Published online Apr 15, 2026. doi: 10.4251/wjgo.v18.i4.116955
Revised: December 16, 2025
Accepted: February 28, 2026
Published online: April 15, 2026
Processing time: 130 Days and 19.7 Hours
Hepatocellular carcinoma (HCC) is the most common primary liver malignancy. It typically develops in patients with advanced chronic liver disease and only rarely occurs during pregnancy.
We report the case of a 46-year-old woman with HBeAg-negative chronic hepatitis B virus (HBV) infection, persistently low HBV DNA levels, and normal aminotransferase values, who developed HCC in a non-cirrhotic liver during a pregnancy achieved through assisted reproduction techniques. Antiviral therapy with Entecavir was initiated shortly before conception because of an increase in HBV DNA levels and was subsequently switched to Tenofovir during pregnancy. Despite a spontaneous miscarriage, alpha-fetoprotein levels continued to rise, prompting magnetic resonance imaging, which revealed a solitary hepatic nodule measuring 18 mm. The patient underwent hepatic resection, and histological examination confirmed a moderately differentiated HCC. Despite complete surgical resection, two early recurrences occurred within 15 months. Tumour and non-tumour liver tissues were collected intra-operatively, snap-frozen, and processed for molecular analyses to assess total HBV DNA, covalently closed circular DNA and HBV RNA levels.
HBV infection and pregnancy-related immunological imbalance may contribute to HCC development, under
Core Tip: Hepatocellular carcinoma is rare during pregnancy and is associated with adverse outcomes for both the mother and the foetus. We report the case of a 46-year-old woman with HBeAg-negative chronic hepatitis B infection (HBV) who developed recurrent hepatocellular carcinoma after a pregnancy achieved through medically assisted reproduction. After surgical resection, immunofluorescence analysis of tumour samples showed no expression of oestrogen receptors, while molecular analyses performed on tumour and non-tumour tissues revealed high levels of total HBV DNA, HBV covalently closed circular DNA and HBV RNA, suggesting a virus-host interaction that may account for the aggressive behaviour of the neoplasm.
- Citation: Asero C, Ciappina G, Franzè MS, Maltese T, Gruttadauria S, Pagano D, Lombardo D, Pitrone A, Musolino C, Berretta M, Cacciola I. Uncommon hepatocellular carcinoma in a non-cirrhotic woman with chronic hepatitis B virus infection after medical assisted reproduction: A case report. World J Gastrointest Oncol 2026; 18(4): 116955
- URL: https://www.wjgnet.com/1948-5204/full/v18/i4/116955.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i4.116955
Hepatocellular carcinoma (HCC) is the most prevalent form of primary liver malignancy and ranks as the sixth most frequently diagnosed cancer and the third leading cause of cancer-related death worldwide[1]. Viral hepatitis remains one of the major risk factors for HCC, particularly chronic infections with hepatitis B virus (HBV)[2], which can promote hepatocarcinogenesis through multiple mechanisms, even in the absence of cirrhosis[3]. HCC diagnosed during pregnancy is a rare clinical condition[4], and is associated with poor outcomes for both the mother and the foetus. Pregnancy represents a unique immunological and hormonal state, and induces the expression of oncofoetal genes normally silenced in adult tissues during embryonic development, potentially influencing tumour initiation and behaviour. Over recent decades, multiple therapeutic strategies for HCC have been developed[5], with treatment selection depending on disease stage, tumour burden and underlying liver function, all of which influence treatment tolerance. Available options include surgical resection, liver transplantation, locoregional therapies such as radiofrequency ablation and transarterial chemoembolization, and systemic treatments including targeted agents and immunotherapy[6].
Pregnancy significantly complicates therapeutic decision-making, limiting the feasibility of several treatment modalities and influencing the choice and timing of intervention[7]. Although no standardized treatment strategy exists for HCC during pregnancy, surgical resection appears to be the most reasonable option based on the available evidence. Radiofrequency ablation may represent a potential alternative; however, no cases have been reported to date. Therapeutic decisions in pregnant patients are further guided by gestational age, particularly foetal lung maturation, which determines whether pregnancy termination or delivery prior to oncological treatment is appropriate. Specifically, early diagnosis may prompt pregnancy termination, whereas later diagnoses may allow for treatment following induced delivery, preferably via vaginal labour[7].
Current literature suggests that chronic HBV infection is associated with an increased risk of infertility, leading a growing number of patients to pursue assisted reproduction techniques (ART) to achieve pregnancy[8]. However, the impact of ART on liver disease progression and HCC onset remains unclear[9]. Here, we report a rare case of HCC diagnosed in a patient with HBeAg-negative chronic HBV following a pregnancy achieved through ART.
A 46-year-old Caucasian woman with HBeAg-negative chronic hepatitis B infection and no clinical signs of liver disease was referred to the Liver Unit of the University Hospital of Messina for specialist consultation after serum testing revealed persistently elevated alpha-fetoprotein (AFP) levels following miscarriage.
The patient had a long-standing chronic HBV infection, previously classified as “inactive carrier” due to persistently low HBV DNA levels and consistently normal aminotransferase values. During a pregnancy induced by ART after twelve months of hormonal stimulation, she developed a highly aggressive HCC in a non-cirrhotic liver.
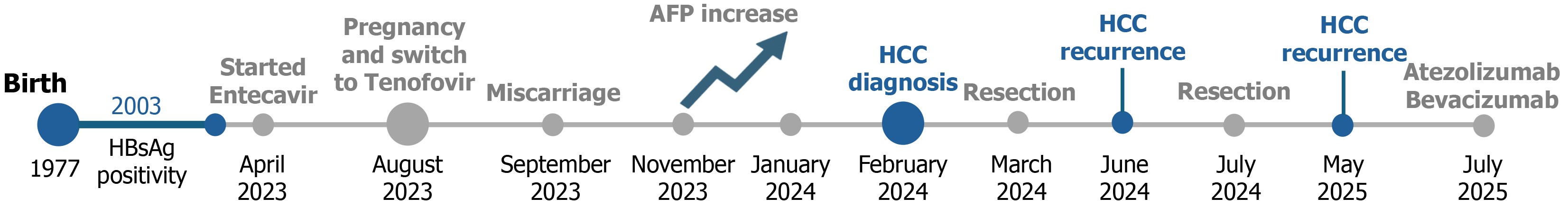
Patient personal history began at age 26, when HBV infection was diagnosed during routine screening, in the absence of any signs of liver disease, as indicated in Figure 1. In 2013, the patient underwent abdominal surgery for ovarian cyst removal and was diagnosed with polycystic ovarian syndrome; in 2021, she underwent myomectomy. The patient had been followed for chronic HBV infection at another centre from 2003 to 2023, undergoing regular laboratory testing and imaging surveillance. During follow-up, HBV DNA levels consistently remained below 2000 IU/mL, and aminotransferase values were within the upper normal limit. The most recent elastography, performed in September 2022, showed liver stiffness values of 3.3 kPa. Between 2013 and 2022, the only treatment received was hormonal therapy with dienogest 2 mg for polycystic ovarian syndrome.
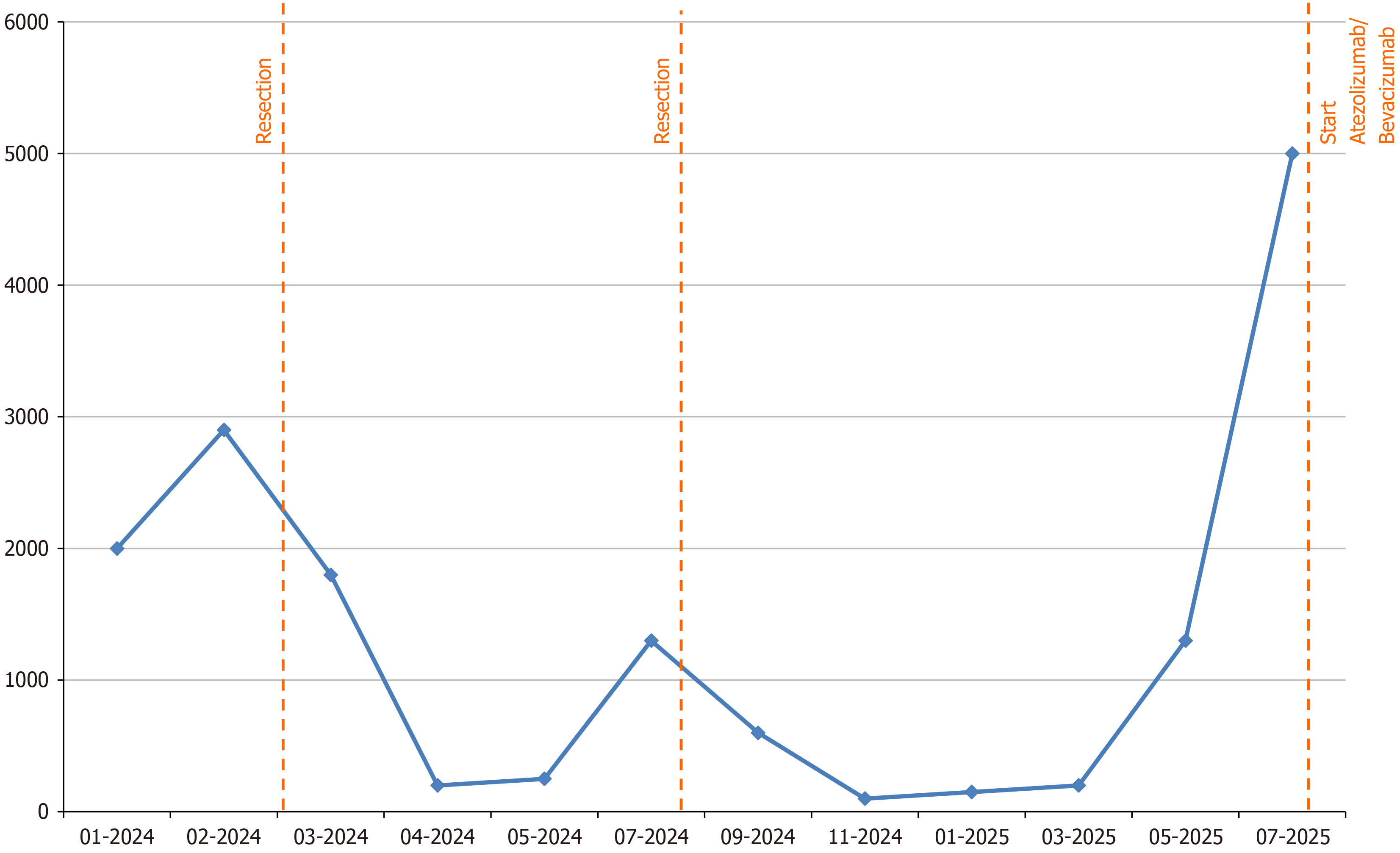
In October 2022, the patient initiated a new hormonal regimen, micronized progesterone 800 mg/day, estradiol valerate 2 mg/day, triptorelina 3.75 mg and prednisone 5 mg/day, in preparation for pregnancy. Routine laboratory testing during medically assisted reproduction revealed elevated AFP levels (16.9 ng/mL; normal range 0-4 ng/mL). Despite the low corticosteroids dose (< 10 mg/day)[8], HBV DNA levels increased in April 2023 (63488 IU/mL) prompting initiation of antiviral therapy with Entecavir 0.5 mg/day. In July 2023, Entecavir was replaced with Tenofovir disoproxil fumarate 245 mg/day after the patient achieved pregnancy through ART. In September 2023, she experienced a spontaneous miscarriage and subsequently underwent uterine curettage (Figure 1).
In November 2023 routine laboratory tests performed in preparation for a new pregnancy showed a marked increase in AFP levels (> 1000 ng/mL) (Figure 2). Abdominal magnetic resonance imaging and positron emission tomography were initially negative for focal lesions. AFP levels continued to rise, reaching 1960 ng/mL in January 2024, despite normal aminotransferase values and undetectable HBV DNA. In February 2024 the patient was referred to our Liver Unit for a second opinion.
The patient had a family history of HBsAg positivity (maternal grandmother, maternal aunt, mother, and two brothers). None of the family members developed advanced chronic liver disease.
On physical examination the patient showed no signs of advanced chronic liver disease, had normal vital parameters, and reported no symptoms.
The patient underwent laboratory testing to assess AFP levels and calculate the PAGE B score (platelet, age, gender). Laboratory analysis showed a progressive increase in AFP levels (2908 ng/mL), while the PAGE B score indicated a low risk of HCC development (age 46 years, female sex, platelet count 295000/mm3). Additional laboratory tests were performed, including quantification of HBV DNA and HBsAg, which revealed values of < 10 IU/mL and 2070 IU/mL, respectively.
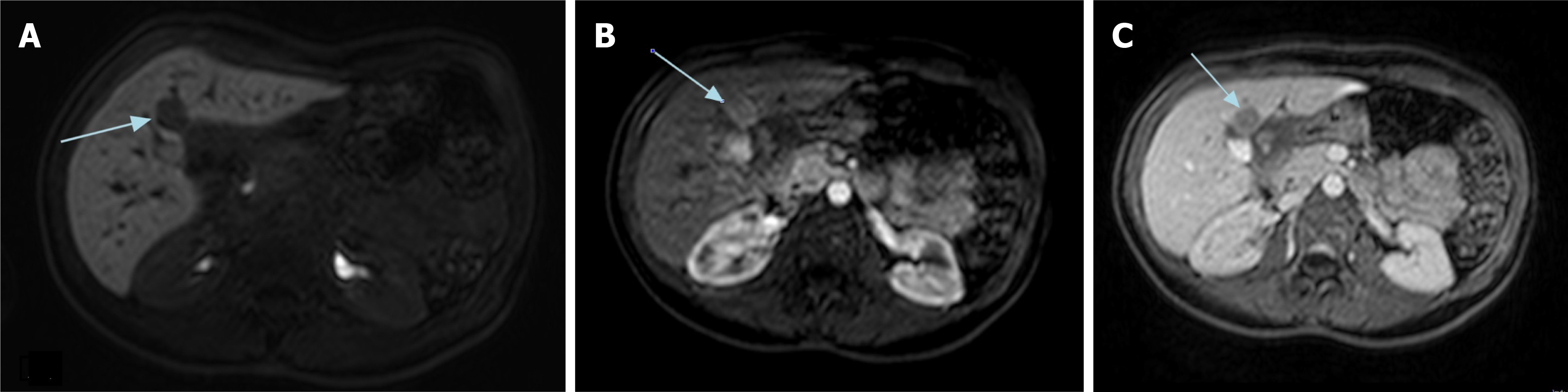
On 9th February 2024 abdominal magnetic resonance imaging was performed. Imaging revealed an HCC nodule (LI-RADS 5) in segment IV, measuring 18 mm in diameter, along with a second smaller nodule of 9 mm in segment II (LI-RADS 2) (Figure 3).
The case was subsequently discussed at the regional tumour board. In line with international guidelines[10], the multidisciplinary team evaluated the patient as having an early stage HCC according to the Barcelona Clinic Liver Cancer classification - a single nodule < 20 mm in diameter, without extra-hepatic localization of disease and/or portal vein invasion, without signs of portal hypertension and associated with a good performance status - and recommended hepatic resection as the most appropriate therapeutic approach.
The patient was diagnosed as early-stage HCC.
On March 2024, the patient underwent surgical treatment with hepatectomy, and histological analysis confirmed a differentiated HCC (pT1a/G2/V1/R0/N0, Figure 1).
Immunofluorescence analysis revealed the absence of oestrogen receptors (ER) in tumour samples. To further investigate the virological profile of the patient and to explore the molecular characteristics of HBV persistence within the liver, frozen tissue specimens were obtained from both the tumour and non-tumour areas. Each sample was divided into two parts, from which total DNA and RNA were extracted as previously described[11]. Quantification of total HBV DNA and HBV covalently closed circular DNA (cccDNA) was performed as previously reported with minor modifications[11]. Briefly, the total intrahepatic HBV DNA or HBV cccDNA were determined using a Rotor-Gene Q (Qiagen, Germany) in a 20 μL reaction volume containing 1 μg DNA, 3 mmol/L MgCl2, 0.5 μmol/L, specific forward and reverse primers, and 0.75 μmol/L 5’-FAM-labelled and 3’-TMR-labelled TaqMan probe (Table 1). Serial dilutions of a plasmid containing a monomeric HBV insert (Alfa Wasserman, Italy) were used as quantification standards. Amplification of total intrahepatic HBV DNA was performed under the following conditions: 94 °C for 3 minutes, followed by 60 cycles of 94 °C for 45 seconds, 58 °C for 40 seconds, and 72 °C for 40 seconds. Before quantitative reverse transcription polymerase chain reaction (qRT-PCR) of HBV cccDNA, aliquots of the liver DNA extracts were treated for 2 hours at 37 °C with 10 U of plasmid-safe DNase (Epicentre, Madison, WI, United States). Amplification of HBV cccDNA was then performed as follows: 94 °C for 3 minutes, 60 cycles of 94 °C for 45 seconds, 60 °C for 40 seconds, and 72 °C for 30 seconds. To normalize the number of viral genomes per liver sample, haploid genome equivalents were quantified using a β-globin gene kit (Light Cycler-Control Kit DNA; Roche Diagnostics, Germany). For HBV RNA analysis, 2 μg of DNase-treated RNA was reverse-transcribed and amplified using the SuperScript First- Strand Synthesis System for qRT-PCR (Invitrogen, CA, United States). Two μL of complementary DNA were quantified by qRT-PCR on the Rotor-Gene Q (Qiagen, Germany) using the same primers and probe designed to quantify total HBV DNA (Table 1), under the following conditions: 94 °C for 3 minutes, followed by 55 cycles of 94 °C for 40 seconds, 60 °C for 30 seconds. Molecular analyses revealed that total HBV DNA concentration levels in the tumour tissue were 4.04 × 10-2 copies/cell, HBV cccDNA concentration values were 8.81 × 10-4 copies/cell, while HBV RNA were 1.14 × 10-3 copies/cell. The corresponding concentration levels in the non-tumour tissue were 3.48 copies/cell for total HBV DNA, 5.36 × 10-4 copies/cell for HBV cccDNA, 90 copies/cell for total HBV RNA. Collectively, molecular analyses highlighted high concentration levels of HBV DNA, HBV cccDNA and RNA in both tumour and non-tumour tissues.
| Name | Polarity | Sequence primer (5’ to 3’) | Position1 |
| HBVDNAfwd | Forward | CTCGTGGTGGACTTCTCTC | 255-273 |
| HBVDNArev | Reverse | CAGCAGGATGAAGAGGAA | 420-403 |
| HBVDNAProbe | Reverse | 6FAM-AACCAGGACAAATTGGAGGACAGGA-TMR | 370-346 |
| cccDNAfwd | Forward | CTCCCCGTCTGTGCCTTCT | 1547-1565 |
| cccDNArev | Reverse | GCCCCAAAGCCACCCAAG | 1902-1885 |
| cccDNAProbe | Reverse | 6FAM-CATGCGACGTGCAGAGGTGAAGCGAA-TMR | 1612-1587 |
| HBVRNAfwd | Forward | CTCGTGGTGGACTTCTCTC | 255-273 |
| HBVRNArev | Reverse | CAGCAGGATGAAGAGGAA | 420-403 |
| HBVRNAProbe | Reverse | 6FAM-AACCAGGACAAATTGGAGGACAGGA-TMR | 370-346 |
Subsequently, AFP levels were monitored monthly, 107 ng/mL in April 2024, 120 ng/mL in May, and 1353 ng/mL in June, while HBsAg and HBV DNA levels measured in April 2024 were 2140 IU/mL and < 10 IU/mL, respectively (Figure 2). In June 2024, a new abdominal computed tomography revealed HCC recurrence with a nodule of 17 mm in segment IVb. Considering the performance status of the patient and the stage of liver disease according to Barcelona Clinic Liver Cancer classification (early-stage HCC), in July 2024, the patient underwent a second hepatic resection for early HCC recurrence (Figure 1).
Over the following year, the patient was monitored using AFP measurements every two months (Figure 2) and abdominal magnetic resonance imaging every three months, with no evidence of HCC recurrence. However, in May 2025, a subcapsular nodule in segment V of 14 mm in diameter was detected, with AFP levels exceeding 5000 ng/mL. Due to disease recurrence and the progressive increase in AFP, the case has been discussed in the multidisciplinary tumour board, with the indication of starting systemic therapy as a bridge therapy for orthotopic liver transplant. Thus, systemic treatment with Atezolizumab plus Bevacizumab was initiated in July 2025. Clinical history of the patient is represented in Figure 1.
This case is particularly remarkable as it represents an extremely rare presentation of HCC, and to our knowledge, is the first reported instance of HCC arising during a pregnancy achieved through ART. Although HCC is the most common primary liver malignancy worldwide[10,12], its incidence is significantly higher in males than in females[7], and is typically related to advanced chronic liver disease, occurring rarely in non-cirrhotic livers, and exceptionally during pregnancy. To date, only 63 cases of HCC diagnosed during pregnancy have been reported in the literature[13]. Prognosis in such cases is generally poor, with maternal survival rates of approximately 23% and fatal loss reported in 42% of cases[14,15].
The molecular mechanisms underlying HCC development during pregnancy are not yet fully understood. In our patient long-term corticosteroid exposure associated with hormonal therapy, followed by the immunotolerant state characteristic of pregnancy, may have contribute to tumour immune escape and disease progression[16]. The timing of diagnosis, within six months of an ART-induced pregnancy, together with the aggressive behaviour and early recurrence, supports the hypothesis that pregnancy may have acted as an oncogenic trigger in this patient.
Immunofluorescence analysis revealed the absence of ER expression in tumour tissue. HCC exhibits marked sexual dimorphism, and is influenced by hormonal factors, with oestrogens exerting tumour-suppressive effects and androgens promoting hepatocarcinogenesis[17]. Oestrogens regulate inflammatory responses, oxidative-stress, and nuclear factor-κB signalling[18], suppress interleukin-6 release, modulate macrophage polarization, and inhibit NOD, LRR and pyrin domain-containing protein 3 inflammasome activation[19]. Furthermore, oestrogens reduce HCC risk and favour a less aggressive disease phenotype by inhibiting signal transducer and activator of transcription 3 signalling also interacting with Foxa1. Previous studies have demonstrated reduced or absent ER expression in HCC cells, a finding confirmed in our case, suggesting loss of hormonal protection and potential contribution to aggressive tumour behaviour[20]. Epigenetic mechanisms further support the link between hormonal signalling and hepatocarcinogenesis. Increased expression of microRNA-22, a small non-coding RNA highly expressed in HCC, suppresses ER activity, attenuating oestrogen-mediated protective effects and promoting interleukin-1α release, thereby facilitating tumour development[21]. Emerging evidence also suggests a role for ER in downregulating HBV RNA transcription, influencing the natural history of HBV infection and HBV-associated HCC development. Collectively, these findings underscore the complex interplay between hormonal signalling, inflammation, viral replication, and tumour risk in patients with chronic HBV infection[22].
Literature data about the impact of medically ART on hormone-sensitive neoplasm risk, are limited. Current evidence is conflicting, due to a lack of standardization in medical regimens applied, and a limited number of studies most of which are observational, elements that don’t allow univocal conclusions[23]. Nowadays, no clinical studies have explored the role of ART in HCC development, about which controlled clinical trials are warranted.
In parallel with the lack of oestrogen protective activity and the potential oncogenic role of pregnancy, chronic HBV infection represents a major oncogenic driver in this patient. Molecular analyses demonstrated high levels of total HBV DNA, HBV cccDNA and HBV RNA levels in both tumour and non-tumour tissues. HBV replication is strongly influenced by the host immune status; therefore, prolonged corticosteroids exposure during ART and the endogenous hypercortisolism of pregnancy may have enhanced viral replication[24,25]. Although antiviral therapy effectively suppresses circulating HBV DNA levels, it does not eradicate intrahepatic viral replication, as reflected by persistent cccDNA. This ongoing virus-host interaction may promote hepatocarcinogenesis through genomic integration, epigenetic modulation, and chronic inflammation.
HBV cccDNA plays a central role in hepatocarcinogenesis, by sustaining viral transcription facilitating viral protein expression, and enabling HBV DNA integration into the host genome. Moreover, cccDNA regulates both viral and host gene expression through epigenetics mechanisms and contributes to liver inflammation, thereby fostering the development of a pro-carcinogenic microenvironment[26]. Molecular analyses revealed the persistence of transcriptionally active HBV forms, which could be associated, in our patients, to viral genome integration[27]. Integration events, present in up to 90% of neoplastic hepatocytes, can drive clonal expansion, oncogene activation and tumour suppression genes inactivation[3,28]. However, a limitation of the present study is the lack of molecular analyses of HBV integration sites, which could have further clarified viral oncogenic mechanisms and strengthened our hypothesis. Taken together, loss of oestrogen-mediated protection, chronic HBV infection, prolonged steroid exposure during ART and the immunological environment of pregnancy, may have synergistically contributed to the emergence of a particularly aggressive HCC phenotype in this patient.
This case describes a unique and highly aggressive presentation of HCC in a non-cirrhotic liver, diagnosed shortly after an ART-induced pregnancy. The rarity of HCC during pregnancy, combined with its poor prognosis and early recurrence, underscores the need for heightened clinical awareness in women of reproductive age with chronic hepatitis B infection. Importantly, this case emphasizes the critical role of active surveillance, not only during pregnancy but also in the preparatory phase of fertility treatments. Early detection through AFP monitoring and imaging, together with multidisciplinary management, may significantly impact maternal and foetal outcomes. Further research is needed elucidate the complex interactions between viral persistence, hormonal modulation, and immune tolerance in HCC pathogenesis, and to refine personalized surveillance strategies for women with chronic HBV undergoing ART. Prospective cohort studies with standardized treatment protocols are warranted to better assess the long-term safety of ART in patients at increased risk of liver-related events.
| 1. | Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76817] [Cited by in RCA: 69601] [Article Influence: 13920.2] [Reference Citation Analysis (42)] |
| 2. | Llovet JM, Kelley RK, Villanueva A, Singal AG, Pikarsky E, Roayaie S, Lencioni R, Koike K, Zucman-Rossi J, Finn RS. Hepatocellular carcinoma. Nat Rev Dis Primers. 2021;7:6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1323] [Reference Citation Analysis (0)] |
| 3. | Rizzo GEM, Cabibbo G, Craxì A. Hepatitis B Virus-Associated Hepatocellular Carcinoma. Viruses. 2022;14:986. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 153] [Article Influence: 38.3] [Reference Citation Analysis (1)] |
| 4. | Lau WY, Leung WT, Ho S, Lam SK, Li CY, Johnson PJ, Williams R, Li AK. Hepatocellular carcinoma during pregnancy and its comparison with other pregnancy-associated malignancies. Cancer. 1995;75:2669-2676. [PubMed] [DOI] [Full Text] |
| 5. | Ducreux M, Abou-Alfa GK, Bekaii-Saab T, Berlin J, Cervantes A, de Baere T, Eng C, Galle P, Gill S, Gruenberger T, Haustermans K, Lamarca A, Laurent-Puig P, Llovet JM, Lordick F, Macarulla T, Mukherji D, Muro K, Obermannova R, O'Connor JM, O'Reilly EM, Osterlund P, Philip P, Prager G, Ruiz-Garcia E, Sangro B, Seufferlein T, Tabernero J, Verslype C, Wasan H, Van Cutsem E. The management of hepatocellular carcinoma. Current expert opinion and recommendations derived from the 24th ESMO/World Congress on Gastrointestinal Cancer, Barcelona, 2022. ESMO Open. 2023;8:101567. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 152] [Cited by in RCA: 129] [Article Influence: 43.0] [Reference Citation Analysis (0)] |
| 6. | Krupa K, Fudalej M, Cencelewicz-Lesikow A, Badowska-Kozakiewicz A, Czerw A, Deptała A. Current Treatment Methods in Hepatocellular Carcinoma. Cancers (Basel). 2024;16:4059. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 17] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 7. | Choi KK, Hong YJ, Choi SB, Park YN, Choi JS, Lee WJ, Kim KS. Hepatocellular carcinoma during pregnancy: is hepatocellular carcinoma more aggressive in pregnant patients? J Hepatobiliary Pancreat Sci. 2011;18:422-431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 21] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 8. | Zhang L, Zhang F, Ma Z, Jin J. Hepatitis B virus infection, infertility, and assisted reproduction. J Zhejiang Univ Sci B. 2024;25:672-685. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 9. | Flemming JA, Velez MP. The ART of medicine: Counselling women with liver disease about assisted reproductive technology. J Hepatol. 2021;74:1283-1285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 10. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines on the management of hepatocellular carcinoma. J Hepatol. 2025;82:315-374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 606] [Cited by in RCA: 515] [Article Influence: 515.0] [Reference Citation Analysis (9)] |
| 11. | Giosa D, Lombardo D, Musolino C, Chines V, Raffa G, Casuscelli di Tocco F, D'Aliberti D, Caminiti G, Saitta C, Alibrandi A, Aiese Cigliano R, Romeo O, Navarra G, Raimondo G, Pollicino T. Mitochondrial DNA is a target of HBV integration. Commun Biol. 2023;6:684. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 20] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 12. | Singal AG, Llovet JM, Yarchoan M, Mehta N, Heimbach JK, Dawson LA, Jou JH, Kulik LM, Agopian VG, Marrero JA, Mendiratta-Lala M, Brown DB, Rilling WS, Goyal L, Wei AC, Taddei TH. AASLD Practice Guidance on prevention, diagnosis, and treatment of hepatocellular carcinoma. Hepatology. 2023;78:1922-1965. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1428] [Cited by in RCA: 1346] [Article Influence: 448.7] [Reference Citation Analysis (3)] |
| 13. | Marasciulo F, Passerini I, Fichera A, Ferrari F, Odicino FE, Prefumo F. Hepatocellular carcinoma in pregnancy: A systematic review. Acta Obstet Gynecol Scand. 2024;103:653-659. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 14. | Norouzi A, Naeimi Tabei M, Tavassoli S, Besharat S. Hepatocellular Carcinoma in Pregnancy withUnusual Presentations. Middle East J Dig Dis. 2012;4:228-231. [PubMed] |
| 15. | Maeda T, Imai D, Wang H, Yugawa K, Kinjo N, Kawata K, Ikeda S, Edahiro K, Takeishi K, Iguchi T, Kayashima H, Harada N, Ninomiya M, Yamaguchi S, Konishi K, Tsutsui S, Matsuda H. Hepatic resection for recurrent hepatocellular carcinoma during pregnancy: a case report. Surg Case Rep. 2020;6:229. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 16. | Jørgensen N, Persson G, Hviid TVF. The Tolerogenic Function of Regulatory T Cells in Pregnancy and Cancer. Front Immunol. 2019;10:911. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 114] [Cited by in RCA: 90] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 17. | Zhao Y, Li Z. Interplay of estrogen receptors and FOXA factors in the liver cancer. Mol Cell Endocrinol. 2015;418 Pt 3:334-339. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 54] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 18. | Hishida M, Nomoto S, Inokawa Y, Hayashi M, Kanda M, Okamura Y, Nishikawa Y, Tanaka C, Kobayashi D, Yamada S, Nakayama G, Fujii T, Sugimoto H, Koike M, Fujiwara M, Takeda S, Kodera Y. Estrogen receptor 1 gene as a tumor suppressor gene in hepatocellular carcinoma detected by triple-combination array analysis. Int J Oncol. 2013;43:88-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 82] [Article Influence: 6.3] [Reference Citation Analysis (1)] |
| 19. | Nevola R, Tortorella G, Rosato V, Rinaldi L, Imbriani S, Perillo P, Mastrocinque D, La Montagna M, Russo A, Di Lorenzo G, Alfano M, Rocco M, Ricozzi C, Gjeloshi K, Sasso FC, Marfella R, Marrone A, Kondili LA, Esposito N, Claar E, Cozzolino D. Gender Differences in the Pathogenesis and Risk Factors of Hepatocellular Carcinoma. Biology (Basel). 2023;12:984. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 71] [Article Influence: 23.7] [Reference Citation Analysis (0)] |
| 20. | Guo Y, Wu G, Yi J, Yang Q, Jiang W, Lin S, Yang X, Cai X, Mao L. Anti-Hepatocellular Carcinoma Effect and Molecular Mechanism of the Estrogen Signaling Pathway. Front Oncol. 2021;11:763539. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 25] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 21. | Montella M, D'Arena G, Crispo A, Capunzo M, Nocerino F, Grimaldi M, Barbieri A, D'Ursi AM, Tecce MF, Amore A, Galdiero M, Ciliberto G, Giudice A. Role of Sex Hormones in the Development and Progression of Hepatitis B Virus-Associated Hepatocellular Carcinoma. Int J Endocrinol. 2015;2015:854530. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 48] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 22. | Liu WC, Liu QY. Molecular mechanisms of gender disparity in hepatitis B virus-associated hepatocellular carcinoma. World J Gastroenterol. 2014;20:6252-6261. [PubMed] [DOI] [Full Text] |
| 23. | Del Pup L, Peccatori FA, Levi-Setti PE, Codacci-Pisanelli G, Patrizio P. Risk of cancer after assisted reproduction: a review of the available evidences and guidance to fertility counselors. Eur Rev Med Pharmacol Sci. 2018;22:8042-8059. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 24. | Gambarin-Gelwan M. Hepatitis B in pregnancy. Clin Liver Dis. 2007;11:945-963, x. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 60] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 25. | Jonas MM. Hepatitis B and pregnancy: an underestimated issue. Liver Int. 2009;29 Suppl 1:133-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 145] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 26. | Lin YH, Lai MW, Yeh CT, Lin WR. Perspectives on NcRNAs in HBV/cccDNA-driven HCC progression. Cancer Cell Int. 2025;25:224. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 27. | Zoulim F, Chen PJ, Dandri M, Kennedy PT, Seeger C. Hepatitis B virus DNA integration: Implications for diagnostics, therapy, and outcome. J Hepatol. 2024;81:1087-1099. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 76] [Article Influence: 38.0] [Reference Citation Analysis (0)] |
| 28. | Jiang Y, Han Q, Zhao H, Zhang J. The Mechanisms of HBV-Induced Hepatocellular Carcinoma. J Hepatocell Carcinoma. 2021;8:435-450. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 183] [Article Influence: 36.6] [Reference Citation Analysis (1)] |