Published online May 19, 2026. doi: 10.5498/wjp.v16.i5.115794
Revised: December 14, 2025
Accepted: January 15, 2026
Published online: May 19, 2026
Processing time: 186 Days and 6.1 Hours
Major depressive disorder (MDD) is one of the most prevalent psychiatric il
Core Tip: Major depressive disorder is one of the most prevalent psychiatric illnesses worldwide which impairs the social functioning of the afflicted patients. Astrocytes play a role in the maintenance of the function of the central nervous system, both physiologically and pathologically. Increasing evidence suggests that the number, volume and function of astrocytes are altered in the depressed brain. The glial fibrillary acidic protein and S100β protein, which are closely associated with astrocytes, also show significant alterations in patients with major depressive disorder. Astrocyte-derived glial cell line-derived neurotrophic factor not only exerts neuroprotective effects in the central and peripheral nervous systems but also plays a role in depressive symptomatology and the pharmacological action of antidepressant treatments.
- Citation: Jing P, Sun C, Yin XP, Liu XC, Tian Q, Zhang XB. Role of astrocytes in the development and progression of major depressive disorder. World J Psychiatry 2026; 16(5): 115794
- URL: https://www.wjgnet.com/2220-3206/full/v16/i5/115794.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i5.115794
Major depressive disorder (MDD) is a type of mood disorder characterized primarily by significant and persistent low mood, slowed thinking, reduced volitional activity, and physical discomfort. Its main clinical manifestations include depressed mood, lack of interest, and anhedonia, accompanied by symptoms such as decreased appetite, early awake
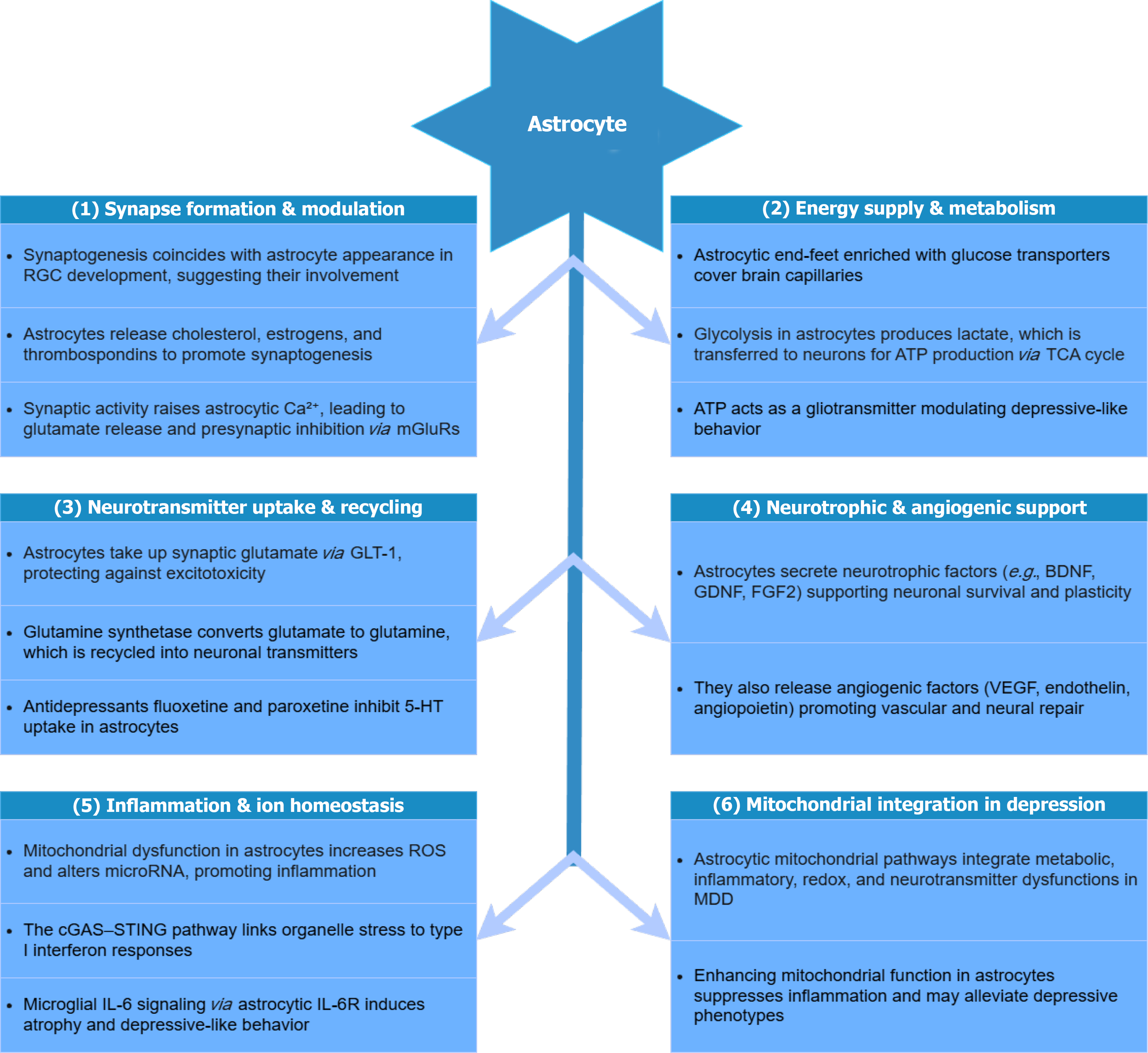
Astrocytes are the predominant type of glial cells. Alongside neurons in the nervous system, there exists a vast population of glial cells that are widely distributed throughout both the central and peripheral nervous systems. Their number far exceeds those of neurons, constituting nearly 90% of all cells in the adult brain. Glial cells in the central nervous system (CNS) include astrocytes, oligodendrocytes, and microglia, with astrocytes constituting the majority and encompassing nearly all functions attributed to glial cells. In recent years, extensive research has been conducted on the morphology and functions of glial cells, with particularly significant advances in the study of astrocytes. For a long time in neurobiological research, astrocytes were considered secondary players, primarily responsible for providing structural support, nourishing neurons, and clearing excess ions and neurotransmitters from synaptic clefts. The term “glia”, derived from the Greek word for “glue”, reflects this historical view of their passive, supportive role. However, in 1997, Pfrieger and Barres[4] first reported that glial cells strongly enhance synaptic connectivity between neurons, marking a new era in understanding the glial function. Advancements in neuroscience research techniques and deeper investigations into astrocytes have demonstrated their crucial roles in nervous system development, synaptic transmission, neural tissue repair and regeneration, and neuroimmunity[5,6]. Astrocytic dysfunction has been implicated in several neurological diseases, including neuropsychiatric disorders such as MDD[7-11]. It contributes to the molecular pa
For a long time, it was believed that synapse formation was solely related to neurons. However, recent studies have demonstrated that astrocytes also play a crucial role in this process[17,18]. For example, during normal development, most retinal ganglion cells extend their axons to the superior colliculus by embryonic day 16 in mice, but significant synapse formation begins only after the appearance of astrocytes nearly postnatal day 7[19]. This temporal correlation between astrocyte maturation and synapse formation suggests a potential connection between the two.
In the CNS, astrocytes guide synapse formation by releasing chemical factors, such as cholesterol, estrogens, and thrombospondins, thereby significantly increasing synapse numbers. Moreover, astrocytes themselves are essential components for synaptic function[20]. Research indicates that astrocytes play a vital role in supporting synaptic activity and promoting synapse maturation. When astrocytes are removed from the culture medium, preformed synapses on retinal ganglion cells fail to mature further, and their numbers decrease to one-fourth of the original count.
Astrocytes also regulate synaptic transmission. It was previously believed that synaptic connections could form solely between the presynaptic and postsynaptic membranes. However, recent studies have found that astrocytes are also a necessary component for synapse formation in the CNS. Astrocytes envelop synaptic terminals, enabling effective communication with synapses. Synaptic activation can trigger responses in astrocytes, which, by taking up glutamate, help modulate the duration of synaptic currents and prevent potential excitotoxic damage.
During high-frequency stimulation, glutamate activates glutamatergic receptors on astrocytes, leading to an increase in intracellular Ca2+ concentration. This, in turn, triggers the release of glutamate from astrocytes and negatively regulates neuronal glutamate release through metabotropic glutamate receptor dependent presynaptic inhibition. An additional effect of elevated Ca2+ levels in astrocytes is the propagation of Ca2+ waves to neighboring astrocytes through gap junctions, allowing signal modulation to spread to nearby synapses[21,22]. Accordingly, astrocytes have the potential to regulate synaptic transmission and synaptic plasticity, mechanisms that are also implicated in patients with MDD[23,24].
Astrocytes possess glucose transporter-rich end-feet that cover all capillary walls in the brain. Through these end-feet, glucose is supplied to meet the energy demands of both glial cells and neurons[25]. Within astrocytes, glucose undergoes glycolysis, providing energy for the conversion of intracellular glutamate to glutamine and for maintaining the Na+ gradient through Na+/K+-ATPase activity[26,27]. Astrocytes then release lactate into neurons, where it enters the tricarboxylic acid cycle and generates ATP through oxidative phosphorylation[28]. In astrocytes, glutamate-mediated synaptic activity is coupled with glucose uptake and utilization[29]. Functional magnetic resonance imaging and positron emission tomography studies allow the observation of signals collectively generated by glucose uptake, glycolysis, and oxidative phosphorylation[30,31]. ATP is an essential gliotransmitter involved in astrocytic modulation of depressive-like behavior[32]. Astrocytes, particularly the astrocyte mitochondrial melatonergic pathway, have been identified as crucial hubs for integrating the wide array of biological underpinnings of MDD[33,34].
Cell culture and in vitro studies have revealed that astrocytes express all the receptors and ion channels found in neuron[35], including key transporters crucial for glutamate uptake at synapses and neuroprotection[36]. A vital function of astrocytes is the uptake of synaptic glutamate through their transporters. By absorbing glutamate, astrocytes terminate its postsynaptic action, reduce extracellular glutamate levels, and play a significant role in neuronal protection[37]. When astrocytes are co-cultured with neurons in vitro, they help protect neurons from death by maintaining low extracellular glutamate levels[38].
Furthermore, mice lacking the glutamate transporter-1 gene exhibit pronounced excitotoxic damage, further demonstrating the protective role of glutamate transport[36]. Antisense knockdown of glutamate transporters in rat brain astrocytes exacerbates neuronal injury[39]. Another glutamate-related function of astrocytes involves the enzyme glutamine synthetase (unique to astrocytes), which converts glutamate into glutamine. The released glutamine is taken up by neuronal terminals and reconverted into glutamate or gamma-aminobutyric acid, replenishing the neurotransmitter pool.
Astrocytes regulate extracellular neurotransmitter levels through specific transporters, modulating both synaptic availability and reuptake. In vitro studies have confirmed that astrocytes can absorb gamma-aminobutyric acid, 5-hydroxytryptamine (5-HT), dopamine (DA), norepinephrine, and histamine, as well as enzymes such as catechol-O-methyltransferase and monoamine oxidase, which further modulate extracellular neurotransmitters[40-48]. Moreover, antidepressants such as fluoxetine and paroxetine can block 5-HT uptake in cultured astrocytes, suggesting that both astrocytes and neurons may contribute to the therapeutic effects of antidepressants in MDD[49-51]. Reduced prefrontal glutamatergic function has been associated with decreased expression of astrocyte-related genes in treatment-resistant depression[52].
Astrocytes synthesize and release various neurotrophic factors and cytokines crucial for maintaining neuronal function, including nerve growth factor, brain-derived neurotrophic factor (BDNF), glial cell line-derived neurotrophic factor (GDNF), fibroblast growth factor 2, and neurotrophins 3, 4, and 5[53-55]. These factors support neuronal survival, growth, differentiation, synaptic plasticity, and efficacy[56]. Reactive astrocytes can also secrete molecules involved in glia-mediated angiogenesis, neuroregeneration, and neuronal precursor migration, such as endothelin, vascular en
Although the exact level of trophic support required for the survival of mature neurons remains unclear, deficiencies in these factors can increase susceptibility to cell death or damage[58]. Preclinical and postmortem human studies strongly suggest that reduced levels of neurotrophic factors, such as BDNF and fibroblast growth factor 2, and impaired neuroplasticity contribute significantly to the pathophysiology of MDD[59-63]. Antidepressant drugs and electroconvulsive therapy have been revealed to enhance BDNF expression in both glial cells and neurons and to stimulate the production of other neurotrophic and angiogenic factors, highlighting glial cells as a potential target for antidepressant treatment[64-66].
Estrogen contributes to the maintenance and restoration of normal brain function and promotes synaptic plasticity, serving as a neuroprotective factor in the CNS[67]. It exerts its neuroprotective effects either directly on neurons or indirectly through glial mediation. In vitro studies have identified estrogen receptors in astrocytes[68]. The absence of glial cells can impair estrogen-mediated neuroprotection, rendering neurons more vulnerable to neurotoxicity. Cell culture studies have demonstrated that estrogen upregulates the expression of glutamate transporters in astrocytes, thereby reducing extracellular glutamate levels and protecting neurons from glutamate-induced excitotoxicity[69]. In ovariectomized rats, estradiol has been revealed to upregulate mRNA and protein levels of glutamine synthetase in the hypothalamus and hippocampus, highlighting the role of astrocytes in hormone-regulated glutamatergic neurotransmission[70]. Consequently, the observed reduction in astrocyte numbers in postmortem brain tissue from patients with MDD may be linked to impaired estrogen-mediated neuroprotection.
Astrocytes are associated with neurodegenerative diseases, inflammatory processes, and regeneration[71,72]. Upon encountering injury, astrocytes become activated, leading to increases in cell number and size, as well as alterations in gene expression patterns, most notably, a marked upregulation of glial fibrillary acidic protein (GFAP). Activated astrocytes may play crucial roles in both inhibiting and promoting damage[73,74]. For example, astrocytes can form glial scars that prevent axonal regeneration; however, these scars may also help limit the spread of injury. Increased astrocyte proliferation does not necessarily indicate enhanced neurodegeneration but may instead reflect nonspecific repair processes that require glial activation. In response to injury, activated astrocytes also release neurotrophic factors that promote neuronal survival and repair.
Astrocytes maintain the neuronal microenvironment at an optimal level by regulating ion concentrations and metabolic substrates. Through gap junctions, they establish connections with numerous CNS cells, allowing the passage of small ions and molecules, such as K+, Ca2+, lactate, glucose, and amino acids. Additionally, astrocytes regulate intracellular pH, electrolyte balance, and extracellular space volume in the CNS through Na+/K+-ATPase activity on their cell membranes[75]. The rapid-acting antidepressant ketamine may exert excitatory effects on neurons by influencing the ability of astrocytes to regulate extracellular K+, thereby alleviating depressive symptoms[76]. One study identifies that the selective knockdown of interleukin-6 (IL-6) in microglia and IL-6 receptors in astrocytes effectively mitigates depression-like behaviors and reduces astrocyte atrophy. Thus, the microglial IL-6 appears to be a key factor contributing to astrocyte apoptosis and depressive symptoms[77].
Recent studies have indicated that astrocyte-mediated inflammation in MDD not only leads to elevated cytokine levels but also involves intracellular and intercellular signaling axes that can impact metabolic homeostasis. For instance, novel findings reveal that the cyclic GMP-AMP synthase-stimulator of interferon genes pathway, acting as a bridge between organelle stress and innate immune responses, links chronic stress signals from mitochondria and the endoplasmic reticulum to type I interferon responses. This finding suggests that chronic organelle stress may progressively transform reversible astrocytic responses into irreversible, inflammation-driven loss of ion regulatory capacity[78,79].
Collectively, these findings suggest that future research should move beyond simple enumeration of cytokine levels to explore how specific inflammatory signaling pathways disrupt astrocytic ion homeostasis and metabolic coupling functions in distinct brain regions and across genders. A research shift of this nature will facilitate a more thorough elucidation of the intrinsic link between neuroinflammation and neural circuit dysfunction in MDD.
A multitude of studies have demonstrated that mitochondrial metabolism and signaling pathways in astrocytes play a pivotal integrative role in the pathology of MDD, thereby linking metabolic, redox, inflammatory, and neurotransmitter-related dysfunctions[34]. Specifically, the inhibition of astrocyte-associated mitochondrial metabolic and signaling pathways results in impaired mitochondrial function, increased reactive oxygen species, and reactive oxygen species-dependent microRNA alterations, thereby promoting a pro-inflammatory phenotype in astrocytes[80]. Conversely, enhancing mitochondrial metabolism and signaling pathways in astrocytes has been shown to improve mitochondrial function and suppress inflammation. This provides a key mechanistic pathway for understanding MDD. This pathway translates intracellular metabolic and inflammatory dysregulation into neural circuit dysfunction and associated behavioral phenotypes[81].
Gender has been identified as a critical factor that modulates the risk of depression and the associated neurobiological responses. The lifetime prevalence of MDD is significantly higher in women than in men, with this disparity emerging around adolescence. This discrepancy is presumably attributable to the synergistic effect of biological factors, including sex hormones, and psychosocial elements[82]. Astrocytes, in particular, have been observed to demonstrate gender disparities not only at rest but also in response to stress or hormonal stimulation.
Recent studies have revealed gender and brain region specific transcriptional and morphological characteristics of astrocytes[83], which may partially explain gender disparities in depression susceptibility. With regard to specific mechanisms, estrogen and other gonadal steroids have been demonstrated to directly regulate astrocytic inflammatory responses, glutamate metabolism, and neurotrophic factor expression. The cyclical or phasic fluctuations of these hormones have been shown to correlate with periods of heightened depression risk. This suggests the possibility that they may influence gender-specific manifestations of depression through interactions with astrocytes[84,85]. Conse
Several postmortem cell-counting studies on brain tissues of patients with MDD have revealed a significant reduction in the density and number of Nissl-stained astrocytes[86]. Moreover, compared with age-matched healthy controls, ultrastructural changes and alterations in the size of astrocyte nuclei have been observed in patients with MDD[87]. The decrease in astrocyte numbers has been primarily identified in specific frontal-limbic brain regions, such as the ventral anterior cingulate cortex (Brodmann area 24)[88], the dorsolateral prefrontal cortex (Brodmann area 9)[89-91], the pregenual anterior cingulate cortex[92], and the orbitofrontal cortex[90]. In contrast, some studies have reported an increased density of astrocytes in the hippocampal cornu ammonis region and the granular cell layer of the dentate gyrus in patients with MDD[93]. This increase in astrocyte density may be associated with a reduction in astrocytic processes rather than a proliferation of astrocytes[94]. Additionally, a significant decrease in astrocyte numbers has been found in the amygdala of patients with MDD and those with untreated bipolar disorder[95]. Suicidal behaviors in individuals with affective disorder may be related to astrocyte loss[96].
Beyond astrocyte density, the volume and morphology of astrocytes are also affected in patients with mood disorders[97]. Among six studies investigating astrocyte somatic volume in patients with MDD or bipolar disorder, three reported a significant enlargement of astrocyte cell bodies in the dorsolateral prefrontal cortex of both patient groups, as well as in the anterior cingulate cortex of patients with MDD, compared with non-psychiatric controls[98,99]. However, the other three studies found no changes in astrocyte somatic volume[100]. Furthermore, alterations in the roundness of astrocyte nuclei have been observed in the dorsolateral prefrontal cortex of patients with bipolar disorder[101]. The decrease in astrocyte density occurs alongside an increase in astrocyte nuclear volume, suggesting possible compensatory mecha
Neuroimaging studies in humans have demonstrated selective hippocampal volume reduction in stress-related psychiatric disorders such as depression[103], a finding that has been replicated in chronic animal stress models[104]. However, the exact mechanism underlying hippocampal volume loss remains unclear. Massive neuronal loss due to repeated exposure to high cortisol levels is unlikely, as postmortem analyses of brain tissues from patients with MDD, steroid-treated individuals, and primates have revealed no significant cell loss or neuropathological changes[105,106]. Studies in chronic stress animal models suggest that hippocampal volume correlates with astrocyte number and cellular morphology. Stress-induced reductions in astrocyte number and size may contribute to hippocampal shrinkage. Notably, fluoxetine has been indicated to reverse these changes, helping maintain normal hippocampal volume in rats[107]. Moreover, one study found that electroacupuncture can prevent the reduction of astrocyte volume, thereby improving depressive-like behaviors in mice[108].
GFAP is an acidic protein with a molecular weight of 50-52 kDa and is a major structural component of astrocytes. Rich in glutamate and aspartate, GFAP exists in glial cells in two forms, intermediate filament protein and soluble protein, and functions as a cytoskeletal element in astrocytes. Its expression increases when astrocytes are stimulated or activated, making GFAP a specific marker for astrocytes and a recognized indicator of astrocyte activation[109]. Elevated GFAP levels in activated astrocytes may have neuroprotective effects. As a reliable marker for astrocyte activity, the intensity of GFAP immunohistochemical staining can reflect the degree of cellular activation. Studies have revealed reduced GFAP immunohistochemical staining in the hippocampus of patients with MDD and stress-induced animal models[10,110]. In the frontal cortex of patients with MDD, decreased gene expression has been observed for GFAP, a specific astrocytic protein, as well as for glutamate transporters in glial cells and glutamine synthetase[111]. Previous clinical studies have demonstrated increased GFAP serum levels in patients with MDD, with a trend toward higher levels correlating with increased severity of depression episodes according to Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition criteria[112]. Serum GFAP may aid in the differential diagnosis of MDD and allow for objective quantification of depression severity. Furthermore, it may serve as a marker for monitoring astroglial pathology throughout the course of MDD[108]. In animal models, treatment with the classical antidepressant fluoxetine or electroacupuncture has been indicated to normalize astrocytic morphology in the prefrontal cortex and alleviate depressive-like behaviors[113].
A substantial body of evidence from human MDD studies supports the hypothesis that GFAP alterations measured in brain tissue primarily reflect changes in parenchymal astrocytes rather than peripheral glial cells. Initially, studies have demonstrated that GFAP immunoreactivity colocalizes with the typical morphology of cortical astrocytes at the protein and morphological levels. Its expression pattern correlates with the downregulation trend of astrocyte-specific markers (e.g., aquaporin-4, connexin 43), and cell density analysis confirms that GFAP-positive cells are highly correlated with astrocyte counts while showing no association with other glial cell types[114,115]. Secondly, at the molecular transcriptional level, studies employing single-nucleus RNA sequencing in postmortem prefrontal cortex tissue have demon
S100β is a 21 kDa acidic calcium-binding protein abundantly present in the CNS, where it performs various biological functions. Under physiological conditions, S100β is primarily produced by astrocytes and acts on neurons and their surrounding microenvironment. In contrast to other astrocytic proteins, excessively high levels of S100β can exert neurotoxic effects[120]. Elevated levels of S100β in cerebrospinal fluid and serum have been consistently correlated with MDD in unmedicated patients, serving as a potential state marker. Some studies have also revealed that S100β levels correlate with symptom severity and that their normalization may predict a successful treatment response[121-123]. Elevated serum S100β levels have been observed in patients with MDD and those with untreated bipolar disorder, whereas antidepressant treatment has been demonstrated to reduce these levels[124,125]. Blood and cerebrospinal fluid levels of S100β are consistently elevated in patients with MDD, and a significant positive correlation has been found between serum S100β levels and Hamilton Depression Rating Scale scores in patients with MDD[126,127]. Conversely, although S100β is highly expressed in astrocytes and correlates with disease status and treatment efficacy in MDD, interpreting it as a specific astrocyte-derived marker requires mechanistic support. This phenomenon is primarily attributable to the multifactorial and complex nature of its release. S100β can be released via exocytosis[128] or passively released during cellular injury or disruption of the blood-brain barrier[129]. Additionally, under certain conditions, its origin can also be traced to non-astrocytic sources[130]. In summary, in research and clinical practice, the interpretation of S100β or GFAP levels in bodily fluids, such as cerebrospinal fluid or serum, should integrate cell-type-specific detection techniques with assessments of blood-brain barrier integrity.
In 1993, Lin et al[131] isolated, purified, and successfully cloned glial cell line-derived GDNF from the rat B49 glial cell line. The human GDNF gene is located at 5p13.1-13.3 and shares 93% amino acid sequence homology with rat GDNF. GDNF is synthesized as a precursor and then processed into a mature 134-amino acid protein before secretion. It contains seven conserved cysteine residues and has a structural arrangement similar to that of the transforming growth factor-β (TGF-β) superfamily, classifying it as a member of this family. GDNF exerts its neurotrophic effects through two receptor subunits: GDNF family receptor alpha (GFR-α) and the receptor tyrosine kinase (Ret). GDNF first binds to GFR-α, forming a GDNF-GFR-α complex, which then recruits Ret to form a ternary complex. This interaction induces Ret dimerization and autophosphorylation of tyrosine residues, thereby initiating downstream signal transduction pathways[132].
GDNF promotes the survival of CNS neurons, including midbrain dopaminergic neurons, motor neurons, and norepinephrine neurons in the locus coeruleus. It also supports peripheral neurons, such as sympathetic and parasympathetic neurons, as well as sensory and enteric neurons. GDNF is the most potent neurotrophic factor identified for cholinergic motor neurons, enhancing their survival and acetylcholinesterase activity in cultured or developing neurons[133]. During neuronal development, GDNF acts synergistically with TGF-β2 and TGF-β3 to provide specific survival signals that guide DA neuron axon migration. In vitro studies, GDNF enhances the growth and differentiation of fetal midbrain DA neurons and increases astrocyte proliferation. Following brain injury, GDNF mRNA expression is altered, kainic acid- or pilocarpine-induced neuronal damage in rats leads to seizures and upregulates GDNF mRNA in the hippocampus, striatum, and cortex, suggesting a neuroprotective role in injury responses.
GDNF is one of the most potent factors for dopaminergic neurons and reveals therapeutic potential in Parkinson’s disease. It improves DA metabolism in the substantia nigra and striatum, repairs neurotoxin-damaged dopaminergic neurons, and enhances neuronal survival and synaptic density in Parkinson’s disease animal models, thereby reducing motor deficits[134].
GDNF knockout mice die shortly after birth, exhibiting deficits in neuronal and neural crest cell differentiation, loss of autonomic, trigeminal, and spinal motor neurons, complete absence of enteric neurons, and kidney malformations[135]. In embryonic kidneys, GDNF functions as a mesenchyme-derived signal that promotes ureteric bud branching. It also regulates spermatogonial differentiation, with its levels in the testes determining the fate of undifferentiated spermatogonia[136].
GDNF knockout mice exhibit abnormal hippocampal synaptic transmission[137]. Other studies have also demonstrated the significant role of GDNF in cognitive dysfunction and drug dependence[138,139]. Both GDNF and its receptors are highly expressed in the hippocampus and prefrontal cortex, brain regions critically involved in the pathogenesis and treatment of MDD[140]. In vitro studies using C6 glioma cell lines and cultured rat astrocytes have indicated that 5-HT, antidepressants, and mood stabilizers (such as lithium and valproate) significantly increase GDNF secretion[141-144]. While numerous biological factors and pharmacological agents can modulate GDNF expression, the intracellular regulatory pathways remain unclear[145]. Notably, research has revealed that blocking the extracellular signal-regulated protein kinase system can partially attenuate fluoxetine-induced upregulation of GDNF mRNA expression in astrocytes, highlighting the crucial role of extracellular signal-regulated protein kinase in cell growth and survival[146].
Animal studies have revealed that antidepressants do not significantly affect the mRNA expression of GDNF and its receptors, whereas mood stabilizers such as lithium can increase GDNF protein levels in the brains of depressed rat models[147]. In animal models of electroconvulsive therapy, electroconvulsive stimulation was found to reduce GDNF concentrations in the hippocampal and striatal regions of rats[148]. However, other studies reported that while electroconvulsive stimulation exhibited no significant effect on GDNF mRNA expression in the rat hippocampus, it markedly increased the mRNA expression of GDNF receptors.
Takebayashi et al[149] investigated serum GDNF levels in patients with partial or complete remission of MDD and bipolar disorder. They found that the serum GDNF levels in these patients were significantly lower than those in the normal control group. In contrast, Rosa et al[150] found that during manic or depressive episodes in patients with bipolar disorder, serum GDNF protein concentration increased. In two recent studies, Otsuki et al[151] reported that during depressive episodes in patients with MDD, GDNF mRNA expression in peripheral blood decreased, whereas, during remission, expression levels did not differ significantly from those of healthy controls. Michel et al[152] found that GDNF concentrations in the parietal cortex of patients with MDD were significantly higher than those in matched controls, whereas GDNF levels in the hippocampus were decreased. Liu et al[153] found that reduced plasma levels of GDNF may serve as a valuable diagnostic marker for first-episode MDD. Moreover, their preliminary finding suggests that baseline plasma GDNF levels could have prognostic value, higher levels may predict a better response to antidepressant treatment in patients with first-episode MDD.
It is noteworthy that GDNF expression demonstrates a high degree of pleiotropy and complexity. In conditions involving neuroinflammation or injury, both reactive astrocytes and microglia have been observed to induce GDNF production. Additionally, peripheral glial cells, such as Schwann cells, have been identified as sources under specific pathological conditions[154,155]. Furthermore, GDNF can be transported via extracellular vesicles[156], providing a mechanistic basis for intercellular communication and its detection in bodily fluids. Consequently, in the context of MDD research, the attribution of GDNF level changes to a particular cell type demands the integration of cell-resolution techniques with meticulous brain region and clinical stratification analyses[152].
Astrocytes perform complex functions in both developing and mature brains, including generating neuronal precursors, supporting neuronal metabolism, regulating neurotransmitter conversion and release, facilitating vascular remodeling, and controlling cerebral blood flow. Dysfunction or astrocytes in the number and morphology in emotion-related brain regions may contribute to MDD pathogenesis or progression.
Although no definitive neuropathological or neurodegenerative changes have been identified in the brains of patients with MDD, accumulating evidence suggests that astrocyte numbers are reduced in the frontal cortex of patients with MDD and those with bipolar disorder compared to non-psychiatric controls. Future research should aim to elucidate the factors that influence astrocyte proliferation and differentiation during brain development, depressive episodes, and disease progression, as well as the role of other glia-derived factors. Furthermore, it is crucial to ascertain whether alterations in glial cell lineage markers (such as GDNF, S100β, and GFAP) reflect astrocyte-specific dysfunction or broader glial and vascular reactivity. This is necessary for distinguishing depression-state-related astrocytic changes from stable characteristics. In addition, mounting evidence indicates that astrocytic reactivity exhibits gender and region-specific patterns, emphasizing the importance of stratified experimental designs. The integration of studies of metabolic, inflammatory, and neurotrophic pathways within this framework has the potential to identify astrocyte-targeted intervention strategies, thereby enhancing precision in depression treatment. Such studies may help determine whether therapies targeting glia-derived factors can alleviate depressive symptoms. Additionally, further investigation is needed to clarify whether changes in astrocyte physiology and number are state markers, present only during depressive episodes, or trait markers that persist even during remission.
We would like to express our gratitude to all the doctors and nurses of the Affective Disorders Research Team at the Affiliated Kangning Hospital of Ningbo University, who participated in this study. We also sincerely thank Jin Li, Wen-Xi Sun, and Peng Chen from Suzhou Guangji Hospital for their support.
| 1. | Marx W, Penninx BWJH, Solmi M, Furukawa TA, Firth J, Carvalho AF, Berk M. Major depressive disorder. Nat Rev Dis Primers. 2023;9:44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 543] [Cited by in RCA: 426] [Article Influence: 142.0] [Reference Citation Analysis (1)] |
| 2. | Huang Y, Wang Y, Wang H, Liu Z, Yu X, Yan J, Yu Y, Kou C, Xu X, Lu J, Wang Z, He S, Xu Y, He Y, Li T, Guo W, Tian H, Xu G, Xu X, Ma Y, Wang L, Wang L, Yan Y, Wang B, Xiao S, Zhou L, Li L, Tan L, Zhang T, Ma C, Li Q, Ding H, Geng H, Jia F, Shi J, Wang S, Zhang N, Du X, Du X, Wu Y. Prevalence of mental disorders in China: a cross-sectional epidemiological study. Lancet Psychiatry. 2019;6:211-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1960] [Cited by in RCA: 1586] [Article Influence: 226.6] [Reference Citation Analysis (1)] |
| 3. | Trautmann S, Rehm J, Wittchen HU. The economic costs of mental disorders: Do our societies react appropriately to the burden of mental disorders? EMBO Rep. 2016;17:1245-1249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 305] [Cited by in RCA: 427] [Article Influence: 42.7] [Reference Citation Analysis (0)] |
| 4. | Pfrieger FW, Barres BA. Synaptic efficacy enhanced by glial cells in vitro. Science. 1997;277:1684-1687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 544] [Cited by in RCA: 569] [Article Influence: 19.6] [Reference Citation Analysis (0)] |
| 5. | Lu CL, Ren J, Cao X. An Astroglial Basis of Major Depressive Disorder: Molecular, Cellular, and Circuit Features. Biol Psychiatry. 2025;97:217-226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 21] [Article Influence: 21.0] [Reference Citation Analysis (1)] |
| 6. | Mo JW, Kong PL, Ding L, Fan J, Ren J, Lu CL, Guo F, Chen LY, Mo R, Zhong QL, Wen YL, Gu TT, Wang QW, Li SJ, Guo T, Gao TM, Cao X. Lysosomal TFEB-TRPML1 Axis in Astrocytes Modulates Depressive-like Behaviors. Adv Sci (Weinh). 2024;11:e2403389. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 7. | Wang Q, Jie W, Liu JH, Yang JM, Gao TM. An astroglial basis of major depressive disorder? An overview. Glia. 2017;65:1227-1250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 181] [Article Influence: 20.1] [Reference Citation Analysis (0)] |
| 8. | Cathomas F, Murrough JW, Nestler EJ, Han MH, Russo SJ. Neurobiology of Resilience: Interface Between Mind and Body. Biol Psychiatry. 2019;86:410-420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 224] [Article Influence: 32.0] [Reference Citation Analysis (1)] |
| 9. | Brown SJ, Brown AM, Purves-Tyson TD, Huang XF, Shannon Weickert C, Newell KA. Alterations in the kynurenine pathway and excitatory amino acid transporter-2 in depression with and without psychosis: Evidence of a potential astrocyte pathology. J Psychiatr Res. 2022;147:203-211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 10. | Aten S, Du Y, Taylor O, Dye C, Collins K, Thomas M, Kiyoshi C, Zhou M. Chronic Stress Impairs the Structure and Function of Astrocyte Networks in an Animal Model of Depression. Neurochem Res. 2023;48:1191-1210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 47] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 11. | Shigetomi E, Koizumi S. The role of astrocytes in behaviors related to emotion and motivation. Neurosci Res. 2023;187:21-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 12. | He S, Shi Y, Ye J, Yin J, Yang Y, Liu D, Shen T, Zeng D, Zhang M, Li S, Xu F, Cai Y, Zhao F, Li H, Peng D. Does decreased autophagy and dysregulation of LC3A in astrocytes play a role in major depressive disorder? Transl Psychiatry. 2023;13:362. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 13. | Ma X, Yang S, Zhang Z, Liu L, Shi W, Yang S, Li S, Cai X, Zhou Q. Rapid and sustained restoration of astrocytic functions by ketamine in depression model mice. Biochem Biophys Res Commun. 2022;616:89-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 14. | Guo F, Fan J, Liu JM, Kong PL, Ren J, Mo JW, Lu CL, Zhong QL, Chen LY, Jiang HT, Zhang C, Wen YL, Gu TT, Li SJ, Fang YY, Pan BX, Gao TM, Cao X. Astrocytic ALKBH5 in stress response contributes to depressive-like behaviors in mice. Nat Commun. 2024;15:4347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 51] [Reference Citation Analysis (0)] |
| 15. | Chi D, Zhang K, Zhang J, He Z, Zhou H, Huang W, Liu Y, Huang J, Zeng W, Bai X, Ou C, Ouyang H. Astrocytic pleiotrophin deficiency in the prefrontal cortex contributes to stress-induced depressive-like responses in male mice. Nat Commun. 2025;16:2528. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 16. | Campos ACP, Pagano RL, Lipsman N, Hamani C. What do we know about astrocytes and the antidepressant effects of DBS? Exp Neurol. 2023;368:114501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 17. | Cohen-Cory S. The developing synapse: construction and modulation of synaptic structures and circuits. Science. 2002;298:770-776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 335] [Cited by in RCA: 307] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 18. | Pfrieger FW, Barres BA. New views on synapse-glia interactions. Curr Opin Neurobiol. 1996;6:615-621. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 111] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 19. | Ullian EM, Sapperstein SK, Christopherson KS, Barres BA. Control of synapse number by glia. Science. 2001;291:657-661. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 952] [Cited by in RCA: 1017] [Article Influence: 40.7] [Reference Citation Analysis (0)] |
| 20. | Sauvageot CM, Stiles CD. Molecular mechanisms controlling cortical gliogenesis. Curr Opin Neurobiol. 2002;12:244-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 282] [Cited by in RCA: 286] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 21. | Hassinger TD, Atkinson PB, Strecker GJ, Whalen LR, Dudek FE, Kossel AH, Kater SB. Evidence for glutamate-mediated activation of hippocampal neurons by glial calcium waves. J Neurobiol. 1995;28:159-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 138] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 22. | Liu J, Mo JW, Wang X, An Z, Zhang S, Zhang CY, Yi P, Leong ATL, Ren J, Chen LY, Mo R, Xie Y, Feng Q, Chen W, Gao TM, Wu EX, Feng Y, Cao X. Astrocyte dysfunction drives abnormal resting-state functional connectivity in depression. Sci Adv. 2022;8:eabo2098. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 61] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 23. | Lin L, Guo Z, Ren Z, Feng Y, Fang P, Wang T, Chen M. Bibliometric insights into astrocytic roles in depression and treatment. Front Cell Neurosci. 2024;18:1521398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 24. | Lei L, Wang YF, Chen CY, Wang YT, Zhang Y. Novel insight into astrocyte-mediated gliotransmission modulates the synaptic plasticity in major depressive disorder. Life Sci. 2024;355:122988. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 25] [Reference Citation Analysis (0)] |
| 25. | Liu D, Guo P, Wang Y, Li W. Regulation of adult neurogenesis: the crucial role of astrocytic mitochondria. Front Mol Neurosci. 2024;17:1516119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 26. | Araque A, Parpura V, Sanzgiri RP, Haydon PG. Tripartite synapses: glia, the unacknowledged partner. Trends Neurosci. 1999;22:208-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1631] [Cited by in RCA: 1760] [Article Influence: 65.2] [Reference Citation Analysis (0)] |
