Published online Mar 15, 2026. doi: 10.4251/wjgo.v18.i3.113757
Revised: November 28, 2025
Accepted: January 4, 2026
Published online: March 15, 2026
Processing time: 191 Days and 9.3 Hours
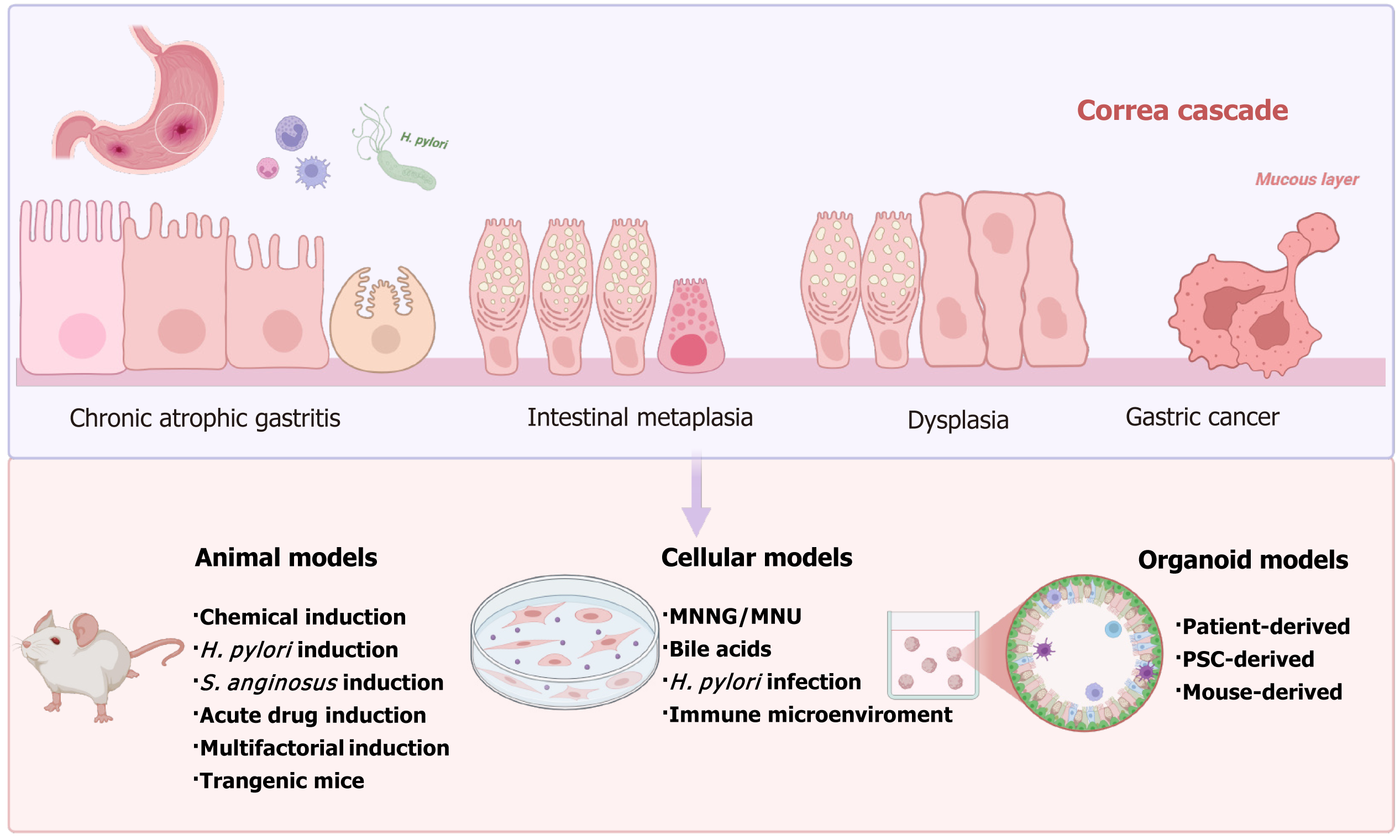
Precancerous lesions of gastric cancer (PLGC), including chronic atrophic gastritis, intestinal metaplasia, and dysplasia, constitute key transitional stages in the Correa cascade toward gastric cancer. Understanding these stages is essential for early detection, prevention, and therapeutic intervention.
To provide a comprehensive and critical overview of current animal, cellular, and organoid models used in PLGC research, emphasizing their applications, translational relevance, and emerging technologies.
A scoping systematic review was performed according to the PRISMA-ScR fra
This study delineates the modeling strategies for PLGC across various platforms. Animal models, including che
Integrated PLGC models provide a robust framework for elucidating the cellular and molecular basis of early gastric tumorigenesis. Their continued evolution will enhance biomarker discovery, immunologic investigation, and therapeutic development.
Core Tip: Precancerous lesions of gastric cancer (GC), including atrophic gastritis, intestinal metaplasia, and dysplasia, represent the pivotal transition in the Correa cascade. Accurate modeling of these lesions is crucial for dissecting early gastric tumorigenesis and identifying chemopreventive strategies. This scoping study systematically summarizes current precancerous lesions of GC models, including chemical- and infection-induced animal systems, transformed gastric epithelial cell lines, and organoid platforms derived from stem cells. We highlight their advantages, limitations, and recent advances such as genome editing, single-cell technologies, and immune co-culture. These insights provide a roadmap for selecting appropriate experimental systems and accelerating translational research in early GC prevention and therapy.
- Citation: Zhou LJ, Hao XY, Wang C, Ren NN, Wang YG. Comprehensive modeling of the Correa cascade in precancerous lesions of gastric cancer: Leveraging animal, cellular, and organoid systems for translational insights. World J Gastrointest Oncol 2026; 18(3): 113757
- URL: https://www.wjgnet.com/1948-5204/full/v18/i3/113757.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i3.113757
Gastric cancer (GC) remains one of the most common digestive malignancies worldwide, ranking fifth in incidence and mortality[1]. Intestinal-type gastric adenocarcinoma, strongly associated with Helicobacter pylori (H. pylori) infection, follows the well-recognized Correa cascade described in 1992: Superficial gastritis, chronic atrophic gastritis (CAG), intestinal metaplasia (IM), dysplasia (Dys), carcinoma[2]. Clinically, CAG, IM, and Dys are grouped as precancerous lesions of GC (PLGC); their severity correlates with subsequent GC risk[3]. Because PLGC represents a critical window between chronic gastritis and malignancy, the “golden turning point”, intervention at this stage may reduce GC incidence[4].
The etiology and pathogenesis of GC have not been fully elucidated. The establishment of a simple and stable gastric epithelial “inflammation-cancer” transformation animal model is the premise and basis for studying the mechanism of GC and conducting intervention research on PLGC. The most commonly used mouse models of PLGC involve chemical carcinogens such as N-methyl-N’-nitro-N-nitrosoguanidine (MNNG) and N-nitroso-N-methylurea (MNU), as well as H. pylori infection to induce lesions; however, these approaches have shortcomings, such as poor stability and long modeling time. In recent years, the rapid development of gene editing technology has provided a new perspective for the development of PLGC mouse models. PLGC cell model is used to simulate the process of GC by culturing PLGC cells in vitro. It is also commonly used in acute and chronic models induced by chemical induction and H. pylori infection of gastric epithelial cell-1 (GES-1) cells to study the mechanisms of cell proliferation, gene mutation and immune response. Although this model is suitable for drug screening and mechanistic studies, it cannot fully reproduce the complex mic
The organoid model is based on the three-dimensional (3D) culture of gastric epithelial structure, which can simulate the function and microenvironment of gastric tissue, closer to human tissue, and has high clinical relevance. However, it is still difficult to fully reproduce the immune and vascular microenvironment of GC. There are great differences among PLGC models, but there are no relevant reviews. In this paper, the common animal, cell and organoid models of PLGC were classified and summarized, the modeling methods, modeling time, pathological changes, observation methods of models were compared and analyzed, and the relevant points of model evaluation were commented, in order to provide reference for PLGC research.
This study was conducted following the PRISMA-ScR checklist. Although the protocol was not prospectively registered, the methodological approach was predefined. We systematically searched PubMed, EMBASE, and Web of Science from January 2000 to June 2025 using a combination of Medical Subject Headings and free-text keywords related to PLGC, the Correa cascade, atrophic gastritis, IM, Dys, animal models, organoids, and cell culture. Boolean operators were applied, for example: (“precancerous lesions” OR “atrophic gastritis” OR “intestinal metaplasia” OR “dysplasia”) AND (“gastric” OR “stomach”) AND (“animal model” OR “rat” OR “mouse” OR “Mongolian gerbils” OR “organoid” OR “cell line”). Only peer-reviewed original studies and reviews were included, and the reference lists were screened for additional relevant articles.
Studies were included if they reported experimental models (animal, cellular, or organoid) of PLGC, described induction methods, histological outcomes, or mechanistic insights related to the Correa cascade. Case reports, clinical-only studies without modeling experiments, non-gastric models, studies focused exclusively on advanced gastric car
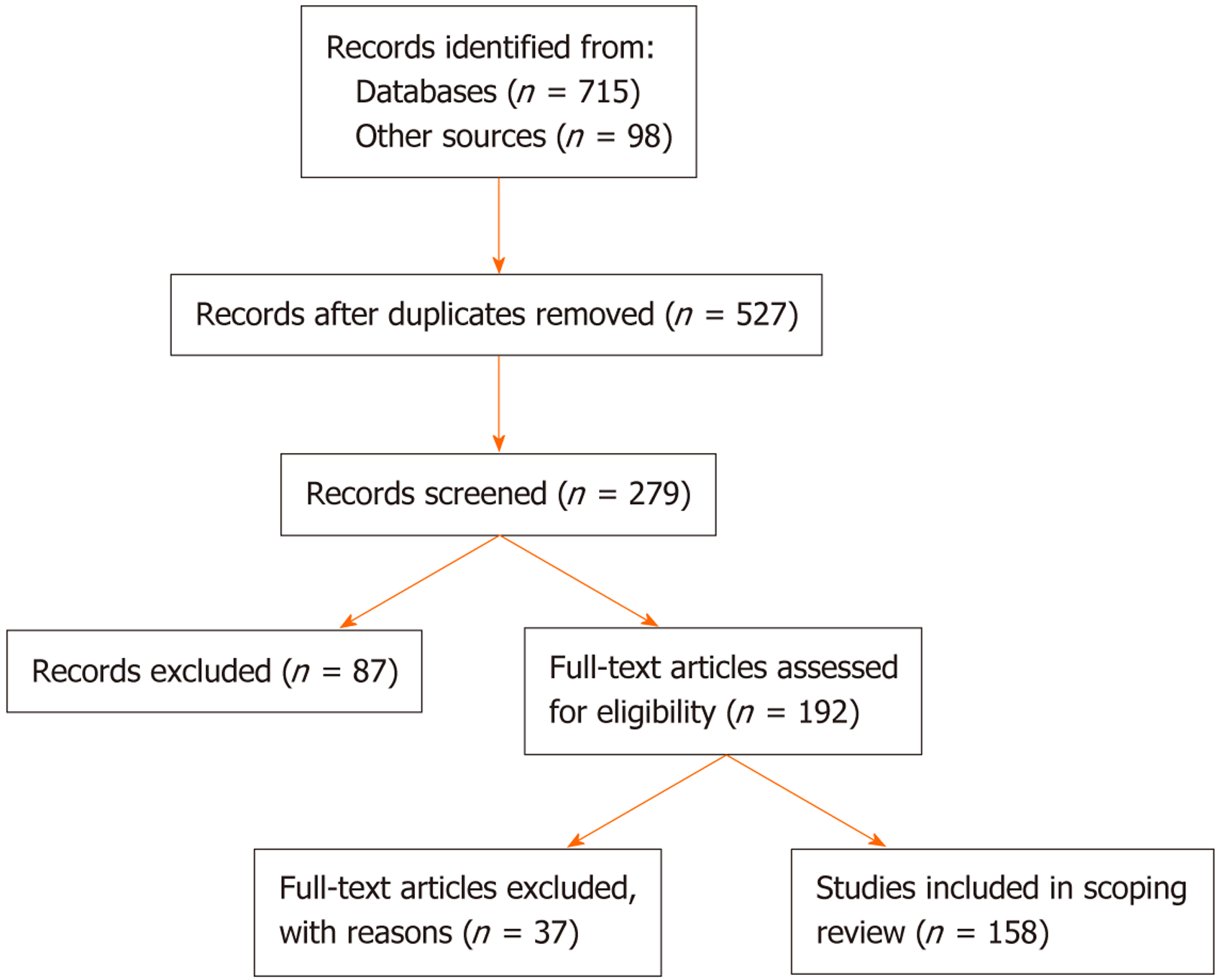
Two reviewers independently screened titles and abstracts, followed by full-text assessment for eligibility, with disagreements resolved by consensus. Data extracted from each included study comprised the type of model, induction methods (chemical, bacterial, dietary, or genetic), duration of observation, histological endpoints (atrophy, metaplasia, Dys, carcinoma), and reported strengths and limitations. The results were narratively synthesized and structured according to model category, with summary tables created to facilitate comparison. The literature selection process is summarized in a PRISMA-ScR flow diagram, showing the number of records identified, screened, excluded, and finally included in the systematic review. The literature screening and selection process and representative experimental models for each stage of the Correa cascade are summarized in Figures 1 and 2.
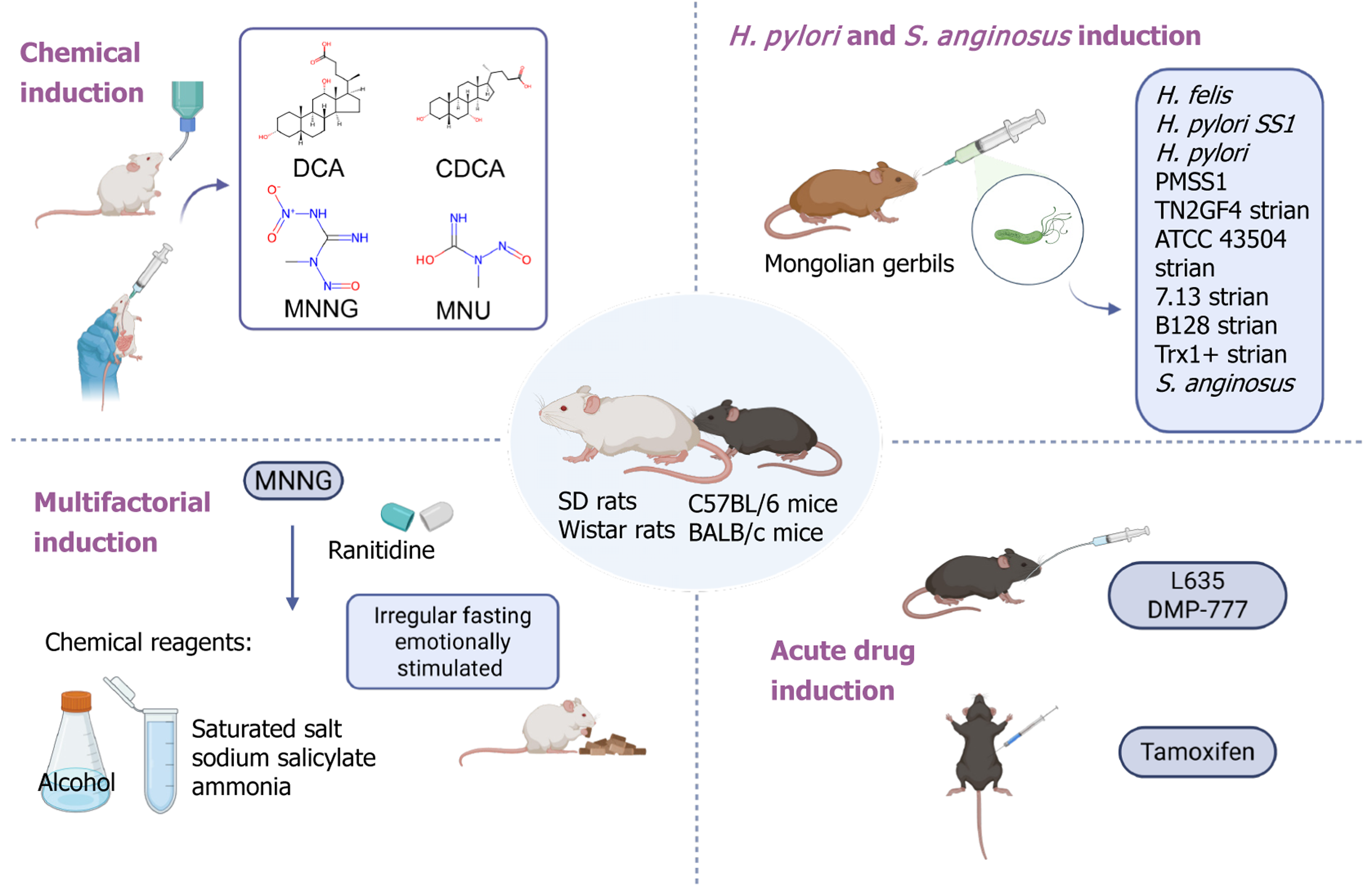
Chemical carcinogens or dietary induction models: MNNG induction model, long-term low-dose exposure to N-nit
Wistar and Sprague-Dawley rats are commonly used due to their docility, resistance to infections, low spontaneous tumor incidence, and suitability for oral administration[8]. IM and Dys can be induced by administering 200 μg/mL MNNG in drinking water for 16 weeks, with progression to gastric adenocarcinoma by 40 weeks[9,10], Alternatively, gavage with 800 mg/L MNNG for 10 weeks can induce PLGC, with progression to carcinoma observed by 34 weeks[11]. However, high-dose gavage increases mortality and reduces model success rate. In mice, free drinking of 150 μg/mL MNNG for 6 weeks has been sufficient to induce PLGC[12,13]. Because PLGC is multifactorial, compound models com
MNNG concentration and administration route are critical. Free drinking reflects natural exposure, while gavage enhances mutagenicity and reduces off-target toxicity[16]. Cai et al[17] demonstrated that 200 μg/mL MNNG for 16 weeks induced IM and Dys, and Wang et al[18] observed mucosal atrophy with focal atypia after 40 weeks of free drinking 100 μg/mL MNNG. Higher doses via gavage (250-50 mg/kg) tend to induce forestomach lesions, whereas free intake targets gastric glandular tissue. Thus, model success depends on dose, duration, and delivery. Free drinking of 200 μg/mL MNNG for 16 weeks induces PLGC efficiently but only partially mimics human pathogenesis due to lack of sustained immune infiltration and stromal remodeling.
MNU induction model: The utility of another N-nitroso compound, MNU as a gastric carcinogen was tested in mice and found to be more effective. Researchers have verified that the MNU-induced PLGC model in mice can be successfully established by short-term gavage of MNU solution (120 ppm). The experimental approach consists of enabling the mice to freely drink the MNU solution for 8 weeks, after which the mice exhibit IM and Dys in the gastric mucosa[19]. When the mice are subjected to the treatment for 30 weeks, GC can be induced[20]. However, the MNU model of gastric carcinogenesis does not proceed through a classical atrophy-metaplasia-Dys sequence, and the time, location, quantity and size of cancer are uncertain, with large genetic heterogeneity, which is the drawback of the MNU mouse model. Therefore, MNU is commonly used in combination with H. pylori infection and a high-salt diet to establish animal models of PLGC and GC[21].
Deoxycholic acid (DCA) and chenodeoxycholic acid (CDCA) induction model: During bile reflux, phospholipase A in pancreatic fluid converts lecithin into lysolecithin, which disrupts the phospholipid bilayer of GESs, increasing per
Acute drug- and H. pylori-induced spasmolytic polypeptide-expressing metaplasia (SPEM) model: SPEM, which is considered a potential neoplastic precursor of IM[27], is characterized by spasmolytic polypeptide (trefoil factor 2) or mucin6 (MUC6)/GSII expression in the basal region of the gland, comprising the chief cells after parietal cell loss[28,29]. DMP-777 is a protonophore that blocks acid secretion and induces rapid parietal cell loss, leading to SPEM after 10-14 days. These changes are reversible within 7-14 days after withdrawal. While these acute chemical injury models effi
High-dose tamoxifen also induces parietal cell loss by acting as a proton carrier. Daily administration of 5 mg emulsified tamoxifen for 3 days reduces parietal cell numbers by 90%, with SPEM emerging within 3 days in mice treated with 250 mg/kg. These lesions are similarly reversible within 2-3 weeks after drug withdrawal[31,32]. Tamoxifen also ablates tuft cells, which play key roles in early carcinogenesis. In gp130Y757F/Y757F mice, two tamoxifen injections at 16 weeks induced tuft cell loss, leading to spontaneous development of SPEM-associated intestinal-type adenomas as early as 4 weeks of age[33]. Recent studies have identified a novel mechanism by which H. pylori induces SPEM through the CagA-telomerase reverse transcriptase (TERT)-Wnt signaling axis. Importantly, this mechanism was elucidated using the CagA-positive Sydney strain 1 (PMSS1). In mice infected with PMSS1, H. pylori stabilizes TERT by suppressing synovial apoptosis inhibitor 1-mediated ubiquitination and degradation, thereby activating the Wnt/β-catenin pathway and driving SPEM development. Genetic deletion of TERT abolished these pathological changes, confirming that the CagA-TERT-Wnt axis is essential for H. pylori-induced metaplastic transformation[34].
High-salt and salting diet induced models: A high-salt and pickled diet is a high-risk factor for GC. High salt diet combined with H. pylori Sydney strain (HpSS1) infection resulted in higher rates of atrophy and epithelial proliferation in Mongolian gerbils[35], and high salt diet also increased the incidence of MNU-induced gastric tumors in C57BL/6 mice[36]. High salt intake alone does not induce GC, and it is more likely to cause cancer synergistically with other factors and increase the risk of GC.
Helicobacter infection model: H. pylori infection is a major etiological factor in gastric carcinogenesis, and various animal models have been developed to study its pathogenic mechanisms. Among them, Mongolian gerbils and C57BL/6 mice are the most widely used due to their reproducible pathological responses. Notably, disease severity and temporal progression in these models are strongly influenced by both bacterial strain virulence and host genetic background, underscoring the importance of strain-host interactions in PLGC modeling.
Watanabe et al[37] first established a Mongolian gerbil model of H. pylori-induced GC, with 37% of animals developing tumors resembling human intestinal-type cancer after 62 weeks of infection. Because C57BL/6 mice are resistant to most H. pylori strains, Helicobacter felis, a closely related species, has been used instead and can cause gastric metaplasia, Dys, and invasive cancer with prolonged infection[38,39]. Lee et al[40] then isolated the HpSS1 carrying CagA and VacA, which successfully colonized C57BL/6 mice, inducing chronic gastritis and atrophy but no carcinoma even after 2 years. Later, Arnold et al[41] introduced PMSS1, a stable mouse-adapted strain that induces metaplasia/SPEM, though with a lower colonization rate than HpSS1.
Effect of H. pylori infection in the mouse stomach: The pathogenic outcome in mice depends on the strain of H. pylori and the duration of infection. In C57BL/6 mice, HpSS1 infection causes lesions ranging from mild inflammation to atrophy and hyperplasia over 5 days to 9 months; hyperplasia and IM were observed at 3 and 18 months, respectively[42]. The more virulent PMSS1 strain accelerates this process, with metaplasia detected at 8 weeks[43] and inflammation by 3-6 months[44]. In BALB/c mice, HpSS1 induces chronic gastritis within 40 days to 8 weeks[45]. These results highlight the influence of host and bacterial factors. Notably, PMSS1 exhibits stronger pathogenicity with accelerated induction of metaplasia and inflammation, whereas HpSS1 favors long-term colonization with relatively milder pathological outcomes[46]. Recent studies also identified SlyD as a novel virulence factor that promotes IM via stabilization of tumor protein translationally-controlled 1 and activation of caudal-related homeobox transcription factor 2 (CDX2)[47].
Effect of H. pylori infection in the Mongolian gerbil: Mongolian gerbils are highly susceptible to H. pylori-induced gastric lesions, closely mimicking human disease progression. Long-term infection leads to gastritis, atrophic gastritis, IM, Dys, and adenocarcinoma[37,48]. Different H. pylori strains exhibit varying carcinogenic potentials. For instance, TN2GF4 strain induces IM and carcinoma after 18 months[49,50], ATCC 43504 strain leads to IM within 3-12 months[51], and strain 7.13 causes rapid progression from IM (7 weeks) to adenocarcinoma (2 months)[52]. These comparisons un
Streptococcus anginosus (S. anginosus) mouse model: Although H. pylori initiates gastric pathology, its influence wanes during later disease stages. Using metagenomic analysis, Fu et al[53] identified S. anginosus as a non-H. pylori species associated with gastric inflammation and tumorigenesis. In murine models, S. anginosus induced acute gastritis within two weeks, and 9-12 months infection led to chronic gastritis, mucinous metaplasia, and Dys in both conventional and germ-free C57BL/6 mice. Notably, co-infection with S. anginosus and MNU significantly accelerated gastric tumorigenesis compared to H. pylori alone.
Multi-factorial induction model: The single-factor modeling method has a single intervention and a long modeling time, which is not fully compatible with the view that atrophic gastritis and its precancerous lesions are complex diseases with multifactorial participation. Therefore, scholars have tried to superimpose other carcinogenic factors on top of MNU and MNNG for composite induction, in order to more closely resemble and mimic the human pathogenic process.
Controversy and exploration of MNU combined Helicobacter infection modeling: Several studies have explored the combined use of MNU and H. pylori infection in inducing gastric carcinogenesis in mice, but results remain inconsistent. Toyoda et al[36] administered 120 ppm MNU via alternate-week free drinking with a 10% NaCl high-salt diet, and a subset received additional H. pylori inoculation biweekly. After 40 weeks, all mice developed antral tumors, suggesting that H. pylori and/or a high-salt diet significantly enhance MNU-induced tumorigenesis, However, whether H. pylori alone synergizes with MNU remains debated.
For this controversy, Lee et al[54] demonstrated that mice co-treated with H. pylori and MNU had the highest GC incidence, significantly exceeding that of either treatment alone. Supporting this, another study reported that 240 ppm MNU combined with oral HpSS1 inoculation induced GC after 50 weeks[55]. Additionally, infection with more virulent strains such as PMSS1[56] and ATCC 43504[57] resulted in gastric mucosal atrophy, IM, and heterotopic hyperplasia. These findings confirm that MNU co-administered with H. pylori accelerates gastric disease progression and tumorigenesis.
MNU combined diet induction model: In terms of modeling of MNU solution combined with dietary factors, Liao et al[58] established a model using 300 ppm MNU in drinking water combined with an irregular feeding schedule (starvation-to-satiation cycles), leading to IM and Dys within 20 weeks. Similarly, Sethi et al[59] developed a long-term model in leucine-rich repeat-containing G protein-coupled receptor 5 (Lgr5)-p53-/- mice exposed to MNU and DCA for 12 months. These mice developed dysplastic and metaplastic lesions, highlighting the critical role of gene–environment interactions in promoting premalignant gastric transformation.
MNNG integrated induction model: The administration mode of MNNG solution and its combined solution is often alternate, and the concentration and dose are also adjusted according to the different administration mode. The concentration is lower and the dose is higher when drinking freely. The concentration was higher and the dose was lower du
(1) Ammonia and H. pylori simulation. The pH of normal gastric mucosa is 2.0, and ammonia can simulate the high ammonia concentration caused by H. pylori infection, destroying the acidic environment of gastric mucosa. Wei et al[16] by compared different mouse models found that the combination of H. pylori, MNNG, and ammonia water is an effective method of developing a mouse model of human CAG. Lv et al[60] MNNG, ammonia and irregular combined with fasting, PLGC rat model was successfully constructed; (2) Diet and lifestyle and stress-emotion factors: High-salt diet can promote gastric mucosal damage and inflammation, and it plays a role in combination with other factors. Yin et al[61] found that saturated salt plus MNNG for 25 weeks induced mild atrophy, while CAG with IM appeared after 35 weeks. Some studies mimicked poor eating habits by alternating fasting and satiation (1-day cycles) alongside MNNG administration for 10 weeks, successfully inducing CAG[62,63]. Zhu et al[64] established a PLGC model combining 24 weeks of MNNG gavage with an irregular diet (2 days satiation, 1 day fasting) and weekly tail-pinching to induce emotional stress. Such stress-emotion PLGC models provide a framework to explore the interactions between physiological, environmental, and psychosomatic factors in gastric precancerous lesion development; (3) Chemical reagents: Sodium salicylate is a non-steroidal anti-inflammatory drug, which causes inflammation by directly stimulating the local gastric mucosa, resulting in the shedding of gastric mucosa cells[18]. Zhu et al[65] combined MNNG with sodium salicylate (2%) to establish a PLGC rat model. At 21 weeks, gastric mucosa atrophy, IM and atypical hyperplasia occurred. In addition, long-term administration of MNNG with 5% aqueous alcohol solution successfully induced GC in rats after 14 months[66]; and (4) Role of ranitidine in accelerating MNNG mutation: Ranitidine can inhibit gastric acid secretion, cause weak acid environment in stomach, promote nitrate to nitrite and accelerate MNNG mutagenesis. The modified MNNG-induced PLGC model was established by oral MNNG, sodium salicylate, 40% alcohol, irregular fasting, and 0.03%-0.05% ranitidine, and such model rats exhibit the phenotypes of gastric mucosa pathology including atrophy, metaplasia, Dys, and adenocarcinoma[67-70]. This composite model is widely used in the evaluation of drug therapy effect.
MNNG combined with Helicobacter infection induction model: In Mongolian gerbils, 50 μg/mL MNNG via free drinking combined with H. pylori infection induced atrophic gastritis by 32 weeks and GC by 52 weeks, although IM was not observed[71]. In contrast, C57BL/6 mice receiving 40 μg/mL MNNG and HpSS1 infection developed moderate gastric hyperplasia at 48 weeks without tumor formation[7]. These findings suggest that H. pylori infection synergizes with chemical carcinogens like MNNG to promote gastric carcinogenesis, though outcomes vary by model and experimental conditions. Commonly used strategies for constructing animal models of precancerous gastric lesions are illu
| Type | Modeling method | Model animals | Modeling time | Observation time | Atrophy | Metaplasia | Dysplasia | Carcinoma | Ref. |
| MNU | MNU (120 ppm) | C57BL/6 mice | 8 weeks | 8 weeks | Yes | IM | Yes | No | [19] |
| MNU (100-120 ppm) | BALB/c and C57BL/6 mice, Wistar rats | 16-30 weeks | 31-54 weeks | Yes | Yes | Yes | Yes | [20,72] | |
| MNU (120 ppm) | db/db mice | 20 weeks | 30 weeks | No | No | Yes | Yes | [73] | |
| MNU (300 ppm) | BALB/c mice | 16 weeks | 16 weeks | No | IM (16 weeks) | No | No | [74] | |
| MNNG | MNNG (200 μg/mL) | SD rats | 16 weeks | 26 weeks | No | IM (16 weeks) | Yes (16 weeks) | No | [10,75] |
| MNNG (200 μg/mL) | SD rats | 40 weeks | 40 weeks | No | No | Yes (40 weeks) | Yes (40 weeks) | [9] | |
| MNNG (800 mg/L) | SD rats | 10 weeks | 34 weeks | Yes (10 weeks) | No | Yes (22 weeks) | Yes (34 weeks) | [11] | |
| MNNG (150 μg/mL) | BALB/c mice, C57BL/6 mice | 6 weeks | 14-36 weeks | Yes | IM | Yes | No | [12,13] | |
| CDCA | DCA (0.2%) | INS-GAS mice | 6 months | 6 months | Yes (5 months) | IM | Yes | No | [23] |
| CDCA (20 mmol/L) + 2% sodium salicylate | SD rats | 12 weeks | 12 weeks | Yes | IM | No | No | [24,25] | |
| CDCA (20 mmol/L) + 0.1% ammonia + irregular diet | SD rats | 16 weeks | 16 weeks | Yes | [26] | ||||
| Acute drug and H. pylori induction | DMP-777 | CD-1 rats, C57BL/6 mice | 7-14 days | 7-14 days | SPEM | No | No | No | [30,76] |
| L635 | C57BL/6 mice | 3 days | 3 days | SPEM | No | No | No | [77] | |
| Tamoxifen (250 mg/kg) | C57BL/6 mice | 3 days | 3 days | SPEM | Yes | No | No | [31,32] | |
| Tamoxifen (50 mg/kg) | gp130Y757F/Y757F mice | 2 days | 16 weeks | SPEM | Yes | Yes | Yes | [33] | |
| CagA-positive PMSS1 | C57BL/6 mice, Tert-/- mice | 12 weeks | 12 weeks | SPEM | No | No | No | [34] | |
| H. pylori strain | H. felis | C57BL/6 mice | 15 months | 15 months | Yes | Yes | Yes | Yes | [39] |
| H. pylori SS1 | C57BL/6 mice | 18 months | 18 months | Yes | Yes | No | No | [42] | |
| H. pylori PMSS1 | C57BL/6 mice | 8 weeks | 8 weeks | Yes | Yes | No | No | [43] | |
| H. pylori HpslyD + strain | C57BL/6 mice | 73 weeks | 73 weeks | Yes | IM | No | No | [47] | |
| H. pylori TN2GF4 strain | Mongolian gerbils | 18 weeks | 18 weeks | Yes (2 weeks) | IM | No | No | [50] | |
| H. pylori ATCC 43504 strain | Mongolian gerbils | 16-18 months | 16-18 months | Yes | IM | Yes | Yes | [49] | |
| H. pylori 7.13 strain | Mongolian gerbils | 7-9 weeks | 7-9 weeks | Yes | IM (7 weeks) | Yes | Yes (9 weeks) | [52] | |
| Non-H. pylori gastric microbiome | S. anginosus | C57BL/6 mice, germ-free BALB/c mice | 48 weeks | 48 weeks | Yes (9 months) | Yes (12 months) | Yes (12 months) | No | [53] |
| S. anginosus + MNU (240 ppm) | C57BL/6 mice | 36 weeks | 36 weeks | Yes | Yes | Yes | Yes (9 months) | [53] | |
| MNU + H. pylori | MNU (240 ppm) + HpSS1 | C57BL/6 mice | 12-16 weeks | 50-80 weeks | Yes | IM | Yes | Yes | [55] |
| MNU (200 ppm) + H. felis | FVB and B6 mice | 36 weeks | 36-50 weeks | Yes | Yes | Yes | Yes | [54,78] | |
| MNU + PMSS1 | C57BL/6 mice | 12 weeks | 40 weeks | Yes | IM | Yes | No | [56] | |
| MNU (10 ppm) + ATCC 43504 | Mongolian gerbil | 24 weeks | 53 weeks | Yes | IM | Yes | No | [57] | |
| MNU + dietary | MNU (120 ppm) + 10% NaCl | C57BL/6J mice | 40 weeks | 40 weeks | Yes | [36] | |||
| MNU (120 ppm) + HpSS1 + 10% NaCl | C57BL/6J mice | 40 weeks | 40 weeks | Yes | [36] | ||||
| MNU + DCA | C57BL/6 mice, Lgr5-p53-/- mice | 48 weeks | 48 weeks | Yes | Yes | No | [59] | ||
| MNU (300 ppm) + irregular diet | BALB/c mice | 20 weeks | 20 weeks | Yes | IM | Yes | No | [58] | |
| MNNG + H. pylori | MNNG (50 μg/mL) + KMUH-G926 | Mongolian gerbil | 20 weeks | 52 weeks | Yes (32 weeks) | No | Yes | Yes (52 weeks) | [71] |
| MNNG (40 μg/mL) + HpSS1 | C57BL/6J mice | 48 weeks | 48 weeks | No | No | Yes (48 weeks) | No | [7] | |
| MNNG composite model | MNNG (200 μg/mL) + saturated salt | Wistar rats | 25 weeks | 35 weeks | Yes (25 weeks) | IM | Yes (35 weeks) | No | [61] |
| MNNG (200 μg/mL) + 0.1% ammonia + irregular fasting | SD rats | 20 weeks | 20 weeks | Yes | IM | Yes | No | [60] | |
| MNNG (120 μg/mL) + H. pylori + ammonia | Kunming mice | 7 days | 120 days | Yes | No | No | No | [16] | |
| MNNG (170-200 μg/mL) + irregular diet | SD rats, Wistar rats | 10-12 weeks | 14 weeks | Yes | No | No | No | [62,63] | |
| MNNG (200 μg/mL) + irregular diet + emotionally stimulated | Wistar rats | 24 weeks | 24 weeks | Yes | IM | Yes | No | [64] | |
| MNNG (100 μg/mL) + 2% sodium salicylate | Wistar rats | 17 weeks | 21 weeks | Yes | IM | Yes | No | [65] | |
| MNNG (120 μg/mL) + 5% aqueous alcohol solution | Wistar rats | 56 weeks | 56 weeks | No | No | No | Yes (14 months) | [66] | |
| MNNG (120-150 μg/mL) + 2% sodium salicylate + irregular fasting + 0.05% ranitidine | Wistar rats | 32-40 weeks | 32-40 weeks | Yes | Yes | Yes | Yes | [67,68,70] | |
| MNNG (200 μg/mL) + 40% alcohol + irregular fasting + 0.03% ranitidine | SD rats | 40 weeks | 40 weeks | Yes | IM | Yes | No | [69] |
Gene editing mouse mode: Gene-edited mouse models are now widely used to study gastric carcinogenesis both domestically and internationally. By inducing aberrant expression of specific inflammatory mediators or mutations in oncogenes and tumor suppressor genes, these models more accurately recapitulate disease pathogenesis and enable functional analysis of key genes and signaling pathways involved in PLGC progression. Compared with Helicobacter infection or chemically induced models, gene-edited models offer advantages such as controlled induction timing and synchronized lesion development.
GAS correlation model: GAS knockout (GAS-/-) mice. GAS, secreted by antral G cells, regulates acid secretion via parietal and enterochromaffin-like cells. GAS-/- mice exhibit antral inflammation, parietal/enterochromaffin-like cell loss, and develop bacterial overgrowth, IM, and spontaneous tumors by 4-5 months under conventional housing[79]. By 12 months, they progress to gastritis, atrophy, IM, and invasive cancer. DMP-777 induces rapid SPEM (1-3 days) in
INS-GAS transgenic mice. INS-GAS transgenic mice, which express human GAS under the insulin promoter, develop sustained hypergastrinemia that initially (1-4 months) increases parietal cell mass and acid secretion, followed by progressive atrophy, metaplasia, Dys, and carcinoma by 20 months[80]. These mice are commonly combined with H. pylori infection to model gastric carcinogenesis, with pathological outcomes depending on bacterial strain and infection duration. For example, HpSS1 infection leads to Dys after 4-9 months[80], while PMSS1 causes invasive carcinoma as early as 4 months[81,82]. B129 strain infection induces Dys and tumors within 3-6 months[83]. INS-GAS and GAS-/- mice are often used together to investigate GAS’s role in tumorigenesis. Notably, tumors in INS-GAS mice localize mainly to the corpus, whereas GAS-/- lesions occur predominantly in the antrum, necessitating precise histological distinction between gastric regions in these models[84]. The accelerated neoplastic progression observed in H. pylori-infected INS-GAS mice, particularly with virulent strains such as PMSS1 or B129, parallels the enhanced cancer risk seen in humans. A cluster-randomized controlled trial has demonstrated that timely eradication of H. pylori reduces the incidence of GC[85] and decreases metachronous tumor development after endoscopic resection[86]. These findings highlight INS-GAS mice as a genetically sensitized host background in which H. pylori strain-dependent effects on PLGC progression are mar
Models related to the induction of inflammatory transmitters. Interleukin-1β-Tg mice (H+/K+-ATPase promoter-driven) develop spontaneous oxyntic atrophy, SPEM, IM, and high-grade Dys or carcinoma[87]. Interferon-γ (IFN-γ) overexpression in parietal cells induces corpus/antral inflammation, parietal/chief cell loss, SPEM, and atypical hyperplasia or adenomatous change[88]. The TxA23 TCR-Tg model, featuring CD4+ T cells against H+/K+-ATPase, mimics autoimmune gastritis with progressive oxyntic atrophy and metaplasia by 2-4 months; nearly all mice develop atypical hyperplasia by 12 months[89]. These models underscore the critical role of chronic cytokine signaling and immune injury in promoting gastric pre-neoplasia.
Oncogene mutation oncogene mutation modeling: Activation of oncogenic Kirsten rat sarcoma viral oncogene homolog (K-ras) in gastric chief cells induces rapid metaplastic transformation, with SPEM appearing within one month and IM by four months[90]. Several transgenic mouse lines expressing mutant K-ras under constitutive or inducible promoters, such as Mist1CreERT[91] and eR1CreERT[92], have been established. In K19-K-ras-V12 mice, parietal cell loss, SPEM, and Dys resemble Helicobacter felis-induced preneoplasia[93], underscoring Ras pathway activation as central to PLGC progression.
Loss of Runt-related transcription factor 3 leads to sequential SPEM and IM, with MNU accelerating adenocarcinoma[94]. Usf1-/- mice infected with H. pylori develop atrophic gastritis, IM, and Dys[95], while p27-/- mice infected with HpSS1 show inflammation, pseudopyloric metaplasia, atrophy, and Dys by 70 weeks[96]. More recently, gastric-specific deletion of SET domain containing 2, a histone methyltransferase linked to poor prognosis, combined with oncogenic Myc mutation (MYCR26StopFL/+; Setd2-/-), produced high-grade IM and Dys as early as 10 weeks, modeling early-onset GC[97].
Glandular cell injury: Suzuki et al[98] generated claudin-18 (stomach-specific claudin-18)-deficient mice, which exhibited increased gastric acid secretion and spontaneous SPEM as early as postnatal day 3. Notably, 20%-30% deve
Pathway misalignment: Several transgenic and knockout mouse models have delineated key pathways in gastric tumorigenesis. Peroxisome proliferator-activated receptor delta-overexpressing mice (villin promoter) sequentially developed SPEM, IM, Dys, and adenocarcinoma by 55 weeks, implicating peroxisome proliferator-activated receptor delta in epithelial transformation[102]. Smad3-/- mice exhibited early SPEM by 6 months and invasive neoplasia by 10 months, particularly at the forestomach/glandular junction[103]. MyD88-deficient mice infected with Helicobacter rapidly developed atrophy, metaplasia, and Dys (25-47 weeks), underscoring the role of innate immunity[104]. Wnt activation via Rnf43 mutation further aggravated Helicobacter-induced lesions[105]. Collectively, these models demon
Models of polygenic mutations or deletions in specific glandular cells. Seidlitz et al[106] developed a gastric-specific Anxa10-CreERT2 mouse model harboring KrasG12D/+, p53R172H/+, and Smad4fl/fl mutations, which showed Dys by 3 weeks, T1-T2 tumors by 2-8 weeks, T3-T4 tumors with liver/Lung metastases by 10 weeks. Li et al[107] inactivated Smad4 and Pten in Lgr5+ stem cells, leading to rapid antral adenoma and intramucosal carcinoma formation. Hayakawa et al[91] introduced Kras activation and/or Apc loss into Mist1+ isthmic stem cells; Kras alone induced Dys with IM, while combined mutations led to intestinal-type GC within 4 months. Fang et al[108] used H+/K+ ATPase-Cre to inactivate Lkb1 and Pten, resulting in hyperplastic polyps (approximately 20 weeks) progressing to metastatic adenocarcinoma (approximately 40 weeks). Genetically engineered mouse models of precancerous gastric lesions and their histological characteristics are summarized in Table 2[109-112].
| Type | Mouse model | Observation time | Intervention | SPEM | Atrophy | Metaplasia | Dysplasia | Carcinoma | Ref. |
| Gastrin-related | Gastrin-/- mice | 12 months | No | Yes | IM | Yes | Yes | [79] | |
| Gastrin-/- mice | 28 days | DMP-777 | Yes | [76] | |||||
| Gastrin-/- mice | 36 weeks | MNU + H. felis | Yes | Yes | Yes | Yes | [78] | ||
| INS-GAS mice | 36 weeks | MNU | Yes | Yes | Yes | Yes | [78] | ||
| INS-GAS mice | 6-18 months | H. pylori SS1 | Yes | Yes | Yes | No | [80] | ||
| INS-GAS mice | 3-4 months | H. pylori PMSS1, B128, NCTC11637 | Yes | IM | Yes | Yes | [81-83] | ||
| Inflammatory transmitter induction | H/K-IL-β transgenic mice | 12 months | No | Yes | Yes | Yes | Yes | Yes | [87,109] |
| H/K-IFN-γ transgenic mice | 15 months | No | Yes | Yes | Yes | Yes | Yes | [88] | |
| TxA23 TCR transgenic mice | 12 months | No | Yes | Yes | Yes | Yes | Yes | [89] | |
| K19-Wnt1/C2mE transgenic mice | 30 weeks | No | Yes | No | Yes | Yes | Yes | [110] | |
| Mutations in oncogenes and tumor suppressors | Mist1-Kras transgenic mice | 1-4 months | No | Yes | Yes | Yes | Yes | Yes | [90] |
| K19-K-ras-V12 transgenic mice | 20 months | No | Yes | Yes | Yes | Yes | [93] | ||
| Runx3-/- mice | 52 weeks | MNU | Yes | No | IM | Yes | Yes | [94] | |
| Usf1-/- mice | 12 months | H. pylori | Yes | IM | Yes | No | [95] | ||
| p27-/- mice | 70 weeks | H. pylori SS1 | Yes | Yes | Yes | No | [96] | ||
| MYCR26StopFL/+; Setd2-/-(AMC) mice | 10 weeks | No | No | IM | Yes | No | [97] | ||
| Glandular cell damage | stCldn18-/- mice | 100 weeks | No | Yes | Yes | IM | Yes | Yes | [98] |
| Slc26a9-/- mice | 18 months | No | Yes | Yes | IM | Yes | Yes | [99] | |
| Tff1-/- mice | 12 months | No | Yes | Yes | Yes | Yes | [100] | ||
| Hip1r-/- mice | 5 weeks | No | Yes | No | No | No | No | [110] | |
| GRIM-19-/- mice | 10 weeks | No | Yes | No | No | No | No | [101] | |
| Dysregulation of signalling pathways | PPARD mice | 55 weeks | No | Yes | Yes | IM | Yes | Yes | [102] |
| Smad3-/- mice | 10 months | No | Yes | Yes | Yes | Yes | Yes | [103] | |
| H/K-noggin mouse | 12 weeks | No | Yes | No | No | No | No | [111] | |
| MyD88-/- mice | 47 weeks | H. felis | Yes | IM | Yes | Yes | [104] | ||
| RNF43H292R/H295R mice | 6 months | H. pylori; PMSS1 | Yes | IM | Yes | Yes | [105] | ||
| Multiple gene mutations or deletions in specific glandular cells | Pgc-Cre mice | 9 months | No | Yes | Yes | Yes | Yes | Yes | [112] |
| Anxa10-Cre mice | 10 weeks | No | No | No | Yes | Yes | [106] | ||
| Lgr5-Cre; Smad4fl/fl; PTENfl/fl mice | 12 months | No | Yes | IM | Yes | Yes | [107] | ||
| Mist1-Cre; LSL-KrasG12D; Apcfl/fl mice | 4 months | No | Yes | IM | Yes | Yes | [91] | ||
| H/K Cre; LKB1L/L; PTENL/L mice | 40 weeks | No | No | IM | Yes | Yes | [108] |
MNNG-induced malignant transformation model of human GES-1: MNNG is commonly used to mimic the in vivo nitrosation process, whereby nitrates are converted into carcinogenic nitrites within the gastric environment. In vitro, MNNG induces malignant transformation in GES-1, promoting enhanced migration, invasion, and survival, as well as activating epithelial-mesenchymal transition (EMT) pathways. This model provides an effective tool for simulating the progression of PLGC[113].
Acute transformation model. Recent studies have established malignantly transformed cells (MCs) by treating GES-1 with MNNG at various concentrations. Doses above 50 μM exhibit significant cytotoxicity, while 30 μM for 24-48 hours is widely accepted for acute induction of transformation[58,74]. In a dose-response assay, MNNG (0-200 μM) exposure led to concentration-dependent reductions in cell viability, with 20-30 μM identified as optimal for inducing DNA damage without compromising survival[114]. At 30 μM for 48 hours, GES-1 cells exhibited approximately 20%-30% viability reduction, sufficient to trigger early transformation events[115,116]. Moreover, exposure to 20 μM MNNG for 24 hours in serum-free medium resulted in extensive cell death, followed by regeneration and stable MC cell lines by the third passage[58,117]. Interestingly, proliferative responses persisted up to 200 μM MNNG, with only 500 μM reversing this trend[118].
Chronic exposure model. Chronic low-dose MNNG effectively induces malignant transformation in GES-1 cells. GES-1 cells treated with 2 μM MNNG for 20 weeks (MC-40) developed malignant phenotypes[119,120]. Another study exposed cells to 1-4 μM MNNG with 0.1% DMSO over 60 passages (approximately 30 weeks), resulting in significantly increased colony formation and EMT marker expression comparable to GC cell line MGC803[121]. Similarly, BGC823 and HGC-27 cells treated chronically with 0.25-0.5 μM MNNG for 60 days showed dose-dependent increases in migration, invasion, vascular mimicry, spheroid formation, and cancer stem cell marker expression[66]. MNU has also been applied in chronic low-dose models to better mimic real-life carcinogen exposure. Song et al[122] cultured GES-1 cells with 0.01 g/L MNU (one-fifth of previously reported doses) for 6 months, successfully inducing malignant transformation while maintaining cell viability. Combined MNNG and H. pylori model. Several studies have combined MNNG treatment with chronic H. pylori infection to enhance transformation efficiency. In a model using the J99 H. pylori strain and daily 5 μM MNNG exposure for 1 year, GES-1 cells exhibited increased proliferation, migration, invasion, EMT, and tumorsphere formation, along with upregulation of cancer stem cell marker CD44[7].
GIM model induced by bile acids in GES-1 cells: Model establishment. DCA and CDCA are used to model bile acid-induced IM in GES-1 cells. GES-1 cells are starved in serum-free medium, then exposed to DCA or CDCA for 24 hours. This method effectively induces IM phenotype[123]. Among various bile acids tested cholic acid, DCA, and CDCA, CDCA was identified as the most potent inducer of IM. Dose- and time-dependent increases in IM markers (e.g., CDX2 and MUC2) were observed following treatment with 0-100 μM of bile acids for 3-24 hours, with CDCA inducing the most significant upregulation of IM-associated markers[123,124]. These findings suggest that CDCA is the optimal inducer for GIM model construction. To optimize the model, GES-1 cells were treated with 50, 100, and 200 μM CDCA. Western blot analysis of CDX2 protein expression revealed a statistically significant increase in CDX2 levels at 100 μM and 200 μM. Based on both efficacy and cell viability, 100 μM CDCA for 24 hours was determined to be the optimal condition for GIM induction[125].
Viability considerations. Cell viability was evaluated using the cell counting kit-8 assay after exposure to various concentrations of DCA and CDCA (50-600 μM) for different durations (6 and 15 minutes; 6, 12, 24, and 48 hours). Interestingly, 200 μM DCA increased GES-1 viability after only 15 minutes of stimulation, simulating the acute onset of bile reflux[23]. However, prolonged exposure (48 hours) to 100 μM or 200 μM of DCA or CDCA significantly reduced cell viability. High concentrations (400-600 μM) were cytotoxic even at short durations. To avoid excessive cytotoxicity while ensuring bile acid stimulation, 24-hour exposure to physiological concentrations (< 200 μM) was selected as the optimal condition for constructing the GIM model[126].
H. pylori-induced transformation model: Acute infection model. Acute H. pylori infection models are commonly esta
Chronic infection model. In the chronic infection model, GES-1 cells are co-cultured with H. pylori at multiplicities of infection of 1:1, 10:1, or 100:1, with daily replacement of fresh bacterial suspension. Co-culture typically continues for up to 6 months. Bacterial density is monitored at OD660 (approximately 2 × 108 CFU/mL per OD). Compared to acute infection, chronic CagA+H. pylori exposure induces profound phenotypic alterations in GES-1 cells, including apoptosis resistance, abnormal proliferation, enhanced migration, colony formation, and acquisition of stem cell-like features[128]. This model mimics in vivo transformation during Correa’s cascade and is increasingly applied in organoid-based gastric carcinogenesis research[129].
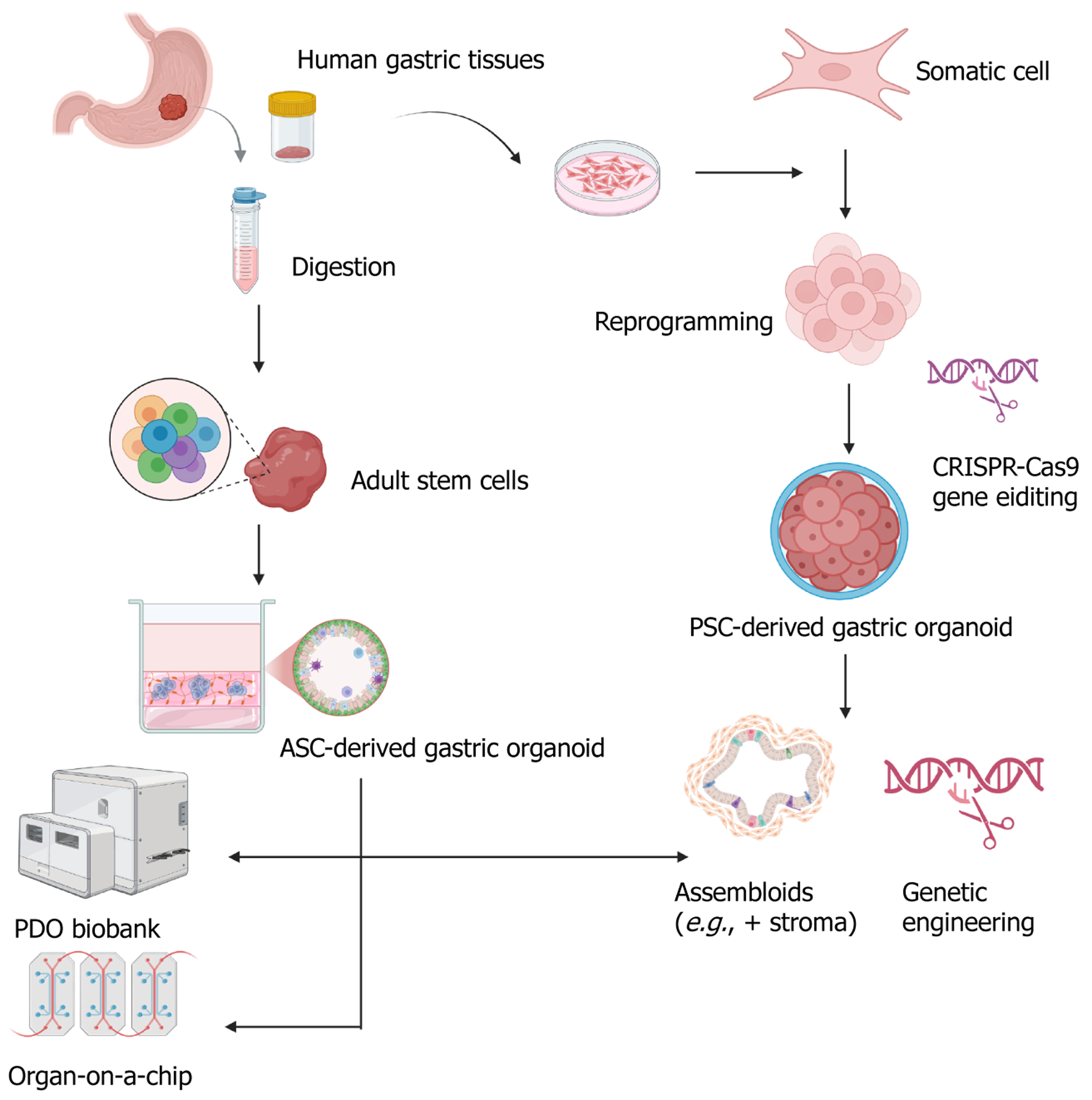
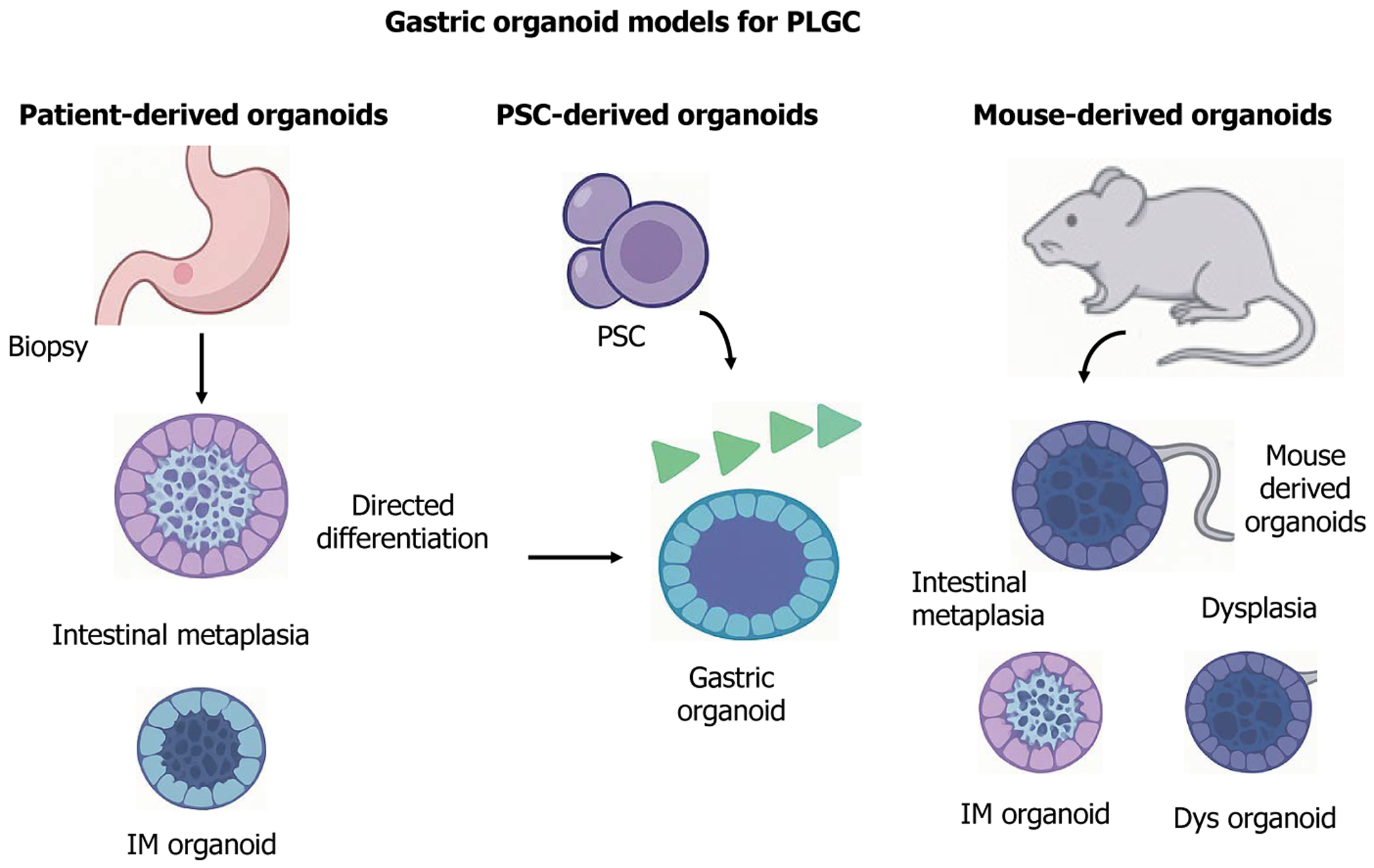
Patient-derived gastric precancerous organoids: Organoids are 3D clusters of organ-specific cells derived from stem cells with self-organizing capabilities. They retain many functional, molecular, and cellular features of their tissue of origin, including its heterogeneity[130]. Organoids derived from adult stem cells (ASCs) and pluripotent stem cells (PSCs) have become essential preclinical models for cancer research and therapeutic development. Importantly, ASC- and PSC-derived organoids are generated via distinct approaches: ASC organoids rely on niche-specific growth factor-enriched media to maintain physiological stem cell homeostasis, whereas PSC organoids undergo stepwise differentiation through sequential morphogen exposure, mimicking embryonic developmental pathways[131].
Precancerous organoids (PDOs) establishment. PDOs are typically derived from surgical or endoscopic gastric samples, processed via mechanical dissociation and enzymatic digestion, such as Dispase II and Collagenase XI[132], EDTA and TrypLE[133], Liberase TH and TrypLE Express[134], or collagenase and hyaluronidase[135], then embedded in extracellular matrix scaffolds (e.g., Matrigel) for 3D culture. Despite variations in media composition, the core aim is to replicate the gastric stem cell niche to sustain growth and differentiation. One common technical challenge is the contamination or overgrowth of normal epithelial organoids. To enrich for precancerous or neoplastic PDOs, several strategies have been employed, including targeted sampling of lesion-rich areas, selection of regions with high tumor purity, and the use of gene-editing tools[136].
ASC-derived organoids of atrophic gastritis, IM, and Dys. Dysfunction of gastric stem cells underlies atrophic gastritis pathogenesis. Li et al[137] successfully expanded organoids from antral biopsies of non-atrophic gastritis, while most atrophic gastritis samples failed to grow, suggesting impaired gastric stem cell self-renewal. Yoshihiro et al[138] gene
IM-derived organoids using optimized stem cell niche factors such as Wnt3A, R-spondin, epidermal growth factor, and Noggin, enabling long-term propagation[139]. Gene-editing of E-cadherin and/or RHOA in normal gastric organoids reproduced IM development[134]. Wei et al[140] group developed IM-mimicking patient-derived organoids revealing OLFM4 and MYH9 cooperation in IM progression. Large-scale RNA-seq revealed heterogeneity and precancerous signatures in IM organoids, identifying STMN1+ isthmus stem cells as potential origins. Dysplastic organoids were also derived from resected tissues, showing chromosomal instability and dysplastic stem cell features[141].
For Dys, Yan et al[135] established 46 GC organoid lines, including dysplastic types, from 34 patients. Serosal sampling improved tumor purity, and a modified STAR protocol achieved > 50% success, validated by whole-exome sequencing. Min et al[142] further demonstrated that Meta4 organoids derived from Mist1-Kras (G12D) mice contain dysplastic stem cells with multilineage potential, functioning as key regulators of the transition from PLGC to cancer. Through comp
PSC-derived organoids. PSC-derived gastric organoids are generated via stepwise differentiation through definitive endoderm and foregut stages into gastric progenitors within a 3D matrix environment[143]. Lgr5, a Wnt-responsive stem cell marker, has facilitated long-term culture of stable organoids[139]. McCracken et al[144] first derived human PSCs into 3D gastric organoids containing progenitor and differentiated cell types, chief, mucous, and endocrine cells, that res
Karlsson et al[145] applied CRISPR-Cas9 technology to knock out both alleles of TP53 in human PSCs into 3D gastric organoids and observed their transformation toward IM and tumor-like phenotypes. Single-cell RNA sequencing confirmed loss of mucosal identity. Koide et al[146] used human-induced pluripotent stem cells with CDX2 overexpression to create an incomplete IM model, showing intestinal marker upregulation, MUC5AC loss, and crypt-like mor
Mouse-derived organoids: Although mouse models cannot fully replicate the physiological and genetic features of the human stomach, organoids derived from murine gastric tissues have become valuable tools for investigating the path
Mouse-derived models of IM and Dys. MNNG-treated mouse gastric organoids exhibit IM-like morphology with CDX2/MUC2 upregulation and ATP4B/MUC6 loss. Lineage tracing confirmed MIST1+ chief cells as the source of met
Min et al[148] developed two organoid lines Meta3 and Meta4 derived from the gastric corpus of Mist1-Kras(G12D) mice, collected three and four months after tamoxifen induction, respectively. Meta3 organoids recapitulated IM phenotypes, while Meta4 organoids exhibited more pronounced dysplastic characteristics. Using CRISPR-Cas9, they further generated Meta4-cortactin knockout dysplastic organoids and found that cortactin was a critical mediator of membrane dynamics, malignant transformation, and exosome secretion in dysplastic cells, implicating it in the tumor microenvironment remodeling and solid tumor formation. Clinically, its aberrant expression may serve as a biomarker for risk stratification in gastric precancerous lesions, and its inhibition could offer a novel preventive strategy to block progression toward invasive cancer.
Transgenic mouse-derived organoids. Although engineered mouse models do not fully capture the entire spectrum of human GC, particularly distant metastasis[59], they remain indispensable tools for dissecting the molecular mechanisms of PLGC. Lu et al[149] developed TMPC (Trp53-/-; Myc; sgPten; sgCdkn2b) gastric organoids that recapitulate Dys-to-metastasis progression post-transplantation. The system allows rapid assembly of driver mutations using CRISPR/Cas9, making it an ideal platform for functional oncogene validation. In a related study, mutations in TP53 and ARID1A were introduced into human gastric organoids, which formed malignant tumors upon subcutaneous transplantation into immunodeficient recipient mice[150]. These findings highlight the power of transplanting genetically modified PDOs into host systems as a versatile strategy for modeling diverse cancer phenotypes.
Organoid models for immune microenvironment and H. pylori infection: Organoid-immune interactions. PLGC deve
Organoid models of H. pylori infection. Gastric organoids, with their 3D architecture closely resembling native glandular structure, offer an ideal platform to study H. pylori-induced carcinogenesis. Guenther et al[153] generated human gastric organoids containing pit mucous, gland mucous, chief, and enteroendocrine cells. H. pylori infection strongly activated nuclear factor-κB signaling and pro-inflammatory cytokine secretion. H. pylori-induced tumorigenesis also involves metabolic pathways. In Smox-/- organoids, reduced inflammation, DNA damage, and β-catenin signaling confirmed the role of polyamine metabolism[154]. The bacterium also upregulates ACVR1, activating the IRF3/POLD1 axis and impairing DNA repair, promoting genomic instability[82].
Single-cell RNA sequencing has revealed distinct cellular compositions across different organoid configurations. While 2D monolayer cultures are enriched in pit-like epithelial cells, 3D organoids are predominantly composed of glandular progenitor cells[155]. Comparative studies demonstrated that apical-out and 2D organoids, with their luminal surface exposed and greater experimental accessibility, significantly enhance H. pylori adhesion and facilitate the study of early epithelial responses to infection, such as barrier disruption and inflammation[156].
To better mimic physiology, a dual-channel organoid-on-chip platform was developed, supporting H. pylori col
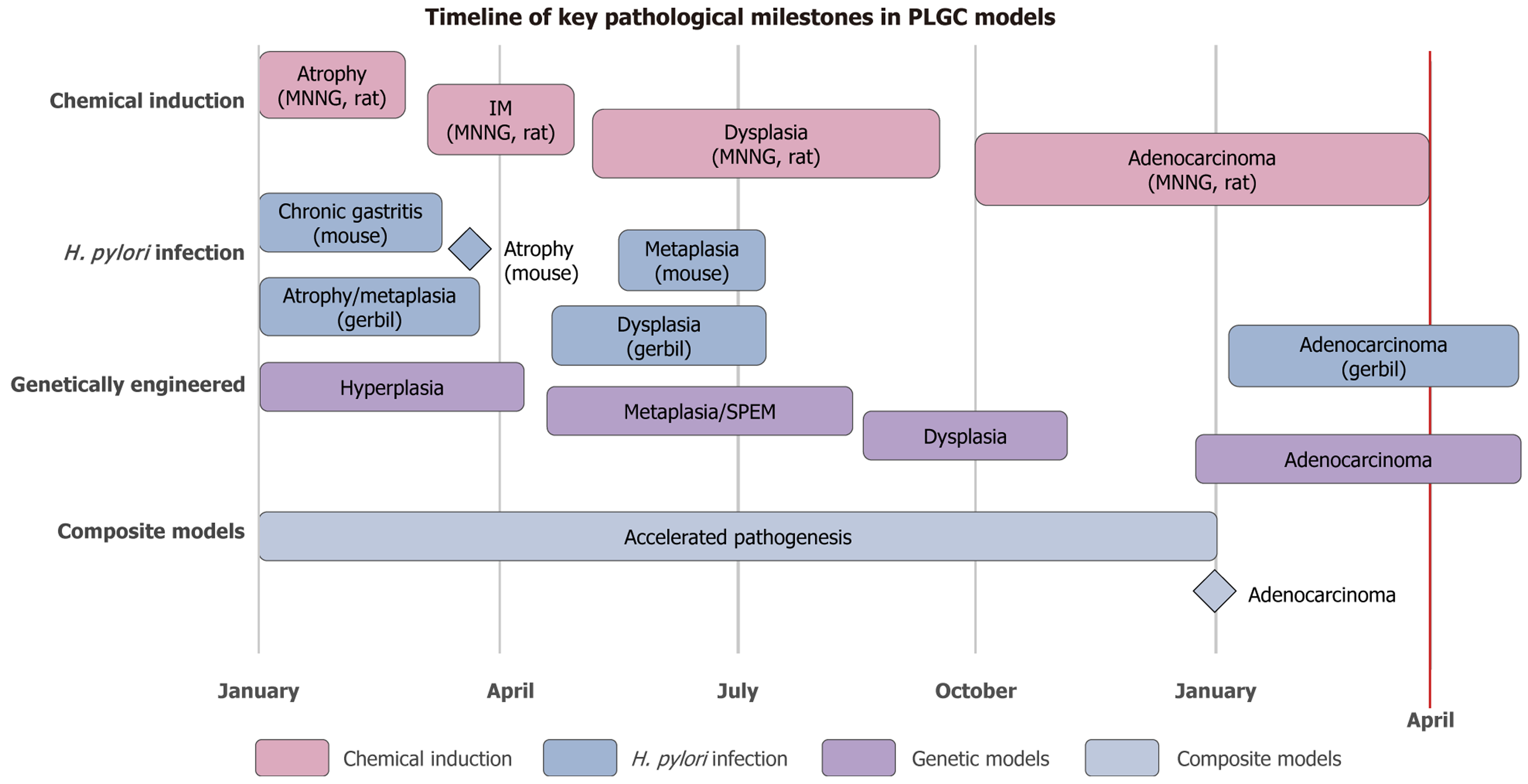
GC develops through the Correa cascade, with PLGC representing a pivotal transitional stage. Accurate PLGC modeling is essential for dissecting early oncogenic events and evaluating preventive strategies. Chemically induced models (e.g., MNNG, MNU, bile acids) have recapitulated epithelial injury and transformation, though outcomes vary depending on dose, duration, and route, highlighting the importance of tailored model selection. A decision framework for selecting precancerous lesions of gastric cancer research models is shown in Table 3. However, these models only partially mimic human PLGC, as they lack the sustained immune infiltration and stromal remodeling characteristic of H. pylori-driven disease. Approximate timeframes for the development of key stages in the Correa cascade across different model systems are summarized in Figure 6, providing guidance for aligning study duration with specific lesion progression.
| Research objective | Model | Induction/approach | Advantages | Limitations |
| SPEM mechanisms | Mouse (acute drug-induced) | DMP-777 (10-14 days), L635 (10-14 days) | Rapid, reversible, specific parietal cell loss | Lacks chronic inflammatory context |
| IM | Mouse (bile acid-induced), GES-1 cells, gastric organoids | Mouse: 0.2% DCA in drinking water (6 months); GES-1: 100 μM CDCA for 24 hours. Organoids: BMP activation | In vivo: Recapitulates IM progression. In vitro: Rapid, high-throughput. Organoids: Human-specific, physiologically relevant | In vivo: Long duration. In vitro: Lack tissue complexity. Organoids: Lack full tumor microenvironment |
| H. pylori pathogenesis | Mongolian gerbil, C57BL/6 mouse | H. pylori inoculation (e.g., TN2GF4, 7.13, PMSS1) | Gerbils: Full Correa cascade to adenocarcinoma. Mice: Genetic tools available, suitable for immune studies | Gerbils: Limited genetic tools. Mice: Resistant to many H. pylori strains |
| Multifactorial carcinogenesis | Composite rat/mouse models | MNNG/MNU + H. pylori; MNNG/MNU + high-salt diet + ranitidine | Models human synergistic etiology; high clinical relevance | Complex setup, multiple variables to control |
| Genetic and molecular mechanisms | GEMMs (e.g., INS-GAS, p53KO), CRISPR-edited organoids | Genetic manipulation (e.g., KrasG12D, TP53-/-) | Definitive genotype-phenotype studies; precise temporal control | High cost, technical complexity |
| Drug screening and toxicity | Cell lines (GES-1, BGC823) | Acute or chronic MNNG/MNU treatment or H. pylori co-culture | High-throughput, low cost, reproducible | Simplified system, lacks in vivo physiology |
| Immune-microenvironment interactions | Immune-organoid co-culture | Co-culture with macrophages (RAW264.7) or PBMCs | Incorporates human immune components; personalized potential | Technically challenging; not fully vascularized |
Compared to chemical agents, H. pylori infection better mimics human pathology, with strain- and host-specific responses in gerbils and mice. Multifactorial models combining H. pylori, carcinogens, diet, and host genetics enhance fidelity by capturing synergistic mechanisms. Acute drug-induced models have elucidated precursor lesions such as SPEM, while genetically engineered mice allow precise manipulation of tumor-related genes and immune dynamics. In addition, stress-related models, such as restraint stress or tail-clamping paradigms, have been used to explore how psychological and neuroendocrine factors modulate gastric mucosal injury and carcinogenic progression, highlighting the influence of host systemic responses[158]. Human GES models offer a practical platform for mechanistic studies and high-throughput screening. While they replicate early molecular alterations induced by carcinogens or infection, their 2D nature and lack of tissue architecture curtail their ability to model disease heterogeneity and cellular crosstalk observed in vivo.
ASC-derived organoids recapitulate renewal, IM, and Dys. CRISPR-Cas9-mediated gene editing has yielded tractable PLGC models for genotype-phenotype correlation studies[134]. PSC-derived organoids mimic developmental signaling and robustly respond to H. pylori infection. Innovations such as organoid-on-chip platforms and apical-out polarity improve physiological relevance[157]. Co-culture with immune cells and organoid assembloids enables exploration of inflammation-driven transformation and tumor-immune interactions[152]. Nevertheless, such co-culture systems remain simplified, typically incorporating limited immune cell subsets and lacking vascular perfusion, thereby constraining their ability to fully recapitulate the spatial and temporal complexity of in vivo immune regulation.
Despite their promise, organoids face limitations, including the absence of vasculature, stromal architecture, and a fully competent immune system, as well as challenges in reproducibility across laboratories. These constraints limit their ability to model chronic inflammation, immune-mediated epithelial remodeling, and stromal-epithelial interactions that are central to human PLGC progression. However, advances in CRISPR gene editing, microfluidics, and single-cell multi-omics are mitigating these barriers. Integration with immune co-culture systems, organoid-on-chip platforms, and PDO biobanks is accelerating biomarker discovery and personalized therapy development[159,160]. Future PLGC modeling will increasingly combine chemical, microbial, dietary, genetic, and stress-related paradigms to recapitulate multifactorial tumorigenesis. Spatial transcriptomics, lineage tracing, and next-generation organoid systems incorporating vasculature, stroma, and immune components are expected to bridge the gap between reductionist models and the human gastric mi
Animal, cellular, and organoid models provide complementary insights into PLGC. Their continued refinement, through genetic engineering, 3D culture, immune modeling, and stress paradigms, will enhance understanding of early gastric tumorigenesis and facilitate development of early detection, prevention, and personalized therapeutic strategies.
| 1. | Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16785] [Cited by in RCA: 14739] [Article Influence: 7369.5] [Reference Citation Analysis (20)] |
| 2. | Correa P. Human gastric carcinogenesis: a multistep and multifactorial process--First American Cancer Society Award Lecture on Cancer Epidemiology and Prevention. Cancer Res. 1992;52:6735-6740. [PubMed] |
| 3. | Song H, Ekheden IG, Zheng Z, Ericsson J, Nyrén O, Ye W. Incidence of gastric cancer among patients with gastric precancerous lesions: observational cohort study in a low risk Western population. BMJ. 2015;351:h3867. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 241] [Cited by in RCA: 227] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 4. | Zhang M, Zhong J, Song Z, Xu Q, Chen Y, Zhang Z. Regulatory mechanisms and potential therapeutic targets in precancerous lesions of gastric cancer: A comprehensive review. Biomed Pharmacother. 2024;177:117068. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 5. | Vermeer IT, Engels LG, Pachen DM, Dallinga JW, Kleinjans JC, van Maanen JM. Intragastric volatile N-nitrosamines, nitrite, pH, and Helicobacter pylori during long-term treatment with omeprazole. Gastroenterology. 2001;121:517-525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 28] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 6. | Wyatt MD, Pittman DL. Methylating agents and DNA repair responses: Methylated bases and sources of strand breaks. Chem Res Toxicol. 2006;19:1580-1594. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 366] [Cited by in RCA: 343] [Article Influence: 17.2] [Reference Citation Analysis (1)] |
| 7. | Lin L, Wei H, Yi J, Xie B, Chen J, Zhou C, Wang L, Yang Y. Chronic CagA-positive Helicobacter pylori infection with MNNG stimulation synergistically induces mesenchymal and cancer stem cell-like properties in gastric mucosal epithelial cells. J Cell Biochem. 2019;120:17635-17649. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 29] [Article Influence: 4.1] [Reference Citation Analysis (1)] |
| 8. | Sato J, Tomonari Y, Doi T, Tsuchitani M. Spontaneous globule leukocyte tumor accompanied by inflammatory cells in a Wistar Hannover rat. J Toxicol Pathol. 2019;32:189-195. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 9. | Isyraqiah F, Kutty MK, Durairajanayagam D, Singh HJ. Leptin enhances N-methyl-N'-nitro-N-nitrosoguanidine (MNNG)-induced tumour growth in gastric mucosa of male Sprague-Dawley rats. Mol Biol Rep. 2019;46:5967-5975. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 10. | Li D, Zhao L, Li Y, Kang X, Zhang S. Gastro-Protective Effects of Calycosin Against Precancerous Lesions of Gastric Carcinoma in Rats. Drug Des Devel Ther. 2020;14:2207-2219. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 11. | Cai D, Yu J, Qiu J, He B, Chen Z, Yan M, Liu Q. Dynamic changes of Sonic Hedgehog signaling pathway in gastric mucosa of rats with MNNG-induced gastric precancerous lesions. J Cell Physiol. 2019;234:10827-10834. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 12. | Yang P, Yang H, Zhou H, Li Q, Wei S, Wang Q, Yan Y, Liu Y, Pan H, Li S. Weipiling decoction alleviates N-methyl-N-nitro-N'-nitrosoguanidine-induced gastric precancerous lesions via NF-κB signalling pathway inhibition. Chin Med. 2022;17:104. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (2)] |
| 13. | Ma Z, Chen X, Xiong M, Wang H, Sun C, Tang W, Li J, Li X, Ma H, Ye X. Cyberpharmacology uncover the mechanism of the total Rhizoma Coptidis extracts ameliorate chronic atrophic gastritis. J Ethnopharmacol. 2024;335:118644. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (1)] |
| 14. | Yu C, Su Z, Li Y, Li Y, Liu K, Chu F, Liu T, Chen R, Ding X. Dysbiosis of gut microbiota is associated with gastric carcinogenesis in rats. Biomed Pharmacother. 2020;126:110036. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 50] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 15. | Weber K. Differences in Types and Incidence of Neoplasms in Wistar Han and Sprague-Dawley Rats. Toxicol Pathol. 2017;45:64-75. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 25] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 16. | Wei X, Feng XP, Wang LY, Huang YQ, Liang LL, Mo XQ, Wei HY. Improved method for inducing chronic atrophic gastritis in mice. World J Gastrointest Oncol. 2019;11:1115-1125. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 2] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 17. | Cai T, Zhang C, Zhao Z, Li S, Cai H, Chen X, Cai D, Liu W, Yan Y, Xie K, Pan H, Zeng X. The gastric mucosal protective effects of astragaloside IV in mnng-induced GPL rats. Biomed Pharmacother. 2018;104:291-299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 53] [Article Influence: 6.6] [Reference Citation Analysis (1)] |
| 18. | Wang XY, Wang LL, Zheng X, Meng LN, Lyu B, Jin HF. Expression of p-STAT3 and vascular endothelial growth factor in MNNG-induced precancerous lesions and gastric tumors in rats. World J Gastrointest Oncol. 2016;8:305-313. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 12] [Cited by in RCA: 19] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 19. | Zhang SX, Tian W, Liu YL, Ni JH, Zhang D, Pan HF, Zhao ZM, Ai B, Chen ZS, Lin LZ, Liu W. Mechanism of N-Methyl-N-Nitroso-Urea-Induced Gastric Precancerous Lesions in Mice. J Oncol. 2022;2022:3780854. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 20. | Yamamoto M, Furihata C, Ogiu T, Tsukamoto T, Inada Ki, Hirano K, Tatematsu M. Independent variation in susceptibilities of six different mouse strains to induction of pepsinogen-altered pyloric glands and gastric tumor intestinalization by N-methyl-N-nitrosourea. Cancer Lett. 2002;179:121-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 34] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 21. | Wang YM, Luo ZW, Shu YL, Zhou X, Wang LQ, Liang CH, Wu CQ, Li CP. Effects of Helicobacter pylori and Moluodan on the Wnt/β-catenin signaling pathway in mice with precancerous gastric cancer lesions. World J Gastrointest Oncol. 2024;16:979-990. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (1)] |
| 22. | He Q, Liu L, Wei J, Jiang J, Rong Z, Chen X, Zhao J, Jiang K. Roles and action mechanisms of bile acid-induced gastric intestinal metaplasia: a review. Cell Death Discov. 2022;8:158. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 34] [Article Influence: 8.5] [Reference Citation Analysis (1)] |
| 23. | Jin D, Huang K, Xu M, Hua H, Ye F, Yan J, Zhang G, Wang Y. Deoxycholic acid induces gastric intestinal metaplasia by activating STAT3 signaling and disturbing gastric bile acids metabolism and microbiota. Gut Microbes. 2022;14:2120744. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 86] [Article Influence: 21.5] [Reference Citation Analysis (1)] |
| 24. | Xu Z, Xiao L, Wang S, Cheng Y, Wu J, Meng Y, Bao K, Zhang J, Cheng C. Alteration of gastric microbiota and transcriptome in a rat with gastric intestinal metaplasia induced by deoxycholic acid. Front Microbiol. 2023;14:1160821. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 14] [Reference Citation Analysis (1)] |
| 25. | Cheng Y, Wang S, Zhu W, Xu Z, Xiao L, Wu J, Meng Y, Zhang J, Cheng C. Deoxycholic acid inducing chronic atrophic gastritis with colonic mucosal lesion correlated to mucosal immune dysfunction in rats. Sci Rep. 2024;14:15798. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 26. | Yu W, Chen S, Guan X, He G, Zhang W, Zhang H, Huang S, Ye Z, Pan H, Zhong Z. Yiqi Huayu Jiedu formula inhibits JAK2/STAT3-mediated partial EMT in treating chronic atrophic gastritis. Phytomedicine. 2025;137:156356. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 27. | Yoshizawa N, Takenaka Y, Yamaguchi H, Tetsuya T, Tanaka H, Tatematsu M, Nomura S, Goldenring JR, Kaminishi M. Emergence of spasmolytic polypeptide-expressing metaplasia in Mongolian gerbils infected with Helicobacter pylori. Lab Invest. 2007;87:1265-1276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 89] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 28. | Nam KT, Lee HJ, Sousa JF, Weis VG, O'Neal RL, Finke PE, Romero-Gallo J, Shi G, Mills JC, Peek RM Jr, Konieczny SF, Goldenring JR. Mature chief cells are cryptic progenitors for metaplasia in the stomach. Gastroenterology. 2010;139:2028-2037.e9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 257] [Cited by in RCA: 249] [Article Influence: 15.6] [Reference Citation Analysis (2)] |
| 29. | Burclaff J, Willet SG, Sáenz JB, Mills JC. Proliferation and Differentiation of Gastric Mucous Neck and Chief Cells During Homeostasis and Injury-induced Metaplasia. Gastroenterology. 2020;158:598-609.e5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 99] [Cited by in RCA: 95] [Article Influence: 15.8] [Reference Citation Analysis (1)] |
| 30. | Goldenring JR, Ray GS, Coffey RJ, Meunier PC, Haley PJ, Barnes TB, Car BD. Reversible drug-induced oxyntic atrophy in rats. Gastroenterology. 2000;118:1080-1093. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 104] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 31. | Huh WJ, Khurana SS, Geahlen JH, Kohli K, Waller RA, Mills JC. Tamoxifen induces rapid, reversible atrophy, and metaplasia in mouse stomach. Gastroenterology. 2012;142:21-24.e7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 214] [Cited by in RCA: 206] [Article Influence: 14.7] [Reference Citation Analysis (1)] |
| 32. | Saenz JB, Burclaff J, Mills JC. Modeling Murine Gastric Metaplasia Through Tamoxifen-Induced Acute Parietal Cell Loss. Methods Mol Biol. 2016;1422:329-339. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 59] [Article Influence: 5.9] [Reference Citation Analysis (1)] |
| 33. | O'Keefe RN, Carli ALE, Baloyan D, Chisanga D, Shi W, Afshar-Sterle S, Eissmann MF, Poh AR, Pal B, Seillet C, Locksley RM, Ernst M, Buchert M. A tuft cell - ILC2 signaling circuit provides therapeutic targets to inhibit gastric metaplasia and tumor development. Nat Commun. 2023;14:6872. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 36] [Article Influence: 12.0] [Reference Citation Analysis (1)] |
| 34. | He L, Zhang X, Zhang S, Wang Y, Hu W, Li J, Liu Y, Liao Y, Peng X, Li J, Zhao H, Wang L, Lv YF, Hu CJ, Yang SM. H. Pylori-Facilitated TERT/Wnt/β-Catenin Triggers Spasmolytic Polypeptide-Expressing Metaplasia and Oxyntic Atrophy. Adv Sci (Weinh). 2025;12:e2401227. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 35. | Gamboa-Dominguez A, Ubbelohde T, Saqui-Salces M, Romano-Mazzoti L, Cervantes M, Domínguez-Fonseca C, de la Luz Estreber M, Ruíz-Palacios GM. Salt and stress synergize H. pylori-induced gastric lesions, cell proliferation, and p21 expression in Mongolian gerbils. Dig Dis Sci. 2007;52:1517-1526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 0.9] [Reference Citation Analysis (2)] |
| 36. | Toyoda T, Tsukamoto T, Yamamoto M, Ban H, Saito N, Takasu S, Shi L, Saito A, Ito S, Yamamura Y, Nishikawa A, Ogawa K, Tanaka T, Tatematsu M. Gene expression analysis of a Helicobacter pylori-infected and high-salt diet-treated mouse gastric tumor model: identification of CD177 as a novel prognostic factor in patients with gastric cancer. BMC Gastroenterol. 2013;13:122. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 39] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 37. | Watanabe T, Tada M, Nagai H, Sasaki S, Nakao M. Helicobacter pylori infection induces gastric cancer in mongolian gerbils. Gastroenterology. 1998;115:642-648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 752] [Cited by in RCA: 664] [Article Influence: 23.7] [Reference Citation Analysis (2)] |
| 38. | Wang TC, Goldenring JR, Dangler C, Ito S, Mueller A, Jeon WK, Koh TJ, Fox JG. Mice lacking secretory phospholipase A2 show altered apoptosis and differentiation with Helicobacter felis infection. Gastroenterology. 1998;114:675-689. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 187] [Article Influence: 6.7] [Reference Citation Analysis (1)] |
| 39. | Fox JG, Sheppard BJ, Dangler CA, Whary MT, Ihrig M, Wang TC. Germ-line p53-targeted disruption inhibits helicobacter-induced premalignant lesions and invasive gastric carcinoma through down-regulation of Th1 proinflammatory responses. Cancer Res. 2002;62:696-702. [PubMed] |
| 40. | Lee A, O'Rourke J, De Ungria MC, Robertson B, Daskalopoulos G, Dixon MF. A standardized mouse model of Helicobacter pylori infection: introducing the Sydney strain. Gastroenterology. 1997;112:1386-1397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 813] [Cited by in RCA: 759] [Article Influence: 26.2] [Reference Citation Analysis (1)] |
| 41. | Arnold IC, Lee JY, Amieva MR, Roers A, Flavell RA, Sparwasser T, Müller A. Tolerance rather than immunity protects from Helicobacter pylori-induced gastric preneoplasia. Gastroenterology. 2011;140:199-209. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 259] [Cited by in RCA: 244] [Article Influence: 16.3] [Reference Citation Analysis (1)] |
| 42. | Bazin T, Krebs A, Jobart-Malfait A, Camilo V, Michel V, Benezeth Y, Marzani F, Touati E, Lamarque D. Multimodal imaging as optical biopsy system for gastritis diagnosis in humans, and input of the mouse model. EBioMedicine. 2021;69:103462. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 43. | Khurana SS, Riehl TE, Moore BD, Fassan M, Rugge M, Romero-Gallo J, Noto J, Peek RM Jr, Stenson WF, Mills JC. The hyaluronic acid receptor CD44 coordinates normal and metaplastic gastric epithelial progenitor cell proliferation. J Biol Chem. 2013;288:16085-16097. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 99] [Article Influence: 7.6] [Reference Citation Analysis (1)] |
| 44. | Kienesberger S, Cox LM, Livanos A, Zhang XS, Chung J, Perez-Perez GI, Gorkiewicz G, Zechner EL, Blaser MJ. Gastric Helicobacter pylori Infection Affects Local and Distant Microbial Populations and Host Responses. Cell Rep. 2016;14:1395-1407. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 150] [Cited by in RCA: 133] [Article Influence: 13.3] [Reference Citation Analysis (1)] |
| 45. | Liu M, Zhong Y, Chen J, Liu Y, Tang C, Wang X, Zhang Y, Wang P, Logan SM, Chen W, Wei B. Oral immunization of mice with a multivalent therapeutic subunit vaccine protects against Helicobacter pylori infection. Vaccine. 2020;38:3031-3041. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 37] [Article Influence: 6.2] [Reference Citation Analysis (1)] |
| 46. | Sigal M, Rothenberg ME, Logan CY, Lee JY, Honaker RW, Cooper RL, Passarelli B, Camorlinga M, Bouley DM, Alvarez G, Nusse R, Torres J, Amieva MR. Helicobacter pylori Activates and Expands Lgr5(+) Stem Cells Through Direct Colonization of the Gastric Glands. Gastroenterology. 2015;148:1392-404.e21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 219] [Cited by in RCA: 199] [Article Influence: 18.1] [Reference Citation Analysis (1)] |
| 47. | Zheng S, Wang Y, Ni C, Guo R, Qiu X, Chen J, Wang L, Sun X, Chen M, Liu Y, Yuan Y, Gong Y. Helicobacter pylori SlyD stabilizes TPT1 via hnRNPK and enhances OCT1-mediated CDX2 transcriptional activation to drive gastric intestinal metaplasia. BMC Med. 2025;23:71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 48. | Amalia R, Panenggak NSR, Doohan D, Rezkitha YAA, Waskito LA, Syam AF, Lubis M, Yamaoka Y, Miftahussurur M. A comprehensive evaluation of an animal model for Helicobacter pylori-associated stomach cancer: Fact and controversy. Helicobacter. 2023;28:e12943. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (2)] |
| 49. | Hirayama F, Takagi S, Iwao E, Yokoyama Y, Haga K, Hanada S. Development of poorly differentiated adenocarcinoma and carcinoid due to long-term Helicobacter pylori colonization in Mongolian gerbils. J Gastroenterol. 1999;34:450-454. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 76] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 50. | Kudo T, Lu H, Wu JY, Ohno T, Wu MJ, Genta RM, Graham DY, Yamaoka Y. Pattern of transcription factor activation in Helicobacter pylori-infected Mongolian gerbils. Gastroenterology. 2007;132:1024-1038. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 35] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 51. | Yamada T, Wei M, Toyoda T, Yamano S, Wanibuchi H. Inhibitory effect of Raphanobrassica on Helicobacter pylori-induced gastritis in Mongolian gerbils. Food Chem Toxicol. 2014;70:107-113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 52. | Bae M, Jang S, Lim JW, Kang J, Bak EJ, Cha JH, Kim H. Protective effect of Korean Red Ginseng extract against Helicobacter pylori-induced gastric inflammation in Mongolian gerbils. J Ginseng Res. 2014;38:8-15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 43] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 53. | Fu K, Cheung AHK, Wong CC, Liu W, Zhou Y, Wang F, Huang P, Yuan K, Coker OO, Pan Y, Chen D, Lam NM, Gao M, Zhang X, Huang H, To KF, Sung JJY, Yu J. Streptococcus anginosus promotes gastric inflammation, atrophy, and tumorigenesis in mice. Cell. 2024;187:882-896.e17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 215] [Cited by in RCA: 187] [Article Influence: 93.5] [Reference Citation Analysis (1)] |
| 54. | Lee JY, Kim N, Choi YJ, Nam RH, Choi YJ, Lee S, Choi D, Lee HS, Kim JW, Lee DH. Effect of N-Methyl-N-Nitrosourea on Helicobacter-induced Gastric Carcinogenesis in C57BL/6 Mice. J Cancer Prev. 2016;21:182-186. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 55. | Su T, Li F, Guan J, Liu L, Huang P, Wang Y, Qi X, Liu Z, Lu L, Wang D. Artemisinin and its derivatives prevent Helicobacter pylori-induced gastric carcinogenesis via inhibition of NF-κB signaling. Phytomedicine. 2019;63:152968. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 39] [Article Influence: 5.6] [Reference Citation Analysis (1)] |
| 56. | Cai Q, Shi P, Yuan Y, Peng J, Ou X, Zhou W, Li J, Su T, Lin L, Cai S, He Y, Xu J. Inflammation-Associated Senescence Promotes Helicobacter pylori-Induced Atrophic Gastritis. Cell Mol Gastroenterol Hepatol. 2021;11:857-880. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 61] [Article Influence: 10.2] [Reference Citation Analysis (2)] |
| 57. | Magari H, Shimizu Y, Inada K, Enomoto S, Tomeki T, Yanaoka K, Tamai H, Arii K, Nakata H, Oka M, Utsunomiya H, Tsutsumi Y, Tsukamoto T, Tatematsu M, Ichinose M. Inhibitory effect of etodolac, a selective cyclooxygenase-2 inhibitor, on stomach carcinogenesis in Helicobacter pylori-infected Mongolian gerbils. Biochem Biophys Res Commun. 2005;334:606-612. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 58. | Liao W, Wen Y, Wang J, Zhao M, Lv S, Chen N, Li Y, Wan L, Zheng Q, Mou Y, Zhao Z, Tang J, Zeng J. Gallic acid alleviates gastric precancerous lesions through inhibition of epithelial mesenchymal transition via Wnt/β-catenin signaling pathway. J Ethnopharmacol. 2023;302:115885. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 39] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 59. | Sethi NS, Kikuchi O, Duronio GN, Stachler MD, McFarland JM, Ferrer-Luna R, Zhang Y, Bao C, Bronson R, Patil D, Sanchez-Vega F, Liu JB, Sicinska E, Lazaro JB, Ligon KL, Beroukhim R, Bass AJ. Early TP53 alterations engage environmental exposures to promote gastric premalignancy in an integrative mouse model. Nat Genet. 2020;52:219-230. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 57] [Article Influence: 9.5] [Reference Citation Analysis (2)] |
| 60. | Lv S, Chen X, Chen Y, Gong D, Mao G, Shen C, Xia T, Cheng J, Luo Z, Cheng Y, Li W, Zeng J. Correction: Ginsenoside Rg3 induces apoptosis and inhibits proliferation by down-regulating TIGAR in rats with gastric precancerous lesions. BMC Complement Med Ther. 2023;23:65. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 61. | Yin J, Yi J, Yang C, Xu B, Lin J, Hu H, Wu X, Shi H, Fei X. Chronic atrophic gastritis and intestinal metaplasia induced by high-salt and N-methyl-N'-nitro-N-nitrosoguanidine intake in rats. Exp Ther Med. 2021;21:315. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (1)] |
| 62. | Tong Y, Wang R, Liu X, Tian M, Wang Y, Cui Y, Zou W, Zhao Y. Zuojin Pill ameliorates chronic atrophic gastritis induced by MNNG through TGF-β1/PI3K/Akt axis. J Ethnopharmacol. 2021;271:113893. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 62] [Article Influence: 12.4] [Reference Citation Analysis (1)] |
| 63. | Tong Y, Jing M, Zhao X, Liu H, Wei S, Li R, Liu X, Wang M, Song H, Zhao Y. Metabolomic Study of Zuojin Pill in Relieving 1-Methyl-3-nitro-1-nitrosoguanidine-Induced Chronic Atrophic Gastritis. Evid Based Complement Alternat Med. 2021;2021:7004798. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 64. | Zhu L, Cai Q, Li G, Zou X. Bromodomain containing 4 inhibition combats gastric precancerous lesions via modulating macrophage polarization. Tissue Cell. 2024;91:102580. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 65. | Zhu F, Xu Y, Pan J, Li M, Chen F, Xie G. Epigallocatechin Gallate Protects against MNNG-Induced Precancerous Lesions of Gastric Carcinoma in Rats via PI3K/Akt/mTOR Pathway. Evid Based Complement Alternat Med. 2021;2021:8846813. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 66. | Wang Q, Huang Y, Jiang M, Tang Y, Wang Q, Bai L, Yu C, Yang X, Ding K, Wang W, Bai J, Chen Y. The demethylase ALKBH5 mediates ZKSCAN3 expression through the m(6)A modification to activate VEGFA transcription and thus participates in MNNG-induced gastric cancer progression. J Hazard Mater. 2024;473:134690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 29] [Article Influence: 14.5] [Reference Citation Analysis (1)] |
| 67. | Li Y, Li T, Chen J, Zheng H, Li Y, Chu F, Wang S, Li P, Lin J, Su Z, Ding X. Manpixiao Decoction Halted the Malignant Transformation of Precancerous Lesions of Gastric Cancer: From Network Prediction to In-Vivo Verification. Front Pharmacol. 2022;13:927731. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 15] [Reference Citation Analysis (1)] |
| 68. | Chu F, Li Y, Meng X, Li Y, Li T, Zhai M, Zheng H, Xin T, Su Z, Lin J, Zhang P, Ding X. Gut Microbial Dysbiosis and Changes in Fecal Metabolic Phenotype in Precancerous Lesions of Gastric Cancer Induced With N-Methyl-N'-Nitro-N-Nitrosoguanidine, Sodium Salicylate, Ranitidine, and Irregular Diet. Front Physiol. 2021;12:733979. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (1)] |
| 69. | Yi Z, Jia Q, Wang Y, Zhang Y, Xie T, Ling J. Elian granules alleviate precancerous lesions of gastric cancer in rats by suppressing M2-type polarization of tumor-associated macrophages through NF-κB signaling pathway. BMC Complement Med Ther. 2023;23:188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 70. | Liu S, Ji H, Zhang T, Huang J, Yin X, Zhang J, Wang P, Wang F, Tang X. Modified Zuojin pill alleviates gastric precancerous lesions by inhibiting glycolysis through the HIF-1α pathway. Phytomedicine. 2025;136:156255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 71. | Kuo CH, Weng BC, Wu CC, Yang SF, Wu DC, Wang YC. Apigenin has anti-atrophic gastritis and anti-gastric cancer progression effects in Helicobacter pylori-infected Mongolian gerbils. J Ethnopharmacol. 2014;151:1031-1039. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 45] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 72. | Mansingh DP, Pradhan S, Biswas D, Barathidasan R, Vasanthi HR. Palliative Role of Aqueous Ginger Extract on N-Nitroso-N-Methylurea-Induced Gastric Cancer. Nutr Cancer. 2020;72:157-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 73. | Zhuang S, Jian YM, Sun YN. Inhibition of N-methyl-N-nitrosourea-induced gastric tumorigenesis by Liuwei Dihuang Pill in db/db mice. World J Gastroenterol. 2017;23:4233-4242. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 5] [Cited by in RCA: 9] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 74. | Zhao M, Wen Y, Yang Y, Pan H, Xie S, Shen C, Liao W, Chen N, Zheng Q, Zhang G, Li Y, Gong D, Tang J, Zhao Z, Zeng J. (-)-Asarinin alleviates gastric precancerous lesions by promoting mitochondrial ROS accumulation and inhibiting the STAT3 signaling pathway. Phytomedicine. 2024;126:155348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 75. | Liu J, Li M, Chen G, Yang J, Jiang Y, Li F, Hua H. Jianwei Xiaoyan granule ameliorates chronic atrophic gastritis by regulating HIF-1α-VEGF pathway. J Ethnopharmacol. 2024;334:118591. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 76. | Nomura S, Yamaguchi H, Ogawa M, Wang TC, Lee JR, Goldenring JR. Alterations in gastric mucosal lineages induced by acute oxyntic atrophy in wild-type and gastrin-deficient mice. Am J Physiol Gastrointest Liver Physiol. 2005;288:G362-G375. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 123] [Article Influence: 5.9] [Reference Citation Analysis (1)] |
| 77. | Petersen CP, Weis VG, Nam KT, Sousa JF, Fingleton B, Goldenring JR. Macrophages promote progression of spasmolytic polypeptide-expressing metaplasia after acute loss of parietal cells. Gastroenterology. 2014;146:1727-38.e8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 126] [Cited by in RCA: 119] [Article Influence: 9.9] [Reference Citation Analysis (2)] |
| 78. | Tomita H, Takaishi S, Menheniott TR, Yang X, Shibata W, Jin G, Betz KS, Kawakami K, Minamoto T, Tomasetto C, Rio MC, Lerkowit N, Varro A, Giraud AS, Wang TC. Inhibition of gastric carcinogenesis by the hormone gastrin is mediated by suppression of TFF1 epigenetic silencing. Gastroenterology. 2011;140:879-891. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 109] [Article Influence: 7.3] [Reference Citation Analysis (2)] |
| 79. | Zavros Y, Eaton KA, Kang W, Rathinavelu S, Katukuri V, Kao JY, Samuelson LC, Merchant JL. Chronic gastritis in the hypochlorhydric gastrin-deficient mouse progresses to adenocarcinoma. Oncogene. 2005;24:2354-2366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 114] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 80. | Lofgren JL, Whary MT, Ge Z, Muthupalani S, Taylor NS, Mobley M, Potter A, Varro A, Eibach D, Suerbaum S, Wang TC, Fox JG. Lack of commensal flora in Helicobacter pylori-infected INS-GAS mice reduces gastritis and delays intraepithelial neoplasia. Gastroenterology. 2011;140:210-220. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 339] [Cited by in RCA: 319] [Article Influence: 21.3] [Reference Citation Analysis (6)] |
| 81. | Chaturvedi R, Asim M, Romero-Gallo J, Barry DP, Hoge S, de Sablet T, Delgado AG, Wroblewski LE, Piazuelo MB, Yan F, Israel DA, Casero RA Jr, Correa P, Gobert AP, Polk DB, Peek RM Jr, Wilson KT. Spermine oxidase mediates the gastric cancer risk associated with Helicobacter pylori CagA. Gastroenterology. 2011;141:1696-708.e1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 187] [Cited by in RCA: 168] [Article Influence: 11.2] [Reference Citation Analysis (1)] |
| 82. | Xu X, Fei X, Wang H, Wu X, Zhan Y, Li X, Zhou Y, Shu C, He C, Hu Y, Liu J, Lv N, Li N, Zhu Y. Helicobacter pylori infection induces DNA double-strand breaks through the ACVR1/IRF3/POLD1 signaling axis to drive gastric tumorigenesis. Gut Microbes. 2025;17:2463581. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 83. | Fox JG, Wang TC, Rogers AB, Poutahidis T, Ge Z, Taylor N, Dangler CA, Israel DA, Krishna U, Gaus K, Peek RM Jr. Host and microbial constituents influence Helicobacter pylori-induced cancer in a murine model of hypergastrinemia. Gastroenterology. 2003;124:1879-1890. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 148] [Article Influence: 6.4] [Reference Citation Analysis (1)] |
| 84. | Takaishi S, Tu S, Dubeykovskaya ZA, Whary MT, Muthupalani S, Rickman BH, Rogers AB, Lertkowit N, Varro A, Fox JG, Wang TC. Gastrin is an essential cofactor for helicobacter-associated gastric corpus carcinogenesis in C57BL/6 mice. Am J Pathol. 2009;175:365-375. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 51] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 85. | Pan KF, Li WQ, Zhang L, Liu WD, Ma JL, Zhang Y, Ulm K, Wang JX, Zhang L, Bajbouj M, Zhang LF, Li M, Vieth M, Quante M, Wang LH, Suchanek S, Mejías-Luque R, Xu HM, Fan XH, Han X, Liu ZC, Zhou T, Guan WX, Schmid RM, Gerhard M, Classen M, You WC. Gastric cancer prevention by community eradication of Helicobacter pylori: a cluster-randomized controlled trial. Nat Med. 2024;30:3250-3260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 66] [Article Influence: 33.0] [Reference Citation Analysis (1)] |
| 86. | Fukase K, Kato M, Kikuchi S, Inoue K, Uemura N, Okamoto S, Terao S, Amagai K, Hayashi S, Asaka M; Japan Gast Study Group. Effect of eradication of Helicobacter pylori on incidence of metachronous gastric carcinoma after endoscopic resection of early gastric cancer: an open-label, randomised controlled trial. Lancet. 2008;372:392-397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1023] [Cited by in RCA: 929] [Article Influence: 51.6] [Reference Citation Analysis (2)] |
| 87. | Tu S, Bhagat G, Cui G, Takaishi S, Kurt-Jones EA, Rickman B, Betz KS, Penz-Oesterreicher M, Bjorkdahl O, Fox JG, Wang TC. Overexpression of interleukin-1beta induces gastric inflammation and cancer and mobilizes myeloid-derived suppressor cells in mice. Cancer Cell. 2008;14:408-419. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 731] [Cited by in RCA: 696] [Article Influence: 38.7] [Reference Citation Analysis (6)] |
| 88. | Syu LJ, El-Zaatari M, Eaton KA, Liu Z, Tetarbe M, Keeley TM, Pero J, Ferris J, Wilbert D, Kaatz A, Zheng X, Qiao X, Grachtchouk M, Gumucio DL, Merchant JL, Samuelson LC, Dlugosz AA. Transgenic expression of interferon-γ in mouse stomach leads to inflammation, metaplasia, and dysplasia. Am J Pathol. 2012;181:2114-2125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 58] [Article Influence: 4.1] [Reference Citation Analysis (2)] |
| 89. | Nguyen TL, Khurana SS, Bellone CJ, Capoccia BJ, Sagartz JE, Kesman RA Jr, Mills JC, DiPaolo RJ. Autoimmune gastritis mediated by CD4+ T cells promotes the development of gastric cancer. Cancer Res. 2013;73:2117-2126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 57] [Article Influence: 4.4] [Reference Citation Analysis (2)] |
| 90. | Choi E, Hendley AM, Bailey JM, Leach SD, Goldenring JR. Expression of Activated Ras in Gastric Chief Cells of Mice Leads to the Full Spectrum of Metaplastic Lineage Transitions. Gastroenterology. 2016;150:918-30.e13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 141] [Article Influence: 14.1] [Reference Citation Analysis (3)] |
| 91. | Hayakawa Y, Ariyama H, Stancikova J, Sakitani K, Asfaha S, Renz BW, Dubeykovskaya ZA, Shibata W, Wang H, Westphalen CB, Chen X, Takemoto Y, Kim W, Khurana SS, Tailor Y, Nagar K, Tomita H, Hara A, Sepulveda AR, Setlik W, Gershon MD, Saha S, Ding L, Shen Z, Fox JG, Friedman RA, Konieczny SF, Worthley DL, Korinek V, Wang TC. Mist1 Expressing Gastric Stem Cells Maintain the Normal and Neoplastic Gastric Epithelium and Are Supported by a Perivascular Stem Cell Niche. Cancer Cell. 2015;28:800-814. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 279] [Cited by in RCA: 266] [Article Influence: 24.2] [Reference Citation Analysis (2)] |
| 92. | Matsuo J, Kimura S, Yamamura A, Koh CP, Hossain MZ, Heng DL, Kohu K, Voon DC, Hiai H, Unno M, So JB, Zhu F, Srivastava S, Teh M, Yeoh KG, Osato M, Ito Y. Identification of Stem Cells in the Epithelium of the Stomach Corpus and Antrum of Mice. Gastroenterology. 2017;152:218-231.e14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 119] [Article Influence: 13.2] [Reference Citation Analysis (1)] |
| 93. | Okumura T, Ericksen RE, Takaishi S, Wang SS, Dubeykovskiy Z, Shibata W, Betz KS, Muthupalani S, Rogers AB, Fox JG, Rustgi AK, Wang TC. K-ras mutation targeted to gastric tissue progenitor cells results in chronic inflammation, an altered microenvironment, and progression to intraepithelial neoplasia. Cancer Res. 2010;70:8435-8445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 75] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 94. | Ito K, Chuang LS, Ito T, Chang TL, Fukamachi H, Salto-Tellez M, Ito Y. Loss of Runx3 is a key event in inducing precancerous state of the stomach. Gastroenterology. 2011;140:1536-46.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 73] [Article Influence: 4.9] [Reference Citation Analysis (1)] |
| 95. | Costa L, Corre S, Michel V, Le Luel K, Fernandes J, Ziveri J, Jouvion G, Danckaert A, Mouchet N, Da Silva Barreira D, Torres J, Camorlinga M, D'Elios MM, Fiette L, De Reuse H, Galibert MD, Touati E. USF1 defect drives p53 degradation during Helicobacter pylori infection and accelerates gastric carcinogenesis. Gut. 2020;69:1582-1591. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 77] [Article Influence: 12.8] [Reference Citation Analysis (1)] |
| 96. | Zhang S, Lee DS, Morrissey R, Aponte-Pieras JR, Rogers AB, Moss SF. Early or late antibiotic intervention prevents Helicobacter pylori-induced gastric cancer in a mouse model. Cancer Lett. 2015;359:345-351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 13] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 97. | Feng W, Ma C, Rao H, Zhang W, Liu C, Xu Y, Aji R, Wang Z, Xu J, Gao WQ, Li L. Setd2 deficiency promotes gastric tumorigenesis through inhibiting the SIRT1/FOXO pathway. Cancer Lett. 2023;579:216470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (1)] |
| 98. | Suzuki K, Sentani K, Tanaka H, Yano T, Suzuki K, Oshima M, Yasui W, Tamura A, Tsukita S. Deficiency of Stomach-Type Claudin-18 in Mice Induces Gastric Tumor Formation Independent of H pylori Infection. Cell Mol Gastroenterol Hepatol. 2019;8:119-142. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 44] [Article Influence: 6.3] [Reference Citation Analysis (1)] |
| 99. | Liu X, Li T, Ma Z, Riederer B, Yuan D, Zhu J, Li Y, An J, Wen G, Jin H, Yang X, Seidler U, Tuo B. SLC26A9 deficiency causes gastric intraepithelial neoplasia in mice and aggressive gastric cancer in humans. Cell Oncol (Dordr). 2022;45:381-398. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (2)] |
| 100. | Katsha A, Soutto M, Sehdev V, Peng D, Washington MK, Piazuelo MB, Tantawy MN, Manning HC, Lu P, Shyr Y, Ecsedy J, Belkhiri A, El-Rifai W. Aurora kinase A promotes inflammation and tumorigenesis in mice and human gastric neoplasia. Gastroenterology. 2013;145:1312-22.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 97] [Article Influence: 7.5] [Reference Citation Analysis (1)] |
| 101. | Zeng X, Yang M, Ye T, Feng J, Xu X, Yang H, Wang X, Bao L, Li R, Xue B, Zang J, Huang Y. Mitochondrial GRIM-19 loss in parietal cells promotes spasmolytic polypeptide-expressing metaplasia through NLR family pyrin domain-containing 3 (NLRP3)-mediated IL-33 activation via a reactive oxygen species (ROS) -NRF2- Heme oxygenase-1(HO-1)-NF-кB axis. Free Radic Biol Med. 2023;202:46-61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 19] [Article Influence: 6.3] [Reference Citation Analysis (1)] |
| 102. | Zuo X, Deguchi Y, Xu W, Liu Y, Li HS, Wei D, Tian R, Chen W, Xu M, Yang Y, Gao S, Jaoude JC, Liu F, Chrieki SP, Moussalli MJ, Gagea M, Sebastian MM, Zheng X, Tan D, Broaddus R, Wang J, Ajami NJ, Swennes AG, Watowich SS, Shureiqi I. PPARD and Interferon Gamma Promote Transformation of Gastric Progenitor Cells and Tumorigenesis in Mice. Gastroenterology. 2019;157:163-178. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 46] [Article Influence: 6.6] [Reference Citation Analysis (1)] |
| 103. | Nam KT, O'Neal R, Lee YS, Lee YC, Coffey RJ, Goldenring JR. Gastric tumor development in Smad3-deficient mice initiates from forestomach/glandular transition zone along the lesser curvature. Lab Invest. 2012;92:883-895. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 34] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 104. | Banerjee A, Thamphiwatana S, Carmona EM, Rickman B, Doran KS, Obonyo M. Deficiency of the myeloid differentiation primary response molecule MyD88 leads to an early and rapid development of Helicobacter-induced gastric malignancy. Infect Immun. 2014;82:356-363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 35] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 105. | Neumeyer V, Vieth M, Gerhard M, Mejías-Luque R. Mutated Rnf43 Aggravates Helicobacter Pylori-Induced Gastric Pathology. Cancers (Basel). 2019;11:372. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 106. | Seidlitz T, Chen YT, Uhlemann H, Schölch S, Kochall S, Merker SR, Klimova A, Hennig A, Schweitzer C, Pape K, Baretton GB, Welsch T, Aust DE, Weitz J, Koo BK, Stange DE. Mouse Models of Human Gastric Cancer Subtypes With Stomach-Specific CreERT2-Mediated Pathway Alterations. Gastroenterology. 2019;157:1599-1614.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 73] [Article Influence: 10.4] [Reference Citation Analysis (1)] |
| 107. | Li XB, Yang G, Zhu L, Tang YL, Zhang C, Ju Z, Yang X, Teng Y. Gastric Lgr5(+) stem cells are the cellular origin of invasive intestinal-type gastric cancer in mice. Cell Res. 2016;26:838-849. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 78] [Article Influence: 7.8] [Reference Citation Analysis (1)] |
| 108. | Fang KT, Hung H, Lau NYS, Chi JH, Wu DC, Cheng KH. Development of a Genetically Engineered Mouse Model Recapitulating LKB1 and PTEN Deficiency in Gastric Cancer Pathogenesis. Cancers (Basel). 2023;15:5893. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 109. | Tu SP, Quante M, Bhagat G, Takaishi S, Cui G, Yang XD, Muthuplani S, Shibata W, Fox JG, Pritchard DM, Wang TC. IFN-γ inhibits gastric carcinogenesis by inducing epithelial cell autophagy and T-cell apoptosis. Cancer Res. 2011;71:4247-4259. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 101] [Cited by in RCA: 102] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 110. | Oshima H, Matsunaga A, Fujimura T, Tsukamoto T, Taketo MM, Oshima M. Carcinogenesis in mouse stomach by simultaneous activation of the Wnt signaling and prostaglandin E2 pathway. Gastroenterology. 2006;131:1086-1095. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 183] [Article Influence: 9.2] [Reference Citation Analysis (1)] |
| 111. | Shinohara M, Mao M, Keeley TM, El-Zaatari M, Lee HJ, Eaton KA, Samuelson LC, Merchant JL, Goldenring JR, Todisco A. Bone morphogenetic protein signaling regulates gastric epithelial cell development and proliferation in mice. Gastroenterology. 2010;139:2050-2060.e2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 61] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 112. | Douchi D, Yamamura A, Matsuo J, Melissa Lim YH, Nuttonmanit N, Shimura M, Suda K, Chen S, Pang S, Kohu K, Abe T, Shioi G, Kim G, Shabbir A, Srivastava S, Unno M, Bok-Yan So J, Teh M, Yeoh KG, Chuang LSH, Ito Y. Induction of Gastric Cancer by Successive Oncogenic Activation in the Corpus. Gastroenterology. 2021;161:1907-1923.e26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 113. | Liang Z, Song J, Xu Y, Zhang X, Zhang Y, Qian H. Hesperidin Reversed Long-Term N-methyl-N-nitro-N-Nitroguanidine Exposure Induced EMT and Cell Proliferation by Activating Autophagy in Gastric Tissues of Rats. Nutrients. 2022;14:5281. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 114. | Zhou X, Zhang C, Shen W, Zhuang R, Liu S, Liu J. Curzerene suppresses epithelial-mesenchymal transition in gastric precancerous lesion cells through targeted regulation of YAP via the long non-coding RNA AFAP1-AS1. Phytomedicine. 2025;143:156879. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 115. | Jiang Q, Fan G, Wu K. Potential Action Mechanism of Erianin in Relieving MNNG-triggered Chronic Atrophic Gastritis. Cell Biochem Biophys. 2025;83:1035-1044. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 116. | Liang D, Tang S, Liu L, Zhao M, Ma X, Zhao Y, Shen C, Liu Q, Tang J, Zeng J, Chen N. Tanshinone I attenuates gastric precancerous lesions by inhibiting epithelial mesenchymal transition through the p38/STAT3 pathway. Int Immunopharmacol. 2023;124:110902. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 117. | Yuan Z, Wang Y, Wang X, Du X, Li G, Luo L, Yao B, Zhang J, Zhao F, Liu D. The fruit of Rosa odorata sweet var. gigantea (Coll. et Hemsl.) Rehd. et Wils attenuates chronic atrophic gastritis induced by MNNG and its potential mechanism. J Ethnopharmacol. 2025;337:118876. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 118. | Xu J, Shen W, Pei B, Wang X, Sun D, Li Y, Xiu L, Liu X, Lu Y, Zhang X, Yue X. Xiao Tan He Wei Decoction reverses MNNG-induced precancerous lesions of gastric carcinoma in vivo and vitro: Regulation of apoptosis through NF-κB pathway. Biomed Pharmacother. 2018;108:95-102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 55] [Article Influence: 6.9] [Reference Citation Analysis (2)] |
| 119. | Jian J, Feng Y, Wang R, Li C, Zhang L, Ruan Y, Luo B, Liang G, Liu T. METTL3-Regulated lncRNA SNHG7 Drives MNNG-Induced Epithelial-Mesenchymal Transition in Gastric Precancerous Lesions. Toxics. 2024;12:573. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 120. | Liu T, Feng YL, Wang RY, Yang S, Ge YL, Zhang TY, Li J, Li CY, Ruan Y, Luo B, Liang GY. Long-term MNNG exposure promotes gastric carcinogenesis by activating METTL3/m6A/miR1184 axis-mediated epithelial-mesenchymal transition. Sci Total Environ. 2024;913:169752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (1)] |
| 121. | Chen Y, Fu R, Xu M, Huang Y, Sun G, Xu L. N-methyl-N-nitro-N-nitrosoguanidine-mediated ING4 downregulation contributed to the angiogenesis of transformed human gastric epithelial cells. Life Sci. 2018;199:179-187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 122. | Song P, Li X, Chen S, Gong Y, Zhao J, Jiao Y, Dai Y, Yang H, Qian J, Li Y, He J, Tang L. YTHDF1 mediates N-methyl-N-nitrosourea-induced gastric carcinogenesis by controlling HSPH1 translation. Cell Prolif. 2024;57:e13619. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 123. | Li T, Guo H, Li H, Jiang Y, Zhuang K, Lei C, Wu J, Zhou H, Zhu R, Zhao X, Lu Y, Shi C, Nie Y, Wu K, Yuan Z, Fan DM, Shi Y. MicroRNA-92a-1-5p increases CDX2 by targeting FOXD1 in bile acids-induced gastric intestinal metaplasia. Gut. 2019;68:1751-1763. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 82] [Article Influence: 11.7] [Reference Citation Analysis (1)] |
| 124. | Yue B, Cui R, Zheng R, Jin W, Song C, Bao T, Wang M, Yu F, Zhao E. Essential role of ALKBH5-mediated RNA demethylation modification in bile acid-induced gastric intestinal metaplasia. Mol Ther Nucleic Acids. 2021;26:458-472. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 125. | Zhao Y, Huang H, Wang X, Hu W, Lu X, Tang S, Liu H, Sun Y. Dendrobium officinale Polysaccharides Inhibit CDCA-Induced Gastric Intestinal Metaplasia through Activating NRF2/HO-1 and Modulating HNF4α/CDX2 Signaling Pathway. J Food Biochem. 2023;2023:1-11. [DOI] [Full Text] |
| 126. | Yu JH, Zheng JB, Qi J, Yang K, Wu YH, Wang K, Wang CB, Sun XJ. Bile acids promote gastric intestinal metaplasia by upregulating CDX2 and MUC2 expression via the FXR/NF-κB signalling pathway. Int J Oncol. 2019;54:879-892. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 60] [Article Influence: 8.6] [Reference Citation Analysis (3)] |
| 127. | Yang T, Wang R, Zhang J, Bao C, Zhang J, Li R, Chen X, Wu S, Wen J, Wei S, Li H, Cai H, Yang X, Zhao Y. Mechanism of berberine in treating Helicobacter pylori induced chronic atrophic gastritis through IRF8-IFN-γ signaling axis suppressing. Life Sci. 2020;248:117456. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 50] [Article Influence: 8.3] [Reference Citation Analysis (2)] |
| 128. | Cheng S, Li H, Chi J, Zhao W, Lin J, Liu X, Xu C. FTO-mediated m(6)A modification promotes malignant transformation of gastric mucosal epithelial cells in chronic Cag A(+) Helicobacter pylori infection. J Cancer Res Clin Oncol. 2023;149:7327-7340. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 129. | He J, Hu W, Ouyang Q, Zhang S, He L, Chen W, Li X, Hu C. Helicobacter pylori infection induces stem cell-like properties in Correa cascade of gastric cancer. Cancer Lett. 2022;542:215764. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 40] [Article Influence: 10.0] [Reference Citation Analysis (1)] |
| 130. | Pang MJ, Burclaff JR, Jin R, Adkins-Threats M, Osaki LH, Han Y, Mills JC, Miao ZF, Wang ZN. Gastric Organoids: Progress and Remaining Challenges. Cell Mol Gastroenterol Hepatol. 2022;13:19-33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 131. | Yan HHN, Chan AS, Lai FP, Leung SY. Organoid cultures for cancer modeling. Cell Stem Cell. 2023;30:917-937. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 98] [Article Influence: 32.7] [Reference Citation Analysis (1)] |
| 132. | Seidlitz T, Merker SR, Rothe A, Zakrzewski F, von Neubeck C, Grützmann K, Sommer U, Schweitzer C, Schölch S, Uhlemann H, Gaebler AM, Werner K, Krause M, Baretton GB, Welsch T, Koo BK, Aust DE, Klink B, Weitz J, Stange DE. Human gastric cancer modelling using organoids. Gut. 2019;68:207-217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 278] [Cited by in RCA: 260] [Article Influence: 37.1] [Reference Citation Analysis (5)] |
| 133. | Vlachogiannis G, Hedayat S, Vatsiou A, Jamin Y, Fernández-Mateos J, Khan K, Lampis A, Eason K, Huntingford I, Burke R, Rata M, Koh DM, Tunariu N, Collins D, Hulkki-Wilson S, Ragulan C, Spiteri I, Moorcraft SY, Chau I, Rao S, Watkins D, Fotiadis N, Bali M, Darvish-Damavandi M, Lote H, Eltahir Z, Smyth EC, Begum R, Clarke PA, Hahne JC, Dowsett M, de Bono J, Workman P, Sadanandam A, Fassan M, Sansom OJ, Eccles S, Starling N, Braconi C, Sottoriva A, Robinson SP, Cunningham D, Valeri N. Patient-derived organoids model treatment response of metastatic gastrointestinal cancers. Science. 2018;359:920-926. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1739] [Cited by in RCA: 1497] [Article Influence: 187.1] [Reference Citation Analysis (4)] |
| 134. | Nanki K, Toshimitsu K, Takano A, Fujii M, Shimokawa M, Ohta Y, Matano M, Seino T, Nishikori S, Ishikawa K, Kawasaki K, Togasaki K, Takahashi S, Sukawa Y, Ishida H, Sugimoto S, Kawakubo H, Kim J, Kitagawa Y, Sekine S, Koo BK, Kanai T, Sato T. Divergent Routes toward Wnt and R-spondin Niche Independency during Human Gastric Carcinogenesis. Cell. 2018;174:856-869.e17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 294] [Cited by in RCA: 254] [Article Influence: 31.8] [Reference Citation Analysis (4)] |
| 135. | Yan HHN, Siu HC, Law S, Ho SL, Yue SSK, Tsui WY, Chan D, Chan AS, Ma S, Lam KO, Bartfeld S, Man AHY, Lee BCH, Chan ASY, Wong JWH, Cheng PSW, Chan AKW, Zhang J, Shi J, Fan X, Kwong DLW, Mak TW, Yuen ST, Clevers H, Leung SY. A Comprehensive Human Gastric Cancer Organoid Biobank Captures Tumor Subtype Heterogeneity and Enables Therapeutic Screening. Cell Stem Cell. 2018;23:882-897.e11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 627] [Cited by in RCA: 558] [Article Influence: 69.8] [Reference Citation Analysis (4)] |
| 136. | Wallaschek N, Niklas C, Pompaiah M, Wiegering A, Germer CT, Kircher S, Brändlein S, Maurus K, Rosenwald A, Yan HHN, Leung SY, Bartfeld S. Establishing Pure Cancer Organoid Cultures: Identification, Selection and Verification of Cancer Phenotypes and Genotypes. J Mol Biol. 2019;431:2884-2893. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 36] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 137. | Li K, Ma X, Li Z, Liu Y, Shen G, Luo Z, Wang D, Xia L, Wang Z, Tian M, Liu H, Geng F, Li B. A Natural Peptide from A Traditional Chinese Medicine Has the Potential to Treat Chronic Atrophic Gastritis by Activating Gastric Stem Cells. Adv Sci (Weinh). 2024;11:e2304326. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (2)] |
| 138. | Yoshihiro T, Yamaguchi K, Ariyama H, Koreishi S, Uehara K, Ohmura H, Ito M, Tsuchihashi K, Isobe T, Shindo K, Ohuchida K, Nakamura M, Nagao Y, Oda Y, Akashi K, Baba E. Elucidation of the mechanism of carcinogenic transformation of human gastric epithelial cells in atrophic gastritis. Biochim Biophys Acta Mol Basis Dis. 2025;1871:167843. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (2)] |
| 139. | Barker N, Huch M, Kujala P, van de Wetering M, Snippert HJ, van Es JH, Sato T, Stange DE, Begthel H, van den Born M, Danenberg E, van den Brink S, Korving J, Abo A, Peters PJ, Wright N, Poulsom R, Clevers H. Lgr5(+ve) stem cells drive self-renewal in the stomach and build long-lived gastric units in vitro. Cell Stem Cell. 2010;6:25-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1348] [Cited by in RCA: 1213] [Article Influence: 75.8] [Reference Citation Analysis (2)] |
| 140. | Wei H, Li W, Zeng L, Ding N, Li K, Yu H, Jiang F, Yin H, Xia Y, Deng C, Cai N, Chen X, Gu L, Chen H, Zhang F, He Y, Li J, Zhang C. OLFM4 promotes the progression of intestinal metaplasia through activation of the MYH9/GSK3β/β-catenin pathway. Mol Cancer. 2024;23:124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 27] [Reference Citation Analysis (1)] |
| 141. | Yue SSK, Tong Y, Siu HC, Ho SL, Law SYK, Tsui WY, Chan D, Huang Y, Chan ASY, Yun SW, Hui HS, Choi JE, Hsu MSS, Lai FPL, Chan AS, Yuen ST, Clevers H, Leung SY, Yan HHN. Divergent lineage trajectories and genetic landscapes in human gastric intestinal metaplasia organoids associated with early neoplastic progression. Gut. 2025;74:522-538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 142. | Min J, Zhang C, Bliton RJ, Caldwell B, Caplan L, Presentation KS, Park DJ, Kong SH, Lee HS, Washington MK, Kim WH, Lau KS, Magness ST, Lee HJ, Yang HK, Goldenring JR, Choi E. Dysplastic Stem Cell Plasticity Functions as a Driving Force for Neoplastic Transformation of Precancerous Gastric Mucosa. Gastroenterology. 2022;163:875-890. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 40] [Article Influence: 10.0] [Reference Citation Analysis (2)] |
| 143. | McCauley HA, Wells JM. Pluripotent stem cell-derived organoids: using principles of developmental biology to grow human tissues in a dish. Development. 2017;144:958-962. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 240] [Article Influence: 26.7] [Reference Citation Analysis (9)] |
| 144. | McCracken KW, Catá EM, Crawford CM, Sinagoga KL, Schumacher M, Rockich BE, Tsai YH, Mayhew CN, Spence JR, Zavros Y, Wells JM. Modelling human development and disease in pluripotent stem-cell-derived gastric organoids. Nature. 2014;516:400-404. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 906] [Cited by in RCA: 744] [Article Influence: 62.0] [Reference Citation Analysis (14)] |
| 145. | Karlsson K, Przybilla MJ, Kotler E, Khan A, Xu H, Karagyozova K, Sockell A, Wong WH, Liu K, Mah A, Lo YH, Lu B, Houlahan KE, Ma Z, Suarez CJ, Barnes CP, Kuo CJ, Curtis C. Deterministic evolution and stringent selection during preneoplasia. Nature. 2023;618:383-393. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 66] [Article Influence: 22.0] [Reference Citation Analysis (1)] |
| 146. | Koide T, Koyanagi-Aoi M, Uehara K, Kakeji Y, Aoi T. CDX2-induced intestinal metaplasia in human gastric organoids derived from induced pluripotent stem cells. iScience. 2022;25:104314. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 25] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 147. | Li Y, Chen J, Li T, Lin J, Zheng H, Johnson N, Yao X, Ding X. Modeling gastric intestinal metaplasia in 3D organoids using nitrosoguanidine. J Mol Cell Biol. 2024;16:mjae030. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 148. | Min J, Vega PN, Engevik AC, Williams JA, Yang Q, Patterson LM, Simmons AJ, Bliton RJ, Betts JW, Lau KS, Magness ST, Goldenring JR, Choi E. Heterogeneity and dynamics of active Kras-induced dysplastic lineages from mouse corpus stomach. Nat Commun. 2019;10:5549. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 41] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 149. | Lu Z, Zhong A, Liu H, Zhang M, Chen X, Pan X, Wang M, Deng X, Gao L, Zhao L, Wang J, Yang Y, Zhang Q, Wu B, Zheng J, Wang Y, Song X, Liu K, Zhang W, Chen X, Yang K, Chen X, Zhao Y, Zhao C, Wang Y, Chen L, Zhou Z, Hu J, Liu Y, Chen C. Dissecting the genetic and microenvironmental factors of gastric tumorigenesis in mice. Cell Rep. 2022;41:111482. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (2)] |
| 150. | Lo YH, Kolahi KS, Du Y, Chang CY, Krokhotin A, Nair A, Sobba WD, Karlsson K, Jones SJ, Longacre TA, Mah AT, Tercan B, Sockell A, Xu H, Seoane JA, Chen J, Shmulevich I, Weissman JS, Curtis C, Califano A, Fu H, Crabtree GR, Kuo CJ. A CRISPR/Cas9-Engineered ARID1A-Deficient Human Gastric Cancer Organoid Model Reveals Essential and Nonessential Modes of Oncogenic Transformation. Cancer Discov. 2021;11:1562-1581. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 128] [Article Influence: 25.6] [Reference Citation Analysis (0)] |
| 151. | Osaki LH, Bockerstett KA, Wong CF, Ford EL, Madison BB, DiPaolo RJ, Mills JC. Interferon-γ directly induces gastric epithelial cell death and is required for progression to metaplasia. J Pathol. 2019;247:513-523. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 68] [Cited by in RCA: 69] [Article Influence: 9.9] [Reference Citation Analysis (1)] |
| 152. | Zhang X, Wen J, Pan Z, Liu Y, Zhu Y. Celastrus orbiculatus Thunb. extract inhibits inflammatory metabolic adaptation in macrophages and regulates polarization via modulating PKM2. Int Immunopharmacol. 2025;144:113665. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 153. | Guenther AA, Ahn S, Min J, Zhang C, Lee HJ, Yang HK, Sung BH, Weaver AM, Choi E. Cortactin Facilitates Malignant Transformation of Dysplastic Cells in Gastric Cancer Development. Cell Mol Gastroenterol Hepatol. 2025;19:101490. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 154. | Sierra JC, Piazuelo MB, Luis PB, Barry DP, Allaman MM, Asim M, Sebrell TA, Finley JL, Rose KL, Hill S, Holshouser SL, Casero RA, Cleveland JL, Woster PM, Schey KL, Bimczok D, Schneider C, Gobert AP, Wilson KT. Spermine oxidase mediates Helicobacter pylori-induced gastric inflammation, DNA damage, and carcinogenic signaling. Oncogene. 2020;39:4465-4474. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 64] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 155. | Aguilar C, Pauzuolis M, Pompaiah M, Vafadarnejad E, Arampatzi P, Fischer M, Narres D, Neyazi M, Kayisoglu Ö, Sell T, Blüthgen N, Morkel M, Wiegering A, Germer CT, Kircher S, Rosenwald A, Saliba AE, Bartfeld S. Helicobacter pylori shows tropism to gastric differentiated pit cells dependent on urea chemotaxis. Nat Commun. 2022;13:5878. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 36] [Reference Citation Analysis (0)] |
| 156. | Yu BM, Lee SD, Hwang BR, Kim JS, Yu S, Nam KT, Lee YC. Application of an organoid-based model to explore Helicobacter pylori-human gastric epithelium interaction in vitro. Front Cell Infect Microbiol. 2025;15:1572244. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 157. | Hofer M, Kim Y, Broguiere N, Gorostidi F, Klein JA, Amieva MR, Lutolf MP. Accessible homeostatic gastric organoids reveal secondary cell type-specific host-pathogen interactions in Helicobacter pylori infections. Nat Commun. 2025;16:2767. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 15] [Article Influence: 15.0] [Reference Citation Analysis (1)] |
| 158. | Lu Y, Cheng D, Pang J, Peng Y, Jin S, Zhang X, Li Y, Zuo Y. Chronic stress promotes gastric cancer progression via the adrenoceptor beta 2/PlexinA1 pathway. Cell Stress Chaperones. 2024;29:201-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 159. | Rezaei Z, Wang N, Yang Y, Govindaraj K, Velasco JJ, Martinez Blanco AD, Bae NH, Lee H, Shin SR. Enhancing organoid technology with carbon-based nanomaterial biosensors: Advancements, challenges, and future directions. Adv Drug Deliv Rev. 2025;222:115592. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 160. | Verstegen MMA, Coppes RP, Beghin A, De Coppi P, Gerli MFM, de Graeff N, Pan Q, Saito Y, Shi S, Zadpoor AA, van der Laan LJW. Clinical applications of human organoids. Nat Med. 2025;31:409-421. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 90] [Article Influence: 90.0] [Reference Citation Analysis (1)] |