Published online Mar 15, 2026. doi: 10.4251/wjgo.v18.i3.113851
Revised: November 18, 2025
Accepted: January 4, 2026
Published online: March 15, 2026
Processing time: 189 Days and 0.8 Hours
Esophageal choristoma is an exceptionally rare congenital non-malignant mass which may present with a range of symptoms and severity according to size, growth and location.
In this article, we describe the case of a 12-day-old patient with an esophageal choristoma. The patient presented to the hospital with intermittent episodes of protrusion of unidentified tissue through the mouth, along with apneic episodes and failure to thrive. The mass was subsequently identified as originating from the upper esophagus. Given the mass size and the severity of symptoms, a de
Endoscopic mucosal resection is feasible and curative in neonatal esophageal symptomatic masses.
Core Tip: Esophageal choristoma is an extremely rare congenital anomaly in neonates. This case report presents the clinical features, challenging yet successful diagnosis, and definitive treatment of a large esophageal choristoma in a 12-day-old infant. Notably, it describes the first documented case of complete endoscopic mucosal resection performed in a neonate using a slim endoscope, demonstrating endoscopic mucosal resection as a safe, effective, and minimally invasive curative option for pediatric esophageal masses.
- Citation: Giz N, Epshtein J, Savin B, Meir K, Slae M. Presentation and endoscopic resection technique of neonatal esophageal choristoma: A case report. World J Gastrointest Oncol 2026; 18(3): 113851
- URL: https://www.wjgnet.com/1948-5204/full/v18/i3/113851.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i3.113851
Choristoma is defined as an uncommon lesion composed of histologically normal tissue derived from germ cell layers that develops in an anatomically abnormal location. In the gastrointestinal tract, choristomas often consist of an abnormal mixture of cartilage and other mesenchymal tissue components in a polypoid (protruding) structure[1]. Choristoma can be found in different locations of the gastrointestinal tract, the duodenal bulb is the most frequent site, but it can also be found in the large intestine, rectum, anus and the esophagus[2]. Case reports indicate that benign esophageal masses, including choristomas, are typically symptomatic[3]. No data currently exist regarding the incidence or prevalence of esophageal choristomas, either in the general population or in neonates.
Overall, esophageal tumours in neonates are uncommon and can pose significant diagnostic and therapeutic challenges, particularly due to their potential to obstruct the airway and disrupt feeding. The differential diagnosis of esophageal benign masses in neonates and infants includes hemangioma, duplication cyst, leiomyoma, esophageal granular cell tumour and on rare occasions, esophageal choristoma. Malignant tumours include neuroblastoma and rhabdomyosarcoma. The management of asymptomatic, benign esophageal masses often involves a decision between conservative observation and proactive resection. However, in the presence of symptoms or suspicion of malignancy, removal is generally indicated. Two therapeutic modalities exist for such cases: Surgical excision, and endoscopic removal, a less common alternative. Endoscopic techniques have advanced significantly over the years, enabling precise excision with minimal invasiveness in both pediatric and adult patients, as demonstrated in published case reports and case series[4]. Herein, we present the case of the youngest reported neonate with a large polypoid esophageal choristoma who underwent successful endoscopic removal.
A 12-day-old full-term female infant presented with repeated episodes of choking and cyanosis.
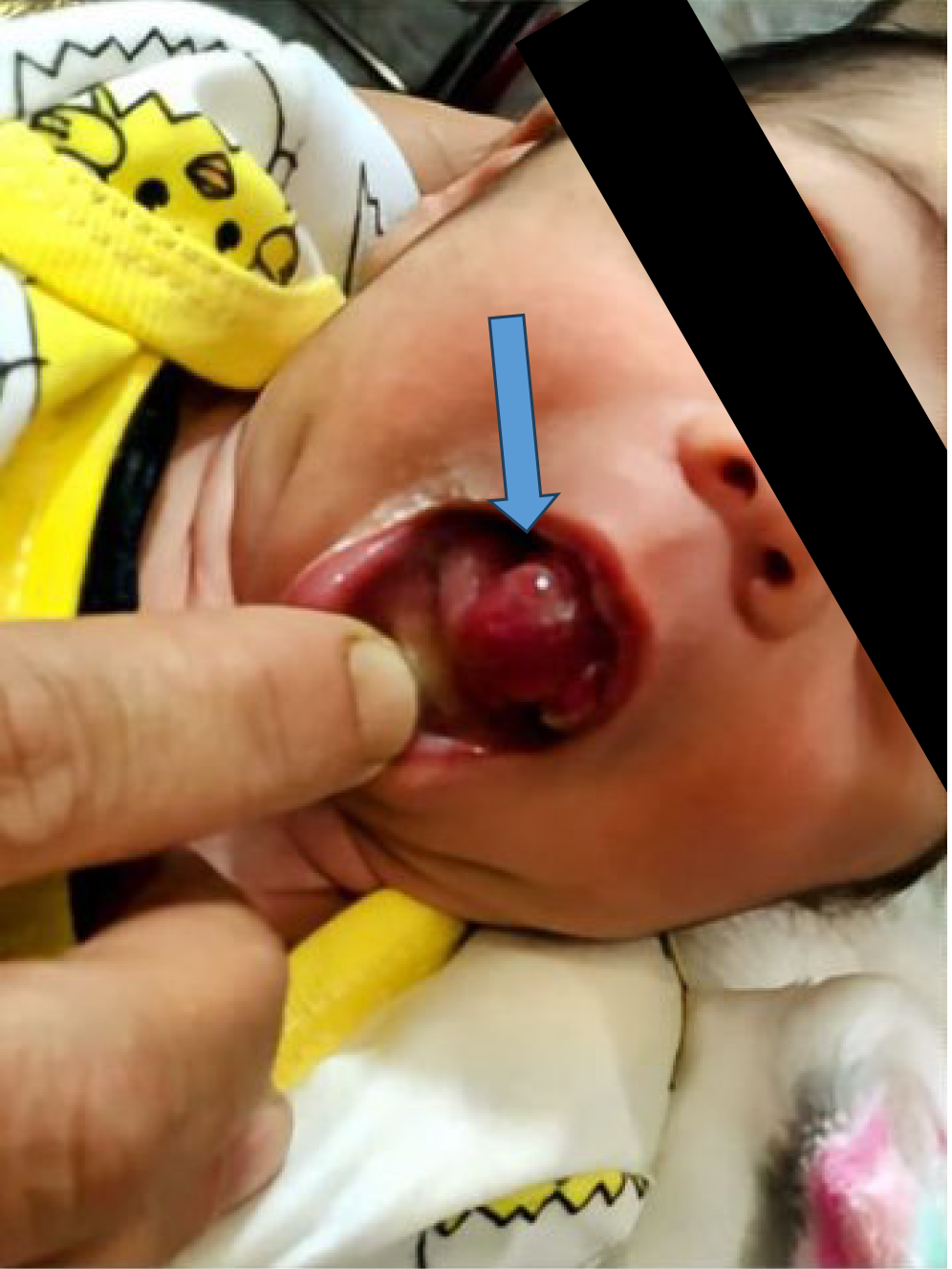
Her parents reported intermittent protrusion of an unidentified tissue from her mouth. On admission, the infant’s weight was unchanged from birth (3.05 kg). Initial assessments by oral and maxillofacial surgery specialists did not reveal a definitive cause, so the infant was admitted to the pediatric ward for monitoring and further evaluation. Cardiac investigations were normal. After two stable days symptoms reappeared, vomiting, expulsion of tissue from the mouth (Figure 1), choking, and cyanosis with oxygen saturation dropping to 70%, which spontaneously resolved. Additional similar episodes were reported, prompting transfer to the pediatric intensive care unit for closer monitoring.
Pregnancy and delivery were unremarkable, and there was no history of polyhydramnios.
The parents are not related, and there are no genetic diseases in the family.
Both the physical examination and the vital signs were within normal limits.
Laboratory workup, including complete blood cell count, glucose, electrolytes, kidney and liver function, calcium, magnesium, phosphorus, albumin, blood gases, and coagulation tests, was normal. C-reactive protein, ammonia, and lactate levels were also within normal limits.
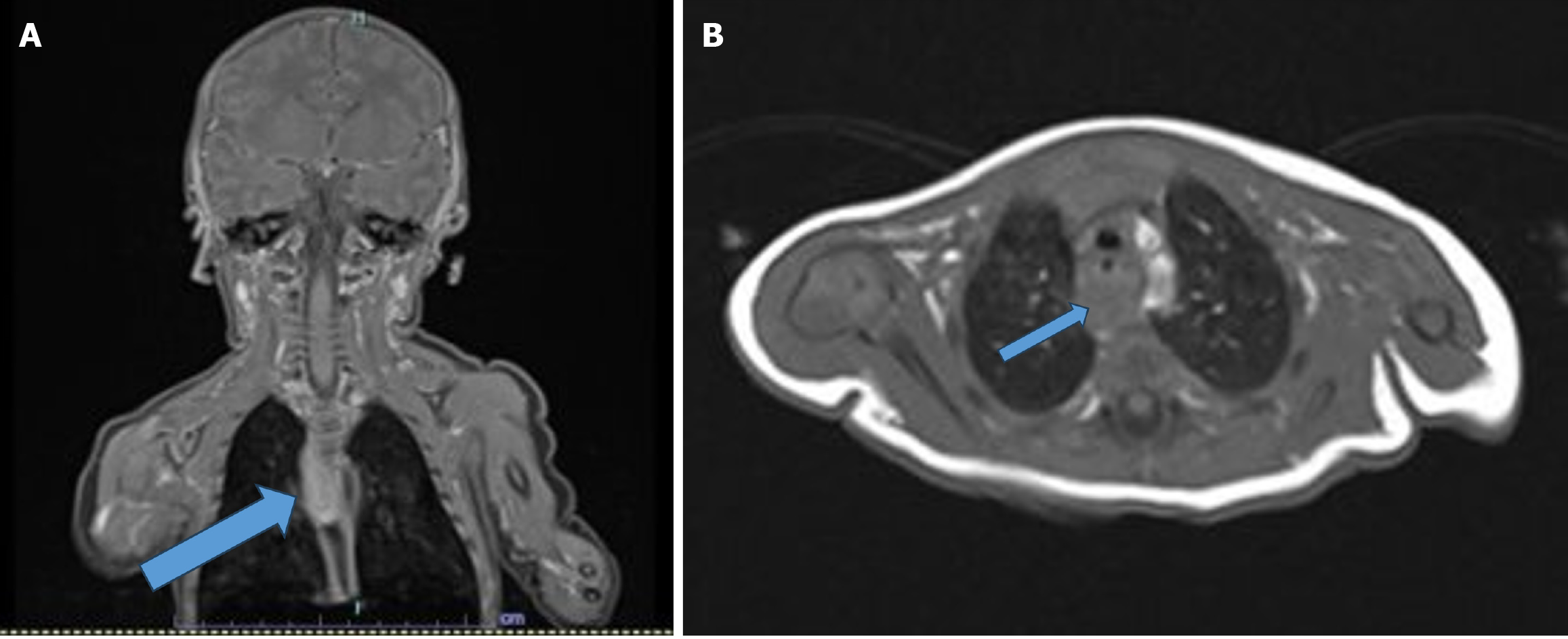
A fiber-optic airway examination showed no abnormalities. However, a neck magnetic resonance imaging, while normal in the cervical region, raised suspicion for a mass in the posterior upper mediastinum (Figure 2).
Subsequent endoscopy revealed a pedunculated mass in the esophagus, likely arising from the upper esophagus or parapharyngeal region.
At 19 days of age, the patient underwent therapeutic endoscopy under general anesthesia. Due to the narrow caliber of an infant’s esophagus, a standard gastroscope (8.9 mm diameter) could not be used. Instead, a slim endoscope (5.9 mm diameter) was required. This smaller scope is more challenging to maneuver, features a narrower working channel, necessitates specialized instruments compatible with its size, and offers a more limited range of endoscopic interventions.
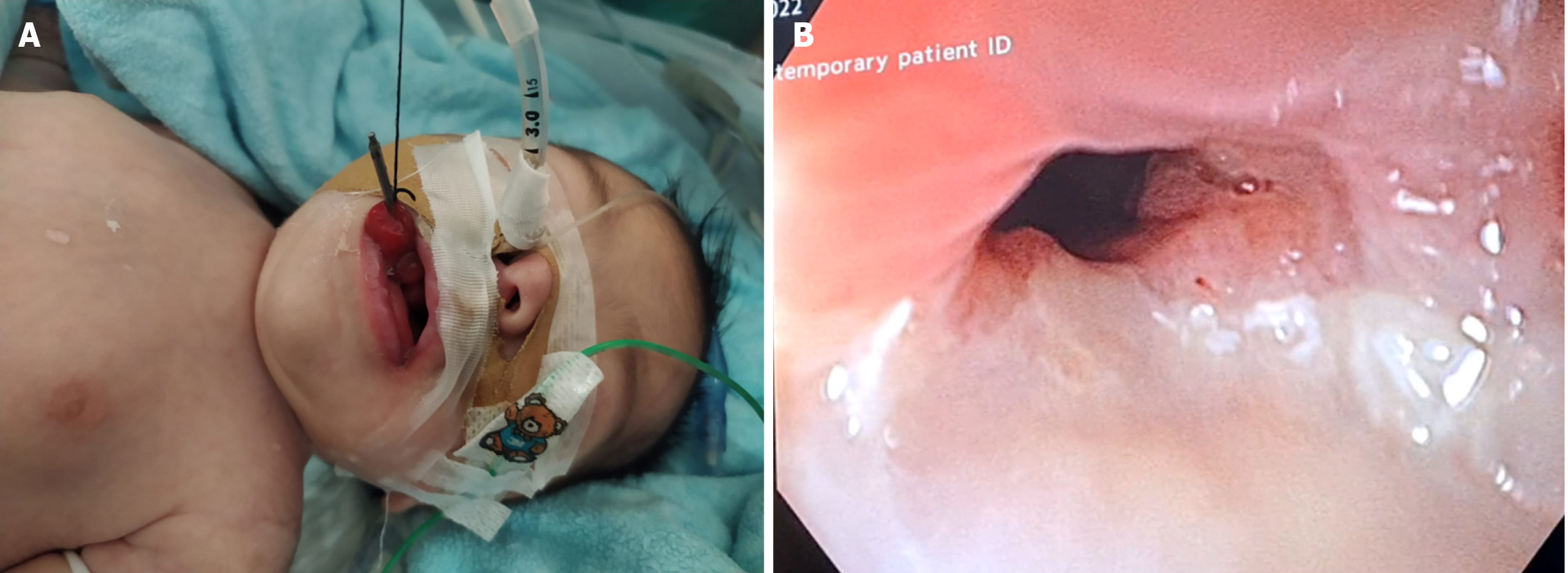
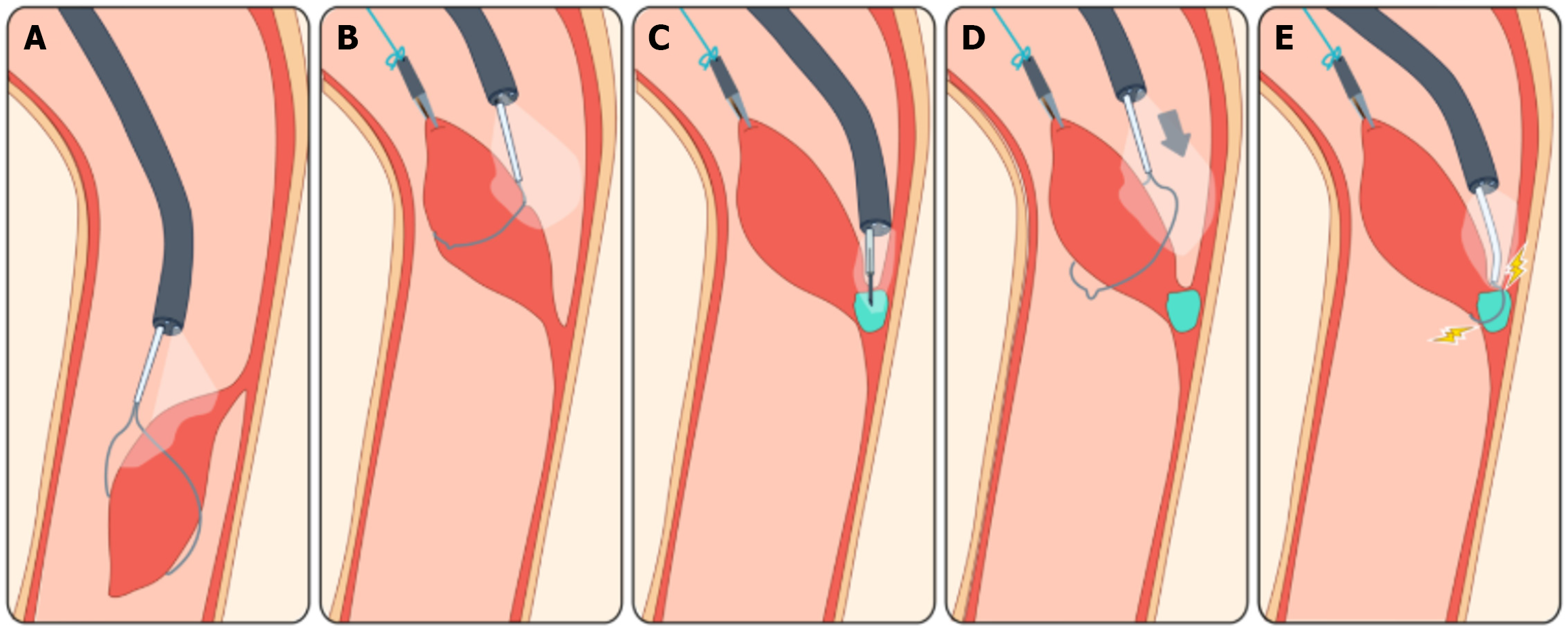
First, a nasal endotracheal tube was inserted. After that, the slim scope was advanced down the esophagus alongside the longitudinal aspect of the mass which was lying in caudal orientation. A slim-scope size snare was used to flip the mass cephalad. Next, we attached, under vision, a hemoclip to the tip of the mass. The hemoclip secured a stitch wire which was used to exert traction on the mass, directing it upwards, and out the oral opening (Figure 3). Following, using a slim-scope sized injection needle, we injected the lesion’s base with an adrenaline-saline mixture (1 mg adrenaline in 10 mL normal saline, total of 2 mL) in order to raise the mass’s base, increase resectability, decrease risk of perforation and provide hemostasis. Then, we inserted a slim-scope size snare through the slim-scope and once the snare came out of the far end of the scope’s working channel, we slid the snare over the stitch-wire, the clip and the body of the lesion, until it reached the base of the mass. We used hot polypectomy technique to resect the mass (the settings used were ERBE viod 300 endocut Q, effect 2, cut duration 1, cut interval 6, upmax 770Vp). The mass was resected completely, with no imme
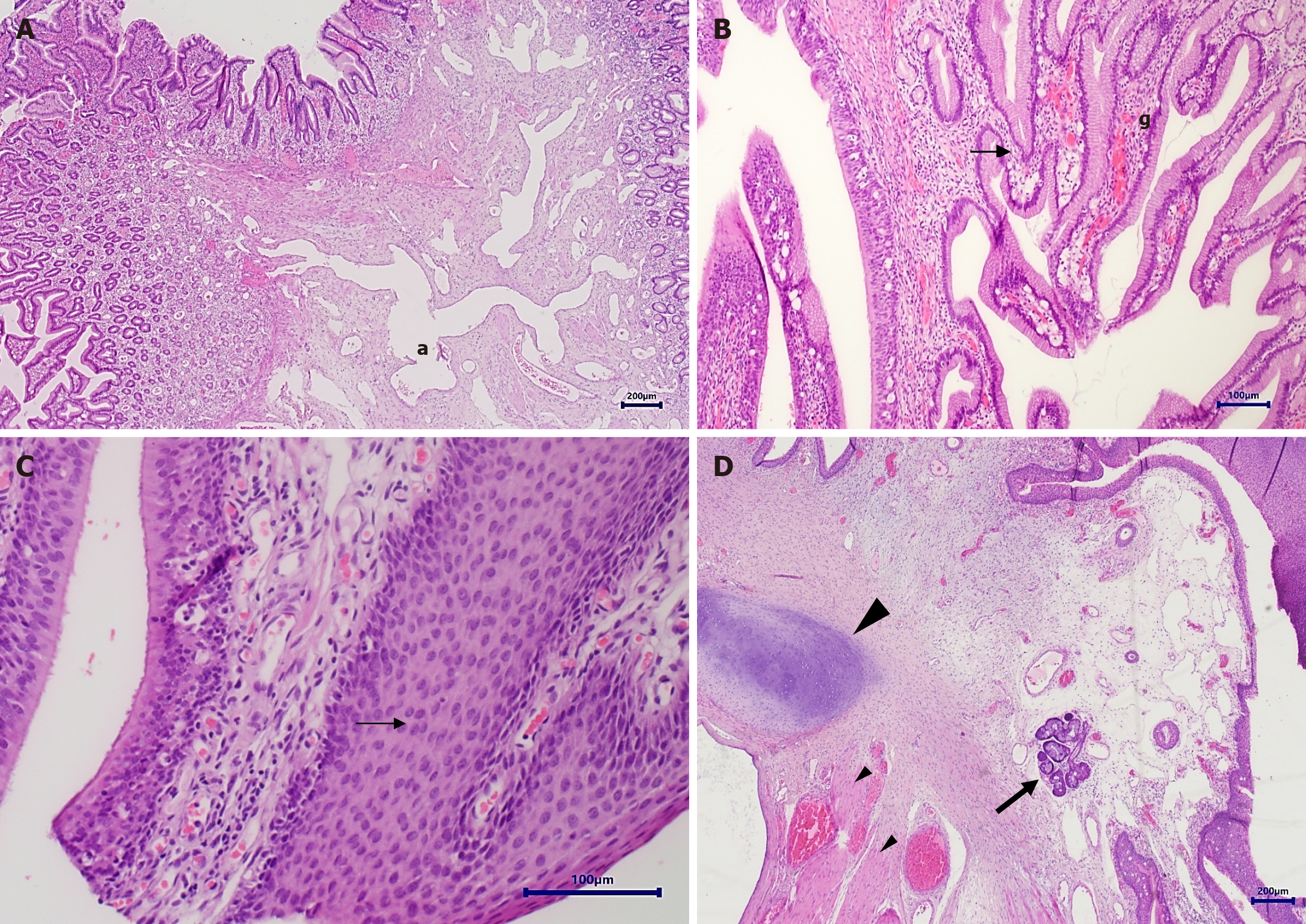
Pathological analysis (Figure 5) demonstrated a 3 cm pedunculated polypoid mass lined by alternating esophageal and gastric mucosa. Foci of layered smooth muscle resembling muscularis propria with intervening ganglion cells were seen. In other areas salivary type glands were observed. In addition, foci of respiratory type epithelium and a cartilage island could be discerned. The findings were consistent with choristoma with tracheobronchial remnants. Importantly, the mass base margins were defined clear (consisting of esophageal mucosa and submucosa). Follow-up at two months showed the infant was thriving, with a weight increase from 3.06 kg to 5 kg and a normal gag reflex. At five-months follow-up, the mother reported occasional choking but noted the child was eating without issues and gaining weight nicely (weight for age increased from the 3rd to the 50th percentile). Examination showed a normal throat and that the gag reflex remained intact. Follow up at the age of 13 months showed normal eating and weight.
The case of a 12-day-old infant with esophageal choristoma highlights a rare and complex pathology posing diagnostic and therapeutic challenges. Choristomas, benign congenital lesions composed of heterotopic tissue, are exceedingly rare in the esophagus, with only few reported cases in the literature (Table 1)[3,5-14]. In children, esophageal masses present with varied symptoms, ranging from asymptomatic babies to choking, cyanosis, and failure to thrive[15]. As in this case, it is difficult to reach a diagnosis, given nonspecific symptoms and rarity of the cause. In our patient, several examinations reported no abnormal findings.
| Ref. | Cases (n) | Age | Sex | Initial symptoms | Location | Histopathology findings | Treatment | Further treatment | Follow-up |
| Zhang et al[3], 2017 | 1 | 64 years | Female | Progressive foreign body sensation | Cervical esophagus | Proliferating hyaline cartilage cells | Transcervical esophagectomy | None | Uneventful recovery |
| Alobid et al[5], 2007 | 1 | 76 years | Male | Sudden dyspnea | Esophagus | Fibrovascular polyp | Endoscopic removal | None | Not specified |
| Marques Dos Santos et al[6], 2024 | 1 | 3 years | Female | Persistent food impaction since 9 months old | Esophagus | Chondroepithelial choristoma | Surgical correction | None | Not specified |
| Goldman and Ban[7], 1972 | 1 | 16 hours | Female | Esophageal atresia | Esophagus | Tracheobronchial remnants | Surgical management | None | Not specified |
| Beckerman et al[8], 1980 | 1 | 11 months | None | Dysphagia and continuous drooling | Esophagus | Fibromuscular hamartoma | Tracheostomy and feeding gastrostomy after surgery | None | 16 months management |
| Halfhide et al[9], 1995 | 1 | 41 years | Male | Progressive dysphagia | Mid esophagus | Hamartoma | Surgical removal | None | Not specified |
| Coury et al[10], 2010 | 1 | 6 weeks | Female | Apnea and bradycardia | Esophagus | Hamartoma | Surgical resection | None | Not specified |
| Zhao and Zhu[11], 2022 | 1 | 46 years | Male | Incidental finding during gastroscopy | Left posterior wall of esophagus | Neuromuscular choristoma | Observation | None | No significant changes over 3 years |
| Tubino et al[12], 1982 | 1 | Not specified | Not specified | Esophageal stenosis | Esophagus | Tracheobronchial remnants | Surgical correction | None | Not specified |
| Lakshman et al[13], 2023 | 1 | 46 years | Female | Chest pain | Middle third of esophagus | Pseudostratified ciliated columnar epithelium with respiratory mucinous glands and skeletal muscle | Surgical removal | None | None |
| Deiraniya[14], 1974 | 1 | 20 months | Male | Esophageal stenosis | Esophagus | Tracheobronchial remnants | Surgical treatment | None | Not specified |
Surgical resection remains the definitive treatment for esophageal masses. However, endoscopic resection can potentially change management by avoiding surgical intervention in pediatric patients with esophageal masses. Endoscopic esophageal tumour resection in children has been reported once, in an adolescent[4]. In our case, therapeutic endoscopy was performed using a snare technique to excise the lesion. The procedure was facilitated by the injection of saline-adrenaline mixture to elevate the mass and minimize bleeding. The pathological confirmation of complete resection and the absence of post-procedure complications, such as esophageal perforation or significant haemorrhage, emphasize the efficacy and safety of endoscopic management when executed with appropriate precautions. This case is unique since it is the first ever documented case of endoscopic mucosal resection of an esophageal mass performed on a neonate, using a slim scope with endoscopic mucosal resection technique.
This approach stands in contrast to open surgical resection, which while effective, carries higher risks of morbidity, prolonged recovery and scarring. For instance, a systematic review analyzing outcomes after esophageal atresia repair reported high complication rates, including esophageal dysmotility in 78% of patients, gastroesophageal reflux disease in 43%, dysphagia in 44%, anastomotic leak in 19%, anastomotic stricture in 26%, and esophagitis in 47%[16]. The complete resolution of symptoms and rapid recovery of this patient support the growing preference for endoscopic methods in managing benign esophageal lesions.
Postoperatively, the infant developed pneumonia and was treated successfully with antibiotics. This complication can potentially result from either aspiration or anesthesia-related factors. Follow-up at two, five, and thirteen months demonstrated sustained improvement, marked by significant weight gain and resolution of major symptoms. These outcomes align with findings in the existing literature, where endoscopic resection of esophageal lesions is associated with excellent long-term prognosis; however, previously reported cases have been limited to adults[10], whereas this case involves a young pediatric patient. The American Society for Gastrointestinal Endoscopy supports the use of endoscopic resection of various benign and early malignant esophageal lesions, emphasizing its safety and efficacy[17]. Endoscopic mucosal resection has been performed in children, however only in the colon. This report carries limitations, including the limited generalizability of a single-case report and the lack of long-term follow-up. Nonetheless, it aims to raise clinical awareness of this rare condition and emphasize the potential of endoscopic management as a safe and effective treatment option in selected pediatric patients.
In summary, this case illustrates the importance of recognizing esophageal tumours in the differential diagnosis in neonates with unexplained choking and feeding difficulties. It also highlights the critical role of endoscopic techniques in achieving diagnosis and curative outcomes while minimizing procedural risks. Since it is likely that studies comparing surgical to endoscopic resection of pediatric esophageal masses will not be performed due to their rarity, case reports such as this case are expected to be the main literature source guiding future directions in managing these cases. We provide a detailed delineation of the procedure, to aid in planning intervention in similar cases. The choristoma’s benign nature in our case was confirmed histologically, showing no risk of recurrence as removed with clear margins. This outcome reinforces the need for a multidisciplinary approach that integrates advanced imaging, skilled endoscopic techniques, experienced pathologist and diligent post-procedure care.
| 1. | Gurzu S, Burlacu D, Jung I. Nonpolypous Hamartomas of the Gastrointestinal Tract: An Updated Review on Classification, Denominations, and Clinical Management. J Oncol. 2022;2022:6983460. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 2. | Johncilla M, Yantiss RK. Malformations, choristomas, and hamartomas of the gastrointestinal tract and pancreas. Semin Diagn Pathol. 2019;36:24-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 3. | Zhang C, Xu J, Wang Z, He Z, Yang H, Hu J. Chondromatous hamartoma of cervical esophagus: a case report and literature review. J Thorac Dis. 2017;9:E236-E244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 4. | Dogom SA, Thongpiya J, Elmassry M, Feist M, Sharma M, Rateb G. Successful endoscopic submucosal dissection of an esophageal granular cell tumor in a pediatric patient: A case report and a therapeutic insight. JPGN Rep. 2024;5:384-388. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 5. | Alobid I, Vilaseca I, Fernández J, Bordas JM. Giant fibrovascular polyp of the esophagus causing sudden dyspnea: endoscopic treatment. Laryngoscope. 2007;117:944-945. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 6. | Marques Dos Santos F, Afonso I, Soares E, Carneiro R. Chondroepithelial Choristoma: A Rare Cause of Congenital Esophageal Stenosis. GE Port J Gastroenterol. 2024;31:217-220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 7. | Goldman RL, Ban JL. Chondroepithelial choristoma (tracheobronchial rest) of the esophagus associated with esophageal atresia. Report of an unusual case. J Thorac Cardiovasc Surg. 1972;63:318-321. [PubMed] |
| 8. | Beckerman RC, Taussig LM, Froede RC, Coulthard SW, Firor H, Tonkin I. Fibromuscular hamartoma of the esophagus in an infant. Am J Dis Child. 1980;134:153-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 9. | Halfhide BC, Ginai AZ, Spoelstra HA, Dees J, Vuzevski VD. Case report: a hamartoma presenting as a giant oesophageal polyp. Br J Radiol. 1995;68:85-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 10. | Coury J, Steinfeld J, Zwillenberg D, Zwillenberg S. Esophageal hamartoma as an unusual cause of neonatal apnea and bradycardia. Ear Nose Throat J. 2010;89:E7-E11. [PubMed] |
| 11. | Zhao W, Zhu X. A case of esophageal neuromuscular choristoma. BMC Gastroenterol. 2022;22:180. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 12. | Tubino P, Marouelli LF, Alves E, Araújo RC. Choristoma: esophageal stenosis, due to tracheobronchial remnants. Z Kinderchir. 1982;35:14-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 13. | Lakshman A, D'Cruze L, Balasubramaniam A, M S, T P. A Unique Presentation of a Thoracic Conundrum: Tracheobronchial Rests in the Esophagus. Cureus. 2023;15:e39096. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 14. | Deiraniya AK. Congenital oesophageal stenosis due to tracheobronchial remnants. Thorax. 1974;29:720-725. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 23] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 15. | Chan CY, Jadcherla SR. Clinical Perspectives on Esophageal Disorders in Infants. ASHA Lead. 2012;21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 16. | Comella A, Tan Tanny SP, Hutson JM, Omari TI, Teague WJ, Nataraja RM, King SK. Esophageal morbidity in patients following repair of esophageal atresia: A systematic review. J Pediatr Surg. 2021;56:1555-1563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 17. | Al-Haddad MA, Elhanafi SE, Forbes N, Thosani NC, Draganov PV, Othman MO, Ceppa EP, Kaul V, Feely MM, Sahin I, Ruan Y, Sadeghirad B, Morgan RL, Buxbaum JL, Calderwood AH, Chalhoub JM, Coelho-Prabhu N, Desai M, Fujii-Lau LL, Kohli DR, Kwon RS, Machicado JD, Marya NB, Pawa S, Ruan W, Sheth SG, Storm AC, Thiruvengadam NR, Qumseya BJ; (ASGE Standards of Practice Committee Chair). American Society for Gastrointestinal Endoscopy guideline on endoscopic submucosal dissection for the management of early esophageal and gastric cancers: methodology and review of evidence. Gastrointest Endosc. 2023;98:285-305.e38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 55] [Article Influence: 18.3] [Reference Citation Analysis (4)] |