Published online Apr 28, 2026. doi: 10.4329/wjr.v18.i4.119851
Revised: March 5, 2026
Accepted: April 7, 2026
Published online: April 28, 2026
Processing time: 75 Days and 21.7 Hours
Pulmonary arteriovenous fistulas (PAVFs) are abnormal vascular communications between pulmonary arteries and veins that may cause hypoxemia and paradoxical embolism. Because many patients are asymptomatic, PAVFs are often detected incidentally on chest computed tomography (CT). Accurate identification of PAVFs is clinically important for appropriate management; however, small or atypical lesions may be overlooked during routine interpretation. Com
To evaluate the detectability of PAVFs on chest CT using an AI-based CAD sys
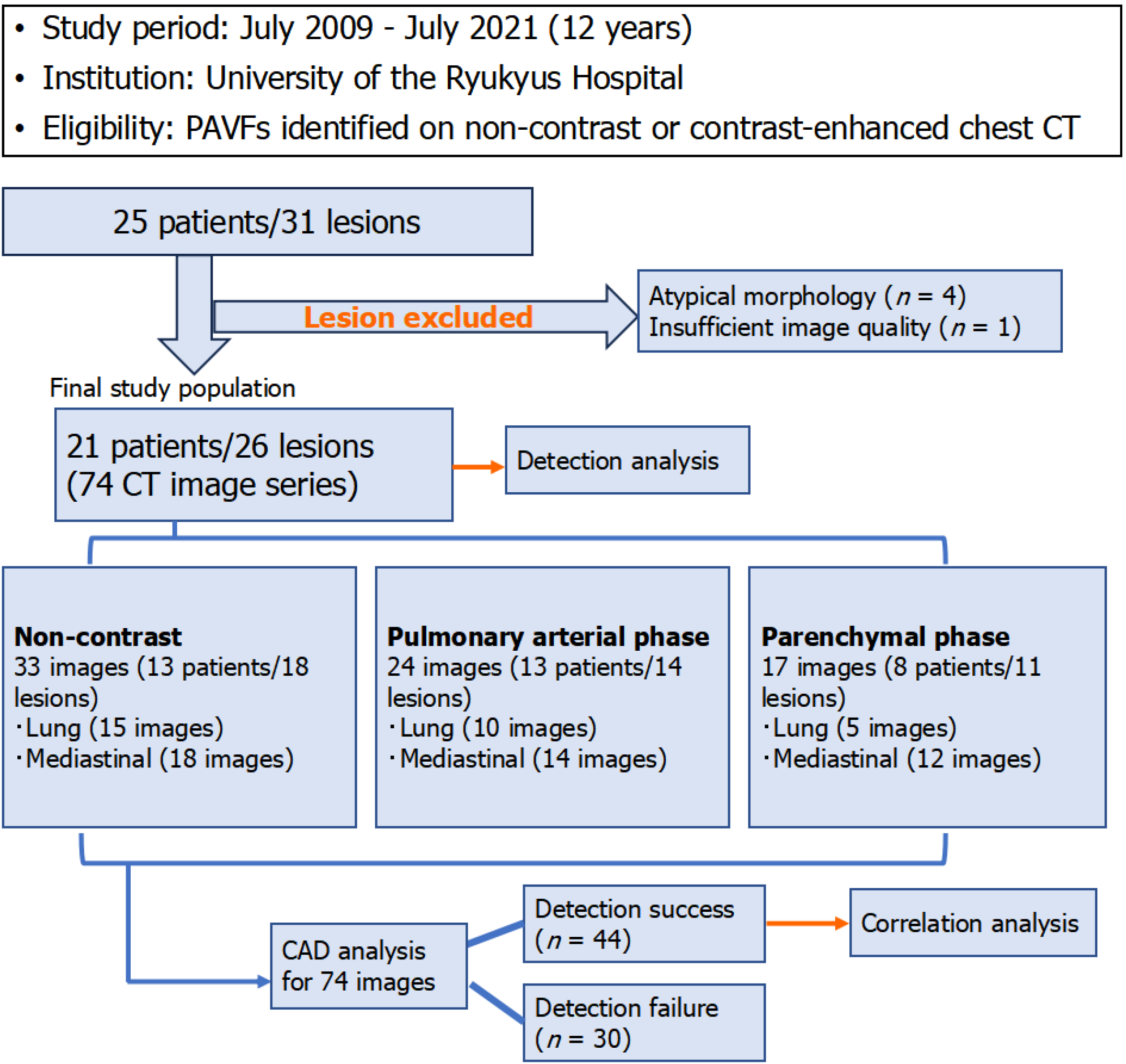
This retrospective observational study included 21 patients with 26 PAVFs identified at University of the Ryukyus Hospital between 2009 and 2021. Chest CT images, including non-contrast and contrast-enhanced scans, were analyzed using a commercially available AI-based lung nodule CAD system. Detection perfor
Among the 26 PAVFs, 15 lesions (58%) were consistently detected, 2 lesions (8%) were detected under certain imaging conditions, and 9 lesions (35%) were not detected, yielding an overall detection success rate of 65% (17/26). Detection rates did not differ significantly according to contrast phase (58% for non-contrast, 71% for pulmonary arterial phase, and 47% for parenchymal phase) or window setting (61% for lung window vs 58% for mediastinal window). Detection success was higher for complex-type lesions than for simple-type lesions (100% vs 59%, P = 0.26). CAD-derived maximum lesion length correlated strongly with manual measurements (r = 0.90, P < 0.001), as did CAD-derived lesion volume (r = 0.92, P < 0.001).
A lung nodule-based AI-CAD system detected a substantial proportion of PAVFs on chest CT and provided reliable quantitative measurements, supporting its potential adjunctive role in PAVF detection and follow-up.
Core Tip: Pulmonary arteriovenous fistulas (PAVFs) are vascular lesions, frequently detected on chest computed tomography. This study demonstrated that an artificial intelligence-based computer-aided detection (CAD) system originally developed for pulmonary nodule detection identified PAVFs with a detection rate of 65% and provided reliable quantitative measurements of lesion length. Although the detection performance was lower than that previously reported for pulmonary nodules, CAD-based analysis showed strong agreement with manual measurements. These findings suggest that lung nodule CAD systems may have broader clinical utility beyond their original purpose, including a potential role as adjunct tools for the opportunistic detection and longitudinal follow-up of PAVFs.
- Citation: Azama K, Tsuchiya N, Toyosato S, Yonemoto K, Nishie A. Artificial intelligence-based lung nodule detection for pulmonary arteriovenous fistulas on chest computed tomography. World J Radiol 2026; 18(4): 119851
- URL: https://www.wjgnet.com/1949-8470/full/v18/i4/119851.htm
- DOI: https://dx.doi.org/10.4329/wjr.v18.i4.119851
Pulmonary arteriovenous fistulas (PAVFs), also referred to as pulmonary arteriovenous malformations, are abnormal vascular communications between the pulmonary artery and vein without an intervening capillary bed. These lesions often include a saccular dilated structure, referred to as a venous sac, located between the feeding artery and the draining vein[1]. Although many patients remain asymptomatic, PAVFs may cause hypoxemia, dyspnea, cyanosis, or paradoxical embolism owing to right-to-left shunting. A feeding artery diameter ≥ 3 mm has been considered an indication for treatment because of the increased risk of complications[2,3]. PAVFs are strongly associated with hereditary hemorrhagic telangiectasia; however, sporadic cases can also occur and are not infrequently encountered in daily clinical practice. On computed tomography (CT), PAVFs are often incidentally detected as nodular to serpiginous vascular lesions[3]. Therefore, accurate identification of PAVFs on CT is crucial for appropriate diagnosis and management.
Computer-aided detection (CAD) systems for pulmonary nodule detection have been increasingly implemented in clinical practice since the early to mid-2000s[4]. The application of artificial intelligence (AI) techniques has further enhanced their performance[5]. The clinical utility of these systems has been increasingly demonstrated, and they are now widely used in daily clinical practice to improve diagnostic efficiency and reduce image interpretation time[6-8]. These systems are designed to extract lesion candidates by combining multiple image features, such as shape, attenuation gradients, and differences from surrounding structures, together with trained algorithms[9]. However, from the user’s perspective, the internal decision-making process of CAD systems is not fully transparent and is often regarded as a “black box”[10]. Previous studies have reported that vascular crossings, vessel curvature, and contact between vessels and the pleura are major sources of false-positive detections in lung nodule CAD systems, indicating that vascular structures may be misidentified as nodule candidates[11]. Conversely, this observation suggests that vascular structures such as PAVFs may also be recognized as nodular lesions by CAD systems.
However, the ability of AI-based lung nodule CAD systems to detect PAVFs has not been systematically evaluated. We hypothesized that such a system could identify PAVFs on chest CT because the venous sac of PAVFs often appears as a nodular structure. Therefore, this study aimed to assess the detectability of PAVFs on chest CT using an AI-based CAD system designed for the detection of lung nodules.
This retrospective observational study evaluated the detectability of PAVFs on chest CT using a commercially available AI-based CAD system for lung nodules. The study protocol was approved by our institutional review board, and the requirement for obtaining informed consent was waived because of the retrospective nature of the study.
Patients suspected of having PAVFs on CT were initially identified through the picture archiving and communication system database and their corresponding radiology reports at our institution between January 2009 and December 2021. The CT images of all candidate patients were reviewed by three radiologists (Kimei Azama, Nanae Tsuchiya, Shun Toyosato), and the presence of PAVFs was determined by consensus. PAVFs were defined as abnormal direct communications between a feeding artery (pulmonary artery) and a draining vein (pulmonary vein) without an intervening pulmo
Multidetector CT was performed using multiple CT scanners, including LightSpeed VCT (64-row scanner; GE HealthCare, Milwaukee, WI, United States), Aquilion Precision (160-row scanner; Canon Medical Systems, Otawara, Japan), and Aquilion ONE (320-row scanner; Canon Medical Systems). CT was performed at 100 kVp or 120 kVp with automatic exposure control. All examinations were conducted with the patient in the supine position during breath-holding at full inspiration.
Depending on the clinical indication, patients underwent either non-contrast CT or contrast-enhanced CT. For contrast-enhanced CT, an iodine-based contrast medium (100 mL; iopamiron 370 mgI/mL, Bayer, Osaka, Japan) was administered via a peripheral venous line at a rate of 1.5-3.0 mL/second. When contrast-enhanced CT was performed, imaging included either a pulmonary arterial phase, a parenchymal phase, or both. All contrast-enhanced CT scans were acquired without bolus tracking. The pulmonary arterial phase was obtained 25 seconds after the initiation of contrast injection, whereas the parenchymal phase was acquired 80 seconds after the initiation of contrast administration. All images were reconstructed with a slice thickness of 5 mm. The GE HealthCare system employed filtered back projection, whereas the Canon systems used a hybrid iterative reconstruction technique (Adaptive Iterative Dose Reduction 3D), weak strength. Lung window images were reconstructed using the FC52 kernel, whereas mediastinal window images were recon
When multiple CT examinations were available, only the initial examination was used for analysis. A total of 74 CT image series were analyzed (Figure 1). As CT protocols varied according to the clinical indication, not all patients underwent all imaging phases. For lung window assessment, lung window images reconstructed from non-contrast CT were used when available; otherwise, lung window images from contrast-enhanced CT were used. In contrast, mediastinal window images were evaluated separately for each imaging phase (non-contrast, pulmonary arterial, and parenchymal phases). Overall, the analyzed CT image series consisted of 33 non-contrast CT series (15 lung window and 18 mediastinal window images), 24 contrast-enhanced pulmonary arterial-phase series (10 lung window and 14 mediastinal window images), and 17 contrast-enhanced parenchymal-phase series (5 lung window and 12 mediastinal window images).
All CT images were analyzed using a commercially available CAD system for lung nodules[12]. This CAD system is based on a deep learning-based algorithm designed to identify pulmonary nodules on chest CT images and output their locations. According to the system specifications, the CAD algorithm is intended for use with chest CT images acquired under the following general conditions: Image matrix size of 512 × 512 pixels, slice thickness < 5 mm, interslice spacing < 5 mm, adult patients, non-contrast scans, and lung kernel reconstruction. The target lesions defined by the CAD system include pulmonary nodules located within the lung fields and meeting the following size criteria: Solid nodules ≥ 3 mm in diameter, part-solid nodules ≥ 5 mm, and pure ground-glass nodules ≥ 5 mm.
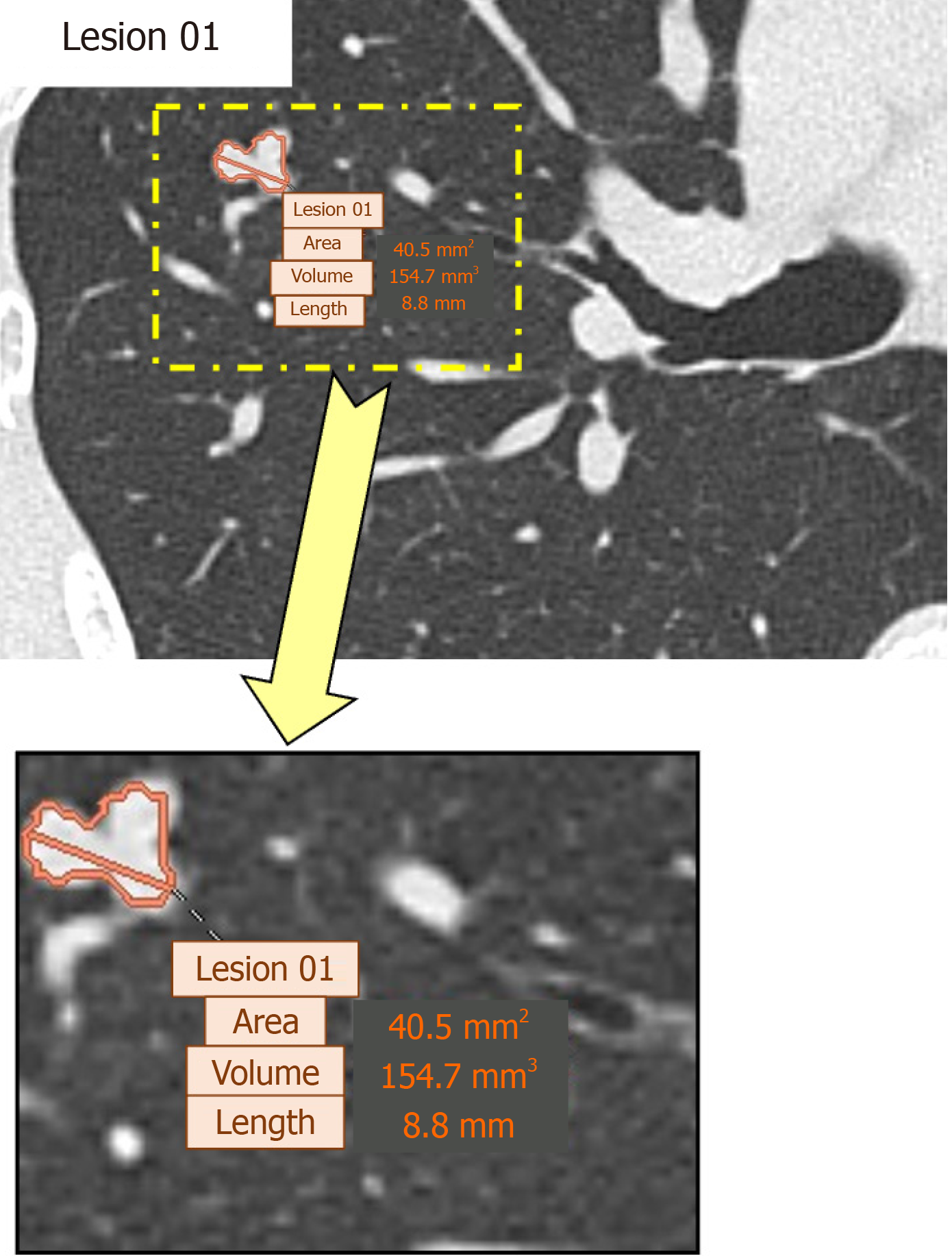
For analysis, the CAD system automatically processed axial CT images reconstructed at a slice thickness of 1 mm and highlighted potential lung nodule locations (Figure 2). The system provided quantitative measurements of the detected lesions, including area, volume, and maximum length; among these parameters, lesion volume and maximum length were recorded for analysis.
One radiologist (Nanae Tsuchiya) who was aware of the locations of the PAVFs evaluated whether the lesions were detected by the CAD system. Detection performance was classified into three categories, as follows: Category A: The PAVF was consistently detected regardless of imaging conditions (contrast phase and window setting); category B: The PAVF was detected under at least one phase/window combination; and category C: The PAVF was not detected under any imaging condition.
For each PAVF lesion, imaging-based evaluations were independently performed by two radiologists (Kimei Azama and Shun Toyosato) who were blinded to the CAD system results. In cases of disagreement, a final consensus was reached through discussion. The evaluated imaging parameters included lesion location (lung lobe) and the presence or absence of contact with adjacent structures, such as the mediastinum, chest wall, and diaphragm. In addition, lesions were classified according to their vascular structure as either simple or complex. The simple type was defined as having a single feeding artery and a single draining vein, whereas the complex type was defined as having multiple feeding arteries and/or draining veins[1]. Furthermore, the diameter of the feeding artery and the maximum lesion length were measured on non-contrast/contrast-enhanced CT images. The diameter of the feeding artery was defined as the maximum short-axis diameter measured at the most dilated portion proximal to the lesion, and the maximum lesion length was defined as the longest dimension of the lesion measured on multiplanar reconstructed images. The presence or absence of a treatment indication was also assessed based on whether the diameter of the feeding artery was ≥ 3 mm[2].
The lesion detection rate by the CAD system was calculated for each imaging condition. Differences in PAVF detection rates were evaluated according to imaging conditions, including non-contrast vs contrast-enhanced CT and lung vs mediastinal windows, using the χ2 test. To investigate factors associated with CAD detection performance, imaging parameters were compared between the successful detection group (categories A and B) and the failed detection group (category C). Categorical variables were compared using Fisher’s exact test, and continuous variables were compared using the Wilcoxon rank-sum test. Correlations between CAD-derived maximum length and lesion volume and the manually measured maximum length were evaluated using Pearson’s correlation coefficient. All data were analyzed using JMP software (version 17.2; SAS Institute Inc., Cary, NC, United States). Statistical significance was defined as a two-sided P value < 0.05. Exact P values are reported where applicable.
A total of 25 consecutive participants (31 lesions) suspected of having PAVFs were initially identified. Four patients with atypical vascular morphology and one patient in whom the feeding arteries and draining veins could not be clearly distinguished because of insufficient image quality were excluded. Ultimately, 21 patients with 26 lesions were enrolled in the study. The sample size was fixed because of the retrospective nature of the study. The baseline characteristics of the 21 included patients are summarized in Table 1. Two patients had been diagnosed with hereditary hemorrhagic telangiectasia (Osler-Weber-Rendu syndrome). Sixteen patients were asymptomatic, four patients had dyspnea, and one patient had hemoptysis. Comorbidities included heart failure in 2 patients and a history of cerebral infarction in 3 patients. Fourteen patients underwent transcatheter embolization, two patients underwent surgical treatment, and the remaining five patients were managed conservatively.
| Characteristic | Value | |
| Age (years) | 59 (16-85) | |
| Sex | Male | 6 |
| Female | 15 | |
| Underlying disease | Hereditary hemorrhagic telangiectasia | 2 |
| Symptoms | Dyspnea | 4 |
| Hemoptysis | 1 | |
| Asymptomatic | 16 | |
| Comorbidities | Heart failure | 2 |
| Cerebral infarction | 3 | |
| SpO2 (%) | 96 (94-99) | |
| Treatment | Endovascular embolization | 14 |
| Surgery | 2 | |
| Conservative management | 5 | |
Among the 26 lesions evaluated, 15 (58%) were detected in all imaging series and were thus classified as category A. Two lesions (8%) were detected in some imaging series and were classified as category B. The remaining nine lesions (35%) were not detected in any imaging series and were classified as category C. Consequently, the overall detection success rate of the CAD system, defined as lesions classified as category A or B, was 65% (17 of 26 lesions). For the two category B lesions, detection performance varied depending on the imaging series (Supplementary Table 1).
Table 2 summarizes the differences in CAD detection rates according to imaging conditions. With respect to contrast use, detection success was observed in 58% (19 of 33) of non-contrast CT images, 71% (17 of 24) of pulmonary arterial phase images, and 47% (8 of 17) of parenchymal phase images. No statistically significant difference in detection success was observed among these contrast phases (P = 0.29).
| Imaging conditions | Overall (image series, | Detection success (image series, n = 44) | Detection failure (image series, n = 30) | P value | |
| Contrast | Non-contrast | 33 | 19 (58) | 14 (42) | 0.29 |
| Pulmonary arterial phase | 24 | 17 (71) | 7 (29) | ||
| Parenchymal phase | 17 | 8 (47) | 9 (53) | ||
| Window setting | Lung window | 31 | 19 (61) | 12 (39) | 0.79 |
| Mediastinal window | 43 | 25 (58) | 18 (42) | ||
Regarding window settings, detection success was achieved in 61% (19 of 31) of lung window settings and 58% (25 of 43) of mediastinal window settings. There was no significant difference in detection rates between lung and mediastinal window settings (P = 0.79).
Factors associated with successful detection by the CAD system are summarized in Table 3. There was no significant difference in detection success according to whether the lesion was in contact with adjacent structures, such as the pleura, pericardium, and diaphragm. With regard to lesion morphology, detection success was higher in complex-type lesions than in simple-type lesions (100% vs 59%); however, this difference was not statistically significant (Fisher’s exact test, P = 0.26). Regarding treatment indication, lesions with a feeding artery diameter of ≥ 3 mm showed a higher detection success rate than those without a treatment indication (72% vs 50%); however, this difference was not statistically significant (P = 0.28).
| Characteristic | Factors | Overall (n = 26) | Detection success (n = 17) | Detection failure (n = 9) | P value |
| Location | Right upper lobe | 7 | 3 | 4 | - |
| Right middle lobe | 3 | 2 | 1 | ||
| Right lower lobe | 4 | 3 | 1 | ||
| Left upper lobe | 3 | 2 | 1 | ||
| Lingular segment | 6 | 4 | 2 | ||
| Left lower lobe | 3 | 3 | 0 | ||
| Contact with adjacent structures | Present | 19 | 13 (68) | 6 (32) | 0.63 |
| Absent | 6 | 3 (50) | 3 (50) | ||
| Morphology1 | Simple type | 22 | 13 (59) | 9 (41) | 0.26 |
| Complex type | 4 | 4 (100) | 0 (0) | ||
| Treatment indication2 | Present | 18 | 13 (72) | 5 (28) | 0.38 |
| Absent | 8 | 4 (50) | 4 (50) | ||
| Feeding artery diameter (mm) | 3.4 (3.0, 4.0) | 3.4 (2.4, 4.8) | 0.47 | ||
| Maximum lesion length (mm) | 6.5 (4.9, 9.6) | 5.5 (2.5, 11.0) | 0.65 | ||
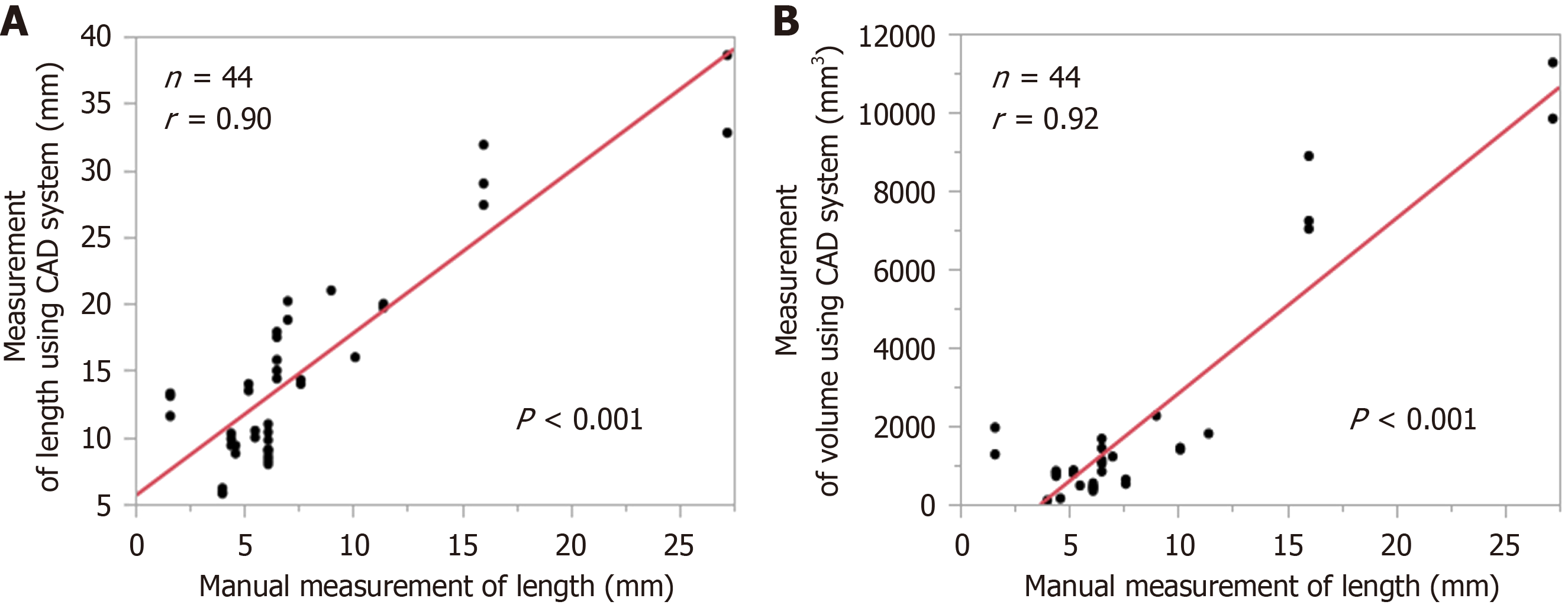
Strong correlations were observed between CAD-derived measurements and manual measurements performed by radiologists. The maximum lesion length measured by the CAD system showed a strong positive correlation with the manually measured maximum lesion length (r = 0.90, P < 0.001; Figure 3A). In addition, CAD-derived lesion volume was strongly correlated with the manually measured maximum lesion length (r = 0.92, P < 0.001; Figure 3B).
In this retrospective study, we evaluated the detectability of PAVFs on chest CT using a commercially available AI-based CAD system originally developed for pulmonary nodule detection. The overall detection success rate of the CAD system was 65%, indicating that a substantial proportion of PAVFs could be identified despite the system not being specifically trained for detecting vascular lesions. However, this detection rate was lower than the approximately 80%-96% detection rates previously reported for pulmonary nodules[5,13,14].
One plausible explanation for the lower detection rate observed for PAVFs compared with pulmonary nodules lies in the fundamental morphological differences between these entities. Pulmonary nodules are typically discrete, approximately spherical lesions, whereas PAVFs represent continuous vascular structures composed of feeding arteries, a venous sac, and draining veins[3,4]. How deep learning-based CAD systems interpret such complex vascular configurations as nodule-like lesions remain unclear, particularly given the limited transparency of the internal decision-making processes of these algorithms.
Another important consideration is that failure of the CAD system to detect a PAVF may also reflect correct system operation. The present CAD system was originally designed for the detection of pulmonary nodules, which are mass-forming lesions, and it is therefore possible that PAVFs were correctly recognized as vascular lesions and intentionally excluded from the detection targets. Accordingly, the observed detection success rate of 65% may reflect not only the proportion of PAVFs detected by the system but also the proportion of PAVFs that were misidentified as nodular lesions. From this perspective, the CAD system could be interpreted as having misclassified up to 65% of PAVF lesions in this dataset.
Detection success rate was higher for complex-type than for simple-type lesions; however, this difference did not reach statistical significance. Simple-type PAVFs are composed of a single feeding artery and a single draining vein, and the aneurysmal portion is often relatively localized and exhibits a nodule-like configuration. In contrast, complex-type PAVFs may involve multiple vessels and therefore present with more complex vascular morphology, resulting in a greater degree of morphological mismatch with nodule detection algorithms. This morphological difference may have contributed to the observed differences in CAD detectability.
At the beginning of this study, we hypothesized that PAVFs in contact with adjacent anatomical structures, such as the mediastinum, chest wall, or diaphragm, would demonstrate reduced detectability owing to overlapping with surrounding tissues and partial volume effects. This hypothesis was based on the concept that the performance of pulmonary nodule CAD systems is strongly influenced by the characteristics of the training datasets used during development[11,15]. Unexpectedly, however, no significant differences in detection rates were observed according to lesion location or contact with adjacent anatomical structures. These findings suggest that anatomical proximity may not represent a major limiting factor for CAD-based detection of PAVFs.
The ability of a lung nodule-based CAD system to detect PAVFs may offer several potential clinical advantages. PAVFs are relatively rare and often asymptomatic, and they are frequently detected incidentally on chest CT examinations[3]. As lung nodule CAD systems are already widely used in routine clinical practice, opportunistic detection of PAVFs using existing CAD systems could improve lesion recognition without requiring additional screening workflows. Moreover, CAD-based detection may serve as a safety net in non-specialized reading environments by drawing attention to potential vascular abnormalities that might otherwise be overlooked[15].
In addition to detection performance, we also evaluated the accuracy of CAD-based quantitative assessment. Strong positive correlations were observed between CAD-derived maximum lesion length and lesion volume and the manually measured maximum lesion length. These findings indicate that CAD-derived quantitative measurements are highly consistent with conventional manual measurements and support the reliability of the CAD system as a quantitative assessment tool.
From a clinical perspective, treatment indications for PAVFs are generally determined based on the diameter of the feeding artery rather than the maximum lesion length or volume[2]. However, changes in lesion size during follow-up are important factors in determining the need for and the timing of therapeutic intervention[3]. In this context, the ability of the CAD system to quantify changes in lesion length and volume in a reproducible manner may be useful for the long-term follow-up of PAVFs.
From a clinical workflow perspective, misclassification of PAVFs as pulmonary nodules may increase radiologist workload. False-positive CAD output can increase image interpretation time and may prompt additional scrutiny or follow-up recommendations. In routine clinical practice, CAD-positive findings are reviewed by radiologists and subsequently accepted or dismissed. Awareness that vascular lesions such as PAVFs may be flagged by nodule-detection algorithms could enable faster reinterpretation of CAD marks and help avoid unnecessary follow-up imaging. Thus, although these detections represent algorithmic misclassification, they may also allow opportunistic identification of clinically significant vascular abnormalities when interpreted appropriately.
Several limitations of this study should be acknowledged. First, not all lesions were confirmed by angiography; angiographic confirmation was available primarily in cases that underwent interventional treatment. Second, the detection performance of the same CAD system for pulmonary nodules was not independently evaluated at our institution, which precluded a direct comparison of detection performance between pulmonary nodules and PAVFs under identical conditions. Third, the relatively small number of cases may have limited the statistical power of subgroup analyses. Finally, the CAD system was originally developed for non-contrast chest CT images[12]. Despite this design constraint, the system demonstrated the potential to detect PAVFs on contrast-enhanced CT images commonly used in routine clinical practice.
This study suggests that a CAD system originally developed for pulmonary nodule detection may have potential clinical utility beyond its intended purpose, including the detection and longitudinal assessment of PAVFs. However, to optimize detection accuracy and interpretability for vascular lesions, the development of CAD systems specifically designed for vascular abnormalities, including PAVFs, is warranted.
| 1. | Gossage JR, Kanj G. Pulmonary arteriovenous malformations. A state of the art review. Am J Respir Crit Care Med. 1998;158:643-661. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 430] [Cited by in RCA: 404] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 2. | White RI Jr, Lynch-Nyhan A, Terry P, Buescher PC, Farmlett EJ, Charnas L, Shuman K, Kim W, Kinnison M, Mitchell SE. Pulmonary arteriovenous malformations: techniques and long-term outcome of embolotherapy. Radiology. 1988;169:663-669. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 345] [Cited by in RCA: 324] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 3. | Shovlin CL. Pulmonary arteriovenous malformations. Am J Respir Crit Care Med. 2014;190:1217-1228. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 181] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 4. | Armato SG 3rd, Giger ML, MacMahon H. Automated detection of lung nodules in CT scans: preliminary results. Med Phys. 2001;28:1552-1561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 127] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 5. | Katase S, Ichinose A, Hayashi M, Watanabe M, Chin K, Takeshita Y, Shiga H, Tateishi H, Onozawa S, Shirakawa Y, Yamashita K, Shudo J, Nakamura K, Nakanishi A, Kuroki K, Yokoyama K. Development and performance evaluation of a deep learning lung nodule detection system. BMC Med Imaging. 2022;22:203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 6. | Wang TW, Wang CK, Hong JS, Chao HS, Chen YM, Wu YT. Deep Learning in Thoracic Oncology: Meta-Analytical Insights into Lung Nodule Early-Detection Technologies. Cancers (Basel). 2025;17:621. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 7. | Ewals LJS, van der Wulp K, van den Borne BEEM, Pluyter JR, Jacobs I, Mavroeidis D, van der Sommen F, Nederend J. The Effects of Artificial Intelligence Assistance on the Radiologists' Assessment of Lung Nodules on CT Scans: A Systematic Review. J Clin Med. 2023;12:3536. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 8. | Geppert J, Asgharzadeh A, Brown A, Stinton C, Helm EJ, Jayakody S, Todkill D, Gallacher D, Ghiasvand H, Patel M, Auguste P, Tsertsvadze A, Chen YF, Grove A, Shinkins B, Clarke A, Taylor-Phillips S. Software using artificial intelligence for nodule and cancer detection in CT lung cancer screening: systematic review of test accuracy studies. Thorax. 2024;79:1040-1049. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 29] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 9. | Yuan R, Vos PM, Cooperberg PL. Computer-aided detection in screening CT for pulmonary nodules. AJR Am J Roentgenol. 2006;186:1280-1287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 91] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 10. | Pesapane F, Volonté C, Codari M, Sardanelli F. Artificial intelligence as a medical device in radiology: ethical and regulatory issues in Europe and the United States. Insights Imaging. 2018;9:745-753. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 390] [Cited by in RCA: 252] [Article Influence: 31.5] [Reference Citation Analysis (0)] |
| 11. | Wormanns D, Fiebich M, Saidi M, Diederich S, Heindel W. Automatic detection of pulmonary nodules at spiral CT: clinical application of a computer-aided diagnosis system. Eur Radiol. 2002;12:1052-1057. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 93] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 12. | Fujifilm Corporation. Lung Nodule Detection Program FS-AI688 [Internet]. Tokyo (2023). May, 2020. [cited 7 February 2026]. Available from: https://www.pmda.go.jp/PmdaSearch/kikiDetail/Resul tDataSetPDF/671001_30200BZX00150000_A_01_09. |
| 13. | Tsuchiya N, Kobayashi S, Nakachi R, Tomori Y, Yogi A, Iida G, Ito J, Nishie A. Application of a pulmonary nodule detection program using AI technology to ultra-low-dose CT: differences in detection ability among various image reconstruction methods. Jpn J Radiol. 2025;43:1303-1312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 14. | Fukumoto W, Yamashita Y, Kawashita I, Higaki T, Sakahara A, Nakamura Y, Awaya Y, Awai K. External validation of the performance of commercially available deep-learning-based lung nodule detection on low-dose CT images for lung cancer screening in Japan. Jpn J Radiol. 2025;43:634-640. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 15. | Kelly CJ, Karthikesalingam A, Suleyman M, Corrado G, King D. Key challenges for delivering clinical impact with artificial intelligence. BMC Med. 2019;17:195. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2018] [Cited by in RCA: 1325] [Article Influence: 189.3] [Reference Citation Analysis (4)] |