Published online Mar 24, 2026. doi: 10.5306/wjco.v17.i3.116673
Revised: January 21, 2026
Accepted: February 27, 2026
Published online: March 24, 2026
Processing time: 126 Days and 0.7 Hours
Cervical cancer remains a major global health burden, and radiotherapy is central across all disease stages. Interfractional organ motion, tumor regression and setup uncertainty can compromise the accuracy of conventional margin-based radio
Core Tip: Online adaptive radiotherapy (oART) enables individualized, fraction-by-fraction plan adaptation in cervical cancer, addressing tumor regression, organ motion, and anatomic variability. By integrating daily imaging, automated or artificial intelligence-assisted contouring, and on-table plan re-optimization, oART improves target coverage, enhances dose precision, and effectively spares pelvic organs at risk. Evidence demonstrates that oART can reduce acute treatment-related toxicities and provides a practical framework for workflow optimization and clinical implementation in modern cervical cancer radiotherapy.
- Citation: Wang GY, Chen YN, Sun S, Hu K. Online adaptive radiotherapy enhances precision and outcomes in the treatment of cervical cancer. World J Clin Oncol 2026; 17(3): 116673
- URL: https://www.wjgnet.com/2218-4333/full/v17/i3/116673.htm
- DOI: https://dx.doi.org/10.5306/wjco.v17.i3.116673
Cervical cancer remains a major global health burden and a leading cause of cancer-related mortality among women[1]. Radiotherapy plays a central role in treatment for cervical cancer across all disease stages[2]. While radical surgery is indicated for early-stage disease, high-risk factors after surgery require adjuvant radiotherapy or chemoradiotherapy[3-5]. For locally advanced tumors, definitive radiotherapy combined with concurrent chemotherapy remains the standard strategy[6]. Over the past few decades, advances in external beam radiotherapy (EBRT) have evolved from conventional two-dimensional approaches to three-dimensional conformal radiotherapy and, subsequently to intensity-modulated radiotherapy (IMRT). The driving force behind these advances is the ability to conform to target volume coverage while sparing organs at risk (OARs), showing dosimetric and clinical benefits[7,8].
Despite these advances, cervical cancer radiotherapy continues to face ongoing challenges. Beyond tumor regression, these challenges are particularly pronounced, as interfractional variations related to bladder and rectal filling can significantly affect target coverage, limit OAR sparing and increase the risk of normal tissue toxicity[9-11]. To compensate for these effects, large margins based on a wide population have traditionally been used, and in many contemporary protocols, these population-based planning target volume (PTV) expansions already approach the limits of acceptable OAR doses, but such an approach inevitably increases the dose to surrounding OARs[12,13]. These limitations emphasize the need for more individualized and adaptive strategies that can dynamically account for anatomical changes during treatment.
Adaptive radiotherapy (ART) has emerged as a promising solution to address these limitations[14]. By incorporating repeated imaging and plan adaptation during treatment, ART allows for individualized dose delivery that accounts for anatomical and biological changes in real time. However, most early implementation of ART was offline and primarily addressed accumulated anatomical deviations over several fractions, which may be suboptimal for the pronounced day-to-day pelvic variability observed in cervical cancer. With the development of cone beam computed tomography
In this narrative review, we provide a critical overview of oART in cervical cancer, structured around three ov
This minireview was designed as a narrative literature review rather than a formal systematic review. The primary objective was to synthesize current knowledge on oART for cervical cancer from three complementary perspectives: (1) Anatomical and clinical rationale for online adaptation in this disease site; (2) Strength and limitations of existing evidence along the continuum from dosimetric endpoints to toxicity and survival; and (3) Practical considerations for implementation of oART across different resource settings.
A comprehensive literature search was conducted in the PubMed, EMBASE and Web of Science databases. The search covered publications from database inception up to December 2025, in order to capture both foundational work on ART and the most recent developments in oART for cervical cancer. The following combinations of keywords and MeSH terms were used: “cervical cancer” OR “cervical carcinoma” AND “adaptive radiotherapy” OR “adaptive radiation therapy” OR “online adaptive radiotherapy” OR “oART” AND “image-guided radiotherapy” OR “cone-beam CT” OR “CBCT” OR “MR-guided” OR “MRI-linac” AND “planning target volume” OR “PTV” OR “organ at risk” OR “toxicity”. Re
We included studies that met the following criteria: (1) Human studies involving patients with cervical cancer or postoperative gynecological malignancies in which cervical cancer represented a clearly defined subgroup; (2) Use of adaptive EBRT (offline ART or oART) and/or image-guided adaptive brachytherapy with explicit discussion of workflow, dosimetry, or clinical outcomes; (3) Original research articles, prospective or retrospective clinical studies, planning or dosimetric analyses, clinical trial protocols, registry reports, consensus guidelines, or high-quality narrative reviews; and (4) Full-text publications in English. Conference abstracts without full text, non-human studies, and articles unrelated to radiotherapy or adaptive techniques were excluded.
Study selection and data extraction were performed by the authors based on title and abstract screening, followed by full-text assessment of potentially relevant articles. Studies that met the above criteria were included and synthesized in this review, corresponding to the reference list used in the manuscript.
For each included article, we extracted information on study design (prospective vs retrospective; single-center vs multicenter); patient population (intact cervix vs postoperative tumor stage); imaging and treatment platforms (CBCT-, FBCT- and MRI-guided systems); adaptive workflow [offline vs online; adapt-to-position (ATP) vs adapt-to-shape (ATS)]; dosimetric endpoints [e.g., clinical target volume (CTV) D95/D98, PTV coverage, OAR dose-volume parameters]; and reported clinical outcomes (acute and late toxicity, local control, progression-free survival, and overall survival when available).
Given the heterogeneity of study designs and endpoints, no formal meta-analysis was undertaken. Instead, we performed a qualitative synthesis organized around: (1) The anatomical and geometric rationale for adaptation in cervical cancer; (2) Technical approaches and platform-specific capabilities; and (3) Clinical evidence and implementation challenges. To enhance critical appraisal and avoid treating all studies as equally informative, we explicitly structured the clinical evidence according to an informal hierarchy, distinguishing randomized controlled trials and prospective cohorts from retrospective dosimetric studies, planning simulations, and expert opinion. Particular emphasis was placed on the extent to which dosimetric advantages were linked to reported toxicity and survival outcomes, and on identifying areas where evidence remains insufficient or conflicting.
As this is a narrative review, no formal risk-of-bias scoring system was applied. However, when interpreting individual studies, we considered sample size, study design, follow-up duration, and the appropriateness of comparators and endpoints. The synthesis also incorporated a conceptual “oART readiness framework” that integrated anatomical complexity, clinical indication stratification, platform suitability, institutional readiness, and patient selection. This framework was used to contextualize the reported evidence and to discuss pragmatic pathways for implementation rather than merely cataloging technical solutions.
Pelvic organ motion significantly contributes to geometric uncertainty during cervical cancer radiotherapy. Variations in bladder and rectal filling, as well as bowel peristalsis, can significantly impact the position of the cervix, uterus and surrounding pelvic anatomy. Tyagi et al[18] evaluated interfractional variation from 10 cervical cancer patients by delineating CTV on CBCT scans obtained before each treatment fraction. They found that increased bladder volume pushed the CTV centroid cranially, while rectal distension caused both posterior and superior shifts. Large-scale prospective trials, such as EMBRACE-II, further highlighted that bladder filling predominantly affects the craniocaudal extent of the target, while rectal filling influences the anterior-posterior dimensions[19,20]. These observations underscore the magnitude of pelvic organ motion and its direct impact on target reproducibility in daily treatment.
To account for these changes, wide margins are often required, with 5-10 mm posterior and 15-25 mm anterior margins recommended when the rectum is empty, and posterior margins up to 15-25 mm needed during rectal distension. However, such expansion inevitably increases radiation exposure to adjacent OARs. Additionally, the small bowel shows significant interfractional variation. Dominello et al[21] assessed small bowel motion in nine patients over 45 computed tomography (CT) scans. They found that caudal movement was maximal and ≥ 4 cm margins were required to encompass 90% of the volume, which complicates the maintenance of both target coverage and OAR sparing. These findings reveal the need for standardized bladder and bowel preparation protocols, including consistent fluid intake, bowel emptying, and dietary management, to reduce positional uncertainties. However, even under strict preparation protocols, substantial interfractional variability persists, particularly in the bladder, rectum and small bowel. This indicates that organ-motion driven uncertainties cannot be fully mitigated by population-based margins alone and require imaging-informed strategies that can adjust for the daily position of pelvic OARs. Offline ART addresses accumulated anatomical shifts but cannot correct daily fluctuations. In the context of cervical cancer, the magnitude and unpredictability of these organ motions define a high level of anatomic complexity within the oART readiness framework and provide a strong rationale for considering real-time online adaptation rather than relying solely on offline replanning in selected patients.
