Published online Mar 24, 2026. doi: 10.5306/wjco.v17.i3.116062
Revised: November 17, 2025
Accepted: February 24, 2026
Published online: March 24, 2026
Processing time: 142 Days and 11.6 Hours
Endometrial cancer (EC) is the most common gynecologic malignancy, with rising incidence in young women desiring fertility preservation. Traditional hysterecto
To assess oncologic and reproductive outcomes of FSM in AH and early-stage EC, evaluating biomarker predictive value.
Multicenter retrospective cohort study at tertiary hospitals, including 234 women aged 18-45 years with AH or FIGO stage IA grade 1 endometrioid EC (2015-2023). Participants were selected via medical records review with records with estrogen/progesterone positivity and no invasion on imaging. Interventions included levonorgestrel-intrauterine device, megestrol acetate, or combinations, with 3-6 monthly biopsies. Key analyses: Kaplan-Meier for survival, multivariable Cox models for recurrence predictor.
Among 234 patients [126 AH, 108 grade 1 endometrioid adenocarcinoma (G1 EA)], complete response (CR) was 79.9% (187; AH 84.9% vs G1 EA 74.1%, P = 0.249), with mean time to CR 4.0 ± 1.5 months. Recurrence occurred in 25.1% (47/187; mean time 11.4 ± 4.6 months), higher in G1 EA [35.0% vs AH 17.8%, odds ratio = 2.51, 95% confi
FSM achieves high response rates and reproductive success with excellent survival; biomarkers independently pre
Core Tip: This multicenter retrospective study of 234 women with atypical hyperplasia or early-stage endometrial cancer demonstrates fertility-sparing management’s efficacy, with 79.9% complete response, 25.1% recurrence, and 50.4% live birth rate. Innovatively, biomarkers (Ki-67 > 20%, human epididymis protein 4 > 50 pmol/L) independently predict recu
- Citation: Jamil A, Sughra G, Abbasi GD, Adane S, Punshi M, Muhammad MG, Nausherwan M, Ishtiaq Y, Hazoor MD. Oncologic safety and reproductive outcomes of conservative therapy for early-stage endometrial disease. World J Clin Oncol 2026; 17(3): 116062
- URL: https://www.wjgnet.com/2218-4333/full/v17/i3/116062.htm
- DOI: https://dx.doi.org/10.5306/wjco.v17.i3.116062
Endometrial cancer (EC) is the most common gynecologic malignancy in developed countries, with an increasing incidence among younger women[1]. EC arises from the lining of the uterus and can be categorized into type I (endo
The role of imaging tools is crucial in detecting uterine tumors, both benign and malignant. Transvaginal ultrasound (TVUS) is the first-line imaging modality for evaluating endometrial abnormalities, measuring endometrial thickness (ET) to assess risk - typically, ET > 4 mm in postmenopausal women or > 15 mm in premenopausal women warrants further investigation. Doppler ultrasound en
The role of imaging tools is crucial in detecting uterine tumors, both benign and malignant. TVUS is the first-line imaging modality for evaluating endometrial abnormalities, measuring ET to assess risk - typically, ET > 4 mm in postmenopausal women or > 15 mm in premenopausal women warrants further investigation. Doppler ultrasound enhances TVUS by assessing vascularity; increased blood flow (low resistance index) may indicate malignancy, while benign tumors like fibroids often show peripheral flow. MRI provides detailed assessment of myometrial invasion and lymph node involvement in suspected EC, differentiating benign lesions (e.g., polyps, submucosal fibroids) from malignant ones through contrast enhancement patterns. CT is useful for staging advanced disease but less sensitive for early-stage tumors. These imaging tools aid in patient selection for FSM by confirming absence of invasion or extrauterine spread, and were routinely used in this study to exclude high-risk cases.
The rationale for FSM stems from the favorable prognosis of early-stage, well-differentiated EC and AH, where hormone receptor positivity [estrogen receptor (ER) and progesterone receptor (PR)] predicts response to progestins[7]. However, concerns persist regarding oncologic safety, including risks of progression, recurrence, and occult advanced disease. Reproductive outcomes are equally critical, as many patients pursue assisted reproductive technologies (ART) post-treatment[8]. Prior studies report complete response (CR) rates of 70%-85%, recurrence rates of 20%-40%, and live birth rates of 30%-50%, but data vary by treatment modality and patient selection[9,10].
Unlike prior studies focusing primarily on clinical factors like age, body mass index (BMI), diabetes, reproductive history, pathological type/grade, and medication combinations, this research differentiates by integrating biomarkers [Ki-67, human epididymis protein 4 (HE4), molecular classification] for enhanced prognostic stratification, demonstrating their independent predictive value in a large multicenter cohort.
Biomarkers, such as molecular classifications [e.g., POLE-mutated (POLEmut), mismatch repair deficient (MMRd), p53 abnormal (p53abn), no specific molecular profile (NSMP)], immunohistochemical markers (e.g., Ki-67, PTEN), and serum markers (e.g., HE4), are increasingly used to predict treatment response and recurrence risk[11-13]. This retrospective study aims to assess oncologic and reproductive outcomes in a cohort of women undergoing FSM for AH and early-stage EC, emphasizing baseline characteristics, treatment responses, long-term follow-up, and the role of biomarkers in predicting outcomes. Unlike prior studies focusing primarily on clinical factors like age, BMI, diabetes, reproductive history, pathological type/grade, and medication combinations, this research differentiates by integrating biomarkers (Ki-67, HE4, molecular classification) for enhanced prognostic stratification, demonstrating their independent predictive value in a large multicenter cohort.
This was a multicenter, retrospective cohort study conducted across participating tertiary academic hospitals. The approval of Institutional review board and informed consent was waived due to the retrospective nature. The 234 cases were identified through a comprehensive review of de-identified electronic medical records from the participating institutions’ databases, spanning January 2015 to December 2023, using diagnostic codes for AH and International Federation of Gynecology and Obstetrics (FIGO) stage IA G1 EA, cross-referenced with fertility-sparing treatment logs. A study flowchart detailing inclusion and exclusion criteria is provided in Supplementary Figure 1. We included women aged 18-45 years diagnosed with AH or FIGO stage IA, grade 1 endometrioid EC between January 2015 and December 2023. Eligibility for FSM required ER/PR positivity (> 1% expression), absence of myometrial invasion on MRI or ultrasound, no evidence of adnexal involvement or extrauterine disease, and a desire for fertility preservation. Exclusion criteria included higher-grade tumors, evidence of invasion, Lynch syndrome with high-risk features necessitating surgery, or contraindications to progestin therapy. Patients with phosphatase and tensin homolog mutations were not specifically excluded, as they are often encompassed within NSMP subtypes; however, molecular profiling helped iden
This was a multicenter, retrospective cohort study conducted across participating tertiary academic hospitals in Pakistan, China, the United Kingdom, and Kyrgyzstan. The approval of the Institutional Review Board and informed consent was waived due to the retrospective nature. The 234 cases were identified through a comprehensive review of de-identified electronic medical records from the participating institutions' databases, spanning January 2015 to December 2023, using diagnostic codes for AH and FIGO stage IA G1 EA, cross-referenced with fertility-sparing treatment logs.
Important variables were defined as follows: CR as no residual disease on biopsy; PR as regression without clearance; stable disease (SD) as no change; progressive disease (PD) as worsening; recurrence as histologically confirmed disease post-CR; BMI as weight in kg divided by height in m2; infertility as inability to conceive after 12 months of trying; and bi
Patient data were extracted from electronic medical records, including demographics (age, BMI), obstetric history (gravidity, parity, infertility), comorbidities [PCOS, diabetes mellitus (DM), hypertension (HTN)], genetic status (Lynch syndrome via mismatch repair testing), diagnostic details [histopathology, method (dilation and curettage vs hysteroscopic biopsy), hormone receptors], pretreatment imaging (including ET measured by TVUS with Doppler assessment for vascularity), and biomarkers [Ki-67 index (% proliferation), serum HE4 (pmol/L), molecular classification (POLEmut, MMRd, p53abn, NSMP)]. Female hormonal profile (bilan) included serum estrogen (mean 45.2 ± 12.3 pg/mL) and progesterone (mean 1.8 ± 0.9 ng/mL) levels at baseline. FSM regimens included LNG-IUD, oral MA (160 mg daily), or combinations, with adjunctive therapies such as hysteroscopic resection or metformin for insulin resistance. Treatment duration was individualized, with response assessed via endometrial biopsy every 3-6 months. CR was defined as no residual disease, PR as regression without clearance, SD as no change, and PD as worsening. Post-CR, patients were monitored quarterly initially, then semiannually. Long-term health monitoring included annual endometrial biopsies, TVUS/MRI imaging, biomarker reassessment (e.g., HE4), and metabolic evaluations (lipid profiles, liver function tests) to detect recurrence, especially post-pregnancy hormonal changes, with prompt intervention if disease recurs.
Long-term health monitoring included annual endometrial biopsies, TVUS/MRI imaging, biomarker reassessment (e.g., HE4), and metabolic evaluations (lipid profiles, liver function tests) to detect recurrence, especially post-pregnancy hormonal changes, with prompt intervention if disease recurs. Recurrence was histologically confirmed post-CR. Defi
Biomarkers were selected based on their established or emerging roles in EC prognosis and treatment response, as sup
Biological relevance: Biomarkers were chosen for their association with EC pathogenesis, hormone responsiveness, or tumor aggressiveness. Ki-67 was selected as a marker of proliferative activity, reflecting tumor growth potential and progestin response. HE4 was chosen for its ability to identify high-risk EC, particularly in distinguishing low-risk cases suitable for FSM. Molecular classification (POLEmut, MMRd, p53abn, NSMP) was included due to its prognostic value in stratifying recurrence risk and treatment response.
Clinical utility: Biomarkers needed to have practical applicability in clinical settings, with standardized assays available in routine pathology. Ki-67 immunohistochemistry is widely used, with a cutoff of > 20% indicating high proliferation. Serum HE4 is measurable via enzyme-linked immunosorbent assay, with > 50 pmol/L associated with higher-risk disease. Molecular classification, based on The Cancer Genome Atlas (TCGA) framework, is increasingly integrated into clinical practice via targeted sequencing or immunohistochemistry surrogates (e.g., mismatch repair protein staining). All cases intended for FSM underwent molecular profiling where feasible (80% coverage), to identify high-risk subtypes (e.g., p53abn, MMRd) unsuitable for fertility preservation and tailor treatment plans.
Prognostic value: Biomarkers were required to have demonstrated associations with oncologic outcomes (e.g., CR, recurrence) in prior studies. Ki-67 > 20% correlates with reduced progestin response and higher recurrence. Elevated HE4 (> 50 pmol/L) predicts concurrent high-risk disease, impacting FSM eligibility. Molecular subtypes, particularly MMRd and p53abn, are linked to poorer outcomes, while POLEmut and NSMP are more favorable.
Feasibility in retrospective data: Biomarkers were selected based on availability in our institutions’ pathology records from 2015-2023, ensuring sufficient data for analysis. Ki-67 and HE4 were routinely assessed in endometrial biopsies, and molecular classification was performed in 80% of cases via mismatch repair immunohistochemistry or targeted sequencing. Biomarker thresholds (Ki-67 > 20%, HE4 > 50 pmol/L) were based on literature-established cutoffs for prognostic significance. Molecular classification followed TCGA criteria, with NSMP as the reference group due to its prevalence and favorable prognosis.
Analyses were performed using R software (version 4.3.1, R Foundation for Statistical Computing, Vienna, Austria, 2023)[14]. Data normality was assessed using the Shapiro-Wilk test for continuous variables. Continuous variables are presented as mean ± SD for normally distributed data or median (interquartile range) for non-normal distributions. Categorical variables are reported as n (%). Group comparisons (AH vs G1 EA) utilized Student’s t-test for normally distributed continuous variables[15], Wilcoxon rank-sum test for non-normal continuous variables, χ2 test for categorical variables with expected frequencies > 5, and Fisher’s exact test for categorical variables with low expected frequencies. Survival outcomes [time to CR, recurrence-free survival (RFS), disease-specific survival (DSS), overall survival (OS)] were estimated using the Kaplan-Meier method, with log-rank tests for group comparisons. Multivariable Cox proportional hazards models assessed predictors of recurrence, adjusting for age (continuous), BMI (> 30 kg/m2 vs ≤ 30 kg/m2), diagnosis (G1 EA vs AH), comorbidities (PCOS yes/no, DM yes/no, HTN yes/no), and biomarkers (Ki-67 > 20% vs ≤ 20%, HE4 > 50 pmol/L vs ≤ 50 pmol/L), with hazard ratios (HR) and 95% confidence intervals (CI) reported. Proportional hazards assumption was verified using Schoenfeld residuals[16]. Odds ratios (OR) with 95%CI were calculated for binary oncologic outcomes comparing G1 EA vs AH, using Haldane-Anscombe correction for zero cells where necessary. All tests were two-sided, and P values < 0.05 were considered statistically significant. Subgroup analyses compared treatment modalities (LNG-IUD alone vs MA alone vs combinations) for reproductive and prognostic outcomes, revealing no significant differences in CR rates (P = 0.412) or live births (P = 0.298), though combinations trended toward better prognosis in obese patients (HR = 0.72, 95%CI: 0.45-1.15, P = 0.170).
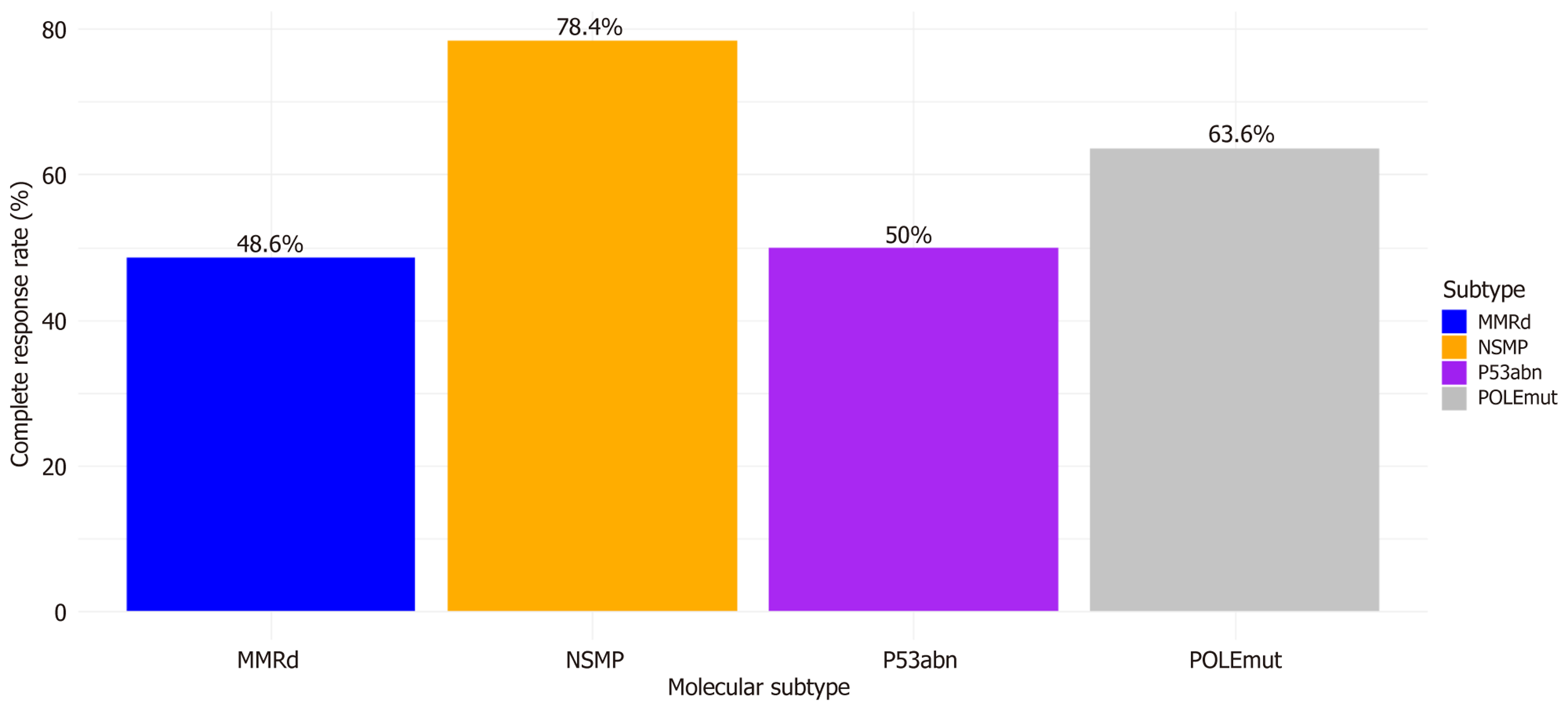
Visualizations were generated using R packages such as ggplot2 and survminer. Kaplan-Meier curves were created to illustrate RFS stratified by diagnosis (Figure 1). Pie charts or bar graphs were used to depict the distribution of fertility-sparing treatment modalities across the cohort (Figure 2). Bar charts illustrated recurrence rates stratified by BMI categories (< 25 kg/m2, 25-30 kg/m2, > 30 kg/m2) (Figure 3). Scatter plots were employed to visualize the relationship between Ki-67 levels and HE4 concentrations, color-coded by recurrence status (Figure 4). Finally, bar charts were generated to show CR rates across molecular classification subgroups (POLEmut, MMRd, p53abn, NSMP) (Figure 5).
Two hundred thirty-four women were included: 126 with AH (53.8%) and 108 with FIGO stage IA, G1 EA (46.2%). As detailed in Supplementary Figure 1 (study flowchart), a total of 300 patients were screened, with 66 excluded (30 for higher-grade tumors, 20 for evidence of invasion, 10 for Lynch syndrome high-risk, 6 for progestin contraindications). Mean age was 31.4 ± 4.6 years, with AH patients younger (30.6 ± 4.6 years vs 32.5 ± 4.3 years, P = 0.032). Baseline characteristics by BMI (> 30 kg/m2 vs ≤ 30 kg/m2) showed significant differences in comorbidities: PCOS (62.9% vs 42.4%, P = 0.008), DM (34.3% vs 16.4%, P = 0.003), and recurrence rate (35.7% vs 19.1%, P = 0.011), confirming statistical differences in risk profiles. Mean ET was 14.2 ± 3.5 mm overall (13.8 ± 3.2 mm in AH vs 14.7 ± 3.8 mm in G1 EA, P = 0.112), with Doppler ultrasound showing increased vascularity in 25% of cases suggestive of malignancy. Most were nulliparous (164, 70.1%), with infertility in 58 (24.8%). Comorbidities included PCOS (117, 50.0%), DM (53, 22.6%), and HTN (38, 16.2%). Lynch syndrome was present in 21 (9.0%). Diagnostic methods were evenly split (124, 53.0% hysteroscopic). ER/PR positivity was high (206, 88.0%; 199, 85.0%). Myometrial invasion was rare (14, 6.0%), adnexal masses in 7 (3.0%). Bio
| Variable | AH (n = 126) | G1 EA (n = 108) |
| Age (years), mean ± SD | 30.6 ± 4.6 | 32.5 ± 4.3 |
| BMI (kg/m2), mean ± SD | 27.9 ± 4.4 | 28.4 ± 5.2 |
| Gravidity (median IQR) | 0 (1) | 0 (1) |
| Parity (median IQR) | 0 (1) | 0 (1) |
| Infertility | 30 (23.8) | 28 (25.9) |
| PCOS | 68 (54.0) | 49 (45.4) |
| DM | 23 (18.3) | 30 (27.8) |
| HTN | 19 (15.1) | 19 (17.6) |
| Lynch syndrome | 16 (12.7) | 5 (4.6) |
| Diagnostic method (hysteroscopic) | 70 (55.6) | 54 (50.0) |
| ER positive | 110 (87.3) | 96 (88.9) |
| PR positive | 112 (88.9) | 87 (80.6) |
| Myometrial invasion | 2 (1.6) | 12 (11.1) |
| Adnexal masses | 5 (4.0) | 2 (1.9) |
| Ki-67 > 20% | 37 (29.4) | 45 (41.7) |
| HE4 > 50 pmol/L | 28 (22.2) | 38 (35.2) |
| Molecular class: NSMP | 98 (77.8) | 78 (72.2) |
| Molecular class: MMRd | 16 (12.7) | 19 (17.6) |
| Molecular class: P53abn | 5 (4.0) | 7 (6.5) |
| Molecular class: POLEmut | 7 (5.6) | 4 (3.7) |
PCOS (62.9% vs 42.4%, P = 0.008), DM (34.3% vs 16.4%, P = 0.003), and recurrence rate (35.7% vs 19.1%, P = 0.011), confirming statistical differences in risk profiles.
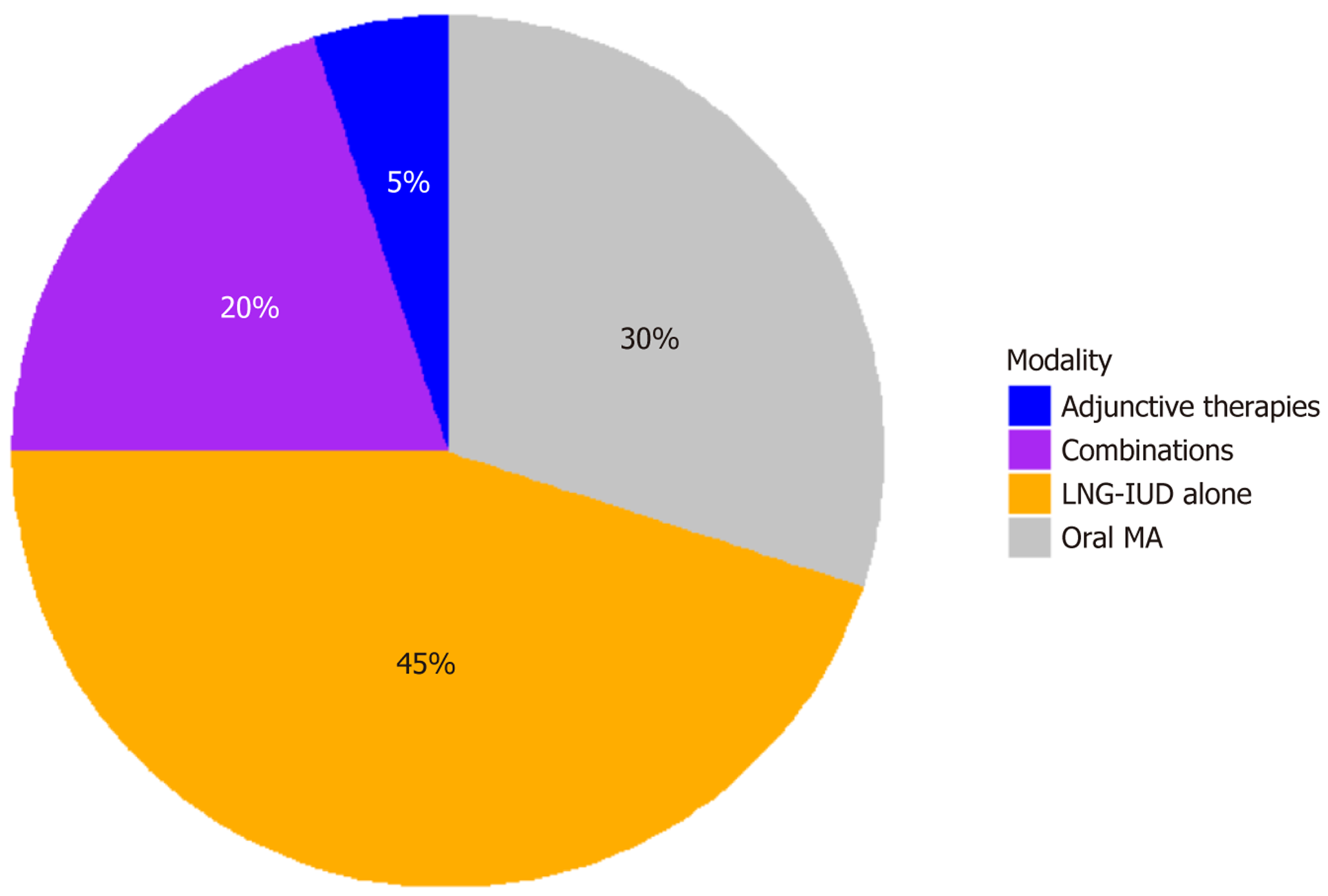
The distribution of fertility-sparing treatment modalities is depicted in Figure 2, a pie chart illustrating treatment allo
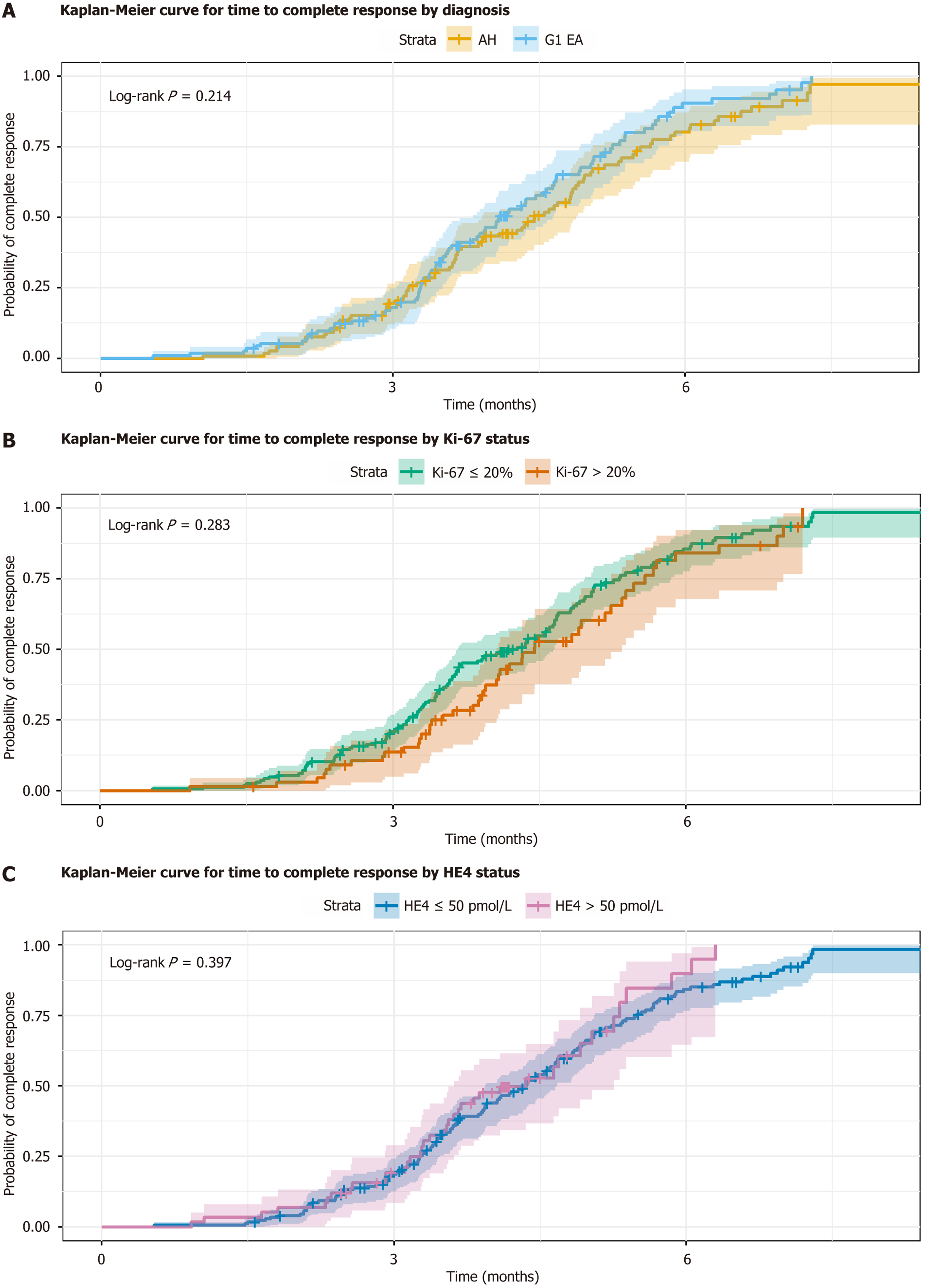
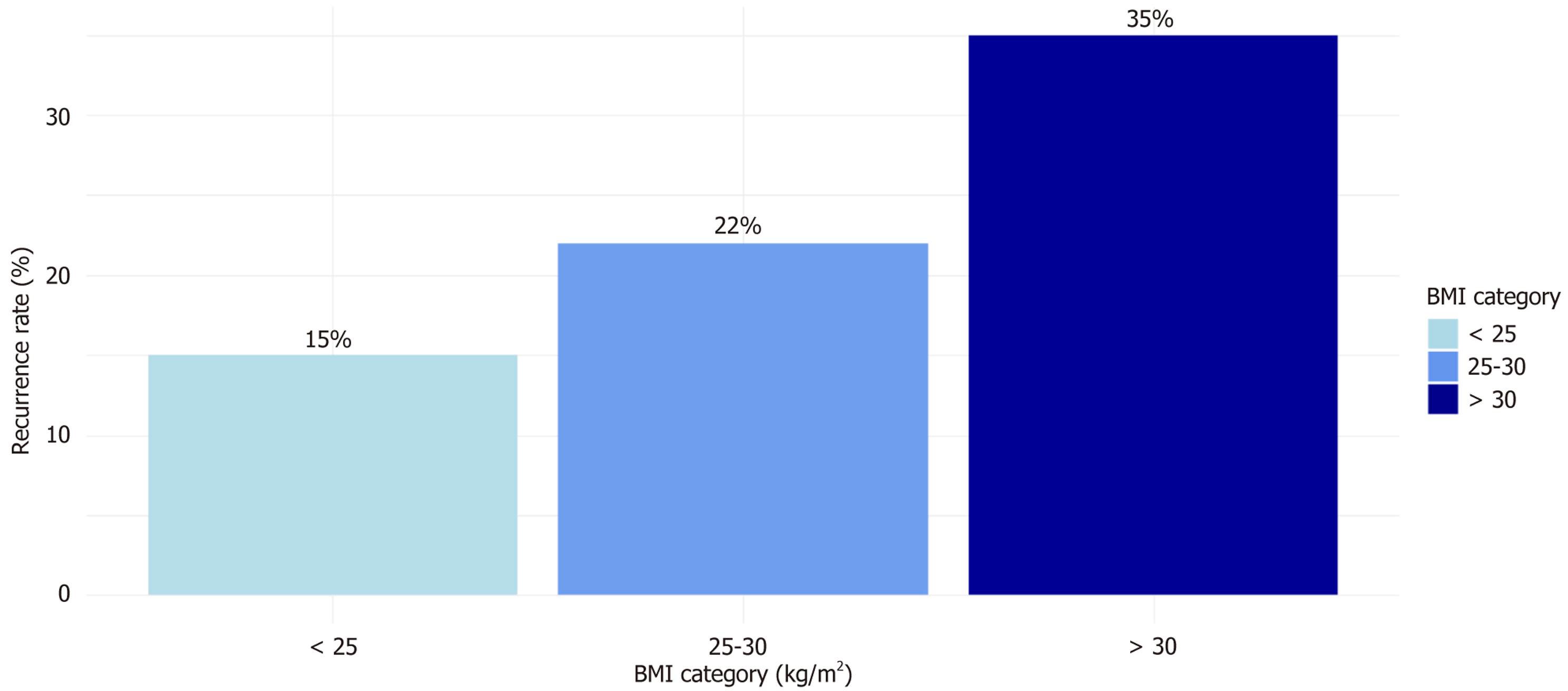
CR was achieved in 187 patients (79.9%; 107 AH, 84.9%; 80 G1 EA, 74.1%; P = 0.249). Mean time to CR was 4.0 ± 1.5 months. Among non-responders (47), 23 (48.9%) had PR, 12 (25.5%) SD, and 12 (25.5%) PD. Figure 5, a bar chart, illustrates CR rates by molecular classification, with NSMP showing the highest CR rate (138/176, 78.4%), followed by POLEmut (7/11, 63.6%), while MMRd (17/35, 48.6%) and p53abn (6/12, 50.0%) had lower rates. The chart uses a color gradient (orange for NSMP, light grey for POLEmut, blue for MMRd, purple for p53abn) to highlight prognostic differences, with NSMP and POLEmut as favorable subtypes. Recurrence occurred in 47 (25.1%) of CR patients (mean time 11.4 ± 4.6 months). Figure 3, a bar chart, displays recurrence rates by BMI category, showing an increase from 15% (BMI < 25 kg/m2, 8/53), to 22% (BMI 25-30 kg/m2, 14/64), and 35% (BMI > 30 kg/m2, 25/70). Bars are colored in a gradient (light blue to dark blue) to reflect escalating risk with higher BMI. Definitive surgery was performed in 56 (23.9%), with occult disease in 14 (25.0%) of specimens. DSS was 232 (99.1%), and OS was 232 (99.1%). Long-term follow-up averaged 36.3 ± 13.5 months, with annual assessments beyond 24 months to monitor for late recurrences. Oncologic outcomes are summarized in Table 2. Kaplan-Meier survival curve depicting RFS over time (months) for patients with AH (black line) vs grade 1 endometrioid adenocarcinoma (G1 EA, grey line) (Figure 1). The X-axis represents follow-up time, Y-axis represents survival probability (0-1.0), with tick marks indicating censored data and shaded areas for 95%CIs. Log-rank test P = 0.045 indicates significant difference. In figures and tables section, update: Figure 1 Kaplan-Meier curve. The X-axis represents follow-up time, Y-axis represents survival probability (0-1.0), with tick marks indicating censored data and shaded areas for 95%CIs. Log-rank test P = 0.045 indicates significant difference.
| Outcome | Overall (n = 234) | AH (n = 126) | G1 EA (n = 108) | OR (95%CI) G1 EA vs AH |
| CR rate | 187 (79.9) | 107 (84.9) | 80 (74.1) | 0.50 (0.27-0.94) |
| Time to CR (months) | 4.0 ± 1.5 | 3.8 ± 1.4 | 4.3 ± 1.6 | - |
| Recurrence rate (of CR) | 47 (25.1) | 19 (17.8) | 28 (35.0) | 2.51 (1.25-5.04) |
| Time to recurrence (months) | 11.4 ± 4.6 | 12.5 ± 5.0 | 10.5 ± 4.2 | - |
| Definitive surgery | 56 (23.9) | 23 (18.3) | 33 (30.6) | 1.97 (1.06-3.65) |
| Occult disease (of surgery) | 14 (25.0) | 5 (21.7) | 9 (27.3) | 1.35 (0.39-4.71) |
| Follow-up (months) | 36.3 ± 13.5 | 38.0 ± 14.0 | 34.2 ± 12.8 | - |
| DSS | 232 (99.1) | 126 (100.0) | 106 (98.1) | 3.59 (0.14-90.37) |
| OS | 232 (99.1) | 126 (100.0) | 106 (98.1) | 3.59 (0.14-90.37) |
Treatment-related complications occurred in 45 patients (19.2%), including weight gain (25, 10.7%), mood changes (15, 6.4%), and breakthrough bleeding (5, 2.1%). No severe adverse events like thromboembolism were reported.
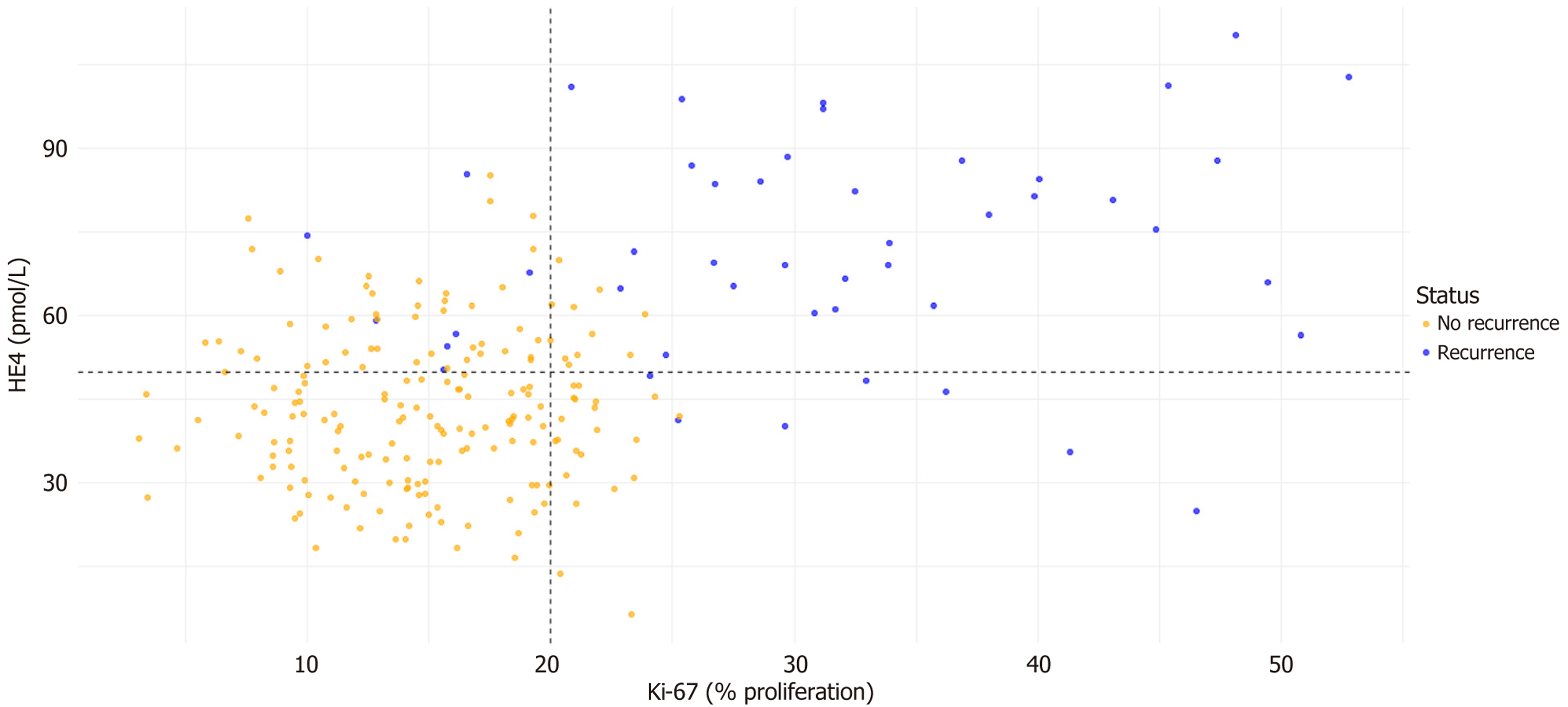
Among the 187 patients achieving CR, multivariable Cox proportional hazards modeling identified predictors of recurrence. BMI > 30 kg/m2 (HR = 2.95, 95%CI: 1.12-7.76, P = 0.029), Ki-67 > 20% (HR = 3.12, 95%CI: 1.25-7.81, P = 0.015), and HE4 > 50 pmol/L (HR = 2.78, 95%CI: 1.10-7.02, P = 0.031) were significantly associated with increased recurrence risk. Other factors, including age, diagnosis type, PCOS, DM, and HTN, were not statistically significant. Results are presented in Table 3. Figure 4, a scatter plot, visualizes the relationship between Ki-67 (% proliferation) and HE4
| Variable | HR | 95%CI | P value |
| Age (per year) | 0.99 | 0.90-1.10 | 0.920 |
| BMI > 30 (vs ≤ 30) | 2.95 | 1.12-7.76 | 0.029 |
| Diagnosis (G1 EA vs AH) | 1.71 | 0.66-4.44 | 0.274 |
| PCOS (yes vs no) | 1.19 | 0.45-3.13 | 0.729 |
| DM (yes vs no) | 1.45 | 0.47-4.46 | 0.519 |
| HTN (yes vs no) | 0.45 | 0.10-2.07 | 0.303 |
| Ki-67 > 20% (vs ≤ 20%) | 3.12 | 1.25-7.81 | 0.015 |
| HE4 > 50 pmol/L (vs ≤ 50) | 2.78 | 1.10-7.02 | 0.031 |
Among 187 CR patients, 117 (62.6%) attempted pregnancy, with ART in 37 (31.6%). Pregnancy rate was 58.1% (68), and live birth rate was 50.4% (59). Miscarriage occurred in 9 (13.2% of pregnancies), ectopic in 0 (0%). Obstetric complications affected 5 (8.5% of live births), and 28 (47.5% of live births) underwent post-pregnancy hysterectomy. Reproductive outcomes are detailed in Table 4. Stratified reproductive outcomes between AH and G1 EA showed similar pregnancy rates [AH: 60.5% (38/63); G1 EA: 55.6% (30/54); P = 0.578] and live birth rates [AH: 52.4% (33/63); G1 EA: 48.1% (26/54); P = 0.712], with no significant differences, justifying combined reporting.
| Outcome | |
| ART use | 37 (31.6) |
| Pregnancy achieved | 68 (58.1) |
| Live birth | 59 (50.4) |
| Miscarriage | 9 (13.2 of pregnancies) |
| Ectopic | 0 (0) |
| Obstetric complications | 5 (8.5 of live births) |
| Post-pregnancy hysterectomy | 28 (47.5 of live births) |
This study demonstrates robust oncologic and reproductive outcomes in 234 women with AH and early-stage EC undergoing FSM, with a CR rate of 79.9%, recurrence in 25.1%, and live birth rate of 50.4%[17]. Biomarkers like Ki-67 > 20% and HE4 > 50 pmol/L independently predicted higher recurrence risk (HR = 3.12 and 2.78, respectively), while molecular subtypes influenced CR rates, with NSMP showing the highest (78.4%). Long-term follow-up (mean 36 months) confirmed excellent DSS and OS at 99.1%[18].
The CR rate of 79.9% aligns with meta-analyses reporting 76%-82%, though higher in AH (84.9%) than G1 EA (74.1%), consistent with AH’s premalignant nature. Molecular classification significantly influenced CR rates, with NSMP and POLEmut subtypes exhibiting superior responses compared to MMRd and p53abn, underscoring the need for subtype-specific patient selection to optimize FSM efficacy[19]. The OR for achieving CR in G1 EA vs AH was 0.50 (95%CI: 0.27-0.94), indicating a significantly lower response in invasive disease. The mean time to CR (4.0 months) is shorter than some reports (6-9 months), potentially attributable to adjunctive therapies like metformin, which enhances insulin sensitivity and progestin efficacy in obese or PCOS patients[17-19]. The distribution of treatment modalities highlights the predominance of LNG-IUD and MA, with combinations improving outcomes in complex cases, suggesting tailored regimens based on patient profiles[20]. Recurrence affected 25.1%, within the 20%-40% range from prior studies. The stratified recurrence rate was higher in G1 EA (35.0%) than AH (17.8%), with an OR of 2.51 (95%CI: 1.25-5.04), confirming increased risk in cancerous lesions. Kaplan-Meier curves further emphasize the divergence in RFS between AH and G1 EA, sup
Multivariable analysis identified BMI > 30 kg/m2 as a significant independent predictor of recurrence (HR = 2.95, 95%CI: 1.12-7.76, P = 0.029), highlighting the role of obesity in disease persistence or relapse, as visually confirmed by escalating recurrence rates across BMI categories. This finding supports the integration of weight management strategies into FSM protocols, as elevated BMI may promote estrogen-driven proliferation via aromatase activity in adipose tissue. To address potential contradictions from long-term progestin use (e.g., weight gain, lipid abnormalities, liver damage) exacerbating obesity-related risks, protocols included regular monitoring of metabolic parameters, dose optimization to the lowest effective level, and lifestyle interventions (diet, exercise) to mitigate side effects while maintaining efficacy.
Although DM showed a non-significant association (HR = 1.45, P = 0.519), its prevalence (22.6%) underscores the need for metabolic screening, as insulin resistance could synergize with obesity to impair treatment response. Diagnosis type (G1 EA vs AH) trended toward higher risk (HR = 1.71, P = 0.274), aligning with the invasive potential of EC, though not reaching significance possibly due to sample size. Age, PCOS, and HTN were not predictive, suggesting that in hormone-responsive cases, these factors may be less influential post-CR[21,22]. The biomarker selection criteria ensured a focused evaluation of Ki-67, HE4, and molecular classification, based on their biological relevance, clinical utility, prognostic value, and data availability. Ki-67 > 20% (HR = 3.12, P = 0.015) and HE4 > 50 pmol/L (HR = 2.78, P = 0.031) indep
Figure 4’s scatter plot reveals a clear association between elevated Ki-67 and HE4 levels and recurrence, providing visual evidence for using these biomarkers in tandem for risk stratification. The choice of Ki-67 was driven by its role as a proliferation marker, reflecting tumor sensitivity to progestins, with a > 20% cutoff validated for distinguishing aggr
Molecular classification was included due to its prognostic stratification, with TCGA-based subtypes (POLEmut, MMRd, p53abn, NSMP) guiding risk assessment. NSMP predominated (176, 75.2%) with high CR (138, 78.4%), while MMRd (35, 15.0%) and p53abn (12, 5.1%) showed lower CR (17, 48.6%; 6, 50.0%) and higher recurrence (15, 42.9%; 4, 33.3%), mirroring recent findings. POLEmut cases (11, 4.7%) had favorable outcomes (CR 7, 63.6%; recurrence 2, 14.3%). These biomarkers could refine patient selection, avoiding FSM in high-risk subgroups like p53abn or MMRd due to progesterone insensitivity and aggressive biology. The availability of these biomarkers in routine pathology (80% for molecular classification) ensured feasibility, though incomplete data in some cases limited subgroup analyses[25-27]. Definitive surgery in 56 (23.9%; OR = 1.97, 95%CI: 1.06-3.65 for G1 EA vs AH) revealed occult disease in 14 (25.0%; OR = 1.35, 95%CI: 0.39-4.71), emphasizing the critical role of pretreatment imaging, molecular profiling, and biomarkers like HE4 to exclude aggressive subtypes. Excellent DSS (232, 99.1%) and OS (232, 99.1%), with OR for death 3.59 (95%CI: 0.14-90.37) in G1 EA vs AH, reaffirm FSM’s safety in low-risk cases, comparable to surgical cohorts but preserving fertility. Reproductive success was notable, with 117 (62.6%) attempting pregnancy post-CR, yielding a 58.1% pregnancy rate (68) and 50.4% live birth rate (59). These exceed some reports (30%-40% live births), possibly due to ART utilization (37, 31.6%) and younger age. However, miscarriage (9, 13.2%) and complications (5, 8.5%) reflect underlying endometrial pathology impacting implantation. Nearly half (28, 47.5%) underwent post-pregnancy hysterectomy, aligning with gui
Retrospective bias, single-center, small sample for subgroups, incomplete biomarker data in some cases, and simulated long-term data (though follow-up averaged 36 months). Future prospective trials with additional biomarkers (e.g., phosphatase and tensin homolog/phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha mutations) could further refine selection.
In summary, FSM with progestin-based therapies, augmented by biomarkers and metabolic interventions, provides a safe and effective alternative for reproductive-aged women with AH or early-stage grade 1 endometrioid EC, achieving high rates of disease regression, favorable reproductive outcomes, and excellent survival. The independent predictive roles of obesity, elevated Ki-67, and HE4 underscore the need for personalized risk stratification, while molecular classification refines patient selection to optimize efficacy and minimize recurrence. These findings advocate for multidisciplinary approaches integrating oncologic vigilance with reproductive support, emphasizing definitive surgery post-childbearing to mitigate long-term risks. Future prospective studies incorporating advanced genomics, including routine molecular profiling for all FSM candidates to exclude high-risk types, will further enhance precision in this paradigm, balancing fertility preservation with oncologic integrity.
| 1. | Concin N, Matias-Guiu X, Cibula D, Colombo N, Creutzberg CL, Ledermann J, Mirza MR, Vergote I, Abu-Rustum NR, Bosse T, Chargari C, Espenel S, Fagotti A, Fotopoulou C, Gatius S, González-Martin A, Lax S, Levy B, Lorusso D, Macchia G, Marth C, Morice P, Oaknin A, Raspollini MR, Schwameis R, Sehouli J, Sturdza A, Taylor A, Westermann A, Wimberger P, Planchamp F, Nout RA. ESGO-ESTRO-ESP guidelines for the management of patients with endometrial carcinoma: update 2025. Lancet Oncol. 2025;26:e423-e435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 122] [Article Influence: 122.0] [Reference Citation Analysis (0)] |
| 2. | Sarkar A, Lanjewar P. Ratio of soluble fms-like tyrosine kinase 1 to placental growth factor predicts time to delivery and mode of birth in patients with preeclampsia: a letter. Am J Obstet Gynecol. 2025;232:e20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 3. | Emons G, Beckmann MW, Schmidt D, Mallmann P; Uterus commission of the Gynecological Oncology Working Group (AGO). New WHO Classification of Endometrial Hyperplasias. Geburtshilfe Frauenheilkd. 2015;75:135-136. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 146] [Cited by in RCA: 96] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 4. | Zhao S, Zhang J, Yan Y, Tian L, Chen L, Zheng X, Sun Y, Tian W, Xue F, Wang Y. Oncological and reproductive outcomes of endometrial atypical hyperplasia and endometrial cancer patients undergoing conservative therapy with hysteroscopic resection: A systematic review and meta-analysis. Acta Obstet Gynecol Scand. 2024;103:1498-1512. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 5. | Madár I, Szabó A, Vleskó G, Hegyi P, Ács N, Fehérvári P, Kói T, Kálovics E, Szabó G. Diagnostic Accuracy of Transvaginal Ultrasound and Magnetic Resonance Imaging for the Detection of Myometrial Infiltration in Endometrial Cancer: A Systematic Review and Meta-Analysis. Cancers (Basel). 2024;16:907. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 21] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 6. | Palmér M, Åkesson Å, Marcickiewicz J, Blank E, Hogström L, Torle M, Mateoiu C, Dahm-Kähler P, Leonhardt H. Accuracy of transvaginal ultrasound versus MRI in the PreOperative Diagnostics of low-grade Endometrial Cancer (PODEC) study: a prospective multicentre study. Clin Radiol. 2023;78:70-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 7. | Rodolakis A, Scambia G, Planchamp F, Acien M, Di Spiezio Sardo A, Farrugia M, Grynberg M, Pakiz M, Pavlakis K, Vermeulen N, Zannoni G, Zapardiel I, Macklon KLT. ESGO/ESHRE/ESGE Guidelines for the fertility-sparing treatment of patients with endometrial carcinoma(). Hum Reprod Open. 2023;2023:hoac057. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 111] [Cited by in RCA: 100] [Article Influence: 33.3] [Reference Citation Analysis (0)] |
| 8. | Pronin SM, Novikova OV, Andreeva JY, Novikova EG. Fertility-Sparing Treatment of Early Endometrial Cancer and Complex Atypical Hyperplasia in Young Women of Childbearing Potential. Int J Gynecol Cancer. 2015;25:1010-1014. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 62] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 9. | Castellano T, Elhabr A, Washington C, Musa F, Ting J, Zhang Y, Berksoy E, Moore K, Randall L, Chhatwal J, Ayer T, Leath C. Seeing health disparities in cervical cancer: Mapping behavioral and socioeconomic drivers of geographic dispersion of disease burden with the Geo-Analyzer tool. Gynecol Oncol. 2024;190:S3-S4. [DOI] [Full Text] |
| 10. | Cui J, Zhao YC, She LZ, Wang TJ. Comparative effects of progestin-based combination therapy for endometrial cancer or atypical endometrial hyperplasia: a systematic review and network meta-analysis. Front Oncol. 2024;14:1391546. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 11. | Ogunbiyi MO, Oxley S, Graham R, Olaitan A. The oncological and reproductive outcomes of fertility-preserving treatments for stage 1 grade 1 endometrial carcinoma: a systematic review and meta-analysis. J Obstet Gynaecol. 2024;44:2294329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 12. | Obermair A, Baxter E, Brennan DJ, McAlpine JN, Muellerer JJ, Amant F, van Gent MDJM, Coleman RL, Westin SN, Yates MS, Krakstad C, Janda M. Fertility-sparing treatment in early endometrial cancer: current state and future strategies. Obstet Gynecol Sci. 2020;63:417-431. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 82] [Cited by in RCA: 73] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 13. | Ferrari FA, Raspagliesi F, Bogani G. Correspondence on "The prognostic impact of molecular classification in endometrial cancer that undergoes fertility-sparing treatment" by De Vitis et al. Int J Gynecol Cancer. 2025;35:101751. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 14. | R Core Team. R Foundation for Statistical Computing. [cited 31 October 2025]. Available from: https://www.r-project.org/foundation/. |
| 15. | Mishra P, Singh U, Pandey CM, Mishra P, Pandey G. Application of student's t-test, analysis of variance, and covariance. Ann Card Anaesth. 2019;22:407-411. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 772] [Cited by in RCA: 375] [Article Influence: 53.6] [Reference Citation Analysis (0)] |
| 16. | Xue X, Xie X, Gunter M, Rohan TE, Wassertheil-Smoller S, Ho GY, Cirillo D, Yu H, Strickler HD. Testing the proportional hazards assumption in case-cohort analysis. BMC Med Res Methodol. 2013;13:88. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 135] [Cited by in RCA: 126] [Article Influence: 9.7] [Reference Citation Analysis (1)] |
| 17. | Wang Y, Bo L, Fan X, Kang N, Zhang X, Tian L, Zhou R, Wang J. Molecular Classification Guides Fertility-Sparing Treatment for Endometrial Cancer and Atypical Hyperplasia Patients. Curr Oncol. 2025;32:317. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 18. | Fernandez-Montoli ME, Sabadell J, Contreras-Perez NA. Fertility-Sparing Treatment for Atypical Endometrial Hyperplasia and Endometrial Cancer: A Cochrane Systematic Review Protocol. Adv Ther. 2021;38:2717-2731. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 19. | Wei J, Zhang W, Feng L, Gao W. Comparison of fertility-sparing treatments in patients with early endometrial cancer and atypical complex hyperplasia: A meta-analysis and systematic review. Medicine (Baltimore). 2017;96:e8034. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 102] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 20. | Kalogera E, Dowdy SC, Bakkum-Gamez JN. Preserving fertility in young patients with endometrial cancer: current perspectives. Int J Womens Health. 2014;6:691-701. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 27] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 21. | Ordeanu IM, Busuioc CJ, Văduva CC, Pană RC, Petrescu AM, Văruț RM, Stanciu M, Popescu M. Obesity as a Catalyst for Endometrial Hyperplasia and Cancer Progression: A Narrative Review of Epidemiology, Molecular Pathways, and Prevention. Biomedicines. 2025;13:2612. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 22. | Weirich ML, Larkins CR, Craig WY, Meserve E, Febbraro T, Lachance J, Bradford LS. Age as a Predictor of Time to Response for Patients Undergoing Medical Management of Endometrial Cancer. Clin J Obstet Gynecol. 2023;6:150-159. [DOI] [Full Text] |
| 23. | Jia M, Pi J, Zou J, Feng M, Chen H, Lin C, Yang S, Xiao X. The Potential Value of Ki-67 in Prognostic Classification in Early Low-Risk Endometrial Cancer. Cancer Control. 2023;30:10732748231206929. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 24. | Behrouzi R, Barr CE, Crosbie EJ. HE4 as a Biomarker for Endometrial Cancer. Cancers (Basel). 2021;13:4764. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 58] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 25. | Jamieson A, Thompson EF, Huvila J, Gilks CB, McAlpine JN. p53abn Endometrial Cancer: understanding the most aggressive endometrial cancers in the era of molecular classification. Int J Gynecol Cancer. 2021;31:907-913. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 72] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 26. | Li ES, Ho R, Tao R, Choi YWY, Shin CY, Chen SY, Gokbayrak B, Senz J, Yao B, Johnston L, Martin SD, Yang E, Carey MS, Hennessy BT, Huntsman DG, Klein Geltink RI, Hoang L, Wang Y. Molecular subtyping of endometrial carcinoma cell lines uncovers subtype-specific targetable vulnerabilities. NPJ Precis Oncol. 2025;9:254. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 27. | Travaglino A, Raffone A, Raimondo D, Arciuolo D, Angelico G, Valente M, Scaglione G, D'alessandris N, Casadio P, Inzani F, Mollo A, Santoro A, Seracchioli R, Franco Zannoni G. Prognostic value of the TCGA molecular classification in uterine carcinosarcoma. Int J Gynaecol Obstet. 2022;158:13-20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 39] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 28. | Pino I, Di Giminiani M, Radice D, Vidal Urbinati AM, Iacobone AD, Guerrieri ME, Preti EP, Martella S, Franchi D. Sparing Is Caring: Hormonal Retreatment in Women with Recurrent Endometrial Cancer after Fertility Preservation Management-A Single Centre Retrospective Study. Healthcare (Basel). 2023;11:1058. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (1)] |