Published online Mar 25, 2026. doi: 10.5501/wjv.v15.i1.116492
Revised: December 8, 2025
Accepted: January 13, 2026
Published online: March 25, 2026
Processing time: 121 Days and 0.3 Hours
This opinion challenges the conventional view that coronavirus disease 2019 behaves as a uniformly winter-dominant respiratory infection. Analysis of multi-year surveillance data across hemispheres reveals that severe acute respiratory syndrome coronavirus-2 exhibits seasonal divergence, with consistent summer surges in tropical regions, such as India, and winter peaks in temperate climates. We propose that this pattern arises primarily from human (host) behavioural re
Core Tip: Coronavirus disease 2019 seasonality is not uniformly winter-driven but shaped by behavioural adaptation to climate and host. Severe acute respiratory syndrome coronavirus-2 exhibits a seasonal pattern, being predominantly summer-predominant in tropical regions and winter-predominant in temperate ones, due to its virological resilience and broad host adaptability. Recognizing this behaviourally modulated seasonality is essential for designing climate- and region-specific surveillance, vaccination timing, and ventilation strategies.
- Citation: Panda PK, Garg R. Rethinking COVID-19 seasonality: A summer respiratory virus in the tropics, contrast to influenza. World J Virol 2026; 15(1): 116492
- URL: https://www.wjgnet.com/2220-3249/full/v15/i1/116492.htm
- DOI: https://dx.doi.org/10.5501/wjv.v15.i1.116492
Seasonality in respiratory viruses has long been characterized by the classical winter dominance of influenza, which exhibits remarkably synchronized peaks across both hemispheres, highlighting the influence of low ambient temperatures, reduced humidity, and increased indoor congregation on transmission[1]. In contrast, tropical climates exhibit more complex and variable respiratory viral patterns, where the rainy season rather than winter often promotes outbreaks of respiratory syncytial virus and parainfluenza[2]. These observations underscore that factors beyond simple meteorological parameters - particularly human behaviour, social structure, and built environments - play decisive roles in shaping viral spread.
The emergence of coronavirus disease 2019 (COVID-19), caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), has further disrupted this classical framework by revealing non-uniform seasonality across regions. Unlike influenza, which consistently peaks in cooler months, SARS-CoV-2 has shown repeated summer surges in tropical and subtropical regions, notably in India, Brazil, and Southeast Asia, while aligning with winter peaks in temperate zones such as Europe and North America[3]. This global divergence challenges the long-held notion of a singular “respiratory virus season” and calls for a contextualized understanding of seasonality that integrates climatic, biological, and sociocultural determinants through the agent-host-environment triad.
Recent multi-country modelling studies have indicated that behavioural-environmental feedback loops - including indoor air recirculation, crowding during heat or cold extremes, and varying compliance with ventilation norms - may outweigh temperature and humidity as predictors of SARS-CoV-2 transmission dynamics[4]. These findings align with the hypothesis that COVID-19 behaves as a “summer-predominant respiratory virus” in tropical settings, driven by human adaptation to high-heat environments rather than the virus’s direct response to meteorological conditions.
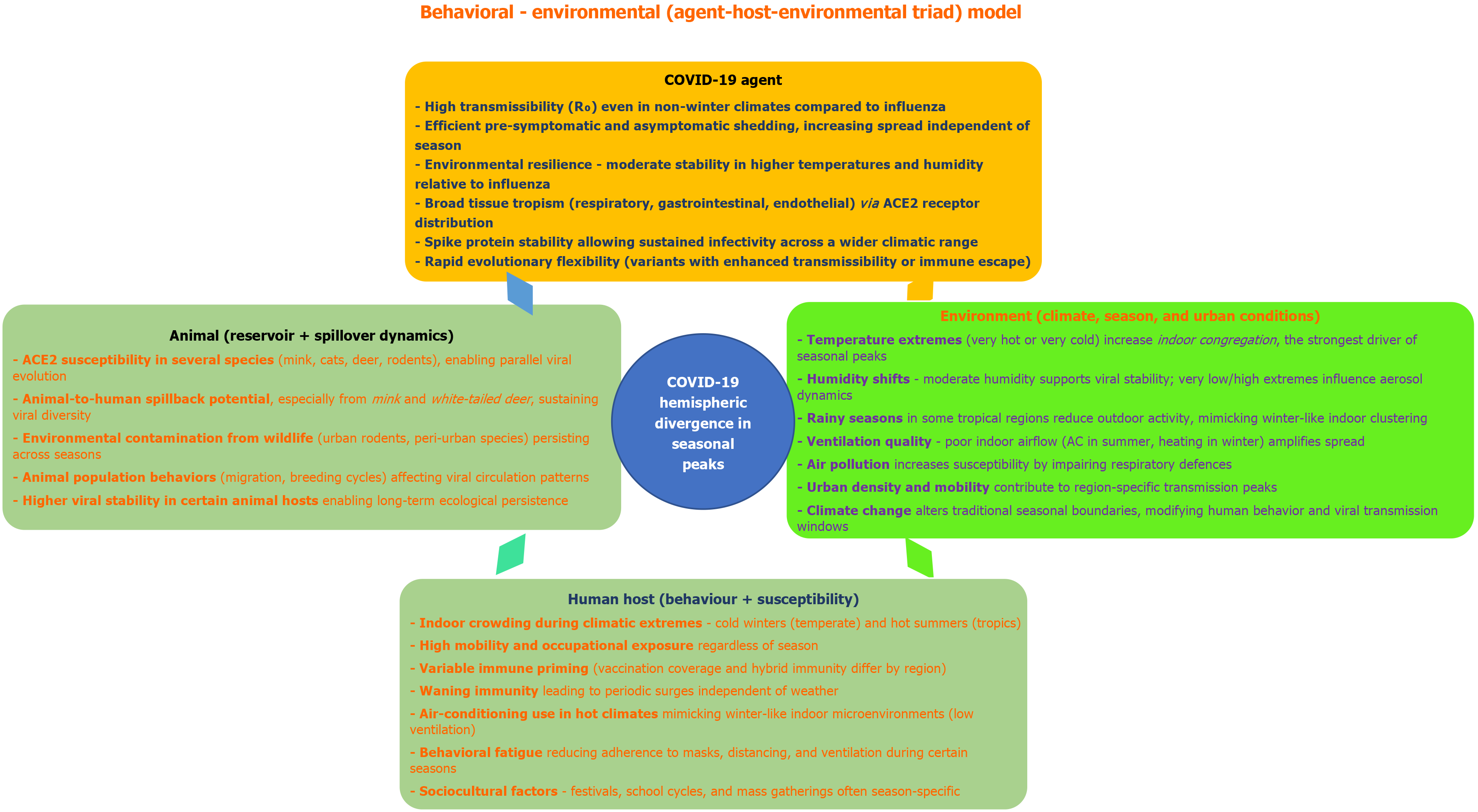
This review revisits COVID-19’s seasonal ecology through a “behavioural-environmental model”, positioning SARS-CoV-2 as a pathogen whose global transmission pattern is sculpted by the intersection of viral resilience (agent), human behaviour (host), and climate (environment). By contrasting these dynamics with influenza’s well-documented winter pattern, we propose that respiratory virus seasonality in the 21st century requires a multifactorial framework that integrates virological adaptability and socio-environmental context, rather than temperature alone, as the primary explanatory axis.
Comparative surveillance data continue to demonstrate a distinct hemispheric divergence in COVID-19 epidemiological trends, underscoring the virus’s context-dependent seasonality. In temperate regions such as the United Kingdom and Canada, recurrent winter surges of COVID-19 are consistently observed between December and February, aligning with periods of low temperature, reduced ultraviolet radiation, and heightened indoor activity[5,6]. Conversely, tropical and subtropical regions - including India, Brazil, and parts of Southeast Asia - exhibit marked summer waves during the pre-monsoon months between April and July, when ambient temperatures exceed 35 °C and humidity remains high[7,8].
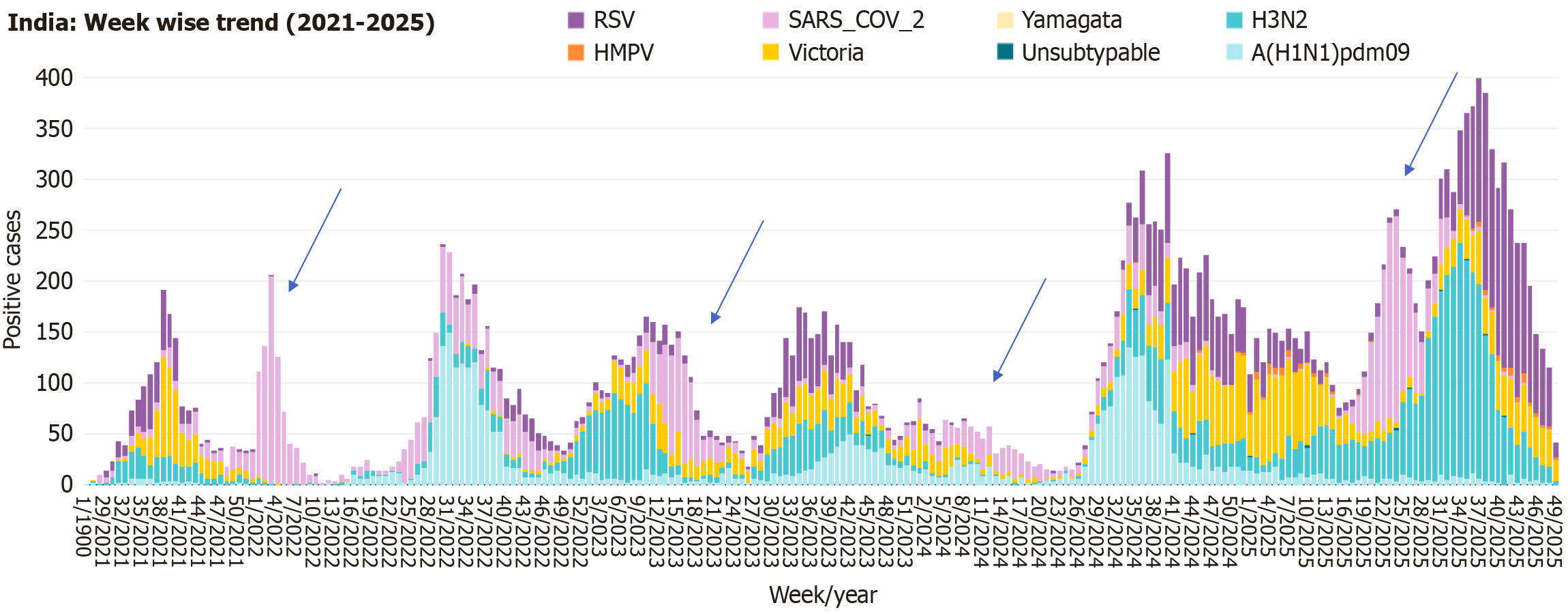
In India, where nearly 80% of the population resides within tropical and subtropical climatic belts, repeated SARS-CoV-2 upsurges were documented during the peak summer seasons from 2022 through 2025 (Figure 1). These surges occurred when influenza and respiratory syncytial virus activity were minimal, as reported by the ICMR-NIV Influenza Surveillance Network[7]. This temporal inversion relative to influenza, where SARS-CoV-2 peaks during months of influenza quiescence, highlights its non-classical and behaviourally modulated seasonality. Similar summer peaks have been observed in southern tropical countries such as Brazil and Indonesia, further reinforcing the association between heat-induced behavioral adaptation - particularly indoor congregation - and COVID-19 transmission[3].
At the global scale, multi-year datasets from Worldometer and the Johns Hopkins Coronavirus Resource Center illustrate that SARS-CoV-2 trends do not conform to a simple winter-bound pattern characteristic of influenza. The aggregation of cases across both hemispheres results in composite global curves that obscure regional seasonal distinctions[9]. For instance, southern hemisphere countries such as Australia, Chile, and South Africa predominantly experience winter waves (June to August), synchronous with their cooler seasons, while northern tropical and equatorial nations like India and Thailand repeatedly show summer-dominant peaks (April to June). This spatial-temporal inversion highlights the strong possibility of local behavioral and environmental contexts, rather than global temperature alone, dictating the timing of COVID-19 resurgences[4].
Collectively, these observations suggest that a uniform climatic determinism does not apply to COVID-19 transmission. Instead, they support a dual-seasonality model: Winter peaks in temperate zones, and summer peaks in tropical regions. This dichotomy marks a clear departure from the influenza model, in which winter predominance is nearly universal irrespective of latitude or economic context.
Despite both being enveloped single-stranded RNA viruses, SARS-CoV-2 and influenza display markedly distinct patterns of environmental resilience and thermal stability, which underpin their divergent seasonal behaviors. Controlled laboratory studies have shown that SARS-CoV-2 retains infectivity over a broad range of temperatures and humidity levels, maintaining viability for nearly 14 days at 4 °C and persisting for several days even at temperatures exceeding
In contrast, influenza viruses exhibit a narrower environmental tolerance. Their hemagglutinin (HA) protein is thermolabile, undergoing conformational destabilization at temperatures above 37 °C, which significantly reduces viral replication and infectivity[12]. Even brief exposure to elevated temperatures or fluctuations in pH can impair influenza’s ability to bind host sialic acid receptors effectively, limiting its spread in warmer climates. This biochemical fragility, combined with a preference for low-humidity environments, explains why influenza transmission peaks robustly during cold, dry winters and declines sharply in hot, humid conditions.
SARS-CoV-2’s spike (S) protein contrasts sharply in this respect. Structural analyses reveal that it maintains conformational integrity across a wider thermal range and resists denaturation under heat stress. Cryo-electron microscopy and molecular dynamics studies suggest that the spike’s glycan shielding and disulfide bonding networks enhance its thermostability, facilitating efficient receptor binding [angiotensin-converting enzyme 2 (ACE2)] even in high-temperature and humid conditions typical of tropical summers[13]. This resilience may partly account for the persistence of COVID-19 transmission in hot, densely populated tropical cities, such as Delhi, Mumbai, and São Paulo, during seasonal peaks when influenza activity remains low.
Beyond absolute temperature, microclimatic factors are increasingly recognized as playing a significant role in shaping viral persistence. Urban heat islands, characterized by elevated night-time temperatures and poor ventilation, can sustain aerosolized particles longer and enhance fomite stability. Furthermore, climate-induced shifts - such as longer warm seasons and altered rainfall patterns - modulate relative humidity, thereby influencing the aerosol physics of respiratory droplets. Recent modeling studies suggest that intermediate humidity (40%-60%) may favor the prolonged survival of SARS-CoV-2 aerosols, whereas influenza degrades rapidly under similar conditions[14].
Morris et al[15] provides a quantitative, physicochemical model of how virus inactivation rates vary with environmental conditions - useful to show that virus survival is environment-dependent. But higher temperature still accelerates decay so survival in heat is not guaranteed; depends on relative humidity, matrix, and viral load. Dabisch et al[16] also suggests that in hot, sunny, low-humidity outdoor settings - virus inactivation is rapid; but indoors (shade, no ultraviolet, recirculated air), survival may persist long enough for transmission. This supports a behavioral/environmental interplay (indoor crowding in heat). Thus, SARS-CoV-2’s virological robustness, coupled with changing climatic and urban conditions and animal adaptation, provides a plausible mechanistic basis for its persistence and transmissibility even during extreme heat.
The distinct receptor affinities and host immune interactions of SARS-CoV-2 and influenza viruses underpin their contrasting ecological and seasonal behaviors. Influenza viruses primarily attach to sialic acid residues on epithelial cells of the upper respiratory tract through their HA glycoprotein, a specificity that restricts infection largely to the nasopharyngeal mucosa and contributes to their dependence on cooler, low-humidity environments conducive to viral stability[12]. Variations in sialic acid linkages - α2,6 in humans and α2,3 in avian species - further constrain influenza’s cross-species adaptability and environmental persistence. This anatomical confinement likely contributes to its sharp seasonality and limited capacity to sustain transmission under tropical heat and humidity.
In contrast, SARS-CoV-2 employs the ACE2 receptor, which exhibits broad tissue distribution beyond the upper airway. ACE2 is expressed in the nasal epithelium, alveolar type II pneumocytes, enterocytes of the small intestine, vascular endothelium, and renal and cardiac tissues[17]. This multiorgan tropism expands potential transmission routes and may allow environmental persistence through fecal-oral contamination and other non-respiratory pathways. Moreover, the high affinity between the viral spike receptor-binding domain and ACE2, which has increased further across Omicron sublineages, enhances cellular entry efficiency even under suboptimal environmental conditions[18].
Thermal and humidity tolerance may also be mediated partly through receptor dynamics. Experimental work suggests that ACE2 expression in airway epithelial cells is upregulated under heat stress and air pollution exposure, conditions prevalent in tropical megacities[19]. Such environmental modulation could favor SARS-CoV-2 binding and replication during hot seasons, whereas sialic acid receptor density and stability decline under similar stressors, limiting influenza’s viability in comparable climates.
From an immunological standpoint, pre-existing population immunity and annual vaccination campaigns play central roles in shaping influenza’s seasonality. The presence of baseline cross-reactive antibodies and memory T cells against influenza A and B limits the virus’s spread during non-optimal seasons. In contrast, SARS-CoV-2 emerged into an immunologically naïve global population, allowing widespread dissemination irrespective of climate or behavioral barriers during its early waves[20]. Although hybrid immunity from infection and vaccination now provides substantial protection, immune escape through spike mutations - notably in Omicron and its descendants - has enabled recurrent resurgences independent of traditional seasonality cues.
Together, these features highlight the virus-host interface as a major determinant of COVID-19’s atypical seasonality. Broad receptor distribution, environmental modulation of ACE2, and evolving immune evasion collectively grant SARS-CoV-2 greater ecological plasticity than influenza, enabling transmission across temperature extremes and climatic zones where classical respiratory viruses remain dormant.
SARS-CoV-2 demonstrates an exceptionally broad mammalian host range, supporting its ecological persistence across diverse climates. Experimental studies show that species with high ACE2-spike compatibility - including cats, ferrets, and mink - develop productive upper-airway infection and efficient respiratory transmission, reflecting strong cross-species adaptability uncommon among respiratory viruses such as influenza[21]. Large-scale mink-farm outbreaks further revealed that high-density indoor environments promote rapid viral evolution, generating mink-associated variants capable of spilling back into humans[22]. Wildlife surveillance has expanded this ecological framework: White-tailed deer in North America exhibit widespread natural infection, sustained intra-species transmission, and weak neutralising responses, suggesting that free-living mammals may function as cold-season reservoirs, maintaining viral lineages independent of human transmission cycles[23]. More recently, Omicron-specific mutations enhancing ACE2 affinity across rodents demonstrate adaptive flexibility that allows SARS-CoV-2 to infect species previously resistant to early variants, widening its ecological footprint[24]. This multi-host adaptability contrasts sharply with influenza’s narrower mammalian susceptibility and provides an additional mechanistic layer to region-specific seasonality. The capacity for SARS-CoV-2 to persist, evolve, and transmit within multiple animal hosts supports its resilience under varied environmental and climatic pressures, reinforcing the behavioural-environmental model proposed for its tropical summer and temperate winter peaks.
Human behavioral adaptation to climate plays a pivotal role in shaping the seasonal expression of respiratory viral infections. The contrasting responses to climatic extremes in temperate vs tropical regions partly explain why SARS-CoV-2 surges occur during summer months in the tropics but winter months in temperate regions, in contrast to influenza’s consistent winter peaks across latitudes.
In temperate regions, winter drives populations indoors into well-insulated and heated environments that inadvertently promote viral transmission. Studies from the United Kingdom and Canada have shown that during cold months, indoor relative humidity drops below 40%, favoring aerosol stability of SARS-CoV-2 and influenza viruses alike[13]. Furthermore, increased crowding in poorly ventilated public spaces, coupled with reduced air exchange rates in energy-efficient buildings, prolongs airborne virus residence times. The use of central heating and sealed windows minimizes natural ventilation, reinforcing conditions conducive to respiratory virus propagation. Social behaviors such as holiday gatherings, school sessions, and workplace clustering also heighten contact intensity during winter, explaining why both COVID-19 and influenza tend to co-circulate in these months[25].
In contrast, tropical regions experience behavioral confinement during the hot, pre-monsoon summers, when outdoor temperatures often exceed 40 °C. To escape heat stress, populations gravitate toward air-conditioned environments - offices, malls, and public transport - where recirculated air and limited filtration create favorable microenvironments for viral persistence[7,26]. The thermal comfort-seeking behavior mimics winter indoor crowding patterns in temperate countries, but driven by heat rather than cold. In lower-income tropical settings, air-conditioning systems are often non-high-efficiency particulate arrestance filtered, and ventilation infrastructure is inadequate, producing stagnant indoor air and facilitating superspreading events.
Socioeconomic gradients further amplify these effects. In developed temperate countries, advanced infrastructure supports mechanical ventilation, humidity regulation, and CO2 monitoring, mitigating airborne transmission risks even during winter peaks. Conversely, in developing tropical nations, frequent power interruptions, overcrowded dwellings, and shared cooling systems substantially elevate exposure to respiratory pathogens. These structural inequities magnify the public health impact of summer outbreaks, as observed in India and Southeast Asia, where urban slums and dense market spaces become focal points for transmission.
Interestingly, the seasonal inversion of risk behavior provides insight into viral ecology. During temperate summers, populations spend more time outdoors under ultraviolet-rich, well-ventilated conditions that inactivate viral particles and dilute airborne concentrations. Outdoor socialization, school breaks, and natural ventilation sharply reduce transmission risks during these months[27]. Thus, the environmental permissiveness of the virus interacts with culturally and climatically conditioned human behavior - a dynamic equilibrium that determines whether a pathogen peaks in winter or summer.
Taken together, these behavioral and societal determinants highlight that temperature itself is not the sole driver of seasonality. Rather, it is the behavioral response to thermal extremes - whether heating in winter or cooling in summer - that recreates similar indoor transmission ecologies under different climatic regimes. This behavioral-environmental (agent-host-environmental triad, Figure 2) coupling explains why COVID-19 exhibits winter peaks in temperate regions and summer peaks in tropical settings, while influenza, constrained by its virological fragility in heat, remains confined to cooler months worldwide.
Despite exposure to similar environmental and behavioral pressures, influenza viruses have remained predominantly winter-dominant pathogens across both temperate and tropical regions. This divergence from SARS-CoV-2’s adaptive summer activity highlights several intrinsic biological and epidemiological distinctions that constrain influenza’s capacity to persist under high-temperature or high-humidity conditions.
First, influenza’s transmission efficiency and pre-symptomatic shedding window are relatively limited compared with SARS-CoV-2. Influenza’s median serial interval of 2-3 days and shorter duration of peak viral load reduce its potential for extended community spread, particularly in marginal environmental conditions. In contrast, SARS-CoV-2 exhibits significant pre-symptomatic and asymptomatic transmission, sustaining infection chains even when symptomatic cases are isolated[28].
Second, thermal and humidity sensitivity critically limit influenza’s environmental survival. Laboratory models have repeatedly shown that influenza virions lose infectivity rapidly at temperatures above 30-33 °C, with steep inactivation at 37 °C and in humid environments above 60% relative humidity. This fragility is linked to the instability of the viral envelope and HA conformation, which denature more readily under heat and moisture stress than the spike glycoprotein of SARS-CoV-2[29]. As a result, outdoor tropical environments are inhospitable to influenza virions, and even indoor air-conditioning fails to fully replicate the cold, dry microclimates that optimize influenza transmission in temperate winters.
Third, population-level immunity plays a central role in constraining influenza’s seasonal reach. Regular exposure to circulating strains and annual vaccination programs confer broad baseline protection in many populations, limiting transmission potential during off-peak periods[7,30]. This immunological landscape contrasts with the initial global naïveté to SARS-CoV-2 and the continuing evolution of immune-evading variants. Furthermore, the immune pressure on influenza’s HA and neuraminidase proteins results in predictable antigenic drift rather than the extensive structural shifts seen in the SARS-CoV-2 spike, reducing its potential for abrupt, season-independent surges.
Epidemiologically, influenza’s behavior in the tropics further explains these biological limits. While some intertropical regions exhibit bimodal or year-round low-level activity, true epidemic peaks in temperate regions still coincide with the onset of cooler, less humid seasons or periods of intense rainfall, conditions that transiently mimic temperate microclimates[31]. Even during extreme summer indoor congregation - such as during heat waves in India or Southeast Asia - surveillance data reveal minimal influenza resurgence compared to concurrent SARS-CoV-2 waves[7].
Taken together, these observations emphasize that influenza’s transmission ecology is tightly constrained by its thermal instability, limited asymptomatic spread and partial herd immunity, whereas SARS-CoV-2 possesses both biophysical resilience and immunological plasticity enabling its persistence and resurgence in hot tropical climates. Thus, even under analogous indoor congregation during summer, influenza lacks the viral durability and behavioral exploitation capacity required to trigger major outbreaks, reinforcing the distinct ecological niche occupied by SARS-CoV-2.
The divergent seasonal peaks of COVID-19 summer surges in tropical countries and winter waves in temperate ones can be understood as the outcome of a dynamic interplay among viral biology, host receptor distribution, immune landscape, and human behavioral adaptation to climate. SARS-CoV-2, unlike influenza, demonstrates marked resilience to heat, broad tissue tropism, multi-animal adaptation, and high transmissibility. When these traits coincide with human behaviors that drive indoor congregation during cold winters in temperate zones and hot summers in the tropics - region-specific surges emerge that defy the classical influenza model. Invisible yet growing influences of global warming and one-health-linked environmental change likely reinforce this evolving pattern. Thus, defining COVID-19 as a strictly “winter virus” oversimplifies its epidemiology. It should instead be viewed as a behaviorally modulated seasonal infection, exhibiting summer predominance in tropical regions, thereby challenging long-held assumptions and informing climate- and region-specific surveillance and prevention strategies.
| 1. | Lofgren E, Fefferman NH, Naumov YN, Gorski J, Naumova EN. Influenza seasonality: underlying causes and modeling theories. J Virol. 2007;81:5429-5436. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 472] [Cited by in RCA: 363] [Article Influence: 19.1] [Reference Citation Analysis (1)] |
| 2. | Paynter S. Humidity and respiratory virus transmission in tropical and temperate settings. Epidemiol Infect. 2015;143:1110-1118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 114] [Article Influence: 10.4] [Reference Citation Analysis (1)] |
| 3. | Gavenčiak T, Monrad JT, Leech G, Sharma M, Mindermann S, Bhatt S, Brauner J, Kulveit J. Seasonal variation in SARS-CoV-2 transmission in temperate climates: A Bayesian modelling study in 143 European regions. PLoS Comput Biol. 2022;18:e1010435. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 30] [Article Influence: 7.5] [Reference Citation Analysis (1)] |
| 4. | Hu MZ, Song T, Gong ZY, Chen L, Che QZ, Guo J, Liang N, Cheng CC, Wang YP, Shi NN, Liu B. Pandemic transmission forecasting: insights from COVID-19 modeling involving environmental factors. Crit Public Health. 2025;35. [DOI] [Full Text] |
| 5. | Government of Canada. Health data in Canada. [cited 3 September 2025]. Available from: https://health-infobase.canada.ca. |
| 6. | Government of UK. National flu and COVID-19 surveillance reports: 2025 to 2026 season. [cited 3 September 2025]. Available from: https://www.gov.uk/government/statistics/national-flu-and-covid-19-surveillance-reports-2025-to-2026-season. |
| 7. | ICMR. Weekly Testing and Virus Wise Positivity with Total Tested Records (Interleaved by Week). [cited 3 September 2025]. Available from: https://influenza.icmr.org.in/public_dashboard/. |
| 8. | Pan American Health Organization. COVID-19 Trends. [cited 3 September 2025]. Available from: https://shiny.pahobra.org/wdc/. |
| 9. | Worldometer. COVID-19 coronavirus pandemic. [cited 3 September 2025]. Available from: https://www.worldometers.info/coronavirus/. |
| 10. | Chan KH, Peiris JS, Lam SY, Poon LL, Yuen KY, Seto WH. The Effects of Temperature and Relative Humidity on the Viability of the SARS Coronavirus. Adv Virol. 2011;2011:734690. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 681] [Cited by in RCA: 533] [Article Influence: 35.5] [Reference Citation Analysis (1)] |
| 11. | Riddell S, Goldie S, Hill A, Eagles D, Drew TW. The effect of temperature on persistence of SARS-CoV-2 on common surfaces. Virol J. 2020;17:145. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 528] [Cited by in RCA: 385] [Article Influence: 64.2] [Reference Citation Analysis (1)] |
| 12. | Weber TP, Stilianakis NI. Inactivation of influenza A viruses in the environment and modes of transmission: a critical review. J Infect. 2008;57:361-373. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 387] [Cited by in RCA: 306] [Article Influence: 17.0] [Reference Citation Analysis (1)] |
| 13. | Moriyama M, Hugentobler WJ, Iwasaki A. Seasonality of Respiratory Viral Infections. Annu Rev Virol. 2020;7:83-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1035] [Cited by in RCA: 746] [Article Influence: 124.3] [Reference Citation Analysis (1)] |
| 14. | Verheyen CA, Bourouiba L. Associations between indoor relative humidity and global COVID-19 outcomes. J R Soc Interface. 2022;19:20210865. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 15. | Morris DH, Yinda KC, Gamble A, Rossine FW, Huang Q, Bushmaker T, Fischer RJ, Matson MJ, Van Doremalen N, Vikesland PJ, Marr LC, Munster VJ, Lloyd-Smith JO. Mechanistic theory predicts the effects of temperature and humidity on inactivation of SARS-CoV-2 and other enveloped viruses. Elife. 2021;10:e65902. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 191] [Cited by in RCA: 134] [Article Influence: 26.8] [Reference Citation Analysis (1)] |
| 16. | Dabisch P, Schuit M, Herzog A, Beck K, Wood S, Krause M, Miller D, Weaver W, Freeburger D, Hooper I, Green B, Williams G, Holland B, Bohannon J, Wahl V, Yolitz J, Hevey M, Ratnesar-Shumate S. The influence of temperature, humidity, and simulated sunlight on the infectivity of SARS-CoV-2 in aerosols. Aerosol Sci Technol. 2021;55:142-153. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 189] [Cited by in RCA: 117] [Article Influence: 19.5] [Reference Citation Analysis (1)] |
| 17. | Hamming I, Timens W, Bulthuis ML, Lely AT, Navis G, van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol. 2004;203:631-637. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4648] [Cited by in RCA: 4075] [Article Influence: 185.2] [Reference Citation Analysis (7)] |
| 18. | Aksenova AY, Likhachev IV, Grishin SY, Galzitskaya OV. The Increased Amyloidogenicity of Spike RBD and pH-Dependent Binding to ACE2 May Contribute to the Transmissibility and Pathogenic Properties of SARS-CoV-2 Omicron as Suggested by In Silico Study. Int J Mol Sci. 2022;23:13502. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 19. | Miyashita L, Foley G, Grigg J. Exposure to Particulate Matter Increases Expression of the Angiotensin converting enzyme-2 (ACE2) Receptor. J Allergy Clin Immunol. 2022;149:AB30. |
| 20. | Kissler SM, Tedijanto C, Goldstein E, Grad YH, Lipsitch M. Projecting the transmission dynamics of SARS-CoV-2 through the postpandemic period. Science. 2020;368:860-868. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2038] [Cited by in RCA: 1521] [Article Influence: 253.5] [Reference Citation Analysis (3)] |
| 21. | Shi J, Wen Z, Zhong G, Yang H, Wang C, Huang B, Liu R, He X, Shuai L, Sun Z, Zhao Y, Liu P, Liang L, Cui P, Wang J, Zhang X, Guan Y, Tan W, Wu G, Chen H, Bu Z. Susceptibility of ferrets, cats, dogs, and other domesticated animals to SARS-coronavirus 2. Science. 2020;368:1016-1020. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1573] [Cited by in RCA: 1333] [Article Influence: 222.2] [Reference Citation Analysis (0)] |
| 22. | Oude Munnink BB, Sikkema RS, Nieuwenhuijse DF, Molenaar RJ, Munger E, Molenkamp R, van der Spek A, Tolsma P, Rietveld A, Brouwer M, Bouwmeester-Vincken N, Harders F, Hakze-van der Honing R, Wegdam-Blans MCA, Bouwstra RJ, GeurtsvanKessel C, van der Eijk AA, Velkers FC, Smit LAM, Stegeman A, van der Poel WHM, Koopmans MPG. Transmission of SARS-CoV-2 on mink farms between humans and mink and back to humans. Science. 2021;371:172-177. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 986] [Cited by in RCA: 797] [Article Influence: 159.4] [Reference Citation Analysis (1)] |
| 23. | Hale VL, Dennis PM, McBride DS, Nolting JM, Madden C, Huey D, Ehrlich M, Grieser J, Winston J, Lombardi D, Gibson S, Saif L, Killian ML, Lantz K, Tell RM, Torchetti M, Robbe-Austerman S, Nelson MI, Faith SA, Bowman AS. SARS-CoV-2 infection in free-ranging white-tailed deer. Nature. 2022;602:481-486. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 364] [Cited by in RCA: 294] [Article Influence: 73.5] [Reference Citation Analysis (1)] |
| 24. | Halfmann PJ, Iida S, Iwatsuki-Horimoto K, Maemura T, Kiso M, Scheaffer SM, Darling TL, Joshi A, Loeber S, Singh G, Foster SL, Ying B, Case JB, Chong Z, Whitener B, Moliva J, Floyd K, Ujie M, Nakajima N, Ito M, Wright R, Uraki R, Warang P, Gagne M, Li R, Sakai-Tagawa Y, Liu Y, Larson D, Osorio JE, Hernandez-Ortiz JP, Henry AR, Ciuoderis K, Florek KR, Patel M, Odle A, Wong LR, Bateman AC, Wang Z, Edara VV, Chong Z, Franks J, Jeevan T, Fabrizio T, DeBeauchamp J, Kercher L, Seiler P, Gonzalez-Reiche AS, Sordillo EM, Chang LA, van Bakel H, Simon V; Consortium Mount Sinai Pathogen Surveillance (PSP) study group, Douek DC, Sullivan NJ, Thackray LB, Ueki H, Yamayoshi S, Imai M, Perlman S, Webby RJ, Seder RA, Suthar MS, García-Sastre A, Schotsaert M, Suzuki T, Boon ACM, Diamond MS, Kawaoka Y. SARS-CoV-2 Omicron virus causes attenuated disease in mice and hamsters. Nature. 2022;603:687-692. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 616] [Cited by in RCA: 508] [Article Influence: 127.0] [Reference Citation Analysis (1)] |
| 25. | Pica N, Bouvier NM. Environmental factors affecting the transmission of respiratory viruses. Curr Opin Virol. 2012;2:90-95. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 275] [Cited by in RCA: 189] [Article Influence: 13.5] [Reference Citation Analysis (1)] |
| 26. | Lu J, Gu J, Li K, Xu C, Su W, Lai Z, Zhou D, Yu C, Xu B, Yang Z. COVID-19 Outbreak Associated with Air Conditioning in Restaurant, Guangzhou, China, 2020. Emerg Infect Dis. 2020;26:1628-1631. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 748] [Cited by in RCA: 509] [Article Influence: 84.8] [Reference Citation Analysis (1)] |
| 27. | Li Y, Cheng P, Jia W. Poor ventilation worsens short-range airborne transmission of respiratory infection. Indoor Air. 2022;32:e12946. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 42] [Article Influence: 10.5] [Reference Citation Analysis (1)] |
| 28. | Bai Y, Tao X. Comparison of COVID-19 and influenza characteristics. J Zhejiang Univ Sci B. 2021;22:87-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 59] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 29. | Zhang L, Fang Z, Li J, Huang Z, Tie X, Li H, Li J, Zhang Y, Zhang Y, Chen K. Research progress on environmental stability of SARS-CoV-2 and influenza viruses. Front Microbiol. 2024;15:1463056. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 30. | Trombetta CM, Kistner O, Montomoli E, Viviani S, Marchi S. Influenza Viruses and Vaccines: The Role of Vaccine Effectiveness Studies for Evaluation of the Benefits of Influenza Vaccines. Vaccines (Basel). 2022;10:714. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 102] [Cited by in RCA: 84] [Article Influence: 21.0] [Reference Citation Analysis (1)] |
| 31. | Saha S, Chadha M, Shu Y; Group of Asian Researchers on Influenza (GARI). Divergent seasonal patterns of influenza types A and B across latitude gradient in Tropical Asia. Influenza Other Respir Viruses. 2016;10:176-184. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 42] [Article Influence: 4.2] [Reference Citation Analysis (1)] |