Published online Mar 24, 2026. doi: 10.5306/wjco.v17.i3.116823
Revised: December 17, 2025
Accepted: January 26, 2026
Published online: March 24, 2026
Processing time: 122 Days and 18.9 Hours
Exosomes are nano-sized extracellular vesicles that play a key role in intercellular communication. Due to their low immunogenicity, good biocompatibility, and tumor-targeting ability, they have shown great potential in cancer therapy. Their applications mainly include three aspects: Exerting direct antitumor effects through their endogenous cargo, serving as engineered drug delivery systems, and functioning as immunomodulators or cancer vaccines. In particular, exo
Core Tip: This minireview elucidates the triple roles of exosomes in cancer therapy: As direct effectors, drug delivery vehicles, and immunomodulators. Focusing on mesen
- Citation: Xiao WZ, Shi JH. Exosomes in cancer: Functional mechanisms and therapeutic perspectives. World J Clin Oncol 2026; 17(3): 116823
- URL: https://www.wjgnet.com/2218-4333/full/v17/i3/116823.htm
- DOI: https://dx.doi.org/10.5306/wjco.v17.i3.116823
Exosomes are nanosized membrane-coated vesicles secreted by a wide range of cell types[1]. With a typical diameter of 30-150 nm, exosomes carry diverse cargo-including plenty of proteins, nucleic acids, lipids, and other bioactive molecules, which makes them a critical mediators of intercellular communication[2]. Exosomes are involved in a variety of physiological and pathological processes. In recent years, exosome-based cancer research has grown rapidly, covering areas such as direct tumor suppression, drug delivery, and immunotherapy[3,4]. Among various cellular sources, exosomes derived from mesenchymal stem cells (MSCs) are regarded as particularly promising natural nanocarriers, due to their ready availability, low immunogenicity, and tumor-homing capacity. Given their translational relevance and well-documented functional complexity, this article reviews the multifaceted roles of exosomes in cancer therapy, focusing on MSC-derived exosomes to illustrate the multifaceted and dual roles of exosomes in cancer therapy.
Exosomes play a multifaceted role in regulating the tumor microenvironment, angiogenesis, tumorigenesis, metastasis, and resistance to chemo- and radiotherapy. In many cases, exosomes can directly influence tumor behavior by transferring functional molecules. On one hand, tumor-derived exosomes often promote malignancy[5]. For example, glioma-derived exosomes under hypoxia induce M2 macrophage polarization, trigger autophagy and facilitate tumor progression[6]; exosomal miRNA-23a from nasopharyngeal carcinoma promotes angiogenesis by inhibiting TSGA10[7]; exosomes from tumor-associated macrophages convey miR-589-3p, which promotes the development of ovarian cancer[8]; exosomes derived from bladder cancer stem cells can enhance the stemness and chemotherapy resistance of bladder cancer cells by delivering LUCAT1[9]; and exosomal miRNA92a-3p from cancer-associated fibroblasts enhances chemoresistance in colorectal cancer via the Wnt/β-catenin pathway[10].
On the other hand, some exosomes exert tumor-suppressive effects. For instance, exosomes carrying miRNA-let7e inhibit migration and invasion in non-small cell lung cancer[11]; exosomes from Plasmodium-infected mice suppress lung cancer angiogenesis by downregulating VEGFR2[12]; and MSC-derived exosomes deliver tumor-suppressive miRNAs such as miR-3940-5p, miR-22-3p and miR-16-5p, inhibiting invasion and progression in colorectal and endometrial cancers[13-17]. In addition, radiotherapy increases the production of exosomes and enhance the expression of miR-3160-5p within the exosomes, which inhibits pancreatic cancer progression[18]. These findings highlight that the functional outcome of exosomes-whether promotive or suppressive-is determined by their specific bioactive cargo. Consequently, appropriately increasing specific genes or substances within exosomes, that is, utilizing exosomes as effective carriers, is a hotspot in the field of exosome-based tumor therapy.
Compared to synthetic nanocarriers, exosomes offer superior biocompatibility, low immunogenicity, and high stability. Their lipid bilayer membrane protects encapsulated drugs or nucleic acids from degradation, making them ideal delivery systems in clinical field. For instance, dendritic cell (DC)-derived exosomes loaded with tumor peptides can eradicate tumors in vivo, leading to clinical trials as early as 2005[19]. Similarly, MSC-derived exosomes carrying siRNA targeting KRASG12D are being evaluated in pancreatic cancer patients[20].
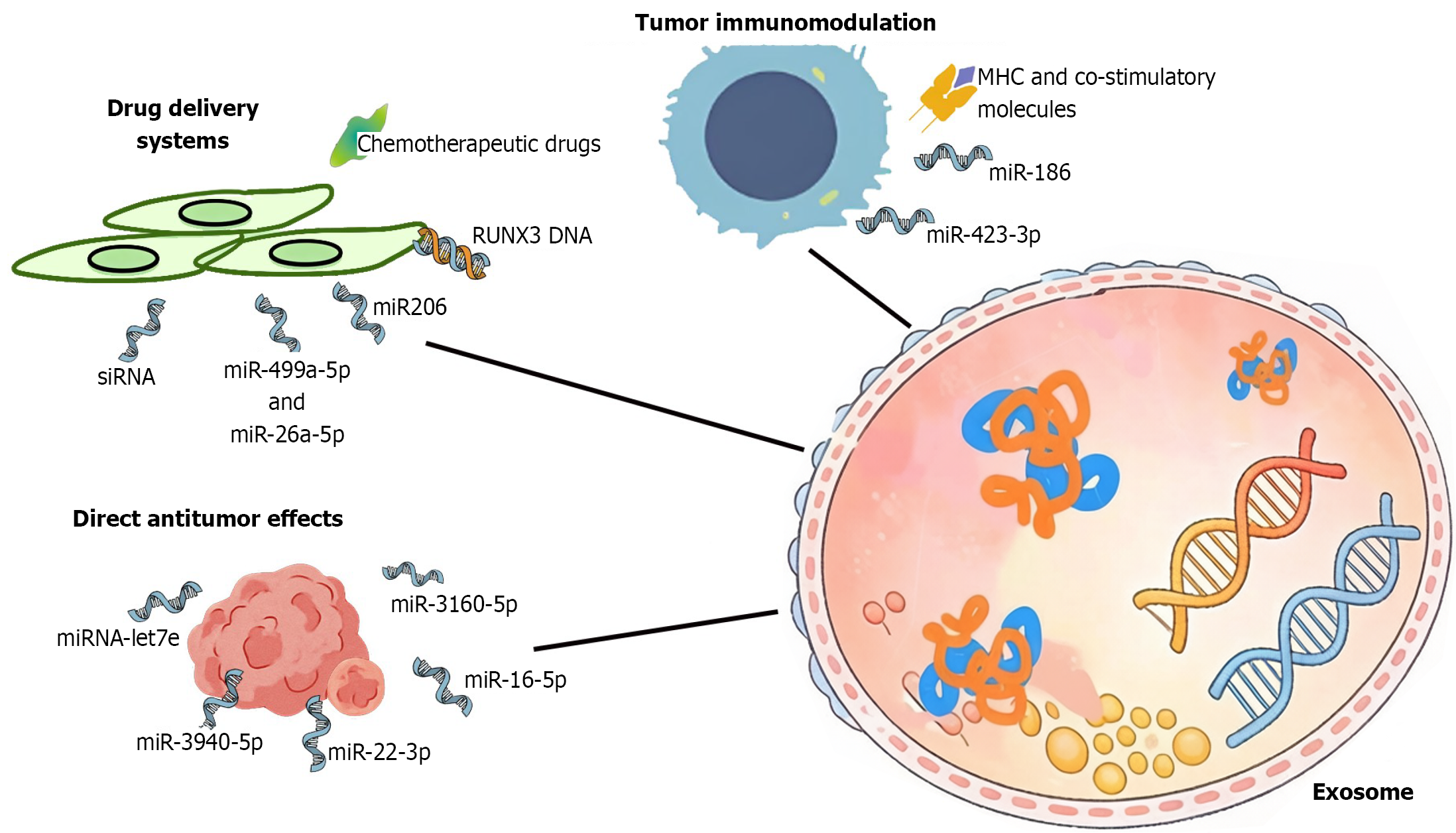
Furthermore, the presence of native surface proteins and ligands enables exosomes to recognize and bind specific cells or tissues, providing an inherent targeting capacity for precise drug delivery. This intrinsic targeting can be further enhanced through bioengineering techniques[21]. Moreover, their bilayer lipid structure helps minimize off-target effects on healthy tissues, which is particularly significant for cancer therapy. In chemotherapy, exosomes can deliver chemotherapeutic drugs such as paclitaxel and doxorubicin, enhancing antitumor efficacy and reducing systemic toxicity[22-24]. Zhang et al[25] utilized MSC-derived exosomes as a delivery system for a triple-drug combination (galectin-9 siRNA/DOGEM/indocyanine green), which substantially enhanced the synergistic effect of chemotherapy, immunotherapy, and phototherapy on pancreatic cancer. Compared to macromolecular drugs, RNA molecules are highly susceptible to degradation, making their delivery a major challenge. By using exosomes as carriers, these small RNA molecules can be shielded and delivered intact to target cells, significantly enhancing their bioavailability and therapeutic efficacy[26-29]. For instance, exosomes carrying the RUNX3 DNA (a key gene for lung adenocarcinoma) could significantly target cancer cells and induce their apoptosis[30]; exosomes derived from miR206-overexpressing tumor cells exhibited antitumor efficacy in colorectal cancer mice by modulating the intercellular interactions, the gut microbiota, and the immune microenvironment[31]; MSC-derived exosomes delivering miR-499a-5p and miR-26a-5p inhibit the migration, invasion, and tumor growth of endometrial cancer by targeting VAV3[32,33]. With their natural tumor-targeting properties and highly efficient delivery, exosomes have been positioned as a next-generation delivery platform for cancer therapy. To translate this potential into clinical application, key translational challenges, including standardized manufacturing, efficient cargo loading, and rigorous clinical validation-must be addressed. Future development should focus on engineering exosomes tailored to tumor-specific biomarkers, offering a versatile new modality for targeted cancer treatment.
Exosomes play a dual role in tumor immunology: They can mediate immunosuppression but can also be engineered for immune activation[34]. Tumor-derived exosomes function as potent immunosuppressive mediators that promote immune escape through multiple mechanisms. A key example is their carrying of immune checkpoint molecules such as programmed death ligand-1, which directly inhibits T cell function[35,36]. Their broad suppressive impact involves inducing T cell apoptosis, impairing the differentiation and maturation of DC, and inactivating natural killer (NK) cells. Furthermore, these exosomes actively shape a pro-tumorigenic microenvironment by enhancing the proliferation of regulatory T cells (Tregs) and regulatory B cells (Bregs), polarizing macrophages toward the immunosuppressive M2 phenotype, and expanding the population of myeloid-derived suppressor cells[5].
Conversely, immune cell-derived exosomes tend to enhance antitumor immunity. For example, DC-derived exosomes expressing MHC and co-stimulatory molecules promote T cell activation and tumor rejection[37]; NK cell-derived exosomes exert cytotoxic effects via miR-186 or induce apoptosis through perforin, granzyme B[38,39]; and T cell-derived bioactive exosomes exert Fas ligand-mediated cytotoxicity[40]. Additionally, exosomes carrying miR-423-3p inhibit M2 macrophage polarization, thereby suppressing cervical cancer growth[41]. These findings underscore the potential of exosomes as immunomodulatory agents in cancer treatment. Consequently, a key goal in the field is to shift the exosomal balance from tumor-promoting to immune-activating, by neutralizing detrimental exosomes and redirecting their natural delivery system for therapeutic purposes. The triple roles of exosomes on cancer therapy were illustrated in Figure 1.
MSCs are a prolific and most important source of exosomes. MSC-derived exosomes possess excellent biocompatibility and tissue penetration, but their effects on tumors are context-dependent[42,43]. Some studies show that MSC exosomes promote tumor progression and therapy resistance. For example, exosomal miR-106a-5p from MSCs promotes accelerates triple-negative breast cancer progression[44]; the lncRNA LINC00461 from MSC exosomes contributes to multiple myeloma pathogenesis[45]; MSC exosomes enhance resistance to 5-FU in gastric cancer by activating the CaM-Ks/Raf/MEK/ERK signaling cascade[46]; and exosomes from bone marrow-derived MSCs in acute myeloid leukemia and chronic myeloid leukemia are associated with resistance to tyrosine kinase inhibitors[47,48].