Beyond daily organ motion, anatomical changes and tumor regression over the course of treatment further challenge consistent dose delivery. The uterus, located between the bladder and rectum, is particularly mobile, with uterine fundal motion reported to reach up to 40 mm[10]. Progressive tumor regression during EBRT alters the geometry of the primary tumor and adjacent structures[22]. This effect is especially marked in squamous cell carcinoma, which demonstrates pronounced radiosensitivity[11,23]. In postoperative patients, positional changes in the vaginal cuff due to variable bladder and rectal filling can also be substantial and are not fully compensated by uniform margins. Jhingran et al[24] placed vaginal markers to correlate bladder and rectal volume with marker shifts, reporting mean shifts of 6.4 mm in anterior-posterior direction, 6.4 mm in superior-inferior direction, and 2.3 mm in left-right directions. Jürgenliemk-Schulz et al[25] reported that vaginal cuff deviation during treatment required anisotropic margins of up to 23 mm in the anterior-posterior direction to ensure adequate coverage. These observations highlight that, unlike organ-motion-driven positional shifts, treatment-course changes reflect progressive geometric changes of the target itself, which cannot be reliably captured by fixed PTV expansions. Therefore, adaptive replanning becomes essential to maintain accurate target coverage throughout both definitive and adjuvant radiotherapy, particularly as the anatomy variation over time. Within the oART readiness framework, such pronounced temporal evolution of target volumes further reinforces cervical cancer as a disease site with intrinsically high demand for online adaptation.
In addition to anatomical variability, treatment accuracy is also affected by setup uncertainties. Daily positioning may be influenced by factors including laser alignment error, subcutaneous fat thickness, skin mark reproducibility, immobilization technique and operator experience. Daily image guidance can substantially reduce systematic setup errors by correcting interfractional variation; however, residual uncertainties persist in absence of adaptive planning, and higher body mass index (BMI) correlates with larger systematic and random setup errors[26]. Bray et al[27] found that obese gynecological patients had significantly larger random setup errors in the right-left direction (obese vs nonobese: 7.6 mm vs 3.9 mm), highlighting the extra positional challenge brought by higher BMI. Mulla et al[28] demonstrated in a gynecological tumor cohort that body mass factors including BMI, umbilical circumference and hip circumference, correlated significantly with lateral and vertical setup errors, suggesting that customized PTV margins or daily CBCT may benefit patients with high BMI. These setup errors interact with organ motion and tumor regression, potentially compromising target coverage and increasing toxicity risk.
Given the limitations of margin-based approaches, ART offers a more robust strategy by incorporating daily imaging and dynamically adjusted treatment plans, thereby ensuring adequate tumor coverage while minimizing unnecessary dose to surrounding organs. To perform ART effectively, various technical approaches to ART have been developed, including advances in imaging, treatment planning, and automation to enable more individualized and precise dose delivery. From a readiness perspective, patients with unfavorable body habitus or large setup uncertainties represent a subgroup in whom the combination of internal and external variability makes online adaptive strategies particularly attractive.
ART defines a dynamic treatment framework that continuously adjusts radiotherapy plans in the entire radiotherapy process, including patient setup, treatment planning, delivery and plan verification[17,29]. Based on image-guided radiation therapy (IGRT), ART monitors anatomical and physiological changes during treatment, including tumor size, shape and position, and compares each fraction with the initial reference plan to inform subsequent plan adaptation. Therefore, ART aims to enhance treatment precision based on patient-specific anatomical and physiological changes, ensuring high-dose coverage of the tumor while minimizing radiation exposure to surrounding normal tissues, thereby improving tumor control and reducing treatment-related toxicity.
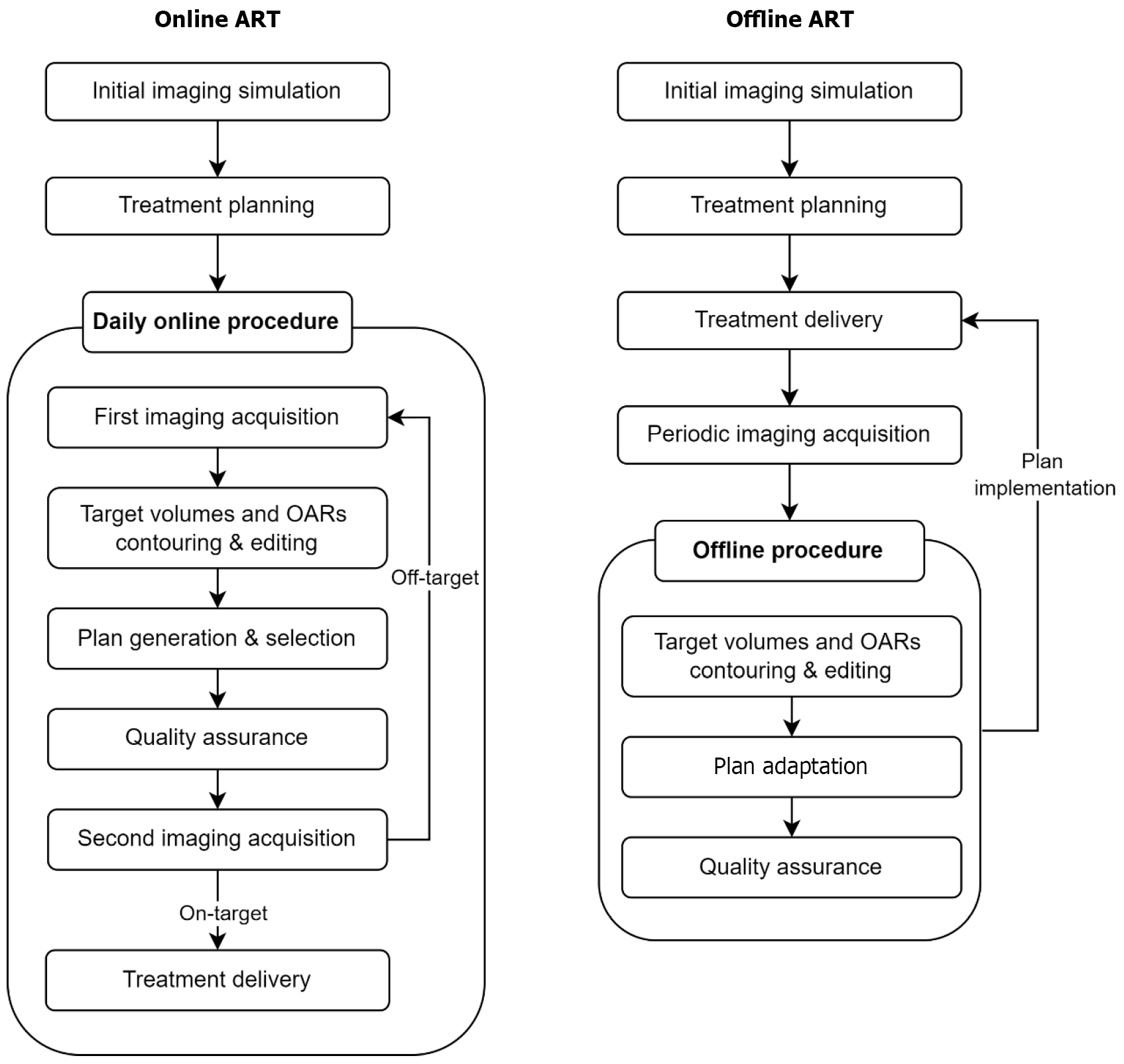
Adaptive strategies and workflows: ART can be performed offline or online, depending on whether treatment plan needs to be adapted between fractions or during the fraction itself (Figure 1). In offline ART, the current fraction is completed before replanning, and the edited plan is applied in subsequent fractions. Nuver et al[30] demonstrated that multiple CT scans during pelvic radiotherapy could establish an offline ART workflow that reduces PTV margins without compromising target coverage. However, offline ART cannot account for real-time anatomical changes that occur during each treatment session.
In contrast, oART offers high individualization and real-time adaptation. By acquiring imaging data during each fraction, oART adjusts the plan immediately to account for changes in tumor and normal tissue position and morphology, ensuring accurate dose delivery throughout treatment[31,32]. Previously, clinical implementation of oART was limited by imaging and computational constraints, but advances in radiation delivery technology and AI have recently made it feasible in practice.
AI-assisted workflows enhance ART by automating key tasks such as contouring, plan optimization and dose calculation, enabling rapid, daily adaptation of radiotherapy plans while maintaining precision, particularly in anatomically variable regions such as the pelvis. These systems can automatically delineate tumors and OARs, generate optimized plans within minutes, and incorporate daily imaging for dose verification and cumulative dose monitoring[33,34]. Offline ART remains useful for accumulated anatomical changes over several fractions, whereas oART ensures that intrafractional variations are corrected in real time. Integration of CBCT, FBCT or MRI guidance with AI-assisted workflows enables a spectrum of ART solutions tailored to institutional capabilities and patient-specific anatomical variations. This spectrum from offline ART to fully oART maps directly onto different levels of technical platform suitability and institutional readiness, and should be selected according to the anatomic complexity and resource profile defined in the oART readiness framework.
Imaging modalities: Currently, ART can be performed through a variety of imaging platforms and workflows, each offering distinct advantages in terms of plan adaptation, treatment precision and clinical feasibility. The choice of modality depends on institutional resources, patient anatomy and the required degree of individualization.
CBCT currently represents the most widely adopted platform for daily oART in cervical cancer due to its availability, speed and compatibility with AI-driven workflows. CBCT-guided ART combines CBCT with iterative reconstruction and AI-driven contours and has been widely used in many centers. Daily imaging allows rapid plan adaptation, with priority given to either target coverage or OAR sparing, while requiring minimal modifications to existing clinical workflow, making it suitable for routine treatment. Based on recent prospective studies, daily CBCT-based ART workflows for pelvic tumors have been shown to be feasible and efficient. Branco et al[35] demonstrated that daily adaptive sessions were completed within 30 minutes, with the majority of time dedicated to contour editing. Similarly, a comprehensive evaluation by Zheng et al[36] reported that > 1000 CBCT-guided ART sessions for pelvic malignancies were performed, highlighting the practicality and effectiveness of this approach in clinical practice. In many institutions, CBCT-based oART therefore represents the most pragmatic entry point for adaptive implementation.
FBCT plays a complementary role in ART, primarily supporting offline or mid-course adaptations rather than routine oART. FBCT-guided ART is typically used for mid-treatment rescans, offline replanning or to establish a library of plans for a “plan-of-the-day” strategy. Importantly, it can correct accumulated deviations without altering the daily treatment process. Sun et al[37] investigated the feasibility of FBCT-guided oART in pelvic tumors and found that FBCT could be effectively integrated into ART workflows and provided high-quality imaging for accurate treatment adaptation. Peng et al[38] also investigated the dosimetric advantages and reliability of FBCT-guided oART in cervical cancer, supporting its feasibility and effectiveness. Clinically, FBCT-based approaches are particularly attractive for centers that already have robust CT-based planning infrastructure and seek to implement adaptive strategies through mid-course replanning or plan libraries, rather than fully online daily adaptation.
MRI-guided platforms enable true oART through superior soft-tissue visualization, particularly in ATS workflows. MRI-guided ART provides superior soft tissue contrast and reduced interobserver variability, with clinical workflows typically categorized into ATP and ATS modes. ATP corrects translational errors and can be completed in approximately 30 minutes, whereas ATS involves recontouring targets and OARs and recalculating dose, requiring up to 1 hour[39-42]. Functional MRI enables individualized dose distribution and identification of tolerance regions, supporting more precise and individualized therapy. These features highlight MRI-guided ART as a typical and practical approach in contemporary clinical practice for patients with highly complex pelvic anatomy or when maximal soft-tissue contrast is critical, but they also imply higher resource requirements and more stringent institutional readiness.
From a clinical decision-making perspective, CBCT-based oART usually offers the best balance between adaptability and throughput for routine cervical cancer care, FBCT-based strategies are well suited for mid-course or plan-of-the-day adaptation, and magnetic resonance (MR)-guided oART is most appropriate when anatomical complexity and soft-tissue contrast requirements justify longer sessions and higher resource consumption (Table 1).
| Dimension | CBCT-guided | FBCT-guided | MRI-guided |
| Soft-tissue contrast | Moderate (limited for cervix/uterus boundaries) | Higher than CBCT | Highest (best for cervix/uterus/vagina) |
| Typical workflow | Fast daily adaptation feasible (routine) | Often mid-course/offline or library-based; can support oART in selected setups | ATP (approximately 30 minutes) to ATS (often approximately 60 minutes) depending on contours/re-optimization |
| Adaptation depth | Often ATP/Limited ATS | Commonly offline replanning/plan library; selected on-table replanning | True ATS with re-contouring and dose recalculation |
| Best-fit clinical scenarios | Broad adoption; high throughput; resource-constrained settings | Centers favoring high-quality CT replanning and plan libraries | High-resource centers; complex anatomy; research-heavy practice |
| Key limitations | Contour uncertainty; CBCT HU/dose calc issues | Less practice in real daily online in many workflows | Time/resource intensive; staffing/QA burden |
Treatment delivery considerations: Conventional EBRT for cervical cancer is challenged by dynamic anatomical changes, organ motion and setup uncertainties, which can reduce tumor coverage and increase toxicity risk. ART addresses these challenges by enabling real-time or fraction-to-fraction plan adaptation, optimizing PTV margins and dose distribution while sparing surrounding organs[43]. Beyond EBRT, ART also enhances brachytherapy planning by providing precise target delineation and dose mapping, facilitating accurate cumulative dose calculation and integration of EBRT and brachytherapy, ultimately improving the precision and safety of the entire radiotherapy course[44,45]. In practice, external-beam oART and MRI-guided adaptive brachytherapy should be viewed as complementary components of a single adaptive treatment pathway: EBRT adaptation improves coverage and sparing during the pelvic phase, while image-guided brachytherapy consolidates local control with highly conformal boost dosing.
Within the oART readiness framework, this integrated perspective underscores that platform selection and workflow design cannot be technology driven alone. They must be aligned with anatomic complexity, clinical indication and institutional readiness, as well as the availability of image-guided brachytherapy, to realize the full potential of ART in cervical cancer.
From dosimetric gain to clinical outcomes: An evolving evidence hierarchy: The clinical evidence base for oART in cervical cancer spans a broad spectrum from retrospective planning analyses to prospective cohorts and ongoing randomized trials. To avoid treating all data as equally informative, the available literature can be conceptually organized into four levels of evidence, reflecting an endpoint cascade from dosimetry to toxicity and survival.
Level 1: Randomized and phase 3 trial evidence (protocol stage): To date, no randomized trial has reported mature survival outcomes specifically comparing oART with nonadaptive EBRT in cervical cancer. However, several phase 2 and 3 studies, such as the SWIFT-1 trial with randomized controlled design, are currently ongoing and are explicitly designed to address this evidence gap. The SWIFT-1 trial (NCT06641635) is a multicenter, open-label phase 3 study comparing moderately hypofractionated oART (43.35 Gy in 17 fractions with online adaptation) vs conventional fractionation (45 Gy in 25 fractions) for locally advanced disease; both combined with concurrent chemotherapy and brachytherapy. The published protocol highlights progression-free survival and late toxicity as primary endpoints and explicitly positions oART not merely as a dosimetric innovation but as a strategy potentially capable of altering long-term outcomes[46]. Although results are pending, the existence of SWIFT-1 strengthens the argument that the field is moving beyond feasibility and dosimetric surrogates toward hard clinical endpoints. In the current hierarchy, however, such trials still provide prospective intent rather than outcome data and thus cannot yet close the evidence gap at the survival level.
Level 2: Prospective single-arm and registry-based clinical cohorts: Most of the strongest current evidence lies at this level. For CBCT-guided oART, Wang et al[47] conducted a prospective single-arm study in 27 patients receiving definitive chemoradiation with daily iterative oART and reduced PTV margins. Adapted plans showed 2%-3% improvement in CTV D95/D98, reductions in rectal V40Gy and bladder V45Gy, and low rates of acute gastrointestinal/genitourinary toxicity, suggesting that dosimetric gains can translate into clinically meaningful reductions in acute morbidity. A related prospective postoperative cohort similarly demonstrated improved vaginal PTV coverage and reductions in bladder and rectal mean doses using a uniform 5-mm PTV margin with daily oART.
Beyond single-institution experiences, Jiang et al[43] reported the first implementation of CT-linac-based oART in cervical cancer, showing improved target coverage and OAR sparing compared with IGRT, with early survival outcomes described as satisfactory over limited follow-up. Prospective feasibility work in locally advanced cervical cancer by Branco et al[35] confirmed that daily CBCT-based oART sessions can typically be completed within 30 minutes, with contour editing as the main time-limiting step, supporting workflow feasibility in busy clinics.
For MRI-guided oART, Ding et al[16] reported the first dedicated prospective experience, demonstrating that MRI-guided daily adaptation can correct day-to-day anatomical variation, maintain target coverage and reduce dose to adjacent organs while remaining clinically tolerable. For oART, the MOMENTUM trial reported the first cervical cancer patient treated with MRI-guided daily adaptation with average adaptation time of 68 minutes, demonstrating the feasibility of this approach in clinical practice[48]. Using data from the international MOMENTUM registry (NCT04075305), Alshamrani et al[49] performed a detailed dosimetric analysis of five node-negative cervical cancer patients treated with daily ATS on a 1.5 T MR-Linac, confirming that in most fractions adaptive plans improved either target coverage or OAR doses, while intrafraction dose deviations remained small. These studies collectively provide level 2 evidence that oART is feasible and safe, with consistent short-term toxicity benefits; however, they are limited by small sample sizes, short follow-up, and absence of randomized comparators.
Level 3: Retrospective dosimetric and planning studies: The majority of oART literature still resides at this level. Early offline ART demonstrated that periodic replanning based on weekly MRI or CT could mitigate accumulated anatomical changes and reduce OAR doses. Kerkhof et al[50] showed that weekly MRI-based replanning modified cervical CTV volumes over time and significantly reduced the volumes of bladder, rectum, small bowel and sigmoid receiving 20-45 Gy. Stewart et al[51] similarly reported that automated weekly replanning improved CTV D98% relative to nonadaptive treatment without increasing OAR exposure. While these offline strategies improved conformality, they inherently lag behind daily anatomical variation and do not leverage the full potential of oART.
More sophisticated CBCT-guided strategies include the “evolutive library” approach by Rigaud et al[52], in which pregenerated adaptive plans are selected based on daily imaging. This method achieved the highest CTV coverage (approximately 98.5% with a 10-mm margin and 96.2% with a 7-mm margin) among several nonadaptive strategies and reduced bowel dose, but still fell short of true online reoptimization. For fully online workflows, Yen et al[53] evaluated daily CBCT-based oART for cervical cancer and showed that a uniform 5-mm PTV margin was sufficient to achieve 98%-99% CTV coverage for most patients, with more complex anatomy requiring selective expansion to 10-15 mm. Guberina et al[54] compared online-onboard ART with reduced PTV margins to IGRT in gynecological tumors, reporting near-ideal generalized equivalent uniform dose preservation for the target with a 0-mm PTV margin in the ART arm, while the IGRT arm showed substantial underdosage at equivalent margins.
Importantly, more recent work has begun to question whether daily adaptation is always necessary. Yen et al[55] investigated adaptation frequency and found that weekly adaptation could recover > 80% of the dosimetric benefit of daily oART relative to IGRT, particularly in terms of bowel sparing, while substantially reducing resource utilization. This highlights an important nuance: While dosimetric superiority of daily oART is clear, the incremental gain over less frequent adaptation may be modest in some cases, raising questions about cost-effectiveness and patient selection.
MRI-based planning and dosimetric analyses reinforce the potential of advanced platforms. Beyond the prospective data mentioned earlier[16,49], planning studies have consistently shown that MRI-guided adaptation enables tighter margins and improved sparing of bladder and rectum compared with photon-based IGRT[48,50]. However, most of these studies rely on surrogate endpoints, including CTV D95/D98 and OAR dose-volume parameters, and cannot directly quantify long-term clinical benefit. Overall, level 3 evidence robustly supports the dosimetric advantages of oART and informs practical questions such as margin selection and adaptation frequency but remains largely surrogate-driven and nonrandomized.
Level 4: Expert consensus, implementation reports and case series: At the base of the hierarchy lie expert reviews, implementation case series and single-center experiences that primarily address feasibility, workflow and conceptual models. Narrative reviews and consensus statements have outlined strategies for adaptive management in cervical cancer and have emphasized the need for individualized PTV margins and integration of EBRT with adaptive brachytherapy[17,44,45]. Implementation reports such as the CBCT-based oART experiences by Sibolt et al[56] and de Jong et al[57] in pelvic and rectal cancer, together with the systematic evaluation by Zhang et al[58] in cervical cancer, which characterized auto-contouring performance and the regions requiring the most manual editing, collectively emphasize that workflow design, AI-assisted automation, and training pathways are at least as critical as hardware capabilities for successful oART implementation.
Case-based reports provide early signals for novel paradigms. Ma et al[59] described the first demand-triggered oART cases in cervical cancer using an FBCT-guided system, where adaptation was activated only when predefined anatomical or dosimetric criteria were violated. Both patients achieved improved target coverage and reduced bladder/rectal doses without grade ≥ 3 toxicity. Ugurluer et al[60] reported that MRI-guided radiotherapy for gynecological cancers, alone or as a boost when brachytherapy is not feasible, can achieve acceptable disease control and toxicity, but with substantial workflow demands. These level 4 sources do not by themselves establish efficacy but are valuable for shaping hypotheses, identifying practical barriers and informing the readiness dimension of oART; particularly around therapist-driven workflows and AI-assisted contouring.
Synthesis across levels: Current evidence provides a coherent but incomplete cascade. Dosimetric gains with oART are consistently demonstrated across platforms (CBCT, FBCT and MR-Linac) and disease settings (intact vs postoperative). Acute toxicity reduction is supported by prospective CBCT-guided oART cohorts, with lower rates of gastrointestinal/genitourinary events compared with historical IMRT data and no apparent increase in acute complications. Late toxicity and survival remain under-reported, with only limited early survival descriptions in single-arm studies and ongoing trials such as SWIFT-1 designed to address this gap. Emerging negative or at least nuanced findings, such as those of Yen et al[55] showing that weekly adaptation can recover most of the benefit of daily oART, emphasize that the field should move from a purely technology-driven pursuit of ever more frequent adaptation, toward a problem-driven, indication-specific approach. In this context, the oART readiness framework proposed in the introduction is useful to interpret these data: Anatomical complexity and clinical indication (level 1 considerations) must be balanced against platform capabilities and institutional resources, informed by the evolving evidence hierarchy rather than by dosimetric surrogates alone.
Toxicity and safety outcomes in the context of the evidence cascade: Within this hierarchy, acute toxicity data currently provide the strongest clinical signal supporting oART. In the prospective single-arm study of daily CBCT-guided oART with reduced PTV margins, Wang et al[47] reported that clinician-reported acute gastrointestinal toxicity was confined to grade 1-3 events (26%, 19% and 4%, respectively), predominantly diarrhea, and genitourinary toxicity was limited to grade 1 in 7% of patients with no grade ≥ 2 events. Patient-reported outcomes showed only transient symptom worsening during treatment, with most quality-of-life domains returning to baseline within 1 month. In the postoperative cohort, acute toxicity was mainly gastrointestinal, with 24% grade 2 events and no grade ≥ 2 genitourinary toxicity; again supporting the safety of margin reduction under daily adaptation. These findings are consistent with the broader pelvic IMRT experience, where larger PTV margins and nonadaptive strategies are associated with higher rates of bowel toxicity, as reflected in trials such as RTOG 1203[61]. Early MRI-guided cohorts, including those reported by Ding et al[16] and within the MOMENTUM registry, similarly suggest low acute toxicity despite long on-table times, although sample sizes remain small and follow-up short[49]. At the same time, the lack of robust late toxicity and survival data - together with observations that less frequent adaptation may recapture most of the dosimetric benefit in some patients - argues against over-generalizing from acute outcomes alone[55]. In other words, current evidence supports the statement that oART can safely reduce margins and improve short-term tolerability, but is still insufficient to claim superior long-term disease control or late toxicity across all cervical cancer indications. This distinction is important in guiding patient selection and in designing implementation models that are proportionate to institutional resources.
Within the oART readiness framework, clinical indication and patient-level characteristics are central to deciding when online adaptation is likely to provide meaningful additional benefit beyond high-quality IGRT or offline ART. Rather than assuming that all cervical cancer patients require daily oART, available data support a more nuanced approach based on anatomic complexity, expected geometric change during treatment, and baseline dosimetric risk.
From an indication perspective, patients with an intact cervix and bulky, locally advanced disease represent a key high-priority group. In this setting, large primary tumors often undergo substantial regression during chemoradiation, while the uterus, cervix and parametria are subject to pronounced day-to-day motion driven by bladder and rectal filling[9-11,18-21]. These combined effects create substantial uncertainty in both the extent and position of the CTV, and PTV margins must otherwise be expanded to 15-25 mm to maintain coverage, at the cost of increased gastrointestinal dose[12-14,21]. Prospective oART cohorts in intact disease have shown that daily adaptation allows margins to be safely reduced to approximately 5 mm in most patients, while maintaining or improving CTV coverage and decreasing bowel and bladder exposure[47,53,58]. In the readiness framework, such scenarios are characterized by high anatomical complexity and a strong rationale for online adaptation.
In the postoperative setting, the geometric problem is different but related. The vaginal cuff and upper vagina can still exhibit substantial motion with variable bladder and rectal filling, and studies have reported anisotropic deviations requiring margins of up to 23 mm in the anterior-posterior direction to ensure coverage[24,25]. At the same time, the target volume is generally smaller and more localized than in intact disease, and nodal basins, particularly pelvic nodes, tend to be geometrically more stable. Prospective postoperative oART data indicate that a uniform 5-mm margin, combined with daily adaptation, can improve vaginal PTV coverage and modestly reduce OAR dose. Thus, in adjuvant radiotherapy, patients with highly mobile vaginal cuff anatomy or close surgical margins may benefit most from oART, whereas cases with more stable geometry and generous margins may be adequately managed with IGRT or offline adaptation.
Lymph node involvement introduces further heterogeneity. Para-aortic and common iliac nodal regions are less affected by organ filling than the primary cervix and vagina, and may not require the same degree of online adaptation. However, in patients with bulky nodal disease abutting radiosensitive structures, or in whom tight constraints are imposed on bowel or kidney dose, even modest geometric shifts can translate into clinically meaningful dosimetric trade-offs. In such high-risk nodal scenarios, oART may provide value by enabling margin reduction around nodal targets while respecting OAR constraints, but the overall priority for daily adaptation is still driven primarily by the behavior of the primary and vaginal targets.
Beyond indication, patient-level selection criteria are equally important for prioritizing oART. Ghimire et al[62] showed that patients with higher baseline bowel dose-volume metrics are more likely to experience larger dosimetric gains from daily online adaptation, suggesting that initial plan quality and proximity of bowel to target should inform selection. Dominello et al[21] highlighted the limitations of standard “bowel bag” contours and demonstrated large small bowel excursions requiring several centimeters of margin to encompass typical motion. These observations imply that patients whose baseline plans already push bowel close to tolerance, because of pelvic anatomy, prior surgery or tumor extent, are logical candidates for oART, as margin reduction and day-to-day reoptimization can most effectively reduce high-dose bowel exposure.
Body habitus and setup reproducibility also modulate potential benefit. Higher BMI, larger umbilical or hip circumference, and greater subcutaneous tissue thickness have all been associated with increased systematic and random setup errors in gynecological radiotherapy, even with daily IGRT[26-28]. These setup uncertainties compound internal organ motion and can lead to underdosage of the target and overdosage of OARs when uniform margins are applied. Within the readiness framework, patients with high BMI or poor external reproducibility, especially when combined with large internal motion, should be viewed as higher-priority candidates for online adaptation or, at minimum, more intensive image-guided strategies.
Finally, adaptation frequency itself may need to be individualized. Yen et al[55] showed that weekly adaptation could recover > 80% of the dosimetric benefit of daily oART compared with IGRT in some settings; particularly for bowel sparing. This suggests that patients with stable anatomy and favorable baseline dosimetry can be adequately managed with less frequent or trigger-based adaptation, reserving daily oART for those with highly variable anatomy, high baseline bowel dose or tight OAR constraints. Thus, clinical indication stratification and patient-level selection, which are two key dimensions of the oART readiness framework, should be considered jointly with the evolving evidence hierarchy, rather than assuming that daily oART is universally required for all cervical cancer patients.
Despite the promising dosimetric and early clinical data, real-world implementation of oART remains constrained by workflow complexity, automation maturity, staffing and training; factors that together define the institutional readiness dimension of the oART readiness framework.
Workflow complexity, automation and training: Even in centers with advanced hardware, the practical delivery of daily oART is strongly influenced by contouring workload and on-table time. Prospective CBCT-based oART studies have reported total session times of 20-30 minutes, with most of that time consumed by contour review and editing rather than dose calculation or beam delivery[35,36]. Failure mode and effects analyses of pelvic oART workflows, such as the study by Zheng et al[36], underscore that most critical vulnerabilities lie in contour quality, image registration and human-machine interaction, rather than in the optimization engine itself.
AI-based auto-contouring tools have been introduced to mitigate this burden[33,34]. While these tools can substantially reduce the time required for initial contour generation, they do not eliminate the need for expert oversight. Zhang et al[58] systematically evaluated autocontouring performance in cervical cancer oART and found generally good overlap with manually corrected contours, but also identified specific structures, including the uterus, upper vaginal cuff, and lower nodal CTV, that required frequent and sometimes extensive edits. This pattern indicates that automation currently functions more as an accelerator than a replacement for human expertise, and that contouring bottlenecks will persist unless training and workflow design explicitly address these high-variability regions.
Staffing and role delineation represent additional challenges. Many early oART implementations have required the simultaneous presence of a radiation oncologist, medical physicist, and experienced radiation therapist at each fraction, placing substantial demands on human resources[38,47]. Emerging models, such as therapist-driven MRI-guided ART reported by Moreira et al[63] in oligometastatic disease, suggest that with structured training and robust protocols, senior therapists can safely assume key responsibilities in contour review and plan approval. For cervical cancer, adapting such role expansion, combined with standardized editing hierarchies, checklists, and quality assurance procedures, may be critical to achieving sustainable oART workflows, particularly in high-volume or resource-constrained settings.
Two-tier implementation model for scalable oART adoption: Given these constraints, it is unlikely that a single oART model will be universally appropriate across all institutions and health systems. Instead, a pragmatic, tiered approach that aligns ambition with available resources is needed, especially to support global adoption where infrastructure and staffing vary widely.
At the upper end of the readiness spectrum, tier 1 programs in high-resource centers may implement daily oART using MRI-guided or advanced CBCT-based systems, supported by AI-assisted autocontouring, deformable registration, rapid plan reoptimization, and comprehensive on-table quality assurance. In this model, patients with the highest anatomical complexity, such as those with bulky intact tumors, highly mobile uteri or vaginal cuffs, or tight bowel or bladder constraints, can receive full ATS workflows on most or all fractions. Early experiences from Ding et al[16] and from MR-Linac cohorts within the MOMENTUM registry indicate that such approaches are feasible and well tolerated, albeit with longer session times and significant workflow demands[53,57].
In contrast, tier 2 programs in resource-constrained settings may adopt a more selective and stepwise strategy. Here, a combination of high-quality IGRT, standardized bladder and bowel preparation, and offline or mid-course adaptation, including plan-of-the-day libraries based on FBCT or planning CTs, can form the backbone of treatment[37,38,50]. CBCT verification can be used to select among pregenerated plans, and online adaptation can be triggered only when predefined anatomical or dosimetric criteria are violated. Case-based experiences, such as the demand-triggered oART report by Ma and Zhou[59] using an FBCT-guided system, demonstrate that such on-demand adaptation can correct major deviations and improve target coverage and OAR sparing without requiring daily full adaptation for all patients. Ugurluer et al[60] similarly showed that MR-guided radiotherapy can be used either as a primary modality or as a boost when brachytherapy is not feasible, but at the cost of substantial workflow complexity, reinforcing the need for judicious patient selection in settings with limited capacity.
Evidence on adaptation frequency further supports this tiered view. As noted above, Yen et al[55] found that weekly adaptation can recapture most of the dosimetric benefit of daily oART for some patients; particularly in terms of bowel sparing. This suggests that tier 2 programs can rationally restrict daily online adaptation to the subset of patients with the most unstable anatomy or highest baseline bowel dose, while using weekly or trigger-based adaptation for others, thereby maximizing benefit per unit of resource.
This tiered implementation model illustrates how the oART readiness framework can be translated into context-sensitive strategies that respect both clinical need and institutional capacity. By explicitly linking anatomical complexity, clinical indication, platform capabilities and staffing realities, it provides a pathway for oART to move from feasibility demonstrations in highly specialized centers toward scalable, globally relevant practice; particularly in regions where cervical cancer burden is high but radiotherapy resources remain limited.
This narrative review had some limitations. First, the available evidence for oART in cervical cancer remains dominated by dosimetric and feasibility studies, with limited prospective data on late toxicity and long-term survival, restricting causal inference on ultimate clinical benefit. Second, heterogeneity in target definitions, imaging platforms, adaptation workflows (ATP vs ATS), and reporting endpoints limits cross-study comparability. Third, implementation feasibility is highly context dependent; workflow time, staffing models and automation maturity vary widely across institutions and may influence generalizability. Finally, publication bias toward positive feasibility experiences may overestimate readiness for routine adoption. These limitations highlight the need for standardized reporting, multicenter registries, and randomized trials to validate clinical endpoints.
oART represents a promising advance in the precision treatment of cervical cancer. By integrating daily imaging, automated contouring assistance, and on-table plan optimization, oART enables individualized dose delivery that more closely reflects the patient’s true anatomy at each fraction. This capability directly addresses long-standing challenges related to pelvic organ motion, tumor regression and setup uncertainty, and consistently improves target coverage and OAR sparing in dosimetric and feasibility studies in ways that are difficult to achieve with conventional margin-based approaches.
Emerging prospective data indicate that oART is feasible, safe and associated with favorable acute toxicity profiles, particularly when coupled with PTV reduction and rigorous image guidance. However, mature evidence on late toxicity and survival remains limited, and not all patients or clinical scenarios are likely to require daily online adaptation. Within an oART readiness framework, the greatest added value is expected in indications characterized by high anatomical complexity and tight normal tissue constraints, and in institutions with sufficient technical and staffing readiness. Pragmatic, tiered implementation models that combine daily oART for selected high-risk patients with less intensive adaptive or image-guided strategies for others may therefore offer the most realistic pathway to broader adoption. As future multicenter cohorts and randomized trials clarify long-term clinical benefit, oART has the potential to become a central component of precision radiotherapy for cervical cancer, improving both therapeutic outcomes and patient experience in a resource-aware, indication-specific manner.
The authors would like to thank the radiation therapists, physicists, and clinical staff involved in the implementation and evaluation of ART workflows.
| 1. | Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16785] [Cited by in RCA: 15060] [Article Influence: 7530.0] [Reference Citation Analysis (23)] |
| 2. | Tewari KS. Cervical Cancer. N Engl J Med. 2025;392:56-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 59] [Article Influence: 59.0] [Reference Citation Analysis (0)] |
| 3. | Estape RE, Angioli R, Madrigal M, Janicek M, Gomez C, Penalver M, Averette H. Close vaginal margins as a prognostic factor after radical hysterectomy. Gynecol Oncol. 1998;68:229-232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 64] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 4. | Diaz ES, Aoyama C, Baquing MA, Beavis A, Silva E, Holschneider C, Cass I. Predictors of residual carcinoma or carcinoma-in-situ at hysterectomy following cervical conization with positive margins. Gynecol Oncol. 2014;132:76-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 33] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 5. | Trifiletti DM, Swisher-McClure S, Showalter TN, Hegarty SE, Grover S. Postoperative Chemoradiation Therapy in High-Risk Cervical Cancer: Re-evaluating the Findings of Gynecologic Oncology Group Study 109 in a Large, Population-Based Cohort. Int J Radiat Oncol Biol Phys. 2015;93:1032-1044. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 6. | Rose PG, Bundy BN, Watkins EB, Thigpen JT, Deppe G, Maiman MA, Clarke-Pearson DL, Insalaco S. Concurrent cisplatin-based radiotherapy and chemotherapy for locally advanced cervical cancer. N Engl J Med. 1999;340:1144-1153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2172] [Cited by in RCA: 1867] [Article Influence: 69.1] [Reference Citation Analysis (0)] |
| 7. | Loiselle C, Koh WJ. The emerging use of IMRT for treatment of cervical cancer. J Natl Compr Canc Netw. 2010;8:1425-1434. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 25] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 8. | Peng Y, Liu Y, Chen Z, Zhang G, Ma C, Xu S, Yin Y. Accuracy Improvement Method Based on Characteristic Database Classification for IMRT Dose Prediction in Cervical Cancer: Scientifically Training Data Selection. Front Oncol. 2022;12:808580. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 9. | Chan P, Dinniwell R, Haider MA, Cho YB, Jaffray D, Lockwood G, Levin W, Manchul L, Fyles A, Milosevic M. Inter- and intrafractional tumor and organ movement in patients with cervical cancer undergoing radiotherapy: a cinematic-MRI point-of-interest study. Int J Radiat Oncol Biol Phys. 2008;70:1507-1515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 164] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 10. | Jadon R, Pembroke CA, Hanna CL, Palaniappan N, Evans M, Cleves AE, Staffurth J. A systematic review of organ motion and image-guided strategies in external beam radiotherapy for cervical cancer. Clin Oncol (R Coll Radiol). 2014;26:185-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 154] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 11. | Beadle BM, Jhingran A, Salehpour M, Sam M, Iyer RB, Eifel PJ. Cervix regression and motion during the course of external beam chemoradiation for cervical cancer. Int J Radiat Oncol Biol Phys. 2009;73:235-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 108] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 12. | Lim K, Small W Jr, Portelance L, Creutzberg C, Jürgenliemk-Schulz IM, Mundt A, Mell LK, Mayr N, Viswanathan A, Jhingran A, Erickson B, De los Santos J, Gaffney D, Yashar C, Beriwal S, Wolfson A, Taylor A, Bosch W, El Naqa I, Fyles A; Gyn IMRT Consortium. Consensus guidelines for delineation of clinical target volume for intensity-modulated pelvic radiotherapy for the definitive treatment of cervix cancer. Int J Radiat Oncol Biol Phys. 2011;79:348-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 392] [Cited by in RCA: 340] [Article Influence: 22.7] [Reference Citation Analysis (0)] |
| 13. | Eminowicz G, Rompokos V, Stacey C, Hall L, McCormack M. Understanding the impact of pelvic organ motion on dose delivered to target volumes during IMRT for cervical cancer. Radiother Oncol. 2017;122:116-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 28] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 14. | Sun R, Mazeron R, Chargari C, Barillot I. CTV to PTV in cervical cancer: From static margins to adaptive radiotherapy. Cancer Radiother. 2016;20:622-628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 15. | Liu H, Schaal D, Curry H, Clark R, Magliari A, Kupelian P, Khuntia D, Beriwal S. Review of cone beam computed tomography based online adaptive radiotherapy: current trend and future direction. Radiat Oncol. 2023;18:144. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 80] [Article Influence: 26.7] [Reference Citation Analysis (0)] |
| 16. | Ding S, Piao Z, Chen M, Li F, Li Y, Liu B, Liu H, Huang X, Li J. MRI guided online adaptive radiotherapy and the dosimetric impact of inter- and intrafractional motion in patients with cervical cancer. Clin Transl Radiat Oncol. 2025;50:100881. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 17. | Shelley CE, Barraclough LH, Nelder CL, Otter SJ, Stewart AJ. Adaptive Radiotherapy in the Management of Cervical Cancer: Review of Strategies and Clinical Implementation. Clin Oncol (R Coll Radiol). 2021;33:579-590. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 43] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 18. | Tyagi N, Lewis JH, Yashar CM, Vo D, Jiang SB, Mundt AJ, Mell LK. Daily online cone beam computed tomography to assess interfractional motion in patients with intact cervical cancer. Int J Radiat Oncol Biol Phys. 2011;80:273-280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 66] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 19. | Pötter R, Tanderup K, Kirisits C, de Leeuw A, Kirchheiner K, Nout R, Tan LT, Haie-Meder C, Mahantshetty U, Segedin B, Hoskin P, Bruheim K, Rai B, Huang F, Van Limbergen E, Schmid M, Nesvacil N, Sturdza A, Fokdal L, Jensen NBK, Georg D, Assenholt M, Seppenwoolde Y, Nomden C, Fortin I, Chopra S, van der Heide U, Rumpold T, Lindegaard JC, Jürgenliemk-Schulz I; EMBRACE Collaborative Group. The EMBRACE II study: The outcome and prospect of two decades of evolution within the GEC-ESTRO GYN working group and the EMBRACE studies. Clin Transl Radiat Oncol. 2018;9:48-60. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 257] [Cited by in RCA: 483] [Article Influence: 60.4] [Reference Citation Analysis (0)] |
| 20. | Tan Mbbs Mrcp Frcr Md LT, Tanderup PhD K, Kirisits PhD C, de Leeuw PhD A, Nout Md PhD R, Duke Mbbs Frcr S, Seppenwoolde PhD Y, Nesvacil PhD N, Georg PhD D, Kirchheiner PhD K, Fokdal Md PhD L, Sturdza Md Frcpc A, Schmid Md M, Swamidas PhD J, van Limbergen Md PhD E, Haie-Meder Md C, Mahantshetty Md U, Jürgenliemk-Schulz Md PhD I, Lindegaard Dm DMSc JC, Pötter Md R. Image-guided Adaptive Radiotherapy in Cervical Cancer. Semin Radiat Oncol. 2019;29:284-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 51] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 21. | Dominello MM, Nalichowski A, Paximadis P, Kaufman I, McSpadden E, Joiner M, Miller S, Konski A. Limitations of the bowel bag contouring technique in the definitive treatment of cervical cancer. Pract Radiat Oncol. 2014;4:e15-e20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 22. | Chen W, Bai P, Pan J, Xu Y, Chen K. Changes in Tumor Volumes and Spatial Locations Relative to Normal Tissues During Cervical Cancer Radiotherapy Assessed by Cone Beam Computed Tomography. Technol Cancer Res Treat. 2017;16:246-252. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 23. | van de Bunt L, van der Heide UA, Ketelaars M, de Kort GA, Jürgenliemk-Schulz IM. Conventional, conformal, and intensity-modulated radiation therapy treatment planning of external beam radiotherapy for cervical cancer: The impact of tumor regression. Int J Radiat Oncol Biol Phys. 2006;64:189-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 146] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 24. | Jhingran A, Salehpour M, Sam M, Levy L, Eifel PJ. Vaginal motion and bladder and rectal volumes during pelvic intensity-modulated radiation therapy after hysterectomy. Int J Radiat Oncol Biol Phys. 2012;82:256-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 59] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 25. | Jürgenliemk-Schulz IM, Toet-Bosma MZ, de Kort GA, Schreuder HW, Roesink JM, Tersteeg RJ, van der Heide UA. Internal motion of the vagina after hysterectomy for gynaecological cancer. Radiother Oncol. 2011;98:244-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 24] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 26. | Lin LL, Hertan L, Rengan R, Teo BK. Effect of body mass index on magnitude of setup errors in patients treated with adjuvant radiotherapy for endometrial cancer with daily image guidance. Int J Radiat Oncol Biol Phys. 2012;83:670-675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 41] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 27. | Bray TS, Kaczynski A, Albuquerque K, Cozzi F, Roeske JC. Role of image guided radiation therapy in obese patients with gynecologic malignancies. Pract Radiat Oncol. 2013;3:249-255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 28. | Mulla Z, Hashem R, AlMohamad A, Weber A, Habibullah H, Abdulmoula G, Mohiuddin MG, Ujaimi R. Effect of Body Mass Factors on Setup Displacement in Gynecologic Tumors and Subsequent Effect on PTV Margins. Adv Radiat Oncol. 2023;8:101108. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 29. | Yan D, Georg D. Adaptive radiation therapy. Z Med Phys. 2018;28:173-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 11] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 30. | Nuver TT, Hoogeman MS, Remeijer P, van Herk M, Lebesque JV. An adaptive off-line procedure for radiotherapy of prostate cancer. Int J Radiat Oncol Biol Phys. 2007;67:1559-1567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 61] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 31. | Green OL, Henke LE, Hugo GD. Practical Clinical Workflows for Online and Offline Adaptive Radiation Therapy. Semin Radiat Oncol. 2019;29:219-227. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 164] [Cited by in RCA: 147] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 32. | Henke LE, Stanley JA, Robinson C, Srivastava A, Contreras JA, Curcuru A, Green OL, Massad LS, Kuroki L, Fuh K, Hagemann A, Mutch D, McCourt C, Thaker P, Powell M, Markovina S, Grigsby PW, Schwarz JK, Chundury A. Phase I Trial of Stereotactic MRI-Guided Online Adaptive Radiation Therapy (SMART) for the Treatment of Oligometastatic Ovarian Cancer. Int J Radiat Oncol Biol Phys. 2022;112:379-389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 36] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 33. | Choi SH, Park JW, Cho Y, Yang G, Yoon HI. Automated Organ Segmentation for Radiation Therapy: A Comparative Analysis of AI-Based Tools Versus Manual Contouring in Korean Cancer Patients. Cancers (Basel). 2024;16:3670. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 34. | Hoque SMH, Pirrone G, Matrone F, Donofrio A, Fanetti G, Caroli A, Rista RS, Bortolus R, Avanzo M, Drigo A, Chiovati P. Clinical Use of a Commercial Artificial Intelligence-Based Software for Autocontouring in Radiation Therapy: Geometric Performance and Dosimetric Impact. Cancers (Basel). 2023;15:5735. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 22] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 35. | Branco D, Mayadev J, Moore K, Ray X. Dosimetric and feasibility evaluation of a CBCT-based daily adaptive radiotherapy protocol for locally advanced cervical cancer. J Appl Clin Med Phys. 2023;24:e13783. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 39] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 36. | Zheng D, Cummings M, Zhang H, Podgorsak A, Li F, Dona Lemus O, Webster M, Joyce N, Hagenbach E, Bylund K, Qiu H, Pacella M, Chen Y, Tanny S. Clinical Practice-Based Failure Modes and Root Cause Analysis of Cone Beam CT-Guided Online Adaptive Radiotherapy of the Pelvis. Cancers (Basel). 2025;17:1462. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 37. | Sun S, Gong X, Liang Y, Sun Y, Que D, Xie Y, He S, He L, Liang H, Wang Y, Wu X, Wang C, Yang B, Qiu J, Hu K, Zhang F. Evaluating the Implementation of fan-beam CT-guided online adaptive re-planning in definitive cervical cancer radiotherapy. Front Oncol. 2025;15:1509619. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 38. | Peng H, Zhang J, Xu N, Zhou Y, Tan H, Ren T. Fan beam CT-guided online adaptive external radiotherapy of uterine cervical cancer: a dosimetric evaluation. BMC Cancer. 2023;23:588. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 39. | Mutic S, Dempsey JF. The ViewRay system: magnetic resonance-guided and controlled radiotherapy. Semin Radiat Oncol. 2014;24:196-199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 822] [Cited by in RCA: 717] [Article Influence: 59.8] [Reference Citation Analysis (0)] |
| 40. | Crijns S, Raaymakers B. From static to dynamic 1.5T MRI-linac prototype: impact of gantry position related magnetic field variation on image fidelity. Phys Med Biol. 2014;59:3241-3247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 41. | Bertelsen AS, Schytte T, Møller PK, Mahmood F, Riis HL, Gottlieb KL, Agergaard SN, Dysager L, Hansen O, Gornitzka J, Veldhuizen E, ODwyer DB, Christiansen RL, Nielsen M, Jensen HR, Brink C, Bernchou U. First clinical experiences with a high field 1.5 T MR linac. Acta Oncol. 2019;58:1352-1357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 84] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 42. | Alongi F, Rigo M, Figlia V, Cuccia F, Giaj-Levra N, Nicosia L, Ricchetti F, Sicignano G, De Simone A, Naccarato S, Ruggieri R, Mazzola R. 1.5 T MR-guided and daily adapted SBRT for prostate cancer: feasibility, preliminary clinical tolerability, quality of life and patient-reported outcomes during treatment. Radiat Oncol. 2020;15:69. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 119] [Cited by in RCA: 106] [Article Influence: 17.7] [Reference Citation Analysis (0)] |
| 43. | Jiang D, Yang C, Sun S, Wang D, Xiao Z, Hu J, Mei Z, Xie C, Liu H, Qiu H, Wang X. First implementation and results of online adaptive radiotherapy for cervical cancer based on CT-Linac combination. Front Oncol. 2024;14:1399468. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 44. | Pötter R, Tanderup K, Schmid MP, Jürgenliemk-Schulz I, Haie-Meder C, Fokdal LU, Sturdza AE, Hoskin P, Mahantshetty U, Segedin B, Bruheim K, Huang F, Rai B, Cooper R, van der Steen-Banasik E, Van Limbergen E, Pieters BR, Tan LT, Nout RA, De Leeuw AAC, Ristl R, Petric P, Nesvacil N, Kirchheiner K, Kirisits C, Lindegaard JC; EMBRACE Collaborative Group. MRI-guided adaptive brachytherapy in locally advanced cervical cancer (EMBRACE-I): a multicentre prospective cohort study. Lancet Oncol. 2021;22:538-547. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 549] [Cited by in RCA: 490] [Article Influence: 98.0] [Reference Citation Analysis (0)] |
| 45. | Wang K, Wang J, Jiang P. High-Dose-Rate Three-Dimensional Image-Guided Adaptive Brachytherapy (3D IGABT) for Locally Advanced Cervical Cancer (LACC): A Narrative Review on Imaging Modality and Clinical Evidence. Curr Oncol. 2023;31:50-65. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 46. | Zeng Z, Chen Y, Qiu J, Yang B, Wang Z, Meng X, Sun Y, Yan J, Hu K, Zhang F. Moderately hypofractionated online adaptive radiotherapy (SWIFT-1) in cervical cancer patients: study protocol for a multi-centered, open-label, two-arm, phase III, randomized controlled study. Radiat Oncol. 2025;20:112. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 47. | Wang G, Chen Y, Wang Z, Zeng Z, Sun Y, Zhou B, Yang B, Qiu J, Yan J, Hu K, Zhang F. A Prospective Single-Arm Study of Daily Online Adaptive Radiation Therapy for Cervical Cancer with Reduced Planning Target Volume Margin: Acute Toxicity and Dosimetric Outcomes. Int J Radiat Oncol Biol Phys. 2025;122:1002-1011. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 48. | de Mol van Otterloo SR, Christodouleas JP, Blezer ELA, Akhiat H, Brown K, Choudhury A, Eggert D, Erickson BA, Faivre-Finn C, Fuller CD, Goldwein J, Hafeez S, Hall E, Harrington KJ, van der Heide UA, Huddart RA, Intven MPW, Kirby AM, Lalondrelle S, McCann C, Minsky BD, Mook S, Nowee ME, Oelfke U, Orrling K, Sahgal A, Sarmiento JG, Schultz CJ, Tersteeg RJHA, Tijssen RHN, Tree AC, van Triest B, Hall WA, Verkooijen HM. The MOMENTUM Study: An International Registry for the Evidence-Based Introduction of MR-Guided Adaptive Therapy. Front Oncol. 2020;10:1328. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 129] [Cited by in RCA: 117] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 49. | Alshamrani A, Chuter R, Aznar M, Hoskin P, Nelder C, Choudhury A, Barraclough L, Eccles CL. Quantifying the dosimetric impact of online daily adaptation for MR-guided RT in cervical cancer. Acta Oncol. 2025;64:693-702. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 50. | Kerkhof EM, Raaymakers BW, van der Heide UA, van de Bunt L, Jürgenliemk-Schulz IM, Lagendijk JJ. Online MRI guidance for healthy tissue sparing in patients with cervical cancer: an IMRT planning study. Radiother Oncol. 2008;88:241-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 60] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 51. | Stewart J, Lim K, Kelly V, Xie J, Brock KK, Moseley J, Cho YB, Fyles A, Lundin A, Rehbinder H, Löf J, Jaffray D, Milosevic M. Automated weekly replanning for intensity-modulated radiotherapy of cervix cancer. Int J Radiat Oncol Biol Phys. 2010;78:350-358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 59] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 52. | Rigaud B, Simon A, Gobeli M, Lafond C, Leseur J, Barateau A, Jaksic N, Castelli J, Williaume D, Haigron P, De Crevoisier R. CBCT-guided evolutive library for cervical adaptive IMRT. Med Phys. 2018;45:1379-1390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 29] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 53. | Yen A, Choi B, Inam E, Yeh A, Lin MH, Park C, Hrycushko B, Nwachukwu C, Albuquerque K. Spare the Bowel, Don't Spoil the Target: Optimal Margin Assessment for Online Cone Beam Adaptive Radiation Therapy (OnC-ART) of the Cervix. Pract Radiat Oncol. 2023;13:e176-e183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 54. | Guberina M, Santiago Garcia A, Khouya A, Pöttgen C, Holubyev K, Ringbaek TP, Lachmuth M, Alberti Y, Hoffmann C, Hlouschek J, Gauler T, Lübcke W, Indenkämpen F, Stuschke M, Guberina N. Comparison of Online-Onboard Adaptive Intensity-Modulated Radiation Therapy or Volumetric-Modulated Arc Radiotherapy With Image-Guided Radiotherapy for Patients With Gynecologic Tumors in Dependence on Fractionation and the Planning Target Volume Margin. JAMA Netw Open. 2023;6:e234066. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 55. | Yen A, Zhong X, Lin MH, Nwachukwu C, Albuquerque K, Hrycushko B. Optimizing Online Adaptation Timing in the Treatment of Locally Advanced Cervical Cancer. Pract Radiat Oncol. 2024;14:e159-e164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 56. | Sibolt P, Andersson LM, Calmels L, Sjöström D, Bjelkengren U, Geertsen P, Behrens CF. Clinical implementation of artificial intelligence-driven cone-beam computed tomography-guided online adaptive radiotherapy in the pelvic region. Phys Imaging Radiat Oncol. 2021;17:1-7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 192] [Cited by in RCA: 166] [Article Influence: 33.2] [Reference Citation Analysis (0)] |
| 57. | de Jong R, Visser J, van Wieringen N, Wiersma J, Geijsen D, Bel A. Feasibility of Conebeam CT-based online adaptive radiotherapy for neoadjuvant treatment of rectal cancer. Radiat Oncol. 2021;16:136. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 66] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 58. | Zhang Y, Wang G, Chang Y, Wang Z, Sun X, Sun Y, Zeng Z, Chen Y, Hu K, Qiu J, Yan J, Zhang F. Prospects for daily online adaptive radiotherapy for cervical cancer: Auto-contouring evaluation and dosimetric outcomes. Radiat Oncol. 2024;19:6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 59. | Ma C, Zhou J. First Application of Demand-Triggered Online Adaptive Radiotherapy in the Treatment of Cervical Cancer: A Clinical Report. Cureus. 2024;16:e69703. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 60. | Ugurluer G, Zoto Mustafayev T, Gungor G, Abacioglu U, Atalar B, Ozyar E. Online Adaptive Magnetic Resonance-guided Radiation Therapy for Gynaecological Cancers: Preliminary Results of Feasibility and Outcome. Clin Oncol (R Coll Radiol). 2024;36:12-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 61. | Klopp AH, Yeung AR, Deshmukh S, Gil KM, Wenzel L, Westin SN, Gifford K, Gaffney DK, Small W Jr, Thompson S, Doncals DE, Cantuaria GHC, Yaremko BP, Chang A, Kundapur V, Mohan DS, Haas ML, Kim YB, Ferguson CL, Pugh SL, Kachnic LA, Bruner DW. Patient-Reported Toxicity During Pelvic Intensity-Modulated Radiation Therapy: NRG Oncology-RTOG 1203. J Clin Oncol. 2018;36:2538-2544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 314] [Cited by in RCA: 271] [Article Influence: 33.9] [Reference Citation Analysis (0)] |
| 62. | Ghimire R, Moore KL, Branco D, Rash DL, Mayadev J, Ray X. Forecasting patient-specific dosimetric benefit from daily online adaptive radiotherapy for cervical cancer. Biomed Phys Eng Express. 2023;9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 63. | Moreira A, Li W, Mansour IR, Faye M, Hosni A, Mesci A, Gutierrez-Valencia E, Lindsay P, Chung P, Winter J. Feasibility of Therapist-Driven MR-Guided Adaptive Radiotherapy for Oligometastatic Disease: Geometric Accuracy and Dosimetric Impact. J Med Imaging Radiat Oncol. 2025;69:777-786. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |