INTRODUCTION
Adrenal incidentaloma (ADI) is a common clinical finding. An ADI is generally defined as an adrenal mass greater than 1 cm in diameter, incidentally detected during imaging studies such as computed tomography (CT), magnetic resonance imaging (MRI), or ultrasonography performed for non-adrenal indications. Although most authors and authorities discuss ADIs larger than 1 cm, some authors propose that all incidentally discovered adrenal lesions should be considered and assessed as ADIs regardless of size, even if they are smaller than 1 cm[1,2]. Lesions identified during screening for hereditary syndromes or during oncologic staging for extra-adrenal malignancies are not considered to be ADIs[3-6]. The reported prevalence of ADIs varies according to the source of data (autopsy, surgical, or radiologic series) and patient selection (general vs specialized populations). In large autopsy series including more than 1000 patients, the prevalence ranges from 1.05% to 8.7%. The rate of ADI detection is increasing. Advances in diagnostic imaging have contributed to the increased detection rate of ADIs over the past three decades; the radiologic prevalence is expected to rise further, potentially approaching that observed in autopsy studies[3,7].
The pathogenesis of ADI/adrenocortical adenomas (ACA) involves a multifaceted interplay between genetic predispositions, advancing age, and modifiable metabolic risk factors[8]. While somatic mutations in genes such as CTNNB1, PRKACA, and GNAS1, alongside syndromes like multiple endocrine neoplasia type 2, are established contributors, the precise mechanisms of development remain under investigation[8]. Recent population-based data identifies increased body mass index (BMI) and tobacco use as significant independent risk factors for subsequent diagnosis, with tobacco use hypothesized to drive ACA by increasing oxidative stress, promoting angiogenesis, or activating the hypothalamic-pituitary-adrenal (HPA) axis[8]. Similarly, obesity-related hyperinsulinemia may exert mitogenic effects on the adrenal cortex via insulin and IGF-1 receptor activation; a bidirectional relationship may exist, since even subclinical cortisol secretion promotes visceral adiposity that further drives tumor growth[8]. Notably, although comorbidities like hypertension and dyslipidemia are frequently correlated with ADIs in cross-sectional studies, these associations may be confounded by imaging bias, as older patients with complex medical histories are more frequently subjected to the cross-sectional imaging that facilitates incidental discovery[8].
The increased prevalence of ADI is also attributed to the aging of the population and increasing medical orders for cross-sectional imaging. For instance, 245 CT scans per 1000 population were performed in 2016 in the United States[3]. This rising prevalence necessitates standardized, evidence-based protocols for evaluation[9].
A critical first step in the assessment of ADIs is distinguishing between “true ADIs” - identified in patients without prior malignancy or clinical suspicion of adrenal pathology - and adrenal lesions detected during oncologic staging[10]. This distinction has substantial clinical implications. In patients with known extra-adrenal malignancy, approximately 50% of adrenal masses represent metastatic disease, whereas in true incidentaloma cohorts the risk of malignancy is generally below 1%[10]. Failure to separate these diagnostic pathways may result in inappropriate extrapolation of benign-prevalence data into high-risk oncologic populations, thereby increasing the risk of false-negative interpretations[10]. The subsequent evaluation of a true ADI must address two overarching objectives: Exclusion of malignancy - including adrenocortical carcinoma (ACC) or metastatic disease - and identification of hormonal hypersecretion, even in the absence of overt clinical manifestations[11,12]. Hormonal excess states such as pheochromocytoma (PHEO), primary aldosteronism (PA), overt Cushing’s syndrome, or mild autonomous cortisol secretion (MACS) are associated with significant cardiovascular and systemic morbidity. Current management frameworks are largely informed by the 2023 European Society of Endocrinology (ESE) guidelines, which advocate risk-stratified imaging and biochemical testing[12]. Diagnostic workup integrates radiographic characterization with endocrine assessment and, in indeterminate or complex cases, often requires multidisciplinary team discussion to guide individualized management[11,12].
EPIDEMIOLOGY: PREVALENCE AND SHIFTING FUNCTIONAL BURDEN
Estimates of ADI prevalence in the general population, derived from imaging and autopsy studies, vary widely, with a mean value of approximately 3%-7%[3,13]. Prevalence generally increases with age[4]. A review of multiple studies reported a median prevalence of 1.9% based on surgical or radiologic series[3]. The high frequency of incidental detection, coupled with a broad differential diagnosis, underscores the associated clinical challenges[9].
Is the epidemiology of ADIs shifting?
Although the underlying etiology remains largely similar, with benign ACA representing the most frequent lesion, the functional profile may differ due to evolving awareness and changes in diagnostic criteria, particularly in older studies. The overall prevalence of hormonally active ADIs is around 15%[4]. MACS, formerly referred to as subclinical Cushing’s syndrome, is commonly identified in patients with ADIs. Reported prevalence varies widely, from approximately 5%-30% in population-based studies to as high as 48% in cohorts specifically examining benign ACAs. Emerging evidence indicates that MACS is more prevalent than previously recognized and represents the most common endocrine disorder associated with ADIs[4,13-15]. PA accounts for around 5%-10% of cases in the general population with hypertension and reaches to 30% among patients with resistant hypertension[16]. PA is demonstrated in a considerable proportion of the patients with ADIs, with the reported prevalence ranging from about 3.3% up to as high as 10% in some series[17]. It constitutes the leading cause of secondary hypertension, although its actual prevalence is probably underestimated, primarily as a result of suboptimal screening[18,19]. Furthermore, the prevalence of PHEO among cohorts with ADIs has been reported to be approximately 3.8%[20].
Partial reasons for heterogeneity between older and newer studies include differences in patient selection and the geographical regions of patients studied. On the other hand, increasing rates in recent publications may reflect heightened clinical awareness and increasingly sensitive biochemical thresholds for establishing a diagnosis of hormonal hyperfunction.
RADIOLOGICAL EVALUATION: CURRENT APPROACHES AND CAVEATS IN IMAGING
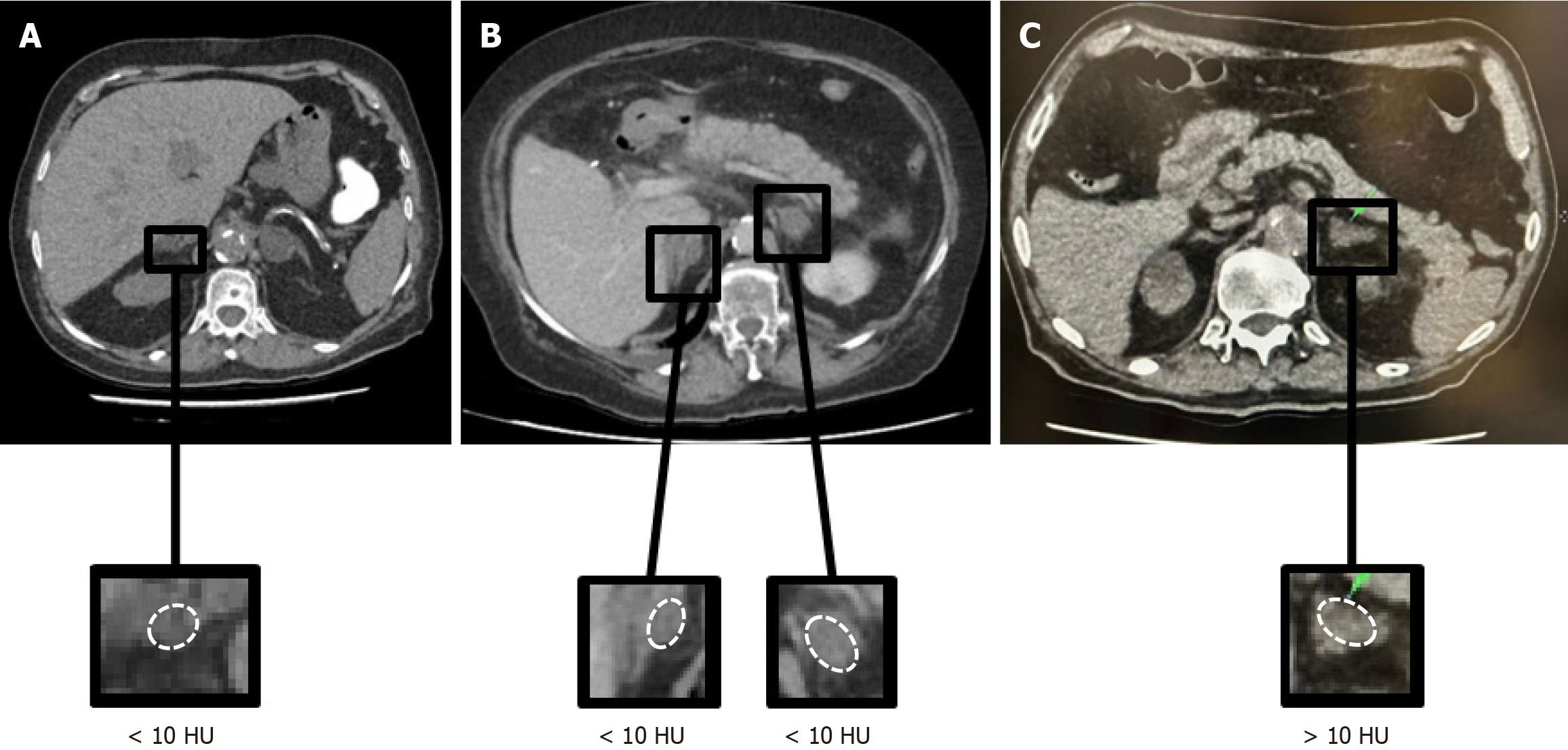
Radiological characterization represents the initial and central step in the evaluation of ADIs. The primary objective is to identify benign lipid-rich ACAs, which account for approximately 80% of ADIs in non-oncologic populations[3]. Non-contrast CT (ncCT) remains the fundamental and preferred first-line modality due to its widespread availability, reproducibility, and robust diagnostic performance[4,12,21]. The attenuation value measured in Hounsfield units (HU) on ncCT reflects tissue density and indirectly indicates intracellular lipid content, a hallmark of benign ACAs. A homogeneous adrenal mass with attenuation ≤ 10 HU is highly specific for a lipid-rich adenoma[3,12,22] (Figure 1). When this criterion is met, current ESE guidelines recommend that no further imaging follow-up is required, irrespective of lesion size, although comprehensive hormonal evaluation remains mandatory[12,22]. Thus, ncCT serves not only as a diagnostic tool but also as a triage instrument, identifying a substantial subgroup of patients in whom additional imaging surveillance can be safely avoided while maintaining vigilance through biochemical screening.
Figure 1 Non-contrast computed tomography is the cornerstone of imaging assessment of adrenal incidentalomas.
A: Non-contrast computed tomography (ncCT) demonstrates an adrenal incidentaloma (ADI) with a density < 10 Hounsfield units (HU); B: Axial ncCT shows bilateral ADIs < 10 HU; C: Axial ncCT shows a lipid-poor left ADI with attenuation > 10 HU. HU: Hounsfield units.
Caveat: Is the 10 HU threshold necessary?
For more than two decades, an attenuation threshold of ≤ 10 HU on ncCT has been considered the gold standard for identifying benign lipid-rich adenomas[3,12]. Systematic reviews encompassing 3723 patients without known malignancy demonstrated that lesions ≤ 10 HU were exclusively benign, supporting a strong negative predictive value for malignancy[12].
However, contemporary analyses suggest that in true ADIs measuring < 4 cm, expanding the benignity threshold to ≤ 20 HU for homogeneous lesions may maintain a positive predictive value between 99.4% and 99.8%[10,23]. In the EURINE-ACT study involving 2017 patients, no ACCs were observed in lesions < 4 cm with attenuation < 20 HU[10,23]. Adoption of this expanded threshold could reduce follow-up imaging by approximately 30%, potentially lowering radiation exposure, healthcare costs, and patient anxiety[10,24].
Nevertheless, the 2023 ESE guidelines retain a conservative approach. Lesions ≤ 10 HU require no further imaging, whereas those measuring 11-20 HU warrant individualized management, including additional imaging [e.g., 18-F-flurodeoxyglucose positron emission tomography/CT (18F-FDG PET/CT)] or repeat imaging at 6-12 months[12]. This cautionary approach reflects rare but documented cases of metastases or ACC presenting with lower attenuation values.
Importantly, the ≤ 20 HU threshold should not be applied in oncology patients, as metastases frequently demonstrate attenuation between 10 HU and 20 HU[10,25]. In such contexts, lesions > 10 HU remain indeterminate and require surveillance aligned with primary malignancy follow-up protocols[12].
Is contrast washout CT necessary?
Adrenal washout CT (AWCT), historically used for indeterminate adrenal lesions with attenuation > 10 HU on ncCT, was long considered a central tool for differentiating ACAs from malignant lesions based on contrast washout kinetics. Rapid contrast washout (absolute washout > 60%) was interpreted as supportive of a benign ACA. However, accumulating evidence has led to critical reassessment of its diagnostic reliability and clinical utility.
A major limitation of AWCT is its poor specificity for excluding PHEO. A systematic review and meta-analysis demonstrated that up to 47% of PHEOs may satisfy “strict” ACA washout criteria (absolute washout > 60%), creating the potential for clinically dangerous misclassification if biochemical metanephrine testing is not performed[10,26]. Given the substantial perioperative risks associated with undiagnosed PHEO, biochemical evaluation remains mandatory irrespective of imaging washout characteristics[4,11].
Furthermore, AWCT has primarily been validated in study populations enriched with malignant lesions and lacks robust validation in true ADIs’ cohorts, where the baseline prevalence of malignancy is very low[10]. In true ADIs < 4 cm, the negative predictive value of washout for malignancy is limited in practical terms, as 98.6% of lesions that “fail” washout criteria (absolute washout < 60%) are nonetheless benign[10]. Thus, failure to meet washout criteria does not reliably indicate malignancy in low-risk populations.
From a health economic perspective, modeling studies involving one million simulated adult patients have demonstrated that strategies incorporating AWCT are not cost-effective compared with ncCT-based follow-up. Only the strategy based on initial ncCT fell below the $100000 per life-year threshold, whereas AWCT strategies were associated with higher costs and fewer life-years[24]. Collectively, these data support a more restricted role for AWCT, emphasizing comprehensive ncCT assessment combined with mandatory biochemical screening rather than routine washout imaging in true ADIs[10,24].
MRI in assessing ADIs
CT remains the preferred first-line imaging modality for ADIs due to its availability, cost-effectiveness, and well-validated attenuation thresholds[12]. Nevertheless, MRI has a complementary role, particularly when CT findings are indeterminate or when radiation exposure or iodinated contrast should be avoided.
Chemical-shift MRI enables detection of microscopic intracellular lipid through signal loss on out-of-phase imaging. A signal intensity index ≥ 16.5% is widely used as a quantitative threshold for identifying adenomas[10,27]. This technique is particularly useful for lipid-poor adenomas with attenuation > 10 HU on ncCT.
MRI may also aid in suspected PHEO, which often - but not invariably - demonstrates marked T2 hyperintensity (“light bulb sign”). However, this sign is absent in approximately 35% of PHEOs, and up to 50% of PHEOs exhibit atypical MRI appearances[10,28]. Therefore, MRI findings should be interpreted as supportive rather than definitive and must always be correlated with biochemical metanephrine testing. In addition, MRI is useful for evaluating hemorrhage, necrosis, and complex lesion morphology. Overall, CT remains the principal diagnostic modality, while MRI refines characterization in selected scenarios and increases diagnostic confidence in indeterminate cases.
What does 18F-FDG PET/CT add to the evaluation of ADIs?
18F-FDG PET/CT evaluates metabolic activity within adrenal lesions and may assist in distinguishing benign from malignant masses, particularly in patients with a history of extra-adrenal malignancy[5,12,29]. Malignant lesions such as ACC and metastases typically demonstrate increased glucose uptake, often reflected in elevated maximum standard uptake value (SUVmax) values or tumor-to-liver standard uptake value ratios[30].
Sensitivity and specificity exceeding 90% have been reported in selected cohorts; however, diagnostic performance depends on appropriate clinical context and correlation with anatomical imaging[31-33]. False positives may occur in inflammatory or infectious conditions and occasionally in benign adenomas, whereas false negatives can be observed in small (< 10 mm) or necrotic tumors.
Thus, 18F-FDG PET/CT should be regarded as a problem-solving modality in indeterminate lesions or oncologic settings rather than a routine first-line investigation. Integration with CT or MRI findings remains essential, and further validation is required before broader application or integration with artificial intelligence (AI)-based analysis.
Alternative functional imaging modalities
Other functional imaging methods, which depend on radiotracers targeting different steroidogenesis enzymes or catecholamine synthesis/metabolism, have demonstrated considerable potential for distinguishing functional subtypes, though they are not mainstream standard of care yet[34], except for some functional imaging modalities for PHEO.
AI
AI and radiomics have been explored as adjunctive tools for non-invasive characterization of adrenal lesions. Several retrospective studies have reported high diagnostic performance, with some models achieving area-under-the-curve values exceeding 0.99 for differentiating benign from malignant masses[35]. Machine learning approaches incorporating tumor diameter, attenuation values, and clinical variables have also been proposed for outcome prediction[35].
In oncological populations - where approximately 50% of adrenal masses represent metastatic disease - machine learning models applied to 18F-FDG PET/CT data have shown promising discrimination between benign and malignant lesions. Ensemble algorithms such as Bagging, Random Forest, and XGBoost have demonstrated high reported accuracy in retrospective analyses, with feature-importance methods (e.g., Shapely Additive exPlanations analysis) identifying adrenal SUVmax, tumor-to-liver SUVmax ratio, and unenhanced CT attenuation as influential predictors[36]. Artificial neural networks have also been evaluated for subtyping malignancies, including differentiation between lung cancer metastases and lymphoma[36].
However, most available studies are small, single-center, and retrospective, and are vulnerable to segmentation bias, data leakage, spectrum bias, and overfitting[35]. External validation across diverse tumor types, imaging protocols, and institutions remains limited. At present, AI-based models should be regarded as investigational tools requiring prospective validation before integration into routine clinical decision-making.
BIOCHEMICAL ASSESSMENT
Comprehensive biochemical evaluation is mandatory for most patients presenting with a true ADI, except in cases of critical illness or limited life expectancy[6,12,29]. Hormonal assessment should investigate cortisol excess (overt Cushing’s syndrome or MACS), PHEO (plasma-free or urinary fractionated metanephrines, including normetanephrine and 3-methoxytyramine), and PA [aldosterone-to-renin ratio (ARR)][4]. When ACC is suspected, evaluation of sex steroids and steroid precursors - preferably via multi-steroid profiling - should be performed[12]. Interpretation of endocrine testing requires awareness of confounders, including diurnal rhythm, assay variability, obesity, psychiatric conditions, and drug interactions - particularly for the dexamethasone suppression test (DST)[11,12].
PHEO
Risk-stratified screening for PHEO is endorsed in the 2023 ESE guidelines. Plasma-free or urinary fractionated metanephrine testing may be omitted in patients with homogeneous, lipid-rich adenomas with attenuation ≤ 10 HU on ncCT[12]. However, metanephrine testing remains mandatory for lesions with attenuation > 10 HU or atypical imaging features[12]. Metanephrine testing demonstrates a sensitivity of 97%-99%, providing reliable biochemical exclusion[10]. Screening must be performed prior to invasive procedures or surgery to avoid hypertensive crisis[11,37,38].
MACS is common
MACS represents adrenocorticotropic hormone-independent cortisol hypersecretion of mild degree. It is defined as post-1 mg overnight DST serum cortisol > 1.8 μg/dL (> 50 nmol/L) in the absence of classic clinical stigmata of overt Cushing’s syndrome[3,11,12]. A value ≤ 1.8 μg/dL effectively excludes autonomous cortisol secretion.
MACS is the most common functional abnormality in ADIs, affecting 20%-50% of patients with ADIs and approximately 0.2%-2% of the general population[4,12,15,39]. The 2023 ESE guidelines replaced the term subclinical Cushing’s syndrome with MACS to reflect the recognition that even mild cortisol excess is clinically meaningful[12,40].
The guidelines recommend interpreting DST results as a continuous variable, as higher post-DST cortisol concentrations correlate with increased cardiometabolic comorbidity[12]. In selected populations (e.g., subjects with obesity or psychiatric disorders), higher cutoffs (3-5 μg/dL) are sometimes used to identify individuals most likely to benefit from intervention[11]. MACS is associated with hypertension, dyslipidemia, type 2 diabetes mellitus, increased mortality, and renal dysfunction independent of traditional cardiovascular risk factors[14,41-43].
PA - are approaches such as adrenal vein sampling for the evaluation of aldosterone-secreting adenomas still valid?
In patients with ADIs and concomitant hypertension or unexplained hypokalemia, screening for PA using the ARR is mandatory[11,19]. A positive ARR should be followed by confirmatory testing. If PA is confirmed, differentiation between unilateral aldosterone-producing adenoma and bilateral adrenal hyperplasia is required, as adrenalectomy is potentially curative only in unilateral disease[18,22]. Adrenal venous sampling remains the gold standard for lateralization prior to surgery, as imaging alone cannot reliably distinguish aldosterone-producing adenoma from bilateral adrenal hyperplasia[18,22,29,44].
Steroid profiling
When ACC is suspected based on size (> 4 cm) or indeterminate imaging features, evaluation of sex steroids and steroid precursors - preferably via mass spectrometry-based multi-steroid profiling - improves diagnostic discrimination compared with nonspecific tumor markers[12,23].
Gas chromatography/mass spectrometry analysis of urinary steroid metabolomics has documented high accuracy in distinguishing ACC from benign ACAs, especially when combined with imaging features of the tumor[3,23]. The EURINE-ACT study was a large, prospective, multicenter diagnostic validation study evaluating the role of urine steroid metabolomics in the assessment of newly identified adrenal masses[23]. In this study, in 2017 adult participants recruited non-selectively across 14 countries, urine steroid metabolomics demonstrated high diagnostic accuracy for ACC, outperforming conventional imaging parameters such as tumor size and imaging characteristics alone. The combination of tumor diameter, imaging characteristics, and urine steroid metabolomics yielded the highest diagnostic performance, substantially increasing positive predictive value for ACC while maintaining excellent ability to exclude malignancy. Implementation of this combined strategy could reduce unnecessary imaging and adrenalectomies for benign lesions, facilitate earlier surgical management of ACC, and decrease patient morbidity and healthcare costs, supporting the integration of urine steroid metabolomics into diagnostic pathways for ADIs[23].
UNEXPLORED ISSUES AND SYSTEMIC MORBIDITY
Besides acute risks of malignancy or hormonal crisis, ADIs are increasingly recognized for subtle but pervasive systemic effects that represent important areas for future clinical research.
Cardiovascular and metabolic morbidity
MACS is increasingly recognized as a driver of systemic morbidity. It is strongly associated with components of the metabolic syndrome, including hypertension, type 2 diabetes, and dyslipidemia[13,14]. Cardiovascular event incidence reaches approximately 15.5% over five years in MACS, compared with 6.4% in non-functional tumors[45,46]. Events include myocardial infarction, stroke, and heart failure[47]. A retrospective study of 3610 patients reported a 4.37-fold increased risk of all-cause mortality among women under 65 with MACS[48]. Higher post-DST cortisol values predict incident cardiovascular events independent of traditional risk factors[44,47].
Musculoskeletal/osseous risks
Even mild cortisol excess adversely affects bone health. MACS is associated with reduced bone mineral density, increased prevalence of osteoporosis and osteopenia, and elevated rates of vertebral and non-vertebral fractures[46,49-52].
Meta-analyses report vertebral fracture prevalence 15%-40% higher than controls and pooled fracture prevalence around 43%, with osteoporosis/osteopenia prevalence approximately 50%[40,50]. Vertebral fracture rates between 34% and 63% have been reported in MACS cohorts[44]. Nevertheless, although morphometric vertebral fractures are more common in subjects with MACS, the risk of clinically overt fragility fractures in them may not be significantly higher than in non-functioning tumors[50,52]. Bearing this in mind, guidelines recommend vertebral fracture screening using lateral spine imaging (X-ray or CT re-evaluation), as many fractures are asymptomatic[12,49].
Psychiatric and mental health implications
Psychiatric morbidity and reduced quality of life (QoL) are increasingly recognized in patients with ADIs. Even mild hypercortisolism within the spectrum of MACS has been biologically linked to adverse neuropsychiatric outcomes[53,54]. Patients with MACS or overt hypercortisolism exhibit higher rates of depressive symptoms, anxiety, cognitive impairment, and reduced health-related QoL compared with matched controls[54,55].
Prospective single-center studies suggest that surgical remission of MACS may improve cognitive performance and QoL[55-57]. However, the 2023 ESE guidelines emphasize the limited understanding of long-term psychiatric impact in both functioning and non-functioning lesions and call for prospective longitudinal research[12].
Bilateral ADIs
Bilateral ADIs represent approximately 20% of incidentally discovered adrenal masses, presenting a complex clinical challenge[58]. The etiology is highly heterogeneous, encompassing benign ACAs, primary bilateral macronodular adrenal disease, PHEOs, and metastatic disease[58]. Critically, bilateral ADIs carry a higher risk of MACS compared to unilateral lesions, which is linked to increased cardiovascular morbidity and mortality[58,59]. Radiological evaluation via ncCT remains the primary modality; a density of ≤ 10 HU typically indicates a benign, lipid-rich phenotype[59]. Current guidelines advocate for a targeted 4-option schema based on imaging morphology to guide biochemical testing, including the 1-mg DST and screening for PA[58]. Genetic screening, particularly for ARMC5 mutations in primary bilateral macronodular adrenal disease, is increasingly recommended to manage familial risk[58]. Management requires multidisciplinary coordination to balance hormonal excess control against the risk of post-surgical adrenal insufficiency[58,59].
Do phosphodiesterase type 5 inhibitors contribute to the appearance of ADIs?
The hypothesis that chronic phosphodiesterase type 5 inhibitor (PDE5i) use may contribute to ADI development remains preliminary. Phosphodiesterase type 5 (PDE5) is expressed in adrenal tissue[60], and PDE5 inhibition influences cyclic guanosine monophosphate signaling pathways. Experimental data suggest potential modulation of endocrine function, including alterations in cortisol and androgen levels[61,62]. Expression of other phosphodiesterase isoforms (PDE2A, PDE11A, PDE8B, PDE8E) in adrenal tissue and hyperplasia has also been documented[63]. Tadalafil exhibits partial inhibitory activity against PDE11A and has been associated with increased cortisol secretion in some studies[61]. Clinical evidence linking PDE5i use to adrenal structural abnormalities derives primarily from a small retrospective study (5/32 men) presented at a European Association of Urology Congress[64]. The study lacked detailed imaging characterization, hormonal evaluation, control groups, and exposure quantification. As such, causality cannot be inferred[65]. At present, this proposed association should be considered hypothesis-generating. Large, controlled cohort studies with dose-response analyses and adjustment for imaging frequency and metabolic confounders are required before any clinical conclusions can be drawn[65].
Surgery for MACS
Surgical management of unilateral MACS remains an area of active investigation. The CHIRACIC trial demonstrated that adrenalectomy was superior to conservative management for blood pressure control: 52% of surgically treated patients discontinued antihypertensive therapy while maintaining normotension, compared with 15.4% in the conservative group, with effects evident within 2.5 months[46,66]. The ITACA study evaluated long-term cardiac remodeling. Adrenalectomy reduced left ventricular mass index (-14.8 g/m2 at 12 months), though this benefit was not sustained at five years[67]. Conservatively managed non-functional tumors exhibited progressive hypertrophy (+22.4 g/m2 over five years), underscoring the importance of longitudinal cardiovascular monitoring. Despite these findings, large randomized controlled trials evaluating long-term hard outcomes remain limited[12].
CONCLUSION
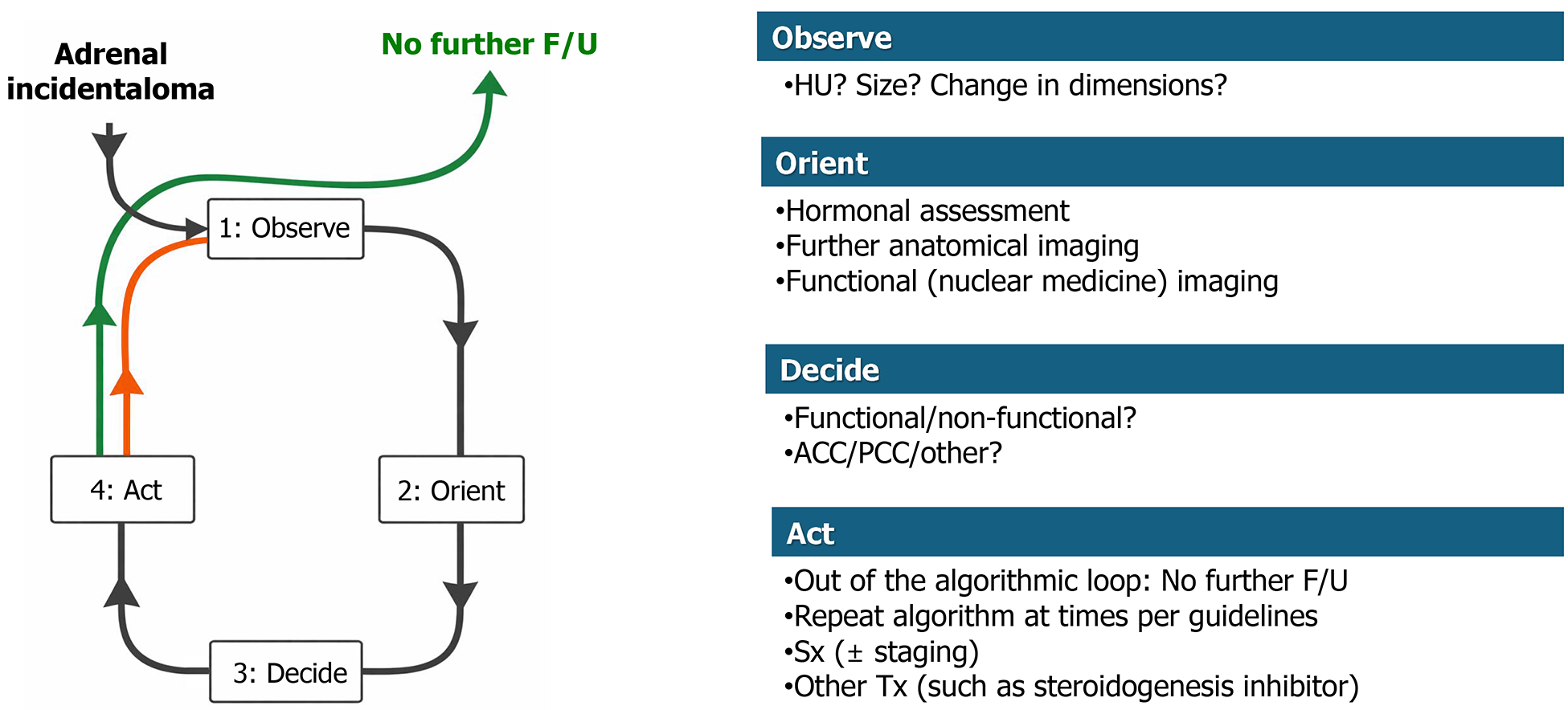
Management of ADIs has evolved toward a risk-stratified framework integrating imaging refinement and comprehensive endocrine evaluation. Adoption of a ≤ 20 HU threshold for homogeneous true ADIs < 4 cm, alongside reduced reliance on washout CT, may decrease unnecessary imaging and associated costs in appropriately selected populations[10,24]. Biochemical screening remains essential, particularly for MACS, which is now recognized as a major contributor to cardiovascular, metabolic, and skeletal morbidity (Figure 2 and Supplementary Figure 1).
Figure 2 Simplified OODA loop (observe, orient, decide, act) management algorithm for adrenal incidentalomas.
For the more detailed form of this algorithm see the Supplementary Figure 1. HU: Hounsfield units; ACC: Adrenocortical carcinoma; PCC: Pheochromocytoma; F/U: Follow-up; Sx: Surgery; Tx: Therapy.
Data from CHIRACIC and ITACA illustrate potential metabolic and cardiac effects of surgical intervention, while also highlighting the need for long-term monitoring[66,67]. Ongoing adaptation of clinical practice based on prospective, high-quality evidence will be necessary to optimize outcomes in this expanding patient population. Further management requires a structured follow-up algorithm balancing avoidance of over-testing with detection of rare malignant transformation. For indeterminate lesions managed non-surgically, repeat unenhanced CT or MRI at 6-12 months is recommended[12]. Surgery is indicated for growth > 20% in maximum diameter with an absolute increase ≥ 5 mm[12]. NcCT-based follow-up is cost-effective in patients without known malignancy, whereas strategies incorporating washout CT or routine MRI are not[24]. In non-operated MACS, annual reassessment of cardiometabolic comorbidities is advised[12,46].
Further studies will be necessary to confirm the potential roles of AI, radiomics, and multi-steroid profiling for the improvement of non-invasive diagnosis, especially differential diagnosis of ACC. Longitudinally prospective studies are needed in order to better define the impact of MACS on QoL and psychiatric morbidity in the long run, and also to formally explore some controversial etiological relationships, including that between PDE5i use and the development of ADIs. Because of the rapidly changing landscape, ongoing adaptation in clinical practice based on high-quality evidence will be required to ensure optimal outcomes in patients.