly Neutralizing Antibodies. Structure. 2016;24:1346-1357. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 34] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 45. | Martin TM, Robinson ST, Huang Y. Discovery medicine - the HVTN's iterative approach to developing an HIV-1 broadly neutralizing vaccine. Curr Opin HIV AIDS. 2023;18:290-299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 46. | HVTN 300: A first-in-human phase 1 clinical trial to evaluate the safety and immunogenicity of stabilized CH505 TF chTrimer in healthy, HIV-uninfected adult participants. [cited 21 January 2026]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/study/NCT04864072 ClinicalTrials.gov Identifier: NCT04864072. |
| 47. | Ray R, Schiffner T, Wang X, Yan Y, Rantalainen K, Lee CD, Parikh S, Reyes RA, Dale GA, Lin YC, Pecetta S, Giguere S, Swanson O, Kratochvil S, Melzi E, Phung I, Madungwe L, Kalyuzhniy O, Warner J, Weldon SR, Tingle R, Lamperti E, Kirsch KH, Phelps N, Georgeson E, Adachi Y, Kubitz M, Nair U, Crotty S, Wilson IA, Schief WR, Batista FD. Affinity gaps among B cells in germinal centers drive the selection of MPER precursors. Nat Immunol. 2024;25:1083-1096. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 26] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 48. | Wang X, Cottrell CA, Hu X, Ray R, Bottermann M, Villavicencio PM, Yan Y, Xie Z, Warner JE, Ellis-Pugh JR, Kalyuzhniy O, Liguori A, Willis JR, Menis S, Rämisch S, Eskandarzadeh S, Kubitz M, Tingle R, Phelps N, Groschel B, Himansu S, Carfi A, Kirsch KH, Weldon SR, Nair U, Schief WR, Batista FD. mRNA-LNP prime boost evolves precursors toward VRC01-like broadly neutralizing antibodies in preclinical humanized mouse models. Sci Immunol. 2024;9:eadn0622. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 26] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 49. | A Study to Evaluate the Safety and Immunogenicity of eOD-GT8 60mer mRNA Vaccine (mRNA-1644). [cited 21 January 2026]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/study/NCT05414786 ClinicalTrials.gov Identifier: NCT05414786. |
| 50. | A Phase 1 Study to Evaluate the Safety and Immunogenicity of eOD-GT8 60mer mRNA Vaccine (mRNA-1644) and Core-g28v2 60mer mRNA Vaccine (mRNA-1644v2-Core). [cited 21 January 2026]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/study/NCT05001373 ClinicalTrials.gov Identifier: NCT05001373. |
| 51. | Kumar S, Singh S, Luthra K. An Overview of Human Anti-HIV-1 Neutralizing Antibodies against Diverse Epitopes of HIV-1. ACS Omega. 2023;8:7252-7261. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 19] [Article Influence: 6.3] [Reference Citation Analysis (1)] |
| 52. | Phase 1 Clinical Trial to Evaluate the Safety and Immunogenicity of HIV-1 Chimp Adenovirus Vaccines Expressing Clade C gp140 & CH505TF gp120 Protein Boost in HIV-uninfected Adult. (HVTN139). [cited 21 January 2026]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/study/NCT05182125 ClinicalTrials.gov Identifier: NCT05182125. |
| 53. | Sanders RW, Dankers MM, Busser E, Caffrey M, Moore JP, Berkhout B. Evolution of the HIV-1 envelope glycoproteins with a disulfide bond between gp120 and gp41. Retrovirology. 2004;1:3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 30] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 54. | Sharma SK, de Val N, Bale S, Guenaga J, Tran K, Feng Y, Dubrovskaya V, Ward AB, Wyatt RT. Cleavage-independent HIV-1 Env trimers engineered as soluble native spike mimetics for vaccine design. Cell Rep. 2015;11:539-550. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 218] [Cited by in RCA: 197] [Article Influence: 17.9] [Reference Citation Analysis (1)] |
| 55. | Torrents de la Peña A, de Taeye SW, Sliepen K, LaBranche CC, Burger JA, Schermer EE, Montefiori DC, Moore JP, Klasse PJ, Sanders RW. Immunogenicity in Rabbits of HIV-1 SOSIP Trimers from Clades A, B, and C, Given Individually, Sequentially, or in Combination. J Virol. 2018;92:e01957-e01917. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 71] [Article Influence: 8.9] [Reference Citation Analysis (1)] |
| 56. | Klasse PJ, Ketas TJ, Cottrell CA, Ozorowski G, Debnath G, Camara D, Francomano E, Pugach P, Ringe RP, LaBranche CC, van Gils MJ, Bricault CA, Barouch DH, Crotty S, Silvestri G, Kasturi S, Pulendran B, Wilson IA, Montefiori DC, Sanders RW, Ward AB, Moore JP. Epitopes for neutralizing antibodies induced by HIV-1 envelope glycoprotein BG505 SOSIP trimers in rabbits and macaques. PLoS Pathog. 2018;14:e1006913. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 131] [Cited by in RCA: 106] [Article Influence: 13.3] [Reference Citation Analysis (1)] |
| 57. | Sanders RW, Derking R, Cupo A, Julien JP, Yasmeen A, de Val N, Kim HJ, Blattner C, de la Peña AT, Korzun J, Golabek M, de Los Reyes K, Ketas TJ, van Gils MJ, King CR, Wilson IA, Ward AB, Klasse PJ, Moore JP. A next-generation cleaved, soluble HIV-1 Env trimer, BG505 SOSIP.664 gp140, expresses multiple epitopes for broadly neutralizing but not non-neutralizing antibodies. PLoS Pathog. 2013;9:e1003618. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 966] [Cited by in RCA: 849] [Article Influence: 65.3] [Reference Citation Analysis (1)] |
| 58. | Beddows S, Franti M, Dey AK, Kirschner M, Iyer SP, Fisch DC, Ketas T, Yuste E, Desrosiers RC, Klasse PJ, Maddon PJ, Olson WC, Moore JP. A comparative immunogenicity study in rabbits of disulfide-stabilized, proteolytically cleaved, soluble trimeric human immunodeficiency virus type 1 gp140, trimeric cleavage-defective gp140 and monomeric gp120. Virology. 2007;360:329-340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 89] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 59. | Reiss EIMM, van der Straten K, Graus LTM, Grobben M, Vlaming KE, van der Veen AIP, Liesdek MH, Ozorowski G, Corcoran M, Gao H, Greene KM, Yates NL, Sawant S, Kerster G, Burger JA, Schonherr S, Cheeseman HM, Evans A, McFarlane LR, Tran AS, Torres JL, Lin RN, Jo G, Tolazzi M, Mundsperger P, Katinger D, Cupo A, Moore JP, Hurks R, Vogt L, Soeters MR, Kootstra NA, Scarlatti G, Tomaras GD, Montefiori DC, Karlsson Hedestam GB, Ward AB, Klouwens M, de Jong MD, Prins JM, Claireaux M, Geijtenbeek TBH, Shattock RJ, van Gils MJ, Sanders RW, de Bree GJ. HIV-1 envelope trimer vaccine induces sex-associated differences in antibody responses: a phase 1 clinical trial. Nat Commun. 2025;16:10250. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 60. | Ramezani-Rad P, Cottrell CA, Marina-Zárate E, Liguori A, Landais E, Torres JL, Myers A, Lee JH, Baboo S, Flynn C, McKenney K, Salcedo E, Zhou X, Kalyuzhniy O, Georgeson E, Phelps N, Lu D, Eskandarzadeh S, Menis S, Kubitz M, Groschel B, Alavi N, Jackson AM, Lee WH, Tran AS, Ben-Akiva E, Michaels KK, Diedrich JK, Enemuo CA, Lewis V, Pradhan A, Kasturi SP, Schiffner T, Steichen JM, Carnathan DG, Himansu S, Yates JR 3rd, Paulson JC, Ozorowski G, Irvine DJ, Silvestri G, Sok D, Ward AB, Crotty S, Schief WR. Vaccination with mRNA-encoded membrane-bound HIV Envelope trimer induces neutralizing antibodies in animal models. bioRxiv. 2025;2025.01.24.634423. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 61. | Ozorowski G, Cupo A, Golabek M, LoPiccolo M, Ketas TA, Cavallary M, Cottrell CA, Klasse PJ, Ward AB, Moore JP. Effects of Adjuvants on HIV-1 Envelope Glycoprotein SOSIP Trimers In Vitro. J Virol. 2018;92:e00381-e00318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 38] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 62. | Upadhyay C, Feyznezhad R, Cao L, Chan KW, Liu K, Yang W, Zhang H, Yolitz J, Arthos J, Nadas A, Kong XP, Zolla-Pazner S, Hioe CE. Signal peptide of HIV-1 envelope modulates glycosylation impacting exposure of V1V2 and other epitopes. PLoS Pathog. 2020;16:e1009185. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (1)] |
| 63. | Torrents de la Peña A, Julien JP, de Taeye SW, Garces F, Guttman M, Ozorowski G, Pritchard LK, Behrens AJ, Go EP, Burger JA, Schermer EE, Sliepen K, Ketas TJ, Pugach P, Yasmeen A, Cottrell CA, Torres JL, Vavourakis CD, van Gils MJ, LaBranche C, Montefiori DC, Desaire H, Crispin M, Klasse PJ, Lee KK, Moore JP, Ward AB, Wilson IA, Sanders RW. Improving the Immunogenicity of Native-like HIV-1 Envelope Trimers by Hyperstabilization. Cell Rep. 2017;20:1805-1817. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 175] [Cited by in RCA: 148] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 64. | Gote V, Bolla PK, Kommineni N, Butreddy A, Nukala PK, Palakurthi SS, Khan W. A Comprehensive Review of mRNA Vaccines. Int J Mol Sci. 2023;24:2700. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 382] [Cited by in RCA: 296] [Article Influence: 98.7] [Reference Citation Analysis (1)] |
| 65. | Ahmed S, Herschhorn A. mRNA-based HIV-1 vaccines. Clin Microbiol Rev. 2024;37:e0004124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 66. | Bloom K, van den Berg F, Arbuthnot P. Self-amplifying RNA vaccines for infectious diseases. Gene Ther. 2021;28:117-129. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 412] [Cited by in RCA: 328] [Article Influence: 65.6] [Reference Citation Analysis (1)] |
| 67. | Phase II Study of AGS-004 as an Immunotherapeutic in Antiretroviral Therapy (ART)-Treated Subjects Infected With HIV. [cited 21 January 2026]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/study/NCT00672191 ClinicalTrials.gov Identifier: NCT00672191. |
| 68. | Gandhi RT, Kwon DS, Macklin EA, Shopis JR, McLean AP, McBrine N, Flynn T, Peter L, Sbrolla A, Kaufmann DE, Porichis F, Walker BD, Bhardwaj N, Barouch DH, Kavanagh DG. Immunization of HIV-1-Infected Persons With Autologous Dendritic Cells Transfected With mRNA Encoding HIV-1 Gag and Nef: Results of a Randomized, Placebo-Controlled Clinical Trial. J Acquir Immune Defic Syndr. 2016;71:246-253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 75] [Article Influence: 7.5] [Reference Citation Analysis (1)] |
| 69. | de Jong W, Aerts J, Allard S, Brander C, Buyze J, Florence E, van Gorp E, Vanham G, Leal L, Mothe B, Thielemans K, Plana M, Garcia F, Gruters R; iHIVARNA consortium. iHIVARNA phase IIa, a randomized, placebo-controlled, double-blinded trial to evaluate the safety and immunogenicity of iHIVARNA-01 in chronically HIV-infected patients under stable combined antiretroviral therapy. Trials. 2019;20:361. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 35] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 70. | Leal L, Guardo AC, Morón-López S, Salgado M, Mothe B, Heirman C, Pannus P, Vanham G, van den Ham HJ, Gruters R, Andeweg A, Van Meirvenne S, Pich J, Arnaiz JA, Gatell JM, Brander C, Thielemans K, Martínez-Picado J, Plana M, García F; iHIVARNA consortium. Phase I clinical trial of an intranodally administered mRNA-based therapeutic vaccine against HIV-1 infection. AIDS. 2018;32:2533-2545. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 69] [Article Influence: 8.6] [Reference Citation Analysis (1)] |
| 71. | A Clinical Trial to Evaluate the Safety and Immunogenicity of BG505 MD39.3, BG505 MD39.3 gp151, and BG505 MD39.3 gp151 CD4KO HIV Trimer mRNA Vaccines in Healthy, HIV-uninfected Adult Participants. [cited 21 January 2026]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/study/NCT05217641 ClinicalTrials.gov Identifier: NCT05217641. |
| 72. | Parks KR, Moodie Z, Allen MA, Yen C, Furch BD, MacPhee KJ, Ozorowski G, Heptinstall J, Hahn WO, Zheng Z, Lu H, Grant S, Domin E, Duff MO, Seese A, Marini-Macouzet C, Ballweber-Fleming L, Lee WH, Cottrell CA, Liguori A, Georgeson E, Alavi N, Kubitz M, Phelps N, Seaton KE, Cohen KW, Anderson MA, Mondal K, Laufer DS, Kublin JG, Ward AB, Hyrien O, De Rosa SC, Himansu S, Leav B, Reuter C, Tomaras GD, Montefiori D, Walsh SR, Frank I, Sobieszczyk ME, Goepfert PA, Stephenson KE, Baden LR, Van Tieu H, Keefer MC, Clark J, Riddler SA, Schief WR, McElrath MJ. Vaccination with mRNA-encoded membrane-anchored HIV envelope trimers elicited tier 2 neutralizing antibodies in a phase 1 clinical trial. Sci Transl Med. 2025;17:eady6831. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 73. | Valentin A, Bergamaschi C, Rosati M, Angel M, Burns R, Agarwal M, Gergen J, Petsch B, Oostvogels L, Loeliger E, Chew KW, Deeks SG, Mullins JI, Pavlakis GN, Felber BK. Comparative immunogenicity of an mRNA/LNP and a DNA vaccine targeting HIV gag conserved elements in macaques. Front Immunol. 2022;13:945706. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 74. | Xu Y, Ferguson T, Masuda K, Siddiqui MA, Smith KP, Vest O, Brooks B, Zhou Z, Obliosca J, Kong XP, Jiang X, Yamashita M, Moriya T, Tison C. Short Carbon Nanotube-Based Delivery of mRNA for HIV-1 Vaccines. Biomolecules. 2023;13:1088. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 75. | Haynes BF, Wiehe K, Alam SM, Weissman D, Saunders KO. Progress with induction of HIV broadly neutralizing antibodies in the Duke Consortia for HIV/AIDS Vaccine Development. Curr Opin HIV AIDS. 2023;18:300-308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 76. | Shim I, Rogowski L, Venketaraman V. Progress and Recent Developments in HIV Vaccine Research. Vaccines (Basel). 2025;13:690. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 77. | Jefferys R. HIV Vaccines and Passive Immunization Pipeline Report 2024. [cited 21 January 2026]. Available from: https://www.treatmentactiongroup.org/wp-content/uploads/2024/07/pipeline_HIV_VAX_2024_final.pdf. |
| 78. | Fries CN, Curvino EJ, Chen JL, Permar SR, Fouda GG, Collier JH. Advances in nanomaterial vaccine strategies to address infectious diseases impacting global health. Nat Nanotechnol. 2021;16:1-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 144] [Article Influence: 28.8] [Reference Citation Analysis (0)] |
| 79. | Karch CP, Matyas GR. The current and future role of nanovaccines in HIV-1 vaccine development. Expert Rev Vaccines. 2021;20:935-944. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (2)] |
| 80. | Kim CG, Lee JC, Ju DB, Kim SK, Yun CH, Cho CS. Enhancement of Immune Responses Elicited by Nanovaccines through a Cross-Presentation Pathway. Tissue Eng Regen Med. 2023;20:355-370. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 13] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 81. | Xu L, Liu Y, Chen Z, Li W, Liu Y, Wang L, Liu Y, Wu X, Ji Y, Zhao Y, Ma L, Shao Y, Chen C. Surface-engineered gold nanorods: promising DNA vaccine adjuvant for HIV-1 treatment. Nano Lett. 2012;12:2003-2012. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 288] [Cited by in RCA: 209] [Article Influence: 14.9] [Reference Citation Analysis (1)] |
| 82. | Damm D, Kostka K, Weingärtner C, Wagner JT, Rojas-Sánchez L, Gensberger-Reigl S, Sokolova V, Überla K, Epple M, Temchura V. Covalent coupling of HIV-1 glycoprotein trimers to biodegradable calcium phosphate nanoparticles via genetically encoded aldehyde-tags. Acta Biomater. 2022;140:586-600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 83. | Li S, Wang B, Jiang S, Pan Y, Shi Y, Kong W, Shan Y. Surface-Functionalized Silica-Coated Calcium Phosphate Nanoparticles Efficiently Deliver DNA-Based HIV-1 Trimeric Envelope Vaccines against HIV-1. ACS Appl Mater Interfaces. 2021;13:53630-53645. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (1)] |
| 84. | Al-Nemrawi NK, Darweesh RS, Al-Shriem LA, Al-Qawasmi FS, Emran SO, Khafajah AS, Abu-Dalo MA. Polymeric Nanoparticles for Inhaled Vaccines. Polymers (Basel). 2022;14:4450. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 85. | Zhu Q, Talton J, Zhang G, Cunningham T, Wang Z, Waters RC, Kirk J, Eppler B, Klinman DM, Sui Y, Gagnon S, Belyakov IM, Mumper RJ, Berzofsky JA. Large intestine-targeted, nanoparticle-releasing oral vaccine to control genitorectal viral infection. Nat Med. 2012;18:1291-1296. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 165] [Cited by in RCA: 139] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 86. | Sliepen K, Ozorowski G, Burger JA, van Montfort T, Stunnenberg M, LaBranche C, Montefiori DC, Moore JP, Ward AB, Sanders RW. Presenting native-like HIV-1 envelope trimers on ferritin nanoparticles improves their immunogenicity. Retrovirology. 2015;12:82. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 183] [Cited by in RCA: 161] [Article Influence: 14.6] [Reference Citation Analysis (1)] |
| 87. | Zhang YN, Paynter J, Antanasijevic A, Allen JD, Eldad M, Lee YZ, Copps J, Newby ML, He L, Chavez D, Frost P, Goodroe A, Dutton J, Lanford R, Chen C, Wilson IA, Crispin M, Ward AB, Zhu J. Single-component multilayered self-assembling protein nanoparticles presenting glycan-trimmed uncleaved prefusion optimized envelope trimmers as HIV-1 vaccine candidates. Nat Commun. 2023;14:1985. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 37] [Article Influence: 12.3] [Reference Citation Analysis (1)] |
| 88. | Jardine JG, Ota T, Sok D, Pauthner M, Kulp DW, Kalyuzhniy O, Skog PD, Thinnes TC, Bhullar D, Briney B, Menis S, Jones M, Kubitz M, Spencer S, Adachi Y, Burton DR, Schief WR, Nemazee D. HIV-1 VACCINES. Priming a broadly neutralizing antibody response to HIV-1 using a germline-targeting immunogen. Science. 2015;349:156-161. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 416] [Cited by in RCA: 365] [Article Influence: 33.2] [Reference Citation Analysis (1)] |
| 89. | Kato Y, Abbott RK, Freeman BL, Haupt S, Groschel B, Silva M, Menis S, Irvine DJ, Schief WR, Crotty S. Multifaceted Effects of Antigen Valency on B Cell Response Composition and Differentiation In Vivo. Immunity. 2020;53:548-563.e8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 267] [Cited by in RCA: 225] [Article Influence: 37.5] [Reference Citation Analysis (1)] |
| 90. | Sliepen K, Schermer E, Bontjer I, Burger JA, Lévai RF, Mundsperger P, Brouwer PJM, Tolazzi M, Farsang A, Katinger D, Moore JP, Scarlatti G, Shattock RJ, Sattentau QJ, Sanders RW. Interplay of diverse adjuvants and nanoparticle presentation of native-like HIV-1 envelope trimers. NPJ Vaccines. 2021;6:103. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 91. | Kasturi SP, Rasheed MAU, Havenar-Daughton C, Pham M, Legere T, Sher ZJ, Kovalenkov Y, Gumber S, Huang JY, Gottardo R, Fulp W, Sato A, Sawant S, Stanfield-Oakley S, Yates N, LaBranche C, Alam SM, Tomaras G, Ferrari G, Montefiori D, Wrammert J, Villinger F, Tomai M, Vasilakos J, Fox CB, Reed SG, Haynes BF, Crotty S, Ahmed R, Pulendran B. 3M-052, a synthetic TLR-7/8 agonist, induces durable HIV-1 envelope-specific plasma cells and humoral immunity in nonhuman primates. Sci Immunol. 2020;5:eabb1025. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 139] [Cited by in RCA: 129] [Article Influence: 21.5] [Reference Citation Analysis (1)] |
| 92. | Xiao Q, Guo D, Chen S. Application of CRISPR/Cas9-Based Gene Editing in HIV-1/AIDS Therapy. Front Cell Infect Microbiol. 2019;9:69. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 145] [Cited by in RCA: 118] [Article Influence: 16.9] [Reference Citation Analysis (1)] |
| 93. | Dash PK, Chen C, Kaminski R, Su H, Mancuso P, Sillman B, Zhang C, Liao S, Sravanam S, Liu H, Waight E, Guo L, Mathews S, Sariyer R, Mosley RL, Poluektova LY, Caocci M, Amini S, Gorantla S, Burdo TH, Edagwa B, Gendelman HE, Khalili K. CRISPR editing of CCR5 and HIV-1 facilitates viral elimination in antiretroviral drug-suppressed virus-infected humanized mice. Proc Natl Acad Sci U S A. 2023;120:e2217887120. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 68] [Cited by in RCA: 54] [Article Influence: 18.0] [Reference Citation Analysis (1)] |
| 94. | European AIDS Treatment Group. CRISPR HIV gene therapy disappoints in early study. [cited 21 January 2026]. Available from: https://www.eatg.org/hiv-news/crispr-hiv-gene-therapy-disappoints-in-early-study/. |
| 95. | Armbruster C, Stiegler GM, Vcelar BA, Jäger W, Michael NL, Vetter N, Katinger HW. A phase I trial with two human monoclonal antibodies (hMAb 2F5, 2G12) against HIV-1. AIDS. 2002;16:227-233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 107] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 96. | Stiegler G, Armbruster C, Vcelar B, Stoiber H, Kunert R, Michael NL, Jagodzinski LL, Ammann C, Jäger W, Jacobson J, Vetter N, Katinger H. Antiviral activity of the neutralizing antibodies 2F5 and 2G12 in asymptomatic HIV-1-infected humans: a phase I evaluation. AIDS. 2002;16:2019-2025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 78] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 97. | Manrique A, Rusert P, Joos B, Fischer M, Kuster H, Leemann C, Niederöst B, Weber R, Stiegler G, Katinger H, Günthard HF, Trkola A. In vivo and in vitro escape from neutralizing antibodies 2G12, 2F5, and 4E10. J Virol. 2007;81:8793-8808. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 80] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 98. | Trkola A, Kuster H, Rusert P, Joos B, Fischer M, Leemann C, Manrique A, Huber M, Rehr M, Oxenius A, Weber R, Stiegler G, Vcelar B, Katinger H, Aceto L, Günthard HF. Delay of HIV-1 rebound after cessation of antiretroviral therapy through passive transfer of human neutralizing antibodies. Nat Med. 2005;11:615-622. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 413] [Cited by in RCA: 399] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 99. | Mehandru S, Vcelar B, Wrin T, Stiegler G, Joos B, Mohri H, Boden D, Galovich J, Tenner-Racz K, Racz P, Carrington M, Petropoulos C, Katinger H, Markowitz M. Adjunctive passive immunotherapy in human immunodeficiency virus type 1-infected individuals treated with antiviral therapy during acute and early infection. J Virol. 2007;81:11016-11031. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 103] [Article Influence: 5.4] [Reference Citation Analysis (1)] |
| 100. | Yang G, Holl TM, Liu Y, Li Y, Lu X, Nicely NI, Kepler TB, Alam SM, Liao HX, Cain DW, Spicer L, VandeBerg JL, Haynes BF, Kelsoe G. Identification of autoantigens recognized by the 2F5 and 4E10 broadly neutralizing HIV-1 antibodies. J Exp Med. 2013;210:241-256. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 171] [Cited by in RCA: 157] [Article Influence: 12.1] [Reference Citation Analysis (1)] |
| 101. | deCamp A, Hraber P, Bailer RT, Seaman MS, Ochsenbauer C, Kappes J, Gottardo R, Edlefsen P, Self S, Tang H, Greene K, Gao H, Daniell X, Sarzotti-Kelsoe M, Gorny MK, Zolla-Pazner S, LaBranche CC, Mascola JR, Korber BT, Montefiori DC. Global panel of HIV-1 Env reference strains for standardized assessments of vaccine-elicited neutralizing antibodies. J Virol. 2014;88:2489-2507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 342] [Cited by in RCA: 299] [Article Influence: 24.9] [Reference Citation Analysis (1)] |
| 102. | Tiller T, Meffre E, Yurasov S, Tsuiji M, Nussenzweig MC, Wardemann H. Efficient generation of monoclonal antibodies from single human B cells by single cell RT-PCR and expression vector cloning. J Immunol Methods. 2008;329:112-124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1071] [Cited by in RCA: 924] [Article Influence: 51.3] [Reference Citation Analysis (0)] |
| 103. | Barouch DH, Whitney JB, Moldt B, Klein F, Oliveira TY, Liu J, Stephenson KE, Chang HW, Shekhar K, Gupta S, Nkolola JP, Seaman MS, Smith KM, Borducchi EN, Cabral C, Smith JY, Blackmore S, Sanisetty S, Perry JR, Beck M, Lewis MG, Rinaldi W, Chakraborty AK, Poignard P, Nussenzweig MC, Burton DR. Therapeutic efficacy of potent neutralizing HIV-1-specific monoclonal antibodies in SHIV-infected rhesus monkeys. Nature. 2013;503:224-228. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 595] [Cited by in RCA: 566] [Article Influence: 43.5] [Reference Citation Analysis (1)] |
| 104. | Shingai M, Nishimura Y, Klein F, Mouquet H, Donau OK, Plishka R, Buckler-White A, Seaman M, Piatak M Jr, Lifson JD, Dimitrov DS, Nussenzweig MC, Martin MA. Antibody-mediated immunotherapy of macaques chronically infected with SHIV suppresses viraemia. Nature. 2013;503:277-280. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 434] [Cited by in RCA: 404] [Article Influence: 31.1] [Reference Citation Analysis (1)] |
| 105. | Nishimura Y, Gautam R, Chun TW, Sadjadpour R, Foulds KE, Shingai M, Klein F, Gazumyan A, Golijanin J, Donaldson M, Donau OK, Plishka RJ, Buckler-White A, Seaman MS, Lifson JD, Koup RA, Fauci AS, Nussenzweig MC, Martin MA. Early antibody therapy can induce long-lasting immunity to SHIV. Nature. 2017;543:559-563. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 294] [Cited by in RCA: 277] [Article Influence: 30.8] [Reference Citation Analysis (0)] |
| 106. | Gautam R, Nishimura Y, Gaughan N, Gazumyan A, Schoofs T, Buckler-White A, Seaman MS, Swihart BJ, Follmann DA, Nussenzweig MC, Martin MA. A single injection of crystallizable fragment domain-modified antibodies elicits durable protection from SHIV infection. Nat Med. 2018;24:610-616. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 112] [Cited by in RCA: 105] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 107. | Caskey M, Klein F, Lorenzi JC, Seaman MS, West AP Jr, Buckley N, Kremer G, Nogueira L, Braunschweig M, Scheid JF, Horwitz JA, Shimeliovich I, Ben-Avraham S, Witmer-Pack M, Platten M, Lehmann C, Burke LA, Hawthorne T, Gorelick RJ, Walker BD, Keler T, Gulick RM, Fätkenheuer G, Schlesinger SJ, Nussenzweig MC. Viraemia suppressed in HIV-1-infected humans by broadly neutralizing antibody 3BNC117. Nature. 2015;522:487-491. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 717] [Cited by in RCA: 663] [Article Influence: 60.3] [Reference Citation Analysis (1)] |
| 108. | Lynch RM, Boritz E, Coates EE, DeZure A, Madden P, Costner P, Enama ME, Plummer S, Holman L, Hendel CS, Gordon I, Casazza J, Conan-Cibotti M, Migueles SA, Tressler R, Bailer RT, McDermott A, Narpala S, O'Dell S, Wolf G, Lifson JD, Freemire BA, Gorelick RJ, Pandey JP, Mohan S, Chomont N, Fromentin R, Chun TW, Fauci AS, Schwartz RM, Koup RA, Douek DC, Hu Z, Capparelli E, Graham BS, Mascola JR, Ledgerwood JE; VRC 601 Study Team. Virologic effects of broadly neutralizing antibody VRC01 administration during chronic HIV-1 infection. Sci Transl Med. 2015;7:319ra206. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 435] [Cited by in RCA: 399] [Article Influence: 36.3] [Reference Citation Analysis (0)] |
| 109. | Caskey M, Schoofs T, Gruell H, Settler A, Karagounis T, Kreider EF, Murrell B, Pfeifer N, Nogueira L, Oliveira TY, Learn GH, Cohen YZ, Lehmann C, Gillor D, Shimeliovich I, Unson-O'Brien C, Weiland D, Robles A, Kümmerle T, Wyen C, Levin R, Witmer-Pack M, Eren K, Ignacio C, Kiss S, West AP Jr, Mouquet H, Zingman BS, Gulick RM, Keler T, Bjorkman PJ, Seaman MS, Hahn BH, Fätkenheuer G, Schlesinger SJ, Nussenzweig MC, Klein F. Antibody 10-1074 suppresses viremia in HIV-1-infected individuals. Nat Med. 2017;23:185-191. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 477] [Cited by in RCA: 420] [Article Influence: 46.7] [Reference Citation Analysis (0)] |
| 110. | Stephenson KE, Julg B, Tan CS, Zash R, Walsh SR, Rolle CP, Monczor AN, Lupo S, Gelderblom HC, Ansel JL, Kanjilal DG, Maxfield LF, Nkolola J, Borducchi EN, Abbink P, Liu J, Peter L, Chandrashekar A, Nityanandam R, Lin Z, Setaro A, Sapiente J, Chen Z, Sunner L, Cassidy T, Bennett C, Sato A, Mayer B, Perelson AS, deCamp A, Priddy FH, Wagh K, Giorgi EE, Yates NL, Arduino RC, DeJesus E, Tomaras GD, Seaman MS, Korber B, Barouch DH. Safety, pharmacokinetics and antiviral activity of PGT121, a broadly neutralizing monoclonal antibody against HIV-1: a randomized, placebo-controlled, phase 1 clinical trial. Nat Med. 2021;27:1718-1724. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 91] [Cited by in RCA: 81] [Article Influence: 16.2] [Reference Citation Analysis (1)] |
| 111. | Pannus P, Rutsaert S, De Wit S, Allard SD, Vanham G, Cole B, Nescoi C, Aerts J, De Spiegelaere W, Tsoumanis A, Couttenye MM, Herssens N, De Scheerder MA, Vandekerckhove L, Florence E. Rapid viral rebound after analytical treatment interruption in patients with very small HIV reservoir and minimal on-going viral transcription. J Int AIDS Soc. 2020;23:e25453. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 46] [Article Influence: 7.7] [Reference Citation Analysis (1)] |
| 112. | Crowell TA, Colby DJ, Pinyakorn S, Sacdalan C, Pagliuzza A, Intasan J, Benjapornpong K, Tangnaree K, Chomchey N, Kroon E, de Souza MS, Tovanabutra S, Rolland M, Eller MA, Paquin-Proulx D, Bolton DL, Tokarev A, Thomas R, Takata H, Trautmann L, Krebs SJ, Modjarrad K, McDermott AB, Bailer RT, Doria-Rose N, Patel B, Gorelick RJ, Fullmer BA, Schuetz A, Grandin PV, O'Connell RJ, Ledgerwood JE, Graham BS, Tressler R, Mascola JR, Chomont N, Michael NL, Robb ML, Phanuphak N, Ananworanich J; RV397 Study Group. Safety and efficacy of VRC01 broadly neutralising antibodies in adults with acutely treated HIV (RV397): a phase 2, randomised, double-blind, placebo-controlled trial. Lancet HIV. 2019;6:e297-e306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 89] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 113. | Scheid JF, Horwitz JA, Bar-On Y, Kreider EF, Lu CL, Lorenzi JC, Feldmann A, Braunschweig M, Nogueira L, Oliveira T, Shimeliovich I, Patel R, Burke L, Cohen YZ, Hadrigan S, Settler A, Witmer-Pack M, West AP Jr, Juelg B, Keler T, Hawthorne T, Zingman B, Gulick RM, Pfeifer N, Learn GH, Seaman MS, Bjorkman PJ, Klein F, Schlesinger SJ, Walker BD, Hahn BH, Nussenzweig MC, Caskey M. HIV-1 antibody 3BNC117 suppresses viral rebound in humans during treatment interruption. Nature. 2016;535:556-560. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 458] [Cited by in RCA: 421] [Article Influence: 42.1] [Reference Citation Analysis (1)] |
| 114. | Bar KJ, Sneller MC, Harrison LJ, Justement JS, Overton ET, Petrone ME, Salantes DB, Seamon CA, Scheinfeld B, Kwan RW, Learn GH, Proschan MA, Kreider EF, Blazkova J, Bardsley M, Refsland EW, Messer M, Clarridge KE, Tustin NB, Madden PJ, Oden K, O'Dell SJ, Jarocki B, Shiakolas AR, Tressler RL, Doria-Rose NA, Bailer RT, Ledgerwood JE, Capparelli EV, Lynch RM, Graham BS, Moir S, Koup RA, Mascola JR, Hoxie JA, Fauci AS, Tebas P, Chun TW. Effect of HIV Antibody VRC01 on Viral Rebound after Treatment Interruption. N Engl J Med. 2016;375:2037-2050. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 451] [Cited by in RCA: 414] [Article Influence: 41.4] [Reference Citation Analysis (0)] |
| 115. | Cale EM, Bai H, Bose M, Messina MA, Colby DJ, Sanders-Buell E, Dearlove B, Li Y, Engeman E, Silas D, O'Sullivan AM, Mann B, Pinyakorn S, Intasan J, Benjapornpong K, Sacdalan C, Kroon E, Phanuphak N, Gramzinski R, Vasan S, Robb ML, Michael NL, Lynch RM, Bailer RT, Pagliuzza A, Chomont N, Pegu A, Doria-Rose NA, Trautmann L, Crowell TA, Mascola JR, Ananworanich J, Tovanabutra S, Rolland M; RV397 Study Group. Neutralizing antibody VRC01 failed to select for HIV-1 mutations upon viral rebound. J Clin Invest. 2020;130:3299-3304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 39] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 116. | Cohen YZ, Butler AL, Millard K, Witmer-Pack M, Levin R, Unson-O'Brien C, Patel R, Shimeliovich I, Lorenzi JCC, Horowitz J, Walsh SR, Lin S, Weiner JA, Tse A, Sato A, Bennett C, Mayer B, Seaton KE, Yates NL, Baden LR, deCamp AC, Ackerman ME, Seaman MS, Tomaras GD, Nussenzweig MC, Caskey M. Safety, pharmacokinetics, and immunogenicity of the combination of the broadly neutralizing anti-HIV-1 antibodies 3BNC117 and 10-1074 in healthy adults: A randomized, phase 1 study. PLoS One. 2019;14:e0219142. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 71] [Article Influence: 10.1] [Reference Citation Analysis (1)] |
| 117. | Mendoza P, Gruell H, Nogueira L, Pai JA, Butler AL, Millard K, Lehmann C, Suárez I, Oliveira TY, Lorenzi JCC, Cohen YZ, Wyen C, Kümmerle T, Karagounis T, Lu CL, Handl L, Unson-O'Brien C, Patel R, Ruping C, Schlotz M, Witmer-Pack M, Shimeliovich I, Kremer G, Thomas E, Seaton KE, Horowitz J, West AP Jr, Bjorkman PJ, Tomaras GD, Gulick RM, Pfeifer N, Fätkenheuer G, Seaman MS, Klein F, Caskey M, Nussenzweig MC. Combination therapy with anti-HIV-1 antibodies maintains viral suppression. Nature. 2018;561:479-484. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 498] [Cited by in RCA: 450] [Article Influence: 56.3] [Reference Citation Analysis (1)] |
| 118. | Wohlwend J, Nathan A, Shalon N, Crain CR, Tano-Menka R, Goldberg B, Richards E, Gaiha GD, Barzilay R. Deep learning enhances the prediction of HLA class I-presented CD8(+) T cell epitopes in foreign pathogens. Nat Mach Intell. 2025;7:232-243. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 19] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 119. | Reynisson B, Alvarez B, Paul S, Peters B, Nielsen M. NetMHCpan-4.1 and NetMHCIIpan-4.0: improved predictions of MHC antigen presentation by concurrent motif deconvolution and integration of MS MHC eluted ligand data. Nucleic Acids Res. 2020;48:W449-W454. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2060] [Cited by in RCA: 1605] [Article Influence: 267.5] [Reference Citation Analysis (1)] |
| 120. | Bai H, Lewitus E, Li Y, Thomas PV, Zemil M, Merbah M, Peterson CE, Thuraisamy T, Rees PA, Hajduczki A, Dussupt V, Slike B, Mendez-Rivera L, Schmid A, Kavusak E, Rao M, Smith G, Frey J, Sims A, Wieczorek L, Polonis V, Krebs SJ, Ake JA, Vasan S, Bolton DL, Joyce MG, Townsley S, Rolland M. Contemporary HIV-1 consensus Env with AI-assisted redesigned hypervariable loops promote antibody binding. Nat Commun. 2024;15:3924. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 121. | Henderson R, Anasti K, Manne K, Stalls V, Saunders C, Bililign Y, Williams A, Bubphamala P, Montani M, Kachhap S, Li J, Jaing C, Newman A, Cain DW, Lu X, Venkatayogi S, Berry M, Wagh K, Korber B, Saunders KO, Tian M, Alt F, Wiehe K, Acharya P, Alam SM, Haynes BF. Engineering immunogens that select for specific mutations in HIV broadly neutralizing antibodies. Nat Commun. 2024;15:9503. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 122. | Slack C, Ndebele P, Allen M, Salzwedel J. Shifts in UNAIDS ethics guidance and implications for ethics review of preventive HIV vaccine trials. J Int AIDS Soc. 2021;24 Suppl 7:e25796. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 123. | Trkola A, Moore PL. Vaccinating people living with HIV: a fast track to preventive and therapeutic HIV vaccines. Lancet Infect Dis. 2024;24:e252-e255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 124. | Voronin Y, Zinszner H, Karg C, Brooks K, Coombs R, Hural J, Holt R, Fast P, Allen M; VISR Working Group of Global HIV Vaccine Enterprise. HIV vaccine-induced sero-reactivity: a challenge for trial participants, researchers, and physicians. Vaccine. 2015;33:1243-1249. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 37] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 125. | Cutrell A, Donnell D, Dunn DT, Glidden DV, Grobler A, Hanscom B, Stancil BS, Meyer RD, Wang R, Cuffe RL. HIV prevention trial design in an era of effective pre-exposure prophylaxis. HIV Clin Trials. 2017;18:177-188. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 126. | Lo YR, Chu C, Ananworanich J, Excler JL, Tucker JD. Stakeholder Engagement in HIV Cure Research: Lessons Learned from Other HIV Interventions and the Way Forward. AIDS Patient Care STDS. 2015;29:389-399. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 65] [Article Influence: 5.9] [Reference Citation Analysis (1)] |
| 127. | Cohen KW, De Rosa SC, Fulp WJ, deCamp AC, Fiore-Gartland A, Mahoney CR, Furth S, Donahue J, Whaley RE, Ballweber-Fleming L, Seese A, Schwedhelm K, Geraghty D, Finak G, Menis S, Leggat DJ, Rahaman F, Lombardo A, Borate BR, Philiponis V, Maenza J, Diemert D, Kolokythas O, Khati N, Bethony J, Hyrien O, Laufer DS, Koup RA, McDermott AB, Schief WR, McElrath MJ. A first-in-human germline-targeting HIV nanoparticle vaccine induced broad and publicly targeted helper T cell responses. Sci Transl Med. 2023;15:eadf3309. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 60] [Article Influence: 20.0] [Reference Citation Analysis (1)] |
| 128. | AVAC. HVTN 300. [cited 23 January 2026]. Available from: Available from: https://avac.org/trial/hvtn-300/. |
| 129. | Evaluating the Safety and Immunogenicity of HIV-1 BG505 SOSIP.664 gp140 With TLR Agonist and/or Alum Adjuvants in Healthy, HIV-uninfected Adults. [cited 23 January 2026]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/study/NCT04177355 ClinicalTrials.gov Identifier: NCT04177355. |
| 130. | AVAC. HVTN 139. [cited 23 January 2026]. Available from: https://avac.org/trial/hvtn-139/. |
| 131. | Houser KV, Gaudinski MR, Happe M, Narpala S, Verardi R, Sarfo EK, Corrigan AR, Wu R, Rothwell RS, Novik L, Hendel CS, Gordon IJ, Berkowitz NM, Cartagena CT, Widge AT, Coates EE, Strom L, Hickman S, Conan-Cibotti M, Vazquez S, Trofymenko O, Plummer S, Stein J, Case CL, Nason M, Biju A, Parchment DK, Changela A, Cheng C, Duan H, Geng H, Teng IT, Zhou T, O'Connell S, Barry C, Carlton K, Gall JG, Flach B, Doria-Rose NA, Graham BS, Koup RA, McDermott AB, Mascola JR, Kwong PD, Ledgerwood JE; VRC 018 Study Team. Safety and immunogenicity of an HIV-1 prefusion-stabilized envelope trimer (Trimer 4571) vaccine in healthy adults: A first-in-human open-label, randomized, dose-escalation, phase 1 clinical trial. EClinicalMedicine. 2022;48:101477. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 38] [Article Influence: 9.5] [Reference Citation Analysis (1)] |
| 132. | A Trial of a Gorilla Adenovirus Vectored Networked Epitopes Vaccine, Administered to Healthy Adults Living Without and With HIV. (IAVI C114). [cited 23 January 2026]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/study/NCT06617091 ClinicalTrials.gov Identifier: NCT06617091. |