Published online Apr 15, 2026. doi: 10.4251/wjgo.v18.i4.115511
Revised: December 9, 2025
Accepted: January 16, 2026
Published online: April 15, 2026
Processing time: 172 Days and 10.1 Hours
This narrative review examines the role of dietary patterns in colorectal cancer (CRC) development and management, with a focus on the diet-microbiome-metabolic reprogramming axis. CRC is one of the most pre
Core Tip: Dietary modulation may represent a relevant preventive and supportive strategy in colorectal cancer. Dietary patterns characterized by higher intake of fiber and bioactive compounds, such as those derived from whole grains, fruits, and vegetables, and lower consumption of ultra-processed foods have been associated with improved intestinal and metabolic homeostasis. In selected patients and under professional supervision, metabolically targeted interventions such as time-restricted eating, caloric restriction, or fasting-mimicking diets may be considered as adjunctive approaches, based on their mechanistic links to tumor metabolism, inflammation, and host metabolic reprogramming. These strategies should be individualized, carefully monitored, and applied as complements to standard oncologic care.
- Citation: Lima RSP, Sousa JDS, Neres MSO, de Sousa DJM, Martins JA, Pereira IC, da Silva ACA, Severo JS, Torres-Leal FL, da Silva MTB. Colorectal cancer therapy and nutrition: From ultra-processed consumption to metabolic reprogramming. World J Gastrointest Oncol 2026; 18(4): 115511
- URL: https://www.wjgnet.com/1948-5204/full/v18/i4/115511.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i4.115511
Colorectal cancer (CRC) represents a significant global health burden, ranking third in incidence and second in mortality among all cancers[1]. Epidemiological projections suggest an alarming rise, with incidence and mortality rates expected to increase by 60% and 71.5%, respectively, by 2035[2,3]. The pathogenesis of CRC involves a complex interplay between clinical, environmental, and genetic determinants, underscoring its multifactorial nature[4,5]. Notably, growing evidence has identified diet as a critical modulator of these processes, linking nutritional patterns to both carcinogenesis and therapeutic responsiveness.
Diet plays a central role in CRC risk, with evidence suggesting that dietary factors may contribute substantially to disease burden. Meta-analysis and systematic reviews of observational studies indicate that unfavourable dietary patterns are associated with an increased risk of CRC. In contrast, healthier dietary behaviours are linked to a reduced risk[6,7]. A systematic review based on data from the European Prospective Investigation into Cancer and Nutrition cohort reported positive associations between alcohol consumption and CRC risk. In contrast, a higher intake of fruits and vegetables, fish, calcium, and yoghurt, as well as lower consumption of red and processed meat, was associated with a reduced risk of CRC[6]. Similar associations were confirmed in an umbrella review of meta-analyses[7]. These findings support diet as a modifiable factor in CRC, providing a conceptual basis for examining both risk-promoting and protective dietary exposures.
In this context, the increasing consumption of ultra-processed foods (UPFs) has emerged as a relevant dietary risk factor for CRC[8-10]. UPFs are defined as “formulations made mostly or entirely from industrial sources of dietary energy and nutrients, plus additives, using a series of processes”[11]. High UPF intake has been mechanistically associated with weight gain, intestinal inflammation, and oxidative stress. In line with these findings, our research group has demonstrated that patients with inflammatory bowel disease exhibit elevated UPF consumption, which is correlated with oxidative stress markers, such as malondialdehyde[12]. However, these findings should be interpreted with caution, given the observational study design and the limited sample size. Considering that inflammatory bowel diseases are well-established risk factors for colorectal carcinogenesis, the paucity of data on UPF intake among CRC patients represents a significant knowledge gap. Two recent systematic reviews have reinforced this association, indicating that higher UPF consumption is associated with an increased risk of CRC[8,9]. Although these findings are primarily based on observational data, they consistently support an association rather than direct causation.
Mechanistically, several ingredients present in UPFs, such as emulsifiers, artificial sweeteners, colorants, and nanoparticles, disrupt intestinal barrier integrity[13]. These compounds have been shown to compromise luminal and mucosal homeostasis by altering microbial abundance and diversity, degrading the mucus layer, impairing epithelial tight junctions, and triggering pro-inflammatory signaling cascades[14]. In this framework, dietary patterns emerge as key determinants of gut health, capable of modulating both protective and deleterious effects that influence CRC risk, clinical outcomes, and therapeutic response in experimental and early clinical settings[15,16].
Taken together, these findings highlight diet as a bidirectional modulator of CRC risk and progression, capable of influencing both carcinogenic processes and therapeutic responsiveness. While UPF-rich dietary patterns may contribute to an unfavourable metabolic and inflammatory milieu[14-16], metabolically targeted nutritional interventions aim to modulate these same biological pathways[17,18] therapeutically. This integrative framework provides the rationale for examining dietary risk factors and metabolic interventions within a unified model of dietary modulation in CRC.
Within this conceptual framework, nutritional interventions have gained prominence as potential therapeutic strategies in CRC, aligning with integrative and personalized medicine approaches, primarily based on preclinical and early-phase clinical evidence. Among these, caloric restriction (CR), fasting, fasting-mimicking diets (FMDs), and ketogenic diets (KD) have demonstrated tumor-suppressive potential through metabolic reprogramming and stress response modulation[17,18]. These dietary interventions vary in intensity, from restrictive FMDs with limited adherence to intermittent or overnight fasting protocols that reduce caloric intake, promote ketone body synthesis, lower plasma glucose and insulin levels, and downregulate IGF-1[19]. Such metabolic adaptations create conditions unfavorable to cancer cell proliferation while enhancing the efficacy of anticancer therapies. Moreover, fasting induces a differential stress response that protects normal cells, fosters tissue regeneration, and mitigates chemotherapy-induced toxicity[20].
Despite these advances, the mechanistic interplay between dietary patterns, metabolic adaptation, and tumor progression in CRC remains insufficiently elucidated. This review, therefore, aims to integrate current evidence on dietary influences in CRC, spanning from risk-promoting UPF consumption to metabolic interventions with therapeutic potential. The central hypothesis guiding this review is that dietary modulation exerts a bidirectional influence on CRC: While high consumption of UPF may increase risk and promote carcinogenesis, targeted nutritional interventions, such as fasting, FMDs, and KD, may therapeutically reprogram tumor metabolism and enhance treatment responsiveness. Accordingly, this review is structured first to discuss dietary risk factors, followed by mechanistic pathways and metabolically targeted nutritional interventions, with an emphasis on translational relevance.
This narrative review was conducted to integrate heterogeneous evidence addressing dietary risk factors and metabolically targeted nutritional interventions in CRC. Given the marked heterogeneity of study designs, endpoints, and mechanistic scopes across the literature addressing dietary patterns and metabolically targeted interventions in CRC, a systematic review or meta-analysis was deemed inappropriate for the objectives of this manuscript. Instead, a narrative review approach was adopted to enable integrative interpretation across preclinical, translational, and clinical contexts. Evidence was qualitatively synthesized using a hierarchy that prioritized prospective human studies, followed by observational investigations, meta-analyses, and mechanistic preclinical models. Conflicting findings were interpreted through comparative analysis of biological plausibility, methodological rigour, and contextual relevance. Although no formal risk-of-bias tool was applied, this limitation is acknowledged and reflects the conceptual and exploratory nature of the present synthesis.
A comprehensive bibliographic search was conducted in PubMed/MEDLINE, Scopus, ScienceDirect, and Web of Science from database inception to August 2025. It was updated in December 2025, without language restrictions or publication date limitations, to retrieve all available evidence on the topic under investigation. Additional relevant studies were identified through manual searching of the reference lists of included articles. The search strategy combined MeSH terms and free-text keywords related to UPF, food additives, the NOVA classification system, CRC, carcinogenesis, dietary patterns, dietary interventions such as calorie restriction, intermittent fasting (IF), time-restricted feeding, FMDs, KDs, amino acid restriction, and mechanistic concepts including metabolic reprogramming, gut microbiota, immune modulation, and oxidative stress.
Studies were eligible for inclusion if they were original articles involving humans, animal models, or cell-based experiments; if they presented mechanistic, physiological, metabolic, or clinical evidence relevant to CRC or dietary exposures; or if they were reviews, meta-analyses, or consensus documents that provided theoretical or contextual support for the topics addressed. No restrictions were applied regarding the language of publication. Exclusion criteria included conference abstracts, studies that did not assess outcomes related to our research question, and articles based on expert opinions. Overall, 868432 records were screened, and 154 articles were included for qualitative synthesis.
The selection process involved an initial screening of titles and abstracts, followed by full-text assessment of potentially eligible studies. For evidence synthesis, the articles were organized into three categories: (1) Preclinical evidence derived from in vitro or animal studies; (2) Evidence from observational or clinical studies; and (3) Narrative and systematic reviews, with or without meta-analysis. Given the narrative nature of this review, no formal risk-of-bias tool or standardized quality assessment framework was applied.
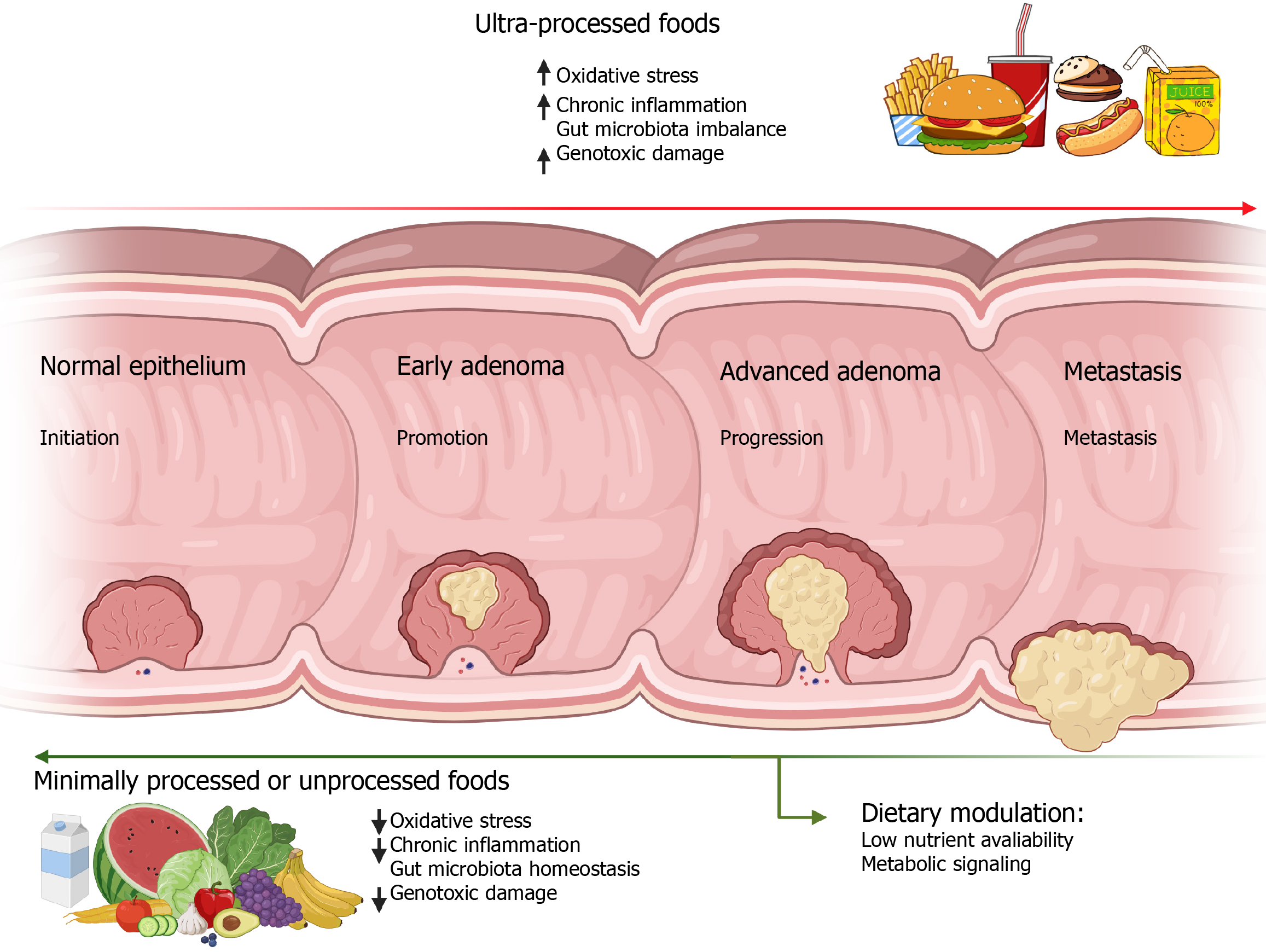
CRC develops through a continuum of genetic and epigenetic alterations in epithelial cells that drive uncontrolled proliferation and disrupt normal tissue homeostasis[21]. This aberrant proliferative state leads to the formation of benign adenomas, which may subsequently progress to malignant adenocarcinomas and metastasize through distinct molecular pathways, including microsatellite instability, chromosomal instability (CIN), and serrated neoplasia[22,23]. The classical adenoma-carcinoma sequence describes this gradual process, beginning with the formation of a polyp or adenoma (stage 0). Approximately 10% of these adenomas undergo malignant transformation, developing into adenocarcinomas that invade the muscularis propria (stage I). Tumor expansion and invasion into the serosa and visceral peritoneum mark stages II and III, respectively, followed by metastatic dissemination through lymphatic or hematogenous routes (stage IV)[21]. Significantly, the transition from benign to malignant lesions is strongly influenced by the local inflammatory and metabolic microenvironment, which dietary exposures may modulate.
The etiological trajectory of CRC represents a multistep biological process comprising four primary stages: Initiation, promotion, progression, and metastasis[24]. The initiation phase is characterized by irreversible genetic alterations, primarily resulting from the formation of DNA adducts during chemical carcinogenesis. These adducts form when carcinogens or their reactive metabolites covalently bind to DNA, generating genotoxic lesions that are detectable even in normal colonic mucosa[25]. DNA repair mechanisms or apoptosis can remove such lesions; however, a dynamic equilibrium between adduct formation and removal persists in target tissues. The sustained accumulation of DNA adducts can trigger mutations in genes that regulate cell proliferation, thereby initiating neoplastic transformation[26]. Diet-related factors, including pro-oxidant compounds, food additives, and microbiota-derived metabolites, may influence this phase by modulating oxidative stress, genotoxic burden, and DNA repair capacity[27-29].
During the promotion phase, these genetically altered cells acquire proliferative advantages, forming benign neoplasms such as adenomas. In the subsequent progression phase, the accumulation of additional genetic and epigenetic events confers invasive and metastatic potential to these lesions. Epigenetic plasticity, influenced by nutrient availability and metabolic signaling, represents a critical interface between environmental exposures and tumor evolution, thereby linking dietary modulation to metabolic reprogramming in CRC. Ultimately, during metastasis, tumor cells disseminate from the primary site to distant organs through either hematogenous or lymphatic routes, establishing secondary tumors[30]. The temporal evolution of these steps varies widely, often spanning several decades, underscoring the prolonged and multifactorial nature of colorectal carcinogenesis.
Evidence from molecular pathological epidemiology further supports the concept that dietary effects on CRC risk are modulated by tumor molecular heterogeneity. In a large prospective cohort, Western dietary patterns, which are characterized by high consumption of UPFs, red and processed meats, refined grains, added sugars, and saturated fats, alongside low intake of fiber-rich foods, fruits, vegetables, and bioactive compounds, were more strongly associated with distal colon and rectal cancers. These associations were particularly evident in tumors characterized by microsatellite stability, wild-type status for KRAS and BRAF genes, and a low or absent CpG island methylator phenotype. In contrast, prudent dietary patterns were associated with a reduced risk of CRC that appeared largely independent of anatomic subsite or molecular subtype. Although formal tests for heterogeneity were not statistically significant, these findings suggest that diet-related metabolic and inflammatory exposures may preferentially influence carcinogenic pathways linked to CIN, thereby the importance of considering molecular context when evaluating dietary modulation in CRC[29]. Figure 1 illustrates how the diet can act as a bidirectional modulator in colorectal carcinogenesis.
Diet constitutes a key modifiable determinant in the prevention and progression of CRC[31]. Parallel to this, UPFs currently account for more than 50% of total caloric intake in industrialized nations such as the United States, Canada, and the United Kingdom[32-34]. UPFs, typically rich in refined sugars and industrial fats, profoundly alter gut microbial composition and diversity, thereby promoting obesity and metabolic dysfunction—conditions mechanistically associated with increased CRC risk[35]. Moreover, these products are generally poor in micronutrients and bioactive compounds with recognized protective roles against colorectal tumorigenesis[36].
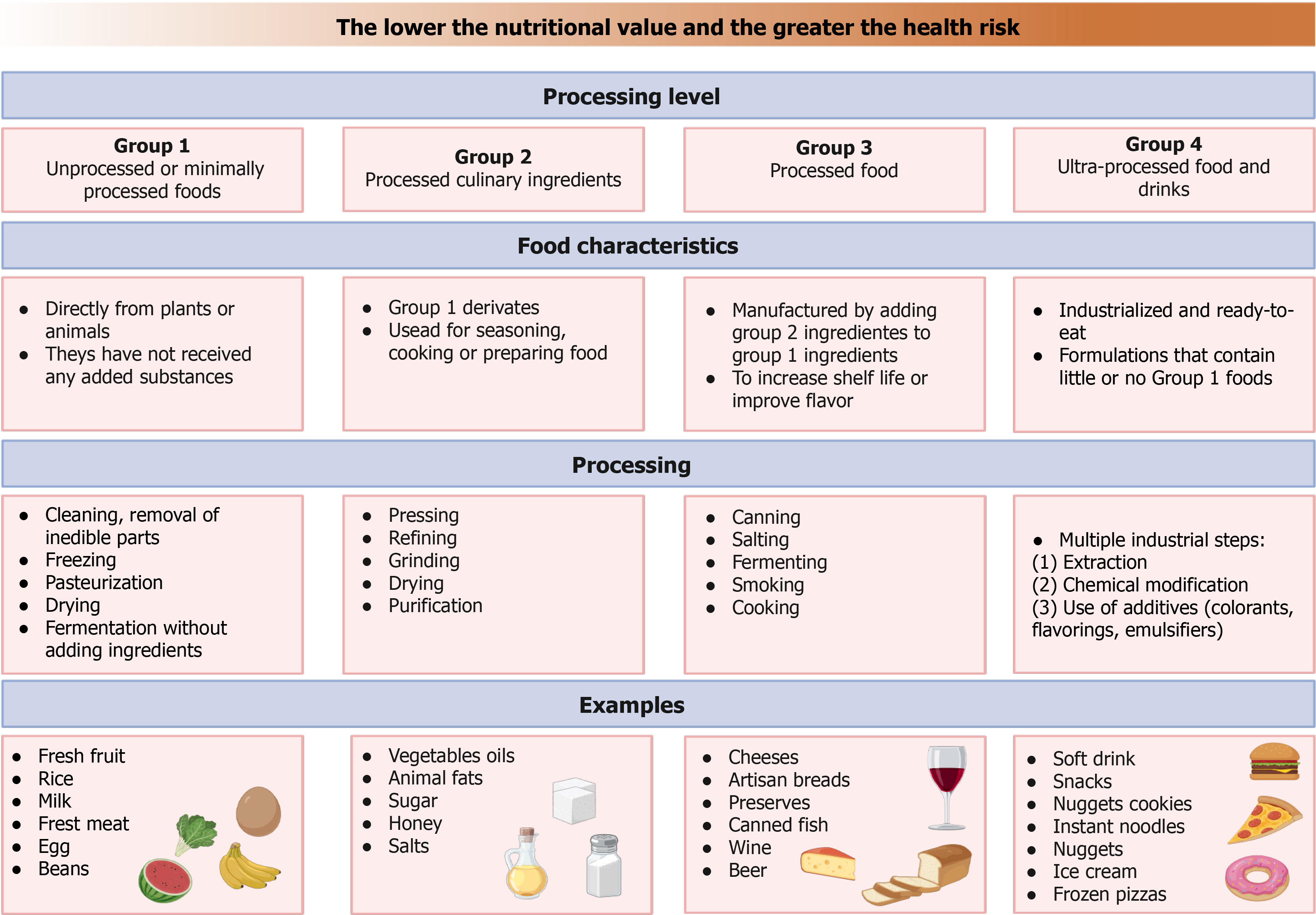
According to the NOVA classification system, foods are categorized into four groups based on the extent and purpose of industrial processing, encompassing physical, biological, and chemical methods, as well as the use of additives[37]. UPFs are defined as complex formulations of ingredients resulting from multiple industrial transformations. Examples include soft drinks, sweet and savory packaged snacks, confectionery, ice creams, industrial breads and cakes, margarines, cookies, pies, breakfast cereals, nuggets, sausages, hamburgers, hot dogs, and other reconstituted products, in addition to powdered or instant soups, noodles, and desserts[12,37]. Figure 2 shows the NOVA classification system.
Characteristic UPFs ingredients fall into two major categories: (1) Food substances rarely or never used in culinary preparations; and (2) Cosmetic additives designed to enhance flavor, color, and texture, often rendering products hyper-palatable. The first group includes refined sugars (fructose, high-fructose corn syrup, fruit juice concentrate, maltodextrin, dextrose, and lactose), industrial oils (hydrogenated or interesterified), and protein isolates (hydrolyzed proteins, soy or whey isolates, gluten, casein, and mechanically processed meats). The second group comprises flavorings, enhancers, colorants, emulsifiers, sweeteners, thickeners, and stabilizers, as well as anti-foaming agents, bulking agents, carbonating agents, and glazing agents. Collectively, these additives mask undesirable sensory properties of ingredients, compensate for nutrient losses during processing, and prolong shelf life, factors that contribute to the metabolic and inflammatory disturbances associated with CRC development[37].
Epidemiological evidence linking UPF consumption to CRC risk is primarily derived from extensive prospective cohort studies, with supportive findings from case-control investigations and meta-analyses. In a prospective cohort study from the United States, men in the highest quintile of UPF consumption had a 29% higher risk of CRC compared with those in the lowest quintile[10]. Similarly, a cohort study including 142052 participants reported a positive association between total UPF intake and CRC risk in men, while specific UPF subgroups were associated with CRC risk in women[36]. Complementing these data, a case-control study conducted in Spain also demonstrated that higher UPF consumption was associated with an increased incidence of CRC[38].
Dose-response relationships further support these associations. A recent meta-analysis integrating data from over half a million participants showed that a 10% increase in UPF consumption was associated with a 4% higher risk of CRC[39]. Subgroup analyses indicated that this association was more pronounced among men. In contrast, no statistically significant relationship was observed in women, highlighting potential sex-specific metabolic and behavioral modifiers of dietary carcinogenic risk. Taken together, these large-scale studies support the notion that UPF consumption may act as an independent nutritional risk factor for CRC. However, residual confounding related to lifestyle clustering cannot be completely ruled out.
Importantly, UPF consumption occurs within a broader context of established CRC risk determinants. Reports from the World Cancer Research Fund and the American Institute for Cancer Research consistently identify smoking, inflammatory bowel disease, obesity, sedentary lifestyle, alcohol intake, and overall dietary patterns as major contributors to CRC risk, alongside protective factors such as physical activity and non-steroidal anti-inflammatory drug use. Height has also emerged as a surrogate marker of early-life growth and hormonal exposures associated with CRC risk. These associations, supported by case-control, cohort, and meta-analytical studies, underscore the multifactorial nature of CRC and emphasize the relevance of dietary modulation within an integrated risk framework[40].
Although a growing body of epidemiological evidence supports an association between UPF consumption and CRC risk, findings are not entirely consistent across studies. Several investigations report attenuated, sex-specific, or statistically null associations after adjustment for socioeconomic status, lifestyle clustering, and overall dietary quality[10,36,39]. These inconsistencies highlight the associative rather than causal nature of the current evidence, suggesting that UPF intake may act as both a potential independent exposure and a marker of broader unfavorable dietary and metabolic patterns. Accordingly, the consumption of UPFs should be interpreted within a multifactorial risk framework rather than as an isolated determinant of colorectal carcinogenesis.
Food additives widely used in UPFs, including emulsifiers, sweeteners, colorants, microparticles, and nanoparticles, have been shown to influence intestinal homeostasis by altering the gut microbiota, increasing intestinal permeability, and promoting inflammatory responses[14]. A controlled feeding trial demonstrated that the consumption of carboxymethylcellulose, a commonly used emulsifier, was sufficient to induce postprandial abdominal discomfort, disrupt microbial diversity, and alter faecal metabolomic profiles in healthy adults[41]. Such findings underscore the ability of individual additives to provoke subclinical inflammation and perturb gut microbial ecology, thereby facilitating carcinogenic microenvironments. However, the doses and exposure patterns used in experimental and short-term feeding studies may not fully reflect chronic, real-world dietary exposure.
Food colorants represent another class of concern, as they lack nutritional value and are primarily used to enhance the appearance of food[14]. Experimental models investigating Red-40 and Yellow-6 revealed that, although these dyes do not independently induce colitis, they exacerbate inflammation in genetically susceptible hosts with elevated interleukin-23 signaling. This effect is microbiota-dependent, requiring bacterial metabolism for activation[42-44]. Collectively, these results highlight the interplay between dietary additives, immune signaling, and microbial metabolism in shaping the intestinal inflammatory landscape. However, in these studies, synthetic colorants such as Red-40 and Yellow-6 or other azo dyes are typically tested at micromolar to millimolar concentrations in cell-based systems or administered acutely via diet supplementation or gavage in murine models, with exposure durations ranging from hours to a few weeks. Observed inflammatory or genotoxic effects generally occur under high-dose conditions or in genetically susceptible hosts, while chronic, low-dose exposure within regulatory limits is not directly assessed. Thus, although these protocols provide mechanistic insight, their translational relevance to habitual human dietary exposure remains limited.
Artificial sweeteners have also been mechanistically associated with metabolic dysregulation. Human studies indicate that non-caloric sweeteners are associated with features of metabolic syndrome, including weight gain, increased waist-to-hip ratio, elevated fasting glucose levels, and higher glycated haemoglobin and alanine aminotransferase levels. These effects exhibit high interindividual variability, likely reflecting differences in gut microbiota composition, functional capacity, and responsiveness to dietary stimuli[45]. These effects exhibit substantial interindividual variability, likely reflecting differences in baseline microbiota composition, functional capacity, and microbiome resilience. Additionally, these findings are derived from short-duration, controlled exposure protocols and do not capture chronic, low-dose dietary intake or long-term carcinogenic outcomes.
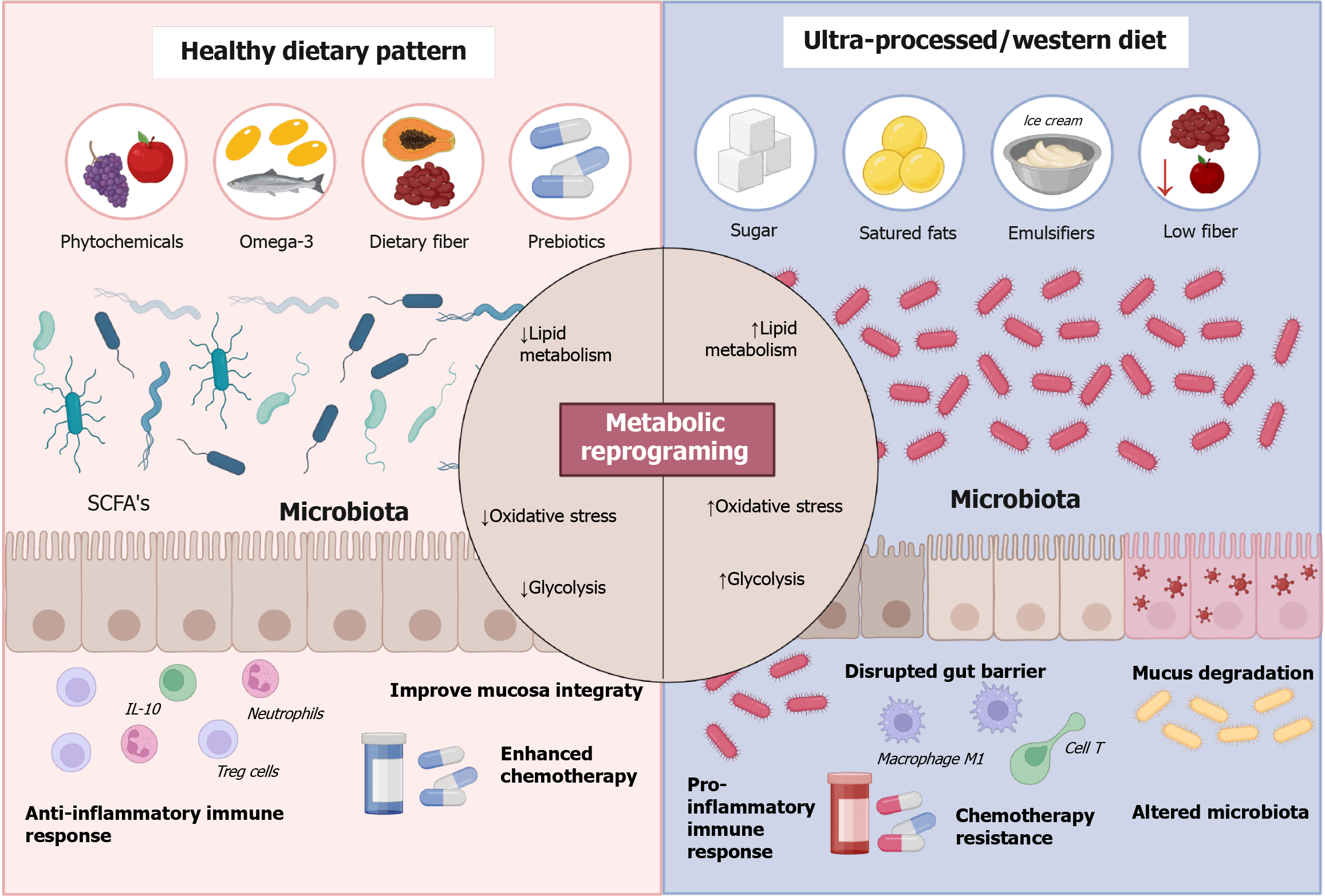
High UPF consumption further contributes to reduced microbial diversity, characterized by the enrichment of the Ruminococcus, Faecalibacterium, Bifidobacterium, Alistipes, Blautia, and Bilophila genera[46]. This dysbiosis, coupled with the typically low fibre content of UPF-based diets, results in diminished short-chain fatty acid production, which is key to metabolites involved in anti-inflammatory and anti-tumorigenic signaling pathways[47]. Together, these findings support a mechanistic cascade in which additive-induced barrier disruption promotes dysbiosis, immune activation, and chronic inflammation, creating a microenvironment conducive to colorectal tumor initiation and promotion. Importantly, not all microbiota alterations represent irreversible carcinogenic remodeling, as transient dysbiosis may be reversible through dietary modification, whereas sustained exposure to UPF-rich diets may promote long-term pro-tumorigenic changes.
Additional mechanistic insights involve the metabolic conversion of red meat-derived compounds, such as choline and carnitine, into trimethylamine (TMA) by gut microbes, which is subsequently oxidized in the liver to TMA N-oxide, a metabolite associated with chronic diseases and carcinogenic processes[48]. Other food stabilizers, such as carrageenan, a commonly used thickener and gelling agent, disrupt the mucus layer and promote intestinal inflammation, further altering microbial composition and barrier integrity[47]. Collectively, these mechanistic findings suggest that chronic exposure to UPF-derived additives promotes a pro-inflammatory, metabolically altered intestinal milieu that is conducive to colorectal carcinogenesis.
Notably, food additive safety assessments are typically conducted for individual compounds and short-term toxicity, with limited consideration of chronic exposure, combined effects, or microbiota-mediated outcomes. These regulatory gaps may underestimate long-term biological effects relevant to colorectal carcinogenesis.
The gut microbiota exerts a profound influence on colorectal tumorigenesis through multiple mechanisms, including genotoxic activity, immune modulation, and the regulation of epithelial proliferation and apoptosis[49]. Advances in metagenomic sequencing have revealed substantial alterations in the microbial composition of patients with CRC compared with healthy controls[50]. These alterations are characterized by an expansion of the oral cavity, associated taxa, and a depletion of butyrate-producing species, which play key roles in maintaining mucosal integrity and regulating inflammation. Moreover, microbial diversity and composition appear to vary according to tumor location and molecular subtype, suggesting that specific microbial signatures may underlie distinct CRC phenotypes[51-53].
Functional metagenomic analyses indicate that the metabolic capacity of the intestinal microbiota in CRC is markedly altered, with enrichment of putrefaction-related pathways, particularly genes involved in ornithine and arginine metabolism, accompanied by a reduction in carbohydrate fermentation pathways observed in tumor-free individuals[51,52,54]. Specific bacterial metabolites, including colibactin produced by pks+ Escherichia coli, the cytolethal distending toxin from Gram-negative species such as Campylobacter jejuni, and indolamines synthesized by Morganella morganii, exert direct genotoxic effects on colonic epithelial cells. These metabolites induce DNA damage, genomic instability, and mutagenic events that promote neoplastic transformation[49].
Microbial regulation of host immunity represents another critical interface linking dysbiosis to colorectal carcinogenesis. Pro-tumorigenic species, including enterotoxigenic Bacteroides fragilis (B. fragilis), Fusobacterium nucleatum, and Clostridioides difficile, stimulate mucosal T helper 17 cell activation and interleukin-17 (IL-17) secretion, fostering epithelial proliferation and chemokine production. IL-17-mediated signaling recruits polymorphonuclear neutrophils (PMNs) and myeloid-derived suppressor cells (MDSCs), which amplify local inflammation and suppress antitumor immune responses, thereby facilitating tumor progression[49]. Additionally, these microorganisms activate oncogenic pathways through virulence factors such as B. fragilis toxin, FadA, and PCWBR2, which interact with epithelial receptors to trigger PI3K-Akt, Wnt-β-catenin, and MAPK-NF-κB signaling cascades[49]. Collectively, these findings underscore the dual role of the microbiota in shaping the pro-inflammatory and oncogenic microenvironment that links diet, dysbiosis, and CRC development. Figure 3 shows the effect of diet on risk and progression of CRC.
Fasting has been discussed as a potential strategy to support cancer treatment, including CRC, where a growing body of evidence, primarily preclinical, has prompted the development of clinical studies suggesting both therapeutic potential and safety. These benefits relate to fasting's ability to reprogram systemic metabolism, modulate antitumor immunity, and enhance treatment efficacy[55-57].
For decades, oncology nutrition has focused mainly on concerns related to malnutrition and cancer cachexia. Although these remain central issues, there is increasing interest in strategies that modulate patient metabolism to improve therapeutic effectiveness. Controlled dietary interventions, including IF, prolonged overnight fasting, and FMDs, have demonstrated the capacity to reconfigure tumor energy metabolism and increase responsiveness to chemotherapy, radiotherapy, and immunotherapy in preclinical CRC models and selected early clinical studies[18,58,59]. Mechanistically, fasting induces systemic adaptations that reduce circulating glucose and insulin levels, as well as downregulate IGF-1 signaling pathways, which are essential for tumor growth and survival and are often associated with treatment resistance or toxicity. These metabolic changes promote selective stress resistance in normal cells while increasing tumor cell vulnerability to oxidative and genotoxic stress[20,60].
Additionally, fasting improves mitochondrial efficiency, restricts anabolic fluxes that fuel cell proliferation, and enhances immune cell function within the tumor microenvironment. By integrating metabolic and immunological reprogramming, fasting protocols create a therapeutic context that can potentiate conventional treatments while mitigating systemic toxicity[61]. Taken together, dietary modulation through fasting represents a conceptual shift in the understanding of metabolic vulnerabilities in CRC, aligning nutritional interventions with the principles of molecular oncology.
Conceptual definitions of dietary interventions used in CRC research: (1) IF: A dietary pattern characterized by the cyclic alternation between periods of food intake and periods of energy restriction, without necessarily reducing total caloric intake over time; (2) Alternate-day fasting (ADF): A form of IF involving alternating days of marked CR (approximately 25%-30% of daily energy requirements) with days of ad libitum feeding. A related approach, the 5:2 model, consists of two nonconsecutive fasting days per week interspersed with five unrestricted days; (3) TRE: A distinct IF variation that confines daily food intake to a fixed time window, typically ranging from 4 hours to 10 hours per day, preferably aligned with circadian rhythms, without mandatory CR; (4) FMDs: Short-term, cyclic dietary protocols designed to mimic the metabolic effects of prolonged fasting through substantial caloric and macronutrient restriction, while allowing limited food intake to improve feasibility and safety; (5) KD: A high-fat, very low-carbohydrate dietary regimen that induces a metabolic shift from glucose to ketone bodies as the primary energy source, resulting in nutritional ketosis; and (6) CR: A sustained reduction in total caloric intake, typically ranging from 20% to 40% below habitual energy consumption, without malnutrition, aimed at modulating metabolic and cellular signaling pathways. Definitions and terminology for IF, ADF, TRE, FMDs, CR, and KD, adapted from Wang et al[62], Wang et al[63], Kibret et al[64], Yüzbaşıoğlu and Özder[65], Habe et al[66], Martínez-Montoro et al[67], Brandhorst et al[68], Rangan et al[69], Bustamante-Marin et al[70], and Malinowska and Żendzian-Piotrowska[71].
IF is defined as a cyclic alternation between periods of food intake and energy restriction, encompassing various patterns, such as ADF and the 5:2 model. In ADF, days of marked CR (approximately 30% of daily energy needs) alternate with days of ad libitum feeding, whereas the 5:2 model prescribes two fasting days interspersed with five unrestricted days[62-64]. Time-restricted eating (TRE), a distinct IF variation, confines the feeding window to 4-10 hours per day, preferably aligned with the circadian rhythm. Evidence indicates that early TRE (feeding between 07:00 AM and 3:00 PM) optimizes glycemic control and insulin sensitivity[65-67]. Hybrid protocols combining IF and TRE, such as intermittent TRE, in which 30% of daily caloric intake occurs between 08:00 AM and 12:00 AM, followed by a 20-hour fast three times per week, promote favorable metabolic adaptations — including reductions in glycemia and insulinemia, increased ketogenesis, and activation of autophagy-related pathways[59,72]. Collectively, these responses create a metabolic environment less favorable for tumor proliferation and more responsive to antineoplastic therapies.
Prolonged overnight fasting, defined as an interval of at least 13 hours between the last meal of the day and the first meal of the next day, has emerged as a feasible approach to synchronize circadian rhythms and enhance metabolic homeostasis[73,74]. Clinical studies demonstrate that this practice improves glycemic control, insulin sensitivity, and inflammatory profiles - effects attributed to the role of feeding as a zeitgeber that entrains peripheral clocks in metabolic tissues[75-77]. In oncology, extended overnight fasting has been inversely associated with the risk of tumor recurrence, possibly due to enhanced coordination between nutrient intake, circadian gene expression, and cellular repair processes during the sleep-wake cycle[77-79]. These findings suggest that fasting regimens that integrate circadian alignment may confer metabolic and therapeutic advantages in the management of CRC.
FMDs are low-calorie, low-protein, and low-carbohydrate regimens enriched with unsaturated fats, designed to elicit metabolic responses analogous to those of complete fasting while maintaining minimal nutrient intake[68,69,80]. Both preclinical and clinical studies have demonstrated that FMD reduces plasma glucose, insulin, and IGF-1 concentrations, while increasing ketone body production and activating cytoprotective stress-response genes[19,81,82]. In CRC models, FMD cycles suppress tumor growth, inhibit cell proliferation, and attenuate angiogenesis[17,18,83]. Clinically, FMD exhibits a favorable safety profile when applied to nutritionally stable patients under professional supervision, showing minimal risk of progressive weight loss[84-86]. Therefore, successful clinical translation of fasting-based interventions requires stringent monitoring and well-defined selection criteria to ensure efficacy and safety. Table 1 shows pre-clinical studies that evaluated fasting and CR diets.
| Ref. | Type of intervention | Main metabolic findings | Effects on tumor/immunity |
| Bensalem et al[59], 2025 and Teong et al[72], 2023 | Intermittent fasting (ADF: 5:2, iTRE) | blood glucose reduction, insulin reduction, ketone bodies increase, autophagy increase | Tumor growth reduction, metabolic environment less favorable to proliferation |
| Tsuda et al[77], 2020 | Prolonged overnight fast | Synchronization of circadian rhythms, insulin sensitivity increase | Systemic inflammation reduction, cellular repair increase |
| Luo et al[17], 2024; Nan et al[18], 2025; and Zhong et al[83], 2023 | Fasting mimicking diet (cycles) | Glucose reduction, insulin reduction, IGF-1 reduction, ketone bodies increase, expression of cellular stress genes increase | Tumor growth reduction, proliferation reduction, angiogenesis reduction, CD8+ infiltration, microbiota remodeling increase |
| Vidoni et al[88], 2021 and Caprara et al[89], 2025 | Caloric restriction (20%-40%) | Glucose reduction, IGF-1 reduction, mTOR reduction, AMPK increase, autophagy increase | Cell proliferation reduction, apoptosis increase, genomic repair increase, inflammation reduction, stress resistance increase |
Despite solid mechanistic rationale and reproducible antitumor effects in CRC preclinical models, clinical translation is constrained by essential limitations. Existing human evidence is associative mainly, derived from small or convenient samples with substantial variability in cancer stage, nutritional status, and concurrent therapies, hindering causal interpretation. Fasting protocols are inconsistently defined across studies (in terms of timing, caloric load, and nutrient composition), adherence remains a significant challenge, and long-term safety outcomes are poorly understood. Moreover, most studies prioritize metabolic and inflammatory biomarkers over objective tumor endpoints. These gaps highlight the necessity for randomized controlled trials with standardized intervention designs and stratification based on metabolic phenotypes.
Effects on immune modulation and therapeutic response: Among the most relevant outcomes of fasting protocols in oncology is their capacity to modulate immune responses, particularly through FMD. This dietary intervention selectively reduces immunosuppressive cell populations, including monocytes, MDSCs (M-MDSCs and PMN-MDSCs), and regulatory T lymphocytes, while expanding effector compartments such as cytotoxic CD8+ T cells and natural killer (NK) cells. These adaptations collectively enhance tumor infiltration by activated cytotoxic lymphocytes and promote a more robust antitumor immune landscape[17,19,83]. However, these findings are primarily based on preclinical models and an early-phase trial, limiting their applicability to routine clinical practice.
In studies from in vitro and preclinical models, FMD has also been shown to inhibit the expansion of IgA+ B cells, thereby enhancing antitumor immunity through increased fatty acid oxidation and transcriptional reprogramming of B cells, which relieves inhibitory signaling toward CD8+ T cells. Additionally, FMD reshapes the gut microbiota, favoring the enrichment of beneficial taxa such as Lactobacillus johnsonii, whose presence is essential to mediate the diet’s immunomodulatory and antitumor effects[17,83].
When combined with chemotherapy, FMD exerts synergistic effects on therapeutic response, improving both radiologic and pathological tumor regression rates while protecting immune cells from DNA damage, even in the absence of corticosteroid co-administration, in early clinical trials involving patients with breast cancer and other cancers[84,85]. In an in vitro and preclinical models, FMD also enhances the efficacy of immune checkpoint blockade, particularly anti-PD-1 therapy, by increasing tumor immunogenicity and CD8+ T-cell infiltration[17,83,87]. Collectively, these findings position fasting-based interventions as potential immunometabolic modulators that can amplify therapeutic efficacy in CRC. However, further studies are warranted to standardize clinical protocols and elucidate their long-term effects on tumor control and patient outcomes.
CR, defined as a reduction of 20%-40% in total energy intake without inducing malnutrition, has been associated with increased longevity and delayed onset of age-related diseases. Beyond its effects on lifespan, CR exhibits significant antineoplastic properties, reducing cancer incidence and enhancing the efficacy of oncologic treatments[70]. These benefits are attributed to metabolic adaptations that improve insulin sensitivity, stimulate autophagy, and reduce glycemia, inflammation, angiogenesis, and growth factor signaling[88,89]. The primary anti-ageing mechanisms underlying CR include neuroendocrine adaptation, suppression of inflammation, activation of hormetic pathways, and protection against oxidative stress-induced damage[90]. Collectively, these responses orchestrate a systemic shift from anabolic to catabolic metabolism, promoting metabolic efficiency and cellular resilience.
At the molecular level, CR upregulates genes responsible for DNA repair, apoptosis, autophagy, stress responses, and antioxidant defenses while downregulating pro-inflammatory genes and energy metabolism pathways[91]. The resulting hypoglycemic state attenuates the secretion of anabolic hormones and suppresses insulin-dependent metabolic activity by inhibiting PI3K and MAPK signaling. This suppression prevents activation of the mTOR, whose inhibition enhances autophagic flux. The increased autophagy induced by CR contributes to immune homeostasis through the downregulation of interferon and cytokine secretion, as well as the inhibition of inflammasome activation[92]. Thus, CR establishes self-reinforcing metabolic feedback that links nutrient sensing to cellular stress resistance.
The metabolic benefits of CR arise from coordinated cellular and systemic adaptations, including reduced insulin and IGF-1 signaling, improved mitochondrial function, enhanced sirtuin activation, and decreased oxidative and inflammatory stress[93]. Central to these effects are the activation of AMPK, the inhibition of mTOR, and the upregulation of SIRT1. These pathways act as metabolic checkpoints that synchronize energy sensing with cellular protection. Hormonal and adipokine regulation—including insulin, IGF-1, and leptin signaling—further mediates CR’s antitumor properties, as these factors strongly influence cell proliferation and survival[70]. Notably, the IGF1R, which is frequently overexpressed in tumors, drives mitogenic activity even at physiological ligand concentrations, while PI3K/Akt and mTOR signaling remain constitutively active across multiple malignancies[94].
Activation of the PI3K-AKT-mTORC1 axis sustains the high glycolytic rate of tumor cells by stabilizing HIF-1α and promoting transcription of glucose transporters and glycolytic enzymes. This pathway also phosphorylates the pro-apoptotic transcription factor FOXO1, triggering its nuclear exclusion and degradation—thereby linking metabolic reprogramming to survival and proliferation[95]. CR mitigates these pro-growth signals by lowering glucose and insulin/IGF-1 levels, ultimately downregulating mTOR activation[96]. Because mTOR drives anabolic metabolism and cell division, its suppression via CR disrupts tumor-promoting signaling, restoring autophagic control and genomic integrity[97]. Through this coordinated modulation, CR generates a cellular state characterized by increased stress tolerance and reduced oncogenic drive.
During CR, glucose depletion triggers the mobilization of glycogen and fatty acids, leading to the production of ketone bodies that serve both as alternative energy substrates and signaling molecules[91]. These metabolites regulate redox balance and inflammatory tone within the tumor microenvironment. Elevated insulin and IGF-1 concentrations promote carcinogenesis via PI3K-AKT-mTORC1-mediated anti-apoptotic signaling, whereas CR interrupts this cascade, shifting metabolism toward oxidative phosphorylation and promoting apoptosis. The nutrient-sensing networks activated by CR detect fluctuations in energy supply and coordinate adaptive responses to scarcity, thereby enhancing resistance to oncogenic stressors and suppressing tumorigenesis[98]. CR also reduces cell proliferation, limits DNA synthesis, and promotes genomic repair, while altering systemic concentrations of estrogen, insulin, IGFBP-3, IGF-1, leptin, and prolactin—all key mediators of cancer pathophysiology[99].
Consistent with preclinical findings, human studies have demonstrated that CR decreases circulating IGF-1, insulin, and glucose levels, thereby attenuating PI3K/Akt signaling, enhancing DNA repair, and improving cancer resistance[100,101]. CR profoundly affects lipid metabolism through the activation of AMPK, sirtuins, and PPARs, which collectively maintain lipid and energy homeostasis under caloric deficit. AMPK activation, triggered by an elevated AMP/ATP ratio, inhibits ACC, thereby reducing malonyl-CoA synthesis and relieving the inhibition of CPT-1, which enhances mitochondrial β-oxidation. This metabolic reprogramming limits lipid availability for membrane synthesis, depriving tumor cells of essential anabolic substrates, while mitigating lipid-induced oxidative stress and inflammation[98].
Unlike normal cells, cancer cells rely heavily on de novo lipogenesis to sustain rapid growth. AMPK activation during CR disrupts this dependency by promoting fatty acid oxidation and suppressing mTOR activity, which acts as a metabolic checkpoint during energy scarcity[91]. mTOR inhibition activates autophagy and fatty acid oxidation while restraining anabolic processes such as protein synthesis, thereby preventing the accumulation of damaged cellular components and maintaining genomic stability[98]. By inducing a reversible quiescent state, CR lowers biosynthetic demands and limits tumor cell proliferation, creating a metabolic landscape incompatible with rapid tumor expansion[102]. Collectively, CR integrates hormonal regulation, nutrient-sensing pathways, and metabolic reprogramming to establish a cellular environment that resists oncogenic transformation while preserving the integrity and adaptability of normal cells[91,103]. Table 2 summarizes clinical evidence linking fasting and CR to therapeutic outcomes.
| Ref | Intervention | Population/Protocol | Metabolic effects | Clinical or therapeutic impact |
| Bensalem et al[59], 2025 and Teong et al[72], 2023 | Intermittent fasting/TRE (iTRE) | Clinical trials in cancer patients | Blood glucose reduction, insulin reduction, ketone bodies increase, autophagy increase | Metabolic environment less favorable to tumor proliferation, with potential response to therapies increase |
| Tsuda et al[77], 2020; Khodabakhshi et al[78], 2021; and Mindikoglu et al[79], 2020 | Prolonged overnight fasting (> 13 hours) | CRC patient cohorts | Insulin sensitivity, systemic inflammation reduction increase | Risk of tumor recurrence reduction |
| Vernieri et al[19], 2022; Zhong et al[83], 2023; Ligorio et al[84], 2022; and Ligorio et al[85], 2025 | Fasting mimicking diet | Clinical trials in CRC | Glucose reduction, insulin reduction, IGF-1 reduction, ketone bodies increase; L-arginine in tumor tissues increase | Favorable safety profile; response to chemotherapy increase; CD8+ infiltration increase; microbiota remodeling; abundance of B. pseudolongum increase; improved patient prognosis |
| Bustamante-Marin et al[70], 2023 and Vidoni et al[88], 2021 | Moderate caloric restriction (20%-40%) | Clinical trials in cancer patients | Glucose reduction, insulin reduction, IGF-1 reduction, insulin sensitivity increase, autophagy increase | Potential for therapeutic response and tumor progression reduction increase |
Although CR has demonstrated consistent antitumor benefits in CRC preclinical models, the translation to clinical practice remains challenging. Human studies are limited by small sample sizes, varying cancer stages, and inconsistent definitions of caloric reduction (ranging from 10% to over 40%). Adherence is a significant barrier, particularly in patients susceptible to unintentional weight loss or treatment-induced anorexia. Moreover, most trials prioritize intermediate metabolic endpoints rather than tumor response or survival, making clinical significance uncertain. Standardized, oncology-specific CR protocols and stratification based on nutritional and metabolic status are needed before CR can be safely recommended in CRC care.
KDs represent a dietary strategy characterized by an extreme reduction in carbohydrate intake and a proportional increase in dietary fat, thereby promoting hepatic ketone body synthesis. In clinical and experimental settings, carbohydrate intake is typically restricted to 20-50 g per day, with an initial fat-to-carbohydrate plus protein ratio of 3:1 or 4:1, adjusted to maintain moderate-to-high urinary ketone concentrations ranging from 80 mg/dL to 160 mg/dL[71]. This nutritional state, known as nutritional ketosis, shifts systemic energy metabolism from glucose dependence toward lipid oxidation and ketone body utilization, thereby recreating metabolic conditions comparable to fasting. The metabolic flexibility induced by KDs has been increasingly explored as a potential therapeutic adjunct in cancer management, particularly for tumors exhibiting high glycolytic flux, such as CRC.
Ketogenesis occurs primarily in the mitochondria of hepatocytes, where fatty acid β-oxidation generates acetyl-CoA that exceeds the tricarboxylic acid (TCA) cycle’s oxidative capacity. This accumulation drives acetyl-CoA condensation into acetoacetate (AcAc) and β-hydroxybutyrate (βHB), which are then released into circulation as alternative energy substrates[104,105]. Each cycle of β-oxidation cleaves two-carbon units from fatty acyl-CoA, producing acetyl-CoA, FADH2, and NADH, which feed into mitochondrial oxidative metabolism[105-107]. In peripheral tissues, βHB is reconverted to AcAc by βHB dehydrogenase (BDH1), then activated to acetoacetyl-CoA by β-ketoacyl-CoA transferase and cleaved into two acetyl-CoA molecules entering the TCA cycle for ATP synthesis—a process termed ketolysis[108-110].
Although the liver is the primary site of ketone body production, extrahepatic organs such as the brain, heart, skeletal muscle, and kidneys possess the enzymatic machinery to utilize ketones as oxidative substrates, particularly under conditions of glucose scarcity[109,111]. The efficiency of this adaptation contrasts sharply with most tumor cells, which exhibit metabolic inflexibility associated with the Warburg phenotype—preferring aerobic glycolysis over oxidative phosphorylation even in the presence of oxygen[112]. Many CRC cells demonstrate partial or complete suppression of ketolytic enzymes, rendering them incapable of effectively oxidizing βHB or AcAc. This metabolic deficiency constitutes a therapeutic vulnerability: While normal cells adapt to ketosis by enhancing mitochondrial respiration, tumor cells experience energetic stress, oxidative imbalance, and apoptotic signaling[71].
Therefore, KD can selectively disadvantage tumor metabolism by simultaneously reducing glucose availability and elevating circulating ketones. In this dual context, healthy tissues sustain energy production through ketolysis, whereas neoplastic cells, unable to metabolize ketone bodies efficiently, undergo bioenergetic crisis and growth arrest. This selective metabolic pressure not only restrains tumor proliferation but may also sensitize cancer cells to cytotoxic and oxidative therapies. Table 3 summarizes the principal methodological features and findings of studies investigating the KD in CRC, detailing experimental models, tumor subtypes, dietary formulations, and antitumor effects[113-116].
| Ref. | Model | Dietary intervention | Main metabolic findings |
| Montroseand Galluzzi[113], 2022 | Immunocompetent and immunodeficient mice, MC38 and CT26 strains (sex not specified) | Ketogenic diet | Effective reduction of MC38 tumor growth; increased sensitivity to immune checkpoint inhibitors (PD-1) |
| Tsenkova et al[114], 2025 | C57BL/6 mice (no sex specified) | Ketogenic diet and stearic acid-supplemented diet | Reduction of colonic tumor burden; induction of apoptosis in cancer cells; ketogenic diet altered the composition and function of the microbiota, enriching stearic acid producers and reducing consumers |
| Sun et al[115], 2022 | Male BALB/c mice | Ketogenic diet | Inhibition of tumor growth; increased antitumor immune response; attenuation of immunosuppression; inhibition of angiogenesis |
| Jiang et al[116], 2024 | Male C57BL/6 mice | Ketogenic diet + chemotherapy | Reduced tumor growth; increased apoptosis; increased sensitivity to chemotherapy |
Despite strong mechanistic rationale and promising tumor-modulating effects in preclinical CRC studies, current human evidence for KD remains preliminary and heterogeneous. Interventional trials often include mixed cancer populations, use distinct macronutrient compositions, and lack long-term endpoints such as recurrence or survival. Risks include exacerbation of cachexia, micronutrient deficiencies, and intolerance in patients with gastrointestinal symptoms related to the tumor or treatment. While KD may benefit select metabolic phenotypes, such as patients with hyperinsulinemia, precision oncology frameworks and controlled clinical trials are necessary to clarify its therapeutic value, optimal duration, and safety profile.
Early clinical trials: Early clinical investigations on the KD in CRC remain scarce. Table 4 summarizes the main findings from the few available studies, of which only two met inclusion criteria, neither reporting microbiome-related outcomes in participants adhering to ketogenic interventions[117,118]. Despite the limited evidence base, preliminary results suggest that KDs may influence tumor metabolism and therapeutic responsiveness, providing a mechanistic rationale for integrating nutritional ketosis with conventional treatments.
| Ref. | Population/protocol | Dietary intervention | Clinical or therapeutic impact |
| Klement et al[117], 2021 | Recurrent stage IV colon cancer | Chemotherapy combined with a modified ketogenic diet | Response rate of 60%; disease control rate of 70%; complete response rate of 50%; trend toward longer overall survival in the responder group (50 months vs 32.5 months in the control group) |
| Egashira et al[118], 2023 | Rectal cancer | Ketogenic diet based on natural foods and standard diet during radiotherapy | Reduction in body weight and fat mass; preservation of skeletal muscle mass; tendency toward greater pathological tumor response (higher degree of Dworak regression and higher percentage of near-complete responses) |
Synergistic effects with chemotherapy and radiotherapy: Tumor cells, characterized by high metabolic rates and elevated biosynthetic demand, sustain intrinsically higher basal oxidative stress than normal cells. Glucose restriction exacerbates this condition, rendering malignant cells more susceptible to chemotherapeutic agents that induce oxidative damage[62,113,119]. The reduction in antioxidant capacity, coupled with sustained production of reactive oxygen species (ROS) through altered metabolic fluxes or chemotherapy exposure, leads to excessive ROS accumulation that damages DNA, proteins, and lipids, culminating in functional impairment and cell death. Agents such as doxorubicin and cisplatin have demonstrated enhanced cytotoxicity under glucose-limited conditions[119].
A central mechanism underlying this potentiation is the concept of differential stress resistance. While tumor cells remain metabolically inflexible and highly glucose-dependent, normal cells exhibit adaptive flexibility, shifting to fatty acid oxidation and ketone utilization under conditions of nutrient deprivation[120]. In response to energy stress, nonmalignant cells activate protective pathways, including AMPK signaling and autophagy, entering a quiescent, repair-oriented metabolic state that enhances survival during chemotherapy exposure. This adaptive transition enables normal cells to withstand cytotoxic doses that would otherwise be lethal in hyperglycemic environments, whereas tumor cells, unable to reprogram their metabolism efficiently, remain selectively vulnerable[121,122].
Oxygen acts as a potent radiosensitizer by stabilizing radiation-induced free radicals and promoting additional ROS generation, intensifying DNA damage. However, many tumors exhibit hypoxic microenvironments that confer relative radioresistance through altered redox balance and glycolytic metabolism[123,124]. Glucose deprivation compromises tumor cells’ ability to generate NADPH, essential for maintaining antioxidant defenses. This reduction in the capacity to neutralize endogenous and exogenous ROS makes tumor cells more susceptible to oxidative damage induced by radiation. ROS accumulation, resulting from the combination of radiation and compromised antioxidant capacity, leads to greater DNA damage and activation of cell death pathways[125].
Additionally, glucose restriction can alter tumor mitochondrial metabolism, forcing cells to rely more on oxidative phosphorylation. While this may increase basal mitochondrial ROS production, it can also render mitochondria more vulnerable to radiation-induced damage, contributing to cell death[126]. Severe carbohydrate restriction is the primary driver of ketosis, but it is also the greatest challenge to patient adherence[127]. Common and culturally important foods, such as bread, pasta, sugars, and many vegetables and fruits, are restricted. This drastic change can be difficult to maintain long-term, leading to non-adherence and consequently, therapy failure. In conclusion, KD therapy in CRC treatment represents a promising strategy based on solid biochemical rationale. However, its translation into clinical practice depends on overcoming adherence barriers, including nutritional complexity, rigorous monitoring, and significant impact on patient quality of life[117].
Despite strong mechanistic rationale and reproducible antitumor effects in preclinical models, clinical evidence supporting fasting-based, CR, and KD interventions in CRC remains limited and heterogeneous. Where available, negative or neutral findings have been reported, particularly in studies characterized by short intervention duration, variable adherence, non-standardized protocols, or reliance on intermediate metabolic endpoints rather than oncologic outcomes. These observations highlight the persistent gap between mechanistic promise and consistent clinical benefit. Consequently, such dietary strategies should currently be regarded as hypothesis-generating and considered only within controlled clinical settings, with careful patient selection and rigorous nutritional monitoring.
Targeting amino acid metabolism has emerged as a promising anticancer strategy based on the observation that many tumor cells exhibit heightened dependence on specific amino acids that function not only as metabolic substrates but also as signaling molecules and epigenetic regulators. These amino acids support macromolecular biosynthesis, redox homeostasis, and transcriptional programs essential for tumor growth and survival. Importantly, however, the majority of evidence supporting amino acid restriction strategies derives from preclinical models, and current findings should therefore be interpreted primarily as hypothesis-generating rather than clinically established interventions[56].
Dietary amino acid restriction offers systemic metabolic modulation but presents challenges related to feasibility and adherence in cancer patients. Pharmacological inhibition of amino acid transporters or metabolic enzymes provides greater specificity and control, but may not fully reproduce the systemic metabolic adaptations induced by dietary interventions. Furthermore, although the combination of exercise and dietary supplementation appears effective for preventing sarcopenia, both dietary restriction and pharmacological approaches raise concerns in cancer patients, particularly those with cachexia or requiring intensive care[128].
Mechanisms of action: One-carbon metabolism, redox balance, and nucleotide synthesis: The one-carbon metabolic network, encompassing the folate and methionine cycles and the transsulfuration pathway, constitutes a central hub supporting tumor proliferation by integrating nucleotide biosynthesis, epigenetic regulation, and redox homeostasis[129-131]. Serine- and glycine-derived one-carbon units fuel folate-mediated reactions that sustain purine and pyrimidine synthesis, regenerate methionine, and generate S-adenosylmethionine (SAM), thereby linking nutrient availability to methylation-dependent gene regulation. In parallel, transsulfuration-derived cysteine supports glutathione (GSH) synthesis, while folate cycle enzymes such as SHMT and MTHFD family members contribute to NADPH production, reinforcing antioxidant capacity and anabolic metabolism[129,132].
This network is tightly regulated by oncogenic signaling pathways, including MYC, NF-κB, and mTORC1, which upregulate key enzymes such as TYMS, DHFR, and MTHFD2, coupling amino acid availability to proliferative signaling. Disruption of one-carbon flux, whether through amino acid restriction or pharmacological inhibition, impairs nucleotide synthesis, induces replication stress, and compromises redox balance in preclinical cancer models[131].
In CRC, evidence supporting amino acid restriction strategies is predominantly preclinical and suggests heightened vulnerability in tumors with dysregulated one-carbon metabolism, particularly those harboring KRAS mutations[133,134]. However, human data remain limited to dietary feasibility studies without tumor-specific outcomes. Potential risks, including sarcopenia, immune dysfunction, and treatment intolerance, constrain the clinical applicability of sustained amino acid restriction. Moreover, metabolic responses vary according to tumor genotype and microbiome composition, underscoring the need for biomarker-guided approaches. Collectively, while targeting one-carbon metabolism represents a mechanistically compelling strategy, its therapeutic relevance in clinical oncology remains hypothesis-generating.
Methionine: Methionine is an essential sulfur-containing amino acid that supports tumor growth primarily through regulation of one-carbon metabolism, epigenetic methylation, and redox homeostasis. As the precursor of SAM, methionine links nutrient availability to DNA, RNA, and protein methylation, polyamine synthesis, and antioxidant capacity via GSH production. Through its interconnection with the folate cycle and the transsulfuration pathway, methionine metabolism integrates methyl group flux with redox control and biosynthetic demands[135-138].
Many tumor cells exhibit methionine dependence, known as the Hoffman effect, characterized by impaired proliferation and induction of apoptosis upon methionine deprivation despite homocysteine availability. This vulnerability is associated with an increased requirement for SAM-mediated chromatin remodeling and is reinforced by elevated expression of MAT2A. Dysregulated methionine metabolism contributes to aberrant DNA methylation patterns, including promoter hypermethylation of tumor suppressor genes, and methionine restriction attenuates these alterations and suppresses tumor growth in preclinical models[135,137].
Beyond tumor-intrinsic effects, methionine-derived metabolites such as SAM and 5′-deoxy-5′-methylthioadenosine modulate antitumor immunity by influencing chromatin accessibility in T lymphocytes and promoting T cell exhaustion. Preclinical studies suggest that dietary methionine restriction may enhance CD8+ T cell infiltration and synergize with chemotherapy. Nonetheless, due to methionine’s essential role in muscle maintenance, neural function, and redox balance, the optimal restriction level and duration require precise definition to avoid adverse systemic effects[139,140].
Serine: Serine is a nonessential amino acid that supports tumor proliferation through its central role in macromolecular biosynthesis and one-carbon metabolism[141]. It provides carbon units for nucleotide synthesis, contributes to cysteine and glycine production, and participates in sphingolipid and phospholipid synthesis. A fraction of glycolytic 3-phosphoglycerate is diverted to serine biosynthesis via PHGDH, PSAT1, and PSPH, while serine SHMT1/2 interconverts serine and glycine to sustain one-carbon flux[129,130].
The resulting glycine supports purine, heme, and GSH biosynthesis, thereby maintaining redox homeostasis and mitochondrial function. Elevated serine consumption, increased expression of PHGDH, PSAT1, PSPH, and the transporters SLC1A4/SLC1A5 are frequently observed in aggressive tumors and correlate with poor prognosis. Serine deprivation, achieved through dietary restriction or inhibition of biosynthetic enzymes, reduces tumor cell proliferation and sensitizes tumors to chemotherapy, highlighting SHMT as a potential therapeutic target[130].
From an immunometabolic perspective, serine availability is required for effector T cell function by supporting GSH synthesis and one-carbon metabolism. In contrast, enhanced serine metabolism in regulatory T cells activates mTOR signaling and promotes their proliferation, underscoring the context-dependent consequences of targeting serine metabolism[140].
Glutamine: Glutamine is the most abundant amino acid in plasma and a key nutrient sustaining tumor proliferation, anaplerosis, and redox balance. It provides carbon and nitrogen for the synthesis of nucleotides, nonessential amino acids, and GSH, and replenishes the TCA cycle through conversion to α-ketoglutarate[129,141]. Tumor cells primarily acquire glutamine via the ASCT2 (SLC1A5) transporter, whose elevated expression correlates with poor clinical outcomes, while de novo synthesis through glutamine synthetase (GS) can further support tumor growth and survival[56].
Glutaminolysis, mediated by glutaminase isoforms GLS1 and GLS2, converts glutamine into glutamate, fueling energy production, redox control, and transamination reactions. GLS1 is frequently upregulated in tumors and promotes GSH synthesis, NADH production, and metastatic potential, whereas GLS2, regulated by p53, exhibits tumor-suppressive functions by inhibiting PI3K/AKT signaling and facilitating ferroptosis. Under glucose-limiting conditions, glutamine metabolism partially compensates for energy demands, thereby limiting full TCA cycle flux and energetic efficiency[142].
Collectively, these features position glutamine restriction or glutaminase inhibition as rational strategies to disrupt tumor metabolism. However, given glutamine’s essential physiological roles, particularly in maintaining systemic metabolic homeostasis, such approaches require careful evaluation of feasibility and toxicity[56].
Arginine: Arginine is a conditionally essential amino acid that supports tumor growth and immune regulation. It can be synthesized de novo from aspartate and citrulline via the urea cycle enzymes ASS1 and ASL, while also serving as a metabolic precursor for nitric oxide, creatine, polyamines, and other amino acids. In many malignancies, reduced ASS1 expression leads to urea cycle dysregulation, redirecting aspartate toward pyrimidine synthesis and fostering rapid cell proliferation. As a result, tumors frequently develop arginine auxotrophy and rely on extracellular uptake through cationic amino acid transporters (CAT-1, CAT-2, and CAT-3), which are commonly overexpressed[56,140,143].
Arginine catabolism is mediated by arginases (ARG1 and ARG2), generating ornithine and urea. Ornithine is further converted into polyamines by ornithine decarboxylase, supporting DNA stabilization, protein synthesis, oxidative stress protection, and proliferation. In parallel, oxidation of arginine by inducible nitric oxide synthase (NOS2) produces nitric oxide, which can promote angiogenesis and immune suppression through peroxynitrite-dependent mechanisms. Together, alterations in arginine biosynthesis, transport, and catabolism shape tumor growth dynamics and immune modulation[56,140,144].
These features position arginine restriction or enzymatic depletion strategies, such as arginine deiminase therapy, as rational antitumor approaches. However, their translational potential depends on tumor-specific metabolic dependencies and careful evaluation of systemic and immunological consequences.
Branched-chain amino acids: Branched-chain amino acids (BCAAs; leucine, isoleucine, and valine) provide both nitrogen and carbon skeletons that support biomass production, energy generation, and epigenetic regulation in tumor cells[145]. Accumulation of BCAAs activates mTORC1, triggering downstream phosphorylation of key effectors including 4E-binding protein 1, ribosomal S6 kinase, and sterol regulatory element-binding protein, thereby promoting protein, lipid, and nucleotide biosynthesis while suppressing autophagy[146].
BCAA catabolism, mediated by BCAT2, generates glutamate and branched-chain α-keto acids that are further converted into acetyl-CoA and succinyl-CoA, sustaining TCA cycle flux and ATP production. This metabolic routing maintains anabolic capacity and supports ribonucleotide reductase subunit M2 activity, facilitating DNA synthesis and tumor proliferation. Accordingly, dysregulated BCAA uptake and catabolism contribute to oncogenic metabolic reprogramming, positioning BCAT2 and BCAA transporters as potential therapeutic targets in colorectal and other cancers[56,143].
Tryptophan: Tryptophan is an essential amino acid that supports tumor metabolism, redox balance, and immune regulation. Its primary catabolic route is the kynurenine pathway, mediated by IDO1 and TDO, which convert tryptophan into kynurenine[147]. Downstream metabolism of kynurenine via kynurenine 3-monooxygenase and kynureninase contributes to de novo NAD+ biosynthesis and enhances oxidative stress resistance, thereby facilitating tumor progression and metastasis[148].
Alternative pathways, including serotonin and indole biosynthesis, play more limited roles but may influence tumor progression and host-microbiota interactions[56]. Dysregulated tryptophan metabolism, partially shaped by the gut microbiota, modulates inflammation, immune evasion, and metastatic potential. Within the tumor microenvironment, tryptophan depletion suppresses cytotoxic CD8+ T cell function, induces apoptosis in T and NK cells, promotes regulatory T cell activation, and recruits MDSCs, resulting in a profoundly immunosuppressive milieu[144]. Collectively, these effects position inhibition of IDO1/TDO or modulation of the tryptophan-kynurenine axis as rational strategies to restore antitumor immunity in gastrointestinal cancers.
Asparagine and aspartate: Asparagine and aspartate are interconvertible amino acids that support tumor proliferation, particularly under hypoxic conditions. Asparagine is a critical substrate for nucleotide biosynthesis and becomes growth-limiting during hypoxia due to restricted mitochondrial export and low membrane permeability. Under these conditions, asparagine metabolism compensates for aspartate scarcity, as its conversion by asparaginase sustains intracellular asparagine availability and tumor growth[149].
Asparagine metabolism intersects with the urea cycle, redirecting nitrogen and carbon toward pyrimidine biosynthesis while asparagine export facilitates the uptake of other amino acids, including serine, arginine, and histidine, thereby supporting protein synthesis and anabolic balance. In addition, asparagine availability regulates epithelial-mesenchymal transition, contributing to metastatic dissemination. At metastatic sites, high asparagine availability combined with glutamine scarcity activates GS, stabilizing HIF1α and c-Myc and supporting the survival and colonization of disseminated tumor cells[56]. Together, these mechanisms position asparagine and aspartate metabolism as key nodes linking amino acid availability, tumor bioenergetics, and metastatic potential.
The strength of evidence supporting amino acid restriction strategies is heterogeneous and strongly influenced by tumor genotype. Methionine and serine/glycine restriction are supported by the most consistent preclinical data, particularly in tumors with dysregulated one-carbon metabolism and oncogenic drivers such as KRAS and MYC. Glutamine targeting also shows robust mechanistic support but raises significant concerns regarding systemic toxicity. In contrast, evidence for restricting arginine, tryptophan, BCAAs, and asparagine/aspartate is more context-dependent and varies according to tumor-specific metabolic dependencies. Overall, clinical evidence remains scarce, indicating that these strategies should currently be viewed as hypothesis-generating rather than clinically established interventions.
The growing body of evidence suggests the integration of mechanistically informed dietary strategies, such as FMDs, TRE, and CR, as complementary therapeutic modalities in CRC management[84,117]. However, the safe translation of these interventions into clinical practice requires precise patient selection and rigorous nutritional supervision to prevent iatrogenic complications. Restrictive protocols are strictly contraindicated for individuals with high dietary risk, including those experiencing unintentional weight loss, cachexia, or severe functional decline, as these conditions predict poor clinical outcomes[84,150]. In real-world oncology workflows, these criteria can be incorporated into routine pre-treatment nutritional screening, with reassessment at each treatment cycle to identify early deterioration and promptly discontinue restrictive protocols. Ethical implementation requires shared decision-making, clear informed consent, and a risk-benefit discussion, particularly in vulnerable patients in whom restrictive diets may aggravate malnutrition or reduce treatment tolerance.
According to Taylor et al[150] in 2022, exclusion criteria include body mass index ≤ 20 kg/m2, Eastern Cooperative Oncology Group performance status ≥ 3, phase angle < 4.5° by bioimpedance, and categories B or C on the Subjective Global Assessment. Eligible patients with preserved nutritional status should undergo continuous metabolic monitoring encompassing blood glucose, insulin, IGF-1, and circulating ketone bodies—critical markers of metabolic adaptation. In addition, regular evaluation of body composition is essential to mitigate the risk of muscle loss, particularly during CR or KD interventions[19,151].
Effective implementation of metabolic dietary strategies requires shared leadership: Oncologists oversee oncologic safety and treatment scheduling, while clinical nutritionists operationalize dietary prescriptions, monitoring, diet composition, nutrient timing, potential side effects, and adherence, with input from metabolic specialists when needed. The therapeutic benefit of FMD, KD, and amino acid restriction protocols hinges on precise synchronization with chemotherapy or radiotherapy cycles. These interventions can alter the tumor microenvironment, reduce metabolic flexibility and promote tumor cell vulnerability, while simultaneously enhancing stress resistance and tissue regeneration in normal cells through differential stress response mechanisms[19,151,152]. Proper timing amplifies the cytotoxic potential of chemotherapeutic and radiotherapeutic agents while protecting healthy tissues. For example, in clinical protocols, short FMD cycles are often scheduled around chemotherapy sessions, whereas TRE or milder CR approaches may be considered during off-cycle periods, always guided by nutritional status and symptom burden. Nevertheless, amino acid restriction strategies targeting methionine or serine require additional caution, as prolonged deficiency may compromise muscle integrity and antioxidant capacity, reinforcing the need for individualized and closely monitored dietary management.
Diet-induced metabolic modulation has shown synergy with chemotherapy and radiotherapy in animal models, enhancing cytotoxic effects while protecting normal tissues. Nevertheless, human data are limited, and most clinical reports evaluate tolerability rather than objective oncologic response. The bidirectional relationship between treatment-induced anorexia and restrictive diets raises safety concerns, particularly regarding weight loss, sarcopenia, and impaired treatment dose-intensity. Future studies must incorporate nutritional risk assessment and body composition monitoring to balance metabolic benefits with patient tolerance, aiming for interventions that improve therapeutic index without compromising clinical outcomes.
Despite their biochemical rationale and therapeutic promise, adherence remains the primary limitation of FMD and KD protocols. The exclusion of culturally central foods, such as bread, pasta, and fruits, poses long-term compliance challenges and increases the risk of discontinuation. Consequently, more flexible approaches like prolonged overnight fasting (> 13 hours) or TRE have gained interest as sustainable alternatives capable of inducing similar metabolic benefits, including improved insulin sensitivity and reduced systemic inflammation[20,151]. These approaches have also been inversely correlated with tumor recurrence risk in CRC cohorts. Therefore, clinical decision-making should balance mechanistic efficacy with patient feasibility to ensure durable adherence, minimize nutritional risks, and preserve quality of life. Implementation is also constrained by access to specialized nutrition care, socioeconomic barriers, cultural food availability, and feasibility of close monitoring, which should be considered when translating trial-based protocols to routine practice.
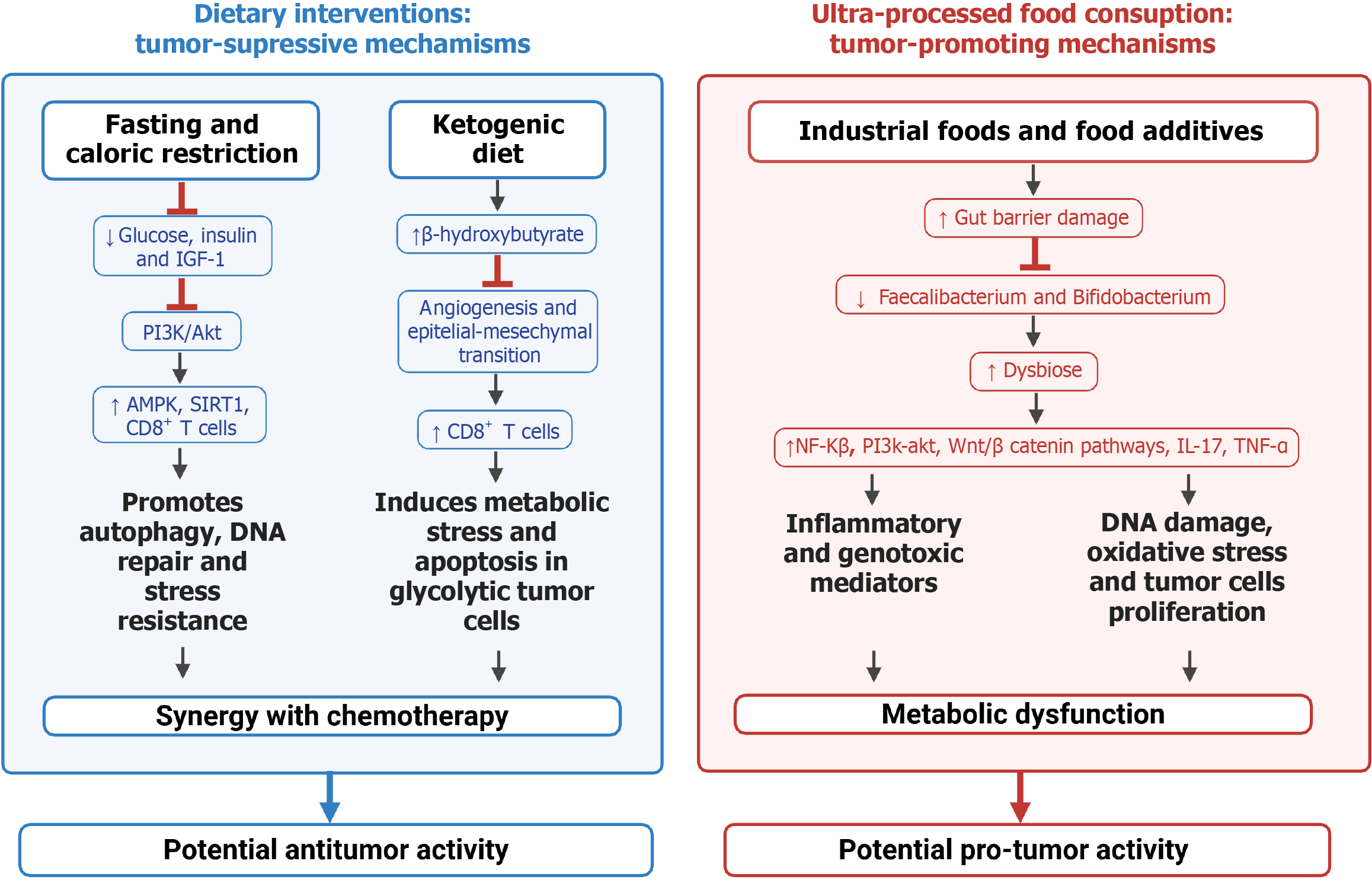
Figure 4 summarizes the effectiveness of dietary interventions on CRC.
This review synthesizes a growing body of evidence connecting UPFs consumption to carcinogenesis and highlights the therapeutic potential of nutrition-based metabolic reprogramming interventions. Nonetheless, some limitations warrant consideration. From a conceptual standpoint, certain interactions among diet, microbiome, and tumor metabolism are still being refined in the literature, reflecting the rapidly evolving nature of the field. From a methodological perspective, differences in study designs and in the standardization of nutritional interventions may influence the comparability of findings. In addition, much of the available evidence is derived from preclinical models or short-term clinical studies, resulting in a relative overrepresentation of mechanistic data compared with long-term oncologic endpoints such as overall or disease-free survival. Microbiome-related findings should also be interpreted with caution, as microbial composition and functional responses are highly context-specific and influenced by host genetics, diet, geography, and treatment exposure.
This narrative review is also subject to inherent limitations, including the risk of confirmation bias, as study selection and interpretation were not guided by a formal systematic protocol. These aspects do not invalidate the conclusions presented but indicate that the topic remains under consolidation, reinforcing the need for future research to deepen and harmonize the available knowledge.
Future progress in this field critically depends on standardized dietary intervention trials in CRC, with harmonized protocols, clearly defined metabolic phenotypes, and clinically meaningful endpoints. Equally important, well-designed negative trials are essential for delineating the limits of benefit, refining patient selection, and preventing the inappropriate extrapolation of mechanistic findings.
Also, future research should integrate multi-omics approaches (genomics, epigenomics, transcriptomics, metabolomics, and metagenomics) to unravel the complex interactions between diet, tumor biology, and the host across different stages of cancer development. Personalized nutrition strategies must be tailored to each phase of the disease, ranging from prevention and early lesions to advanced and cachectic states, considering tumor molecular profiles, metabolic dependencies, and host nutritional status. Omics-guided biomarkers are crucial for predicting individual responses and ensuring safety when applying metabolic interventions, such as fasting, KD, or amino acid modulation. Next-generation clinical trials should incorporate longitudinal omics sampling, microbiome analyses, and adaptive designs to test the synergistic effects of nutritional interventions combined with standard therapies. Additionally, studies should explore microbiome-targeted nutrition, aiming to restore gut integrity and enhance immune and metabolic resilience, while ensuring cultural adaptability, feasibility, and equity of access.
Ultimately, the promise of dietary modulation in CRC lies not in universal dietary prescriptions, but in the development of precision nutrition strategies aligned with tumor biology, host metabolism, and clinical context.
In summary, nutrition exerts a dual and fundamental influence on CRC. On the one hand, the high intake of UPFs acts as a metabolic and inflammatory driver of carcinogenesis by disrupting microbiome equilibrium. On the other hand, structured dietary interventions, such as CR, fasting, and KD, provide therapeutic opportunities by reprogramming host metabolism. These interventions selectively sensitize tumor cells to chemotherapy and radiotherapy while enhancing immune competence. Although clinical implementation demands careful patient selection, metabolic monitoring, and adherence support, dietary modulation emerges as a key pillar in the advancement of personalized and metabolically targeted therapies against CRC. At present, restrictive dietary interventions should not be routinely recommended outside clinical trials, particularly in patients at risk of malnutrition or sarcopenia. Conversely, clinicians should not disregard nutrition altogether, but rather integrate metabolic and dietary considerations within multidisciplinary, supervised, and patient-centered frameworks.
Artificial intelligence tools [ChatGPT, model GPT-5, OpenAI (United States), and Gemini, Google DeepMind (United States)] were used exclusively to support the synthesis, language refinement, and translation of this manuscript from Portuguese to English. All outputs generated by these tools were carefully reviewed, edited, and validated by the authors to ensure scientific accuracy, coherence, and fidelity to the study’s objectives. The AI tools were not used to generate original scientific ideas, data analysis, or conclusions. All intellectual content, interpretations, and final revisions are the sole responsibility of the authors.
| 1. | Patel SG, Karlitz JJ, Yen T, Lieu CH, Boland CR. The rising tide of early-onset colorectal cancer: a comprehensive review of epidemiology, clinical features, biology, risk factors, prevention, and early detection. Lancet Gastroenterol Hepatol. 2022;7:262-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 664] [Cited by in RCA: 554] [Article Influence: 138.5] [Reference Citation Analysis (6)] |
| 2. | Miller KD, Nogueira L, Mariotto AB, Rowland JH, Yabroff KR, Alfano CM, Jemal A, Kramer JL, Siegel RL. Cancer treatment and survivorship statistics, 2019. CA Cancer J Clin. 2019;69:363-385. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3000] [Cited by in RCA: 3117] [Article Influence: 445.3] [Reference Citation Analysis (2)] |
| 3. | Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76817] [Cited by in RCA: 69585] [Article Influence: 13917.0] [Reference Citation Analysis (41)] |
| 4. | Vaghari-Tabari M, Targhazeh N, Moein S, Qujeq D, Alemi F, Majidina M, Younesi S, Asemi Z, Yousefi B. From inflammatory bowel disease to colorectal cancer: what's the role of miRNAs? Cancer Cell Int. 2022;22:146. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (3)] |
| 5. | Zhou RW, Harpaz N, Itzkowitz SH, Parsons RE. Molecular mechanisms in colitis-associated colorectal cancer. Oncogenesis. 2023;12:48. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 71] [Article Influence: 23.7] [Reference Citation Analysis (0)] |
| 6. | Ubago-Guisado E, Rodríguez-Barranco M, Ching-López A, Petrova D, Molina-Montes E, Amiano P, Barricarte-Gurrea A, Chirlaque MD, Agudo A, Sánchez MJ. Evidence Update on the Relationship between Diet and the Most Common Cancers from the European Prospective Investigation into Cancer and Nutrition (EPIC) Study: A Systematic Review. Nutrients. 2021;13:3582. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 137] [Cited by in RCA: 111] [Article Influence: 22.2] [Reference Citation Analysis (2)] |
| 7. | Veettil SK, Wong TY, Loo YS, Playdon MC, Lai NM, Giovannucci EL, Chaiyakunapruk N. Role of Diet in Colorectal Cancer Incidence: Umbrella Review of Meta-analyses of Prospective Observational Studies. JAMA Netw Open. 2021;4:e2037341. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 247] [Cited by in RCA: 205] [Article Influence: 41.0] [Reference Citation Analysis (0)] |
| 8. | Isaksen IM, Dankel SN. Ultra-processed food consumption and cancer risk: A systematic review and meta-analysis. Clin Nutr. 2023;42:919-928. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 99] [Article Influence: 33.0] [Reference Citation Analysis (0)] |
| 9. | Meine GC, Picon RV, Espírito Santo PA, Sander GB. Ultra-Processed Food Consumption and Gastrointestinal Cancer Risk: A Systematic Review and Meta-Analysis. Am J Gastroenterol. 2024;119:1056-1065. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 25] [Article Influence: 12.5] [Reference Citation Analysis (1)] |
| 10. | Hang D, Wang L, Fang Z, Du M, Wang K, He X, Khandpur N, Rossato SL, Wu K, Hu Z, Shen H, Ogino S, Chan AT, Giovannucci EL, Zhang FF, Song M. Ultra-processed food consumption and risk of colorectal cancer precursors: results from 3 prospective cohorts. J Natl Cancer Inst. 2023;115:155-164. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 39] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 11. | Monteiro CA, Cannon G, Moubarac JC, Levy RB, Louzada MLC, Jaime PC. The UN Decade of Nutrition, the NOVA food classification and the trouble with ultra-processing. Public Health Nutr. 2018;21:5-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1726] [Cited by in RCA: 1372] [Article Influence: 171.5] [Reference Citation Analysis (1)] |
| 12. | Severo JS, da Silva Barros VJ, Moraes Mendes PH, Dos Santos BLB, da Silva ACA, de Oliveira KBV, de Moura MSB, de Almeida Fonseca Viola PC, do Nascimento Nogueira N, Luz Parente JM, Lima MM, Dos Santos AA, Silva MTB. Phase angle values and ultra-processed food consumption are associated with changes in oxidative stress in inflammatory bowel disease patients. Clin Nutr ESPEN. 2023;57:10-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 13. | Spiller AL, Costa BGD, Yoshihara RNY, Nogueira EJZ, Castelhano NS, Santos A, Brusco De Freitas M, Magro DO, Yukie Sassaki L. Ultra-Processed Foods, Gut Microbiota, and Inflammatory Bowel Disease: A Critical Review of Emerging Evidence. Nutrients. 2025;17:2677. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (2)] |
| 14. | Whelan K, Bancil AS, Lindsay JO, Chassaing B. Ultra-processed foods and food additives in gut health and disease. Nat Rev Gastroenterol Hepatol. 2024;21:406-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 210] [Cited by in RCA: 154] [Article Influence: 77.0] [Reference Citation Analysis (2)] |
| 15. | Wong CC, Yu J. Gut microbiota in colorectal cancer development and therapy. Nat Rev Clin Oncol. 2023;20:429-452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 536] [Cited by in RCA: 453] [Article Influence: 151.0] [Reference Citation Analysis (1)] |
| 16. | Qu R, Zhang Y, Ma Y, Zhou X, Sun L, Jiang C, Zhang Z, Fu W. Role of the Gut Microbiota and Its Metabolites in Tumorigenesis or Development of Colorectal Cancer. Adv Sci (Weinh). 2023;10:e2205563. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 212] [Cited by in RCA: 166] [Article Influence: 55.3] [Reference Citation Analysis (1)] |
| 17. | Luo M, Wang Q, Sun Y, Jiang Y, Wang Q, Gu Y, Hu Z, Chen Q, Xu J, Chen S, Hou T, Feng L. Fasting-mimicking diet remodels gut microbiota and suppresses colorectal cancer progression. NPJ Biofilms Microbiomes. 2024;10:53. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 22] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 18. | Nan K, Zhong Z, Yue Y, Shen Y, Zhang H, Wang Z, Zhuma K, Yu B, Fu Y, Wang L, Sun X, Qu M, Chen Z, Guo M, Zhang J, Chu Y, Liu R, Miao C. Fasting-mimicking diet-enriched Bifidobacterium pseudolongum suppresses colorectal cancer by inducing memory CD8(+) T cells. Gut. 2025;74:775-786. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 24] [Article Influence: 24.0] [Reference Citation Analysis (1)] |
| 19. | Vernieri C, Fucà G, Ligorio F, Huber V, Vingiani A, Iannelli F, Raimondi A, Rinchai D, Frigè G, Belfiore A, Lalli L, Chiodoni C, Cancila V, Zanardi F, Ajazi A, Cortellino S, Vallacchi V, Squarcina P, Cova A, Pesce S, Frati P, Mall R, Corsetto PA, Rizzo AM, Ferraris C, Folli S, Garassino MC, Capri G, Bianchi G, Colombo MP, Minucci S, Foiani M, Longo VD, Apolone G, Torri V, Pruneri G, Bedognetti D, Rivoltini L, de Braud F. Fasting-Mimicking Diet Is Safe and Reshapes Metabolism and Antitumor Immunity in Patients with Cancer. Cancer Discov. 2022;12:90-107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 288] [Cited by in RCA: 254] [Article Influence: 63.5] [Reference Citation Analysis (1)] |
| 20. | Nencioni A, Caffa I, Cortellino S, Longo VD. Fasting and cancer: molecular mechanisms and clinical application. Nat Rev Cancer. 2018;18:707-719. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 443] [Cited by in RCA: 380] [Article Influence: 47.5] [Reference Citation Analysis (2)] |
| 21. | Hossain MS, Karuniawati H, Jairoun AA, Urbi Z, Ooi J, John A, Lim YC, Kibria KMK, Mohiuddin AKM, Ming LC, Goh KW, Hadi MA. Colorectal Cancer: A Review of Carcinogenesis, Global Epidemiology, Current Challenges, Risk Factors, Preventive and Treatment Strategies. Cancers (Basel). 2022;14:1732. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 744] [Cited by in RCA: 572] [Article Influence: 143.0] [Reference Citation Analysis (0)] |
| 22. | Malki A, ElRuz RA, Gupta I, Allouch A, Vranic S, Al Moustafa AE. Molecular Mechanisms of Colon Cancer Progression and Metastasis: Recent Insights and Advancements. Int J Mol Sci. 2020;22:130. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 320] [Cited by in RCA: 269] [Article Influence: 44.8] [Reference Citation Analysis (9)] |
| 23. | Nguyen HT, Duong HQ. The molecular characteristics of colorectal cancer: Implications for diagnosis and therapy. Oncol Lett. 2018;16:9-18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 141] [Article Influence: 17.6] [Reference Citation Analysis (2)] |
| 24. | Li Q, Geng S, Luo H, Wang W, Mo YQ, Luo Q, Wang L, Song GB, Sheng JP, Xu B. Signaling pathways involved in colorectal cancer: pathogenesis and targeted therapy. Signal Transduct Target Ther. 2024;9:266. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 398] [Cited by in RCA: 319] [Article Influence: 159.5] [Reference Citation Analysis (2)] |
| 25. | Pfohl-Leszkowicz A, Grosse Y, Carrière V, Cugnenc PH, Berger A, Carnot F, Beaune P, de Waziers I. High levels of DNA adducts in human colon are associated with colorectal cancer. Cancer Res. 1995;55:5611-5616. [PubMed] |
| 26. | Keum N, Giovannucci E. Global burden of colorectal cancer: emerging trends, risk factors and prevention strategies. Nat Rev Gastroenterol Hepatol. 2019;16:713-732. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2156] [Cited by in RCA: 1836] [Article Influence: 262.3] [Reference Citation Analysis (2)] |
| 27. | Sánchez-Alcoholado L, Ramos-Molina B, Otero A, Laborda-Illanes A, Ordóñez R, Medina JA, Gómez-Millán J, Queipo-Ortuño MI. The Role of the Gut Microbiome in Colorectal Cancer Development and Therapy Response. Cancers (Basel). 2020;12:1406. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 291] [Cited by in RCA: 238] [Article Influence: 39.7] [Reference Citation Analysis (4)] |
| 28. | Loke YL, Chew MT, Ngeow YF, Lim WWD, Peh SC. Colon Carcinogenesis: The Interplay Between Diet and Gut Microbiota. Front Cell Infect Microbiol. 2020;10:603086. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 106] [Cited by in RCA: 81] [Article Influence: 13.5] [Reference Citation Analysis (2)] |
| 29. | Mehta RS, Song M, Nishihara R, Drew DA, Wu K, Qian ZR, Fung TT, Hamada T, Masugi Y, da Silva A, Shi Y, Li W, Gu M, Willett WC, Fuchs CS, Giovannucci EL, Ogino S, Chan AT. Dietary Patterns and Risk of Colorectal Cancer: Analysis by Tumor Location and Molecular Subtypes. Gastroenterology. 2017;152:1944-1953. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 171] [Cited by in RCA: 157] [Article Influence: 17.4] [Reference Citation Analysis (1)] |
| 30. | Carethers JM, Jung BH. Genetics and Genetic Biomarkers in Sporadic Colorectal Cancer. Gastroenterology. 2015;149:1177-1190. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 356] [Cited by in RCA: 362] [Article Influence: 32.9] [Reference Citation Analysis (1)] |
| 31. | Toledo E, de A Carmona-Torre F, Alonso A, Puchau B, Zulet MA, Martinez JA, Martinez-Gonzalez MA. Hypothesis-oriented food patterns and incidence of hypertension: 6-year follow-up of the SUN (Seguimiento Universidad de Navarra) prospective cohort. Public Health Nutr. 2010;13:338-349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 40] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 32. | Baraldi LG, Martinez Steele E, Canella DS, Monteiro CA. Consumption of ultra-processed foods and associated sociodemographic factors in the USA between 2007 and 2012: evidence from a nationally representative cross-sectional study. BMJ Open. 2018;8:e020574. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 362] [Cited by in RCA: 314] [Article Influence: 39.3] [Reference Citation Analysis (1)] |
| 33. | Moubarac JC, Batal M, Louzada ML, Martinez Steele E, Monteiro CA. Consumption of ultra-processed foods predicts diet quality in Canada. Appetite. 2017;108:512-520. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 531] [Cited by in RCA: 445] [Article Influence: 49.4] [Reference Citation Analysis (2)] |
| 34. | Rauber F, da Costa Louzada ML, Steele EM, Millett C, Monteiro CA, Levy RB. Ultra-Processed Food Consumption and Chronic Non-Communicable Diseases-Related Dietary Nutrient Profile in the UK (2008⁻2014). Nutrients. 2018;10:587. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 493] [Cited by in RCA: 407] [Article Influence: 50.9] [Reference Citation Analysis (1)] |
| 35. | Zinöcker MK, Lindseth IA. The Western Diet-Microbiome-Host Interaction and Its Role in Metabolic Disease. Nutrients. 2018;10:365. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 503] [Cited by in RCA: 483] [Article Influence: 60.4] [Reference Citation Analysis (8)] |
| 36. | Wang L, Du M, Wang K, Khandpur N, Rossato SL, Drouin-Chartier JP, Steele EM, Giovannucci E, Song M, Zhang FF. Association of ultra-processed food consumption with colorectal cancer risk among men and women: results from three prospective US cohort studies. BMJ. 2022;378:e068921. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 69] [Article Influence: 17.3] [Reference Citation Analysis (1)] |
| 37. | Monteiro CA, Cannon G, Levy RB, Moubarac JC, Louzada ML, Rauber F, Khandpur N, Cediel G, Neri D, Martinez-Steele E, Baraldi LG, Jaime PC. Ultra-processed foods: what they are and how to identify them. Public Health Nutr. 2019;22:936-941. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2199] [Cited by in RCA: 1723] [Article Influence: 246.1] [Reference Citation Analysis (1)] |
| 38. | Romaguera D, Fernández-Barrés S, Gracia-Lavedán E, Vendrell E, Azpiri M, Ruiz-Moreno E, Martín V, Gómez-Acebo I, Obón M, Molinuevo A, Fresán U, Molina-Barceló A, Olmedo-Requena R, Tardón A, Alguacil J, Solans M, Huerta JM, Ruiz-Dominguez JM, Aragonés N, Fernández-Villa T, Dierssen-Sotos T, Moreno V, Guevara M, Vanaclocha-Espi M, Lozano-Lorca M, Fernández-Tardón G, Castaño-Vinyals G, Pérez-Gómez B, Molina AJ, Llorca J, Gil L, Castilla J, Pollán M, Kogevinas M, Amiano P. Consumption of ultra-processed foods and drinks and colorectal, breast, and prostate cancer. Clin Nutr. 2021;40:1537-1545. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 72] [Article Influence: 14.4] [Reference Citation Analysis (1)] |
| 39. | Lian Y, Wang GP, Chen GQ, Chen HN, Zhang GY. Association between ultra-processed foods and risk of cancer: a systematic review and meta-analysis. Front Nutr. 2023;10:1175994. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 40. | Murphy CC, Zaki TA. Changing epidemiology of colorectal cancer - birth cohort effects and emerging risk factors. Nat Rev Gastroenterol Hepatol. 2024;21:25-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 141] [Article Influence: 70.5] [Reference Citation Analysis (1)] |
| 41. | Chassaing B, Compher C, Bonhomme B, Liu Q, Tian Y, Walters W, Nessel L, Delaroque C, Hao F, Gershuni V, Chau L, Ni J, Bewtra M, Albenberg L, Bretin A, McKeever L, Ley RE, Patterson AD, Wu GD, Gewirtz AT, Lewis JD. Randomized Controlled-Feeding Study of Dietary Emulsifier Carboxymethylcellulose Reveals Detrimental Impacts on the Gut Microbiota and Metabolome. Gastroenterology. 2022;162:743-756. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 285] [Cited by in RCA: 235] [Article Influence: 58.8] [Reference Citation Analysis (1)] |
| 42. | Feng J, Cerniglia CE, Chen H. Toxicological significance of azo dye metabolism by human intestinal microbiota. Front Biosci (Elite Ed). 2012;4:568-586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 85] [Article Influence: 6.1] [Reference Citation Analysis (1)] |
| 43. | He Z, Chen L, Catalan-Dibene J, Bongers G, Faith JJ, Suebsuwong C, DeVita RJ, Shen Z, Fox JG, Lafaille JJ, Furtado GC, Lira SA. Food colorants metabolized by commensal bacteria promote colitis in mice with dysregulated expression of interleukin-23. Cell Metab. 2021;33:1358-1371. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 72] [Article Influence: 14.4] [Reference Citation Analysis (1)] |
| 44. | Zou L, Spanogiannopoulos P, Pieper LM, Chien HC, Cai W, Khuri N, Pottel J, Vora B, Ni Z, Tsakalozou E, Zhang W, Shoichet BK, Giacomini KM, Turnbaugh PJ. Bacterial metabolism rescues the inhibition of intestinal drug absorption by food and drug additives. Proc Natl Acad Sci U S A. 2020;117:16009-16018. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 45] [Article Influence: 7.5] [Reference Citation Analysis (1)] |
| 45. | Suez J, Korem T, Zeevi D, Zilberman-Schapira G, Thaiss CA, Maza O, Israeli D, Zmora N, Gilad S, Weinberger A, Kuperman Y, Harmelin A, Kolodkin-Gal I, Shapiro H, Halpern Z, Segal E, Elinav E. Artificial sweeteners induce glucose intolerance by altering the gut microbiota. Nature. 2014;514:181-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1762] [Cited by in RCA: 1446] [Article Influence: 120.5] [Reference Citation Analysis (2)] |
| 46. | Frioux C, Ansorge R, Özkurt E, Ghassemi Nedjad C, Fritscher J, Quince C, Waszak SM, Hildebrand F. Enterosignatures define common bacterial guilds in the human gut microbiome. Cell Host Microbe. 2023;31:1111-1125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 87] [Article Influence: 29.0] [Reference Citation Analysis (1)] |
| 47. | Ross FC, Patangia D, Grimaud G, Lavelle A, Dempsey EM, Ross RP, Stanton C. The interplay between diet and the gut microbiome: implications for health and disease. Nat Rev Microbiol. 2024;22:671-686. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 440] [Cited by in RCA: 337] [Article Influence: 168.5] [Reference Citation Analysis (1)] |
| 48. | Zhou X, Qiao K, Wu H, Zhang Y. The Impact of Food Additives on the Abundance and Composition of Gut Microbiota. Molecules. 2023;28:631. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 40] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 49. | White MT, Sears CL. The microbial landscape of colorectal cancer. Nat Rev Microbiol. 2024;22:240-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 102] [Article Influence: 51.0] [Reference Citation Analysis (1)] |
| 50. | Zhang H, Jin K, Xiong K, Jing W, Pang Z, Feng M, Cheng X. Disease-associated gut microbiome and critical metabolomic alterations in patients with colorectal cancer. Cancer Med. 2023;12:15720-15735. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 51. | Thomas AM, Manghi P, Asnicar F, Pasolli E, Armanini F, Zolfo M, Beghini F, Manara S, Karcher N, Pozzi C, Gandini S, Serrano D, Tarallo S, Francavilla A, Gallo G, Trompetto M, Ferrero G, Mizutani S, Shiroma H, Shiba S, Shibata T, Yachida S, Yamada T, Wirbel J, Schrotz-King P, Ulrich CM, Brenner H, Arumugam M, Bork P, Zeller G, Cordero F, Dias-Neto E, Setubal JC, Tett A, Pardini B, Rescigno M, Waldron L, Naccarati A, Segata N. Metagenomic analysis of colorectal cancer datasets identifies cross-cohort microbial diagnostic signatures and a link with choline degradation. Nat Med. 2019;25:667-678. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 848] [Cited by in RCA: 687] [Article Influence: 98.1] [Reference Citation Analysis (2)] |
| 52. | Liu NN, Jiao N, Tan JC, Wang Z, Wu D, Wang AJ, Chen J, Tao L, Zhou C, Fang W, Cheong IH, Pan W, Liao W, Kozlakidis Z, Heeschen C, Moore GG, Zhu L, Chen X, Zhang G, Zhu R, Wang H. Multi-kingdom microbiota analyses identify bacterial-fungal interactions and biomarkers of colorectal cancer across cohorts. Nat Microbiol. 2022;7:238-250. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 245] [Cited by in RCA: 212] [Article Influence: 53.0] [Reference Citation Analysis (5)] |
| 53. | Mouradov D, Greenfield P, Li S, In EJ, Storey C, Sakthianandeswaren A, Georgeson P, Buchanan DD, Ward RL, Hawkins NJ, Skinner I, Jones IT, Gibbs P, Ma C, Liew YJ, Fung KYC, Sieber OM. Oncomicrobial Community Profiling Identifies Clinicomolecular and Prognostic Subtypes of Colorectal Cancer. Gastroenterology. 2023;165:104-120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 69] [Article Influence: 23.0] [Reference Citation Analysis (1)] |
| 54. | Wirbel J, Pyl PT, Kartal E, Zych K, Kashani A, Milanese A, Fleck JS, Voigt AY, Palleja A, Ponnudurai R, Sunagawa S, Coelho LP, Schrotz-King P, Vogtmann E, Habermann N, Niméus E, Thomas AM, Manghi P, Gandini S, Serrano D, Mizutani S, Shiroma H, Shiba S, Shibata T, Yachida S, Yamada T, Waldron L, Naccarati A, Segata N, Sinha R, Ulrich CM, Brenner H, Arumugam M, Bork P, Zeller G. Meta-analysis of fecal metagenomes reveals global microbial signatures that are specific for colorectal cancer. Nat Med. 2019;25:679-689. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1160] [Cited by in RCA: 902] [Article Influence: 128.9] [Reference Citation Analysis (5)] |
| 55. | de Groot S, Vreeswijk MP, Welters MJ, Gravesteijn G, Boei JJ, Jochems A, Houtsma D, Putter H, van der Hoeven JJ, Nortier JW, Pijl H, Kroep JR. The effects of short-term fasting on tolerance to (neo) adjuvant chemotherapy in HER2-negative breast cancer patients: a randomized pilot study. BMC Cancer. 2015;15:652. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 214] [Cited by in RCA: 181] [Article Influence: 16.5] [Reference Citation Analysis (1)] |
| 56. | Chen J, Su R, He Y, Chen J. Intermittent fasting inhibits the development of colorectal cancer in APC (Min/+) mice through gut microbiota and its related metabolites. Front Microbiol. 2025;16:1563224. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 57. | Song Q, Gao Y, Liu K, Tang Y, Man Y, Wu H. Gut microbial and metabolomics profiles reveal the potential mechanism of fecal microbiota transplantation in modulating the progression of colitis-associated colorectal cancer in mice. J Transl Med. 2024;22:1028. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 23] [Article Influence: 11.5] [Reference Citation Analysis (1)] |
| 58. | Peñalver Bernabé B, Oliveira ML, Wolf PG, McLeod A, Gabel K, Cares K, Robinson N, DiPiazza B, Varady K, Tussing-Humphreys L. Intermittent Fasting: Implications for Obesity-Related Colorectal Tumorigenesis. Endocrinol Metab Clin North Am. 2025;54:61-83. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 59. | Bensalem J, Teong XT, Hattersley KJ, Hein LK, Fourrier C, Dang LVP, Singh S, Liu K, Wittert GA, Hutchison AT, Heilbronn LK, Sargeant TJ. Intermittent time-restricted eating may increase autophagic flux in humans: an exploratory analysis. J Physiol. 2025;603:3019-3032. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 60. | Di Biase S, Lee C, Brandhorst S, Manes B, Buono R, Cheng CW, Cacciottolo M, Martin-Montalvo A, de Cabo R, Wei M, Morgan TE, Longo VD. Fasting-Mimicking Diet Reduces HO-1 to Promote T Cell-Mediated Tumor Cytotoxicity. Cancer Cell. 2016;30:136-146. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 369] [Cited by in RCA: 333] [Article Influence: 33.3] [Reference Citation Analysis (1)] |
| 61. | Caccialanza R, Aprile G, Cereda E, Pedrazzoli P. Fasting in oncology: a word of caution. Nat Rev Cancer. 2019;19:177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 26] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 62. | Wang Q, Li K, Li L, Li Q, Qi Y, Liu K, Yuan H, Lin P. The role and therapeutic potential of glucose metabolism in multidrug resistance of cancer. Front Cell Dev Biol. 2025;13:1584630. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 63. | Wang B, Wang C, Li H. The impact of intermittent fasting on body composition and cardiometabolic outcomes in overweight and obese adults: a systematic review and meta-analysis of randomized controlled trials. Nutr J. 2025;24:120. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 64. | Kibret KT, Peeters A, Tegegne TK, Mesfin YM, Nichols M. Intermittent Fasting for the Prevention of Cardiovascular Disease Risks: Systematic Review and Network Meta-Analysis. Curr Nutr Rep. 2025;14:93. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 65. | Yüzbaşıoğlu M, Özder A. Comparison of time-restricted eating and a six-meal diet: Effects on body composition and biochemical parameters. Asia Pac J Clin Nutr. 2025;34:589-595. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 66. | Habe B, Črešnovar T, Petelin A, Kenig S, Mohorko N, Jenko Pražnikar Z. Comparing the influence of early and late time-restricted eating with energy restriction and energy restriction alone on cardiometabolic markers, metabolic hormones and appetite in adults with overweight/obesity: per-protocol analysis of a 3-month randomized clinical trial. Nutr Metab (Lond). 2025;22:85. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 67. | Martínez-Montoro JI, Bandera B, Gutiérrez-Bedmar M, Gómez-Pérez AM, Macías-González M, Moreno-Indias I, Tinahones FJ. Effect of a ketogenic diet, time-restricted eating, or alternate-day fasting on weight loss in adults with obesity: a randomized clinical trial. BMC Med. 2025;23:368. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 68. | Brandhorst S, Levine ME, Wei M, Shelehchi M, Morgan TE, Nayak KS, Dorff T, Hong K, Crimmins EM, Cohen P, Longo VD. Fasting-mimicking diet causes hepatic and blood markers changes indicating reduced biological age and disease risk. Nat Commun. 2024;15:1309. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 35] [Article Influence: 17.5] [Reference Citation Analysis (1)] |
| 69. | Rangan P, Lobo F, Parrella E, Rochette N, Morselli M, Stephen TL, Cremonini AL, Tagliafico L, Persia A, Caffa I, Monacelli F, Odetti P, Bonfiglio T, Nencioni A, Pigliautile M, Boccardi V, Mecocci P, Pike CJ, Cohen P, LaDu MJ, Pellegrini M, Xia K, Tran K, Ann B, Chowdhury D, Longo VD. Fasting-mimicking diet cycles reduce neuroinflammation to attenuate cognitive decline in Alzheimer's models. Cell Rep. 2022;40:111417. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 52] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 70. | Bustamante-Marin X, Devlin KL, McDonell SB, Dave O, Merlino JL, Grindstaff EJ, Ho AN, Rezeli ET, Coleman MF, Hursting SD. Regulation of IGF1R by MicroRNA-15b Contributes to the Anticancer Effects of Calorie Restriction in a Murine C3-TAg Model of Triple-Negative Breast Cancer. Cancers (Basel). 2023;15:4320. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 71. | Malinowska D, Żendzian-Piotrowska M. Ketogenic Diet: A Review of Composition Diversity, Mechanism of Action and Clinical Application. J Nutr Metab. 2024;2024:6666171. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 26] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 72. | Teong XT, Liu K, Vincent AD, Bensalem J, Liu B, Hattersley KJ, Zhao L, Feinle-Bisset C, Sargeant TJ, Wittert GA, Hutchison AT, Heilbronn LK. Intermittent fasting plus early time-restricted eating versus calorie restriction and standard care in adults at risk of type 2 diabetes: a randomized controlled trial. Nat Med. 2023;29:963-972. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 91] [Article Influence: 30.3] [Reference Citation Analysis (1)] |
| 73. | Schüssler IC, Pappe CL, von Scheidt C, Peters B, Dommisch H, Kessler C, Michalsen A, Koppold DA, Pivovarova-Ramich O. Dietary assessment in intermittent fasting: validation of a short food frequency questionnaire vs. food records in diurnal dry fasting and time-restricted eating. Front Nutr. 2025;12:1552990. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 74. | Gonzalo-Encabo P, Giobbie-Hurder A, Sears DD, Mayer EL, Partridge AH, Poorvu PD, Waks AG, Tanasijevic AM, Crane TE, Dieli-Conwright CM, Ligibel JA. Combined intermittent fasting and exercise intervention in patients with metastatic, hormone receptor-positive breast cancer (FastER): a pilot study. Breast Cancer Res Treat. 2025;214:171-180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 75. | Elahy V, Wang Y, Flanders WD, Zhong C, McCullough ML. Overnight Fasting and Body Weight: Emulated Target Trial Using Cancer Prevention Study-3 Data. Nutrients. 2025;17:1559. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 76. | Meng W, Leung JW, Wang Z, Li Q, Zhang L, Zhang K, Wang X, Wang M, Wang Q, Shao Y, Zhang J, Yue P, Zhang L, Zhu K, Zhu X, Zhang H, Hou S, Cai K, Sun H, Xue P, Liu W, Wang H, Zhang L, Ding S, Yang Z, Zhang M, Weng H, Wu Q, Chen B, Jiang T, Wang Y, Zhang L, Wu K, Yang X, Wen Z, Liu C, Miao L, Wang Z, Li J, Yan X, Wang F, Zhang L, Bai M, Mi N, Zhang X, Zhou W, Yuan J, Suzuki A, Tanaka K, Liu J, Nur U, Weiderpass E, Li X. Safety of high-carbohydrate fluid diet 2 h versus overnight fasting before non-emergency endoscopic retrograde cholangiopancreatography: A single-blind, multicenter, randomized controlled trial. Chin Med J (Engl). 2024;137:1437-1446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 77. | Tsuda M, Ishiguro H, Toriguchi N, Masuda N, Bando H, Ohgami M, Homma M, Morita S, Yamamoto N, Kuroi K, Yanagita Y, Takano T, Shimizu S, Toi M. Overnight fasting before lapatinib administration to breast cancer patients leads to reduced toxicity compared with nighttime dosing: a retrospective cohort study from a randomized clinical trial. Cancer Med. 2020;9:9246-9255. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 78. | Khodabakhshi A, Akbari ME, Mirzaei HR, Seyfried TN, Kalamian M, Davoodi SH. Effects of Ketogenic metabolic therapy on patients with breast cancer: A randomized controlled clinical trial. Clin Nutr. 2021;40:751-758. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 90] [Article Influence: 18.0] [Reference Citation Analysis (1)] |
| 79. | Mindikoglu AL, Abdulsada MM, Jain A, Jalal PK, Devaraj S, Wilhelm ZR, Opekun AR, Jung SY. Intermittent fasting from dawn to sunset for four consecutive weeks induces anticancer serum proteome response and improves metabolic syndrome. Sci Rep. 2020;10:18341. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 46] [Article Influence: 7.7] [Reference Citation Analysis (1)] |
| 80. | Rangan P, Choi I, Wei M, Navarrete G, Guen E, Brandhorst S, Enyati N, Pasia G, Maesincee D, Ocon V, Abdulridha M, Longo VD. Fasting-Mimicking Diet Modulates Microbiota and Promotes Intestinal Regeneration to Reduce Inflammatory Bowel Disease Pathology. Cell Rep. 2019;26:2704-2719. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 273] [Cited by in RCA: 234] [Article Influence: 33.4] [Reference Citation Analysis (1)] |
| 81. | Caffa I, Spagnolo V, Vernieri C, Valdemarin F, Becherini P, Wei M, Brandhorst S, Zucal C, Driehuis E, Ferrando L, Piacente F, Tagliafico A, Cilli M, Mastracci L, Vellone VG, Piazza S, Cremonini AL, Gradaschi R, Mantero C, Passalacqua M, Ballestrero A, Zoppoli G, Cea M, Arrighi A, Odetti P, Monacelli F, Salvadori G, Cortellino S, Clevers H, De Braud F, Sukkar SG, Provenzani A, Longo VD, Nencioni A. Fasting-mimicking diet and hormone therapy induce breast cancer regression. Nature. 2020;583:620-624. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 304] [Cited by in RCA: 275] [Article Influence: 45.8] [Reference Citation Analysis (6)] |
| 82. | de Groot S, Lugtenberg RT, Cohen D, Welters MJP, Ehsan I, Vreeswijk MPG, Smit VTHBM, de Graaf H, Heijns JB, Portielje JEA, van de Wouw AJ, Imholz ALT, Kessels LW, Vrijaldenhoven S, Baars A, Kranenbarg EM, Carpentier MD, Putter H, van der Hoeven JJM, Nortier JWR, Longo VD, Pijl H, Kroep JR; Dutch Breast Cancer Research Group (BOOG). Fasting mimicking diet as an adjunct to neoadjuvant chemotherapy for breast cancer in the multicentre randomized phase 2 DIRECT trial. Nat Commun. 2020;11:3083. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 281] [Cited by in RCA: 235] [Article Influence: 39.2] [Reference Citation Analysis (1)] |
| 83. | Zhong Z, Zhang H, Nan K, Zhong J, Wu Q, Lu L, Yue Y, Zhang Z, Guo M, Wang Z, Xia J, Xing Y, Fu Y, Yu B, Zhou W, Sun X, Shen Y, Chen W, Zhang J, Zhang J, Ma D, Chu Y, Liu R, Miao C. Fasting-Mimicking Diet Drives Antitumor Immunity against Colorectal Cancer by Reducing IgA-Producing Cells. Cancer Res. 2023;83:3529-3543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 36] [Article Influence: 12.0] [Reference Citation Analysis (1)] |
| 84. | Ligorio F, Fucà G, Provenzano L, Lobefaro R, Zanenga L, Vingiani A, Belfiore A, Lorenzoni A, Alessi A, Pruneri G, de Braud F, Vernieri C. Exceptional tumor responses to fasting-mimicking diet combined with standard anticancer therapies: A sub-analysis of the NCT03340935 trial. Eur J Cancer. 2022;172:300-310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 57] [Article Influence: 14.3] [Reference Citation Analysis (1)] |
| 85. | Ligorio F, Vingiani A, Torelli T, Sposetti C, Drufuca L, Iannelli F, Zanenga L, Depretto C, Folli S, Scaperrotta G, Capri G, Bianchi GV, Ferraris C, Martelli G, Maugeri I, Provenzano L, Nichetti F, Agnelli L, Lobefaro R, Fucà G, Fotia G, Mariani L, Morelli D, Ladisa V, De Santis MC, Lozza L, Trecate G, Belfiore A, Brich S, Bertolotti A, Lorenzini D, Ficchì A, Martinetti A, Sottotetti E, Arata A, Corsetto P, Sorrentino L, Rediti M, Salvadori G, Minucci S, Foiani M, Apolone G, Pagani M, Pruneri G, de Braud F, Vernieri C. Early downmodulation of tumor glycolysis predicts response to fasting-mimicking diet in triple-negative breast cancer patients. Cell Metab. 2025;37:330-344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 14.0] [Reference Citation Analysis (1)] |
| 86. | Micarelli A, Mrakic-Sposta S, Vezzoli A, Malacrida S, Caputo S, Micarelli B, Misici I, Carbini V, Iennaco I, Granito I, Longo VD, Alessandrini M. Chemosensory and cardiometabolic improvements after a fasting-mimicking diet: A randomized cross-over clinical trial. Cell Rep Med. 2025;6:101971. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 87. | Bush CO, Perry RJ. Molecular Mechanism of Fasting-Mimicking Diet in Inhibiting Colorectal Cancer Progression: Implications for Immune Therapy and Metabolic Regulation. Cancer Res. 2023;83:3493-3494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 88. | Vidoni C, Ferraresi A, Esposito A, Maheshwari C, Dhanasekaran DN, Mollace V, Isidoro C. Calorie Restriction for Cancer Prevention and Therapy: Mechanisms, Expectations, and Efficacy. J Cancer Prev. 2021;26:224-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 46] [Article Influence: 9.2] [Reference Citation Analysis (1)] |
| 89. | Caprara G, Pallavi R, Sanyal S, Pelicci PG. Dietary Restrictions and Cancer Prevention: State of the Art. Nutrients. 2025;17:503. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 90. | Pistollato F, Forbes-Hernandez TY, Iglesias RC, Ruiz R, Elexpuru Zabaleta M, Dominguez I, Cianciosi D, Quiles JL, Giampieri F, Battino M. Effects of caloric restriction on immunosurveillance, microbiota and cancer cell phenotype: Possible implications for cancer treatment. Semin Cancer Biol. 2021;73:45-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (1)] |
| 91. | Ibrahim EM, Al-Foheidi MH, Al-Mansour MM. Energy and caloric restriction, and fasting and cancer: a narrative review. Support Care Cancer. 2021;29:2299-2304. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 92. | Napoleão A, Fernandes L, Miranda C, Marum AP. Effects of Calorie Restriction on Health Span and Insulin Resistance: Classic Calorie Restriction Diet vs. Ketosis-Inducing Diet. Nutrients. 2021;13:1302. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 57] [Article Influence: 11.4] [Reference Citation Analysis (1)] |
| 93. | Yong-Quan Ng G, Yang-Wei Fann D, Jo DG, Sobey CG, Arumugam TV. Dietary Restriction and Epigenetics: Part I. Cond Med. 2019;2:284-299. [PubMed] |
| 94. | Chaudhary S, Chaudhary MR, Jena MK, Rath PK, Mishra BP, Paital B, Vyas A, Sahoo DK, Singh P, Mehdi MM. Calorie restriction mimetics against aging and inflammation. Biogerontology. 2025;26:126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 95. | Werner H. Tumor suppressors govern insulin-like growth factor signaling pathways: implications in metabolism and cancer. Oncogene. 2012;31:2703-2714. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 105] [Article Influence: 7.5] [Reference Citation Analysis (1)] |
| 96. | Klement RJ, Fink MK. Dietary and pharmacological modification of the insulin/IGF-1 system: exploiting the full repertoire against cancer. Oncogenesis. 2016;5:e193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 80] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 97. | Pomatto-Watson LCD, Bodogai M, Carpenter M, Chowdhury D, Krishna P, Ng S, Bosompra O, Kato J, Wong S, Reyes-Sepulveda C, Bernier M, Price NL, Biragyn A, de Cabo R. Replenishment of myeloid-derived suppressor cells (MDSCs) overrides CR-mediated protection against tumor growth in a murine model of triple-negative breast cancer. Geroscience. 2022;44:2471-2490. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 98. | Brandhorst S, Longo VD. Fasting and Caloric Restriction in Cancer Prevention and Treatment. Recent Results Cancer Res. 2016;207:241-266. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 135] [Cited by in RCA: 112] [Article Influence: 11.2] [Reference Citation Analysis (1)] |
| 99. | Koc Yildirim E, Balkaya M. Dynamics of breast tumor incidence, tumor volume and serum metabolic hormones in calorie restricted rats. Biotech Histochem. 2021;96:339-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 100. | Lin S, Zhang Q, Shao X, Zhang T, Xue C, Shi S, Zhao D, Lin Y. IGF-1 promotes angiogenesis in endothelial cells/adipose-derived stem cells co-culture system with activation of PI3K/Akt signal pathway. Cell Prolif. 2017;50:e12390. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 132] [Cited by in RCA: 116] [Article Influence: 12.9] [Reference Citation Analysis (1)] |
| 101. | Piscitello D, Varshney D, Lilla S, Vizioli MG, Reid C, Gorbunova V, Seluanov A, Gillespie DA, Adams PD. AKT overactivation can suppress DNA repair via p70S6 kinase-dependent downregulation of MRE11. Oncogene. 2018;37:427-438. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 45] [Article Influence: 5.6] [Reference Citation Analysis (1)] |
| 102. | Saxton RA, Sabatini DM. mTOR Signaling in Growth, Metabolism, and Disease. Cell. 2017;168:960-976. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5642] [Cited by in RCA: 4890] [Article Influence: 543.3] [Reference Citation Analysis (2)] |
| 103. | De-Leon-Covarrubias UE, Perez-Trujillo JJ, Villa-Cedillo SA, Martinez-Perez AG, Montes-de-Oca-Saucedo CR, Loera-Arias MJ, Garcia-Garcia A, Saucedo-Cardenas O, Montes-de-Oca-Luna R. Unlocking the Potential: Caloric Restriction, Caloric Restriction Mimetics, and Their Impact on Cancer Prevention and Treatment. Metabolites. 2024;14:418. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 104. | Jensen NJ, Wodschow HZ, Nilsson M, Rungby J. Effects of Ketone Bodies on Brain Metabolism and Function in Neurodegenerative Diseases. Int J Mol Sci. 2020;21:8767. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 388] [Cited by in RCA: 303] [Article Influence: 50.5] [Reference Citation Analysis (1)] |
| 105. | Cantrell CB, Mohiuddin SS. Biochemistry, Ketone Metabolism. 2023 Apr 24. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. [PubMed] |
| 106. | Kolb H, Kempf K, Röhling M, Lenzen-Schulte M, Schloot NC, Martin S. Ketone bodies: from enemy to friend and guardian angel. BMC Med. 2021;19:313. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 298] [Cited by in RCA: 254] [Article Influence: 50.8] [Reference Citation Analysis (1)] |
| 107. | Houten SM, Wanders RJ. A general introduction to the biochemistry of mitochondrial fatty acid β-oxidation. J Inherit Metab Dis. 2010;33:469-477. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 872] [Cited by in RCA: 748] [Article Influence: 46.8] [Reference Citation Analysis (2)] |
| 108. | Newman JC, Verdin E. Ketone bodies as signaling metabolites. Trends Endocrinol Metab. 2014;25:42-52. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 858] [Cited by in RCA: 757] [Article Influence: 63.1] [Reference Citation Analysis (2)] |
| 109. | Puchalska P, Crawford PA. Multi-dimensional Roles of Ketone Bodies in Fuel Metabolism, Signaling, and Therapeutics. Cell Metab. 2017;25:262-284. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1433] [Cited by in RCA: 1245] [Article Influence: 138.3] [Reference Citation Analysis (5)] |
| 110. | Hwang CY, Choe W, Yoon KS, Ha J, Kim SS, Yeo EJ, Kang I. Molecular Mechanisms for Ketone Body Metabolism, Signaling Functions, and Therapeutic Potential in Cancer. Nutrients. 2022;14:4932. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 94] [Cited by in RCA: 76] [Article Influence: 19.0] [Reference Citation Analysis (1)] |
| 111. | Fulghum K, Salathe SF, Davis X, Thyfault JP, Puchalska P, Crawford PA. Ketone body metabolism and cardiometabolic implications for cognitive health. NPJ Metab Health Dis. 2024;2:29. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 112. | Liang Z, Deng L, Zhou X, Zhang Z, Zhao W. Comprehensive Overview of Ketone Bodies in Cancer Metabolism: Mechanisms and Application. Biomedicines. 2025;13:210. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 113. | Montrose DC, Galluzzi L. Restored Ketosis Drives Anticancer Immunity in Colorectal Cancer. Cancer Res. 2022;82:1464-1466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 114. | Tsenkova M, Brauer M, Pozdeev VI, Kasakin M, Busi SB, Schmoetten M, Cheung D, Meyers M, Rodriguez F, Gaigneaux A, Koncina E, Gilson C, Schlicker L, Herebian D, Schmitz M, de Nies L, Mayatepek E, Haan S, de Beaufort C, Cramer T, Meiser J, Linster CL, Wilmes P, Letellier E. Ketogenic diet suppresses colorectal cancer through the gut microbiome long chain fatty acid stearate. Nat Commun. 2025;16:1792. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 26] [Article Influence: 26.0] [Reference Citation Analysis (1)] |
| 115. | Sun W, Yang J, Liu B, Liu Q, Wang T, Wang Q, Liu M, Li L, Wang Z, Li S, Siebert H, Zhang N. Ketogenic diet inhibits tumor growth by enhancing immune response, attenuating immunosuppression, inhibiting angiogenesis and EMT in CT26 colon tumor allografts mouse model. J Funct Foods. 2022;92:105067. [RCA] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 116. | Jiang H, Zeng Y, Jiang X, Xu X, Zhao L, Yuan X, Xu J, Zhao M, Wu F, Li G. Ketogenesis attenuated KLF5 disrupts iron homeostasis via LIF to confer oxaliplatin vulnerability in colorectal cancer. Biochim Biophys Acta Mol Basis Dis. 2024;1870:167210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 117. | Klement RJ, Koebrunner PS, Meyer D, Kanzler S, Sweeney RA. Impact of a ketogenic diet intervention during radiotherapy on body composition: IV. Final results of the KETOCOMP study for rectal cancer patients. Clin Nutr. 2021;40:4674-4684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (1)] |
| 118. | Egashira R, Matsunaga M, Miyake A, Hotta S, Nagai N, Yamaguchi C, Takeuchi M, Moriguchi M, Tonari S, Nakano M, Saito H, Hagihara K. Long-Term Effects of a Ketogenic Diet for Cancer. Nutrients. 2023;15:2334. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 119. | Attique I, Haider Z, Khan M, Hassan S, Soliman MM, Ibrahim WN, Anjum S. Reactive Oxygen Species: From Tumorigenesis to Therapeutic Strategies in Cancer. Cancer Med. 2025;14:e70947. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 34] [Article Influence: 34.0] [Reference Citation Analysis (1)] |
| 120. | Stine ZE, Schug ZT, Salvino JM, Dang CV. Targeting cancer metabolism in the era of precision oncology. Nat Rev Drug Discov. 2022;21:141-162. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1022] [Cited by in RCA: 878] [Article Influence: 219.5] [Reference Citation Analysis (1)] |
| 121. | Buono R, Longo VD. Starvation, Stress Resistance, and Cancer. Trends Endocrinol Metab. 2018;29:271-280. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 141] [Cited by in RCA: 118] [Article Influence: 14.8] [Reference Citation Analysis (1)] |
| 122. | Hernandez GA, Perera RM. Autophagy in cancer cell remodeling and quality control. Mol Cell. 2022;82:1514-1527. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 58] [Article Influence: 14.5] [Reference Citation Analysis (1)] |
| 123. | Kim W, Lee S, Seo D, Kim D, Kim K, Kim E, Kang J, Seong KM, Youn H, Youn B. Cellular Stress Responses in Radiotherapy. Cells. 2019;8:1105. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 272] [Cited by in RCA: 239] [Article Influence: 34.1] [Reference Citation Analysis (1)] |
| 124. | Bian C, Zheng Z, Su J, Wang H, Chang S, Xin Y, Jiang X. Targeting Mitochondrial Metabolism to Reverse Radioresistance: An Alternative to Glucose Metabolism. Antioxidants (Basel). 2022;11:2202. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 125. | Read GH, Bailleul J, Vlashi E, Kesarwala AH. Metabolic response to radiation therapy in cancer. Mol Carcinog. 2022;61:200-224. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 126. | Falcone M, Uribe AH, Papalazarou V, Newman AC, Athineos D, Stevenson K, Sauvé CG, Gao Y, Kim JK, Del Latto M, Kierstead M, Wu C, Smith JJ, Romesser PB, Chalmers AJ, Blyth K, Maddocks ODK. Sensitisation of cancer cells to radiotherapy by serine and glycine starvation. Br J Cancer. 2022;127:1773-1786. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 43] [Article Influence: 10.8] [Reference Citation Analysis (1)] |
| 127. | Mundi MS, Mohamed Elfadil O, Patel I, Patel J, Hurt RT. Ketogenic diet and cancer: Fad or fabulous? JPEN J Parenter Enteral Nutr. 2021;45:26-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 128. | Hu J, Wang Y, Ji X, Zhang Y, Li K, Huang F. Non-Pharmacological Strategies for Managing Sarcopenia in Chronic Diseases. Clin Interv Aging. 2024;19:827-841. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 15] [Article Influence: 7.5] [Reference Citation Analysis (1)] |
| 129. | Pan S, Fan M, Liu Z, Li X, Wang H. Serine, glycine and onecarbon metabolism in cancer (Review). Int J Oncol. 2021;58:158-170. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 177] [Cited by in RCA: 156] [Article Influence: 31.2] [Reference Citation Analysis (1)] |
| 130. | Amelio I, Cutruzzolá F, Antonov A, Agostini M, Melino G. Serine and glycine metabolism in cancer. Trends Biochem Sci. 2014;39:191-198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 933] [Cited by in RCA: 837] [Article Influence: 69.8] [Reference Citation Analysis (2)] |
| 131. | Petrova B, Maynard AG, Wang P, Kanarek N. Regulatory mechanisms of one-carbon metabolism enzymes. J Biol Chem. 2023;299:105457. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 63] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 132. | Locasale JW. Serine, glycine and one-carbon units: cancer metabolism in full circle. Nat Rev Cancer. 2013;13:572-583. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1469] [Cited by in RCA: 1294] [Article Influence: 99.5] [Reference Citation Analysis (2)] |
| 133. | Fang L, Hao Y, Yu H, Gu X, Peng Q, Zhuo H, Li Y, Liu Z, Wang J, Chen Y, Zhang J, Tian H, Gao Y, Gao R, Teng H, Shan Z, Zhu J, Li Z, Liu Y, Zhang Y, Yu F, Lin Z, Hao Y, Ge X, Yuan J, Hu HG, Ma Y, Qin HL, Wang P. Methionine restriction promotes cGAS activation and chromatin untethering through demethylation to enhance antitumor immunity. Cancer Cell. 2023;41:1118-1133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 91] [Article Influence: 30.3] [Reference Citation Analysis (0)] |
| 134. | Wang QL, Chen Z, Lu X, Lin H, Feng H, Weng N, Chen L, Liu M, Long L, Huang L, Deng Y, Zheng K, Zheng X, Li Y, Cai T, Zheng J, Yang W. Methionine Metabolism Dictates PCSK9 Expression and Antitumor Potency of PD-1 Blockade in MSS Colorectal Cancer. Adv Sci (Weinh). 2025;12:e2501623. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 135. | Ma C, Xu A, Zuo L, Li Q, Fan F, Hu Y, Sun C. Methionine Dependency and Restriction in Cancer: Exploring the Pathogenic Function and Therapeutic Potential. Pharmaceuticals (Basel). 2025;18:640. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 136. | Tassinari V, Jia W, Chen WL, Candi E, Melino G. The methionine cycle and its cancer implications. Oncogene. 2024;43:3483-3488. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 137. | Lauinger L, Kaiser P. Sensing and Signaling of Methionine Metabolism. Metabolites. 2021;11:83. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 120] [Cited by in RCA: 101] [Article Influence: 20.2] [Reference Citation Analysis (1)] |
| 138. | Pascale RM, Peitta G, Simile MM, Feo F. Alterations of Methionine Metabolism as Potential Targets for the Prevention and Therapy of Hepatocellular Carcinoma. Medicina (Kaunas). 2019;55:296. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 34] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 139. | Jin X, Liu L, Liu D, Wu J, Wang C, Wang S, Wang F, Yu G, Jin X, Xue YW, Jiang D, Ni Y, Yang X, Wang MS, Wang ZW, Orlov YL, Jia W, Melino G, Liu JB, Chen WL. Unveiling the methionine cycle: a key metabolic signature and NR4A2 as a methionine-responsive oncogene in esophageal squamous cell carcinoma. Cell Death Differ. 2024;31:558-573. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 12.0] [Reference Citation Analysis (1)] |
| 140. | Yu M, Zhang S. Influenced tumor microenvironment and tumor immunity by amino acids. Front Immunol. 2023;14:1118448. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (1)] |
| 141. | Luengo A, Gui DY, Vander Heiden MG. Targeting Metabolism for Cancer Therapy. Cell Chem Biol. 2017;24:1161-1180. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 815] [Cited by in RCA: 716] [Article Influence: 79.6] [Reference Citation Analysis (1)] |
| 142. | Wang Z, Liu F, Fan N, Zhou C, Li D, Macvicar T, Dong Q, Bruns CJ, Zhao Y. Targeting Glutaminolysis: New Perspectives to Understand Cancer Development and Novel Strategies for Potential Target Therapies. Front Oncol. 2020;10:589508. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 133] [Cited by in RCA: 127] [Article Influence: 21.2] [Reference Citation Analysis (1)] |
| 143. | Wei Z, Liu X, Cheng C, Yu W, Yi P. Metabolism of Amino Acids in Cancer. Front Cell Dev Biol. 2020;8:603837. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 311] [Cited by in RCA: 259] [Article Influence: 51.8] [Reference Citation Analysis (1)] |
| 144. | Zhu M, Hu Y, Gu Y, Lin X, Jiang X, Gong C, Fang Z. Role of amino acid metabolism in tumor immune microenvironment of colorectal cancer. Am J Cancer Res. 2025;15:233-247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 18.0] [Reference Citation Analysis (1)] |
| 145. | Sivanand S, Vander Heiden MG. Emerging Roles for Branched-Chain Amino Acid Metabolism in Cancer. Cancer Cell. 2020;37:147-156. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 417] [Cited by in RCA: 382] [Article Influence: 63.7] [Reference Citation Analysis (1)] |
| 146. | Tian T, Li X, Zhang J. mTOR Signaling in Cancer and mTOR Inhibitors in Solid Tumor Targeting Therapy. Int J Mol Sci. 2019;20:755. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 519] [Cited by in RCA: 463] [Article Influence: 66.1] [Reference Citation Analysis (2)] |
| 147. | Liu XH, Zhai XY. Role of tryptophan metabolism in cancers and therapeutic implications. Biochimie. 2021;182:131-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 50] [Article Influence: 10.0] [Reference Citation Analysis (1)] |
| 148. | Xue C, Li G, Zheng Q, Gu X, Shi Q, Su Y, Chu Q, Yuan X, Bao Z, Lu J, Li L. Tryptophan metabolism in health and disease. Cell Metab. 2023;35:1304-1326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 789] [Cited by in RCA: 634] [Article Influence: 211.3] [Reference Citation Analysis (1)] |
| 149. | Sullivan LB, Luengo A, Danai LV, Bush LN, Diehl FF, Hosios AM, Lau AN, Elmiligy S, Malstrom S, Lewis CA, Vander Heiden MG. Aspartate is an endogenous metabolic limitation for tumor growth. Nat Cell Biol. 2018;20:782-788. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 325] [Cited by in RCA: 286] [Article Influence: 35.8] [Reference Citation Analysis (1)] |
| 150. | Taylor SR, Falcone JN, Cantley LC, Goncalves MD. Developing dietary interventions as therapy for cancer. Nat Rev Cancer. 2022;22:452-466. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 148] [Cited by in RCA: 129] [Article Influence: 32.3] [Reference Citation Analysis (0)] |
| 151. | Klement RJ, Meyer D, Kanzler S, Sweeney RA. Ketogenic diets consumed during radio-chemotherapy have beneficial effects on quality of life and metabolic health in patients with rectal cancer. Eur J Nutr. 2022;61:69-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 152. | Xiang Y, Wang M, Miao H. Ketogenic diet: new avenues to overcome colorectal cancer. Signal Transduct Target Ther. 2022;7:262. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (1)] |