Published online Mar 24, 2026. doi: 10.5306/wjco.v17.i3.116534
Revised: December 2, 2025
Accepted: January 13, 2026
Published online: March 24, 2026
Processing time: 130 Days and 1.6 Hours
Gastric cancer remains a global health challenge with high mortality. While surgery is the primary treatment, postoperative complications and recurrence sig
Core Tip: This article comprehensively summarizes the multifaceted role of traditional Chinese medicine in postoperative gastric cancer care. We detail how traditional Chinese medicine strategies, ranging from herbal formulas to strengthen body resistance, eliminate toxins, and prevent metastasis, to acupuncture for improving gastrointestinal function, contribute to enhanced recovery, reduced recurrence, and improved quality of life. The paper also explores the bioactive compounds of herbs and their molecular mechanisms, alongside ongoing clinical trials, advocating for an evidence-based integrative approach to improve patient outcomes.
- Citation: Zhao XF, Zhang YJ, Wang SL, Lian FM, Liu Z. Adjuvant role of traditional Chinese medicine in postoperative gastric cancer. World J Clin Oncol 2026; 17(3): 116534
- URL: https://www.wjgnet.com/2218-4333/full/v17/i3/116534.htm
- DOI: https://dx.doi.org/10.5306/wjco.v17.i3.116534
Gastric cancer (GC) is one of the most commonly occurring malignant neoplasms of the digestive tract. Recent epidemiological studies have demonstrated that GC has one of the highest incidence and mortality rates among all cancers[1]. According to the GLOBOCAN 2022 database, GC ranks as the fifth most common cancer globally in terms of incidence and the fifth leading cause of cancer death. In 2022, GC accounted for 4.9% of all cancers worldwide (968350 new cases) and 6.8% of all cancer-related deaths (659000 deaths)[2]. The development of GC is the result of a multifactorial process influenced by various factors, including Helicobacter pylori infection, age, and dietary influences such as high salt intake, the consumption of foods preserved by salting, processed meat, grilled or barbecued meat, and diets with low intake of fruits and vegetables[3]. In the early stage of GC, clinical symptoms are not significant, specific indicators are poor, and the condition is hidden and easily overlooked, thus making clinical diagnosis difficult. Once detected, the cancer has already reached the middle and late stages, which is difficult to treat and has high mortality rate.
With regard to therapeutic interventions, gastrectomy constitutes the primary treatment approach for patients with non-metastatic GC and the only treatment with curative intent for advanced GC. For early-stage GC, the primary treatment modality is endoscopic resection. The survival rate of non-early operable GC can be enhanced through surgical intervention in conjunction with lymph node dissection and perioperative or adjuvant chemotherapy. For advanced GC, the use of sequential chemotherapy and molecular targeted therapy has been established[3].
Traditional Chinese medicine (TCM) has attracted interest as a potential treatment for cancer. Historically, TCM views GC through the lens of “accumulation” and “deficiency”. The integration of TCM into GC care has evolved from purely symptomatic relief in ancient texts to a modern adjuvant role focusing on correcting the “spleen deficiency” (digestive compromise) and “blood stasis” (microenvironment stagnation) often exacerbated by surgical stress. Both internal herbal medicines and external TCM treatments have been shown to enhance body function following GC surgery. Herbal medicine and acupuncture have a long-standing presence in East Asia, and their clinical benefits have been recognized in several countries. The characteristic multi-component and multi-target nature of TCM offers a wide range of biologically active compounds. These compounds, often used in synergistic combinations, have demonstrated multifaceted pharmacological activities, including anti-inflammatory, anti-angiogenic, and immunomodulatory effects, showing promise in anticancer applications[4]. Guided by the holistic view of TCM, which addresses not only the etiology of the disease, but also supports the vital energy, TCM has improved clinical symptoms and quality of life in patients with advanced GC, prolonged survival, alleviated postoperative weakness and reduced toxic side effects associated with radiotherapy and chemotherapy[5]. A meta-analysis encompassing 13 randomized controlled trials (RCT) of oral TCM for resectable gastric tumors revealed that postoperative adjuvant herbal therapy was associated with a diminished risk of mortality in patients with GC[6].
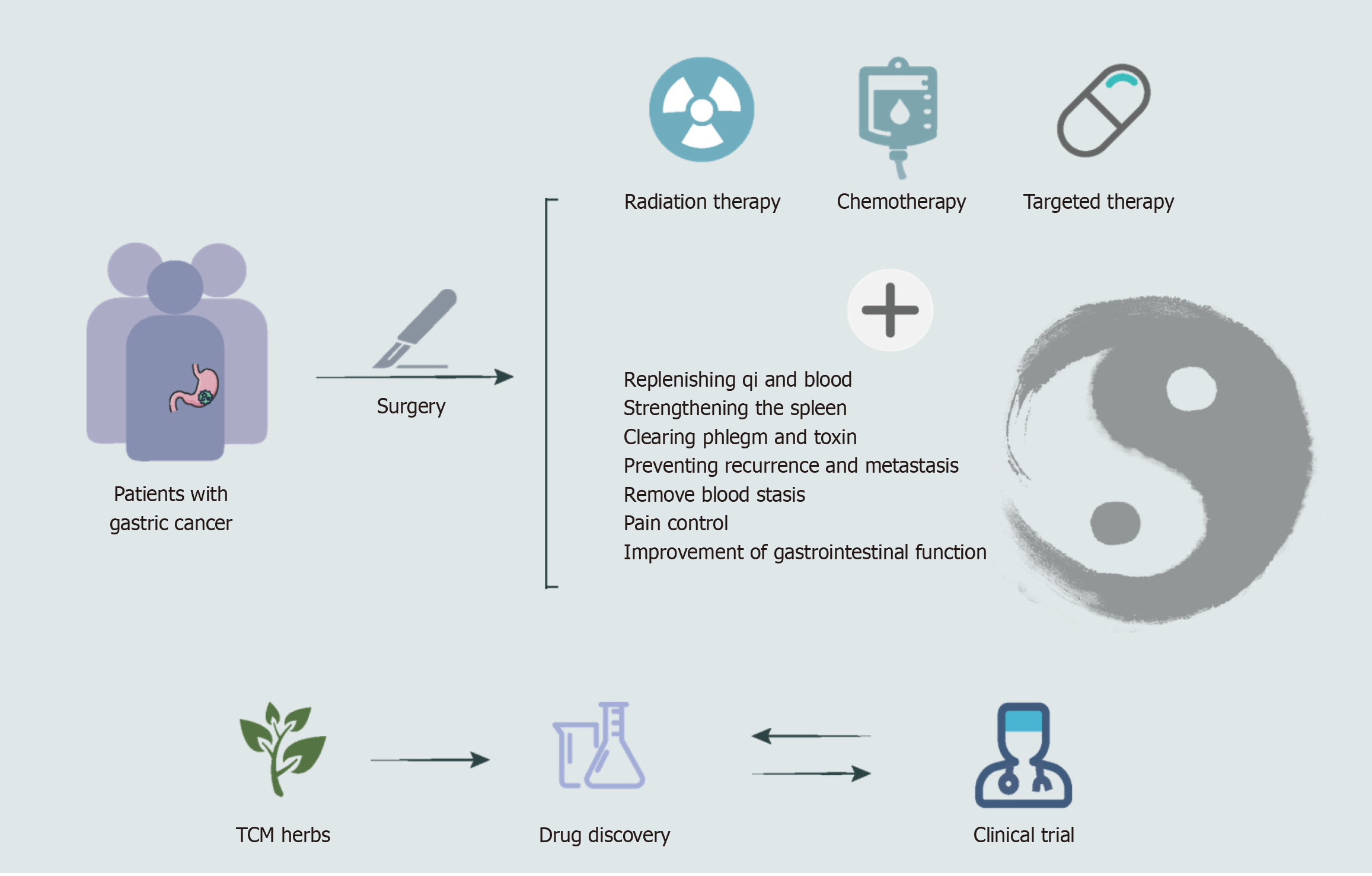
Given the significant global burden of GC and a growing interest in integrative oncology, this narrative review aims to synthesize the current landscape of TCM as an adjuvant therapy in postoperative GC management. We provide a comprehensive overview of clinical studies evaluating herbal formulations and other TCM modalities, delve into the mechanistic research on their bioactive components, and summarize key ongoing clinical trials. Furthermore, we critically discuss the challenges facing the clinical application of TCM, including issues of standardization and evidence quality, and outline future directions for research. Ultimately, this review serves as a resource for clinicians and researchers by providing an updated understanding of TCM’s potential role in improving patient outcomes, reducing recurrence, and enhancing quality of life, as visually summarized in Figure 1.
Modern medicine primarily eliminates cancer foci through surgery, radiotherapy, and chemotherapy. In contrast, TCM offers an adjunctive approach for advanced and postoperative GC, focusing on strengthening spleen function (in TCM, the spleen system governs digestion and nutrient absorption) and benefiting qi (vital energy), anti-tumor, and anti-metastasis effects, while also alleviating treatment side effects, improving quality of life, reducing recurrence, and potentially prolonging survival[7]. From a TCM perspective, cancer toxicity (pathogenic factors believed to cause inflammation and masses) is believed to deplete qi (vital energy) and blood (nutritive essence), hindering the body’s ability to eliminate toxins and fostering a pathological cycle.
Following gastrectomy, TCM theory posits that surgical trauma and anesthesia can precipitate qi and Blood disorders or deficiencies, leading to a spectrum of pain and visceral dysfunctions. Patients often exhibit pronounced “spleen deficiency” (a syndrome indicating impaired digestion, fatigue, and weakness) and “blood deficiency” (a syndrome of insufficient nutritive essence, leading to paleness and fatigue)[8,9]. Consequently, the typical pathological state of postoperative GC patients is characterized by “qi deficiency” (a syndrome of insufficient vital energy), “blood stasis” (a condition of impaired blood circulation), and “residual toxin” (lingering pathogenic factors or tumor burden), mani
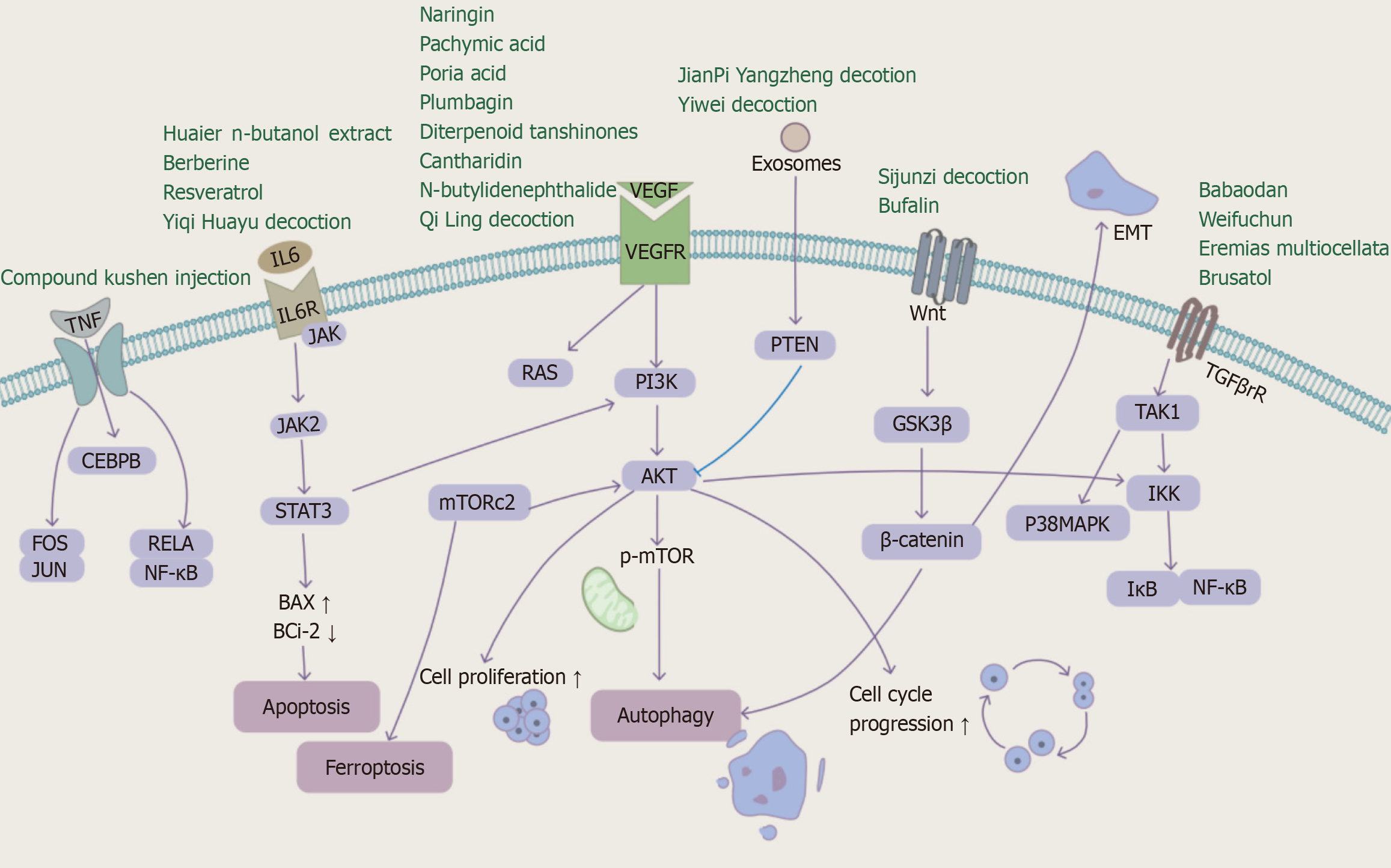
Table 1 summarizes the clinical studies of TCM in the treatment of GC after surgery, and Table 2 summarizes the anti-GC effects of TCM formulas and the corresponding mechanisms. Figure 2 illustrates the bioactive compounds identified from representative Chinese herbal medicines and formulas, along with their potential targets and pharmacological effects.
| TCM | Compounds/constituents | Intervention | Intervention time | Postoperative condition | Study design | Outcome | Ref. |
| Sijunzi decoction | Radix Codonopsis (Dangshen), Poria cocos (Fuling), Rhizoma Atractylodis Macrocephalae (Baizhu), and Radix Glycyrrhizae (Gancao) | Sijunzi decoction + enteral nutrition vs enteral nutrition alone | 8-10 days after operation | POCs after gastrectomy, Postoperative nutritional therapy | 10 RCTs (688 participants), meta | Enhancing immune function, reducing the incidence of postoperative complications, and improving body nutrition status | [9] |
| Jianpi Yangzheng decoction | 15 g Dangshen, 10 g Baizhu, 10 g Fuling, 15 g Shengyiren, 10 g Danggui, 15 g Shanyao, 10 g Muxiang, 10 g Baishao, 6 g Chenpi, 10 g Baqia, 15 g Shijianchuan and 3 g Zhigancao | TCM nursing intervention group (n = 520) vs the routine nursing intervention group (n = 512) | Every day after gastrectomy for at least 6 months | POCs after gastrectomy | Prospective, randomized controlled study | Improving POCs, mental status, long-term survival and reduces the recurrence | [25] |
| Yiqi Huayu Jiedu decoction | Astragalus membranaceus, 15 g; Codonopsis pilosula, 15 g; Atractylodes macrocephala, 10 g; Angelica, 10 g; Radices paeoniae alba, 10 g; Pericarpium citri reticulatae, 6 g; Pinellia, 10 g; Rhizoma sparganii, 10 g; Curcuma zedoary, 10 g; Chinese sage herb, 30 g; Hedyotis diffusa, 30 g; and Radix glycyrrhizae preparata, 5 g | Chemotherapy alone (n = 238) vs chemotherapy combined with Yiqi Huayu Jiedu decoction (n = 251) | More than 6 months | Stage II or III GC after radical gastrectomy | Multicenter, prospective, cohort study | Improve DFS rate in patients with GC stage III after radical gastrectomy and reduce the risk of recurrence and metastasis and improve the quality of life in patients with stage II or III GC after radical gastrectomy | [21] |
| Weifuchun | Radix Ginseng Rubra (Red Ginseng), Rabdosia amethystoides H. Hara (Xiangchacai), and fried Fructus Aurantii (Zhike) | Standard chemotherapy supplemented with Weifuchun tablets (n = 32) vs chemotherapy alone (n = 31) | 6 months | Stages II and III GC and having undergonecurative resection within 8 weeks | Randomized, controlled, single-blind study | Reduce the 2-year recurrence and metastasis rates | [26] |
| TCM therapy | TCM therapies such as TCM decoction, moxibustion therapy, auricular-plaster therapy, hot ironing therapy, personalized food therapy, etc. | N/A | N/A | After the GC surgery | Meta-analysis (32 RCTs) | Reduce the risks of postoperative Wound infections and help shorten the time of wound healing; help patients rebuild a normal immune system and nutritional state | [38] |
| TCM therapy | The most common single herb prescriptions are Hwang-Chyi (Radix Hedysari; 11.8%), Dan-Shen (Radix Salviae Miltiorrhizae; 9.8%), Yan-Hu-Suo (Rhizoma Corydalis; 9.4%), Bai-Hua-She-She-Cao (Herba Hedyotidis Diffusae; 9.2%), and Hou-Pu (Cortex Magnoliae; 9.0%). The most common herbal formula prescriptions are Xiang-Sha-Liu-Jun-Zi-Tang (15.5%), Ping-Wei-San (12.6%), Ban-Xia-Xie-Xin-Tang (11.8%), Bu-Zhong-Yi-Qi-Tang (10.3%), and Shen-Lin-Bai-Zhu-San (10.2%) | Short-term TCM users (TCM use 30-179 days) (n = 1178) vs long-term TCM users (TCM use ≥ 180 days) (n = 523) vs nonusers (n = 5103) | N/A | Patients with GC following surgery and adjuvant chemotherapy | Nationwide matched cohort study | Short-term and long-term TCM users were independently associated with a decreased risk of death with HRs of 0.59 (95%CI: 0.55-0.65) in TCM users and 0.41 (95%CI: 0.36-0.47) in TCM nonusers | [100] |
| Zhipu Liujunzi decoction | Ginseng, solomonseal rhizome, figwort root, Ganoderma sinense, glabrous greenbrier rhizome, appendiculate cremastra pseudobulb and bamboo shavings (15 g each), 30 g of Mongolian milkvetch root, 20 g of large-head atractylodes rhizome, 6 g of villous amomum fruit, 6 g of finger citron, 12 g of dried tangerine peel, and 12 g of ginger processed pinellia | Early chemotherapy (oxaliplatin + capecitabine) (n = 50) vs Zhipu Liujunzi decoction + early chemotherapy (n = 50) | 2 weeks | After radical gastrectomy | Retrospective analysis | Improve the immune function and quality of life for GC patients with higher safety | [15] |
| Decoction with Astragalus mongholicus and Semen Cuscutae | Radix Pseudostellariae 45 g, Rhizoma Atractylodis Macrocephalae 45 g, Rhizoma Zingiberis 45 g, Radix Glycyrrhizae Preparata 45 g, Rhizoma Pinellinae Praeparata 15 g, Fructus Amomi 15 g, Rhiizoma Dioscoreae 60 g, Astragalus mongholicus 120 g, Semen Cuscutae 60 g, Lignum Millettiae 60 g | Each N-of-1 trial consisted of two or three cycles with treatment decotion and control decotion assigned randomly | 20-30 weeks (3-week standard chemotherapy and then used 3-day treatment periods) | After radical gastrectomy | A series of N-of-1 trials with randomization, double-blind, and controlled treatment | The QoL score after the trial was reported is a little higher than before | [101] |
| San Jie Pai Shi decoction | 10 g of Codonopsis pilosula, 6 g of Radix glycyrrhiza, 12 g of the rhizome of Atrac_x005f tylodis macrocephalae, 9 g of the cortex of Magnoliae officinalis, 12 g of immature bitter orange, 12 g of the endothelium cor neum of Gigeriae galli, and 9 g of Rheum officinale | N/A | 2 weeks | Gastric bezoar after Billroth II gastrectomy for GC | Case report | Gastroscopic examination showed that the gastric bezoar had dissolved | [22] |
| Xuesaitong injection | Extract from the roots of Pannax notoginseng (Sanqi) | Conventional anti-infection and transfusion treatment + 400 mg Xuesaitong Injection (n = 25) vs conventional anti-infection and transfusion treatment (n = 25) | 6 days from the first day after surgery | After laparoscopic surgery for gastric cancer | RCT | The occurrence rate of deep vein thrombosis on day 7 after operation in the Xuesaitong group was lower than that in the control group | [41] |
| Rikkunshito (Liujunzi decoction) | Dried Atractyloidis lanceae rhizoma, Ginseng radix, Pi_x005f nelliae tuber, Hoelen, Zizyphi fructus, Aurantii nobilis percarpium, Glycyrrhizae radix, and Zingiberis rhizome | Rikkunshito and then off treatment (n = 4) vs off treatment and then Rikkunshito (n = 7) | On treatment and off treatment for 4 weeks each | Underwent PPG for early GC more than 5 years ago | Crossover design study | Improved gastric emptying and ameliorated postoperative symptoms of patients who had undergone a PPG | [39] |
| Dai-kenchu-to (Dajianzhong decotion) | Dried ginger root, ginseng, and zanthoxylum fruit | Dai-kenchu-to and then off treatment (n = 10) vs off treatment and then Dai-kenchu-to (n = 7) | On treatment and off treatment for 2 weeks each | Underwent total gastrectomy with jejunal pouch interposition for GC | Crossover design study | Reduced stasis-related symptoms, accelerated emptying of both liquid and solid meals from the pouch, increased intestinal motility and decreased postoperative symptoms | [42] |
| Fuzheng Huoxue anticancer prescription | Codonopsis pilosula (Franch.) N annf (15 g), Astragalus membranaceus (Fisch.) Bge (15 g), Atractylodes macrocephala Koidz (12 g), Poria cocos (Schw.) Wolf (12 g), Rehmannia glutinosa (Gaertn.) Lib osch (12 g), Adenophoratetraphylla (Thunb.) Fisch (15 g), Salvia miltiorrhiza Bge (15 g), and Angelica sinensis (Oliv.) Diels (12 g) | Fuzheng Huoxue anticancer prescription (n = 35) vs control prescription (n = 34) | Five days, starting at one month after an operation | Mid- or late-stage (23 cases received palliative surgery and 11 cases radical surgery) | RCT | Replenish vital functions (Zhengqi), correct a hypercoagulatory state, improve immunologic function, and extend patient survival times, directly inhibit gastric tumor growth without producing toxic side effects | [13] |
| Powder of eight Noble Ingredients and cinnamon twig and Poria pills | Ginseng, white atractylodes rhizome, Poria, Pinellia, 4 g each; tangerine peel and Fructus ZiZiphi Jujubae, 2 g each; liquorice, 1 g; dried ginger, 0.5 g; cinnamon twig, Poria, Moutan bark, peach seed, peony root, 4 g each | FAP regimen (5-fluorouracil, 300 mg/m2 intravenous gutta for five consecutive days; adriamycin, 40 mg/m2 for the first day), or the CODP regimens (cyclophosphamide, vincristine, daunorubicin, prednisone) (n = 31) vs FAP regimen and TCM (n = 32) | 6 months | Metastasis formation after surgery for GC | RCT | Metastatic relapse was reduced and the ornithine decarboxylase levels of the gastric mucosa were sharply reduced | [27] |
| Modified ShengYangYiwei decoction | 30 g ginseng, 9 g atractylodes macrocephala, 9 g poria, 60 g astragalus, 15 g white peony, 30 g pinellia ternata, 6 g rhizoma coptidis, 9 g rhizoma alismatis, 12 g dried tangerine peel, 10 g magnolia officinalis, 9 g rhizoma Notopterygii, 9 g angelica pubescens, 9 g fangfeng, 9 g bupleurum chinense, 5 g ginger, 6 g jujube (denuded), 6 g cohosh, 9 g kudzu, 9 g pueraria lobata, 15 g Shijian Chuan, 12 g divine koji, and 6 g raw licorice | Modified ShengYangYiwei decoction one week before the examination (n = 56) vs routine gastrointestinal endoscopy (n = 50) | One week before the examination | GC patients after painless gastroscopy | Prospective, randomized controlled study | Optimize the anesthesia program during painless gastroscopy, improve the gastrointestinal function of patients after the operation, reduce the occurrence of examination-related complications | [102] |
| Acupuncture | Bilateral ST-36 (Zusanli), SP-6 (Sanyinjiao), LI-4 (Hegu), TE-6 (Ziagou), LV-3 (Taichong), LI-11 (Quchi), and unilaterally at GV-20 (Baihui), EX-HN3 (Yintang), GV-26 (Shuigou), and CV-24 (Chengjiang) | Acupuncture (n = 18) vs non-acupuncture (n = 18) | Once daily for 5 consecutive days starting at postoperative day 1 | Postoperative Ileus after Distal Gastrectomy for GC | Prospective, randomized controlled, phase III study | Promoting the passage of sitz markers in small intestine; reducing the duration of POI after gastric cancer surgery, by earlier recovery of small bowel movement | [45] |
| Acupuncture therapy including electroacupuncture, manual acupuncture, moxibustion, TEAS, warm needling, and ear acupressure | Acupuncture therapy vs standard perioperative care or sham/placebo acupuncture | N/A | Postoperative gastrointestinal dysfunction in patients with gastric and colorectal cancer | Umbrella review (six systematic reviews/meta-analyses) | Improve postoperative gastrointestinal function for postoperative GC patients | [44] | |
| Zusanli (ST 36), Shangjuxu (ST 37), Xiajuxu (ST 39), Gongsun (SP 4), Sanyinjiao (SP 6) | Acupuncture + routine treatment (n = 40) vs routine treatment (continuous gastrointestinal decompression) (n = 40) | 30 minutes each time, once a day, 5 days as a course, 1-3 courses | Functional delayed gastric emptying after GC surgery | RCT | The first exhaust time, remove gastric tube time, liquid food intake time and hospital stay were shortened | [103] | |
| Baihui (GV 20), Zusanli (ST 36), Neiguan (PC 6), etc. | High-dose acupuncture (n = 19) vs low-dose acupuncture (n = 20) vs control group (n = 19) | Chemotherapy cycle of 21 days, 3 courses | Patients with GC during adjuvant chemotherapy after gastrectomy | RCT | The total score and the scores of feeling of non-well being, pain and shortness of breath of ESAS in the acupuncture group were lower than the control group | [104] | |
| Bilateral ST 36 (Zusanli), SP 6 (Sanyinjiao), LI 4 (Hegu), TE 6 (Ziagou), LV 3 (Taichong), LI 11 (Quchi), and unilaterally at GV 20 (Baihui), EX HN3 (Yintang), GV 26 (Shuigou), and CV 24 (Chengjiang) | Acupuncture (n = 5) vs nonacupuncture (n = 5) | 5 consecutive days starting on postoperative day 1 | POI after GC surgery | Prospective, randomized, controlled pilot study | Reduce duration of POI after GC surgery, by earlier recovery of small bowel movement | [105] | |
| Simo decoction + acupuncture | Fructus aurantii (Zhike), Radix aucklandiae (Muxiang), Semen arecae (Binlang), Radix linderae (Wuyao)PC6 (Neiguan), ST36 (Zusanli), LV-3 (Taichong), SP-6 (Sanyinjiao) | Control group (n = 30) vs Simo decoction (n = 30) vs acupuncture + Simo decoction (n = 30) | 10 days | After radical surgeries for GC | RCT | Accelerate early air exhaustion and defecation, improve clinical symptoms, bi-directional regulate peripheral white blood cells | [106] |
| TEAS | Hegu (LI4), Neiguan (PC6), Weishu (BL21), Xiaochangshu (BL27), Zusanli (ST36), Shangjuxu (ST37) | TEAS therapy on postoperative day 1-3 (n = 41) vs usual care (n = 40) | 3 days | Patients underwent radical surgery for GC (laparoscopy/robot) | Unblinded randomized controlled trial | Relieve postoperative pain and promote the recovery of gastrointestinal function | [49] |
| Electro-acupuncture | Bilateral ST36, PC6, SP4, and DU20, EX-HN3, and selected Back-shu points | High-dose EA (seven times each chemotherapy cycle for three cycles) + adjuvant chemotherapy vs low-dose EA (three times each chemotherapy cycle) + adjuvant chemotherapy vs or adjuvant chemotherapy | 3 chemotherapy cycles | During adjuvant chemotherapy following gastrectomy | Open-label, multicenter, parallel controlled trial | Improve HRQOL, controlling symptom burden, and reducing toxicity during adjuvant chemotherapy in GC patients | [107] |
| Fu’s subcutaneous needling | Erector spinae muscle and rectus abdominis muscle | N/A | Once a day for three days | Progressive nausea, vomiting and stomach fullness, with a bloating abdomen ten days after laparoscopic radical gastrectomy for GC | Case report | Gastrointestinal motility enhancement and gastric drainage volume decrement | [46] |
| Acupuncture and moxibustion | N/A | Experimental group (n = 638) vs control group (n = 621) | N/A | Undergone any type of surgical treatment (e.g., partial gastrectomy, and total gastrectomy) and/or any chemotherapy regimen | Meta-analysis (fifteen RCTs) | Aid in gastrointestinal function recovery, reduce the incidence of AEs of surgery and chemotherapy, and improve immune function | [47] |
| Acupoint injection | Zusanli (ST36) acupoint injection with Neostigmine | ST36 acupoint injection with neostigmine 05 mg per side (n = 67) vs gluteal intramuscular injection with 1.0 mg neostigmine (n = 67) vs ST36 acupuncture alone (n = 67) vs standard therapy (n = 67) | Once a day until recovery of peristalsis | Patients with paralytic POI following gastrectomy for GC | RCT | Shorter time to bowel sound recovery, shorter time to first flatus and first defecation | [48] |
| Auricular point-pressing with bean | Acupoints of the stomach, spleen, large intestine, endocrine, subcortex, and small intestine | Auricular point-pressing with bean plus 20 mg EM tablets (n = 41) vs routine EM tablets (n = 37) | The therapist conducted compression once every 6 hours | Gastrointestinal dysfunction after ESD | Retrospective study | Alleviate the gastrointestinal dysfunction of early GC patients after ESD and help them to maintain normal gastrointestinal function | [57] |
| Spleen acupoint, large intestine acupoint, and gastrointestinal pain point | Acupoint patch combined with ear acupoint bean pressing burrowing (n = 41) vs conventional nursing methods (n = 41) | Postoperative until 1 day before patient discharge | After laparoscopic surgery for gastric cancer | Retrospective study | Better adapt to the treatment process, reduce anxiety, and improve the treatment effect and quality of life | [108] | |
| Auricular acupuncture | Shenmen, sympathetic nerve, thalamus, point zero, and omega 2 | Sham auricular acupuncture (n = 20) vs auricular acupuncture (n = 20) | 6 days | Postoperative movement-evoked pain after open radical gastrectomy | Single-blind randomized controlled pilot trial | Have a certain relief effect on mild postoperative pain at rest with pain score below 3 | [62] |
| Traditional Chinese medicine nursing | (1) TCM psychological care; (2) Syndrome differentiation nursing; (3) Dietetic nursing; and (4) Nursing of auricular-plaster therapy of TCM: Auricular points: Spleen, stomach, sympathy, endocrine, large and small intestines | TCM nursing (n = 52) vs routine nursing (n = 51) | Until the patient was discharged from hospital | Postoperative patients with GC after radical gastrectomy | Prospective, randomized controlled study | Improve postoperative gastrointestinal dysfunction, alleviate acute inflammation, improve postoperative unhealthy mental state, reduce the occurrence of postoperative complications | [58] |
| TCM formulae | Compounds/constituents | Functions | Clinical stage | Experimental model | Effect | Specific mechanisms (pathways/targets) | Ref. |
| Sijunzi decoction | Radix Glycyrrhiizae (Gancao), Radix Ginseng (Renshen), Poria cocos Schw. (Fuling) and Rhizoma Atractylodes (Baizhu) | Nourishing spleen and enrich qi | GC tumor recurrence and treatment resistance | In vitro (MKN74 cellsand MKN45 cells) | Anti-tumor | Inhibit the nuclear accumulation and DNA-binding of β-catenin | [14] |
| Fuzheng Huoxue anticancer prescription | Codonopsis pilosula (Franch.) N annf Astragalus membranaceus (Fisch.) Bge, Atractylodes macrocephala Koidz, Poria cocos (Schw.) Wolf, Rehmannia glutinosa (Gaertn.) Lib osch, Adenophoratetraphylla (Thunb.) Fisch, Salvia miltiorrhiza Bge, and Angelica sinensis (Oliv.) Diels | Replenish vital functions (Zhengqi), improve blood stasis and circulation | Mid- or late-stage GC | In vitro (SGC-7901). In vivo (female nude mice) | Correct a hypercoagulatory state, improve immunologic function, extend patient survival times, and inhibit gastric tumor growth | NS | [13] |
| SRRS | Atractylodes macrocephala koidz (Baizhu), Poria cocos Schw. (Fuling), Sargentodoxa cuneata (Oliv.) Rehd. Et Wils. (Daxueteng), Prunella vulgaris L. (Xiakucao) and etc | Nourishing spleen | Advanced GC | In vitro (SGC-7901). In vivo (female BALB/C-nu/nu mice) | Inhibition of gastric cancer cell growth | Decrease the expression of bcl-2 mRNA inducing apoptosis | [109] |
| Composed Chinese medicine of Huachansu | Arenobufagin (11.14%), bufalin (18.67%), bufotalin (7.33%), cinobufagin (16.67%), cinobufotalin (16.74%), gamabufotalin (8.45%), resibufogenin (12.03%), and telocinobufagin (8.97%) | Anticancer | Advanced GC | In vitro (SGC7901, MGC803 and NCM460) | Suppression of proteasome activities and increase of ROS levels | Regulating PI3K/AKT and MAPK signaling pathways | [110] |
| Xiaojianzhong decoction | cassia twigs, paeonia, roasted licorice, ginger, jujube, and maltose sugar | Nourishing spleen and stomach, anticancer | GC | In vitro (AGS, HGC-27, and GES-1) | Suppressing the viability of GC cells, inducing apoptosis, arresting these cells in the G0/G1 phase, and inhibiting cell clone formation | Inhibiting the expression of IL6, PTGS2, MMP9, MMP2, and CCL2 proteins and promoted the expression of the heme oxygenase-1 protein | [111] |
| Yiqi Huayu decoction | Huangqi, Dangshen, Chenpi, Banxia, Baizhu, Baishao, Danggui, Sanleng, Ezhu, Sheshecao, Shijianchuan, Fulin, Muxiang, Sharen, and Gancao | Invigorating qi and invigorating spleen and removing blood stasis | GC after stage II and III operations | In vitro (AGS) | Anti-recurrence and metastasis of GC | Affecting the JAK2-STAT3 pathway and the expression of ACSL4, and induction of ferroptosis | [35] |
| Weifuchun | Radix Ginseng Rubra (red ginseng), Rabdosia amethystoides H. Hara (Xiangchacai), and fried Fructus Aurantii (Zhike) | Strengthen the spleen and replenish qi, promote blood circulation and detoxification, and alleviate flatulence and phlegm | GC | In vitro (AGS, MKN45, and SGC7901) | Inhibiting proliferation, migration, and inducing apoptosis in GC cells | Targeting candidate genes (TNFa, IL6, VEGFA, NFKB, MAPK1, and BAX) | [26] |
| In vitro (NCI-N87, SNU-5, SNU-16, GES-1 cells, and Hs.738. St/Int cells) | Suppressing the malignant cellular phenotypes of GC cells | miR-26a-5p-mediated KPNA2 destabilization and the disruption of the MAPK pathway also enhances the repression of KPNA2 | [32] | ||||
| Daikenchuto | Zanthoxylum fruit (Huajiao), processed dried ginger (Ganjiang), ginseng (Renshen), and malt sugar (Maiyatang) | Stimulates intestinal motility | POI following abdominal surgery | In vivo (male BALB/c mice) | Recovery of the delayed intestinal transit | Inhibited the infiltration of neutrophils and CD68-positive macrophages, and inhibited mRNA expressions of TNF-α and MCP-1 may be partly mediated by activation of α7nAChR | [40] |
| Jianpi Yangzheng decoction | Astragali Radix (Huang Qi) (60 g), Codonopsis Radix (Dang Shen) (30 g), Sparganii Rhizoma (San Leng) (15 g), Curcumae Rhizoma (E Zhu) (15 g), Atractylodis Macrocephalae Rhizoma (Bai Zhu) (10 g), Angelicae Sinensis Radix (Dang Gui) (10 g), Paeoniae Alba Radix (Bai Shao) (10 g), Aucklandiae Radix (Mu Xiang) (10 g), Citri Reticulatae Pericarpium (Chen Pi) (10 g), and Glycyrrhizae Radix (Gan Cao) (5 g) | Anticancer, anti-metastasis | GC after chemotherapy | In vitro (MFC murine GC cell). In vivo (615-strain mice) | Impeding pre-metastatic niche formation and exhibiting anti-tumor metastasis effect | Reshaping the gut microbiota structure, enhancing SCFA production, and inhibiting the formation of the pre-metastatic microenvironment | [30] |
| In vitro (MKN28, AGS, HGC-27, MKN74, MKN45 and MFC). In vivo (male BALB/c-nude mice) | Inhibitory effect on GC growth and metastasis | Elevates the abundance of CLDN18.2 in gastric cancer, suppresses the activity of YAP/TAZ signaling | [31] | ||||
| In vitro (AGS, HGC -27). In vivo (BALB/c nude mice) | Inhibit the invasion and migration of GC | Regulating TAM-exos miR-513 b-5p mediated PTEN/AKT signaling | [29] | ||||
| Modified Jian-pi-yang-zheng decoction | - | Qi-invigorating, spleen-strengthening and stasis-removing | Advanced GC | In vitro (HGC-27 and THP-1 monocytes). In vivo (mice (615-strain) | Suppressed GC growth and metastasis | Inhibit GC cell EMT via PI3Kγ-dependent TAM reprogramming | [28] |
| Modified Gexia-Zhuyu Tang | Peach kernel (9 g), Safflower (9 g), Angelica sinensis (9 g), Chuanxiong (6 g), Red peony (6 g), Peony bark (6 g), YanhuSuo (3 g), Wulingzhi (6 g), Wuyao (6 g), Fructus Aurantii (4.5 g), Xiangfu (4.5 g), Licorice (9 g), Rhizoma atractylodis macrocephalae (12 g), Rhizoma curcumae (9 g), Hedyotis diffusa (15 g), and Scutellaria barbata (15 g) | Anticancer | GC | In vitro (MFC). In vivo (BALB/c male mice) | Inhibited GC tumor progression | Inhibits the growth, proliferation, metastasis, and invasion of GC by regulating the intestinal flora and promotes pyroptosis of GC | [43] |
| Xiaotan Sanjie recipe | Rhizoma Arisaematis, Pinellia Ternata, Atractylodes Macrocephala, Poria Cocos, Fritillaria sichuanensis, White Mustard Seed, Fructus aurantii, Pericarpium Citri Reticulatae, Radish Seed, Galli Gigerii Endothelium Corneum and Medicated Leaven (5:5:4:4:3:3:3:3:2:2:1) | Anti-metastasis | Advanced GC | In vitro (GC-7901, HGC-27, MKN-28, MKN-45, MGC80-3, BGC-823, MKN-7 and KAO-II). In vivo (Female BALB/c-nude mice) | Prevented GC metastasis | Inhibits the GnT-V-mediated E-cadherin glycosylation and promotes the E-cadherin accumulation at cell-cell junctions | [17] |
| In vitro (SGC-7901 cells and HUVEC) | Inhibit angiogenesis in GC | Inhibit angiogenesis in gastric cancer through IL-8-linked regulation of the VEGF pathway | [18] | ||||
| Banxia Xiexin decoction | Pinellia ternata (Thunb.) Makino 12 g, Zingiber officinale Roscoe 9 g, Scutellaria baicalensis Georgi 9 g, Coptis chinensis Franch. 3 g, Panax ginseng C.A.Mey. 9 g, Ziziphus jujuba Mill 12 g, and Glycyrrhiza uralensis Fisch 9 g | Protecting the gastrointestinal mucosa, improving gastrointestinal diseases, regulating human endocrine metabolism | Advanced GC | In vitro (AGS and GES-1) | Inhibits invasion, metastasis, and epithelial mesenchymal transition in GC | Inhibition of lncRNA TUC338 expression | [37] |
| Compound kushen injection | Kushen (Radix Sophorae Flavescentis) and Baituling (Rhizoma Heterosmilacis) | Anticancer, improve body immunity, relieve of cancer pain and bleeding | Advanced GC | In vitro (AGS, HGC-27, MKN-45, MKN-74, GES-1). In vivo (male mice) | Inhibit GC growth and metastasis, improve body's immunity, and protect normal tissues from damage | Regulating VCAM1 induced by the TNF signaling pathway to inhibit epithelial-mesenchymal transition of GC | [34] |
| In vitro (BGC-803/MKN-28) | Inhibited GC cell growth and migration and induced GC cell apoptosis | Regulating the EMT process in GC cells through the PI3K/AKT signalling pathway | [33] | ||||
| Yiwei decoction | Astragalus Membranaceus, Pinellia Ternate, Tetrastigma Hemsleyanum Diels et Gilg, Actinidia chinensis Planch, Ophiopogon Japonicus, Taraxacum mongolicum Hand, Paeonia lactiflora Pall, Coix lacryma-jobi L.var.ma-yuen (Roman.) Stapf, and Rabdosia amethstoides (Benth.) Hara | Anticancer, supplementing qi and strengthening the body | GC recurrence and metastasis | In vitro (HGC-27 , MFC, S180 cells). In vivo (Wistar rats) | Inhibit the proliferation of tumor cells | Inhibition of GC cell proliferation by spleen-derived exosomes inducing apoptosis | [11] |
| Ziyin Huatan recipe | Lilii Bulbus (Baihe.), Pinelliae Rhizoma (Fabanxia) and Hedyotis Diffusa (Baihuasheshecao) | Anti-metastasis | Advanced GC | In vitro (MGC-803 and SGC-790). In vivo (male BALB/c nude mice) | Inhibit migration and invasion of GC | Relate to the upregulation of RUNX3 expression | [19] |
| In vitro (HGC27 and MGC803). In vivo (male BALB/c nude mice) | Inhibit cell proliferation and promote apoptosis in GC | Decrease the expression of Bcl-2 and Cyclin D1, modulate PI3K/AKT signaling pathway by inhibiting PI3K expression | [20] | ||||
| Qi Ling decoction | Astragalus, Smilacisglabrae, Atractylodesmacrocephalae, Scutellariabarbata, Salvia chinensis, Benth, Eupolyphagasteleophaga, Agrimonia, and Forsythia suspense | Anti-metastasis, anti-tumor | Advanced GC | In vitro (BGC-823 and SGC-7901) | Inhibit the invasion, migration, and adhesion of GC cells | Inhibition of MMP-9 expression through the PI3K/AKT signaling pathway | [24] |
| Babao Dan | Natural bezoar, snake gall, antelope horn, pearl, musk, and Panax notoginseng | Anti-metastasis, anti-tumor | Advanced GC | In vitro (AGS and MGC80-3) | Inhibits the migration and invasion of GC cells | Inhibit TGF-β-induced EMT and inactivating TGF-β/ | [36] |
Surgical intervention may result in impaired vital energy and exacerbated splenic dysfunction due to deficiency of qi and blood, leading to diminished immune function and malnutrition[9]. Furthermore, a proportion of patients with GC are diagnosed at progressive or advanced stages, and these patients exhibit diminished physical function. Consequently, surgery or chemotherapy will aggravate immune impairment and adversely affect quality of life. The TCM concept of replenishing qi and blood implies to strengthen the patient’s physique and enhance the body’s resistance to diseases, related to the function of immune regulation of the spleen[11]. TCM increases the patient’s cellular immunity, relieves cancer-related fatigue and improves anorexia and cachexia improving the patient’s quality of life[12].
The Fuzheng Huoxue Anticancer Prescription has demonstrated the ability to restore vital functions (Zhengqi) and directly inhibit tumor growth in GC, resulting in prolonged patient survival. In a study of mid- to late-stage GC patients treated with this formula, a significant improvement in the immunological function of T lymphocytes was observed in the treatment group (P < 0.01), indicating improved immune function[13]. Sijunzi decoction, which consists of Radix Codonopsis (Dangshen), Poria cocos (Fuling), Rhizoma Atractylodis Macrocephalae (Baizhu), and Radix Glycyrrhizae (Gancao), is a TCM formula recognized for nourishing the spleen and enriching qi. A meta-analysis of 10 RCTs showed that GC patients who received Sijunzi decoction along with enteral nutrition had significantly higher levels of albumin, prealbumin, transferrin, immunoglobulins (IgG, IgA, IgM), and CD3+ T cells, thereby enhancing their immune response[9]. GC stem cells (MKN74 and MKN45) were employed to evaluate the effect and mechanism of Sijunzi decoction in repressing GC cell growth and inducing apoptosis. The results indicated that the mechanism underlying this effect involves the inhibition of β-catenin transcriptional activity[14]. Further, Zhipu Liujunzi Decoction, an improved version of Sijunzi Decoction, was administered to patients after radical gastrectomy and chemotherapy for two weeks, resulting in significantly higher objective remission and disease control rates (P < 0.05) and increased levels of immune parameters (P < 0.001)[15].
The spleen is the largest secondary lymphoid organ in the body and has a wide range of immune functions. Yiwei decoction-treated spleen-derived exosomes inhibited the proliferation of MFC cells, S180 cells and HGC-27 cells in vitro, and induced apoptosis of HGC-27 cells. The cell counting kit-8 assay confirmed that the relative tumor inhibition rate of the Yiwei decoction-treated spleen-derived exosomes was 70.78% in comparison to the control exomes[11].
Shi and Wei[16] proposed that GC is closely associated with phlegm and developed a phlegm theory of GC. According to this theory, phlegm stagnation consists of a phlegm core, phlegm collateral, and phlegm contamination, which together explain the mechanisms underlying the development, recurrence, and metastasis of GC. Xiaotan Sanjie recipe is an empirical compound prescription is modified from the classic Dao-Tan decoction for eliminating phlegm. Following the administration of the Xiaotan Sanjie recipe, there was a marked decrease in the migration and invasion abilities of GC cells. Both in vivo and in vitro investigations indicate that the regulation of E-cadherin accumulation at cell-cell junctions through the GnT-V/b1,6 GlcNAc-E-cadherin axis may be a key mechanism involved[17]. Additionally, another study suggested that the Xiaotan Sanjie recipe could potentially inhibit angiogenesis in GC by suppressing interleukin (IL)-8-induced expressions of vascular endothelial growth factor (VEGF)-A and VEGF receptor-1 proteins[18].
The Ziyin Huatan recipe, comprising Lilii Bulbus, Pinelliae Rhizoma, and Hedyotis Diffusa, was developed based on the hypothesis of “tumor-phlegm microenvironment”, has demonstrated significant efficacy in enhancing disease-free survival (DFS) in GC patients following surgical intervention in combination with chemotherapy. The recipe has been shown to not only reduce the likelihood of recurrence and metastasis but also effectively prolong overall survival[19]. In vitro studies suggest that the Ziyin Huatan Recipe can impede the proliferation, migration, and invasion of GC cells. Furthermore, in vivo experiments have demonstrated that it can reduce the metastatic spread of GC cells to the lungs and increase the survival time of nude mice. The underlying molecular mechanism appears to involve the upregulation of RUNX3 expression and the inhibition of cell proliferation and promotion of apoptosis in GC by suppressing the phosphatidylinositol 3-kinase (PI3K)/protein kinase B (AKT) pathway[19,20].
To eliminate stasis and cancerous toxins, the Yiqi Huayu Jiedu decoction was developed as a treatment for GC. A multi-center study conducted over nearly eight years demonstrated that this formulation, when used in conjunction with chemotherapy, significantly reduced the risk of recurrence and metastasis in patients with advanced GC, especially those with stage III disease[21]. In addition, a case report of a Billroth II gastrectomy for GC highlighted the efficacy of the San Jie Pai Shi decoction, which has functions such as strengthening the spleen, regulating the stomach and resolving phlegm imbalances. This decoction successfully dissolved a gastric bezoar within two weeks[22].
Qi Ling decoction is a TCM formula composed of eight herbs, which is predicated on the principle of “detoxification” according to the cancer toxicity theory in TCM oncology. The primary active components of this decoction include diosgenin, catechins, and calycosin, all of which have been shown to reduce both the rate of closure and the number of migrated cells, thereby inhibiting the invasion and migration of BGC-823 cells. Furthermore, diosgenin and catechin have been observed to impede the adhesion of BGC-823 cells. The PI3K/AKT axis plays a role in metastasis and tumor invasion by activating nuclear factor kappa B-mediated matrix metalloproteinase (MMP)-9[23]. The in vitro observations of Qi Ling decoction’s impact on cell migration, invasion, and adhesion may be attributable to its ability to suppress MMP-9 expression, via the PI3K/AKT signaling pathway[24].
Some herbal formulas have been recommended for long-term use after GC surgery to prevent metastasis and recurrence to increase long-term survival, e.g. Jianpi Yangzheng decoction[25], Yiqi Huayu Jiedu decoction[21], Weifuchun[26] and a powder of eight noble ingredients and cinnamon twig and Poria pills[27] can reduce recurrence and/or metastasis rates when used for 6 months in clinical trials.
Jianpi Yangzheng decoction is a TCM formula for the treatment of advanced GC, which plays the role of promoting qi, invigorating the spleen, and removing stasis according to TCM theory[28]. A clinical study indicated that TCM nursing interventions incorporating oral administration of the Jianpi Yangzheng decoction offer benefits in enhancing postoperative gastrointestinal function, metabolic status, and immune activity, while also reducing postoperative complications and inflammation, while concomitantly improving survival rates in postoperative patients with GC[25]. The inhibitory effect of Jianpi Yangzheng decoction on GC growth and metastasis, along with its underlying mechanisms, has been explored through various in vivo and in vitro studies. The Jianpi Yangzheng decoction has demonstrated antitumor properties by decreasing the levels of miR-513b-5p in tumor-associated macrophage exosome, thereby alleviating the activation of the phosphatase and tensin homolog/AKT signaling pathway and influencing the progression of GC[29]. The complex characterization of the gut microbial community plays a key role as a potential biomarker and response predictor in gastrointestinal cancer therapy, and some TCM therapies work against tumors by modifying gut microbiota homeostasis. The Jianpi Yangzheng decoction has been shown to target the gut microbiota, enhance the production of short-chain fatty acids, and modulate immune activity, thereby inhibiting GC growth and metastasis[30,31]. Additionally, it has been observed to prevent the accumulation of myeloid-derived suppressor cells in lung tissues, thereby blocking the excessive production of inflammatory factors in serum and lung tissues. In vitro studies suggest that Jianpi Yangzheng decoction may inhibit the formation of an immunosuppressive microenvironment and prevent GC metastasis to the lungs[30]. Moreover, the Modified Jianpi Yangzheng decoction inhibited GC cell epithelial-mesenchymal transition (EMT) via PI3Kγ-dependent macrophages reprogramming and eventually suppressed GC growth and metastasis[28].
Weifuchun is composed of Radix Ginseng Rubra, Rabdosia amethystoides H. Hara, and fried Fructus Aurantii. Clinical experience in TCM suggests that its functions include strengthening the spleen and replenishing qi, promoting blood circulation and detoxification, as well as alleviating flatulence and phlegm[26]. Through network pharmacology and GC cell experiments, we discovered that Weifuchun exhibits antitumor effects by targeting candidate genes such as tumor necrosis factor α (TNF-α), IL-6, vascular endothelial growth factor A, nuclear factor kappa-light-chain-enhancer of activated B cells, mitogen-activated protein kinase (MAPK) 1, and Bcl-2-associated X protein. These effects involve the AMP-activated protein kinase-dependent extracellular signal-regulated kinases 1/2 and nuclear factor kappa-light-chain-enhancer of activated B cells pathways, which regulate proliferation, migration, and apoptosis in GC cells[26]. In another in vitro study, Weifuchun was found to suppress the malignant cellular phenotypes of GC cells by reducing the levels of karyopherin alpha 2[32].
Compound kushen injection (CKI), derived from Kushen (Radix Sophorae Flavescentis) and Baituling (Rhizoma Heterosmilacis), has demonstrated significant effects on cancer and tumors. Research indicates that CKI may inhibit the growth and migration of GC cells while inducing apoptosis in these cells. Furthermore, CKI regulates the EMT process in GC cells via the PI3K/AKT signaling pathway[33]. Another study has confirmed that CKI effectively suppresses the growth and metastasis of GC, with its molecular mechanism involving the downregulation of the TNF signaling pathway, which in turn inhibits the expression of vascular cell adhesion molecule 1 and suppresses EMT in GC[34].
Yiqi Huayu decoction can induce ferroptosis in GC by affecting the Janus kinase 2 (JAK2)-signal transducer and activator of transcription 3 (STAT3) pathway and the expression of Acyl-CoA synthetase long-chain family member 4 which may be one of the possible mechanisms of its anti-recurrence and metastasis of GC[35]. Babao Dan inhibited the viability, migration, and invasion of AGS cells and MGC80-3 cells, by inhibiting transforming growth factor β-induced EMT and deactivating the transforming growth factor β/Smad signaling pathway[36]. Banxia Xiexin Decoction may serve as a potential inhibitor of TUC338, demonstrating a dose-dependent reduction of long non-coding RNA TUC338 expression in AGS cells, which would reduce the invasion and migration of GC cells. Silencing TUC338 effectively inhibited the activation of the PI3K/AKT signaling pathway, resulting in reduced tumor proliferation and migration[37].
Several clinical studies have focused on the perioperative management of GC, particularly emphasizing the role of TCM in preventing and treating postoperative complications. TCM has been shown to have a positive impact on postoperative care by reducing the incidence of infection and promoting wound healing. A meta-analysis of 32 RCTs evaluating various TCM therapies, including decoction, moxibustion therapy, and auricular plaster therapy, demonstrated their potential to reduce the risk of postoperative wound infection and accelerate the wound healing process[38]. In addition, postoperative gastroparesis syndrome, also known as gastric atony or delayed gastric emptying, is a common postoperative complication. In patients who have undergone pylorus-preserving gastrectomy, Rikkunshito (Liujunzi Decoction) has been shown to improve gastric emptying and alleviate postoperative symptoms[39]. Daikenchuto, derived from the Dajianzhong decoction in TCM, promotes intestinal motility by inhibiting the infiltration of neutrophils and CD68-positive macrophages. Additionally, it reduces the mRNA expression of TNF-α and monocyte chemoattractant protein-1, a process that may be partially mediated by the activation of the α7 nicotinic acetylcholine receptor[40].
Another issue following GC surgery is the potential for high hypercoagulable state, which can increase the risk of deep vein thrombosis. Laparoscopic GC surgery typically has a longer operative time compared to open surgery, resulting in persistent postoperative elevations in fibrinogen and D-dimer levels, while prothrombin time is significantly shortened, potentially leading to venous stasis[41]. Research has shown that five days after laparoscopic GC surgery, both the Xuesaitong injection group and the control group showed a continuous decrease in fibrinolysis indices (fibrinogen, D-dimer). However, patients who received Xuesaitong injection immediately postoperatively showed a greater reduction and lower incidence of deep vein thrombosis, with statistically significant differences between the two groups[41]. In a small-sample Crossover design study, the use of Daikenchuto not only reduced stasis-related symptoms, but also accelerated gastric emptying, increased intestinal motility, and decreased postoperative symptoms[42]. Gexia-Zhuyu Tang was utilized to treat blood stasis. Modified Gexia-Zhuyu Tang facilitated the expression of pro-inflammatory factors TNF-α, IL-1β, and IL-18 by modulating gut microbiota, thereby promoting caspase-1-dependent pyroptosis, an inflammatory form of programmed cell death, which in turn inhibited the progression of GC[43].
TCM external use, exemplified by acupuncture, have shown efficacy in pain control and the improvement of gas
Postoperative ileus (POI) is a prevalent postoperative complication following GC surgery that can adversely affect patient recovery and prolong hospital stays. The etiology is multifactorial, including surgical interventions, such as vagus nerve damage and physiological structural changes, as well as CO2 pneumoperitoneum, which is caused by ischemia due to intra-abdominal hypertension. These factors can trigger a molecular inflammatory response in the intestinal tract, resulting in the recruitment of leukocytes and other inflammatory cells to the intestinal muscular layer. These inflammatory cells express inducible nitric oxide synthase, leading to the production of nitric oxide and subsequent gas
Rest pain and exercise-induced pain that occurs after tumor resection and lymph node dissection, if inadequately treated, may lead to poor outcomes by preventing patients from participating in rehabilitation programs and delaying discharge and recovery. However, postoperative pain and opioid use will further prolong the recovery of gastrointestinal function[49]. Acupuncture therapy in TCM has potential for postoperative pain control. A clinical study exploring auricular acupuncture for the treatment of postoperative pain after open radical gastrectomy in patients with GC demonstrated that auricular acupuncture provided relief of mild resting pain with a postoperative Numerical Rating Scale for pain score of 3 or less[62]. Pain, inflammation and immunity are interrelated and complementary. After a patient suffers a surgical injury, the body releases a variety of injurious mediators that cause pain. The varying degrees of stress induced by surgery, anesthesia and pain will further lead to immune suppression. The analgesic and anti-inflammatory effects of acupuncture on inflammatory pain are a complex process involving the peripheral and central nervous system, and the main research hotspots are opioid peptides system, ephedrine and its receptors, and Purinergic signal, etc.[63]. IL-2 plays an important role in promoting the maturation of lymphocytes and regulating immunity. EA, a combination of traditional acupuncture and modern electrotherapy, promotes IL-2 production, attenuates inflammatory factors, and reduces the degree of immunosuppression[64].
Naringin, derived from the peel and pulp of Citrus grandis, Citrus paradise, and orange (Zhike, Chenpi), has been investigated for its role in inhibiting GC development through both cellular and animal studies. EMT is the process by which epithelial cells transform into mesenchymal cells in response to certain stimuli, thereby enabling tumour cells to become invasive and metastatic[65]. Naringin has been shown to arrest the cell cycle in the G0/G1 phase, induce apoptosis in cancer cells, and inhibit the EMT by targeting the PI3K-AKT/zinc finger E-box binding homeobox 1 pathway in GC cells[66].
Pachymic acid and Poria acid are natural triterpenoid compounds derived from the TCM herb Poria cocos, exhibiting significant biological activity against GC. Pachymic acid induces ferroptosis and inhibits GC progression by suppressing the platelet-derived growth factor receptor beta-mediated PI3K/AKT pathway, thereby demonstrating its antitumor properties[67]. After treatment with Poria acid, both the proliferation and adhesion of GC cells were significantly reduced, as evidenced by thiazolyl blue (MTT) assay and cell adhesion experiments, respectively. The mechanism of Poria acid against GC lies in its ability to inhibit the expression of EMT and metastasis-associated proteins (e.g., MMPs) in GC cells[68].
Cimifugin, derived from Cimicifugae Rhizoma (Shengma), demonstrates promising effects on GC, suggesting its potential for clinical application. A study evaluated the inhibitory effects of cimifugin on proliferation, invasion, and migration in MKN28 cells. Through network pharmacology and cellular molecular biology experiments, it was found that cimifugin may influence various metabolic pathways in GC, showing moderate binding affinities to three key targets: Aldo-keto reductase family 1 member C2, monoamine oxidase B, and phosphodiesterase 2A[69].
Plumbagin, obtained from the roots of Plumbago zeylanica, is a medicinal plant recognized for its extensive therapeutic properties, including antimicrobial, hepatoprotective, anticancer, antifertility, antiulcer, and wound healing effects. An In vitro study utilizing the human GC cell line SGC-7901 have demonstrated that Plumbagin exerts its anti-apoptotic effects by modulating the expression of various genes associated with cell proliferation and survival. The study found that concentrations ranging from 5 mmol/L to 20 mmol/L significantly reduced the viability of GC cells, which may be attributed to its ability to suppress the phosphorylation of STAT3 and AKT[70].
Triptonoterpene, an active compound extracted from Celastrus orbiculatus Thunb., exhibits significant anti-GC effects. It reduces GC cell proliferation, as evidenced by cell counting kit-8 assays and colony formation experiments. Additionally, Triptonoterpene inhibits the invasion and migration of GC cells by modulating proteins associated with EMT and MMPs[71]. Additionally, both in vitro and in vivo studies have demonstrated that the extract of Celastrus orbiculatus Thunb. enhances CFL1 degradation, thereby disrupting actin cytoskeleton remodeling in GC cells, which ultimately impedes EMT progression and suppresses metastasis[72].
Salvia miltiorrhiza Bunge, known as Danshen in TCM, is recognized for its ability to activate blood circulation and alleviate blood stasis. Diterpenoid tanshinones are extracted from Danshen. Both in vitro and in vivo studies have explored its anti-GC effects, measuring changes in the expression of angiogenesis-related factors. It was found that diterpenoid tanshinones inhibit tumor growth and angiogenesis by modulating the expression of the angiogenic factor VEGF via the PI3K/AKT/mTOR pathway[73].
Another TCM herb extract, Huaier (Trametes robiniophila Murr.), has been suggested in some studies to potentially inhibit the growth and metastasis of GC, especially in cases of liver metastasis, while also showing a lack of significant toxicity. An overview of systematic reviews and meta-analyses reported that Huaier granules, when combined with conventional chemotherapy, significantly reduced the incidence of common adverse events - including gastrointestinal reactions, myelosuppression, hepatotoxicity, and nausea and vomiting - compared to chemotherapy alone[74]. It is important to note, however, that the authors of that overview graded the quality of evidence for these safety outcomes as “moderate” to “low”, indicating that while the results are promising, further high-quality research is needed to solidify these findings. Mechanistically, Huaier demonstrates a range of anti-cancer effects, including anti-proliferation, anti-angiogenesis, and the induction of apoptosis[75]. More specifically, its action against GC has been linked to Syntenin, a protein whose high expression correlates with metastasis and poor prognosis. The mechanism of action of Huaier n-butanol extract involves the inhibition of the syntenin/STAT3 signaling pathway and the reversal of EMT[76].
Berberine, the primary active ingredient in Rhizoma coptidis, has been shown to possess anti-inflammatory, anti-tumor, and hypoglycemic properties. It has been demonstrated that berberine inhibits the progression and development of GC by modulating the IL-6/JAK2/STAT3 signaling pathway. In vitro studies have shown that berberine significantly promotes apoptosis in GC cells and induces G0/G1 phase cell cycle arrest. In addition, in a mouse model of human GC, berberine has been shown to effectively suppress tumor growth and reduce IL-6 levels[77].
Paeonol, a natural bioactive compound extracted from Paeonia suffruticosa Andr., has been shown to inhibit the malignancy of GC cells. In an in vivo study using a tumor-bearing mouse model of GC, intragastric administration of paeonol resulted in a reduction of tumor growth. Furthermore, cell cycle analysis indicated a decreased proportion of cells in the G0/G1 phase[78]. Beyond these direct anticancer effects, paeonol holds significant promise as an adjuvant therapy due to its dual beneficial actions: It exhibits synergistic effects with various traditional anticancer drugs and, crucially, protects vital organs from the toxic side effects of chemoradiotherapy. This selectivity is particularly note
Brusatol, a balsam lactone compound isolated from the traditional Chinese herb Brucea javanica, is known to have antitumor properties. It has been observed to induce an EMT in response to lipopolysaccharide through the deactivation of the PI3K/AKT/nuclear factor kappa B signaling pathway. In addition, brusatol has been shown to promote an accumulation of reactive oxygen species, resulting in the inhibition of GC cell growth and the induction of caspase-dependent apoptosis in vitro[83].
β-Elemene, a sesquiterpene derived from the TCM herb Curcuma Wenyujin, exhibits a wide spectrum of therapeutic effects, including anti-inflammatory and anti-tumour properties, with low toxicity levels[84]. It has been categorized as a Class II anti-tumour drug due to its significant anti-tumour efficacy and favourable safety profile[85]. As a novel anticancer drug, it has demonstrated significant potential, exhibiting a broad-spectrum of activity and an enhanced capacity to combat metastasis in cases of multidrug-resistant GC. A study conducted in vitro and confirmed by immunohistochemistry using lung tissues from mice demonstrated that β-Elemene inhibits the metastasis of multidrug-resistant GC cells by modulating the microRNA-1323/casitas B-lineage lymphoma b/epidermal growth factor receptor signaling pathway[86]. Furthermore, β-Elemene has been identified as an effective agent against peritoneal metastasis, a common cause of poor prognosis following GC surgery. In a nude mouse model of GC peritoneal metastasis, intraperitoneal injection of β-Elemene significantly reduced the formation of diffuse peritoneal tumours and metastatic nodules in abdominal organs. This effect is associated with its ability to decrease the expression of the tight junction protein claudin-1 and down-regulate phosphorylated focal adhesion kinase[87].
Cantharidin, a terpenoid derived from the dried bodies of highly toxic beetles in the family Meloidae, has been shown to have antineoplastic properties, including the ability to inhibit the growth of various tumor cell lines. Cancer-associated transcript 1, which has a high expression level in GC, has been found to be closely related to clinicopathological features such as tumor-node-metastasis stage, lymph node metastasis, and distant metastasis[88]. Experimental cell studies have shown Ethat cantharidin induces apoptosis in GC cells and inhibits metastasis by suppressing the PI3K/AKT signaling pathway through the downregulation of cancer-associated transcript 1[89]. Despite its origin from highly toxic insects, clinical studies with its derivative, Cantharidin sodium (e.g., Qinin®), combined with chemotherapy for GC patients, suggest a favorable safety profile. This combination not only enhances clinical benefits but also significantly reduces common chemotherapy-induced side effects like gastrointestinal reactions and leukopenia. Treatment-related toxicities are generally reversible, with no serious adverse events leading to treatment discontinuation or death[90]. However, large-scale studies remain lacking.
Resveratrol, a natural polyphenolic phytoalexin, can be extracted, isolated, and purified from sources such as the skin of grapes (Vitis vinifera Linn.) and peanuts (Arachis hypogaea Linn.), or it can be synthesized artificially. This compound is known for its potential health benefits, particularly its multiple anticancer properties, which include the prevention of tumor formation across various cancer types. Metastasis-associated lung adenocarcinoma transcript 1 (MALAT1) has emerged as a promising biomarker for distant metastasis of GC, with high expression levels of MALAT1 being independently associated with poor prognosis in GC patients[91]. Resveratrol has been shown to inhibit cell migration and invasion by suppressing MALAT1-mediated EMT in the BGC823 GC cell line[92]. IL-6, a pivotal inflammatory cytokine, has been demonstrated to stimulate cancer cell invasion and is associated with cancer development. Resveratrol has been shown to impede IL-6-induced invasion by obstructing the Raf-MAPK signaling pathway[93].
N-butylidenephthalide is a bioactive compound isolated from the extract of Radix Angelica Sinensis (Danggui) which is widely used in TCM prescriptions in China. According to data from Taiwan’s National Health Insurance Research Database, GC patients treated with Danggui exhibited a notable decline in mortality, with an adjusted hazard ratio of 0.72 [95% confidence interval (CI): 0.57-0.92; P = 0.009]. The underlying mechanisms of action of N-butylidenephthalide include the inhibition of the proliferation of GC cells and the induction of mitochondrial-dependent apoptosis, leading to the expression of regulated in development and DNA damage response 1 and the activation of the mTOR signaling pathway. Additionally, it regulates EMT, thereby inhibiting the migration and invasion of GC cells. Furthermore, N-butylidenephthalide suppresses the growth of AGS cell xenograft tumors in NOD-SCID mice[94].
Venenum Bufonis (Chansu), derived from the skin and parotid venom glands of toads, contains the bioactive polyhydroxysteroid bufalin. Research has demonstrated that, although bufalin is less effective than oxaliplatin, it significantly reduces the invasion and metastatic potential of GC cells following treatment. The in vivo study demonstrated that bufalin inhibited the growth of subcutaneously transplanted tumor cells in the nude mouse model. The mechanism underlying bufalin’s anti-cancer activity may involve the down-regulation of the Wnt/β-catenin signaling pathway, which is fol
Ginsenoside Rg3, derived from the traditional Chinese herb ginseng, has been found to inhibit cancer cell proliferation and induce apoptosis. In an orthotopic mouse model of GC assessed through fluorescence imaging, the Ginsenoside Rg3 group demonstrated a significant inhibition of primary tumor growth and a marked reduction in lymph node metastasis compared to the vehicle control group[96].
Table 3 summarizes the efficacy and specific mechanisms of single Chinese herbal medicines and their components in the treatment of GC.
| Herb | Active component/extract | Functions | Clinical stage | Experimental model | Effect | Specific mechanism | Ref. |
| Peel and pulp of citrus grandis, citrus paradise and orange (Zhike, Chenpi) | Naringin | Anti-tumor | GC | In vitro (MGC803 and MKN45 GC cells). In vivo (BALB/c female nude mice) | Block the cell-cycle of GC cells | Induce cancer cell apoptosis, and inhibit the epithelial mesenchymal transition process by inhibiting the PI3K-AKT/Zeb1 pathway | [66] |
| Poria cocos (Fuling) | Pachymic acid | Anti-tumor | GC | In vitro (SGC-7901, AGS, and GES-1). In vivo (BALB/c nude mice) | Ferroptosis in GC cells | Suppressing the PDGFRB-mediated PI3K/AKT pathway | [67] |
| Poria acid | In vitro (AGS and MKN-28) | Inhibit the invasion and metastasis of GC cells | Inhibit the EMT process and expressions of metastasis-related proteins matrix metalloproteinase in GC cells | [68] | |||
| Cimicifugae Rhizoma (Shengma) | Cimifugin | Anti-tumor | GC | In vitro (MKN28) | Inhibits GC cell proliferation, invasion, and migration | Interacts with AKR1C2, MAOB and PDE2A to modulate various metabolic pathways | [69] |
| the roots of Plumbago zeylanica (Banlangen) | Plumbagin | Anti-tumor | GC | In vitro (SGC-7901 cells) | Inhibits cell apoptosis in human GC cells | Suppressed the expression of BAX, BCL-2, pro-caspase-3, and cleaved-caspase-3; restrained the expression and phosphorylation of STAT3 and decreased the phosphorylation of AKT1 | [70] |
| Eremias multiocellata | N/A | Removing blood stasis, eliminating nodules | Cisplatin-resistant GC | In vitro (MKN45/DDP). In vivo (BALB/c nude mice) | Reversing DDP resistance and increasing the sensitivity of gastric cancer drug-resistant cells to DDP | Regulating the NF-κB/Snail signaling pathway, PI3K/AKT/mTOR signaling pathway, and the expression of drug resistance-related proteins and genes | [112] |
| Salvia miltiorrhiza Bunge (Danshen) | Diterpenoid tanshinones | Activating blood circulation and removing blood stasis | GC | In vitro (SGC-7901, HUVECs). In vivo (BALB/c-nu nude mice) | Inhibit tumor growth and angiogenesis | Affecting the expression of the angiogenic factor VEGF through the PI3K/AKT/mTOR pathway | [73] |
| Celastrus orbiculatus Thunb (Leigongteng) | Triptonoterpene | Anti-metastasis, anti-tumor | Advanced GC | In vitro (BGC-823, MKN-28) | Inhibit the migration and invasion of GC cells | Inhibit the changes in EMT-related and invasion and metastasis-related proteins | [71] |
| In vitro (BGC-823 and AGS). In vivo (nude mice) | Inhibit the metastasis and EMT of GC cells | Inhibited the remodeling of the actin skeleton of gastriccancer cells by promoting the degradation of CFL1 | [72] | ||||
| Trametes robiniophila Murr. (Huaier) | Huaier n-butanol extract | Anti-metastasis, anti-tumor | Advanced GC | In vitro (GES-1, MGC803, MKN74, AZ-521, and MKN28). In vivo (nude mice) | Inhibits GC growth and hepatic metastasis | Inhibiting the syntenin/STAT3 signaling pathway and reversing EMT | [76] |
| Rhizoma coptidis (Chaihu) | Berberine | Anti-inflammatory, anti-tumor | Advanced GC | In vitro (MKN-45 and HGC-27 and GES-1). In vivo (male BALB/C nude mice) | Inhibits GC cell proliferation | Modulating the signaling pathways related to IL-6/JAK2/STAT3 | [77] |
| Paeonia suffruticosa Andr. (Mudanpi) | Paeonol | Anti-tumor, anti-inflammation, immune regulation | Apatinib-resistant GC | In vitro (BGC-823 and MGC-80). In vivo (female BALB/C nude mice) | Inhibit the malignancy of Apatinib-resistant GC cells | Regulating the LINC00665/miR-665/MAPK1 axis | [81] |
| Brucea javanica (Yadanzi) | Brusatol | Antitumor | Advanced GC | In vitro (SGC-7901) | Suppress GC cell proliferation, migration, invasion, and EMT formation | Inhibit LPS-induced EMT via the deactivation of the PI3K/AKT/NF-кB signaling pathway | [83] |
| Curcuma (wenyujin) | β-Elemene | Anti-metastasis, anti-tumor | Multidrug resistant GC | In vitro (SGC7901, SGC7901/ADR). In vivo (male BALB/C nude mice) | Inhibits the metastasis of multidrug resistant GC cells | Modulating the miR-1323/Cbl-b/EGFR signaling axis | [86] |
| Highly toxic beetles of the family Meloidae | Cantharidin | Anti-metastasis, anti-tumor | Advanced GC | In vitro (MGC803 and BGC823) | Suppress GC cell growth and migration/invasion | Inhibited the activation of the PI3K/AKT signaling pathway by downregulating CCAT1 targeting | [89] |
| Arachis hypogaea Linn. and Vitis vinifera Linn. | Resveratrol | Anti-metastasis, anti-tumor | Advanced GC | In vitro (SGC7901, BGC823 and GES1) | Suppress the invasion and migration of human GC cells | Inhibition of MALAT1-mediated epithelial-to-mesenchymal transition | [92] |
| Interleukin-6 induced GC metastasis | In vitro (SGC-7901). In vivo (male NOD/SCID mice) | Prevent the IL-6 induced GC metastasis | Block the IL-6 induced invasion through the blocking of Raf-MAPK signaling pathway | [93] | |||
| Radix Angelica Sinensis (danggui) | N-butylidenephthalide | Anti-metastasis, anti-tumor | Advanced GC | In vitro (AGS, NCI-N87 and TSGH-9201). In vivo (NOD-SCID mice) | Inhibit proliferation and induces mitochondrial-dependent apoptosis in GC cells | Activation of mitochondria-intrinsic pathway and induced the REDD1 expression leading to mTOR signal pathway inhibition in GC cells | [94] |
| Venenum Bufonis (Chan Su) | Bufalin | Anti-invasion, anti-metastasis | Advanced GC | In vitro (AGS). In vivo (male BALB/c mice) | Arrests GC invasion and metastasis | Down-regulation of the Wnt/β-catenin signaling pathway, followed by the inhibition of ASCL2 expression and EMT | [95] |
| Ginseng | Ginsenoside Rg3 | Anti-metastasis, anti-tumor | Metastatic GC | In vitro (NUGC-4). In vivo (female BALB/c mice) | Inhibite tumor growth and reduced lymphatic metastasis | Suppress expression of VEGF-C, VEGF-D and VEGFR-3 | [96] |
To explore the potential of TCM in future cancer treatment, we summarize the current clinical research status of TCM-based GC therapies (Table 4). Among the ongoing studies, the Phase 4 trial of Huaier granules (NCT05498766) is noteworthy for its potential, employing a prospective, multi-center, open-label, randomized controlled design. This study has enrolled 702 participants from 30 research centers who have undergone radical surgery (R0, D2 resection, with more than 16 lymph nodes detected) for gastric adenocarcinoma. The objective of this study is to evaluate the efficacy and safety of Huaier granules as postoperative adjuvant therapy, with the primary outcome being the 3-year DFS rate. Additionally, another prospective, multicenter, randomized, open-label trial (NCT03607656) is the first clinical investigation assessing the effects of a TCM collaborative model on DFS and quality of life in patients with stage IIIb and IIIc GC undergoing curative D2 gastrectomy[97]. Among the ongoing projects, four studies focus on acupuncture or EA (registration numbers NCT03753399, NCT02480361, NCT04467528, and NCT03291574) to evaluate the effects of acupuncture on gastrointestinal function recovery post-GC surgery and its underlying mechanisms. These ongoing trials may provide a deeper understanding of the safety and efficacy of TCM in adjunctive GC treatment, reflecting the growing interest and recognition of the potential of TCM in cancer therapy.
| TCM | Compounds/constituents | Condition | Intervention | Study design | Time frame | Phase | Status | Primary outcome | Secondary outcomes | NCT number |
| TCM collaborative model | Radix Pseudostellariae (12 g), Rhizoma Atractylodis Macrocephalae (12 g), Poria (30 g), Rhizome Pinelliae Preparata (9 g), green tangerine peel (4.5 g), Concha Ostreae (30 g), and Prunella vulgaris (9 g) | Stage IIIb and IIIc gastric cancer who undergo radical surgery for D2 lymphadenectomy | Adjuvant chemotherapy (oxaliplatin plus capecitabine or S-1; or docetaxel plus S-1; docetaxel plus oxaliplatin plus 5-FU) (n = 130) vs adjuvant chemotherapy + TCM formula + auricular acupressure and acupoint therapy (n = 130) | Prospective, multicenter, randomized, open-label trial | 36 months | Phase 2, phase 3 | Completed | 3-year DFS rate | Quality of life, side effects caused by chemotherapy, and safety-related measures | NCT03607656 |
| Yiqi Wenyang Jiedu prescription | Astragalus membranaceus (30 g), Codonopsis pilosula (15 g), Angelica dahurica (10 g), Curcuma zedoary (9 g), Rhizoma nardostachyos (10 g), Polygonum cuspidatum (10 g), Radix Actinidiae chinensis (15 g), and Paris polyphylla (9 g) | Completed adjuvant chemotherapy within 8 months of radical gastrectomy | YWJP (n = 106) vs YWJP placebo (n = 106) | Multicenter, randomized, double-blind, placebo-parallel-controlled clinical trial | 24 weeks | Phase 4 | Unknown | DFS rate 2 years after surgery | DFS time, overall survival, annual cumulative recurrence and rate of metastasis after 1-3 years, cumulative annual survival after 1-3 years, fat distribution-related indicators, tumor markers, peripheral blood inflammatory indicators, prognostic nutritional index, symptoms and quality of life evaluation, medication compliance, and adverse reaction rate | NCT05229809 |
| Hou Gu Mi Xi (modified Shen Ling Bai Zhu San) | Ginseng, tuckahoe, baked licorice, coixenolide, Chinese yam, lotus seed, shrinkage fructus amomi, white hyacinth bean, dried orange peel, and perilla leaf | Spleen qi deficiency status in GC patients after radical gastrectomy | Dietary supplement: Hou Gu Mi Xi (n = 65) vs Other: Placebo (n = 65) | Multicenter, randomized, double-blinded, parallel-group, placebo-controlled trial | 104 weeks | N/A | Terminated (high attrition rate) | Spleen Qi deficiency symptoms grading and quantifying scale | Quality of life assessed by the Short Form 36 scale, performance status as assessed by the Eastern Cooperative Oncology Group Performance Status scale, body weight, and body mass index | NCT03025152 |
| Modified Banxia Xiexin decoction | N/A | Stage IV GC | Modified Banxia Xiexin decoction combined chemotherapy (n = 73) vs placebo granules combined chemotherapy (n = 73) | Randomized clinical trial | 18 weeks | Early phase 1 | Completed | Overall survival | Progression-free survival, solid tumor efficacy, TCM syndrome score, quality of life score, tumor markers, Immune function and adverse reactions | NCT05908838 |
| Huaier granule | N/A | Patients with stage II or III gastric adenocarcinoma who undergo radical gastric adenocarcinoma within 2 months of surgery | Huaier granule (n = 351) vs SOX regimen (n = 351) | Prospective, multi-center, open-label, randomised controlled study | 3 years | Phase 4 | Not yet recruiting | 3-year DFS rate | 1-, 2-, 3-year OS rate, 1-, 2-, 3-year local recurrence-free survival rate, 1-, 2-, 3-year distant metastasis-free survival rate, quality of life score | NCT05498766 |
| Weifichun | N/A | Gastric adenocarcinoma patients undergoing chemotherapy | Weifichun combined chemotherapy (n = 36) vs chemotherapy (n = 36) | Randomized clinical trial | 24 weeks | Early phase 1 | Completed | OS rate | Progression-free survival, EuroPean organization for Research and Treatment Quality of Life Scale, clinical symptoms, tumor markers, immune function | NCT05888675 |
| Acupuncture | Acupuncture at back-shu points according to heat-pain threshold measurement at 24 well-points, combining with electro-acupuncture at fixted acupoints | GC patients after R0 resection and D2 lymph node dissection | High-dose acupuncture + chemotherapy vs low-dose acupuncture + chemotherapy vs none-acupuncture | Randomized, open label, pilot study | 3 cycles of adjuvant chemotherapy | N/A | Completed | FACT-gastric trial outcome index, C-ESAS | Incidence of treatment-emergent adverse events, adherence to chemotherapy, concentration of inflammatory factors in plasma, circulating myeloid-derived suppressor cells, circulating CD8+ T lymph cells, circulating tumor cells | NCT03753399 |
| N/A | POI after gastrectomy in patients with GC | Acupuncture vs none-acupuncture | Prospective randomized pilot study | 5 postoperative days | N/A | Completed | Remnant sitz markers in small intestine | Time to first flatus, start of water intake, start of soft diet, hospital stay | NCT02480361 | |
| For participants with abdominal distension: LI4, PC6, ST36, SP6. For participants with post-operative ileus: LI4, SJ6, ST36, ST37 | Underwent abdominal surgery within one month | Electroacupuncture combined with metoclopramide vs metoclopramide | Non-randomized, parallel assignment | 3 days | N/A | Unknown | Visual Analogue Scale, daily feeding volume, gastric residual volume | Acute Physiology and Chronic Health Evaluation II score, Sequential Organ Failure Assessment score, Physiological and operative severity scores for the enumeration of mortality and morbidity score, blood examination | NCT04467528 | |
| Electroacupuncture at Baihui, Nei guan, bilateral Zu sanli and bilateral Tian shu | Patients with gastric cancer who undergoing gastrectomy | Electroacupuncture vs Sham electroacupuncture | Randomized, parallel assignment | 7 days | N/A | Unknown | Gastrointestinal function score | Physical fitness index | NCT03291574 |
This article systematically examines the evidence for the potential clinical benefits of TCM therapies (including herbal formulas, acupuncture, and external therapies) in managing postoperative GC. It critically evaluates the limitations of the existing evidence base and explores the role of compounds derived from TCM in the development of anti-GC drugs.
Studies indicate that the 5-year survival rate for GC patients is below 30%, with a postoperative recurrence and metastasis rate of approximately 50% within one year, escalating to a staggering 70% by the end of two years[98,99]. According to a database from Taiwan, short-term TCM users (30-179 days) and long-term TCM users (180 days or more) among patients diagnosed with GC and undergoing surgery were independently associated with a reduced risk of mortality, with hazard ratio of 0.59 (95%CI: 0.55-0.65) and 0.41 (95%CI: 0.36-0.47), respectively[100]. However, such real-world data, while suggestive, are prone to confounding factors, and the observed associations require validation through prospective, randomized trials.
In the context of integrative medicine for cancer treatment, modern medicine typically provides targeted evidence-based interventions, while TCM offers methods to enhance the overall health of patients. TCM can tonify qi (vital energy) and blood, promote blood circulation, clear phlegm and toxins, and remove blood stasis, thereby contributing to postoperative recovery, enhancement of immune function, detoxification, and synergistic effects with chemoradiotherapy. Nevertheless, a critical gap exists between these holistic TCM concepts and their validation through modern scientific paradigms. Previous studies have demonstrated that many herbal extracts can inhibit the proliferation, angiogenesis, metastasis, and invasion of cancer cells, and can reverse the resistance to targeted drugs. However, it is crucial to interpret these findings with caution. Many mechanistic findings are derived from in vitro or in vivo models using purified compounds at concentrations that may not be achievable via oral administration of herbal decoctions in humans. Therefore, translational gaps remain. The extrapolation of these reductionist findings to the complex, multi-component, and systemic effects of whole TCM formulas in humans represents a significant oversimplification. For instance, while the PI3K/AKT pathway is frequently reported as a target, this may reflect a common investigative focus rather than a uniquely specific action of TCM. Additionally, research has shown that herbal extracts and TCM formulas benefit resistance to recurrence and metastasis, prolong patient survival, and improve quality of life. TCM can inhibit GC metastasis through various mechanisms. The mechanisms summarized in this review include the inhibition of EMT, promotion of apoptosis, induction of cell cycle arrest, and suppression of GC cell proliferation, migration, and invasion. It is noteworthy that a number of TCM external therapies, including diverse acupuncture techniques, acupoint injection, moxibustion, and auricular point-pressing with beans, have been utilized postoperatively for GC, leading to a reduction in the duration of POI, the restoration of intestinal function, and an enhancement in immune function. Yet, the clinical evidence supporting these external treatments, as summarized in Table 1, is often constrained by studies with small sample sizes, lack of adequate blinding, and heterogeneity in acupuncture techniques and outcome measures, limiting the strength of conclusions.
A substantial body of evidence suggests the potential efficacy of TCM in GC; however, the field is fraught with challenges that must be rigorously addressed. The globalization of TCM is impeded by several factors, including the translation, interpretation, standardization, and validation of TCM theories and practices. A primary concern is the lack of standardization and reproducibility. Variations in the geographical origin, processing, and preparation of herbal medicines can lead to significant inconsistencies in the composition and potency of active ingredients, making it difficult to replicate clinical results. The limited sample sizes and methodological weaknesses of many current clinical trials hinder their ability to provide strong evidence support, despite the existence of studies with promising results. Rigorous scientific investigations and further design of well-structured multicenter, large cohort, and placebo-controlled randomized clinical trials are necessary to validate the efficacy and safety of TCM. Unlike standard chemotherapy, TCM focuses on host modulation. A critical gap in current research is the comparison or combination of TCM with emerging immunotherapies, such as PD-1 inhibitors. Theoretical evidence suggests TCM could enhance the tumor microenvironment to make immunotherapies more effective (“turning cold tumors hot”), but clinical data is lacking. Furthermore, the issue of safety and potential herb-drug interactions demands greater emphasis. Particularly, some anticancer herbal remedies exhibit toxicity, raising concerns about long-term safety and potential interactions with standard GC treatments. For example, compounds like cantharidin and bufalin, while showing anti-tumor activity, have narrow therapeutic windows. The concurrent use of TCM with chemotherapy necessitates comprehensive pharmacovigilance to monitor for herb-drug interactions and cumulative toxicity, especially in patients with compromised liver and kidney function. Moreover, within the “black box” of TCM formulations, it is often challenging to determine the specific mechanisms regulated by biomolecular compounds, and the precise mechanisms and potential adverse reactions of some herbal medicines remain unclear.
In conclusion, while the pre-clinical and clinical data compiled herein are promising, they should not be overinterpreted. The path forward requires a concerted effort to bridge traditional wisdom with modern scientific rigor. In the future, it is essential to integrate knowledge and practices from both modern and traditional medicine to establish standardized protocols that can fully utilize the potential of TCM in cancer treatment. Future research must prioritize high-quality clinical trials, sophisticated pharmacological studies to deconvolute the synergistic actions of multi-herb formulations, and the development of robust quality control standards. Only through such rigorous and critical approaches can the full integrative potential of TCM in oncology be reliably assessed and realized.
This paper summarizes the clinical evidence of TCM-related drugs and external treatments, detailing how they intervene at multiple levels and affect multiple targets to influence the development and metastasis of GC cells, and also includes a compendium of ongoing clinical trials. However, the multimodal antitumor effects and complex composition of TCM make it difficult to identify their active ingredients, thus posing significant challenges to fully realize their therapeutic potential. Additionally, the paucity of in-depth research and standardized evaluation criteria in TCM clinical studies has often resulted in insufficient scientific validation, underscoring the necessity for conclusive evidence substantiating the efficacy, safety, and reproducibility of these interventions. While the evidence is preliminary, it nonetheless suggests that integrating TCM with modern medicine represents a promising approach. However, more rigorous clinical and mechanistic studies are imperative to validate its efficacy, safety, and to establish standardized protocols. It is anticipated that subsequent rigorous studies will further substantiate the clinical relevance of TCM in the treatment of GC metastasis and will serve as a guide for rational drug discovery.
| 1. | Sundar R, Nakayama I, Markar SR, Shitara K, van Laarhoven HWM, Janjigian YY, Smyth EC. Gastric cancer. Lancet. 2025;405:2087-2102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 191] [Article Influence: 191.0] [Reference Citation Analysis (0)] |
| 2. | Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16785] [Cited by in RCA: 15061] [Article Influence: 7530.5] [Reference Citation Analysis (23)] |
| 3. | Smyth EC, Nilsson M, Grabsch HI, van Grieken NC, Lordick F. Gastric cancer. Lancet. 2020;396:635-648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4042] [Cited by in RCA: 3475] [Article Influence: 579.2] [Reference Citation Analysis (13)] |
| 4. | Li L, Yang L, Yang L, He C, He Y, Chen L, Dong Q, Zhang H, Chen S, Li P. Network pharmacology: a bright guiding light on the way to explore the personalized precise medication of traditional Chinese medicine. Chin Med. 2023;18:146. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 109] [Reference Citation Analysis (0)] |
| 5. | Lee YK, Bae K, Yoo HS, Cho SH. Benefit of Adjuvant Traditional Herbal Medicine With Chemotherapy for Resectable Gastric Cancer. Integr Cancer Ther. 2018;17:619-627. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 36] [Article Influence: 4.5] [Reference Citation Analysis (2)] |
| 6. | Huang Z, Zhang Q, Fan Y, Zhou J, Liang M, Deng X, Liang J. Effect of Traditional Chinese Medicine Injection on Cancer-Related Fatigue: A Meta-Analysis Based on Existing Evidence. Evid Based Complement Alternat Med. 2020;2020:2456873. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 7. | Ye HN, Liu XY, Qin BL. Research progress of integrated traditional Chinese and Western medicine in the treatment of advanced gastric cancer. World J Gastrointest Oncol. 2023;15:69-75. [PubMed] [DOI] [Full Text] |
| 8. | Jiang C, Lin SY, Zhao JL. [Retrospective study on the dynamic development of Chinese medical syndrome types of gastric cancer]. Zhongguo Zhong Xi Yi Jie He Za Zhi. 2013;33:44-46. [PubMed] |
| 9. | Chen X, Yang K, Yang J, Li K. Meta-Analysis of Efficacy of Sijunzi Decoction Combined with Enteral Nutrition for the Treatment of Gastric Cancer. Nutr Cancer. 2020;72:723-733. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 10. | Liu Y, Yu X, Shen H, Hong Y, Hu G, Niu W, Ge J, Xuan J, Qin JJ, Li Q. Mechanisms of traditional Chinese medicine in the treatment and prevention of gastric cancer. Phytomedicine. 2024;135:156003. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 22] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 11. | Chen Y, Li Y, Wu Y, Chen S, Jin X, Chen X, Fei B, Xue X, Wu R, Chai K. Yiwei decoction promotes apoptosis of gastric cancer cells through spleen-derived exosomes. Front Pharmacol. 2023;14:1144955. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 12. | Xu Y, Zhao AG, Li ZY, Zhao G, Cai Y, Zhu XH, Cao ND, Yang JK, Zheng J, Gu Y, Han YY, Zhu YJ, Yang JZ, Gao F, Wang Q. Survival benefit of traditional Chinese herbal medicine (a herbal formula for invigorating spleen) for patients with advanced gastric cancer. Integr Cancer Ther. 2013;12:414-422. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 34] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 13. | Zhou AG, Huang DW, Ding YX, Jiang H, Tang ML. Treatment of postoperative gastric cancer with the Fuzheng Huoxue anticancer prescription. World J Gastroenterol. 1997;3:189-191. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 14. | Li YJ, Liao LL, Liu P, Tang P, Wang H, Peng QH. Sijunzi Decoction Inhibits Stemness by Suppressing β-Catenin Transcriptional Activity in Gastric Cancer Cells. Chin J Integr Med. 2022;28:702-710. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 15. | Wang M, Wang S, Su Q, Ma T. Effect of Combining Early Chemotherapy with Zhipu Liujunzi Decoction under the Concept of Strengthening and Consolidating Body Resistance for Gastric Cancer Patients and Nursing Strategy. Contrast Media Mol Imaging. 2021;2021:2135924. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 16. | Shi J, Wei PK. [The phlegm theory of gastric cancer]. Zhong Xi Yi Jie He Xue Bao. 2011;9:581-587. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 17. | Huang N, He HW, He YY, Gu W, Xu MJ, Liu L. Xiaotan Sanjie recipe, a compound Chinese herbal medicine, inhibits gastric cancer metastasis by regulating GnT-V-mediated E-cadherin glycosylation. J Integr Med. 2023;21:561-574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (2)] |
| 18. | Shi J, Lu Y, Wei P. Xiaotan Sanjie decoction inhibits angiogenesis in gastric cancer through Interleukin-8-linked regulation of the vascular endothelial growth factor pathway. J Ethnopharmacol. 2016;189:230-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 15] [Article Influence: 1.5] [Reference Citation Analysis (3)] |
| 19. | Song SJ, Liu X, Ji Q, Sun DZ, Xiu LJ, Xu JY, Yue XQ. Ziyin Huatan Recipe, a Chinese herbal compound, inhibits migration and invasion of gastric cancer by upregulating RUNX3 expression. J Integr Med. 2022;20:355-364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 20. | Yu J, Song S, Jiao J, Liu X, Zhu H, Xiu L, Sun D, Li Q, Yue X. ZiYinHuaTan Recipe Inhibits Cell Proliferation and Promotes Apoptosis in Gastric Cancer by Suppressing PI3K/AKT Pathway. Biomed Res Int. 2020;2020:2018162. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 21. | Shu P, Tang H, Zhou B, Wang R, Xu Y, Shao J, Qi M, Xia Y, Huang W, Liu S. Effect of Yiqi Huayu Jiedu decoction on stages II and III gastric cancer: A multicenter, prospective, cohort study. Medicine (Baltimore). 2019;98:e17875. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 25] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 22. | Gao F, Gao R, Hao JY, Jia JD. Gastric bezoar: a case of a patient treated with traditional Chinese medicine. J Altern Complement Med. 2012;18:93-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 23. | Yang HL, Thiyagarajan V, Shen PC, Mathew DC, Lin KY, Liao JW, Hseu YC. Anti-EMT properties of CoQ0 attributed to PI3K/AKT/NFKB/MMP-9 signaling pathway through ROS-mediated apoptosis. J Exp Clin Cancer Res. 2019;38:186. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 112] [Cited by in RCA: 104] [Article Influence: 14.9] [Reference Citation Analysis (4)] |
| 24. | Chen W, Zhang Y, Gu X, Qian P, Liu W, Shu P. Qi Ling decoction reduces gastric cancer cell metastasis by inhibiting MMP-9 through the PI3K/Akt signaling pathway. Am J Transl Res. 2021;13:4591-4602. [PubMed] |
| 25. | Fan L, He Y, Li Y, Li X, Liu D, Wang R. Efficacy and safety of traditional Chinese medicine nursing intervention in postoperative patients after gastrectomy. Oncol Lett. 2023;26:537. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 26. | Wang Z, Zhang Z, Azami NLB, Hui D, Wang Z, Xie D, Ye G, Liu N, Sun M. An Integrated Approach Using Network Pharmacology and Experimental Validation to Reveal the Therapeutic Mechanism of Weifuchun in Treating Gastric Cancer. J Med Food. 2024;27:1168-1182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 27. | Bu P. Effect of remedies for enhancing resistance and relieving blood stasis on metastasis in postoperative gastric cancer and ornithine decarboxylase levels. World J Gastroenterol. 1997;3:129-130. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 28. | Yuan M, Zou X, Liu S, Xu X, Wang H, Zhu M, Xie X, Wang H, Wu J, Sun Q. Modified Jian-pi-yang-zheng decoction inhibits gastric cancer progression via the macrophage immune checkpoint PI3Kγ. Biomed Pharmacother. 2020;129:110440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 29. | Zhang R, Chen X, Miao C, Chen Y, Li Y, Shen J, Yuan M, Chen M, Cheng J, Liu S, Sun Q, Wu J. Tumor-associated macrophage-derived exosomal miR-513b-5p is a target of jianpi yangzheng decoction for inhibiting gastric cancer. J Ethnopharmacol. 2024;318:117013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 30. | Zhu X, Zhang X, Shen J, Zheng S, Li H, Han B, Zhang C, Chen M, Sun Q, Wu J. Gut microbiota-dependent modulation of pre-metastatic niches by Jianpi Yangzheng decoction in the prevention of lung metastasis of gastric cancer. Phytomedicine. 2024;128:155413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 31. | Xu X, Li Y, Zhang R, Chen X, Shen J, Yuan M, Chen Y, Chen M, Liu S, Wu J, Sun Q. Jianpi Yangzheng decoction suppresses gastric cancer progression via modulating the miR-448/CLDN18.2 mediated YAP/TAZ signaling. J Ethnopharmacol. 2023;311:116450. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 32. | Ma L, Hu X, Zhang W, Qi D, Chen L, Yin M. Weifuchun suppresses the malignancy of gastric cancer cells by targeting KPNA2 through miR-26a-5p-mediated destabilization and the deactivation of the MAPK signaling pathway. J Ethnopharmacol. 2024;334:118538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 33. | Li L, Wang K, Liu Z, Lü Y, Wang C, Yi X, Guo J. Compound Kushen injection inhibits EMT of gastric cancer cells via the PI3K/AKT pathway. World J Surg Oncol. 2022;20:161. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 34. | Huang Z, Wu C, Zhou W, Lu S, Tan Y, Wu Z, You R, Stalin A, Guo F, Zhang J, Liu P, Wang W, Duan X, You L, Wu J. Compound Kushen Injection inhibits epithelial-mesenchymal transition of gastric carcinoma by regulating VCAM1 induced by the TNF signaling pathway. Phytomedicine. 2023;118:154984. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 35. | Song S, Wen F, Gu S, Gu P, Huang W, Ruan S, Chen X, Zhou J, Li Y, Liu J, Shu P. Network Pharmacology Study and Experimental Validation of Yiqi Huayu Decoction Inducing Ferroptosis in Gastric Cancer. Front Oncol. 2022;12:820059. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 36. | Liu J, Chen Y, Cao Z, Guan B, Peng J, Chen Y, Zhan Z, Sferra TJ, Sankararaman S, Lin J. Babao Dan inhibits the migration and invasion of gastric cancer cells by suppressing epithelial-mesenchymal transition through the TGF-β/Smad pathway. J Int Med Res. 2020;48:300060520925598. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 37. | Dai X, Yu Y, Zou C, Pan B, Wang H, Wang S, Wang X, Wang C, Liu D, Liu Y. Traditional Banxia Xiexin decoction inhibits invasion, metastasis, and epithelial mesenchymal transition in gastric cancer by reducing lncRNA TUC338 expression. Heliyon. 2023;9:e21064. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (1)] |
| 38. | Chen S, Tian X, Li S, Wu Z, Li Y, Guo J, Liao Z. The role of traditional Chinese medicine in postoperative wound complications of gastric cancer. Int Wound J. 2024;21:e14847. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 39. | Takahashi T, Endo S, Nakajima K, Souma Y, Nishida T. Effect of rikkunshito, a chinese herbal medicine, on stasis in patients after pylorus-preserving gastrectomy. World J Surg. 2009;33:296-302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 33] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 40. | Endo M, Hori M, Ozaki H, Oikawa T, Hanawa T. Daikenchuto, a traditional Japanese herbal medicine, ameliorates postoperative ileus by anti-inflammatory action through nicotinic acetylcholine receptors. J Gastroenterol. 2014;49:1026-1039. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 52] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 41. | Wang Y, Wang YT, Lin YH, Zou ZD. [Effects of Xuesaitong injection on coagulation function of gastric cancer patients undergoing laparoscopic surgery: a randomized controlled trial]. Zhong Xi Yi Jie He Xue Bao. 2009;7:514-517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 42. | Endo S, Nishida T, Nishikawa K, Nakajima K, Hasegawa J, Kitagawa T, Ito T, Matsuda H. Dai-kenchu-to, a Chinese herbal medicine, improves stasis of patients with total gastrectomy and jejunal pouch interposition. Am J Surg. 2006;192:9-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 57] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 43. | Zhao T, Yu Z. Modified Gexia-Zhuyu Tang inhibits gastric cancer progression by restoring gut microbiota and regulating pyroptosis. Cancer Cell Int. 2024;24:21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 44. | Wang Y, Wang L, Ni X, Jiang M, Zhao L. Effect of acupuncture therapy for postoperative gastrointestinal dysfunction in gastric and colorectal cancers: an umbrella review. Front Oncol. 2024;14:1291524. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 13] [Reference Citation Analysis (9)] |
| 45. | Jung SY, Chae HD, Kang UR, Kwak MA, Kim IH. Effect of Acupuncture on Postoperative Ileus after Distal Gastrectomy for Gastric Cancer. J Gastric Cancer. 2017;17:11-20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 28] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 46. | Liang Y, Huang H, Sun J, Fu Z, Chou LW. Treatment of postsurgical gastroparesis syndrome with Fu's subcutaneous needling: A case report. Explore (NY). 2023;19:827-831. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 47. | Yu C, Mao X, Zhou C. Influence of acupuncture and moxibustion on gastrointestinal function and adverse events in gastric cancer patients after surgery and chemotherapy: a meta-analysis. Support Care Cancer. 2024;32:524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 48. | You X, Wang Y, Wu J, Liu Q, Liu Y, Qian Y, Chen J, Tang D, Wang D. Zusanli (ST36) Acupoint Injection with Neostigmine for Paralytic Postoperative Ileus following Radical Gastrectomy for Gastric Cancer: a Randomized Clinical Trial. J Cancer. 2018;9:2266-2274. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 49. | Zhou X, Cao SG, Tan XJ, Liu XD, Li ZQ, Kong LX, Tian YL, Liu D, Shen S, Sun YQ, Jiang HT, Zhou YB. Effects of Transcutaneous Electrical Acupoint Stimulation (TEAS) on Postoperative Recovery in Patients with Gastric Cancer: A Randomized Controlled Trial. Cancer Manag Res. 2021;13:1449-1458. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 42] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 50. | Wehner S, Behrendt FF, Lyutenski BN, Lysson M, Bauer AJ, Hirner A, Kalff JC. Inhibition of macrophage function prevents intestinal inflammation and postoperative ileus in rodents. Gut. 2007;56:176-185. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 206] [Cited by in RCA: 196] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 51. | Bauer AJ, Boeckxstaens GE. Mechanisms of postoperative ileus. Neurogastroenterol Motil. 2004;16 Suppl 2:54-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 300] [Cited by in RCA: 244] [Article Influence: 11.1] [Reference Citation Analysis (4)] |
| 52. | Boeckxstaens GE, de Jonge WJ. Neuroimmune mechanisms in postoperative ileus. Gut. 2009;58:1300-1311. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 228] [Cited by in RCA: 270] [Article Influence: 15.9] [Reference Citation Analysis (4)] |
| 53. | Luo D, Liu S, Xie X, Hou X. Electroacupuncture at acupoint ST-36 promotes contractility of distal colon via a cholinergic pathway in conscious rats. Dig Dis Sci. 2008;53:689-693. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 67] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 54. | Chen JDZ, Ni M, Yin J. Electroacupuncture treatments for gut motility disorders. Neurogastroenterol Motil. 2018;30:e13393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 74] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 55. | Yang NN, Yang JW, Ye Y, Huang J, Wang L, Wang Y, Su XT, Lin Y, Yu FT, Ma SM, Qi LY, Lin LL, Wang LQ, Shi GX, Li HP, Liu CZ. Electroacupuncture ameliorates intestinal inflammation by activating α7nAChR-mediated JAK2/STAT3 signaling pathway in postoperative ileus. Theranostics. 2021;11:4078-4089. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 175] [Cited by in RCA: 163] [Article Influence: 32.6] [Reference Citation Analysis (4)] |
| 56. | Parthasarathy G, Ravi K, Camilleri M, Andrews C, Szarka LA, Low PA, Zinsmeister AR, Bharucha AE. Effect of neostigmine on gastroduodenal motility in patients with suspected gastrointestinal motility disorders. Neurogastroenterol Motil. 2015;27:1736-1746. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 43] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 57. | Su J, Chen L, Song N, Li X, Huang J, Duan P. Auricular Point-Pressing With Bean Plus Esomeprazole Magnesium in Relieving Gastrointestinal Dysfunction. Altern Ther Health Med. 2022;28:16-22. [PubMed] |
| 58. | Zhang Y, Wang X, Yang H. Effect of traditional Chinese medicine nursing on postoperative patients with gastric cancer and its impact on quality of life. Am J Transl Res. 2021;13:5589-5595. [PubMed] |
| 59. | Usichenko TI, Lehmann Ch, Ernst E. Auricular acupuncture for postoperative pain control: a systematic review of randomised clinical trials. Anaesthesia. 2008;63:1343-1348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 63] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 60. | Elliott T, Merlano Gomez M, Morris D, Wilson C, Pilitsis JG. A scoping review of mechanisms of auricular acupuncture for treatment of pain. Postgrad Med. 2024;136:255-265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 61. | Mao JJ, Liou KT, Baser RE, Bao T, Panageas KS, Romero SAD, Li QS, Gallagher RM, Kantoff PW. Effectiveness of Electroacupuncture or Auricular Acupuncture vs Usual Care for Chronic Musculoskeletal Pain Among Cancer Survivors: The PEACE Randomized Clinical Trial. JAMA Oncol. 2021;7:720-727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 157] [Article Influence: 31.4] [Reference Citation Analysis (0)] |
| 62. | Wang XQ, Xiao L, Duan PB, Xu Q, Yang LH, Wang AQ, Wang Y. The feasibility and efficacy of perioperative auricular acupuncture technique via intradermal needle buried for postoperative movement-evoked pain after open radical gastrectomy: A randomized controlled pilot trial. Explore (NY). 2022;18:36-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 63. | Zhang Q, Zhou M, Huo M, Si Y, Zhang Y, Fang Y, Zhang D. Mechanisms of acupuncture-electroacupuncture on inflammatory pain. Mol Pain. 2023;19:17448069231202882. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 48] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 64. | Cabioğlu MT, Cetin BE. Acupuncture and immunomodulation. Am J Chin Med. 2008;36:25-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 63] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 65. | Mittal V. Epithelial Mesenchymal Transition in Tumor Metastasis. Annu Rev Pathol. 2018;13:395-412. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1238] [Cited by in RCA: 1119] [Article Influence: 139.9] [Reference Citation Analysis (3)] |
| 66. | Zhu L, Shi J, Mu M, Chen Z, Zhao C, Li X, Qu C, Ye C, Zhao W, Sun X, Wang X. Naringin Inhibits the Proliferation, Migration, Invasion and Epithelial-to-Mesenchymal Transition of Gastric Cancer Cells via the PI3K/AKT Signaling Pathway. Altern Ther Health Med. 2023;29:191-197. [PubMed] |
| 67. | Nie J, Zhang H, Li X, Qin J, Zhou J, Lu Y, Yang N, Li Y, Li H, Li C. Pachymic acid promotes ferroptosis and inhibits gastric cancer progression by suppressing the PDGFRB-mediated PI3K/Akt pathway. Heliyon. 2024;10:e38800. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (2)] |
| 68. | Wang H, Luo Y, Chu Z, Ni T, Ou S, Dai X, Zhang X, Liu Y. Poria Acid, Triterpenoids Extracted from Poria cocos, Inhibits the Invasion and Metastasis of Gastric Cancer Cells. Molecules. 2022;27:3629. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 29] [Article Influence: 7.3] [Reference Citation Analysis (2)] |
| 69. | Zhu Z, Zhang Y, Zhang X, Chen Q, Tang S, Zhou X, Li X, Wen J, Bai Y, Zhang T. Potential molecular metabolic mechanisms underlying the effects of cimifugin in gastric cancer through single-cell and bulk RNA sequencing combined with network pharmacology. J Gastrointest Oncol. 2024;15:1409-1430. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 70. | Li J, Li J, Cai G, Shen L, Lu F. Proapoptotic and Growth-inhibitory Effects of Plumbagin on Human Gastric Cancer Cells Via Suppression of Signal Transducer and Activator of Transcription 3 and Protein Kinase B. Altern Ther Health Med. 2017;23:42-48. [PubMed] |
| 71. | Wang H, Luo Y, Hu Y, Feng X, Feng J, Chu Z, Ou S, Dai X, Wang X, Liu Y. Triptonoterpene, a Natural Product from Celastrus orbiculatus Thunb, Has Biological Activity against the Metastasis of Gastric Cancer Cells. Molecules. 2022;27:8005. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 72. | Wang H, Luo Y, Ou S, Ni T, Chu Z, Feng X, Dai X, Zhang X, Liu Y. Celastrus orbiculatus Thunb. extract inhibits EMT and metastasis of gastric cancer by regulating actin cytoskeleton remodeling. J Ethnopharmacol. 2023;301:115737. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 73. | Yu JR, Liu YY, Gao YY, Qian LH, Qiu JL, Wang PP, Zhang GJ. Diterpenoid tanshinones inhibit gastric cancer angiogenesis through the PI3K/Akt/mTOR signaling pathway. J Ethnopharmacol. 2024;324:117791. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 17] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 74. | Chen J, Chen S, Zhou Y, Wang S, Wu W. Efficacy and Safety of Huaier Granule as an Adjuvant Therapy for Cancer: An Overview of Systematic Reviews and Meta-Analyses. Integr Cancer Ther. 2022;21:15347354221083910. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 75. | Song X, Li Y, Zhang H, Yang Q. The anticancer effect of Huaier (Review). Oncol Rep. 2015;34:12-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 65] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 76. | Shi Y, Yuan L, Xu J, Xu H, Wang L, Huang L, Xu Z, Cheng X. Huaier Inhibits Gastric Cancer Growth and Hepatic Metastasis by Reducing Syntenin Expression and STAT3 Phosphorylation. J Oncol. 2022;2022:6065516. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 77. | Xu M, Ren L, Fan J, Huang L, Zhou L, Li X, Ye X. Berberine inhibits gastric cancer development and progression by regulating the JAK2/STAT3 pathway and downregulating IL-6. Life Sci. 2022;290:120266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 72] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 78. | Li N, Fan LL, Sun GP, Wan XA, Wang ZG, Wu Q, Wang H. Paeonol inhibits tumor growth in gastric cancer in vitro and in vivo. World J Gastroenterol. 2010;16:4483-4490. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 45] [Cited by in RCA: 48] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 79. | Wang Y, Li BS, Zhang ZH, Wang Z, Wan YT, Wu FW, Liu JC, Peng JX, Wang HY, Hong L. Paeonol repurposing for cancer therapy: From mechanism to clinical translation. Biomed Pharmacother. 2023;165:115277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 21] [Reference Citation Analysis (0)] |
| 80. | Gao L, Wang Z, Lu D, Huang J, Liu J, Hong L. Paeonol induces cytoprotective autophagy via blocking the Akt/mTOR pathway in ovarian cancer cells. Cell Death Dis. 2019;10:609. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 71] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 81. | Li M, Cai O, Yu Y, Tan S. Paeonol inhibits the malignancy of Apatinib-resistant gastric cancer cells via LINC00665/miR-665/MAPK1 axis. Phytomedicine. 2022;96:153903. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 38] [Article Influence: 9.5] [Reference Citation Analysis (3)] |
| 82. | Hu X, Ding L, Cao S, Cheng L, Wang K, Guang C, Li W, Koike K, Qiu F. Pharmacokinetics, Tissue Distribution and Excretion of Paeonol and Its Major Metabolites in Rats Provide a Further Insight Into Paeonol Effectiveness. Front Pharmacol. 2020;11:190. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (3)] |
| 83. | Chen H, Jiang T, Chen H, Su J, Wang X, Cao Y, Li Q. Brusatol reverses lipopolysaccharide-induced epithelial-mesenchymal transformation and induces apoptosis through PI3K/Akt/NF-кB pathway in human gastric cancer SGC-7901 cells. Anticancer Drugs. 2021;32:394-404. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 84. | Zhai B, Zhang N, Han X, Li Q, Zhang M, Chen X, Li G, Zhang R, Chen P, Wang W, Li C, Xiang Y, Liu S, Duan T, Lou J, Xie T, Sui X. Molecular targets of β-elemene, a herbal extract used in traditional Chinese medicine, and its potential role in cancer therapy: A review. Biomed Pharmacother. 2019;114:108812. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 220] [Cited by in RCA: 184] [Article Influence: 26.3] [Reference Citation Analysis (1)] |
| 85. | Zhai B, Zeng Y, Zeng Z, Zhang N, Li C, Zeng Y, You Y, Wang S, Chen X, Sui X, Xie T. Drug delivery systems for elemene, its main active ingredient β-elemene, and its derivatives in cancer therapy. Int J Nanomedicine. 2018;13:6279-6296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 126] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 86. | Deng M, Liu B, Song H, Yu R, Zou D, Chen Y, Ma Y, Lv F, Xu L, Zhang Z, Lv Q, Yang X, Che X, Qu X, Liu Y, Zhang Y, Hu X. β-Elemene inhibits the metastasis of multidrug-resistant gastric cancer cells through miR-1323/Cbl-b/EGFR pathway. Phytomedicine. 2020;69:153184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 54] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 87. | Deng M, Zhang Y, Liu B, Chen Y, Song H, Yu R, Che X, Qu X, Liu Y, Hu X, Xu X. β-Elemene inhibits peritoneal metastasis of gastric cancer cells by modulating FAK/Claudin-1 signaling. Phytother Res. 2019;33:2448-2456. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 32] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 88. | Shi D, Wu F, Gao F, Qing X, Shao Z. Prognostic value of long non-coding RNA CCAT1 expression in patients with cancer: A meta-analysis. PLoS One. 2017;12:e0179346. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 17] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 89. | Song M, Wang X, Luo Y, Liu Z, Tan W, Ye P, Fu Z, Lu F, Xiang W, Tang L, Yao L, Nie Y, Xiao J. Cantharidin suppresses gastric cancer cell migration/invasion by inhibiting the PI3K/Akt signaling pathway via CCAT1. Chem Biol Interact. 2020;317:108939. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 47] [Article Influence: 7.8] [Reference Citation Analysis (4)] |
| 90. | Zhan YP, Huang XE, Cao J, Lu YY, Wu XY, Liu J, Xu X, Xu L, Xiang J, Ye LH. Clinical study on safety and efficacy of Qinin® (cantharidin sodium) injection combined with chemotherapy in treating patients with gastric cancer. Asian Pac J Cancer Prev. 2012;13:4773-4776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 91. | Xia H, Chen Q, Chen Y, Ge X, Leng W, Tang Q, Ren M, Chen L, Yuan D, Zhang Y, Liu M, Gong Q, Bi F. The lncRNA MALAT1 is a novel biomarker for gastric cancer metastasis. Oncotarget. 2016;7:56209-56218. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 90] [Cited by in RCA: 97] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 92. | Yang Z, Xie Q, Chen Z, Ni H, Xia L, Zhao Q, Chen Z, Chen P. Resveratrol suppresses the invasion and migration of human gastric cancer cells via inhibition of MALAT1-mediated epithelial-to-mesenchymal transition. Exp Ther Med. 2019;17:1569-1578. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 34] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 93. | Yang T, Zhang J, Zhou J, Zhu M, Wang L, Yan L. Resveratrol inhibits Interleukin-6 induced invasion of human gastric cancer cells. Biomed Pharmacother. 2018;99:766-773. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 39] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 94. | Liao KF, Chiu TL, Huang SY, Hsieh TF, Chang SF, Ruan JW, Chen SP, Pang CY, Chiu SC. Anti-Cancer Effects of Radix Angelica Sinensis (Danggui) and N-Butylidenephthalide on Gastric Cancer: Implications for REDD1 Activation and mTOR Inhibition. Cell Physiol Biochem. 2018;48:2231-2246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 38] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 95. | Wang J, Cai H, Xia Y, Wang S, Xing L, Chen C, Zhang Y, Xu J, Yin P, Jiang Y, Zhao R, Zuo Q, Chen T. Bufalin inhibits gastric cancer invasion and metastasis by down-regulating Wnt/ASCL2 expression. Oncotarget. 2018;9:23320-23333. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 27] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 96. | Dai X, Liu D, Liu M, Zhang X, Wang W, Jin F, Qian Y, Wang X, Zhao J, Wu Y, Xiong F, Chang NA, Sun YU, Yang Z, Hoffman RM, Liu Y. Anti-metastatic Efficacy of Traditional Chinese Medicine (TCM) Ginsenoside Conjugated to a VEFGR-3 Antibody on Human Gastric Cancer in an Orthotopic Mouse Model. Anticancer Res. 2017;37:979-986. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 16] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 97. | Li Z, Zhang G, Cao N, Xu J, Dong J, Li J, Zhu X, Xu Y, Han C, Wang R, Xia X, Zhao G, Huan X, Fan J, Zhao A. Effects of traditional Chinese medicine collaborative model (TCMCM) combined with adjuvant chemotherapy on IIIb and IIIc gastric cancer: a protocol for a randomized controlled trial. Trials. 2022;23:68. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 98. | Mehlen P, Puisieux A. Metastasis: a question of life or death. Nat Rev Cancer. 2006;6:449-458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1538] [Cited by in RCA: 1367] [Article Influence: 68.4] [Reference Citation Analysis (0)] |
| 99. | Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76817] [Cited by in RCA: 69798] [Article Influence: 13959.6] [Reference Citation Analysis (50)] |
| 100. | Shih WT, Yang PR, Shen YC, Yang YH, Wu CY. Traditional Chinese Medicine Enhances Survival in Patients with Gastric Cancer after Surgery and Adjuvant Chemotherapy in Taiwan: A Nationwide Matched Cohort Study. Evid Based Complement Alternat Med. 2021;2021:7584631. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 101. | Li J, Niu J, Yang M, Ye P, Zhai J, Yuan W, Feng L, Tian G, Hu J, Wang Y, Shang H. Using single-patient (n-of-1) trials to determine effectiveness of traditional Chinese medicine on chemotherapy-induced leukopenia in gastric cancer: a feasibility study. Ann Transl Med. 2019;7:124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 102. | Mi SC, Wu LY, Xu ZJ, Zheng LY, Luo JW. Effect of modified ShengYangYiwei decoction on painless gastroscopy and gastrointestinal and immune function in gastric cancer patients. World J Gastrointest Endosc. 2023;15:376-385. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 103. | Guo JF, Zhang Y, Wu GC, Guo H. [Effect of acupuncture on functional delayed gastric emptying after gastric cancer surgery based on enhance recovery after surgery]. Zhongguo Zhen Jiu. 2023;43:141-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 104. | Chang XS, Zhu YJ, Peng JJ, Wu XY, Wang W, Liu YH, Zhan DD, Chen YD, Zhang HB. [Clinical observation on acupuncture for symptom burden in gastric cancer patients undergoing adjuvant chemotherapy after gastrectomy]. Zhongguo Zhen Jiu. 2022;42:1226-1232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 105. | Chae HD, Kwak MA, Kim IH. Effect of Acupuncture on Reducing Duration of Postoperative Ileus After Gastrectomy in Patients with Gastric Cancer: A Pilot Study Using Sitz Marker. J Altern Complement Med. 2016;22:465-472. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 106. | Yin SH, Du YQ, Liu B. [Clinical study on acupuncture combined with medication in restoration of gastrointestinal functions for postoperative patients with gastric cancer]. Zhongguo Zhen Jiu. 2009;29:459-462. [PubMed] |
| 107. | Zhu YJ, Wu XY, Wang W, Chang XS, Zhan DD, Diao DC, Xiao J, Li Y, Ma D, Hu M, Li JC, Wan J, Wu GN, Ke CF, Sun KY, Huang ZL, Cao TY, Zhai XH, Chen YD, Peng JJ, Mao JJ, Zhang HB. Acupuncture for Quality of Life in Gastric Cancer Patients Undergoing Adjuvant Chemotherapy. J Pain Symptom Manage. 2022;63:210-220. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 30] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 108. | Ren G, Yang L. The effect of TCM triple rehabilitation and ear acupoint bean pressing on laparoscopic surgery for gastric cancer. Medicine (Baltimore). 2024;103:e39423. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 109. | Zhao AG, Zhao HL, Jin XJ, Yang JK, Tang LD. Effects of Chinese Jianpi herbs on cell apoptosis and related gene expression in human gastric cancer grafted onto nude mice. World J Gastroenterol. 2002;8:792-796. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 38] [Cited by in RCA: 37] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 110. | Deng YQ, Gao M, Lu D, Liu QP, Zhang RJ, Ye J, Zhao J, Feng ZH, Li QZ, Zhang H. Compound-composed Chinese medicine of Huachansu triggers apoptosis of gastric cancer cells through increase of reactive oxygen species levels and suppression of proteasome activities. Phytomedicine. 2024;123:155169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 32] [Reference Citation Analysis (1)] |
| 111. | Chen GQ, Nan Y, Ning N, Huang SC, Bai YT, Zhou ZY, Qian G, Li WQ, Yuan L. Network pharmacology study and in vitro experimental validation of Xiaojianzhong decoction against gastric cancer. World J Gastrointest Oncol. 2024;16:3932-3954. [PubMed] [DOI] [Full Text] |
| 112. | Cheng FE, Li Z, Bai X, Jing Y, Zhang J, Shi X, Li T, Li W. Investigation on the mechanism of the combination of eremias multiocellata and cisplatin in reducing chemoresistance of gastric cancer based on in vitro and in vivo experiments. Aging (Albany NY). 2024;16:3386-3403. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |