Published online Mar 15, 2026. doi: 10.4251/wjgo.v18.i3.115737
Revised: December 17, 2025
Accepted: January 19, 2026
Published online: March 15, 2026
Processing time: 133 Days and 10.8 Hours
Sporadic nonampullary duodenal tumors (SNADTs) are rare in clinical practice. Endoscopic resection (ER) is widely used in the treatment of SNADTs. However, the clinical outcome following ER for SNADTs remains unclear.
To evaluate the clinical efficacy of ER for SNADTs.
We retrospectively analyzed clinical data and follow-up information for SNADT patients who underwent ER at Peking University First Hospital between January 2013 and May 2024. Patients were grouped by technique: Endoscopic mucosal resection (EMR), endoscopic submucosal dissection (ESD), or ESD with snare assistance (ESD-S). The outcomes included procedure time, complications, en bloc resection, complete resection, and recurrence during short- and long-term follow-up.
A total of 124 consecutive patients were included (EMR 66; ESD 26; ESD-S 32). Procedure time differed significantly among the three groups. The en bloc re
ER for SNADTs is safe and effective. In addition to EMR and ESD, a modified ESD technique (ESD-S) is effective. Endoscopic therapy is a viable and effective option for recurrence.
Core Tip: Sporadic nonampullary duodenal tumors (SNADTs) are uncommon neoplasms in clinical practice. Endoscopic resection (ER) has become a widely adopted therapeutic modality for these lesions. Our study evaluates the outcomes of ER for nonampullay duodenal tumors to assess its efficacy and safety. ER for SNADTs is a safe and effective treatment strategy. Endoscopic therapy represents a viable first-line option for appropriately selected patients, offering favorable oncologic outcomes. Long-term follow-up and careful endoscopic surveillance are recommended to ensure early detection and management of potential recurrences.
- Citation: Yu H, Rong L, Cai YL, Liu GY, Guo XY. Short- and long-term outcomes after endoscopic resection of sporadic nonampullary duodenal tumors: An 11-year single-center retrospective study. World J Gastrointest Oncol 2026; 18(3): 115737
- URL: https://www.wjgnet.com/1948-5204/full/v18/i3/115737.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i3.115737
Sporadic nonampullary duodenal tumors (SNADTs)-including nonampullary duodenal adenomas, duodenal neuroendocrine tumors, Brunner’s gland adenoma, and follicular lymphoma-are relatively rare, with an overall prevalence of 1.0%-4.6%[1,2]. As endoscopic imaging and diagnostic capabilities improve, the detection of SNADTs has increased. Duodenal adenomas typically present as flat or slightly elevated lesions with irregular whitish opaque contents, a milky-white appearance, and abnormal microvascular patterns[3,4]. Duodenal neuroendocrine tumors are commonly found in the bulb and often present a depressed surface and vascular dilation.
Endoscopic resection (ER) is widely used for SNADTs, yet duodenal ER remains technically demanding and carries greater risk than procedures at other gastrointestinal sites. A thin and pliable muscle layer, narrow lumen, sharp angulation, the presence of Brunner’s glands, and rich submucosal vascularity complicate treatment[5,6]. Current ER techniques include endoscopic mucosal resection (EMR), endoscopic submucosal dissection (ESD), and ESD with snare assistance (ESD-S). In this study, the outcomes following ER for SNADTs at our center are retrospectively examined to assess its efficacy and safety.
This retrospective included consecutive patients with endoscopically diagnosed primary SNADTs who underwent ER at the endoscopic center of Peking University First Hospital between January 2013 and May 2024. The exclusion criteria were gastrointestinal polyposis syndromes (familial adenomatous polyposis or Peutz-Jeghers syndrome) and advanced malignant tumors. All patients underwent comprehensive preoperative evaluation, and informed consent (or an approved alternatives) was obtained. The study complied with the Declaration of Helsinki and was approved by the Institutional Review Board of Peking University First Hospital (No. 2025110).
All procedures were performed by experienced endoscopists with at least 10000 prior gastroscopies. Sedation was administered with continuous monitoring of vital signs. High-definition white-light endoscopy and narrow-band imaging were used to identify the lesion and determine its relationship to the ampulla of Vater. The resection strategy was individualized after the lesion size, location, and procedural risk were assessed. For small lesions and those exhibiting a protruding gross morphology, EMR is generally considered the treatment of choice. However, in the case of larger submucosal tumors (SMT) or flat lesions, EMR is associated with a reduced probability of achieving complete en bloc resection and carries an increased risk of residual neoplastic tissue. Consequently, ESD or ESD-S is typically preferred in these scenarios to enhance resection completeness and curative potential. When technical challenges-such as difficult anatomical access, poor lesion visibility, or anticipated fibrosis-are anticipated during endoscopic intervention, ESD-S may be specifically selected to balance the need for adequate tumor resection with the minimization of procedural complications, including perforation and intraoperative bleeding.
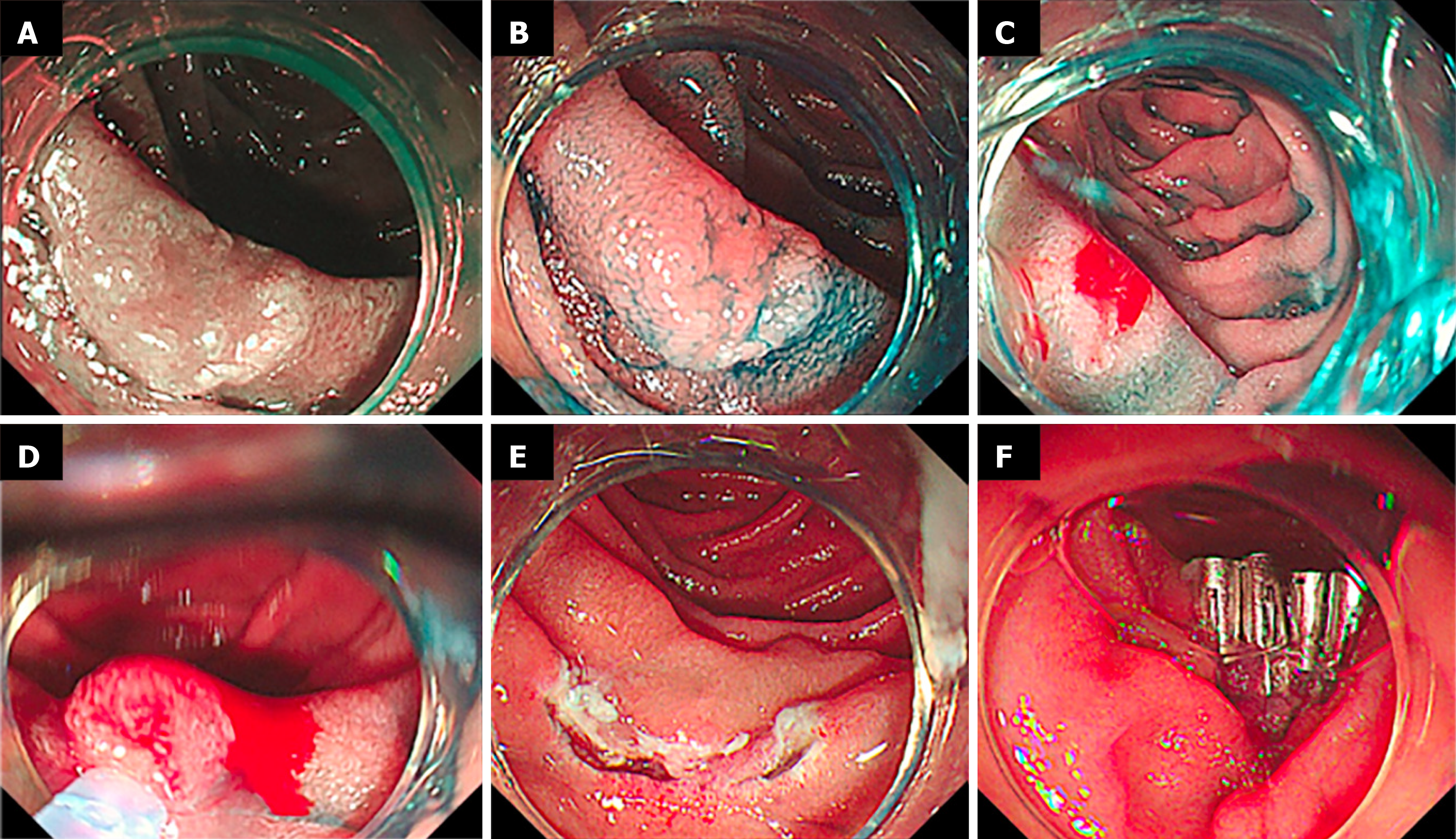
EMR: Submucosal lifting with saline containing methylene blue was followed by snare resection (Figure 1).
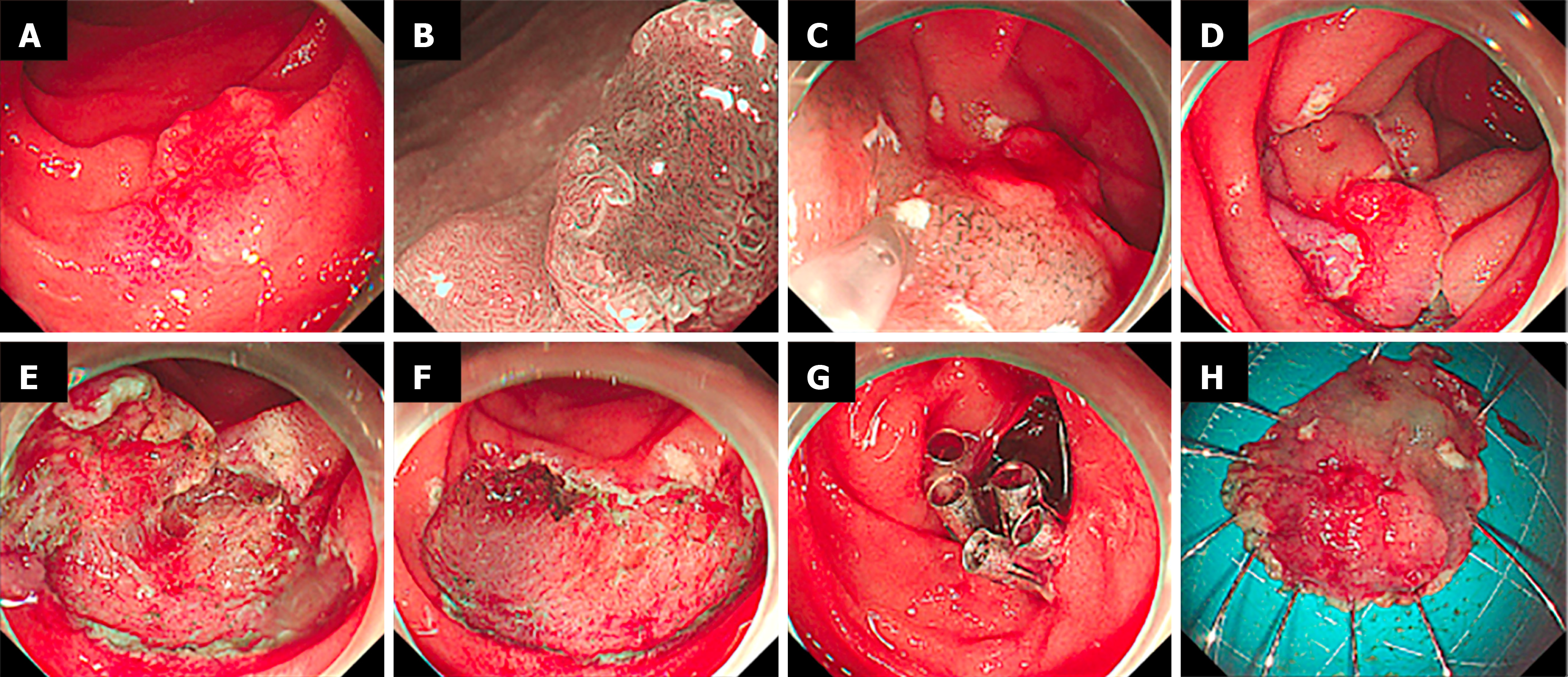
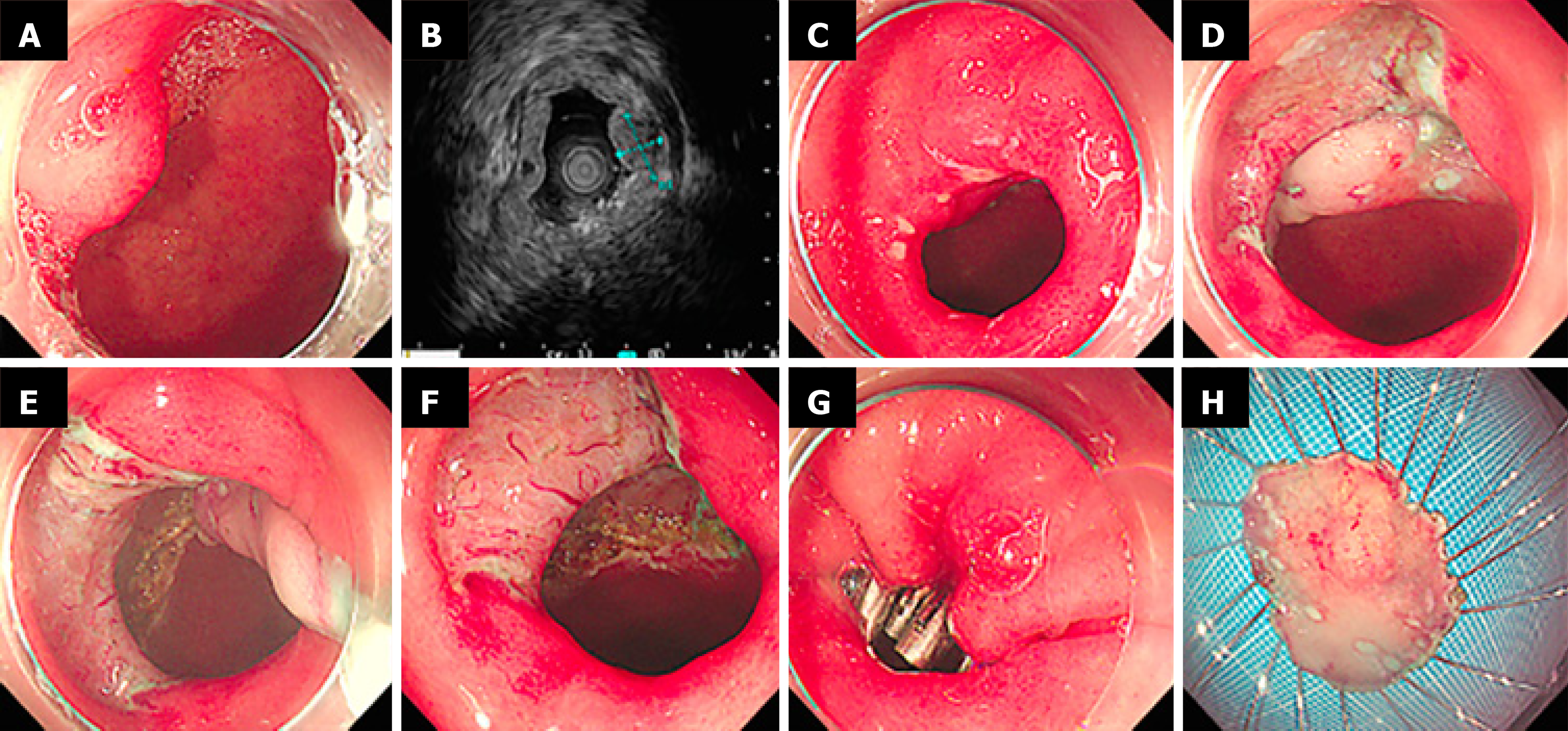
ESD: Marking dots were placed outside the lesion, and methylene blue saline was then injected into the submucosa; sodium hyaluronate was added to maintain a durable cushion. Circumferential incision and submucosal dissection were performed using a dual knife (Olympus, Tokyo, Japan) or a gold knife (Micro-Tech, Nanjing, China) (Figures 2 and 3).
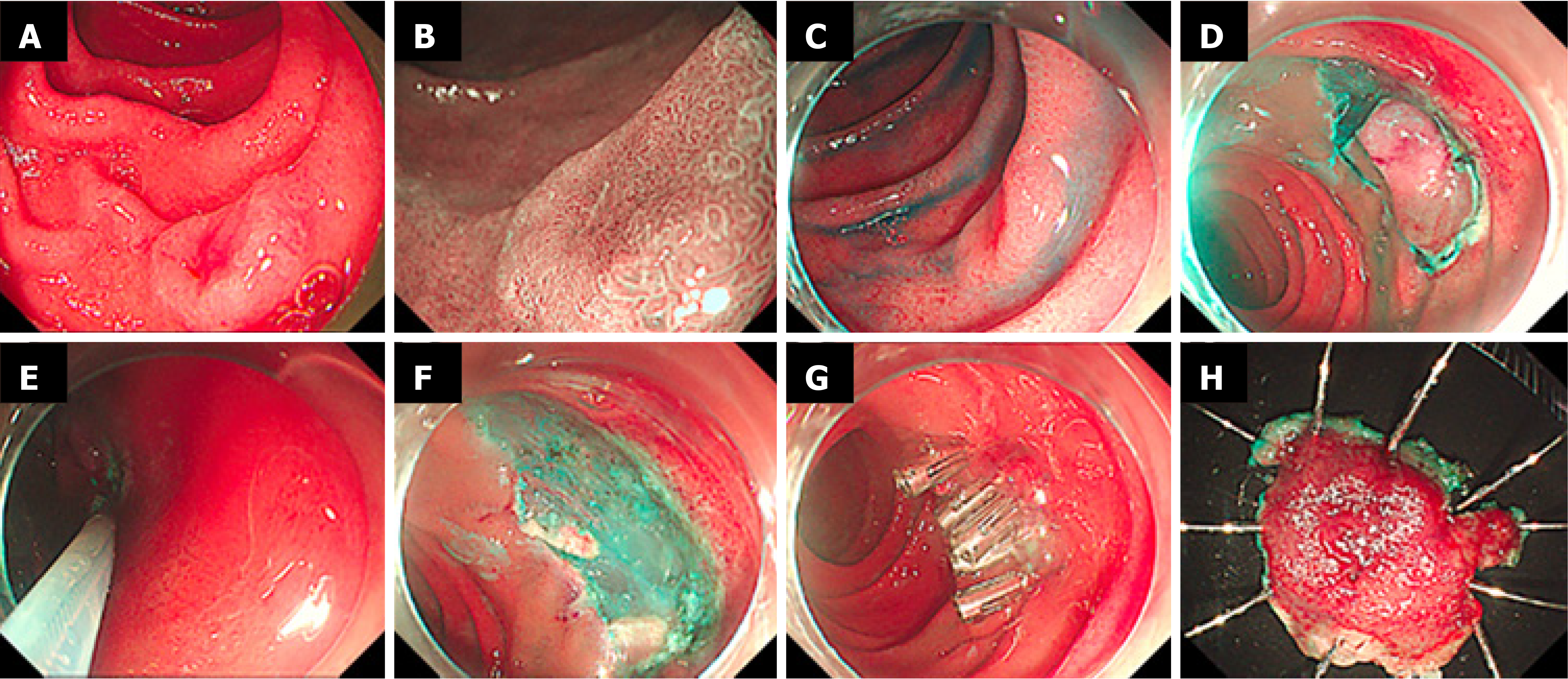
ESD-S: After marking and submucosal elevation with saline containing methylene blue and sodium hyaluronate, a circumferential incision was made with the above-mentioned knives. Most of the submucosal dissection was performed using high-frequency knives, and the lesion was ultimately removed with a snare (Figure 4).
After resection, hemostasis was achieved with electrocoagulation using hot-biopsy forceps or the tip of the snare to reduce the risk of delayed bleeding. Prophylactic clip closure or purse-string suturing was applied to minimize delayed perforation. In high-risk patients, a nasogastric tube was used to drain gastric and pancreaticobiliary secretions to mitigate wound erosion. Perioperative adverse events were carefully assessed.
After discharge, outpatient and endoscopic follow-up were scheduled at 6 and 12 months during the first year and annually thereafter to assess local recurrence. If recurrence was suspected and confirmed by tissue sampling, additional endoscopic treatment was recommended.
Lesions were classified by macroscopic morphology as the protruding, flat, or SMT type. Procedure time was measured from the initial submucosal injection to the completion of resection. Intraprocedural perforation was defined as a visible muscularis propria defect during the procedure. Delayed perforation was diagnosed by postprocedural CT evidence of free air accompanied by severe abdominal pain. Delayed bleeding was defined as postoperative hemorrhage requiring medical intervention. En bloc resection denoted removal in a single piece without fragmentation. Complete resection was defined as tumor-free vertical and horizontal margins, irrespective of en bloc status.
Outcomes were divided into short-term and long-term categories. Short-term outcomes included procedural success (en bloc and complete resection) and procedure-related complications (bleeding and perforation). The long-term outcome of interest was local recurrence, defined as tumor regrowth at the resection scar, among patients with at least 12 months of surveillance. For patients who were followed up at outside hospitals, information was collected via telephone and e-mail from patients or referring physicians.
Analyses were conducted using IBM SPSS Statistics for Mac, version 25.0 (IBM Corp., Armonk, NY, United States). Normally distributed continuous variables (e.g., age and lesion size) are expressed as the mean ± SD and were compared using one-way ANOVA. Non-normally distributed variables (e.g., operative time) are presented as median (range) and were compared using the Mann-Whitney U test. Categorical variables are summarized as n (%) and were compared using the χ2 test or Fisher’s exact test, as appropriate. A two-sided P value < 0.05 was considered statistically significant to indicate statistical significance.
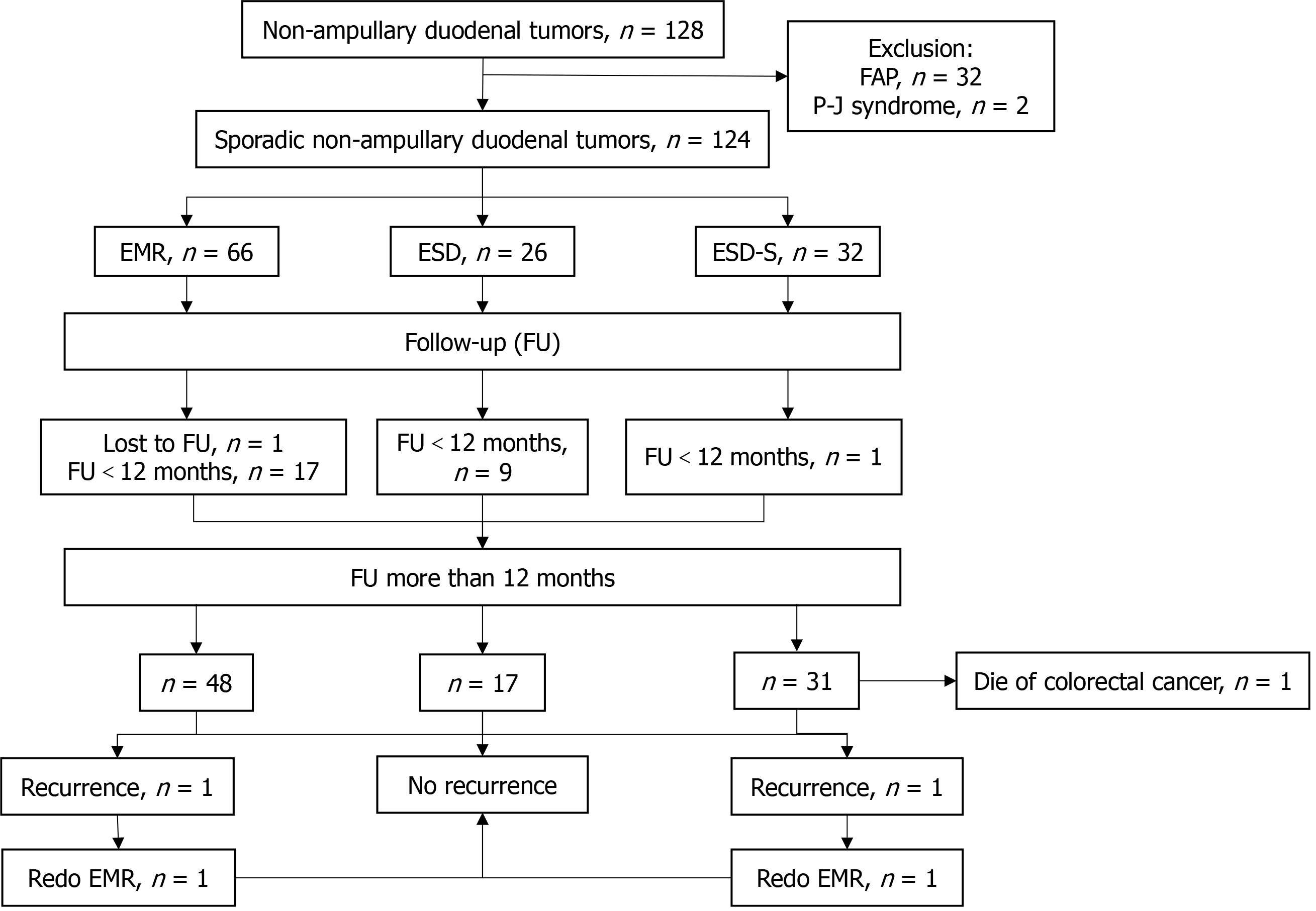
During the study period, 128 patients underwent ER for SNADTs. Four patients were excluded (two with familial adenomatous polyposis and two with Peutz-Jeghers syndrome), leaving 124 consecutive patients for analysis. A flowchart of the study is shown in Figure 5. Men constituted 57.3% of the cohort, and the median age was 59.9 years (range, 29-79). The lesion locations were the duodenal bulb (29.8%), descending segment (69.4%), and horizontal segment (0.8%). The mean lesion size was 1.2 ± 0.7 cm (range, 0.5-3.5 cm). Morphologically, 32.3% were protruding, 58.9% flat, and 8.9% SMT type. Pathology revealed 96 (77.4%) adenomas/adenocarcinomas, 15 (12.1%) Brunner’s gland adenomas, 12 (9.7%) neuroendocrine tumors, and one (0.8%) follicular lymphoma. The treatments included EMR in 66 cases (53.3%), ESD in 26 (21.0%), and ESD-S in 32 (25.8%). The baseline characteristics are summarized in Table 1.
| Patients/Lesions | 124/124 |
| Age, years | 59.9 ± 10.6 |
| Gender | |
| Male | 71 (57.3) |
| Female | 53 (42.7) |
| Tumor location | |
| Bulb | 37 (29.8) |
| Descending segment | 86 (69.4) |
| Horizontal segment | 1 (0.8) |
| Macroscopic morphology type | |
| Protrude | 40 (32.3) |
| Flat | 73 (58.9) |
| SMT | 11 (8.9) |
| Tumor size, cm | 1.2 ± 0.7 |
| Pathology | |
| Adenoma/adenocarcinoma | 96 (77.4) |
| Brunner’s gland adenoma | 15 (12.1) |
| Neuroendocrine tumor | 12 (9.7) |
| Follicular lymphoma | 1 (0.8) |
| Endoscopic treatment | |
| EMR | 66 (53.2) |
| ESD | 26 (21.0) |
| ESD-S | 32 (25.8) |
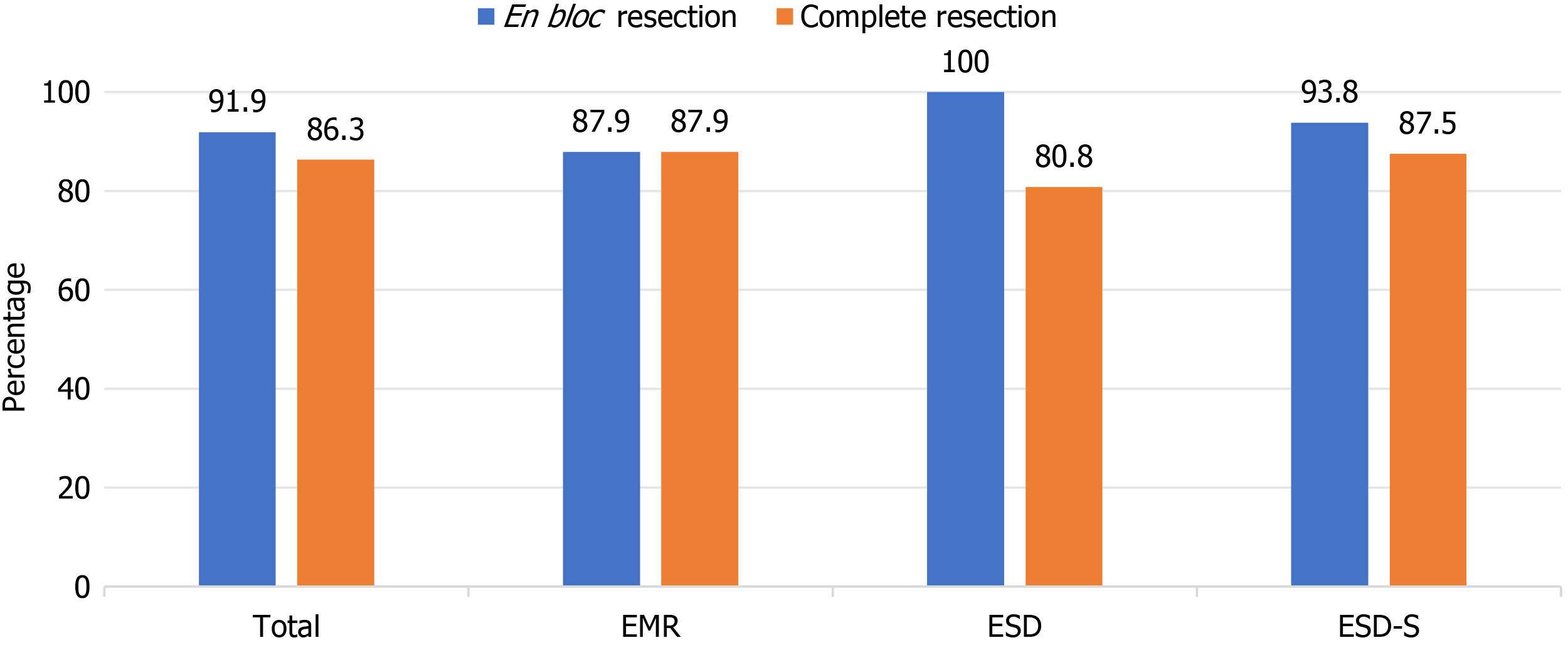
Significant differences in procedure time were observed among the three groups. En bloc resection was achieved in 114/124 patients (91.9%), specifically 87.9% with EMR, 100% with ESD, and 93.8% with ESD-S (P = 0.14). Pathology revealed four lesions with positive horizontal margins and seven with indeterminate horizontal margins; for vertical margins, two were positive, and six were indeterminate. Margin assessment was not applicable in one follicular lymphoma case. The complete resection rates were 87.9% (EMR), 80.8% (ESD), and 87.5% (ESD-S) (P = 0.73; Figure 6).
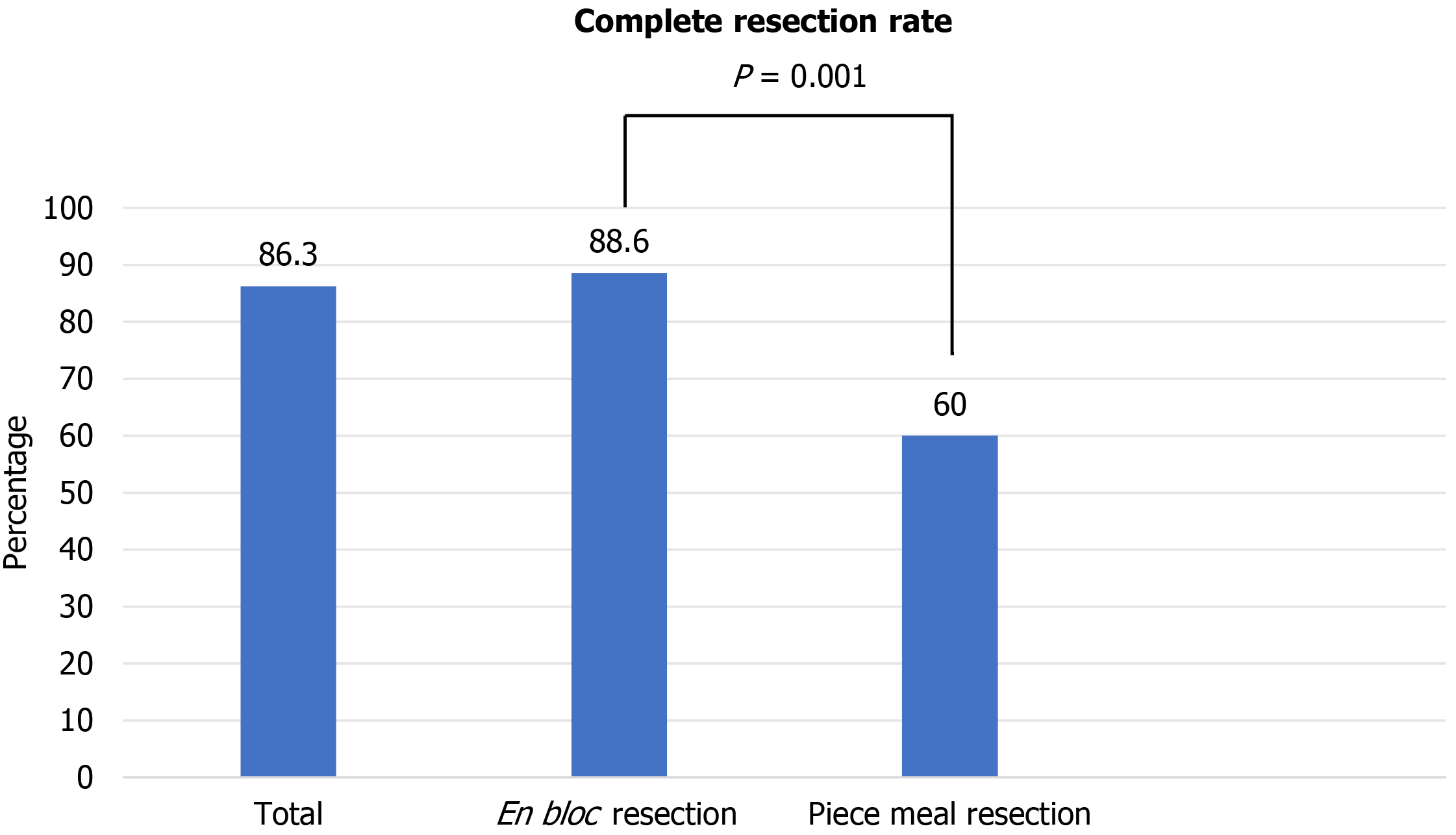
Patients were further stratified by resection method. Among the 114 patients who underwent en bloc resection, 101 (88.6%) achieved complete resection; among the 10 who underwent piecemeal resection, 6 (60%) achieved complete resection (P = 0.001; Figure 7).
No delayed bleeding occurred. Perforation was observed in five patients (4%): Four in the ESD-S group and one in the EMR group. The single intraprocedural perforation was managed conservatively with endoscopic clip closure, gas
| EMR (n = 66) | ESD (n = 26) | ESD-S (n = 32) | Statistic value | P value | |
| Age, years | 63.0 ± 9.4 | 55.7 ± 9.4 | 57.2 ± 12.3 | F = 6.314 | 0.002 |
| Gender, male/female | 39/27 | 15/11 | 17/15 | χ2 = 0.316 | 0.859 |
| Tumor location | χ2 = 1.221 | 0.946 | |||
| Bulb | 21 | 7 | 9 | ||
| Descending segment | 44 | 19 | 23 | ||
| Horizontal segment | 1 | 0 | 0 | ||
| Tumor size, cm | 1.16 (0.99-1.32) | 1.50 (1.16-1.83) | 1.13 (0.87-1.38) | F = 2.498 | 0.087 |
| Procedure time, minute | 5.8 (4.4-7.2) | 50.7 (35.3-66.2) | 32.6 (20.0-45.1) | Hc = 78.517 | < 0.001 |
| Pathology | |||||
| Adenoma/adenocarcinoma | 53 (80.3) | 19 (73.1) | 24 (75.0) | χ2 = 0.702 | 0.743 |
| Brunner’s gland adenoma | 12 (18.2) | 1 (3.8) | 2 (6.3) | χ2 = 4.991 | 0.083 |
| Neuroendocrine tumor | 1 (1.5) | 5 (19.2) | 6 (18.6) | χ2 = 10.759 | 0.006 |
| Follicular lymphoma | 0 (0) | 1 (3.8) | 0 (0) | χ2 = 3.800 | 0.210 |
| En bloc resection | 58 (87.9) | 26 (100) | 30 (93.8) | χ2 = 3.888 | 0.129 |
| Complete resection | 58 (87.9) | 21 (80.8) | 28 (87.5) | χ2 = 0.850 | 0.734 |
| Adverse events | |||||
| Intraprocedural perforation | 0 (0) | 0 (0) | 1 (3.1) | χ2 = 2.898 | 0.468 |
| Delayed perforation | 1 (1.5) | 0 (0) | 3 (9.4) | χ2 = 5.361 | 0.081 |
Twenty-seven of the 124 patients had < 12 months of follow-up and were excluded from long-term analysis. One patient (EMR for a 2.0 cm adenoma) was lost to follow-up. Ninety-six patients were followed up for ≥ 12 months (median 34.5 months; range, 12-134). The overall survival was 99.0%, and the disease-specific survival was 100%. One patient who underwent ESD-S for a 1.0 cm duodenal adenoma died of advanced colorectal cancer 40 months after the procedure.
Two patients experienced local recurrence. One patient who underwent ESD-S for a 3.5 cm adenoma in the descending duodenum with an indeterminate vertical margin developed recurrence at 4 months; repeat piecemeal EMR was performed, and there was no further recurrence over an additional 34-month follow-up. Another patient who underwent EMR for a 2.0 cm adenoma in the descending segment with indeterminate vertical and horizontal margins developed recurrence at 16 months; repeat piecemeal EMR was performed, and no recurrence was observed over the next 12 months. The clinical course is shown in Figure 5. Table 3 presents data for the 14 patients who did not achieve complete resection.
| No. | Gender/age | Pathology | Location | Tumor size, cm | Resection method | En bloc resection | Reason for non-complete resection | Recurrence | Follow-up period, months |
| 1 | M/76 | Net | Bulb | 0.8 | ESD-S | Yes | HM (+) | No | 59 |
| 2 | M/40 | Adenoma | Descending segment | 3.0 | EMR | No | HM (+) | No | 19 |
| 3 | M/71 | Net | Bulb | 1.0 | ESD | Yes | VM (+) | No | 12 |
| 4 | M/55 | Adenoma | Descending segment | 1.5 | EMR | No | Indeterminate HM and VM | No | 48 |
| 5 | M/71 | Adenoma | Bulb | 3.0 | EMR | No | Indeterminate HM and VM | No | 14 |
| 6 | F/54 | Adenoma | Bulb | 3.5 | ESD-S | Yes | Indeterminate VM | Yes | 44 |
| 7 | M/54 | Adenoma | Descending segment | 1.5 | EMR | Yes | HM (+) | No | 32 |
| 8 | F/73 | Net | Bulb | 0.6 | ESD-S | Yes | VM (+) | No | 39 |
| 9 | F/57 | Adenoma | Descending segment | 2.0 | EMR | No | Indeterminate HM and VM | Yes | 19 |
| 10 | M/52 | Adenoma | Descending segment | 1.5 | ESD | Yes | Indeterminate HM | No | 21 |
| 11 | F/42 | Follicular lymphoma | Descending segment | 3.5 | ESD | Yes | Not evaluated | No | 22 |
| 12 | M/57 | Adenoma | Descending segment | 1.0 | ESD-S | Yes | Indeterminate HM | No | 19 |
| 13 | F/65 | Adenoma | Bulb | 0.8 | EMR | Yes | Indeterminate HM | No | 21 |
| 14 | M/71 | Adenoma | Descending segment | 1.0 | EMR | Yes | HM (+) | No | 12 |
This 11-year single-center retrospective study supports the feasibility and effectiveness of ER for SNADTs, with favorable outcomes and no deaths attributable to primary SNADTs. The recurrence rate was 1.6%, and all recurrent cases were successfully retreated endoscopically without subsequent recurrence.
Although uncommon, SNADTs are detected with increasing frequency in contemporary practice. European Society of Gastrointestinal Endoscopy guidelines recommend resection of all duodenal adenomas because of malignant potential, analogous to the adenoma-carcinoma sequence in the colon, with malignant transformation rates reported at 30%-85%[2,7]. Most duodenal neuroendocrine tumors are indolent, yet when the diameter exceeds 10 mm or invasion reaches the submucosa, the risk of distant metastasis increases, warranting treatment. Brunner’s gland adenomas may enlarge and rarely undergo malignant transformation; therefore, removal is recommended upon diagnosis.
Endoscopic therapy results in less trauma, faster recovery, and a better prognosis than open surgery does. However, several anatomical and physiological features of the duodenum complicate ER: A thin, vascular wall favoring bleeding and perforation; Brunner’s gland–related submucosal fibrosis complicating dissection; proximity to critical structures such as the common bile duct, pancreas, and major vessels; and exposure of resection ulcers to bile and pancreatic secretions, which heightens delayed perforation risk[2,8-11]. Although advances in endoscopic techniques have reduced perioperative complications and expanded the use of endoscopic procedures for SNADTs, large long-term datasets remain limited.
In addition to standard EMR and ESD, we adopted a modified approach, ESD-S, in which a snare is used for final removal after most of the lesion has been dissected with an electrosurgical knife. This hybrid technique retains the key advantages of ESD, including en bloc resection and high curative resection rates, while reducing procedural complexity and duration. In the present study, more than half of the lesions in the EMR group were of the protruding morphological type, whereas the majority of lesions treated with the ESD or ESD-S approach were flat lesions or SMTs. This distribution largely reflects the limitations of EMR in managing non-protruding lesions, which are associated with lower rates of complete resection and a higher likelihood of residual or recurrent disease due to piecemeal resection. Consequently, to maximize the probability of curative resection, ESD or ESD-S is preferentially employed for flat lesions and SMTs. Notably, for lesions that present substantial technical challenges-such as those involving fibrosis, poor endoscopic visibility, or difficult anatomical locations-the ESD-S technique may be favored over conventional ESD. This approach helps mitigate the risk of adverse events, such as perforation or intraoperative bleeding, while preserving the high efficacy and completeness of resection that characterize ESD. In our cohort, the en bloc resection rates were 87.9% for EMR, 100% for ESD, and 93.8% for ESD-S, with complete resection rates of 87.9%, 80.8%, and 87.5%, respectively. These figures slightly exceed those in earlier reports[1,5,12-16]. The comparatively low complete resection rate in the ESD group is likely attributable to the case mix: ESD was preferentially used for SMTs with deeper growth, increasing the likelihood of positive or indeterminate vertical margins on histology. Although no statistically significant difference was found, ESD-S showed numerically higher en bloc and complete resection rates than EMR, suggesting a potential advantage for larger or higher-risk lesions. Moreover, subgroup analysis confirmed that en bloc resection markedly improves the probability of complete resection compared with piecemeal removal, underscoring the importance of single-piece excision to minimize residual disease and recurrence.
Perforation remains a major concern during duodenal ER, with reported rates for duodenal ESD ranging from 9.4%-28.6%[2,10,17]. In our series, one intraprocedural perforation occurred in the ESD-S group and was successfully managed with endoscopic clips, decompression, acid suppression, and antibiotics, requiring no surgery. Prompt recognition and definitive closure of defects during the procedure are critical for favorable outcomes. Delayed perforation, a serious adverse event with an incidence of approximately 1.0%-2.0%[18,19], occurred in four patients; three underwent surgical repair with primary closure and enteral tube placement, and one improved with conservative treatment. The pathogenesis of delayed perforation is likely multifactorial, including a thin duodenal wall, thermal injury from cautery, and exposure of the ulcer bed to bile and pancreatic secretions. Prophylactic closure of the defect and diversion of di
During follow-up, two patients (2.1%) developed local recurrence; both had incomplete initial resection. Each was successfully managed with repeat endoscopic therapy, and no further recurrence was observed. These findings emphasize the importance of achieving complete resection initially and support endoscopic retreatment as an effective strategy to address recurrence.
This study has several limitations inherent to its retrospective single-center design. First, the choice of ER technique was determined by the endoscopist without a standardized protocol, introducing potential selection bias. Second, generalizability may be limited by the single-institution setting despite the relatively large sample size. Third, the imbalance in group sizes-particularly the smaller ESD cohort-may have affected the comparative analyses. Even so, our data provide a comprehensive appraisal of both short- and long-term outcomes for ER in SNADTs. Multicenter prospective studies are warranted to validate these results and facilitate standardization of SNADT management.
ER of SNADTs is safe and effective. In addition to EMR and ESD, the modified ESD-S technique offers a novel therapeutic strategy that may reduce the technical difficulty of ER for challenging duodenal lesions to a certain extent. Tailoring technique selection to lesion size, location, and risk profile can maximize en bloc and complete resection rates while minimizing residual disease and recurrence. Prompt detection and proper closure of perforations, combined with effective diversion of digestive fluids, can reduce the need for conversion to surgery. In cases of recurrence, repeat ER is feasible and effective.
We thank all authors listed in this manuscript for their contributions.
| 1. | Jung SH, Chung WC, Kim EJ, Kim SH, Paik CN, Lee BI, Cho YS, Lee KM. Evaluation of non-ampullary duodenal polyps: comparison of non-neoplastic and neoplastic lesions. World J Gastroenterol. 2010;16:5474-5480. [PubMed] [DOI] [Full Text] |
| 2. | Vanbiervliet G, Moss A, Arvanitakis M, Arnelo U, Beyna T, Busch O, Deprez PH, Kunovsky L, Larghi A, Manes G, Napoleon B, Nalankilli K, Nayar M, Pérez-Cuadrado-Robles E, Seewald S, Strijker M, Barthet M, van Hooft JE. Endoscopic management of superficial nonampullary duodenal tumors: European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy. 2021;53:522-534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 82] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 3. | Kikuchi D, Hoteya S, Iizuka T, Kimura R, Kaise M. Diagnostic algorithm of magnifying endoscopy with narrow band imaging for superficial non-ampullary duodenal epithelial tumors. Dig Endosc. 2014;26 Suppl 2:16-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 62] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 4. | Toya Y, Endo M, Matsumoto T. Revised diagnostic algorithm of magnifying endoscopy with crystal violet staining for non-ampullary duodenal epithelial tumors. Dig Endosc. 2022;34:245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 5. | Nonaka S, Oda I, Tada K, Mori G, Sato Y, Abe S, Suzuki H, Yoshinaga S, Nakajima T, Matsuda T, Taniguchi H, Saito Y, Maetani I. Clinical outcome of endoscopic resection for nonampullary duodenal tumors. Endoscopy. 2015;47:129-135. [PubMed] [DOI] [Full Text] |
| 6. | Yamasaki Y, Uedo N, Takeuchi Y, Ishihara R, Okada H, Iishi H. Current Status of Endoscopic Resection for Superficial Nonampullary Duodenal Epithelial Tumors. Digestion. 2018;97:45-51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 61] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 7. | Seifert E, Schulte F, Stolte M. Adenoma and carcinoma of the duodenum and papilla of Vater: a clinicopathologic study. Am J Gastroenterol. 1992;87:37-42. [PubMed] |
| 8. | Shibagaki K, Ishimura N, Kinoshita Y. Endoscopic submucosal dissection for duodenal tumors. Ann Transl Med. 2017;5:188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 39] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 9. | Tsutsumi K, Kato M, Kakushima N, Iguchi M, Yamamoto Y, Kanetaka K, Uraoka T, Fujishiro M, Sho M; Japan Duodenal Cancer Guideline Committee. Efficacy of endoscopic preventive procedures to reduce delayed adverse events after endoscopic resection of superficial nonampullary duodenal epithelial tumors: a meta-analysis of observational comparative trials. Gastrointest Endosc. 2021;93:367-374.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 10. | Valerii G, Tringali A, Landi R, Boškoski I, Familiari P, Bizzotto A, Perri V, Petruzziello L, Costamagna G. Endoscopic mucosal resection of non-ampullary sporadic duodenal adenomas: a retrospective analysis with long-term follow-up. Scand J Gastroenterol. 2018;53:490-494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (1)] |
| 11. | Cho JH, Lim KY, Lee EJ, Lee SH. Clinical outcomes of endoscopic resection of superficial nonampullary duodenal epithelial tumors: A 10-year retrospective, single-center study. World J Gastrointest Surg. 2022;14:329-340. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (5)] |
| 12. | Hara Y, Goda K, Dobashi A, Ohya TR, Kato M, Sumiyama K, Mitsuishi T, Hirooka S, Ikegami M, Tajiri H. Short- and long-term outcomes of endoscopically treated superficial non-ampullary duodenal epithelial tumors. World J Gastroenterol. 2019;25:707-718. [PubMed] [DOI] [Full Text] |
| 13. | Na HK, Kim DH, Ahn JY, Lee JH, Jung KW, Choi KD, Song HJ, Lee GH, Jung HY. Clinical Outcomes following Endoscopic Treatment for Sporadic Nonampullary Duodenal Adenoma. Dig Dis. 2020;38:364-372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 14. | Santos-Antunes J, Morais R, Marques M, Macedo G. Underwater Duodenal ESD of a Large Adenoma Using the Pocket-Creation Method. GE Port J Gastroenterol. 2021;28:367-369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 15. | Tomizawa Y, Ginsberg GG. Clinical outcome of EMR of sporadic, nonampullary, duodenal adenomas: a 10-year retrospective. Gastrointest Endosc. 2018;87:1270-1278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 63] [Article Influence: 7.9] [Reference Citation Analysis (2)] |
| 16. | Udd M, Lindström O, Tenca A, Rainio M, Kylänpää L. Endoscopic therapy of sporadic non-ampullary duodenal adenomas, single centre retrospective analysis. Scand J Gastroenterol. 2023;58:208-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 17. | Mukherjee S, Kocher HM, Hutchins RR, Bhattacharya S, Abraham AT. Impact of hospital volume on outcomes for pancreaticoduodenectomy: a single UK HPB centre experience. Eur J Surg Oncol. 2009;35:734-738. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 18. | Hoteya S, Furuhata T, Takahito T, Fukuma Y, Suzuki Y, Kikuchi D, Mitani T, Matsui A, Yamashita S, Nomura K, Kuribayashi Y, Iizuka T, Kaise M. Endoscopic Submucosal Dissection and Endoscopic Mucosal Resection for Non-Ampullary Superficial Duodenal Tumor. Digestion. 2017;95:36-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 102] [Article Influence: 11.3] [Reference Citation Analysis (1)] |
| 19. | Pérez-Cuadrado-Robles E, Quénéhervé L, Margos W, Shaza L, Ivekovic H, Moreels TG, Yeung R, Piessevaux H, Coron E, Jouret-Mourin A, Deprez PH. Comparative analysis of ESD versus EMR in a large European series of non-ampullary superficial duodenal tumors. Endosc Int Open. 2018;6:E1008-E1014. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 49] [Article Influence: 6.1] [Reference Citation Analysis (0)] |