Published online Apr 28, 2026. doi: 10.4329/wjr.v18.i4.119347
Revised: February 20, 2026
Accepted: April 2, 2026
Published online: April 28, 2026
Processing time: 89 Days and 13 Hours
Breast magnetic resonance imaging (MRI) provides valuable information for tumor detection, as well as potential applications in molecular characterization and prognostication. A key feature detectable on MRI is the presence of breast edema, which has been associated with tumor aggressiveness and poorer clinical outcomes.
To determine the correlation between intramammary edema patterns as observed on breast MRI and the histopathological and molecular characteristics of the tumor.
In this retrospective single-center study, 123 women with biopsy-proven breast cancer underwent preoperative breast MRI from June 2022 to June 2025. The classification of edema was determined on T2-weighted images, divided into four breast edema score (BES) categories: BES-1 (no edema), BES-2 (peritumoral edema), BES-3 (prepectoral edema), and BES-4 (subcutaneous edema). The MRI findings were correlated with histological type, molecular subtype, receptor status, Ki67 index, and lymph node involvement. Data analysis was conducted using IBM SPSS Statistics for Windows, version 28.
Edema was observed in 45.5% of patients. Statistically significant correlation was observed between BES and molecular subtypes (P < 0.001), hormone receptor status (P < 0.001), and human epidermal growth factor receptor 2 expression (P < 0.001). Higher BES categories (BES 2-4) exhibited a higher prevalence in human epidermal growth factor receptor 2-positive and triple-negative tumors, while the absence of edema (BES-1) demonstrated a predominance in hormone receptor-positive subtypes.
The presence and severity of MRI-based breast edema score have been found to correlate with aggressive molecular subtypes, underscoring the potential role of BES in prognostic stratification and guiding tailored treat
Core Tip: Magnetic resonance imaging-based assessment of intramammary edema may serve as a practical imaging biomarker reflecting breast cancer biology. Using a simple four-level breast edema score on T2-weighted images, edema patterns can be linked to key histopathological and molecular features, including tumor subtype, receptor status, proliferation index, and nodal involvement. Higher breast edema score categories tend to accompany more aggressive molecular profiles, whereas the absence of edema is more frequently observed in hormone receptor-positive disease. Therefore, incorporating breast edema scoring into routine preoperative magnetic resonance imaging interpretation could support risk stratification and help guide individualized treatment planning in clinical practice.
- Citation: Vasilska A, Tsvetkova S, Doykova K, Chervenkov L, Velikova T, Vasilska M, Sariyan S. Magnetic resonance imaging as a non-invasive breast cancer tool - the relevance of breast edema score: A retrospective study. World J Radiol 2026; 18(4): 119347
- URL: https://www.wjgnet.com/1949-8470/full/v18/i4/119347.htm
- DOI: https://dx.doi.org/10.4329/wjr.v18.i4.119347
Breast cancer remains the most frequently diagnosed malignancy among women globally. In 2022, an estimated 2.3 million new cases were recorded worldwide, accounting for 23.8% of all cancer cases in women[1]. Incidence rates continue to rise, especially in industrialized countries. The disease poses a significant threat to both public health and socioeconomic systems. Early detection is crucial for improving survival and patient outcomes. Characterization of prognostic and predictive markers enables clinicians to create effective therapeutic strategies.
The most sensitive imaging modality for breast cancer detection is magnetic resonance imaging (MRI), outperforming mammography and ultrasound[2-4]. Although histological verification via biopsy remains indispensable, MRI contributes beneficial, additional information- enhanced visualization of tumor characteristics and identification of associated factors relevant to prediction and prognosis[2].
Breast edema, which is detected by MRI, is a clinically important radiological finding. Its presence and degree are often associated with more aggressive tumor subtypes. It is related to poorer prognosis and can indicate a higher risk profile[5-7]. Non-invasive evaluation of tumor biology and potential aggressiveness is enabled via such an advanced imaging modality. Verifying breast edema at an early stage may guide clinicians to opt for more intensive therapeutic in
This study aims to investigate the association between intramammary edema appearance on breast MRI and the histopathological and molecular profile of the tumor. The distribution of different breast edema score (BES) categories was specifically evaluated, and their associations with histological and molecular subtypes, receptor status, tumor grade, proliferative index, and lymph node involvement were analyzed.
This is a retrospective, single-center, present study, which includes 123 women who were diagnosed with breast cancer through either an excisional or Tru-Cut biopsy from June 2022 to June 2025. All women underwent preoperative MRI at a minimum of 6 weeks following the biopsy procedure. In accordance with the current guidelines, all MRI examinations of premenopausal women were conducted within the timeframe spanning from the seventh to the fourteenth day of the menstrual cycle. The inclusion criteria comprised patients over the age of 18 years who had undergone a breast MRI and had a proven diagnosis of breast cancer, as confirmed by histology and immunohistochemistry. Exclusion criteria include cases within complete clinical, imaging, and pathological data, patients who also underwent neoadjuvant chemo therapy before the MRI, and examinations within adequate image quality. In addition, cases with clinical or imaging findings suggestive of alternative causes of edema, such as inflammatory or infectious processes or systemic conditions, were excluded. This approach was adopted to reduce potential confounding and to ensure that the analyzed edema reflected tumor-associated changes.
The MRI examinations were conducted using a 1.5 T MRI scanner (Magnetom Amira, Siemens Healthcare, Erlangen, Germany), an 18-channel dedicated breast coil, and the women were positioned prone, with the head first. The examination encompasses the following sequences: A non-fat-suppressed T1-weighted transversal sequence; a fat-suppressed turbo inversion recovery magnitude (TIRM) transversal sequence, diffusion-weighted images with apparent diffusion coefficient maps, and axial dynamic fat-suppressed contrast-enhanced 3D T1 with subtraction images.
In order to evaluate the breast edema, the following sequences with their respective parameters were analyzed: Fat-suppressed TIRM transversal sequence - field of view 340/100 mm, time of repetition 3000 milliseconds, time of echo 55 milliseconds, matrix size 336/448, slice thickness 3 mm, and scanning time 156 seconds. Axial dynamic fat-suppressed contrast-enhanced 3D T1 - weighted fast low-angle l sequence-field of view 240/150 mm, time of repetition 4.35 milliseconds, time of echo 1.76 milliseconds, matrix size 384/256, slice thickness 1.1 mm, and scanning time 398 seconds.
Five contrast series were obtained - one pre-contrast and four post-contrasts. The contrast media utilized in this procedure were Gadovist 1 mmol/mL (a gadolinium-based contrast agent), with an administration rate of 3 mL/second, injected through a peripheral venous access, followed by 15 mL of saline.
MRI assessments were conducted by two radiologists. One radiologist had five years of experience in breast imaging, and the other had 15 years of experience. Both radiologists worked independently from one another and were unaware of the histopathological results. In cases of discrepant interpretations, a consensus review was performed to reach a final agreed score.
Regions with edema were assessed by comparison of TIRM and were compared with post-contrast T1-weighted images. On the TIRM sequence, edematous changes appeared as hyperintense areas, indicative of increased interstitial fluid. Those zones lacked enhancement after contrast administration.
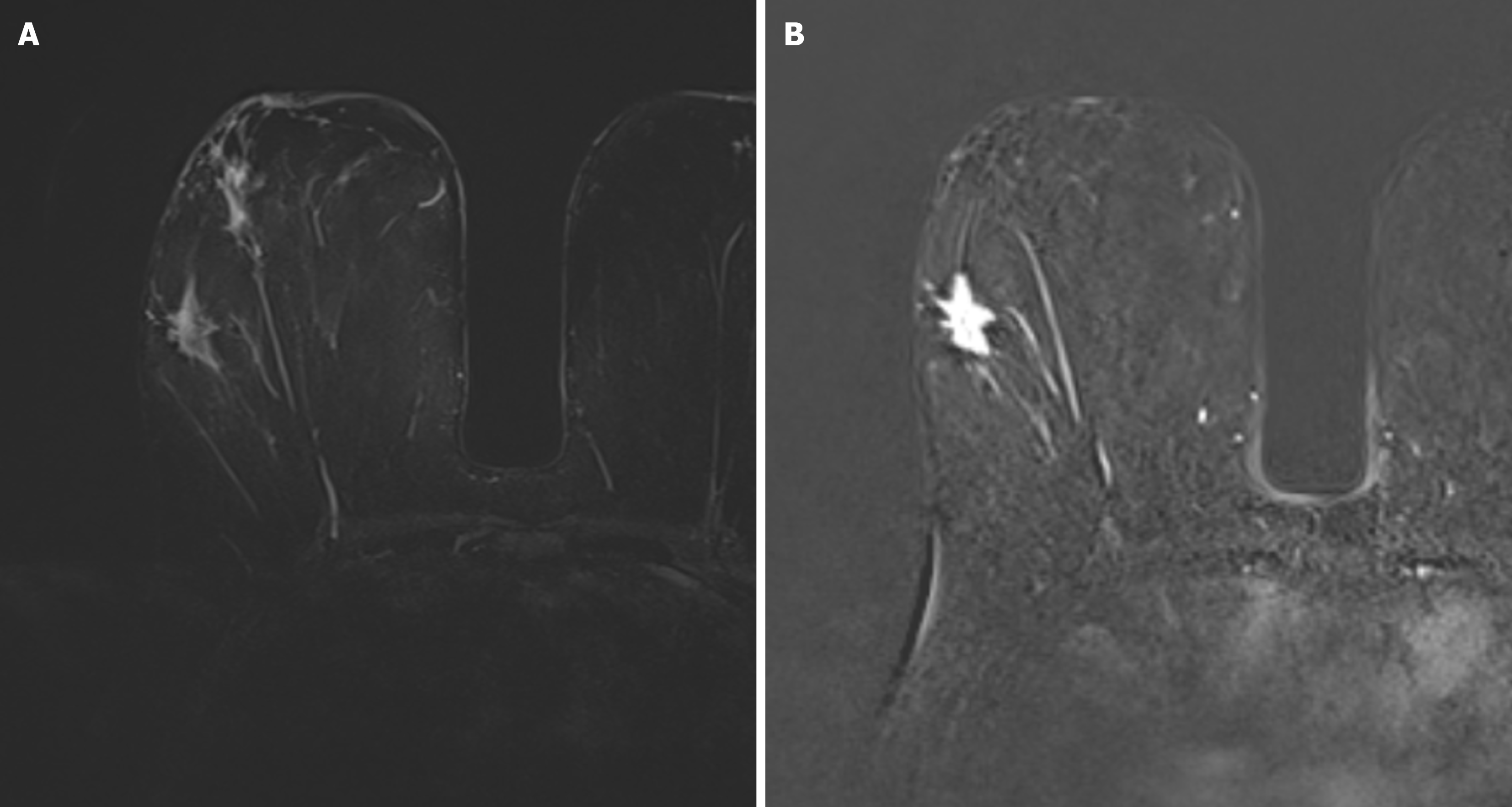
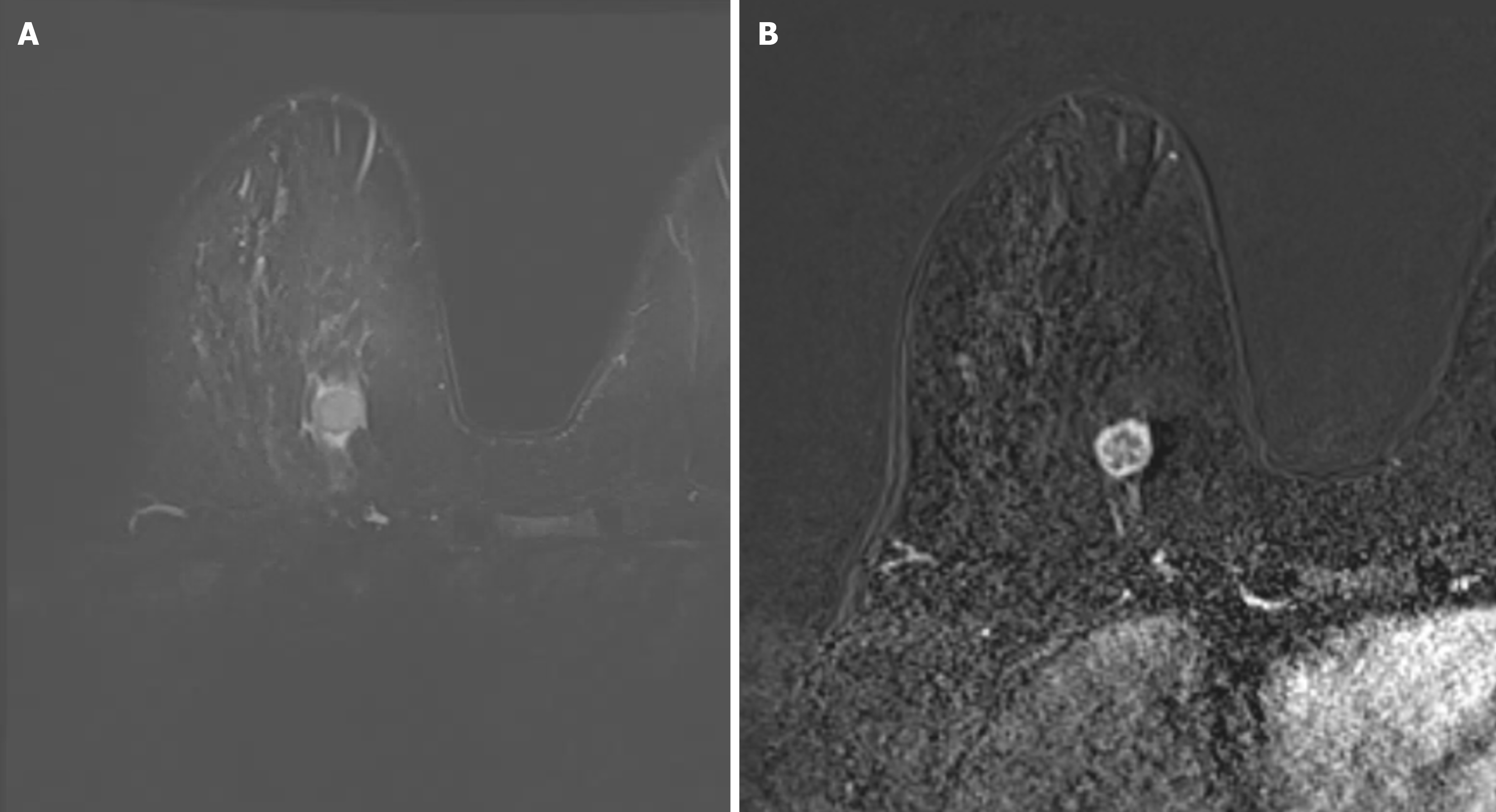
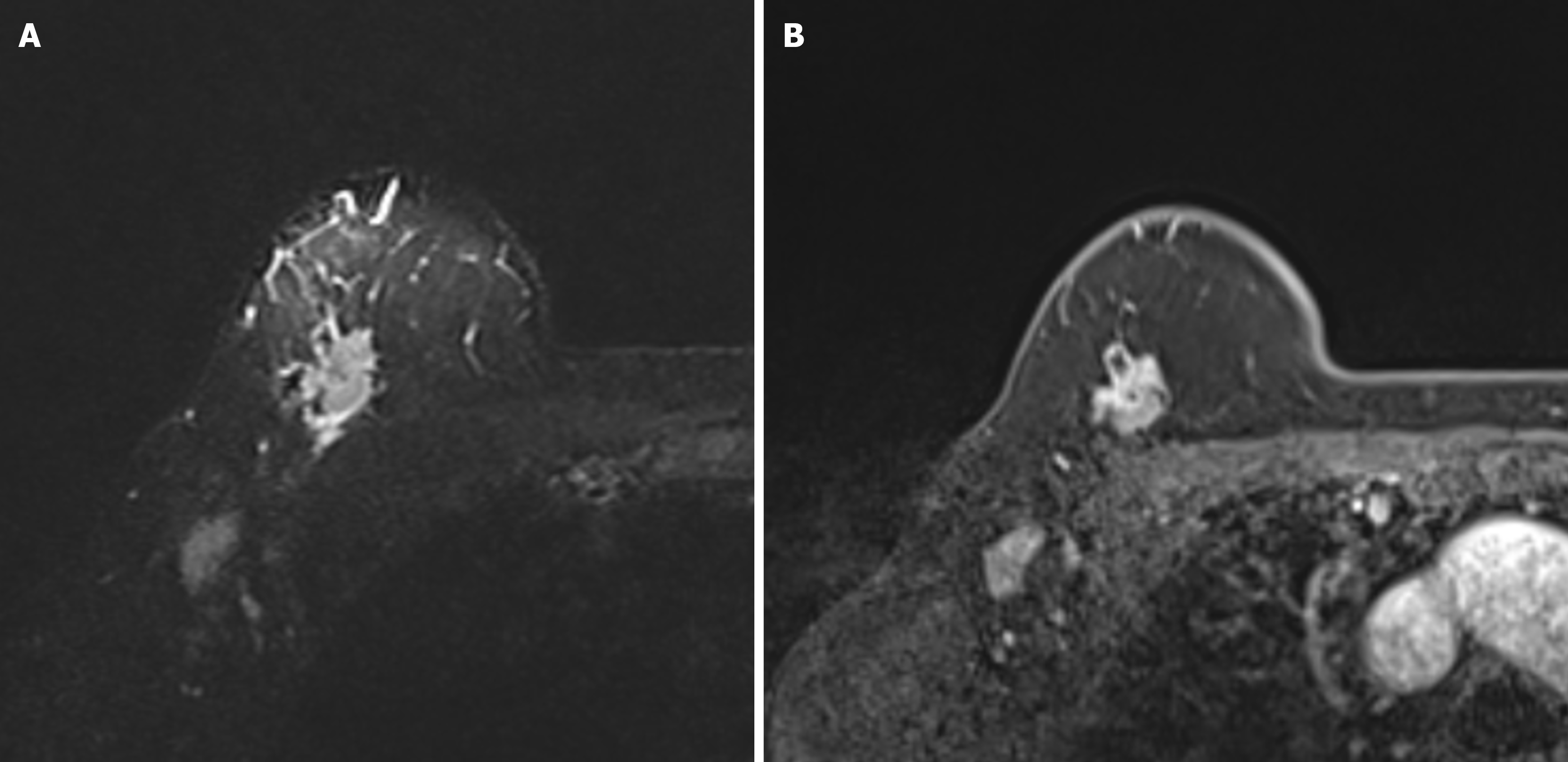
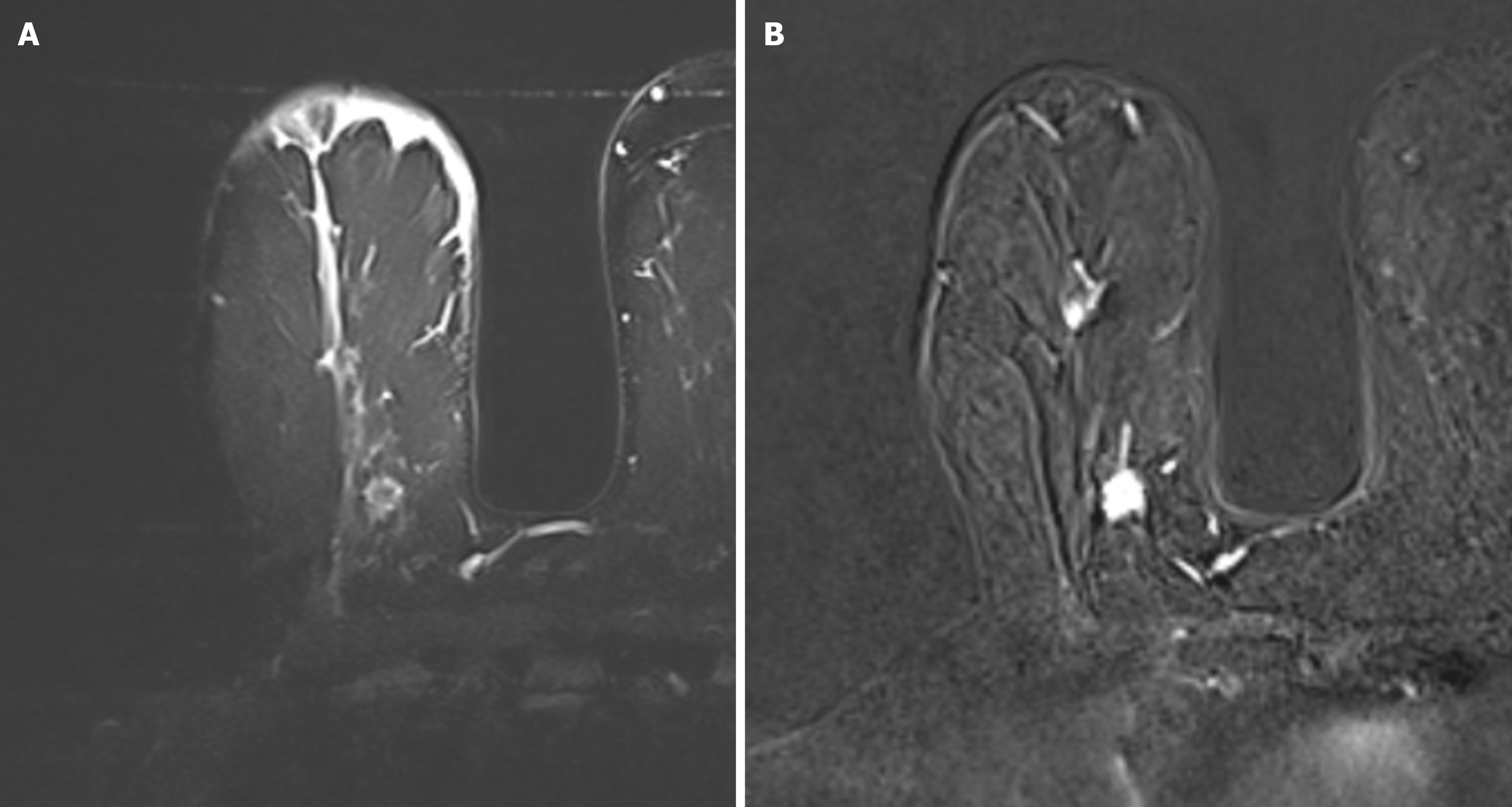
Based on the existence and type of edema the patients were subdivided into four categories: (1) BES-1-without edema (Figure 1); and (2) BES-2 with peritumoral edema (Figure 2), BES-3 with prepectoral edema (Figure 3) and BES-4 with subcutaneous edema (Figure 4). If two types of edemas were found in one patient the one with the higher score was taken into account.
Furthermore, an evaluation of the lymph nodes was conducted, encompassing the assessment of their morphology, namely the presence of a round shape, the thickness of the cortical layer, and the absence of the conventional fat-equivalent center within the lymph node. The presence of any of these abnormalities was deemed indicative of metastatic disease.
The following data were collected for the study: Histologic type, histologic grade, tumor grading, and lymph node status. Immunohistochemistry was also employed, including estrogen receptor, progesterone receptor, human epidermal growth factor receptor 2 (HER2), and Ki-67 index, which were used to classify tumors according to the 2013 St. Gallen International Expert Consensus Guidelines for Immunohistochemistry in breast cancer. The cut-off value of the Ki67 index was determined to be 20%. All histopathological and immunohistochemical analyses were performed as part of the routine clinical diagnosis in a certified pathology laboratory, following institutional protocols and internationally accepted guidelines.
Data analysis was conducted using IBM SPSS Statistics for Windows, version 28.0. Age distribution, being the only continuous variable, showed a lack of normality, and the central tendency was described with the median and interquartile range (interquartile range) and compared among BES lesion categories using the Kruskal-Wallis test. The remaining data consisted of categorical or ordinal variables, which were presented as n (%). Z-tests were employed to compare category proportions. To establish associations between variables, Fisher’s exact test was utilized for di
The study included 123 female patients diagnosed with breast cancer, with a median age of 47 years (interquartile range = 20), an age range of 26-80 years, and the age group 41-60 constituted a significant majority (P < 0.001). Luminal B (HER2 negative) and luminal A were the two most prevalent molecular subtypes of breast cancer, whereas invasive ductal carcinoma was the dominant histological type (89.40%) against all other types (P < 0.001). According to tumor grading, grade II was significantly more prevalent than grade III and grade I (P < 0.001). The majority of the patients were estrogen (estrogen receptor) and progesterone (progesterone receptor) positive (P < 0.001 for both). HER2 receptors 1+ were detected in the majority of cases, followed by 0+ and 3+. Patients with a high proliferative index (≥ 20%) dominated significantly over those with a low proliferative index (< 20%), P < 0.001. Edema was observed in 45.50%, among which peritumoral edema (BES-2) was the most frequently present, vs prepectoral edema (BES-3) and subcutaneous edema (BES-4) (P < 0.001). Lymphadenopathy was detected in 56.30% of the cases (P = 0.030) (Table 1).
| Variables | Statistics, n (%) | P value |
| Age | 4 | |
| Median (interquartile range) | 7 (20) | |
| Minimum-maximum | 26-80 | |
| Age categories | ||
| ≥ 40 years | 29 (23.60)a | |
| 41-60 years | 61 (49.60)b | < 0.001 |
| 61-80 years | 33 (26.80)a | |
| Molecular subtype | ||
| HER2+ | 11 (8.90)a | Luminal B vs luminal A: P = 0.02; all others: P < 0.001 |
| Luminal A | 35 (28.50)b | |
| Luminal B (HER2+) | 52 (42.30)c | Luminal A vs HER2+, luminal B, and triple-negative breast cancer: P < 0.001 |
| Luminal B (HER2-) | 9 (7.30)a | |
| Triple negative (triple-negative breast cancer) | 16 (13.00)a | |
| Histological type | ||
| Invasive ductal carcinoma | 110 (89.40)a | |
| Invasive lobular carcinoma | 8 (6.50)b | |
| Medullary carcinoma | 2 (1.60)b | |
| Invasive papillary carcinoma | 1 (0.80)b | |
| Invasive tabular carcinoma | 1 (0.80)b | |
| Mucinous carcinoma | 1 (0.80)b | IDC vs each of the other types: P < 0.001 |
| Tumor grading (G) | ||
| Grade I | 5 (4.10)a | GI vs GII: P < 0.001 |
| Grade II | 84 (68.30)b | GI vs GIII: P < 0.001 |
| Grade III | 34 (27.60)c | GII vs GIII: P < 0.001 |
| Estrogen status (estrogen receptor) | ||
| Positive (> 1) | 96 (78.00) | |
| Negative (0) | 27 (22. 00) | < 0.001 |
| Progesteron status (progesterone receptor) | ||
| Positive (> 1) | 90 (73.20) | |
| Negative (0) | 33 (26.80) | < 0.001 |
| Human epidermal growth factor receptor 2-receptors | ||
| 0+ | 49 (39.80)a | |
| 1+ | 54 (43.90)a | < 0.001 |
| 3+ | 20 (16.30)b | |
| Proliferative Ki67-index | ||
| < 20 | 35 (28.50) | |
| ≥ 20 | 88 (71.50) | < 0.001 |
| Edema | ||
| Yes | 56 (45.50) | |
| No | 67 (54.50) | 0.160 |
| BES | ||
| BES-1 (no edema) | 67 (54.50)a | |
| BES-2 (peritumoral edema) | 37 (30.10)b | |
| BES-3 (prepectoral edema) | 7 (5.70)c | |
| BES-4 (subcutaneous edema) | 12 (9.80)c | < 0.001 |
| Lymphadenopathy | ||
| Yes | 70 (56.90) | |
| No | 53 (43.10) | 0.030 |
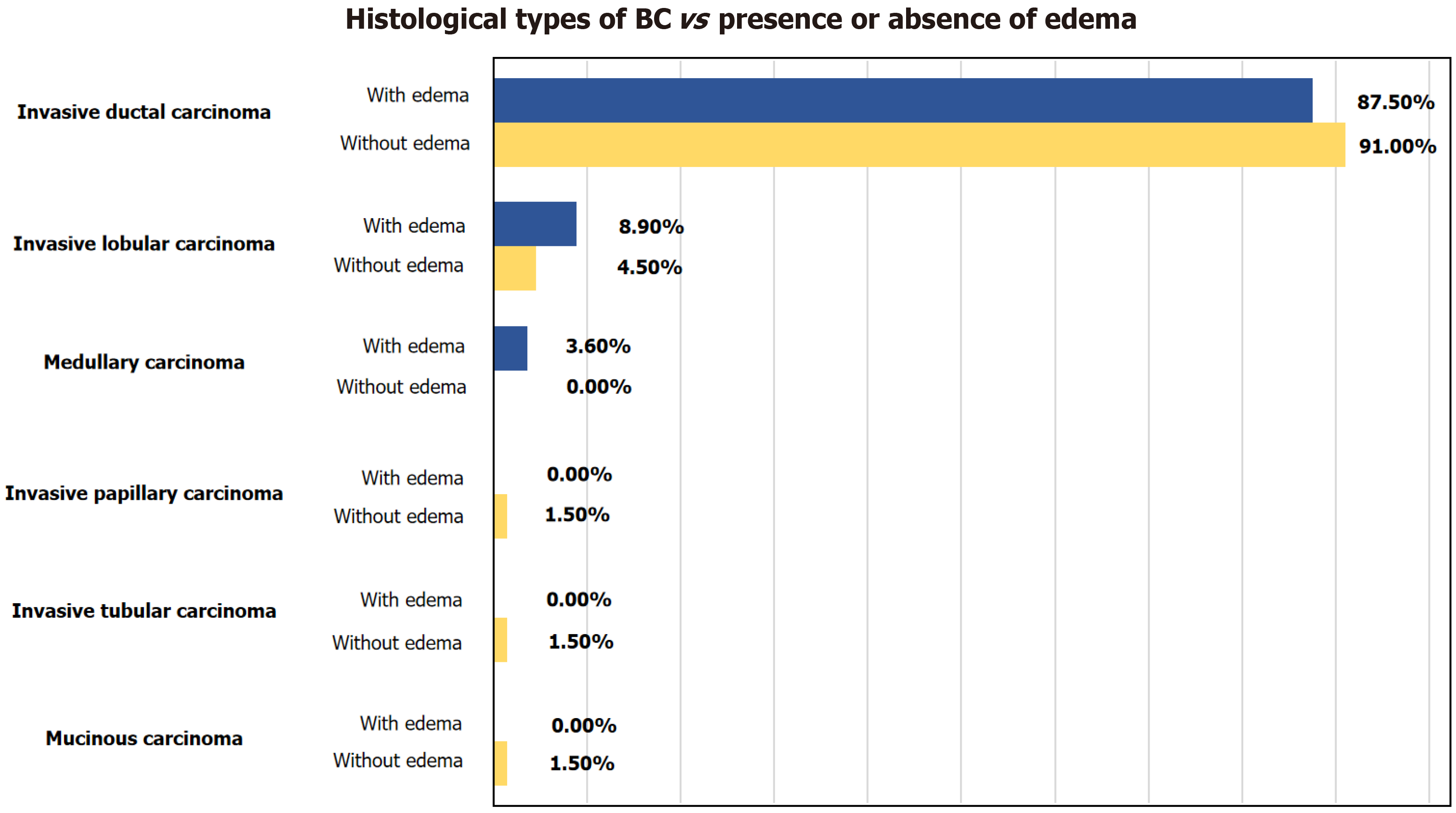
There was no significant association between the presence or absence of edema and the histological types of breast cancer (P = 0.275). Invasive ductal carcinoma was the most prevalent histological type in both cases with edema (87.50%, n = 49) and those without edema (91.00%, n = 61). Invasive lobular carcinoma was identified in 8.90% (n = 5) of the cases with edema and in 4.50% (n = 3) of those without edema. Medullary carcinoma was found in 3.60% (n = 2) of the edema cases and was not observed in cases without edema. Invasive papillary carcinoma, invasive tubular carcinoma, and mucinous carcinoma were absent in edema cases and present in 1.5% (n = 1) of those without edema (Figure 5).
No significant age difference was found among the four BES categories (P = 0.612). Molecular subtype showed a significant association with BES categories (P < 0.001). Luminal A and luminal B (HER2-) were significantly more prevalent in lesions with BES-1. BES-2 was more frequent in luminal B (HER2-) and triple-negative breast cancer (TNBC). Lesions with BES-3 were mostly characterized by luminal B (HER2+) and TNBC, whereas lesions with BES-4 were mostly distinguished by the absence of luminal A and the presence of all other subtypes.
Tumor grading (G) did not show a significant association with BES categories (P = 0.326). Grade II was most commonly observed in all BES, followed by grade III and grade I in frequency. Estrogen receptor and progesterone receptor expressions were significantly associated with BES (P < 0.001 for both estrogen receptor and progesterone receptor). Positive estrogen receptor and progesterone receptor expressions were prevalent in lesions without edema, BES-1.
HER2 receptors were also significantly related to BES categories (P < 0.001). The main differences were observed in relation to HER2 1+ receptors, which were significantly more frequent in lesions with BES-1, as well as in relation to 3+ receptors, which were more often identified in BES-3 and BES-4.
Ki67 levels greater than 20% were common across all BES categories, while lymph node involvement was more frequently observed in lesions with BES-3 and BES-4. However, no significant associations were identified between BES categories and the proliferative Ki67 index (P = 0.184) or lymphadenopathy (P = 0.130) (Table 2).
| Variables | BES-1 (n = 67) | BES-2 (n = 37) | BES-3 (n = 7) | BES-4 (n = 12) | P value |
| Age, median (interquartile range) | 47 (20) | 50 (21) | 44 (29) | 54.50 (22) | 0.6121 |
| Molecular subtype | |||||
| HER2+ | 2 (3.0)a | 6 (16.2)a | 1 (14.3)a | 2 (16.7)a | |
| Luminal A | 30 (44.8)a | 4 (10.8)b | 1 (14.3)b | 0 (0.0)b | |
| Luminal B (HER2-) | 33 (49.3)a | 14 (37.8)a | 1 (14.3)b | 4 (33.3)a | |
| Luminal B (HER2+) | 1 (1.5)a | 2 (5.4)a | 3 (42.9)b | 3 (25.0)b | |
| Triple negative | 1 (1.5)a | 11 (29.7)b | 3 (42.9)b | 3 (25.0)b | < 0.0012 |
| Tumor grading (G) | |||||
| Grade I | 2 (3.0) | 2 (5.4) | 1 (14.3) | 0 (0.0) | |
| Grade II | 51 (76.1) | 22 (59.5) | 4 (57.1) | 7 (58.3) | |
| Grade III | 14 (20.9) | 13 (35.1) | 2 (29.6) | 5 (41.7) | 0.3262 |
| Estrogen status (estrogen receptor) | |||||
| Positive (> 1%) | 64 (95.5)a | 20 (54.1)b | 5 (71.4)a,b | 7 (58.3)b | |
| Negative (0%) | 3 (4.5) | 17 (45.9) | 2 (28.6) | 5 (41.7) | < 0.0013 |
| Progesteron status (progesterone receptor) | |||||
| Positive (> 1%) | 61 (91.0)a | 18 (48.6)b | 4 (57.1)b | 7 (58.3)b | |
| Negative (0%) | 6 (9.0) | 19 (51.4) | 3 (42.9) | 5 (41.7) | < 0.0013 |
| Human epidermal growth factor receptor 2-receptors | |||||
| 0+ | 26 (38.8)a | 18 (48.6)a | 2 (28.6)a | 3 (25.0)a | |
| 1+ | 38 (56.7)a | 11 (29.7)b | 1 (14.3)b | 3 (33.3)b | |
| 3+ | 3 (4.5)a | 8 (21.6)b | 4 (57.1)b | 5 (41.7)b | < 0.0012 |
| Proliferative Ki67-index | |||||
| < 20% | 24 (35.8) | 6 (16.2) | 2 (28.6) | 3 (25.0) | |
| ≥ 20% | 43 (64.2) | 31 (83.8) | 5 (71.4) | 9 (75.0) | 0.1843 |
| Lymphadenopathy | |||||
| Yes | 33 (49.3) | 22 (59.5) | 5 (71.4) | 10 (83.3) | |
| No | 34 (50.7) | 15 (40.5) | 2 (28.6) | 2 (16.7) | 0.1303 |
In the context of modern medicine, diagnostic imaging has found its valuable place as a source of additional essential information. In the management of breast diseases, while the pathological result remains paramount for patient treatment, advancements in imaging technology, such as MRI, have enabled the extraction of data regarding tumors, their surroundings, and the entire breast, all in vivo. At present, several predictive and prognostic biomarkers from the breast MRI related to duration of response, progression-free survival, and overall survival are documented in the extant literature. These factors include the presence of hypervascularization, feeding vessels, hyperintensity on T2, washout, central necrosis, rim enhancement, and peritumoral edema[8,9]. What’s more, the presence of peritumoral edema is reported as an independent prognostic factor for breast cancer[5,7,10].
In breast cancer, breast edema is the buildup of interstitial fluid caused by disruption of the balance between vascular permeability, lymphatic drainage, and tissue compliance due to malignant infiltration, tumor-associated angiogenesis, or treatment-induced changes in the breast’s vasculature and lymphatic architecture and function[11-13]. The different patterns of edema define different types of diagnostic and prognostic importance.
Peritumoral edema is a result of two main mechanisms: Increased vascular permeability and early lymphatic obstruction. Factors like vascular endothelial growth factor activate tumor angiogenesis, which causes the production of immature, leaky vessels that allow protein-rich plasma to leak into the stroma[5,8]. Meanwhile, by invading lym
When superficial dermal lymphatics are blocked, usually because of tumor emboli or widespread inflammatory changes, subcutaneous edema occurs. It can also show up in advanced locally invasive disease, but it is typical of inflammatory breast cancer. The accumulation of protein-rich interstitial fluid in the skin and subcutaneous fat is caused by impaired lymphatic outflow. This results in the classic clinical sign - peau d’orange[15,17]. Skin thickening and an increase in signal in the superficial fat layer are imaging signs of subcutaneous edema[12,16]. Its presence indicates widespread lymphovascular spread and is strongly linked to a poor prognosis[5,17].
Prepectoral edema happens when enlarged metastatic nodes hinder fluid return along prepectoral pathways, or when malignant obstruction spreads into lymphatics draining the axillary region or chest wall. When fluid builds up in the tissues close to the pectoral muscles, it can be seen on MRI as a high signal intensity[12]. For staging and treatment planning, this finding has important ramifications as it implies advanced locoregional disease, involvement of the chest wall, or nodal metastasis[15,18].
According to the findings from the present study, there is no statistically significant difference in the presence of edema among various histological subtypes. This suggests that the development of breast edema is more closely related to tumor biology andmicro environment changes rather than to histological type alone. Similar to our results, other studies have also reported lower rates of peritumoral edema in invasive lobular carcinoma, presumably due to its distinctive growth pattern- although this association has not been conclusively demonstrated[5,6,19-21].
When comparing molecular subtypes, we found that the vast majority of tumors without associated edema belonged to luminal subtypes, comprising 44.8% luminal A and 49.3% luminal B HER2-negative carcinomas. Furthermore, a statistically significant association was identified between the presence of estrogen and progesterone receptor expression and the absence of edema. This finding suggests that tumors lacking edema on MRI are more likely to correspond to luminal, hormone receptor-positive subtypes, which are generally linked to more favorable prognostic profiles. Similarly, many other authors have reported a significant correlation between breast edema and non-luminal tumors[6,19,22]. A possible explanation is the typically slower growth kinetics and lower angiogenic potential of luminal tumors, which may lead to less impairment in lymphatic drainage and lower vascular permeability, thus reducing interstitial fluid accumulation.
Our study demonstrated that nearly all HER2- positives cases exhibited edema, further supporting the hypothesis that HER2-driven angiogenesis contributes not only to tumor growth but also to stromal changes that promote fluid accumulation visible on T2-weighted images. The receptor tyrosine kinase HER2 is part of the family of human epidermal growth factor receptors. Overexpression of HER2 has been shown to upregulate vascular endothelial growth factor, thereby facilitating neovascularization and increasing vascular permeability, processes that lead to increased extracellular fluid accumulation[6,11,22,23].
An association between breast edema and TNBC is also demonstrated. TNBC, characterized by the absence of estrogen receptor, progesterone receptor, and HER2 receptor expression, is well known for its aggressive clinical course and poor prognosis[24]. In our cohort, nearly all tumors of this molecular subtype presented with edema on MRI. A potential explanation may be the high Ki67 expression, which reflects increased proliferative activity, rapid tumor growth, and increased angiogenesis. Several authors, including Abdelbary et al[6], Uematsu et al[15], Panzironi et al[21], Huang et al[22], and Baltzer et al[25] have further reported a correlation between tumor size and the presence of edema. Although no statistically significant difference was identified between Ki67 expression and BES in our study, lesions with Ki67 values above 20% tended to present more frequently with edema. This trend might still be biologically relevant, as a high proliferative index reflects more aggressive tumor behavior, increased metabolic demand, and angiogenic signaling, all of which may promote vascular leakage and lymphatic drainage. The absence of statistical significance in our cohort is likely related to the limited number of cases within some BES subgroups, particularly in the higher edema categories, rather than the absence of a true biological association. In addition, the presence of lymphovascular invasion may contribute to this phenomenon by obstructing vessels and lymphatic drainage, increasing vascular permeability, and thus fluid extravasation[5,8,26,27]. Wei et al[28] identified peritumoral edema in 67.6% TNBC cases in comparison to none of the cases with fibroadenomas. Abdelbary et al[6] further demonstrated that all TNBC cases presented with peritumoral edema with a significantly higher prevalence of moderate (prepectoral) and severe (subcutaneous) edema. Similarly, Chen et al[12] reported an association between higher grades of edema and more biologically aggressive tumors.
The existing literature provides heterogeneous and conflicting evidence regarding the relationship between breast edema and lymph node involvement. While several studies have demonstrated a statistically significant association between edema and higher nodal stage[6,7,12,20], others have failed to confirm such a correlation[5,19]. In this study, lymphadenopathy was observed to be more prevalent in higher BES grades (3 and 4), although this did not reach statistical significance. This pattern may reflect progressive lymphatic obstruction and locoregional tumor spread, which are likely mechanisms behind the development of prepectoral and subcutaneous edema.
Overall, these findings suggest that breast edema seen on MRI reflects underlying tumor biology rather than being a nonspecific imaging feature. The observed associations with molecular subtype and hormone receptor status indicate that BES, as a structured method for grading the extent and distribution of edema, may serve as an indirect imaging biomarker of tumor aggressiveness and tumor-related microenvironmental alterations.
This study has several limitations. First, it is a single-center retrospective analysis, which may limit the generalizability of the findings. Second, the number of patients in certain subgroups was relatively small, potentially reducing the statistical power for subgroup comparisons. These limitations highlight the need for larger, prospective, multicenter studies to validate the observed associations.
MRI can be utilized to noninvasively assess the tumor microenvironment, thereby providing invaluable additional information. Routine evaluation and reporting of breast edema, including its BES, can be applied as an adjunct imaging marker to support preoperative risk stratification, to suggest the presence of more aggressive tumor biology, and to improve prediction of axillary nodal involvement and unfavorable molecular sub types. Thus, BES can complement established MRI descriptors and clinic pathological factors, potentially aiding treatment planning and supporting tailored therapeutic strategies.
| 1. | Global Cancer Observatory. Statistics at a glance, 2022. Top 5 most frequent cancers. [cited 5 February 2026]. Available from: https://gco.iarc.who.int/media/globocan/factsheets/populations/900-world-fact-sheet.pdf. |
| 2. | Sardanelli F, Magni V, Rossini G, Kilburn-Toppin F, Healy NA, Gilbert FJ. The paradox of MRI for breast cancer screening: high-risk and dense breasts-available evidence and current practice. Insights Imaging. 2024;15:96. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 31] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 3. | Schoub PK. Understanding indications and defining guidelines for breast magnetic resonance imaging. SA J Radiol. 2018;22:1353. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 4. | Iacob R, Manolescu DL, Stoicescu ER, Fabian A, Malita D, Oancea C. Breast Cancer-How Can Imaging Help? Healthcare (Basel). 2022;10:1159. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 5. | Cheon H, Kim HJ, Kim TH, Ryeom HK, Lee J, Kim GC, Yuk JS, Kim WH. Invasive Breast Cancer: Prognostic Value of Peritumoral Edema Identified at Preoperative MR Imaging. Radiology. 2018;287:68-75. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 118] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 6. | Abdelbary EAM, Ibrahim AR, Rezk KM, Omar NN. Breast edema score at breast MRI: its value in prediction of molecular subtype of breast cancer and its impact on axillary LN metastasis. Egypt J Radiol Nucl Med. 2024;55:76. [DOI] [Full Text] |
| 7. | Moradi B, Gity M, Etesam F, Borhani A, Ahmadinejad N, Kazemi MA. Correlation of apparent diffusion coefficient values and peritumoral edema with pathologic biomarkers in patients with breast cancer. Clin Imaging. 2020;68:242-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 8. | Park NJ, Jeong JY, Park JY, Kim HJ, Park CS, Lee J, Park HY, Jung JH, Kim WW, Chae YS, Lee SJ, Kim WH. Peritumoral edema in breast cancer at preoperative MRI: an interpretative study with histopathological review toward understanding tumor microenvironment. Sci Rep. 2021;11:12992. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 9. | Dietzel M, Trimboli RM, Zanardo M, Schultz-Wendtland R, Uder M, Clauser P, Sardanelli F, Baltzer PAT. The potential of predictive and prognostic breast MRI (P2-bMRI). Eur Radiol Exp. 2022;6:42. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 10. | Harada TL, Uematsu T, Nakashima K, Kawabata T, Nishimura S, Takahashi K, Tadokoro Y, Hayashi T, Tsuchiya K, Watanabe J, Sugino T. Evaluation of Breast Edema Findings at T2-weighted Breast MRI Is Useful for Diagnosing Occult Inflammatory Breast Cancer and Can Predict Prognosis after Neoadjuvant Chemotherapy. Radiology. 2021;299:53-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 53] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 11. | Gupta SS, Mayrovitz HN. The Breast Edema Enigma: Features, Diagnosis, Treatment, and Recommendations. Cureus. 2022;14:e23797. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 12. | Chen Y, Wang L, Luo R, Liu H, Zhang Y, Wang D. Focal breast edema and breast edema score on T2-weighted images provides valuable biological information for invasive breast cancer. Insights Imaging. 2023;14:73. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 16] [Reference Citation Analysis (0)] |
| 13. | Koyama H, Kobayashi N, Harada M, Takeoka M, Kawai Y, Sano K, Fujimori M, Amano J, Ohhashi T, Kannagi R, Kimata K, Taniguchi S, Itano N. Significance of tumor-associated stroma in promotion of intratumoral lymphangiogenesis: pivotal role of a hyaluronan-rich tumor microenvironment. Am J Pathol. 2008;172:179-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 96] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 14. | Kwak JY, Kim EK, Chung SY, You JK, Oh KK, Lee YH, Kwon TH, Jung HK. Unilateral breast edema: spectrum of etiologies and imaging appearances. Yonsei Med J. 2005;46:1-7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 37] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 15. | Uematsu T. Focal breast edema associated with malignancy on T2-weighted images of breast MRI: peritumoral edema, prepectoral edema, and subcutaneous edema. Breast Cancer. 2015;22:66-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 110] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 16. | Le-Petross HT, Cristofanilli M, Carkaci S, Krishnamurthy S, Jackson EF, Harrell RK, Reed BJ, Yang WT. MRI features of inflammatory breast cancer. AJR Am J Roentgenol. 2011;197:W769-W776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 49] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 17. | Uematsu T. MRI findings of inflammatory breast cancer, locally advanced breast cancer, and acute mastitis: T2-weighted images can increase the specificity of inflammatory breast cancer. Breast Cancer. 2012;19:289-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 31] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 18. | Sharma S, Vicenty-Latorre FG, Elsherif S, Sharma S. Role of MRI in Breast Cancer Staging: A Case-Based Review. Cureus. 2021;13:e20752. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 19. | Kushvaha S, Renganathan R. Presence of peritumoral edema on T2w MRI: a poor non-invasive prognostic marker in breast cancer patients. Egypt J Radiol Nucl Med. 2022;53:210. [RCA] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 20. | Liang T, Hu B, Du H, Zhang Y. Predictive value of T2-weighted magnetic resonance imaging for the prognosis of patients with mass-type breast cancer with peritumoral edema. Oncol Lett. 2020;20:314. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 21. | Panzironi G, Moffa G, Galati F, Marzocca F, Rizzo V, Pediconi F. Peritumoral edema as a biomarker of the aggressiveness of breast cancer: results of a retrospective study on a 3 T scanner. Breast Cancer Res Treat. 2020;181:53-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 52] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 22. | Huang Z, Tu X, Lin Q, Zhan Z, Tang L, Liu J, Lin D, Luo S, Zhang D, Ruan C. Intramammary edema of invasive breast cancers on MRI T(2)-weighted fat suppression sequence: Correlation with molecular subtypes and clinical-pathologic prognostic factors. Clin Imaging. 2022;83:87-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 23. | Mendelsohn J, Baselga J. Epidermal growth factor receptor targeting in cancer. Semin Oncol. 2006;33:369-385. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 562] [Cited by in RCA: 501] [Article Influence: 25.1] [Reference Citation Analysis (0)] |
| 24. | Adrada BE, Moseley TW, Kapoor MM, Scoggins ME, Patel MM, Perez F, Nia ES, Khazai L, Arribas E, Rauch GM, Guirguis MS. Triple-Negative Breast Cancer: Histopathologic Features, Genomics, and Treatment. Radiographics. 2023;43:e230034. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 37] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 25. | Baltzer PA, Yang F, Dietzel M, Herzog A, Simon A, Vag T, Gajda M, Camara O, Kaiser WA. Sensitivity and specificity of unilateral edema on T2w-TSE sequences in MR-Mammography considering 974 histologically verified lesions. Breast J. 2010;16:233-239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 75] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 26. | Akdogan Gemıcı A, Tokgoz Ozal S, Hocaoğlu E, Arslan G, Sen E, Altınay S, İnci E. Relation of peritumoral, prepectoral and diffuse edema with histopathologic findings of breast cancer in preoperative 3T magnetic resonance imaging. J Surg Med. 2019;3:49-53. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 27. | Sung P, Lee JY, Cheun JH, Choi IS, Park JH, Park JH, Kim BH, Oh S, Chu AJ, Hwang KT. Prognostic Implication of Focal Breast Edema on Preoperative Breast Magnetic Resonance Imaging in Breast Cancer Patients. J Breast Cancer. 2023;26:479-491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 28. | Wei Z, Chen X, Yang Y, Yang L, Ma X. The potential role of breast MRI in evaluation of triple-negative breast cancer and fibroadenoma of less than 3 cm. Transl Cancer Res. 2024;13:4042-4051. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |