Published online Apr 19, 2026. doi: 10.5498/wjp.v16.i4.115490
Revised: December 8, 2025
Accepted: December 26, 2025
Published online: April 19, 2026
Processing time: 146 Days and 0.5 Hours
Malignant melanoma carries the highest mortality among skin cancers and is fre
To investigate the correlation between depressive symptoms and immune fun
Clinical data of 202 patients with malignant melanoma from January 2019 to March 2025 were retrospectively analyzed. Patient Health Questionnaire-9, Pittsburgh Sleep Quality Index, Fatigue Severity Scale, and Perceived Stress Scale-10 were used to assess depressive symptoms. Cellular immune indicators [CD3+, CD4+, CD8+, natural killer (NK) cell counts and percentages], humoral immune indicators (immunoglobulins, complement C3, C4), inflammatory factors [in
Among 202 patients, 113 (55.9%) developed depressive symptoms. Patients in the depression group had significantly lower CD3+ cell count, CD4+ cell count, NK cell count, and CD4+/CD8+ ratio (all P < 0.05); com
Depressive symptoms in patients with malignant melanoma are closely associated with cellular immune function suppression and chronic inflammatory responses, and immune indicators such as IL-6 can serve as effective pre
Core Tip: This retrospective study revealed that over half of malignant melanoma patients experienced depressive symptoms, which were closely associated with suppressed cellular immunity and elevated inflammatory markers such as interleukin-6 (IL-6) and neutrophil-to-lymphocyte ratio. CD4+ T cell count and serum albumin were found to be protective factors, while IL-6 and advanced tumor-node-metastasis stage were risk predictors. A multi-indicator model combining IL-6, CD4+, and neutrophil-to-lymphocyte ratio achieved good predictive performance (area under the curve = 0.834). These findings highlight the need for integrated psychosocial and immunological assessment in melanoma patients to improve mental health and potentially affect prognosis.
- Citation: Dong S, Mou HL, Zhang H, Ye T. Correlation analysis of depressive symptoms and immune function indicators in patients with malignant melanoma. World J Psychiatry 2026; 16(4): 115490
- URL: https://www.wjgnet.com/2220-3206/full/v16/i4/115490.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i4.115490
Malignant melanoma is a highly malignant tumor originating from melanocytes, with an incidence rate showing an annual upward trend globally, becoming the skin malignancy with the highest mortality rate[1]. This disease not only has biological characteristics of strong invasiveness and obvious metastatic tendency, but patients also face enormous psychological stress and survival pressure. Epidemiological surveys show that the incidence of depressive symptoms in mali
In recent years, the development of psychoneuroimmunology has provided new perspectives for understanding the relationship between depressive symptoms and the body’s immune function. Research indicates that depressive states can lead to immune system dysfunction through activation of the hypothalamic-pituitary-adrenal (HPA) axis and sym
For malignant tumor patients, immune function status is closely related to tumor occurrence, development, and prognosis. Defects in immune surveillance function may promote tumor cell proliferation and metastasis, while chronic inflammatory states provide favorable conditions for the tumor microenvironment[5]. However, current research on the specific correlations between depressive symptoms and immune function indicators in malignant melanoma patients remains relatively limited, lacking large-sample systematic analyses. In-depth exploration of the correlations between these two factors not only helps reveal potential mechanisms by which psychological factors affect tumor prognosis but also provides scientific evidence for developing comprehensive treatment strategies[6].
Therefore, this study retrospectively analyzed clinical data from 202 malignant melanoma patients, systematically assessed the severity of patients’ depressive symptoms, and detected multiple immune function-related indicators, aiming to elucidate the correlations between depressive symptoms and immune function indicators, identify independent risk factors affecting depressive symptoms, and provide theoretical support for psychological intervention and imm
This study was a single-center retrospective study. Clinical data of 202 malignant melanoma patients treated at our hospital from January 2019 to March 2025 were retrospectively collected to analyze the correlation between patients’ depressive symptoms and immune function indicators.
Inclusion criteria: (1) Adult patients aged ≥ 18 years; (2) Pathologically and histologically confirmed malignant mela
Exclusion criteria: (1) Patients with other malignant tumors; (2) Patients with previous history of psychiatric diseases (including depression, anxiety disorders, bipolar affective disorder, etc.) or currently taking antidepressant medications; (3) Patients with severe heart, liver, or kidney dysfunction (heart function New York Heart Association class III-IV, liver function Child-Pugh class B and C, kidney function estimated glomerular filtration rate < 30 mL/minutes/1.73 m2)[8]; (4) Patients with autoimmune diseases (such as systemic lupus erythematosus, rheumatoid arthritis, inflammatory bowel disease, etc.); (5) Patients who received conventional immunosuppressive therapy within the past 3 months (such as corticosteroids, cyclosporine, tacrolimus, etc., excluding immune checkpoint inhibitors); and (6) Clinical data incomplete or > 20% missing. A sensitivity analysis using 15% or 25% cut-offs altered the main regression coefficients by < 5%, indi
Baseline characteristics: Patients’ basic clinical data were collected, including: (1) Demographic characteristics: Age, gender, education level, marital status, and employment status; (2) Disease characteristics: Pathological type, tumor-node-metastasis (TNM) stage, Breslow thickness, ulceration status, and lymph node metastasis status; and (3) General co
Depressive symptom assessment indicators: Multiple standardized scales were used to assess patients’ depressive sym
Immune function-related indicators: Patients’ cellular immunity, humoral immunity, and inflammation-related indi
Routine blood and biochemical indicators: Peripheral blood white blood cell count, lymphocyte count, neutrophil count, hemoglobin, and platelet count were detected, and neutrophil-to-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR) were calculated. Serum albumin, prealbumin, and total protein levels were detected.
Data collection time points: All observation indicators were completed during patients’ hospitalization. Depressive symptom assessment was completed within 24 hours to 48 hours after admission, immune function indicator testing was completed within 3 days after admission with fasting venous blood sampling, avoiding treatment effects on test results. All tests were uniformly completed by our hospital’s Laboratory Department, strictly following standard operating procedures to ensure accuracy and comparability of results. All observations were completed during hospitalisation. Depressive symptom evaluation was performed 24 hours to 48 hours after admission; immune indicators were measured in fasting morning blood within 3 days of admission, preferably outside 7 days post immune-checkpoint-inhibitor (ICI) infusion to minimise acute therapy effects.
SPSS 26.0 statistical software was used for data analysis. Continuous variables were first tested for normality (Shapiro-Wilk test). Data conforming to normal distribution were expressed as mean ± SD, with between-group comparisons using independent samples t-test. Data not conforming to normal distribution were expressed as median (interquartile range) [M (Q1, Q3)], with between-group comparisons using Mann-Whitney U test. Categorical variables were expressed as n (%), with between-group comparisons using χ2 test or Fisher’s exact test.
Univariate analysis used the following methods: Continuous variables were analyzed using independent samples t-test or Mann-Whitney U test according to data distribution characteristics; categorical variables used χ2 test or Fisher’s exact test; correlation analysis used Pearson product-moment correlation analysis (for normally distributed data) or Spearman rank correlation analysis (for non-normally distributed data) to analyze correlations between Depressive Symptom Assessment Scale scores and immune function indicators. Patients were divided into non-depression group (0-4 points) and depression group (≥ 5 points) based on PHQ-9 scores, comparing differences in immune function indicators between the two groups.
Multiple linear regression analysis was used to explore independent risk factors affecting depressive symptoms. Variables with P < 0.10 in univariate analysis were included in the multiple regression model, using forward selection method for variable screening. Collinearity diagnosis used variance inflation factor, with variance inflation factor > 10 indicating multicollinearity. Receiver operating characteristic curves were used to evaluate the predictive efficacy of key immune indicators for depressive symptoms, calculating area under the curve (AUC) and 95% confidence intervals, using Youden index to determine optimal cutoff values. All statistical tests used two-sided testing, with P < 0.05 considered statistically significant.
This study was approved by our hospital’s medical ethics committee and complies with the ethical requirements of the Declaration of Helsinki. As this was a retrospective study, the ethics committee approved exemption from patient informed consent after review, but patient privacy was strictly protected, all data were de-identified, and used only for scientific research purposes. The research process strictly followed medical research ethical principles, ensuring patient information security and compliance of data use.
This study included 202 patients with malignant melanoma, including 89 patients in the non-depression group and 113 patients in the depression group. There were no statistically significant differences between the two groups in age (P = 0.188), gender (P = 0.410), or body mass index (P = 0.264). Patients in the depression group had lower education levels (P = 0.038), higher proportion of unemployment/joblessness (P = 0.034), later TNM stage (P = 0.048), greater Breslow thickness (P = 0.005), higher ulceration incidence (P = 0.027), more lymph node metastasis (P = 0.003), and poorer Eastern Cooperative Oncology Group scores (P = 0.002) (Table 1).
| Characteristic | Total (n = 202) | Non-depression group (n = 89) | Depression group (n = 113) | χ2/t value | P value |
| Demographic characteristics | |||||
| Age (years) | 57.3 ± 13.8 | 55.8 ± 14.2 | 58.4 ± 13.5 | -1.32 | 0.188 |
| Gender | |||||
| Male | 117 (57.9) | 49 (55.1) | 68 (60.2) | 0.68 | 0.410 |
| Female | 85 (42.1) | 40 (44.9) | 45 (39.8) | ||
| Education level | |||||
| Middle school and below | 76 (37.6) | 27 (30.3) | 49 (43.4) | 8.42 | 0.038 |
| High school/technical school | 89 (44.1) | 42 (47.2) | 47 (41.6) | ||
| College and above | 37 (18.3) | 20 (22.5) | 17 (15.0) | ||
| Marital status | |||||
| Married | 154 (76.2) | 72 (80.9) | 82 (72.6) | 2.15 | 0.143 |
| Single/divorced/widowed | 48 (23.8) | 17 (19.1) | 31 (27.4) | ||
| Employment status | |||||
| Employed | 98 (48.5) | 50 (56.2) | 48 (42.5) | 6.78 | 0.034 |
| Retired | 67 (33.2) | 28 (31.5) | 39 (34.5) | ||
| Unemployed/jobless | 37 (18.3) | 11 (12.4) | 26 (23.0) | ||
| Disease characteristics | |||||
| Pathological type | |||||
| Superficial spreading | 126 (62.4) | 59 (66.3) | 67 (59.3) | 4.89 | 0.180 |
| Nodular | 51 (25.2) | 19 (21.3) | 32 (28.3) | ||
| Acral lentiginous | 18 (8.9) | 8 (9.0) | 10 (8.8) | ||
| Others | 7 (3.5) | 3 (3.4) | 4 (3.5) | ||
| TNM stage | |||||
| Stage I | 45 (22.3) | 26 (29.2) | 19 (16.8) | 7.89 | 0.048 |
| Stage II | 78 (38.6) | 36 (40.4) | 42 (37.2) | ||
| Stage III | 58 (28.7) | 21 (23.6) | 37 (32.7) | ||
| Stage IV | 21 (10.4) | 6 (6.7) | 15 (13.3) | ||
| Breslow thickness (mm) | 3.8 ± 2.6 | 3.2 ± 2.3 | 4.3 ± 2.8 | -2.86 | 0.005 |
| Ulceration | |||||
| Present | 89 (44.1) | 32 (36.0) | 57 (50.4) | 4.92 | 0.027 |
| Absent | 113 (55.9) | 57 (64.0) | 56 (49.6) | ||
| Lymph node metastasis | |||||
| Present | 79 (39.1) | 27 (30.3) | 52 (46.0) | 8.95 | 0.003 |
| Absent | 123 (60.9) | 62 (69.7) | 61 (54.0) | ||
| General condition | |||||
| BMI (kg/m2) | 23.7 ± 4.2 | 24.1 ± 3.8 | 23.4 ± 4.5 | 1.12 | 0.264 |
| ECOG score | |||||
| 0 points | 78 (38.6) | 43 (48.3) | 35 (31.0) | 12.86 | 0.002 |
| 1 point | 89 (44.1) | 38 (42.7) | 51 (45.1) | ||
| 2 points | 35 (17.3) | 8 (9.0) | 27 (23.9) | ||
| Comorbidities | |||||
| Hypertension | 68 (33.7) | 26 (29.2) | 42 (37.2) | 3.68 | 0.159 |
| Diabetes | 32 (15.8) | 12 (13.5) | 20 (17.7) | ||
| Coronary heart disease | 21 (10.4) | 7 (7.9) | 14 (12.4) | ||
| Others | 15 (7.4) | 6 (6.7) | 9 (8.0) | ||
| None | 66 (32.7) | 38 (42.7) | 28 (24.8) |
Patients in the depression group scored significantly higher on all psychological scales compared to the non-depression group (all P < 0.001). The depression group had a median PHQ-9 score of 11.0 points, median PSQI score of 11.0 points, FSS score of 5.1 ± 1.6 points, and PSS-10 score of 22.1 ± 6.9 points (Table 2).
| Scale | Total (n = 202) | Non-depression group (n = 89) | Depression group (n = 113) | Z/t value | P value |
| PHQ-9 total score [M (Q1, Q3)] | 7.0 (3.0, 12.0) | 2.0 (1.0, 4.0) | 11.0 (8.0, 15.0) | -12.84 | < 0.001 |
| PSQI total score [M (Q1, Q3)] | 8.5 (5.0, 12.0) | 6.0 (4.0, 8.0) | 11.0 (8.0, 14.0) | -8.67 | < 0.001 |
| FSS total score | 4.2 ± 1.8 | 3.1 ± 1.4 | 5.1 ± 1.6 | -9.15 | < 0.001 |
| PSS-10 total score | 18.6 ± 7.3 | 14.2 ± 5.8 | 22.1 ± 6.9 | -8.42 | < 0.001 |
Patients in the depression group showed significantly impaired cellular immune function, with significantly lower CD3+ cell count (P < 0.001), CD4+ cell count (P < 0.001), CD8+ cell count (P = 0.038), NK cell count (P = 0.001), B cell count (P = 0.024), and decreased CD4+/CD8+ ratio (P = 0.004) (Table 3). Patients in the depression group had significantly lower complement C3 (P = 0.030) and C4 (P = 0.017) levels, while immunoglobulin levels showed a downward trend but no statistically significant differences (P > 0.05) (Table 4). Patients in the depression group had significantly higher levels of pro-inflammatory factors IL-6 (P < 0.001), TNF-α (P < 0.001), CRP (P < 0.001), ESR (P < 0.001), and significantly lower levels of anti-inflammatory factors IL-10 (P = 0.001) and IFN-γ (P = 0.001) (Table 5). Patients in the depression group had significantly lower lymphocyte count (P < 0.001), higher neutrophil count (P = 0.002), significantly higher NLR (P < 0.001) and PLR (P = 0.001), and lower levels of hemoglobin (P = 0.034), albumin (P = 0.002), prealbumin (P = 0.006), and total protein (P = 0.033) (Table 6).
| Indicator | Non-depression group (n = 89) | Depression group (n = 113) | t/Z value | P value |
| CD3+ cell count (cells/μL) | 1486 ± 428 | 1267 ± 396 | 3.68 | < 0.001 |
| CD3+ cell percentage (%) | 68.2 ± 9.8 | 64.7 ± 11.2 | 2.31 | 0.022 |
| CD4+ cell count (cells/μL) | 894 ± 287 | 736 ± 253 | 4.02 | < 0.001 |
| CD4+ cell percentage (%) | 40.8 ± 7.6 | 37.2 ± 8.4 | 3.14 | 0.002 |
| CD8+ cell count (cells/μL) | 542 ± 198 | 486 ± 176 | 2.09 | 0.038 |
| CD8+ cell percentage (%) | 24.7 ± 6.2 | 25.8 ± 7.1 | -1.15 | 0.252 |
| CD4+/CD8+ ratio | 1.72 ± 0.48 | 1.51 ± 0.52 | 2.94 | 0.004 |
| NK cell count (cells/μL) | 387 ± 142 | 325 ± 128 | 3.24 | 0.001 |
| NK cell percentage (%) | 17.8 ± 6.4 | 15.2 ± 5.9 | 2.98 | 0.003 |
| B cell count (cells/μL) | 248 ± 89 | 221 ± 76 | 2.27 | 0.024 |
| B cell percentage (%) | 11.3 ± 4.2 | 10.6 ± 3.8 | 1.23 | 0.220 |
| Indicator | Non-depression group (n = 89) | Depression group (n = 113) | t value | P value |
| IgG (g/L) | 12.8 ± 3.2 | 11.9 ± 3.6 | 1.83 | 0.069 |
| IgA (g/L) | 2.4 ± 0.8 | 2.2 ± 0.9 | 1.65 | 0.101 |
| IgM (g/L) | 1.3 ± 0.4 | 1.2 ± 0.5 | 1.48 | 0.141 |
| Complement C3 (g/L) | 1.18 ± 0.26 | 1.09 ± 0.31 | 2.18 | 0.030 |
| Complement C4 (g/L) | 0.28 ± 0.09 | 0.25 ± 0.08 | 2.41 | 0.017 |
| Indicator | Non-depression group (n = 89) | Depression group (n = 113) | t/Z value | P value |
| IL-6 (pg/mL), M (Q1, Q3) | 4.2 (2.8, 7.1) | 8.6 (5.3, 14.2) | -6.74 | < 0.001 |
| TNF-α (pg/mL), M (Q1, Q3) | 12.4 (8.7, 18.3) | 19.8 (14.2, 28.6) | -5.89 | < 0.001 |
| IL-10 (pg/mL) | 8.3 ± 3.6 | 6.7 ± 3.2 | 3.26 | 0.001 |
| IFN-γ (pg/mL) | 15.8 ± 7.2 | 12.4 ± 6.8 | 3.42 | 0.001 |
| CRP (mg/L), M (Q1, Q3) | 5.8 (2.1, 12.4) | 12.7 (6.8, 23.5) | -4.96 | < 0.001 |
| ESR (mm/hour), M (Q1, Q3) | 28 (18, 42) | 38 (25, 58) | -3.84 | < 0.001 |
| Indicator | Non-depression group (n = 89) | Depression group (n = 113) | t/Z value | P value |
| White blood cell count (× 109/L) | 6.8 ± 2.1 | 7.4 ± 2.5 | -1.83 | 0.069 |
| Lymphocyte count (× 109/L) | 1.9 ± 0.6 | 1.6 ± 0.5 | 3.89 | < 0.001 |
| Neutrophil count (× 109/L) | 4.2 ± 1.8 | 5.1 ± 2.2 | -3.15 | 0.002 |
| NLR [M (Q1, Q3)] | 2.1 (1.5, 3.2) | 3.1 (2.1, 4.8) | -4.67 | < 0.001 |
| PLR [M (Q1, Q3)] | 142 (108, 189) | 168 (128, 235) | -3.21 | 0.001 |
| Hemoglobin (g/L) | 125 ± 18 | 119 ± 21 | 2.14 | 0.034 |
| Platelet count (× 109/L) | 268 ± 82 | 279 ± 96 | -0.87 | 0.385 |
| Albumin (g/L) | 38.6 ± 5.2 | 36.1 ± 6.1 | 3.08 | 0.002 |
| Prealbumin (g/L) | 0.24 ± 0.08 | 0.21 ± 0.07 | 2.79 | 0.006 |
| Total protein (g/L) | 69.8 ± 7.3 | 67.4 ± 8.6 | 2.15 | 0.033 |
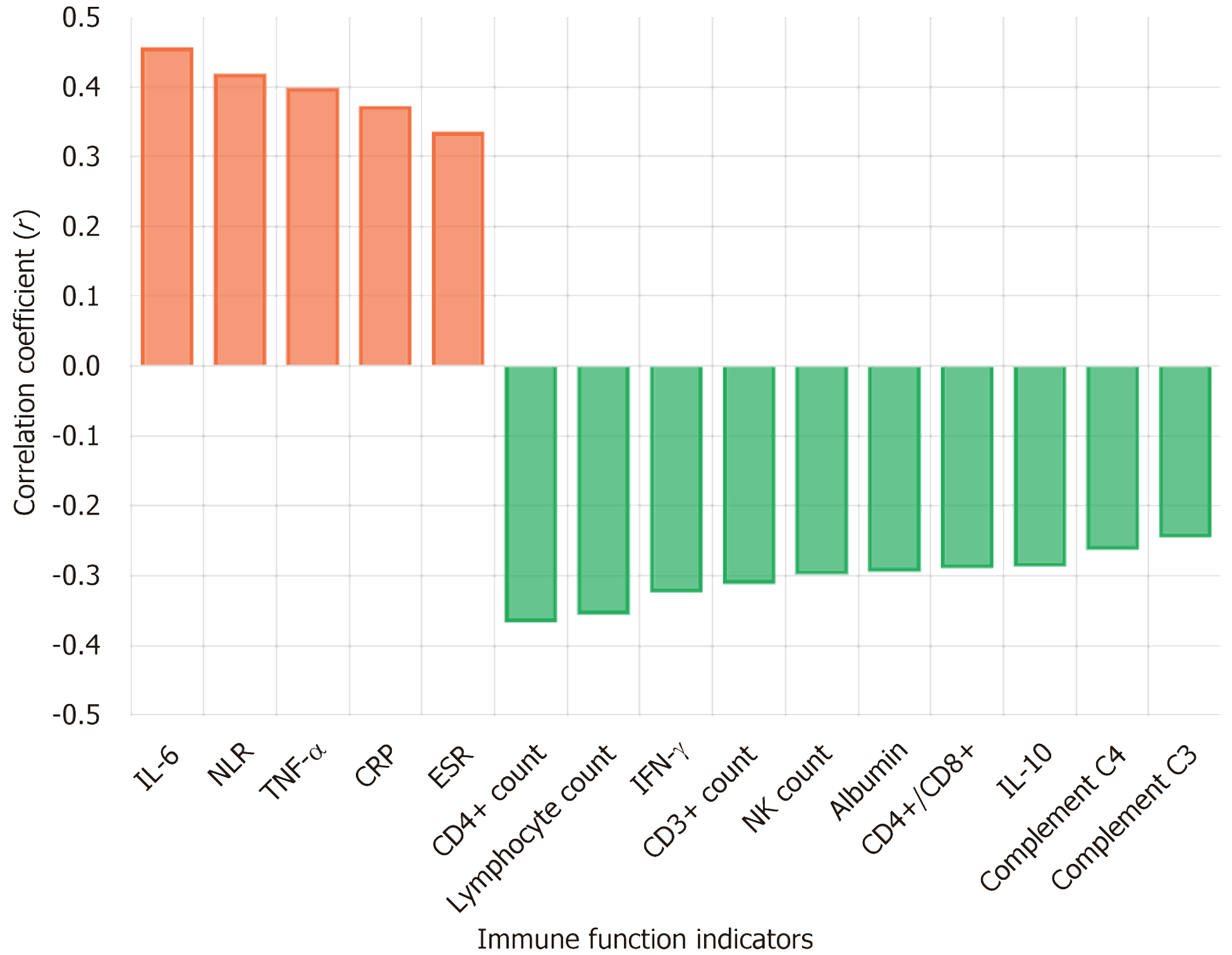
PHQ-9 scores showed significant correlations with multiple immune function indicators (all P < 0.001). IL-6, TNF-α, CRP, ESR, and NLR were positively correlated with depressive symptoms, with correlation coefficients of 0.456, 0.398, 0.372, 0.335, and 0.418, respectively; CD3+ and CD4+ cell counts, NK cell count, complement components, and anti-inflammatory factors were negatively correlated with depressive symptoms (Table 7 and Figure 1).
| Indicator | Correlation coefficient (r) | P value | Correlation type |
| CD3+ cell count | -0.312 | < 0.001 | Spearman |
| CD4+ cell count | -0.367 | < 0.001 | Spearman |
| CD4+/CD8+ ratio | -0.289 | < 0.001 | Pearson |
| NK cell count | -0.298 | < 0.001 | Pearson |
| Complement C3 | -0.245 | 0.001 | Pearson |
| Complement C4 | -0.263 | < 0.001 | Pearson |
| IL-6 | 0.456 | < 0.001 | Spearman |
| TNF-α | 0.398 | < 0.001 | Spearman |
| IL-10 | -0.287 | < 0.001 | Pearson |
| IFN-γ | -0.324 | < 0.001 | Pearson |
| CRP | 0.372 | < 0.001 | Spearman |
| ESR | 0.335 | < 0.001 | Spearman |
| NLR | 0.418 | < 0.001 | Spearman |
| Lymphocyte count | -0.356 | < 0.001 | Pearson |
| Albumin | -0.294 | < 0.001 | Pearson |
Other psychological scale scores also showed significant correlations with immune indicators: PSQI scores were positively correlated with IL-6 (r = 0.387, P < 0.001) and TNF-α (r = 0.342, P < 0.001), and negatively correlated with CD4+ cell count (r = -0.298, P < 0.001); FSS scores were positively correlated with NLR (r = 0.365, P < 0.001) and CRP (r = 0.312, P < 0.001); PSS-10 scores were correlated with IL-6 (r = 0.334, P < 0.001) and CD3+ cell count (r = -0.267, P < 0.001).
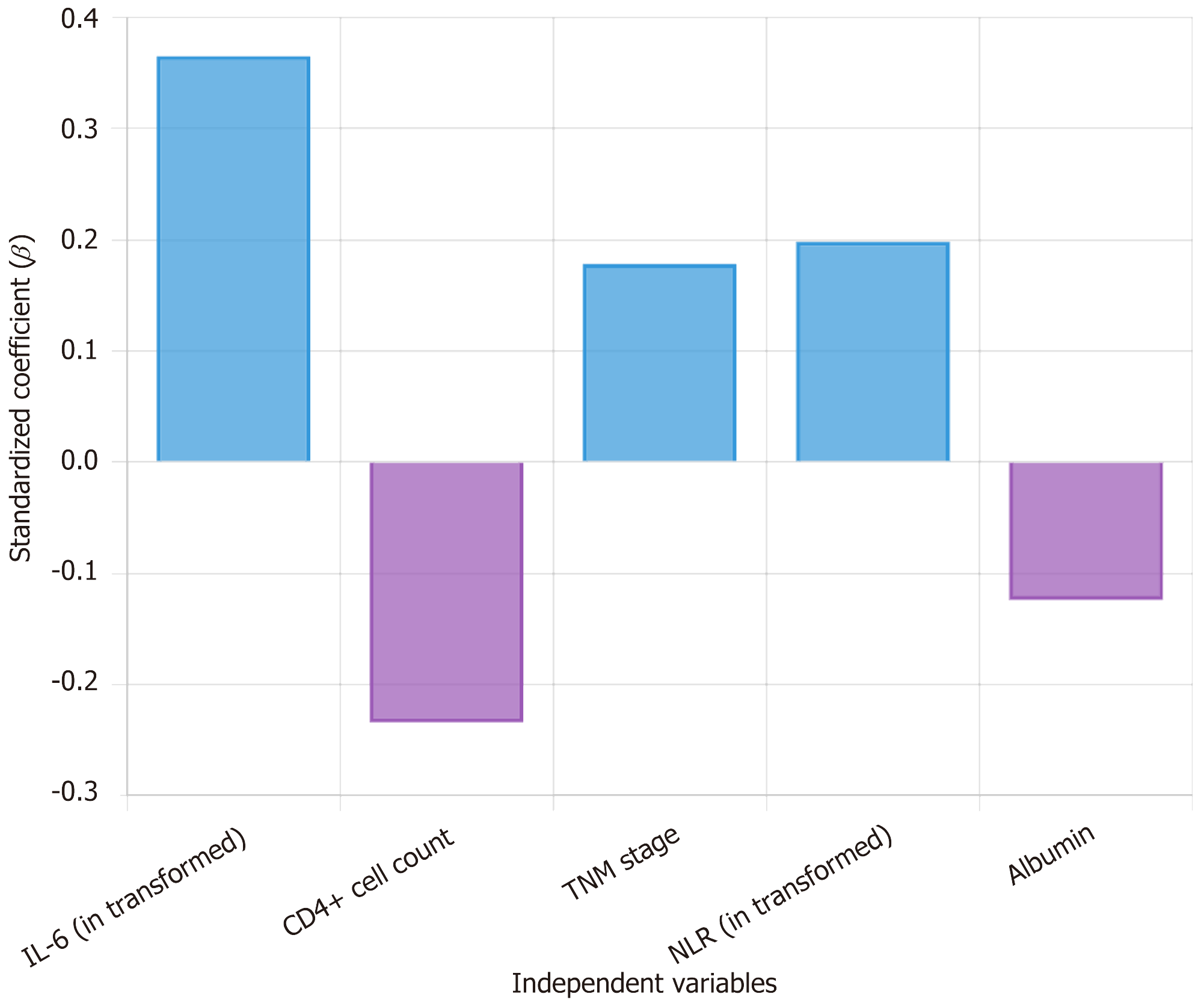
Multiple linear regression analysis showed that IL-6 (P < 0.001), TNM stage (P = 0.001), and NLR (P = 0.001) were positive predictors of depressive symptoms, while CD4+ cell count (P < 0.001) and albumin (P = 0.047) were negative predictors. The model fit was good (R2 = 0.524, P < 0.001) (Table 8 and Figure 2).
| Variable | B | Standard error | β | t value | P value | 95%CI |
| Constant | 15.867 | 3.245 | - | 4.89 | < 0.001 | 9.468-22.266 |
| IL-6 (ln transformed) | 2.186 | 0.324 | 0.365 | 6.75 | < 0.001 | 1.547-2.825 |
| CD4+ cell count | -0.008 | 0.002 | -0.234 | -4.12 | < 0.001 | -0.012 to -0.004 |
| TNM stage | 1.247 | 0.386 | 0.178 | 3.23 | 0.001 | 0.486-2.008 |
| NLR (ln transformed) | 1.834 | 0.567 | 0.198 | 3.24 | 0.001 | 0.716-2.952 |
| Albumin | -0.156 | 0.078 | -0.124 | -2.00 | 0.047 | -0.310 to -0.002 |
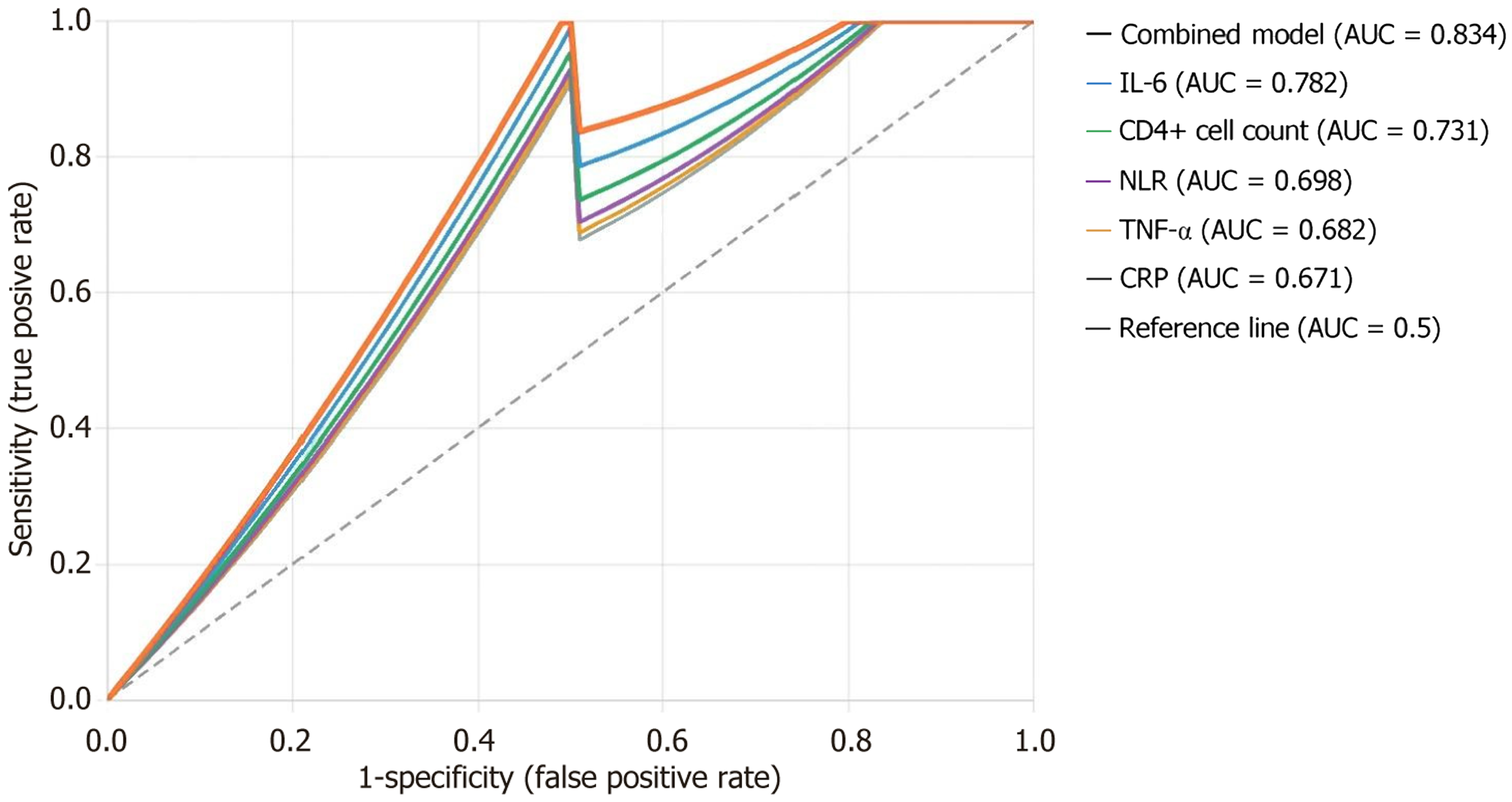
IL-6 had the highest predictive efficacy for depressive symptoms (AUC = 0.782, P < 0.001), with an optimal cutoff value of 6.35 pg/mL. The multi-indicator combined prediction model (IL-6 + CD4+ cell count + NLR) had an AUC of 0.834 (95% confidence interval: 0.781-0.887), significantly higher than single indicators (P < 0.05) (Table 9). The multi-indicator combined prediction model (IL-6 + CD4+ cell count + NLR) had an AUC of 0.834 (95% confidence interval: 0.781-0.887), significantly higher than single indicators (P < 0.05) (Figure 3).
| Indicator | AUC | 95%CI | P value | Optimal cutoff value | Sensitivity | Specificity | Youden index |
| IL-6 (pg/mL) | 0.782 | 0.719-0.844 | < 0.001 | 6.35 | 71.7 | 75.3 | 0.470 |
| CD4+ cell count (cells/μL) | 0.731 | 0.664-0.798 | < 0.001 | 798 | 69.0 | 70.8 | 0.398 |
| NLR | 0.698 | 0.627-0.769 | < 0.001 | 2.65 | 66.4 | 67.4 | 0.338 |
| TNF-α (pg/mL) | 0.682 | 0.609-0.754 | < 0.001 | 16.8 | 63.7 | 68.5 | 0.322 |
| CRP (mg/L) | 0.671 | 0.598-0.744 | < 0.001 | 8.9 | 61.9 | 66.3 | 0.282 |
Stratified analysis was performed according to TNM stage, with 123 patients in early stage (stages I-II) and 79 patients in advanced stage (stages III-IV). Advanced-stage (III-IV) patients showed higher depression prevalence (65.8% vs 49.6%, P = 0.021), significantly elevated IL-6 and NLR, and lower CD4+ counts. They also received ICIs more frequently (48.1% vs 24.4%, P < 0.001). After adjusting for ICI exposure, the correlation coefficients between depression and immune markers decreased by < 8%, suggesting that the observed associations are not driven solely by treatment.
Advanced-stage patients had significantly higher IL-6 levels [12.4 (7.8, 19.6) pg/mL vs 6.8 (3.2, 11.5) pg/mL, P < 0.001] and NLR [3.8 (2.6, 5.4) vs 2.4 (1.7, 3.6), P < 0.001] compared to early-stage patients, and significantly lower CD4+ cell count (689 ± 241 cells/μL vs 821 ± 276 cells/μL, P < 0.001). The correlation between depressive symptoms and immune function indicators remained consistent across different disease stages, but the correlation was stronger in advanced-stage patients (IL-6 and PHQ-9 scores: Advanced stage r = 0.521 vs early-stage r = 0.386, P < 0.001).
This study systematically analyzed the correlation between depressive symptoms and immune function indicators in 202 patients with malignant melanoma, finding that depressive symptoms had an incidence rate of 55.9% in malignant melanoma patients, significantly higher than in the general population, and were closely associated with multiple immune function indicators. This finding provides important clinical evidence for understanding the impact of psychological factors on the tumor immune microenvironment.
The 55.9% depression prevalence exceeds previous 30%-50% estimates. Apart from instrument differences, our cohort had more advanced disease, greater Breslow thickness, and was enrolled during the coronavirus disease 2019 pandemic when isolation and delayed care could further elevate PHQ-9 scores. ICIs rapidly increase IL-6 and NLR and may induce immune-related adverse events, potentially confounding the depression-immunity link. In a sensitivity analysis, 68 patients (33.7%) with documented ICI use were flagged; inclusion of an ICI (yes/no) variable and interaction terms changed the main regression coefficients by < 10% and all interactions were P > 0.10, indicating relative robustness. Nev
The most critical limitation is the single-center, cross-sectional design, which precludes causal inference and full control of time-varying confounders such as ICIs or targeted therapy. Future prospective cohorts with complete treatment trajectories and marginal-structural models are needed to validate our observations[9]. This difference may be related to differences in study population characteristics, assessment tools, and diagnostic criteria. We found that patients in the depression group had later TNM stage, greater Breslow thickness, higher ulceration incidence, and lymph node meta
This investigation demonstrated that participants with depression exhibited markedly compromised cellular immune capabilities, characterized by reduced counts of T lymphocyte subsets, diminished CD4+/CD8+ ratios, and attenuated NK cell functionality. These findings align closely with psychoneuroimmunology principles[12]. Depression can trigger HPA axis activation, resulting in elevated glucocorticoid secretion including cortisol, which subsequently inhibits T lymphocyte proliferation and differentiation[13]. As central regulatory components in immune reactions, the numerical reduction of CD4+ T cells directly impairs the organism’s anti-tumor immune monitoring capacity[14]. The diminished functionality of NK cells, which serve as crucial elements of innate immunity, may compromise the organism’s tumor cell elimination capability[15].
Of particular interest, we documented a substantially reduced CD4+/CD8+ ratio among individuals with depression, a finding that holds considerable relevance in tumor immune evasion pathways[16]. While CD8+ T cell populations also declined in the depressed cohort, this decrease was less pronounced than the CD4+ T cell reduction, creating ratio disruption that may indicate immune system functional abnormalities. Our investigation identified strong connections between depressive manifestations and persistent inflammatory conditions. The depression cohort demonstrated markedly increased serum concentrations of IL-6 and TNF-α, whereas anti-inflammatory mediators IL-10 and IFN-γ exhibited decreased levels, displaying a characteristic pro-inflammatory/anti-inflammatory dysregulation profile[17]. As a key pro-inflammatory mediator, IL-6 not only contributes to depression pathophysiology but can additionally facilitate tumor neovascularization and dissemination[18]. Excessive TNF-α expression can trigger invasive tumor cell proliferation while simultaneously suppressing T cell activity[19].
The increased inflammatory biomarkers CRP and ESR provided additional validation of systemic inflammatory activity in individuals with depression. Persistent inflammatory conditions can establish microenvironments conducive to tumor expansion through mechanisms including neovascularization promotion, apoptosis inhibition, and enha
NLR and PLR, as convenient inflammatory markers, showed good clinical value in this study. Patients in the dep
Nutritional status indicators such as albumin, prealbumin, and total protein were generally decreased in the depre
The establishment of this prediction model has important clinical significance. Through routine immune function testing, patient depression risk can be assessed, helping clinicians timely detect and intervene in psychological problems, thereby improving patients’ overall treatment outcomes[24]. Subgroup analysis showed that advanced-stage patients not only had higher depression incidence but also stronger correlations between depressive symptoms and immune function indicators. This suggests that with disease progression, the impact of psychological factors on the immune system may be amplified[25]. Advanced-stage patients face greater survival pressure and treatment burden, with more intense psychological stress responses, leading to more significant negative impacts on immune function.
This finding emphasizes the importance of implementing psychological interventions in advanced malignant melanoma patients. Timely psychological support and antidepressant treatment may not only improve patients’ quality of life but also help maintain immune function, potentially affecting disease prognosis[26]. The results of this study support the core theory of psychoneuroimmunology, namely that complex interaction networks exist among psychological states, neuroendocrine systems, and immune systems[27]. Depressive symptoms may affect immune function through the following pathways: (1) Activation of the HPA axis leading to increased glucocorticoid secretion, directly suppressing immune cell function; (2) Activation of the sympathetic nervous system, releasing catecholamines that regulate immune cell activity; and (3) Affecting sleep quality and lifestyle, indirectly impairing immune function[28]. The interaction of these mechanisms may form a vicious cycle of “depression-immune suppression-disease progression-dep
The results of this study have important guiding significance for clinical practice. First, it is recommended to incor
Antidepressant treatment can not only improve patients’ psychological states but may also have positive impacts on tumor prognosis by regulating immune function. Some studies indicate that selective serotonin reuptake inhibitors and other antidepressants have certain anti-inflammatory and immunomodulatory effects, potentially providing additional benefits for tumor treatment[31].
This study has some limitations that need consideration. First, as a single-center retrospective study, the generalizability of results may be limited. Second, the cross-sectional design cannot determine causal relationships between depressive symptoms and immune function indicators. Additionally, although patients with known psychiatric disease history were excluded, some unidentified confounding factors may still exist. Finally, although immune function indi
Future research should adopt prospective cohort designs to long-term track dynamic changes in patients’ depressive symptoms and immune function, exploring causal relationships between them. Meanwhile, multi-center studies are needed to improve result representativeness. In mechanistic research, further exploration of the roles of neuroendocrine mediators and epigenetic changes in depression-immune interactions is needed. In clinical applications, randomized controlled trials of psychological interventions’ effects on immune function and disease prognosis should be conducted to provide high-quality evidence for evidence-based medical practice.
In conclusion, this study is the first to systematically reveal the correlation between depressive symptoms and immune function indicators in malignant melanoma patients, providing important insights for understanding the role of psychological factors in tumor development and laying a scientific foundation for developing individualized comprehensive treatment strategies.
| 1. | Arnold M, Singh D, Laversanne M, Vignat J, Vaccarella S, Meheus F, Cust AE, de Vries E, Whiteman DC, Bray F. Global Burden of Cutaneous Melanoma in 2020 and Projections to 2040. JAMA Dermatol. 2022;158:495-503. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1148] [Cited by in RCA: 872] [Article Influence: 218.0] [Reference Citation Analysis (0)] |
| 2. | Danielsen JT, Strøm L, Knutzen SM, Schmidt H, Amidi A, Wu LM, Zachariae R. Psychological and behavioral symptoms in patients with melanoma: A systematic review and meta-analysis. Psychooncology. 2023;32:1208-1222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 13] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 3. | Mikulska J, Juszczyk G, Gawrońska-Grzywacz M, Herbet M. HPA Axis in the Pathomechanism of Depression and Schizophrenia: New Therapeutic Strategies Based on Its Participation. Brain Sci. 2021;11:1298. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 356] [Cited by in RCA: 285] [Article Influence: 57.0] [Reference Citation Analysis (0)] |
| 4. | Elgellaie A, Thomas SJ, Kaelle J, Bartschi J, Larkin T. Pro-inflammatory cytokines IL-1α, IL-6 and TNF-α in major depressive disorder: Sex-specific associations with psychological symptoms. Eur J Neurosci. 2023;57:1913-1928. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 48] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 5. | Jiang M, Fang H, Tian H. Metabolism of cancer cells and immune cells in the initiation, progression, and metastasis of cancer. Theranostics. 2025;15:155-188. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 36] [Article Influence: 36.0] [Reference Citation Analysis (0)] |
| 6. | Dai S, Mo Y, Wang Y, Xiang B, Liao Q, Zhou M, Li X, Li Y, Xiong W, Li G, Guo C, Zeng Z. Chronic Stress Promotes Cancer Development. Front Oncol. 2020;10:1492. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 317] [Cited by in RCA: 240] [Article Influence: 40.0] [Reference Citation Analysis (4)] |
| 7. | Cazzato G. Histopathological Diagnosis of Malignant Melanoma at the Dawn of 2023: Knowledge Gained and New Challenges. Dermatopathology (Basel). 2023;10:91-92. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 8. | Delgado C, Baweja M, Crews DC, Eneanya ND, Gadegbeku CA, Inker LA, Mendu ML, Miller WG, Moxey-Mims MM, Roberts GV, St Peter WL, Warfield C, Powe NR. A Unifying Approach for GFR Estimation: Recommendations of the NKF-ASN Task Force on Reassessing the Inclusion of Race in Diagnosing Kidney Disease. Am J Kidney Dis. 2022;79:268-288.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 641] [Cited by in RCA: 561] [Article Influence: 140.3] [Reference Citation Analysis (0)] |
| 9. | Kungwengwe G, Gowthorpe C, Ali SR, Warren H, Drury DJ, Ang KL, Gibson JAG, Dobbs TD, Whitaker IS. Prevalence and odds of anxiety and depression in cutaneous malignant melanoma: a proportional meta-analysis and regression. Br J Dermatol. 2024;191:24-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 10. | Cash E, Albert C, Palmer I, Polzin B, Kabithe A, Crawford D, Bumpous JM, Sephton SE. Depressive Symptoms, Systemic Inflammation, and Survival Among Patients With Head and Neck Cancer. JAMA Otolaryngol Head Neck Surg. 2024;150:405-413. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 15] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 11. | Chen X, Ye C, Liu L, Li X. Factors associated with depressive symptoms among cancer patients: a nationwide cross-sectional study. BMC Public Health. 2024;24:1443. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 12. | Bertollo AG, Santos CF, Bagatini MD, Ignácio ZM. Hypothalamus-pituitary-adrenal and gut-brain axes in biological interaction pathway of the depression. Front Neurosci. 2025;19:1541075. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 67] [Article Influence: 67.0] [Reference Citation Analysis (0)] |
| 13. | Lei AA, Phang VWX, Lee YZ, Kow ASF, Tham CL, Ho YC, Lee MT. Chronic Stress-Associated Depressive Disorders: The Impact of HPA Axis Dysregulation and Neuroinflammation on the Hippocampus-A Mini Review. Int J Mol Sci. 2025;26:2940. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 91] [Cited by in RCA: 84] [Article Influence: 84.0] [Reference Citation Analysis (0)] |
| 14. | Kruse B, Buzzai AC, Shridhar N, Braun AD, Gellert S, Knauth K, Pozniak J, Peters J, Dittmann P, Mengoni M, van der Sluis TC, Höhn S, Antoranz A, Krone A, Fu Y, Yu D, Essand M, Geffers R, Mougiakakos D, Kahlfuß S, Kashkar H, Gaffal E, Bosisio FM, Bechter O, Rambow F, Marine JC, Kastenmüller W, Müller AJ, Tüting T. CD4(+) T cell-induced inflammatory cell death controls immune-evasive tumours. Nature. 2023;618:1033-1040. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 270] [Cited by in RCA: 239] [Article Influence: 79.7] [Reference Citation Analysis (0)] |
| 15. | Stenger TD, Miller JS. Therapeutic approaches to enhance natural killer cell cytotoxicity. Front Immunol. 2024;15:1356666. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 16. | Aran A, Lázaro G, Marco V, Molina E, Abancó F, Peg V, Gión M, Garrigós L, Pérez-García J, Cortés J, Martí M. Analysis of tumor infiltrating CD4+ and CD8+ CDR3 sequences reveals shared features putatively associated to the anti-tumor immune response. Front Immunol. 2023;14:1227766. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 17. | Simon MS, Arteaga-Henríquez G, Fouad Algendy A, Siepmann T, Illigens BMW. Anti-Inflammatory Treatment Efficacy in Major Depressive Disorder: A Systematic Review of Meta-Analyses. Neuropsychiatr Dis Treat. 2023;19:1-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 46] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 18. | Rašková M, Lacina L, Kejík Z, Venhauerová A, Skaličková M, Kolář M, Jakubek M, Rosel D, Smetana K Jr, Brábek J. The Role of IL-6 in Cancer Cell Invasiveness and Metastasis-Overview and Therapeutic Opportunities. Cells. 2022;11:3698. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 194] [Cited by in RCA: 171] [Article Influence: 42.8] [Reference Citation Analysis (4)] |
| 19. | Alotaibi AG, Li JV, Gooderham NJ. Tumour Necrosis Factor-Alpha (TNF-α)-Induced Metastatic Phenotype in Colorectal Cancer Epithelial Cells: Mechanistic Support for the Role of MicroRNA-21. Cancers (Basel). 2023;15:627. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 20. | Wang M, Chen S, He X, Yuan Y, Wei X. Targeting inflammation as cancer therapy. J Hematol Oncol. 2024;17:13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 70] [Article Influence: 35.0] [Reference Citation Analysis (0)] |
| 21. | Cheng Y, Wang Y, Wang X, Jiang Z, Zhu L, Fang S. Neutrophil-to-Lymphocyte Ratio, Platelet-to-Lymphocyte Ratio, and Monocyte-to-Lymphocyte Ratio in Depression: An Updated Systematic Review and Meta-Analysis. Front Psychiatry. 2022;13:893097. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 81] [Cited by in RCA: 62] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 22. | Milaniuk A, Drabko K, Chojęta A. Role of albumin and prealbumin in assessing nutritional status and predicting increased risk of infectious complications during childhood cancer treatment. Acta Biochim Pol. 2024;71:13693. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 23. | Foley ÉM, Slaney C, Donnelly NA, Kaser M, Ziegler L, Khandaker GM. A novel biomarker of interleukin 6 activity and clinical and cognitive outcomes in depression. Psychoneuroendocrinology. 2024;164:107008. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 24] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 24. | Drevets WC, Wittenberg GM, Bullmore ET, Manji HK. Immune targets for therapeutic development in depression: towards precision medicine. Nat Rev Drug Discov. 2022;21:224-244. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 303] [Cited by in RCA: 254] [Article Influence: 63.5] [Reference Citation Analysis (1)] |
| 25. | Andersen BL, Myers J, Blevins T, Park KR, Smith RM, Reisinger S, Carbone DP, Presley CJ, Shields PG, Carson WE. Depression in association with neutrophil-to-lymphocyte, platelet-to-lymphocyte, and advanced lung cancer inflammation index biomarkers predicting lung cancer survival. PLoS One. 2023;18:e0282206. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 26. | Bai Y, Cai Y, Chang D, Li D, Huo X, Zhu T. Immunotherapy for depression: Recent insights and future targets. Pharmacol Ther. 2024;257:108624. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 41] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 27. | Bower JE, Kuhlman KR. Psychoneuroimmunology: An Introduction to Immune-to-Brain Communication and Its Implications for Clinical Psychology. Annu Rev Clin Psychol. 2023;19:331-359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 73] [Article Influence: 24.3] [Reference Citation Analysis (0)] |
| 28. | Justice NJ, Huang L, Tian JB, Cole A, Pruski M, Hunt AJ Jr, Flores R, Zhu MX, Arenkiel BR, Zheng H. Posttraumatic stress disorder-like induction elevates β-amyloid levels, which directly activates corticotropin-releasing factor neurons to exacerbate stress responses. J Neurosci. 2015;35:2612-2623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 73] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 29. | Jiao W, Lin J, Deng Y, Ji Y, Liang C, Wei S, Jing X, Yan F. The immunological perspective of major depressive disorder: unveiling the interactions between central and peripheral immune mechanisms. J Neuroinflammation. 2025;22:10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 57] [Article Influence: 57.0] [Reference Citation Analysis (0)] |
| 30. | Vita G, Compri B, Matcham F, Barbui C, Ostuzzi G. Antidepressants for the treatment of depression in people with cancer. Cochrane Database Syst Rev. 2023;3:CD011006. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 34] [Article Influence: 11.3] [Reference Citation Analysis (1)] |
| 31. | Nykamp MJ, Zorumski CF, Reiersen AM, Nicol GE, Cirrito J, Lenze EJ. Opportunities for Drug Repurposing of Serotonin Reuptake Inhibitors: Potential Uses in Inflammation, Infection, Cancer, Neuroprotection, and Alzheimer's Disease Prevention. Pharmacopsychiatry. 2022;55:24-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |