Published online Mar 25, 2026. doi: 10.5501/wjv.v15.i1.115210
Revised: November 22, 2025
Accepted: December 29, 2025
Published online: March 25, 2026
Processing time: 153 Days and 21 Hours
Rift Valley fever (RVF) is an acute, mosquito-borne zoonotic disease caused by the RVF virus (RVFV), which belongs to the Phlebovirus genus of the Bunyaviridae family. The infection mainly targets domestic ruminants such as sheep, goats, cattle, and camels, with sheep showing the highest vulnerability. The virus is spread predominantly through the bites of Aedes and Culex mosquitoes, although humans may also become infected by direct exposure to blood, organs, or se
Core Tip: Rift Valley fever is a re-emerging zoonotic disease with significant health and economic impacts across Africa and the Middle East. This mini-review comprehensively discusses its virology, transmission, clinical manifestations, and challenges in diagnosis, treatment, and vaccine development. It highlights current outbreak trends, environmental and socioeconomic factors, and emphasizes the urgent need for global surveillance, intersectoral collaboration, and the development of effective prevention and control strategies for Rift Valley fever.
- Citation: Uppala PK, Karanam SK, Kandra NV, Edhi S. Rift Valley fever: An emerging zoonotic threat to global health and agriculture. World J Virol 2026; 15(1): 115210
- URL: https://www.wjgnet.com/2220-3249/full/v15/i1/115210.htm
- DOI: https://dx.doi.org/10.5501/wjv.v15.i1.115210
Rift Valley fever (RVF) is a mosquito-transmitted zoonotic infection that significantly impacts public health, livestock productivity, and local economies. The disease is caused by the RVF virus (RVFV), a single-stranded RNA virus classified under the Phlebovirus genus of the Bunyaviridae family[1]. This viral family also contains several other important pathogens, including the Punta Toro virus and the severe fever with thrombocytopenia syndrome virus.
RVF predominantly impacts domestic ruminants, including sheep, goats, cattle, and camels. The highest vulnerability is observed in young animals and pregnant females. In livestock, the disease typically presents with sudden onset of high fever, hepatic dysfunction, abortion storms, and high neonatal mortality rates. These manifestations often lead to major financial setbacks in regions heavily reliant on animal husbandry.
Humans can contract RVF either through the bite of infected mosquitoes, mainly species of the Aedes and Culex genera, or through direct exposure to infected animals. High-risk activities include handling blood, tissues, or body fluids during slaughtering, animal births, or veterinary practices. While the majority of human infections are mild and resolve without complications, a subset of patients may progress to severe conditions, including hemorrhagic manifestations, neurological involvement such as encephalitis, or eye-related complications that may result in long-term effects[2].
Due to its zoonotic nature, potential for outbreak expansion, and capacity to disrupt food systems, RVF is considered a disease of high concern. Enhanced surveillance, public awareness, vector control, and livestock vaccination programs are critical to mitigating its spread and impact. As global environmental changes continue to alter mosquito habitats, the risk of RVF emergence in previously unaffected regions remains an ongoing challenge for both animal and human health sectors.
RVFV was first detected in 1931 during an outbreak among sheep in the Rift Valley of Kenya. Since then, the virus has been linked to several major epidemics that have impacted both animals and humans in various parts of Africa. Countries where large-scale transmission has been recorded include Egypt, Kenya, South Africa, Madagascar, Mauritania, Senegal, and The Gambia. These outbreaks highlight the virus’s ability to cause serious public health crises and economic losses in regions where it becomes established[3].
The emergence and intensity of RVF outbreaks are strongly associated with specific environmental conditions, particularly periods of excessive rainfall, flooding, and increased vegetation. These factors create optimal breeding grounds for mosquito vectors, especially those of the Aedes and Culex genera, which play a central role in virus transmission to susceptible animal hosts[4].
Until the mid-1970s, RVF was largely considered an animal disease confined to Africa, with human infections being rare and usually mild. The situation changed in 1975, when South Africa recorded a severe outbreak involving hemorrhagic fever and deaths in humans. Similar large-scale outbreaks were later documented in Egypt in 1977 and Mauritania in 1987. A particularly devastating epidemic occurred in East Africa in late 1997, where unexplained human deaths were reported in Kenya’s Northeastern Province and Southern Somalia.
In Egypt, the 1977 outbreak was linked to the introduction of infected livestock through trade along the Nile irrigation system. Between 1997 and 1998, extensive flooding caused by the El Niño phenomenon led to a major RVF outbreak in Kenya, Somalia, and Tanzania. In September 2000, the disease crossed continental borders for the first time, spreading to Saudi Arabia and Yemen through livestock trade from the Horn of Africa. This demonstrated the virus’s ability to expand beyond Africa when ecological and trade conditions are favorable. Further outbreaks followed in 2006-2007 in Kenya, later extending to Tanzania and Somalia, while Madagascar and South Africa experienced epidemics in 2007 and 2008[5].
As of now, there have been no documented RVF outbreaks in Europe.
In Mayotte, a French territory in the Indian Ocean, the first human cases of RVF were documented in 2007. Retro
Serological studies in countries bordering the Mediterranean – including Turkey, Tunisia, Iran, Iraq, Algeria, and the Western Sahara – have indicated RVFV exposure in humans and animals. However, due to limited data and regional va
| Category | Details |
| Mosquito vectors | Aedes species: Primary vectors, capable of vertical transmission via eggs. Culex, Mansonia, Anopheles: Secondary/mechanical vectors that amplify outbreaks in new regions |
| Transmission ecology | Primary foci: Virus maintained between Aedes mosquitoes and ruminant hosts through vertical transmission. Secondary foci: Spread through livestock movement and dispersal of infected mosquitoes; outbreaks amplified by local mosquito species in irrigated/flood-prone areas |
| Host animals | Affects cattle, sheep, goats, and camels. Sheep and goats more susceptible than cattle and camels. Outbreaks often marked by sudden abortion waves in livestock |
| Human transmission | Direct/indirect contact with infected animal blood, tissues, or organs during slaughtering, butchering, veterinary work, or carcass handling. Consumption of raw/unpasteurized milk from infected animals. Bites from infected mosquitoes (Aedes, Culex) and possibly blood-feeding flies |
| Human-to-human spread | No documented human-to-human transmission. No transmission to healthcare workers when standard infection control is used |
Although most human cases of RVF are mild or asymptomatic, some individuals develop severe forms of the disease, such as hemorrhagic fever, encephalitis, retinitis, or jaundice (icterus). These complications can result in fatal outcomes, particularly among patients experiencing hemorrhagic or icteric manifestations. While the case fatality rate varies by outbreak, it typically remains below 1%, with the highest mortality linked to severe clinical forms[8-10].
Over the last two decades, numerous outbreaks involving severe human infections and significant mortality have been documented.
Niger (2016): By October 11, the Ministry of Health reported 105 suspected cases in the Tahoua region, including 28 deaths.
Mauritania (2012): Between September 16 and November 13, six regions reported 36 confirmed cases with 18 deaths.
South Africa (2010): From February to July, a total of 237 laboratory-confirmed cases and 26 deaths were recorded across nine provinces.
Madagascar (2008-2009): Between December 2008 and May 2009, authorities reported 236 suspected cases and 7 deaths. A separate cluster from January to June 2008 recorded 476 suspected cases and 19 fatalities in four provinces.
Sudan (2007-2008): An outbreak beginning in late October 2007 led to 738 reported cases and 230 deaths by January 2008.
Kenya, Somalia, and Tanzania (2006-2007): (1) Kenya: 684 cases with 234 deaths; (2) Somalia: 114 cases with 51 deaths; and (3) Tanzania: 264 cases with 109 deaths.
Egypt (2003): A total of 148 cases and 27 deaths were reported.
Saudi Arabia and Yemen (2000): (1) Saudi Arabia: 516 confirmed cases, 87 deaths; and (2) Yemen: 1087 suspected cases, 121 deaths.
These outbreaks demonstrate RVF’s potential to cause widespread epidemics with considerable public health and economic consequences. Regions characterized by favorable ecological conditions for mosquito breeding and close human-animal interactions are particularly vulnerable. Therefore, robust surveillance systems, early warning mechanisms, and vector control interventions are critical to mitigating future outbreaks (Figure 1).
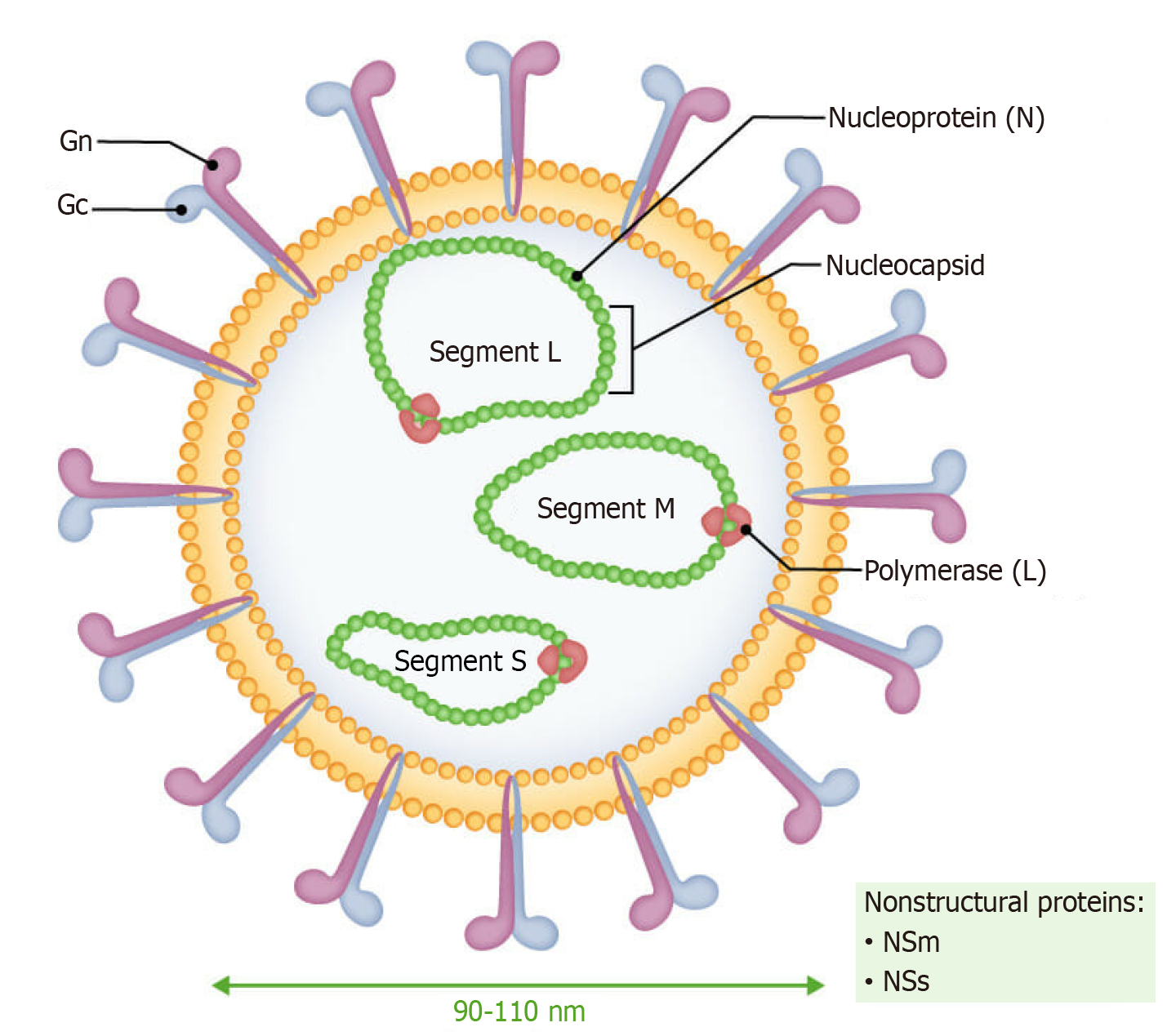
RVFV is a notable pathogen within the Bunyaviridae family, recognized for its genetic diversity. It is classified under the Phlebovirus genus, which also contains several clinically important viruses such as Punta Toro virus, Sandfly fever virus, and severe fever with thrombocytopenia syndrome virus. Although the genus name is associated with phlebotomine sandflies as vectors, RVFV differs in its mode of transmission, being spread primarily by mosquitoes, especially species of the Aedes and Culex genera[11].
Like other members of the Bunyaviridae family, RVFV possesses a segmented, single-stranded, negative-sense RNA genome. The genome is divided into three distinct segments: (1) Large (L); (2) Medium (M); and (3) Small (S).
L segment: This segment codes for the RNA-dependent RNA polymerase, an enzyme required for both transcription and replication of the viral RNA.
M segment: This segment is responsible for producing the envelope glycoproteins Gn and Gc, which play a vital role in viral entry by enabling host cell binding and membrane fusion. Additionally, it encodes the nonstructural protein m, with all products synthesized from the negative-sense RNA strand.
S segment: It exhibits an ambisense coding mechanism, producing the nucleoprotein from the negative strand and the nonstructural (NSs) protein from the positive strand. NSs is particularly important for suppressing host interferon responses and facilitating viral immune evasion[12,13].
The viral RNA segments are encapsidated by the N protein to form ribonucleoprotein complexes. Complementary terminal sequences enable circularization of each RNA segment. Viral entry into host cells is initiated by receptor-mediated endocytosis, followed by fusion within endosomes triggered by low pH. Replication, transcription, and protein synthesis occur exclusively in the cytoplasm. A distinct feature of RVFV is that virion assembly and budding occur at the Golgi apparatus[14].
Genomic studies of RVFV isolates spanning from 1944 to 2007 have demonstrated relatively low genetic variation (approximately 5%), suggesting overall genomic stability. Although seven genetic lineages have been identified, they do not exhibit strict geographic clustering, implying frequent regional movement and genetic intermixing. While reassortment between genome segments has been documented, no confirmed cases of recombination among RVFV strains have been reported to date (Figures 2 and 3)[15,16].
RVF presents with a range of clinical signs that vary in severity depending on the age and species of the affected animals. One of the most prominent features in livestock is the sudden onset of abortion storms, particularly among pregnant females. Young lambs under five to six days old are especially vulnerable, often experiencing mortality rates approaching 100%. In adult animals, common symptoms include high fever, lymph node enlargement, and nasal and ocular discharges. Affected animals may also exhibit profuse, foul-smelling diarrhea, which is frequently hemorrhagic, along with vomiting, abdominal pain (colic), and signs of severe weakness or prostration. Additional findings may include dysgalactia (reduced milk production), jaundice, and in some cases, neurological symptoms. The typical epizootic duration ranges from 8 weeks to 16 weeks, often coinciding with seasonal environmental conditions favorable for mosquito vector proliferation[17].
The incubation period for RVF in humans typically ranges from 2 days to 6 days following exposure to the virus. In many cases, infection results in either asymptomatic presentation or a mild, self-limiting illness. Individuals with the mild form commonly experience sudden-onset fever, accompanied by flu-like symptoms such as muscle aches, joint pain, and headache. In some patients, additional signs like neck stiffness, photophobia (sensitivity to light), loss of appetite, and vomiting may occur, potentially leading to an initial misdiagnosis as meningitis. The symptomatic phase usually resolves within 4-7 days, and most individuals recover without the need for specialized medical intervention[18].
Although the majority of RVF cases in humans are mild, a small proportion of patients may progress to severe disease, presenting in one or more of three clinical syndromes. The first is ocular involvement, affecting approximately 0.5%-2% of patients, which may lead to visual disturbances or, in some cases, permanent vision loss. The second form is meningoencephalitis, a serious neurological condition that occurs in fewer than 1% of cases, characterized by symptoms such as confusion, seizures, and coma. The third and most life-threatening presentation is haemorrhagic fever, also seen in less than 1% of infected individuals, and is marked by bleeding disorders, shock, and multi-organ failure. These severe manifestations require prompt medical attention and carry a higher risk of mortality[19].
In animals, particularly in domestic ruminants such as sheep, goats, and cattle, RVF presents with a range of clinical symptoms that vary in severity depending on age and species. Common signs include the onset of fever, loss of appetite, vomiting, and diarrhea, which may progress to dehydration and weakness. In more severe cases, the disease can lead to systemic bleeding, particularly in sheep and goats, often accompanied by sudden death. These hemorrhagic manifestations are especially pronounced in younger or immunocompromised animals. High mortality rates, particularly in newborn lambs and kids, are a hallmark of RVF outbreaks. The disease not only affects animal health but also poses a significant threat to livestock-dependent communities due to sudden and extensive losses (Table 2)[20-22].
| Parameter | Mild form | Severe form |
| Onset | 2-6 days after infection | Usually develops after initial mild symptoms |
| Symptoms | Fever, muscle and joint pain, headache, vomiting, photophobia | Ocular disease, meningoencephalitis, haemorrhagic fever |
| Prevalence | Majority of infected individuals | Occurs in a small percentage (< 2%) of cases |
| Ocular involvement | Rare | Seen in 0.5%-2% of cases; may cause blurred vision or blindness |
| Neurological involvement | Absent | Occurs in < 1% of cases; symptoms include seizures, confusion, and coma |
| Bleeding disorders | Not observed | Occurs in < 1% of cases; may include internal bleeding and multi-organ failure |
| Duration | 4-7 days | Variable; prolonged and potentially life-threatening |
| Prognosis | Full recovery | May lead to permanent damage or death |
Ocular (eye) form (0.5%-2% of patients): In some patients, mild RVF symptoms are followed by retinal lesions that typically develop 1-3 weeks after the initial illness. These eye complications often lead to blurred or reduced vision. Most cases improve within 10-12 weeks, but if the lesions involve the macula, about half of the affected individuals may suffer from permanent vision loss.
Meningoencephalitis form (less than 1% of patients): This neurological form usually begins 1-4 weeks after the initial symptoms. Patients may experience severe headaches, memory loss, hallucinations, confusion, disorientation, dizziness, seizures, lethargy, and even coma. Although the death rate is generally low, long-term neurological complications and disabilities are frequently observed, and in some cases, these may appear months later.
Hemorrhagic fever form (less than 1% of patients): This is the most severe form, with symptoms appearing 2-4 days after illness onset. It starts with acute liver damage, followed by signs of internal and external bleeding, such as blood in vomit or stool, skin rashes, gum or nosebleeds, heavy menstrual bleeding, and bleeding at injection sites. The fatality rate is extremely high, estimated at around 50%, with death often occurring within 3-6 days of symptom onset.
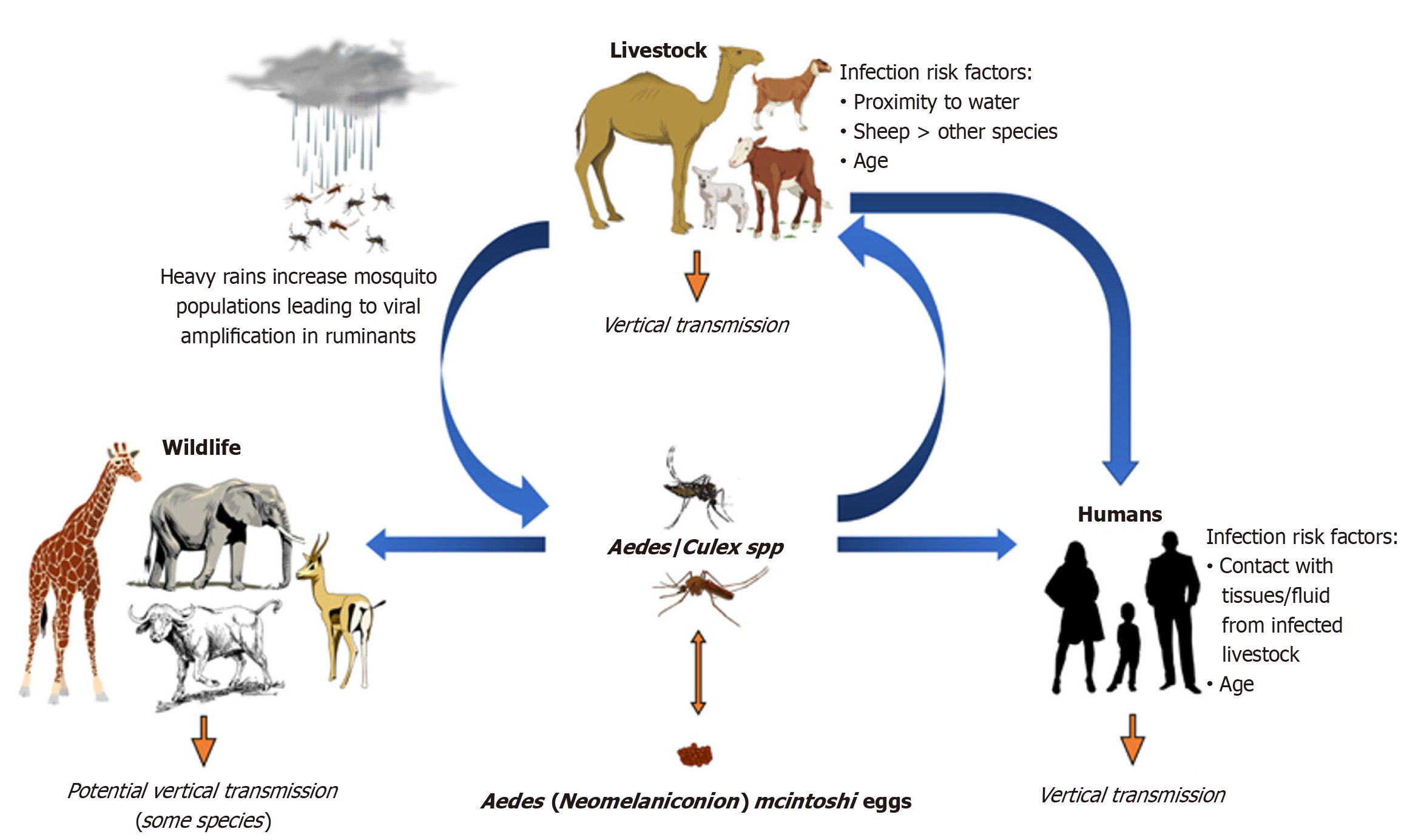
RVFV spreads via multiple routes, impacting both humans and animals. In humans, the most common transmission pathway is direct exposure to blood, tissues, body fluids, or aborted material from infected livestock, particularly during slaughter, animal handling, or veterinary practices. Aerosol transmission is also possible, placing individuals in farming, animal care, and veterinary fields at higher occupational risk. Additionally, vertical transmission of the virus – from mother to offspring – has been documented in humans, animals, and even mosquito vectors[23-25].
Drinking raw or unpasteurized milk has been identified as a possible source of RVFV transmission, particularly during outbreaks, with studies indicating that infected milk may play a role in human infections in endemic areas. Mosquito-borne transmission remains the most established route, primarily through bites from infected Aedes species – especially those in the Aedimorphus and Neomelaniconion sub-genera, known as floodwater-breeding mosquitoes – as well as Culex species, which act as important vectors in maintaining and spreading the virus.
At present, there is no definitive proof of human-to-human transmission of RVFV, although occasional instances of vertical transmission from mother to child have been documented. The exact wildlife reservoirs of the virus remain uncertain; however, African buffaloes (Syncerus caffer), other wild ruminants, rodents, and bats are being investigated for their potential role in the virus’s ecology and spread. While their involvement has not been confirmed, it cannot be excluded, highlighting the need for ongoing monitoring to better understand how the virus persists in nature.
Humans face a high risk of RVF infection through exposure to fetal tissues and body fluids of infected animals, especially after livestock abortions, when contamination around the udder and perineal region is significant. Laboratory workers handling blood or tissues from suspected RVF-infected carcasses have been among the most commonly affected due to accidental exposure. Veterinary professionals also face considerable danger during post-mortem examinations of infected animals. Consumption of milk is generally not regarded as a transmission risk. However, the potential role of blood meal, bone meal, or offal products in spreading the virus has not yet been clearly established.
Effective diagnosis of RVF is essential for controlling outbreaks, especially in endemic areas where human infections often follow animal outbreaks. Surveillance of livestock populations and timely case reporting are critical for early detection. In humans, diagnosis during the acute phase can be confirmed using several laboratory techniques, including reverse transcription polymerase chain reaction (RT-PCR), antigen detection enzyme-linked immunosorbent assay (ELISA), or virus isolation from blood samples. These methods are most effective during the viremic phase. Once viremia subsides, immunoglobulin M (IgM) antibodies can be detected transiently, while immunoglobulin G (IgG) antibodies typically persist for years, aiding in retrospective serosurveillance[26,27].
ELISAs are widely used for detecting IgG and IgM antibodies and are suitable for diagnosing both recent and past infections. Advanced dual-target ELISAs can differentiate between natural infection and vaccination by targeting both the nucleoprotein and NSs proteins, as antibodies to NSs are absent in animals vaccinated with attenuated strains.
Virus isolation may be performed using whole blood, serum, or organ tissues (e.g., brain, liver, spleen) obtained from recently deceased or aborted animals. The virus may also be present in milk, though diagnostic assays are not specifically validated for milk samples. Virus isolation involves inoculation of susceptible cell lines such as Vero (African green monkey kidney), BHK-21 (baby hamster kidney), or AP61 (mosquito cells), where cytopathic effects typically appear within five days, confirmed by immunostaining.
Molecular techniques such as nested RT-PCR, quantitative real-time PCR, loop-mediated isothermal amplification, multiplex PCR, and recombinase polymerase amplification have been developed for high-sensitivity detection. These methods are not only used in clinical diagnosis but also in mosquito surveillance programs. Although newer tools like next-generation sequencing, TaqMan array cards, and colorimetry-based assays have been introduced, their high cost and complexity limit their use to well-equipped laboratories with trained personnel.
In addition, point-of-care diagnostics such as lateral flow immuno-chromatographic strip tests have been developed to detect RVFV nucleoprotein antigens. These rapid tests are especially useful in field settings, facilitating early diagnosis and improved outbreak management in resource-limited areas.
Diagnosing RVF can be challenging, as its early symptoms closely resemble those of other infectious diseases such as malaria, typhoid fever, shigellosis, yellow fever, and various viral hemorrhagic fevers. Therefore, confirmation relies on laboratory-based methods rather than clinical signs alone.
The main diagnostic techniques include: (1) RT-PCR: For direct detection of viral genetic material; (2) IgG and IgM antibody detection by ELISA: To identify the immune response against RVF infection; and (3) Virus isolation using cell culture: For definitive confirmation, though it requires specialized facilities.
Processing and handling of patient specimens present a high biohazard risk. Laboratory testing on non-inactivated samples must be carried out under maximum biosafety containment conditions. To ensure safe national and international transport, all biological specimens should be packaged and shipped using the triple packaging system, in accordance with biosafety regulations.
Given the nonspecific nature of RVF symptoms, laboratory confirmation is essential to differentiate it from other febrile illnesses and to establish an accurate diagnosis.
During the 2016 RVF outbreak in Niger, the World Health Organization (WHO) mobilized a multisectoral national rapid response team that included experts from the Ministry of Health, veterinary departments, and the Centre de Recherche Médicale et Sanitaire. The team initiated field investigations on August 31, 2016 to assess the magnitude of the outbreak and implement effective control and containment strategies[28].
The WHO Country Office in Niger played a central role in supporting the national response by providing technical and financial assistance. Key activities included strengthening disease surveillance, facilitating outbreak investigations, offering guidance on case definitions and management protocols, coordinating the transport of diagnostic samples, and enhancing risk communication strategies.
Acknowledging the zoonotic characteristics of RVF, the WHO partnered with the Food and Agriculture Organization and the World Organisation for Animal Health (WOAH). This collaboration aimed to coordinate human and animal health initiatives and strengthen the nation’s capacity to respond effectively to the outbreak.
Additionally, the WHO utilized its partnerships within the Global Outbreak Alert and Response Network to ensure effective coordination of international support efforts. Humanitarian organizations such as the International Federation of Red Cross and Red Crescent Societies and United Nations Children's Fund contributed by providing logistical support, engaging communities, and promoting public health awareness to strengthen the outbreak response.
This coordinated approach underscores the importance of multi-agency collaboration in managing zoonotic outbreaks and minimizing their impact on public health and livestock.
Vaccination is considered the most effective approach to control RVF. Since outbreaks are often linked to specific ecological conditions, early detection of high-risk periods allows authorities to plan targeted vaccination campaigns[29].
The modified live Smithburn neurotropic strain vaccine is widely used due to its strong immune response. However, it has limitations, as it may cause fetal damage and abortions in susceptible pregnant sheep, with up to 30% of such animals being affected.
Inactivated vaccines have also been developed, though they generally provide weaker immunity. One example is the inactivated vaccine produced by Onderstepoort Biological Products (South Africa), which is derived from a bovine virulent RVF isolate adapted in cell culture. It is later inactivated and formulated with aluminium hydroxide gel as an adjuvant. This vaccine is safer for pregnant ewes and is also recommended for cattle, since it enhances colostral immunity in calves. To maintain protection, animals require a booster dose within three to six months after the initial vaccination, followed by annual boosters.
Routine vaccination is advised when animals are not pregnant, as this avoids reproductive complications. While the Smithburn neurotropic strain vaccine is safe and effective in cattle, vaccination during an active outbreak is discouraged. Administering vaccines at that stage carries the risk of spreading the virus through contaminated needles, in addition to being too late to prevent disease spread.
In addition to existing live and inactivated vaccines, research is focused on creating modified live virus and molecular-based vaccines. These candidates have shown promise in experimental models but are not yet available for field application[30].
MP-12 strain: This strain was created through mutagenic modifications of the ZH548 RVFV strain. Along with clone-13, which was derived from a mild human infection in the Central African Republic, it has demonstrated strong immunogenicity in mice. Both strains elicit detectable antibody responses, confirmed by ELISA and plaque reduction neutralization tests.
For clone-13, the protective dose (PD50) was 100.1 TCID50, while for MP-12, it was 103.
The S segment of the virus controls virulence. In clone-13, deletion of the NSs protein gene results in attenuation, as NSs normally acts as an antagonist to type I interferon.
Experiments show that strains with intact NSs fail to stimulate interferon production, while NSs-deficient strains like clone-13 trigger high interferon levels.
Clone-13 is considered safer due to its low risk of reverting to a virulent form. However, its L and M segments do not carry markers for attenuation. This raises concerns about reassortment with circulating virulent strains, which could theoretically lead to the emergence of new pathogenic viruses.
R566 strain: R566 is a reassortant virus developed from clone-13 and MP-12 in Vero cells. It carries the S segment of clone-13 (attenuated due to NSs deletion) and the L and M segments of MP-12, which have multiple point mutations.
These mutations introduce both attenuation and thermosensitivity, making the strain less likely to revert to virulence.
Laboratory studies show that R566 is safe and provides strong protection against RVF in mice, suggesting potential for future vaccine development.
One important strategy to limit RVF transmission is the larvicidal treatment of mosquito breeding sites[31]. Two effective options include: (1) Hormonal inhibitors such as Methoprene; and (2) Larvicidal toxins produced by Bacillus thuringiensis.
Both methods have shown good results and are widely available commercially. However, their use can be challenging in regions with extensive flooding, where mosquito breeding habitats are widespread and difficult to manage.
The application of ultra-low-volume insecticides using vehicles or aerial spraying has been attempted, but such measures have shown limited effectiveness in reducing RVF transmission rates or significantly targeting the main adult mosquito species responsible for spreading the virus.
Restricting animal movement within an affected country has little to no impact on the progression of an outbreak. However, it plays an important role in the international trade of livestock. When animals from enzootic or epizootic regions are moved for trade, there is a risk of introducing the virus into previously unaffected countries[32].
If viraemic animals (infected but still in the incubation phase) are transported to regions where competent mosquito vectors are present, the likelihood of RVF being established in the new area becomes high.
Therefore, to minimize this risk, it is recommended that all livestock exports be suspended during RVF epizootic periods.
Preventing RVF outbreaks in animals relies mainly on sustained vaccination programs. Both live attenuated and inactivated vaccines are available for veterinary use.
Vaccination should be carried out before an outbreak to prevent epizootics. During an active outbreak, vaccination is discouraged as the use of multi-dose vials and re-use of needles may worsen virus transmission[31-33].
Movement restrictions or livestock trade bans are recommended to prevent the spread of infection from affected to unaffected areas.
Since animal cases typically appear before human infections, establishing an active animal health surveillance system is crucial to provide early warnings for veterinary and public health authorities.
Awareness campaigns play a key role in reducing human infections by highlighting risk factors and protective measures.
Risk reduction strategies include: (1) Practicing good hygiene and using protective equipment (gloves, masks, protective clothing) when handling sick animals, their tissues, or carcasses; (2) Avoiding consumption of raw milk, undercooked meat, or fresh animal blood in affected regions; (3) All animal products should be properly cooked, preventing mosquito bites through insecticide-treated bed nets, personal repellents, wearing light-colored protective clothing, and avoiding outdoor exposure during peak biting hours; and (4) Implementing vector control measures such as larviciding mosquito breeding sites where feasible. However, widespread flooding often makes large-scale larviciding impractical.
While human-to-human transmission of RVF has not been documented, healthcare workers remain at risk when in contact with infected blood or tissues.
It is essential to adhere to standard infection control procedures when managing specimens from patients suspected or confirmed to have RVF.
RVF outbreaks are closely associated with climatic factors, especially excessive rainfall and flooding.
In East Africa, these outbreaks frequently coincide with the El Niño-Southern Oscillation phenomenon.
Predictive models that utilize satellite imagery and climate data are useful for identifying high-risk periods, enabling early detection of cases in animals and allowing authorities to implement preventive interventions before large-scale epidemics develop.
In most human cases, RVF is mild and short-lived, requiring no specific medical intervention. However, in patients with severe illness, supportive care remains the mainstay of treatment, focusing on fluid management and relief of clinical symptoms.
During outbreaks, the highest risk of infection arises from direct or indirect contact with infected animals or their body fluids. Preventive measures include practicing safe animal husbandry, proper hygiene, and the avoidance of raw or unprocessed animal products such as blood, milk, or meat[34].
The severity of RVF in animals depends largely on their species and age. Common clinical signs include fever, loss of appetite, reduced movement, abortions, and high fatality rates among young livestock, especially newborn sheep and goats. Owing to its serious economic and health implications, RVF is categorized as a WOAH-notifiable disease and must be reported in accordance with the WOAH Terrestrial Animal Health Code (chapters 1.1 and 8.15). Because the virus tends to reappear after extended periods of inactivity, it is also considered a re-emerging infectious disease[35].
The most effective preventive approach is the vaccination of susceptible livestock, which indirectly reduces the risk of human infection. Without vaccination, human exposure is more likely, especially during veterinary procedures, slaughtering, necropsy, or butchering of infected animals.
No specific antiviral treatment currently exists for RVF infection[36]. Most cases are mild and self-limiting, requiring only symptomatic care such as rest, fluids, and fever management.
For severe cases, especially those with hemorrhagic fever, encephalitis, or ocular complications, intensive supportive therapy is crucial. This includes: (1) Careful fluid and electrolyte balance; (2) Monitoring and management of liver and kidney function; (3) Treatment of bleeding complications; (4) Neurological monitoring in meningoencephalitis cases; and (5) Early medical intervention significantly improves patient outcomes.
An inactivated vaccine has been developed for human use but is not licensed for general public use. It has mainly been used experimentally for veterinarians, laboratory staff, and high-risk workers.
Several candidate vaccines are under research, including live attenuated and recombinant vaccines, but none are yet commercially available for widespread immunization.
Controlling RVF in animals is key to preventing human infections since outbreaks in livestock usually precede human cases[37].
Available vaccines include: (1) Live attenuated vaccines (e.g., Smithburn strain) – provide strong protection but may cause abortions or birth defects if used in pregnant animals; and (2) Inactivated vaccines – safer for pregnant animals but require multiple doses and regular boosters to maintain immunity.
Preventive vaccination programs should be carried out before outbreaks occur. Vaccination during active outbreaks is not recommended because of the risk of spreading infection through shared needles and equipment.
In the United States, an inactivated RVF vaccine was once produced to protect military and laboratory personnel, as the virus is considered a potential biological threat. However, manufacturing of this vaccine was discontinued due to logistical challenges[38].
Since the primary transmission cycle begins in domestic ruminants, controlling infection in animals is the most effective strategy to prevent human disease. Vaccination of livestock is therefore prioritized. Both live attenuated and inactivated vaccines are available: (1) Smithburn vaccine: A modified live virus vaccine effective in sheep, goats, and cattle, offering protection against abortion induced by wild strains. Despite its immunogenicity, it can cause adverse effects, including flu-like symptoms in humans and reproductive issues (abortions and congenital malformations) in ruminants; (2) Inactivated vaccines: Safer alternatives but provide weaker immunity, require multiple doses, and are more costly to produce; and (3) Candidate vaccines: Newer options such as clone-13 are currently under evaluation for use in ruminants.
Additional non-vaccine preventive strategies include[39]: (1) Prohibiting slaughtering and butchering of animals during outbreaks; (2) Promoting the use of mosquito control methods such as repellents, insecticide-treated bed nets, and protective clothing; (3) Advising sleeping in screened or air-conditioned rooms to reduce mosquito exposure; (4) Implementing awareness campaigns for high-risk groups such as farmers, veterinarians, butchers, and slaughterhouse workers; and (5) Ensuring safe disposal of infected carcasses to prevent further spread.
The growing impact of international trade, environmental changes, climate warming, and extreme weather events may increase the risk of RVF spreading beyond its traditional tropical zones into subtropical and even temperate regions.
To improve preparedness, there is a need for field-based evaluations of currently available diagnostic methods and animal vaccines, focusing on their efficacy, practicality, and cost-effectiveness in real-world conditions.
In addition, the potential use of vaccines for high-risk human populations, such as farmers, veterinarians, and other individuals with frequent animal contact during outbreaks, should be carefully assessed. These assessments must consider epidemiological evidence and sociological factors to ensure appropriate and acceptable vaccination strategies[40].
RVF was first identified during an outbreak in 1930, and despite more than eight decades of research, the disease continues to cause recurrent epidemics in several African nations and the Arabian Peninsula. Considerable progress has been made in understanding the clinical manifestations, pathology, and virology of RVF; however, gaps remain in achieving complete control.
The development of safe, cost-effective, and highly immunogenic vaccines for both humans and animals remain one of the most effective strategies to prevent future outbreaks in endemic regions. To advance RVF prevention and management, key scientific questions must be addressed, including: (1) Identifying the underlying mechanisms responsible for hemorrhagic fever in RVF; (2) Understanding the pathways of viral entry into the brain; (3) Explaining why some patients experience prolonged illness despite the presence of neutralizing antibodies; and (4) Assessing the role of vaccination in preventing disease after aerosol exposure.
Addressing these research gaps will significantly improve knowledge of RVF pathogenesis and support the development of effective therapeutic agents and preventive tools against the virus.
| 1. | Kwaśnik M, Rożek W, Rola J. Rift Valley Fever - a Growing Threat To Humans and Animals. J Vet Res. 2021;65:7-14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 51] [Article Influence: 10.2] [Reference Citation Analysis (1)] |
| 2. | Peng K, Lozach P. Rift Valley Fever Virus: A New Avenue of Research on the Biological Functions of amyloids? Future Virol. 2021;16:677-689. [RCA] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 3. | Balenghien T, Cardinale E, Chevalier V, Elissa N, Failloux AB, Jean Jose Nipomichene TN, Nicolas G, Rakotoharinome VM, Roger M, Zumbo B. Towards a better understanding of Rift Valley fever epidemiology in the south-west of the Indian Ocean. Vet Res. 2013;44:78. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 52] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 4. | Balkema-Buschmann A, Rissmann M, Kley N, Ulrich R, Eiden M, Groschup MH. Productive Propagation of Rift Valley Fever Phlebovirus Vaccine Strain MP-12 in Rousettus aegyptiacus Fruit Bats. Viruses. 2018;10:681. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 5. | Brinkmann A, Ergünay K, Radonić A, Kocak Tufan Z, Domingo C, Nitsche A. Development and preliminary evaluation of a multiplexed amplification and next generation sequencing method for viral hemorrhagic fever diagnostics. PLoS Negl Trop Dis. 2017;11:e0006075. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 3.1] [Reference Citation Analysis (1)] |
| 6. | Birnberg L, Talavera S, Aranda C, Núñez AI, Napp S, Busquets N. Field-captured Aedes vexans (Meigen, 1830) is a competent vector for Rift Valley fever phlebovirus in Europe. Parasit Vectors. 2019;12:484. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 24] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 7. | Bosworth A, Ghabbari T, Dowall S, Varghese A, Fares W, Hewson R, Zhioua E, Chakroun M, Tiouiri H, Ben Jemaa M, Znazen A, Letaief A. Serologic evidence of exposure to Rift Valley fever virus detected in Tunisia. New Microbes New Infect. 2016;9:1-7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 21] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 8. | Faburay B, Gaudreault NN, Liu Q, Davis AS, Shivanna V, Sunwoo SY, Lang Y, Morozov I, Ruder M, Drolet B, Scott McVey D, Ma W, Wilson W, Richt JA. Development of a sheep challenge model for Rift Valley fever. Virology. 2016;489:128-140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 33] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 9. | Faburay B, LaBeaud AD, McVey DS, Wilson WC, Richt JA. Current Status of Rift Valley Fever Vaccine Development. Vaccines (Basel). 2017;5:29. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 96] [Article Influence: 10.7] [Reference Citation Analysis (1)] |
| 10. | Fagre AC, Kading RC. Can Bats Serve as Reservoirs for Arboviruses? Viruses. 2019;11:215. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 58] [Article Influence: 8.3] [Reference Citation Analysis (1)] |
| 11. | Fakour S, Naserabadi S, Ahmadi E. The first positive serological study on rift valley fever in ruminants of Iran. J Vector Borne Dis. 2017;54:348-352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 21] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 12. | Fawzy M, Helmy YA. The One Health Approach is Necessary for the Control of Rift Valley Fever Infections in Egypt: A Comprehensive Review. Viruses. 2019;11:139. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 43] [Article Influence: 6.1] [Reference Citation Analysis (1)] |
| 13. | Grossi-Soyster EN, Lee J, King CH, LaBeaud AD. The influence of raw milk exposures on Rift Valley fever virus transmission. PLoS Negl Trop Dis. 2019;13:e0007258. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 32] [Article Influence: 4.6] [Reference Citation Analysis (1)] |
| 14. | Gür S, Kale M, Erol N, Yapici O, Mamak N, Yavru S. The first serological evidence for Rift Valley fever infection in the camel, goitered gazelle and Anatolian water buffaloes in Turkey. Trop Anim Health Prod. 2017;49:1531-1535. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 29] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 15. | Ahmed Zehairy A, Sartaj Sohrab S, Al-Judaibi AA, Ibraheem Azhar E. Rift Valley Fever Virus: An update on current status and future prospects. Rev Argent Microbiol. 2025;S0325-7541(25)00095. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 16. | Kumalija MS, Chilongola JO, Budodo RM, Horumpende PG, Mkumbaye SI, Vianney JM, Mwakapuja RS, Mmbaga BT. Detection of Rift Valley Fever virus inter-epidemic activity in Kilimanjaro Region, North Eastern Tanzania. Glob Health Action. 2021;14:1957554. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 17. | Hartman A. Rift Valley Fever. Clin Lab Med. 2017;37:285-301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 99] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 18. | Linthicum KJ, Britch SC, Anyamba A. Rift Valley Fever: An Emerging Mosquito-Borne Disease. Annu Rev Entomol. 2016;61:395-415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 231] [Article Influence: 23.1] [Reference Citation Analysis (1)] |
| 19. | Kainulainen M, Lau S, Samuel CE, Hornung V, Weber F. NSs Virulence Factor of Rift Valley Fever Virus Engages the F-Box Proteins FBXW11 and β-TRCP1 To Degrade the Antiviral Protein Kinase PKR. J Virol. 2016;90:6140-6147. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 50] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 20. | Liu J, Ochieng C, Wiersma S, Ströher U, Towner JS, Whitmer S, Nichol ST, Moore CC, Kersh GJ, Kato C, Sexton C, Petersen J, Massung R, Hercik C, Crump JA, Kibiki G, Maro A, Mujaga B, Gratz J, Jacob ST, Banura P, Scheld WM, Juma B, Onyango CO, Montgomery JM, Houpt E, Fields B. Development of a TaqMan Array Card for Acute-Febrile-Illness Outbreak Investigation and Surveillance of Emerging Pathogens, Including Ebola Virus. J Clin Microbiol. 2016;54:49-58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 106] [Article Influence: 9.6] [Reference Citation Analysis (1)] |
| 21. | Lumley S, Horton DL, Hernandez-Triana LLM, Johnson N, Fooks AR, Hewson R. Rift Valley fever virus: strategies for maintenance, survival and vertical transmission in mosquitoes. J Gen Virol. 2017;98:875-887. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 83] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 22. | Alomar AA, Campbell LP, Mathias DK, Burkett-Cadena ND. Rift Valley Fever Virus: A Zoonotic Vector-Borne Pathogen Affecting Human and Livestock Health. EDIS. 2023;. [DOI] [Full Text] |
| 23. | Mansfield KL, Banyard AC, McElhinney L, Johnson N, Horton DL, Hernández-Triana LM, Fooks AR. Rift Valley fever virus: A review of diagnosis and vaccination, and implications for emergence in Europe. Vaccine. 2015;33:5520-5531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 117] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 24. | Martinet JP, Ferté H, Failloux AB, Schaffner F, Depaquit J. Mosquitoes of North-Western Europe as Potential Vectors of Arboviruses: A Review. Viruses. 2019;11:1059. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 77] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 25. | Sang R, Arum S, Chepkorir E, Mosomtai G, Tigoi C, Sigei F, Lwande OW, Landmann T, Affognon H, Ahlm C, Evander M. Distribution and abundance of key vectors of Rift Valley fever and other arboviruses in two ecologically distinct counties in Kenya. PLoS Negl Trop Dis. 2017;11:e0005341. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 47] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 26. | Vloet RPM, Vogels CBF, Koenraadt CJM, Pijlman GP, Eiden M, Gonzales JL, van Keulen LJM, Wichgers Schreur PJ, Kortekaas J. Transmission of Rift Valley fever virus from European-breed lambs to Culex pipiens mosquitoes. PLoS Negl Trop Dis. 2017;11:e0006145. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 41] [Article Influence: 4.6] [Reference Citation Analysis (1)] |
| 27. | Wilson WC, Davis AS, Gaudreault NN, Faburay B, Trujillo JD, Shivanna V, Sunwoo SY, Balogh A, Endalew A, Ma W, Drolet BS, Ruder MG, Morozov I, McVey DS, Richt JA. Experimental Infection of Calves by Two Genetically-Distinct Strains of Rift Valley Fever Virus. Viruses. 2016;8:145. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 29] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 28. | Wilson WC, Kim IJ, Trujillo JD, Sunwoo SY, Noronha LE, Urbaniak K, McVey DS, Drolet BS, Morozov I, Faburay B, Schirtzinger EE, Koopman T, Indran SV, Balaraman V, Richt JA. Susceptibility of White-Tailed Deer to Rift Valley Fever Virus. Emerg Infect Dis. 2018;24:1717-1719. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 35] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 29. | Wright D, Kortekaas J, Bowden TA, Warimwe GM. Rift Valley fever: biology and epidemiology. J Gen Virol. 2019;100:1187-1199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 140] [Article Influence: 20.0] [Reference Citation Analysis (1)] |
| 30. | Youssouf H, Subiros M, Dennetiere G, Collet L, Dommergues L, Pauvert A, Rabarison P, Vauloup-Fellous C, Le Godais G, Jaffar-Bandjee MC, Jean M, Paty MC, Noel H, Oliver S, Filleul L, Larsen C. Rift Valley Fever Outbreak, Mayotte, France, 2018-2019. Emerg Infect Dis. 2020;26:769-772. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 31. | Zaher MR, Ahmed HA, Hamada KEZ, Tammam RH. Colorimetric Detection of Unamplified Rift Valley Fever Virus Genetic Material Using Unmodified Gold Nanoparticles. Appl Biochem Biotechnol. 2018;184:898-908. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 12] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 32. | Cohen J. Unfilled Vials. Science. 2016;351:16-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 37] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 33. | Tinto B, Quellec J, Cêtre-Sossah C, Dicko A, Salinas S, Simonin Y. Rift Valley fever in West Africa: A zoonotic disease with multiple socio-economic consequences. One Health. 2023;17:100583. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 26] [Reference Citation Analysis (1)] |
| 34. | Venter M. Assessing the zoonotic potential of arboviruses of African origin. Curr Opin Virol. 2018;28:74-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 36] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 35. | Grossi-Soyster EN, Banda T, Teng CY, Muchiri EM, Mungai PL, Mutuku FM, Gildengorin G, Kitron U, King CH, Desiree Labeaud A. Rift Valley Fever Seroprevalence in Coastal Kenya. Am J Trop Med Hyg. 2017;97:115-120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 16] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 36. | Njenga MK, Njagi L, Thumbi SM, Kahariri S, Githinji J, Omondi E, Baden A, Murithi M, Paweska J, Ithondeka PM, Ngeiywa KJ, Dungu B, Donadeu M, Munyua PM. Randomized controlled field trial to assess the immunogenicity and safety of rift valley fever clone 13 vaccine in livestock. PLoS Negl Trop Dis. 2015;9:e0003550. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 37. | Matsiela MS, Naicker L, Khoza T, Mokoena N. Safety and immunogenicity of inactivated Rift Valley Fever Smithburn viral vaccine in sheep. Virol J. 2023;20:221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 38. | LaBeaud AD, Pfeil S, Muiruri S, Dahir S, Sutherland LJ, Traylor Z, Gildengorin G, Muchiri EM, Morrill J, Peters CJ, Hise AG, Kazura JW, King CH. Factors associated with severe human Rift Valley fever in Sangailu, Garissa County, Kenya. PLoS Negl Trop Dis. 2015;9:e0003548. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 58] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 39. | Anywaine Z, Lule SA, Hansen C, Warimwe G, Elliott A. Clinical manifestations of Rift Valley fever in humans: Systematic review and meta-analysis. PLoS Negl Trop Dis. 2022;16:e0010233. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 45] [Article Influence: 11.3] [Reference Citation Analysis (1)] |
| 40. | Nanyingi MO, Munyua P, Kiama SG, Muchemi GM, Thumbi SM, Bitek AO, Bett B, Muriithi RM, Njenga MK. A systematic review of Rift Valley Fever epidemiology 1931-2014. Infect Ecol Epidemiol. 2015;5:28024. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 103] [Cited by in RCA: 139] [Article Influence: 12.6] [Reference Citation Analysis (1)] |