Published online Apr 15, 2026. doi: 10.4251/wjgo.v18.i4.115614
Revised: December 23, 2025
Accepted: February 10, 2026
Published online: April 15, 2026
Processing time: 165 Days and 23.1 Hours
Gastric cancer is the sixth most common malignancy worldwide. Metastatic small intestinal malignancy is more common than primary small intestinal malignancy; however, metastasis from gastric cancer to the small intestine is extremely rare. Triple primary gastrointestinal malignancies have been sporadically reported and linked to rare genetic mutations. Rarely, tumor-to-tumor metastasis (TTM) occurs when a primary donor tumor metastasizes to another distinctive recipient pri
A 57-year-old male with a history of colon cancer remained disease-free for eight years before presenting with abdominal pain, easy fatigability, and diarrhea. Upper endoscopy showed a fungating gastric mass, which was diagnosed as moderately differentiated primary gastric adenocarcinoma. After four cycles of neoadjuvant chemotherapy, exploratory laparotomy revealed an ileal mass, histopathologically diagnosed as moderately differentiated primary small intestinal adenocarcinoma with metastatic mucinous adenocarcinoma of gastric origin, consistent with TTM. The patient received 20 cycles of chemotherapy but unfortunately passed away secondary to septic shock.
TTM of gastric adenocarcinoma to small intestinal adenocarcinoma is a rare and challenging clinical entity that requires a high index of suspicion and meticulous histopathological and immunohistochemical evaluation. Its pathophysiology is not fully understood, and both prognosis and treatment intent depend on the type of donor tumor and the nature of the recipient tumor, whether benign or malignant.
Core Tip: Metastatic involvement of the small intestine usually results from direct invasion by adjacent organs, while hematogenous spread is rare. Interestingly, metastatic tumors are more common than primary small bowel malignancies due to protective mucosal and immunological factors. Tumor-to-tumor metastasis is an exceptionally rare event, defined as the growth of a metastatic tumor within another primary neoplasm. Among reported cases, renal cell carcinoma is the most frequent recipient, and gastric primaries are exceedingly uncommon. This report presents the first documented case of Recurrent gastric adenocarcinoma metastasizing into a primary small bowel adenocarcinoma, highlighting the importance of thorough histopathological evaluation.
- Citation: AlQattan AS, Binkhamis L, Foula MS, Alsalim A, Alselwi WM, Alzahir AA. Triple gastrointestinal malignancies with tumor-to-tumor metastasis from gastric adenocarcinoma to small bowel adenocarcinoma: A case report. World J Gastrointest Oncol 2026; 18(4): 115614
- URL: https://www.wjgnet.com/1948-5204/full/v18/i4/115614.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i4.115614
Gastric cancer is the sixth most common malignancy worldwide and remains the third leading cause of cancer-related mortality, despite significant advances in systemic therapies[1,2]. The prognosis for metastatic gastric cancer remains poor, with a median overall survival of approximately nine months. Patterns of metastasis in gastric cancer are not fully established and may vary across different parts of the world, but the most reported metastatic sites include the peritoneum, liver, distant lymph nodes, bone, and brain[3]. In contrast, primary small bowel adenocarcinoma is rare, accounting for only 4% of all gastrointestinal (GI) malignancies, yet they carry a poor prognosis, with five-year survival rates as low as 32% in non-metastatic cases[4]. Interestingly, secondary involvement of the small bowel through meta
A 57-year-old male presented with a complain of epigastric abdominal pain associated with symptomatic anaemia.
Upon presentation he was complaining of a 1-month history of epigastric abdominal pain, diarrhea and symptoms of anaemia.
The patient had a history of colon cancer for which he underwent open extended left hemicolectomy (pT3N0M0, with no high-risk features) in 2014, with no evidence of recurrence or metastasis during 5 years of continuous surveillance.
Upon presentation, the patient’s vital signs were within normal limits. Abdominal examination revealed a midline laparotomy scar from the previous surgery, with no palpable masses.
Laboratory investigations were unremarkable, except for a haemoglobin level of 8.5 g/dL.
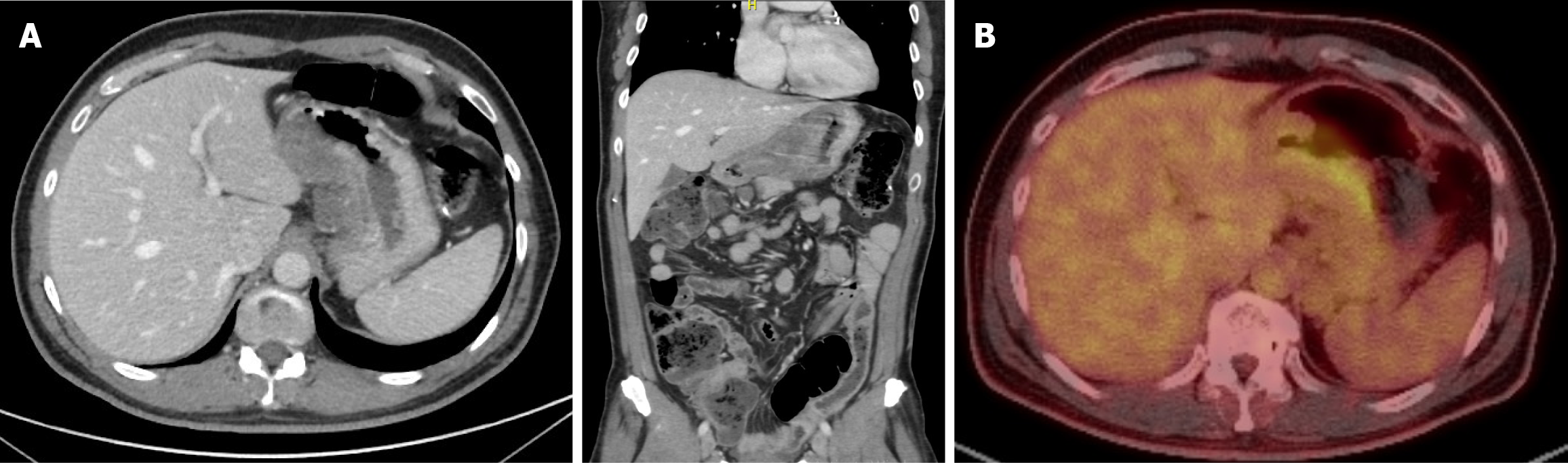
Computed tomography (CT) scan of the chest, abdomen and pelvis was done for complete staging and showed: A diffuse mural thickening of the lesser curvature of stomach reaching the antrum, associated with locoregional. Whole body positron emission tomography (PET)/CT scan showed FDG uptake in the gastric lesser curvature thickening seen on the CT scan as well as in the left para-aortic lymph nodes (cT3N3M1) (Figure 1).
An-esophago gastroduodenoscopy (EGD) was showed a large fungating, ulcerated mass occupying the fundus extending down to the antrum. Biopsy from the mass showed a moderately differentiated adenocarcinoma with mucinous features. The tumor cells were positive for CDX2 immunostaining while being negative for CK20. Programmed death-ligand 1: Positive (Combined Positive Score = 15), microsatellite stable, HER 2 was negative. Complete colonoscopy showed no evidence of recurrence or new lesions. An EGD was showed a large fungating, ulcerated mass occupying the fundus extending down to the antrum.
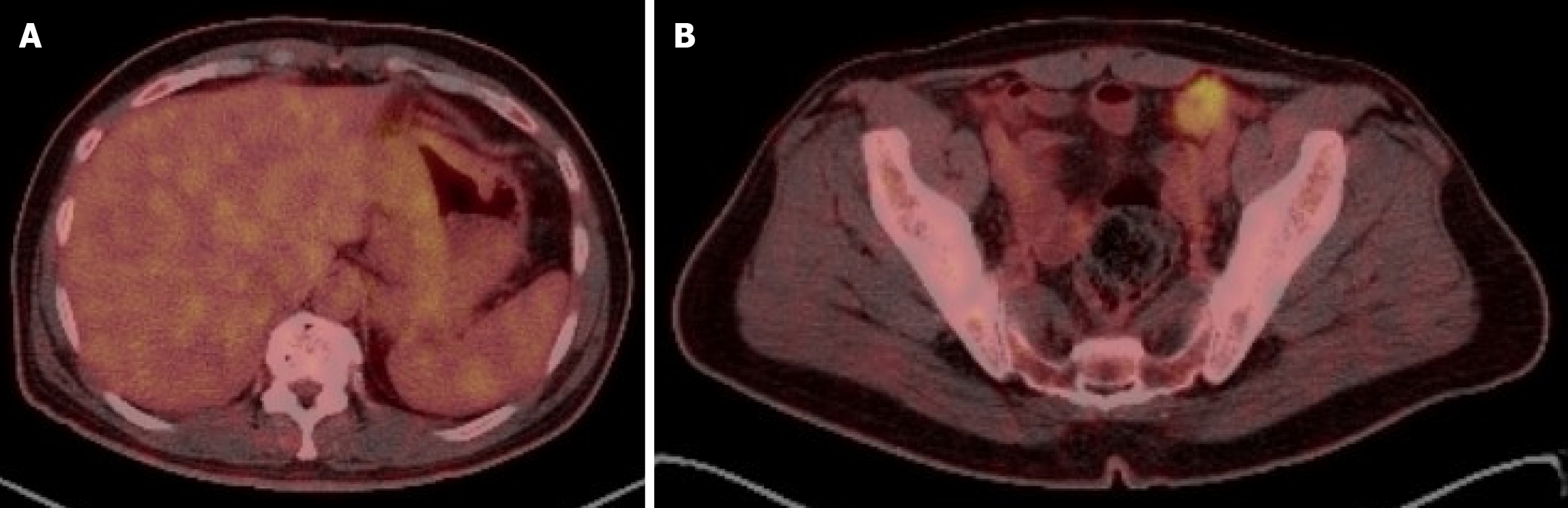
The case was discussed in the multidisciplinary tumor board, and the consensus were to start with a neoadjuvant chemotherapy 4 cycles of FLOT followed by restaging and possible surgery since it was oligo-metastatic disease. Pre-treatment diagnostic laparoscopy was omitted as pre-treatment radiological staging was clearly consistent with locally advanced disease. After completion of neoadjuvant significant interval metabolic regression of the previously noted gastric curvature FDG avid thickening with complete metabolic remission of the previously noted locoregional lymph nodes. However, there was a new FGD uptake in an ileal bowel loop that was reported as inflammatory/infectious (Figure 2).
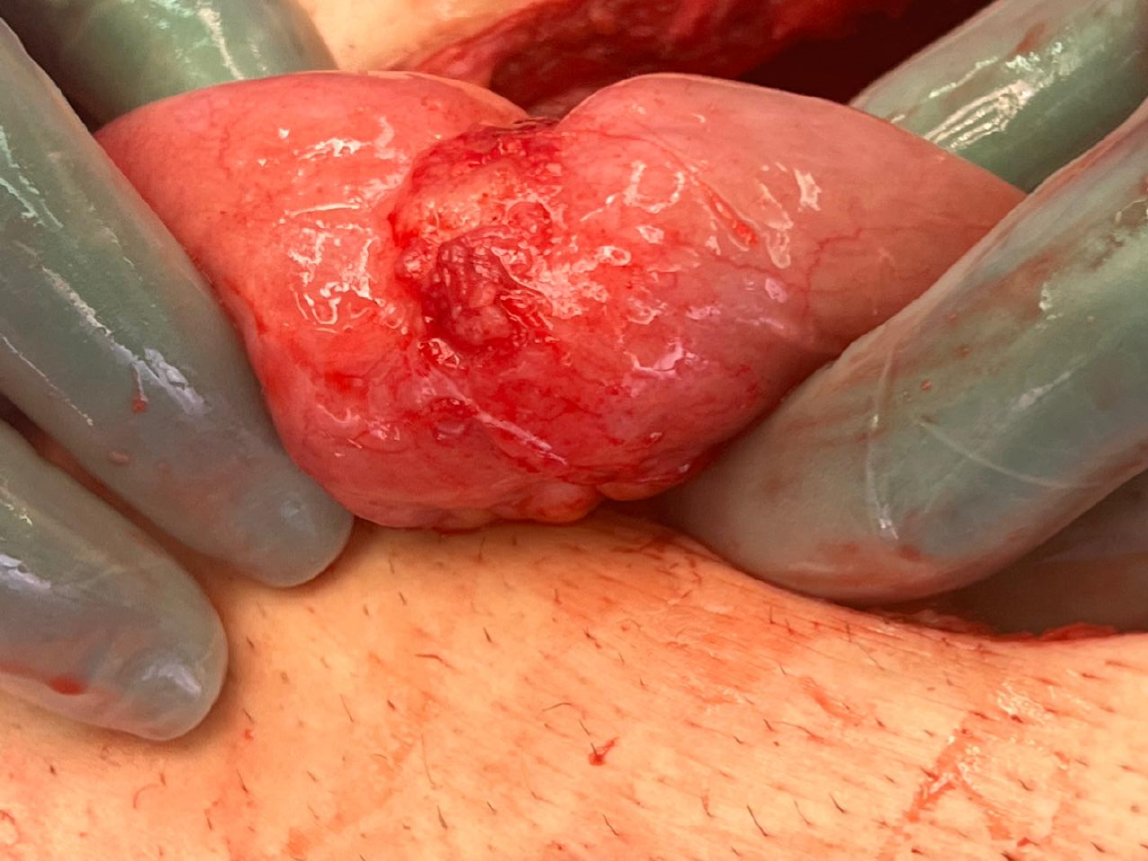
Diagnostic laparoscopy was done which revealed multiple adhesions between the small bowel and anterior abdominal wall, so it was converted to laparotomy, and the adhesions were found to be between a small bowel loops located 40 cm proximal to the ileocecal valve with no evidence of solid organ or peritoneal metastases. A sharp adhesiolysis was done which revealed a mass at the same location, with no evidence of any peritoneal or liver metastasis (Figure 3). With the presumption that it’s a single metastatic deposit from the primary gastric cancer and the possibility of bowel obstruction, so we proceeded with a limited resection of that bowel segment with primary anastomosis while the gastrectomy was aborted, as the goal of care was shifted toward palliative intent.
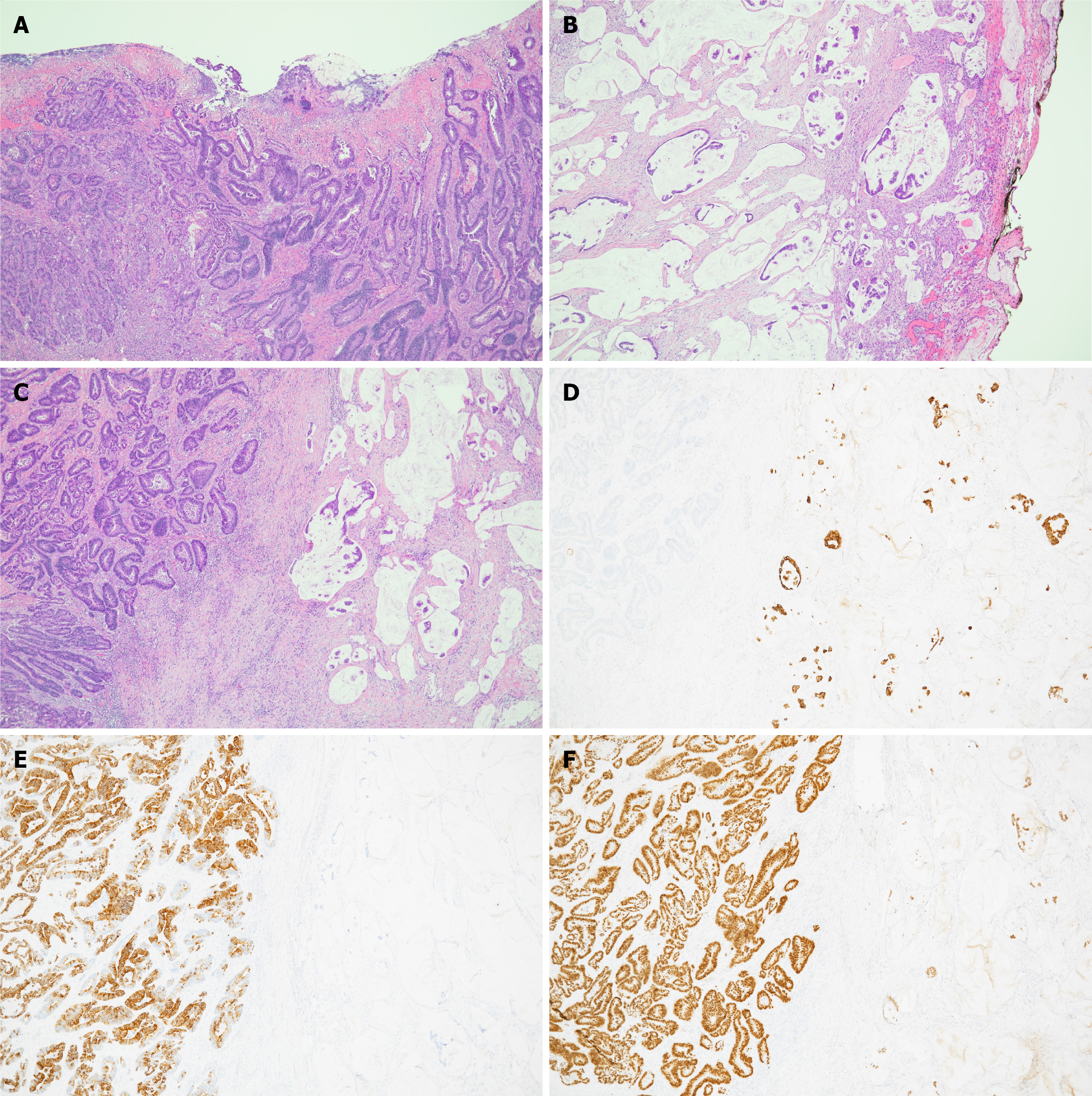
The final histopathology showed a 4 cm tumor consisting of: (1) Small bowel adenocarcinoma, moderately differentiated. The immunohistochemistry of the tumor from the mucosal side was positive for CK20 and CDX2, while being negative for CK7 consistent with adenocarcinoma of lower GI origin, i.e.; a primary small bowel adenocarcinoma (pT3NxM0); and (2) Metastatic mucinous adenocarcinoma of gastric origin invading the small bowel through the serosal surface. With an immunohistochemistry profile of positive for CK7 and CDX2, while being negative for CK20 consistent with metastasis from the patient’s known primary gastric adenocarcinoma. Both proximal and distal resection were negative. Consistent with TTM (Figure 4).
The case was re-discussed again in the multidisciplinary team, and the decision was to proceed with additional 4 cycles of FLOT which was followed by a 2nd diagnostic laparoscopy and possible gastrectomy. However, the procedure was terminated due to widespread carcinomatosis all-over the abdomen. The patient was then kept on FOLFIRI and Nivolumab, after completing 20 cycles the latest CT scan showed a stable local gastric disease with interval progression of the peritoneal deposits.
The patient unfortunately presented with a clinical picture of septic shock secondary to pneumonia and passed away after 39 months from time of diagnosis.
The small intestine is a frequently affected site of GI metastasis, predominantly due to direct extension from adjacent organs such as the stomach or pancreas, whereas lymphatic or hematogenous spread is relatively uncommon. In
The process by which malignant tumors disseminate to establish secondary neoplastic foci in anatomically distant sites, termed metastasis, was first recognized in 1848. However, a well-documented instance of TTM was later reported by Berent in 1902, detailing the metastatic spread of a squamous cell carcinoma originating in the jaw to a pre-existing renal cell carcinoma[9]. TTM is defined as the hematogenous spread of a secondary (donor) tumor into a primary neoplasm, referred to as the recipient tumor. In contrast, a collision tumor is a rare pathological entity characterized by the coexistence of two histologically distinct tumor types within the same anatomical region, without evidence of one invading the other[10,11]. Although uncommon, these two entities are often misinterpreted as synonymous in the literature. The criteria established remain the standard for diagnosing dual tumors (TTM) and distinguishing it from other tumor interactions[10,11]. These include: (1) At least two histologically confirmed primary tumors; (2) Evidence of metastatic (donor) tumor growth within the host (recipient) tumor; (3) Confirmation that the recipient tumor is a true neoplasm; and (4) The recipient tumor must not be a lymph node associated with or involved in leukaemia or lymphoma[10,11]. These criteria aid in differentiating TTM from collision tumors.
A systematic review and meta-analysis conducted by Kunc et al[11] included all the published cases of TTM where Campbell’s criteria were fulfilled. Cases where there was only a tumor embolus in a secondary tumor or metastasis to a lymph node basin already involved by a systemic lymphatic malignancy were excluded. A total of 685 cases of TTM were included in the meta-analysis. The age at time of diagnosis ranged from 16-97 years, of which 57.5% were females. In the early reported cases (prior to 2000), more than half were diagnosed at autopsy, while only 6.2% were diagnosed post-mortem in the following years, which can be attributed to advancements in imaging modalities and screening protocols over time.
The underlying pathogenesis of TTM is not fully elucidated, though several mechanisms have been proposed. One of the most widely cited is the “seed and soil” hypothesis, first introduced by Stephen Paget in 1889. This theory suggests that metastatic tumor cells “seeds” require a receptive microenvironment “soil” to establish secondary growth, which may be provided by certain primary tumors due to factors such as rich vascularity, slow growth, hormonal expression, and the presence of abundant lipids and collagen-all of which may facilitate donor tumor survival by providing essential nutrients. Supporting this concept, a meta-analysis of patients meeting the criteria for TTM found that most recipient tumors exhibited high vascularity and angiogenic potential[12,13].
Notably, donor tumor histopathological type showed a predominance of adenocarcinoma from breast (29%), followed by lung (25%) and kidney (14%), while gastric primaries accounted for less than 3% of all reported cases (20 cases). The recipient tumors were benign in two-thirds of cases, with meningiomas being the most common[9,11]. In the existing literature, multiple authors have identified renal cell carcinoma as the predominant malignant recipient of TTM[12,13]. Honma et al[14] described the first case of primary gastric adenocarcinoma metastasizing to a cerebral hemangioma, discovered incidentally at autopsy. As previously noted, donor tumors in TTM typically Favor recipients with rich vascularity. In contrast, Nomura et al[13] reported a case involving a poorly vascularized neck mass in an elderly male, which was ultimately revealed as well-differentiated adipose tissue consistent with a lipoma. However, the lesion also contained signet ring-like cells, and immunohistochemical staining confirmed the presence of metastatic gastric adenocarcinoma[14]. In support of our case rationale, Sakai et al[15] reported a case involving a female who presented with a renal mass six years after distal gastrectomy for gastric adenocarcinoma. Histopathological analysis following nephron-sparing surgery revealed clear-cell renal cell carcinoma containing metastatic signet-ring cells. The patient subsequently received three months of chemotherapy without reported complications.
A noteworthy consideration is the potential role of genetic mutations in increasing the risk of TTM. One reported case involved a patient with triple GI malignancies, including primary sigmoid adenocarcinoma and small bowel adenocarcinoma, with an isolated gastric metastasis from the small bowel linked to lynch syndrome[16]. Conversely, our case also presented with triple GI malignancies, but lynch syndrome was excluded based on a negative Modified Amsterdam Criteria, suggesting a different underlying predisposition.
The preoperative diagnosis of TTM is often incidental, since these lesions usually lack distinctive clinical and radio
Due to its extreme rarity, TTM is described almost exclusively through case reports. As a result, treatment decisions remain challenging and are largely determined by whether the recipient tumor is benign or malignant. Most cases in the literature have been treated surgically, often combined with radiotherapy, chemotherapy, immunotherapy, or hormone therapy as appropriate. Prognosis depends mainly on the biological behaviour of both the donor and recipient tumors[17]. In regard to the survival analysis of this patient population, the following factors were associated with inferior overall survival: (1) Occult donor primary; (2) Donor metastasis to multiple recipient sites (disseminated disease); and (3) Shorter disease-free interval from primary to metastasis (< 3 years). Interestingly, the nature of the recipient tumor (benign vs malignant) was not associated with decreased survival[11].
To the best of our knowledge, this is the first reported case of gastric adenocarcinoma metastasizing to the small intestine coexisting with a primary small bowel adenocarcinoma, forming a TTM phenomenon-an exceedingly rare occurrence with no similarly reported cases in the literature. Table 1 highlights all the published cases of TTM from a primary gastric cancer[9,11,13-19]. Recognizing the distinct histological components is essential, as they have a significant impact on treatment planning and prognosis. This case highlights the importance of comprehensive pathological eva
| Ref. | Age/sex | Primary tumor (donor) | Recipient tumor site |
| Kunc et al[11] | Gastric adenocarcinoma | Reported cases included: 5 renal cell tumors, 2 meningiomas, 1 soft tissue tumor, 1 brain tumor, 4 uterine tumors, 2 pituitary gland tumors, 2 thyroid tumors, 1 lung tumor, and 2 skin tumors | |
| Mei and Zhang[18] | 52/F | Gastric adenocarcinoma | Chromophobe renal cell carcinoma |
| Nomura et al[13] | 71/M | Gastric adenocarcinoma | Lipoma |
| Kiyokoba et al[17] | 62/F | Gastric adenocarcinoma | Uterine lipoleiomyoma |
| Shi et al[19] | 41/F | Gastric adenocarcinoma | Bilateral ovarian granulosa cell tumors |
| Sakai et al[15] | 75/F | Gastric adenocarcinoma | Renal cells carcinoma |
| Pamphlett[14] | 74/F | Gastric adenocarcinoma | Meningioma |
| Petraki et al[9] | 66/F | Gastric adenocarcinoma | Renal cells carcinoma |
| Petraki et al[9] | 45/M | Gastric adenocarcinoma | Pre-existent lympho-sarcoma at regional nodes |
TTM of a donor gastric adenocarcinoma to a recipient small intestinal adenocarcinoma is a challenging clinical entity that necessitates a high index of suspicion and meticulous histopathological examination and immunohistochemistry assessment. Recognizing this rare metastatic behavior is important for accurate diagnosis, appropriate treatment planning, and understanding the unique biological interactions that may underlie such cases. Its pathophysiology is not fully understood yet and both the prognosis and intent of treatment depend on the type of malignancy of donor tumor and the nature of recipient tumor, whether it a begin or malignant.
| 1. | Narita Y, Muro K, Takahari D. Practical management of oligometastatic gastric cancer. ESMO Gastrointest Oncol. 2024;6:100108. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 2. | Huang RJ, Laszkowska M, In H, Hwang JH, Epplein M. Controlling Gastric Cancer in a World of Heterogeneous Risk. Gastroenterology. 2023;164:736-751. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 92] [Cited by in RCA: 91] [Article Influence: 30.3] [Reference Citation Analysis (1)] |
| 3. | Sirody J, Kaji AH, Hari DM, Chen KT. Patterns of gastric cancer metastasis in the United States. Am J Surg. 2022;224:445-448. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 44] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 4. | Alfagih A, Alrehaili M, Asmis T. Small Bowel Adenocarcinoma: 10-Year Experience in a Cancer Center-The Ottawa Hospital (TOH). Curr Oncol. 2022;29:7439-7449. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 5. | Wei SC, Su WC, Chang MC, Chang YT, Wang CY, Wong JM. Incidence, endoscopic morphology and distribution of metastatic lesions in the gastrointestinal tract. J Gastroenterol Hepatol. 2007;22:827-831. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 27] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 6. | Fan Q, Su M. Isolated Small Bowel Metastasis From Gastric Cancer Detected by 18F-FDG PET/CT. Clin Nucl Med. 2019;44:840-841. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 7. | Kamiyama H, Ichikawa R, Arima S, Sugiyama Y, Takei M, Namekata K, Tsumura H, Takase M, Okada M, Matsumoto F. Gastric cancer with multiple intramural metastases and metastasis to the small intestine which eventually developed Trousseau's syndrome: report of a case. Clin J Gastroenterol. 2014;7:117-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 8. | Hatzaras I, Palesty JA, Abir F, Sullivan P, Kozol RA, Dudrick SJ, Longo WE. Small-bowel tumors: epidemiologic and clinical characteristics of 1260 cases from the connecticut tumor registry. Arch Surg. 2007;142:229-235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 147] [Article Influence: 7.7] [Reference Citation Analysis (3)] |
| 9. | Petraki C, Vaslamatzis M, Argyrakos T, Petraki K, Strataki M, Alexopoulos C, Sotsiou F. Tumor to tumor metastasis: report of two cases and review of the literature. Int J Surg Pathol. 2003;11:127-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 92] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 10. | Patel Z, Bhatia SS, Han P, McCloskey J, Kassar A. A rare case of tumor-to-tumor metastasis of esophageal adenocarcinoma into meningioma. Radiol Case Rep. 2024;19:2874-2878. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 11. | Kunc M, Skrzypkowska P, Pęksa R, Biernat W. Tumor-to-tumor metastases: systematic review and meta-analysis of 685 reported cases. Clin Exp Metastasis. 2025;42:14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 12. | Minezaki S, Misawa T, Tsukayama H, Shibuya M, Wada K, Sano K, Mochizuki M, Sasajima Y, Kondo H. Tumor-to-tumor metastasis: an extremely rare combination with renal cell carcinoma as the donor and a pancreatic neuroendocrine tumor as the recipient. Surg Case Rep. 2022;8:8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 13. | Nomura S, Kurihara N, Ishikawa T, Tateda M, Sakurada J. Gastric cancer metastatic to neck lipoma: a case report with imaging consideration. Skeletal Radiol. 2018;47:575-578. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 14. | Pamphlett R. Carcinoma metastasis to meningioma. J Neurol Neurosurg Psychiatry. 1984;47:561-563. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 57] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 15. | Sakai Y, Kanomata N, Itami H, Kajimoto K, Sakuma T, Ohbayashi C. Signet-ring cell carcinoma of the stomach metastasizing to renal cell carcinoma: a case report and review of the literature. Kobe J Med Sci. 2010;55:E122-E131. [PubMed] |
| 16. | Matsumoto A, Shimada Y, Kondo S, Mizuno KI, Nakano M, Yamai D, Nakano M, Nyuzuki H, Umezu H, Wakai T. Gastric metastasis from small bowel adenocarcinoma in a Lynch syndrome patient. Clin J Gastroenterol. 2022;15:575-581. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 17. | Kiyokoba R, Yagi H, Yahata H, Kawano Y, Kaneki E, Okugawa K, Sonoda K, Kato K. Tumor-To-Tumor Metastasis of Poorly Differentiated Gastric Carcinoma to Uterine Lipoleiomyoma. Case Rep Obstet Gynecol. 2015;2015:352369. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 18. | Mei W, Zhang D. Unraveling Tumor-to-Tumor Metastasis: Insights into Pathogenesis, Diagnostic Challenges, and Treatment Modalities. Biologics. 2025;19:43-58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 19. | Shi L, Guo Z, Wei X, Jia Y, Bao L, Lu J. Gastric signet-ring cell carcinoma metastasis to bilateral ovarian granulosa cell tumors. Int J Gynecol Pathol. 2015;34:126-131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 1.2] [Reference Citation Analysis (0)] |