Published online Mar 24, 2026. doi: 10.5306/wjco.v17.i3.116093
Revised: January 2, 2026
Accepted: February 9, 2026
Published online: March 24, 2026
Processing time: 141 Days and 16.9 Hours
Liver metastases are common in advanced cancer and are consistently associated with poorer survival and limited treatment options. Although immunotherapy has transformed outcomes for many malignancies, patients with liver involvement respond less well, highlighting the liver as a uniquely challenging site for effective antitumor immuni
Core Tip: This review highlights how the liver’s unique vascular and immune environment fosters metastasis. We summarize key cellular interactions, the concept of the hepatic pre-metastatic niche, and emerging therapeutic strategies that may improve outcomes for patients with liver metastases.
- Citation: Pham HT, Dong ZY, De Alba M, Cottle C, Zhang MY, Parker JB, Morecroft-Phillipps R, Chaludiya K, Ingram D, Ahmad AN, Zeng Z, Mohseni MM, Zhang ZZ, Rueda Prada L, Ali M, Yang V. Immunotherapy in liver metastases: Challenges, emerging evidence, and future directions. World J Clin Oncol 2026; 17(3): 116093
- URL: https://www.wjgnet.com/2218-4333/full/v17/i3/116093.htm
- DOI: https://dx.doi.org/10.5306/wjco.v17.i3.116093
Liver metastases are a common and clinically significant challenge in patients with advanced malignancies[1]. Tumors originating from the gastrointestinal (GI) tract, such as those of the pancreas, stomach, and colon, as well as from extra-GI sites including the breast, lung, and skin, frequently metastasize to the liver due to its unique dual blood supply and immuno-tolerant microenvironment[2]. Despite therapeutic advances, liver metastases often confer a poor prognosis and remain challenging to treat effectively[3]. Management of liver metastases often requires a multimodal approach tailored to tumor origin and disease extent: Therapies include surgical resection, thermal ablation (radiofrequency or microwave), regional interventions such as transarterial chemoembolization, radioembolization, or hepatic arterial infusion, and systemic therapies including chemotherapy, molecularly targeted agents, and immunotherapy[4]. Immune checkpoint inhibitors (ICIs) have transformed cancer therapy, offering durable responses in several malignancies[5]. However, emerging data consistently show that patients with liver metastases derive less benefit from immunotherapy compared to those without hepatic involvement[3]. This discrepancy is observed across multiple tumor types and is supported by clinical trial subgroup analyses and meta-analyses[3]. The liver’s immunosuppressive microenvironment-characterized by cytotoxic T-cell and natural killer (NK) cell exhaustion and exclusion, along with the expansion of multiple immu
The liver’s anatomy and microarchitecture create a distinct environment for tumor cell settlement. One of the most salient anatomical features is its dual blood supply: Approximately 75% of hepatic inflow comes via the portal vein (draining the GI tract), and 25% via the hepatic artery[7,8]. This configuration exposes the liver to a continuous influx of dietary antigens, microbial products, and-importantly-circulating tumor cells (CTCs) originating from both GI and non-GI primaries[9]. Because the portal venous network channels blood from abdominal organs first through the liver, it makes the liver a primary “first-pass filter” for tumor cells arising in colorectal, gastric, pancreatic cancers, and even extra-GI malignancies such as breast, lung, and melanoma[10].
However, intact portal flow appears to be a prerequisite for efficient metastatic seeding in hepatic sinusoids. In a large study of 1453 cirrhotic livers with liver masses, only 1.7% turned out to be metastases (24 cases), in contrast to the preponderance of primary hepatic tumors[11]. Among those metastases, the majority (71%) occurred in livers with mild to moderate fibrosis (Laennec stages 4A/4B) and patent portal veins; fewer cases occurred in severe cirrhosis (4C) with reversal or obstruction of portal flow[11]. These findings imply that disturbed portal hemodynamics and fibrotic remo
This concept is reinforced by older post-mortem data showing lower rates of metastases in cirrhotic compared to non-cirrhotic livers: The odds ratio for metastatic incidence in cirrhosis was 0.47[12]. In other words, cirrhotic architecture-despite being pathologic-may confer a degree of “metastatic resistance” via altered flow and vascular remodeling.
Beyond macro-hemodynamics, the sinusoidal microarchitecture provides additional layers of complexity. Liver sinusoidal endothelial cells (LSECs) form the fenestrated lining of hepatic sinusoids, maintaining immune tolerance and vascular homeostasis through antioxidant and anti-inflammatory activity[13-15]. When injured, LSECs lose fenestrae and develop intracellular gaps, up-regulating adhesion molecules such as ICAM-1 and MMP9 that facilitate tumor cell adhesion and transmigration. The diverse functions of LSECs in liver metastasis will be further elaborated below in a separate subsection below[14,16,17].
The liver is uniquely organized into lobular zones with distinct oxygen, nutrient, and metabolic gradients that shape its physiological and pathological responses. Peri-portal hepatocytes, located near the portal triad, exhibit high oxidative metabolism and oxygen consumption, whereas peri-venous hepatocytes, situated near the central vein, reside in relatively hypoxic regions and rely predominantly on glycolysis for ATP generation[18]. This zonal heterogeneity esta
Together, these anatomical and micro-architectural features, including the liver’s dual blood supply, uniquely fene
Pre-metastatic niche (PMN) refers to a permissive microenvironment established in distant organs prior to the arrival of CTCs, creating fertile “soil” for metastatic seeding and immune evasion[22-24]. In the liver, PMN formation is orchestrated by tumor-derived soluble factors, extracellular vesicles (EVs), and myeloid cell recruitment, resulting in distinctive molecular, stromal, and immunologic remodeling that distinguishes hepatic niches from those in the lung or bone.
Tumor-derived exosomes enriched with integrins-particularly αvβ5-selectively home to Kupffer cells (KCs), where they trigger transforming growth factor-beta (TGF-β) release, activation of hepatic stellate cells (HSCs), and fibronectin deposition. This extracellular matrix (ECM) remodeling recruits bone marrow-derived myeloid progenitors, establishing a fibrotic and immunosuppressive scaffold that supports later colonization[22,25]. In parallel, cytokines and chemokines such as CCL2, CXCL1, and CX3CL1 drive the accumulation of myeloid-derived suppressor cells (MDSCs), which dampen cytotoxic T-cell activity and promote regulatory T-cell expansion[26]. Within this evolving microenvironment, KCs undergo phenotypic reprogramming toward an M2-like, programmed death ligand-1 (PD-L1)+ state, driven by metabolic stress and tumor-derived signals, amplifying T-cell exhaustion and immune escape[27]. The liver’s intrinsic tolerogenic landscape-shaped by its dual blood supply and constant exposure to gut-derived antigens-further enhances these sup
The metastatic cascade is a multi-step biological process by which cancer cells detach from a primary tumor, survive in circulation, and colonize distant organs[31]. Although this cascade occurs across multiple metastatic sites, liver meta
Tumor invasion and intravasation initiate when malignant cells degrade the ECM and breach vascular or lymphatic barriers. This process is mediated by matrix metalloproteinases (MMPs), epithelial-mesenchymal transition (EMT), and dynamic interactions with endothelial and stromal components that facilitate motility and vessel penetration[33]. Once within the circulation, CTCs encounter substantial hemodynamic shear stress and immune surveillance[34]. To evade destruction, CTCs frequently aggregate with platelets, forming protective micro-thrombi that shield them from neu
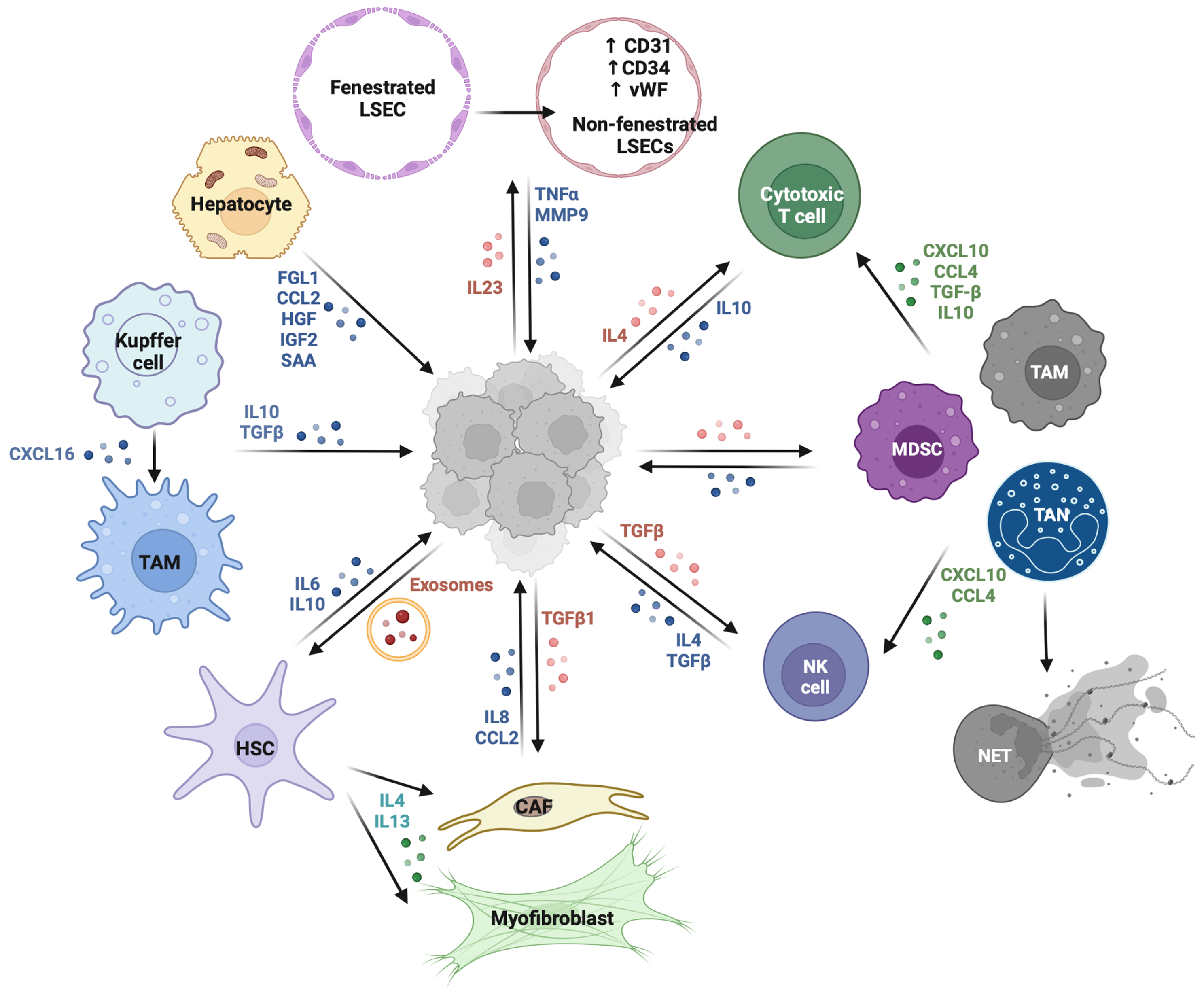
Within the liver, hemodynamic and structural features favor metastatic arrest. The hepatic sinusoids, characterized by low shear stress, fenestrated endothelium, and abundant adhesion molecules such as VCAM-1, ICAM-1, and selectins, provide an ideal microvascular niche for tumor cell adhesion[36-38]. Tumor extravasation is further enhanced by inflammation-induced up-regulation of adhesion molecules-particularly ICAM-1-on LSECs. Experimental and translational models demonstrate that ICAM-1 expression on LSECs induces tumor cells to secrete prostaglandin E2, interleukin (IL)-6, vascular endothelial growth factor (VEGF), and MMPs, which collectively promote endothelial transmigration and remodeling of the peri-sinusoidal matrix, ultimately facilitating hepatic colonization[37]. Following extravasation, the hepatic microenvironment initiates a unique sequence of immunosuppressive responses that differ from other organs (Figure 1 and Table 1).
| Cell type | Key functions in liver metastasis | Roles in metastatic colonization & immune suppression |
| KCs[39-46] | Liver-resident macrophages that are the first immune cells to contact CTCs in hepatic sinusoids | Can exert early tumoricidal activity but may become reprogrammed into an immunosuppressive phenotype promoting T-cell exhaustion and MDSC accumulation |
| LSECs[13-16,47-49] | Fenestrated endothelial cells regulating leukocyte trafficking, antigen presentation, and vascular tone | Under pathological activation, LSECs upregulate adhesion molecules and promote tumor cell arrest, transmigration, and angiogenesis |
| HSCs[50-55,109] | Perisinusoidal cells that can transition into myofibroblast-like CAFs and remodel ECM | Activated by tumor-derived factors to secrete IL-8, deposit collagen, and create a fibrotic, immune-excluded microenvironment |
| Hepatocytes[56-61] | Primary parenchymal liver cells central to metabolism and paracrine signaling | Modulate Fas/FasL, IGF2, HGF, and FGL1 to support tumor survival, proliferation, and suppression of CD8+ T-cell and NK-cell responses |
The liver’s complex architecture and diverse cellular ecosystem create a unique environment for metastatic colonization. Beyond hepatocytes, a variety of non-parenchymal cells-including KCs, LSECs, HSCs (Ito cells), and infiltrating immune cells-coordinate local immune, metabolic, and stromal responses that dictate whether disseminated tumor cells are cleared or colonize successfully. Each cell type contributes distinct yet interconnected roles: KCs and LSECs regulate immune surveillance and vascular adhesion, while stellate cells mediate fibrotic remodeling and extracellular-matrix de
KCs, the liver’s resident macrophages, play a dual role in metastatic progression, initially anti-tumorigenic but later becoming immunosuppressive. Their activation is governed by a balance of activating and inhibitory receptors that normally protect hepatic tissue from excessive inflammation but can shift toward immune tolerance during metastasis. In early liver metastasis, KCs are highly phagocytic and eliminate tumor cells. However, this capacity declines at later stages due to induction of musculoaponeurotic fibrosarcoma oncogene homolog B and c-musculoaponeurotic fibrosarcoma oncogene homolog, which limit their tumor infiltration and phagocytosis[39]. The lineage-defining factor inhibitor of DNA binding 3 (ID3) maintains KC phagocytic competence and promotes recruitment and activation of NK and CD8+ T cells to suppress tumor growth; loss of ID3 skews KCs toward an immunosuppressive phenotype[40].
Once reprogrammed, KCs actively support metastasis. Tumor-derived deleted in malignant brain tumors 1 binds to mucin 1 on KCs, triggering nuclear factor kappa B activation and induction of chemokine (C-C motif) ligand 8 and L-selectin, which recruit neutrophils and promote neutrophil extracellular trap (NET) formation, establishing a pro-metastatic niche[41]. KCs can also undergo CD206+ M2-like polarization in response to tumor-secreted cytokines such as C-X-C motif chemokine ligand 16, secrete immunosuppressive mediators (IL-10, TGF-β), and express Fas ligand (FasL), inducing apoptosis of Fas+ CD8+ T cells-a mechanism distinctive to the hepatic metastatic environment (Figure 1 and Table 1)[42-45]. Given their inherent phagocytic capacity, KCs have emerged as a target for therapeutic re-activation. β-glucans, pathogen-associated molecular patterns that signal through Dectin-1, can reprogram macrophages toward anti-tumor activity. In murine models, treatment with soluble β-1,3/1,6-glucan (odetiglucan) combined with immune che
LSECs form the highly specialized lining of hepatic sinusoids, serving as the anatomical interface between the liver’s dual blood supply-the portal vein and hepatic artery-and the parenchyma[13]. Unlike continuous endothelial cells found in other organs, LSECs possess fenestrations that allow for efficient molecular exchange and maintain a non-inflammatory, tolerogenic environment[15,47]. Fenestrated LSECs exert antioxidant and anti-inflammatory functions, scavenging reactive oxygen species and producing nitric oxide, which helps preserve hepatic microvascular homeostasis[15,47]. They also suppress HSC activation, thereby preventing fibrogenesis and indirectly impeding metastatic colonization[14,48]. In contrast, non-fenestrated LSECs exhibit a marked increase in the expression of CD31, CD34, and molecules such as von Willebrand factor, which is accompanied with development of basement membranes and ultimate transformation into a continuous endothelium[16]. However, the role of LSECs phenotype switching, i.e., from fenestrated to capillarized LSECs, remains unclear in liver metastases. The evidence we discussed above showed the cirrhosis is a protective factor against liver metastases, which seems to suggested the capillarized LSECs prevents metastatic seeding. However, recent studies revealed that pathological liver injury induces intracellular gap formation in LSECs through the destruction of fenestrae, creating permissive entry points for tumor cells. Cancer cells stimulate IL-23-dependent TNF-α secretion from LSECs, leading to F-actin depolymerization and up-regulation of MMP9, ICAM-1, and CXCLs-molecular changes that facilitate tumor cell transmigration and metastasis (Figure 1)[49].
HSCs, or Ito cells, which differentiate into myofibroblasts/cancer-associated fibroblasts (CAFs) and deposit dense collagen-rich ECM, are important cell types to suppress T-cell driven anti-tumor immunity and create “cold” tumor microenvironment (TME) by forming dense collagen-based stroma to hinder immune cell infiltration and to shield tumor cells from damages inflicted by many anti-cancer therapies[50]. The process cell-derived fibrosis is driven by complex molecular and immune mechanisms, and dynamic communications of tumor cells, HSCs and other cell types in the TME. In the liver metastasis of gastro-intestinal malignancies, the HSC-CAF conversion often starts off with tumor cell-derived signalings. Tumor cells can produce exosomes, or secrete soluble factors such as cytokines or chemokines, to influence HSC behavior. For example, AMIGO2-containing sEVs derived from gastric cancer cells actively modify the hepatic microenvironment by activating HSCs and inducing IL-8 secretion, which promotes gastric cancer cell migration into the liver parenchyma[51]. Exosomal sphingosine kinase 1 (SPHK1) increased the migration of colorectal cells, and activated HSCs by regulating pAKT[52]. For example, POU6F2 over-expressing gastric tumor cells promotes the conversion of HSCs into CAFs via transcriptional upregulation of insulin-like growth factor 2 (IGF2) and subsequent activation of PI3K/Akt signaling as well as[53]. Similarly, suprabasin (SBSN)-expressing gastric tumor cells activate HSCs into CAFs via EGF/EGFR axis, and cause subsequent productions of CCL2 from HSCs to feed tumor cell growth via CCR2/JAK2 pathway[54]. Sometimes, the HSCs cell activation is not directly driven by tumor cells. Certain immune cells, such as in
In summary, multiple tumor-derived factors converge to activate HSCs during metastatic colonization. These include soluble proteins and EV-associated cargo such as AMIGO2, SPHK1, and POU6F2- or SBSN-regulated signaling mole
Hepatocytes, which make up nearly 70% of liver mass, are central to the formation of the hepatic metastatic niche through complex interactions with tumor, stromal, and immune cells. Although the precise mechanisms remain incompletely understood, emerging evidence highlights their dual role in modulating immune responses and remodeling the local microenvironment to support metastatic seeding. During early colonization, tumor-secreted FasL can induce apoptosis of neighboring hepatocytes, creating localized areas of parenchymal disruption that facilitate tumor cell invasion[56]. Once in contact with hepatocytes, cancer cells exploit hepatocyte-derived growth and inflammatory factors, including IGF2, hepatocyte growth factor (HGF), fibrinogen-like protein 1 (FGL1), CCL2, and serum amyloid A1/A2 (SAA), to establish a protective and metabolically supportive niche that shelters tumor cells from immune clearance and therapy-induced stress (Figure 1)[57-59].
Activation of the IL-6/signal transducer and activator of transcription 3 (STAT3) pathway in hepatocytes further amplifies this process. STAT3-driven production of SAA recruits myeloid cells and promotes fibrosis, reinforcing an immunosuppressive and fibrotic microenvironment conducive to metastasis[59]. Recent single-cell analyses also reveal a distinct “proinflammatory hepatocyte” subset, primarily derived from periportal zones, that recruits macrophages via the CCL2-CCR2 axis. These recruited macrophages release cytokines such as IL-6, TNF-α, and IL-17, which upregulate PD-L1 on hepatocytes, further suppressing T-cell responses and promoting immune tolerance within the metastatic microenvironment[60]. Beyond cytokine signaling, hepatocytes also exert immunoregulatory control through FGL1, a ligand for lymphocyte activation gene 3. Under physiological conditions, FGL1 maintains hepatic immune tolerance; however, in the metastatic setting, elevated FGL1 inhibits cytotoxic CD8+ T cells and NK cells, promoting immune evasion[61].
Collectively, hepatocytes actively sculpt the hepatic niche through inflammatory, metabolic, and immune interactions-transforming from passive parenchymal cells into orchestrators of tumor accommodation and immune suppression.
Angiogenesis within liver metastases is a complex, tumor-driven process shaped by both pre-metastatic conditioning and direct tumor-endothelial interactions. Tumor cells secrete proangiogenic mediators such as VEGF, TGF-β, and IL-10 to reprogram local immune and stromal cells, fostering vascular growth and immune evasion[62-64]. In the hepatic metastatic niche, this neovascularization is often aberrant-characterized by disorganized, pericyte-poor, and hyperpermeable (leaky) vessels driven by VEGF-mediated endothelial destabilization and immature vascular architecture[65]. Such dysfunctional vasculature enhances plasma extravasation and stromal remodeling, further facilitating tumor exp
Beyond KCs, HSCs, LSECs and hepatocytes discussed above, a diverse network of immune cells, including MDSCs, regulatory T cells, M2-like macrophages, IgA+ plasma B cells, and neutrophils, contributes to the immunosuppressive landscape of liver metastases. These cells coordinate closely with hepatic stromal elements such as fibroblasts and sinusoidal endothelial cells to suppress cytotoxic lymphocyte activity through cytokine secretion (IL-10, TGF-β), meta
Overall, the coordinated response involving KCs, LSECs, and HSCs-along with a fibrotic, hypoxic, immune-sup
Many clinical studies have been conducted to improve outcome for those with liver metastasis, and the evidence is sum
Multiple meta-analyses have consistently shown that liver metastases predict reduced efficacy of ICIs across a range of tumor types. A comprehensive meta-analysis by Tian et al[77] including 163 studies across diverse cancers found that liver metastases were associated with significantly worse overall survival (OS; HR = 1.82, 95%CI: 1.59-2.08) and progression-free survival (PFS; HR = 1.68, 95%CI: 1.49-1.89) in ICI-treated patients (Table 2). In a pan-cancer analysis by Chen et al[78], stratified subgroup analysis of patients receiving ICI monotherapy showed markedly shorter OS for those with liver metastases: 10 months vs 20 months in patients without liver disease (P < 0.0001; Table 1).
| Tumor type | Analysis | Treatment/comparison | Endpoint | Liver metastasis subgroup result | Overall population result (for comparison) | Key finding regarding liver metastases |
| Pan-cancer[77] | Meta-analysis | ICI-treated patients (vs without liver mets) | OS, PFS | OS: HR = 1.82, 95%CI: 1.59-2.08; PFS: HR = 1.68, 95%CI: 1.49-1.89 | N/A | Liver metastases associated with significantly worse OS and PFS |
| Pan-cancer[78] | Pan-cancer analysis | ICI monotherapy | OS | 10 months | 20 months (patients without liver disease) | Markedly shorter OS for those with liver metastases |
| NSCLC[81] | Meta-analysis | ICIs vs control | PFS, OS | PFS: HR = 0.64, 95%CI: 0.55-0.75; OS: HR = 0.82, 95%CI: 0.72-0.94 | PFS: HR = 0.56, 95%CI: 0.50-0.63; OS: HR = 0.73, 95%CI: 0.66-0.81 | Benefit is present but attenuated compared to those without liver mets (larger HR values) |
| NSCLC[82] | Pooled CheckMate 017/057 | Nivolumab vs docetaxel | OS | HR = 0.68, 95%CI: 0.50-0.91 | HR = 0.70 | Nivolumab maintained an OS benefit, similar to the overall population |
| NSCLC[83] | KEYNOTE-189 | Pembro + chemo vs chemo | OS | HR = 0.62, 95%CI: 0.44-0.87) | HR = 0.56 | OS benefit was present but less pronounced than in the overall population |
| NSCLC[84-87] | Meta-analyses of KEYNOTE-001, KEYNOTE-010, and KEYNOTE-024 | ICIs vs control (pooled analysis) | OS | Pooled OS: HR = 0.78, 95%CI: 0.68-0.90 | N/A | Survival benefit is present but attenuated relative to patients without liver involvement (ratio of OS-HRs = 1.10) |
| NSCLC[88] | Real-world cohorts | ICI-treated patients (with vs without liver mets) | OS | N/A | N/A | Approximately 21% higher risk of death (OS: HR = 1.21, 95%CI: 1.17-1.27) with liver metastases |
| NSCLC[89-91] | IMpower150 (final analysis) | ABCP (atezolizumab + bevacizumab + chemo) vs chemo | OS | HR = 0.68, 95%CI: 0.45-1.02 | N/A | Suggested clinically meaningful benefit by adding anti-VEGF (bevacizumab) to the regimen in this subgroup |
| NSCLC[92,93] | CheckMate 9 LA | Nivo + ipi + chemo vs chemo | OS (5-year rate) | Numerically lower survival than those without hepatic mets; 18% OS rate (vs 11% with chemo alone) | N/A | Durable OS benefit across metastatic subgroups, including those with liver involvement |
| Melanoma[94] | - | Anti-PD-1 therapy (pooled analyses) | ORR | Markedly reduced | N/A | Liver metastases significantly dampen responses, associated with reduced CD8+ T-cell infiltration |
| CRC[99] | KEYNOTE-177 (MSI-H/dMMR subset) | Pembrolizumab vs chemotherapy | PFS | Improved PFS (specific HR not provided) | N/A | Pembrolizumab significantly improved PFS, including those with liver metastases |
| CRC[106] | Real-world, MSI-H/dMMR) | ICI-treated patients (with vs without liver mets) | ORR, PFS | ORR = 58%; PFS: HR = 3.18, 95%CI: 1.52-6.67 | ORR 66% | Liver involvement impairs outcomes (lower ORR, higher HR for progression) |
| CRC[108] | REGONIVO (regorafenib + nivolumab, early-phase) | ICI + VEGF inhibitor | ORR | 7%-19% (North American cohorts) | Up to 33% (Japanese cohorts) | Limited generalizability; ORR significantly lower, especially in patients with liver metastases |
| PDAC[123,125] | KEYNOTE-158 (MSI-H/dMMR subset) | Pembrolizumab | OS, ORR | ORR = 18% (4 of 22 patients); median OS = 4.0 months, 95%CI: 2.1-8.7 | N/A | Limited data; liver metastases exacerbate resistance due to fibrosis-driven immune exclusion |
| Gastric/GEJ cancer[129] | KEYNOTE 859 | Chemo-immunotherapy combination | OS | HR = 0.83, 95%CI: 0.70-0.90 | HR = 0.73, 95%CI: 0.63-0.84 | Survival benefits regardless of liver metastasis status; difference in benefit was not statistically significant |
| Gastric/GEJ cancer[131] | Retrospective cohort | ICI-treated patients (with vs without liver mets) | OS | 10.53 months | 13.43 months | No significant difference in OS. Other factors (peritoneal mets, burden) were more strongly associated with reduced PFS |
| Gastric/GEJ[126] | Mechanistic profiling | - | Immune composition (CD4+, CD8+, monocytic MDSC) | - | Stomach/GEJ show increase proliferating CD4+/CD8+ and decrease monocytic MDSC vs EAC | Proinflammatory TME may blunt LM’s negative effect |
| Gastric/GEJ[127] | JAVELIN gastric 100 - phase 3 (maintenance) | Avelumab maintenance vs maintenance chemotherapy | OS, PFS | Not reported specifically by LM status | No improvement in OS or PFS with avelumab maintenance | Maintenance ICI alone did not improve outcomes |
| Gastric/GEJ[128-130] | Meta-analysis | Chemoimmunotherapy vs chemotherapy | OS, PFS | Benefit observed irrespective of LM status across included trials | ChemoIO improved OS and PFS (KEYNOTE859, CheckMate 649) | Supports chemoIO regardless of LM status |
| Gastric/GEJ[130] | CheckMate 649 - phase 3 | Nivolumab + chemo vs chemo | OS, PFS | No dedicated LM subgroup analysis reported | Significant OS/PFS benefit overall (approximately 96% metastatic; approximately 40% LM) | ChemoIO improves outcomes; LMspecific effect not isolated |
| Gastric/GEJ[129] | KEYNOTE859 - phase 3 | Pembrolizumab + chemo vs chemo | OS | HR = 0.83, 95%CI: 0.70-0.90 - benefit regardless of LM | NoLM HR = 0.73, 95%CI: 0.63-0.84 | Difference by LM not statistically significant |
| Gastric/GEJ[131] | Retrospective cohort | Immunotherapy (with LM vs without LM) | OS (factors for PFS) | OS = 10.53 months (LM) - no significant difference vs noLM | OS = 13.43 months (noLM); P = 0.584 | Reduced PFS associated with MMR mutations, > 3 metastatic sites, peritoneal metastases |
Together, these meta-analyses confirm that liver metastasis is a robust and independent negative predictive factor for ICI response. These data provide a clear rationale for the investigation and development of liver-specific combination str
Approximately 20% of patients with non-small cell lung cancer (NSCLC) develop liver metastases, which are an established negative prognostic factor and are associated with inferior responses to ICIs[79,80]. Xu et al[81] conducted a systematic review and meta-analysis of 17 randomized controlled trials in NSCLC patients. In those with liver metastases, ICIs improved PFS (HR = 0.64, 95%CI: 0.55-0.75) and OS (HR = 0.82, 95%CI: 0.72-0.94; Table 1). In comparison, patients without liver metastases experienced greater benefits from ICIs, with PFS (HR = 0.56, 95%CI: 0.50-0.63) and OS (HR = 0.73, 95%CI: 0.66-0.81; Table 1)[81].
Standard first-line treatment for advanced NSCLC without actionable driver mutations typically includes PD-1/PD-L1 inhibitors-such as pembrolizumab, nivolumab, or atezolizumab-administered alone or in combination with platinum-based chemotherapy, depending on PD-L1 expression status. Subgroup analysis from the pivotal trials, including KEYNOTE-189 and the pooled CheckMate 017/057 data, consistently demonstrated that the benefit of ICIs is attenuated, but still present, in patients with liver metastases. For instance, in the pooled CheckMate 017/057 analysis, nivolumab maintained an OS benefit over docetaxel in the liver metastasis subgroup (HR = 0.68, 95%CI: 0.50-0.91), which was similar to the HR observed in the overall population (HR = 0.70; Table 1)[82]. Similarly, KEYNOTE-189 showed OS benefit with the combination regimen in patients with liver metastases (HR = 0.62, 95%CI: 0.44-0.87), which was less pronounced than in the overall population (HR = 0.56; Table 1)[83]. Across major pembrolizumab trials, including KEYNOTE-001, KEYNOTE-010, and KEYNOTE-024, patients with hepatic metastases consistently demonstrated lower objective response rate (ORR) and shorter OS compared to those without liver involvement[84-87]. Meta-analyses of these and related studies show that while ICIs improve survival in NSCLC patients with liver metastases (pooled OS: HR = 0.78, 95%CI: 0.68-0.90), the magnitude of benefit is attenuated relative to patients without liver involvement (ratio of OS: HR = 1.10, 95%CI: 0.94-1.29; Table 1)[88]. Real-world cohorts similarly report an approximately 21% higher risk of death among NSCLC patients with liver metastases treated with ICIs vs those without (OS: HR = 1.21, 95%CI: 1.17-1.27; Table 1)[88].
Importantly, IMpower150 demonstrated that adding bevacizumab (anti-VEGF) to atezolizumab and platinum-based chemotherapy (ABCP regimen) improved survival among patients with liver metastases, underscoring the value of vas
| Strategy | Mechanistic target | Representative trial/evidence | Primary cancer(s) | Outcome summary | Clinical implication |
| VEGF blockade + ICI[137,138] | Vascular normalization, myeloid suppression | IMpower150 (ABCP regimen) | NSCLC (with liver mets) | OS: HR = 0.68; improved immune infiltration | Supports VEGF + PD-L1 combination |
| Dual checkpoint blockade[92] | T-cell priming & exhaustion | CheckMate 9 LA | NSCLC, melanoma, CRC | Higher ORR & PFS in liver mets | Increased efficacy but higher toxicity |
| TGF-β + PD-L1 blockade[158-160] | Fibrosis reversal, stromal remodeling | Bintrafusp alfa trials | Mixed metastatic | Disease control approximately 10%-15% | Promising mechanism; modest efficacy |
| CSF1R/CCR2 blockade + PD-1[162] | Myeloid reprogramming | Ongoing phase I/II | CRC, NSCLC | Preclinical synergy with PD-1 | Pending translation |
| Epigenetic modulation + ICI[163] | Antigen presentation, viral mimicry | Early-phase trials | Multiple cancers | MHC expression & T-cell infiltration increase | Converts cold to hot lesions |
| Adoptive cell therapy[166] | Enhanced intrahepatic trafficking | Hepatic artery CAR-T delivery | CRC, HCC | Feasible; improved local homing | Useful for liver-limited disease |
Similarly, in CheckMate 9 LA, dual checkpoint blockade with nivolumab plus ipilimumab alongside chemotherapy provided durable OS benefits across metastatic subgroups, including those with liver involvement (Table 3)[92]. At five years, the combination achieved a 18% OS rate vs 11% with chemotherapy alone, with consistent but numerically lower survival in the hepatic metastasis cohort (Table 2)[93]. Together, these findings indicate that although liver metastases confer immunotherapy resistance and attenuated efficacy, the integration of anti-angiogenic or dual checkpoint blockade strategies may partially restore responsiveness and improve outcomes in this challenging subset.
Immunotherapy has revolutionized the treatment of advanced melanoma, with anti-PD-1 monotherapy achieving ORRs of 30%-40% and combination anti-PD-1/CTLA-4 therapy reaching approximately 50% in some settings (Table 2)[94,95]. However, liver metastases significantly dampen these responses[94].
In 2017, Tumeh et al[94] demonstrated that melanoma patients with liver metastases had markedly reduced CD8+ T-cell infiltration and experienced inferior responses to anti-PD-1 therapy compared to those without liver involvement. Although the specific ORR numbers in that study represent pooled analyses rather than a single randomized trial, the correlation between poor CD8+ infiltration and reduced clinical benefit is robust[94]. There is no dedicated meta-analysis exclusively focused on melanoma liver metastases, but pan-cancer analyses consistently identify liver metastasis as a negative predictive factor for ICI efficacy across tumor types, including melanoma[78].
A recent multi-organ genomic and transcriptomic analysis of therapy-resistant melanoma further substantiates the biological basis of this clinical resistance[96]. In rapid autopsy specimens, liver metastases displayed a distinct immune-desert phenotype characterized by CD8+ T-cell depletion, type-2-skewed immunity, and profound T-cell exhaustion[96]. Compared with other metastatic sites, hepatic lesions showed frequent B2M, CDKN2A, and JAK2 loss-alterations ass
Approximately 50%-60% of patients with CRC develop liver metastases during their disease course[97,98]. Standard sys
Oligometastasis represents an intermediate state between localized and widespread disease, defined by a limited number of metastatic lesions amenable to curative local therapy[101]. This state is particularly common in CRC because venous drainage from the colon and rectum flows directly into the portal circulation, making the liver the predominant and often the only site of metastasis, found as the sole metastatic site in about one-third of patients at autopsy[102]. Approximately 20%-30% of CRC patients with liver metastases are candidates for resection, which remains the only established curative option, achieving five-year OS rates of 45%-60%, recurrence-free survival of 22%-36%, and long-term cure in 15%-25% of selected patients[103]. Ablative therapies such as stereotactic body radiation therapy, radiofrequency ablation (RFA), cryoablation, and yttrium-90 radioembolization provide effective alternatives for patients who are not surgical candidates, achieving five-year OS rates of 30%-46% and excellent local control for small lesions[104]. The curative potential of liver-only oligometastases is largely unique to CRC, reflecting its slower progression, frequent liver-limited spread, and immune-enriched/desmoplastic microenvironment, features not seen in more aggressive primaries such as pancreatic, lung, or gastric cancers, where liver involvement typically signifies systemic, incurable disease[105].
In KEYNOTE-177, pembrolizumab significantly improved PFS compared to chemotherapy in MSI-H/dMMR meta
Pancreatic ductal adenocarcinoma (PDAC) exhibits profound desmoplasia that extends to its liver metastases, where metastatic cells reprogram hepatic stellate (Ito) cells into α-SMA+ myofibroblasts via TGF-β, PDGF, and FGF2 signaling, driving dense collagen- and fibronectin-rich fibrosis that mirrors the primary tumor’s stroma[109,110]. This fibrosis establishes a physical and immunologic barrier to drug delivery and T-cell infiltration, reinforced by feedback loops bet
Gastric cancer appears to be less dependent on liver metastasis status for its response to immunotherapy than several other tumor types[77]. A proposed mechanism is the presence of a more pro-inflammatory TME in gastric malignancies. Supporting this, Groen-van Schooten et al[126] demonstrated significantly increased proliferating CD4+ T helper and CD8+ cytotoxic T cells, along with decreased monocytic MDSCs, in tumors located in the stomach and gastroesophageal junction (GEJ) compared with esophageal adenocarcinoma (Table 2)[126].
In gastric cancer, the JAVELIN 100 trial failed to show improvements in OS or PFS with maintenance immunotherapy compared with maintenance chemotherapy (Table 2)[127,128]. Consequently, clinical trial efforts have shifted toward evaluating immunotherapy in combination with chemotherapy, which has shown more promising outcomes.
A meta-analysis by Pu et al[128] of patients with advanced, HER2-negative gastric and GEJ cancers found that com
A separate meta-analysis by Tian et al[77] further confirmed that liver metastases exert a smaller negative effect on immunotherapy efficacy in gastric and GEJ cancers compared with other tumor types. Instead, peritoneal metastases and the total number of metastatic sites were more strongly correlated with decreased OS. Similarly, a retrospective cohort study by Liang et al[131] reported no significant difference in OS for patients with gastric cancer receiving immunotherapy with or without liver metastases (13.43 months vs 10.53 months; P = 0.584). In this study, MMR mutations (HR = 2.31, 95%CI: 1.09-25.9), metastases involving more than three sites (HR = 2.24, 95%CI: 1.00-5.01), and peritoneal met
Collectively, these findings indicate that combined chemotherapy and immunotherapy represents a viable treatment option for patients with gastric and GEJ cancers, regardless of liver metastasis status, while other metastatic characteristics, particularly peritoneal involvement and overall metastatic burden, play a more decisive role in determining pro
With evidence that show liver metastases strongly associated with poor response to immunotherapy, several therapeutic strategies have been designed to counteract this resistance by targeting specific pathways and immune barriers.
A well-established way to modulate the hepatic microenvironment is through VEGF blockade. VEGF drives abnormal, leaky vasculature in liver metastases and recruits immunosuppressive myeloid cells[132-135]. The addition of the anti-VEGF antibody bevacizumab to immunotherapy normalizes vasculature, enhances cytotoxic T cell trafficking and red
In patients with liver metastases, clinical and translational studies have shown that adding anti-VEGF therapy to ICIs improves immune cell recruitment and disease control compared to ICI monotherapy[137,138]. Clinical studies eva
Dual checkpoint inhibition targeting CTLA-4 and PD-1/PD-L1 pathways addresses two distinct phases of T-cell dys
Retrospective and prospective analyses across multiple tumor types including melanoma, hepatocellular carcinoma, and CRC demonstrate that dual immune checkpoint blockade (CTLA-4 plus PD-1/PD-L1 inhibition) yields higher ORR and improved PFS in patients with liver metastases compared to monotherapy (Table 2)[143-145]. Mechanistic studies in murine models of liver metastasis show that CTLA-4 blockade preferentially expands tissue-resident effector memory CD4+ and CD8+ T-cells and reduces intrahepatic Tregs, while PD-1/PD-L1 blockade restores the cytotoxic function of exhausted T-cells within the metastatic niche[144,146]. The combination leads to increased intratumoral infiltration of CD8+ and CD4+ T-cells, enhanced Th1/M1 cytokine profiles, and a reduction in immunosuppressive myeloid and regu
Despite encouraging signals, the benefit of dual ICIs in liver metastases remains modest in magnitude[148,149]. The main challenges are hepatotoxicity and systemic immune-related adverse events, which are more frequent when both checkpoints are inhibited (Table 2)[150,151]. To mitigate toxicity, newer regimens such as “priming” with a single low-dose CTLA-4 antibody followed by PD-1/PD-L1 maintenance are under evaluation in ongoing phase II trials specifically enrolling patients with liver-dominant metastases[152-154].
TGF-β plays a central role in shaping the fibrotic and immune-excluded phenotype of liver metastases[62]. It drives HSC activation, ECM deposition, and conversion of KCs into M2-like macrophages[155]. Therapeutic targeting of this pathway aims to dismantle the fibrotic stroma and restore immune access to the tumor core (Table 3)[156,157].
Dual TGF-β/PD-L1 blockade has shown promising mechanistic results. Agents such as bintrafusp alfa colocalizes a TGF-β “trap” to the TME via PD-L1 binding, resulting in more effective local TGF-β sequestration and simultaneous checkpoint inhibition (Table 2)[158-160]. This approach enhances antitumor immunity in liver metastases by increasing T-cell infiltration, reducing immunosuppressive myeloid activity, and downregulating pathways such as EMT and fibrosis, thereby overcoming resistance to immunotherapy[159,160]. Although clinical responses remain modest, with disease control rates around 10%-15% in heavily pretreated cohorts, these studies validate the biological relevance of TGF-β in liver-specific immune resistance (Table 3)[160].
Alternative approaches include selective inhibition of TGF-β receptor I kinase (e.g., galunisertib) and macrophage reprogramming agents targeting CSF1R or CCR2 signaling to reduce immunosuppressive myeloid infiltration[161]. Preclinical liver metastasis models show that such inhibitors synergize with PD-1 blockade to restore CD8+ cytotoxic function and diminish fibrosis. Clinical translation is ongoing, with several phase I/II trials testing these combinations specifically in metastatic colorectal or lung cancers with liver involvement[162].
Epigenetic modulation represents a frontier strategy to enhance tumor immunogenicity in the liver microenvironment. DNA methyltransferase and histone deacetylase inhibitors can upregulate antigen-presentation machinery (e.g., MHC class I, β2-microglobulin) and reactivate endogenous retroviral elements, leading to “viral mimicry” and interferon sig
Similarly, adoptive cell therapies, such as chimeric antigen receptor-T (CAR-T) or T cell receptor-engineered T-cells face unique trafficking and survival barriers within the liver (Table 2)[166]. New strategies utilize chemokine receptor engineering (e.g., CXCR3, CXCR6) to improve intrahepatic homing, and metabolic reprogramming to resist TGF-β-mediated exhaustion. Trials delivering CAR-T-cells intra-arterially via the hepatic artery are underway to bypass the sinusoidal barrier and achieve higher local concentrations (Table 2)[167,168].
Ablative therapies-including RFA, microwave ablation (MWA), cryoablation, and stereotactic body radiotherapy (SBRT)-offer effective local control for liver metastases across multiple primary cancers, with particularly strong evidence in CRC (Table 4)[169]. For small, oligometastatic CRLM, ablation can be potentially curative, achieving local control rates of 80%-95% and OS comparable to surgical resection (Table 3)[170]. The phase III COLLISION trial confirmed non-inferiority of ablation vs surgery for OS (HR = 1.05, P = 0.83) and local control, while the CLOCC trial demonstrated a durable OS benefit when RFA was added to systemic therapy in unresectable CRLM (8-year OS 35.9% vs 8.9%, HR = 0.58, P = 0.01; Table 3)[171,172]. For other primaries (breast, lung, neuroendocrine), ablation provides high local control (≥ 85%) and symptom palliation, though no randomized data confirm an OS benefit[173]. SBRT achieves local control rates of 83%-94% for small lesions (< 3 cm) with minimal grade ≥ 3 toxicity (< 10%), and MWA may outperform RFA for perivascular tumors due to reduced heat-sink effects (Table 3)[174,175]. Emerging strategies combine ablation with ICIs to augment systemic immunity[176]. In hepatocellular carcinoma, IMbrave050 and Qin et al[177] and Zhu et al[178] trials showed improved recurrence-free or OS with ablation + ICI compared to ICI alone, though OS benefit remains unconfirmed in long-term follow-up (Table 3). In contrast, the EORTC-1560 ILOC trial in CRC failed to demonstrate responses in untreated metastases, suggesting liver-specific immune tolerance limits efficacy[179]. Overall, ablation offers excellent local disease control and safety across cancer types, potential cure for limited CRLM, and serves as a promising immu
| Technique | Typical local control rate | Any OS benefit in RCTs | Comments |
| RFA[169] | 80%-95% (CRLM) | Yes (CLOCC: 8-year OS 35.9% vs 8.9%, HR = 0.58, P = 0.01) | Effective for small, oligometastatic CRLM; potentially curative; added to systemic therapy in unresectable CRLM |
| MWA[174,175] | ≥ 85% (various primaries) | Not reported | May outperform RFA for perivascular tumors due to reduced heat-sink effects |
| Cryoablation[169] | ≥ 85% (various primaries) | Not reported | High local control for breast, lung, neuroendocrine metastases; symptom palliation |
| SBRT[169] | 83%-94% (small lesions < 3 cm) | Not reported | Minimal grade ≥ 3 toxicity (< 10%) |
| Ablation (general)[171,172] | 80%-95% (CRLM) | Yes (collision: HR = 1.05, P = 0.83, non-inferior to surgery) | Non-inferior to surgery for OS and local control in CRLM; promising with ICI |
| Ablation + ICI[177,178] | Not specified | Yes (IMbrave050: Improved RFS/OS vs ICI alone) | Improved RFS/OS in HCC; unconfirmed long-term OS benefit; EORTC-1560 ILOC showed no response in untreated metastases |
Despite encouraging mechanistic progress, the clinical translation of these strategies faces several barriers. First, biomarker validation remains limited: Traditional markers such as PD-L1 expression or tumor mutational burden fail to predict ICI response in hepatic metastases[45,94]. Emerging tissue, and blood-based biomarkers such as CD8+ T-cell density, interferon-γ-related gene signatures, and post-treatment reduction in exhausted T-cell subsets (PD-1+ TIM-3+, TIGIT+) show promise but require standardization[181].
Second, trial design complexity impedes progress. Most ICI studies stratify by primary tumor type rather than by metastatic organ site, diluting the statistical power to detect liver-specific effects. Dedicated liver-metastasis cohorts or platform trials incorporating multi-omic profiling could clarify which patients benefit from combination strategies.
Finally, safety and tolerability pose practical challenges. The liver’s dual role as a metabolic and immune organ heightens the risk of immune-related hepatotoxicity, particularly with dual checkpoint or TGF-β-targeting regimens[182-184]. Rational sequencing, such as using VEGF or radiation “priming” before full systemic immunotherapy may help mitigate toxicity while maximizing efficacy[185].
Liver metastases represent a biologically distinct and clinically formidable barrier to effective immunotherapy. Their resistance arises not from tumor-intrinsic factors alone, but from the liver’s unique immune architecture, which favors tolerance over inflammation. Throughout this review, we have highlighted how hepatic-specific features such as sinusoidal structure, KC-mediated T-cell deletion, and stellate cell-driven fibrosis collectively generates a profoundly immunosuppressive niche that limits the efficacy of ICIs across multiple tumor types. Mechanistic and clinical evidence converge on a key insight: The liver does not merely host metastases but actively reprograms immune dynamics systemically. This realization has reframed therapeutic development from conventional ICI monotherapy toward strategies that remodel the hepatic microenvironment itself. Approaches such as VEGF inhibition, dual checkpoint blockade, and liver-directed radiation aim to normalize vasculature, restore cytotoxic T-cell trafficking, and reduce myeloid-mediated suppression. Meanwhile, next-generation interventions like targeting TGF-β signaling, reprogramming suppressive macrophages, and employing epigenetic or adoptive cellular therapies are redefining how immune exclusion in liver metastases can be reversed. Despite promising preclinical data, clinical translation remains limited by heterogeneous trial designs, lack of validated biomarkers, and concerns over hepatotoxicity. The path forward will require mechanism-guided trials specifically stratified by hepatic involvement, integration of multi-omic profiling to capture immune and stromal evolution, and rational sequencing of locoregional and systemic therapies. Ultimately, overcoming immunotherapy resistance in liver metastases demands a paradigm shift from treating the liver as a passive site of disease to understanding it as an active immune organ that must itself be therapeutically reconditioned. Success will depend on the ability to integrate immunologic, vascular, and stromal modulation into unified treatment strategies that transform the hepatic niche from a site of immune privilege control.
| 1. | Xu W, Xu J, Liu J, Wang N, Zhou L, Guo J. Liver Metastasis in Cancer: Molecular Mechanisms and Management. MedComm (2020). 2025;6:e70119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 2. | Zeng X, Ward SE, Zhou J, Cheng ASL. Liver Immune Microenvironment and Metastasis from Colorectal Cancer-Pathogenesis and Therapeutic Perspectives. Cancers (Basel). 2021;13:2418. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 49] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 3. | Gao Y, Chen S, Wang H, Wu C, An R, Li G, Yang M, Zhou Y, Zhou Y, Xie X, Yu H, Zhang J. Liver metastases across cancer types sharing tumor environment immunotolerance can impede immune response therapy and immune monitoring. J Adv Res. 2024;61:151-164. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 19] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 4. | Dai M, Tiu BY, Chen M, Sing C, Sharma V, Hoyos JP, Scherr R, Lin WJ, Breuer JA, Dayyani F, Imagawa D, Abi-Jaoudeh N. Novel combination approaches to locoregional and systemic therapy in the management of primary and metastatic liver tumors. Clin Adv Hematol Oncol. 2023;21:633-643. [PubMed] |
| 5. | He B, Ganss R. Modulation of the Vascular-Immune Environment in Metastatic Cancer. Cancers (Basel). 2021;13:810. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 6. | Ciner AT, Jones K, Muschel RJ, Brodt P. The unique immune microenvironment of liver metastases: Challenges and opportunities. Semin Cancer Biol. 2021;71:143-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 47] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 7. | Valenzuela-Fuenzalida JJ, Rodríguez-Osorio B, Salgado-Torres C, Nova-Baeza P, Orellana-Donoso M, Bruna-Mejías A, Avila-Sepulveda R, Becerra-Farfán Á, Rodríguez-Luengo M, Sanchis-Gimeno J, Granite G, Gutiérrez-Espinoza H. A systematic review and meta-analysis: prevalence and clinical implications of anatomical variants of the hepatic portal vein. Sci Rep. 2024;14:30002. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 8. | Eipel C, Abshagen K, Vollmar B. Regulation of hepatic blood flow: the hepatic arterial buffer response revisited. World J Gastroenterol. 2010;16:6046-6057. [PubMed] [DOI] [Full Text] |
| 9. | Zheng M, Tian Z. Liver-Mediated Adaptive Immune Tolerance. Front Immunol. 2019;10:2525. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 197] [Cited by in RCA: 177] [Article Influence: 25.3] [Reference Citation Analysis (1)] |
| 10. | Wu K, Zhang G, Shen C, Zhu L, Yu C, Sartorius K, Ding W, Jiang Y, Lu Y. Role of T cells in liver metastasis. Cell Death Dis. 2024;15:341. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 11. | Mahdi Z, Ettel MG, Gonzalez RS, Hart J, Alpert L, Fang J, Liu N, Hammer ST, Panarelli N, Cheng J, Greenson JK, Swanson PE, Westerhoff M. Metastases can occur in cirrhotic livers with patent portal veins. Diagn Pathol. 2021;16:18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 12. | Seymour K, Charnley RM. Evidence that metastasis is less common in cirrhotic than normal liver: a systematic review of post-mortem case-control studies. Br J Surg. 1999;86:1237-1242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 44] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 13. | Wilkinson AL, Qurashi M, Shetty S. The Role of Sinusoidal Endothelial Cells in the Axis of Inflammation and Cancer Within the Liver. Front Physiol. 2020;11:990. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 112] [Cited by in RCA: 98] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 14. | Yang M, Zhang C. The role of liver sinusoidal endothelial cells in cancer liver metastasis. Am J Cancer Res. 2021;11:1845-1860. [PubMed] |
| 15. | Shetty S, Lalor PF, Adams DH. Liver sinusoidal endothelial cells - gatekeepers of hepatic immunity. Nat Rev Gastroenterol Hepatol. 2018;15:555-567. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 444] [Cited by in RCA: 387] [Article Influence: 48.4] [Reference Citation Analysis (1)] |
| 16. | Ruan B, Duan JL, Xu H, Tao KS, Han H, Dou GR, Wang L. Capillarized Liver Sinusoidal Endothelial Cells Undergo Partial Endothelial-Mesenchymal Transition to Actively Deposit Sinusoidal ECM in Liver Fibrosis. Front Cell Dev Biol. 2021;9:671081. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 51] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 17. | Qu J, Wang L, Li Y, Li X. Liver sinusoidal endothelial cell: An important yet often overlooked player in the liver fibrosis. Clin Mol Hepatol. 2024;30:303-325. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 38] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 18. | Jungermann K, Kietzmann T. Oxygen: modulator of metabolic zonation and disease of the liver. Hepatology. 2000;31:255-260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 365] [Cited by in RCA: 307] [Article Influence: 11.8] [Reference Citation Analysis (3)] |
| 19. | Chen C, Wang Z, Ding Y, Qin Y. Tumor microenvironment-mediated immune evasion in hepatocellular carcinoma. Front Immunol. 2023;14:1133308. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 241] [Cited by in RCA: 221] [Article Influence: 73.7] [Reference Citation Analysis (0)] |
| 20. | Loo JM, Scherl A, Nguyen A, Man FY, Weinberg E, Zeng Z, Saltz L, Paty PB, Tavazoie SF. Extracellular metabolic energetics can promote cancer progression. Cell. 2015;160:393-406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 231] [Cited by in RCA: 317] [Article Influence: 28.8] [Reference Citation Analysis (0)] |
| 21. | Dupuy F, Tabariès S, Andrzejewski S, Dong Z, Blagih J, Annis MG, Omeroglu A, Gao D, Leung S, Amir E, Clemons M, Aguilar-Mahecha A, Basik M, Vincent EE, St-Pierre J, Jones RG, Siegel PM. PDK1-Dependent Metabolic Reprogramming Dictates Metastatic Potential in Breast Cancer. Cell Metab. 2015;22:577-589. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 356] [Cited by in RCA: 457] [Article Influence: 41.5] [Reference Citation Analysis (0)] |
| 22. | Liu Y, Cao X. Characteristics and Significance of the Pre-metastatic Niche. Cancer Cell. 2016;30:668-681. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1047] [Cited by in RCA: 925] [Article Influence: 92.5] [Reference Citation Analysis (3)] |
| 23. | Wang Y, Jia J, Wang F, Fang Y, Yang Y, Zhou Q, Yuan W, Gu X, Hu J, Yang S. Pre-metastatic niche: formation, characteristics and therapeutic implication. Signal Transduct Target Ther. 2024;9:236. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 155] [Cited by in RCA: 152] [Article Influence: 76.0] [Reference Citation Analysis (4)] |
| 24. | Ormseth B, Onuma A, Zhang H, Tsung A. The Hepatic Pre-Metastatic Niche. Cancers (Basel). 2022;14:3731. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 25. | Yu Z, Zhao S, Ren L, Wang L, Chen Z, Hoffman RM, Zhou J. Pancreatic cancer-derived exosomes promote tumor metastasis and liver pre-metastatic niche formation. Oncotarget. 2017;8:63461-63483. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 105] [Cited by in RCA: 107] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 26. | Ma C, Zhang Q, Greten TF. MDSCs in liver cancer: A critical tumor-promoting player and a potential therapeutic target. Cell Immunol. 2021;361:104295. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 66] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 27. | Trehan R, Huang P, Zhu XB, Wang X, Soliman M, Strepay D, Nur A, Kedei N, Arhin M, Ghabra S, Rodríguez-Matos F, Benmebarek MR, Ma C, Korangy F, Greten TF. SPP1 + macrophages cause exhaustion of tumor-specific T cells in liver metastases. Nat Commun. 2025;16:4242. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 26] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 28. | Gottwick C, Carambia A, Herkel J. Harnessing the liver to induce antigen-specific immune tolerance. Semin Immunopathol. 2022;44:475-484. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 24] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 29. | Lee JC, Green MD, Huppert LA, Chow C, Pierce RH, Daud AI. The Liver-Immunity Nexus and Cancer Immunotherapy. Clin Cancer Res. 2022;28:5-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 95] [Article Influence: 23.8] [Reference Citation Analysis (0)] |
| 30. | Lin J, Rao D, Zhang M, Gao Q. Metabolic reprogramming in the tumor microenvironment of liver cancer. J Hematol Oncol. 2024;17:6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 164] [Cited by in RCA: 164] [Article Influence: 82.0] [Reference Citation Analysis (1)] |
| 31. | Le XF, Merchant O, Bast RC, Calin GA. The Roles of MicroRNAs in the Cancer Invasion-Metastasis Cascade. Cancer Microenviron. 2010;3:137-147. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 78] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 32. | Milette S, Sicklick JK, Lowy AM, Brodt P. Molecular Pathways: Targeting the Microenvironment of Liver Metastases. Clin Cancer Res. 2017;23:6390-6399. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 98] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 33. | Shenoy AK, Lu J. Cancer cells remodel themselves and vasculature to overcome the endothelial barrier. Cancer Lett. 2016;380:534-544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 69] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 34. | Ring A, Nguyen-Sträuli BD, Wicki A, Aceto N. Biology, vulnerabilities and clinical applications of circulating tumour cells. Nat Rev Cancer. 2023;23:95-111. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 288] [Cited by in RCA: 250] [Article Influence: 83.3] [Reference Citation Analysis (2)] |
| 35. | Raskov H, Orhan A, Agerbæk MØ, Gögenur I. The impact of platelets on the metastatic potential of tumour cells. Heliyon. 2024;10:e34361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 36. | Auguste P, Fallavollita L, Wang N, Burnier J, Bikfalvi A, Brodt P. The host inflammatory response promotes liver metastasis by increasing tumor cell arrest and extravasation. Am J Pathol. 2007;170:1781-1792. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 131] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 37. | Benedicto A, Herrero A, Romayor I, Marquez J, Smedsrød B, Olaso E, Arteta B. Liver sinusoidal endothelial cell ICAM-1 mediated tumor/endothelial crosstalk drives the development of liver metastasis by initiating inflammatory and angiogenic responses. Sci Rep. 2019;9:13111. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 64] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 38. | Benedicto A, Romayor I, Arteta B. Role of liver ICAM-1 in metastasis. Oncol Lett. 2017;14:3883-3892. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 81] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 39. | Liu W, Zhou X, Yao Q, Chen C, Zhang Q, Ding K, Li L, Zeng Z. In situ expansion and reprogramming of Kupffer cells elicit potent tumoricidal immunity against liver metastasis. J Clin Invest. 2023;133:e157937. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 42] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 40. | Deng Z, Loyher PL, Lazarov T, Li L, Shen Z, Bhinder B, Yang H, Zhong Y, Alberdi A, Massague J, Sun JC, Benezra R, Glass CK, Elemento O, Iacobuzio-Donahue CA, Geissmann F. The nuclear factor ID3 endows macrophages with a potent anti-tumour activity. Nature. 2024;626:864-873. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 59] [Article Influence: 29.5] [Reference Citation Analysis (0)] |
| 41. | Tian P, Wu Q, He D, Zhao W, Luo L, Jia Z, Luo W, Lv X, Liu Y, Wang Y, Wang Q, Zhang P, Liang Y, Yang Q, Hu G. Breast cancer induces CD62L(+) Kupffer cells via DMBT1 to promote neutrophil extracellular trap formation and liver metastasis. Cell Discov. 2025;11:68. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 42. | Liu Y, Zhai Y, Zhang Y, Song L, Zhang H, Cao J, Zhao S, Wu Y, Liang R, Zhu R, Wang W, Sun Y. High metastatic tumor-derived CXCL16 mediates liver colonization metastasis by inducing Kupffer cell polarization via the PI3K/AKT/FOXO3a pathway. Neoplasia. 2025;65:101174. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 43. | Yin Y, Feng W, Chen J, Chen X, Wang G, Wang S, Xu X, Nie Y, Fan D, Wu K, Xia L. Immunosuppressive tumor microenvironment in the progression, metastasis, and therapy of hepatocellular carcinoma: from bench to bedside. Exp Hematol Oncol. 2024;13:72. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 78] [Article Influence: 39.0] [Reference Citation Analysis (6)] |
| 44. | Arvanitakis K, Koletsa T, Mitroulis I, Germanidis G. Tumor-Associated Macrophages in Hepatocellular Carcinoma Pathogenesis, Prognosis and Therapy. Cancers (Basel). 2022;14:226. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 103] [Article Influence: 25.8] [Reference Citation Analysis (0)] |
| 45. | Yu J, Green MD, Li S, Sun Y, Journey SN, Choi JE, Rizvi SM, Qin A, Waninger JJ, Lang X, Chopra Z, El Naqa I, Zhou J, Bian Y, Jiang L, Tezel A, Skvarce J, Achar RK, Sitto M, Rosen BS, Su F, Narayanan SP, Cao X, Wei S, Szeliga W, Vatan L, Mayo C, Morgan MA, Schonewolf CA, Cuneo K, Kryczek I, Ma VT, Lao CD, Lawrence TS, Ramnath N, Wen F, Chinnaiyan AM, Cieslik M, Alva A, Zou W. Liver metastasis restrains immunotherapy efficacy via macrophage-mediated T cell elimination. Nat Med. 2021;27:152-164. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 870] [Cited by in RCA: 809] [Article Influence: 161.8] [Reference Citation Analysis (5)] |
| 46. | Thomas SK, Wattenberg MM, Choi-Bose S, Uhlik M, Harrison B, Coho H, Cassella CR, Stone ML, Patel D, Markowitz K, Delman D, Chisamore M, Drees J, Bose N, Beatty GL. Kupffer cells prevent pancreatic ductal adenocarcinoma metastasis to the liver in mice. Nat Commun. 2023;14:6330. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 38] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 47. | Czyzynska-Cichon I, Kotlinowski J, Blacharczyk O, Giergiel M, Szymanowski K, Metwally S, Wojnar-Lason K, Dobosz E, Koziel J, Lekka M, Chlopicki S, Zapotoczny B. Early and late phases of liver sinusoidal endothelial cell (LSEC) defenestration in mouse model of systemic inflammation. Cell Mol Biol Lett. 2024;29:139. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 48. | Deleve LD, Wang X, Guo Y. Sinusoidal endothelial cells prevent rat stellate cell activation and promote reversion to quiescence. Hepatology. 2008;48:920-930. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 355] [Cited by in RCA: 312] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 49. | Huu Hoang T, Sato-Matsubara M, Yuasa H, Matsubara T, Thuy LTT, Ikenaga H, Phuong DM, Hanh NV, Hieu VN, Hoang DV, Hai H, Okina Y, Enomoto M, Tamori A, Daikoku A, Urushima H, Ikeda K, Dat NQ, Yasui Y, Shinkawa H, Kubo S, Yamagishi R, Ohtani N, Yoshizato K, Gracia-Sancho J, Kawada N. Cancer cells produce liver metastasis via gap formation in sinusoidal endothelial cells through proinflammatory paracrine mechanisms. Sci Adv. 2022;8:eabo5525. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 50. | Fabregat I, Caballero-Díaz D. Transforming Growth Factor-β-Induced Cell Plasticity in Liver Fibrosis and Hepatocarcinogenesis. Front Oncol. 2018;8:357. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 305] [Cited by in RCA: 283] [Article Influence: 35.4] [Reference Citation Analysis (0)] |
| 51. | Izutsu R, Osaki M, Seong H, Sato R, Okada F. Hepatic Stellate Cells Activated by Cancer Cell-derived AMIGO2-containing Small Extracellular Vesicles Promote Cancer Cell Migration by Producing IL-8. Anticancer Res. 2025;45:1435-1446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 52. | Zhang W, Xu C. Exosomal SphK1 from colorectal cancer cells promotes cancer cell migration and activates hepatic stellate cells. Mol Med Rep. 2025;31:73. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 53. | Yang C, Gao Z, Tang R, Zhou L, Zhou P, Shi W, Ren D, Chen H, Zhang Z, Xie X, Lin J, Ye Y, Feng Z, Feng X, He Y, Zhi Y, Liu D, Zhang X, Jiang L. POU6F2 promotes liver metastasis of gastric adenocarcinoma by dual mechanism of transcriptional upregulation of SNAI1 and IGF2/PI3K/AKT signaling-induced conversion of hepatic stellate cells into cancer-associated fibroblasts. Br J Cancer. 2025;133:14-26. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 54. | Li D, Gao Z, Zhang Z, Chen H, Tang R, Zhou L, Ye Y, Lin J, Zhou P, Wang C, Feng X, He Y, Meng Z, Zheng M, Lu W, Feng Z, Wang L, Pei Y, Yang J, Tao T, Zhang X, Jiang L. Suprabasin promotes gastric cancer liver metastasis via hepatic stellate cells-mediated EGF/CCL2/JAK2 intercellular signaling pathways. Oncogene. 2025;44:1975-1989. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 55. | Nater M, Brügger M, Cecconi V, Pereira P, Forni G, Köksal H, Dimakou D, Herbst M, Calvanese AL, Lucchiari G, Schneider C, Valenta T, van den Broek M. Hepatic iNKT cells facilitate colorectal cancer metastasis by inducing a fibrotic niche in the liver. iScience. 2025;28:112364. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 56. | Yoong KF, Afford SC, Randhawa S, Hubscher SG, Adams DH. Fas/Fas ligand interaction in human colorectal hepatic metastases: A mechanism of hepatocyte destruction to facilitate local tumor invasion. Am J Pathol. 1999;154:693-703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 76] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 57. | Kawamoto K, Onodera H, Kan S, Kondo S, Imamura M. Possible paracrine mechanism of insulin-like growth factor-2 in the development of liver metastases from colorectal carcinoma. Cancer. 1999;85:18-25. [PubMed] [DOI] [Full Text] |
| 58. | Huang CY, Zhou QY, Hu Y, Wen Y, Qiu ZW, Liang MG, Mo JL, Xu JH, Sun C, Liu FB, Chen XL. Hepatocyte growth factor is a prognostic marker in patients with colorectal cancer: a meta-analysis. Oncotarget. 2017;8:23459-23469. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 59. | Lee JW, Stone ML, Porrett PM, Thomas SK, Komar CA, Li JH, Delman D, Graham K, Gladney WL, Hua X, Black TA, Chien AL, Majmundar KS, Thompson JC, Yee SS, O'Hara MH, Aggarwal C, Xin D, Shaked A, Gao M, Liu D, Borad MJ, Ramanathan RK, Carpenter EL, Ji A, de Beer MC, de Beer FC, Webb NR, Beatty GL. Hepatocytes direct the formation of a pro-metastatic niche in the liver. Nature. 2019;567:249-252. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 343] [Cited by in RCA: 311] [Article Influence: 44.4] [Reference Citation Analysis (4)] |
| 60. | Sun X, Wu J, Liu L, Chen Y, Tang Y, Liu S, Chen H, Jiang Y, Liu Y, Yuan H, Lu Y, Chen Z, Cai J. Transcriptional switch of hepatocytes initiates macrophage recruitment and T-cell suppression in endotoxemia. J Hepatol. 2022;77:436-452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 46] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 61. | Xi F, Sun H, Peng H, Lian Z, Wei H, Tian Z, Sun R, Chen Y. Hepatocyte-derived FGL1 accelerates liver metastasis and tumor growth by inhibiting CD8+ T and NK cells. JCI Insight. 2024;9:e173215. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (4)] |
| 62. | Marvin DL, Heijboer R, Ten Dijke P, Ritsma L. TGF-β signaling in liver metastasis. Clin Transl Med. 2020;10:e160. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 43] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 63. | Shiri AM, Zhang T, Bedke T, Zazara DE, Zhao L, Lücke J, Sabihi M, Fazio A, Zhang S, Tauriello DVF, Batlle E, Steglich B, Kempski J, Agalioti T, Nawrocki M, Xu Y, Riecken K, Liebold I, Brockmann L, Konczalla L, Bosurgi L, Mercanoglu B, Seeger P, Küsters N, Lykoudis PM, Heumann A, Arck PC, Fehse B, Busch P, Grotelüschen R, Mann O, Izbicki JR, Hackert T, Flavell RA, Gagliani N, Giannou AD, Huber S. IL-10 dampens antitumor immunity and promotes liver metastasis via PD-L1 induction. J Hepatol. 2024;80:634-644. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 105] [Article Influence: 52.5] [Reference Citation Analysis (0)] |
| 64. | Shimabukuro-Vornhagen A, Draube A, Liebig TM, Rothe A, Kochanek M, von Bergwelt-Baildon MS. The immunosuppressive factors IL-10, TGF-β, and VEGF do not affect the antigen-presenting function of CD40-activated B cells. J Exp Clin Cancer Res. 2012;31:47. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 43] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 65. | Weis SM, Cheresh DA. Pathophysiological consequences of VEGF-induced vascular permeability. Nature. 2005;437:497-504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 752] [Cited by in RCA: 692] [Article Influence: 33.0] [Reference Citation Analysis (0)] |
| 66. | Zhang Y, Yang X, Zhang S, Huang Q, Liu S, Qiu L, Wei M, Deng X, Meng W, Chen HN, Zhang Y, Han J, Wang Z. MicroRNA-6084 orchestrates angiogenesis and liver metastasis in colorectal cancer via extracellular vesicles. JCI Insight. 2025;10:e189503. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 67. | Yang L, Zhu H, Chen L, Zhang J, Huang J, Zeng L, Zong Z. Exosomal miR-382-5p prevents pre-metastatic niche formation by inhibiting GPR176/GNAS-CXCR1/CXCR2 axis in colorectal cancer liver metastasis. Cell Signal. 2025;134:111963. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 68. | Zhang LP, Zhang ZG, Guan J, Li LQ. Thrombospondin 2 drives liver metastasis in skin cutaneous melanoma via regulation of angiogenesis and extracellular matrix remodeling. Melanoma Res. 2025;35:306-316. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 69. | Zhou S, Zhao Z, Zhong H, Ren Z, Li Y, Wang H, Qiu Y. The role of myeloid-derived suppressor cells in liver cancer. Discov Oncol. 2023;14:77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 21] [Reference Citation Analysis (0)] |
| 70. | Groth C, Hu X, Weber R, Fleming V, Altevogt P, Utikal J, Umansky V. Immunosuppression mediated by myeloid-derived suppressor cells (MDSCs) during tumour progression. Br J Cancer. 2019;120:16-25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 721] [Cited by in RCA: 676] [Article Influence: 96.6] [Reference Citation Analysis (4)] |
| 71. | Ireland AS, Oliver TG. Neutrophils Create an ImpeNETrable Shield between Tumor and Cytotoxic Immune Cells. Immunity. 2020;52:729-731. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 42] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 72. | Zhang J, Miao C, Zhang H. Targeting neutrophil extracellular traps in cancer progression and metastasis. Theranostics. 2025;15:5846-5869. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 73. | Jiang W, Guan B, Sun H, Mi Y, Cai S, Wan R, Li X, Lian P, Li D, Zhao S. WNT11 Promotes immune evasion and resistance to Anti-PD-1 therapy in liver metastasis. Nat Commun. 2025;16:1429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 74. | Ho WJ, Erbe R, Danilova L, Phyo Z, Bigelow E, Stein-O'Brien G, Thomas DL 2nd, Charmsaz S, Gross N, Woolman S, Cruz K, Munday RM, Zaidi N, Armstrong TD, Sztein MB, Yarchoan M, Thompson ED, Jaffee EM, Fertig EJ. Multi-omic profiling of lung and liver tumor microenvironments of metastatic pancreatic cancer reveals site-specific immune regulatory pathways. Genome Biol. 2021;22:154. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 57] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 75. | Conway JW, Rawson RV, Lo S, Ahmed T, Vergara IA, Gide TN, Attrill GH, Carlino MS, Saw RPM, Thompson JF, Spillane AJ, Shannon KF, Shivalingam B, Menzies AM, Wilmott JS, Long GV, Scolyer RA, Pires da Silva I. Unveiling the tumor immune microenvironment of organ-specific melanoma metastatic sites. J Immunother Cancer. 2022;10:e004884. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 39] [Reference Citation Analysis (0)] |
| 76. | Ye J, Guo W, Wang C, Egelston CA, D'Apuzzo M, Shankar G, Fakih MG, Lee PP. Peritumoral Immune-suppressive Mechanisms Impede Intratumoral Lymphocyte Infiltration into Colorectal Cancer Liver versus Lung Metastases. Cancer Res Commun. 2023;3:2082-2095. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 77. | Tian BW, Han CL, Wang HC, Yan LJ, Ding ZN, Liu H, Mao XC, Tian JC, Xue JS, Yang LS, Tan SY, Dong ZR, Yan YC, Wang DX, Li T. Effect of liver metastasis on the efficacy of immune checkpoint inhibitors in cancer patients: a systemic review and meta-analysis. Clin Exp Metastasis. 2023;40:255-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 20] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 78. | Chen XJ, Ren A, Zheng L, Zheng ED, Jiang T. Pan-Cancer Analysis Identifies Liver Metastases as Negative Predictive Factor for Immune Checkpoint Inhibitors Treatment Outcome. Front Immunol. 2021;12:651086. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 79. | Yin Q, Dai L, Sun R, Ke P, Liu L, Jiang B. Clinical Efficacy of Immune Checkpoint Inhibitors in Non-Small Cell Lung Cancer Patients with Liver Metastases: A Network Meta-Analysis of Nine Randomized Controlled Trials. Cancer Res Treat. 2022;54:803-816. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 80. | Wang JF, Lu HD, Wang Y, Zhang R, Li X, Wang S. Clinical characteristics and prognosis of non-small cell lung cancer patients with liver metastasis: A population-based study. World J Clin Cases. 2022;10:10882-10895. [PubMed] [DOI] [Full Text] |
| 81. | Xu H, Ming P, Zhao Z, Zhao N, Zhou D, Tang X, Cao D. Assessing the Relationship Between Liver Metastases and the Survival of Patients With Non-Small Cell Lung Cancer After Immune Checkpoint Inhibitors Treatment: A Systematic Review and Meta-Analysis. Integr Cancer Ther. 2023;22:15347354231164584. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 82. | Vokes EE, Ready N, Felip E, Horn L, Burgio MA, Antonia SJ, Arén Frontera O, Gettinger S, Holgado E, Spigel D, Waterhouse D, Domine M, Garassino M, Chow LQM, Blumenschein G Jr, Barlesi F, Coudert B, Gainor J, Arrieta O, Brahmer J, Butts C, Steins M, Geese WJ, Li A, Healey D, Crinò L. Nivolumab versus docetaxel in previously treated advanced non-small-cell lung cancer (CheckMate 017 and CheckMate 057): 3-year update and outcomes in patients with liver metastases. Ann Oncol. 2018;29:959-965. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 263] [Cited by in RCA: 382] [Article Influence: 54.6] [Reference Citation Analysis (0)] |
