Published online Mar 15, 2026. doi: 10.4251/wjgo.v18.i3.113781
Revised: November 1, 2025
Accepted: December 23, 2025
Published online: March 15, 2026
Processing time: 190 Days and 21.6 Hours
Gastric cancer (GC) remains one of the most prevalent and deadly cancers glo
To explore the effect and potential mechanism of miR-27b-5p on the occurrence and development of GC by targeting CREBZF to regulate the signal transducer and activator of transcription signal transducer and activator of transcription 3 (STAT3) pathway.
The regulatory roles of miR-27b-5p on CREBZF were validated through dual-luciferase reporter assays, real-time quantitative polymerase chain reaction, and western blotting to examine changes in the signaling axis. Cell models were created by overexpressing or knocking down miR-27b-5p and its target gene in lentivirus-transfected AGS and NCI-N87 cells. The changes in cellular proliferation were detected using cell counting kit-8 assays and flow cytometry, whereas cell migration was evaluated utilizing Transwell and wound healing experiments. The function of miR-27b-5p in the proliferation of GC cells was investigated in vivo employing a subcutaneous xenograft model in nude mice.
MiR-27b-5p showed higher expression levels in GC tissue compared to nearby non-cancerous tissue, while CREBZF displayed significant downregulation. Elevated miR-27b-5p levels inhibited CREBZF expression, as revealed by analyses of mRNA and protein, thereby promoting the proliferation and migratory capabilities of GC cell lines. Excess CREBZF counteracted this effect. Inhibition of miR-27b-5p diminished the proliferation and migration of GC cell lines, whereas CREBZF knockdown counteracted this outcome. Excessive miR-27b-5p ex
MiR-27b-5p expression is elevated in GC and operates as an oncogene by targeting CREBZF to regulate the STAT3 pathway, promoting malignancy in GC. MiR-27b-5p is a potential therapeutic target.
Core Tip: Our findings provide preliminary evidence that reduction of miR-27b-5p restrains proliferation and migration of gastric cancer cells. Our investigation also indicates that elevated expression level of miR-27b-5p suppresses gene exp
- Citation: Ma FC, Zhang GL, Chen GQ, Ling JW, Li YF, Chi BT, Tang YL, Peng W, Chen G, Wei DM, Rong MH, Ge LY. MiR-27b-5p promotes gastric cancer progression by targeting CREBZF to regulate the STAT3 pathway. World J Gastrointest Oncol 2026; 18(3): 113781
- URL: https://www.wjgnet.com/1948-5204/full/v18/i3/113781.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i3.113781
Gastric cancer (GC) remains a major global health burden due to its high incidence and mortality. In accordance with the recent data published from the International Agency for Research on Cancer in 2020[1], GC ranks third both in incidence and mortality in China. There are approximately 480000 new cases (10.5% of all newly diagnosed cancer cases) and approximately 370000 fatalities (12.4% of all deaths caused by cancer). Recent advances in molecular biology highlight the critical role of microRNAs (miRNAs) in regulating gene expression and tumorigenesis. MiR-27b-5p is overexpressed in low-grade and high-grade gastric dysplasia, suggesting it acts as a possible biomarker for GC progression alongside cAMP response element-binding protein zinc finger (CREBZF) mRNA[2]. Although miR-27b-5p has been implicated in various tumor types, its exact role and mechanism in GC remain unclear. A better comprehension of the function of miR-27b-5p in GC could offer new perspectives on the molecular foundations of the disease and possible treatment targets.
Earlier research has shown that CREBZF affects circular RNA biogenesis in breast cancer via regulating the signal transducer and activator of transcription 3 (STAT3) pathway[3]. Our preliminary research indicated a notable rise in miR-27b-5p levels in GC tissue when compared to nearby non-cancerous tissue, along with a marked decrease in CREBZF mRNA expression in GC cells. Dual-luciferase reporter assays validated that miR-27b-5p reduces CREBZF transcription, confirming that CREBZF is directly targeted by miR-27b-5p. CREBZF has been shown to inhibit tumor progression, while the STAT3 signaling pathway enhances the growth and invasion of malignant cells. CREBZF suppresses STAT3 pathway activation; therefore, it is likely that miR-27b-5p promotes malignant behavior by influencing CREBZF and modulating the STAT3 pathway in GC cells. However, the precise mechanism by which miR-27b-5p targets CREBZF to regulate the STAT3 pathway and influence GC progression remains unclear.
Thus, this research aimed to excavate the functions and molecular mechanisms of miR-27b-5p in the biological behavior of GC by targeting CREBZF and regulating the STAT3 pathway, utilizing in vitro and in vivo experimental approaches. GC cell models with overexpressed or silenced miR-27b-5p and CREBZF were established to assess the impact on GC cell proliferation, migration, as well as invasion. Xenograft models were conducted to investigate the impacts on tumor growth following interference of miR-27b-5p and CREBZF. Additionally, real-time quantitative polymerase chain reaction (RT-qPCR) and western blotting were utilized to estimate the regulation of proteins related to the STAT3 pathway.
Plasma specimens and the corresponding clinical information of GC patients during 2021 to 2023 from Guangxi Medical University Cancer Hospital, Guilin People’s Hospital, and Youjiang Medical University were collected. GC tissue along with adjacent non-cancerous tissue specimens were gathered from 15 individuals who had undergone GC resection surgery at Guangxi Medical University Cancer Hospital. Inclusion criteria included patients with a confirmed diagnosis of GC via pathological examination, absence of prior chemotherapy, radiotherapy, or any form of treatment before surgery, and absence of cancer cells at resected tissue margins. The study protocol obtained the consent of the Ethics Committees of Guangxi Medical University Cancer Hospital, No. KY2020148; Guilin People’s Hospital, No. 2020-102KY; and Youjiang Medical University, No. YYFY-LL-2024-005. The specimens were collected and preserved at a temperature of -80 °C.
The miRNA sequencing data and the prognostic information of GC and non-cancer tissue were acquired from The Cancer Genome Atlas database (https://portal.gdc.cancer.gov/). For the data processing, the counts of miRNAs were obtained and normalized by log2(x + 1) method. For the overall survival time, live patients were identified based on the last follow-up time, and dead patients were identified based on the time of death. The expression status of miR-27b-5p between GC and non-cancerous tissue were compared, and the association between miR-27b-5p expression level and overall survival was investigated utilizing Kaplan-Meier curves.
NCI-N87, AGS, and 293T cell lines were procured from the Cell Bank of the Chinese Academy of Sciences. The cell lines were maintained in Roswell Park Memorial Institute (RPMI) 1640 culture medium or Dulbecco’s modified Eagle’s medium (DMEM), both supplemented with 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin. The cell lines were cultured at a temperature of 37 °C with an environment enriched with 5% CO2.
Procured from the Guangxi Medical University Animal Center, nude mice (BALB/c) aged of 6-8 weeks were fed under specific pathogen-free condition. Simultaneously, the mice were distributed into four groups through a random allocation process, each consisting of five mice: LV-miR-27b-5p, LV-negative control (NC), anti-LV-miR-27b-5p, and anti-LV-NC. A 100 μL cell suspension was injected subcutaneously into the rear axilla and upper inguinal regions. Tumor formation was assessed weekly by weighing and photographing the mice. The tumor’s length (a) and width (b) were measured every four days, and its volume was computed utilizing the following formula: 1/2 × a × b. Tumor growth curves were generated, and monitoring was discontinued once the tumor diameter reached 2.0 cm. The research obtained approval from the Ethics Committee of the Guangxi Medical University Animal Center, No. KY2020149.
Total RNAs were isolated from plasma specimens, tissue specimens and cells utilizing TRIzol reagent. Complementary DNA was synthesized through reverse transcription utilizing the miRNA First-Strand complementary DNA Kit (Monad, China). The RT-qPCR was performed utilizing the Hieff® qPCR SYBR Green Master Mix (Yeasen, China) following a specific protocol: Pre-denaturation at 95 °C for 30 seconds, succeeded by 40 cycles comprising denaturation at 95 °C for 5 seconds, and annealing at 60 °C for 30 seconds. For internal controls, the expression status of miRNAs was standardized by a control gene U6, and the expression of mRNAs was normalized by a control gene β-actin. The relative miRNA or mRNA expression was assessed utilizing the 2-ΔΔCT method. The primer sequences employed in this research are detailed in Table 1.
| Primers | Sequences (5’-3’) |
| H-miR-27b-5p | AGCTTAGCTGATTGGTGAAC |
| H-cyclin D1-F | GAGGAACAGAAGTGCGAGGAG |
| H-cyclin D1-R | TGGAGTTGTCGGTGAGATGC |
| H-c-myc-F | GGGTAGTGGAAAACCAGCCT |
| H-c-myc-R | AGAAATACGGCTGCACCGAG |
| H-Bax-F | CAGACCGTGACCATCTTTGTG |
| H-Bax-R | CTCAGCCCATCTTCTTCCAG |
| H-Bcl-2-F | TGGCCTTCTTTGAGTTCGG |
| H-Bcl-2-R | TCCACAGGGCGATGTTGTC |
| H-survivin-F | TGAGAACGAGCCAGACTTGG |
| H-survivin-R | TTCCTTTGCATGGGGTCGTC |
| H-CREBZF-F | GGGAAAGCAGAAGCAGGAC |
| H-CREBZF-R | GAGCAGAACTCCACCGACAC |
| H-ACTB-F | AGTCATTCCAAATATGAGATGCGTT |
| H-ACTB-R | TGCTATCACCTCCCCTGTGT |
| H-U6-F | CTCGCTTCGGCAGCACA |
| H-U6-R | AAAATATGGAACGCTTCACG |
Lentiviral plasmids were purchased from Hanbio (Shanghai, China) for overexpressing miR-27b-5p, referred to as LV-miR-27b-5p, or inhibiting it, termed anti-LV-miR-27b-5p. The plasmids were designed to carry the negative controls labeled as LV-NC and anti-LV-NC, and to upregulate CREBZF denoted as OE-CREBZF, along with the matching negative control referred to as OE-NC. Cells were cultured in six-well plates at 5 × 104 cells/mL, succeeded by trans
The TargetScan tool (http://www.targetscan.org/vert_72/) was employed to analyze the potential binding domains of miR-27b-5p within the 3’-untranslated region (UTR) of the CREBZF mRNAs. Both the wild-type 3’-UTR of CREBZF and the mutated 3’-UTR that did not contain the binding domains for CREBZF, were cloned downstream of the luciferase coding sequence within the pMIR-REPORT plasmids. These constructs were transfected into 293T cells alongside the miR-27b-5p-mimic or NC-mimic, utilizing lipofectamine 2000 (Invitrogen, Carlsbad, CA, United States). Following a 48-hour incubation period, the activity of luciferase was assessed through a dual-luciferase reporter assay system (E1960; Promega, Madison, WI, United States). The relative activities of luciferase were quantified by calculating the proportion of firefly luciferase activity to renilla luciferase activity.
A suspension of cells previously transfected with the plasmids was assembled and centrifuged and plated into 96-well plates at 2 × 103 cells per well (100 μL) in DMEM or RPMI 1640 supplemented with 10% FBS. After 24, 48, 72, and 96 hours of incubation, 10 μL cell counting kit-8 (CCK-8) solution was added to each individual well, followed by a 2-h incubation at 37 °C in a 5% CO2 environment. The optical density at 450 nm (OD450) was quantified utilizing an automated microplate reader.
Cell migration was assessed using the Transwell assay. The cells were cultured in serum-free DMEM or RPMI 1640 at 105 cells/mL and 200 μL cell suspension was added to the upper chamber. Simultaneously, 500 μL DMEM or RPMI 1640 with 10% FBS was added to the lower chamber. These cultures were incubated for 48 hours at 37 °C and an environment containing 5% CO2. The cells were wiped off and removed from the upper surface of the filter. The membranes were treated by 30 minutes incubation with 4% paraformaldehyde, and 30 minutes incubation with 0.1% crystal violet solution. Following the drying process, the number of migrated cells was counted using microscope.
GC cells that had been stably transfected (5 × 105) were seeded into six-well culture plates. After the cells were adhered to the surface, linear scratches were made by sterile pipette tips with a volume of 200 μL. The plates were rinsed several times utilizing phosphate-buffered saline (PBS) to eliminate the suspended cells, and serum-free medium was used for GC cell culture. The wound images were captured at the same location under the microscope (Olympus, Japan) at 0 and 24 hours. The distance between the wound edges was measured. All procedures were conducted in triplicate.
Apoptosis was examined by cell cycle and apoptosis analysis kit (Beyotime Biotechnology, Shanghai, China). GC cells were detached using trypsin without EDTA, and collected by centrifugation and resuspended with ice-cold PBS. A cell suspension at a concentration of 5 × 106 cells/mL was mixed with 0.5 mL solution of propidium iodide. The mixture was subsequently incubated in darkness for 30 minutes at a temperature of 37 °C. The labeled cells were subjected to flow cytometric analysis using a Beckman Coulter instrument (Brea, CA, United States). The apoptotic cell percentage was calculated by summing the proportions of cells in both the early and late apoptotic stages.
Cells and tissues were subjected to lysis using radioimmunoprecipitation lysis buffer, supplemented with phenylmethylsulfonyl fluoride at a dilution ratio of 100:1. Total protein concentration was determined utilizing a bicinchoninic acid protein assay kit. The protein obtained was resolved by 10% sodium-dodecyl sulfate gel electrophoresis. The proteins were transferred to the polyvinylidene difluoride membranes with a pore diameter of 0.22 μm. The membranes were blocked for 1.5 hours in Tris-buffered saline with Tween and 5% skimmed milk. Following three washes in PBS, the membranes underwent overnight incubation at 4 °C with specific antibodies (Abcam, Cambridge, United Kingdom) targeting various proteins: β-actin, STAT3, phosphorylated STAT3 (p-STAT3), cellular myelocytomatosis oncogene (c-myc), B-cell lymphoma 2 (Bcl-2), Bcl-2-associated X protein (Bax), cyclin D1, survivin, and CREBZF. After washing with PBS for three additional times, the membranes underwent treatment with a horseradish-peroxidase-conjugated anti-rabbit IgG secondary antibody for 1 hour at ambient temperature. The enhanced chemiluminescence was employed to quantify the protein bands, with the relative concentrations of the target proteins being standardized against β-actin.
Statistical analyses were conducted utilizing R software 4.3.2 and GraphPad Prism 9.0. The expression data were shown as mean ± SEM. Independent sample t tests or Welch’s t tests were used to compare two independent groups, whereas one-way analysis of variance was utilized for analysis of distinctions among multiple groups. Nonparametric data were subjected to analysis using the Mann-Whitney U or Kruskal-Wallis H test. Statistical significance was demonstrated by P < 0.05.
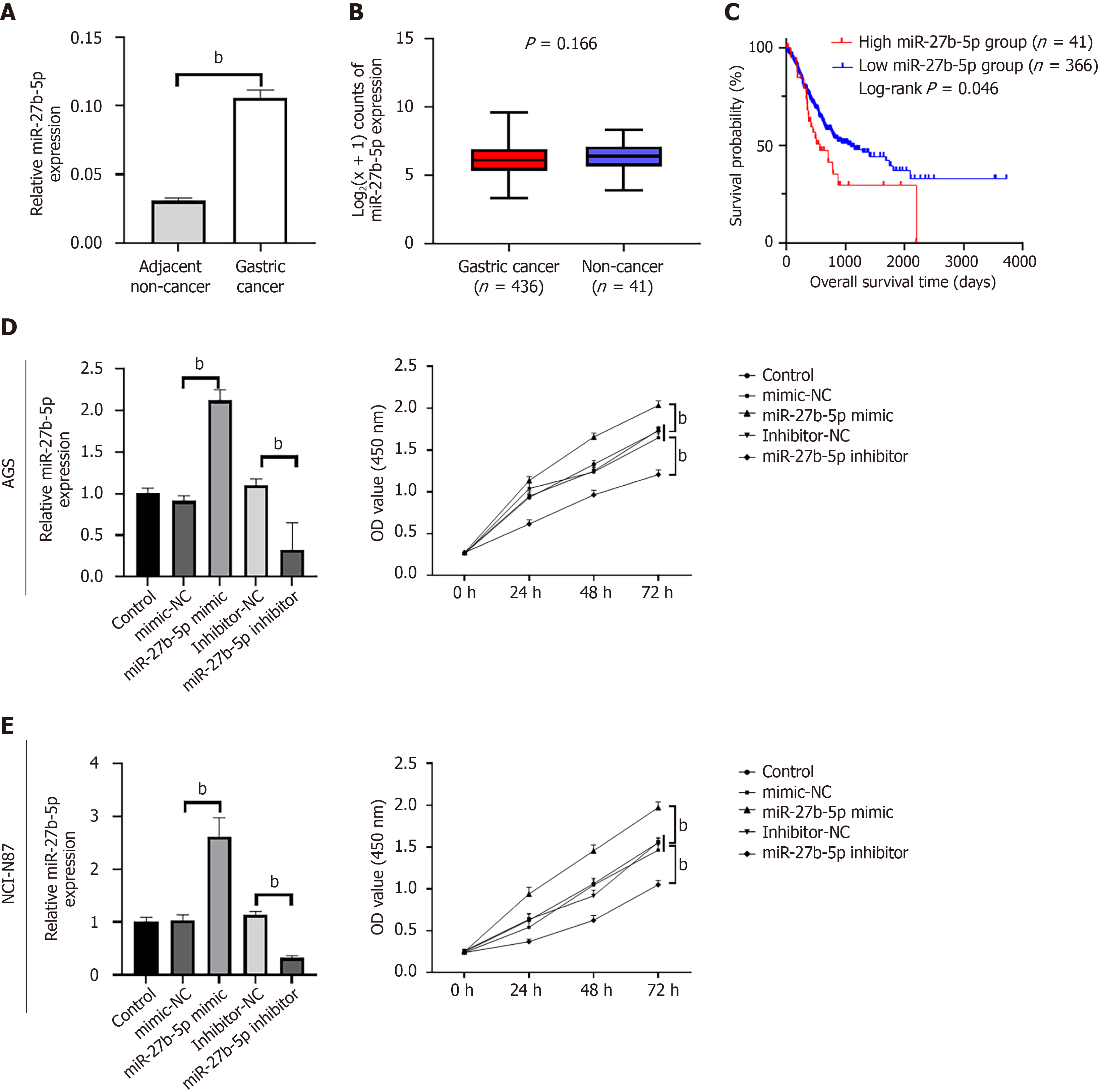
The study included 168 plasma GC specimens (Supplementary Table 1), 15 GC, and 15 adjacent non-cancerous tissue specimens. Totally 436 GC and 41 non-cancerous tissue were collected from The Cancer Genome Atlas database. In contrast to the adjacent non-cancerous tissue group, the transcriptional expression status of miR-27b-5p was upregulated in GC tissue (Figure 1A), but no statistically significant difference was observed according to the public miRNA-sequencing data (Figure 1B). Based on the results of clinical characteristics analysis, higher tumor invasion depth (T2 + T3 + T4 vs T1; P < 0.0001), regional lymph node metastasis (N1 + N2 + N3 vs N0; P = 0.0098), metastasis (M1 vs M0; P = 0.0228) and clinical stage (II + III + IV vs I; P = 0.0004) were all associated with elevated expression of miR-27b-5p, with significant differences (Table 2). High miR-27b-5p appeared to be correlated to the poorer prognosis in patients with GC (log-rank P = 0.046, Figure 1C). This indicated that increased levels of miR-27b-5p expression correlated with advanced GC.
| Characteristics | n | MiR-27b-5p expression | Mann-Whitney U | P value |
| Gender | ||||
| Male | 108 | 0.1183 | 3071 | 0.5763 |
| Female | 60 | 0.1162 | ||
| Age | ||||
| ≥ 60 | 48 | 0.1134 | 2603 | 0.3315 |
| < 60 | 120 | 0.1177 | ||
| T | ||||
| T1 | 71 | 0.0934 | 1871 | P < 0.0001 |
| T2 + T3 + T4 | 97 | 0.2031 | ||
| N | ||||
| N0 | 113 | 0.1088 | 2347 | 0.0098 |
| N1 + N2 + N3 | 55 | 0.1984 | ||
| M | ||||
| M0 | 137 | 0.1132 | 1569 | 0.0228 |
| M1 | 31 | 0.2404 | ||
| Clinical stages | ||||
| I | 107 | 0.1063 | 2208 | 0.0004 |
| II + III + IV | 61 | 0.2243 |
To explore the biological significance of miR-27b-5p, lentiviruses containing miR-27b-5p mimics and inhibitors were transfected into AGS and NCI-N87 cell lines, respectively. RT-qPCR indicated that the cell lines were successfully transfected. Relative to the mimic-NC group, the miR-27b-5p mimic group exhibited significant elevation of miR-27b-5p, while the miR-27b-5p inhibitor group displayed a marked decrease of miR-27b-5p when compared to the inhibitor-NC group (Figure 1D and E).
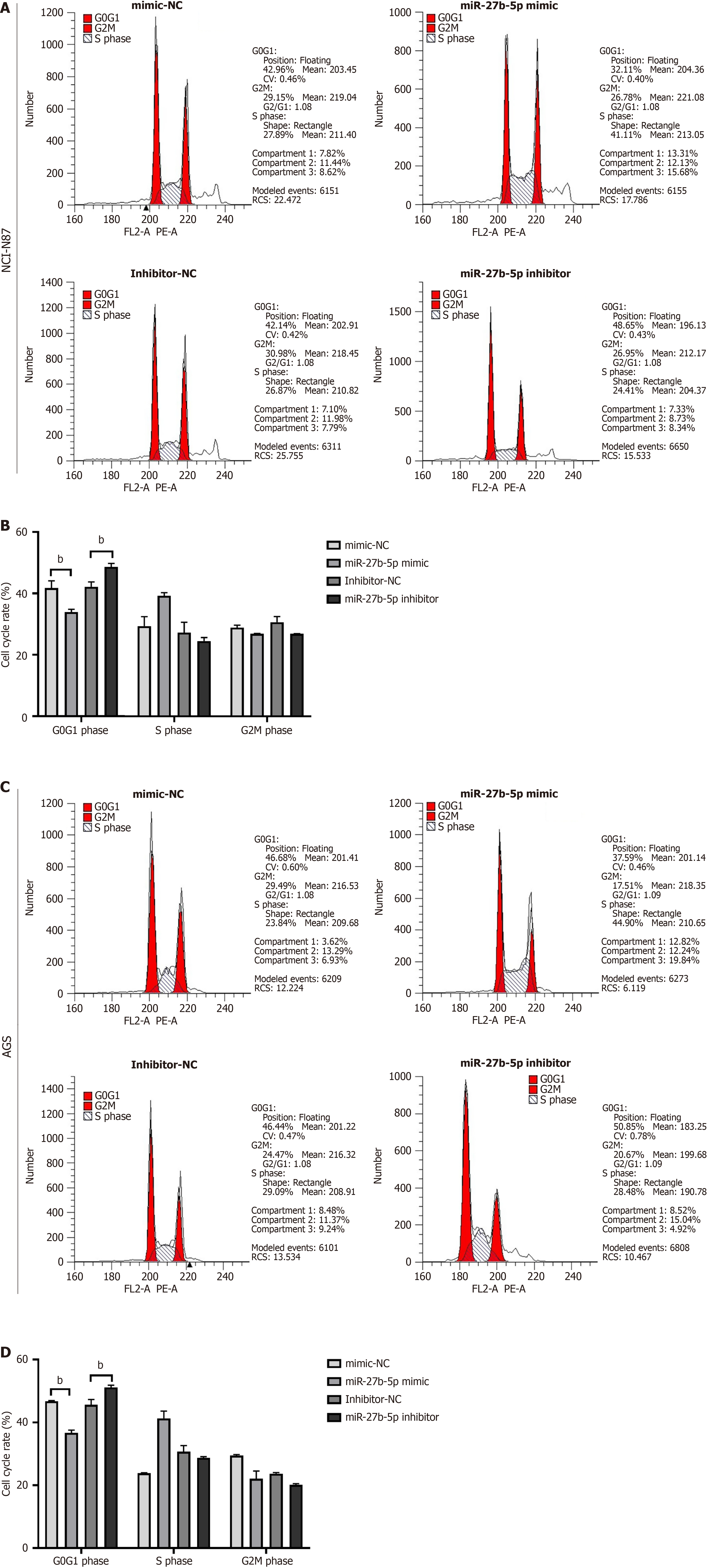
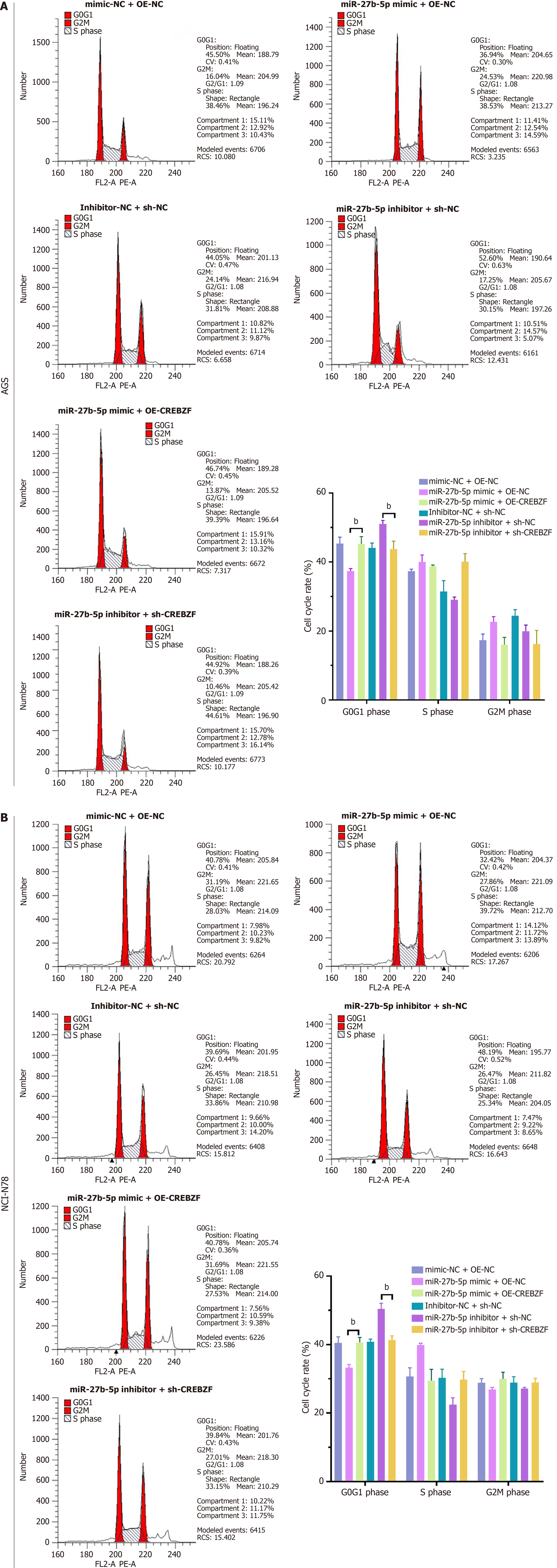
Based on the CCK-8 assay, OD450 in the miR-27b-5p mimic group was notably elevated when compared to that in the control groups, while OD450 in the miR-27b-5p inhibitor group was significantly decreased, in both NCI-N87 and AGS cell lines (Figure 1D and E; P < 0.01). Flow cytometry analysis was performed to explore the impact of miR-27b-5p on the cell cycle. In comparison to the mimic-NC group, the miR-27b-5p mimic group presented a significant decrease in the proportions of NCI-N87 and AGC cell lines in G0/G1 phase, while the percentage in S phase was increased (Figure 2). Inversely, compared to the inhibitor-NC group, the percentages of NCI-N87 and AGC cell lines in the G0/G1 phase exhibited a notable increase in the miR-27b-5p inhibitor group, accompanied by a decrease in the S phase. The findings in the CCK-8 assay and flow cytometric analysis were consistent, which demonstrated that increased miR-27b-5p levels facilitated proliferation of AGS and NCI-N87 cell lines, while silencing miR-27b-5p produced contrary effects.
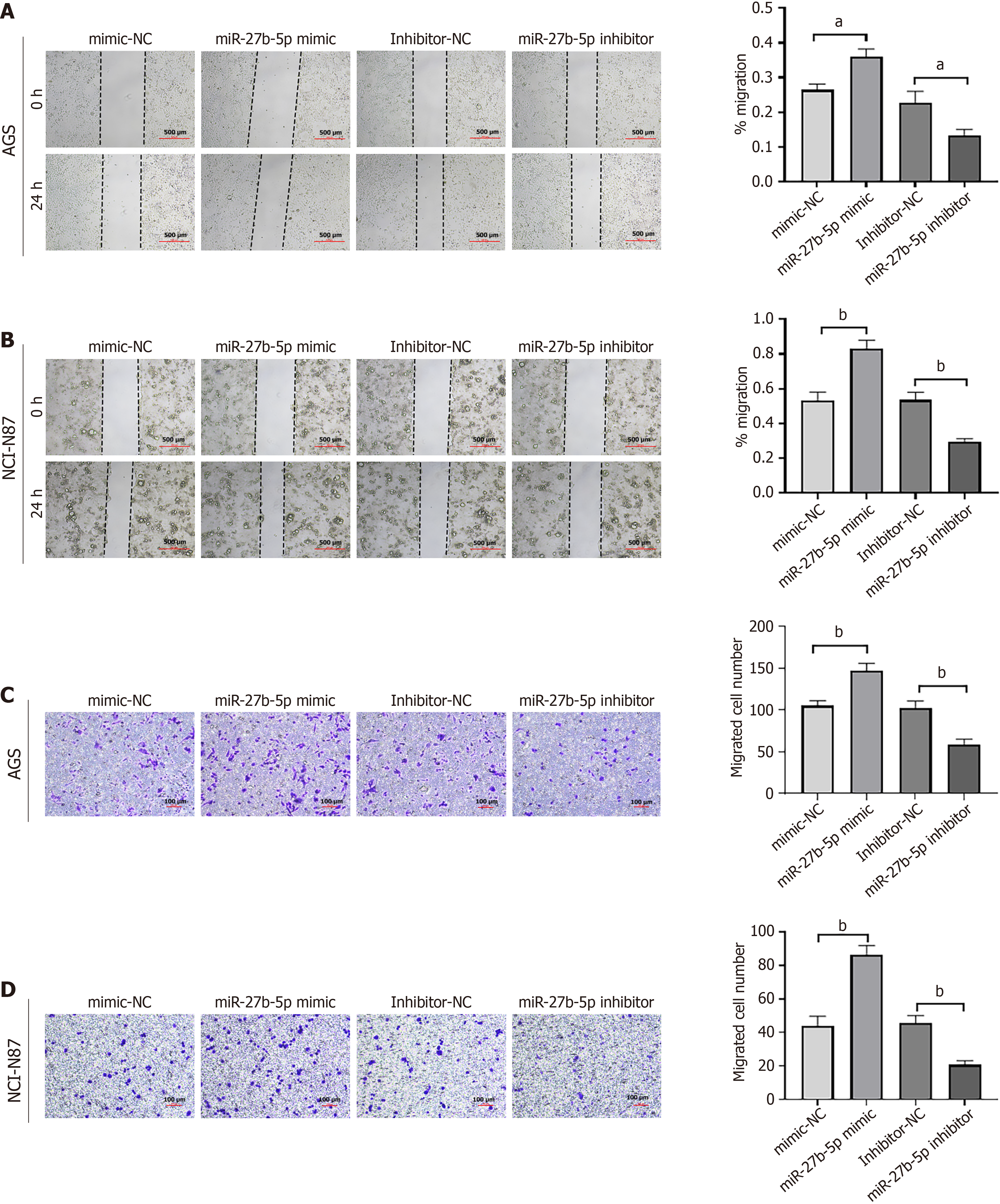
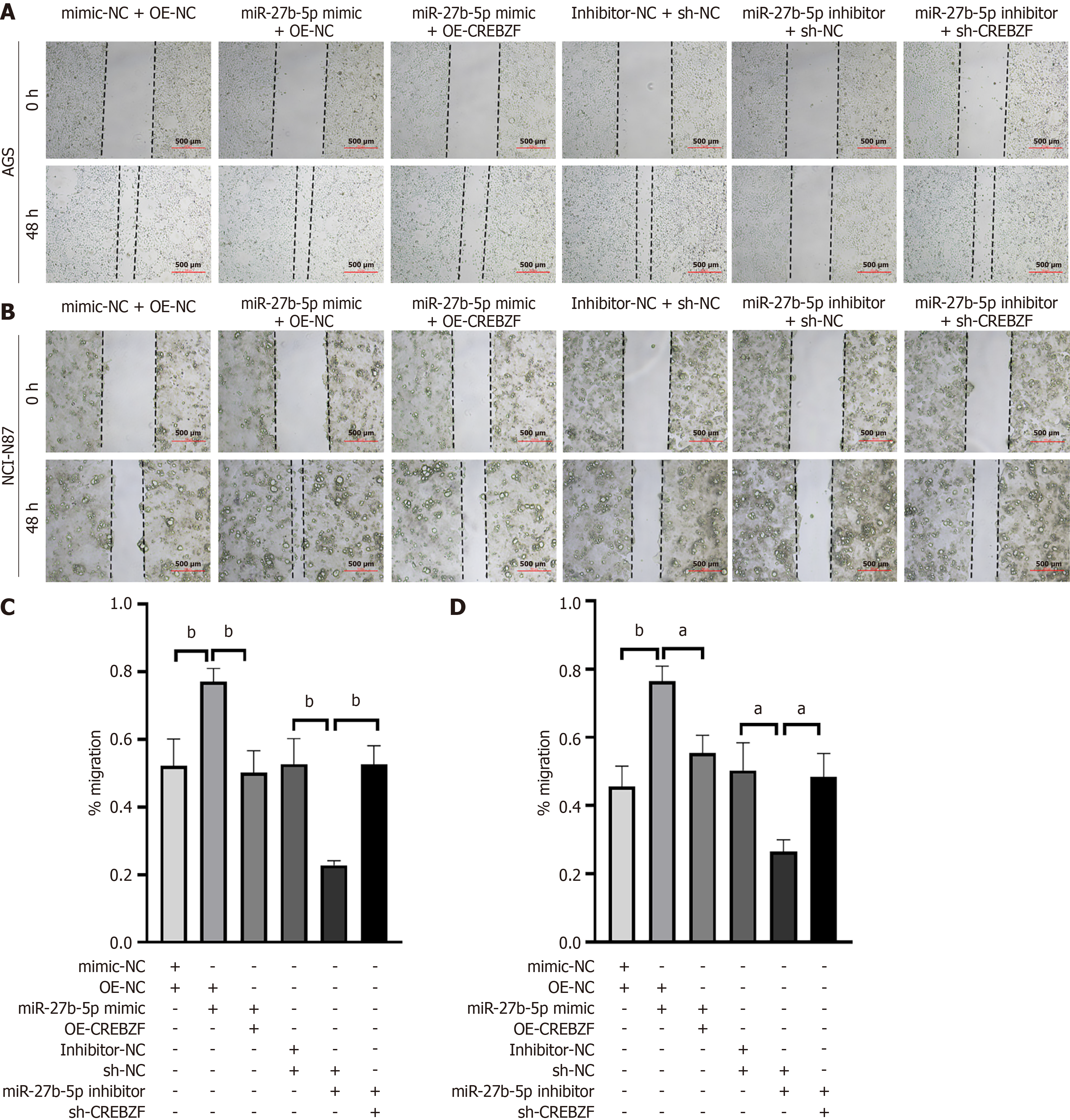
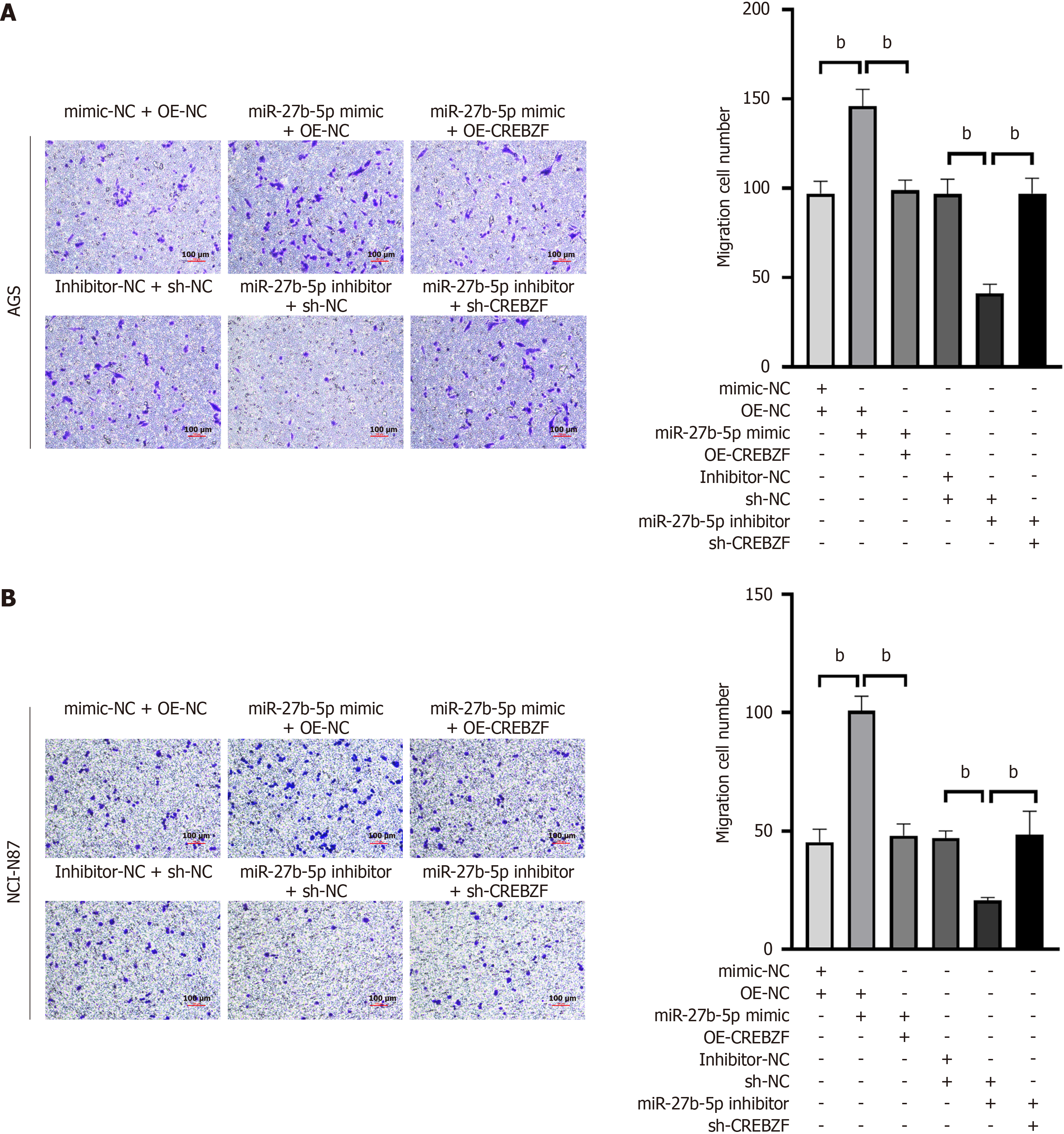
The wound healing experiment demonstrated that, as the level of miR-27b-5p increased, the distance and rate of GC cell migration also increased. Conversely, suppression of miR-27b-5p diminished cell migration (Figure 3A and B). In line with the results of wound healing assay, similar findings were observed in the Transwell assay (Figure 3C and D). Overexpression of miR-27b-5p in AGS or NCI-N87 cell lines enhanced cell migration, whereas a decrease in miR-27b-5p resulted in lower migration rates than those observed in the control group.
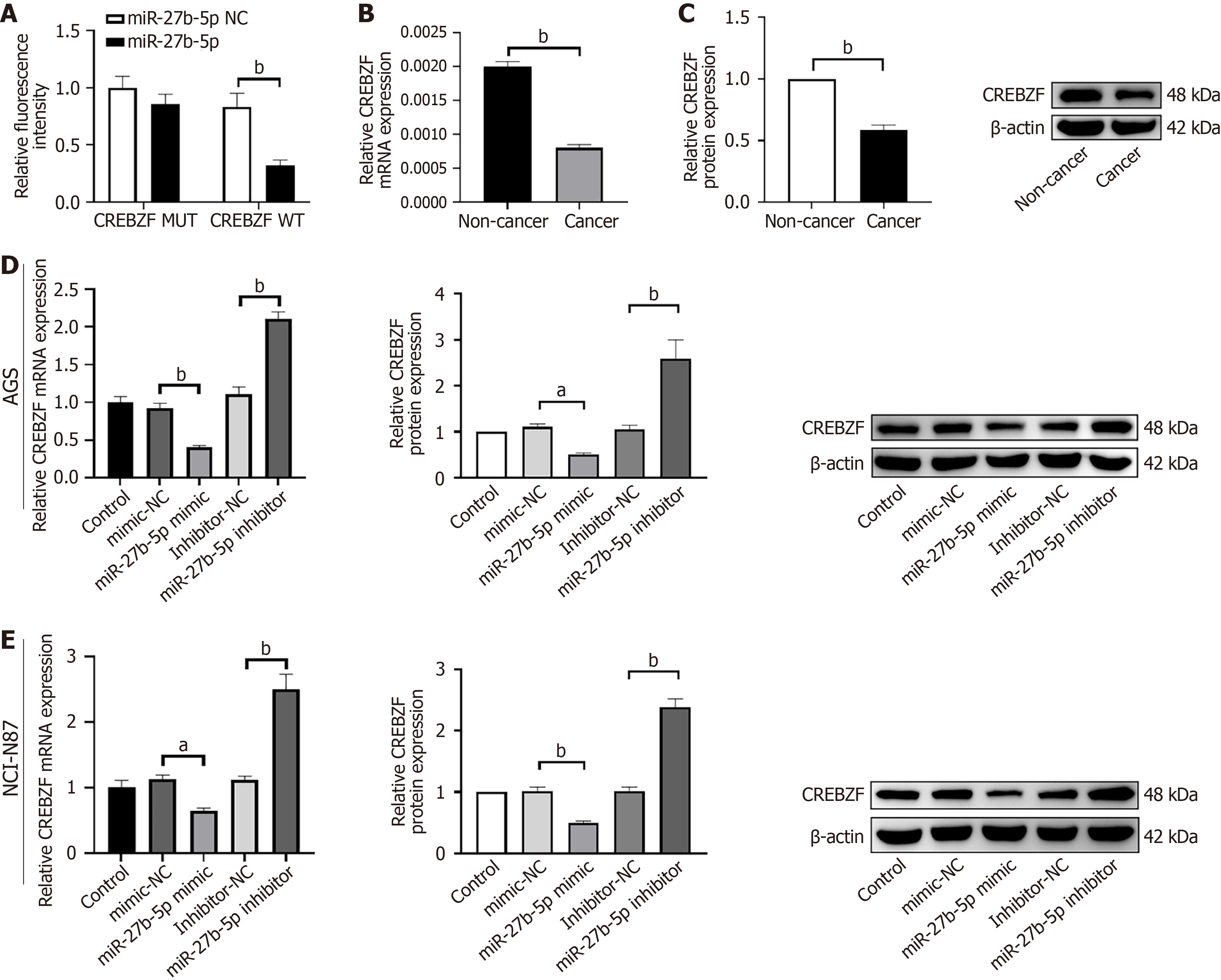
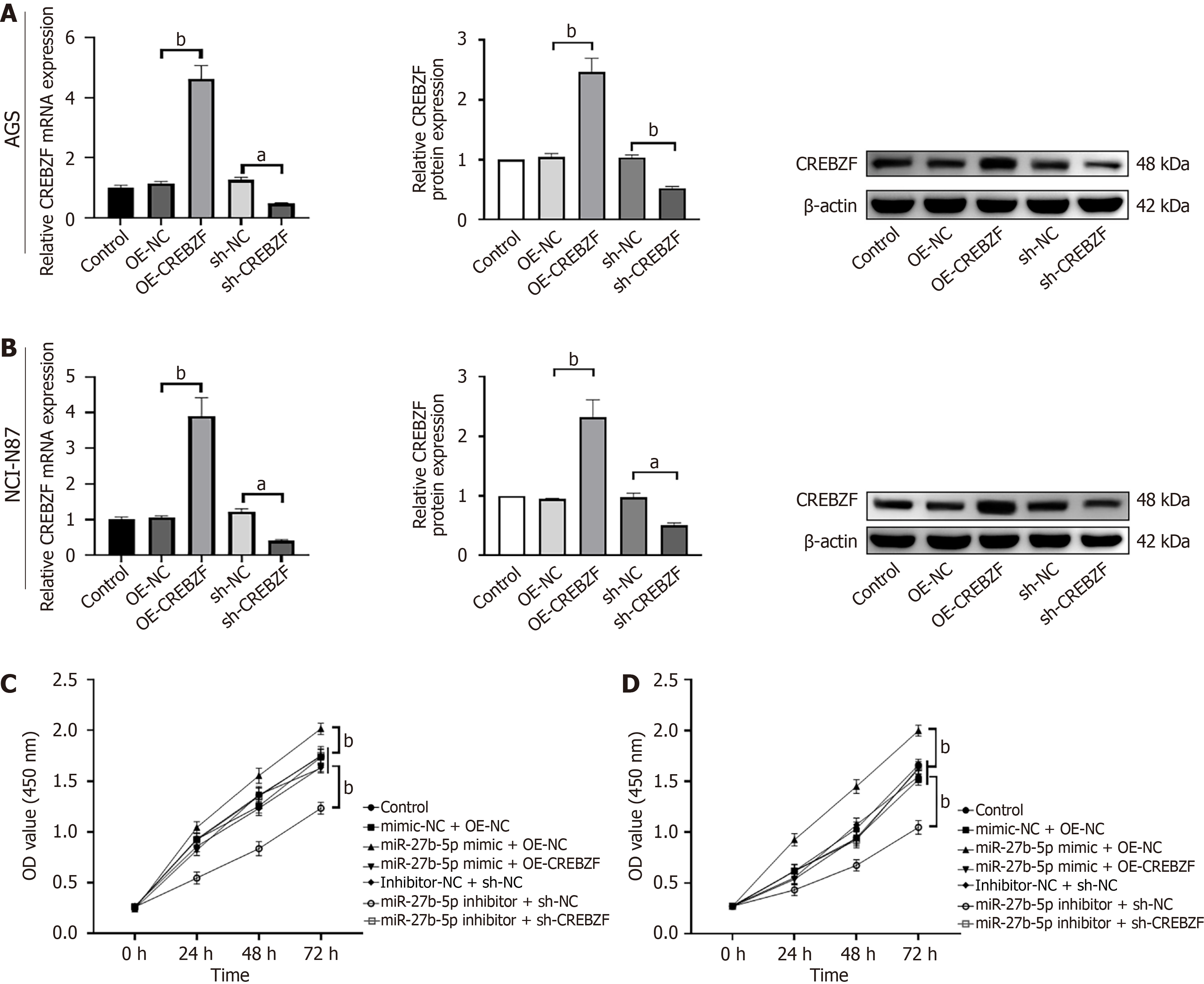
A promising binding domain for miR-27b-5p was predicted by TargetScan within the 3’-UTR of CREBZF. To investigate this potential interaction, miR-27b-5p or NC mimics were co-transfected alongside the luciferase reporter constructs containing either the wild-type or mutated 3’-UTR of CREBZF. Insertion of miR-27b-5p mimics with wild-type 3’-UTR of CREBZF showed decreased luciferase activity, which was completely reversed with mutant-type 3’-UTR of CREBZF (Figure 4A). The findings suggest that miR-27b-5p may directly target CREBZF, thereby suppressing its activity. In the subsequent RT-qPCR and western blotting experiments, expression of CREBZF was reduced in GC tissue in contrast to adjacent noncancerous tissue (Figure 4B and C). Excess of miR-27b-5p led to reduction in both mRNA and protein levels of CREBZF, and inhibition of miR-27b-5p expression produced a contrary effect (Figure 4D and E).
To investigate the functions of miR-27b-5p in promoting proliferation and migration in GC via regulating CREBZF, we constructed the OE-CREBZF vector to upregulate expression of CREBZF. NCI-N87 and AGS cell lines were transfected with the miR-27b-5p mimic, mimic-NC, miR-27b-5p inhibitor, inhibitor-NC, OE-CREBZF, OE-NC, sh-NC, and sh-CREBZF. Expression status of CREBZF was assessed utilizing RT-qPCR and western blotting (Figure 5A and B). Cotransfection of OE-CREBZF with the miR-27b-5p mimic attenuated the functions of miR-27b-5p on cellular proliferation, based on the CCK-8 assay (Figure 5C and D) and flow cytometry (Figure 6). The same results were observed in the wound healing assay (Figure 7) and Transwell assay (Figure 8), which demonstrated that CREBZF overexpression reversed the migratory effects induced by miR-27b-5p in GC cell lines. The findings demonstrated that miR-27b-5p stimulated the proliferation and migration in GC cells through its direct interaction with CREBZF.
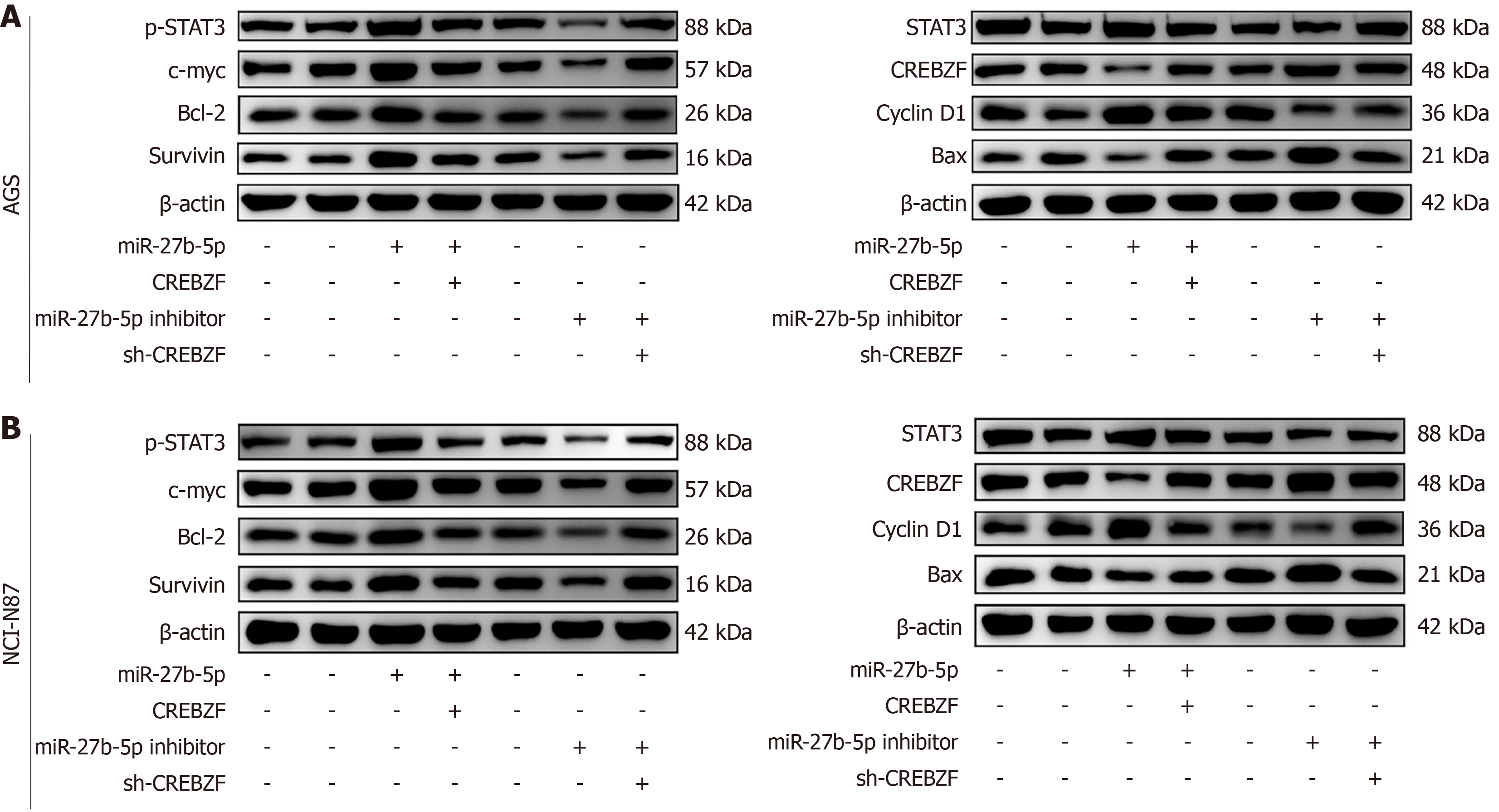
CREBZF acts as a negative regulator of the STAT3 signaling pathway, and we detected expression of STAT3-pathway-related proteins (Figure 9, Supplementary Figures 1 and 2). Excessive miR-27b-5p expression in AGS and NCI-N87 cell lines significantly promoted expression of STAT3, p-STAT3, c-myc, cyclin D1, Bcl-2, and survivin, and downregulated Bax and CREBZF. The p-STAT3/STAT3 ratio was increased and the Bax/Bcl-2 ratio was decreased. Excessive CREBZF expression counteracted the effects induced by miR-27b-5p overexpression. CREBZF significantly downregulated expression of STAT3, p-STAT3, c-myc, cyclin D1, Bcl-2, and survivin, but enhanced expression of Bax and CREBZF. The inhibition of miR-27b-5p significantly reduced the expression status of p-STAT3, p-STAT3/STAT3 ratio, c-myc, cyclin D1, Bcl-2, and survivin, while elevated Bax, Bax/Bcl-2 ratio, and CREBZF expression. Conversely, disrupting expression of CREBZF attenuated the effects of the miR-27b-5p inhibitor, leading to significant increases of p-STAT3, p-STAT3/STAT3 ratio, c-myc, cyclin D1, Bcl-2, and survivin expression, and decreases in Bax, Bax/Bcl-2 ratio, and CREBZF expression. These results elucidated that miR-27b-5p modulates the STAT3 signaling pathway via regulating CREBZF to affect cellular biological processes.
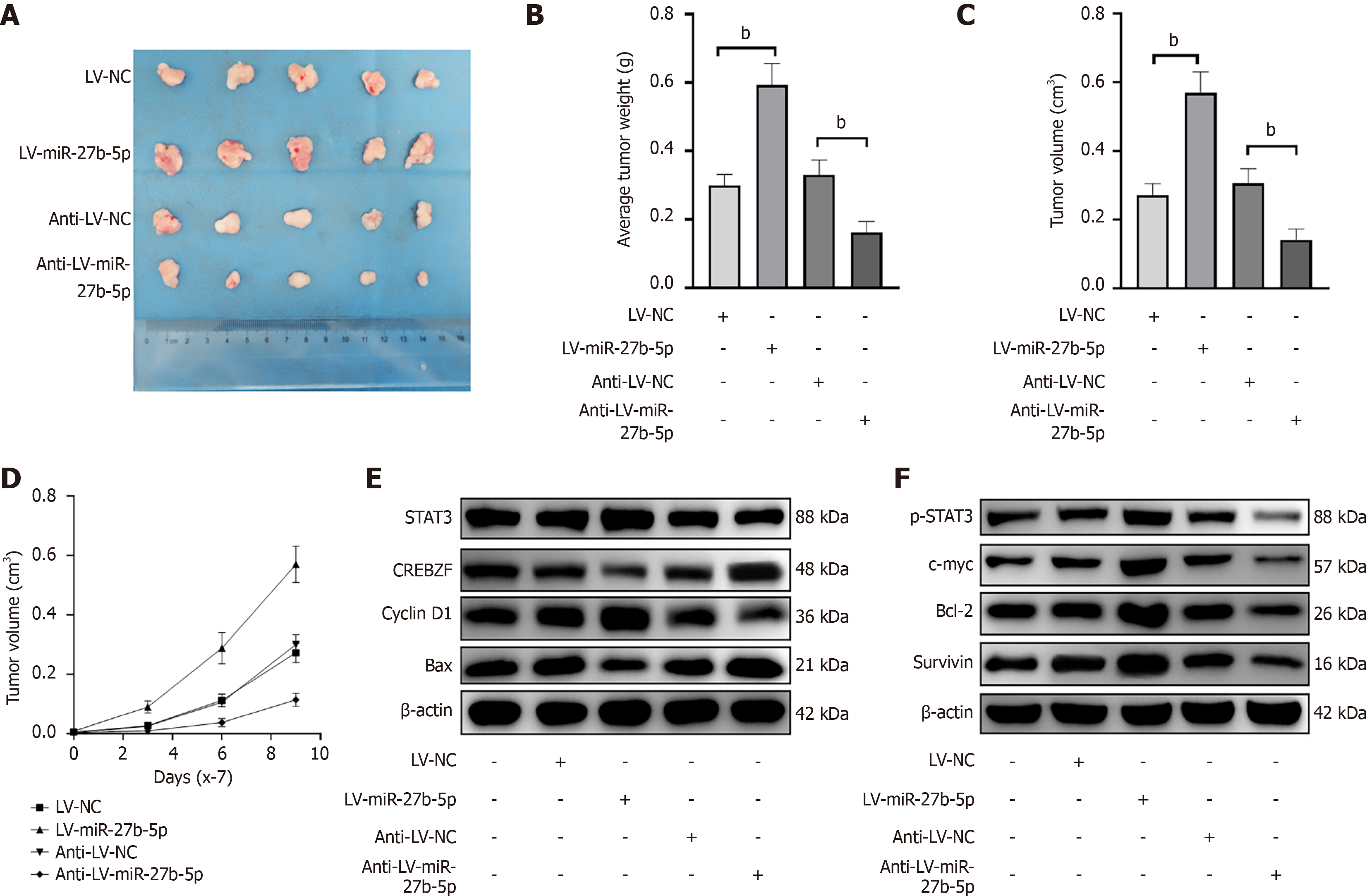
The pro-tumorigenic functions of miR-27b-5p were examined in vivo. AGS cells transfected by LV-miR-27b-5p, LV-NC, anti-LV-miR-27b-5p, and anti-LV-NC were administered via subcutaneous injection into four groups of BALB/c nude mice. By day 7 post-injection, subcutaneous tumors approximately the size of a green pea were evident. The sizes of xenografts were assessed every 4 days using calipers. The mice were killed after 18 days, and the xenografts are shown in Figure 10. Elevated miR-27b-5p expression led to enhanced tumor proliferation, whereas decreased levels of miR-27b-5p suppressed tumorigenesis. Tumor weight and volume were markedly greater in the miR-27b-5p overexpression group (Figure 10A-D), whereas absence of miR-27b-5p corresponded with a notable decrease in these metrics. Western blotting (Figure 10E, Supplementary Figure 3) showed that in comparison to the LV-NC group, transplantation tumors overexpressing miR-27b-5p showed significant upregulation of STAT3, p-STAT3, p-STAT3/STAT3, c-myc, cyclin D1, Bcl-2, and survivin expression (all P < 0.01), while Bax (P < 0.01), Bax/Bcl-2 (P < 0.05), and CREBZF (P < 0.01) expression was significantly downregulated. Conversely, after miR-27b-5p knockdown, transplantation tumors exhibited significant downregulation of p-STAT3, p-STAT3/STAT3, c-myc, cyclin D1, Bcl-2 (the above P < 0.01), and survivin (P < 0.05) ex
There is evidence that miRNAs regulate target genes’ expression through their interaction with the 3’-UTR, and function as oncogenes or tumor suppressors in cancer evolution. For instance, He and Zou[4] found that miR-96-5p promotes the growth of GC cells by targeting forkhead box O3. Ding et al[5] reported that miR-146b-5p inhibits the aggressive development of GC by targeting TNFR-associated factor. In addition, Meng et al[6] discovered that miR-93-5p promotes the biological progression of GC cells through its regulation of the Hippo signaling pathway. Therefore, exploring the biological functions of miRNAs is critical for understanding the carcinogenesis processes of GC and developing new cancer therapies.
MiR-27b-5p appears to be a crucial factor in the advancement of gastric adenocarcinoma from precancerous adenomas. The precise molecular mechanism by which miR-27b-5p operates in GC has yet to be fully elucidated. RT-qPCR demonstrated that higher miR-27b-5p expression levels were observed in GC tissue than in the paired non-cancerous tissue. This observation prompted further exploration into the role of the 5’ end-derived miR-27b-5p in GC. A series of proliferation and migration assays in this research demonstrated that excess expression of miR-27b-5p encouraged GC progression, whereas its inhibition had contrary effects. The subsequent findings of in vivo experiments supported the in vitro results. Collectively, these results manifest that miR-27b-5p may represent a promising therapeutic target in GC.
To comprehend the influence of miR-27b-5p on cellular proliferation and migratory capacity, bioinformatics analysis aimed at identifying potential target genes within GC cells was conducted. Among the identified candidates, we focused on the gene CREBZF. CREBZF belongs to the bZIP transcription factor family, which includes the cAMP response element-binding protein and activating transcription factor members. CREBZF is crucial for its anti-tumor effects. For example, previous studies have demonstrated that CREBZF has anti-cancer properties in breast cancer[3]. In our research, it was found that the level of CREBZF was markedly reduced in GC tissue. Additionally, dual luciferase reporter assays have validated that miR-27b-5p directly interacts with and targets CREBZF, resulting in reduced CREBZF levels. CREBZF levels were notably diminished in AGS and NCI-N87 cells exhibiting overexpression of miR-27b-5p, and enhancing CREBZF expression reversed the influence of miR-27b-5p on cellular proliferation and migration. The results of our research indicate that miR-27b-5p play a contributory role in the development of GC through downregulating CREBZF.
Previous studies have demonstrated that CREBZF activation promotes P53 stimulation and elicits anticancer effects[7]. The deletion of CREBZF in hepatic cells promotes cell cycle progression and cellular proliferation[8]. Increasing CREBZF expression by inhibiting miR-421 and miR-29b-1-5p may provide a promising approach to prevent progression of early GC[9]. Therefore, CREBZF is posited to exert tumor-suppressive effects in the initiation and progression of GC.
After being activated by upstream signaling molecules, STAT3 functions as a transcriptional activator to regulate downstream target genes expression. Recent studies have demonstrated that persistent activation of STAT3 is commonly detected in various tumor types, which is closely associated with tumor growth, invasion, metastasis, as well as the emergence of resistance to chemotherapy and radiotherapy. STAT3 is crucial to the function of breast cancer stem cells and the development of tamoxifen resistance, contributing to breast cancer progression, proliferation, metastasis, and chemotherapy resistance[10]. Inhibition of the STAT3–ferroptosis negative regulatory axis could suppress tumor proliferation and chemotherapy resistance in GC[11]. STAT3 activation in the tumor stroma could induce immunosuppressive effect, promote a carcinogenic phenotype in fibroblasts and then facilitate tumor progression[12]. STAT3 also facilitates nuclear-factor-κB-mediated immune evasion, which is reversed by vemurafenib[13]. These findings suggest that STAT3 activation is implicated in the malignant biological behavior of GC.
In the current research, we found that CREBZF was involved in inhibiting STAT3 transcriptional activity in vitro or in vivo. Previous studies have indicated that CREBZF directly interacts with the linker domain of STAT3, and inhibits its dimerization and transcriptional activity in the process of liver regeneration in mice[8]. It was also found in our research that excess expression of miR-27b-5p directly inhibited expression of CREBZF and weakened its activity, leading to CREBZF-mediated inhibition of STAT3. Ultimately, the STAT3 pathway is upregulated. This regulation not only increased STAT3 levels but also further elevated p-STAT3 and p-STAT3/STAT3 ratio. In addition, this regulatory mechanism promotes the upregulation of c-myc, cyclin D1, Bcl-2, and survivin, which collectively facilitate cell proliferation and inhibit apoptosis. The downregulation of the pro-apoptotic gene Bax and the ratio of Bcl-2/Bax mediates anti-apoptotic signaling in GC cells. The animal models confirmed that miR-27b-5p positively regulated STAT3 by targeting CREBZF and promoting tumor proliferation in nude mice. The modulation of gene expression associated with c-myc, cyclin D1, Bcl-2, survivin, and Bax in vivo was consistent with that in cell experiments. Overall, these results offer novel perspectives on the molecular mechanisms underlying GC pathogenesis. Therefore, miR-27b-5p promotes the carcinogenic processes and involves in the initiation and progression of GC by regulating the STAT3 pathway via targeting CREBZF.
With the development of research, miRNAs have a broad prospect in the clinical diagnosis and treatment of certain malignant tumors as an effective alternative to current clinical practices. The therapies targeting miRNAs have entered clinical development phases. For instance, the tumor suppressor miR-16 has progressed to phase 1 clinical trials for pleural mesothelioma therapy[14], and a therapy targeting miR-155 is under evaluation in a phase 2 clinical trials for T-cell lymphoma therapy (https://clinicaltrials.gov/show/NCT03713320). Our study found that miR-27b-5p promoted cellular proliferation and migration in GC, indicating its potential as an innovative therapeutic target for GC in the future. Nonetheless, further investigation based on a larger clinical sample cohort and the exploration of additional associated pathways are necessary to deepen the understanding of miR-27b-5p. This research elucidates the effects and possible mechanisms of miR-27b-5p in modulating the STAT3 signaling pathway via targeting CREBZF, thereby regulating the biological processes underlying the initiation and progression of GC. In the future, we will attempt to translate these insights into clinical practice, with the goal of providing more effective therapeutic strategies for GC patients.
There were some limitations to our study. First, the functions of miR-27b-5p and CREBZF were verified in vitro and in vivo experimental approaches; however, these models are not identical to the complicated internal environment of the human body. For cell apoptosis, more direct evidence demonstrating the involvement of the miR-27b-5p-CREBZF axis in apoptosis and cytotoxic effects of drugs on tumor cells should be supplemented in the future research. Second, histological evidence was lacking in this study, because protein expression was analyzed exclusively by western blotting, and the capacity of cell proliferation was only investigated at the cellular level. Immunohistochemical staining and histological analysis would strengthen the research. Third, although this study extends preliminary understanding of the carcinogenic mechanisms associated with miR-27b-5p, the complex molecular regulatory network remains insufficiently elucidated. The initiation and progression of GC is a multifactorial and multi-stage process influenced by numerous factors, and further exploration should focus on additional factors and pathways. Consequently, future investigations should adopt a comprehensive perspective on these variables to attain a more comprehensive understanding.
MiR-27b-5p may target CREBZF and regulate the STAT3 signaling pathway, thereby enhancing the proliferation, migration, progression, and malignant conversion of GC cells in vitro. MiR-27b-5p also facilitates the proliferation and metastatic capacity of xenograft tumors, and enhances the malignant progression of GC in vivo. Therefore, miR-27b-5p is a potential therapeutic target for GC.
The authors thank the Guangxi Zhuang Autonomous Region Clinical Medicine Research Center for Molecular Pathology and Intelligent Pathology Precision Diagnosis.
| 1. | Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76817] [Cited by in RCA: 69588] [Article Influence: 13917.6] [Reference Citation Analysis (42)] |
| 2. | Kim YJ, Hwang KC, Kim SW, Lee YC. Potential miRNA-target interactions for the screening of gastric carcinoma development in gastric adenoma/dysplasia. Int J Med Sci. 2018;15:610-616. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 3. | Zhou B, Xue J, Wu R, Meng H, Li R, Mo Z, Zhai H, Chen X, Liu R, Lai G, Chen X, Li T, Zheng S. Correction: CREBZF mRNA nanoparticles suppress breast cancer progression through a positive feedback loop boosted by circPAPD4. J Exp Clin Cancer Res. 2023;42:158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 4. | He X, Zou K. MiRNA-96-5p contributed to the proliferation of gastric cancer cells by targeting FOXO3. J Biochem. 2020;167:101-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 41] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 5. | Ding JN, Zang YF, Ding YL. MiRNA-146b-5p inhibits the malignant progression of gastric cancer by targeting TRAF6. Eur Rev Med Pharmacol Sci. 2021;25:3151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 6. | Meng H, Li YY, Han D, Zhang CY. MiRNA-93-5p promotes the biological progression of gastric cancer cells via Hippo signaling pathway. Eur Rev Med Pharmacol Sci. 2019;23:4763-4769. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 7. | López-Mateo I, Villaronga MÁ, Llanos S, Belandia B. The transcription factor CREBZF is a novel positive regulator of p53. Cell Cycle. 2012;11:3887-3895. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 32] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 8. | Hu Z, Han Y, Liu Y, Zhao Z, Ma F, Cui A, Zhang F, Liu Z, Xue Y, Bai J, Wu H, Bian H, Chin YE, Yu Y, Meng Z, Wang H, Liu Y, Fan J, Gao X, Chen Y, Li Y. CREBZF as a Key Regulator of STAT3 Pathway in the Control of Liver Regeneration in Mice. Hepatology. 2020;71:1421-1436. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 43] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 9. | Kim YJ, Jeong S, Jung WY, Choi JW, Hwang KC, Kim SW, Lee YC. miRNAs as potential biomarkers for the progression of gastric cancer inhibit CREBZF and regulate migration of gastric adenocarcinoma cells. Int J Med Sci. 2020;17:693-701. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 10. | Zamanian MY, Golmohammadi M, Alalak A, Kamiab Z, Obaid R, Ramírez-Coronel AA, Hjazi A, Abosaooda M, Mustafa Y, Heidari M, Verma A, Nazari Y, Bazmandegan G. STAT3 Signaling Axis and Tamoxifen in Breast Cancer: A Promising Target for Treatment Resistance. Anticancer Agents Med Chem. 2023;23:1819-1828. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 11. | Ouyang S, Li H, Lou L, Huang Q, Zhang Z, Mo J, Li M, Lu J, Zhu K, Chu Y, Ding W, Zhu J, Lin Z, Zhong L, Wang J, Yue P, Turkson J, Liu P, Wang Y, Zhang X. Inhibition of STAT3-ferroptosis negative regulatory axis suppresses tumor growth and alleviates chemoresistance in gastric cancer. Redox Biol. 2022;52:102317. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 369] [Article Influence: 92.3] [Reference Citation Analysis (0)] |
| 12. | Lefler JE, MarElia-Bennett CB, Thies KA, Hildreth BE 3rd, Sharma SM, Pitarresi JR, Han L, Everett C, Koivisto C, Cuitino MC, Timmers CD, O'Quinn E, Parrish M, Romeo MJ, Linke AJ, Hobbs GA, Leone G, Guttridge DC, Zimmers TA, Lesinski GB, Ostrowski MC. STAT3 in tumor fibroblasts promotes an immunosuppressive microenvironment in pancreatic cancer. Life Sci Alliance. 2022;5:e202201460. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 37] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 13. | Gowrishankar K, Gunatilake D, Gallagher SJ, Tiffen J, Rizos H, Hersey P. Inducible but not constitutive expression of PD-L1 in human melanoma cells is dependent on activation of NF-κB. PLoS One. 2015;10:e0123410. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 149] [Cited by in RCA: 192] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 14. | van Zandwijk N, Pavlakis N, Kao SC, Linton A, Boyer MJ, Clarke S, Huynh Y, Chrzanowska A, Fulham MJ, Bailey DL, Cooper WA, Kritharides L, Ridley L, Pattison ST, MacDiarmid J, Brahmbhatt H, Reid G. Safety and activity of microRNA-loaded minicells in patients with recurrent malignant pleural mesothelioma: a first-in-man, phase 1, open-label, dose-escalation study. Lancet Oncol. 2017;18:1386-1396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 352] [Cited by in RCA: 552] [Article Influence: 61.3] [Reference Citation Analysis (0)] |