Copyright: ©Author(s) 2026.
World J Virol. Mar 25, 2026; 15(1): 116055
Published online Mar 25, 2026. doi: 10.5501/wjv.v15.i1.116055
Published online Mar 25, 2026. doi: 10.5501/wjv.v15.i1.116055
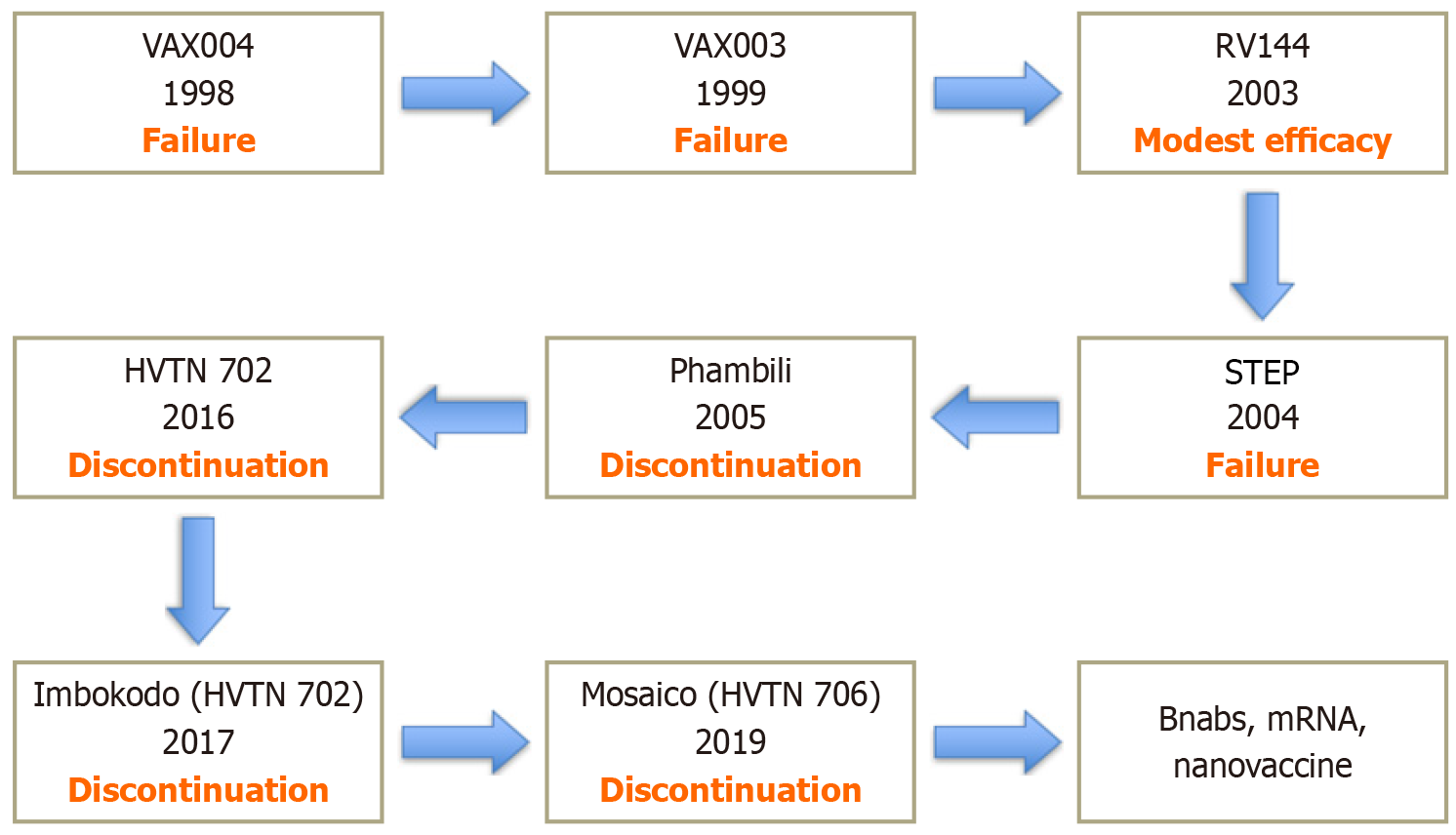
Figure 1 This timeline illustrates over 25 years of human immunodeficiency virus vaccine development efforts.
Early protein vaccines (VAX004, VAX003) failed. RV144 showed modest 31% protection-the only success so far. Adenovirus vectors (STEP, Phambili) were stopped early due to safety risk. Recent poxvirus and mosaic Ad269 (Adenovirus serotype 26) human immunodeficiency virus vaccine trials (HVTN 702, Imbokodo, Mosaico) have also failed. Now, researchers focus on bnAbs, mRNA, and nanoparticle vaccines.
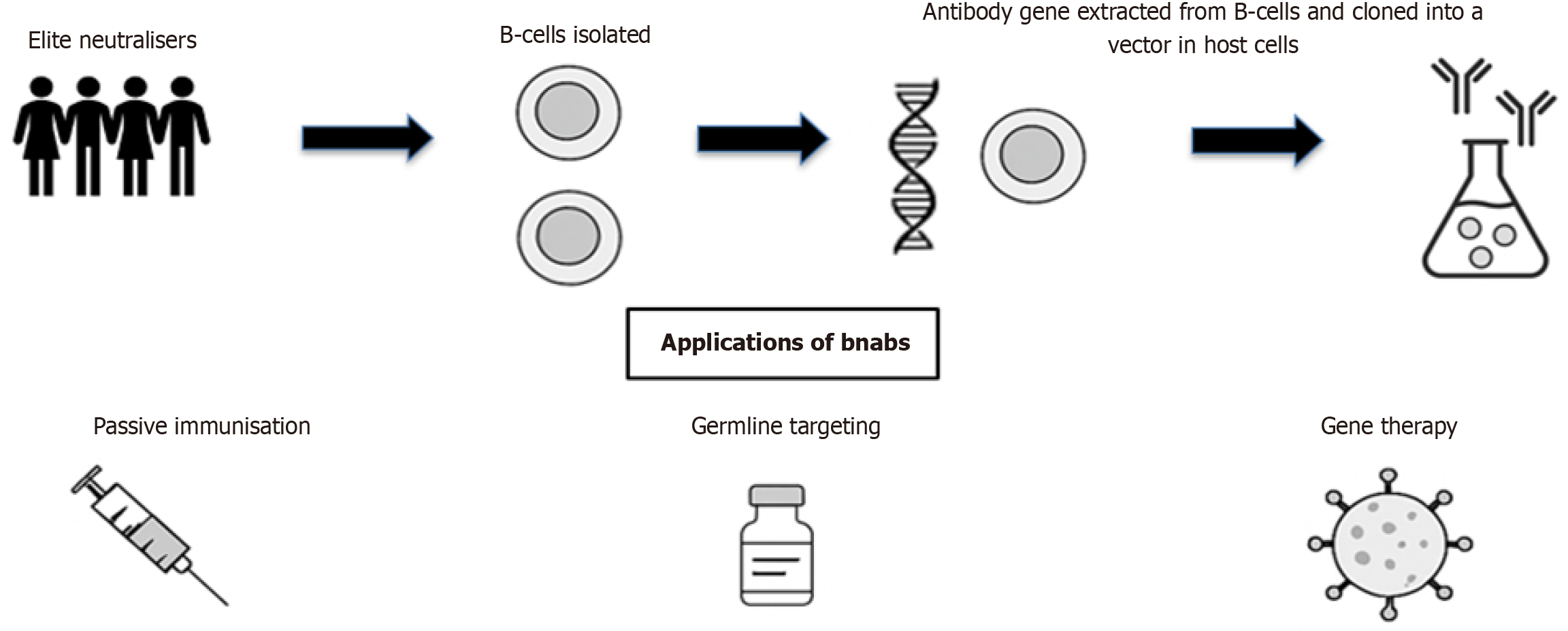
Figure 2 From elite neutralisers, B-cells producing broad neutralising antibodies are isolated, and antibody genes are extracted and cloned into expression vectors for production.
Applications include: (1) Passive immunisation via bnAb infusions for immediate viral control; (2) Germline targeting vaccines designed to activate bnAb precursor B-cells; and (3) Gene therapy delivering bnAb genes for long-term endogenous production.
- Citation: Varatharajan S, Krishnasai S, Girish C. Human immunodeficiency virus vaccines: Advances, challenges and future perspectives. World J Virol 2026; 15(1): 116055
- URL: https://www.wjgnet.com/2220-3249/full/v15/i1/116055.htm
- DOI: https://dx.doi.org/10.5501/wjv.v15.i1.116055