Published online Apr 15, 2026. doi: 10.4251/wjgo.v18.i4.116821
Revised: January 4, 2026
Accepted: January 12, 2026
Published online: April 15, 2026
Processing time: 138 Days and 16.5 Hours
The diagnostic value of serum chitinase-3-like protein 1 (CHI3L1) in liver fibrosis and cirrhosis, as well as its prognostic significance in hepatocellular carcinoma (HCC), remains incompletely understood.
To assess the diagnostic value of CHI3L1 for liver cirrhosis in liver disease patients and its prognostic significance in HCC.
A retrospective study was conducted at a tertiary hospital to measure serum CHI3L1 levels in 551 patients from different cohorts (viral/non-viral hepatitis, cirrhosis, HCC, post-radical hepatectomy HCC, and HCC recurrence) and healthy controls using electrochemiluminescence. Receiver operating characteristic (ROC) curve analysis was used to evaluate the diagnostic performance of CHI3L1, and its correlation with liver stiffness measurement (LSM) and the fibrosis-4 index (FIB-4) was analyzed.
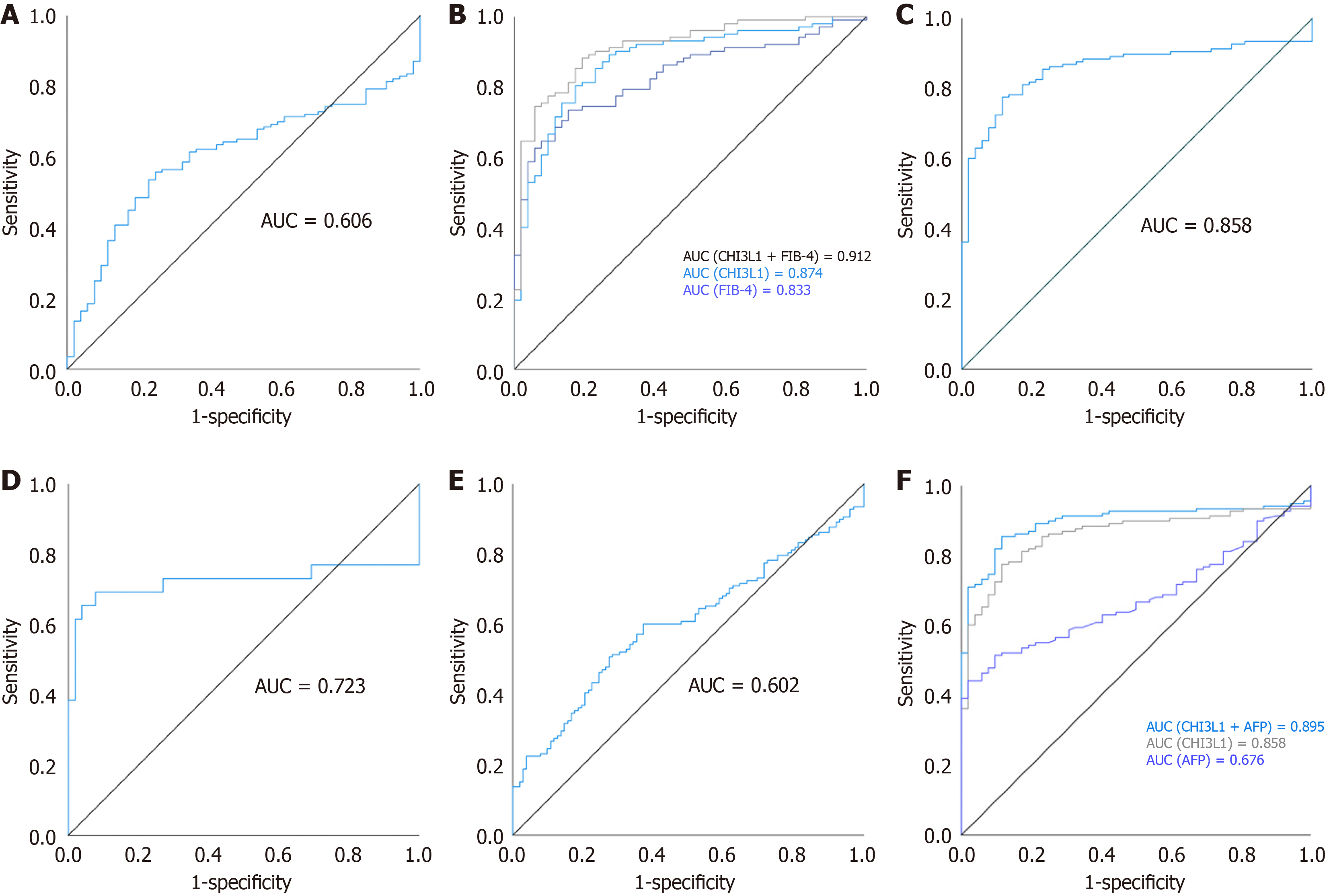
Serum CHI3L1 levels were significantly higher in the cirrhosis, HCC, post-radical hepatectomy HCC, and HCC recurrence groups than in the control group (all P < 0.05). The cirrhosis group had higher serum CHI3L1 levels than the viral hepatitis and non-viral hepatitis groups (both P < 0.01), and the HCC group had higher serum CHI3L1 levels than the post-radical hepatectomy HCC group (P < 0.001). The areas under the ROC curve of CHI3L1 for diagnosing cirrhosis, HCC, and HCC recurrence were 0.874, 0.858, and 0.723, respectively. CHI3L1 levels were positively correlated with LSM (r = 0.424, P < 0.001) and FIB-4 (r = 0.370, P < 0.001). CHI3L1 levels differed significantly between fibrosis grades F3 and F4 (P < 0.01).
Serum CHI3L1 levels hold significant diagnostic value for liver fibrosis, cirrhosis, and HCC, serving as an effective non-invasive marker for these conditions as well as HCC recurrence.
Core Tip: Serum chitinase-3-like protein 1 (CHI3L1) demonstrates robust diagnostic performance for liver cirrhosis [area under the receiver operating characteristic curve (AUC) = 0.874], hepatocellular carcinoma (HCC; AUC = 0.858) and HCC recurrence (AUC = 0.723) in liver disease patient cohorts. CHI3L1 exhibits positive correlations with liver stiffness measurement and the fibrosis-4 index, effectively differentiates fibrosis grades F3 and F4, and emerged as a reliable non-invasive biomarker for liver disease stratification, treatment monitoring and prognosis prediction, with superior performance than single traditional indices.
- Citation: Zhang HY, Zhang RL, Jia YY, Liu Y, Zhu L, Liu F, Rao HY. Diagnostic value of serum chitinase-3-like protein 1 for liver fibrosis/cirrhosis and hepatocellular carcinoma. World J Gastrointest Oncol 2026; 18(4): 116821
- URL: https://www.wjgnet.com/1948-5204/full/v18/i4/116821.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i4.116821
Chronic hepatitis B (CHB) has become a major global health challenge, having a profound impact on morbidity and mortality rates worldwide. Hepatitis B virus (HBV) infection, as a major public health issue globally, causes approximately 1.5 million new infections each year[1]. The global HBV infection rate in 2022 was approximately 3.2%, equivalent to 257 million people being positive for hepatitis B surface antigen. The focus of concern for CHB lies in its potential risk of progression, which might lead to serious liver diseases such as hepatocellular carcinoma (HCC) and cirrhosis[2].
In clinical diagnosis and treatment, rapid, accurate, and comprehensive diagnosis is crucial for delaying the progression of HBV-related liver diseases. Hepatic fibrosis is a chronic repair process of liver cell damage caused by viral infection, toxins, alcohol, and other factors. The activation of hepatic stellate cells (HSCs) and the excessive deposition of fibrous tissue in the liver can lead to serious complications, including abnormal liver function, splenomegaly, portal hypertension, and even HCC[3]. Among them, liver cirrhosis, as an advanced stage of liver fibrosis, has been confirmed to be an important risk factor for HCC[4]. The cumulative risk of liver cirrhosis progressing to HCC within 5 years ranges 5%-30%[5]. Because of difficulties in early diagnosis, rapid progression, and complex treatment, the survival period of patients with HCC is usually short. Most patients have missed the opportunity for radical surgery at the initial diagnosis, and the survival rate after diagnosis is less than 5%[6]. Numerous domestic and international studies have revealed that if the cause can be eliminated or early intervention is implemented, the progression from liver fibrosis to cirrhosis can be reversed[7]. Conversely, if the “optimal treatment window period” is missed, then irreversible liver damage might occur, eventually developing into advanced liver cirrhosis or HCC. Therefore, timely and effective diagnosis, accurate assessment of the severity of liver fibrosis, and the adoption of appropriate management and treatment measures are crucial for alleviating extensive liver damage.
Liver biopsy is recognized as the gold standard for evaluating liver necrosis, inflammation (grading), and fibrosis (staging)[8]. However, factors such as invasiveness, potential complications, and sampling errors limit its wide application[9]. In recent years, a series of non-invasive detection technologies have emerged, such as transient elastography (TE) and the fibrosis-4 index (FIB-4). TE technology has been widely used and verified because of its good repeatability and high accuracy in diagnosing liver cirrhosis. However, this technique requires specialized equipment, and its accuracy is susceptible to the levels of transaminase and bilirubin, the status of ascites, the degree of obesity in patients, and the professional skills of operators[10]. Therefore, compared with serum biomarkers, the overall applicability of TE is somewhat reduced. Composite indicators such as FibroTest aim to provide a comprehensive assessment, but they also have deficiencies in precisely grading liver fibrosis. Research by Poynard revealed a 28.7% difference between FibroTest results and those of traditional clinical and pathological examinations[11]. Therefore, in China, the demand for serum markers that can effectively identify the stages of liver fibrosis is rising. Furthermore, clinical symptoms, physical examinations, imaging screenings, and routine laboratory indicators also have deficiencies in HCC detection[12]. The 5-year overall survival rate of patients with HCC remains unsatisfactory, even after radical resection, at less than 50%[13]. Therefore, in the clinical treatment of HBV-related liver diseases, it is particularly crucial to identify non-invasive, safe, and highly specific diagnostic methods. At the same time, there is an urgent need to discover new biomarkers to accurately predict the survival of patients and improve the clinical management of HCC.
Chitinase-3-like protein 1 (CHI3L1), a member of the glycoside hydrolase family, is a secretory glycoprotein in humans. In liver tissue, CHI3L1 is mainly produced by hepatic macrophages and HSCs[14,15]. This protein, as a growth-promoting factor for fibroblasts and HSCs, can activate HSCs and promote their differentiation into myofibroblasts. During differentiation, HSCs express α-smooth muscle actin and globin and secrete a large amount of extracellular matrix, especially types I and III collagen fibers, thereby promoting the development of liver fibrosis[16]. Among all proteins secreted by the liver into the extracellular matrix, CHI3L1 has the highest expression level[17,18], and its upregulation and accumulation are involved in the process of liver fibrosis. In addition, CHI3L1 plays a crucial role in resisting pathogens and antigen- and oxidation-induced damage responses and participates in inflammation and tissue remodeling[19]. Clinical studies have found that CHI3L1 levels are significantly elevated in liver diseases related to CHB, and its levels increase with worsening disease severity[3,20]. Furthermore, serum CHI3L1 has been recommended as a non-invasive marker for liver diseases. A meta-analysis found that serum CHI3L1 is a promising biomarker for the diagnosis of liver fibrosis, and serum CHI3L1 levels represent a feasible diagnostic tool for liver fibrosis[21].
Therefore, this study explored the correlation of CHI3L1 levels in different liver diseases patients with liver fibrosis, cirrhosis, liver cancer, and HCC recurrence after radical resection and compared serum CHI3L1 measurement with other assessment methods for liver fibrosis and cirrhosis [e.g., FIB-4, liver stiffness measurement (LSM)].
This study included patients treated for various chronic liver diseases from August 2019 to December 2024. The study population included 55 cases of viral hepatitis, 85 cases of non-viral hepatitis, 102 cases of liver cirrhosis, 138 cases of primary liver cancer, 93 cases of radical resection of liver cancer, and 26 cases of recurrent liver cancer. Additionally, 52 healthy subjects who underwent physical examinations at the Physical Examination Center of Peking University People's Hospital were selected. The patients with viral hepatitis included 46 patients with chronic HBV infection and 9 patients with hepatitis C virus (HCV) infection. Among patients with non-viral hepatitis, 85 patients had steatohepatitis.
The diagnostic criteria for CHB and liver cirrhosis were based on the “Guidelines for the Prevention and Treatment of Chronic Hepatitis B (Second Edition)”. The diagnostic criteria for HCV were based on “Diagnosis of Hepatitis C” WS 213-2018.
The inclusion criteria for patients with liver cancer[22] were as follows: (1) Pathological confirmation of HCC; (2) Patients with complete clinical and follow-up data; and (3) Informed consent of the patients. The exclusion criteria were as follows: (1) Cholangiocarcinoma or mixed HCC; (2) Severe concurrent diseases of vital organs such as heart, lung, and kidney; or (3) Use of procoagulants or anticoagulants for various reasons before sampling.
The inclusion criteria for patients who underwent radical resection of liver cancer were as follows: (1) Pathological diagnosis of HCC; (2) Age of 18-85 years; and (3) Complete surgical resection[23]. Patients meeting the following criteria were excluded: (1) HCC with distant metastasis; (2) Preoperative chemotherapy, radiotherapy, or transcatheter arterial chemoembolization; or (3) No follow-up or incomplete clinical data.
All patients underwent regular follow-up after liver cancer surgery in the outpatient department, including continuous monitoring of serum alpha-fetoprotein (AFP) levels, liver function tests, chest X-ray, and computed tomography every 3 months to check for intrahepatic recurrence or distant metastasis. Suspected tumor recurrence was confirmed by fine-needle biopsy or cytological examination[24].
This study was approved by the Ethics Committee of Peking University People's Hospital (Approval No. 2025PHB541-001), and informed consent was obtained from all patients. All methods were carried out in accordance with relevant guidelines and regulations.
All patients underwent standardized medical history assessment, laboratory assessment, anthropometric measurements, and physical examinations. Biochemical tests included platelet (PLT) counts and aspartate aminotransferase (AST) and alanine aminotransferase (ALT) levels. FIB-4 was derived using the following formula: FIB-4 = [age × AST (IU/L)]/[PLT (109/L) × ALT (IU/L)1/2]. Liver pathological diagnosis and fibrosis staging results were also collected. Serum AST and ALT levels were measured via the rate method using the AU5800 automatic biochemical analyzer (Beckman Coulter Inc., United States) using reagents provided by Beckman Coulter Inc. The PLT count was determined by the electrical impedance method using the XN 2800 automatic blood analyzer (Sysmex Corporation, Japan). All procedures were strictly conducted in accordance with the instructions of the reagent kits.
The FibroScan TE scanner (Shenzhen Echo Medical Devices Co., LTD) was used to measure liver elasticity. The detection area was from the anterior axillary line to the midaxillary line at the 7th to 9th ribs on the right side. At least 10 valid measurements were recorded for each patient. The system automatically took the median as the final LSM. A detection success rate lower than 60% or deviation greater than 30% was considered invalid. According to the liver elasticity measurement results of patients with liver fibrosis, the classification was made in accordance with the “Expert Consensus on the Clinical Application of Transient Elastography (2015 Edition)” guidelines: F0-1: < 7.5 kPa; F2: 7.5-9.8 kPa; F3: 9.8-12.5 kPa; F4: > 17.3 kPa.
All tests were strictly conducted in accordance with standard operating procedures under the conditions of normal instrument operation and passing internal quality control, with standardized operational procedures applied for measurement. The CHI3L1 detection kit from Hangzhou Puwang Biotechnology Co., Ltd. and the fully automated chemiluminescence immunoassay analyzer (Wan200+, Xiamen U-Medical Instruments Co., Ltd.) were used to measure the serum CHI3L1 levels. The samples were tested in batches by the experimenters. The CHI3L1 detection is an immunoassay that employs the principle of a double-antibody sandwich method. According to the manufacturer's instructions, we loaded the samples, reagents, and other materials related to the CHI3L1 detection into the Wan200+. The sample volume for each test was 10 µL. The instrument automatically aspirated and diluted the samples 20-fold with the accompanying sample diluent before performing the detection, and automatically provided the test results (multiplied by 20).
All statistical analyses were conducted using SPSS 27.0. Normally distributed data were presented as the mean ± SD. Otherwise, the median (interquartile range) was used for representation. Variables were compared between the groups using the Mann-Whitney U test or Kruskal-Wallis test. After adjusting for age and sex using logistic regression, the receiver operating characteristic (ROC) curve was drawn to evaluate the diagnostic effect of serum CHI3L1. We used Spearman’s correlation analysis to evaluate whether the CHI3L1 concentration was correlated with LSM and FIB-4. P < 0.05 was considered statistically significant.
The research participants included in this study were recruited from clinical cases at several reputable tertiary hospitals in Beijing. These patients originated from diverse geographical regions across China, thereby ensuring a certain degree of regional variability and population representativeness. A total of 551 patients were included for this study, included 55 cases of viral hepatitis, 85 cases of non-viral hepatitis, 102 cases of liver cirrhosis, 138 cases of liver cancer, 93 patients after radical resection of liver cancer and 26 patients with recurrence of liver cancer (Table 1). All patients underwent fiber scanning and serum CHI3L1 detection. In addition, a control group of 52 healthy subjects was enrolled. The clinical characteristics of all enrolled individuals are presented in Table 1. The study population included significantly more men than women (P < 0.001). The HCC recurrence group had the oldest mean age among all groups, with an average age of 64 years. Our study statistics demonstrated that the positive rate of CHI3L1 exhibited a marked upward trend with the progression of liver lesions. Specifically, the positive rate was merely 0.91% (5/551) in healthy individuals; patients with chronic viral hepatitis (1.81% for hepatitis B and 1.27% for hepatitis C) and fatty liver disease presented with relatively low positive rates (3.45%); in contrast, the positive rates increased dramatically in patients with liver cirrhosis and HCC, reaching 14.88% (82/551) and 17.42% (96/551), respectively.
| Group | n | Gender (male/female) | Age (year; median) |
| Health control | 52 | 1.36:1 (30/22) | 39.0 |
| Virus hepatitis | 55 | 1:0.90 (26/29) | 49.0 |
| Non-virus hepatitis | 85 | 1.83:1 (55/30) | 43.0 |
| Liver cirrhosis | 102 | 2.52:1 (73/29) | 43.5 |
| HCC | 138 | 4.52:1 (113/25) | 61.0 |
| HCC patients after radical resection | 93 | 2.88:1 (69/24) | 62.0 |
| HCC recurrence | 26 | 3.33:1 (20/6) | 64.0 |
| χ2 | 29.265 | ||
| P value | < 0.001 |
Serum CHI3L1 levels in patients with different liver diseases and healthy controls are presented in Table 2. Serum CHI3L1 levels were significantly higher in the cirrhosis, HCC, post-radical hepatectomy for HCC, and HCC recurrence groups than in the healthy control group (P < 0.001, P < 0.001, P < 0.05, P < 0.001, respectively). Serum CHI3L1 levels were significantly higher in the cirrhosis group than in the viral hepatitis and non-viral hepatitis groups (P < 0.01, P < 0.001, respectively). Serum CHI3L1 levels were significantly higher in the HCC group than in the post-radical hepatectomy for HCC group (P < 0.001). The CHI3L1 levels in the healthy control group were significantly different from those in all hepatitis groups (P < 0.05). There were no significant differences in serum CHI3L1 levels between the viral hepatitis and healthy control groups, between the non-viral hepatitis and the healthy control groups, between the HCC and cirrhosis groups, or between the HCC recurrence and post-radical hepatectomy for HCC groups (all P > 0.05).
| Group | Group serum CHI3L1 level (ng/mL) | ||
| 25th percentile | 50th percentile | 75th percentile | |
| Health control | 36.79 | 40.99 | 51.47 |
| Virus hepatitis | 36.81 | 58.91 | 85.39 |
| Non-virus hepatitis | 34.50 | 50.89 | 77.34 |
| Liver cirrhosis | 61.58 | 100.13 | 161.38a,b,c |
| HCC | 67.38 | 141.07 | 271.77a,b,c |
| HCC patients after radical resection | 32.77 | 64.27 | 130.78d,e |
| HCC recurrence | 35.09 | 156.00 | 554.6a,b,c |
The hepatitis group was compared with the healthy control group, and ROC curve analysis was used to assess the diagnostic effectiveness of changes in serum CHI3L1 levels in hepatitis patients. The receiver operating characteristic curve (AUC) was 0.606. At a cutoff value of 50.49 ng/mL, the sensitivity was 55.7% and the specificity was 75.0% (Figure 1A).
In comparison with the healthy control group, the AUC of serum CHI3L1 for detecting cirrhosis was 0.874. At a cutoff of 57.45 ng/mL, the sensitivity was 80.4%, and the specificity was 82.7% (Figure 1B). For FIB-4 in the diagnosis of liver cirrhosis, the AUC was 0.833; with an optimal cutoff of 1.12, the sensitivity and specificity were 73.5% and 84.6%, respectively. The sensitivity and specificity of the combined predictors were 88.2% and 80.8%, and the AUC was 0.912 (Figure 1B).
ROC curve analysis was used to assess the diagnostic effectiveness of changes in serum CHI3L1 levels for diagnosing HCC in comparison with the healthy control group. The AUC was 0.858. At a cutoff of 65.171 ng/mL, the sensitivity was 77.5% and the specificity was 88.5% (Figure 1C).
ROC curve analysis was used to evaluate the diagnostic effectiveness of changes in serum CHI3L1 levels for diagnosing liver cancer recurrence in comparison with the heathy control group. The AUC was 0.723. At a cutoff of 95.818 ng/mL, the sensitivity was 65.4% and the specificity was 62% (Figure 1D).
ROC curve analysis was used to assess the diagnostic effectiveness of changes in serum CHI3L1 levels for HCC and liver cirrhosis patients. The AUC was 0.602. At a cutoff of 140.755 ng/mL, the sensitivity was 50.7% and the specificity was 72.5% (Figure 1E).
To evaluate the diagnostic significance of CHI3L1 and AFP levels in HCC, we screened 138 subjects in the HCC and control groups who underwent measurements of both AFP and CHI3L1 levels. As illustrated in Figure 1F, an AUC of CHI3L1 was 0.858; the sensitivity and specificity were 77.5% and 88.5% when the optimal cutoff was 65.171 ng/mL. For the diagnosis of HCC, the AUC of AFP was 0.676; the sensitivity and specificity were 44.2% and 98.1% when the optimal cutoff was 6.62 ng/mL. The sensitivity of the combined predictors reached 84.1%, and the AUC was 0.895 (Figure 1F).
The potential of CHI3L1 as a biomarker for liver fibrosis and cirrhosis in liver diseases patients was assessed in comparison with LSM and FIB-4. Spearman’s correlation analysis revealed that CHI3L1 levels were positively correlated with LSM (r = 0.424, P < 0.001) and FIB-4 (r = 0.370, P < 0.001). Meanwhile, LSM was positively correlated with FIB-4 (r = 0.319, P = 0.004).
Patients with liver fibrosis were classified by liver elasticity as follows: F0-1, < 7.5 kPa; F2, 7.5-9.8 kPa; F3, 9.8-12.5 kPa; and F4, > 17.3 kPa. In total, 5, 13, 25, and 36 patients were categorized into grades F0-1, F2, F3, and F4, respectively. The statistical analysis results revealed a statistically significant difference in CHI3L1 levels between grades F3 and F4 (P < 0.01).
In the clinical treatment and prognosis assessment of chronic liver diseases, timely, accurate, and comprehensive diagnosis is of vital significance. This study established serum CHI3L1 as an innovative biomarker for the full-cycle management of chronic liver diseases. CHI3L1 levels exhibited a stepwise increase during progression of chronic hepatitis, liver cirrhosis, and HCC but significantly decreased after radical resection of HCC. In addition, CHI3L1 displayed excellent diagnostic efficacy for liver cirrhosis and liver cancer, and it was positively correlated with liver fibrosis indicators (LSM, FIB-4). This “one standard throughout the entire process” feature, covering the entire process from early screening to non-invasive diagnosis and postoperative dynamic monitoring, provides an efficient and consistent individualized management tool for clinical practice, significantly enhancing the precision of liver disease diagnosis and treatment.
Our research found that CHI3L1 levels increased successively from chronic hepatitis to liver cirrhosis and then HCC, suggesting that CHI3L1 might have clinical value in the assessment of different stages of chronic liver diseases and serve as an indicator of disease evolution. This finding is consistent with previous research results[25]. Additionally, in our study, no significant difference was observed between the viral hepatitis group and the healthy control group. However, the CHI3L1 levels in the healthy control group showed significant differences compared to all hepatitis groups (P < 0.05). This finding is consistent with the results of previous studies by Johansen and colleagues, who found that serum CHI3L1 levels were higher in patients with chronic hepatitis than in normal controls[17]. This finding suggests that the impact of hepatitis etiology in liver diseases on the potential biomarker application of CHI3L1 should be considered in clinical practice. Our study demonstrated that serum CHI3L1 levels significantly decrease after radical resection of HCC vs the preoperative levels, suggesting that successful surgical removal of tumor tissue effectively reduces the release of this biomarker. Zhu et al[24] reported that serum CHI3L1 represents an independent prognostic factor for overall survival and recurrence-free survival in patients undergoing radical resection of liver cancer. During follow-up after curative resection, patients with HCC and higher serum CHI3L1 levels within 6 months postoperatively exhibited significantly worse overall survival. Collectively, this evidence suggests that continuous monitoring of dynamic changes in serum CHI3L1 after radical surgery can provide critical information for early warning of tumor recurrence and optimization of follow-up strategies.
To investigate the diagnostic value of serum CHI3L1 for liver cirrhosis, we compared serum CHI3L1 levels between the liver cirrhosis and healthy control groups using ROC curve analysis. The AUC of serum CHI3L1 for diagnosing liver cirrhosis reached 0.874 with 80.4% sensitivity and 82.7% specificity, suggesting the potential of CHI3L1 as an effective non-invasive diagnostic marker. By contrast, Qiu and Zhang[25] reported an AUC of 0.782 (95%CI: 0.721-0.844) along with sensitivity of 64.4% and specificity of 96.7% at a cutoff of 63 ng/mL. The AUC and sensitivity in this study were significantly higher than those reported in the literature, further confirming that serum CHI3L1 possesses superior potential for liver cirrhosis screening. Higher sensitivity is particularly crucial for early screening of liver cirrhosis, and this study achieved significantly improved sensitivity while maintaining good specificity (> 80%), demonstrating clear clinical value. When CHI3L1 was combined with FIB-4 for joint detection, the AUC increased to 0.912 and the sensitivity reached 88.2%. These findings indicated that the combined detection of serum CHI3L1 and FIB-4 could serve as an effective biomarker panel for the diagnosis of liver cirrhosis. In addition, in a systematic meta-analysis that included 11 published research papers and involved 1897 patients over 18 years old with liver cirrhosis from four countries, the overall diagnostic performance of CHI3L1 for liver cirrhosis showed that its sensitivity and specificity were 0.72 and 0.74, respectively and AUC was 0.85[21]. In contrast, the results of this study conducted in the Chinese population show that the diagnostic sensitivity and specificity of CHI3L1 are significantly higher than those of the above meta-analysis, and the AUC value is also better, further suggesting that CHI3L1 may have a higher diagnostic value for liver cirrhosis in the Chinese population.
Recent studies have revealed that HBV integration-driven genomic instability serves as a key driver in the clonal evolution of HCC[26]. Our observation of elevated CHI3L1 levels in patients with postoperative recurrence suggests that the protein can act as a microenvironment factor secreted by tumor cells or related macrophages. CHI3L1 is a key oncogenic molecule implicated in the progression of HCC. Through multidimensional and multi-pathway cross-regulation, it plays a critical role in the initiation, proliferation, invasion, and metastasis of HCC. CHI3L1 interacts with various cell surface receptors, including IL-13Rα2, αvβ3/αvβ5, galectin-3, and CD44v3, leading to the activation of multiple signaling pathways such as MAPK (p42/p44 and p38), NF-κB, and PI3K/Akt[27]. These pathways collectively regulate cellular processes including survival, proliferation, migration, inflammation, fibrosis, and tumorigenesis. Furthermore, CHI3L1 can enhance its oncogenic potential by promoting SMAD2/3 phosphorylation through kinase activation[28]. CHI3L1 activates fibroblasts to increase extracellular matrix protein production and facilitate tissue remodeling[29]. It also recruits macrophages and induces their polarization toward the M2 phenotype, thereby contributing to tumor proliferation, immunosuppression, and angiogenesis[30]. In addition, CHI3L1 directly stimulates angiogenesis and maintains vascular integrity, supporting tumor growth and metastatic dissemination[29]. Evidence indicates that CHI3L1 enhances the production of inflammatory cytokines via an interaction with RAGE, thereby exacerbating chronic inflammation and fostering the development of a pro-tumorigenic microenvironment[31]. By modulating the cell cycle, CHI3L1 promotes HCC proliferation by increasing the proportion of cells in S-phase[28]. As a pro-fibrotic mediator, it is highly expressed in liver cirrhosis, and it acts on HSCs to drive fibrogenesis, linking the protein to both liver fibrosis and HCC pathogenesis[3].
To explore the applicability of serum CHI3L1 in the diagnosis of HCC, a ROC curve was constructed. CHI3L1 exhibited strong specificity for HCC, reaching 88.5%, suggesting that serum CHI3L1 has certain diagnostic efficacy for patients with HCC. Liu et al[22] also analyzed the diagnostic value of multiple liver cancer markers, and CHI3L1 demonstrated the highest specificity at 86%, consistent with our research data. Furthermore, according to ROC curve analysis, serum AFP demonstrated superior performance at a cutoff of 6.62 ng/mL (AUC = 0.676). When combining CHI3L1 with AFP for detection, the AUC increased to 0.890 with 84.1% sensitivity, indicating that the combined detection of serum CHI3L1 and AFP represents an efficient biomarker for diagnosing HCC. Additionally, serum CHI3L1 serves as a promising and valuable biomarker for the prognosis of patients after HCC resection[23]. In our study, ROC curve analysis of serum CHI3L1 for diagnosing HCC recurrence revealed 62% specificity. AFP has long been recognized as an important biomarker for HCC prognosis evaluation, and it has been extensively studied and applied in the medical field[23]. However, studies have found that AFP levels are elevated in approximately 10% of patients with early-stage HCC, highlighting the limitations of relying solely on AFP as a prognostic indicator[32]. Furthermore, a significant proportion of patients with HCC (38.1%-39.4%) yield negative AFP test results. For this subset of patients, AFP cannot provide effective diagnostic assistance[33,34]. In light of this, researchers have proposed CHI3L1 as a novel potential biomarker that has been demonstrated to represent an effective prognostic marker in cases in which AFP is not useful, thereby compensating for the limitations of AFP in HCC prognosis evaluation. Surgical resection, as the most effective treatment with curative potential for HCC, produces significant therapeutic outcomes. However, the overall prognosis of patients with HCC remains unsatisfactory, primarily because of the high postoperative recurrence and metastasis rates. Monitoring serum CHI3L1 levels in patients after radical resection might provide crucial information for prognostic assessment[23]. Although CHI3L1 demonstrated moderate diagnostic performance in predicting HCC recurrence (AUC = 0.723), its advantages include its non-invasiveness and low cost. Although CHI3L1 might not be suitable as a standalone screening tool, it could potentially serve as an effective supplement to existing monitoring methods (such as imaging) for identifying high-risk patients requiring closer follow-up.
Liver fibrosis, as a key predictor of chronic liver disease progression and clinical outcomes, holds critical significance for the clinical management and prognostic evaluation of chronic liver diseases. This study revealed a positive correlation between CHI3L1 levels and liver fibrosis indicators (LSM and FIB-4) in patients with liver fibrosis, with the strongest correlation observed with LSM. This finding is consistent with previous research conclusions[20,35,36]. The moderate correlations of CHI3L1 with LSM and FIB-4 aligned with our expectations, suggesting an association between CHI3L1 and liver fibrosis. LSM and FIB-4 primarily reflect the degree of liver fibrosis, whereas CHI3L1, as an inflammatory and immune regulatory factor, might be influenced by fibrosis as well as inflammatory activity, immune responses, and other pathological processes. Therefore, CHI3L1 provides additional biological information beyond mere fibrosis assessment, and it can serve as a “supplementary indicator” for fibrosis. Although histopathological evaluation is considered the gold standard for diagnosing liver fibrosis and it can provide critical evidence for definitive diagnosis, as an invasive procedure, liver puncture biopsy carries risks of postoperative complications, including pain, bleeding, infection, and even death. Additionally, liver biopsy has inherent limitations in terms of sampling scope and interobserver variability, which restrict its ability to accurately diagnose and periodically assess the degree of liver fibrosis[15]. In recent years, a series of non-invasive imaging techniques, such as TE and magnetic resonance elastography (MRE), have been widely adopted as testing methods for LSM. It is well known that factors such as hepatic congestion, hyperbilirubinemia, elevated transaminase levels, and prolonged prothrombin time might all influence the detection results of TE[36]. Although MRE demonstrates better overall efficacy than TE, no diagnostic threshold guidelines incorporating liver stiffness values from MRE have been established to date for liver fibrosis. Furthermore, the diagnostic value of serological markers such as FIB-4 and the AST-to-PLT ratio index (APRI) in assessing intermediate stages of liver fibrosis (particularly fibrosis caused by other etiologies) remains inadequately defined[37]. This study reveals the correlation between serum CHI3L1 levels and liver fibrosis assessment indicators LSM and FIB-4. Jiang and colleagues also observed that CHI3L1 surpasses other non-invasive diagnostic indicators, including LSM, FIB-4, and APRI, in diagnosing significant fibrosis, demonstrating high specificity and sensitivity[23]. These findings suggest that serum CHI3L1 levels might provide a potential, effective non-invasive means for monitoring fibrosis progression, potentially compensating for the limitations of LSM and FIB-4. Moreover, the application prospects of CHI3L1 in diagnosing and staging hepatitis B-related liver fibrosis are highly promising. Our study recorded statistically significant differences in CHI3L1 levels between the F3 and F4 groups. In a recent study conducted by Jin et al[36] in China, patients with CHB and significant liver fibrosis exhibited significantly higher serum CHI3L1 levels than those without significant fibrosis; Huang et al[19] also observed higher CHI3L1 levels in subjects with higher histological grades of liver fibrosis. These observations align closely with the results of the present study. Beyond serving as a non-invasive biomarker for assessing liver fibrosis in CHB patients prior to treatment, Wang et al[35] found that CHI3L1 is a potentially useful marker for monitoring fibrosis changes during therapy. Therefore, CHI3L1 might be valuable for the diagnosis and staging of liver fibrosis, and it can help guide treatment strategies for patients with CHB.
This study had several limitations. This study was conducted at a single center, which inherently imposes limitations on the representativeness of the study population. The geographical concentration and clinical homogeneity of the enrolled participants may have restricted the inclusion of demographically and clinically diverse patient subgroups, thereby constraining the generalizability of the findings. Furthermore, the standardized diagnostic and therapeutic protocols adopted by a single institution fail to account for interinstitutional variations in clinical practice, which could compromise the consistency of assessments and the robustness of the results. To validate the core findings of this study, a prospective multi-center cohort study is therefore planned. This initiative will involve collaboration with three to four tertiary grade-A hospitals across distinct geographical regions, encompassing patients with chronic hepatitis, liver cirrhosis, newly diagnosed HCC, as well as those undergoing postoperative follow-up. Standardized protocols for serum sample collection and laboratory testing will be uniformly implemented, and CHI3L1 levels will be dynamically monitored throughout the entire study period. By integrating longitudinal clinical outcome data, the diagnostic and prognostic value of CHI3L1 will be further rigorously evaluated, with the aim of enhancing the universality and reliability of the conclusions. Additionally, although LSM holds significant clinical value in diagnosing liver fibrosis, its results can be influenced by factors such as hepatic congestion, cholestasis, and elevated transaminase levels. These potential confounding factors also apply to serum CHI3L1, and they potentially affected the conclusions of this study. Therefore, future research should focus on conducting multi-center prospective studies with expanded sample sizes to further validate the diagnostic efficacy of CHI3L1 in liver fibrosis while establishing diagnostic thresholds for different fibrosis stages. Identifying factors influencing CHI3L1 levels is also crucial for diagnosing and assessing the degree of liver fibrosis. Future studies should extend the research population to other liver diseases, including alcoholic liver disease and metabolic dysfunction-associated fatty liver disease, to further explore the diagnostic applicability of CHI3L1.
This study revealed the potential of serum CHI3L1 levels as a promising biomarker for liver diseases. In patients with chronic hepatitis, serum CHI3L1 levels increased with disease progression from cirrhosis to HCC while declining after radical hepatectomy. Furthermore, CHI3L1 demonstrated feasibility in disease progression monitoring, serving as an effective predictive indicator for assessing the progression of chronic liver diseases and predicting the prognosis of patients with liver cancer. We also revealed that serum CHI3L1 levels could represent a novel and effective serological biomarker for diagnosing fibrosis, with CHI3L1 levels displaying positive correlations with the liver fibrosis indicators LSM and FIB-4, particularly LSM.
We thank the patients included in the study.
| 1. | Huang C, Lu Y, Wang Z, Jiang Q, Dong Y, Cao L, Yan J, Xu Z, Wang F, Gao Y, Fu J, Zhang M, Wang FS. Correlation Between Clinical Indicators and Liver Pathology in Children with Chronic Hepatitis B. Biomedicines. 2024;12:2903. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 2. | Trépo C, Chan HL, Lok A. Hepatitis B virus infection. Lancet. 2014;384:2053-2063. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1369] [Cited by in RCA: 1208] [Article Influence: 100.7] [Reference Citation Analysis (5)] |
| 3. | Wang S, Hu M, Qian Y, Jiang Z, Shen L, Fu L, Hu Y. CHI3L1 in the pathophysiology and diagnosis of liver diseases. Biomed Pharmacother. 2020;131:110680. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 42] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 4. | Addolorato G, Abenavoli L, Dallio M, Federico A, Germani G, Gitto S, Leandro G, Loguercio C, Marra F, Stasi E. Alcohol associated liver disease 2020: A clinical practice guideline by the Italian Association for the Study of the Liver (AISF). Dig Liver Dis. 2020;52:374-391. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 26] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 5. | Fattovich G, Stroffolini T, Zagni I, Donato F. Hepatocellular carcinoma in cirrhosis: incidence and risk factors. Gastroenterology. 2004;127:S35-S50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1979] [Cited by in RCA: 1789] [Article Influence: 81.3] [Reference Citation Analysis (7)] |
| 6. | Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76817] [Cited by in RCA: 69607] [Article Influence: 13921.4] [Reference Citation Analysis (45)] |
| 7. | Li C, Li R, Zhang W. Progress in non-invasive detection of liver fibrosis. Cancer Biol Med. 2018;15:124-136. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 82] [Cited by in RCA: 79] [Article Influence: 9.9] [Reference Citation Analysis (1)] |
| 8. | Liu S, Peng C, Xia S, Li C, Dai X, Liu X, Zhang M, Li X, Tang L. Chitinase 3-like protein 1: a diagnostic biomarker for early liver fibrosis in autoimmune liver diseases. Front Immunol. 2025;16:1504066. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 9. | Sripongpun P, Pongpaibul A, Charatcharoenwitthaya P. Value and risk of percutaneous liver biopsy in patients with cirrhosis and clinical suspicion of autoimmune hepatitis. BMJ Open Gastroenterol. 2021;8:e000701. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (2)] |
| 10. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines on non-invasive tests for evaluation of liver disease severity and prognosis - 2021 update. J Hepatol. 2021;75:659-689. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1546] [Cited by in RCA: 1383] [Article Influence: 276.6] [Reference Citation Analysis (6)] |
| 11. | Poynard T, Lassailly G, Diaz E, Clement K, Caïazzo R, Tordjman J, Munteanu M, Perazzo H, Demol B, Callafe R, Pattou F, Charlotte F, Bedossa P, Mathurin P, Ratziu V; FLIP consortium. Performance of biomarkers FibroTest, ActiTest, SteatoTest, and NashTest in patients with severe obesity: meta analysis of individual patient data. PLoS One. 2012;7:e30325. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 118] [Cited by in RCA: 103] [Article Influence: 7.4] [Reference Citation Analysis (1)] |
| 12. | Liao Y, Peng S, Huang L, Li Z, Hu J, Xu R, Tang W, Zhuang J. Analytical and Clinical Evaluation of a Chemiluminescent Immunoassay to Detect Serum Chitinase-3-like Protein 1 in HBV-Related Liver Diseases. Int J Anal Chem. 2024;2024:6688819. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 13. | Forner A, Reig M, Bruix J. Hepatocellular carcinoma. Lancet. 2018;391:1301-1314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4664] [Cited by in RCA: 4427] [Article Influence: 553.4] [Reference Citation Analysis (8)] |
| 14. | Higashiyama M, Tomita K, Sugihara N, Nakashima H, Furuhashi H, Nishikawa M, Inaba K, Wada A, Horiuchi K, Hanawa Y, Shibuya N, Kurihara C, Okada Y, Nishii S, Mizoguchi A, Hozumi H, Watanabe C, Komoto S, Yamamoto J, Seki S, Miura S, Hokari R. Chitinase 3-like 1 deficiency ameliorates liver fibrosis by promoting hepatic macrophage apoptosis. Hepatol Res. 2019;49:1316-1328. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 37] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 15. | Hu X, Liu W, Liu J, Wang B, Qin X. Research advances in serum chitinase-3-like protein 1 in liver fibrosis. Front Med (Lausanne). 2024;11:1372434. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 16. | Bao J, Ouyang Y, Qiao L, He J, Liu F, Wang Y, Miao L, Fu A, Lou Z, Zang Q, Huang W, Huang J, Li Z. Serum CHI3L1 as a Biomarker for Non-invasive Diagnosis of Liver Fibrosis. Discov Med. 2022;33:41-49. [PubMed] |
| 17. | Johansen JS, Christoffersen P, Møller S, Price PA, Henriksen JH, Garbarsch C, Bendtsen F. Serum YKL-40 is increased in patients with hepatic fibrosis. J Hepatol. 2000;32:911-920. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 209] [Cited by in RCA: 198] [Article Influence: 7.6] [Reference Citation Analysis (1)] |
| 18. | Nishimura N, De Battista D, McGivern DR, Engle RE, Tice A, Fares-Gusmao R, Kabat J, Pomerenke A, Nguyen H, Sato S, Bock KW, Moore IN, Kleiner DE, Zamboni F, Alter HJ, Govindarajan S, Farci P. Chitinase 3-like 1 is a profibrogenic factor overexpressed in the aging liver and in patients with liver cirrhosis. Proc Natl Acad Sci U S A. 2021;118:e2019633118. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (1)] |
| 19. | Huang H, Wu T, Mao J, Fang Y, Zhang J, Wu L, Zheng S, Lin B, Pan H. CHI3L1 Is a Liver-Enriched, Noninvasive Biomarker That Can Be Used to Stage and Diagnose Substantial Hepatic Fibrosis. OMICS. 2015;19:339-345. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 54] [Article Influence: 4.9] [Reference Citation Analysis (1)] |
| 20. | Jiang Z, Wang S, Jin J, Ying S, Chen Z, Zhu D, Xiao B, Hu Y, Qian Y, Cai T, Fu L. The clinical significance of serum chitinase 3-like 1 in hepatitis B-related chronic liver diseases. J Clin Lab Anal. 2020;34:e23200. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (2)] |
| 21. | Huang X, Zhuang J, Yang Y, Jian J, Ai W, Liu C, Tang W, Jiang C, He Y, Huang L, Peng S. Diagnostic Value of Serum Chitinase-3-Like Protein 1 for Liver Fibrosis: A Meta-analysis. Biomed Res Int. 2022;2022:3227957. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 22. | Liu M, Qiu Y, Xie E, Qian P, Yang S, Zhao S, Yan W, Huang X, Han S. Development of a chitosanase 3-like protein 1 assay kit and study of its application in patients with hepatocellular carcinoma. BMC Biotechnol. 2025;25:35. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 23. | Jiang Y, Gong W, Liu Y, Zhou Z, Liang X, Lin Q, Qiu M, Lin B, Qiu X, Yu H. Serum CHI3L1 Levels Predict Overall Survival of Hepatocellular Carcinoma Patients after Hepatectomy. J Cancer. 2024;15:6315-6325. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 24. | Zhu CB, Chen LL, Tian JJ, Su L, Wang C, Gai ZT, Du WJ, Ma GL. Elevated serum YKL-40 level predicts poor prognosis in hepatocellular carcinoma after surgery. Ann Surg Oncol. 2012;19:817-825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 33] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 25. | Qiu H, Zhang X. The Value of Serum CHI3L1 for the Diagnosis of Chronic Liver Diseases. Int J Gen Med. 2022;15:5835-5841. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 26. | Chen L, Li W, Zai W, Zheng X, Meng X, Yao Q, Li W, Liang Y, Ye M, Zhou K, Liu M, Yang Z, Mao Z, Wei H, Yang S, Shi G, Yuan Z, Yu W. HBV sequence integrated to enhancer acting as oncogenic driver epigenetically promotes hepatocellular carcinoma development. J Exp Clin Cancer Res. 2025;44:155. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 27. | Mizoguchi E, Wang S. Future Perspectives and Conclusions from Animal Models of CHI3L1-Related Inflammation-Associated Cancer. Cells. 2025;14:982. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 28. | Qiu QC, Wang L, Jin SS, Liu GF, Liu J, Ma L, Mao RF, Ma YY, Zhao N, Chen M, Lin BY. CHI3L1 promotes tumor progression by activating TGF-β signaling pathway in hepatocellular carcinoma. Sci Rep. 2018;8:15029. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 70] [Article Influence: 8.8] [Reference Citation Analysis (1)] |
| 29. | Chang MC, Chen CT, Chiang PF, Chiang YC. The Role of Chitinase-3-like Protein-1 (YKL40) in the Therapy of Cancer and Other Chronic-Inflammation-Related Diseases. Pharmaceuticals (Basel). 2024;17:307. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 30] [Article Influence: 15.0] [Reference Citation Analysis (1)] |
| 30. | Zhou Y, He CH, Yang DS, Nguyen T, Cao Y, Kamle S, Lee CM, Gochuico BR, Gahl WA, Shea BS, Lee CG, Elias JA. Galectin-3 Interacts with the CHI3L1 Axis and Contributes to Hermansky-Pudlak Syndrome Lung Disease. J Immunol. 2018;200:2140-2153. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 54] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 31. | He CH, Lee CG, Ma B, Kamle S, Choi AMK, Elias JA. N-Glycosylation Regulates Chitinase 3-like-1 and IL-13 Ligand Binding to IL-13 Receptor α2. Am J Respir Cell Mol Biol. 2020;63:386-395. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 28] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 32. | Singal AG, Hoshida Y, Pinato DJ, Marrero J, Nault JC, Paradis V, Tayob N, Sherman M, Lim YS, Feng Z, Lok AS, Rinaudo JA, Srivastava S, Llovet JM, Villanueva A. International Liver Cancer Association (ILCA) White Paper on Biomarker Development for Hepatocellular Carcinoma. Gastroenterology. 2021;160:2572-2584. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 147] [Cited by in RCA: 137] [Article Influence: 27.4] [Reference Citation Analysis (1)] |
| 33. | Zhang B, Zhang B, Zhang Z, Huang Z, Chen Y, Chen M, Bie P, Peng B, Wu L, Wang Z, Li B, Fan J, Qin L, Chen P, Liu J, Tang Z, Niu J, Yin X, Li D, He S, Jiang B, Mao Y, Zhou W, Chen X. 42,573 cases of hepatectomy in China: a multicenter retrospective investigation. Sci China Life Sci. 2018;61:660-670. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 63] [Article Influence: 7.9] [Reference Citation Analysis (1)] |
| 34. | Chan MY, She WH, Dai WC, Tsang SHY, Chok KSH, Chan ACY, Fung J, Lo CM, Cheung TT. Prognostic value of preoperative alpha-fetoprotein (AFP) level in patients receiving curative hepatectomy- an analysis of 1,182 patients in Hong Kong. Transl Gastroenterol Hepatol. 2019;4:52. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 46] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 35. | Wang L, Liu T, Zhou J, You H, Jia J. Changes in serum chitinase 3-like 1 levels correlate with changes in liver fibrosis measured by two established quantitative methods in chronic hepatitis B patients following antiviral therapy. Hepatol Res. 2018;48:E283-E290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 36. | Jin X, Fu B, Wu ZJ, Zheng XQ, Hu JH, Jin LF, Tang LL. Serum chitinase-3-like protein 1 is a biomarker of liver fibrosis in patients with chronic hepatitis B in China. Hepatobiliary Pancreat Dis Int. 2020;19:384-389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 37. | Dong XQ, Wu Z, Zhao H, Wang GQ; China HepB-Related Fibrosis Assessment Research Group. Evaluation and comparison of thirty noninvasive models for diagnosing liver fibrosis in chinese hepatitis B patients. J Viral Hepat. 2019;26:297-307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 34] [Article Influence: 4.9] [Reference Citation Analysis (1)] |