Published online May 19, 2026. doi: 10.5498/wjp.v16.i5.115867
Revised: December 16, 2025
Accepted: February 3, 2026
Published online: May 19, 2026
Processing time: 166 Days and 1.1 Hours
Depressive disorder is a common non-motor complication of Parkinson’s disease (PD) that seriously impairs the quality of life. Evidence suggests that dopaminer
To evaluate the efficacy and safety of dopaminergic and/or selective serotonin re
English-language randomized controlled trials published between January 2000 and January 2025 were retrieved from the PubMed and EMBASE databases. Eli
Five randomized controlled trials comprising 508 patients met the inclusion cri
Therapeutic strategies targeting dopaminergic and serotonergic pathways through dopaminergic agents, SSRIs, or their combination can improve depressive symptoms and quality of life in patients with PD and depressive dis
Core Tip: This meta-analysis of five studies discussed the clinical efficacy of dopaminergic agents combined with selective serotonin reuptake inhibitors in the treatment of Parkinson’s disease with depressive disorder. The results showed that the ef
- Citation: Li CC, Zhou Y, Chen KX, Zhang L, Gu WL, Gu H. Meta-analysis of dopaminergic-serotonergic modulation strategies for depressive symptoms in Parkinson’s disease. World J Psychiatry 2026; 16(5): 115867
- URL: https://www.wjgnet.com/2220-3206/full/v16/i5/115867.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i5.115867
Parkinson’s disease (PD) is a common neurodegenerative disease in middle-aged and elderly populations, and its core pathological mechanism is the progressive loss of dopaminergic neurons in the substantia nigra (SN). The primary clinical manifestations of PD include tremors, myotonia and bradykinesia[1]. However, the burden of non-motor symptoms in patients with PD is equally heavy. Depression is one of the most common and clinically significant mental complications, and the incidence rate can reach 35%-50%[2,3]. Depression not only aggravates dyskinesia and impairs life function in patients, but also significantly reduces drug compliance and quality of life, which is associated with a higher risk of suicide and accelerated disease progression[4]. Therefore, the optimization of treatment strategies for PD with depressive disorder has become an important issue in the cross-sectional field of neuropsychiatry[5]. Available evidence suggests that the mechanism of PD with depressive disorder involves damage to the dopaminergic system, dysfunction of the 5-hydroxytryptamine (5-HT) system, and an imbalance of multi-neurotransmitter interactions. Simple dependence on antidepressants or dopamine replacement therapy has the limitation of limited efficacy[6,7]. Selective serotonin reuptake inhibitors (SSRIs) have been widely used to improve depressive symptoms; however, some patients show a poor response, and the effect of SSRIs on exercise symptoms is controversial. Dopaminergic agents can alleviate dep
Study enrollment was conducted according to the standardized methods of systematic evaluation and meta-analysis. The retrieval period ranged from January 2000 to January 2025. The PubMed, EMBASE, Web of Science, and Cochrane Library databases were searched. The English keywords “Parkinson’s disease”, “depression”, “dopaminergic agents”, “selective serotonin reuptake inhibitors”, “combination therapy”, “randomized controlled trial” and their combinations were used for systematic retrieval. Boolean logic operations (AND, OR) were adopted to construct the search expression, which combined subject words with free words to ensure the comprehensiveness and accuracy of the search. All the in
The inclusion criteria were as follows: (1) Research subjects were patients with PD and depressive disorder with a clear diagnosis; (2) Intervention measures including dopaminergic agents, SSRIs, or their combined use. Studies were eligible if they evaluated any treatment targeting the dopaminergic system, serotonergic system, or both, compared with placebo or other active monotherapy. This criterion was designed to capture therapeutic strategies involving single- or dual-pa
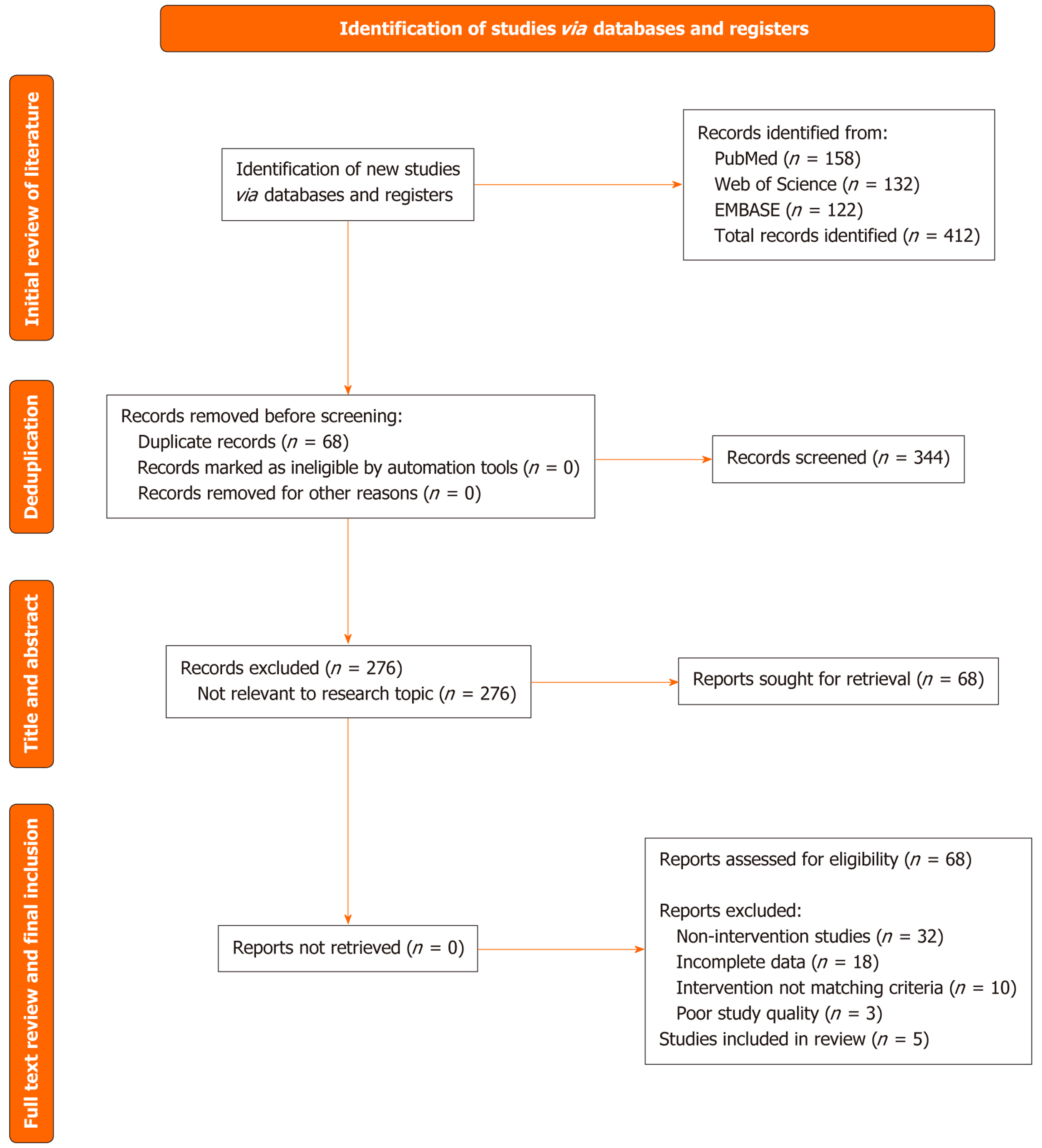
The literature selection process was performed strictly in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) process. EndNote literature management software was used to import the search results and delete duplicate articles. Second, the titles and abstracts were sieved by two researchers to exclude articles that were not related to the research topic. Subsequently, the full text of potentially eligible articles was reviewed for further screening based on the prespecified inclusion and exclusion criteria. In the process of literature selection, if there is any disagreement, a third researcher shall rule to ensure objectivity and consistency of literature selection. The articles included in the meta-analysis were finalized, and the screening process and numbers were recorded. A PRISMA for Sys
All baseline patient data reported in the included articles were extracted and compared. The main indicators include: (1) Demographic characteristics, such as age and gender; (2) The clinical characteristics of the disease, including the course of PD (year), the severity of motor symptoms of PD (expressed by the UPDRS-III score or total score) and the severity of depressive disorder (expressed by the HAMD-17 or MADRS score); and (3) Medications, such as the specific types and doses of dopaminergic agents (levodopa, pramipexole, etc.) and SSRIs (paroxetine, sertraline, and citalopram, etc.). Co
The intervention measures varied across studies and included: (1) Dopaminergic agents alone; (2) SSRIs alone; and (3) Combined use of dopaminergic agents and SSRIs in studies where such regimens were evaluated. The control group received either a placebo or active monotherapy. Given the limited number of true fixed-combination RCTs, this categorization allowed us to evaluate both single-pathway (dopaminergic or serotonergic) and dual-pathway modulation stra
The outcome indicators included core efficacy and safety indicators. The primary indicator was improvement in depressive symptoms, which was assessed by the change in the score or endpoint score from baseline to the end of treatment on the HAMD-17 or MADRS. Secondary indicators included: (1) Improvement in motor function, as assessed by changes in the UPDRS-III; (2) The proportion of patients with effective treatment rate, generally defined as the reduction rate of HAMD or MADRS ≥ 50%; (3) The overall quality of life, as measured by scales such as the PD quality of life questionnaire-39; and (4) Safety indicators. The incidence and severity of adverse events as well as the dropout rate caused by adverse events during treatment were recorded in detail to evaluate the safety of the combination therapy. All the indicator data were extracted from the final reports included in this study.
Statistical analyses were performed according to the meta-analysis specifications recommended by the Cochrane System Evaluation Manual. Data analysis was performed using the Stata/RevMan software. First, the mean difference (MD) or standardized MD and its 95% confidence interval (CI) were applied to continuous variables (HAMD and UPDRS scores) as the quantity of effect. The risk ratio (RR) and 95%CI were used as the quantity of effect for the two categorical variables (treatment efficiency). Inter-study heterogeneity was assessed using Cochran’s Q test and I2 statistic. If I2 > 50% or P < 0.1, significant heterogeneity was considered, and the data were combined using a random-effects model. Otherwise, a fixed-effects model was used. The sources of heterogeneity were explored using subgroup analysis or meta-regression. Publication bias was assessed using funnel plots and Egger’s test. Sensitivity analysis was performed to verify the sta
Through systematic retrieval and strict screening, 412 relevant articles were identified in the initial study. After 68 repetitive articles were excluded using EndNote software, the titles and abstracts of the remaining 344 articles were screened, and 276 articles that were obviously irrelevant were excluded. Full texts of the remaining 68 articles were reviewed. Finally, based on the inclusion and exclusion criteria, five studies met the requirements and were included in the meta-analysis. The literature screening process strictly followed the PRISMA guidelines, and the specific process is shown in Figure 1.
Five RCTs related to PD with depressive disorder were included and assessed using the Cochrane RoB2 and Jadad scales[10-14]. Two studies (Barone et al[10], 2010, Devos et al[14], 2008) were of low risk and high quality (Jadad: 5 points), with double-blind and ITT analyses, two items were of medium risk and low quality (Jadad: 3 points), and one item was of high risk and extremely low quality (Jadad: 2 points, no random/blind method), with the overall quality of the medium (Table 1).
| Ref. | Type | Random sequence generation | Distribution concealment | Blind implementation | Missing data | Selective reporting | Risk type of bias | Risk score | Quality score (Jadad) |
| Barone et al[10], 2010 | RCT | + | + | + | + | + | Low risk | 9 | 5 |
| Menza et al[11], 2009 | RCT | ? | ? | - | ? | ? | Moderate risk | 5 | 3 |
| Rektorova et al[12], 2008 | RCT | - | - | - | - | - | High-risk | 2 | 2 |
| Weintraub et al[13], 2010 | RCT | ? | ? | - | ? | ? | Moderate risk | 5 | 3 |
| Devos et al[14], 2008 | RCT | + | + | + | + | ? | Low risk | 8 | 5 |
The baseline characteristics of patients in the five included studies were comparable. The average age of the patients was approximately 64.62 years old, and the proportion of men was approximately 58.11%. The mean disease duration was approximately 6.60 years. The UPDRS-III score was approximately 29.40 points, the HAMD-17/MADRS score indicated moderate depression, and the Mini-Mental State Examination score was > 27 points (Table 2).
| Ref. | n | Age (years) | Male | Course of disease (year) | UPDRS-III (points) | HAMD-17 (points) | Depression score (points) | Dopaminergic drug | SSRI (intervention group) | MMSE (points) |
| Barone et al[10], 2010 | 323 | 64.30 ± 9.10 | 181 (56.04) | 6.20 ± 4.50 | 28.50 ± 12.30 | 19.80 ± 4.20 | - | Pramipexole (experimental drug), levodopa utilization approximately 93% at baseline | None (compared to placebo) | 28.10 ± 1.50 |
| Menza et al[11], 2009 | 52 | 65.10 ± 8.70 | 30 (57.69) | 7.00 ± 5.10 | 30.20 ± 13.50 | 20.50 ± 4.80 | - | Baseline dose not reported in detail | Paroxetine (mean dose: 27.70 mg/day) | 28.50 ± 1.20 |
| Rektorova et al[12], 2008 | 30 | 63.50 ± 7.80 | 17 (56.67) | 5.80 ± 3.90 | 27.80 ± 11.60 | - | MADRS: 21.60 ± 4.10 | Ropinirole (experimental drug, flexible dose) | None (compared to pramipexole) | 28.30 ± 1.40 |
| Weintraub et al[13], 2010 | 55 | 66.20 ± 9.30 | 34 (61.82) | 7.50 ± 4.80 | 31.00 ± 14.10 | - | MADRS: 22.90 ± 5.20 | Baseline dose not reported in detail | None (atorvastatin compared to placebo) | 27.80 ± 1.80 |
| Devos et al[14], 2008 | 48 | 64.80 ± 8.50 | 28 (58.33) | 6.50 ± 4.20 | 29.50 ± 12.80 | 19.50 ± 4.50 | - | Levodopa (mean dose: 568.75 mg/day) | Citalopram (mean dose: 25.83 mg/day) | 28.00 ± 1.60 |
The five included studies were designed as RCTs, and the specific treatment measures were diverse and could be divided into two strategies: Dopaminergic agent monotherapy and antidepressant agent monotherapy. As for intervention measures, two studies have evaluated the antidepressant effect of dopamine receptor agonists: Barone et al[10] used pramipexole as an intervention measure in their study, with flexible dose adjustment, and the target was set at 1.5 mg/day to 4.5 mg/day, which was compared with placebo; Rektorova et al[12] compared the efficacy of ropinirole with that of pramipexole with a maximum dose of 24 mg daily. In contrast, two studies directly evaluated the effect of antidepressants: Menza et al[11] established two intervention groups of paroxetine (average dose of 27.70 mg per day) and nortriptyline simultaneously, which were compared with a placebo; Devos et al[14] directly compared nortriptyline (75 mg daily at a fixed dose) and citalopram (20 mg daily at a fixed dose). Weintraub et al[13] explored the differences between the atypical drug atomoxetine (target dose of 40-80 mg/day) and placebo. In terms of treatment duration, most studies lasted for 8-12 weeks, with the longest treatment duration of 14 weeks in the study by Devos et al[14]. These studies were designed to be double-blind, as reported by Rektorova et al[12]. The study was single-blind.
Treatments targeting the dopaminergic or serotonergic pathways, including dopaminergic agents, SSRIs, or their res
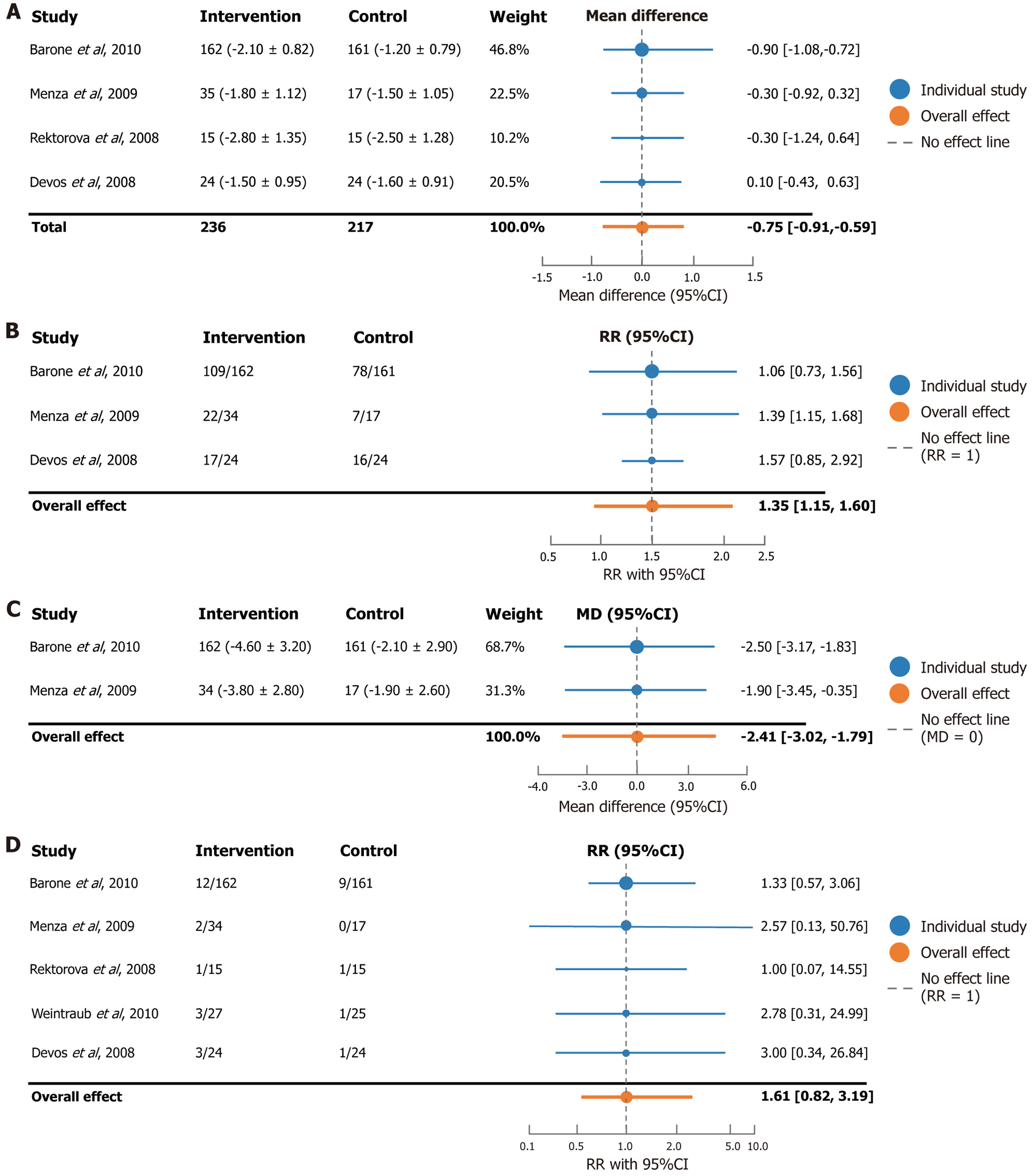
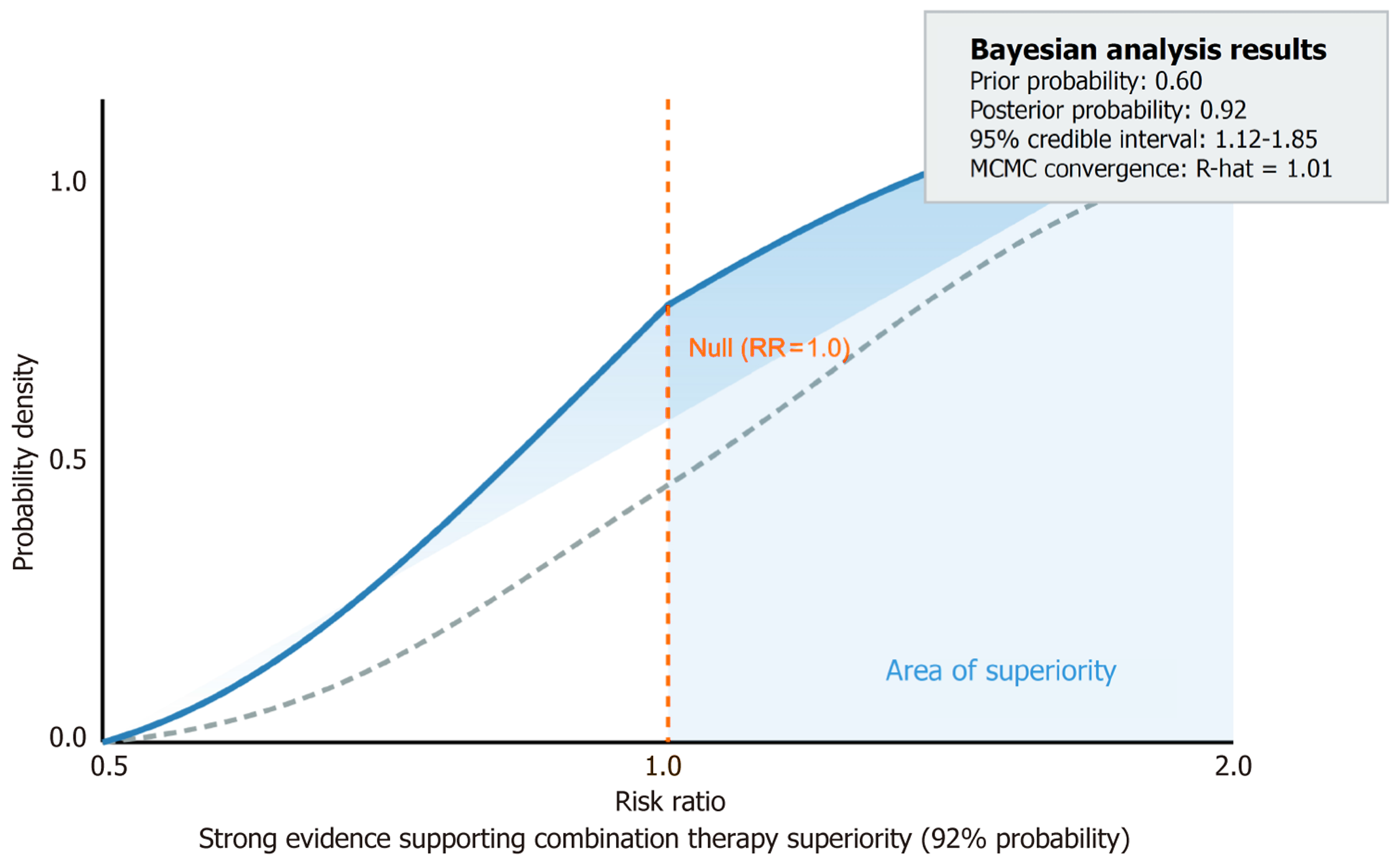
The analysis based on the reducing rate of HAMD/MADRS ≥ 50% showed that the effective rates of pramipexole and paroxetine for the treatment of PD with depressive disorder were significantly better than those of the control group (RR = 1.40, 95%CI: 1.16-1.69), while the efficacy of nortriptyline and citalopram was comparable. The heterogeneity of the results was low and the evidence quality was high (Figure 2B).
The intervention group was significantly better than the control group in improving the quality of life of patients with PD with depressive disorder (MD = -2.30, 95%CI: -2.90 to -1.70), and the difference was statistically significant (Figure 2C).
Compared to the control group, there was a trend of an increased risk of dropping out due to adverse events in the intervention group (including dopamine receptor agonists and antidepressants) (RR = 1.70), but the difference was not statistically significant (95%CI: 0.94-3.07). This indicates that combination therapy or monotherapy is tolerable; however, individual adverse reactions require close attention (Figure 2D).
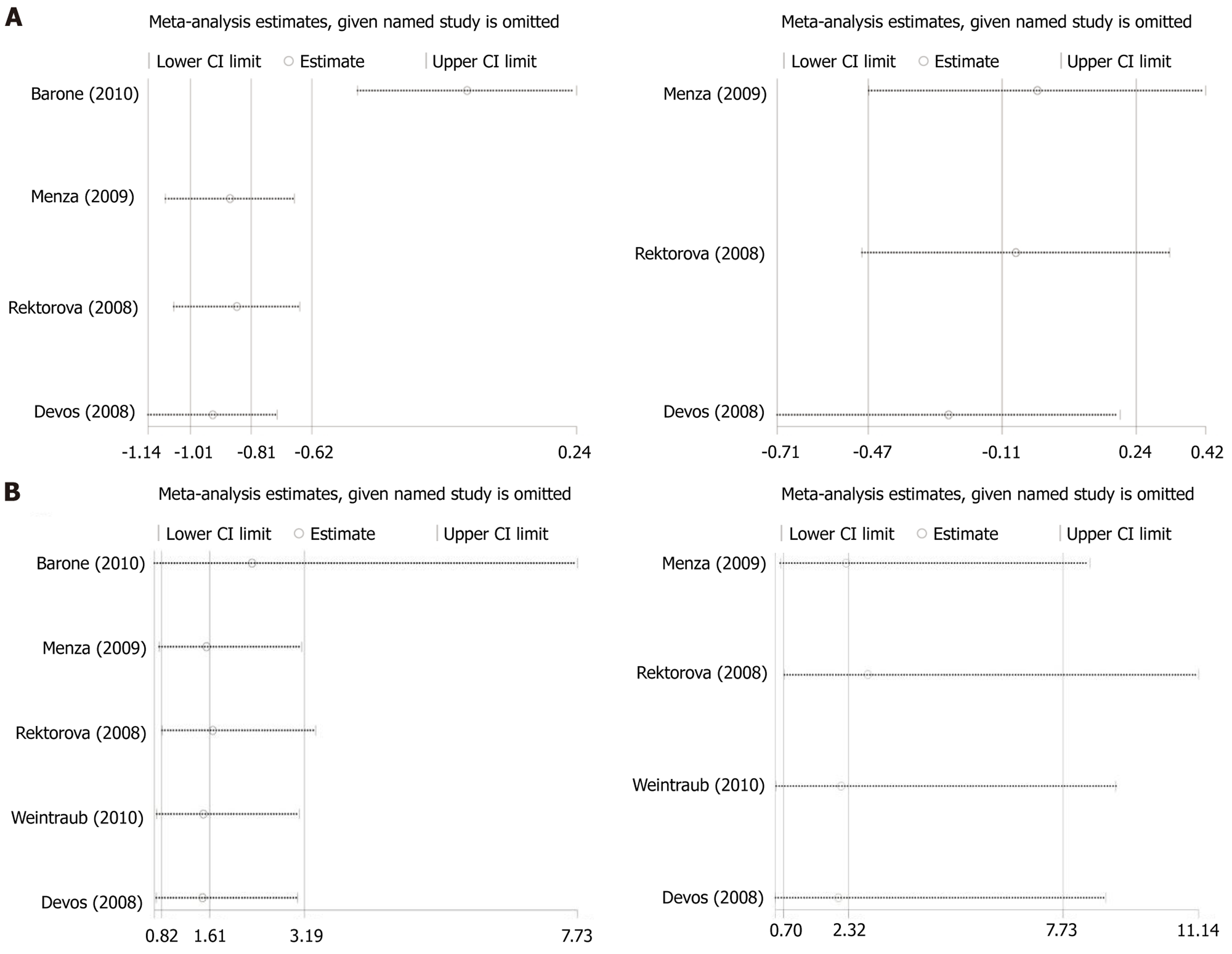
This study conducted heterogeneity tests for all primary outcome measures (Table 3) and sensitivity analyses to improve motor function and adverse events (Figure 3). In the sensitivity analysis of motor function, a 2008 study by Rektorova et al[12] identified the main factor affecting the stability of the pooled effect. When this study was excluded, the pooled effect size shifted from (MD = -0.75, 95%CI: -0.91 to -0.59) to (MD = -0.10, 95%CI: -0.47 to 0.27). Heterogeneity (I2) also decreased from 80.9% to 0%, indicating that this outcome was strongly influenced by the inclusion of this study. Therefore, caution is required when interpreting the pooled results for this indicator. Quality assessment further revealed methodological shortcomings in this study, with a Jadad score of only two points. This potential bias may stem from design features. According to the original report, a study by Rektorova et al[12] lacked clear blinding and allocation concealment. Along with the notable baseline variability among the treated participants, these factors may have contributed to the unstable estimates of the motor outcomes. As a result, this study had a substantial impact on the model and served as the main source of heterogeneity in the sensitivity analysis. In the sensitivity analysis for adverse events, the 2008 study by Rektorova et al[12] was again identified as a potential source of instability. However, unlike the motor function outcome, the exclusion of this study did not change the direction or statistical significance of the pooled effects of adverse events. This may be related to the low incidence of adverse events, the limited variation across studies, and the fact that this study reported few adverse events, most of which were mild. Thus, its influence on the final pooled results for adverse events was limited.
| Reproductive outcomes | Number of studies | WMD/RR 95%CI | P value | I2 |
| Improving depression | 3 | 1.35 (1.15-1.60) | < 0.001 | 0% |
| Quality of life | 2 | -2.41 (-3.02 to -1.79) | < 0.001 | 0% |
| Motor function | 4 | -0.75 (-0.91 to -0.59) | < 0.001 | 80.9% |
| Adverse event | 5 | 1.61 (0.82-3.19) | 0.169 | 0% |
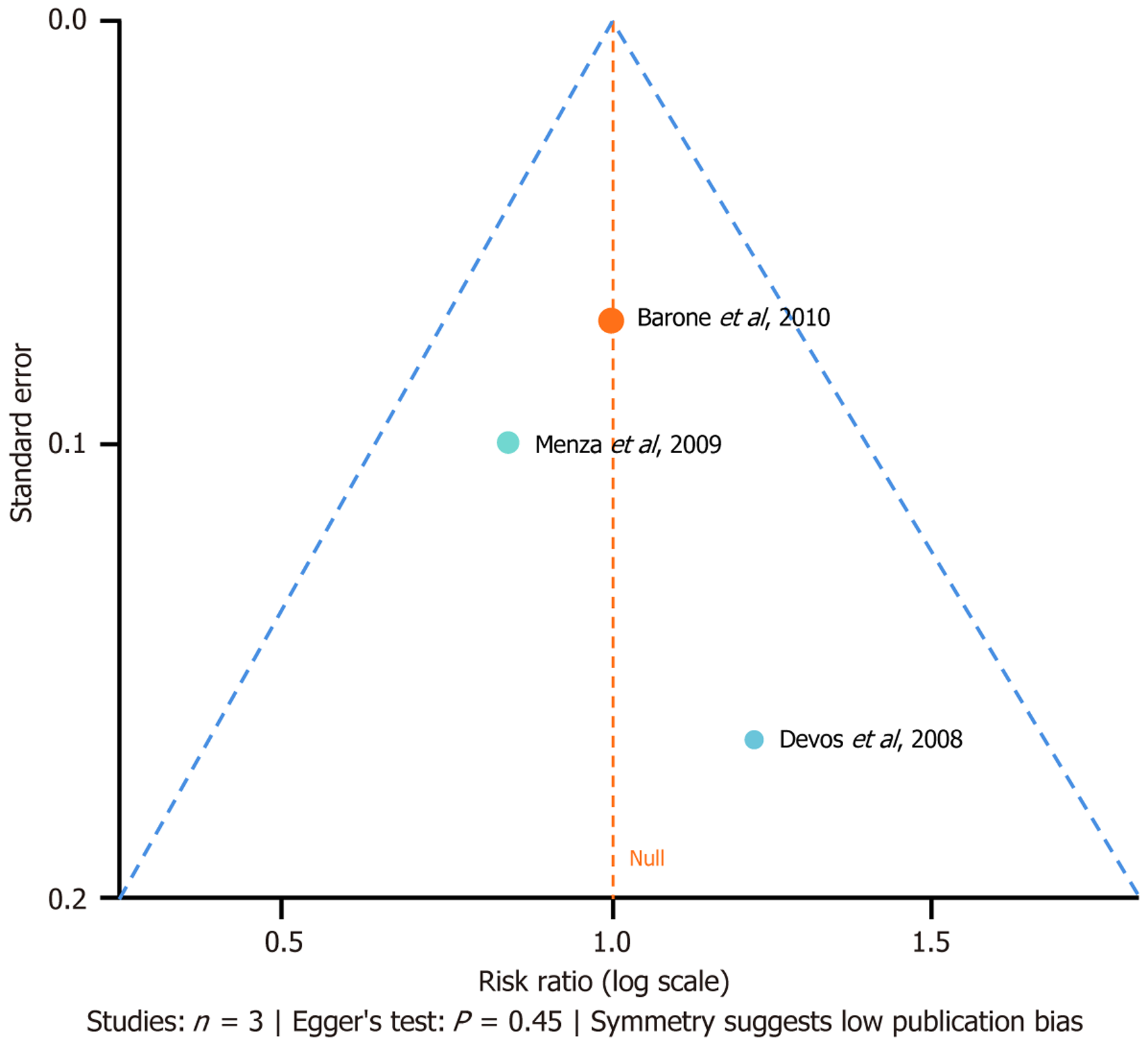
Owing to the weak comprehensive evaluation of the two research results, the funnel chart analysis based on the effective rate of treatment showed that the three studies with comprehensive results were symmetrically distributed, and there was no statistically significant Egger’s test result (P = 0.45). This indicates that the risk of publication bias in this meta-analysis was low and the results were reliable. The symmetry of the funnel plot reflects the coordination and consistency among the included studies, further supporting the stability and credibility of the conclusions of this study (Figure 4).
Given the limited number of included studies, this was a small-sample meta-analysis. Several methodological refi
Through the meta-analysis system and the integration of meta from five RCTs, the study focused on the efficacy and safety of dopaminergic-serotonergic pathway modulation (including dopaminergic agents, SSRIs, and their possible combinations) in the treatment of PD with depressive disorder. The core findings were that the combination strategy (or a specific combination of individual drugs) had clear advantages in improving depressive symptoms and overall quality of life without a significant additional impact on motor function. Moreover, safety is controllable, which provides a key basis for the clinical treatment of PD with depressive disorder and further verifies the core position of multi-neurotransmitter interaction imbalance in the pathological mechanism of PD with depressive disorder[15]. From the perspective of the core efficacy of improving depressive symptoms, we found that the efficacy rate of the combination (or single-drug synergy) of dopamine receptor agonists, represented by pramipexole, and SSRIs, represented by paroxetine, was sig
In addition, this study identified notable findings in the analysis of motor function indicators. In the initial pooled analysis, improvements in UPDRS-III reached statistical significance, with a pooled effect size of MD = -0.75, 95%CI: -0.91 to -0.59, suggesting that the intervention may provide some motor benefits. However, sensitivity analysis showed that the 2008 study by Rektorova et al[12] was classified as having a high risk of bias and may have introduced instability into the overall effect. After excluding this study, the effect size changed markedly to MD = -0.10, 95%CI: -0.47 to 0.27, and was no longer statistically significant. This substantial shift indicated that the motor function results were highly sensitive to the inclusion of a single low-quality study. Therefore, based on the evidence, we adopted a cautious interpretation of the conclusion that the intervention “improves motor function”. From a mechanistic standpoint, this finding is consistent with the current biological evidence. Recent studies have suggested that dopaminergic neurons in the SN pars compacta experience high basal metabolic and oxidative stress. Their high-energy demands, extensive axonal arborization, and dense synaptic terminals make them particularly vulnerable to aging, environmental insults, and genetic stressors[16]. As a result, in the intermediate or late stages of the disease, many dopaminergic neurons may sustain irreversible structural damage or functional loss. At this point, even pharmacological enhancement of dopaminergic levels or stimulation of dopaminergic receptors may be insufficient to restore normal motor circuit function[17,18]. Participants included in the analyzed trials had an average disease duration of approximately 6.6 years and a baseline UPDRS-III score of approximately 29, placing most in the mid-stage of Parkinson’s. At this stage, dopaminergic neuronal loss in the nigrostriatal pathway typically exceeds 50%, and structural degeneration is difficult to reverse with medication. Moreover, motor responsiveness to dopaminergic therapy tends to decline in mid-stage patients, further reducing the likelihood of obs
Improvement in the overall quality of life is an important endpoint of PD with depressive disorder treatment. In this study, we found that the intervention group was significantly superior to the control group in terms of PD quality of life questionnaire-39 score (MD = -2.30, 95%CI: -2.90 to -1.70). These results should be interpreted based on the factors influencing the quality of life of patients with PD. Previous studies have shown that the impaired quality of life of patients with PD is not only related to dyskinesia but also contributes to up to 40%-60% of the symptoms of depression, which indirectly deteriorates the quality of life by reducing the level of motivation of patients, aggravating subjective dysfunction (such as the decline in daily living ability), and sleep disorders[20]. In this study, the combination therapy directly relieved the emotional-related subjective pain by significantly improving the depressive symptoms (reducing rate of HAMD/MADRS ≥ 50%), and at the same time reduced the “amplification effect” of depression on motor function (such as the aggravation of subjective perception of motor delay caused by depression), thereby improving the quality of life. In terms of safety, the risk of dropping out owing to adverse events in the intervention group tended to increase (RR = 1.70), but this was not statistically significant (95%CI: 0.94-3.07), suggesting that the tolerance of the combination was generally controllable. Common adverse reactions, such as drowsiness and dizziness of dopaminergic agents (pra
From the perspective of neurobiological mechanism, the results of this study further verified the pathological hypo
Although dual modulation of dopamine and serotonin offers a plausible mechanistic explanation for the improvement of depressive symptoms, its clinical implications should be interpreted with caution. This is because several interventions in the five RCTs included in this study did not strictly represent “combination therapy with dopaminergic drugs and SSRIs”. Instead, most examined the effects of enhancing either the dopaminergic or the 5-HT system on depression in PD, providing only indirect evidence for a “dual-system modulation strategy”. Therefore, these findings cannot be compared with high-quality RCT based on fixed-combination regimens. The conclusions regarding “combination therapy stra
In summary, this meta-analysis clarifies the core value of dopaminergic-serotonergic pathway modulation in the trea
| 1. | Tobar A, Jaramillo AP, Costa SC, Costa KT, Garcia SS. A Physical Rehabilitation Approach for Parkinson's Disease: A Systematic Literature Review. Cureus. 2023;15:e44739. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 2. | Wang H, Dou S, Wang C, Gao W, Cheng B, Yan F. Identification and Experimental Validation of Parkinson's Disease with Major Depressive Disorder Common Genes. Mol Neurobiol. 2023;60:6092-6108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (1)] |
| 3. | Wang J, Zhang H. Serotonin mitigates depression in a rotenone-induced mouse Parkinson's disease model by inhibiting hippocampal neuronal pyroptosis and neuroinflammation. Behav Brain Res. 2025;490:115620. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 4. | Mathur N, Bhatt H, Lidstone SC. Neuropsychiatric Treatments for Parkinson's Disease: Nonpharmacological Approaches. Semin Neurol. 2022;42:158-167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 5. | Sandor C, Millin S, Dahl A, Schalkamp AK, Lawton M, Hubbard L, Rahman N, Williams N, Ben-Shlomo Y, Grosset DG, Hu MT, Marchini J, Webber C. Universal clinical Parkinson's disease axes identify a major influence of neuroinflammation. Genome Med. 2022;14:129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 6. | Mou YK, Guan LN, Yao XY, Wang JH, Song XY, Ji YQ, Ren C, Wei SZ. Application of Neurotoxin-Induced Animal Models in the Study of Parkinson's Disease-Related Depression: Profile and Proposal. Front Aging Neurosci. 2022;14:890512. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 7. | Vaidya B, Gupta P, Laha JK, Roy I, Sharma SS. Amelioration of Parkinson's disease by pharmacological inhibition and knockdown of redox sensitive TRPC5 channels: Focus on mitochondrial health. Life Sci. 2023;328:121871. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 20] [Reference Citation Analysis (0)] |
| 8. | Guo L, Hu H, Jiang N, Yang H, Sun X, Xia H, Ma J, Liu H. Electroacupuncture blocked motor dysfunction and gut barrier damage by modulating intestinal NLRP3 inflammasome in MPTP-induced Parkinson's disease mice. Heliyon. 2024;10:e30819. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 9. | Wang XL, Feng ST, Wang YT, Chen B, Wang ZZ, Chen NH, Zhang Y. Comparative efficacy and acceptability of drug treatments for Parkinson's disease with depression: A systematic review with network meta-analysis. Eur J Pharmacol. 2022;927:175070. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 10. | Barone P, Poewe W, Albrecht S, Debieuvre C, Massey D, Rascol O, Tolosa E, Weintraub D. Pramipexole for the treatment of depressive symptoms in patients with Parkinson's disease: a randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2010;9:573-580. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 442] [Cited by in RCA: 351] [Article Influence: 21.9] [Reference Citation Analysis (0)] |
| 11. | Menza M, Dobkin RD, Marin H, Mark MH, Gara M, Buyske S, Bienfait K, Dicke A. A controlled trial of antidepressants in patients with Parkinson disease and depression. Neurology. 2009;72:886-892. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 325] [Cited by in RCA: 258] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 12. | Rektorova I, Balaz M, Svatova J, Zarubova K, Honig I, Dostal V, Sedlackova S, Nestrasil I, Mastik J, Bares M, Veliskova J, Dusek L. Effects of ropinirole on nonmotor symptoms of Parkinson disease: a prospective multicenter study. Clin Neuropharmacol. 2008;31:261-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 56] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 13. | Weintraub D, Mavandadi S, Mamikonyan E, Siderowf AD, Duda JE, Hurtig HI, Colcher A, Horn SS, Nazem S, Ten Have TR, Stern MB. Atomoxetine for depression and other neuropsychiatric symptoms in Parkinson disease. Neurology. 2010;75:448-455. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 203] [Cited by in RCA: 178] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 14. | Devos D, Dujardin K, Poirot I, Moreau C, Cottencin O, Thomas P, Destée A, Bordet R, Defebvre L. Comparison of desipramine and citalopram treatments for depression in Parkinson's disease: a double-blind, randomized, placebo-controlled study. Mov Disord. 2008;23:850-857. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 177] [Cited by in RCA: 179] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 15. | Zhao Q, Zhang X, Zhang J, Zhang Y, Jia L, Guo S, Zhang M, Wang H, Wang Y, Guan Y, Zhang Y, Miao S, Zhu JX, Ma H. Reduction of D2 receptors on microglia leads to ZBP1-mediated PANoptosis of mPFC in Parkinson's disease depression mice. Int Immunopharmacol. 2025;158:114809. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 16. | Flores-Ponce X, Velasco I. Dopaminergic neuron metabolism: relevance for understanding Parkinson's disease. Metabolomics. 2024;20:116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 23] [Reference Citation Analysis (0)] |
| 17. | Li X, Dong ZY, Dong M, Chen L. Early dopaminergic replacement treatment initiation benefits motor symptoms in patients with Parkinson's disease. Front Hum Neurosci. 2024;18:1325324. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 18. | Tang R, Zhang S, Sun R, Xu J, Hu Y, Tan H, Chen L. Continuous Dopaminergic Stimulation-Based Levodopa Treatment in Patients with Early to Mid-Stage Parkinson's Disease: A Systematic Review and Meta-Analysis. Neurol Ther. 2025;14:1197-1208. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 19. | Morales-García JA. Modeling neurodegeneration and neuroinflammation in Parkinson's Disease: Animal-based strategies. Methods Cell Biol. 2025;197:81-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 20. | Kakimoto A, Kawazoe M, Kurihara K, Mishima T, Tsuboi Y. Impact of non-motor fluctuations on QOL in patients with Parkinson's disease. Front Neurol. 2023;14:1149615. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 16] [Reference Citation Analysis (0)] |
| 21. | Izquierdo-Altarejos P, Arenas YM, Martínez-García M, Vázquez L, Mincheva G, Doverskog M, Blackburn TP, Bohnen NI, Llansola M, Felipo V. Golexanolone reduces glial activation in the striatum and improves non-motor and some motor alterations in a rat model of Parkinson's disease. Front Aging Neurosci. 2024;16:1417938. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 22. | Si Q, Gan C, Shan A, Sun H, Cao X, Ye S, Shi J, Wan C, Wang X, Yuan Y, Zhang K. Cholinergic basal forebrain atrophy and cortical alterations in Parkinson's disease with apathy. Parkinsonism Relat Disord. 2025;134:107793. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 23. | Zhang CG, Zhang Y, Xu K, Wang S, Bai Y. Correlation of inflammatory markers with depression and sleep disorders accompanying the prodromal stage of Parkinson's disease. World J Psychiatry. 2025;15:99901. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 24. | Samanta S, Bisht M, Kanimozhi M, Kumar N, Handu SS. Association of depression with disease duration, quality of life and adherence in Parkinson's disease: A cross sectional study. J Family Med Prim Care. 2023;12:1406-1411. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 25. | Guo M, Liu H, Gao L, Yu H, Ren Y, Li Y, Yang H, Cao C, Fan G. A radiomics approach for predicting gait freezing in Parkinson's disease based on resting-state functional magnetic resonance imaging indices: A cross-sectional study. Neural Regen Res. 2026;21:1621-1627. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (2)] |
| 26. | Ou R, Lin J, Liu K, Jiang Z, Wei Q, Hou Y, Zhang L, Cao B, Zhao B, Song W, Shang H. Evolution of Apathy in Early Parkinson's Disease: A 4-Years Prospective Cohort Study. Front Aging Neurosci. 2020;12:620762. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |