Published online Apr 15, 2026. doi: 10.4251/wjgo.v18.i4.115576
Revised: December 29, 2025
Accepted: February 4, 2026
Published online: April 15, 2026
Processing time: 169 Days and 20.6 Hours
Hepatocellular carcinoma (HCC) is typically associated with advanced liver fibrosis or cirrhosis. However, the clinical characteristics, prognosis, and recur
To clarify the clinical characteristics, prognosis, and risk factors for recurrence of F0 vs F4 HCC.
We retrospectively analyzed 315 patients [F0 HCC (n = 42); F4 HCC (n = 105)] who underwent curative hepatic resection for HCC at the Oita University (January 2010-December 2021). Fibrosis stage was determined histologically using Hematoxylin–Eosin and Azan staining according to the New Inuyama Classification. Clinical characteristics, laboratory data, and tumor features were compared. Survival analyses were performed using Kaplan-Meier curves and log-rank tests. Cox proportional hazards regression was applied to identify recurrence risk factors in F0 HCC. The mean observation period was 1463 ± 1110 days.
Patients with F0 HCC were significantly older; more often male; and more frequently had negative for both hepatitis B and C etiology compared with F4 cases (P < 0.05). Platelet counts and prothrombin activity were higher in the F0 group (P < 0.001). Tumor size was significantly larger in F0 HCC, whereas tumor number did not differ between groups. Overall survival (OS) and recurrence-free survival were comparable between F0 and F4 HCC. Cases of F0 and F4 HCC had no significant difference in OS. Among patients with F0 HCC, those without re
F0 HCC presents with distinct clinical features, including older age, male predominance, negative for both hepatitis B and C background, and larger tumor size. Despite preserved liver function, recurrence significantly compro
Core Tip: Hepatocellular carcinoma (HCC) arising in nonfibrotic (F0) livers is uncommon and its clinical behavior remains unclear. This study directly compared F0 and cirrhotic (F4) HCC, revealing that F0 HCC typically occurs in older men with negative for both hepatitis B and C etiology and larger tumors. Despite preserved liver function, recurrence significantly worsened prognosis. Tumor number was the sole independent risk factor for recurrence in F0 HCC. These findings emphasize the need for early detection strategies, even in non-cirrhotic populations.
- Citation: Endo M, Honda K, Tokumaru T, Saito T, Uchida T, Iwao M, Arakawa M, Seike M, Kodama M, Masuda T, Endo Y, Inomata M, Murakami K, Mizukami K. Hepatocellular carcinoma in F0 livers: Clinical and prognostic insights. World J Gastrointest Oncol 2026; 18(4): 115576
- URL: https://www.wjgnet.com/1948-5204/full/v18/i4/115576.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i4.115576
Hepatocellular carcinoma (HCC) is the most common primary liver malignancy and a leading cause of cancer-related mortality worldwide[1]. It typically occurs in the setting of chronic liver disease, particularly cirrhosis, which is strongly associated with chronic hepatitis B and C infection, alcohol-related liver disease, and nonalcoholic fatty liver disease[2]. The close association of HCC with liver fibrosis and cirrhosis is well-established, with approximately 80%-90% of cases developing in fibrotic livers and one-third of patients with cirrhosis predicted to develop HCC during their lifetime[3,4]. The fibrotic microenvironment plays a critical role in hepatocarcinogenesis, particularly through mechanisms such as extracellular matrix remodeling, immune evasion, and enhanced angiogenesis[5,6]. Chronic inflammation, such as in the setting of viral hepatitis or metabolic disorders, further accelerates the progression of fibrosis and increases the risk of HCC[7].
A distinct subset of HCC arises in nonfibrotic livers, following an alternative oncogenic pathway. This nonfibrotic HCC is often triggered by direct genotoxic or metabolic insults, bypassing the conventional fibrosis-cirrhosis sequence. Histologically, these tumors have minimal changes in the surrounding liver tissue and lack the regenerative nodules or pre
Surgical resection is the primary treatment for HCC in nonfibrotic livers. Due to their preserved liver function, such cases have more favorable long-term outcomes vs HCC in cirrhotic livers[19]. However, challenges remain for F0 HCC, specifically in terms of optimizing surveillance strategies and understanding its underlying molecular mechanisms[9]. Accordingly, a deeper understanding of their differences can facilitate the development of more targeted therapeutic strategies and preventive measures for this unique patient population.
In this study, we aimed to define the clinical characteristics and prognosis of F0 HCC using a cohort of histologically confirmed cases, with comparisons made to patients with F4 HCC.
This retrospective analysis included 315 patients who underwent curative hepatic resection for HCC at the Department of Gastroenterological and Pediatric Surgery, Faculty of Medicine, Oita University, between January 2010 and December 2021. The staging of liver fibrosis was determined by experienced liver pathologists via hematoxylin-eosin staining and Azan staining, with the analysis including patients classified as either stage F0 or F4 based on the New Inuyama Classification[20]. Among these patients, only those with background liver histologically diagnosed as either stage F0 (non
Data regarding the clinical background, laboratory tests, and tumor characteristics at the time of HCC onset were collected. The preoperative laboratory data included liver function markers [i.e., albumin, total bilirubin, aspartate aminotransferase, alanine aminotransferase (ALT), prothrombin time], tumor markers (i.e., alpha-fetoprotein, des-gamma-carboxy prothrombin), and platelet count. Tumor characteristics (i.e., tumor size, number of tumors, tumor differentiation and vascular invasion) were recorded. Postoperative recurrence was diagnosed based on imaging studies such as contrast-enhanced computed tomography and magnetic resonance imaging. Follow-up imaging was performed every 3-6 months after surgery. The factors contributing to postoperative recurrence were analyzed using the Cox proportional hazards model.
Overall survival (OS) and recurrence-free survival (RFS) were compared between patients with F0 and F4 HCC. OS was defined as the time from surgery until death from any cause, while RFS was defined as the time from surgery until the first documentation of recurrence or last follow-up without recurrence.
As a sensitivity analysis, age-adjusted and sex-adjusted Cox proportional hazards regression analyses were performed to evaluate the impact of recurrence on OS in patients with F0 HCC.
All statistical analyses were performed using statistical analysis software (SPSS 27.0, IBM Corp., Armonk, NY, United States). Categorical variables were analyzed using the χ² test, while continuous variables were analyzed using the Mann-Whitney U test or Student’s t-test depending on the normality of distribution. Survival curves were estimated using the Kaplan-Meier method, and differences between groups were assessed using the log-rank test. The multivariate analysis included Cox proportional hazards regression to identify independent factors associated with recurrence in patients with F0 HCC. Statistical significance was set at P < 0.05.
This study was approved by the Institutional Review Board of Oita University. Informed consent was obtained using an opt-out method, wherein information about the study was disclosed on the institutional website, and participants were given the opportunity to decline participation. This study was conducted in accordance with the Declaration of Helsinki.
Among a total of 315 patients who underwent liver resection for HCC, 42 (13%) and 105 (33%) patients were classified as having F0 and F4 liver fibrosis, respectively. The baseline characteristics were compared between patients with F0 and F4 HCC (Table 1). Patients with F0 HCC were significantly older (P < 0.001) and had a higher proportion of male patients (P = 0.002). Regarding etiology, negative for both hepatitis B and C (NBNC) disease was significantly higher in the F0 group (P < 0.001), while hepatitis C infection was more common in the F4 group (P < 0.001). The F0 group exhibited significantly higher platelet counts (P < 0.001) and greater prothrombin activity (P < 0.001), indicating better preserved liver function and the absence of advanced fibrosis or portal hypertension. Consistent with these findings, both the fibrosis-4 index (P < 0.001) and the aspartate aminotransferase to platelet ratio index score (P < 0.001) were significantly higher in the F4 group than in the F0 group, reflecting advanced fibrosis in patients with F4.
| F0 HCC (n = 42) | F4 HCC (n = 105) | P value | |
| Sex (male/female) | 36/6 | 62/43 | 0.002 |
| Age (year) | 77.9 ± 7.5 | 70.2 ± 8.1 | < 0.001 |
| Body mass index (kg/m2) | 23.6 ± 3.2 | 24.1 ± 3.6 | 0.481 |
| Platelet count (104/μL) | 20.7 ± 5.3 | 11.9 ± 7.8 | < 0.001 |
| Albumin (g/dL) | 3.9 ± 0.6 | 3.8 ± 0.4 | 0.302 |
| Total bilirubin (g/dL) | 0.7 ± 0.3 | 0.9 ± 0.3 | 0.011 |
| Aspartate aminotransferase (U/L) | 42.3 ± 48.0 | 42.0 ± 21.6 | 0.958 |
| Alanine aminotransferase (U/L) | 28.1 ± 32.4 | 34.1 ± 23.2 | 0.216 |
| Gamma-glutamyl transpeptidase (U/L) | 114 ± 171 | 81.4 ± 82.1 | 0.249 |
| Prothrombin time (%) | 101 ± 17.7 | 87.7 ± 17.7 | < 0.001 |
| Alpha-fetoprotein (ng/mL) | 2981 ± 6988 | 2804 ± 18478 | 0.956 |
| Des-gamma-carboxy prothrombin (mAU/mL) | 34031 ± 138072 | 4333 ± 28027 | 0.222 |
| Albumin-bilirubin grade | -2.61 ± 0.46 | -2.46 ± 0.39 | 0.056 |
| Fibrosis-4 index | 3.14 ± 2.52 | 5.47 ± 2.99 | < 0.001 |
| Aspartate aminotransferase to platelet ratio index | 0.74 | 1.45 | < 0.001 |
| Hypertension (yes/no) | 28/14 | 59/46 | 0.243 |
| Diabetes mellitus (yes/no) | 19/23 | 35/70 | 0.176 |
| Cardiovascular disease (yes/no) | 10/32 | 16/89 | 0.219 |
| Significant alcohol use (yes/no) | 16/26 | 37/68 | 0.744 |
| Etiology (B/C/negative for both hepatitis B and C) | 5/4/33 | 22/54/30 | < 0.001 |
| Tumor number (solitary/multiple) | 37/5 | 79/26 | 0.371 |
| Largest tumor diameter (mm) | 84.7 ± 44.2 | 57.2 ± 37.6 | 0.035 |
| Tumor differentiation (well/moderate/poor) | 13/26/3 | 20/65/12 | 0.337 |
| Vascular invasion (yes/no) | 26/16 | 84/21 | 0.034 |
Regarding tumor characteristics, the maximum tumor diameter was significantly larger in cases of F0 HCC (P = 0.035), although both groups had a similar proportion of solitary vs multiple tumors. Additionally, vascular invasion was significantly more frequent in the F0 group (P = 0.034), while tumor differentiation did not differ significantly between the two groups.
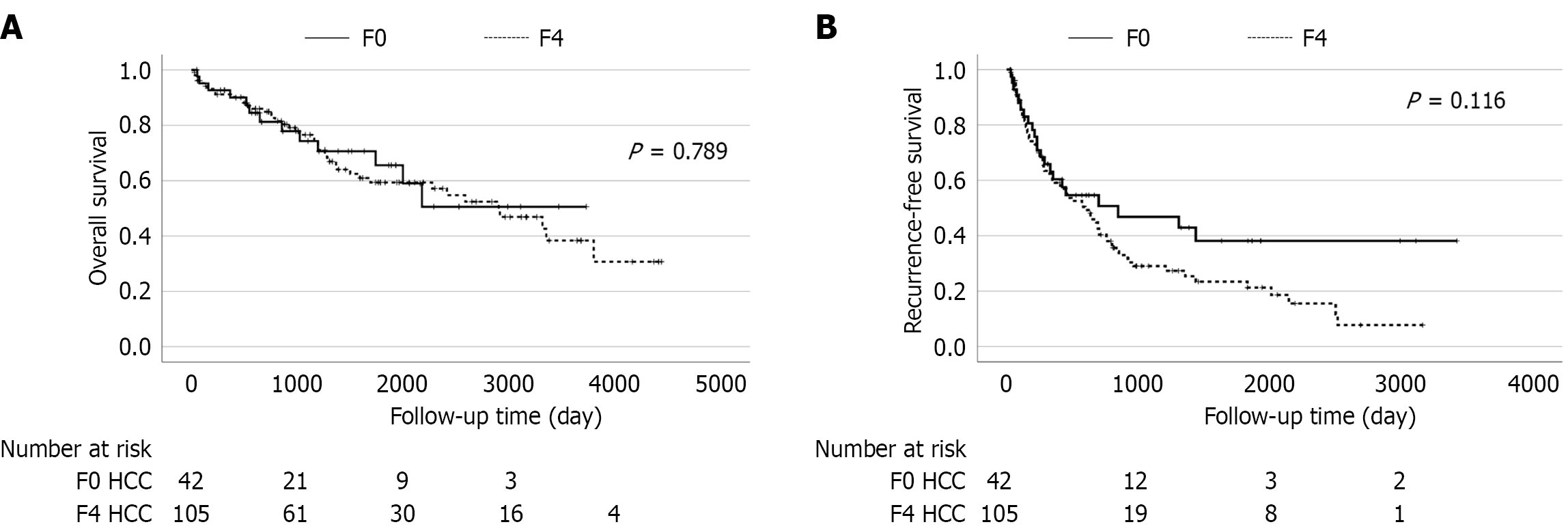
Cases of F0 and F4 HCC had no significant difference in OS (P = 0.789; Figure 1A). A total of 13 patients died in the F0 group during the observation period. Of them, 8 deaths were attributable to HCC progression, while 5 were due to non-liver-related causes. Notably, no liver failure-related deaths were observed in the F0 group. By contrast, 42 deaths occurred in the F4 group, comprising 26 from HCC progression, 7 from liver failure, and 9 from other causes. There were 22 cases and 73 cases of recurrence observed in the F0 and F4 groups, respectively, although there was no significant difference in RFS (P = 0.116; Figure 1B).
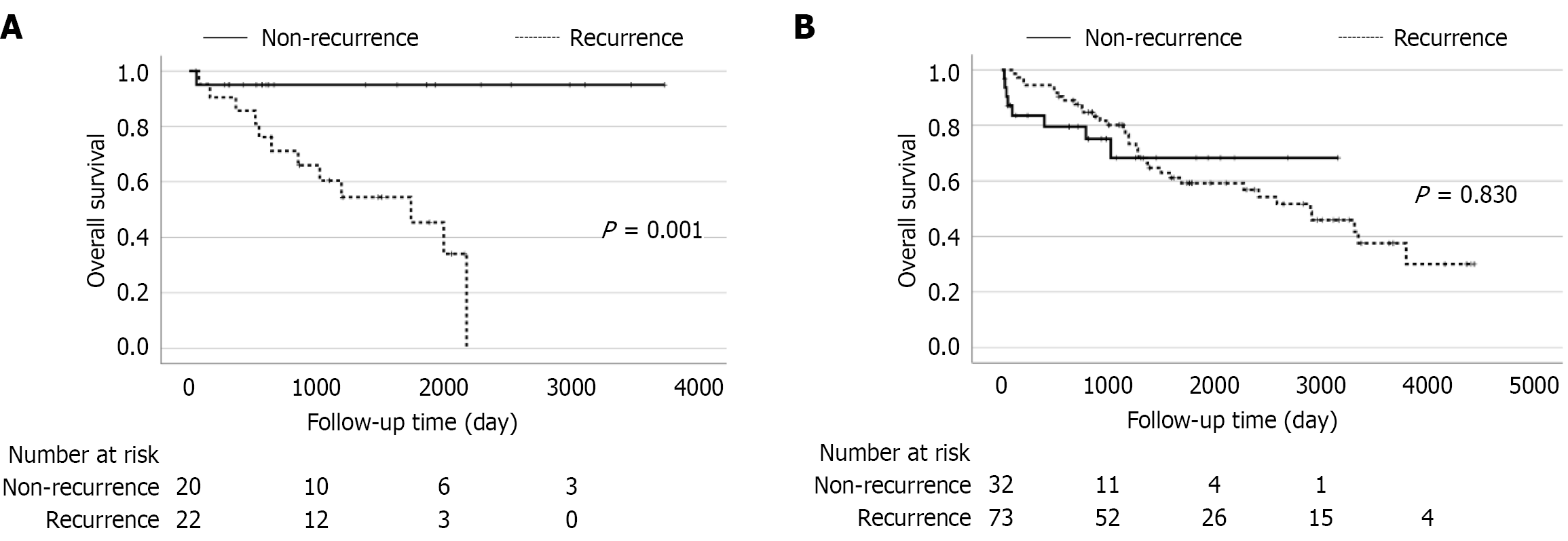
Among cases of F0 HCC, OS was significantly shorter among cases with recurrence vs those without (P = 0.001; Figure 2A), indicating the strong negative impact of recurrence on the survival of these patients. However, among cases of F4 HCC, OS was similar regardless of recurrence (Figure 2B).
As a sensitivity analysis, after adjustment for age and sex, recurrence remained a significant independent predictor of OS in patients with F0 HCC (P = 0.003). Sex was also significantly associated with OS (P = 0.009), whereas age was not a significant predictor.
A Cox proportional hazards model was used to identify factors contributing to recurrence in patients with F0 HCC. On univariate analysis, the significant risk factors for recurrence were ALT, hemoglobin A1c (HbA1c), tumor number, maximum tumor diameter and vascular invasion (P < 0.05). On multivariate analysis, only tumor number remained an independent risk factor for recurrence (P < 0.05) (Table 2).
| Univariate analysis | Multivariate analysis | |||
| HR (95%CI) | P value | HR (95%CI) | P value | |
| Sex (male) | 1.261 (0.606-1.261) | 0.535 | ||
| Age | 1.031 (0.974-1.092) | 0.292 | ||
| Body mass index (kg/m2) | 0.906 (0.788-1.042) | 0.166 | ||
| Platelet count (104/μL) | 0.990 (0.907-1.082) | 0.828 | ||
| Albumin (g/dL) | 0.538 (0.231-1.255) | 0.151 | ||
| Total bilirubin (g/dL) | 1.809 (0.473-6.925) | 0.386 | ||
| Aspartate aminotransferase (U/L) | 1.005 (0.999-1.010) | 0.122 | ||
| Alanine aminotransferase (U/L) | 1.021 (1.005-1.037) | 0.008 | 1.010 (0.990-1.031) | 0.319 |
| Gamma-glutamyl transpeptidase (U/L) | 1.001 (0.999-1.003) | 0.195 | ||
| Prothrombin time (%) | 0.997 (0.951-1.004) | 0.089 | ||
| Alpha-fetoprotein (ng/mL) | 1.000 (1.000-1.000) | 0.810 | ||
| Des-gamma-carboxy prothrombin (mAU/mL) | 1.000 (1.000-1.000) | 0.625 | ||
| Hemoglobin A1c (%) | 0.440 (0.205-0.945) | 0.035 | 0.624 (0.310-1.254) | 0.185 |
| Albumin-bilirubin grade | 2.568 (0.978-6.742) | 0.056 | ||
| Fibrosis-4 index | 1.108 (0.987-1.244) | 0.083 | ||
| Hypertension (yes) | 1.232 (0.489-3.103) | 0.658 | ||
| Diabetes mellitus (yes) | 1.938 (0.771-4.869) | 0.159 | ||
| Cardiovascular disease (yes) | 0.641 (0.245-1.673) | 0.363 | ||
| Smoking (yes) | 1.265 (0.516-3.101) | 0.608 | ||
| Significant alcohol use (yes) | 1.334 (0.531-3.350) | 0.540 | ||
| Negative for both hepatitis B and C (yes) | 0.785 (0.262-2.353) | 0.666 | ||
| Hepatitis C virus (yes) | 25.586 (0.083-7906) | 0.268 | ||
| Tumor number (multiple) | 5.217 (1.801-15.117) | 0.002 | 4.452 (1.288-15.393) | 0.018 |
| Largest tumor diameter(mm) | 1.012 (1.001-1.022) | 0.028 | 1.003 (0.989-1.018) | 0.269 |
| Vascular invasion (yes) | 2.530 (1.077-5.940) | 0.033 | 2.133 (0.644-7.062) | 0.682 |
This study provides a comprehensive analysis of HCC in nonfibrotic (F0) livers, focusing on clinical characteristics, prognostic outcomes, and recurrence risk factors. Our findings highlight the key differences between F0 and F4 HCC, emphasizing the unique biological behavior of tumors in non-cirrhotic backgrounds.
Regarding background characteristics, F0 HCC was more frequently observed in older male patients, with a higher prevalence of NBNC etiology and a larger tumor size compared to F4 HCC. This clinical profile supports the hypothesis that F0 HCC follows a different oncogenic pathway, possibly influenced by metabolic or environmental factors, rather than fibrosis-associated mechanisms. Consistent with this hypothesis, HCC arising in nonfibrotic livers suggests the involvement of carcinogenic mechanisms distinct from the traditional inflammation-fibrosis-cirrhosis sequence[8,21]. Metabolic dysfunction constitutes one such mechanism, as metabolic syndrome and metabolic dysfunction-associated steatotic liver disease have been associated with an increased risk of HCC even without advanced fibrosis[22]. Insulin resistance, chronic inflammation, and oxidative stress may promote hepatocarcinogenesis independently of fibrosis progression[23].
Another important mechanism relevant to F0-HCC is direct viral carcinogenesis mediated by hepatitis B virus DNA integration. Unlike hepatitis C virus-related hepatocarcinogenesis, hepatitis B virus can induce malignant transformation through genomic integration, resulting in genomic instability and oncogene activation without the requirement for advanced fibrosis[24].
In addition to metabolic and viral factors, genetic and epigenetic alterations may play a critical role in the pathogenesis of F0-HCC. Tumor-intrinsic molecular abnormalities, including somatic mutations and epigenetic dysregulation, are recognized as central events in hepatocarcinogenesis[25]. Although molecular analyses were not conducted in the present study, future investigations incorporating genomic profiling may help elucidate the biological heterogeneity of F0-HCC.
Consistent with our findings, Naganuma and Ishida[10] also reported a predominance of older male patients with solitary large tumors among cases of F0 HCC. However, their review noted a second, smaller peak in young female patients, which was not observed in our cohort. This difference may reflect population differences or referral biases.
Despite these differences in background and tumor characteristics, the OS and RFS did not significantly differ between F0 and F4 HCC. However, within the F0 HCC group, those without recurrence demonstrated a significantly more favorable prognosis (P = 0.001), emphasizing the critical impact of recurrence on survival. Considering that these patients have well-preserved liver function, recurrence is likely the primary determinant of long-term prognosis in F0 HCC. This interpretation is further supported by differences in the causes of death between the two groups. In the F0 group, most deaths were attributable to tumor progression, and no deaths due to liver failure were observed. By contrast, in the F4 group, 17% of the deaths were related to liver failure, indicating the presence of competing risks distinct from tumor progression. Importantly, the prognostic impact of recurrence in F0 HCC remained significant even after adjustment for age and sex, underscoring the robustness of our findings. This underscores the need for improved surveillance and early intervention strategies in these patients. Using a Cox proportional hazards model, ALT, HbA1c, tumor number, maximum tumor diameter and vascular invasion were significant risk factors for recurrence on univariate analysis, but only tumor number remained significant on multivariate analysis. Thus, tumor burden at diagnosis is a crucial deter
These findings highlight the distinct characteristics of HCC in nonfibrotic livers and its clinical challenges. Given the high recurrence rates and the significant impact of recurrence on OS, enhanced surveillance strategies are urgently needed in this population. In contrast to patients with cirrhosis, those without cirrhosis do not undergo routine imaging-based surveillance, which often leads to a delayed diagnosis. Although the early detection of HCC is crucial for im
There is currently no established strategy for systematically screening individuals at risk for F0 HCC. Additionally, most patients do not undergo regular follow-up for liver disease, making early detection even more difficult. Therefore, collaboration across medical specialties is essential to ensure that patients who are not under the care of hepatologists can still undergo appropriate imaging for early detection. Increasing awareness among general practitioners, diabetologists, and gastroenterologists regarding the risk of HCC in non-cirrhotic individuals can help in the earlier identification of the disease. In 2023, the Japan Society of Hepatology issued the Nara Declaration, an initiative that encourages the general public to seek medical consultation if their serum ALT levels exceed 30 U/L, as well as advises primary care physicians to perform appropriate evaluations and refer patients to gastroenterologists when necessary. These efforts aimed to promote the early detection of chronic liver diseases, including those associated with metabolic syndrome, alcohol use, or immune-mediated liver injury, helping prevent the development of cirrhosis and HCC. These early detection strategies can facilitate the diagnosis of nonfibrotic HCC cases that would otherwise go unnoticed. This underscores the critical need for interdisciplinary cooperation to improve the early diagnosis and outcomes of this unique patient population.
By strictly defining fibrosis stage through histological evaluation of resected background liver specimens and focusing on recurrence-driven prognosis in F0 HCC, the present study provides a refined clinical framework for understanding outcomes in this distinct patient population. Although related to prior fibrosis studies, this work shifts the emphasis toward prognostic stratification within histologically confirmed F0 HCC[26,27].
Several limitations should be acknowledged. First, the retrospective study design could introduce selection bias. Second, the relatively small sample size of F0 HCC cases compared to the F4 cases potentially limits the statistical power of our findings. Although propensity score matching based on age and sex was explored to address baseline imbalances, the matched cohort was substantially reduced in size and therefore was not adopted as the primary analytical approach. Third, molecular analyses were not performed, and thus, the genetic drivers of HCC in F0 livers remain unclear. To validate these findings, future prospective studies should incorporate larger cohorts and include molecular profiling.
This study provides valuable insights into the clinical characteristics, prognosis, and recurrence risk factors of HCC in nonfibrotic livers. Compared to F4 HCC, cases of F0 HCC were more common in older male patients, characterized by an NBNC etiology and larger tumor. Although OS and RFS were comparable between F0 and F4 HCC, recurrence sig
The authors are grateful to the pathology team at Oita University for their expertise in histological assessment of liver fibrosis.
| 1. | McGlynn KA, Petrick JL, El-Serag HB. Epidemiology of Hepatocellular Carcinoma. Hepatology. 2021;73 Suppl 1:4-13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1711] [Cited by in RCA: 1556] [Article Influence: 311.2] [Reference Citation Analysis (8)] |
| 2. | Samant H, Amiri HS, Zibari GB. Addressing the worldwide hepatocellular carcinoma: epidemiology, prevention and management. J Gastrointest Oncol. 2021;12:S361-S373. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 188] [Cited by in RCA: 144] [Article Influence: 28.8] [Reference Citation Analysis (2)] |
| 3. | Fattovich G, Stroffolini T, Zagni I, Donato F. Hepatocellular carcinoma in cirrhosis: incidence and risk factors. Gastroenterology. 2004;127:S35-S50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1979] [Cited by in RCA: 1789] [Article Influence: 81.3] [Reference Citation Analysis (5)] |
| 4. | El-Serag HB. Hepatocellular carcinoma. N Engl J Med. 2011;365:1118-1127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3139] [Cited by in RCA: 3069] [Article Influence: 204.6] [Reference Citation Analysis (1)] |
| 5. | Zhang DY, Friedman SL. Fibrosis-dependent mechanisms of hepatocarcinogenesis. Hepatology. 2012;56:769-775. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 373] [Cited by in RCA: 340] [Article Influence: 24.3] [Reference Citation Analysis (1)] |
| 6. | Baglieri J, Brenner DA, Kisseleva T. The Role of Fibrosis and Liver-Associated Fibroblasts in the Pathogenesis of Hepatocellular Carcinoma. Int J Mol Sci. 2019;20:1723. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 275] [Cited by in RCA: 261] [Article Influence: 37.3] [Reference Citation Analysis (0)] |
| 7. | Roehlen N, Crouchet E, Baumert TF. Liver Fibrosis: Mechanistic Concepts and Therapeutic Perspectives. Cells. 2020;9:875. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1035] [Cited by in RCA: 909] [Article Influence: 151.5] [Reference Citation Analysis (2)] |
| 8. | Bralet MP, Régimbeau JM, Pineau P, Dubois S, Loas G, Degos F, Valla D, Belghiti J, Degott C, Terris B. Hepatocellular carcinoma occurring in nonfibrotic liver: epidemiologic and histopathologic analysis of 80 French cases. Hepatology. 2000;32:200-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 178] [Cited by in RCA: 162] [Article Influence: 6.2] [Reference Citation Analysis (1)] |
| 9. | Desai A, Sandhu S, Lai JP, Sandhu DS. Hepatocellular carcinoma in non-cirrhotic liver: A comprehensive review. World J Hepatol. 2019;11:1-18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 259] [Cited by in RCA: 228] [Article Influence: 32.6] [Reference Citation Analysis (1)] |
| 10. | Naganuma H, Ishida H. Hepatocellular Carcinoma in Non-Fibrotic Liver: A Narrative Review. Diagnostics (Basel). 2023;13:3426. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 11. | Schulze K, Imbeaud S, Letouzé E, Alexandrov LB, Calderaro J, Rebouissou S, Couchy G, Meiller C, Shinde J, Soysouvanh F, Calatayud AL, Pinyol R, Pelletier L, Balabaud C, Laurent A, Blanc JF, Mazzaferro V, Calvo F, Villanueva A, Nault JC, Bioulac-Sage P, Stratton MR, Llovet JM, Zucman-Rossi J. Exome sequencing of hepatocellular carcinomas identifies new mutational signatures and potential therapeutic targets. Nat Genet. 2015;47:505-511. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1516] [Cited by in RCA: 1407] [Article Influence: 127.9] [Reference Citation Analysis (1)] |
| 12. | Dyson J, Jaques B, Chattopadyhay D, Lochan R, Graham J, Das D, Aslam T, Patanwala I, Gaggar S, Cole M, Sumpter K, Stewart S, Rose J, Hudson M, Manas D, Reeves HL. Hepatocellular cancer: the impact of obesity, type 2 diabetes and a multidisciplinary team. J Hepatol. 2014;60:110-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 509] [Cited by in RCA: 463] [Article Influence: 38.6] [Reference Citation Analysis (2)] |
| 13. | Kensler TW, Roebuck BD, Wogan GN, Groopman JD. Aflatoxin: a 50-year odyssey of mechanistic and translational toxicology. Toxicol Sci. 2011;120 Suppl 1:S28-S48. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 564] [Cited by in RCA: 428] [Article Influence: 28.5] [Reference Citation Analysis (0)] |
| 14. | Bedossa P, Poynard T. An algorithm for the grading of activity in chronic hepatitis C. The METAVIR Cooperative Study Group. Hepatology. 1996;24:289-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3413] [Cited by in RCA: 3112] [Article Influence: 103.7] [Reference Citation Analysis (1)] |
| 15. | Van Roey G, Fevery J, Van Steenbergen W. Hepatocellular carcinoma in Belgium: clinical and virological characteristics of 154 consecutive cirrhotic and non-cirrhotic patients. Eur J Gastroenterol Hepatol. 2000;12:61-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 16] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 16. | Okuda K, Nakashima T, Kojiro M, Kondo Y, Wada K. Hepatocellular carcinoma without cirrhosis in Japanese patients. Gastroenterology. 1989;97:140-146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 76] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 17. | Pinyopornpanish K, Al-Yaman W, Dasarathy S, Romero-Marrero C, McCullough A. Hepatocellular Carcinoma in Patients Without Cirrhosis: The Fibrosis Stage Distribution, Characteristics and Survival. Dig Dis Sci. 2022;67:2677-2687. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 18. | Yip VS, Gomez D, Tan CY, Staettner S, Terlizzo M, Fenwick S, Malik HZ, Ghaneh P, Poston G. Tumour size and differentiation predict survival after liver resection for hepatocellular carcinoma arising from non-cirrhotic and non-fibrotic liver: a case-controlled study. Int J Surg. 2013;11:1078-1082. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 29] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 19. | Gaddikeri S, McNeeley MF, Wang CL, Bhargava P, Dighe MK, Yeh MM, Dubinsky TJ, Kolokythas O, Lalwani N. Hepatocellular carcinoma in the noncirrhotic liver. AJR Am J Roentgenol. 2014;203:W34-W47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 65] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 20. | Ichida F, Tsuji T, Omata M, Ichida T, Inoue K, Kamimura T, Yamada G, Hino K, Yokosuka O, Suzuki H. New Inuyama classification; new criteria for histological assessment of chronic hepatitis. Int Hepatol Commun. 1996;6:112-119. [RCA] [DOI] [Full Text] [Cited by in Crossref: 253] [Cited by in RCA: 276] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 21. | Aryan M, Ruli T, Shoreibah M. HCC in patients without cirrhosis: A review. Clin Liver Dis (Hoboken). 2024;23:e0224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 22. | Piscaglia F, Svegliati-Baroni G, Barchetti A, Pecorelli A, Marinelli S, Tiribelli C, Bellentani S; HCC-NAFLD Italian Study Group. Clinical patterns of hepatocellular carcinoma in nonalcoholic fatty liver disease: A multicenter prospective study. Hepatology. 2016;63:827-838. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 501] [Cited by in RCA: 484] [Article Influence: 48.4] [Reference Citation Analysis (1)] |
| 23. | Baffy G, Brunt EM, Caldwell SH. Hepatocellular carcinoma in non-alcoholic fatty liver disease: an emerging menace. J Hepatol. 2012;56:1384-1391. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 712] [Cited by in RCA: 659] [Article Influence: 47.1] [Reference Citation Analysis (1)] |
| 24. | Ma L, Chen S, Wang H, Chen L. Hepatitis B virus integration and hepatocarcinogenesis. Liver Res. 2025;9:189-198. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 25. | Lin HY, Jeon AJ, Chen K, Lee CJM, Wu L, Chong SL, Anene-Nzelu CG, Foo RS, Chow PK. The epigenetic basis of hepatocellular carcinoma - mechanisms and potential directions for biomarkers and therapeutics. Br J Cancer. 2025;132:869-887. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 26] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 26. | Yasukawa K, Shimizu A, Kubota K, Notake T, Hosoda K, Hayashi H, Soejima Y. Impact of Liver Fibrosis Severity on Oncological Prognosis in Hepatocellular Carcinoma. Liver Cancer. 2024;13:150-160. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 27. | Kamarajah SK. Fibrosis score impacts survival following resection for hepatocellular carcinoma (HCC): A Surveillance, End Results and Epidemiology (SEER) database analysis. Asian J Surg. 2018;41:551-561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 2.6] [Reference Citation Analysis (1)] |