| 83. | Gadgeel S, Rodríguez-Abreu D, Speranza G, Esteban E, Felip E, Dómine M, Hui R, Hochmair MJ, Clingan P, Powell SF, Cheng SY, Bischoff HG, Peled N, Grossi F, Jennens RR, Reck M, Garon EB, Novello S, Rubio-Viqueira B, Boyer M, Kurata T, Gray JE, Yang J, Bas T, Pietanza MC, Garassino MC. Updated Analysis From KEYNOTE-189: Pembrolizumab or Placebo Plus Pemetrexed and Platinum for Previously Untreated Metastatic Nonsquamous Non-Small-Cell Lung Cancer. J Clin Oncol. 2020;38:1505-1517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 881] [Cited by in RCA: 797] [Article Influence: 132.8] [Reference Citation Analysis (0)] |
| 84. | Kang SP, Gergich K, Lubiniecki GM, de Alwis DP, Chen C, Tice MAB, Rubin EH. Pembrolizumab KEYNOTE-001: an adaptive study leading to accelerated approval for two indications and a companion diagnostic. Ann Oncol. 2017;28:1388-1398. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 82] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 85. | Herbst RS, Garon EB, Kim DW, Cho BC, Gervais R, Perez-Gracia JL, Han JY, Majem M, Forster MD, Monnet I, Novello S, Gubens MA, Boyer M, Su WC, Samkari A, Jensen EH, Kobie J, Piperdi B, Baas P. Five Year Survival Update From KEYNOTE-010: Pembrolizumab Versus Docetaxel for Previously Treated, Programmed Death-Ligand 1-Positive Advanced NSCLC. J Thorac Oncol. 2021;16:1718-1732. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 258] [Cited by in RCA: 234] [Article Influence: 46.8] [Reference Citation Analysis (0)] |
| 86. | Reck M, Rodríguez-Abreu D, Robinson AG, Hui R, Csőszi T, Fülöp A, Gottfried M, Peled N, Tafreshi A, Cuffe S, O'Brien M, Rao S, Hotta K, Vandormael K, Riccio A, Yang J, Pietanza MC, Brahmer JR. Updated Analysis of KEYNOTE-024: Pembrolizumab Versus Platinum-Based Chemotherapy for Advanced Non-Small-Cell Lung Cancer With PD-L1 Tumor Proportion Score of 50% or Greater. J Clin Oncol. 2019;37:537-546. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1350] [Cited by in RCA: 1243] [Article Influence: 177.6] [Reference Citation Analysis (3)] |
| 87. | Reck M, Rodríguez-Abreu D, Robinson AG, Hui R, Csőszi T, Fülöp A, Gottfried M, Peled N, Tafreshi A, Cuffe S, O'Brien M, Rao S, Hotta K, Leal TA, Riess JW, Jensen E, Zhao B, Pietanza MC, Brahmer JR. Five-Year Outcomes With Pembrolizumab Versus Chemotherapy for Metastatic Non-Small-Cell Lung Cancer With PD-L1 Tumor Proportion Score ≥ 50. J Clin Oncol. 2021;39:2339-2349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 944] [Cited by in RCA: 868] [Article Influence: 173.6] [Reference Citation Analysis (3)] |
| 88. | Xia H, Zhang W, Zhang Y, Shang X, Liu Y, Wang X. Liver metastases and the efficacy of immune checkpoint inhibitors in advanced lung cancer: A systematic review and meta-analysis. Front Oncol. 2022;12:978069. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 34] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 89. | Socinski MA, Jotte RM, Cappuzzo F, Orlandi F, Stroyakovskiy D, Nogami N, Rodríguez-Abreu D, Moro-Sibilot D, Thomas CA, Barlesi F, Finley G, Kelsch C, Lee A, Coleman S, Deng Y, Shen Y, Kowanetz M, Lopez-Chavez A, Sandler A, Reck M; IMpower150 Study Group. Atezolizumab for First-Line Treatment of Metastatic Nonsquamous NSCLC. N Engl J Med. 2018;378:2288-2301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3160] [Cited by in RCA: 2954] [Article Influence: 369.3] [Reference Citation Analysis (3)] |
| 90. | Socinski MA, Nishio M, Jotte RM, Cappuzzo F, Orlandi F, Stroyakovskiy D, Nogami N, Rodríguez-Abreu D, Moro-Sibilot D, Thomas CA, Barlesi F, Finley G, Kong S, Lee A, Coleman S, Zou W, McCleland M, Shankar G, Reck M. IMpower150 Final Overall Survival Analyses for Atezolizumab Plus Bevacizumab and Chemotherapy in First-Line Metastatic Nonsquamous NSCLC. J Thorac Oncol. 2021;16:1909-1924. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 425] [Cited by in RCA: 374] [Article Influence: 74.8] [Reference Citation Analysis (0)] |
| 91. | Nogami N, Barlesi F, Socinski MA, Reck M, Thomas CA, Cappuzzo F, Mok TSK, Finley G, Aerts JG, Orlandi F, Moro-Sibilot D, Jotte RM, Stroyakovskiy D, Villaruz LC, Rodríguez-Abreu D, Wan-Teck Lim D, Merritt D, Coleman S, Lee A, Shankar G, Yu W, Bara I, Nishio M. IMpower150 Final Exploratory Analyses for Atezolizumab Plus Bevacizumab and Chemotherapy in Key NSCLC Patient Subgroups With EGFR Mutations or Metastases in the Liver or Brain. J Thorac Oncol. 2022;17:309-323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 244] [Cited by in RCA: 219] [Article Influence: 54.8] [Reference Citation Analysis (0)] |
| 92. | Paz-Ares L, Ciuleanu TE, Cobo M, Schenker M, Zurawski B, Menezes J, Richardet E, Bennouna J, Felip E, Juan-Vidal O, Alexandru A, Sakai H, Lingua A, Salman P, Souquet PJ, De Marchi P, Martin C, Pérol M, Scherpereel A, Lu S, John T, Carbone DP, Meadows-Shropshire S, Agrawal S, Oukessou A, Yan J, Reck M. First-line nivolumab plus ipilimumab combined with two cycles of chemotherapy in patients with non-small-cell lung cancer (CheckMate 9LA): an international, randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22:198-211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1178] [Cited by in RCA: 1021] [Article Influence: 204.2] [Reference Citation Analysis (3)] |
| 93. | Reck M, Ciuleanu TE, Schenker M, Bordenave S, Cobo M, Juan-Vidal O, Reinmuth N, Richardet E, Felip E, Menezes J, Cheng Y, Mizutani H, Zurawski B, Alexandru A, Carbone DP, Lu S, John T, Aoyama T, Grootendorst DJ, Hu N, Eccles LJ, Paz-Ares LG. Five-year outcomes with first-line nivolumab plus ipilimumab with 2 cycles of chemotherapy versus 4 cycles of chemotherapy alone in patients with metastatic non-small cell lung cancer in the randomized CheckMate 9LA trial. Eur J Cancer. 2024;211:114296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 56] [Article Influence: 28.0] [Reference Citation Analysis (0)] |
| 94. | Tumeh PC, Hellmann MD, Hamid O, Tsai KK, Loo KL, Gubens MA, Rosenblum M, Harview CL, Taube JM, Handley N, Khurana N, Nosrati A, Krummel MF, Tucker A, Sosa EV, Sanchez PJ, Banayan N, Osorio JC, Nguyen-Kim DL, Chang J, Shintaku IP, Boasberg PD, Taylor EJ, Munster PN, Algazi AP, Chmielowski B, Dummer R, Grogan TR, Elashoff D, Hwang J, Goldinger SM, Garon EB, Pierce RH, Daud A. Liver Metastasis and Treatment Outcome with Anti-PD-1 Monoclonal Antibody in Patients with Melanoma and NSCLC. Cancer Immunol Res. 2017;5:417-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 491] [Cited by in RCA: 481] [Article Influence: 53.4] [Reference Citation Analysis (3)] |
| 95. | Pires da Silva I, Ahmed T, McQuade JL, Nebhan CA, Park JJ, Versluis JM, Serra-Bellver P, Khan Y, Slattery T, Oberoi HK, Ugurel S, Haydu LE, Herbst R, Utikal J, Pföhler C, Terheyden P, Weichenthal M, Gutzmer R, Mohr P, Rai R, Smith JL, Scolyer RA, Arance AM, Pickering L, Larkin J, Lorigan P, Blank CU, Schadendorf D, Davies MA, Carlino MS, Johnson DB, Long GV, Lo SN, Menzies AM. Clinical Models to Define Response and Survival With Anti-PD-1 Antibodies Alone or Combined With Ipilimumab in Metastatic Melanoma. J Clin Oncol. 2022;40:1068-1080. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 70] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 96. | Liu S, Dharanipragada P, Lomeli SH, Wang Y, Zhang X, Yang Z, Lim RJ, Dumitras C, Scumpia PO, Dubinett SM, Moriceau G, Johnson DB, Moschos SJ, Lo RS. Multi-organ landscape of therapy-resistant melanoma. Nat Med. 2023;29:1123-1134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 25] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 97. | Zarour LR, Anand S, Billingsley KG, Bisson WH, Cercek A, Clarke MF, Coussens LM, Gast CE, Geltzeiler CB, Hansen L, Kelley KA, Lopez CD, Rana SR, Ruhl R, Tsikitis VL, Vaccaro GM, Wong MH, Mayo SC. Colorectal Cancer Liver Metastasis: Evolving Paradigms and Future Directions. Cell Mol Gastroenterol Hepatol. 2017;3:163-173. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 233] [Cited by in RCA: 218] [Article Influence: 24.2] [Reference Citation Analysis (4)] |
| 98. | Engstrand J, Nilsson H, Strömberg C, Jonas E, Freedman J. Colorectal cancer liver metastases - a population-based study on incidence, management and survival. BMC Cancer. 2018;18:78. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 747] [Cited by in RCA: 682] [Article Influence: 85.3] [Reference Citation Analysis (8)] |
| 99. | André T, Shiu KK, Kim TW, Jensen BV, Jensen LH, Punt C, Smith D, Garcia-Carbonero R, Benavides M, Gibbs P, de la Fouchardiere C, Rivera F, Elez E, Bendell J, Le DT, Yoshino T, Van Cutsem E, Yang P, Farooqui MZH, Marinello P, Diaz LA Jr; KEYNOTE-177 Investigators. Pembrolizumab in Microsatellite-Instability-High Advanced Colorectal Cancer. N Engl J Med. 2020;383:2207-2218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2522] [Cited by in RCA: 2184] [Article Influence: 364.0] [Reference Citation Analysis (6)] |
| 100. | Van Cutsem E, Cervantes A, Adam R, Sobrero A, Van Krieken JH, Aderka D, Aranda Aguilar E, Bardelli A, Benson A, Bodoky G, Ciardiello F, D'Hoore A, Diaz-Rubio E, Douillard JY, Ducreux M, Falcone A, Grothey A, Gruenberger T, Haustermans K, Heinemann V, Hoff P, Köhne CH, Labianca R, Laurent-Puig P, Ma B, Maughan T, Muro K, Normanno N, Österlund P, Oyen WJ, Papamichael D, Pentheroudakis G, Pfeiffer P, Price TJ, Punt C, Ricke J, Roth A, Salazar R, Scheithauer W, Schmoll HJ, Tabernero J, Taïeb J, Tejpar S, Wasan H, Yoshino T, Zaanan A, Arnold D. ESMO consensus guidelines for the management of patients with metastatic colorectal cancer. Ann Oncol. 2016;27:1386-1422. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2828] [Cited by in RCA: 2564] [Article Influence: 256.4] [Reference Citation Analysis (13)] |
| 101. | Hellman S, Weichselbaum RR. Oligometastases. J Clin Oncol. 1995;13:8-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2176] [Cited by in RCA: 1976] [Article Influence: 63.7] [Reference Citation Analysis (0)] |
| 102. | Valderrama-Treviño AI, Barrera-Mera B, Ceballos-Villalva JC, Montalvo-Javé EE. Hepatic Metastasis from Colorectal Cancer. Euroasian J Hepatogastroenterol. 2017;7:166-175. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 192] [Cited by in RCA: 176] [Article Influence: 19.6] [Reference Citation Analysis (0)] |
| 103. | Maher B, Ryan E, Little M, Boardman P, Stedman B. The management of colorectal liver metastases. Clin Radiol. 2017;72:617-625. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 25] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 104. | Torielli P, McGale J, Liao MJ, Rhaiem R, Bouche O, Botsen D, Gerin O, Lamane A, Lawrence Y, Madelis G, Rozenblum L, Sajan A, Tordjman M, Dercle L, Beddok A. Hepatic metastases management: A comparative review of surgical resection, thermal ablation, and stereotactic body radiation therapy. Eur J Cancer. 2025;228:115691. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 105. | Renz BW, Boeck S, Roeder F, Trumm C, Heinemann V, Werner J. Oligometastatic Disease in Pancreatic Cancer - How to Proceed? Visc Med. 2017;33:36-41. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 36] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 106. | Saberzadeh-Ardestani B, Jones JC, McWilliams RR, Tougeron D, Halfdanarson TR, Guimbaud R, Hubbard JM, Flecchia C, Shi Q, Alouani E, Sonbol MB, Ticku J, Jin Z, Taieb J, Sinicrope FA. Metastatic site and clinical outcome of patients with deficient mismatch repair metastatic colorectal cancer treated with an immune checkpoint inhibitor in the first-line setting. Eur J Cancer. 2024;196:113433. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 11.0] [Reference Citation Analysis (3)] |
| 107. | Chen EX, Jonker DJ, Loree JM, Kennecke HF, Berry SR, Couture F, Ahmad CE, Goffin JR, Kavan P, Harb M, Colwell B, Samimi S, Samson B, Abbas T, Aucoin N, Aubin F, Koski SL, Wei AC, Magoski NM, Tu D, O'Callaghan CJ. Effect of Combined Immune Checkpoint Inhibition vs Best Supportive Care Alone in Patients With Advanced Colorectal Cancer: The Canadian Cancer Trials Group CO.26 Study. JAMA Oncol. 2020;6:831-838. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 342] [Cited by in RCA: 320] [Article Influence: 53.3] [Reference Citation Analysis (4)] |
| 108. | Fukuoka S, Hara H, Takahashi N, Kojima T, Kawazoe A, Asayama M, Yoshii T, Kotani D, Tamura H, Mikamoto Y, Hirano N, Wakabayashi M, Nomura S, Sato A, Kuwata T, Togashi Y, Nishikawa H, Shitara K. Regorafenib Plus Nivolumab in Patients With Advanced Gastric or Colorectal Cancer: An Open-Label, Dose-Escalation, and Dose-Expansion Phase Ib Trial (REGONIVO, EPOC1603). J Clin Oncol. 2020;38:2053-2061. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 673] [Cited by in RCA: 615] [Article Influence: 102.5] [Reference Citation Analysis (3)] |
| 109. | Kang N, Gores GJ, Shah VH. Hepatic stellate cells: partners in crime for liver metastases? Hepatology. 2011;54:707-713. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 157] [Cited by in RCA: 145] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 110. | Liu C, Li J, Xiang X, Guo L, Tu K, Liu Q, Shah VH, Kang N. PDGF receptor-α promotes TGF-β signaling in hepatic stellate cells via transcriptional and posttranscriptional regulation of TGF-β receptors. Am J Physiol Gastrointest Liver Physiol. 2014;307:G749-G759. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 62] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 111. | Sherman MH, Beatty GL. Tumor Microenvironment in Pancreatic Cancer Pathogenesis and Therapeutic Resistance. Annu Rev Pathol. 2023;18:123-148. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 366] [Cited by in RCA: 319] [Article Influence: 106.3] [Reference Citation Analysis (1)] |
| 112. | Jiang H, Hegde S, DeNardo DG. Tumor-associated fibrosis as a regulator of tumor immunity and response to immunotherapy. Cancer Immunol Immunother. 2017;66:1037-1048. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 204] [Cited by in RCA: 193] [Article Influence: 21.4] [Reference Citation Analysis (0)] |
| 113. | Wang H, Qiu B, Li X, Ying Y, Wang Y, Chen H, Zeng F, Shi J, Huang J, Wu Z, Chen Z, Che X, Li Q, Fan Y, Li B, Wang Q, Huang C, Chen Y, Li T, Mo K, Wang Q, Cui C. Single cell analysis reveals that SPP1(+) macrophages enhance tumor progression by triggering fibroblast extracellular vesicles. Transl Oncol. 2025;55:102347. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 114. | Li E, Cheung HCZ, Ma S. CTHRC1(+) fibroblasts and SPP1(+) macrophages synergistically contribute to pro-tumorigenic tumor microenvironment in pancreatic ductal adenocarcinoma. Sci Rep. 2024;14:17412. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 26] [Reference Citation Analysis (0)] |
| 115. | Wei D, Wang L, Zuo X, Maitra A, Bresalier RS. A Small Molecule with Big Impact: MRTX1133 Targets the KRASG12D Mutation in Pancreatic Cancer. Clin Cancer Res. 2024;30:655-662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 117] [Article Influence: 58.5] [Reference Citation Analysis (0)] |
| 116. | Wasko UN, Jiang J, Curiel-Garcia A, Wang Y, Lee B, Orlen M, Drizyte-Miller K, Menard M, Dilly J, Sastra SA, Palermo CF, Dalton T, Hasselluhn MC, Decker-Farrell AR, Chang S, Jiang L, Wei X, Yang YC, Helland C, Courtney H, Gindin Y, Zhao R, Kemp SB, Clendenin C, Sor R, Vostrejs W, Amparo AA, Hibshman PS, Rees MG, Ronan MM, Roth JA, Bakir B, Badgley MA, Chabot JA, Kluger MD, Manji GA, Quintana E, Wang Z, Smith JAM, Holderfield M, Wildes D, Aguirre AJ, Der CJ, Vonderheide RH, Stanger BZ, Singh M, Olive KP. Tumor-selective effects of active RAS inhibition in pancreatic ductal adenocarcinoma. bioRxiv. 2023;2023.12.03.569791. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 117. | Jiang X, Wang T, Zhao B, Sun H, Dong Y, Ma Y, Li Z, Wu Y, Wang K, Guan X, Long B, Qin L, Shi W, Shi L, He Q, Liu W, Li M, Xiao L, Zhou C, Sun H, Yang J, Guan J, Zhou H, Yu Z, Jiao Z. KRAS(G12D)-driven pentose phosphate pathway remodeling imparts a targetable vulnerability synergizing with MRTX1133 for durable remissions in PDAC. Cell Rep Med. 2025;6:101966. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 12] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 118. | Liu S, Li F, Deng L, Ma Q, Lu W, Zhao Z, Liu H, Zhou Y, Hu M, Wang H, Yan Y, Zhao M, Zhang H, Du M. Claudin18.2 bispecific T cell engager armed oncolytic virus enhances antitumor effects against pancreatic cancer. Mol Ther Oncolytics. 2023;30:275-285. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 119. | Ma Z, Zhou Z, Duan W, Yao G, Sheng S, Zong S, Zhang X, Li C, Liu Y, Ou F, Dahar MR, Huang Y, Yu L. DR30318, a novel tri-specific T cell engager for Claudin 18.2 positive cancers immunotherapy. Cancer Immunol Immunother. 2024;73:82. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 120. | Hosein AN, Brekken RA, Maitra A. Pancreatic cancer stroma: an update on therapeutic targeting strategies. Nat Rev Gastroenterol Hepatol. 2020;17:487-505. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 800] [Cited by in RCA: 747] [Article Influence: 124.5] [Reference Citation Analysis (5)] |
| 121. | Conroy T, Desseigne F, Ychou M, Bouché O, Guimbaud R, Bécouarn Y, Adenis A, Raoul JL, Gourgou-Bourgade S, de la Fouchardière C, Bennouna J, Bachet JB, Khemissa-Akouz F, Péré-Vergé D, Delbaldo C, Assenat E, Chauffert B, Michel P, Montoto-Grillot C, Ducreux M; Groupe Tumeurs Digestives of Unicancer; PRODIGE Intergroup. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med. 2011;364:1817-1825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6475] [Cited by in RCA: 5916] [Article Influence: 394.4] [Reference Citation Analysis (4)] |
| 122. | Von Hoff DD, Ervin T, Arena FP, Chiorean EG, Infante J, Moore M, Seay T, Tjulandin SA, Ma WW, Saleh MN, Harris M, Reni M, Dowden S, Laheru D, Bahary N, Ramanathan RK, Tabernero J, Hidalgo M, Goldstein D, Van Cutsem E, Wei X, Iglesias J, Renschler MF. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N Engl J Med. 2013;369:1691-1703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5577] [Cited by in RCA: 5194] [Article Influence: 399.5] [Reference Citation Analysis (4)] |
| 123. | Marabelle A, Le DT, Ascierto PA, Di Giacomo AM, De Jesus-Acosta A, Delord JP, Geva R, Gottfried M, Penel N, Hansen AR, Piha-Paul SA, Doi T, Gao B, Chung HC, Lopez-Martin J, Bang YJ, Frommer RS, Shah M, Ghori R, Joe AK, Pruitt SK, Diaz LA Jr. Efficacy of Pembrolizumab in Patients With Noncolorectal High Microsatellite Instability/Mismatch Repair-Deficient Cancer: Results From the Phase II KEYNOTE-158 Study. J Clin Oncol. 2020;38:1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2532] [Cited by in RCA: 2280] [Article Influence: 380.0] [Reference Citation Analysis (9)] |
| 124. | Bear AS, Vonderheide RH, O'Hara MH. Challenges and Opportunities for Pancreatic Cancer Immunotherapy. Cancer Cell. 2020;38:788-802. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 554] [Cited by in RCA: 501] [Article Influence: 83.5] [Reference Citation Analysis (4)] |
| 125. | Taïeb J, Sayah L, Heinrich K, Kunzmann V, Boileve A, Cirkel G, Lonardi S, Chibaudel B, Turpin A, Beller T, Hautefeuille V, Vivaldi C, Mazard T, Bauguion L, Niger M, Prager GW, Coutzac C, Benedikt Westphalen C, Auclin E, Pilla L. Efficacy of immune checkpoint inhibitors in microsatellite unstable/mismatch repair-deficient advanced pancreatic adenocarcinoma: an AGEO European Cohort. Eur J Cancer. 2023;188:90-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 66] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 126. | Groen-van Schooten TS, Harrasser M, Seidel J, Bos EN, Fleitas T, van Mourik M, Pouw RE, Goedegebuure RSA, Doeve BH, Sanders J, Bos J, van Berge Henegouwen MI, Thijssen VLJL, van Grieken NCT, van Laarhoven HWM, de Gruijl TD, Derks S. Phenotypic immune characterization of gastric and esophageal adenocarcinomas reveals profound immune suppression in esophageal tumor locations. Front Immunol. 2024;15:1372272. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 127. | Moehler M, Dvorkin M, Boku N, Özgüroğlu M, Ryu MH, Muntean AS, Lonardi S, Nechaeva M, Bragagnoli AC, Coşkun HS, Cubillo Gracian A, Takano T, Wong R, Safran H, Vaccaro GM, Wainberg ZA, Silver MR, Xiong H, Hong J, Taieb J, Bang YJ. Phase III Trial of Avelumab Maintenance After First-Line Induction Chemotherapy Versus Continuation of Chemotherapy in Patients With Gastric Cancers: Results From JAVELIN Gastric 100. J Clin Oncol. 2021;39:966-977. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 188] [Cited by in RCA: 180] [Article Influence: 36.0] [Reference Citation Analysis (0)] |
| 128. | Pu W, Li S, Zhang J, Huang J, Li J, Jiang Y, Xu Z, Yi F, Lan Y, Xiao Q, Chen W, Jin J. The efficacy and safety of PD-1/PD-L1 inhibitors in combination with chemotherapy as a first-line treatment for unresectable, locally advanced, HER2-negative gastric or gastroesophageal junction cancer: a meta-analysis of randomized controlled trials. Front Immunol. 2025;16:1566939. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 129. | Rha SY, Oh DY, Yañez P, Bai Y, Ryu MH, Lee J, Rivera F, Alves GV, Garrido M, Shiu KK, Fernández MG, Li J, Lowery MA, Çil T, Cruz FM, Qin S, Luo S, Pan H, Wainberg ZA, Yin L, Bordia S, Bhagia P, Wyrwicz LS; KEYNOTE-859 investigators. Pembrolizumab plus chemotherapy versus placebo plus chemotherapy for HER2-negative advanced gastric cancer (KEYNOTE-859): a multicentre, randomised, double-blind, phase 3 trial. Lancet Oncol. 2023;24:1181-1195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 547] [Cited by in RCA: 509] [Article Influence: 169.7] [Reference Citation Analysis (3)] |
| 130. | Janjigian YY, Shitara K, Moehler M, Garrido M, Salman P, Shen L, Wyrwicz L, Yamaguchi K, Skoczylas T, Campos Bragagnoli A, Liu T, Schenker M, Yanez P, Tehfe M, Kowalyszyn R, Karamouzis MV, Bruges R, Zander T, Pazo-Cid R, Hitre E, Feeney K, Cleary JM, Poulart V, Cullen D, Lei M, Xiao H, Kondo K, Li M, Ajani JA. First-line nivolumab plus chemotherapy versus chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (CheckMate 649): a randomised, open-label, phase 3 trial. Lancet. 2021;398:27-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2501] [Cited by in RCA: 2292] [Article Influence: 458.4] [Reference Citation Analysis (5)] |
| 131. | Liang H, Li Z, Huang Z, Wu C, Qiu Y, Liang Y, Chen X, Li F, Xu Z, Li G, Liu H, Zhao L. Prognostic characteristics and clinical response to immunotherapy targeting programmed cell death 1 for patients with advanced gastric cancer with liver metastases. Front Immunol. 2022;13:1015549. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 132. | Zhu AX, Duda DG, Sahani DV, Jain RK. HCC and angiogenesis: possible targets and future directions. Nat Rev Clin Oncol. 2011;8:292-301. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 456] [Cited by in RCA: 457] [Article Influence: 30.5] [Reference Citation Analysis (4)] |
| 133. | Zhang Y, Huang H, Coleman M, Ziemys A, Gopal P, Kazmi SM, Brekken RA. VEGFR2 activity on myeloid cells mediates immune suppression in the tumor microenvironment. JCI Insight. 2021;6:e150735. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 46] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 134. | Okikawa S, Morine Y, Saito Y, Yamada S, Tokuda K, Teraoku H, Miyazaki K, Yamashita S, Ikemoto T, Imura S, Shimada M. Inhibition of the VEGF signaling pathway attenuates tumorassociated macrophage activity in liver cancer. Oncol Rep. 2022;47:71. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 47] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 135. | Zhang Y, Brekken RA. Direct and indirect regulation of the tumor immune microenvironment by VEGF. J Leukoc Biol. 2022;111:1269-1286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 94] [Article Influence: 23.5] [Reference Citation Analysis (0)] |
| 136. | Nishida N. Clinical implications of the dual blockade of the PD-1/PD-L1 and vascular endothelial growth factor axes in the treatment of hepatocellular carcinoma. Hepatobiliary Surg Nutr. 2020;9:640-643. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 137. | Lee WS, Yang H, Chon HJ, Kim C. Combination of anti-angiogenic therapy and immune checkpoint blockade normalizes vascular-immune crosstalk to potentiate cancer immunity. Exp Mol Med. 2020;52:1475-1485. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 341] [Cited by in RCA: 510] [Article Influence: 85.0] [Reference Citation Analysis (0)] |
| 138. | Kudo M. Scientific Rationale for Combined Immunotherapy with PD-1/PD-L1 Antibodies and VEGF Inhibitors in Advanced Hepatocellular Carcinoma. Cancers (Basel). 2020;12:1089. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 174] [Cited by in RCA: 169] [Article Influence: 28.2] [Reference Citation Analysis (0)] |
| 139. | Hegde PS, Karanikas V, Evers S. The Where, the When, and the How of Immune Monitoring for Cancer Immunotherapies in the Era of Checkpoint Inhibition. Clin Cancer Res. 2016;22:1865-1874. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 801] [Cited by in RCA: 736] [Article Influence: 73.6] [Reference Citation Analysis (0)] |
| 140. | Fukumura D, Kloepper J, Amoozgar Z, Duda DG, Jain RK. Enhancing cancer immunotherapy using antiangiogenics: opportunities and challenges. Nat Rev Clin Oncol. 2018;15:325-340. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1703] [Cited by in RCA: 1586] [Article Influence: 198.3] [Reference Citation Analysis (8)] |
| 141. | Rahma OE, Hodi FS. The Intersection between Tumor Angiogenesis and Immune Suppression. Clin Cancer Res. 2019;25:5449-5457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 424] [Cited by in RCA: 376] [Article Influence: 53.7] [Reference Citation Analysis (3)] |
| 142. | Wei SC, Levine JH, Cogdill AP, Zhao Y, Anang NAS, Andrews MC, Sharma P, Wang J, Wargo JA, Pe'er D, Allison JP. Distinct Cellular Mechanisms Underlie Anti-CTLA-4 and Anti-PD-1 Checkpoint Blockade. Cell. 2017;170:1120-1133.e17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1117] [Cited by in RCA: 1039] [Article Influence: 115.4] [Reference Citation Analysis (4)] |
| 143. | Pires da Silva I, Li I, Ugurel S, Serra-Bellver P, Andhale A, Burnette H, Aya F, Conway JW, Braden J, Carlino MS, Menzies AM, Weichenthal M, Mohr P, Gutzmer R, Arance AM, Johnson DB, Lorigan P, Schadendorf D, Lo SN, Long GV. Anti-PD-1 alone or in combination with anti-CTLA-4 for advanced melanoma patients with liver metastases. Eur J Cancer. 2024;205:114101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 144. | Fiegle E, Doleschel D, Koletnik S, Rix A, Weiskirchen R, Borkham-Kamphorst E, Kiessling F, Lederle W. Dual CTLA-4 and PD-L1 Blockade Inhibits Tumor Growth and Liver Metastasis in a Highly Aggressive Orthotopic Mouse Model of Colon Cancer. Neoplasia. 2019;21:932-944. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 77] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 145. | Sové RJ, Verma BK, Wang H, Ho WJ, Yarchoan M, Popel AS. Virtual clinical trials of anti-PD-1 and anti-CTLA-4 immunotherapy in advanced hepatocellular carcinoma using a quantitative systems pharmacology model. J Immunother Cancer. 2022;10:e005414. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 37] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 146. | Buchbinder EI, Desai A. CTLA-4 and PD-1 Pathways: Similarities, Differences, and Implications of Their Inhibition. Am J Clin Oncol. 2016;39:98-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2065] [Cited by in RCA: 1854] [Article Influence: 185.4] [Reference Citation Analysis (1)] |
| 147. | Curran MA, Montalvo W, Yagita H, Allison JP. PD-1 and CTLA-4 combination blockade expands infiltrating T cells and reduces regulatory T and myeloid cells within B16 melanoma tumors. Proc Natl Acad Sci U S A. 2010;107:4275-4280. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1625] [Cited by in RCA: 1514] [Article Influence: 94.6] [Reference Citation Analysis (4)] |
| 148. | Zhao W, Li B, Gu Y, Jin X, Li Z, Guo W, Lu X, Jiang J. The efficacy and safety of immune combination therapy in patients with driver gene-negative non-small cell lung cancer with liver metastasis: a systematic review and network meta-analysis. BMC Cancer. 2025;25:1332. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 149. | Wu RY, Wang BC, Wang K, Xia F, Zhang ZY, Wan JF, Zhang Z. Immunotherapy and tumor mutational burden in cancer patients with liver metastases: A meta and real word cohort analysis. Front Oncol. 2022;12:994276. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 150. | Wolchok JD, Kluger H, Callahan MK, Postow MA, Rizvi NA, Lesokhin AM, Segal NH, Ariyan CE, Gordon RA, Reed K, Burke MM, Caldwell A, Kronenberg SA, Agunwamba BU, Zhang X, Lowy I, Inzunza HD, Feely W, Horak CE, Hong Q, Korman AJ, Wigginton JM, Gupta A, Sznol M. Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med. 2013;369:122-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3590] [Cited by in RCA: 3318] [Article Influence: 255.2] [Reference Citation Analysis (3)] |
| 151. | Hassel JC, Heinzerling L, Aberle J, Bähr O, Eigentler TK, Grimm MO, Grünwald V, Leipe J, Reinmuth N, Tietze JK, Trojan J, Zimmer L, Gutzmer R. Combined immune checkpoint blockade (anti-PD-1/anti-CTLA-4): Evaluation and management of adverse drug reactions. Cancer Treat Rev. 2017;57:36-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 288] [Cited by in RCA: 255] [Article Influence: 28.3] [Reference Citation Analysis (0)] |
| 152. | Kirchberger MC, Moreira A, Erdmann M, Schuler G, Heinzerling L. Real world experience in low-dose ipilimumab in combination with PD-1 blockade in advanced melanoma patients. Oncotarget. 2018;9:28903-28909. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 37] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 153. | Lebbé C, Meyer N, Mortier L, Marquez-Rodas I, Robert C, Rutkowski P, Menzies AM, Eigentler T, Ascierto PA, Smylie M, Schadendorf D, Ajaz M, Svane IM, Gonzalez R, Rollin L, Lord-Bessen J, Saci A, Grigoryeva E, Pigozzo J. Evaluation of Two Dosing Regimens for Nivolumab in Combination With Ipilimumab in Patients With Advanced Melanoma: Results From the Phase IIIb/IV CheckMate 511 Trial. J Clin Oncol. 2019;37:867-875. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 158] [Cited by in RCA: 294] [Article Influence: 42.0] [Reference Citation Analysis (0)] |
| 154. | Long GV, Robert C, Butler MO, Couture F, Carlino MS, O'Day S, Atkinson V, Cebon JS, Brown MP, Dalle S, Hill AG, Gibney GT, McCune S, Menzies AM, Niu C, Ibrahim N, Moreno BH, Diab A. Standard-Dose Pembrolizumab Plus Alternate-Dose Ipilimumab in Advanced Melanoma: KEYNOTE-029 Cohort 1C, a Phase 2 Randomized Study of Two Dosing Schedules. Clin Cancer Res. 2021;27:5280-5288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 155. | Wang W, Gao Y, Chen Y, Cheng M, Sang Y, Wei L, Dai R, Wang Y, Zhang L. TGF-β inhibitors: the future for prevention and treatment of liver fibrosis? Front Immunol. 2025;16:1583616. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 156. | Peng D, Fu M, Wang M, Wei Y, Wei X. Targeting TGF-β signal transduction for fibrosis and cancer therapy. Mol Cancer. 2022;21:104. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 924] [Cited by in RCA: 846] [Article Influence: 211.5] [Reference Citation Analysis (4)] |
| 157. | Leonardo-Sousa C, Barriga R, Florindo HF, Acúrcio RC, Guedes RC. Structural insights and clinical advances in small-molecule inhibitors targeting TGF-β receptor I. Mol Ther Oncol. 2025;33:200945. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 158. | Khasraw M, Weller M, Lorente D, Kolibaba K, Lee CK, Gedye C, I de La Fuente M, Vicente D, Reardon DA, Gan HK, Scott AM, Dussault I, Helwig C, Ojalvo LS, Gourmelon C, Groves M. Bintrafusp alfa (M7824), a bifunctional fusion protein targeting TGF-β and PD-L1: results from a phase I expansion cohort in patients with recurrent glioblastoma. Neurooncol Adv. 2021;3:vdab058. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (4)] |
| 159. | Gameiro SR, Strauss J, Gulley JL, Schlom J. Preclinical and clinical studies of bintrafusp alfa, a novel bifunctional anti-PD-L1/TGFβRII agent: Current status. Exp Biol Med (Maywood). 2022;247:1124-1134. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 160. | Morris VK, Overman MJ, Lam M, Parseghian CM, Johnson B, Dasari A, Raghav K, Kee BK, Huey R, Wolff RA, Shen JP, Li J, Zorrilla I, Tzeng CD, Tran Cao HS, Chun YS, Newhook TE, Vauthey N, Duose D, Luthra R, Haymaker C, Kopetz S. Bintrafusp alfa, an anti-PD-L1:TGF-β trap fusion protein, in patients with ctDNA-positive, liver-limited metastatic colorectal cancer. Cancer Res Commun. 2022;2:979-986. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 25] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 161. | Holmgaard RB, Schaer DA, Li Y, Castaneda SP, Murphy MY, Xu X, Inigo I, Dobkin J, Manro JR, Iversen PW, Surguladze D, Hall GE, Novosiadly RD, Benhadji KA, Plowman GD, Kalos M, Driscoll KE. Targeting the TGFβ pathway with galunisertib, a TGFβRI small molecule inhibitor, promotes anti-tumor immunity leading to durable, complete responses, as monotherapy and in combination with checkpoint blockade. J Immunother Cancer. 2018;6:47. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 295] [Cited by in RCA: 286] [Article Influence: 35.8] [Reference Citation Analysis (4)] |
| 162. | Mariathasan S, Turley SJ, Nickles D, Castiglioni A, Yuen K, Wang Y, Kadel EE III, Koeppen H, Astarita JL, Cubas R, Jhunjhunwala S, Banchereau R, Yang Y, Guan Y, Chalouni C, Ziai J, Şenbabaoğlu Y, Santoro S, Sheinson D, Hung J, Giltnane JM, Pierce AA, Mesh K, Lianoglou S, Riegler J, Carano RAD, Eriksson P, Höglund M, Somarriba L, Halligan DL, van der Heijden MS, Loriot Y, Rosenberg JE, Fong L, Mellman I, Chen DS, Green M, Derleth C, Fine GD, Hegde PS, Bourgon R, Powles T. TGFβ attenuates tumour response to PD-L1 blockade by contributing to exclusion of T cells. Nature. 2018;554:544-548. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4457] [Cited by in RCA: 4124] [Article Influence: 515.5] [Reference Citation Analysis (5)] |
| 163. | Chiappinelli KB, Strissel PL, Desrichard A, Li H, Henke C, Akman B, Hein A, Rote NS, Cope LM, Snyder A, Makarov V, Budhu S, Slamon DJ, Wolchok JD, Pardoll DM, Beckmann MW, Zahnow CA, Merghoub T, Chan TA, Baylin SB, Strick R. Inhibiting DNA Methylation Causes an Interferon Response in Cancer via dsRNA Including Endogenous Retroviruses. Cell. 2015;162:974-986. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1290] [Cited by in RCA: 1428] [Article Influence: 129.8] [Reference Citation Analysis (0)] |
| 164. | Akce M, El-Rayes BF, Wajapeyee N. Combinatorial targeting of immune checkpoints and epigenetic regulators for hepatocellular carcinoma therapy. Oncogene. 2023;42:1051-1057. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 19] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 165. | Topper MJ, Vaz M, Chiappinelli KB, DeStefano Shields CE, Niknafs N, Yen RC, Wenzel A, Hicks J, Ballew M, Stone M, Tran PT, Zahnow CA, Hellmann MD, Anagnostou V, Strissel PL, Strick R, Velculescu VE, Baylin SB. Epigenetic Therapy Ties MYC Depletion to Reversing Immune Evasion and Treating Lung Cancer. Cell. 2017;171:1284-1300.e21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 431] [Cited by in RCA: 398] [Article Influence: 44.2] [Reference Citation Analysis (4)] |
| 166. | Liu Y, Yang H, Li T, Zhang N. Immunotherapy in liver cancer: overcoming the tolerogenic liver microenvironment. Front Immunol. 2024;15:1460282. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 24] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 167. | Wang Y, Guan Y, Hu Y, Li Y, Lu N, Zhang C. Murine CXCR3(+)CXCR6(+)γδT Cells Reside in the Liver and Provide Protection Against HBV Infection. Front Immunol. 2021;12:757379. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 168. | Katz SC, Burga RA, McCormack E, Wang LJ, Mooring W, Point GR, Khare PD, Thorn M, Ma Q, Stainken BF, Assanah EO, Davies R, Espat NJ, Junghans RP. Phase I Hepatic Immunotherapy for Metastases Study of Intra-Arterial Chimeric Antigen Receptor-Modified T-cell Therapy for CEA+ Liver Metastases. Clin Cancer Res. 2015;21:3149-3159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 339] [Cited by in RCA: 322] [Article Influence: 29.3] [Reference Citation Analysis (3)] |
| 169. | Takahashi H, Berber E. Role of thermal ablation in the management of colorectal liver metastasis. Hepatobiliary Surg Nutr. 2020;9:49-58. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 135] [Cited by in RCA: 124] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 170. | De Baere T, Tselikas L, Delpla A, Roux C, Varin E, Kobe A, Yevich S, Deschamps F. Thermal ablation in the management of oligometastatic colorectal cancer. Int J Hyperthermia. 2022;39:627-632. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 171. | van der Lei S, Puijk RS, Dijkstra M, Schulz HH, Vos DJW, De Vries JJJ, Scheffer HJ, Lissenberg-Witte BI, Aldrighetti L, Arntz M, Barentsz MW, Besselink MG, Bracke B, Bruijnen RCG, Buffart TE, Burgmans MC, Chapelle T, Coolsen MME, de Boer SW, de Cobelli F, de Jong K, de Wilt JHW, Diederik AL, Dooper AMC, Draaisma WA, Eker HH, Erdmann JI, Futterer JJ, Geboers B, Groot GMC, Hagendoorn J, Hartgrink HH, Horsthuis K, Hurks R, Jenniskens SFM, Kater M, Kazemier G, Kist JW, Klaase JM, Knapen RRMM, Kruimer JWH, Lamers ABGN, Leclercq WKG, Liefers GJ, Manusama ER, Meier MAJ, Melenhorst MCAM, Mieog JSD, Molenaar QI, Nielsen K, Nijkamp MW, Nieuwenhuijs VB, Nota IMGC, Op de Beeck B, Overduin CG, Patijn GA, Potters FH, Ratti F, Rietema FJ, Ruiter SJS, Schouten EAC, Schreurs WH, Serafino G, Sietses C, Slooter GD, Smits MLJ, Soykan EA, Spaargaren GJ, Stommel MWJ, Timmer FEF, van Baardewijk LJ, van Dam RM, van Delden OM, van den Bemd BAT, van den Bergh JE, van den Boezem PB, van der Leij C, van der Meer RW, van der Meijs BBM, van der Ploeg APT, van der Reijden JJ, van Duijvendijk P, van Erkel AR, van Geel AM, Van Heek NT, van Manen CJ, van Rijswijk CSP, van Waesberghe JHTM, Versteeg KS, Vink T, Zijlstra IAJ, Zonderhuis BM, Swijnenburg RJ, van den Tol MP, Meijerink MR. Thermal ablation versus surgical resection of small-size colorectal liver metastases (COLLISION): an international, randomised, controlled, phase 3 non-inferiority trial. Lancet Oncol. 2025;26:187-199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 81] [Article Influence: 81.0] [Reference Citation Analysis (1)] |
| 172. | Ruers T, Van Coevorden F, Punt CJ, Pierie JE, Borel-Rinkes I, Ledermann JA, Poston G, Bechstein W, Lentz MA, Mauer M, Folprecht G, Van Cutsem E, Ducreux M, Nordlinger B; European Organisation for Research and Treatment of Cancer (EORTC); Gastro-Intestinal Tract Cancer Group; Arbeitsgruppe Lebermetastasen und tumoren in der Chirurgischen Arbeitsgemeinschaft Onkologie (ALM-CAO); National Cancer Research Institute Colorectal Clinical Study Group (NCRI CCSG). Local Treatment of Unresectable Colorectal Liver Metastases: Results of a Randomized Phase II Trial. J Natl Cancer Inst. 2017;109:djx015. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 542] [Cited by in RCA: 495] [Article Influence: 55.0] [Reference Citation Analysis (5)] |
| 173. | Wong J, Cooper A. Local Ablation for Solid Tumor Liver Metastases: Techniques and Treatment Efficacy. Cancer Control. 2016;23:30-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 174. | Long Y, Liang Y, Li S, Guo J, Wang Y, Luo Y, Wu Y. Therapeutic outcome and related predictors of stereotactic body radiotherapy for small liver-confined HCC: a systematic review and meta-analysis of observational studies. Radiat Oncol. 2021;16:68. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 175. | van Tilborg AA, Scheffer HJ, de Jong MC, Vroomen LG, Nielsen K, van Kuijk C, van den Tol PM, Meijerink MR. MWA Versus RFA for Perivascular and Peribiliary CRLM: A Retrospective Patient- and Lesion-Based Analysis of Two Historical Cohorts. Cardiovasc Intervent Radiol. 2016;39:1438-1446. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 68] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 176. | Mustafa AR, Miyasato D, Wehrenberg-Klee E. Synergizing Thermal Ablation Modalities with Immunotherapy: Enough to Induce Systemic Antitumoral Immunity? J Vasc Interv Radiol. 2024;35:185-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 177. | Qin S, Chen M, Cheng AL, Kaseb AO, Kudo M, Lee HC, Yopp AC, Zhou J, Wang L, Wen X, Heo J, Tak WY, Nakamura S, Numata K, Uguen T, Hsiehchen D, Cha E, Hack SP, Lian Q, Ma N, Spahn JH, Wang Y, Wu C, Chow PKH; IMbrave050 investigators. Atezolizumab plus bevacizumab versus active surveillance in patients with resected or ablated high-risk hepatocellular carcinoma (IMbrave050): a randomised, open-label, multicentre, phase 3 trial. Lancet. 2023;402:1835-1847. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 418] [Cited by in RCA: 370] [Article Influence: 123.3] [Reference Citation Analysis (9)] |
| 178. | Zhu XD, Li KS, Sun HC. Adjuvant therapies after curative treatments for hepatocellular carcinoma: Current status and prospects. Genes Dis. 2020;7:359-369. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 48] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 179. | Seligmann J, Koessler T, Mauer M, Evrard S, Freedman J, Gootjes EC, Guckenberger M, Govaerts AS, Giraut A, Ricke J, Folprecht G, Arnold D, Giasafaki P, Ducreux M, Antunes S, Ruers T. Durvalumab and tremelimumab plus local partial tumour ablation (radiofrequency ablation or stereotactic radiotherapy) in patients with unresectable liver metastases from metastatic colorectal cancer: results of the EORTC-1560-GITCG multicentre, single-arm phase II study (ILOC). ESMO Open. 2025;10:105508. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 180. | Nieuwenhuizen S, Dijkstra M, Puijk RS, Geboers B, Ruarus AH, Schouten EA, Nielsen K, de Vries JJJ, Bruynzeel AME, Scheffer HJ, van den Tol MP, Haasbeek CJA, Meijerink MR. Microwave Ablation, Radiofrequency Ablation, Irreversible Electroporation, and Stereotactic Ablative Body Radiotherapy for Intermediate Size (3-5 cm) Unresectable Colorectal Liver Metastases: a Systematic Review and Meta-analysis. Curr Oncol Rep. 2022;24:793-808. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 42] [Article Influence: 10.5] [Reference Citation Analysis (1)] |
| 181. | Ayers M, Lunceford J, Nebozhyn M, Murphy E, Loboda A, Kaufman DR, Albright A, Cheng JD, Kang SP, Shankaran V, Piha-Paul SA, Yearley J, Seiwert TY, Ribas A, McClanahan TK. IFN-γ-related mRNA profile predicts clinical response to PD-1 blockade. J Clin Invest. 2017;127:2930-2940. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3229] [Cited by in RCA: 2989] [Article Influence: 332.1] [Reference Citation Analysis (4)] |
| 182. | Liu Z, Zhu Y, Xie H, Zou Z. Immune-mediated hepatitis induced by immune checkpoint inhibitors: Current updates and future perspectives. Front Pharmacol. 2022;13:1077468. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 51] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 183. | Bolte FJ, Hall RD, Shah NL. Immune checkpoint inhibitor-related liver toxicity. Clin Liver Dis (Hoboken). 2022;20:93-96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 184. | Zheng C, Huang S, Lin M, Hong B, Ni R, Dai H, Lin X, Yang J. Hepatotoxicity of immune checkpoint inhibitors: What is Currently Known. Hepatol Commun. 2023;7:e0063. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 37] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 185. | Li S, Li K, Wang K, Yu H, Wang X, Shi M, Liang Z, Yang Z, Hu Y, Li Y, Liu W, Li H, Cheng S, Ye L, Yang Y. Low-dose radiotherapy combined with dual PD-L1 and VEGFA blockade elicits antitumor response in hepatocellular carcinoma mediated by activated intratumoral CD8(+) exhausted-like T cells. Nat Commun. 2023;14:7709. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 63] [Article Influence: 21.0] [Reference Citation Analysis (0)] |