However, many other studies confirm that MSC-derived exosomes inhibit tumor growth and improve responses to chemo- and radiotherapy. For instance, exosomes from human umbilical cord MSCs carrying miR-375 inhibit the proliferation, invasion, and migration of esophageal cancer cells[49]; miR-503-3p and miR302a in human umbilical cord blood MSC exosomes inhibit endometrial cancer progression[16,17]; adipose tissue-derived MSC exosomes delivering miR-122 enhance sorafenib efficacy in hepatocellular carcinoma[50]; and MSC-derived exosomes transfected with miR34c inhibit the invasion, migration, proliferation, and epithelial-mesenchymal transition of nasopharyngeal carcinoma while reducing radioresistance[51]. Therefore, the application of MSC-derived exosomes must be carefully evaluated based on tumor type and molecular context. Ultimately, MSC-derived exosomes constitute a complex, context-dependent signaling network rather than a simple delivery vehicle. Harnessing their full potential requires a nuanced understanding of the molecular switches that dictate their dual roles. The future of this field lies in learning to decode these signals and programmatically engineer exosomes-or the MSCs that produce them-to ensure a reliably therapeutic outcome against cancer.
Despite the considerable promise of exosomes in cancer treatment, no exosome-based therapeutic has yet received full market approval, and the field remains in an active phase of clinical translation. Several engineered exosome-based strategies have advanced into early-stage clinical trials. For instance, building on promising preclinical results that demonstrated enhanced tumor retention and systemic anti-tumor immunity with minimal systemic inflammation[52], the exoSTING platform utilizes engineered exosomes to deliver STING agonists precisely to antigen-presenting cells within the tumor microenvironment, activating anti-tumor immunity. This candidate is currently under evaluation in a phase I/II trial (NCT04592484) for advanced solid tumors. In another study, curcumin encapsulated in exosomes was investigated for its effects on immune modulation and cellular metabolism in patients undergoing surgery for colon cancer (NCT01294072). Such studies provide preliminary evidence of safety and potential efficacy.
However, significant challenges must be addressed to enable broad clinical application. On the production front, establishing scalable manufacturing processes that comply with Good Manufacturing Practice is essential to ensure batch-to-batch consistency in exosome yield, purity, and function. Regarding quality control, standardized analytical methods for critical attributes-such as particle size, drug loading, and biological potency-are urgently needed. Furthermore, key translational and regulatory gaps remain, including a clearer understanding of in vivo pharmacokinetics, long-term safety profiles, and defined regulatory pathways for these complex biologics. Moving forward, advances in closed bioreactor systems, refined engineering strategies, and rigorously designed clinical studies will be crucial to translate exosome-based therapies from promising experimental agents into practical clinical tools.
As a natural information transfer system, exosomes play multifaceted roles in cancer therapy. They can directly affect tumor cells, serve as efficient drug carriers, and regulate antitumor immunity. MSC-derived exosomes, with their low immunogenicity and tumor-targeting capacity, are of particular translational interest, though their functional duality demands careful investigation. The clinical translation of exosome-based therapies, while promising, remains at an early stage, facing challenges in scalable manufacturing, standardization, and regulatory clarity. Future studies should focus on precise engineering and functional standardization of exosomes to develop safe and effective therapeutic agents in oncology.
| 1. | Zaborowski MP, Balaj L, Breakefield XO, Lai CP. Extracellular Vesicles: Composition, Biological Relevance, and Methods of Study. Bioscience. 2015;65:783-797. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1123] [Cited by in RCA: 976] [Article Influence: 88.7] [Reference Citation Analysis (1)] |
| 2. | Lai JJ, Chau ZL, Chen SY, Hill JJ, Korpany KV, Liang NW, Lin LH, Lin YH, Liu JK, Liu YC, Lunde R, Shen WT. Exosome Processing and Characterization Approaches for Research and Technology Development. Adv Sci (Weinh). 2022;9:e2103222. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 483] [Cited by in RCA: 423] [Article Influence: 105.8] [Reference Citation Analysis (0)] |
| 3. | Yong T, Zhang X, Bie N, Zhang H, Zhang X, Li F, Hakeem A, Hu J, Gan L, Santos HA, Yang X. Tumor exosome-based nanoparticles are efficient drug carriers for chemotherapy. Nat Commun. 2019;10:3838. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 749] [Cited by in RCA: 610] [Article Influence: 87.1] [Reference Citation Analysis (4)] |
| 4. | Lee KWA, Chan LKW, Hung LC, Phoebe LKW, Park Y, Yi KH. Clinical Applications of Exosomes: A Critical Review. Int J Mol Sci. 2024;25:7794. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 71] [Article Influence: 35.5] [Reference Citation Analysis (0)] |
| 5. | Saad SH, Kashanchi A, Zadeh MA, Williams A, Batrakova EV. Exosome-Mediated Crosstalk Between Cancer Cells and Tumor Microenvironment. Cells. 2025;14:1750. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 6. | Xu J, Zhang J, Zhang Z, Gao Z, Qi Y, Qiu W, Pan Z, Guo Q, Li B, Zhao S, Guo X, Qian M, Chen Z, Wang S, Gao X, Zhang S, Wang H, Guo X, Zhang P, Zhao R, Xue H, Li G. Hypoxic glioma-derived exosomes promote M2-like macrophage polarization by enhancing autophagy induction. Cell Death Dis. 2021;12:373. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 194] [Cited by in RCA: 183] [Article Influence: 36.6] [Reference Citation Analysis (0)] |
| 7. | Bao L, You B, Shi S, Shan Y, Zhang Q, Yue H, Zhang J, Zhang W, Shi Y, Liu Y, Wang X, Liu D, You Y. Metastasis-associated miR-23a from nasopharyngeal carcinoma-derived exosomes mediates angiogenesis by repressing a novel target gene TSGA10. Oncogene. 2018;37:2873-2889. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 155] [Cited by in RCA: 160] [Article Influence: 20.0] [Reference Citation Analysis (5)] |
| 8. | Wang J, Zhu Y, He Y, Shao W. TAM-derived exosomal miR-589-3p accelerates ovarian cancer progression through BCL2L13. J Ovarian Res. 2025;18:36. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 9. | Zhan Y, Zhou Z, Zhu Z, Zhang L, Yu S, Liu Y, Zhang X. Exosome-transmitted LUCAT1 promotes stemness transformation and chemoresistance in bladder cancer by binding to IGF2BP2. J Exp Clin Cancer Res. 2025;44:80. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 10. | Hu JL, Wang W, Lan XL, Zeng ZC, Liang YS, Yan YR, Song FY, Wang FF, Zhu XH, Liao WJ, Liao WT, Ding YQ, Liang L. CAFs secreted exosomes promote metastasis and chemotherapy resistance by enhancing cell stemness and epithelial-mesenchymal transition in colorectal cancer. Mol Cancer. 2019;18:91. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 640] [Cited by in RCA: 625] [Article Influence: 89.3] [Reference Citation Analysis (8)] |
| 11. | Xu S, Zheng L, Kang L, Xu H, Gao L. microRNA-let-7e in serum-derived exosomes inhibits the metastasis of non-small-cell lung cancer in a SUV39H2/LSD1/CDH1-dependent manner. Cancer Gene Ther. 2021;28:250-264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 27] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 12. | Yang Y, Liu Q, Lu J, Adah D, Yu S, Zhao S, Yao Y, Qin L, Qin L, Chen X. Exosomes from Plasmodium-infected hosts inhibit tumor angiogenesis in a murine Lewis lung cancer model. Oncogenesis. 2017;6:e351. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 50] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 13. | Harrell CR, Jovicic N, Djonov V, Volarevic V. Therapeutic Use of Mesenchymal Stem Cell-Derived Exosomes: From Basic Science to Clinics. Pharmaceutics. 2020;12:474. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 108] [Cited by in RCA: 94] [Article Influence: 15.7] [Reference Citation Analysis (1)] |
| 14. | Shakerian N, Darzi-Eslam E, Afsharnoori F, Bana N, Noorabad Ghahroodi F, Tarin M, Mard-Soltani M, Khalesi B, Hashemi ZS, Khalili S. Therapeutic and diagnostic applications of exosomes in colorectal cancer. Med Oncol. 2024;41:203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 15. | Guo G, Tan Z, Liu Y, Shi F, She J. The therapeutic potential of stem cell-derived exosomes in the ulcerative colitis and colorectal cancer. Stem Cell Res Ther. 2022;13:138. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 114] [Cited by in RCA: 101] [Article Influence: 25.3] [Reference Citation Analysis (0)] |
| 16. | Pan Y, Wang X, Li Y, Yan P, Zhang H. Human umbilical cord blood mesenchymal stem cells-derived exosomal microRNA-503-3p inhibits progression of human endometrial cancer cells through downregulating MEST. Cancer Gene Ther. 2022;29:1130-1139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 17. | Li X, Liu LL, Yao JL, Wang K, Ai H. Human Umbilical Cord Mesenchymal Stem Cell-Derived Extracellular Vesicles Inhibit Endometrial Cancer Cell Proliferation and Migration through Delivery of Exogenous miR-302a. Stem Cells Int. 2019;2019:8108576. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 72] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 18. | Nakaoka A, Salah M, Ruixian Z, Nakahana M, Shimizu Y, Qin Q, Zhang Q, Mukumoto N, Kobayashi K, Ikegaki Y, Akasaka H, Inubushi S, Koma YI, Ishihara T, Miyawaki D, Sasayama T, Sasaki R. Radiation suppresses liver metastases via exosome secretion in pancreatic cancer cells. Sci Rep. 2025;15:23712. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 19. | Zhang M, Hu S, Liu L, Dang P, Liu Y, Sun Z, Qiao B, Wang C. Engineered exosomes from different sources for cancer-targeted therapy. Signal Transduct Target Ther. 2023;8:124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 378] [Cited by in RCA: 342] [Article Influence: 114.0] [Reference Citation Analysis (0)] |
| 20. | Kamerkar S, LeBleu VS, Sugimoto H, Yang S, Ruivo CF, Melo SA, Lee JJ, Kalluri R. Exosomes facilitate therapeutic targeting of oncogenic KRAS in pancreatic cancer. Nature. 2017;546:498-503. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2260] [Cited by in RCA: 2072] [Article Influence: 230.2] [Reference Citation Analysis (4)] |
| 21. | Li J, Wang J, Chen Z. Emerging role of exosomes in cancer therapy: progress and challenges. Mol Cancer. 2025;24:13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 177] [Cited by in RCA: 163] [Article Influence: 163.0] [Reference Citation Analysis (0)] |
| 22. | Pascucci L, Coccè V, Bonomi A, Ami D, Ceccarelli P, Ciusani E, Viganò L, Locatelli A, Sisto F, Doglia SM, Parati E, Bernardo ME, Muraca M, Alessandri G, Bondiolotti G, Pessina A. Paclitaxel is incorporated by mesenchymal stromal cells and released in exosomes that inhibit in vitro tumor growth: a new approach for drug delivery. J Control Release. 2014;192:262-270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 805] [Cited by in RCA: 721] [Article Influence: 60.1] [Reference Citation Analysis (3)] |
| 23. | Coccè V, Franzè S, Brini AT, Giannì AB, Pascucci L, Ciusani E, Alessandri G, Farronato G, Cavicchini L, Sordi V, Paroni R, Dei Cas M, Cilurzo F, Pessina A. In Vitro Anticancer Activity of Extracellular Vesicles (EVs) Secreted by Gingival Mesenchymal Stromal Cells Primed with Paclitaxel. Pharmaceutics. 2019;11:61. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 51] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 24. | Tian Y, Li S, Song J, Ji T, Zhu M, Anderson GJ, Wei J, Nie G. A doxorubicin delivery platform using engineered natural membrane vesicle exosomes for targeted tumor therapy. Biomaterials. 2014;35:2383-2390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1585] [Cited by in RCA: 1416] [Article Influence: 118.0] [Reference Citation Analysis (0)] |
| 25. | Zhang R, Zhang Y, Hao F, Su Z, Duan X, Song X. Exosome-mediated triple drug delivery enhances apoptosis in pancreatic cancer cells. Apoptosis. 2025;30:1893-1911. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 26. | Jafari A, Babajani A, Abdollahpour-Alitappeh M, Ahmadi N, Rezaei-Tavirani M. Exosomes and cancer: from molecular mechanisms to clinical applications. Med Oncol. 2021;38:45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 66] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 27. | Pi F, Binzel DW, Lee TJ, Li Z, Sun M, Rychahou P, Li H, Haque F, Wang S, Croce CM, Guo B, Evers BM, Guo P. Nanoparticle orientation to control RNA loading and ligand display on extracellular vesicles for cancer regression. Nat Nanotechnol. 2018;13:82-89. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 445] [Cited by in RCA: 390] [Article Influence: 48.8] [Reference Citation Analysis (4)] |
| 28. | Shu D, Li H, Shu Y, Xiong G, Carson WE 3rd, Haque F, Xu R, Guo P. Systemic Delivery of Anti-miRNA for Suppression of Triple Negative Breast Cancer Utilizing RNA Nanotechnology. ACS Nano. 2015;9:9731-9740. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 240] [Cited by in RCA: 214] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 29. | Yin H, Wang H, Li Z, Shu D, Guo P. RNA Micelles for the Systemic Delivery of Anti-miRNA for Cancer Targeting and Inhibition without Ligand. ACS Nano. 2019;13:706-717. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 66] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 30. | Jeon J, Byun H, Lee MC, Kim J, Park SG, Madrigal-Salazar AD, González ML, Lopez-Pacheco MC, Liu W, Nasr ML, Lee SH, Lee YS, Lee LP, Kim WJ, Kim D, Shin SR. Exosome-Mediated RUNX3 DNA Delivery for Lung Cancer Therapy. ACS Appl Mater Interfaces. 2025;17:66339-66357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 31. | Tian J, Liu M, Yang X, Wang T, Zhou R, Liu J, Ma B, Su C, Duan X. Tumor-derived exosome-based microRNA-206 delivery system as dual modulators of the immune microenvironment and gut microbiota for colorectal cancer therapy. Int J Biol Macromol. 2025;321:146489. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 32. | Wang J, Gong X, Yang L, Li L, Gao X, Ni T, Yang X, Fan Q, Sun X, Wang Y. Loss of exosomal miR-26a-5p contributes to endometrial cancer lymphangiogenesis and lymphatic metastasis. Clin Transl Med. 2022;12:e846. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 33. | Jing L, Hua X, Yuanna D, Rukun Z, Junjun M. Exosomal miR-499a-5p Inhibits Endometrial Cancer Growth and Metastasis via Targeting VAV3. Cancer Manag Res. 2020;12:13541-13552. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 59] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 34. | Abedi A, Moosazadeh Moghaddam M, Kachuei R, Imani Fooladi AA. Exosomes as a Therapeutic Strategy in Cancer: Potential Roles as Drug Carriers and Immune Modulators. Biochim Biophys Acta Rev Cancer. 2025;1880:189238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 35. | Wang C, Huang X, Wu Y, Wang J, Li F, Guo G. Tumor Cell-associated Exosomes Robustly Elicit Anti-tumor Immune Responses through Modulating Dendritic Cell Vaccines in Lung Tumor. Int J Biol Sci. 2020;16:633-643. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 77] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 36. | Li C, Qiu S, Jin K, Zheng X, Zhou X, Jin D, Xu B, Jin X. Tumor-derived microparticles promote the progression of triple-negative breast cancer via PD-L1-associated immune suppression. Cancer Lett. 2021;523:43-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 44] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 37. | Zitvogel L, Regnault A, Lozier A, Wolfers J, Flament C, Tenza D, Ricciardi-Castagnoli P, Raposo G, Amigorena S. Eradication of established murine tumors using a novel cell-free vaccine: dendritic cell-derived exosomes. Nat Med. 1998;4:594-600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1839] [Cited by in RCA: 1715] [Article Influence: 61.3] [Reference Citation Analysis (3)] |
| 38. | Neviani P, Wise PM, Murtadha M, Liu CW, Wu CH, Jong AY, Seeger RC, Fabbri M. Natural Killer-Derived Exosomal miR-186 Inhibits Neuroblastoma Growth and Immune Escape Mechanisms. Cancer Res. 2019;79:1151-1164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 290] [Cited by in RCA: 269] [Article Influence: 38.4] [Reference Citation Analysis (0)] |
| 39. | Kim IY, Kim HY, Song HW, Park JO, Choi YH, Choi E. Functional enhancement of exosomes derived from NK cells by IL-15 and IL-21 synergy against hepatocellular carcinoma cells: The cytotoxicity and apoptosis in vitro study. Heliyon. 2023;9:e16962. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 20] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 40. | Seo N, Shirakura Y, Tahara Y, Momose F, Harada N, Ikeda H, Akiyoshi K, Shiku H. Activated CD8(+) T cell extracellular vesicles prevent tumour progression by targeting of lesional mesenchymal cells. Nat Commun. 2018;9:435. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 185] [Cited by in RCA: 176] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 41. | Zhou Y, Zhang Y, Gong H, Luo S, Cui Y. The Role of Exosomes and Their Applications in Cancer. Int J Mol Sci. 2021;22:12204. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 145] [Cited by in RCA: 133] [Article Influence: 26.6] [Reference Citation Analysis (0)] |
| 42. | Yeo RW, Lai RC, Zhang B, Tan SS, Yin Y, Teh BJ, Lim SK. Mesenchymal stem cell: an efficient mass producer of exosomes for drug delivery. Adv Drug Deliv Rev. 2013;65:336-341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 718] [Cited by in RCA: 659] [Article Influence: 50.7] [Reference Citation Analysis (0)] |
| 43. | Lin Z, Wu Y, Xu Y, Li G, Li Z, Liu T. Mesenchymal stem cell-derived exosomes in cancer therapy resistance: recent advances and therapeutic potential. Mol Cancer. 2022;21:179. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 255] [Cited by in RCA: 237] [Article Influence: 59.3] [Reference Citation Analysis (0)] |
| 44. | Xing L, Tang X, Wu K, Huang X, Yi Y, Huan J. LncRNA HAND2-AS1 suppressed the growth of triple negative breast cancer via reducing secretion of MSCs derived exosomal miR-106a-5p. Aging (Albany NY). 2020;13:424-436. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 39] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 45. | Deng M, Yuan H, Liu S, Hu Z, Xiao H. Exosome-transmitted LINC00461 promotes multiple myeloma cell proliferation and suppresses apoptosis by modulating microRNA/BCL-2 expression. Cytotherapy. 2019;21:96-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 89] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 46. | Ji R, Zhang B, Zhang X, Xue J, Yuan X, Yan Y, Wang M, Zhu W, Qian H, Xu W. Exosomes derived from human mesenchymal stem cells confer drug resistance in gastric cancer. Cell Cycle. 2015;14:2473-2483. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 194] [Cited by in RCA: 201] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 47. | Viola S, Traer E, Huan J, Hornick NI, Tyner JW, Agarwal A, Loriaux M, Johnstone B, Kurre P. Alterations in acute myeloid leukaemia bone marrow stromal cell exosome content coincide with gains in tyrosine kinase inhibitor resistance. Br J Haematol. 2016;172:983-986. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 74] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 48. | Zhang X, Yang Y, Yang Y, Chen H, Tu H, Li J. Exosomes from Bone Marrow Microenvironment-Derived Mesenchymal Stem Cells Affect CML Cells Growth and Promote Drug Resistance to Tyrosine Kinase Inhibitors. Stem Cells Int. 2020;2020:8890201. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 51] [Article Influence: 8.5] [Reference Citation Analysis (1)] |
| 49. | He Z, Li W, Zheng T, Liu D, Zhao S. Human umbilical cord mesenchymal stem cells-derived exosomes deliver microRNA-375 to downregulate ENAH and thus retard esophageal squamous cell carcinoma progression. J Exp Clin Cancer Res. 2020;39:140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 93] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 50. | Lou G, Song X, Yang F, Wu S, Wang J, Chen Z, Liu Y. Exosomes derived from miR-122-modified adipose tissue-derived MSCs increase chemosensitivity of hepatocellular carcinoma. J Hematol Oncol. 2015;8:122. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 616] [Cited by in RCA: 579] [Article Influence: 52.6] [Reference Citation Analysis (0)] |
| 51. | Wan FZ, Chen KH, Sun YC, Chen XC, Liang RB, Chen L, Zhu XD. Exosomes overexpressing miR-34c inhibit malignant behavior and reverse the radioresistance of nasopharyngeal carcinoma. J Transl Med. 2020;18:12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 93] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 52. | Jang SC, Economides KD, Moniz RJ, Sia CL, Lewis N, McCoy C, Zi T, Zhang K, Harrison RA, Lim J, Dey J, Grenley M, Kirwin K, Ross NL, Bourdeau R, Villiger-Oberbek A, Estes S, Xu K, Sanchez-Salazar J, Dooley K, Dahlberg WK, Williams DE, Sathyanarayanan S. ExoSTING, an extracellular vesicle loaded with STING agonists, promotes tumor immune surveillance. Commun Biol. 2021;4:497. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 148] [Cited by in RCA: 133] [Article Influence: 26.6] [Reference Citation Analysis (0)] |