Published online Apr 15, 2026. doi: 10.4251/wjgo.v18.i4.116949
Revised: January 14, 2026
Accepted: February 9, 2026
Published online: April 15, 2026
Processing time: 134 Days and 4.2 Hours
Screening can play a role in primary prevention by mitigating modifiable risk factors. However, whether upper gastrointestinal (UGI) endoscopic screening can reduce the burden of modifiable risk factors is unknown.
To evaluate the effect of UGI endoscopic screening on modifiable risk factors for cancer and its potential role in primary prevention.
We collected data from 11842 participants in the screening arm (received standard UGI endoscopy) and 12793 participants in the control arm (received abdominal B-ultrasound examination) of a randomized controlled trial. The participants com
UGI endoscopic screening significantly increased the cessation rate of cigarette smoking, alcohol drinking and unhealthy dietary habits [odds ratio (OR)smoking = 1.20 (1.06-1.36), ORdrinking = 1.33 (1.19-1.50) and ORunhealthy dietary habits = 1.07 (1.02-1.13)]. Subgroup analysis showed that the cessation effects were dominant among subjects who com
UGI endoscopic screening increased the cessation of modifiable risk factors; thus, contributing to primary pre
Core Tip: This study provides novel evidence that upper gastrointestinal (UGI) endoscopic screening contributes to primary cancer prevention by reducing modifiable risk factors. Based on a population-based randomized controlled trial, we showed that UGI screening significantly promoted the reduction of cigarette smoking, alcohol drinking, and unhealthy dietary habits, particularly among women and older adults. These findings highlight the potential of UGI endoscopic screening to modify behavioral risk factors at the population level, underscoring its broader preventive value in cancer control strategies. Integrating health education into UGI screening programs may further enhance its effectiveness in reducing the cancer burden.
- Citation: Cheng XW, Liu Z, Guo CH, Li FL, Pan YQ, Liu Y, Liu FF, Chen HY, Yang WL, Tian HR, Shi C, He ZH, Liu MF, Ke Y. Upper gastrointestinal endoscopic screening serves as a primary prevention approach by reducing modifiable risk factors. World J Gastrointest Oncol 2026; 18(4): 116949
- URL: https://www.wjgnet.com/1948-5204/full/v18/i4/116949.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i4.116949
Modifiable risk factors refer to behaviors and exposures that can be altered to reduce the risk of developing diseases. These factors can be classified into three main types: Lifestyle risk factors (include tobacco smoking, physical inactivity, and excessive alcohol use), dietary risk factors and metabolic risk factors[1]. National representative surveys in the United States reported that 40.0% of all incident cancers in adults aged 30 years and above were attributable to modifiable risk factors. Among these factors, cigarette smoking, alcohol consumption and dietary habits were the main contributors to cancer, accounting for 19.3%, 5.4% and 4.2% of the burden of incident cancer cases, respectively[2]. Mitigating modifiable risk factors is a key approach for the primary prevention of cancer. A large-scale prospective cohort study reported that adherence to a healthy lifestyle recommended by the cancer prevention guidelines was associated with reductions in all-cancer incidence with hazard ratios (95%CI) of 0.90 (0.87-0.93) in men and 0.81 (0.77-0.84) in women[3].
Screening, which aims to improve early detection and treatment, is generally regarded as a method of secondary prevention for cancer[4]. However, screening practice also offers a teachable moment (TM), a situation that has strong potential to motivate positive change in health behaviors, and contributes to primary prevention for all cancers by mitigating modifiable risk factors. Knudsen et al[5] found a modest improvement in lifestyle factors after endoscopic screening for colorectal cancer (CRC), suggesting that endoscopic screening could serve as a TM for primary prevention. Similar results were also reported in computed tomography screening for lung cancer, mammogram screening for breast cancer and other screening studies[6-8].
Upper gastrointestinal tract (UGI) cancers impose significant burdens in East Asia, and accounted for 12.08%, 14.59% and 11.25% of all-cancer cases in China, Japan and Korea in 2022, respectively[9,10]. As the gold-standard screening approach for UGI cancer, large-scale population-level UGI endoscopic screening has been conducted in these countries since 1980s[11-14]. However, studies evaluating the effect of UGI endoscopic screening on modifiable risk factors for cancers are currently lacking. Although it is already known that endoscopic screening for CRC can lead to a decrease in modifiable risk factors, conclusions derived from CRC screening cannot be directly applied to UGI cancers, due to significant differences regarding the features of populations undergoing endoscopies[5,15,16]. CRC is more prevalent in developed regions, where individuals typically exhibit higher health consciousness; however, UGI cancers are common in economically undeveloped areas, characterized by a low education level and health awareness[16], which may result in differences in changing attitude and behavior.
Our previous reports based on a large-scale community-based cluster randomized controlled trial (RCT) indicate that UGI endoscopic screening decreased the incidence and mortality of UGI cancer[17,18]. In this study, we investigated the effect of UGI endoscopic screening on modifiable risk factors for cancer, including cigarette smoking, alcohol drinking and unhealthy diet habits, based on repeated cross-sectional questionnaire data before and after screening from this RCT. We also performed subgroup analysis and stratified analysis to identify the optimal timing for teaching and which populations are prone to changing their risk behaviors.
The current study was based on the Endoscopic Screening for Esophageal Cancer in China (ESECC) cluster RCT conducted since 2012 in Hua County, a high-risk area for esophageal squamous cell carcinoma in China (Clinical
A total of 17151 subjects in the screening arm and 16797 in the control arm were enrolled in the ESECC trial. For the current study, we enrolled 11842 and 12793 ESECC participants in the screening and control arms, respectively, who had completed the baseline and a repeat questionnaire after a median interval of 6.55 years.
The exposure variable in this study was defined as the intended allocation of UGI endoscopic screening. Participants in the screening arm were invited to accept standard UGI endoscopy with iodine staining using 1.2% Lugol’s solution. The entire esophagus and stomach were systematically examined, and biopsies were taken from suspected lesions before or after staining. Biopsy specimens were diagnosed by two experienced pathologists in the Anyang Cancer Hospital and discrepancies were resolved by consultation. The Anyang Cancer Hospital is the sole tertiary cancer hospital in the Taihang Mountain region and participates in the national screening program for UGI cancers in China[11]. Immediately after the endoscopic examination, endoscopic reports were given to the participant along with a unified recommendation informing him/her to consult a physician at a local hospital or their village doctor for advice.
In the control arm, an abdominal ultrasound scan was performed for each individual, which had no role in detecting UGI cancers. Individuals with detected abnormalities were informed immediately after the examination, and were recommended to consult a physician in a local hospital or his/her village doctor.
Our primary outcome was the change regarding the status of modifiable risk factors for cancer (including cigarette smoking, alcohol drinking and unhealthy dietary habits) after baseline examination, which were further classified as cessation and incident events. In this study, each participant completed a computer-aided one-on-one questionnaire survey conducted by a well-trained interviewer on the same day before the baseline examination. A repeat questionnaire was completed by these individuals at the second cross-sectional investigation (including questionnaire survey and blood sampling in all enrolled participants) of the ESECC trial, by the same team of interviewers using the same questionnaire a median of 6.55 years after baseline examination.
For cigarette smoking and alcohol drinking, dichotomous variables were applied to reflect their status. Cessation was defined as those reporting as current smokers/drinkers at baseline but not currently smoking/drinking at the repeat questionnaire. An incident event was defined as those reporting as non-smokers/drinkers at baseline but currently smoking/drinking at the repeat questionnaire.
The unhealthy dietary habit score was constructed to assess the overall effect of modifiable dietary factors on UGI cancers. The score was the summation of self-reported status of well-known dietary habits associated with UGI cancer risk, including the regularity and speed of food intake; the preference of food temperature and hardness; the ingestion of salty, spicy, fried, moldy, preserved food and leftovers; and the intake of fruit, vegetables and meat/egg/milk[19,20]. The coding and points assignment of each dietary variable can be found in Supplementary Table 1. Changes in unhealthy dietary habit score were defined as the unhealthy dietary score difference (repeat questionnaire minus baseline ques
Covariates collected in this study that may influence the change in modifiable risk factors, included age, sex, education, marital status, occupation, household income, body mass index, gastrointestinal symptoms score, family history of cancer, self-reported personal disease history, cigarette smoking status, years of cigarette smoking, lifetime cigarette consumption, cigarette smoking in family members and alcohol drinking status at the baseline questionnaire investigation. The coding of each variable can be found in the Supplementary material.
The median (interquartile range), mean (SD) and n (%) were applied to describe baseline characteristics of the enrolled participants. The Wilcoxon test and χ2 test were used to test differences between the screening arm and control arm for continuous and categorical variables, respectively.
Multivariable logistic regression analysis was applied to assess the effect of UGI endoscopic screening on the cessation and incidence of cigarette smoking, alcohol drinking and unhealthy dietary habits separately, using the respective changes in the randomized control arm as the reference. For cessation, subjects without the modifiable risk factors at baseline were excluded from the respective analysis. However, these persons were subject to the incidence analysis of modifiable risk factors.
All analyses were adjusted for the following covariates potentially related to the change in health risk behaviors: (1) Baseline age; (2) Sex; (3) Baseline body mass index; (4) Household income; (5) Education; (6) Marital status; (7) Occupa
In addition to the primary analysis following the intention-to-treat principal, we also performed subgroup analysis according to the uptake of UGI endoscopic screening, which can be grouped into subjects who declined UGI endoscopy, completed UGI endoscopy but without biopsy, and completed UGI endoscopy with biopsy, to evaluate the discrepancy in screening effects on these modifiable risk factors. Stratified analyses were performed to evaluate the impact of biopsy results on behavioral changes. We also performed stratified analysis according to baseline age (< 60 years and ≥ 60 years), sex (male and female) and baseline gastrointestinal symptoms score (0 and ≥ 1), to assess target populations that are prone to changing their risk behaviors. Interaction terms between each stratified variable and the screening status were tested using the Wald-test. All analyses were conducted by R 4.3.3. P values were two-sided and P < 0.05 was considered statistically significant.
A total of 11842 ESECC trial participants in the screening arm and 12793 participants in the control arm were enrolled in this study. In the screening arm, 390 subjects declined UGI endoscopy, 10593 subjects completed UGI endoscopy but without biopsy, and 859 subjects completed UGI endoscopy with biopsy. The two arms were well balanced in terms of age, marital status and occupation but had significant differences in other characteristics and risk factors at the baseline investigation (Supplementary Table 2).
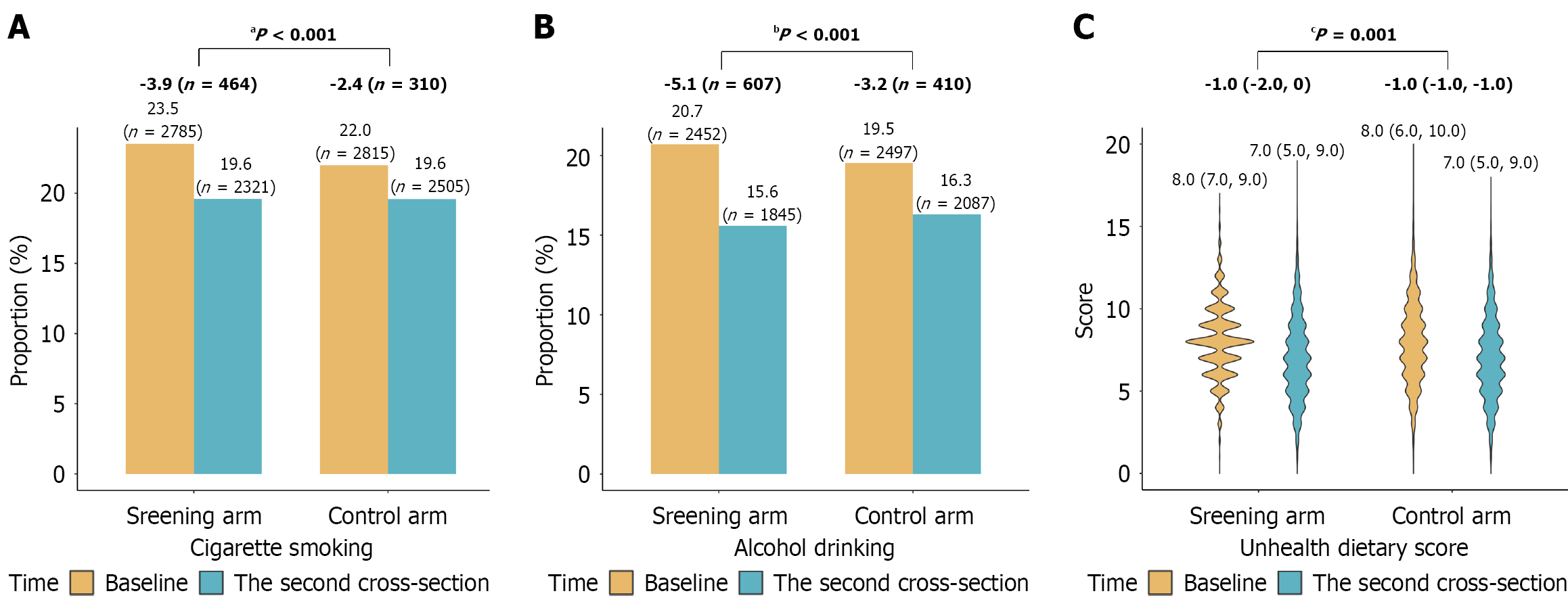
As compared to the baseline, the prevalence of cigarette smoking, alcohol drinking and unhealthy dietary habits decreased both in the screening and control arms at a median of 6.55 years after examination. However, the extent of the decreases in prevalence regarding all three modifiable risk factors were significantly larger in the screening arm as com
A total of 5600 current smokers at baseline were included in the smoking cessation analysis. UGI endoscopic screening was associated with a 20% increase in cessation rate as compared to the unscreened control arm [odds ratio (OR) (95%CI) = 1.20 (1.06-1.36), Table 1 and Supplementary Table 3]. The association was dominant in subjects who completed the endoscopic examination, with an OR of 1.15 (1.01-1.31) without biopsy, and 1.77 (1.32-2.35) for subjects with biopsy. However, the association between UGI endoscopic screening and smoking cessation was not significant in subjects who declined UGI endoscopy [OR = 1.09 (0.65-1.83), Table 2]. For the stratified analysis on results of biopsy, subjects with high grade intraepithelial neoplasia (HGIN) had the largest effect size on cessation [OR = 15.21 (3.32-69.71), Supplementary Table 4]. In contrast, stratified analyses on sex, age, and baseline gastrointestinal symptom scores showed no significant heterogeneity in the associations (Table 3). In multivariable analysis of incidence, 19035 non-smokers were analyzed and no significant effect of UGI endoscopic screening was observed (Supplementary Table 5).
| Variable | Cessation of cigarette smoking (n = 5600)1 | Cessation of alcohol drinking (n = 4949)2 | Reduction of unhealthy dietary habit | |||
| Outcome events/total (%) | Adjusted OR (95%CI) | Outcome events/total (%) | Adjusted OR (95%CI) | Outcome events/total (%) | Adjusted OR (95%CI) | |
| Screening | ||||||
| Control arm | 681/2815 (24.19) | Reference | 960/2497 (38.45) | Reference | 5059/12790 (39.55) | Reference |
| Screening arm | 775/2785 (27.83) | 1.20 (1.06-1.36) | 1114/2452 (45.43) | 1.33 (1.19-1.50) | 4870/11839 (41.14) | 1.07 (1.02-1.13) |
| Variable | Cessation of cigarette smoking (n = 5600)1 | Cessation of alcohol drinking (n = 4949)2 | Reduction of unhealthy dietary habit (n = 24629)3 | |||
| Outcome events/total (%) | Adjusted OR (95%CI) | Outcome events/total (%) | Adjusted OR (95%CI) | Outcome events/total (%) | Adjusted OR (95%CI) | |
| Screening status | ||||||
| Control arm | 681/2815 (24.19) | Reference | 960/2497 (38.45) | Reference | 5059/12790 (39.55) | Reference |
| Subjects decline UGI endoscopy | 20/88 (22.73) | 1.09 (0.65-1.83) | 33/85 (38.82) | 1.18 (0.75-1.87) | 156/389 (40.10) | 1.04 (0.85-1.28) |
| Subjects complete UGI endoscopy but without biopsy | 663/2465 (26.90) | 1.15 (1.01-1.31) | 974/2145 (45.41) | 1.35 (1.19-1.52) | 4314/10591 (40.73) | 1.06 (1.00-1.11) |
| Subjects complete UGI endoscopy with biopsy | 92/232 (39.66) | 1.77 (1.32-2.35) | 107/222 (48.20) | 1.27 (0.96-1.69) | 400/859 (46.57) | 1.31 (1.14-1.50) |
| Stratified variable | Cessation of cigarette smoking (n = 5600)1 | Cessation of alcohol drinking (n = 4949)2 | Reduction of unhealthy dietary habit | ||||||
| Outcome events/total (%) | Adjusted OR (95%CI) | P value | Outcome events/total (%) | Adjusted OR (95%CI) | P value | Outcome events/total (%) | Adjusted OR (95%CI) | P value | |
| Sex | |||||||||
| Female | 48/94 (51.06) | 1.50 (0.58-3.85) | 0.399 | 42/63 (66.67) | 1.56 (0.37-6.53) | 0.546 | 5550/13255 (41.87) | 1.12 (1.04-1.20) | 0.002 |
| Male | 1408/5506 (25.57) | 1.19 (1.05-1.35) | 0.007 | 2032/4886 (41.59) | 1.32 (1.17-1.49) | < 0.001 | 4379/11374 (38.50) | 1.02 (0.94-1.10) | 0.617 |
| Interaction4 | 0.557 | 0.181 | 0.033 | ||||||
| Baseline age | |||||||||
| < 60 | 700/3241 (21.60) | 1.16 (0.98-1.38) | 0.089 | 1108/3026 (36.62) | 1.16 (0.99-1.35) | 0.061 | 5752/14515 (39.63) | 1.03 (0.96-1.10) | 0.451 |
| ≥ 60 | 756/2359 (32.05) | 1.23 (1.03-1.47) | 0.024 | 966/1923 (50.23) | 1.66 (1.38-2) | < 0.001 | 4177/10114 (41.30) | 1.14 (1.05-1.24) | 0.001 |
| Interaction4 | 0.589 | 0.004 | 0.130 | ||||||
| Baseline gastrointestinal symptoms score | |||||||||
| 0 | 795/3092 (25.71) | 1.16 (0.98-1.37) | 0.087 | 997/2586 (38.55) | 1.41 (1.20-1.67) | < 0.001 | 5322/13578 (39.20) | 1.08 (1.01-1.16) | 0.029 |
| ≥ 1 | 661/2508 (26.36) | 1.25 (1.04-1.51) | 0.018 | 1077/2363 (45.58) | 1.26 (1.07-1.49) | 0.007 | 4607/11051 (41.69) | 1.06 (0.98-1.14) | 0.158 |
| Interaction4 | 0.488 | 0.436 | 0.662 | ||||||
For alcohol drinking cessation, 4949 current drinkers at baseline were included and an increase in cessation rate associated with screening was observed with an OR of 1.33 (1.19-1.50) (Table 1 and Supplementary Table 3). In subgroup analysis, the association was only significant in subjects who completed UGI endoscopy but without biopsy [OR = 1.35 (1.19-1.52), Table 2]. In stratified analyses on biopsy results, no significant differences were observed among individuals with non-dysplasia, low-grade intraepithelial neoplasia, and HGIN (Supplementary Table 4). Age was identified as an effect modifier for the association between UGI endoscopic screening and drinking cessation, which indicate that individuals aged > 60 years are more inclined to stop drinking (Pinteraction = 0.004; Table 3). In multivariable analysis of incidence, 19686 non-drinkers were analyzed and no significant effect of UGI endoscopic screening was observed (Supplementary Table 5).
In multivariable analysis of unhealthy dietary habit reduction, 24629 individuals with unhealthy dietary habits were included, and a greater reduction associated with screening was observed [OR = 1.07 (1.02-1.13), Table 1 and Supplementary Table 3]. This association was also dominant among individuals who underwent UGI endoscopic screening, with an OR of 1.04 (0.85-1.28) for subjects who declined UGI endoscopy, 1.06 (1.00-1.11) for subjects who completed UGI endoscopy but without biopsy, and 1.31 (1.14-1.50) for those who completed UGI endoscopy with biopsy (Table 2). In stratified analyses on biopsy results, subjects with HGIN showed most significant changes regarding their behaviors [OR = 2.48 (1.39-4.42), Supplementary Table 4]. In addition, sex-stratified analysis revealed that females were more likely than males to reduce unhealthy dietary habits (Pinteraction = 0.033) (Table 3). In multivariable analysis of incidence, 24635 individuals with the possibility of reductions in dietary habits were included in the analysis and decreases in incidence rate due to screening were detected with an OR of 0.85 (0.80-0.91) (Supplementary Table 5).
Although screening is typically regarded as a secondary prevention approach, it also plays a role in health education through knowledge dissemination from medical staff, and the impact of screening positive in the participant or their relatives, thereby provides primary prevention benefits across multiple cancers[15,22]. The current study found that UGI endoscopic screening can increase the cessation rate of modifiable risk factors including cigarette smoking, alcohol drinking and unhealthy dietary habits, thus acts as an effective approach for primary prevention of various cancers driven by these risk factors. Moreover, the endoscopic procedure itself was the key step in influencing the changes in modifiable risk factors, independent of screening outcomes. Females and individuals aged > 60 years were more inclined to change their risk behaviors in rural China. Our findings fill the gap in understanding the role of UGI endoscopic screening in primary prevention of cancer, and sheds light on the optimal timing and target populations for health education.
In multivariable analysis, it was found that UGI endoscopic screening played a more significant role in increasing cessation than decreasing incidence to mitigate modifiable risk factors. This can be explained by the self-referential effect, which claims that individuals have a tendency to remember information related to themselves, and disregard information perceived as irrelevant to their behaviors[23]. Thus, subjects with modifiable risk behaviors may be a better target popu
Several studies have shown that screening may have adverse effects in screening negative subjects. Negative findings at screening may make one to perceive that he/she is in good general health, and thus more prone to indulge in unhealthy behaviors, known as the “health certificate” effect[24,25]. However, our subgroup analysis revealed that while greater improvements were observed in subjects who completed UGI endoscopy with biopsy, significant increases in cessation rates were also noted in subjects who completed UGI endoscopy but without biopsy compared to the control arm, suggesting that the act of screening itself serves as a TM, independent of screening outcomes in this rural popula
This discrepancy between our results and others can mainly be explained by differences in health awareness and accessibility of health education among populations. Older individuals in rural China (i.e., our study population) often have low education levels, limited health awareness, few opportunities to receive health education but higher burdens of modifiable risk factors[26-29]. These features make the older individuals more vulnerable and susceptible to the influence of health education efforts, even when endoscopy results are negative[30,31]. Therefore, integrating health education into screening programs in rural areas may promote the adoption of a healthy lifestyle and maximize the efficacy of cancer screening.
The stratified analysis in this study revealed that females and individuals aged > 60 years were more inclined to improve their risk behavior, which indicates that the old and women are more vulnerable to health promotion modalities in this rural population, as they usually have low social economic status[32,33]. In line with our findings, Townsend et al[6] demonstrated that older people were associated with a higher possibility of smoking abstinence after chest computed tomography screening. Studies have also shown that women had higher risk perception and adherence compared to men due to social expectations[34,35]. Thus, among the vast populations in rural areas in China, conveying health education knowledge to females and participants aged > 60 years at endoscopic screening may be a more effective choice.
UGI cancer, including esophageal and stomach cancers, has a high incidence in China[9]. Organized community-based endoscopic screening for UGI cancer has been conducted in high-risk rural areas since 2005[12]. However, to date, the population coverage of UGI endoscopic screening in China is still limited, due to a shortage of endoscopists and health resources, especially in the vast undeveloped rural areas. Opportunistic UGI endoscopic screening in outpatient clinics has been proposed by the national cancer center in China since 2019. Characterized by its high endoscopic compliance and relatively low cost, opportunistic UGI endoscopic screening has rapidly been expanded nationwide and 1180000 individuals completed the screening in a single year in 2020[36,37]. With the increase in accessibility, UGI endoscopic screening could offer an integrated benefit, not only by decreasing UGI-specific mortality through detection and treat
This study has two main strengths. First, it was based on an ongoing large-scale community-based RCT, which pro
The limitations of our study should also be noted. First, the modifiable risk factors we assessed were limited due to unavailable data in the questionnaire. Crucial risk factors, such as physical activity, were not included in the analysis. Second, although the impact of recall bias on our conclusion may be largely eliminated by use of the control arm as a reference, this bias cannot be completely excluded since we used self-reported status of cigarette smoking, alcohol drinking and unhealthy dietary habits in this study.
UGI endoscopic screening not only has a secondary prevention effect via early detection of UGI cancers, but also serves as a TM for primary prevention by mitigating modifiable risk factors. Integration of health education efforts into UGI endo
The authors thank all the following team members and collaborators for their contributions to the field work done for this study (in alphabetical order by last name and first name): Chan-Yuan Zhang, Dong Hang, Fuxiao Li, Hui Wang, Linlin Liang, Min-Min Wang, Na Shen, Qiu-Ju Deng, Ren Zhou, Wen-Qing Yuan, Xiang Li and Zi-Fan Qi, from Peking University Cancer Hospital & Institute. The authors would also like to thank the government and the Health and Family Planning Commission of Anyang City and Hua County, Henan Province, all the community leaders, village doctors and participants in the Endoscopic Screening for Esophageal Cancer in China randomized controlled trial.
| 1. | Danaei G, Ding EL, Mozaffarian D, Taylor B, Rehm J, Murray CJ, Ezzati M. The preventable causes of death in the United States: comparative risk assessment of dietary, lifestyle, and metabolic risk factors. PLoS Med. 2009;6:e1000058. [PubMed] [DOI] [Full Text] |
| 2. | Islami F, Marlow EC, Thomson B, McCullough ML, Rumgay H, Gapstur SM, Patel AV, Soerjomataram I, Jemal A. Proportion and number of cancer cases and deaths attributable to potentially modifiable risk factors in the United States, 2019. CA Cancer J Clin. 2024;74:405-432. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 104] [Article Influence: 52.0] [Reference Citation Analysis (0)] |
| 3. | Kabat GC, Matthews CE, Kamensky V, Hollenbeck AR, Rohan TE. Adherence to cancer prevention guidelines and cancer incidence, cancer mortality, and total mortality: a prospective cohort study. Am J Clin Nutr. 2015;101:558-569. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 131] [Cited by in RCA: 119] [Article Influence: 10.8] [Reference Citation Analysis (4)] |
| 4. | Sepodes B, Rebelo T, Santos F, Oliveira D, Catalão C, Águas F, Fernandes G. Optimization of HPV-positive women triage with p16/Ki67 dual staining cytology in an organized cervical cancer screening program in the center region of Portugal. Eur J Obstet Gynecol Reprod Biol. 2024;302:111-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 5. | Knudsen MD, Wang L, Wang K, Wu K, Ogino S, Chan AT, Giovannucci E, Song M. Changes in Lifestyle Factors After Endoscopic Screening: A Prospective Study in the United States. Clin Gastroenterol Hepatol. 2022;20:e1240-e1249. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 6. | Townsend CO, Clark MM, Jett JR, Patten CA, Schroeder DR, Nirelli LM, Swensen SJ, Hurt RD. Relation between smoking cessation and receiving results from three annual spiral chest computed tomography scans for lung carcinoma screening. Cancer. 2005;103:2154-2162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 87] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 7. | Khanna D, Shruti T, Tiwari M, Sharma P, Khan A, Ranjan S, Balasundaram P, Khargekar N, Chaturvedi P, Mishra A. Prevalence of Oral Potentially Malignant Lesions, Tobacco use, and Effect of Cessation Strategies among Solid Waste Management workers in Northern India: a pre-post intervention study. BMC Oral Health. 2024;24:1292. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 8. | Sinclair J, McCann M, Sheldon E, Gordon I, Brierley-Jones L, Copson E. The acceptability of addressing alcohol consumption as a modifiable risk factor for breast cancer: a mixed method study within breast screening services and symptomatic breast clinics. BMJ Open. 2019;9:e027371. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 38] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 9. | Han B, Zheng R, Zeng H, Wang S, Sun K, Chen R, Li L, Wei W, He J. Cancer incidence and mortality in China, 2022. J Natl Cancer Cent. 2024;4:47-53. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1395] [Cited by in RCA: 1423] [Article Influence: 711.5] [Reference Citation Analysis (5)] |
| 10. | Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16785] [Cited by in RCA: 14770] [Article Influence: 7385.0] [Reference Citation Analysis (21)] |
| 11. | He Z, Liu Z, Liu M, Guo C, Xu R, Li F, Liu A, Yang H, Shen L, Wu Q, Duan L, Li X, Zhang C, Pan Y, Cai H, Ke Y. Efficacy of endoscopic screening for esophageal cancer in China (ESECC): design and preliminary results of a population-based randomised controlled trial. Gut. 2019;68:198-206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 114] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 12. | Chen R, Liu Y, Song G, Li B, Zhao D, Hua Z, Wang X, Li J, Hao C, Zhang L, Liu S, Wang J, Zhou J, Zhang Y, Li B, Li Y, Feng X, Li L, Dong Z, Wei W, Wang G. Effectiveness of one-time endoscopic screening programme in prevention of upper gastrointestinal cancer in China: a multicentre population-based cohort study. Gut. 2021;70:251-260. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 123] [Article Influence: 24.6] [Reference Citation Analysis (0)] |
| 13. | Hamashima C, Shibuya D, Yamazaki H, Inoue K, Fukao A, Saito H, Sobue T. The Japanese guidelines for gastric cancer screening. Jpn J Clin Oncol. 2008;38:259-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 304] [Cited by in RCA: 271] [Article Influence: 15.1] [Reference Citation Analysis (3)] |
| 14. | Leung WK, Wu MS, Kakugawa Y, Kim JJ, Yeoh KG, Goh KL, Wu KC, Wu DC, Sollano J, Kachintorn U, Gotoda T, Lin JT, You WC, Ng EK, Sung JJ; Asia Pacific Working Group on Gastric Cancer. Screening for gastric cancer in Asia: current evidence and practice. Lancet Oncol. 2008;9:279-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 680] [Cited by in RCA: 642] [Article Influence: 35.7] [Reference Citation Analysis (2)] |
| 15. | Sriphanlop P, Jandorf L, Thompson H, Valdimarsdottir H, Redd W, Shelton RC. Preventive Health Behaviors Among Low-Income African American and Hispanic Populations: Can Colonoscopy Screening Serve as a Teachable Moment? J Racial Ethn Health Disparities. 2018;5:179-186. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 16. | Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53448] [Cited by in RCA: 56091] [Article Influence: 7011.4] [Reference Citation Analysis (10)] |
| 17. | Liu M, Yang W, Guo C, Liu Z, Li F, Liu A, Yang H, Shen L, Wu Q, Duan L, Wang H, Tian H, Shi C, Pan Y, Liu Y, Liu F, Weiss NS, Cai H, He Z, Ke Y. Effectiveness of Endoscopic Screening on Esophageal Cancer Incidence and Mortality: A 9-Year Report of the Endoscopic Screening for Esophageal Cancer in China (ESECC) Randomized Trial. J Clin Oncol. 2024;42:1655-1664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 33] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 18. | Liu M, He Z, Guo C, Xu R, Li F, Ning T, Pan Y, Li Y, Ding H, Zheng L, Zhou Y, Tian X, Yang W, Wang X, Lu F, Zhang Y, Zhao Y, Guo F, Chen K, Gao L, Sun M, Liu Y, Liu F, Hang D, Shen N, Li J, Xu Z, Wang Q, Zhang C, Abliz A, Deng Q, Li X, Liu Z, Zhang C, Yuan W, Wang H, Weiss NS, Cai H, Ke Y. Effectiveness of Intensive Endoscopic Screening for Esophageal Cancer in China: A Community-Based Study. Am J Epidemiol. 2019;188:776-784. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 59] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 19. | He J, Chen WQ, Li ZS, Li N, Ren JS, Tian JH, Tian WJ, Hu FL, Peng J; Expert Group of China Guideline for the Screening, Early Detection and Early Treatment of Esophageal Cancer; Work Group of China Guideline for the Screening,Early Detection and Early Treatment of Esophageal Cancer. [China guideline for the screening, early detection and early treatment of esophageal cancer (2022, Beijing)]. Zhonghua Zhong Liu Za Zhi. 2022;44:491-522. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 21] [Reference Citation Analysis (0)] |
| 20. | He J, Chen WQ, Li ZS, Li N, Ren JS, Tian JH, Tian WJ, Hu FL, Peng J; Expert Group of China Guideline for the Screening,Early Detection and Early Treatment of Gastric Cancer; Working Group of China Guideline for the Screening, Early Detection and Early Treatment of Gastric Cancer. [China guideline for the screening, early detection and early treatment of gastric cancer (2022, Beijing)]. Zhonghua Zhong Liu Za Zhi. 2022;44:634-666. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 21. | Glick JH Jr. Expression of random analytical error as a precentage of the range of clinical interest. Clin Chem. 1976;22:475-83. [PubMed] |
| 22. | Vernon M, Coughlin SS, Tingen M, Jones S, Heboyan V. Cancer health awareness through screening and education: A community approach to healthy equity. Cancer Med. 2024;13:e7357. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 23. | Kim K, Song H, Suh B. Self-Referential Review: Exploring the Impact of Self-Reference Effect in Review. In: Yang GH, Wang HN, Han S, Hauff C, Zuccon G, Zhang Y, editors. Proceedings of the 47th International ACM SIGIR Conference on Research and Development in Information Retrieval. United States: Association for Computing Machinery, 2024. [DOI] [Full Text] |
| 24. | Berstad P, Løberg M, Larsen IK, Kalager M, Holme Ø, Botteri E, Bretthauer M, Hoff G. Long-term lifestyle changes after colorectal cancer screening: randomised controlled trial. Gut. 2015;64:1268-1276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 37] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 25. | Larsen IK, Grotmol T, Almendingen K, Hoff G. Impact of colorectal cancer screening on future lifestyle choices: a three-year randomized controlled trial. Clin Gastroenterol Hepatol. 2007;5:477-483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 49] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 26. | Yu C. [Investigation and Analysis of Cognitive Status and Health Service Needs for Middle Aged and Elderly People with Chronic Diseases in Rural Area]. Jiefangjun Huli Zazhi. 2013;30:18-21. |
| 27. | Hu XY, Wu JP, Gong X, Xia QH, Qin SL, Li L, Yu SY, Gan T, Hu J. The characteristics of health Literacy of residents in hubei province. 2011 IEEE International Symposium on IT in Medicine and Education. 2011 December 9-11, Guangzhou, China. Bengaluru: IEEE, 2011: 512-516. [DOI] [Full Text] |
| 28. | Luo D, Du J, Wang P, Yang W. Urban-rural comparisons in health risk factor, health status and outcomes in Tianjin, China: A cross-sectional survey (2009-2013). Aust J Rural Health. 2019;27:535-541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 29. | Ma SH. An investigation of the present situation of urban and rural residents' health literacy in guangzhou city. PhD Thesis. Sun Yat-sen University. |
| 30. | Walton-Moss B, Samuel L, Nguyen TH, Commodore-Mensah Y, Hayat MJ, Szanton SL. Community-based cardiovascular health interventions in vulnerable populations: a systematic review. J Cardiovasc Nurs. 2014;29:293-307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 40] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 31. | Waller M, Blomstrand A, Högberg T, Ariai N, Thorn J, Hange D, Björkelund C. A primary care lifestyle programme suitable for socioeconomically vulnerable groups - an observational study. Scand J Prim Health Care. 2016;34:352-359. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 32. | Malnar B, Hafner-fink M. Thirty years of gender differences in self-assessed health: the case of Slovenia/Trideset let razlik v samoocenah zdravja med spoloma: primer Slovenije. SJPH. 2013;52:99-107. [RCA] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 33. | Cala-Montoya CA, Hernández R, Vázquez JJ. Economic situation, sources of income and coverage of basic needs among older adults in a vulnerable community in Cuba. J Poverty Soc Justice. 2025;33:139-154. [DOI] [Full Text] |
| 34. | Jayawardana S, Esquivel M, Orešković T, Mossialos E. Gender differences in COVID-19 preventative measures and vaccination rates in the United States: A longitudinal survey analysis. Vaccine. 2024;42:126044. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 35. | Bird CE, Rieker PP. Gender and Health. United Kingdom: Cambridge University Press, 2008. [DOI] [Full Text] |
| 36. | Wang GQ, Wei WW. [A new transition of the screening, early diagnosis and early treatment project of the upper gastrointestinal cancer: opportunistic screening]. Zhonghua Yu Fang Yi Xue Za Zhi. 2019;53:1084-1087. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 37. | Li MJ, Wei WQ. [Promoting the Prevention and Control of Esophageal Cancer with Screening as a Pointcut]. Zhongliu Yufang Yu Zhiliao. 2021;34:587-590. [DOI] [Full Text] |