| 27. | Lu CL, Ren J, Mo JW, Fan J, Guo F, Chen LY, Wen YL, Li SJ, Fang YY, Wu ZF, Li YL, Gao TM, Cao X. Glucocorticoid Receptor-Dependent Astrocytes Mediate Stress Vulnerability. Biol Psychiatry. 2022;92:204-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 71] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 28. | Tsacopoulos M, Magistretti PJ. Metabolic coupling between glia and neurons. J Neurosci. 1996;16:877-885. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 726] [Cited by in RCA: 666] [Article Influence: 22.2] [Reference Citation Analysis (0)] |
| 29. | Schurr A, Miller JJ, Payne RS, Rigor BM. An increase in lactate output by brain tissue serves to meet the energy needs of glutamate-activated neurons. J Neurosci. 1999;19:34-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 222] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 30. | Magistretti PJ, Pellerin L, Rothman DL, Shulman RG. Energy on demand. Science. 1999;283:496-497. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 899] [Cited by in RCA: 819] [Article Influence: 30.3] [Reference Citation Analysis (0)] |
| 31. | Rossi DJ. Another BOLD role for astrocytes: coupling blood flow to neural activity. Nat Neurosci. 2006;9:159-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 30] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 32. | Zhao YF, Verkhratsky A, Tang Y, Illes P. Astrocytes and major depression: The purinergic avenue. Neuropharmacology. 2022;220:109252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 56] [Reference Citation Analysis (0)] |
| 33. | Zhang H, Liu S, Qin Q, Xu Z, Qu Y, Wang Y, Wang J, Du Z, Yuan S, Hong S, Chang Z, He W, Yan X, Lang Y, Tang R, Wang Y, Zhu L, Jiang X. Genetic and Pharmacological Inhibition of Astrocytic Mysm1 Alleviates Depressive-Like Disorders by Promoting ATP Production. Adv Sci (Weinh). 2022;10:e2204463. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 34. | Anderson G. Depression Pathophysiology: Astrocyte Mitochondrial Melatonergic Pathway as Crucial Hub. Int J Mol Sci. 2022;24:350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 35. | Tanaka K, Watase K, Manabe T, Yamada K, Watanabe M, Takahashi K, Iwama H, Nishikawa T, Ichihara N, Kikuchi T, Okuyama S, Kawashima N, Hori S, Takimoto M, Wada K. Epilepsy and exacerbation of brain injury in mice lacking the glutamate transporter GLT-1. Science. 1997;276:1699-1702. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1301] [Cited by in RCA: 1368] [Article Influence: 47.2] [Reference Citation Analysis (0)] |
| 36. | Verkhratsky A, Orkand RK, Kettenmann H. Glial calcium: homeostasis and signaling function. Physiol Rev. 1998;78:99-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 526] [Cited by in RCA: 514] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 37. | Seifert G, Schilling K, Steinhäuser C. Astrocyte dysfunction in neurological disorders: a molecular perspective. Nat Rev Neurosci. 2006;7:194-206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 565] [Cited by in RCA: 589] [Article Influence: 29.5] [Reference Citation Analysis (0)] |
| 38. | Rothstein JD, Dykes-Hoberg M, Pardo CA, Bristol LA, Jin L, Kuncl RW, Kanai Y, Hediger MA, Wang Y, Schielke JP, Welty DF. Knockout of glutamate transporters reveals a major role for astroglial transport in excitotoxicity and clearance of glutamate. Neuron. 1996;16:675-686. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1871] [Cited by in RCA: 1979] [Article Influence: 66.0] [Reference Citation Analysis (8)] |
| 39. | Rao VL, Dogan A, Todd KG, Bowen KK, Kim BT, Rothstein JD, Dempsey RJ. Antisense knockdown of the glial glutamate transporter GLT-1, but not the neuronal glutamate transporter EAAC1, exacerbates transient focal cerebral ischemia-induced neuronal damage in rat brain. J Neurosci. 2001;21:1876-1883. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 173] [Cited by in RCA: 169] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 40. | Hirst WD, Price GW, Rattray M, Wilkin GP. Serotonin transporters in adult rat brain astrocytes revealed by [3H]5-HT uptake into glial plasmalemmal vesicles. Neurochem Int. 1998;33:11-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 61] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 41. | Minelli A, DeBiasi S, Brecha NC, Zuccarello LV, Conti F. GAT-3, a high-affinity GABA plasma membrane transporter, is localized to astrocytic processes, and it is not confined to the vicinity of GABAergic synapses in the cerebral cortex. J Neurosci. 1996;16:6255-6264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 195] [Cited by in RCA: 211] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 42. | Wu Q, Wada M, Shimada A, Yamamoto A, Fujita T. Functional characterization of Zn2(+)-sensitive GABA transporter expressed in primary cultures of astrocytes from rat cerebral cortex. Brain Res. 2006;1075:100-109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 14] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 43. | Bal N, Figueras G, Vilaró MT, Suñol C, Artigas F. Antidepressant drugs inhibit a glial 5-hydroxytryptamine transporter in rat brain. Eur J Neurosci. 1997;9:1728-1738. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 40] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 44. | Takeda H, Inazu M, Matsumiya T. Astroglial dopamine transport is mediated by norepinephrine transporter. Naunyn Schmiedebergs Arch Pharmacol. 2002;366:620-623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 75] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 45. | Inazu M, Takeda H, Matsumiya T. Functional expression of the norepinephrine transporter in cultured rat astrocytes. J Neurochem. 2003;84:136-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 67] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 46. | Inazu M, Takeda H, Matsumiya T. Expression and functional characterization of the extraneuronal monoamine transporter in normal human astrocytes. J Neurochem. 2003;84:43-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 61] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 47. | Hansson E, Muyderman H, Leonova J, Allansson L, Sinclair J, Blomstrand F, Thorlin T, Nilsson M, Rönnbäck L. Astroglia and glutamate in physiology and pathology: aspects on glutamate transport, glutamate-induced cell swelling and gap-junction communication. Neurochem Int. 2000;37:317-329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 117] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 48. | González-Arias C, Sánchez-Ruiz A, Esparza J, Sánchez-Puelles C, Arancibia L, Ramírez-Franco J, Gobbo D, Kirchhoff F, Perea G. Dysfunctional serotonergic neuron-astrocyte signaling in depressive-like states. Mol Psychiatry. 2023;28:3856-3873. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 84] [Article Influence: 28.0] [Reference Citation Analysis (0)] |
| 49. | Dave V, Kimelberg HK. Na(+)-dependent, fluoxetine-sensitive serotonin uptake by astrocytes tissue-printed from rat cerebral cortex. J Neurosci. 1994;14:4972-4986. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 36] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 50. | Ali DN, Ali HM, Lopez MR, Kang S, Choi DS. Astrocytic GABAergic Regulation in Alcohol Use and Major Depressive Disorders. Cells. 2024;13:318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 51. | Fang Y, Ding X, Zhang Y, Cai L, Ge Y, Ma K, Xu R, Li S, Song M, Zhu H, Liu J, Ding J, Lu M, Hu G. Fluoxetine inhibited the activation of A1 reactive astrocyte in a mouse model of major depressive disorder through astrocytic 5-HT(2B)R/β-arrestin2 pathway. J Neuroinflammation. 2022;19:23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 74] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 52. | Wada M, Nakajima S, Honda S, Takano M, Taniguchi K, Homma S, Ueda R, Tobari Y, Mimura Y, Fujii S, Mimura M, Noda Y. Decreased prefrontal glutamatergic function is associated with a reduced astrocyte-related gene expression in treatment-resistant depression. Transl Psychiatry. 2024;14:478. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 53. | Malin SA, Molliver DC, Koerber HR, Cornuet P, Frye R, Albers KM, Davis BM. Glial cell line-derived neurotrophic factor family members sensitize nociceptors in vitro and produce thermal hyperalgesia in vivo. J Neurosci. 2006;26:8588-8599. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 210] [Cited by in RCA: 206] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 54. | Erickson JT, Brosenitsch TA, Katz DM. Brain-derived neurotrophic factor and glial cell line-derived neurotrophic factor are required simultaneously for survival of dopaminergic primary sensory neurons in vivo. J Neurosci. 2001;21:581-589. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 115] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 55. | Turner CA, Akil H, Watson SJ, Evans SJ. The fibroblast growth factor system and mood disorders. Biol Psychiatry. 2006;59:1128-1135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 102] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 56. | Li K, Wang K, Xu SX, Xie XH, Tang Y, Zhang L, Liu Z. Investigating Neuroplasticity Changes Reflected by BDNF Levels in Astrocyte-Derived Extracellular Vesicles in Patients with Depression. Int J Nanomedicine. 2024;19:8971-8985. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 57. | Acker T, Beck H, Plate KH. Cell type specific expression of vascular endothelial growth factor and angiopoietin-1 and -2 suggests an important role of astrocytes in cerebellar vascularization. Mech Dev. 2001;108:45-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 98] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 58. | Rawani NS, Chan AW, Todd KG, Baker GB, Dursun SM. The Role of Neuroglia in the Development and Progression of Schizophrenia. Biomolecules. 2024;15:10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 59. | Fuchs E, Czéh B, Kole MH, Michaelis T, Lucassen PJ. Alterations of neuroplasticity in depression: the hippocampus and beyond. Eur Neuropsychopharmacol. 2004;14 Suppl 5:S481-S490. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 193] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 60. | Duman RS, Monteggia LM. A neurotrophic model for stress-related mood disorders. Biol Psychiatry. 2006;59:1116-1127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2783] [Cited by in RCA: 2517] [Article Influence: 125.9] [Reference Citation Analysis (4)] |
| 61. | Manji HK, Quiroz JA, Sporn J, Payne JL, Denicoff K, A Gray N, Zarate CA Jr, Charney DS. Enhancing neuronal plasticity and cellular resilience to develop novel, improved therapeutics for difficult-to-treat depression. Biol Psychiatry. 2003;53:707-742. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 352] [Cited by in RCA: 341] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 62. | Dwivedi Y, Rizavi HS, Conley RR, Roberts RC, Tamminga CA, Pandey GN. Altered gene expression of brain-derived neurotrophic factor and receptor tyrosine kinase B in postmortem brain of suicide subjects. Arch Gen Psychiatry. 2003;60:804-815. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 716] [Cited by in RCA: 633] [Article Influence: 27.5] [Reference Citation Analysis (0)] |
| 63. | Wang H, Yang Y, Pei G, Wang Z, Chen N. Neurotrophic basis to the pathogenesis of depression and phytotherapy. Front Pharmacol. 2023;14:1182666. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 64. | Newton SS, Girgenti MJ, Collier EF, Duman RS. Electroconvulsive seizure increases adult hippocampal angiogenesis in rats. Eur J Neurosci. 2006;24:819-828. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 45] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 65. | Chen B, Dowlatshahi D, MacQueen GM, Wang JF, Young LT. Increased hippocampal BDNF immunoreactivity in subjects treated with antidepressant medication. Biol Psychiatry. 2001;50:260-265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 914] [Cited by in RCA: 827] [Article Influence: 33.1] [Reference Citation Analysis (0)] |
| 66. | Ongür D, Heckers S. A role for glia in the action of electroconvulsive therapy. Harv Rev Psychiatry. 2004;12:253-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 19] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 67. | Mhyre AJ, Dorsa DM. Estrogen activates rapid signaling in the brain: role of estrogen receptor alpha and estrogen receptor beta in neurons and glia. Neuroscience. 2006;138:851-858. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 82] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 68. | Pawlak J, Karolczak M, Krust A, Chambon P, Beyer C. Estrogen receptor-alpha is associated with the plasma membrane of astrocytes and coupled to the MAP/Src-kinase pathway. Glia. 2005;50:270-275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 85] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 69. | Pawlak J, Brito V, Küppers E, Beyer C. Regulation of glutamate transporter GLAST and GLT-1 expression in astrocytes by estrogen. Brain Res Mol Brain Res. 2005;138:1-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 144] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 70. | Garcia-Segura LM, McCarthy MM. Minireview: Role of glia in neuroendocrine function. Endocrinology. 2004;145:1082-1086. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 115] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 71. | Xie XH, Lai WT, Xu SX, Di Forti M, Zhang JY, Chen MM, Yao LH, Wang P, Hao KK, Rong H. Hyper-inflammation of astrocytes in patients of major depressive disorder: Evidence from serum astrocyte-derived extracellular vesicles. Brain Behav Immun. 2023;109:51-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 45] [Reference Citation Analysis (0)] |
| 72. | Rana T, Behl T, Shamsuzzaman M, Singh S, Sharma N, Sehgal A, Alshahrani AM, Aldahish A, Chidambaram K, Dailah HG, Bhatia S, Bungau S. Exploring the role of astrocytic dysfunction and AQP4 in depression. Cell Signal. 2022;96:110359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 73. | Eddleston M, Mucke L. Molecular profile of reactive astrocytes--implications for their role in neurologic disease. Neuroscience. 1993;54:15-36. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1131] [Cited by in RCA: 1078] [Article Influence: 32.7] [Reference Citation Analysis (11)] |
| 74. | Laping NJ, Teter B, Nichols NR, Rozovsky I, Finch CE. Glial fibrillary acidic protein: regulation by hormones, cytokines, and growth factors. Brain Pathol. 1994;4:259-275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 163] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 75. | Cornell-Bell AH, Finkbeiner SM, Cooper MS, Smith SJ. Glutamate induces calcium waves in cultured astrocytes: long-range glial signaling. Science. 1990;247:470-473. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1329] [Cited by in RCA: 1317] [Article Influence: 36.6] [Reference Citation Analysis (0)] |
| 76. | Stenovec M, Li B, Verkhratsky A, Zorec R. Astrocytes in rapid ketamine antidepressant action. Neuropharmacology. 2020;173:108158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 35] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 77. | Shen SY, Liang LF, Shi TL, Shen ZQ, Yin SY, Zhang JR, Li W, Mi WL, Wang YQ, Zhang YQ, Yu J. Microglia-Derived Interleukin-6 Triggers Astrocyte Apoptosis in the Hippocampus and Mediates Depression-Like Behavior. Adv Sci (Weinh). 2025;12:e2412556. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 32] [Article Influence: 32.0] [Reference Citation Analysis (0)] |
| 78. | Li H, Cai R, Zhou Y, Jiang Y, Tan S. cGAS-STING signaling in brain aging and neurodegeneration: molecular links and therapeutic perspectives. J Neuroinflammation. 2025;22:235. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 20] [Reference Citation Analysis (0)] |
| 79. | Miyata S, Ishino Y, Shimizu S, Tohyama M. Involvement of inflammatory responses in the brain to the onset of major depressive disorder due to stress exposure. Front Aging Neurosci. 2022;14:934346. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 80. | Reiter RJ, Ma Q, Sharma R. Melatonin in Mitochondria: Mitigating Clear and Present Dangers. Physiology (Bethesda). 2020;35:86-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 74] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 81. | Rose J, Brian C, Pappa A, Panayiotidis MI, Franco R. Mitochondrial Metabolism in Astrocytes Regulates Brain Bioenergetics, Neurotransmission and Redox Balance. Front Neurosci. 2020;14:536682. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 123] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 82. | Salk RH, Hyde JS, Abramson LY. Gender differences in depression in representative national samples: Meta-analyses of diagnoses and symptoms. Psychol Bull. 2017;143:783-822. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2063] [Cited by in RCA: 1676] [Article Influence: 186.2] [Reference Citation Analysis (0)] |
| 83. | Gozlan E, Lewit-Cohen Y, Frenkel D. Sex Differences in Astrocyte Activity. Cells. 2024;13:1724. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 22] [Reference Citation Analysis (0)] |
| 84. | Sun Q, Li G, Zhao F, Dong M, Xie W, Liu Q, Yang W, Cui R. Role of estrogen in treatment of female depression. Aging (Albany NY). 2024;16:3021-3042. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 22] [Reference Citation Analysis (0)] |
| 85. | Kundakovic M, Rocks D. Sex hormone fluctuation and increased female risk for depression and anxiety disorders: From clinical evidence to molecular mechanisms. Front Neuroendocrinol. 2022;66:101010. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 203] [Article Influence: 50.8] [Reference Citation Analysis (0)] |
| 86. | Zhang X, Alnafisah RS, Hamoud AA, Shukla R, Wen Z, McCullumsmith RE, O'Donovan SM. Role of Astrocytes in Major Neuropsychiatric Disorders. Neurochem Res. 2021;46:2715-2730. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 87. | Pan Y, Xiang L, Zhu T, Wang H, Xu Q, Liao F, He J, Wang Y. Prefrontal cortex astrocytes in major depressive disorder: exploring pathogenic mechanisms and potential therapeutic targets. J Mol Med (Berl). 2024;102:1355-1369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 88. | Ongür D, Drevets WC, Price JL. Glial reduction in the subgenual prefrontal cortex in mood disorders. Proc Natl Acad Sci U S A. 1998;95:13290-13295. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1155] [Cited by in RCA: 1066] [Article Influence: 38.1] [Reference Citation Analysis (0)] |
| 89. | Cotter D, Mackay D, Chana G, Beasley C, Landau S, Everall IP. Reduced neuronal size and glial cell density in area 9 of the dorsolateral prefrontal cortex in subjects with major depressive disorder. Cereb Cortex. 2002;12:386-394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 508] [Cited by in RCA: 453] [Article Influence: 18.9] [Reference Citation Analysis (0)] |
| 90. | Rajkowska G, Miguel-Hidalgo JJ, Wei J, Dilley G, Pittman SD, Meltzer HY, Overholser JC, Roth BL, Stockmeier CA. Morphometric evidence for neuronal and glial prefrontal cell pathology in major depression. Biol Psychiatry. 1999;45:1085-1098. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1202] [Cited by in RCA: 1042] [Article Influence: 38.6] [Reference Citation Analysis (0)] |
| 91. | Rajkowska G, Legutko B, Moulana M, Syed M, Romero DG, Stockmeier CA, Miguel-Hidalgo JJ. Astrocyte pathology in the ventral prefrontal white matter in depression. J Psychiatr Res. 2018;102:150-158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 56] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 92. | Cotter D, Mackay D, Landau S, Kerwin R, Everall I. Reduced glial cell density and neuronal size in the anterior cingulate cortex in major depressive disorder. Arch Gen Psychiatry. 2001;58:545-553. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 559] [Cited by in RCA: 548] [Article Influence: 21.9] [Reference Citation Analysis (1)] |
| 93. | Stockmeier CA, Mahajan GJ, Konick LC, Overholser JC, Jurjus GJ, Meltzer HY, Uylings HB, Friedman L, Rajkowska G. Cellular changes in the postmortem hippocampus in major depression. Biol Psychiatry. 2004;56:640-650. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 589] [Cited by in RCA: 543] [Article Influence: 24.7] [Reference Citation Analysis (0)] |
| 94. | Zeb S, Ye H, Liu Y, Du HP, Guo Y, Zhu YM, Ni Y, Zhang HL, Xu Y. Necroptotic kinases are involved in the reduction of depression-induced astrocytes and fluoxetine's inhibitory effects on necroptotic kinases. Front Pharmacol. 2022;13:1060954. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 95. | Bowley MP, Drevets WC, Ongür D, Price JL. Low glial numbers in the amygdala in major depressive disorder. Biol Psychiatry. 2002;52:404-412. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 401] [Cited by in RCA: 356] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 96. | Zhang X, Alnafisah RS, Hamoud AA, Shukla R, McCullumsmith RE, O'Donovan SM. Astrocytes in Neuropsychiatric Disorders: A Review of Postmortem Evidence. Adv Neurobiol. 2021;26:153-172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 97. | Yamamoto M, Sakai M, Yu Z, Nakanishi M, Yoshii H. Glial Markers of Suicidal Behavior in the Human Brain-A Systematic Review of Postmortem Studies. Int J Mol Sci. 2024;25:5750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 98. | Chana G, Landau S, Beasley C, Everall IP, Cotter D. Two-dimensional assessment of cytoarchitecture in the anterior cingulate cortex in major depressive disorder, bipolar disorder, and schizophrenia: evidence for decreased neuronal somal size and increased neuronal density. Biol Psychiatry. 2003;53:1086-1098. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 186] [Cited by in RCA: 188] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 99. | Rajkowska G, Halaris A, Selemon LD. Reductions in neuronal and glial density characterize the dorsolateral prefrontal cortex in bipolar disorder. Biol Psychiatry. 2001;49:741-752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 436] [Cited by in RCA: 397] [Article Influence: 15.9] [Reference Citation Analysis (0)] |
| 100. | Cotter D, Hudson L, Landau S. Evidence for orbitofrontal pathology in bipolar disorder and major depression, but not in schizophrenia. Bipolar Disord. 2005;7:358-369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 103] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 101. | Rajkowska G, Selemon LD, Goldman-Rakic PS. Neuronal and glial somal size in the prefrontal cortex: a postmortem morphometric study of schizophrenia and Huntington disease. Arch Gen Psychiatry. 1998;55:215-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 380] [Cited by in RCA: 368] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 102. | Miguel-Hidalgo JJ, Wei J, Andrew M, Overholser JC, Jurjus G, Stockmeier CA, Rajkowska G. Glia pathology in the prefrontal cortex in alcohol dependence with and without depressive symptoms. Biol Psychiatry. 2002;52:1121-1133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 114] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 103. | Sheline YI. Neuroimaging studies of mood disorder effects on the brain. Biol Psychiatry. 2003;54:338-352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 494] [Cited by in RCA: 425] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 104. | Ohl F, Michaelis T, Vollmann-Honsdorf GK, Kirschbaum C, Fuchs E. Effect of chronic psychosocial stress and long-term cortisol treatment on hippocampus-mediated memory and hippocampal volume: a pilot-study in tree shrews. Psychoneuroendocrinology. 2000;25:357-363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 74] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 105. | Lucassen PJ, Müller MB, Holsboer F, Bauer J, Holtrop A, Wouda J, Hoogendijk WJ, De Kloet ER, Swaab DF. Hippocampal apoptosis in major depression is a minor event and absent from subareas at risk for glucocorticoid overexposure. Am J Pathol. 2001;158:453-468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 206] [Cited by in RCA: 211] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 106. | Leverenz JB, Wilkinson CW, Wamble M, Corbin S, Grabber JE, Raskind MA, Peskind ER. Effect of chronic high-dose exogenous cortisol on hippocampal neuronal number in aged nonhuman primates. J Neurosci. 1999;19:2356-2361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 103] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 107. | Czéh B, Simon M, Schmelting B, Hiemke C, Fuchs E. Astroglial plasticity in the hippocampus is affected by chronic psychosocial stress and concomitant fluoxetine treatment. Neuropsychopharmacology. 2006;31:1616-1626. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 368] [Cited by in RCA: 346] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 108. | Lin SS, Zhou B, Chen BJ, Jiang RT, Li B, Illes P, Semyanov A, Tang Y, Verkhratsky A. Electroacupuncture prevents astrocyte atrophy to alleviate depression. Cell Death Dis. 2023;14:343. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 72] [Article Influence: 24.0] [Reference Citation Analysis (0)] |
| 109. | Ajtai BM, Kálmán M. Reactive glia support and guide axon growth in the rat thalamus during the first postnatal week. A sharply timed transition from permissive to non-permissive stage. Int J Dev Neurosci. 2001;19:589-597. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 110. | Müller MB, Lucassen PJ, Yassouridis A, Hoogendijk WJ, Holsboer F, Swaab DF. Neither major depression nor glucocorticoid treatment affects the cellular integrity of the human hippocampus. Eur J Neurosci. 2001;14:1603-1612. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 192] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 111. | Choudary PV, Molnar M, Evans SJ, Tomita H, Li JZ, Vawter MP, Myers RM, Bunney WE Jr, Akil H, Watson SJ, Jones EG. Altered cortical glutamatergic and GABAergic signal transmission with glial involvement in depression. Proc Natl Acad Sci U S A. 2005;102:15653-15658. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 558] [Cited by in RCA: 506] [Article Influence: 24.1] [Reference Citation Analysis (0)] |
| 112. | Zhao J, Verwer RW, van Wamelen DJ, Qi XR, Gao SF, Lucassen PJ, Swaab DF. Prefrontal changes in the glutamate-glutamine cycle and neuronal/glial glutamate transporters in depression with and without suicide. J Psychiatr Res. 2016;82:8-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 66] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 113. | Steinacker P, Al Shweiki MR, Oeckl P, Graf H, Ludolph AC, Schönfeldt-Lecuona C, Otto M. Glial fibrillary acidic protein as blood biomarker for differential diagnosis and severity of major depressive disorder. J Psychiatr Res. 2021;144:54-58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 65] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 114. | Miguel-Hidalgo JJ, Baucom C, Dilley G, Overholser JC, Meltzer HY, Stockmeier CA, Rajkowska G. Glial fibrillary acidic protein immunoreactivity in the prefrontal cortex distinguishes younger from older adults in major depressive disorder. Biol Psychiatry. 2000;48:861-873. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 228] [Cited by in RCA: 238] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 115. | Rajkowska G, Stockmeier CA. Astrocyte pathology in major depressive disorder: insights from human postmortem brain tissue. Curr Drug Targets. 2013;14:1225-1236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 423] [Cited by in RCA: 483] [Article Influence: 37.2] [Reference Citation Analysis (0)] |
| 116. | Nagy C, Maitra M, Tanti A, Suderman M, Théroux JF, Davoli MA, Perlman K, Yerko V, Wang YC, Tripathy SJ, Pavlidis P, Mechawar N, Ragoussis J, Turecki G. Single-nucleus transcriptomics of the prefrontal cortex in major depressive disorder implicates oligodendrocyte precursor cells and excitatory neurons. Nat Neurosci. 2020;23:771-781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 480] [Cited by in RCA: 374] [Article Influence: 62.3] [Reference Citation Analysis (0)] |
| 117. | Maitra M, Mitsuhashi H, Rahimian R, Chawla A, Yang J, Fiori LM, Davoli MA, Perlman K, Aouabed Z, Mash DC, Suderman M, Mechawar N, Turecki G, Nagy C. Cell type specific transcriptomic differences in depression show similar patterns between males and females but implicate distinct cell types and genes. Nat Commun. 2023;14:2912. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 97] [Article Influence: 32.3] [Reference Citation Analysis (0)] |
| 118. | Michel M, Fiebich BL, Kuzior H, Meixensberger S, Berger B, Maier S, Nickel K, Runge K, Denzel D, Pankratz B, Schiele MA, Domschke K, van Elst LT, Endres D. Increased GFAP concentrations in the cerebrospinal fluid of patients with unipolar depression. Transl Psychiatry. 2021;11:308. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 53] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 119. | O'Leary LA, Belliveau C, Davoli MA, Ma JC, Tanti A, Turecki G, Mechawar N. Widespread Decrease of Cerebral Vimentin-Immunoreactive Astrocytes in Depressed Suicides. Front Psychiatry. 2021;12:640963. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 120. | Barger SW, Van Eldik LJ. S100 beta stimulates calcium fluxes in glial and neuronal cells. J Biol Chem. 1992;267:9689-9694. [PubMed] |
| 121. | Gulen B, Serinken M, Eken C, Karcıoglu Ö, Kucukdagli OT, Kilic E, Akpinar G, Nogay S, Kuh M. Serum S100B as a Surrogate Biomarker in the Diagnoses of Burnout and Depression in Emergency Medicine Residents. Acad Emerg Med. 2016;23:786-789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 16] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 122. | Güleş E, Iosifescu DV, Tural Ü. Plasma Neuronal and Glial Markers and Anterior Cingulate Metabolite Levels in Major Depressive Disorder: A Pilot Study. Neuropsychobiology. 2020;79:214-221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 123. | O'Leary LA, Mechawar N. Implication of cerebral astrocytes in major depression: A review of fine neuroanatomical evidence in humans. Glia. 2021;69:2077-2099. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 89] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 124. | Schroeter ML, Abdul-Khaliq H, Diefenbacher A, Blasig IE. S100B is increased in mood disorders and may be reduced by antidepressive treatment. Neuroreport. 2002;13:1675-1678. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 102] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 125. | Machado-Vieira R, Lara DR, Portela LV, Gonçalves CA, Soares JC, Kapczinski F, Souza DO. Elevated serum S100B protein in drug-free bipolar patients during first manic episode: a pilot study. Eur Neuropsychopharmacol. 2002;12:269-272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 48] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 126. | Grabe HJ, Ahrens N, Rose HJ, Kessler C, Freyberger HJ. Neurotrophic factor S100 beta in major depression. Neuropsychobiology. 2001;44:88-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 62] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 127. | Jang BS, Kim H, Lim SW, Jang KW, Kim DK. Serum S100B Levels and Major Depressive Disorder: Its Characteristics and Role in Antidepressant Response. Psychiatry Investig. 2008;5:193-198. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 40] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 128. | Sakatani S, Seto-Ohshima A, Shinohara Y, Yamamoto Y, Yamamoto H, Itohara S, Hirase H. Neural-activity-dependent release of S100B from astrocytes enhances kainate-induced gamma oscillations in vivo. J Neurosci. 2008;28:10928-10936. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 90] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 129. | Gayger-Dias V, Vizuete AF, Rodrigues L, Wartchow KM, Bobermin L, Leite MC, Quincozes-Santos A, Kleindienst A, Gonçalves CA. How S100B crosses brain barriers and why it is considered a peripheral marker of brain injury. Exp Biol Med (Maywood). 2023;248:2109-2119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 30] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 130. | Sorci G, Riuzzi F, Arcuri C, Tubaro C, Bianchi R, Giambanco I, Donato R. S100B protein in tissue development, repair and regeneration. World J Biol Chem. 2013;4:1-12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 77] [Cited by in RCA: 83] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 131. | Lin LF, Doherty DH, Lile JD, Bektesh S, Collins F. GDNF: a glial cell line-derived neurotrophic factor for midbrain dopaminergic neurons. Science. 1993;260:1130-1132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2423] [Cited by in RCA: 2399] [Article Influence: 72.7] [Reference Citation Analysis (0)] |
| 132. | Bespalov MM, Saarma M. GDNF family receptor complexes are emerging drug targets. Trends Pharmacol Sci. 2007;28:68-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 137] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 133. | Lapchak PA, Miller PJ, Jiao S. Glial cell line-derived neurotrophic factor induces the dopaminergic and cholinergic phenotype and increases locomotor activity in aged Fischer 344 rats. Neuroscience. 1997;77:745-752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 52] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 134. | Grondin R, Gash DM. Glial cell line-derived neurotrophic factor (GDNF): a drug candidate for the treatment of Parkinson's disease. J Neurol. 1998;245:P35-P42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 82] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 135. | Pichel JG, Shen L, Sheng HZ, Granholm AC, Drago J, Grinberg A, Lee EJ, Huang SP, Saarma M, Hoffer BJ, Sariola H, Westphal H. Defects in enteric innervation and kidney development in mice lacking GDNF. Nature. 1996;382:73-76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 975] [Cited by in RCA: 857] [Article Influence: 28.6] [Reference Citation Analysis (0)] |
| 136. | Meng X, Lindahl M, Hyvönen ME, Parvinen M, de Rooij DG, Hess MW, Raatikainen-Ahokas A, Sainio K, Rauvala H, Lakso M, Pichel JG, Westphal H, Saarma M, Sariola H. Regulation of cell fate decision of undifferentiated spermatogonia by GDNF. Science. 2000;287:1489-1493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1146] [Cited by in RCA: 972] [Article Influence: 37.4] [Reference Citation Analysis (1)] |
| 137. | Nanobashvili A, Airaksinen MS, Kokaia M, Rossi J, Asztély F, Olofsdotter K, Mohapel P, Saarma M, Lindvall O, Kokaia Z. Development and persistence of kindling epilepsy are impaired in mice lacking glial cell line-derived neurotrophic factor family receptor alpha 2. Proc Natl Acad Sci U S A. 2000;97:12312-12317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 32] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 138. | Messer CJ, Eisch AJ, Carlezon WA Jr, Whisler K, Shen L, Wolf DH, Westphal H, Collins F, Russell DS, Nestler EJ. Role for GDNF in biochemical and behavioral adaptations to drugs of abuse. Neuron. 2000;26:247-257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 124] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 139. | Gerlai R, McNamara A, Choi-Lundberg DL, Armanini M, Ross J, Powell-Braxton L, Phillips HS. Impaired water maze learning performance without altered dopaminergic function in mice heterozygous for the GDNF mutation. Eur J Neurosci. 2001;14:1153-1163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 93] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 140. | Pochon NA, Menoud A, Tseng JL, Zurn AD, Aebischer P. Neuronal GDNF expression in the adult rat nervous system identified by in situ hybridization. Eur J Neurosci. 1997;9:463-471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 127] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 141. | Hisaoka K, Nishida A, Koda T, Miyata M, Zensho H, Morinobu S, Ohta M, Yamawaki S. Antidepressant drug treatments induce glial cell line-derived neurotrophic factor (GDNF) synthesis and release in rat C6 glioblastoma cells. J Neurochem. 2001;79:25-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 96] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 142. | Bartholomä P, Erlandsson N, Kaufmann K, Rössler OG, Baumann B, Wirth T, Giehl KM, Thiel G. Neuronal cell death induced by antidepressants: lack of correlation with Egr-1, NF-kappa B and extracellular signal-regulated protein kinase activation. Biochem Pharmacol. 2002;63:1507-1516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 39] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 143. | Hisaoka K, Nishida A, Takebayashi M, Koda T, Yamawaki S, Nakata Y. Serotonin increases glial cell line-derived neurotrophic factor release in rat C6 glioblastoma cells. Brain Res. 2004;1002:167-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 30] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 144. | Castro LM, Gallant M, Niles LP. Novel targets for valproic acid: up-regulation of melatonin receptors and neurotrophic factors in C6 glioma cells. J Neurochem. 2005;95:1227-1236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 88] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 145. | Liang C, Wei S, Ji Y, Lin J, Jiao W, Li Z, Yan F, Jing X. The role of enteric nervous system and GDNF in depression: Conversation between the brain and the gut. Neurosci Biobehav Rev. 2024;167:105931. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 146. | Mercier G, Lennon AM, Renouf B, Dessouroux A, Ramaugé M, Courtin F, Pierre M. MAP kinase activation by fluoxetine and its relation to gene expression in cultured rat astrocytes. J Mol Neurosci. 2004;24:207-216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 120] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 147. | Angelucci F, Aloe L, Jiménez-Vasquez P, Mathé AA. Lithium treatment alters brain concentrations of nerve growth factor, brain-derived neurotrophic factor and glial cell line-derived neurotrophic factor in a rat model of depression. Int J Neuropsychopharmacol. 2003;6:225-231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 102] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 148. | Angelucci F, Aloe L, Jiménez-Vasquez P, Mathé AA. Electroconvulsive stimuli alter the regional concentrations of nerve growth factor, brain-derived neurotrophic factor, and glial cell line-derived neurotrophic factor in adult rat brain. J ECT. 2002;18:138-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 46] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 149. | Takebayashi M, Hisaoka K, Nishida A, Tsuchioka M, Miyoshi I, Kozuru T, Hikasa S, Okamoto Y, Shinno H, Morinobu S, Yamawaki S. Decreased levels of whole blood glial cell line-derived neurotrophic factor (GDNF) in remitted patients with mood disorders. Int J Neuropsychopharmacol. 2006;9:607-612. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 103] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 150. | Rosa AR, Frey BN, Andreazza AC, Ceresér KM, Cunha AB, Quevedo J, Santin A, Gottfried C, Gonçalves CA, Vieta E, Kapczinski F. Increased serum glial cell line-derived neurotrophic factor immunocontent during manic and depressive episodes in individuals with bipolar disorder. Neurosci Lett. 2006;407:146-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 70] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 151. | Otsuki K, Uchida S, Watanuki T, Wakabayashi Y, Fujimoto M, Matsubara T, Funato H, Watanabe Y. Altered expression of neurotrophic factors in patients with major depression. J Psychiatr Res. 2008;42:1145-1153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 140] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 152. | Michel TM, Frangou S, Camara S, Thiemeyer D, Jecel J, Tatschner T, Zoechling R, Grünblatt E. Altered glial cell line-derived neurotrophic factor (GDNF) concentrations in the brain of patients with depressive disorder: a comparative post-mortem study. Eur Psychiatry. 2008;23:413-420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 61] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 153. | Liu X, Li P, Ma X, Zhang J, Sun X, Luo X, Zhang Y. Association between plasma levels of BDNF and GDNF and the diagnosis, treatment response in first-episode MDD. J Affect Disord. 2022;315:190-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 154. | Duarte Azevedo M, Sander S, Tenenbaum L. GDNF, A Neuron-Derived Factor Upregulated in Glial Cells during Disease. J Clin Med. 2020;9:456. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 95] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 155. | Rocha SM, Cristovão AC, Campos FL, Fonseca CP, Baltazar G. Astrocyte-derived GDNF is a potent inhibitor of microglial activation. Neurobiol Dis. 2012;47:407-415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 137] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 156. | Zhao Y, Haney MJ, Fallon JK, Rodriguez M, Swain CJ, Arzt CJ, Smith PC, Loop MS, Harrison EB, El-Hage N, Batrakova EV. Using Extracellular Vesicles Released by GDNF-Transfected Macrophages for Therapy of Parkinson Disease. Cells. 2022;11:1933. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |